Methylation of Estrogen Receptor 1 Gene in the Paraspinal Muscles of Girls with Idiopathic Scoliosis and Its Association with Disease Severity
Abstract
1. Introduction
2. Results
2.1. Patient Characteristics
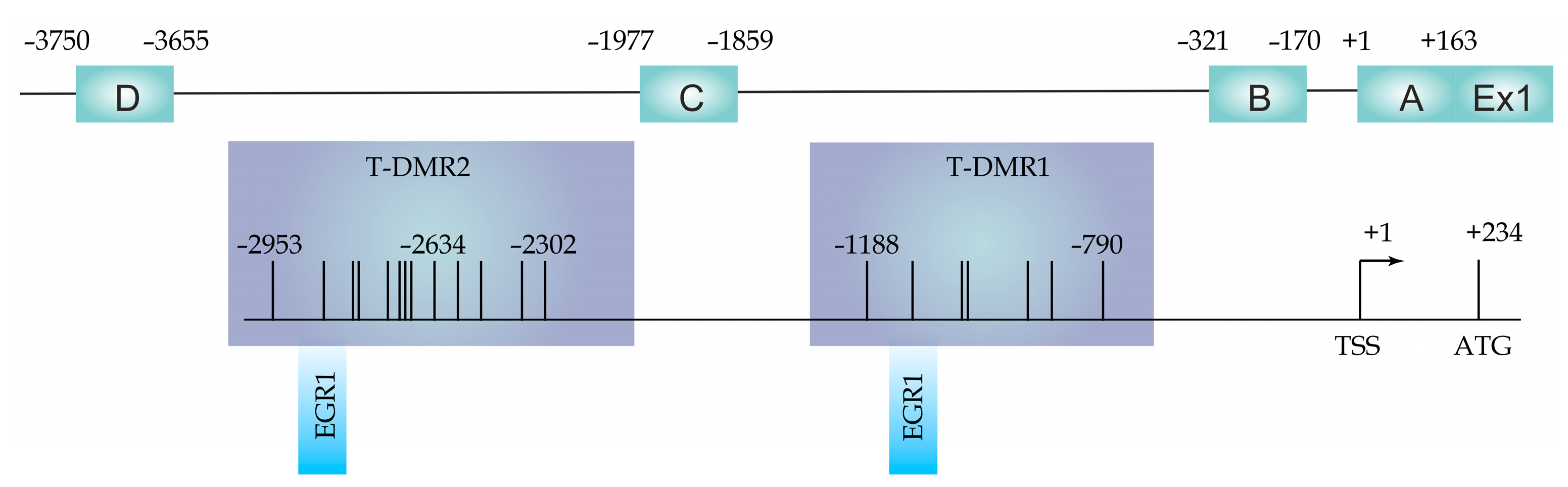
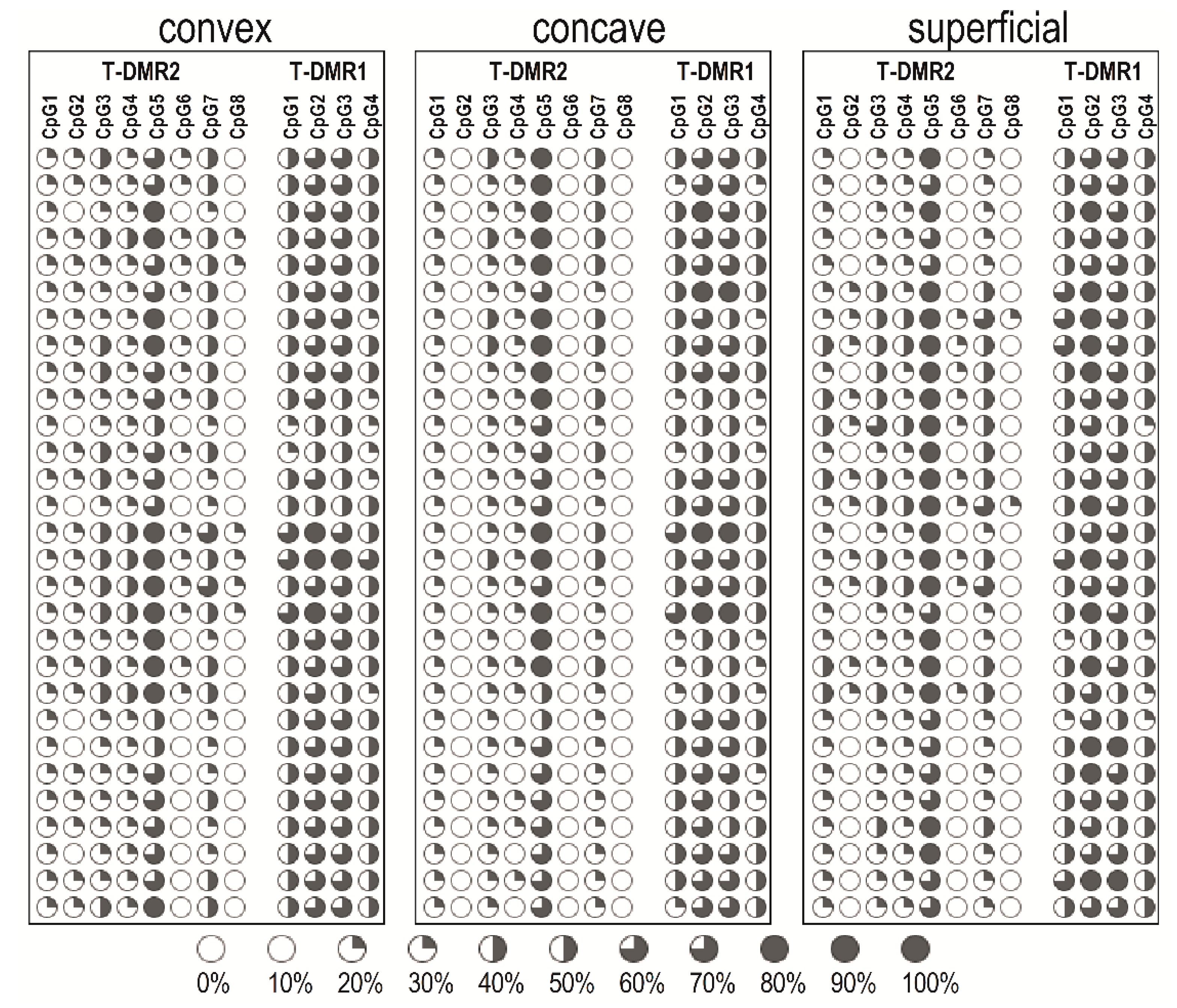
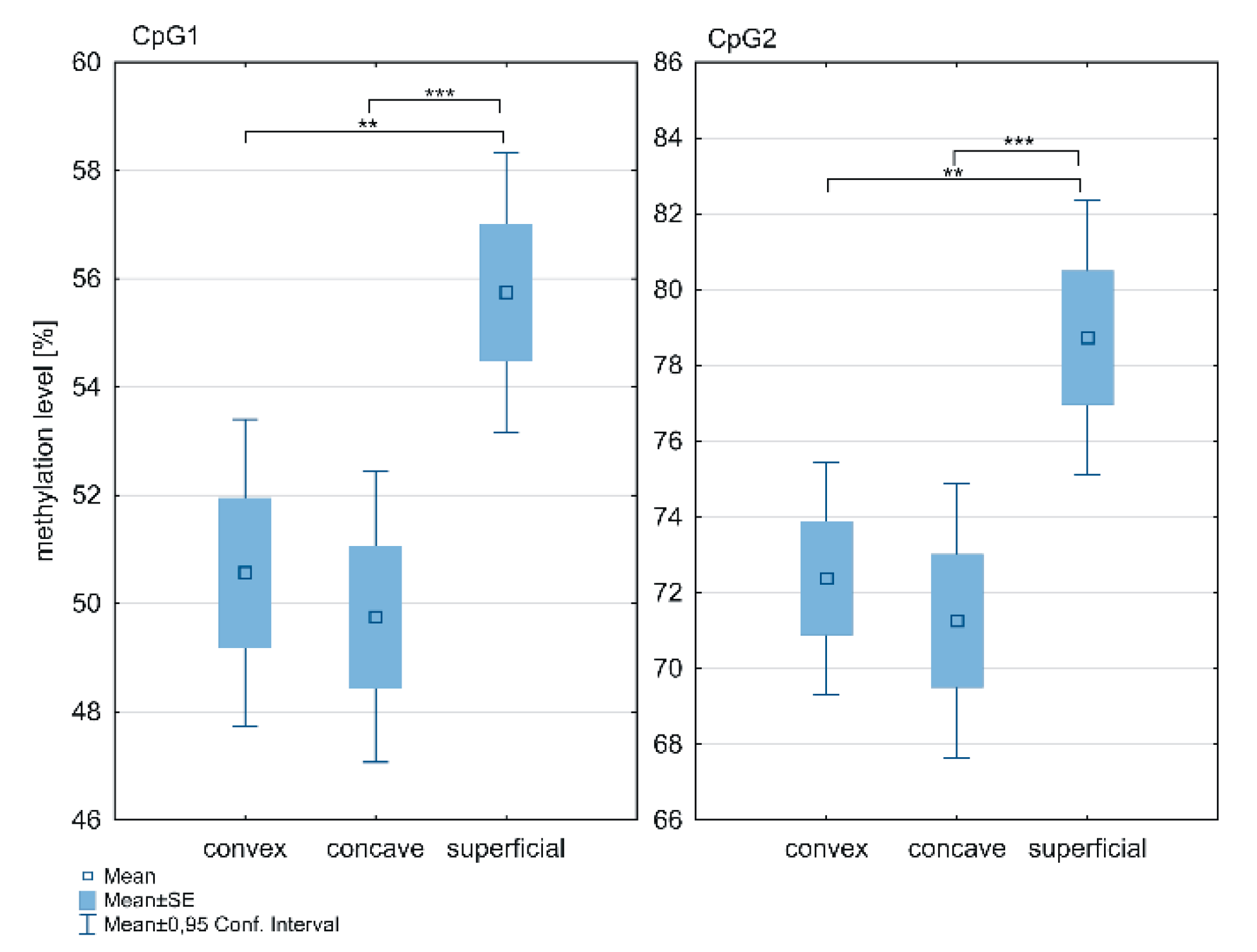
2.2. DNA Methylation at the ESR1 T-DMR1 and T-DMR2
2.3. Correlation between ESR1 Methylation Levels and Relative Expression of the ESR1 Gene
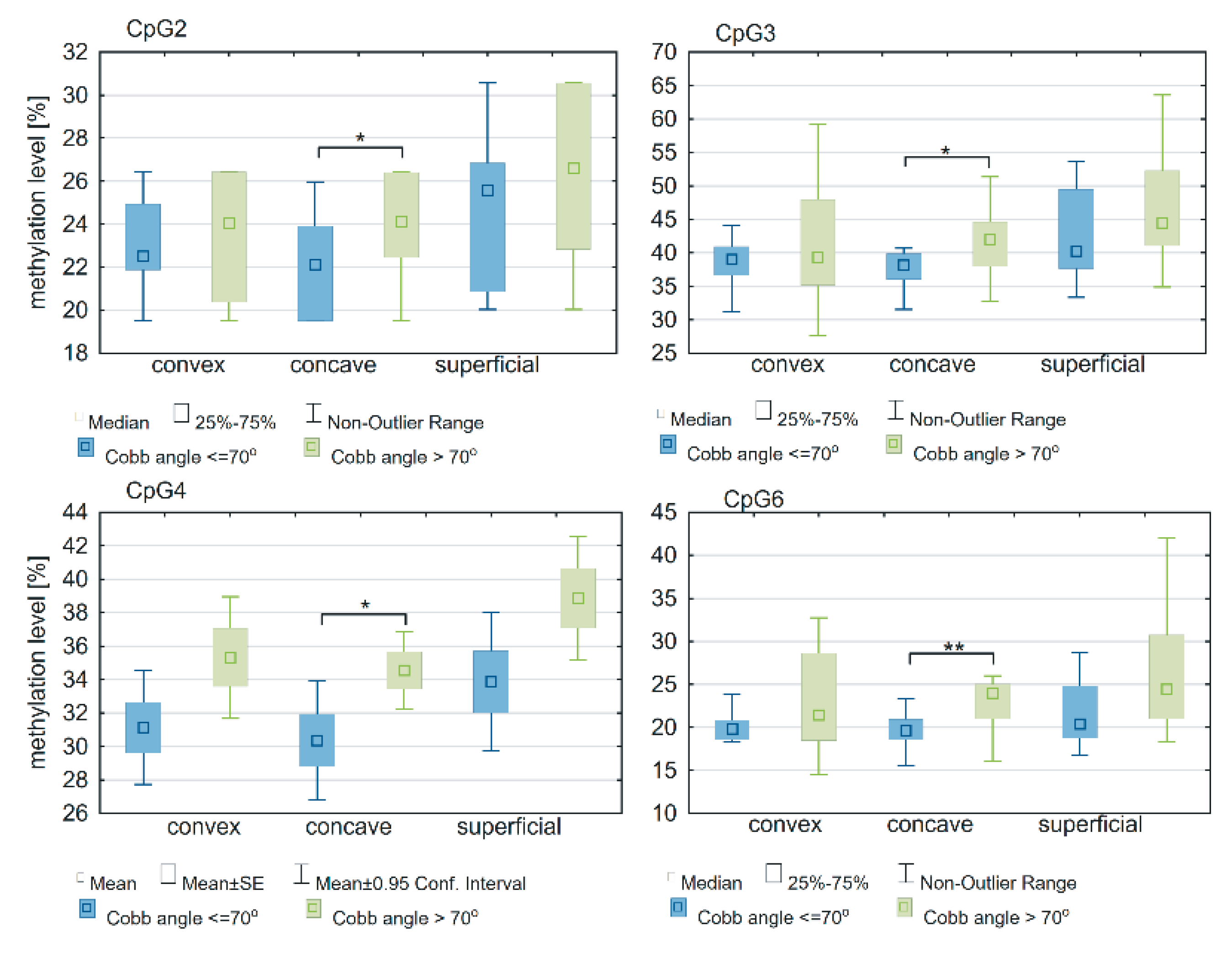
2.4. Association between Methylation Status of ESR1 and Cobb Angle
3. Discussion
4. Materials and Methods
4.1. Patient Population
4.2. Tissue Samples
4.3. Genomic DNA Methylation Analysis
4.3.1. Genomic DNA Isolation and Bisulfite Conversion
4.3.2. Polymerase Chain Reaction and Pyrosequencing Analysis
4.4. Analysis of ESR1 mRNA Expression
4.4.1. Total RNA Isolation and Reverse Transcription
4.4.2. Quantitative Polymerase Chain Reaction
4.5. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Weinstein, S.L.; Dolan, L.A.; Cheng, J.C.Y.; Danielsson, A.; Morcuende, J.A. Adolescent idiopathic scoliosis. Lancet 2008, 371, 1527–1537. [Google Scholar] [CrossRef]
- Newton Ede, M.M.P.; Jones, S.W. Adolescent idiopathic scoliosis: Evidence for intrinsic factors driving aetiology and progression. Int. Orthop. 2016, 40, 2075–2080. [Google Scholar] [CrossRef] [PubMed]
- Weinstein, S.L.; Zavala, D.C.; Ponseti, I.V. Idiopathic scoliosis. Long-term follow-up and prognosis in untreated patients. J. Bone Jt. Surg. Ser. A 1981, 63, 702–712. [Google Scholar] [CrossRef]
- Huh, S.; Eun, L.Y.; Kim, N.K.; Jung, J.W.; Choi, J.Y.; Kim, H.S. Cardiopulmonary function and scoliosis severity in idiopathic scoliosis children. Korean J. Pediatr. 2015, 58, 218–223. [Google Scholar] [CrossRef]
- Nepple, J.J.; Lenke, L.G. Severe idiopathic scoliosis with respiratory insufficiency treated with preoperative traction and staged anteroposterior spinal fusion with a 2-level apical vertebrectomy. Spine J. 2009, 9, e9–e13. [Google Scholar] [CrossRef]
- Noshchenko, A.; Hoffecker, L.; Lindley, E.M.; Burger, E.L.; Cain, C.M.J.; Patel, V.V.; Bradford, A.P. Predictors of spine deformity progression in adolescent idiopathic scoliosis: A systematic review with meta-analysis. World J. Orthop. 2015, 6, 537–558. [Google Scholar] [CrossRef]
- Roye, B.D.; Wright, M.L.; Matsumoto, H.; Yorgova, P.; McCalla, D.; Hyman, J.E.; Roye, D.P.; Shah, S.A.; Vitale, M.G. An Independent Evaluation of the Validity of a DNA-Based Prognostic Test for Adolescent Idiopathic Scoliosis. J. Bone Jt. Surg. Am. 2015, 97, 1994–1998. [Google Scholar] [CrossRef]
- Fadzan, M.; Bettany-Saltikov, J. Etiological Theories of Adolescent Idiopathic Scoliosis: Past and Present. Open Orthop. J. 2017, 11, 1466–1489. [Google Scholar] [CrossRef]
- Lowe, T.G.; Edgar, M.; Margulies, J.Y.; Miller, N.H.; Raso, V.J.; Reinker, K.A.; Rivard, C.H. Etiology of idiopathic scoliosis: Current trends in research. J. Bone Jt. Surg. Ser. A 2000, 82, 1157–1168. [Google Scholar] [CrossRef]
- Liu, G.; Liu, S.; Lin, M.; Li, X.; Chen, W.; Zuo, Y.; Liu, J.; Niu, Y.; Zhao, S.; Long, B.; et al. Genetic polymorphisms of GPR126 are functionally associated with PUMC classifications of adolescent idiopathic scoliosis in a Northern Han population. J. Cell. Mol. Med. 2018, 22, 1964–1971. [Google Scholar] [CrossRef]
- Liu, G.; Liu, S.; Li, X.; Chen, J.; Chen, W.; Zuo, Y.; Liu, J.; Niu, Y.; Lin, M.; Zhao, S.; et al. Genetic polymorphisms of PAX1 are functionally associated with different PUMC types of adolescent idiopathic scoliosis in a northern Chinese Han population. Gene 2019, 688, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhang, Y.; Zhao, S.; Dong, X.; Li, X.; You, Y.; Yan, Z.; Liu, G.; Tong, B.; Chen, Y.; et al. Estrogen receptors (ESRs) mutations in adolescent idiopathic scoliosis: A cross-sectional study. Med. Sci. Monit. 2020, 26, e921611-1–e921611-7. [Google Scholar] [PubMed]
- Cheng, J.C.; Castelein, R.M.; Chu, W.C.; Danielsson, A.J.; Dobbs, M.B.; Grivas, T.B.; Gurnett, C.A.; Luk, K.D.; Moreau, A.; Newton, P.O.; et al. Adolescent idiopathic scoliosis. Nat. Rev. Dis. Prim. 2015, 1, 15030. [Google Scholar] [CrossRef] [PubMed]
- Burwell, R.G.; Dangerfield, P.H.; Moulton, A.; Grivas, T.B.; Meredith, D.; Wägele, B.; Altmaier, E.; Deloukas, P.; Erdmann, J.; Grundberg, E.; et al. Adolescent idiopathic scoliosis (AIS), environment, exposome and epigenetics: A molecular perspective of postnatal normal spinal growth and the etiopathogenesis of AIS with consideration of a network approach and possible implications for medical therapy. Scoliosis 2011, 6, 26. [Google Scholar] [CrossRef]
- Liu, G.; Wang, L.; Wang, X.; Yan, Z.; Yang, X.; Lin, M.; Liu, S.; Zuo, Y.; Niu, Y.; Zhao, S.; et al. Whole-Genome Methylation Analysis of Phenotype Discordant Monozygotic Twins Reveals Novel Epigenetic Perturbation Contributing to the Pathogenesis of Adolescent Idiopathic Scoliosis. Front. Bioeng. Biotechnol. 2019, 7, 364. [Google Scholar] [CrossRef] [PubMed]
- Mao, S.-H.; Qian, B.-P.; Shi, B.; Zhu, Z.-Z.; Qiu, Y. Quantitative evaluation of the relationship between COMP promoter methylation and the susceptibility and curve progression of adolescent idiopathic scoliosis. Eur. Spine J. 2018, 27, 272–277. [Google Scholar] [CrossRef] [PubMed]
- Shi, B.; Xu, L.; Mao, S.; Xu, L.; Liu, Z.; Sun, X.; Zhu, Z.; Qiu, Y. Abnormal PITX1 gene methylation in adolescent idiopathic scoliosis: A pilot study. BMC Musculoskelet. Disord. 2018, 19, 138. [Google Scholar] [CrossRef]
- Shi, B.; Mao, S.; Xu, L.; Li, Y.; Sun, X.; Liu, Z.; Zhu, Z.; Qiu, Y. Quantitation Analysis of PCDH10 Methylation In Adolescent Idiopathic Scoliosis Using Pyrosequencing Study. Spine 2020, 45, E373–E378. [Google Scholar] [CrossRef]
- Meng, Y.; Lin, T.; Liang, S.; Gao, R.; Jiang, H.; Shao, W.; Yang, F.; Zhou, X. Value of DNA methylation in predicting curve progression in patients with adolescent idiopathic scoliosis. EBioMedicine 2018, 36, 489–496. [Google Scholar] [CrossRef]
- Konieczny, M.R.; Senyurt, H.; Krauspe, R. Epidemiology of adolescent idiopathic scoliosis. J. Child. Orthop. 2013, 7, 3–9. [Google Scholar] [CrossRef]
- Dayer, R.; Haumont, T.; Belaieff, W.; Lascombes, P. Idiopathic scoliosis: Etiological concepts and hypotheses. J. Child. Orthop. 2013, 7, 11–16. [Google Scholar] [CrossRef]
- Lemoine, S.; Granier, P.; Tiffoche, C.; Rannou-Bekono, F.; Thieulant, M.-L.; Delamarche, P. Estrogen Receptor Alpha mRNA in Human Skeletal Muscles. Med. Sci. Sports Exerc. 2003, 35, 439–443. [Google Scholar] [CrossRef] [PubMed]
- Wiik, A.; Glenmark, B.; Ekman, M.; Esbjornsson-Liljedahl, M.; Johansson, O.; Bodin, K.; Enmark, E.; Jansson, E. Oestrogen receptor beta is expressed in adult human skeletal muscle both at the mRNA and protein level. Acta Physiol. Scand. 2003, 179, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Wiik, A.; Ekman, M.; Johansson, O.; Jansson, E.; Esbjörnsson, M. Expression of both oestrogen receptor alpha and beta in human skeletal muscle tissue. Histochem. Cell Biol. 2009, 131, 181–189. [Google Scholar] [CrossRef]
- Skibinska, I.; Tomaszewski, M.; Andrusiewicz, M.; Urbaniak, P.; Czarnecka-Klos, R.; Shadi, M.; Kotwicki, T.; Kotwicka, M. Expression of Estrogen Receptor Coactivator Proline-, Glutamic Acid- and Leucine-Rich Protein 1 within Paraspinal Muscles in Adolescents with Idiopathic Scoliosis. PLoS ONE 2016, 11, e0152286. [Google Scholar] [CrossRef]
- Lee, C.-J.; Evans, J.; Kim, K.; Chae, H.; Kim, S. Determining the Effect of DNA Methylation on Gene Expression in Cancer Cells. Methods Mol. Biol. 2014, 1101, 161–178. [Google Scholar] [CrossRef]
- Chen, Z.; Li, S.; Subramaniam, S.; Shyy, J.Y.-J.; Chien, S. Epigenetic Regulation: A New Frontier for Biomedical Engineers. Annu. Rev. Biomed. Eng. 2017, 19, 195–219. [Google Scholar] [CrossRef]
- Miller, J.L.; Grant, P.A. The role of DNA methylation and histone modifications in transcriptional regulation in humans. Subcell. Biochem. 2013, 61, 289–317. [Google Scholar] [CrossRef]
- Rakyan, V.K.; Down, T.A.; Thorne, N.P.; Flicek, P.; Kulesha, E.; Graf, S.; Tomazou, E.M.; Backdahl, L.; Johnson, N.; Herberth, M.; et al. An integrated resource for genome-wide identification and analysis of human tissue-specific differentially methylated regions (tDMRs). Genome Res. 2008, 18, 1518–1529. [Google Scholar] [CrossRef]
- Maunakea, A.K.; Nagarajan, R.P.; Bilenky, M.; Ballinger, T.J.; D’Souza, C.; Fouse, S.D.; Johnson, B.E.; Hong, C.; Nielsen, C.; Zhao, Y.; et al. Conserved Role of Intragenic DNA Methylation in Regulating Alternative Promoters. Nature 2010, 466, 253–257. [Google Scholar] [CrossRef]
- Illingworth, R.S.; Gruenewald-Schneider, U.; Webb, S.; Kerr, A.R.W.; James, K.D.; Turner, D.J.; Smith, C.; Harrison, D.J.; Andrews, R.; Bird, A.P. Orphan CpG islands identify numerous conserved promoters in the mammalian genome. PLoS Genet. 2010, 6, e1001134. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.-J.; Maekawa, M.; Horii, T.; Morita, M. The multiple promoter methylation profile of PR gene and ERα gene in tumor cell lines. Life Sci. 2003, 73, 1963–1972. [Google Scholar] [CrossRef]
- Sogon, T.; Masamura, S.; Hayashi, S.; Santen, R.J.; Nakachi, K.; Eguchi, H. Demethylation of promoter C region of estrogen receptor α gene is correlated with its enhanced expression in estrogen-ablation resistant MCF-7 cells. J. Steroid Biochem. Mol. Biol. 2007, 105, 106–114. [Google Scholar] [CrossRef] [PubMed]
- Maekawa, R.; Sato, S.; Okada, M.; Lee, L.; Tamura, I.; Jozaki, K.; Kajimura, T.; Asada, H.; Yamagata, Y.; Tamura, H.; et al. Tissue-Specific Expression of Estrogen Receptor 1 Is Regulated by DNA Methylation in a T-DMR. Mol. Endocrinol. 2016, 30, 335–347. [Google Scholar] [CrossRef]
- Weinstein, S.L. The Natural History of Adolescent Idiopathic Scoliosis. J. Pediatr. Orthop. 2019, 39, S44–S46. [Google Scholar] [CrossRef]
- Grauers, A.; Einarsdottir, E.; Gerdhem, P. Genetics and pathogenesis of idiopathic scoliosis. Scoliosis Spinal Disord. 2016, 11, 45. [Google Scholar] [CrossRef]
- Zhao, L.; Roffey, D.M.; Chen, S. Genetics of adolescent idiopathic scoliosis in the post-genome-wide association study era. Ann. Transl. Med. 2015, 3, S35. [Google Scholar] [CrossRef]
- Gorman, K.F.; Julien, C.; Moreau, A. The genetic epidemiology of idiopathic scoliosis. Eur. Spine J. 2012, 21, 1905–1919. [Google Scholar] [CrossRef]
- Grauers, A.; Rahman, I.; Gerdhem, P. Heritability of scoliosis. Eur. Spine J. 2012, 21, 1069–1074. [Google Scholar] [CrossRef]
- Wajchenberg, M.; Martins, D.E.; De Paiva Luciano, R.; Puertas, E.B.; Del Curto, D.; Schmidt, B.; De Souza Oliveira, A.B.; Faloppa, F. Histochemical analysis of paraspinal rotator muscles from patients with adolescent idiopathic scoliosis. Medicine 2015, 94, e598. [Google Scholar] [CrossRef] [PubMed]
- Stetkarova, I.; Zamecnik, J.; Bocek, V.; Vasko, P.; Brabec, K.; Krbec, M. Electrophysiological and histological changes of paraspinal muscles in adolescent idiopathic scoliosis. Eur. Spine J. 2016, 25, 3146–3153. [Google Scholar] [CrossRef]
- Gibson, J.N.A.; McMaster, M.J.; Scrimgeour, C.M.; Stoward, P.J.; Rennie, M.J. Rates of muscle protein synthesis in paraspinal muscles: Lateral disparity in children with idiopathic scoliosis. Clin. Sci. 1988, 75, 79–83. [Google Scholar] [CrossRef] [PubMed]
- Esposito, T.; Uccello, R.; Caliendo, R.; Di Martino, G.F.; Gironi Carnevale, U.A.; Cuomo, S.; Ronca, D.; Varriale, B. Estrogen receptor polymorphism, estrogen content and idiopathic scoliosis in human: A possible genetic linkage. J. Steroid Biochem. Mol. Biol. 2009, 116, 56–60. [Google Scholar] [CrossRef] [PubMed]
- Kulis, A.; Goździalska, A.; Drąg, J.; Jaśkiewicz, J.; Knapik-Czajka, M.; Lipik, E.; Zarzycki, D. Participation of sex hormones in multifactorial pathogenesis of adolescent idiopathic scoliosis. Int. Orthop. 2015, 39, 1227–1236. [Google Scholar] [CrossRef]
- Janusz, P.; Kotwicki, T.; Andrusiewicz, M.; Kotwicka, M. XbaI and PvuII Polymorphisms of Estrogen Receptor 1 Gene in Females with Idiopathic Scoliosis: No Association with Occurrence or Clinical Form. PLoS ONE 2013, 8, e76806. [Google Scholar] [CrossRef]
- Takahashi, Y.; Matsumoto, M.; Karasugi, T.; Watanabe, K.; Chiba, K.; Kawakami, N.; Tsuji, T.; Uno, K.; Suzuki, T.; Ito, M.; et al. Replication study of the association between adolescent idiopathic scoliosis and two estrogen receptor genes. J. Orthop. Res. 2010, 29, 834–837. [Google Scholar] [CrossRef]
- Sobhan, M.R.; Mahdinezhad-Yazdi, M.; Dastgheib, S.A.; Jafari, M.; Raee-Ezzabadi, A.; Neamatzadeh, H. Association of ESRα XbaI A > G, ESRα PvuII T > C and ESRβ AlwNI T > C Polymorphisms with the Risk of Developing Adolescent Idiopathic Scoliosis: A Systematic Review and Genetic Meta-analysis. Rev. Bras. Ortop. 2020, 55, 8–16. [Google Scholar] [CrossRef]
- Slieker, R.C.; Bos, S.D.; Goeman, J.J.; Bovée, J.V.; Talens, R.P.; Van Der Breggen, R.; Suchiman, H.E.D.; Lameijer, E.W.; Putter, H.; Van Den Akker, E.B.; et al. Identification and systematic annotation of tissue-specific differentially methylated regions using the Illumina 450k array. Epigenet. Chromatin 2013, 6, 26. [Google Scholar] [CrossRef] [PubMed]
- Hogg, K.; Price, E.M.; Robinson, W.P. Improved reporting of DNA methylation data derived from studies of the human placenta. Epigenetics 2014, 9, 333–337. [Google Scholar] [CrossRef]
- Huse, S.M.; Gruppuso, P.A.; Boekelheide, K.; Sanders, J.A. Patterns of gene expression and DNA methylation in human fetal and adult liver. BMC Genom. 2015, 16, 981. [Google Scholar] [CrossRef]
- Cheung, J.; Veldhuizen, A.G.; Halbertsma, J.P.K.; Maurits, N.M.; Sluiter, W.J.; Cool, J.C.; Van Horn, J.R. The Relation Between Electromyography and Growth Velocity of the Spine in the Evaluation of Curve Progression in Idiopathic Scoliosis. Spine 2004, 29, 1011–1016. [Google Scholar] [CrossRef]
- Jiang, H.; Meng, Y.; Jin, X.; Zhang, C.; Zhao, J.; Wang, C.; Gao, R.; Zhou, X. Volumetric and fatty infiltration imbalance of deep paravertebral muscles in adolescent idiopathic scoliosis. Med. Sci. Monit. 2017, 23, 2089–2095. [Google Scholar] [CrossRef]
- Mannion, A.F.; Meier, M.; Grob, D.; Muntener, M. Paraspinal muscle fibre type alterations associated with scoliosis: An old problem revisited with new evidence. Eur. Spine J. 1998, 7, 289–293. [Google Scholar] [CrossRef]
- Cheung, K.M.C.; Wang, T.; Qiu, G.X.; Luk, K.D.K. Recent advances in the aetiology of adolescent idiopathic scoliosis. Int. Orthop. 2008, 32, 729–734. [Google Scholar] [CrossRef] [PubMed]
- Leboeuf, D.; Letellier, K.; Alos, N.; Edery, P.; Moldovan, F. Do estrogens impact adolescent idiopathic scoliosis? Trends Endocrinol. Metab. 2009, 20, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Weinstein, S.L.; Ponseti, I.V. Curve progression in idiopathic scoliosis. J. Bone Jt. Surg. Ser. A 1983, 65, 447–455. [Google Scholar] [CrossRef]
- Asher, M.A.; Burton, D.C. Adolescent idiopathic scoliosis: Natural history and long term treatment effects. Scoliosis 2006, 1, 2. [Google Scholar] [CrossRef]
- Luhmann, S.J.; Lenke, L.G.; Kim, Y.J.; Bridwell, K.H.; Schootman, M. Thoracic adolescent idiopathic scoliosis curves between 70° and 100°: Is anterior release necessary? Spine 2005, 30, 2061–2067. [Google Scholar] [CrossRef]
- Suk, S.I.; Kim, J.H.; Cho, K.J.; Kim, S.S.; Lee, J.J.; Han, Y.T. Is anterior release necessary in severe scoliosis treated by posterior segmental pedicle screw fixation? Eur. Spine J. 2007, 16, 1359–1365. [Google Scholar] [CrossRef] [PubMed]
- Solla, F.; Clement, J.L.; Doria, C.; Bertoncelli, C.; Rosello, O.; Rampal, V. Adolescent idiopathic scoliosis exceeding 70°: A single unit surgical experience. Minerva Ortop. Traumatol. 2018, 69, 69–77. [Google Scholar] [CrossRef]
- Wan, J.; Oliver, V.F.; Wang, G.; Zhu, H.; Zack, D.J.; Merbs, S.L.; Qian, J. Characterization of tissue-specific differential DNA methylation suggests distinct modes of positive and negative gene expression regulation. BMC Genom. 2015, 16, 49. [Google Scholar] [CrossRef]
- Cobb, J.R. Outline for the study of scoliosis. In Instructional Course Lectures; Literary Licensing, LLC: Whitefish, MT, USA, 1948; Volume 5, pp. 261–275. [Google Scholar]
- Risser, J.C.; Brand, R.A. The iliac apophysis: An invaluable sign in the management of scoliosis. Clin. Orthop. Relat. Res. 2010, 468, 646–653. [Google Scholar] [CrossRef]


| Primer | Sequence | Tm (°C) | GC (%) | PCR Product Size | Location with Respect to TSS | Location with Respect to ATG | ||
|---|---|---|---|---|---|---|---|---|
| ESR1 T-DMR1 | → PCR | GGGTGTATGTGAGTGTGTATGTTTAA | 26 | 58.8 | 38.5 | 256 bp | −1107 | −1341 |
| ← PCR B | ATAAAATATAACCTTTTCATACCAAACAT | 29 | 56.8 | 20.7 | −851 | −1085 | ||
| → SEQ | GTATGTGAGTGTGTATGTTTAAT | 23 | 44.7 | 30.4 | - | −1105 | −1337 | |
| ESR1 T-DMR2 | → PCR | GTTTTTATTGGGTGTTATGTGTTTTGG | 27 | 56.8 | 24.1 | 307 bp | −2886 | −3120 |
| ← PCR B | AAACCTTTCCATAAATAACTCAATTAACT | 29 | 56.8 | 20.7 | −2579 | −2813 | ||
| → SEQ | GTTATGTGTTTTGGGAT | 17 | 47.2 | 53.3 | - | −2874 | −3108 |
| Component | Initial Concentration | Volume Added | Final Concentration | Mixture Volume |
|---|---|---|---|---|
| ZymoTaqTMPremix | 2× | 5 µL | 1× | 10 µL |
| →PCR | 10 µM | 1 µL | 1 µM | |
| ←PCR | 10 µM | 1 µL | 1 µM | |
| DNA | 100 ng/µL | 0.2 µL | 2 ng/µL | |
| Nuclease-free water | 2.8 µL | |||
| Thermal profile of the reactions | ||||
| Number of cycles | Step | Duration, temperature | ||
| 1 | Initial denaturation | 10 min, 95 °C | ||
| 37 | Denaturation | 30 s, 95 °C | ||
| Annealing | 30 s, 54 °C | |||
| Extension | 60 s, 72 °C | |||
| 1 | Final extension | 7 min, 72 °C | ||
| 1 | Hold | ∞, 4 °C | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Janusz, P.; Chmielewska, M.; Andrusiewicz, M.; Kotwicka, M.; Kotwicki, T. Methylation of Estrogen Receptor 1 Gene in the Paraspinal Muscles of Girls with Idiopathic Scoliosis and Its Association with Disease Severity. Genes 2021, 12, 790. https://doi.org/10.3390/genes12060790
Janusz P, Chmielewska M, Andrusiewicz M, Kotwicka M, Kotwicki T. Methylation of Estrogen Receptor 1 Gene in the Paraspinal Muscles of Girls with Idiopathic Scoliosis and Its Association with Disease Severity. Genes. 2021; 12(6):790. https://doi.org/10.3390/genes12060790
Chicago/Turabian StyleJanusz, Piotr, Małgorzata Chmielewska, Mirosław Andrusiewicz, Małgorzata Kotwicka, and Tomasz Kotwicki. 2021. "Methylation of Estrogen Receptor 1 Gene in the Paraspinal Muscles of Girls with Idiopathic Scoliosis and Its Association with Disease Severity" Genes 12, no. 6: 790. https://doi.org/10.3390/genes12060790
APA StyleJanusz, P., Chmielewska, M., Andrusiewicz, M., Kotwicka, M., & Kotwicki, T. (2021). Methylation of Estrogen Receptor 1 Gene in the Paraspinal Muscles of Girls with Idiopathic Scoliosis and Its Association with Disease Severity. Genes, 12(6), 790. https://doi.org/10.3390/genes12060790







