Specialized Metabolites and Valuable Molecules in Crop and Medicinal Plants: The Evolution of Their Use and Strategies for Their Production
Abstract
1. Valuable Molecules from Plants: An Ancient Resource for Modern Challenges
2. Molecules in Crop and Medicinal Plants: The Evolution of Their Use
3. Production Improvement of Plant-Derived Compounds by Stress Elicitation
3.1. Laboratory Scale
3.2. Industrial Scale
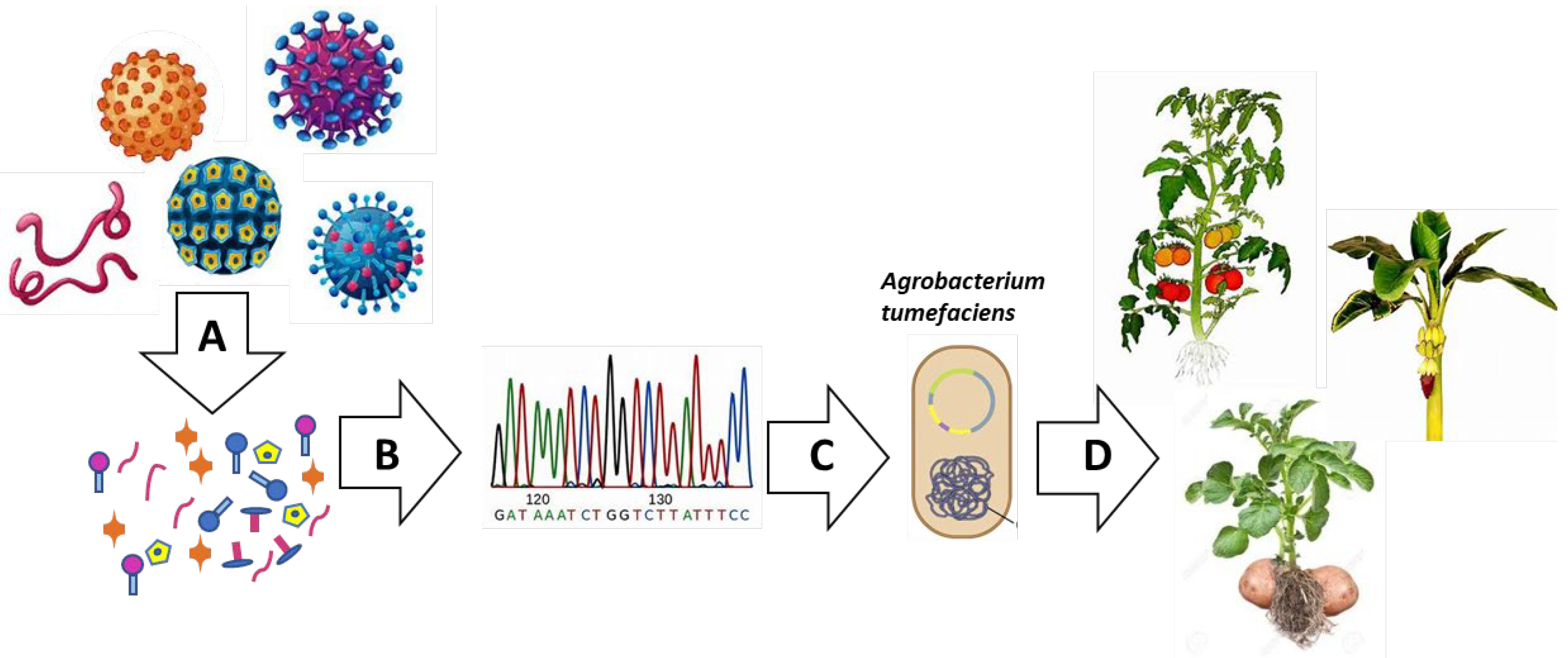
4. Plant as Biofactories
5. Novel Perspectives in the Production of Plant-Derived Compounds
5.1. Genome Editing
5.2. Synthetic Biology
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Câmara, J.S.; Albuquerque, B.R.; Aguiar, J.; Corrêa, R.C.G.; Gonçalves, J.L.; Granato, D.; Pereira, J.A.M.; Barros, L.; Ferreira, I.C.F.R. Food Bioactive Compounds and Emerging Techniques for Their Extraction: Polyphenols as a Case Study. Foods 2020, 10, 37. [Google Scholar] [CrossRef]
- Guiné, R.P.F.; Florença, S.G.; Barroca, M.J.; Anjos, O. The Link between the Consumer and the Innovations in Food Product Development. Foods 2020, 9, 1317. [Google Scholar] [CrossRef]
- Pisanello, D. Chemistry of Foods: EU Legal and Regulatory Approaches; SpringerBriefs in Molecular Science; Springer International Publishing: Cham, Switzerland, 2014; ISBN 978-3-319-03433-1. [Google Scholar]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs from 1981 to 2014. J. Nat. Prod. 2016, 79, 629–661. [Google Scholar] [CrossRef]
- Balunas, M.J.; Kinghorn, A.D. Drug discovery from medicinal plants. Life Sci. 2005, 78, 431–441. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the nearly four decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770–803. [Google Scholar] [CrossRef] [PubMed]
- K, L. Botanical and plant-derived drugs: Global markets. BCC, Res. 2017.
- Hamilton, A.C. Medicinal plants, conservation and livelihoods. Biodivers. Conserv. 2004, 13, 1477–1517. [Google Scholar] [CrossRef]
- Aizen, M.A.; Aguiar, S.; Biesmeijer, J.C.; Garibaldi, L.A.; Inouye, D.W.; Jung, C.; Martins, D.J.; Medel, R.; Morales, C.L.; Ngo, H.; et al. Global agricultural productivity is threatened by increasing pollinator dependence without a parallel increase in crop diversification. Glob. Chang. Biol. 2019, 25, 3516–3527. [Google Scholar] [CrossRef] [PubMed]
- Dauber, J.; Jones, M.B.; Stout, J.C. The impact of biomass crop cultivation on temperate biodiversity. GCB Bioenergy 2010, 2, 289–309. [Google Scholar] [CrossRef]
- Zhang, G.; Liu, X.; Quan, Z.; Cheng, S.; Xu, X.; Pan, S.; Xie, M.; Zeng, P.; Yue, Z.; Wang, W.; et al. Genome sequence of foxtail millet (Setaria italica) provides insights into grass evolution and biofuel potential. Nat. Biotechnol. 2012, 30, 549–554. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Liu, C.; Zhao, G.; Gong, X.; Dang, K.; Yang, Q.; Feng, B. Transcriptome analysis reveals the mechanism associated with dynamic changes in fatty acid and phytosterol content in foxtail millet (Setaria italica) during seed development. Food Res. Int. 2021, 145, 110429. [Google Scholar] [CrossRef] [PubMed]
- Ghimire, B.K.; Yu, C.Y.; Kim, S.-H.; Chung, I.-M. Assessment of Diversity in the Accessions of Setaria italica L. Based on Phytochemical and Morphological Traits and ISSR Markers. Molecules 2019, 24, 1486. [Google Scholar] [CrossRef]
- Saito, K. Phytochemical genomics—A new trend. Curr. Opin. Plant Biol. 2013, 16, 373–380. [Google Scholar] [CrossRef]
- Stavrianidi, A. A classification of liquid chromatography mass spectrometry techniques for evaluation of chemical composition and quality control of traditional medicines. J. Chromatogr. A 2020, 1609, 460501. [Google Scholar] [CrossRef]
- Chen, S.-L.; Yu, H.; Luo, H.-M.; Wu, Q.; Li, C.-F.; Steinmetz, A. Conservation and sustainable use of medicinal plants: Problems, progress, and prospects. Chin. Med. 2016, 11, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Petrovska, B.B. Historical review of medicinal plants′ usage. Pharmacogn. Rev. 2012, 6, 1–5. [Google Scholar] [CrossRef]
- Rizzo, P.; Altschmied, L.; Ravindran, B.M.; Rutten, T.; D’Auria, J.C. The Biochemical and Genetic Basis for the Biosynthesis of Bioactive Compounds in Hypericum Perforatum L., One of the Largest Medicinal Crops in Europe. Genes 2020, 11, 1210. [Google Scholar] [CrossRef] [PubMed]
- Leonti, M.; Verpoorte, R. Traditional Mediterranean and European herbal medicines. J. Ethnopharmacol. 2017, 199, 161–167. [Google Scholar] [CrossRef]
- Dreher, K. Putting the Plant Metabolic Network Pathway Databases to Work: Going Offline to Gain New Capabilities. Adv. Struct. Saf. Stud. 2014, 1083, 151–171. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, J.; He, S.; Gao, Y.; Ma, X.; Gao, Y.; Zhang, G.; Kui, L.; Wang, W.; Wang, Y.; et al. HMOD: An Omics Database for Herbal Medicine Plants. Mol. Plant 2018, 11, 757–759. [Google Scholar] [CrossRef]
- Chae, L.; Kim, T.; Nilo-Poyanco, R.; Rhee, S.Y. Genomic Signatures of Specialized Metabolism in Plants. Science 2014, 344, 510–513. [Google Scholar] [CrossRef]
- Schläpfer, P.; Zhang, P.; Wang, C.; Kim, T.; Banf, M.; Chae, L.; Dreher, K.; Chavali, A.K.; Nilo-Poyanco, R.; Bernard, T.; et al. Genome-Wide Prediction of Metabolic Enzymes, Pathways, and Gene Clusters in Plants. Plant Physiol. 2017, 173, 2041–2059. [Google Scholar] [CrossRef]
- Nützmann, H.-W.; Osbourn, A. Gene clustering in plant specialized metabolism. Curr. Opin. Biotechnol. 2014, 26, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Gao, M.; Kang, S.; Yang, C.; Meng, H.; Yang, Y.; Zhao, X.; Gao, Z.; Xu, Y.; Jin, Y.; et al. Molecular Mechanism Underlying Mechanical Wounding-Induced Flavonoid Accumulation in Dalbergia odorifera T. Chen, an Endangered Tree That Produces Chinese Rosewood. Genes 2020, 11, 478. [Google Scholar] [CrossRef] [PubMed]
- Andrzejewska, J.; Martinelli, T.; Sadowska, K. Silybum marianum: Non-medical exploitation of the species. Ann. Appl. Biol. 2015, 167, 285–297. [Google Scholar] [CrossRef]
- Gad, D.; Elhaak, M.; Pompa, A.; Mattar, M.; Zayed, M.; Fraternale, D.; Dietz, K.-J. A New Strategy to Increase Production of Genoprotective Bioactive Molecules from Cotyledon-Derived Silybum marianum L. Callus. Genes 2020, 11, 791. [Google Scholar] [CrossRef]
- Christaki, E.; Bonos, E.; Florou-Paneri, P. Nutritional and Functional Properties of Cynara Crops (Globe Artichoke and Car-doon) and Their Potential Applications: A Review. Int. J. Appl. Sci. Technol. 2012, 2, 64–70. [Google Scholar]
- Graziani, G.; Docimo, T.; De Palma, M.; Sparvoli, F.; Izzo, L.; Tucci, M.; Ritieni, A. Changes in Phenolics and Fatty Acids Composition and Related Gene Expression during the Development from Seed to Leaves of Three Cultivated Cardoon Genotypes. Antioxidants 2020, 9, 1096. [Google Scholar] [CrossRef] [PubMed]
- Docimo, T.; De Stefano, R.; Cappetta, E.; Piccinelli, A.L.; Celano, R.; De Palma, M.; Tucci, M. Physiological, Biochemical, and Metabolic Responses to Short and Prolonged Saline Stress in Two Cultivated Cardoon Genotypes. Plants 2020, 9, 554. [Google Scholar] [CrossRef]
- Gominho, J.; Lourenco, A.; Palma, P.; Lourenço, M.; Curt, M.; Fernández, J.; Pereira, H. Large scale cultivation of Cynara cardunculus L. for biomass production—A case study. Ind. Crops Prod. 2011, 33, 1–6. [Google Scholar] [CrossRef]
- Bouis, H.E.; Saltzman, A. Improving nutrition through biofortification: A review of evidence from HarvestPlus, 2003 through 2016. Glob. Food Secur. 2017, 12, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Siwela, M.; Pillay, K.; Govender, L.; Lottering, S.; Mudau, F.N.; Modi, A.T.; Mabhaudhi, T. Biofortified Crops for Combating Hidden Hunger in South Africa: Availability, Acceptability, Micronutrient Retention and Bioavailability. Foods 2020, 9, 815. [Google Scholar] [CrossRef] [PubMed]
- D’Amelia, V.; Aversano, R.; Chiaiese, P.; Carputo, D. The antioxidant properties of plant flavonoids: Their exploitation by molecular plant breeding. Phytochem. Rev. 2018, 17, 611–625. [Google Scholar] [CrossRef]
- Romano, A.; D’Amelia, V.; Gallo, V.; Palomba, S.; Carputo, D.; Masi, P. Relationships between composition, microstructure and cooking performances of six potato varieties. Food Res. Int. 2018, 114, 10–19. [Google Scholar] [CrossRef] [PubMed]
- De Masi, L.; Bontempo, P.; Rigano, D.; Stiuso, P.; Carafa, V.; Nebbioso, A.; Piacente, S.; Montoro, P.; Aversano, R.; D’Amelia, V.; et al. Comparative Phytochemical Characterization, Genetic Profile, and Antiproliferative Activity of Polyphenol-Rich Extracts from Pigmented Tubers of Different Solanum tuberosum Varieties. Molecules 2020, 25, 233. [Google Scholar] [CrossRef] [PubMed]
- Russo, M.; Moccia, S.; Spagnuolo, C.; Tedesco, I.; Russo, G.L. Roles of flavonoids against coronavirus infection. Chem. Interactions 2020, 328, 109211. [Google Scholar] [CrossRef] [PubMed]
- Iorizzo, M.; Curaba, J.; Pottorff, M.; Ferruzzi, M.G.; Simon, P.; Cavagnaro, P.F. Carrot Anthocyanins Genetics and Genomics: Status and Perspectives to Improve Its Application for the Food Colorant Industry. Genes 2020, 11, 906. [Google Scholar] [CrossRef] [PubMed]
- Brumbley, S.M. Transgenic Plants used as a Bioreactor System. U.S. Patent No. 7,754,943, 13 July 2010. [Google Scholar]
- Bringi, V. Enhanced Production of Paclitaxel and Taxanes by Cell Cultures of Taxus Species. U.S. Patent No. 8,338,143, 25 December 2012. [Google Scholar]
- Kossmann, J.; Lorberth, R. Methods of Using Tubers Having Genetically Modified Potato Plant Cells. U.S. Patent No. 8,586,722, 19 November 2013. [Google Scholar]
- Luo, J. Method for Modifying Anthocyanin Expression in Solanaceous Plants. U.S. Patent No. 8,802,925, 12 August 2014. [Google Scholar]
- Cirpus, P. Oils, Lipids and Fatty Acids Produced in Transgenic Brassica Plant. U.S. Patent No. 10,533,182, 14 January 2020. [Google Scholar]
- Philippe, R. Microbial Production of Steviol Glycosides. U.S. Patent No. 10,743,567, 18 August 2020. [Google Scholar]
- Yanay, Y.; Raviv, L. Methods and Compositions for Producing Cannabinoids. W.O. Patent No. WO2020222233A1, 5 November 2020. [Google Scholar]
- Ajikumar, P.K.; Stephanopoulos, G.; Heng, P.T. Microbial Engineering for the Production of Chemical and Pharmaceutical Products from the Isoprenoid Pathway. U.S. Patent No. 8,512,988, 20 August 2013. [Google Scholar]
- Rademacher, T. Method for the Generation and Cultivation of a Plant Cell Pack. U.S. Patent No. 10,638,679, 5 May 2020. [Google Scholar]
- Huang, N. Monocot Seed Product Comprising a Human Serum Albumin Protein. U.S. Patent No. 8,158,857, 17 April 2012. [Google Scholar]
- He, Q.; Ren, Y.; Zhao, W.; Li, R.; Zhang, L. Low Temperature Promotes Anthocyanin Biosynthesis and Related Gene Expression in the Seedlings of Purple Head Chinese Cabbage (Brassica rapa L.). Genes 2020, 11, 81. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.; Li, M.; Dai, S. Ectopic Expression of Multiple Chrysanthemum (Chrysanthemum × morifolium) R2R3-MYB Transcription Factor Genes Regulates Anthocyanin Accumulation in Tobacco. Genes 2019, 10, 777. [Google Scholar] [CrossRef] [PubMed]
- Su, H.; Zhang, X.; He, Y.; Li, L.; Wang, Y.; Hong, G.; Xu, P. Transcriptomic Analysis Reveals the Molecular Adaptation of Three Major Secondary Metabolic Pathways to Multiple Macronutrient Starvation in Tea (Camellia sinensis). Genes 2020, 11, 241. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.; Li, M.; Dai, S. iTRAQ-Based Protein Profiling Provides Insights into the Mechanism of Light-Induced Anthocyanin Biosynthesis in Chrysanthemum (Chrysanthemum × morifolium). Genes 2019, 10, 1024. [Google Scholar] [CrossRef] [PubMed]
- Holzmeyer, L.; Hartig, A.-K.; Franke, K.; Brandt, W.; Muellner-Riehl, A.N.; Wessjohann, L.A.; Schnitzler, J. Evaluation of plant sources for antiinfective lead compound discovery by correlating phylogenetic, spatial, and bioactivity data. Proc. Natl. Acad. Sci. USA 2020, 117, 12444–12451. [Google Scholar] [CrossRef]
- Carillo, P.; Cirillo, C.; De Micco, V.; Arena, C.; De Pascale, S.; Rouphael, Y. Morpho-anatomical, physiological and biochemical adaptive responses to saline water of Bougainvillea spectabilis Willd. trained to different canopy shapes. Agric. Water Manag. 2019, 212, 12–22. [Google Scholar] [CrossRef]
- Golldack, D.; Li, C.; Mohan, H.; Probst, N. Tolerance to drought and salt stress in plants: Unraveling the signaling networks. Front. Plant Sci. 2014, 5, 151. [Google Scholar] [CrossRef] [PubMed]
- Julkowska, M.; Testerink, C. Tuning plant signaling and growth to survive salt. Trends Plant Sci. 2015, 20, 586–594. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Lv, Y.; Jahan, N.; Chen, G.; Ren, D.; Guo, L. Sensing of Abiotic Stress and Ionic Stress Responses in Plants. Int. J. Mol. Sci. 2018, 19, 3298. [Google Scholar] [CrossRef]
- Fang, C.; Fernie, A.R.; Luo, J. Exploring the Diversity of Plant Metabolism. Trends Plant Sci. 2019, 24, 83–98. [Google Scholar] [CrossRef] [PubMed]
- Pichersky, E.; Gang, D.R. Genetics and biochemistry of secondary metabolites in plants: An evolutionary perspective. Trends Plant Sci. 2000, 5, 439–445. [Google Scholar] [CrossRef]
- Pichersky, E.; Lewinsohn, E. Convergent Evolution in Plant Specialized Metabolism. Annu. Rev. Plant Biol. 2011, 62, 549–566. [Google Scholar] [CrossRef] [PubMed]
- Malhi, Y.; Franklin, J.; Seddon, N.; Solan, M.; Turner, M.G.; Field, C.B.; Knowlton, N. Climate change and ecosystems: Threats, opportunities and solutions. Philos. Trans. R. Soc. B Biol. Sci. 2020, 375, 20190104. [Google Scholar] [CrossRef]
- Alagoz, Y.; Gurkok, T.; Zhang, B.; Unver, T. Manipulating the Biosynthesis of Bioactive Compound Alkaloids for Next-Generation Metabolic Engineering in Opium Poppy Using CRISPR-Cas 9 Genome Editing Technology. Sci. Rep. 2016, 6, 30910. [Google Scholar] [CrossRef] [PubMed]
- Weng, J.-K.; Noel, J.P. The Remarkable Pliability and Promiscuity of Specialized Metabolism. Cold Spring Harb. Symp. Quant. Biol. 2012, 77, 309–320. [Google Scholar] [CrossRef]
- Di Ferdinando, M.; Brunetti, C.; Fini, A.; Tattini, M. Flavonoids as Antioxidants in Plants Under Abiotic Stresses. In Abiotic Stress Responses in Plants; Springer: Berlin, Germany, 2011; pp. 159–179. [Google Scholar]
- Hernández, I.; Alegre, L.; Van Breusegem, F.; Munné-Bosch, S. How relevant are flavonoids as antioxidants in plants? Trends Plant Sci. 2009, 14, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Holopainen, J.K.; Virjamo, V.; Ghimire, R.P.; Blande, J.D.; Julkunen-Tiitto, R.; Kivimäenpää, M. Climate Change Effects on Secondary Compounds of Forest Trees in the Northern Hemisphere. Front. Plant Sci. 2018, 9, 1445. [Google Scholar] [CrossRef]
- Mittler, R. Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci. 2002, 7, 405–410. [Google Scholar] [CrossRef]
- Zhu, J.-K. Abiotic Stress Signaling and Responses in Plants. Cell 2016, 167, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Baxter, A.; Mittler, R.; Suzuki, N. ROS as key players in plant stress signalling. J. Exp. Bot. 2014, 65, 1229–1240. [Google Scholar] [CrossRef] [PubMed]
- Agati, G.; Azzarello, E.; Pollastri, S.; Tattini, M. Flavonoids as antioxidants in plants: Location and functional significance. Plant Sci. 2012, 196, 67–76. [Google Scholar] [CrossRef]
- Jalmi, S.K.; Sinha, A.K. ROS mediated MAPK signaling in abiotic and biotic stress- striking similarities and differences. Front. Plant Sci. 2015, 6, 769. [Google Scholar] [CrossRef]
- Minibayeva, F.; Beckett, R.P.; Kranner, I. Roles of apoplastic peroxidases in plant response to wounding. Phytochemistry 2015, 112, 122–129. [Google Scholar] [CrossRef]
- Balestrini, R.; Brunetti, C.; Cammareri, M.; Caretto, S.; Cavallaro, V.; Cominelli, E.; De Palma, M.; Docimo, T.; Giovinazzo, G.; Grandillo, S.; et al. Strategies to Modulate Specialized Metabolism in Mediterranean Crops: From Molecular Aspects to Field. Int. J. Mol. Sci. 2021, 22, 2887. [Google Scholar] [CrossRef]
- D’Amelia, V.; Aversano, R.; Ruggiero, A.; Batelli, G.; Appelhagen, I.; Dinacci, C.; Hill, L.; Martin, C.; Carputo, D. Subfunctionalization of duplicate MYB genes in Solanum commersonii generated the cold-induced ScAN2 and the anthocyanin regulator ScAN1. Plant Cell Environ. 2017, 41, 1038–1051. [Google Scholar] [CrossRef]
- Cirillo, V.; D’Amelia, V.; Esposito, M.; Amitrano, C.; Carillo, P.; Carputo, D.; Maggio, A. Anthocyanins Are Key Regulators of Drought Stress Tolerance in Tobacco. Biology 2021, 10, 139. [Google Scholar] [CrossRef] [PubMed]
- Landi, M.; Tattini, M.; Gould, K.S. Multiple functional roles of anthocyanins in plant-environment interactions. Environ. Exp. Bot. 2015, 119, 4–17. [Google Scholar] [CrossRef]
- Ahmed, N.U.; Park, J.-I.; Jung, H.-J.; Hur, Y.; Nou, I.-S. Anthocyanin biosynthesis for cold and freezing stress tolerance and desirable color in Brassica rapa. Funct. Integr. Genom. 2014, 15, 383–394. [Google Scholar] [CrossRef]
- He, Q.; Lu, Q.; He, Y.; Wang, Y.; Zhang, N.; Zhao, W.; Zhang, L. Dynamic Changes of the Anthocyanin Biosynthesis Mechanism During the Development of Heading Chinese Cabbage (Brassica rapa L.) and Arabidopsis Under the Control of BrMYB2. Front. Plant Sci. 2020, 11, 3766. [Google Scholar] [CrossRef] [PubMed]
- Jaakola, L. New insights into the regulation of anthocyanin biosynthesis in fruits. Trends Plant Sci. 2013, 18, 477–483. [Google Scholar] [CrossRef] [PubMed]
- Takos, A.M.; Jaffé, F.W.; Jacob, S.R.; Bogs, J.; Robinson, S.; Walker, A.R. Light-Induced Expression of a MYB Gene Regulates Anthocyanin Biosynthesis in Red Apples. Plant Physiol. 2006, 142, 1216–1232. [Google Scholar] [CrossRef]
- Zhang, F.; Zhu, G.; Du, L.; Shang, X.; Cheng, C.; Yang, B.; Hu, Y.; Cai, C.; Guo, W. Genetic regulation of salt stress tolerance revealed by RNA-Seq in cotton diploid wild species, Gossypium davidsonii. Sci. Rep. 2016, 6, 20582. [Google Scholar] [CrossRef] [PubMed]
- Ang, L.-H.; Chattopadhyay, S.; Wei, N.; Oyama, T.; Okada, K.; Batschauer, A.; Deng, X.-W. Molecular Interaction between COP1 and HY5 Defines a Regulatory Switch for Light Control of Arabidopsis Development. Mol. Cell 1998, 1, 213–222. [Google Scholar] [CrossRef]
- Fasano, R.; Gonzalez, N.; Tosco, A.; Piaz, F.D.; Docimo, T.; Serrano, R.; Grillo, S.; Leone, A.; Inzé, D. Role of Arabidopsis UV RESISTANCE LOCUS 8 in Plant Growth Reduction under Osmotic Stress and Low Levels of UV-B. Mol. Plant 2014, 7, 773–791. [Google Scholar] [CrossRef]
- Legris, M.; Nieto, C.; Sellaro, R.; Prat, S.; Casal, J.J. Perception and signalling of light and temperature cues in plants. Plant J. 2017, 90, 683–697. [Google Scholar] [CrossRef]
- Ochoa-Villarreal, M.; Howat, S.; Hong, S.; Jang, M.O.; Jin, Y.-W.; Lee, E.-K.; Loake, G.J. Plant cell culture strategies for the production of natural products. BMB Rep. 2016, 49, 149–158. [Google Scholar] [CrossRef] [PubMed]
- Davies, K.M.; Deroles, S.C. Prospects for the use of plant cell cultures in food biotechnology. Curr. Opin. Biotechnol. 2014, 26, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Krasteva, G.; Georgiev, V.; Pavlov, A. Recent applications of plant cell culture technology in cosmetics and foods. Eng. Life Sci. 2021, 21, 68–76. [Google Scholar] [CrossRef]
- Eibl, R.; Meier, P.; Stutz, I.; Schildberger, D.; Hühn, T.; Eibl, D. Plant cell culture technology in the cosmetics and food industries: Current state and future trends. Appl. Microbiol. Biotechnol. 2018, 102, 8661–8675. [Google Scholar] [CrossRef]
- Ramirez-Estrada, K.; Vidal-Limon, H.; Hidalgo, D.; Moyano, E.; Golenioswki, M.; Cusidó, R.M.; Palazon, J. Elicitation, an ef-fective strategy for the biotechnological production of bioactive high-added value compounds in plant cell factories. Molecules 2016, 21, 182. [Google Scholar] [CrossRef] [PubMed]
- Wilson, S.A.; Roberts, S.C. Recent advances towards development and commercialization of plant cell culture processes for the synthesis of biomolecules. Plant Biotechnol. J. 2011, 10, 249–268. [Google Scholar] [CrossRef]
- Nazir, M.; Ullah, M.A.; Younas, M.; Siddiquah, A.; Shah, M.; Giglioli-Guivarc’H, N.; Hano, C.; Abbasi, B.H. Light-mediated biosynthesis of phenylpropanoid metabolites and antioxidant potential in callus cultures of purple basil (Ocimum basilicum L. var purpurascens). Plant Cell Tissue Organ Cult. (PCTOC) 2020, 142, 107–120. [Google Scholar] [CrossRef]
- Srivastava, M.; Singh, G.; Sharma, S.; Shukla, S.; Misra, P. Elicitation Enhanced the Yield of Glycyrrhizin and Antioxidant Activities in Hairy Root Cultures of Glycyrrhiza glabra L. J. Plant Growth Regul. 2019, 38, 373–384. [Google Scholar] [CrossRef]
- Hegazi, G.; Ghareb, H.; Gabr, M. Ephedrine production from suspension cultures of Ephedra alata L. callus. Biotechnology 2020, 101, 25–33. [Google Scholar] [CrossRef]
- Skrzypczak-Pietraszek, E.; Piska, K.; Pietraszek, J. Enhanced production of the pharmaceutically important polyphenolic compounds inVitex agnus castusL. shoot cultures by precursor feeding strategy. Eng. Life Sci. 2018, 18, 287–297. [Google Scholar] [CrossRef]
- Nadeem, M.; Ahmed, W.; Zahir, A.; Hano, C.; Abbasi, B.H. Salicylic acid-enhanced biosynthesis of pharmacologically important lignans and neo lignans in cell suspension culture of Linum ussitatsimum L. Eng. Life Sci. 2018, 19, 168–174. [Google Scholar] [CrossRef]
- Teptat, P.; Tansakul, P.; Sakunphueak, A. Effects of Sequential and Simultaneous Double Elicitation on Aloe-Emodin Produc-tion in Cassia Tora Root Cultures. Res. Sq. 2020. [Google Scholar] [CrossRef]
- Razavizadeh, R.; Adabavazeh, F.; Komatsu, S. Chitosan effects on the elevation of essential oils and antioxidant activity of Carum copticum L. seedlings and callus cultures under in vitro salt stress. J. Plant Biochem. Biotechnol. 2020, 29, 473–483. [Google Scholar] [CrossRef]
- Linden, J.C.; Phisalaphong, M. Oligosaccharides potentiate methyl jasmonate-induced production of paclitaxel in Taxus canadensis. Plant Sci. 2000, 158, 41–51. [Google Scholar] [CrossRef]
- Hao, X.; Shi, M.; Cui, L.; Xu, C.; Zhang, Y.; Kai, G. Effects of methyl jasmonate and salicylic acid on tanshinone production and biosynthetic gene expression in transgenicSalvia miltiorrhizahairy roots. Biotechnol. Appl. Biochem. 2014, 62, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Wang, B.; Li, H.; Peng, L.; Ru, M.; Liang, Z.; Yan, X.; Zhu, Y. Establishment of Salvia castanea Diels f. tomentosa Stib. hairy root cultures and the promotion of tanshinone accumulation and gene expression with Ag+, methyl jasmonate, and yeast extract elicitation. Protoplasma 2016, 253, 87–100. [Google Scholar] [CrossRef] [PubMed]
- Bor, M.; Seckin, B.; Ozgur, R.; Yilmaz, Ö.; Ozdemir, F.; Turkan, I. Comparative effects of drought, salt, heavy metal and heat stresses on γ-aminobutryric acid levels of sesame (Sesamum indicum L.). Acta Physiol. Plant. 2009, 31, 655–659. [Google Scholar] [CrossRef]
- Fatima, S.; Mujib, A.; Tonk, D. NaCl amendment improves vinblastine and vincristine synthesis in Catharanthus roseus: A case of stress signalling as evidenced by antioxidant enzymes activities. Plant Cell Tissue Organ Cult. (PCTOC) 2015, 121, 445–458. [Google Scholar] [CrossRef]
- Gupta, P.; Sharma, S.; Saxena, S. Biomass Yield and Steviol Glycoside Production in Callus and Suspension Culture of Stevia rebaudiana Treated with Proline and Polyethylene Glycol. Appl. Biochem. Biotechnol. 2015, 176, 863–874. [Google Scholar] [CrossRef] [PubMed]
- Naik, P.M.; Al-Khayri, J.M. Abiotic and Biotic Elicitors–Role in Secondary Metabolites Production through In Vitro Culture of Medicinal Plants. In Abiotic and Biotic Stress in Plants-Recent Advances and Future Perspectives; InTech: London, UK, 2016; Volume 10, pp. 247–277. [Google Scholar]
- Häkkinen, S.T.; Nygren, H.; Nohynek, L.; Puupponen-Pimiä, R.; Heiniö, R.-L.; Maiorova, N.; Rischer, H.; Ritala, A. Plant cell cultures as food—aspects of sustainability and safety. Plant Cell Rep. 2020, 39, 1655–1668. [Google Scholar] [CrossRef] [PubMed]
- Shanmugaraj, B.; Bulaon, C.J.I.; Phoolcharoen, W. Plant Molecular Farming: A Viable Platform for Recombinant Biopharmaceutical Production. Plants 2020, 9, 842. [Google Scholar] [CrossRef]
- Capell, T.; Twyman, R.M.; Armario-Najera, V.; Ma, J.K.-C.; Schillberg, S.; Christou, P. Potential Applications of Plant Biotechnology against SARS-CoV-2. Trends Plant Sci. 2020, 25, 635–643. [Google Scholar] [CrossRef]
- Clark, M.; Maselko, M. Transgene Biocontainment Strategies for Molecular Farming. Front. Plant Sci. 2020, 11, 210. [Google Scholar] [CrossRef] [PubMed]
- Fischer, R.; Buyel, J. Molecular farming—The slope of enlightenment. Biotechnol. Adv. 2020, 40, 107519. [Google Scholar] [CrossRef] [PubMed]
- Qiu, X.; Wong, G.; Audet, J.; Bello, A.; Fernando, L.; Alimonti, J.B.; Fausther-Bovendo, H.; Wei, H.; Aviles, J.; Hiatt, E.; et al. Reversion of advanced Ebola virus disease in nonhuman primates with ZMapp. Nature 2014, 514, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Qiang, W.; Gao, T.; Lan, X.; Guo, J.; Noman, M.; Li, Y.; Guo, Y.; Kong, J.; Li, H.; Du, L.; et al. Molecular Pharming of the Recombinant Protein hEGF-hEGF Concatenated with Oleosin Using Transgenic Arabidopsis. Genes 2020, 11, 959. [Google Scholar] [CrossRef] [PubMed]
- Arya, S.S.; Rookes, J.E.; Cahill, D.M.; Lenka, S.K. Next-generation metabolic engineering approaches towards development of plant cell suspension cultures as specialized metabolite producing biofactories. Biotechnol. Adv. 2020, 45, 107635. [Google Scholar] [CrossRef] [PubMed]
- Menary, J.; Hobbs, M.; De Albuquerque, S.M.; Pacho, A.; Drake, P.M.W.; Prendiville, A.; Ma, J.K.-C.; Fuller, S.S. Shotguns vs. Lasers: Identifying barriers and facilitators to scaling-up plant molecular farming for high-value health products. PLoS ONE 2020, 15, e0229952. [Google Scholar] [CrossRef] [PubMed]
- Adli, M. The CRISPR tool kit for genome editing and beyond. Nat. Commun. 2018, 9, 1–13. [Google Scholar] [CrossRef] [PubMed]
- D’Amelia, V.; Villano, C.; Aversano, R. Emerging Genetic Technologies to Improve Crop Productivity. In Encyclopedia of Food Security and Sustainability; Elsevier: Amsterdam, The Netherlands, 2019; pp. 152–158. [Google Scholar]
- Sander, J.D.; Joung, J.K. CRISPR-Cas systems for editing, regulating and targeting genomes. Nat. Biotechnol. 2014, 32, 347–355. [Google Scholar] [CrossRef]
- Guo, L.; Winzer, T.; Yang, X.; Li, Y.; Ning, Z.; He, Z.; Teodor, R.; Lu, Y.; Bowser, T.A.; Graham, I.A.; et al. The opium poppy genome and morphinan production. Science 2018, 362, 343–347. [Google Scholar] [CrossRef]
- Singh, A.; Menéndez-Perdomo, I.M.; Facchini, P.J. Benzylisoquinoline alkaloid biosynthesis in opium poppy: An update. Phytochem. Rev. 2019, 18, 1457–1482. [Google Scholar] [CrossRef]
- Hunziker, J.; Nishida, K.; Kondo, A.; Kishimoto, S.; Ariizumi, T.; Ezura, H. Multiple gene substitution by Target-AID base-editing technology in tomato. Sci. Rep. 2020, 10, 1–12. [Google Scholar] [CrossRef]
- Li, R.; Li, R.; Li, X.; Fu, D.; Zhu, B.; Tian, H.; Luo, Y.; Zhu, H. Multiplexed CRISPR/Cas9-mediated metabolic engineering of γ-aminobutyric acid levels in Solanum lycopersicum. Plant Biotechnol. J. 2018, 16, 415–427. [Google Scholar] [CrossRef] [PubMed]
- Etakayama, M.; Eezura, H. How and why does tomato accumulate a large amount of GABA in the fruit? Front. Plant Sci. 2015, 6, 612. [Google Scholar] [CrossRef]
- Bachtiar, V.; Near, J.; Johansen-Berg, H.; Stagg, C.J. Modulation of GABA and resting state functional connectivity by transcranial direct current stimulation. eLife 2015, 4, e08789. [Google Scholar] [CrossRef] [PubMed]
- Cravens, A.; Payne, J.; Smolke, C.D. Synthetic biology strategies for microbial biosynthesis of plant natural products. Nat. Commun. 2019, 10, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Gomes, A.M.V.; Carmo, T.S.; Carvalho, L.S.; Bahia, F.M.; Parachin, N.S. Comparison of Yeasts as Hosts for Recombinant Protein Production. Microorganisms 2018, 6, 38. [Google Scholar] [CrossRef]
- Ajikumar, P.K.; Xiao, W.-H.; Tyo, K.E.J.; Wang, Y.; Simeon, F.; Leonard, E.; Mucha, O.; Phon, T.H.; Pfeifer, B.; Stephanopoulos, G. Isoprenoid Pathway Optimization for Taxol Precursor Overproduction in Escherichia coli. Science 2010, 330, 70–74. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, G. Strategies of Isoprenoids Production in Engineered Bacteria. J. Appl. Microbiol. 2016, 121, 932–940. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, P.; Smolke, C.D. Biosynthesis of medicinal tropane alkaloids in yeast. Nat. Cell Biol. 2020, 585, 1–6. [Google Scholar] [CrossRef]
- Bhatia, S.K.; Bhatia, R.K.; Choi, Y.-K.; Kan, E.; Kim, Y.-G.; Yang, Y.-H. Biotechnological potential of microbial consortia and future perspectives. Crit. Rev. Biotechnol. 2018, 38, 1209–1229. [Google Scholar] [CrossRef]
- Chae, T.U.; Choi, S.Y.; Kim, J.W.; Ko, Y.-S.; Lee, S.Y. Recent advances in systems metabolic engineering tools and strategies. Curr. Opin. Biotechnol. 2017, 47, 67–82. [Google Scholar] [CrossRef] [PubMed]
- Romanowski, S.; Eustáquio, A.S. Synthetic biology for natural product drug production and engineering. Curr. Opin. Chem. Biol. 2020, 58, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Yang, S.; Zhang, L.; Zhou, Y.J. Advanced Strategies for Production of Natural Products in Yeast. iScience 2020, 23, 100879. [Google Scholar] [CrossRef] [PubMed]
- Lennen, R.M.; Pfleger, B.F. Microbial production of fatty acid-derived fuels and chemicals. Curr. Opin. Biotechnol. 2013, 24, 1044–1053. [Google Scholar] [CrossRef] [PubMed]
- Birchfield, A.S.; McIntosh, C.A. Metabolic engineering and synthetic biology of plant natural products—A minireview. Curr. Plant Biol. 2020, 24, 100163. [Google Scholar] [CrossRef]


| Patent Number | Molecule Targeted | Methodology Used or Proposed | Inventors | Assignee/Applicant |
|---|---|---|---|---|
| US7754943B2 | Several plant molecules | Method for culturing in bioreactor C4 transgenic grasses | Brumbley et al., 2010 [39] | BSES Limited, Indooroopilly (AU) |
| US8338143B2 | Taxol | Elicitors to enhance production in cell culture of taxus species | Bringi et al., 2012 [40] | Phyton Holdings, LLC, SanAntonio, TX (US) |
| US8586722B2 | Starch/sugars | Antisense silencing technology to reduce amount of reducing sugars in tubers | Kossmann et al., 2013 [41] | Bayer Cropscience AG, Monheim (DE) |
| US8802925B2 | Flavonoids | Recombinant DNA technology in Solanaceae | Luo et al., 2014 [42] | Norfolk Plant Sciences Limited, Norfolk (GB) |
| US10533182B2 | Polyunsaturated fatty acids | Recombinant DNA technology in Brassica | Cirpus et al., 2019 [43] | BASF Plant Science GmbH, Ludwigshafen (DE) |
| US10743567B2 | Steviol glycosides | Engineered and synthetic biology technology for production in host cells | Philippe et al., 2020 [44] | MANUS BIO INC., Cambridge, MA (US) |
| WO2020222233A1 | Cannabinoids | Plant cell culture | Yanay and Raviv, 2020 [45] | Pluristem LTD., Haifa (IL) |
| US9796980B2 | Isoprenoid pathway | Synthetic biology technology for production in E. coli | Ajikumar et al., 2017 [46] | Massachusetts Institute of Technology Cambridge, Cambridge, MA (US); National University of Singapore, Singapore (SG) |
| US10638679B2 | Several plant-derived compounds | Cell pack cultivation | Rademacher, 2020 [47] | Fraunhofer-Gesellschaftzur Förderungderangewandten Forschung e.V., München (DE) |
| US8158857B2 | Human protein | Human protein | Huang et al., 2012 [48] | Ventria Bioscience, Sacramento, CA (US) |
| Secondary Metabolites | Species | Industrial Manufacturer | Use/Notes |
|---|---|---|---|
| Anthocyanins | Euphorbia milii, Aralia cordata | Nippon Paint Co., Ltd. (Japan) | Textile dyes, colouring agents for beverages |
| Arbutin | Catharanthus roseus | Mitsui Chemicals, Inc. (Japan) | Pigment, antiseptic |
| Betacyanins | β vulgaris | Nippon Shinyaku Co., Ltd. (Japan) | Food colorant and dye |
| Carthamin | Carthamus tinctorius | Kibun Foods, Inc. (Japan) | Food colorant and dye |
| Geraniol | Geraminea spp. | Mitsui Chemicals, Inc. (Japan) | Essential oil |
| Ginseng | Panax ginseng, Wild ginseng | Nitto Denko Corporation (Japan) | Dietary supplement, cosmetics |
| Sanguinarin | Papaverum sonniferum | Vipont research laboratories (Canada) | Cosmetic anti-inflammatory |
| Glabridin | Glycyrrhiza glabra | Lonza (Switzerland) | Skin brightening |
| Rosmarinic chlorogenic caffeic acid | Symphytum officinale L., Saponaria Pumila | Mibelle AG Biochemistry (Switzerland) | Cosmetics, skin care products |
| Phenols and flavonoids | Gossypium herbaceum L. | Vytrus Biotech (Spain) | Cosmetics, |
| Berberines | Coptis | Mitsui Chemicals, Inc. (Japan) | Anti-cancer, antibiotic, anti-inflammatory |
| Echinacea polysaccharides | Echinacea purpurea, Echinacea angustifolia | Diversa (Germany) | Immunostimulant, anti-inflammatory |
| Paclitaxel | Taxus spp. | Phyton Biotech, Inc. (USA/Germany), Samyang, Genex (Korea) | Anti-cancer |
| Podophyllotoxin | Podophyllum spp. | Nippon Oil (Japan) | Anti-cancer |
| Rosmarinic acid | Coleus blumei | A. Nattermann & Cie. GmbH (Germany) | Anti-inflammatory |
| Scopolamine | Duboisia spp. | Sumitomo Chemical Co., Ltd. (Japan) | Anticholinergic, anti-muscarinic |
| Shikonin | Lithospermum erythrorhizon | Mitsui Chemicals, Inc. (Japan) | Pigment, antibiotic |
| Echinacoside | Echinacea angustifolia | Institute of Biotechnological research (IRB) by Sederma (Italy) | Food supplement ingredient |
| Verbascoside | Lippia citriodora | Active Botanicals Research (ABR) (Italy) | Food supplement ingredient |
| Vanillin | Vanilla planifolia | Esca genetics corporation (USA) | Food ingredients, flavour |
| Bioactive Compounds | Plant | Tissue Type | Elicitors | References |
|---|---|---|---|---|
| Rosmarinic acid, chicoric acid, anthocyanins | Purple basil Ocimum basilicum L. var. purpurascen | Callus cultures | Melatonin; and UV-C irradiation | [91] |
| Glycyrrhizin | Glycyrrhiza glabra | Hairy root cultures | Polyethylene glycol CdCl2, cellulase, mannan | [92] |
| Ephedrine | Ephedra alata | Suspension cultures | Aspergillus niger and yeast extract | [93] |
| Caffeic acid; cynaroside | Vitex agnus castus | Agitated shoot cultures | L-phenylalanine | [94] |
| Lignans | Linum ussitatsimum L. | Cell suspension cultures | Salicylic acid (SA) | [95] |
| Aloe-emodin | Cassia tora | Root cultures | Chitosan; yeast extract | [96] |
| Thymol; p-cymene | Carum copticum L. | Callus cultures | Salt stress and chitosan | [97] |
| Paclitaxel | Taxus canadensis | Plant | Chitosan-derived oligosaccharides | [98] |
| Tanshinone | Salvia miltiorrhiza, S. officinalis, S. castanea | Plant | MeJA | [99,100] |
| GABA | Sesamum indicum | Plant | Salt stress | [101] |
| Vinblastine, vincristine | Catharanthus roseus | Plant | Salt stress | [102] |
| Steviol glycosides | Stevia rebaudiana | Plant | PEG | [103] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
D’Amelia, V.; Docimo, T.; Crocoll, C.; Rigano, M.M. Specialized Metabolites and Valuable Molecules in Crop and Medicinal Plants: The Evolution of Their Use and Strategies for Their Production. Genes 2021, 12, 936. https://doi.org/10.3390/genes12060936
D’Amelia V, Docimo T, Crocoll C, Rigano MM. Specialized Metabolites and Valuable Molecules in Crop and Medicinal Plants: The Evolution of Their Use and Strategies for Their Production. Genes. 2021; 12(6):936. https://doi.org/10.3390/genes12060936
Chicago/Turabian StyleD’Amelia, Vincenzo, Teresa Docimo, Christoph Crocoll, and Maria Manuela Rigano. 2021. "Specialized Metabolites and Valuable Molecules in Crop and Medicinal Plants: The Evolution of Their Use and Strategies for Their Production" Genes 12, no. 6: 936. https://doi.org/10.3390/genes12060936
APA StyleD’Amelia, V., Docimo, T., Crocoll, C., & Rigano, M. M. (2021). Specialized Metabolites and Valuable Molecules in Crop and Medicinal Plants: The Evolution of Their Use and Strategies for Their Production. Genes, 12(6), 936. https://doi.org/10.3390/genes12060936






