Abstract
The aim of this study was to characterize the ophthalmic and genetic features of Bardet Biedl (BBS) syndrome in a cohort of patients from a German specialized ophthalmic care center. Sixty-one patients, aged 5–56 years, underwent a detailed ophthalmic examination including visual acuity and color vision testing, electroretinography (ERG), visually evoked potential recording (VEP), fundus examination, and spectral domain optical coherence tomography (SD-OCT). Adaptive optics flood illumination ophthalmoscopy was performed in five patients. All patients had received diagnostic genetic testing and were selected upon the presence of apparent biallelic variants in known BBS-associated genes. All patients had retinal dystrophy with morphologic changes of the retina. Visual acuity decreased from ~0.2 (decimal) at age 5 to blindness 0 at 50 years. Visual field examination could be performed in only half of the patients and showed a concentric constriction with remaining islands of function in the periphery. ERG recordings were mostly extinguished whereas VEP recordings were reduced in about half of the patients. The cohort of patients showed 51 different likely biallelic mutations—of which 11 are novel—in 12 different BBS-associated genes. The most common associated genes were BBS10 (32.8%) and BBS1 (24.6%), and by far the most commonly observed variants were BBS10 c.271dup;p.C91Lfs*5 (21 alleles) and BBS1 c.1169T>G;p.M390R (18 alleles). The phenotype associated with the different BBS-associated genes and genotypes in our cohort is heterogeneous, with diverse features without genotype–phenotype correlation. The results confirm and expand our knowledge of this rare disease.
1. Introduction
Over 100 years ago, Georges Bardet first published a report on “Congenital obesity syndrome with polydactyly and retinitis pigmentosa (RP)” [1]. Two years later, in 1922, Artur Biedl reported on a “Sibling pair with adiposo-genital dystrophy, along with illustrations of a third case” [2]. Both papers describe a syndrome that was subsequently named the Bardet–Biedl syndrome (BBS, OMIM 2099000), a form of the similar manifesting Laurence–Moon syndrome [3], although there is research suggesting that the two conditions are not distinct, but variable expressions of the same disease [4,5,6,7]. Therefore, the disorder is sometimes also acknowledged as Laurence–Moon–Bardet–Biedl syndrome.
BBS is a rare autosomal recessive genetic disorder belonging to the ciliopathies. It has a prevalence of around 1 in 125,000–160,000 in Europe [8,9]; however, this is considerably higher in closed communities such as the Faroe islands at 1:3700 [10], Bedouin communities of Kuwait at 1:65,000 [11] and the island of Newfoundland at 1:17,500 [12].
BBS has a complex phenotype and heterogeneous genotype. It can affect multiple organ systems and has a wide variability of phenotypic expression. Beales, in his notable survey, described 109 patients and their families in the UK [5], and lists five main features of the syndrome: rod-cone dystrophy, polydactyly, short stature along with obesity, learning difficulties and renal tract abnormalities. Moreover, there is a wide range of other medical conditions which may be present including neuropsychiatric abnormalities, colonic disorders, gallstone disease and asthma, and there is considerable inter- and intra-familial variation in phenotype [7,9,12,13]. The Laurence–Moon syndrome may differ from BBS by the presence of spasticity and the absence of polydactyly and obesity [4]. Some symptoms of BBS generally are not present at birth but appear gradually, and progressively worsen during or after the first decade of life, making the average age of diagnosis relatively late at 9 years [5]. Although the combination of symptoms in BBS is variable, a retinal dystrophy develops in almost all patients [5].
More than 20 BBS- or BBS-like associated genes have been identified to date, all following an autosomal recessive mode of inheritance (https://sph.uth.edu/retnet/, accessed on 17 July 2022) [14,15]. BBS proteins are necessary for the development of many organs [6]. Ansley [16] was the first to show that BBS is caused by a defect at the basal body of ciliated cells, with BBS proteins found in the basal body and cilia of cells. Subsequently, studies on animal models of BBS confirmed the early findings and have clarified the primary role of the BBS proteins in mediating and regulating intraflagellar transport, a microtubule-based intracellular transport process [17,18,19,20,21,22,23]. Seven of the gene products assemble together with the protein BBIP1/BBIP10 into the BBSome, an octameric protein complex localized at the basal body and involved in trafficking of cargo to and from primary cilia [24,25]. Therefore, BBS is considered to belong to the ciliopathies.
After the initial descriptions of BBS, several case reports on this extremely rare disease were published [26,27,28,29,30,31]. However, over the past 30 years not only has there been a vast increase in the number of genes identified, but also in the number of cases reported [5,8,9,12,32,33]. This is due to development of next-generation sequencing genetic technologies, which has accelerated the identification of the genetic causes of the disease including effective diagnostic genetic testing. In this study, we report on 61 patients that had attended the specialized out-patients’ clinic for inherited retinal dystrophies (IRD) of the University of Tübingen Eye Hospital for diagnosis and therapy.
2. Materials and Methods
2.1. Patients
Sixty-one BBS patients with likely biallelic mutations in any known BBS-associated gene and a clinical diagnosis of BBS were included in the study, performed at the Centre for Ophthalmology, University Tübingen, a German specialized ophthalmic care center for inherited retinal diseases. The minimum clinical criteria to establish a clinical diagnosis of BBS were at least four major features (i.e., visual disorder, limb defect, small stature/overweight, learning difficulties, and renal tract abnormalities) or three major and two minor features (e.g., developmental delay, neurological and motor defect, behavioral abnormality, speech and/or hearing deficits, dental anomaly, asthma, facial features, hypogonadism, heart defect, and diabetes mellitus), as suggested by Beales and co-workers in 1999 [5]. All patients received diagnostic genetic testing, either by IRD, BBS or allied disease gene panel sequencing or virtual BBS panel analysis based on whole-genome sequencing.
The patients in this study were selected upon the presence of (likely) biallelic mutations in known BBS-associated genes and upon the diagnosis of BBS based on at least four major features or three major and two minor features [5]. In the initially selected cohort, there were five patients, however, who were non-syndromic and thus these were subsequently not included in this analysis.
The study was approved by the ethics committee of the University of Tübingen. It was carried out in accordance with the Declaration of Helsinki. Written consent was obtained from all patients (or their parents/guardians if underaged or intellectually disabled) for both the research study and for the diagnostic genetic testing.
2.2. Procedure
The medical history of the patients was recorded, and a comprehensive ophthalmic examination was performed including: full-field electroretinography (ERG, scotopic and photopic) and visually evoked potential recording (VEPs) according to ISCEV standards with the Diagnosys system (Lowell, MA, USA) or the RETeval® device (LKC Technologies, Inc., Gaithersburg, MD, USA). Fundus autofluorescence (AF, 30° or 55°) and spectral domain optical coherence tomography (SD-OCT) were performed with the Spectralis HRA+OCT (Heidelberg Engineering Inc., Heidelberg, Germany). Adaptive optics flood illumination ophthalmoscopy (AO-FIO) images were additionally obtained from five patients using the adaptive optics flood illuminated camera rtx1TM (Imagine Eyes, Orsay, France). Visual acuity was tested with Snellen or Lea charts, and color vision testing was carried out with the panel D-15 or Lea tests. Additionally, visual field testing was performed with the Octopus 900 (Haag-Streit, Wedel, Germany) using the III4e or I4e stimulus. Of note, some patients with accompanying disabilities were not able to perform all tests. The number of patients that performed each examination is given in the results section.
3. Results
Sixty-one patients from 57 families with likely biallelic mutations in known BBS-associated genes were included in the study (see Supplementary Tables S1–S3). In Supplementary Table S4, a detailed description of the ophthalmological findings for each patient is given, while Supplementary Table S5 summarizes all systemic findings.
The patient’s mean age at the time of the ophthalmic examination was 24.5 years +/− 12.3 SD (median 23 years), with 35 males and 26 females ranging from 5 years to 56 years. Figure 1A depicts the age distribution for each gender. In Figure 1B, the visual acuity for both eyes are plotted against age. Visual acuity is extremely compromised at all ages and decreases by approximately 0.06 (decimal) per year. The best visual acuities recorded were 0.63 in a 21-year-old patient with BBS12, 0.5 in a BBS2 patient aged 8 years and 0.4 in a BBS16 patient aged 42 years.
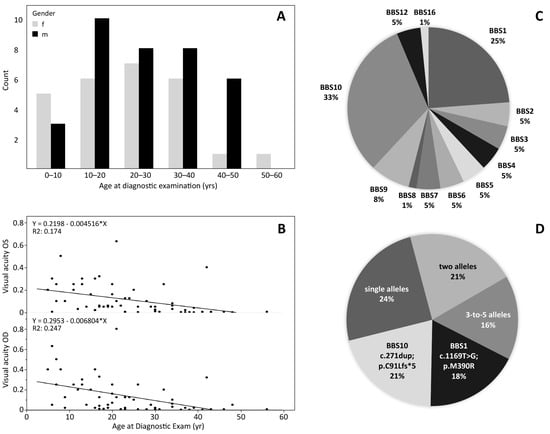
Figure 1.
Demographics of the 61 BBS patients. (A): Age of the patients, for male and female separately. (B): Decimal visual acuity of the left and right eyes plotted against age. (C): Frequency of BBS genes carrying biallelic mutations in the patients of this study. (D): Common variants presented as percentage of total allele counts. BBS10 c.271dup;p.C91Lfs*5 (21%) and BBS1 c.1169T>G;p.M390R (18%) are most common, but also other variants with up to 5 alleles were observed recurrently.
We evaluated the spherical equivalent in diopter (D) of the patient’s refractive error (Table 1), and the number of patients with myopia and hyperopia for each genetic subgroup are provided separately (of note, nine patients could not perform this test). A myopia grade −3D–−6D is the most common along with a hyperopia <+3D. The patient numbers for the other genes are too low to draw conclusions, but interestingly, the three patients in the subgroups BBS3, BBS4 and BBS5 are myopic. The values for the left eye were similar to those of the right (Supplementary Table S4).

Table 1.
Spherical equivalent (dpt) of the right eye. Number of patients and % of cases for each genetic subgroup.
The cohort showed 51 different disease-causing variants in twelve different BBS genes, as depicted in Figure 1C,D: Twenty patients (32.8%) carried likely biallelic (apparent homozygous or two heterozygous) variants in the BBS10 gene and 15 (24.6%) in the BBS1 gene, five patients (8.2%) in the BBS9 gene, three patients (4.9%) each in the genes BBS2, BBS3, BBS4, BBS7, BBS5 and BBS12, whereas only one patient each (1.6%) carried apparent homozygous disease-causing variants in the BBS6, BBS8, and BBS16 genes.
The most common associated genes were BBS10 (32.8%) and BBS1 (24.6%), and by far the most commonly observed variants were BBS10 c.271dup;p.C91Lfs*5 (21 alleles) and BBS1 c.1169T>G;p.M390R (18 alleles) (see Supplementary Tables S1 and S2). In contrast, 45% of all variants were observed only once or twice. Thirty-nine patients were (apparent) homozygous (63.9%) and 22 patients carried two heterozygous mutations. Segregation to confirm homozygosity or compound-heterozygosity was available in 34.4% (21/61) of the cases. Missense variants were the most common mutation type (47%), followed by frame-shifting small insertion, deletion and duplication mutations (19.6%), variants likely resulting in mis-splicing (13.7%) and nonsense mutations (7.8%). Large deletions were observed in the BBS1 and the BBS9 gene.
In 12 patients, additional heterozygous variants in other BBS- or IRD-related genes were observed (Supplementary Table S1). It has been implied that the mutational load or digenic, triallelic variants contribute to the BBS phenotype [34,35]. We have classified the variants (Supplementary Table S3), and present only variants that are either classified as variants of uncertain significance, likely pathogenic or pathogenic. Whether these variants contribute to the disease-phenotype could not be established. Of note, both brothers of family BBS56 were homozygous both for the BBS5 variant c.143-4_143-2ins400-500;p.? and the BBS12 missense variant of uncertain significance c.1139C>T;p.T380I. Segregation analysis showed that the unaffected mother is also homozygous for the BBS12 missense variant c.1139C>T;p.T380I; therefore, we conclude that the BBS phenotype is primarily caused by BBS5 variant c.143-4_143-2ins400-500;p.?, and the BBS12 variant might—if at all—only modulate the phenotype.
In Figure 2 and Figure 3, we show the retinal images obtained from representative patients. Two patients did not perform the OCT examination and 18 the AF.
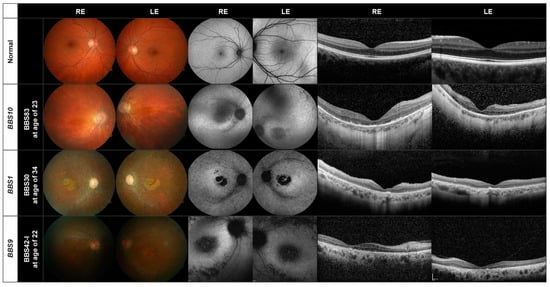
Figure 2.
Fundus (left), AF (center) and OCT (right) images of representative patients with disease-causing variants in the BBS1, BBS10, and the BBS9 genes. The images of a normal eye are shown in the first row.
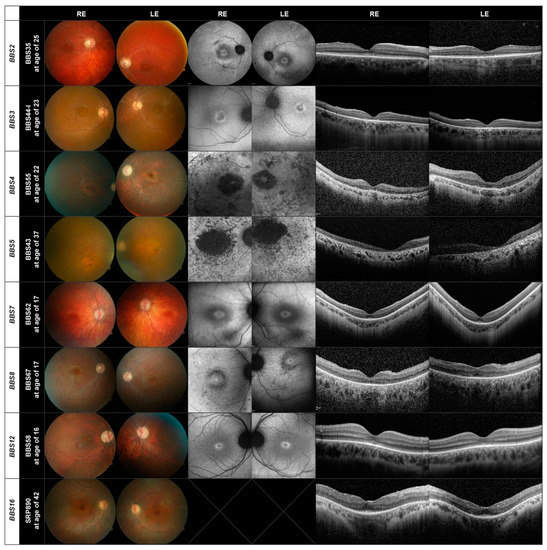
Figure 3.
Fundus, AF and OCT images of one patient each carrying disease-causing variants in the BBS2, BBS3, BBS4, BBS5, BBS7, BBS8, BBS12, and BBS16 genes.
The fundus, AF and OCT images of one patient (BBS83) harboring disease-causing variants in the BBS10 gene are shown in the second row in Figure 2. For comparison, normal images can be seen in the upper row. Peripapillary atrophy, retinal pigment epithelial (RPE) changes with macular atrophy and attenuated vessels, typical of RP, can be seen with a normal optic disc; 14 of the 20 patients with variants in the BBS10 gene showed a pallor optic disc. A perimacular hyperfluorescent ring can be seen in the AF images (center) which was found in 13 of the 20 patients. Hypoautofluorescent spots are found in the periphery. The OCT images of all BBS10 patients showed atrophy of the photoreceptor layer and loss of photoreceptor cells, mostly along with internal limiting membrane wrinkling.
All 15 patients with mutations in the BBS1 gene show a large macular atrophy with diffuse RPE atrophy throughout the retina and attenuated vessels (third row, Figure 2). Pallor of the optic nerve can also be seen; four of the 15 patients had a normal optic disc. In the AF images, a perimacular hyperfluorescent ring is present, as found in all but three of the 15 patients. All patients also exhibited hypoautofluorescent spots. The OCT images show again foveal atrophy or loss of the photoreceptor layer in all patients. ONL thinning is evident in the BBS1 patient (BBS30) in the third row in Figure 2.
In the lower panel in Figure 2, we show the retinal images of one of the five patients carrying disease-causing variants in the BBS9 gene (BBS42-I). Loss of the photoreceptor layer, foveal atrophy and a pallor optic disc can be seen, as in the patients with mutations in the BBS10 or BBS1 genes. The BBS9 patient also exhibits a bull’s eye maculopathy, seen as a dark macula surrounded by paler rings in the AF images, in addition to hypoautofluorescent spots. This was also found in another two patients with this variant. The OCT displays a loss of the photoreceptor cell layer and foveal atrophy. Two of the patients were brothers (BBS42-I, BBS42-II).
Thus, for the three largest subgroups of patients BBS10 (20 patients), BBS1 (15 patients) and BBS9 (five patients) there is a similar pattern of retinal degeneration. This also holds true for the other genes present in our patient cohort, as demonstrated in Figure 3, where there are only three or fewer patients in each subgroup. Of interest is that the fundus images of patients BBS44-I (BBS3) and BBS58 (BBS12) show evidence of cone dystrophy. The patient with the BBS12 mutations shows additionally a peripapillary myopic conus.
Although the results of the retinal imaging indicate that the phenotype of the retinal dystrophy does not appear to depend greatly on the mutations present, two features deserve further mentioning. The first is the presence of a bull’s eye macula, observed in nine (15%) of the patients, which although not uncommon in IRD, appears to be unevenly distributed between subgroups. It was found in 3/5 patients with disease-causing variants in the BBS9 gene and 2/3 patients with variants in the BBS7 gene, but was not present in any of the 20 patients with disease-causing genotypes in the BBS10 gene. A bull’s eye was further found in 1/15 BBS1 patients, 1/3 BBS2 patients, 1/3 BBS3 patients and 1/3 BBS4 patients (see Supplementary Table S4). A second feature is the presence of cone-rod dystrophy. A total of 16 patients (26%) were found to have a cone or cone-rod dystrophy, but it appears to be distributed evenly between BBS subtypes (3/15 BBS1, 2/3 BBS2, 3/3 BBS3, 1/5 BBS9, 5/20 BBS10, and 2/3 BBS12).
To explore the photoreceptor abnormalities on a cellular level, adaptive optics imaging was performed in five patients. In at least one eye of each patient, we could acquire a montage out of five single 4 degrees × 4 degrees sized images. The images of three patients are shown in Figure 4. Due to the nystagmus in three of the five patients, the imaging was difficult. However, in all images a disrupted cone photoreceptor mosaic dominated by dark patchy areas was observable. In one patient (BBS10-II), we detected a hemicycle shaped RPE clumping in both eyes, which is also observable in the fundus and infrared imaging. In patient BBS85 (third panel), the AO images showed a typical central blur followed by a reduced but still visible cone photoreceptor mosaic followed by parafoveal blur. The images correlate with the still delineable ellipsoid zone and external limiting membrane in the foveola in the OCT, which disintegrate parafoveally. Furthermore, many small hyperreflective spots were detectable in the AO imaging. In another patient (BBS75), the AO imaging revealed the appearance of so-called “puffy cones”; these cones appear bigger in size but in a still intact mosaic. The fixation of patient BBS75 was too unstable to overlay the AO imaging with the fundus autofluorescence or the infrared imaging.
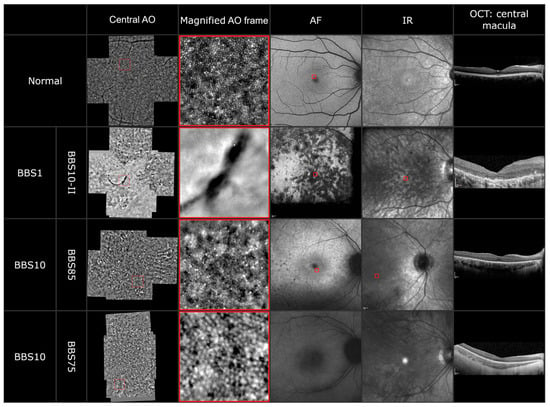
Figure 4.
AO montage, magnified AO imaging (highlighted by a red frame), AF, IR and OCT of a healthy subject and three BBS patients carrying disease-causing variants in the BBS1 and BBS10 genes.
3.1. Further Examinations
In all patients, the first symptoms were night blindness and/or photophobia along with eye abnormalities such as strabismus, cataracts, and nystagmus (Supplementary Table S4). Twenty of the 61 patients (33%) suffered from strabismus with two having had it as a child and two having had surgery. Thirty-two of the 61 patients have or have had a cataract, nine of whom had received surgery, but they were distributed evenly between genotypes. Fourteen (70%) of the 20 BBS10 patients suffered from nystagmus, and 4/5 (80%) of the BBS9 patients but only 4/15 (27%) patients with BBS1.
Visual field testing could not be performed in 35 of the patients. Of the rest, most showed a concentric constriction, with some showing good function or remaining islands of function in the periphery. The ERG was extinguished in 54 of the 61 patients; five patients (two BBS12 (BBS58, 16 y and BBS59, 21 y), one BBS10 (RCD768 29 y), two BBS3 (BBS44-I, 23 y and BBS44-I, 13 y) showed a scotopic response. Recording was not possible in two patients. The VEP was performed in 24 subjects (four pattern VEP, 20 flash VEP). Thirteen showed a good flash VEP response and a reduction in VEP amplitude was found in 10 patients. Color vision testing was not possible in nine patients and only five gave normal results. Forty-four patients were totally color blind.
3.2. Systemic Features
In the Supplementary Table S5, the systemic features for each patient are listed. An evaluation of primary and secondary features of the cohort is given in Table 2, showing that all patients suffered from retinal dystrophy, with polydactyly and obesity also being common. Brachy-/syndactyly is the most common (55%) among the secondary features. None of our patients reported a hearing impairment and only one patient reported anosmia.

Table 2.
Primary and secondary features of the BBS study cohort.
4. Discussion
In this study, we report on the ophthalmic and genetic features of a cohort of 61 BBS patients, ranging from 5 to 56 years in age. The initial symptoms reported by almost all of our patients were night blindness and photophobia with only seven patients reporting additional amblyopia or visual acuity impairment.
The visual impairment of the BBS patients is severe. The visual acuity of the BBS cohort decreased from an average of around 0.2 in young children to 0 at age 50 (Figure 1). This is comparable to that reported elsewhere [14,36,37,38,39]. All of the patients showed changes in the morphology of the fundus in the fundus photographs and AF images, demonstrating that retinal dystrophy is a general deficiency in BBS patients [14,36,37,38,39]. RP along with “general imperfections of development” were the first features to be reported about the syndrome [3]. The retinal dystrophy occurring after early childhood can be explained by a theory that proposes that the dendritic processes of retinal neurons are supported by intraflagellar transport protein complexes [40,41] which are responsible for the assembly and replacement of cilia [42], as well as signaling in the cilium [43]. Mutations in the BBS genes disturb ciliary assembly (see e.g., [44,45,46]). The degeneration was also evident in the reduced cone and rod responses in the ERG of most patients. We find extinguished responses in 89% of the patients, aged between 13 and 29 years, which is consistent with the results of Fawcett et al. [47], indicating that there is no correlation between age and the degree of retinal dysfunction revealed by electrophysiological recordings. The VEP response, on the other hand, was reduced in about half of the recordings. These results are in agreement with previous studies [47,48,49,50]. The visual field could not be measured accurately in around half of the patients due to inadequate fixation and nystagmus. Most of the remainder showed a central scotoma with peripheral islands of remaining function.
Observations of the cone photoreceptor mosaic with an AO flood-illuminated camera or an AO-SLO in retinitis pigmentosa patients have shown that the cone density can range from normal to severely reduced [51,52,53,54,55,56]. Additionally, four main patterns could be described, which could be correlated to the progressive phases of retinal degeneration [54]. The AO images of our BBS patients complement the standard clinical examinations and the findings of the OCT and fundus photography. The disrupted cone photoreceptor mosaic, dark patchy areas and clumping of the RPE demonstrate the severe dystrophy of the retina. A publication of two siblings with BBS due to a mutation in the BBS7 gene also observed a reduced photoreceptor density by adaptive optics scanning light ophthalmoscopy (AOSLO) [57]. Detection of changes in cone appearance in one patient could also show the further progression of degeneration.
EURO-WABB, a rare disease registry, has published guidelines for the management of BBS and the assessment of the many affected organs and systems (www.euro-wabb.org, accessed on 14 February 2022). Generally, our patients have similar features (Table 2) to those in the guidelines, but the frequencies with which we find them differ. Thirty-one percent of our patients show renal abnormalities, whereas in the guidelines this figure is only 9%. We also find a larger number of patients with a speech disorder (25% vs. 2%), developmental delay (34% vs. 9%) and brachydactyly/syndactyly (55% vs. 4%) and a lower number of patients with heart disease (0% vs. 6%). The Laurence–Moon syndrome generally differs from BBS by the presence of spasticity and the absence of polydactyly and obesity [4], which was not observed in any of our patients.
The results of five non-syndromic patients were not included in this analysis. Most interesting is the 20-year-old brother of patient BBS40-I, aged 43, who only displayed retinal symptoms of the disease despite his brother showing developmental delay, mental retardation and polydactyly. The brothers were both homozygous for the BBS9 c.263+1G>T;p.(?) variant. In line with previous studies, 3/5 non-syndromic patients carried disease-causing variants in the BBS1 gene, all homozygous for BBS1 c.1169T>G;p.(M390R) [58,59]. The fifth patient who appeared non-syndromic was heterozygous for two variants in BBS4 c.883C>T;p.(R295*) and c.1107-10_-7delTCTG;p.(?). Non-syndromic retinitis pigmentosa has also been reported in patients with other BBS mutations [60,61,62].
BBS1 and BBS10 were the most common genotypes in our cohort, as found by others [9,13,63], together making up 57.4% of our patients. Mutations in BBS9 were our next most common cause of BBS, at 8% (five patients). Other studies, on the other hand, have reported significant proportions of BBS2 and BBS12-related disease [63,64]. Ethnic factors including common founder mutations and rate of consanguinity are expected to be the cause of this discrepancy.
The mutation spectrum is comparable to that of other recent studies [35,65,66], and 51 different variants were observed, either (apparent) homozygous or two (compound) heterozygous variants found in 12 different genes (Supplementary Tables S1 and S2). Of these, 11 variants have never been reported before and deserve further discussion. In BBS1, four novel variants were observed; one near splice site c.479+4A>G;p.(?), a duplication of 10 bp c.784_793dup;p.(N269Gfs*95) and a 17 bp deletion c.1431_1447del;p.(L478Rfs*17), both resulting in frame-shift and premature termination codon, as well as a large deletion covering exons 14 to 17. While the latter three are predicted pathogenic or likely pathogenic according to ACMG classification, the splice site variant c.479+4A>G;p.(?) is classified as variant of uncertain significance. Of note, the variant is predicted to result in mis-splicing by two of three queried prediction software and we therefore suggest this compound-heterozygous variant in patient BBS78 to be likely disease-causing together with the deletion of exons 14–17 on the BBS1 counter allele.
In the BBS5 gene, an intronic insertion of 400–500 bp was observed in three patients of two independent families. Both families have documented consanguinity, but it could not be established whether these patients are distantly related. Unfortunately, the exact extent and sequence content of the insertion could not be established due to repetitive sequences, and therefore also prediction on the effect of this insertion for example on splicing could not be predicted. In addition, segregation analysis showed that the unaffected mother of both brothers of family BBS56 was homozygous for the variant. Still, we cannot exclude that this BBS5 insertion may contribute to the disease, e.g., as a hypomorphic allele, as the variant may alter the splice acceptor of exon 3, was observed in multiple patients and families, and was absent from healthy control individuals (i.e., gnomAD browser database).
Another putative splicing variant was observed in the BBS7 gene. The variant c.1037+29T>A;p.(?) was found in patient BBS60, compound heterozygous to the 4 bp deletion c.712_715delAGAG;p.(R238Efs*59). It is not predicted to result in mis-splicing but was the only rare heterozygous variant found in this patient by BBS panel sequencing. We cannot exclude that another BBS7 variant, for example, a deep intronic variant, was missed and only cDNA analysis or minigene splice assays could finally elucidate the effect of this variant.
The TTC8 (BBS8) missense variant c.694G>A;p.(G232R) was found apparent homozygously in patient BBS67 and was classified as a variant of uncertain significance. No further support for its pathogenicity than the output of the prediction tools, which include consideration to conservation and biophysical properties of the amino acid residue, and frequency of the variant in normal population, can be added.
Last but not least, three new variants were observed in BBS10, which is the most frequently mutated gene in our study. The missense variants c.686C>T;p.(P229L), c.901C>T;p.(L301V) and c.1802C>T;p.(P601L) also fulfil the same criteria as those just described for the novel TTC8 missense variants to classify these as variants of uncertain significance. Further cases, segregation analysis and functional studies will be needed to confirm the pathogenic effect of these variants. In contrast, the BBS10 2 bp duplication c.858_859dup;p.(Q287Lfs*12) will result in frameshift and premature termination codon, resulting in loss of important structural and functional domains and rendering this variant possibly a null allele.
We find that the phenotype of the different BBS genes in our cohort is heterogeneous, with diverse features occurring in all mutations. This is characteristic of the syndrome [6,38,67]; however, in recent years, by analyzing larger patient cohorts, some associations have been published.
Generally, BBS10 and BBS2 patients have been reported as having more severe features than BBS1 [63,68,69] with lower risk of cardiovascular disease [70]. We did find a significant difference between BBS gene-association with respect to the occurrence of nystagmus (p = 0.005), with 68% of BBS10 patients suffering from nystagmus and only 21% of those with BBS1. Our BBS10 patients also had a greater chance of strabismus 37% (compared to the BBS1 21%). This difference between BBS10 and BBS1 also tended to be true for the systemic symptoms, but the lack of sufficient numbers does not allow a more detailed analysis. In addition, renal anomalies have been found to be more prevalent in BBS2, BBS7 and BBS9 patients [63,68], and a relatively low penetrance of polydactyly in patients with mutations in BBS1, which does not appear to be the case in our patients.
The visual prognosis for BBS patients is poor. However, genetic therapeutics offer a more promising future, with research ongoing in animal models. In Bbs1 and Bbs4 mouse models, the use of gene therapy to preserve the retinal function, especially at early stages of the disease, has been shown to be successful [71,72,73]. Clinical trials using antisense-oligonucleotide therapy for the common deep intronic variant c.2991+1655A->G in CEP290 (BBS14/NPHP6/LCA10/MKS4/SLSN6/JBTS5) have shown promising results in halting degeneration of the photoreceptors, although the recent press release by ProQR on the phase II/III trial is unfortunately less encouraging as primary endpoints were not met [74]. The olfactory system has also been shown to be sensitive to gene therapy in mice [75,76]. A macaque model of RP due to mutations in BBS7 has been recently discovered which will help in testing treatments for this subtype [77] and further the search for adequate therapies.
5. Conclusions
The results of the 61 BBS patients in this German cohort, confirm and expand our knowledge on the phenotype and genotype of BBS and will aid in providing information for future studies to help combat the devastating effects of this rare disease.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/genes13071218/s1, Table S1, Genotypes of BBS patients in this study; Table S2: Mutation spectrum in BBS-related genes in this study cohort; Table S3: Classification of additional heterozygous variants in BBS- or IRD-related genes. Tables S1–S3 (genetics) [78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94,95,96,97,98,99], Table S4 (ophthalmology) and Table S5 (systemic findings).
Author Contributions
Conceptualization, F.N. and E.Z.; validation, F.N., M.K., S.K.; formal analysis, F.N., A.K., S.K., N.W.; investigation, F.N., M.K., S.B., T.Z., T.B.H., S.K., N.W.; writing—original draft preparation, A.K., F.N., S.K., K.S., E.Z.; writing—review and editing, all authors; visualization, F.N., A.K., S.K., M.K.; supervision, E.Z.; project administration, F.N.; funding acquisition, K.S., E.Z. All authors have read and agreed to the published version of the manuscript.
Funding
The research was supported by grants from the Tistou and Charlotte Kerstan Foundation to F.N. and A.K. and from the German Research Council (project 399487883) to M.K. and K.S.
Institutional Review Board Statement
The study was approved by the ethics committee of the University of Tübingen. Germany Project no. 349/2003V, reviewed 22 September 2003 and project no. 116/2015BO2116/2015BO2, last update and review February 2022. It was carried out in accordance with the Declaration of Helsinki. Written consent was obtained from all patients (or their parents/guardians if underaged or intellectually disabled) for both the research study and for the diagnostic genetic testing.
Informed Consent Statement
Written informed consent was obtained from all patients (or their parents/guardians if underaged or intellectually disabled) for both the research study and for the diagnostic genetic testing.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Bardet, G. Sur Un Syndrome D’obesite Congenitale Avec Polydactylie et Retinite Pigmentaire. Ph.D. Thesis, University of Paris, Paris, France, 1920. [Google Scholar]
- Biedl, A. Geschwisterpaar Mit Adiposo-Genitalier Dystrophie. Deutsch. Med. Wschr. 1922, 48, 1630. [Google Scholar]
- Laurence, J.Z.; Moon, R.C. Four Cases of “Retinitis Pigmentosa” Occurring in the Same Family, and Accompanied by General Imperfections of Development. Ophthalmic Rev. 1866, 2, 32–41. [Google Scholar]
- Schachat, A.P.; Maumenee, I.H. Bardet-Biedl Syndrome and Related Disorders. Arch. Ophthalmol. 1982, 100, 285–288. [Google Scholar] [CrossRef]
- Beales, P.L.; Elcioglu, N.; Woolf, A.S.; Parker, D.; Flinter, F.A. New Criteria for Improved Diagnosis of Bardet-Biedl Syndrome: Results of a Population Survey. J. Med. Genet. 1999, 36, 437–446. [Google Scholar] [CrossRef] [PubMed]
- Moore, S.J.; Green, J.S.; Fan, Y.; Bhogal, A.K.; Dicks, E.; Fernandez, B.A.; Stefanelli, M.; Murphy, C.; Cramer, B.C.; Dean, J.C.S.; et al. Clinical and Genetic Epidemiology of Bardet-Biedl Syndrome in Newfoundland: A 22-Year Prospective, Population-Based, Cohort Study. Am. J. Med. Genet. Part A 2005, 132A, 352–360. [Google Scholar] [CrossRef]
- Riise, R. Laurence-Moon-Bardet-Biedl Syndrome: Clinical Electrophysiology and Genetic Aspects. Acta Ophthalmol. Scand Suppl. 1998, 226, 1–28. [Google Scholar]
- Klein, D.; Ammann, F. The Syndrome of Laurence-Moon-Bardet-Biedl and Allied Diseases in Switzerland. J. Neurol. Sci. 1969, 9, 479–513. [Google Scholar] [CrossRef]
- Beales, P.L.; Warner, A.M.; Hitman, G.A.; Thakker, R.; Flinter, F.A. Bardet-Biedl Syndrome: A Molecular and Phenotypic Study of 18 Families. J. Med. Genet. 1997, 34, 92–98. [Google Scholar] [CrossRef]
- Hjortshoj, T.D.; Gronskov, K.; Brondum-Nielsen, K.; Rosenberg, T. A Novel Founder BBS1 Mutation Explains a Unique High Prevalence of Bardet-Biedl Syndrome in the Faroe Islands. Br. J. Ophthalmol. 2009, 93, 409–413. [Google Scholar] [CrossRef]
- Farag, T.I.; Teebi, A.S. High Incidence of Bardet Biedl Syndrome among the Bedouin. Clin. Genet. 2008, 36, 463–464. [Google Scholar] [CrossRef]
- Green, J.S.; Parfrey, P.S.; Harnett, J.D.; Farid, N.R.; Cramer, B.C.; Johnson, G.; Heath, O.; McManamon, P.J.; O’Leary, E.; Pryse-Phillips, W. The Cardinal Manifestations of Bardet–Biedl Syndrome, a Form of Laurence–Moon–Biedl Syndrome. N. Engl. J. Med. 1989, 321, 1002–1009. [Google Scholar] [CrossRef] [PubMed]
- Forsythe, E.; Beales, P.L. Bardet–Biedl Syndrome. Eur. J. Hum. Genet. 2013, 21, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Weihbrecht, K.; Goar, W.A.; Pak, T.; Garrison, J.E.; DeLuca, A.P.; Stone, E.M.; Scheetz, T.E.; Sheffield, V.C. Keeping an Eye on Bardet-Biedl Syndrome: A Comprehensive Review of the Role of Bardet-Biedl Syndrome Genes in the Eye. Med. Res. Arch. 2017, 5. [Google Scholar] [CrossRef]
- Rohrschneider, K.; Bolz, H.J. Bardet-Biedl-Syndrom–Diagnose Und Klinischer Verlauf. Klin. Monbl. Augenheilkd. 2020, 237, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Ansley, S.J.; Badano, J.L.; Blacque, O.E.; Hill, J.; Hoskins, B.E.; Leitch, C.C.; Chul Kim, J.; Ross, A.J.; Eichers, E.R.; Teslovich, T.M.; et al. Basal Body Dysfunction Is a Likely Cause of Pleiotropic Bardet–Biedl Syndrome. Nature 2003, 425, 628–633. [Google Scholar] [CrossRef]
- Blacque, O.E.; Reardon, M.J.; Li, C.; McCarthy, J.; Mahjoub, M.R.; Ansley, S.J.; Badano, J.L.; Mah, A.K.; Beales, P.L.; Davidson, W.S.; et al. Loss of C. elegans BBS-7 and BBS-8 Protein Function Results in Cilia Defects and Compromised Intraflagellar Transport. Genes Dev. 2004, 18, 1630–1642. [Google Scholar] [CrossRef] [PubMed]
- Ou, G.; Blacque, O.E.; Snow, J.J.; Leroux, M.R.; Scholey, J.M. Functional Coordination of Intraflagellar Transport Motors. Nature 2005, 436, 583–587. [Google Scholar] [CrossRef]
- Nachury, M.V.; Loktev, A.V.; Zhang, Q.; Westlake, C.J.; Peränen, J.; Merdes, A.; Slusarski, D.C.; Scheller, R.H.; Bazan, J.F.; Sheffield, V.C.; et al. A Core Complex of BBS Proteins Cooperates with the GTPase Rab8 to Promote Ciliary Membrane Biogenesis. Cell 2007, 129, 1201–1213. [Google Scholar] [CrossRef]
- Mockel, A.; Perdomo, Y.; Stutzmann, F.; Letsch, J.; Marion, V.; Dollfus, H. Retinal Dystrophy in Bardet–Biedl Syndrome and Related Syndromic Ciliopathies. Prog. Retin. Eye Res. 2011, 30, 258–274. [Google Scholar] [CrossRef]
- Hildebrandt, F.; Benzing, T.; Katsanis, N. Ciliopathies. N. Engl. J. Med. 2011, 364, 1533–1543. [Google Scholar] [CrossRef]
- Novas, R.; Cardenas-Rodriguez, M.; Irigoín, F.; Badano, J.L. Bardet-Biedl Syndrome: Is It Only Cilia Dysfunction? FEBS Lett. 2015, 589, 3479–3491. [Google Scholar] [CrossRef] [PubMed]
- Dean, S.; Moreira-Leite, F.; Varga, V.; Gull, K. Cilium Transition Zone Proteome Reveals Compartmentalization and Differential Dynamics of Ciliopathy Complexes. Proc. Natl. Acad. Sci. USA 2016, 113, E5135–E5143. [Google Scholar] [CrossRef]
- Wei, Q.; Zhang, Y.; Li, Y.; Zhang, Q.; Ling, K.; Hu, J. The BBSome Controls IFT Assembly and Turnaround in Cilia. Nat. Cell Biol. 2012, 14, 950–957. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.; Zhang, Y.; Wei, Q.; Huang, Y.; Li, Y.; Ling, K.; Hu, J. BBS4 and BBS5 Show Functional Redundancy in the BBSome to Regulate the Degradative Sorting of Ciliary Sensory Receptors. Sci. Rep. 2015, 5, 11855. [Google Scholar] [CrossRef] [PubMed]
- Solis-Cohen, S.; Weiss, E. Dystrophia Adiposogenitalis, with Atypical Retinitis Pigmentosa and Mental Deficiency- the Laurence-Biedl Syndrome: A Report of Four Cases in One Family. Am. J. Med. Sci. 1925, 169, 499–504. [Google Scholar] [CrossRef]
- Uoti, I. Two Cases of Dystrophia Adiposo-Genitalis, Type Laurence-Moon-Biedl-Bardet. Ann. Med. Intern. Fenn. 1947, 36, 711–714. [Google Scholar]
- Siegler, A.M.; Weisfogel, E. Laurence-Moon-Bardet-Biedl Syndrome; a Family Group with Three Affected Siblings. Obstet. Gynecol. 1956, 8, 332–335. [Google Scholar]
- Fan, Y.; Esmail, M.A.; Ansley, S.J.; Blacque, O.E.; Boroevich, K.; Ross, A.J.; Moore, S.J.; Badano, J.L.; May-Simera, H.; Compton, D.S.; et al. Mutations in a Member of the Ras Superfamily of Small GTP-Binding Proteins Causes Bardet-Biedl Syndrome. Nat. Genet. 2004, 36, 989–993. [Google Scholar] [CrossRef]
- Sahu, J.; Jain, V. Laurence-Moon-Bardet-Biedl Syndrome. J. Nepal Med. Assoc. 2008, 47, 235–237. [Google Scholar] [CrossRef]
- Chakrabarty, S.; Savantre, S.B.; Ramachandra Bhat, C.; Satyamoorthy, K. Multiple Genetic Mutations Implicate Spectrum of Phenotypes in Bardet-Biedl Syndrome. Gene 2020, 725, 144164. [Google Scholar] [CrossRef]
- Griffiths, G.M. The lawrence-biedl syndrome. J. Neurol. Neurosurg. Psychiatry 1931, 12, 53–58. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Beales, P.L.; Badano, J.L.; Ross, A.J.; Ansley, S.J.; Hoskins, B.E.; Kirsten, B.; Mein, C.A.; Froguel, P.; Scambler, P.J.; Lewis, R.A.; et al. Genetic Interaction of BBS1 Mutations with Alleles at Other BBS Loci Can Result in Non-Mendelian Bardet-Biedl Syndrome. Am. J. Hum. Genet. 2003, 72, 1187–1199. [Google Scholar] [CrossRef] [PubMed]
- Katsanis, N. Triallelic Inheritance in Bardet-Biedl Syndrome, a Mendelian Recessive Disorder. Science 2001, 293, 2256–2259. [Google Scholar] [CrossRef] [PubMed]
- Manara, E.; Paolacci, S.; D’Esposito, F.; Abeshi, A.; Ziccardi, L.; Falsini, B.; Colombo, L.; Iarossi, G.; Pilotta, A.; Boccone, L.; et al. Mutation Profile of BBS Genes in Patients with Bardet-Biedl Syndrome: An Italian Study. Ital. J. Pediatr. 2019, 45, 72. [Google Scholar] [CrossRef] [PubMed]
- Fulton, A.B. Natural Course of Visual Functions in the Bardet-Biedl Syndrome. Arch. Ophthalmol. 1993, 111, 1500. [Google Scholar] [CrossRef]
- Denniston, A.K.; Beales, P.L.; Tomlins, P.J.; Good, P.; Langford, M.; Foggensteiner, L.; Williams, D.; Tsaloumas, M.D. Evaluation of Visual Function and Needs in Adult Patients with Bardet-Biedl Syndrome. Retina 2014, 34, 2282–2289. [Google Scholar] [CrossRef]
- Meng, X.; Long, Y.; Ren, J.; Wang, G.; Yin, X.; Li, S. Ocular Characteristics of Patients with Bardet–Biedl Syndrome Caused by Pathogenic BBS Gene Variation in a Chinese Cohort. Front. Cell Dev. Biol. 2021, 9, 635216. [Google Scholar] [CrossRef]
- Berezovsky, A.; Rocha, D.; Sacai, P.; Watanabe, S.; Cavascan, N.; Salomao, S. Visual Acuity and Retinal Function in Patients with Bardet-Biedl Syndrome. Clinics 2012, 67, 145–149. [Google Scholar] [CrossRef]
- Sedmak, T.; Wolfrum, U. Intraflagellar Transport Molecules in Ciliary and Nonciliary Cells of the Retina. J. Cell Biol. 2010, 189, 171–186. [Google Scholar] [CrossRef]
- Orozco, J.T.; Wedaman, K.P.; Signor, D.; Brown, H.; Rose, L.; Scholey, J.M. Movement of Motor and Cargo along Cilia. Nature 1999, 398, 674. [Google Scholar] [CrossRef]
- Qin, H.; Diener, D.R.; Geimer, S.; Cole, D.G.; Rosenbaum, J.L. Intraflagellar Transport (IFT) Cargo: IFT Transports Flagellar Precursors to the Tip and Turnover Products to the Cell Body. J. Cell Biol. 2004, 164, 255–266. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Pan, J.; Snell, W.J. Intraflagellar Transport Particles Participate Directly in Cilium-Generated Signaling in Chlamydomonas. Cell 2006, 125, 549–562. [Google Scholar] [CrossRef] [PubMed]
- Cole, D.G.; Diener, D.R.; Himelblau, A.L.; Beech, P.L.; Fuster, J.C.; Rosenbaum, J.L. Chlamydomonas Kinesin-II-Dependent Intraflagellar Transport (IFT): IFT Particles Contain Proteins Required for Ciliary Assembly in Caenorhabditis Elegans Sensory Neurons. J. Cell Biol. 1998, 141, 993–1008. [Google Scholar] [CrossRef] [PubMed]
- Pazour, G.J.; Rosenbaum, J.L. Intraflagellar Transport and Cilia-Dependent Diseases. Trends Cell Biol. 2002, 12, 551–555. [Google Scholar] [CrossRef]
- Tsujikawa, M.; Malicki, J. Intraflagellar Transport Genes Are Essential for Differentiation and Survival of Vertebrate Sensory Neurons. Neuron 2004, 42, 703–716. [Google Scholar] [CrossRef]
- Fawcett, R.J.; Liasis, A.; Moore, W.; Sparks, K.L.; Walters, B.; Beales, P.; Thompson, D.A. Visual Function in Patients with Bardet Biedl Syndrome. Doc. Ophthalmol. 2012, 124, 40. [Google Scholar] [CrossRef]
- Lavy, T.; Harris, C.M.; Shawkat, F.; Thompson, D.; Taylor, D.; Kriss, A. Electrophysiological and Eye-Movement Abnormalities in Children with the Bardet-Biedl Syndrome. J. Pediatr. Ophthalmol. Strabismus 1995, 32, 364–367. [Google Scholar] [CrossRef]
- Iannaccone, A.; Vingolo, E.M.; Rispoli, E.; Propris, G.; Tanzilli, P.; Pannarale, M.R. Electroretinographic Alterations in the Laurence-Moon-Bardet-Biedl Phenotype. Acta Ophthalmol. Scand. 2009, 74, 8–13. [Google Scholar] [CrossRef]
- Praidou, A.; Hagan, R.; Nayak, H.; Chandna, A. Multifocal Electroretinogram Contributes to Differentiation of Various Clinical Pictures within a Family with Bardet-Biedl Syndrome. Eye 2014, 28, 1136–1142. [Google Scholar] [CrossRef][Green Version]
- Makiyama, Y.; Ooto, S.; Hangai, M.; Takayama, K.; Uji, A.; Oishi, A.; Ogino, K.; Nakagawa, S.; Yoshimura, N. Macular Cone Abnormalities in Retinitis Pigmentosa with Preserved Central Vision Using Adaptive Optics Scanning Laser Ophthalmoscopy. PLoS ONE 2013, 8, e79447. [Google Scholar] [CrossRef]
- Tojo, N.; Nakamura, T.; Fuchizawa, C.; Oiwake, T.; Hayashi, A. Adaptive Optics Fundus Images of Cone Photoreceptors in the Macula of Patients with Retinitis Pigmentosa. Clin. Ophthalmol. 2013, 7, 203–210. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Park, S.P.; Lee, W.; Bae, E.J.; Greenstein, V.; Sin, B.H.; Chang, S.; Tsang, S.H. Early Structural Anomalies Observed by High-Resolution Imaging in Two Related Cases of Autosomal-Dominant Retinitis Pigmentosa. Ophthalmic Surg. Lasers Imaging Retin. 2014, 45, 469–473. [Google Scholar] [CrossRef] [PubMed]
- Gale, M.J.; Feng, S.; Titus, H.E.; Smith, T.B.; Pennesi, M.E. Interpretation of Flood-Illuminated Adaptive Optics Images in Subjects with Retinitis Pigmentosa BT—Retinal Degenerative Diseases. In Proceedings of the Advances in Experimental Medicine and Biology; Bowes Rickman, C., LaVail, M.M., Anderson, R.E., Grimm, C., Hollyfield, J., Ash, J., Eds.; Springer: New York, NY, USA, 2016; Volume 854, pp. 291–297. [Google Scholar]
- Sun, L.W.; Johnson, R.D.; Langlo, C.S.; Cooper, R.F.; Razeen, M.M.; Russillo, M.C.; Dubra, A.; Connor, T.B.; Han, D.P.; Pennesi, M.E.; et al. Assessing Photoreceptor Structure in Retinitis Pigmentosa and Usher Syndrome. Investig. Ophthalmol. Vis. Sci. 2016, 57, 2428–2442. [Google Scholar] [CrossRef] [PubMed]
- Nakatake, S.; Murakami, Y.; Funatsu, J.; Koyanagi, Y.; Akiyama, M.; Momozawa, Y.; Ishibashi, T.; Sonoda, K.-H.; Ikeda, Y. Early Detection of Cone Photoreceptor Cell Loss in Retinitis Pigmentosa Using Adaptive Optics Scanning Laser Ophthalmoscopy. Graefe’s Arch. Clin. Exp. Ophthalmol. 2019, 257, 1169–1181. [Google Scholar] [CrossRef] [PubMed]
- Aleman, T.S.; O’Neil, E.C.; O’Connor, K.; Jiang, Y.Y.; Aleman, I.A.; Bennett, J.; Morgan, J.I.W.; Toussaint, B.W. Bardet-Biedl Syndrome-7 (BBS7) Shows Treatment Potential and a Cone-Rod Dystrophy Phenotype That Recapitulates the Non-Human Primate Model. Ophthalmic Genet. 2021, 42, 252–265. [Google Scholar] [CrossRef] [PubMed]
- Estrada-Cuzcano, A.; Koenekoop, R.K.; Senechal, A.; De Baere, E.B.W.; de Ravel, T.; Banfi, S.; Kohl, S.; Ayuso, C.; Sharon, D.; Hoyng, C.B.; et al. BBS1 Mutations in a Wide Spectrum of Phenotypes Ranging from Nonsyndromic Retinitis Pigmentosa to Bardet-Biedl Syndrome. Arch. Ophthalmol. 2012, 130, 1425–1432. [Google Scholar] [CrossRef]
- Wang, X.; Wang, H.; Sun, V.; Tuan, H.-F.; Keser, V.; Wang, K.; Ren, H.; Lopez, I.; Zaneveld, J.E.; Siddiqui, S.; et al. Comprehensive Molecular Diagnosis of 179 Leber Congenital Amaurosis and Juvenile Retinitis Pigmentosa Patients by Targeted next Generation Sequencing. J. Med. Genet. 2013, 50, 674–688. [Google Scholar] [CrossRef]
- Riazuddin, S.A.; Iqbal, M.; Wang, Y.; Masuda, T.; Chen, Y.; Bowne, S.; Sullivan, L.S.; Waseem, N.H.; Bhattacharya, S.; Daiger, S.P.; et al. A Splice-Site Mutation in a Retina-Specific Exon of BBS8 Causes Nonsyndromic Retinitis Pigmentosa. Am. J. Hum. Genet. 2010, 86, 805–812. [Google Scholar] [CrossRef]
- Pretorius, P.R.; Aldahmesh, M.A.; Alkuraya, F.S.; Sheffield, V.C.; Slusarski, D.C. Functional Analysis of BBS3 A89V That Results in Non-Syndromic Retinal Degeneration. Hum. Mol. Genet. 2011, 20, 1625–1632. [Google Scholar] [CrossRef]
- Shevach, E.; Ali, M.; Mizrahi-Meissonnier, L.; McKibbin, M.; El-Asrag, M.; Watson, C.M.; Inglehearn, C.F.; Ben-Yosef, T.; Blumenfeld, A.; Jalas, C.; et al. Association between Missense Mutations in the BBS2 Gene and Nonsyndromic Retinitis Pigmentosa. JAMA Ophthalmol. 2015, 133, 312–318. [Google Scholar] [CrossRef]
- Niederlova, V.; Modrak, M.; Tsyklauri, O.; Huranova, M.; Stepanek, O. Meta-analysis of Genotype-phenotype Associations in Bardet-Biedl Syndrome Uncovers Differences among Causative Genes. Hum. Mutat. 2019, 40, 2068–2087. [Google Scholar] [CrossRef]
- Billingsley, G.; Deveault, C.; Héon, E. BBS Mutational Analysis: A Strategic Approach. Ophthalmic Genet. 2011, 32, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Fattahi, Z.; Rostami, P.; Najmabadi, A.; Mohseni, M.; Kahrizi, K.; Akbari, M.R.; Kariminejad, A.; Najmabadi, H. Mutation Profile of BBS Genes in Iranian Patients with Bardet-Biedl Syndrome: Genetic Characterization and Report of Nine Novel Mutations in Five BBS Genes. J. Hum. Genet. 2014, 59, 368–375. [Google Scholar] [CrossRef] [PubMed]
- Tao, T.; Wang, L.; Chong, W.; Yang, L.; Li, G. Characteristics of Genotype and Phenotype in Chinese Patients with Bardet–Biedl Syndrome. Int. Ophthalmol. 2020, 40, 2325–2343. [Google Scholar] [CrossRef] [PubMed]
- Suspitsin, E.N.; Imyanitov, E.N. Bardet-Biedl Syndrome. Mol. Syndromol. 2016, 7, 62–71. [Google Scholar] [CrossRef]
- Forsyth, R.L.; Gunay-Aygun, M. Bardet-Biedl Syndrome Overview. In GeneReviews® [Internet]; Adam, M.P., Mirzaa, G.M., Pagon, R.A., Eds.; University of Washington: Seattle, WA, USA, 2003; updated 2020. Available online: https://www.ncbi.nlm.nih.gov/books/NBK1363/ (accessed on 14 February 2022).
- Castro-Sánchez, S.; Álvarez-Satta, M.; Cortón, M.; Guillén, E.; Ayuso, C.; Valverde, D. Exploring Genotype-Phenotype Relationships in Bardet-Biedl Syndrome Families. J. Med. Genet. 2015, 52, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Forsythe, E.; Sparks, K.; Hoskins, B.E.; Bagkeris, E.; Mcgowan, B.M.; Carroll, P.V.; Huda, M.S.B.; Mujahid, S.; Peters, C.; Barrett, T.; et al. Genetic Predictors of Cardiovascular Morbidity in Bardet-Biedl Syndrome. Clin. Genet. 2015, 87, 343–349. [Google Scholar] [CrossRef]
- Simons, D.L.; Boye, S.L.; Hauswirth, W.W.; Wu, S.M. Gene Therapy Prevents Photoreceptor Death and Preserves Retinal Function in a Bardet-Biedl Syndrome Mouse Model. Proc. Natl. Acad. Sci. USA 2011, 108, 6276–6281. [Google Scholar] [CrossRef]
- Seo, S.; Mullins, R.F.; Dumitrescu, A.V.; Bhattarai, S.; Gratie, D.; Wang, K.; Stone, E.M.; Sheffield, V.; Drack, A.V. Subretinal Gene Therapy of Mice With Bardet-Biedl Syndrome Type 1. Investig. Opthalmology Vis. Sci. 2013, 54, 6118. [Google Scholar] [CrossRef] [PubMed]
- Datta, P.; Hendrickson, B.; Seo, S. Effective Arrest of Retinal Degeneration Depends on the Timing of Gene Restoration in a BBS17 Mouse Model. Investig. Ophthalmol. Vis. Sci. 2019, 60, 3394. [Google Scholar]
- Zhang, W.; Linjing, L.; Guangping, G.; Khanna, H. Gene Therapy Using a miniCEP290 Fragment Delays Photoreceptor Degeneration in a Mouse Model of Leber Congenital Amaurosis. Hum. Gene Ther. 2017, 29, 42–50. [Google Scholar] [CrossRef]
- Williams, C.L.; Uytingco, C.R.; Green, W.W.; McIntyre, J.C.; Ukhanov, K.; Zimmerman, A.D.; Shively, D.T.; Zhang, L.; Nishimura, D.Y.; Sheffield, V.C.; et al. Gene Therapeutic Reversal of Peripheral Olfactory Impairment in Bardet-Biedl Syndrome. Mol. Ther. 2017, 25, 904–916. [Google Scholar] [CrossRef]
- Uytingco, C.R.; Williams, C.L.; Green, W.W.; Shively, D.; Zhang, L.; Sheffield, V.C. Restoration of Olfactory Epithelium Ciliation and Odorant Detection in BBS4 Loss-of-Function Model of Bardet-Biedl Syndrome. Chem. Senses 2018, 43, e35. [Google Scholar]
- Peterson, S.M.; McGill, T.J.; Puthussery, T.; Stoddard, J.; Renner, L.; Lewis, A.D.; Colgin, L.M.A.; Gayet, J.; Wang, X.; Prongay, K.; et al. Bardet-Biedl Syndrome in Rhesus Macaques: A Nonhuman Primate Model of Retinitis Pigmentosa. Exp. Eye Res. 2019, 189, 107825. [Google Scholar] [CrossRef]
- Weisschuh, N.; Obermaier, C.; Battke, F. Genetic Architecture of Inherited Retinal Degeneration in Germany: A Large Cohort Study from a Single Diagnostic Center over a 9-year Period. Hum. Mutat. 2020, 69, 722–737. [Google Scholar] [CrossRef]
- Sharon, D.; Banin, E. Nonsyndromic Retinitis Pigmentosa Is Highly Prevalent in the Jerusalem Region with a High Frequency of Founder Mutations. Mol Vis 2015, 21, 783–792. [Google Scholar]
- Mykytyn, K.; Nishimura, D.Y.; Searby, C.C.; Shastri, M.; Yen, H.J.; Beck, J.S.; Braun, T.; Streb, L.M.; Cornier, A.S.; Cox, G.F.; et al. Identification of the Gene (BBS1) Most Commonly Involved in Bardet-Biedl Syndrome, a Complex Human Obesity Syndrome. Nat Genet 2002, 31, 435–438. [Google Scholar] [CrossRef]
- Deveault, C.; Billingsley, G.; Duncan, J.L.; Bin, J.; Theal, R.; Vincent, A.; Fieggen, K.J.; Gerth, C.; Noordeh, N.; Traboulsi, E.I.; et al. BBS Genotype-Phenotype Assessment of a Multiethnic Patient Cohort Calls for a Revision of the Disease Definition. Hum Mutat 2011, 32, 610–619. [Google Scholar] [CrossRef]
- Hjortshoj, T.D.; Gronskov, K.; Philp, A.R.; Nishimura, D.Y.; Riise, R.; Sheffield, V.C.; Rosenberg, T.; Brondum-Nielsen, K. Bardet-Biedl Syndrome in Denmark—Report of 13 Novel Sequence Variations in Six Genes. Hum. Mutat. 2010, 31, 429–436. [Google Scholar] [CrossRef]
- Glockle, N.; Kohl, S.; Mohr, J.; Scheurenbrand, T.; Sprecher, A.; Weisschuh, N.; Bernd, A.; Rudolph, G.; Schubach, M.; Poloschek, C.; et al. Panel-Based next Generation Sequencing as a Reliable and Efficient Technique to Detect Mutations in Unselected Patients with Retinal Dystrophies. Eur. J. Hum. Genet. 2014, 22, 99–104. [Google Scholar] [CrossRef]
- Karmous-Benailly, H.; Martinovic, J.; Gubler, M.C.; Sirot, Y.; Clech, L.; Ozilou, C.; Auge, J.; Brahimi, N.; Etchevers, H.; Detrait, E.; et al. Antenatal Presentation of Bardet-Biedl Syndrome May Mimic Meckel Syndrome. Am. J. Hum. Genet. 2005, 76, 493–504. [Google Scholar] [CrossRef]
- Stone, D.L.; Slavotinek, A.; Bouffard, G.G.; Banerjee-Basu, S.; Baxevanis, A.D.; Barr, M.; Biesecker, L.G. Mutation of a Gene Encoding a Putative Chaperonin Causes McKusick-Kaufman Syndrome. Nat. Genet. 2000, 25, 79–82. [Google Scholar] [CrossRef]
- Bin, J.; Madhavan, J.; Ferrini, W.; Mok, C.A.; Billingsley, G.; Heon, E. BBS7 and TTC8 (BBS8) Mutations Play a Minor Role in the Mutational Load of Bardet-Biedl Syndrome in a Multiethnic Population. Hum. Mutat. 2009, 30, E737–E746. [Google Scholar] [CrossRef]
- Badano, J.L.; Ansley, S.J.; Leitch, C.C.; Lewis, R.A.; Lupski, J.R.; Katsanis, N. Identification of a Novel Bardet-Biedl Syndrome Protein, BBS7, That Shares Structural Features with BBS1 and BBS2. Am. J. Hum. Genet. 2003, 72, 650–658. [Google Scholar] [CrossRef]
- Weisschuh, N.; Mayer, A.K.; Strom, T.M.; Kohl, S.; Glöckle, N.; Schubach, M.; Andreasson, S.; Bernd, A.; Birch, D.G.; Hamel, C.P.; et al. Mutation Detection in Patients with Retinal Dystrophies Using Targeted Next Generation Sequencing. PLoS ONE 2016, 11, e0145951. [Google Scholar] [CrossRef]
- Stoetzel, C.; Laurier, V.; Davis, E.E.; Muller, J.; Rix, S.; Badano, J.L.; Leitch, C.C.; Salem, N.; Chouery, E.; Corbani, S.; et al. BBS10 Encodes a Vertebrate-Specific Chaperonin-like Protein and Is a Major BBS Locus. Nat. Genet. 2006, 38, 521–524. [Google Scholar] [CrossRef]
- Esposito, G.; Testa, F.; Zacchia, M.; Crispo, A.A.; Di Iorio, V.; Capolongo, G.; Rinaldi, L.; D’Antonio, M.; Fioretti, T.; Iadicicco, P.; et al. Genetic Characterization of Italian Patients with Bardet-Biedl Syndrome and Correlation to Ocular, Renal and Audio-Vestibular Phenotype: Identification of Eleven Novel Pathogenic Sequence Variants. BMC Med. Genet. 2017, 18, 10. [Google Scholar] [CrossRef]
- Harville, H.M.; Held, S.; Diaz-Font, A.; Davis, E.E.; Diplas, B.H.; Lewis, R.A.; Borochowitz, Z.U.; Zhou, W.; Chaki, M.; MacDonald, J.; et al. Identification of 11 Novel Mutations in Eight BBS Genes by High-Resolution Homozygosity Mapping. J. Med. Genet. 2010, 47, 262–267. [Google Scholar] [CrossRef]
- Janssen, S.; Ramaswami, G.; Davis, E.E.; Hurd, T.; Airik, R.; Kasanuki, J.M.; Van Der Kraak, L.; Allen, S.J.; Beales, P.L.; Katsanis, N.; et al. Mutation Analysis in Bardet-Biedl Syndrome by DNA Pooling and Massively Parallel Resequencing in 105 Individuals. Hum. Genet. 2011, 129, 79–90. [Google Scholar] [CrossRef]
- Stoetzel, C.; Muller, J.; Laurier, V.; Davis, E.E.; Zaghloul, N.A.; Vicaire, S.; Jacquelin, C.; Plewniak, F.; Leitch, C.C.; Sarda, P.; et al. Identification of a Novel BBS Gene (BBS12) Highlights the Major Role of a Vertebrate-Specific Branch of Chaperonin-Related Proteins in Bardet-Biedl Syndrome. Am. J. Hum. Genet. 2007, 80, 1–11. [Google Scholar] [CrossRef]
- Dulfer, E.; Hoefsloot, L.H.; Timmer, A.; Mom, C.; van Essen, A.J. Two Sibs with Bardet-Biedl Syndrome Due to Mutations in BBS12: No Clues for Modulation by a Third Mutation in BBS10. Am. J. Med. Genet. A 2010, 152A, 2666–2669. [Google Scholar] [CrossRef] [PubMed]
- Otto, E.A.; Hurd, T.W.; Airik, R.; Chaki, M.; Zhou, W.; Stoetzel, C.; Patil, S.B.; Levy, S.; Ghosh, A.K.; Murga-Zamalloa, C.A.; et al. Candidate Exome Capture Identifies Mutation of SDCCAG8 as the Cause of a Retinal-Renal Ciliopathy. Nat. Genet. 2010, 42, 840–850. [Google Scholar] [CrossRef] [PubMed]
- Richards, S.; Aziz, N.; Bale, S.; Bick, D.; Das, S.; Gastier-Foster, J.; Grody, W.W.; Hegde, M.; Lyon, E.; Spector, E.; et al. Standards and Guidelines for the Interpretation of Sequence Variants: A Joint Consensus Recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. 2015, 17, 405–424. [Google Scholar] [CrossRef] [PubMed]
- Allikmets, R.; Singh, N.; Sun, H.; Shroyer, N.F.; Hutchinson, A.; Chidambaram, A.; Gerrard, B.; Baird, L.; Stauffer, D.; Peiffer, A.; et al. A Photoreceptor Cell-Specific ATP-Binding Transporter Gene (ABCR) Is Mutated in Recessive Stargardt Macular Dystrophy. Nat. Genet. 1997, 15, 236–246. [Google Scholar] [CrossRef] [PubMed]
- Stenirri, S.; Fermo, I.; Battistella, S.; Galbiati, S.; Soriani, N.; Paroni, R.; Manitto, M.P.; Martina, E.; Brancato, R.; Allikmets, R.; et al. Denaturing HPLC Profiling of the ABCA4 Gene for Reliable Detection of Allelic Variations. Clin. Chem. 2004, 50, 1336–1343. [Google Scholar] [CrossRef] [PubMed]
- Shroyer, N.F.; Lewis, R.A.; Lupski, J.R. Complex Inheritance of ABCR Mutations in Stargardt Disease: Linkage Disequilibrium, Complex Alleles, and Pseudodominance. Hum. Genet. 2000, 106, 244–248. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).