Genome-Wide Association Study Reveals the Genetic Basis of Duck Plumage Colors
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling
2.2. Genotyping and SNP Calling
2.3. GWAS
2.4. Analysis of SNP–SNP Epistasis
2.5. Functional Annotation
3. Results
3.1. Associations between SNPs and Plumage Color
3.2. MITF–MC1R Interaction and Epistasis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Seddon, N.; Botero, C.A.; Tobias, J.; Dunn, P.O.; MacGregor, H.E.A.; Rubenstein, D.; Uy, J.A.C.; Weir, J.T.; Whittingham, L.A.; Safran, R.J. Sexual selection accelerates signal evolution during speciation in birds. Proc. R. Soc. B Boil. Sci. 2013, 280, 20131065. [Google Scholar] [CrossRef] [PubMed]
- Wilkins, M.R.; Karaardıç, H.; Vortman, Y.; Parchman, T.L.; Albrecht, T.; Petrželková, A.; Özkan, L.; Pap, P.L.; Hubbard, J.K.; Hund, A.K.; et al. Phenotypic differentiation is associated with divergent sexual selection among closely related barn swallow populations. J. Evol. Biol. 2016, 29, 2410–2421. [Google Scholar] [CrossRef] [PubMed]
- Hubbard, J.K.; Jenkins, B.R.; Safran, R.J. Quantitative genetics of plumage color: Lifetime effects of early nest environment on a colorful sexual signal. Ecol. Evol. 2015, 5, 3436–3449. [Google Scholar] [CrossRef] [PubMed]
- Lancaster, F.M. The Inheritance of Plumage Colour in the Common Duck; Bibliographia Genetica XIX; Springer: Berlin/Heidelberg, Germany, 1963; pp. 317–404. [Google Scholar]
- Gluckman, T.-L.; Mundy, N.I. Evolutionary pathways to convergence in plumage patterns. BMC Evol. Biol. 2016, 16, 172. [Google Scholar] [CrossRef] [PubMed]
- Kushimoto, T.; Valencia, J.; Costin, G.-E.; Toyofuku, K.; Watabe, H.; Yasumoto, K.-I.; Rouzaud, F.; Vieira, W.; Hearing, V. The Seiji memorial lecture: The melanosome: An ideal model to study cellular differentiation. Pigment. Cell Res. 2003, 16, 237–244. [Google Scholar] [CrossRef]
- Le Douarin, N.M.; Dupin, E. Multipotentiality of the neural crest. Curr. Opin. Genet. Dev. 2003, 13, 529–536. [Google Scholar] [CrossRef]
- Wakamatsu, K.; Ito, S. Advanced Chemical Methods in Melanin Determination. Pigment. Cell Res. 2002, 15, 174–183. [Google Scholar] [CrossRef]
- McGraw, K.J.; Safran, R.J.; Wakamatsu, K. How feather colour reflects its melanin content. Funct. Ecol. 2005, 19, 816–821. [Google Scholar] [CrossRef]
- Hearing, V.J.; Tsukamoto, K. Enzymatic control of pigmentation in mammals. FASEB J. 1991, 5, 2902–2909. [Google Scholar] [CrossRef]
- Raposo, G.; Marks, M.S. Melanosomes—Dark organelles enlighten endosomal membrane transport. Nat. Rev. Mol. Cell Biol. 2007, 8, 786–797. [Google Scholar] [CrossRef]
- Campbell, R.; Reinhard, B.; Jerome, F. A “New” Allele of the Mallard Plumage Pattern in Ducks. Poult. Sci. 1984, 63, 19–24. [Google Scholar] [CrossRef]
- Jaap, R.G. Inheritance of White Spotting in Ducks. Poult. Sci. 1933, 12, 233–241. [Google Scholar] [CrossRef]
- Gong, Y.; Yang, Q.; Li, S.; Feng, Y.; Gao, C.; Tu, G.; Peng, X. Grey plumage colouration in the duck is genetically determined by the alleles on two different, interacting loci. Anim. Genet. 2010, 41, 105–108. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, S.; Suzuki, H.; Yabuuchi, M.; Takahashi, S. A possible involvement of melanocortin 1-receptor in regulating feather color pigmentation in the chicken. Biochim. Biophys. Acta (BBA) Gene Struct. Expr. 1996, 1308, 164–168. [Google Scholar] [CrossRef]
- Kerje, S.; Lind, J.; Schütz, K.; Jensen, P.; Andersson, L. Melanocortin 1-receptor (MC1R) mutations are associated with plumage colour in chicken. Anim. Genet. 2003, 34, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Hallsson, J.H.; Haflidadóttir, B.S.; Schepsky, A.; Arnheiter, H.; Steingrímsson, E. Evolutionary sequence comparison of the Mitf gene reveals novel conserved domains. Pigment. Cell Res. 2007, 20, 185–200. [Google Scholar] [CrossRef] [PubMed]
- Hodgkinson, C.A.; Moore, K.J.; Nakayama, A.; Steingrímsson, E.; Copeland, N.G.; Jenkins, N.A.; Arnheiter, H. Mutations at the mouse microphthalmia locus are associated with defects in a gene encoding a novel basic-helix-loop-helix-zipper protein. Cell 1993, 74, 395–404. [Google Scholar] [CrossRef]
- Moore, K.J. Insight into the microphthalmia gene. Trends Genet. 1995, 11, 442–448. [Google Scholar] [CrossRef]
- Steingrímsson, E.; Moore, K.J.; Lamoreux, M.L.; Ferré-D’Amaré, A.R.; Burley, S.; Zimring, D.C.S.; Skow, L.; Hodgkinson, C.A.; Arnheiter, H.; Copeland, N.G.; et al. Molecular basis of mouse microphthalmia (mi) mutations helps explain their developmental and phenotypic consequences. Nat. Genet. 1994, 8, 256–263. [Google Scholar] [CrossRef]
- Minvielle, F.; Bed’Hom, B.; Coville, J.-L.; Ito, S.; Inoue-Murayama, M.; Gourichon, D. The “silver” Japanese quail and the MITF gene: Causal mutation, associated traits and homology with the “blue” chicken plumage. BMC Genet. 2010, 11, 15. [Google Scholar] [CrossRef]
- Li, S.; Wang, C.; Yu, W.; Zhao, S.; Gong, Y. Identification of Genes Related to White and Black Plumage Formation by RNA-Seq from White and Black Feather Bulbs in Ducks. PLoS ONE 2012, 7, e36592. [Google Scholar] [CrossRef] [PubMed]
- Lin, R.; Lin, W.; Zhou, S.; Chen, Q.; Pan, J.; Miao, Y.; Zhang, M.; Huang, Z.; Xiao, T. Integrated Analysis of mRNA Expression, CpG Island Methylation, and Polymorphisms in the MITF Gene in Ducks (Anas platyrhynchos). BioMed Res. Int. 2019, 2019, 8512467. [Google Scholar] [CrossRef]
- Sultana, H.; Seo, D.; Choi, N.-R.; Alam Bhuiyan, S.; Lee, S.H.; Heo, K.N.; Lee, J.H. Identification of polymorphisms in MITF and DCT genes and their associations with plumage colors in Asian duck breeds. Asian-Australas. J. Anim. Sci. 2018, 31, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Mo, C.; Shen, W.; Du, X.; Bhuiyan, A.A.; Li, L.; Li, N.; Gong, Y.; Li, S. The recessive C locus in the MITF gene plays a key regulatory role in the plumage colour pattern of duck (Anas platyrhynchos). Br. Poult. Sci. 2019, 60, 105–108. [Google Scholar] [CrossRef]
- Zhang, Z.; Jia, Y.; Almeida, P.; E Mank, J.; van Tuinen, M.; Wang, Q.; Jiang, Z.; Chen, Y.; Zhan, K.; Hou, S.; et al. Whole-genome resequencing reveals signatures of selection and timing of duck domestication. Gigascience 2018, 7, giy027. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Li, M.; Cheng, H.; Fan, W.; Yuan, Z.; Gao, Q.; Xu, Y.; Guo, Z.; Zhang, Y.; Hu, J.; et al. An intercross population study reveals genes associated with body size and plumage color in ducks. Nat. Commun. 2018, 9, 2648. [Google Scholar] [CrossRef]
- Robbins, L.S.; Nadeau, J.H.; Johnson, K.R.; Kelly, M.A.; Roselli-Rehfuss, L.; Baack, E.; Mountjoy, K.G.; Cone, R.D. Pigmentation phenotypes of variant extension locus alleles result from point mutations that alter MSH receptor function. Cell 1993, 72, 827–834. [Google Scholar] [CrossRef]
- Schiöth, H.B.; Raudsepp, T.; Ringholm, A.; Fredriksson, R.; Takeuchi, S.; Larhammar, D.; Chowdhary, B.P. Remarkable synteny conservation of melanocortin receptors in chicken, human, and other vertebrates. Genomics 2003, 81, 504–509. [Google Scholar] [CrossRef]
- Dávila, S.G.; Gil, M.G.; Resino-Talaván, P.; Campo, J.L. Association between polymorphism in the melanocortin 1 receptor gene and E locus plumage color phenotype. Poult. Sci. 2014, 93, 1089–1096. [Google Scholar] [CrossRef]
- Zhang, L.Y.; Huang, M.Y.; Li, Y.; Yang, X.D.; Luo, Y.S.; Shi, X.W. Molecular characteristics of MC1R gene in tile-grey plumage of domestic chicken. Br. Poult. Sci. 2020, 61, 382–389. [Google Scholar] [CrossRef]
- Zhang, X.; Pang, Y.; Zhao, S.; Xu, H.; Li, Y.; Xu, Y.; Guo, Z.; Wang, D. The relationship of plumage colours with MC1R (Melanocortin 1 Receptor) and ASIP (Agouti Signaling Protein) in Japanese quail (Coturnix coturnix japonica). Br. Poult. Sci. 2013, 54, 306–311. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.; Wang, C.; Xin, Q.; Li, S.; Feng, Y.; Peng, X.; Gong, Y. Non-synonymous SNPs in MC1R gene are associated with the extended black variant in domestic ducks (Anas platyrhynchos). Anim. Genet. 2012, 44, 214–216. [Google Scholar] [CrossRef] [PubMed]
- Sultana, H.; Seo, D.-W.; Park, H.-B.; Choi, N.-R.; Hoque, R.; Alam Bhuiyan, S.; Heo, K.-N.; Lee, S.-H.; Lee, J.-H. Identification of MC1R SNPs and their Association with Plumage Colors in Asian Duck. J. Poult. Sci. 2017, 54, 111–120. [Google Scholar] [CrossRef]
- Wright, D.; Rubin, C.-J.; Barrio, A.M.; Schütz, K.; Kerje, S.; Brändström, H.; Kindmark, A.; Jensen, P.; Andersson, L. The genetic architecture of domestication in the chicken: Effects of pleiotropy and linkage. Mol. Ecol. 2010, 19, 5140–5156. [Google Scholar] [CrossRef] [PubMed]
- Cordell, H.J. Epistasis: What it means, what it doesn’t mean, and statistical methods to detect it in humans. Hum. Mol. Genet. 2002, 11, 2463–2468. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Richards, S.; Carbone, M.A.; Zhu, D.; Anholt, R.R.H.; Ayroles, J.F.; Duncan, L.; Jordan, K.W.; Lawrence, F.; Magwire, M.M.; et al. Epistasis dominates the genetic architecture of Drosophila quantitative traits. Proc. Natl. Acad. Sci. USA 2012, 109, 15553–15559. [Google Scholar] [CrossRef]
- Zhang, H.; Yu, J.-Q.; Yang, L.-L.; Kramer, L.M.; Zhang, X.-Y.; Na, W.; Reecy, J.M.; Li, H. Identification of genome-wide SNP-SNP interactions associated with important traits in chicken. BMC Genom. 2017, 18, 892. [Google Scholar] [CrossRef]
- Mackay, T.F.C. Epistasis and quantitative traits: Using model organisms to study gene–gene interactions. Nat. Rev. Genet. 2013, 15, 22–33. [Google Scholar] [CrossRef]
- Manolio, T.A.; Collins, F.S.; Cox, N.J.; Goldstein, D.B.; Hindorff, L.A.; Hunter, D.J.; McCarthy, M.I.; Ramos, E.M.; Cardon, L.R.; Chakravarti, A.; et al. Finding the missing heritability of complex diseases. Nature 2009, 461, 747–753. [Google Scholar] [CrossRef]
- Chen, S.; Zhou, Y.; Chen, Y.; Gu, J. fastp: An ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 2018, 34, i884–i890. [Google Scholar] [CrossRef]
- Li, H.; Durbin, R. Fast and accurate short read alignment with Burrows—Wheeler transform. Bioinformatics 2009, 25, 1754–1760. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Ruan, J.; Durbin, R. Mapping short DNA sequencing reads and calling variants using mapping quality scores. Genome Res. 2008, 18, 1851–1858. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Stephens, M. Genome-wide efficient mixed-model analysis for association studies. Nat. Genet. 2012, 44, 821–824. [Google Scholar] [CrossRef] [PubMed]
- McLaren, W.; Pritchard, B.; Rios, D.; Chen, Y.; Flicek, P.; Cunningham, F. Deriving the consequences of genomic variants with the Ensembl API and SNP Effect Predictor. Bioinformatics 2010, 26, 2069–2070. [Google Scholar] [CrossRef] [PubMed]
- Nadeau, N.J.; Minvielle, F.; Mundy, N.I. Association of a Glu92Lys substitution in MC1R with extended brown in Japanese quail (Coturnix japonica). Anim. Genet. 2006, 37, 287–289. [Google Scholar] [CrossRef]
- Ran, J.-S.; You, X.-Y.; Jin, J.; Zhou, Y.-G.; Wang, Y.; Lan, D.; Ren, P.; Liu, Y.-P. The Relationship between MC1R Mutation and Plumage Color Variation in Pigeons. BioMed Res. Int. 2016, 2016, 3059756. [Google Scholar] [CrossRef]
- Wang, X.; Li, D.; Song, S.; Zhang, Y.; Li, Y.; Wang, X.; Liu, D.; Zhang, C.; Cao, Y.; Fu, Y.; et al. Combined transcriptomics and proteomics forecast analysis for potential genes regulating the Columbian plumage color in chickens. PLoS ONE 2019, 14, e0210850. [Google Scholar] [CrossRef]
- Vachtenheim, J.; Borovanský, J. “Transcription physiology” of pigment formation in melanocytes: Central role of MITF. Exp. Dermatol. 2010, 19, 617–627. [Google Scholar] [CrossRef]
- Guo, X.; Li, X.; Li, Y.; Gu, Z.; Zheng, C.; Wei, Z.; Wang, J.; Zhou, R.; Li, L.; Zheng, H. Genetic variation of chicken MC1R gene in different plumage colour populations. Br. Poult. Sci. 2010, 51, 734–739. [Google Scholar] [CrossRef]
- Guo, Q.; Jiang, Y.; Wang, Z.; Bi, Y.; Chen, G.; Bai, H.; Chang, G. Genome-Wide Analysis Identifies Candidate Genes Encoding Feather Color in Ducks. Genes 2022, 13, 1249. [Google Scholar] [CrossRef]
- Ding, Y.; Berrocal, A.; Morita, T.; Longden, K.D.; Stern, D.L. Natural courtship song variation caused by an intronic retroelement in an ion channel gene. Nature 2016, 536, 329–332. [Google Scholar] [CrossRef] [PubMed]
- Feuk, L.; Carson, A.R.; Scherer, S. Structural variation in the human genome. Nat. Rev. Genet. 2006, 7, 85–97. [Google Scholar] [CrossRef] [PubMed]
- Vialle, R.A.; Lopes, K.D.P.; Bennett, D.A.; Crary, J.F.; Raj, T. Integrating whole-genome sequencing with multi-omic data reveals the impact of structural variants on gene regulation in the human brain. Nat. Neurosci. 2022, 25, 504–514. [Google Scholar] [CrossRef] [PubMed]
- Phillips, P.C. Epistasis—The essential role of gene interactions in the structure and evolution of genetic systems. Nat. Rev. Genet. 2008, 9, 855–867. [Google Scholar] [CrossRef]
- Aoki, H.; Moro, O. Involvement of microphthalmia-associated transcription factor (MITF) in expression of human melanocortin-1 receptor (MC1R). Life Sci. 2002, 71, 2171–2179. [Google Scholar] [CrossRef]
- Dürig, N.; Letko, A.; Lepori, V.; Hadji Rasouliha, S.; Loechel, R.; Kehl, A.; Hytönen, M.K.; Lohi, H.; Mauri, N.; Dietrich, J. Two MC1R loss-of-function alleles in cream-coloured Australian Cattle Dogs and white Huskies. Anim. Genet. 2018, 49, 284–290. [Google Scholar] [CrossRef]
- Lancaster, F.M. Mutations and major variants in domestic ducks. In Poultry Breeding and Genetics; Crawford, R.D., Ed.; Elsevier: Amsterdam, The Netherlands, 1990; pp. 381–388. [Google Scholar]
- Lin, R.L.; Chen, H.P.; Rouvier, R.; Poivey, J.P. Selection and Crossbreeding in Relation to Plumage Color Inheritance in Three Chinese Egg Type Duck Breeds (Anas Platyrhynchos). Asian-Australas. J. Anim. Sci. 2014, 27, 1069–1074. [Google Scholar] [CrossRef]
- Klungland, H.; Våge, D.I.; Gomez-Raya, L.; Adalsteinsson, S.; Lien, S. The role of melanocyte-stimulating hormone (MSH) receptor in bovine coat color determination. Mamm. Genome 1995, 6, 636–639. [Google Scholar] [CrossRef]
- Kijas, J.M.H.; Wales, R.; Törnsten, A.; Chardon, P.; Moller, M.; Andersson, L. Melanocortin Receptor 1 (MC1R) Mutations and Coat Color in Pigs. Genetics 1998, 150, 1177–1185. [Google Scholar] [CrossRef]
- Zhu, T.; Qi, X.; Chen, Y.; Wang, L.; Lv, X.; Yang, W.; Zhang, J.; Li, K.; Ning, Z.; Jiang, Z.; et al. Positive selection of skeleton-related genes during duck domestication revealed by whole genome sequencing. BMC Ecol. Evol. 2021, 21, 165. [Google Scholar] [CrossRef]

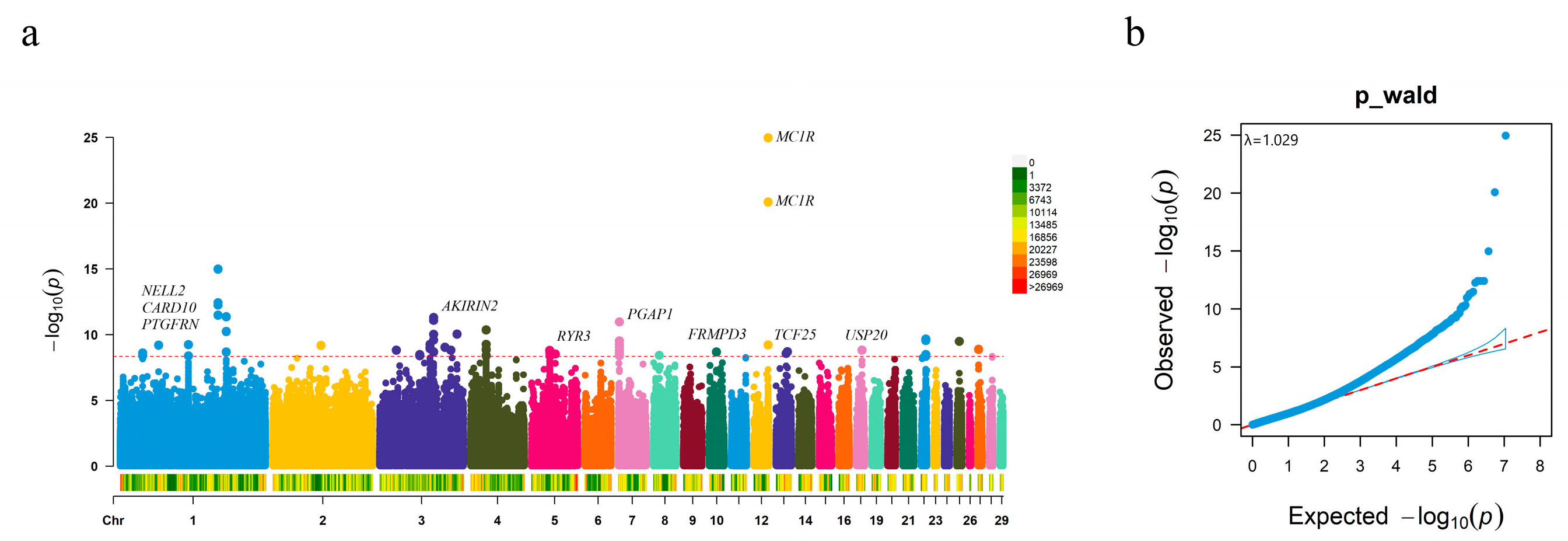
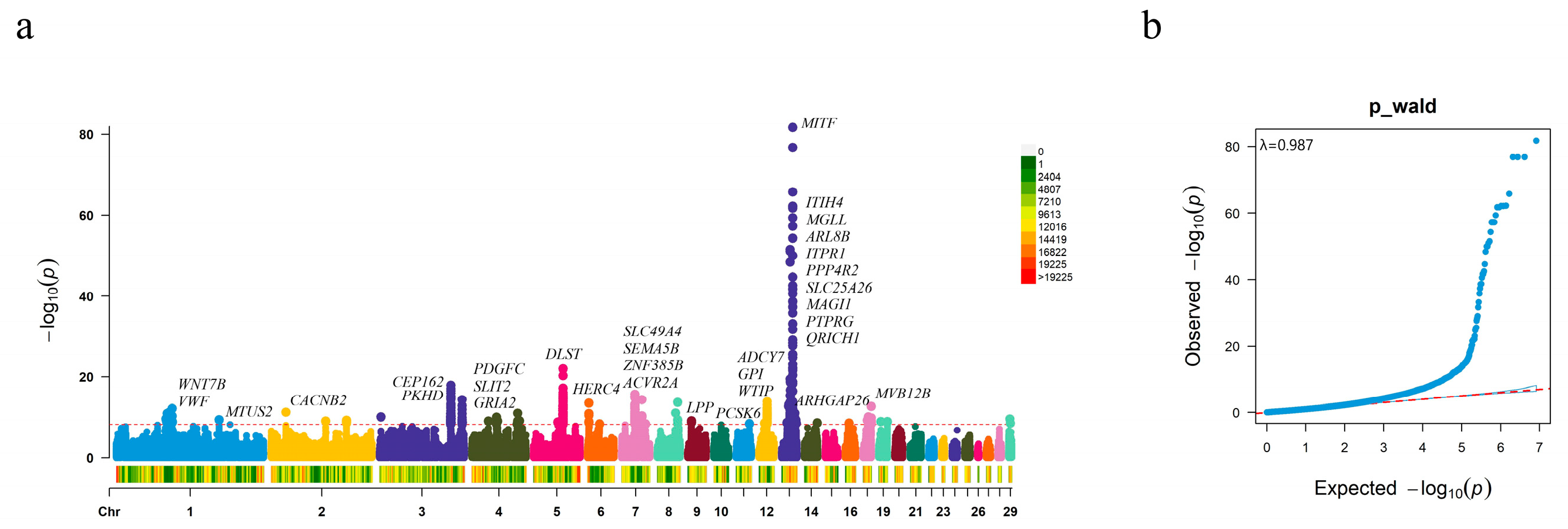
| Population | Geographic Origin | Classification | Number |
|---|---|---|---|
| Spot-billed duck (SB) | Ningxia, China | Spotty plumage | 7 |
| Mallard (MD) | Zhejiang, China | Spotty plumage | 8 |
| Peking duck (PK) | Beijing, China | White plumage | 8 |
| Maple Leaf duck (FY) | Beijing, China | White plumage | 8 |
| Cherry Valley duck (CV) | Beijing, China | White plumage | 8 |
| Wendeng black duck (WD) | Shandong, China | Black plumage | 8 |
| Putian black duck (PT) | Fujian, China | Black plumage | 8 |
| Gaoyou duck (GY) | Jiangsu, China | Spotty plumage | 8 |
| Jinding duck (JD) | Fujian, China | Spotty plumage | 8 |
| Mei duck (MEI) | Anhui, China | Spotty plumage | 9 |
| Shanma duck (SM) | Fujian, China | Spotty plumage | 8 |
| Shaoxing duck (SX) | Jiangsu, China | Spotty plumage | 8 |
| Total | 96 |
| MITF | MC1R | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Phenotype | Breeds | 6.6-kb Intron Insertion | chr13:15411658 A>G | chr13:15412570 T>C | chr13:15412592 C>G | c.52 G>A | c.376 G>A | ||||||||||
| AA | AG | GG | TT | TC | CC | CC | CG | GG | GG | GA | AA | GG | GA | AA | |||
| Recessive white | CV | insertion | 8 | 0 | 0 | 8 | 0 | 0 | 8 | 0 | 0 | 0 | 1 | 6 | 0 | 1 | 7 |
| Recessive white | FY | insertion | 8 | 0 | 0 | 8 | 0 | 0 | 8 | 0 | 0 | 2 | 3 | 3 | 1 | 3 | 4 |
| Recessive white | PK | insertion | 8 | 0 | 0 | 8 | 0 | 0 | 8 | 0 | 0 | 6 | 0 | 2 | 5 | 2 | 1 |
| Wild-type | GY | ⁄ | 0 | 0 | 8 | 0 | 0 | 8 | 0 | 0 | 6 | 8 | 0 | 0 | 8 | 0 | 0 |
| Wild-type | JD | ⁄ | 0 | 0 | 8 | 0 | 0 | 8 | 0 | 0 | 8 | 8 | 0 | 0 | 8 | 0 | 0 |
| Wild-type | MD | ⁄ | 0 | 0 | 8 | 0 | 0 | 8 | 0 | 0 | 8 | 8 | 0 | 0 | 8 | 0 | 0 |
| Wild-type | MEI | ⁄ | 0 | 0 | 9 | 0 | 0 | 9 | 0 | 0 | 9 | 9 | 0 | 0 | 9 | 0 | 0 |
| Wild-type | SM | ⁄ | 0 | 0 | 8 | 0 | 0 | 8 | 0 | 0 | 8 | 8 | 0 | 0 | 8 | 0 | 0 |
| Wild-type | SX | ⁄ | 0 | 0 | 8 | 0 | 0 | 8 | 0 | 0 | 8 | 8 | 0 | 0 | 8 | 0 | 0 |
| Wild-type | SB | ⁄ | 0 | 0 | 7 | 0 | 0 | 7 | 0 | 0 | 7 | 7 | 0 | 0 | 7 | 0 | 0 |
| Extended black | PT | ⁄ | 0 | 1 | 7 | 0 | 1 | 7 | 0 | 1 | 7 | 0 | 3 | 5 | 0 | 3 | 5 |
| Extended black | WD | ⁄ | 0 | 3 | 5 | 0 | 3 | 4 | 0 | 3 | 5 | 2 | 6 | 0 | 1 | 6 | 1 |
| Group | Chr1 | Gene1 | SNP1 | Chr2 | Gene2 | SNP2 | p |
|---|---|---|---|---|---|---|---|
| 13 | MITF | 13:15411658 | 12 | MC1R | 12:20222793 | 0.226 | |
| 13 | MITF | 13:15411658 | 12 | MC1R | 12:20223117 | 0.164 | |
| white vs. non-white | 13 | MITF | 13:15412570 | 12 | MC1R | 12:20222793 | 0.271 |
| 13 | MITF | 13:15412570 | 12 | MC1R | 12:20223117 | 0.212 | |
| 13 | MITF | 13:15412592 | 12 | MC1R | 12:20222793 | 0.298 | |
| 13 | MITF | 13:15412592 | 12 | MC1R | 12:20223117 | 0.234 | |
| 13 | MITF | 13:15411658 | 12 | MC1R | 12:20222793 | 0.451 | |
| 13 | MITF | 13:15411658 | 12 | MC1R | 12:20223117 | 0.509 | |
| white vs. black | 13 | MITF | 13:15412570 | 12 | MC1R | 12:20222793 | 0.438 |
| 13 | MITF | 13:15412570 | 12 | MC1R | 12:20223117 | 0.490 | |
| 13 | MITF | 13:15412592 | 12 | MC1R | 12:20222793 | 0.451 | |
| 13 | MITF | 13:15412592 | 12 | MC1R | 12:20223117 | 0.509 | |
| 13 | MITF | 13:15411658 | 12 | MC1R | 12:20222793 | 0.025 | |
| 13 | MITF | 13:15411658 | 12 | MC1R | 12:20223117 | 0.036 | |
| white vs. spotty | 13 | MITF | 13:15412570 | 12 | MC1R | 12:20222793 | 0.038 |
| 13 | MITF | 13:15412570 | 12 | MC1R | 12:20223117 | 0.054 | |
| 13 | MITF | 13:15412592 | 12 | MC1R | 12:20222793 | 0.041 | |
| 13 | MITF | 13:15412592 | 12 | MC1R | 12:20223117 | 0.057 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, X.; Zhu, T.; Wang, L.; Lv, X.; Yang, W.; Qu, C.; Li, H.; Wang, H.; Ning, Z.; Qu, L. Genome-Wide Association Study Reveals the Genetic Basis of Duck Plumage Colors. Genes 2023, 14, 856. https://doi.org/10.3390/genes14040856
Zhang X, Zhu T, Wang L, Lv X, Yang W, Qu C, Li H, Wang H, Ning Z, Qu L. Genome-Wide Association Study Reveals the Genetic Basis of Duck Plumage Colors. Genes. 2023; 14(4):856. https://doi.org/10.3390/genes14040856
Chicago/Turabian StyleZhang, Xinye, Tao Zhu, Liang Wang, Xueze Lv, Weifang Yang, Changqing Qu, Haiying Li, Huie Wang, Zhonghua Ning, and Lujiang Qu. 2023. "Genome-Wide Association Study Reveals the Genetic Basis of Duck Plumage Colors" Genes 14, no. 4: 856. https://doi.org/10.3390/genes14040856
APA StyleZhang, X., Zhu, T., Wang, L., Lv, X., Yang, W., Qu, C., Li, H., Wang, H., Ning, Z., & Qu, L. (2023). Genome-Wide Association Study Reveals the Genetic Basis of Duck Plumage Colors. Genes, 14(4), 856. https://doi.org/10.3390/genes14040856






