Translational Bioinformatics Applied to the Study of Complex Diseases
Abstract
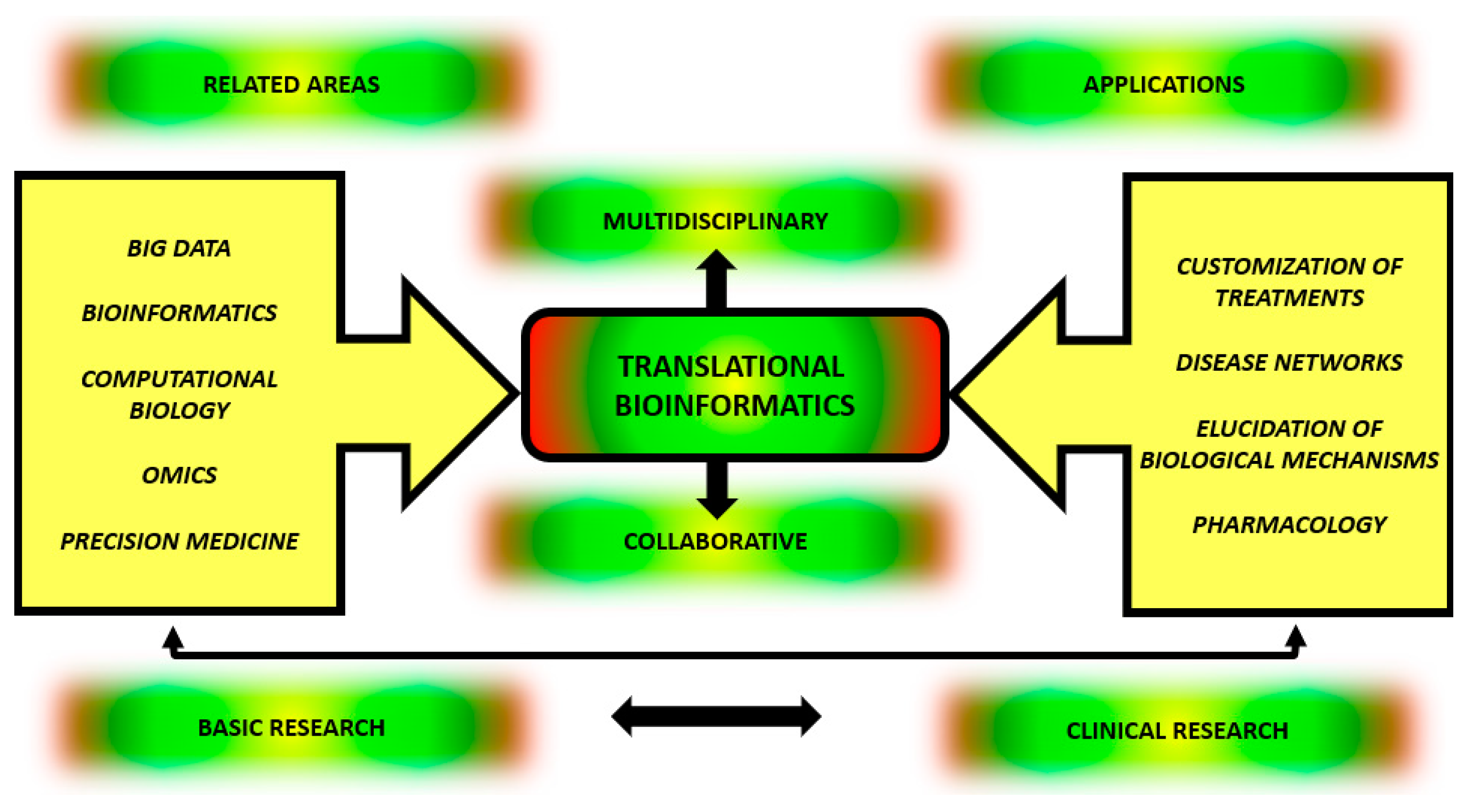
1. Introduction
2. TBI: From Definition to Application
| Authors | Study Description |
|---|---|
| Ahmed [13]; Kang; Ko; Mersha [14]; Savoska; Ristevski; Trajkovik [15]. | Integration of collective and individualized clinical data with patient-specific multi-omic data, AI algorithms and cloud electronic health record databases. |
| Baruah; Deka; Mahanta [16]. | Multidisciplinary cooperation between laboratory and clinical researchers, aiming to meet community needs. |
| Bellazzi et al. [17]. | Clinical bioinformatics (CBI) seeks to integrate molecular and clinical data, using bioinformatics to understand molecular mechanisms and potential therapies. |
| Bruggemann et al. [18]. | Pharmacogenomics provides personalized patient care by selecting specific drugs for diseases, such as non-small cell lung cancer. |
| Han; Liu [19]. | AI unravelling latent data behavior and generating new insights and optimal strategies in decision-making. |
| Liu et al. [20]. | TBI used in reproductive medicine. |
| Liu; Wang; Lai [21]. | Single-cell total RNA sequencing (scRNA-seq) and bulk total RNA sequencing (RNA-seq) associated with machine learning for detection of tumor origin. |
| Lussier; Butte; Hunter [22]. | TBI effects on: (i) availability and cost reduction of molecular measurements; (ii) accessibility to measurements of health and disease status; (iii) sharing data and molecular tools; (iv) interpretation of new clinical molecular discoveries; (v) research funding. |
| Mitra et al. [23]. | In silico analysis of acquired, archived, and retrieved biological data, as well as dynamic molecular docking experiments affecting pharmacology, biotechnology, bioengineering and education. |
| Sheikh; Ramlal; Khan [24]. | Predictive modeling of antineoplastic drugs through computational analysis of microarrays. |
| Tang et al. [25]. | TBI encompassing issues of equity and inclusion, by means of phenotyping applications, characterization of disease subtypes, predictive modeling, biomarker discovery and selection of better treatments. |
| Torkamannia; Omidi; Ferdousi [26]. | Combined pharmacotherapy with synergistic/additive effect as a powerful treatment strategy for complex diseases. |
| Yang et al. [27]. | High-throughput technologies/TBI in the biological interpretation of Big Data, uncovering mechanistic landscapes of complex diseases. |
3. Translational Biomedical Knowledge
4. Precision Medicine and TBI
5. Omics Revolution in Complex Diseases
6. Integrating Complex Diseases through TBI
7. Application of TBI in Complex Diseases
7.1. Relationship between Next-Generation Sequencing (NGS) and TBI in the Study of Complex Diseases
7.2. Multi-Omics, Single Cells, and TBI in the Study of Complex Diseases
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lin, H.; Peng, S.; Huang, J. Special issue on computational resources and methods in biological sciences. Int. J. Biol. Sci. 2018, 14, 807–810. [Google Scholar] [CrossRef] [PubMed]
- Robinson, P.N. Genomic data sharing for translational research and diagnostics. Genome Med. 2014, 6, 78. [Google Scholar] [CrossRef]
- Londin, E.R.; Barash, C.I. What is Translational Bioinformatics? Appl. Transl. Genom. 2015, 6, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Mooney, S.D.; Tenenbaum, J.D.; Altman, R.B. Translational Bioinformatics. In Biomedical Informatics: Computer Applications in Health Care and Biomedicine; Springer: Berlin/Heidelberg, Germany, 2014; pp. 867–911. [Google Scholar]
- Peng, J.; Bai, K.; Shang, X.; Wang, G.; Xue, H.; Jin, S.; Cheng, L.; Wang, Y.; Chen, J. Predicting disease-related genes using integrated biomedical networks. BMC Genom. 2017, 18, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Kimmel, C.; Visweswaran, S. An algorithm for network-based gene prioritization that encodes knowledge both in nodes and in links. PLoS ONE 2013, 8, e79564. [Google Scholar] [CrossRef] [PubMed]
- Bellazzi, R. Big data and biomedical informatics: A challenging opportunity. Yearb. Med. Inform. 2014, 23, 8–13. [Google Scholar] [CrossRef]
- Azuaje, F. Computational models for predicting drug responses in cancer research. Brief. Bioinform. 2017, 18, 820–829. [Google Scholar] [CrossRef]
- Tenenbaum, J.D. Translational Bioinformatics: Past, present, and future. Genom. Proteom. Bioinform. 2016, 14, 31–41. [Google Scholar] [CrossRef]
- Sarkar, I.N. Biomedical informatics and translational medicine. J. Transl. Med. 2010, 8, 22. [Google Scholar] [CrossRef]
- Altman, R.B. TBI: Linking the molecular world to the clinical world. Clin. Pharmacol. Ther. 2012, 91, 994–1000. [Google Scholar] [CrossRef]
- Su, C.; Tong, J.; Zhu, Y.; Cui, P.; Wang, F. Network embedding in biomedical data science. Brief. Bioinform. 2020, 21, 182–197. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, Z. Multi-omics strategies for personalized and predictive medicine: Past, current, and future translational opportunities. Emerg. Top. Life Sci. 2022, 6, 215–225. [Google Scholar] [CrossRef]
- Kang, M.; Ko, E.; Mersha, T.B. A roadmap for multi-omics data integration using deep learning. Brief. Bioinform. 2022, 23, bbab454. [Google Scholar] [CrossRef] [PubMed]
- Savoska, S.; Ristevski, B.; Trajkovik, V. Personal Health Record Data-Driven Integration of Heterogeneous Data. In Data-Driven Approach for Bio-Medical and Healthcare; Springer: Singapore, 2023; 21p. [Google Scholar]
- Baruah, C.; Deka, B.; Mahanta, S. A Review of Recent Advances in TBI and Systems Biomedicine. Inf. Retr. Bioinform. 2022, 1, 37–62. [Google Scholar]
- Bellazzi, R.; Masseroli, M.; Murphy, S.; Shabo, A.; Romano, P. Clinical Bioinformatics: Challenges and opportunities. BMC Bioinform. 2012, 13, S1. [Google Scholar] [CrossRef] [PubMed]
- Bruggemann, L.; Falls, Z.; Mangione, W.; Schwartz, S.A.; Battaglia, S.; Aalinkeel, R.; Mahajan, S.D.; Samudrala, R. Multiscale Analysis and Validation of Effective Drug Combinations Targeting Driver KRAS Mutations in Non-Small Cell Lung Cancer. Int. J. Mol. Sci. 2023, 24, 997. [Google Scholar] [CrossRef]
- Han, H.; Liu, X. The challenges of explainable AI in biomedical data science. BMC Bioinform. 2022, 22, 1–3. [Google Scholar] [CrossRef]
- Liu, K.; Zhang, Y.; Martin, C.; Ma, X.; Shen, B. Translational Bioinformatics for Human Reproductive Biology Research: Examples, Opportunities and Challenges for a Future Reproductive Medicine. Int. J. Mol. Sci. 2023, 24, 4. [Google Scholar] [CrossRef]
- Liu, H.-P.; Wang, D.; Lai, H.-M. Can we infer tumor presence of single cell transcriptomes and their tumor of origin from bulk transcriptomes by machine learning? Comput. Struct. Biotechnol. J. 2022, 20, 2672–2679. [Google Scholar] [CrossRef]
- Lussier, Y.A.; Butte, A.J.; Hunter, L. Current methodologies for TBI. J. Biomed. Inform. 2010, 43, 355. [Google Scholar] [CrossRef]
- Mitra, D.; Mitra, D.; Bensaad, M.S.; Sinha, S.; Pant, K.; Pant, M.; Priyadarshini, A.; Singh, P.; Dassamiour, S.; Hambaba, L.; et al. Evolution of Bioinformatics and its impact on modern bio-science in the twenty-first century: Special attention to pharmacology, plant science and drug discovery. Comput. Toxicol. 2022, 24, 100248. [Google Scholar] [CrossRef]
- Sheikh, K.; Ramlal, A.; Khan, F.N. Computational Resources for Oncology Research: A Comprehensive Analysis. In Computational Intelligence in Oncology; Springer: Singapore, 2022; Volume 1016, pp. 65–92. [Google Scholar]
- Tang, A.; Woldemariam, S.; Roger, J.; Sirota, M. Translational Bioinformatics to Enable Precision Medicine for All: Elevating Equity across Molecular, Clinical, and Digital Realms. Yearb. Med. Inform. 2022, 31, 106–115. [Google Scholar] [CrossRef]
- Torkamannia, A.; Omidi, Y.; Ferdousi, R. A review of machine learning approaches for drug synergy prediction in cancer. Brief. Bioinform. 2022, 23, bbac075. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.Y.; Dunker, A.K.; Liu, J.S.; Qin, X.; Arabnia, H.R.; Yang, W.; Niemierko, A.; Chen, Z.; Luo, Z.; Wang, L.; et al. Advances in Translational Bioinformatics facilitate revealing the landscape of complex disease mechanisms. BMC Bioinform. 2014, 15, I1. [Google Scholar] [CrossRef]
- Smaïl-Tabbone, M.; Rance, B. Contributions from the 2018 literature on bioinformatics and translational informatics. Yearb. Med. Inform. 2019, 28, 190–193. [Google Scholar] [CrossRef]
- Smaïl-Tabbone, M.; Rance, B. Contributions from the 2019 Literature on Bioinformatics and Translational Informatics. Yearb. Med. Inform. 2020, 29, 188–192. [Google Scholar] [CrossRef]
- Hutchins, B.I.; Davis, M.T.; Meseroll, R.A.; Santangelo, G.M. Predicting translational progress in biomedical research. PLoS Biol. 2019, 17, e3000416. [Google Scholar] [CrossRef]
- Shameer, K.; Badgeley, M.A.; Miotto, R.; Glicksberg, B.S.; Morgan, J.W.; Dudley, J.T. Translational Bioinformatics in the era of real-time biomedical, health care and wellness data streams. Brief. Bioinform. 2017, 18, 105–124. [Google Scholar] [CrossRef]
- Wu, H.Y.; Shendre, A.; Zhang, S.; Zhang, P.; Wang, L.; Zeruesenay, D.; Rocha, L.M.; Shatkay, H.; Quinney, S.K.; Ning, X.; et al. Translational knowledge discovery between drug interactions and pharmacogenetics. Clin. Pharmacol. Ther. 2020, 107, 886–902. [Google Scholar] [CrossRef]
- König, I.R.; Fuchs, O.; Hansen, G.; von Mutius, E.; Kopp, M.V. What is precision medicine? Eur. Respir. J. 2017, 50, 4. [Google Scholar] [CrossRef]
- Kosorok, M.R.; Laber, E.B. Precision medicine. Annu. Rev. Stat. Its Appl. 2019, 6, 263–286. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Ma, D.; Yang, Y.S.; Yang, F.; Ding, J.H.; Gong, Y.; Jiang, L.; Ge, L.P.; Wu, S.Y.; Yu, Q.; et al. Comprehensive metabolomics expands precision medicine for triple-negative breast cancer. Cell Res. 2022, 32, 477–490. [Google Scholar] [CrossRef] [PubMed]
- Milluzzo, A.; Vigneri, P.; Martorana, F.; Vigneri, R.; Sciacca, L. Type 2 diabetes and cancer: Problems and suggestions for best patient management. Explor. Med. 2020, 1, 184–204. [Google Scholar] [CrossRef]
- Qazi, S.; Raza, K. Translational Bioinformatics in healthcare: Past, present, and future. In Translational Bioinformatics in Healthcare and Medicine; Academic Press: Cambridge, MA, USA, 2021; Volume 13, p. 12. [Google Scholar]
- Djordjevic, M.; Rodic, A.; Graovac, S. From biophysics to ‘omics and systems biology. Eur. Biophys. J. 2019, 48, 413–424. [Google Scholar] [CrossRef]
- Noor, E.; Cherkaoui, S.; Sauer, U. Biological insights through omics data integration. Curr. Opin. Syst. Biol. 2019, 15, 39–47. [Google Scholar] [CrossRef]
- Parsons, J.; Francavilla, C. ′Omics approaches to explore the breast cancer landscape. Front. Cell Dev. Biol. 2020, 7, 395. [Google Scholar] [CrossRef]
- Peng, A.; Mao, X.; Zhong, J.; Fan, S.; Hu, Y. Single-Cell Multi-Omics and Its Prospective Application in Cancer Biology. Proteomics 2020, 20, 1900271. [Google Scholar] [CrossRef]
- Subramanian, I.; Verma, S.; Kumar, S.; Jere, A.; Anamika, K. Multi-omics data integration, interpretation, and its application. Bioinform. Biol. Insights 2020, 14, 1177932219899051. [Google Scholar] [CrossRef]
- Hinderer III, E.W.; Flight, R.M.; Dubey, R.; MacLeod, J.N.; Moseley, H.N. Advances in gene ontology utilization improve statistical power of annotation enrichment. PLoS ONE 2019, 14, e0220728. [Google Scholar] [CrossRef]
- Gene Ontology Consortium. The gene ontology resource: 20 years and still GOing strong. Nucleic Acids Res. 2019, 47, D330–D338. [Google Scholar] [CrossRef]
- Manda, P. Data mining powered by the gene ontology. Wiley Interdiscip. Rev. Data Min. Knowl. Discov. 2020, 10, e1359. [Google Scholar] [CrossRef]
- Wang, L.L. Ontology-driven pathway data integration. Ph.D. Dissertation, University of Washington, Seattle, WA, USA, 2019. [Google Scholar]
- Wang, L.; Wang, B.; Quan, Z. Identification of aberrantly methylateddifferentially expressed genes and gene ontology in prostate cancer. Mol. Med. Rep. 2020, 21, 744–758. [Google Scholar] [PubMed]
- Schriml, L.M.; Mitraka, E.; Munro, J.; Tauber, B.; Schor, M.; Nickle, L.; Felix, V.; Jeng, L.; Bearer, C.; Lichenstein, R.; et al. Human Disease Ontology 2018 update: Classification, content and workflow expansion. Nucleic Acids Res. 2019, 47, D955–D962. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Singh, A.P.; Gupta, D. Unsupervised subtyping and methylation landscape of pancreatic ductal adenocarcinoma. Heliyon 2021, 7, e06000. [Google Scholar] [CrossRef]
- Liu, D.; Li, B.; Shi, X.; Zhang, J.; Chen, A.M.; Xu, J.; Wang, W.; Huang, K.; Gao, J.; Zheng, Z.; et al. Cross-platform genomic identification and clinical validation of breast cancer diagnostic biomarkers. Aging 2021, 13, 4258. [Google Scholar] [CrossRef]
- Cai, Z.; Guldbrandtsen, B.; Lund, M.S.; Sahana, G. Prioritizing candidate genes post-GWAS using multiple sources of data for mastitis resistance in dairy cattle. BMC Genom. 2018, 19, 656. [Google Scholar] [CrossRef]
- Yu, L.; Zhao, J.; Gao, L. Predicting potential drugs for breast cancer based on miRNA and tissue specificity. Int. J. Biol. Sci. 2018, 14, 971–982. [Google Scholar] [CrossRef]
- Zheng, X.; Wang, X.; Zheng, L.; Zhao, H.; Li, W.; Wang, B.; Xue, L.; Tian, Y.; Xie, Y. Construction and analysis of the tumor-specific mRNA–miRNA–lncRNA network in gastric cancer. Front. Pharmacol. 2020, 11, 1112. [Google Scholar] [CrossRef]
- Zhou, H.; Chen, J.X.; Yang, C.S.; Yang, M.Q.; Deng, Y.; Wang, H. Gene regulation mediated by microRNAs in response to green tea polyphenol EGCG in mouse lung cancer. BMC Genom. 2014, 15, S3. [Google Scholar] [CrossRef]
- Akgün, M.; Ünal, A.B.; Ergüner, B.; Pfeifer, N.; Kohlbacher, O. Identifying disease-causing mutations with privacy protection. Bioinformatics 2020, 36, 5205–5213. [Google Scholar] [CrossRef]
- Immel, A.; Key, F.M.; Szolek, A.; Barquera, R.; Robinson, M.K.; Harrison, G.F.; Palmer, W.H.; Spyrou, M.A.; Susat, J.; Krause-Kyora, B.; et al. Analysis of genomic DNA from medieval plague victims suggests long-term effect of Yersinia pestis on human immunity genes. Mol. Biol. Evol. 2021, 38, 4059–4076. [Google Scholar] [CrossRef]
- Pooja, M.; Reddy, G.J.; Hema, K.; Dodoala, S.; Koganti, B. Unravelling high-affinity binding compounds towards transmembrane protease serine 2 enzyme in treating SARS-CoV-2 infection using molecular modelling and docking studies. Eur. J. Pharmacol. 2021, 890, 173688. [Google Scholar]
- Jaballah, A.; Soltani, I.; Bahia, W.; Dandana, A.; Hasni, Y.; Miled, A.; Ferchichi, S. The Relationship Between Menopause and Metabolic Syndrome: Experimental and Bioinformatics Analysis. Biochem. Genet. 2021, 59, 1558–1581. [Google Scholar] [CrossRef] [PubMed]
- Djeddi, S.; Reiss, D.; Menuet, A.; Freismuth, S.; de Carvalho Neves, J.; Djerroud, S.; Massana-Muñoz, X.; Sosson, A.S.; Kretz, C.; Raffelsberger, W.; et al. Multi-omics comparisons of different forms of centronuclear myopathies and the effects of several therapeutic strategies. Mol. Ther. 2021, 29, 2514–2534. [Google Scholar] [CrossRef]
- Huang, Z.; Han, Z.; Wang, T.; Shao, W.; Xiang, S.; Salama, P.; Rizkalla, M.; Huang, K.; Zhang, J. TSUNAMI: Translational Bioinformatics tool suite for network analysis and mining. Genom. Proteom. Bioinform. 2021, 19, 1023–1031. [Google Scholar] [CrossRef] [PubMed]
- Thalor, A.; Joon, H.K.; Singh, G.; Roy, S.; Gupta, D. Machine learning assisted analysis of breast cancer gene expression profiles reveals novel potential prognostic biomarkers for triple-negative breast cancer. Comput. Struct. Biotechnol. J. 2022, 20, 1618–1631. [Google Scholar] [CrossRef] [PubMed]
- Ullah, M.A.; Islam, N.N.; Sarkar, B.; Araf, Y.; Zohora, U.S.; Rahman, M.S. Bioinformatics analysis on the prognostic significance of SOX9 gene and its transcriptional product in colorectal cancer. Hum. Gene 2022, 33, 201043. [Google Scholar] [CrossRef]
- Kaur, P. Cancer informatics survey of different grades and subtypes of glioma (P14-9.003). Neurology 2022, 98, 825. [Google Scholar]
- Yi, S.; Yan, Y.; Jin, M.; Bhattacharya, S.; Wang, Y.; Wu, Y.; Yang, L.; Gine, E.; Clot, G.; Chen, L.; et al. Genomic and transcriptomic profiling reveals distinct molecular subsets associated with outcomes in mantle cell lymphoma. J. Clin. Investig. 2022, 132, 3. [Google Scholar] [CrossRef]
- Yu, L.; Lopez, G.; Rassa, J.; Wang, Y.; Basavanhally, T.; Browne, A.; Huang, C.P.; Dorsey, L.; Jen, J.; Hersey, S. Direct comparison of circulating tumor DNA sequencing assays with targeted large gene panels. PLoS ONE 2022, 17, e0266889. [Google Scholar] [CrossRef]
- Xu, H.; Buckeridge, D.L.; Wang, F.; Tarczy-Hornoch, P. Novel Informatics Approaches to COVID-Research: From methods to applications. J. Biomed. Inform. 2022, 129, 104028. [Google Scholar] [CrossRef] [PubMed]
- Battineni, G.; Hossain, M.A.; Chintalapudi, N.; Amenta, F. A Survey on the Role of Artificial Intelligence in Biobanking Studies. Diagnostics 2022, 12, 1179. [Google Scholar] [CrossRef] [PubMed]
- Branco, I.; Choupina, A. Bioinformatics: New tools and applications in life science and personalized medicine. Appl. Microbiol. Biotechnol. 2021, 105, 937–951. [Google Scholar] [CrossRef] [PubMed]
- Pervez, M.T.; Abbas, S.H.; Moustafa, M.F.; Aslam, N.; Shah, S.S.M. A Comprehensive Review of Performance of Next-Generation Sequencing Platforms. BioMed Res. Int. 2022, 2022, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Pereira, R.; Oliveira, J.; Sousa, M. Bioinformatics and computational tools for next-generation sequencing analysis in clinical genetics. J. Clin. Med. 2020, 9, 132. [Google Scholar] [CrossRef]
- Petersen, L.M.; Martin, I.W.; Moschetti, W.E.; Kershaw, C.M.; Tsongalis, G.J. Third-generation sequencing in the clinical laboratory: Exploring the advantages and challenges of nanopore sequencing. J. Clin. Microbiol. 2019, 58, e01315-19. [Google Scholar] [CrossRef]
- Suzuki, Y. Advent of a new sequencing era: Long-read and on-site sequencing. J. Hum. Genet. 2020, 65, 1. [Google Scholar] [CrossRef]
- Xiao, T.; Zhou, W. The third generation sequencing: The advanced approach to genetic diseases. Transl. Pediatr. 2020, 9, 163. [Google Scholar] [CrossRef]
- Klee, E.W.; Cousin, M.A.; Pinto e Vairo, F.; Morales-Rosado, J.A.; Macke, E.L.; Jenkinson, W.G.; Ferrer, A.; Schultz-Rogers, L.E.; Olson, R.J.; Oliver, G.R.; et al. Impact of integrated translational research on clinical exome sequencing. Genet. Med. 2021, 23, 498–507. [Google Scholar] [CrossRef]
- Lightbody, G.; Haberland, V.; Browne, F.; Taggart, L.; Zheng, H.; Parkes, E.; Blayney, J.K. Review of applications of high-throughput sequencing in personalized medicine: Barriers and facilitators of future progress in research and clinical application. Brief. Bioinform. 2019, 20, 1795–1811. [Google Scholar] [CrossRef]
- Sarma, M.K.; Ningthoujam, R.; Panda, M.K.; Babu, P.J.; Srivastava, A.; Das, M.; Singh, Y.D. Translational healthcare system through bioinformatics. In Translational Bioinformatics Applications in Healthcare; CRC Press: Boca Raton, FL, USA, 2021; Volume 1, pp. 3–21. [Google Scholar]
- Hu, Y.; An, Q.; Sheu, K.; Trejo, B.; Fan, S.; Guo, Y. Single cell multi-omics technology: Methodology and application. Front. Cell Dev. Biol. 2018, 6, 28. [Google Scholar] [CrossRef] [PubMed]
- Bock, C.; Farlik, M.; Sheffield, N.C. Multi-omics of single cells: Strategies and applications. Trends Biotechnol. 2016, 34, 605–608. [Google Scholar] [CrossRef] [PubMed]
- Kashima, Y.; Sakamoto, Y.; Kaneko, K.; Seki, M.; Suzuki, Y.; Suzuki, A. Single-cell sequencing techniques from individual to multiomics analyses. Exp. Mol. Med. 2020, 52, 1419–1427. [Google Scholar] [CrossRef]
- Nam, A.S.; Chaligne, R.; Landau, D.A. Integrating genetic and non-genetic determinants of cancer evolution by single-cell multi-omics. Nat. Rev. Genet. 2021, 22, 3–18. [Google Scholar] [CrossRef] [PubMed]

| Study Title | Study Purpose | Conditions | Interventions | Locations | Study Number |
|---|---|---|---|---|---|
| Preliminary Study on Plasma Markers for Early Diagnosis of Lung Cancer | Molecular features of liquid biopsy and clinical data from lung cancer patients using multi-omics assays, validated a Machine Learning method able to discriminate lung cancer patients from healthy subjects. | Lung Cancer | Diagnostic test: A machine-learning tool to detect early-stage lung cancer. | China | NCT04558255 |
| Advanced Therapies for Liver Metastases | State-of-the-art omics used to characterize the immune and non-immune microenvironment of primary tumor and liver metastases, correlating with activation status of peripheral blood leukocytes. | Pancreatic Ductal Adenocarcinoma, Colorectal Cancer, Liver Metastasis | Not provided. | Italy | NCT04622423 |
| AI used to optimize early-stage hepatocellular carcinoma treatment based on multi-modal imaging. | AI omics imaging, traditional omics imaging and clinical information used to predict prognosis of different treatment options for early liver cancer. | Hepatocellular Carcinoma | Method: Contrast-enhanced ultrasound (CEUS) and Contrast-enhanced MRI examination (CEMRI). | China | NCT05627297 |
| Electronic cigarettes cancer risk | Integrative multiomics approach used to determine the carcinogenic potential of e-cig, relative to cigarette smoking in oral epithelium. | Cigarette Smoking | Device: NIDA Standard Research E-cigarette (SREC) | United States | NCT03750825 |
| Artificial Intelligence system for assessment of tumor risk, diagnosis, and treatment | AI used to establish a medical database of standardized and structured clinical diagnosis based on multiomics information. | Lung, Stomach, and Colon Cancer | Not provided | China | NCT05426135 |
| Liquid Biopsy early detection of Gastric cancer | Prospective, observational, multiomics study focused on detection of gastric cancer by combined assays using serum protein markers, cfDNA deep sequencing, ctDNA mutation and circulating RNA. Bioinformatics tools like PEAR and Bismark are used to process FASTQ files. | Gastric Cancer | Diagnostic test: blood-based biomarkers | China | NCT05224596 |
| Liquid biopsy early detection of pancreatic cancer (ASCEND-PANCREATIC) | Multiomic prospective study aims to detect early pancreatic cancer using combined assays for cfDNA methylation biomarkers, circulating tumor DNA (ctDNA) mutations, serum protein and miRNA markers using bioinformatics tools PEAR and Bismark. | Pancreatic Cancer | Not provided | China | NCT05556603 |
| Breast Cancer, Omics, and Precision Medicine (BR(E)2ASTOME) | To evaluate the clinical utility of the early use of network-oriented BR(E)2ASTOME algorithm which combines the power of liquid-based assays, advanced epi-genomics, and network analysis to improve personalized therapies in BC. | Breast Cancer | Biological: Next-generation sequencing and network analysis | Italy | NCT04996836 |
| Comprehensive Omics Analysis of Pediatric Solid Tumors/Repository for Related Biological Studies | To create a repository of blood, serum, tissue, urine, and tumors to perform omics profiling. | Sarcoma, Endocrine Tumors, Neuroblastoma, Retinoblastoma, Renal Cancer | Not provided | United States | NCT01109394 |
| Concurrent Radiochemotherapy and Anlotinib for Locally Advanced Cervical Cancer | To observe the efficacy and safety of a new treatment option for cervical cancer. Multi-omics technology and bioinformatics tools were used to analyze the patients. | Uterine Cervical Cancer | Drug: Hydrochloride anlotinib, Drug: cis Platinum/carboplatin, Radiation: External beam radiotherapy and brachytherapy | China | NCT04772001 |
| Lethal Prostate Cancer Biology–Urine Metabolomics | A prospective, observational and investigational study that aims to find new markers/targets for screening prostate cancer. | Prostate Cancer | Dietary supplement: Multi-carotenoids | Taiwan | NCT03237702 |
| Artificial Intelligence System for Pathological Diagnosis and Therapeutic Effect Prediction Based on Multimodal Data Fusion of Common Tumors and Major Infectious Diseases in the Respiratory System. | To create a large medical database that includes standardized and structured clinical diagnosis and treatment information, and to develop a multi-modal data fusion-based technology system for individualized intelligent pathological diagnosis and therapeutic effect prediction using artificial intelligence technology. | Lung Cancer, Pulmonary Tuberculosis, Covid19 | Not provided | China | NCT05046366 |
| Early Diagnosis of Small Pulmonary Nodules by Multi-omics | To analyze the immunological repertoire and genetic mutations of pulmonary nodules using imaging tests, three-dimensional reconstruction, bioinformatics R-scripts and algorithms (OptiType algorithm). | Non-small-cell Lung Cancer | Not provided | China | NCT03320044 |
| AI early screening of Colorectal Cancer Based on Plasma Multi-omics. | AI algorithm to evaluate accuracy and effectiveness of a novel screening method based on plasma multi-omics to detect colorectal cancer and advanced adenomas. | Colorectal Adenoma, Colorectal Cancer | Diagnostic test: Colonoscopy, Diagnostic test: ctDNA methylation, Diagnostic test: characteristics of ctDNA fragment | China | NCT05587452 |
| Evaluation of Clinical Treatment of Multiple Myeloma Based on Multi-omics | Multi-omics method to find biomarkers of clinical efficacy, adverse reactions, and blood concentration of bortezomib in peripheral blood samples. | Multiple Myeloma, Bortezomib | Drug: Bortezomib | China | NCT04678089 |
| Gut Microbiota and Metabolomic (MBS) | To evaluate the correlation between intestinal microbiota and metabolites in Borrmann type IV gastric cancer and to use Machine Learning to build models of intestinal microbiota and metabolomics. | Stomach Neoplasms, Gut Microbiota, Metabolomics | Procedure: Healthy control specimen collection, Procedure: Non-Borrmann IV patient specimen collection, Procedure: Borrmann IV patient specimen collection | China | NCT05205187 |
| I3LUNG: Integrative Science, Intelligent Data Platform for Individualized lung cancer immunotherapy | AI international project that aims to predict immunotherapy efficacy for NSCLC patients using the integration of multisource data (real-word and multi-omics data). | Lung Cancer | Not provided | United States, Greece, Israel, Spain | NCT05537922 |
| Integrative Omics Analysis for Colorectal Cancer and Metastasis | Integrative omics to analyze and predict candidate biomarkers of colorectal cancer and distant metastasis. | Colorectal Cancer | Other: Integrative omics | China | NCT05482529 |
| Multi-omics Characterization of Pancreatic Neuroendocrine Tumors | Integrated multi-omics to identify tumor subgroups in pancreatic neuroendocrine tumors and carcinomas regardless of their grade and stage. | Cancer of Pancreas | Not provided | France | NCT05234450 |
| Multi-omics Sequencing in Neoadjuvant Immunotherapy of Gastrointestinal Tumors | To find new biomarkers of efficacy of combined immunotherapy. | Immunotherapy, Gastric Cancer, Rectal Cancer, Chemotherapy Effect, Radiotherapy | Drug: Terelizumab, Drug: CapeOx, Drug: Trastuzumab, Radiation: Radiotherapy | China | NCT05515796 |
| Multi-Omics Noninvasive Inspection of Tumor Risk for Gastric Cancer | A prospective, case-control study intended to develop and validate a blood-based multi-omics assay and computational model for early detection of gastric cancer. | Gastric Cancer | Device: ctDNA multi-omics test | China | NCT04947995 |
| Omics of Cancer: Onco Genomics | To create a registry of genomic/clinical data of cancer or cancer predisposition patients. | Neoplasms Cancer | Not provided | United States | NCT05431439 |
| Pleural Carcinomatosis Tissue Banking | To create a biocollection of tissues from pleural carcinomatosis and characterize intratumoral heterogeneity through multi-omics and bioinformatics analysis. | Pleural Effusion, Malignant | Pleural biopsies | France | NCT04844827 |
| Predictive Biomarkers in Patients with Advanced Hepatocellular Carcinoma Treated with Systemic Therapy | Multi-omics to find biomarkers of treatment response in hepatocellular carcinoma (HCC). | Hepatocellular Carcinoma | Drug: atezolizumab plus bevacizumab | Korea | NCT05197504 |
| Project CADENCE (CAncer Detected Early caN be CurEd) (CADENCE) | To develop and validate multi-cancer screening tests based on multi-omics (single-cell early cancer detection algorithms). | Thoracic, Ovarian, Liver, Prostate, Gastric, Colorectal, Breast, Esophageal, and Pancreatic Cancer | Not provided | Singapore | NCT05633342 |
| Prospectively Predict Gastrointestinal Tumor Treatment Efficacy Based on Peripheral Multi-omics Liquid Biopsy | To predict and monitor immunotherapeutic outcomes of gastrointestinal tumors. | Advanced Gastric Adenocarcinoma, Immunotherapy | Device: EV-array | China | NCT04993378 |
| ML radiomic and pathomic study of Pituitary Adenomas | Machine learning to study multi-dimensional and multi-omics data, to train a risk prediction algorithm for refractory pituitary adenomas. | Pituitary Neoplasms | Diagnostic test: Artificial intelligence model | China | NCT05108064 |
| Multi-omics immune prevention and treatment of gliomas | Omics sequencing and molecular biology technologies to study glioma treatment efficacy. | Transcriptomics, Radiomics, Glioma | Procedure: surgery | China | NCT04792437 |
| Esophageal Cancer Neoadjuvant Chemoradiation response prediction using Artificial Intelligence & Machine Learning (QARC) | To predict treatment response in esophageal cancer patients using radiomics AI modeling. | Esophageal Cancer | Radiation: Neo-adjuvant radiotherapy, Drug: Neo-adjuvant chemotherapy, Procedure: Esophagectomy | India | NCT04489368 |
| Easy-to-use Adrenal Cancer/Tumor Identity Card | To provide an easy-to-use “identity card” of adrenal tumors for personalized patient management. | Adrenal Gland Neoplasms | Biological: omics identity card | France | NCT02672020 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Casotti, M.C.; Meira, D.D.; Alves, L.N.R.; Bessa, B.G.d.O.; Campanharo, C.V.; Vicente, C.R.; Aguiar, C.C.; Duque, D.d.A.; Barbosa, D.G.; Santos, E.d.V.W.d.; et al. Translational Bioinformatics Applied to the Study of Complex Diseases. Genes 2023, 14, 419. https://doi.org/10.3390/genes14020419
Casotti MC, Meira DD, Alves LNR, Bessa BGdO, Campanharo CV, Vicente CR, Aguiar CC, Duque DdA, Barbosa DG, Santos EdVWd, et al. Translational Bioinformatics Applied to the Study of Complex Diseases. Genes. 2023; 14(2):419. https://doi.org/10.3390/genes14020419
Chicago/Turabian StyleCasotti, Matheus Correia, Débora Dummer Meira, Lyvia Neves Rebello Alves, Barbara Gomes de Oliveira Bessa, Camilly Victória Campanharo, Creuza Rachel Vicente, Carla Carvalho Aguiar, Daniel de Almeida Duque, Débora Gonçalves Barbosa, Eldamária de Vargas Wolfgramm dos Santos, and et al. 2023. "Translational Bioinformatics Applied to the Study of Complex Diseases" Genes 14, no. 2: 419. https://doi.org/10.3390/genes14020419
APA StyleCasotti, M. C., Meira, D. D., Alves, L. N. R., Bessa, B. G. d. O., Campanharo, C. V., Vicente, C. R., Aguiar, C. C., Duque, D. d. A., Barbosa, D. G., Santos, E. d. V. W. d., Garcia, F. M., Paula, F. d., Santana, G. M., Pavan, I. P., Louro, L. S., Braga, R. F. R., Trabach, R. S. d. R., Louro, T. S., Carvalho, E. F. d., & Louro, I. D. (2023). Translational Bioinformatics Applied to the Study of Complex Diseases. Genes, 14(2), 419. https://doi.org/10.3390/genes14020419






