Molecular Analysis Reveals a High Diversity of Anopheline Mosquitoes in Yanomami Lands and the Pantanal Region of Brazil
Abstract
1. Introduction
2. Materials and Methods
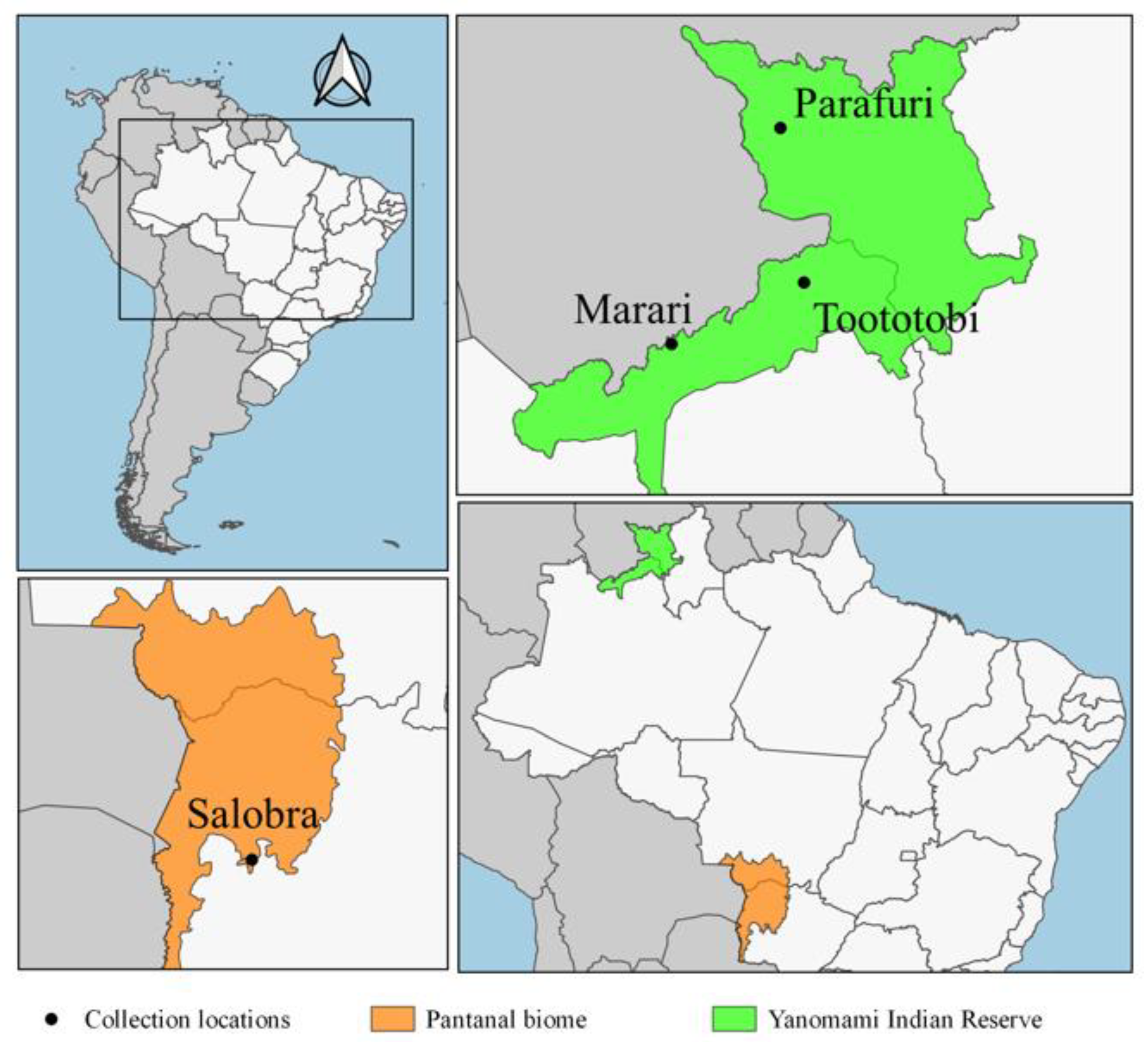
2.1. Study Areas
2.2. Mosquito Collections and Species Identification
2.3. DNA Extraction
2.4. DNA Amplification
2.5. Sequencing and Alignment
2.6. Species Delimitation
3. Results
3.1. Species Identification
3.2. Genus Anopheles
3.3. Genus Nyssorhynchus
3.4. Genus Kerteszia
3.5. Summary of Species Delimitation for Collection Specimens
4. Discussion
4.1. Nyssorhynchus and Kerteszia Genera
4.2. Anopheles Genus
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shretta, R.; Liu, J.; Cotter, C.; Cohen, J.; Dolenz, C.; Makomva, K.; Newby, G.; Ménard, D.; Phillips, A.; Tatarsky, A.; et al. Malaria Elimination and Eradication. In Major Infectious Diseases, 3rd ed.; Holmes, K.K., Bertozzi, S., Bloom, B.R., Jha, P., Eds.; The International Bank for Reconstruction and Development/The World Bank: Washington, DC, USA, 2017; Chapter 12. [Google Scholar]
- World Malaria Report 2021; Licence: CC BY-NC-SA 3.0 IGO; World Health Organization: Geneva, Switzerland, 2021.
- SIVEP-Malaria. Malaria Data; Ministry of Health of Brazil: Brasilia, Brazil, 2021. [Google Scholar]
- Oliveira, T.M.P.; Laporta, G.Z.; Bergo, E.S.; Chaves, L.S.M.; Antunes, J.L.F.; Bickersmith, S.A.; Conn, J.E.; Massad, E.; Sallum, M.A.M. Vector role and human biting activity of Anophelinae mosquitoes in different landscapes in the Brazilian Amazon. Parasites Vectors 2021, 14, 236. [Google Scholar] [CrossRef] [PubMed]
- Chaves, L.S.M.; Conn, J.E.; López, R.V.M.; Sallum, M.A.M. Abundance of impacted forest patches less than 5 km2 is a key driver of the incidence of malaria in Amazonian Brazil. Sci. Rep. 2018, 8, 7077. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, A.J.; Mordecai, E.A. Amazon deforestation drives malaria transmission, and malaria burden reduces forest clearing. Proc. Natl. Acad. Sci. USA 2019, 116, 22212–22218. [Google Scholar] [CrossRef] [PubMed]
- Souza, P.F.; Xavier, D.R.; Suarez Mutis, M.C.; da Mota, J.C.; Peiter, P.C.; de Matos, V.P.; Magalhães, M.A.F.M.; Barcellos, C. Spatial spread of malaria and economic frontier expansion in the Brazilian Amazon. PLoS ONE. 2019, 14, e0217615. [Google Scholar] [CrossRef] [PubMed]
- Guimarães, A.E.; Gentile, C.; Alencar, J.; Lopes, C.M.; de Mello, R.P. Ecology of Anopheline (Diptera, Culicidae), malaria vectors around the Serra da Mesa Reservoir; State of Goiás, Brazil. 1-Frequency and climatic factors. Cad. Saude Publica 2004, 20, 291–302. [Google Scholar] [CrossRef]
- Cella, W.; Baia-da-Silva, D.C.; Melo, G.C.; Tadei, W.P.; Sampaio, V.S.; Pimenta, P.; Lacerda, M.V.G.; Monteiro, W.M. Do climate changes alter the distribution and transmission of malaria? Evidence assessment and recommendations for future studies. Rev. Soc. Bras. Med. Trop. 2019, 52, e20190308. [Google Scholar] [CrossRef] [PubMed]
- Consoli, R.A.G.B.; Oliveira, R.L. Principais Mosquitos de Importância Sanitaria No Brasil; SciELO Books; Editora Fiocruz: Rio de Janeiro, Brazil, 1994; 228p, ISBN 85-85676-03-5. Available online: htpp://books.scielo.org (accessed on 4 December 2021).
- Foster, P.G.; de Oliveira, T.M.P.; Bergo, E.S.; Conn, J.E.; Sant’Ana, D.C.; Nagaki, S.S.; Nihei, S.; Lamas, C.E.; González, C.; Moreira, C.C.; et al. Phylogeny of Anophelinae using mitochondrial protein coding genes. R. Soc. Open Sci. 2017, 4, 170758. [Google Scholar] [CrossRef]
- Marrelli, M.; Malafronte, R.; Sallum, M.A.M.; Delsio, N. Kerteszia subgenus of Anopheles associated with the Brazilian Atlantic rainforest: Current knowledge and future challenges. Malar. J. 2007, 6, 127. [Google Scholar] [CrossRef]
- Bourke, B.P.; Conn, J.E.; de Oliveira, T.M.P.; Chaves, L.S.M.; Bergo, E.S.; Laporta, G.Z.; Sallum, M.A.M. Exploring malaria vector diversity on the Amazon Frontier. Malar. J. 2018, 17, 342. [Google Scholar] [CrossRef] [PubMed]
- Pithan, O.A.; Confalonieri, U.E.C.; Morgado, A.F. A situação de saúde dos índios Yanomámi: Diagnóstico a partir da Casa do Índio de Boa Vista, Roraima, 1987–1989. Cad. Saude Publica 1991, 7, 563–580. [Google Scholar] [CrossRef] [PubMed]
- Leandro-Reguillo, P.; Thomson-Luque, R.; Monteiro, W.M.; de Lacerda, M.V. Urban and architectural risk factors for malaria in indigenous Amazonian settlements in Brazil: A typological analysis. Malar. J. 2015, 14, 284. [Google Scholar] [CrossRef]
- Vittor, Y.; Laporta, G.Z.; Sallum, M.A.M.; Walker, R.T. The COVID-19 crisis and Amazonia’s indigenous people: Implications for conservation and global health. World Dev. 2021, 145, 105533. [Google Scholar] [CrossRef]
- Marcano, T.J.; Morgado, A.; Tosta, C.E.; Coura, J.R. Cross-sectional study defines difference in malaria morbidity in two Yanomami communities on Amazonian boundary between Brazil and Venezuela. Mem. Inst. Oswaldo Cruz 2004, 99, 369–376. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Robortella, D.R.; Calvet, A.A.; Amaral, L.C.; Fantin, R.F.; Guimarães, L.F.F.; França Dias, M.H.; Brito, C.F.A.; Sousa, T.N.; Herzog, M.M.; Oliveira-Ferreira, J.; et al. Prospective assessment of malaria infection in a semi-isolated Amazonian indigenous Yanomami community: Transmission heterogeneity and predominance of submicroscopic infection. PLoS ONE. 2020, 15, e0230643. [Google Scholar] [CrossRef]
- De Pina-Costa, A.; Brasil, P.; Di Santi, S.M.; de Araujo, M.P.; Suárez-Mutis, M.C.; Santelli, A.C.; Oliveira-Ferreira, J.; Lourenço-de-Oliveira, R.; Daniel-Ribeiro, C.T. Malaria in Brazil: What happens outside the Amazonian endemic region. Mem. Inst. Oswaldo Cruz 2014, 109, 618–633. [Google Scholar] [CrossRef] [PubMed]
- Bergier, I.; Assine, M.L.; McGlue, M.M.; Alho, C.J.R.; Silva, A.; Guerreiro, R.L.; Carvalho, J.C. Amazon rainforest modulation of water security in the Pantanal wetland. Sci Total Environ. 2018, 619–620, 1116–1125. [Google Scholar] [CrossRef] [PubMed]
- Junk, W.J.; da Cunha, C.N.; Wantzen, K.M.; Petermann, P.; Strüssmann, C.; Marques, M.I.; Adis, J. Biodiversity and its conservation in the Pantanal of Mato Grosso, Brazil. Aquat. Sci. 2006, 68, 278–309. [Google Scholar] [CrossRef]
- Travassos, L. Relatório da terceira excursão à zona da estrada de ferro Noroeste do Brasil realizada em fevereiro e março de 1940: I—Introdução. Mem. Inst. Oswaldo Cruz 1940, 35, 607–713. [Google Scholar] [CrossRef]
- Travassos, L.; Freitas, J.F.T. Relatório da excursão científica realizada na zona da Estrada de Ferro Noroeste do Brasil em julho de 1939. Mem. Inst. Oswaldo Cruz 1940, 35, 525–556. [Google Scholar] [CrossRef]
- Alencar, J.; Lorosa, E.S.; Silva, J.D.S.; Lopes, C.M.; Guimarães, A.E. Observações sobre padrões alimentares de mosquitos (Diptera: Culicidae) no Pantanal Mato-Grossense. Neotrop. Entomol. 2005, 34, 681–687. [Google Scholar] [CrossRef]
- Silva-do-Nascimento, T.F.; Lourenço-de-Oliveira, R. Diverse population dynamics of three Anopheles species belonging to the Triannulatus Complex (Diptera: Culicidae). Mem. Inst. Oswaldo Cruz 2007, 102, 975–982. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pauvolid-Corrêa, A.; Tavares, F.N.; Alencar, J.; Silva, J.d.S.; Murta, M.; Serra-Freire, N.M.; Pellegrin, A.O.; Gil-Santana, H.; Guimarães, A.E.; Silva, E.E. Preliminary investigation of Culicidae species in South Pantanal, Brazil and their potential importance in arbovirus transmission. Rev. Inst. Med. Trop Sao Paulo 2010, 52, 17–24. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Marinho-e-Silva, M.; Sallum, M.A.M.; Rosa-Freitas, M.G.; Lourenço-de-Oliveira, R.; Silva-do-Nascimento, T.F. Anophelines species and the receptivity and vulnerability to malaria transmission in the Pantanal wetlands, Central Brazil. Mem. Inst. Oswaldo Cruz 2018, 113, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Beebe, N.W.; Cooper, R.D. Systematics of malaria vectors with particular reference to the Anopheles punctulatus group. Int. J. Parasitol. 2000, 30, 1–17. [Google Scholar] [CrossRef]
- Motoki, M.T.; Linton, Y.-M.; Ruiz, F.; Flores-Mendoza, C.; Sallum, M.A.M. Redescription of Anopheles oswaldoi (Peryassú, 1922) (Diptera: Culicidae), with formal lectotype designation. Zootaxa 2007, 1588, 31–51. [Google Scholar] [CrossRef]
- Tennessen, J.A.; Ingham, V.A.; Toé, K.H.; Guelbéogo, W.M.; Sagnon, N.; Kuzma, R.; Ranson, H.; Neafsey, D.E. A population genomic unveiling of a new cryptic mosquito taxon within the malaria-transmitting Anopheles gambiae complex. Mol. Ecol. 2021, 30, 775–790. [Google Scholar] [CrossRef]
- Mohammed, B.R.; Yayo, A.M.; Ajanusi, O.J.; Lawal, I.A. Relative abundance and molecular identification of Culex pipiens complex (Diptera: Culicidae), in Kura Local Government Area, North-western Nigeria. Parasite Epidemiol. Control 2021, 14, e00213. [Google Scholar] [CrossRef] [PubMed]
- Abdelwhab, O.F.; Elaagip, A.; Albsheer, M.M.; Ahmed, A.; Paganotti, G.M.; Abdel Hamid, M.M. Molecular and morphological identification of suspected Plasmodium vivax vectors in Central and Eastern Sudan. Malar. J. 2021, 20, 132. [Google Scholar] [CrossRef]
- Tananchai, C.; Manguin, S.; Bangs, M.J.; Chareonviriyaphap, T. Malaria vectors and species complexes in Thailand: Implications for vector control. Trends Parasitol. 2019, 35, 544–558. [Google Scholar] [CrossRef]
- Foster, P.G.; Bergo, E.S.; Bourke, B.P.; Oliveira, T.M.; Nagaki, S.S.; Sant’Ana, D.C.; Sallum, M.A. Phylogenetic analysis and DNA-based species confirmation in Anopheles (Nyssorhynchus). PLoS ONE 2013, 8, e54063. [Google Scholar] [CrossRef]
- Ruiz-Lopez, F.; Wilkerson, R.C.; Ponsonby, D.J.; Herrera, M.; Sallum, M.A.; Velez, I.D.; Quiñones, M.L.; Flores-Mendoza, C.; Chadee, D.D.; Alarcon, J.; et al. Systematics of the Oswaldoi complex (Anopheles, Nyssorhynchus) in South America. Parasites Vectors 2013, 6, 324. [Google Scholar] [CrossRef][Green Version]
- Shahhosseini, N.; Wong, G.; Frederick, C.; Kobinger, G.P. Mosquito species composition and abundance in Quebec; eastern Canada. J. Med. Entomol. 2020, 57, 1025–1031. [Google Scholar] [CrossRef]
- Laurito, M.; Oliveira, T.M.; Almirón, W.R.; Sallum, M.A. COI barcode versus morphological identification of Culex (Culex) (Diptera: Culicidae) species: A case study using samples from Argentina and Brazil. Mem. Inst. Oswaldo Cruz 2013, 108, 110–122. [Google Scholar] [CrossRef] [PubMed]
- Ratnasingham, S.; Hebert, P.D. A DNA-based registry for all animal species: The barcode index number (BIN) system. PLoS ONE 2013, 8, e66213. [Google Scholar] [CrossRef]
- Puillandre, N.; Lambert, A.; Brouillet, S.; Achaz, G. ABGD, Automatic Barcode Gap Discovery for primary species delimitation. Mol. Ecol. 2012, 21, 1864–1877. [Google Scholar] [CrossRef]
- Puillandre, N.; Brouillet, S.; Achaz, G. ASAP: Assemble species by automatic partitioning. Mol. Ecol. Resour. 2021, 21, 609–620. [Google Scholar] [CrossRef]
- Pons, J.; Barraclough, T.G.; Gomez-Zurita, J.; Cardoso, A.; Duran, D.P.; Hazell, S.; Kamoun, S.; Sumlin, W.D.; Vogler, A.P. Sequence-based species delimitation for the DNA taxonomy of undescribed insects. Syst. Biol. 2006, 55, 595–609. [Google Scholar] [CrossRef] [PubMed]
- Fujisawa, T.; Barraclough, T.G. Delimiting Species Using Single-Locus Data and the Generalized Mixed Yule Coalescent Approach: A Revised Method and Evaluation on Simulated Data Sets. Syst. Biol. 2013, 62, 707–724. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Kapli, P.; Pavlidis, P.; Stamatakis, A. A general species delimitation method with applications to phylogenetic placements. Bioinformatics 2013, 29, 2869–2876. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Ribas, J.; Oliveira-Ferreira, J.; Rosa-Freitas, M.G.; Trilla, L.; Silva-do-Nascimento, T.F. New classification of natural breeding habitats for Neotropical anophelines in the Yanomami Indian Reserve; Amazon Region, Brazil and a new larval sampling methodology. Mem. Inst. Oswaldo Cruz 2015, 110, 760–770. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Ribas, J.; Oliveira-Ferreira, J.; Gimnig, J.E.; Pereira-Ribeiro, C.; Santos-Neves, M.S.A.; Silva-do-Nascimento, T.F. Environmental variables associated with anopheline larvae distribution and abundance in Yanomami villages within unaltered areas of the Brazilian Amazon. Parasites Vectors 2017, 10, 571. [Google Scholar] [CrossRef]
- Rosa-Freitas, M.; Tsouris, P.; Peterson, A.; Honorio, N.; Barros, F.; Aguiar, D.; Gurgel, H.; Arruda, M.; Vasconcelos, S.; Moura, J. An ecoregional classification for the state of Roraima, Brazil: The importance of landscape in malaria biology. Mem. Inst. Oswaldo Cruz 2007, 102, 349–358. [Google Scholar] [CrossRef]
- Alho, C.J. Biodiversity of the Pantanal: Response to seasonal flooding regime and to environmental degradation. Braz. J. Biol. 2008, 68, 957–966. [Google Scholar] [CrossRef]
- Guerra, A.; Roque, F.O.; Garcia, L.C.; Ochoa-Quintero, J.M.; Oliveira, P.T.S.; Guariento, R.D.; Rosa, I. Drivers and projections of vegetation loss in the Pantanal and surrounding ecosystems. Land Use Policy 2020, 91, 104388. [Google Scholar] [CrossRef]
- Forattini, O.P. Culicidologia Médica: Identificação, Biologia, Epidemiologia, 1st ed.; USP: São Paulo, Brazil, 2002. [Google Scholar]
- Lamas, C.J.E.; Nihei, S.S.; Foster, P.G.; Conn, J.E.; Sallum, M.A.M. Anopheles darlingi versus Nyssorhynchus darlingi, the discussion continues. Trends Parasitol. 2021, 37, 847–848. [Google Scholar] [CrossRef]
- Edgar, R.C. MUSCLE: A multiple sequence alignment method with reduced time and space complexity. BMC Bioinform. 2004, 5, 113. [Google Scholar] [CrossRef]
- Gouy, M.; Guindon, S.; Gascuel, O. SeaView version 4: A multiplatform graphical user interface for sequence alignment and phylogenetic tree building. Mol. Biol. Evol. 2010, 27, 221–224. [Google Scholar] [CrossRef]
- Abascal, F.; Zardoya, R.; Telford, M.J. TranslatorX: Multiple alignment of nucleotide sequences guided by amino acid translations. Nucleic Acids Res. 2010, 38, W7–W13. [Google Scholar] [CrossRef]
- Kapli, P.; Lutteropp, S.; Zhang, J.; Kobert, K.; Pavlidis, P.; Stamatakis, A.; Flouri, T. Multi-rate Poisson tree processes for single-locus species delimitation under maximum likelihood and Markov chain Monte Carlo. Bioinformatics 2017, 33, 1630–1638. [Google Scholar] [CrossRef]
- Stamatakis, A. RAxML version 8: A tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 2014, 30, 1312–1313. [Google Scholar] [CrossRef]
- Miller, M.A.; Pfeiffer, W.; Schwartz, T. Creating the CIPRES Science Gateway for Inference of Large Phylogenetic Trees. In Proceedings of the Gateway Computing Environments Workshop (GCE), New Orleans, LA, USA, 14 November 2010; pp. 1–8. [Google Scholar] [CrossRef]
- Murillo, E.; Muskus, C.; Agudelo, L.A.; Vélez, I.D.; Ruiz-Lopez, F. A new high-resolution melting analysis for the detection and identification of Plasmodium in human and Anopheles vectors of malaria. Sci. Rep. 2019, 9, 1674. [Google Scholar] [CrossRef]
- Galardo, A.K.; Arruda, M.; D’Almeida Couto, A.A.; Wirtz, R.; Lounibos, L.P.; Zimmerman, R.H. Malaria vector incrimination in three rural riverine villages in the Brazilian Amazon. Am. J. Trop. Med. Hyg. 2007, 76, 461–469. [Google Scholar] [CrossRef]
- Dusfour, I.; Issaly, J.; Carinci, R.; Gaborit, P.; Girod, R. Incrimination of Anopheles (Anopheles) intermedius Peryassú, An. (Nyssorhynchus) nuneztovari Gabaldón, An. (Nys.) oswaldoi Peryassú as natural vectors of Plasmodium falciparum in French Guiana. Mem. Inst. Oswaldo Cruz 2012, 107, 429–432. [Google Scholar] [CrossRef]
- Quiñones, M.L.; Ruiz, F.; Calle, D.A.; Harbach, R.E.; Erazo, H.F.; Linton, Y.-M. Incrimination of Anopheles (Nyssorhynchus) rangeli and An.(Nys.) oswaldoi as natural vectors of Plasmodium vivax in Southern Colombia. Mem. Inst. Oswaldo Cruz 2006, 101, 617–623. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Carvajal, H.; de Herrera, M.A.; Quintero, J.; Alzate, A.; Herrera, S. Anopheles neivai: A vector of malaria in the Pacific lowlands of Colombia. Trans. R. Soc. Trop. Med. Hyg. 1989, 83, 609. [Google Scholar] [CrossRef]
- Tadei, W.P.; Dutary-Thatcher, B. Malaria vectors in the Brazilian Amazon: Anopheles of the subgenus Nyssorhynchus. Rev. Inst. Med. Trop. 2000, 42, 94–97. [Google Scholar] [CrossRef]
- Prussing, C.; Moreno, M.; Saavedra, M.; Bickersmith, S.; Gamboa, D.; Alava, F.; Schlichting, C.; Emerson, K.; Vinetz, J.; Conn, J. Decreasing proportion of Anopheles darlingi biting outdoors between long-lasting insecticidal net distributions in peri-Iquitos, Amazonian Peru. Malar. J. 2018, 17, 86. [Google Scholar] [CrossRef]
- Naranjo-Diaz, N.; Altamiranda-Saavedra, M.; Correa, M.M. Anopheles species composition and entomological parameters in malaria endemic localities of North West Colombia. Acta Trop. 2019, 190, 13–21. [Google Scholar] [CrossRef]
- Altamiranda-Saavedra, M.; Arboleda, S.; Parra, J.L.; Peterson, A.T.; Correa, M.M. Potential distribution of mosquito vector species in a primary malaria endemic region of Colombia. PLoS ONE 2017, 12, e0179093. [Google Scholar] [CrossRef] [PubMed]
- Carlos, B.C.; Rona, L.D.P.; Christophides, G.K.; Souza-Neto, J.A. A comprehensive analysis of malaria transmission in Brazil. Pathog. Glob Health 2019, 113, 1–13. [Google Scholar] [CrossRef]
- Marrelli, M.T.; Floeter-Winter, L.M.; Malafronte, R.S.; Tadei, W.P.; Lourenço-de-Oliveira, R.; Flores-Mendoza, C.; Marinotti, O. Amazonian malaria vector anopheline relationships interpreted from ITS2 rDNA sequences. Med. Vet. Entomol. 2005, 19, 208–218. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, F.; Quiñones, M.L.; Erazo, H.F.; Calle, D.A.; Alzate, J.F.; Linton, Y.M. Molecular differentiation of Anopheles (Nyssorhynchus) benarrochi and An. (N.) oswaldoi from southern Colombia. Mem. Inst. Oswaldo Cruz 2005, 100, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Conn, J. A genetic study of the malaria vector Anopheles nuneztovari from western Venezuela. J. Am. Mosq. Control Assoc. 1990, 6, 400–405. [Google Scholar]
- Conn, J.; Puertas, Y.R.; Seawright, J.A. A new cytotype of Anopheles nunezatovari from western Venezuela and Colombia. J. Am. Mosq. Control Assoc. 1993, 9, 294–301. [Google Scholar] [PubMed]
- Calado, D.C.; Foster, P.G.; Bergo, E.S.; Santos, C.L.; Galardo, A.K.; Sallum, M.A. Resurrection of Anopheles goeldii from synonymy with Anopheles nuneztovari (Diptera, Culicidae) and a new record for Anopheles dunhami in the Brazilian Amazon. Mem. Inst. Oswaldo Cruz 2008, 103, 791–799. [Google Scholar] [CrossRef]
- Branquinho, M.S.; Araújo, M.S.; Natal, D.; Marrelli, M.T.; Rocha, R.M.; Taveira, F.A.; Kloetzel, J.K. Anopheles oswaldoi a potential malaria vector in Acre; Brazil. Trans. R. Soc. Trop. Med. Hyg. 1996, 90, 233. [Google Scholar] [CrossRef]
- Harbach, R. The Culicidae (Diptera): A Review of Taxonomy; Classification and Phylogeny. Zootaxa 2007, 1668, 591–638. [Google Scholar] [CrossRef]
- Torres-Gutierrez, C.; Bergo, E.S.; Emerson, K.J.; de Oliveira, T.M.P.; Greni, S.; Sallum, M.A.M. Mitochondrial COI gene as a tool in the taxonomy of mosquitoes Culex subgenus Melanoconion. Acta Trop. 2016, 164, 137–149. [Google Scholar] [CrossRef]
- Adeniran, A.A.; Hernández-Triana, L.M.; Ortega-Morales, A.I.; Garza-Hernández, J.A.; Cruz-Ramos, J.; Chan-Chable, R.J.; Vázquez-Marroquín, R.; Huerta-Jiménez, H.; Nikolova, N.I.; Fooks, A.R.; et al. Identification of mosquitoes (Diptera: Culicidae) from Mexico State; Mexico using morphology and COI DNA barcoding. Acta Trop. 2021, 213, 105730. [Google Scholar] [CrossRef]
- Sallum, M.A.M.; Conn, J.E.; Bergo, E.S.; Laporta, G.Z.; Chaves, L.S.M.; Bickersmith, S.A.; de Oliveira, T.M.P.; Figueira, E.A.G.; Moresco, G.; Olívêr, L.; et al. Vector competence, vectorial capacity of Nyssorhynchus darlingi and the basic reproduction number of Plasmodium vivax in agricultural settlements in the Amazonian Region of Brazil. Malar. J. 2019, 18, 117. [Google Scholar] [CrossRef]
- Mendes, A.M.; Lima, M.D.S.; Maciel, A.G.P.; Menezes, R.A.O.; Eugênio, N.C.C. Malaria among indigenous peoples on the Brazil-French Guiana border, 2007–2016: A descriptive study. Epidemiol. Serv. Saude 2020, 29, e2019056. [Google Scholar] [CrossRef]
- Meireles, B.M.; de Souza Sampaio, V.; Monteiro, W.M.; Gonçalves, M.J.F. Factors associated with malaria in indigenous populations: A retrospective study from 2007 to 2016. PLoS ONE 2020, 15, e0240741. [Google Scholar] [CrossRef] [PubMed]
- Laporta, G.Z.; Ilacqua, R.C.; Bergo, E.S.; Chaves, L.S.M.; Rodovalho, S.R.; Moresco, G.G.; Figueira, E.A.G.; Massad, E.; de Oliveira, T.M.P.; Bickersmith, S.A.; et al. Malaria transmission in landscapes with varying deforestation levels and timelines in the Amazon: A longitudinal spatiotemporal study. Sci. Rep. 2021, 11, 6477. [Google Scholar] [CrossRef] [PubMed]
- Demari-Silva, B.; Laporta, G.Z.; Oliveira, T.M.P.; Sallum, M.A.M. Plasmodium infection in Kerteszia cruzii (Diptera: Culicidae) in the Atlantic tropical rain forest; southeastern Brazil. Infect. Genet. Evol. 2020, 78, 104061. [Google Scholar] [CrossRef]
- Saraiva, J.F.; Souto, R.N.P.; Scarpassa, V.M. Molecular taxonomy and evolutionary relationships in the Oswaldoi-Konderi complex (Anophelinae: Anopheles: Nyssorhynchus) from the Brazilian Amazon region. PLoS ONE 2018, 5, e0193591. [Google Scholar] [CrossRef]
- Flores-Mendoza, C.; Peyton, E.L.; Wilkerson, R.C.; Lourenço-de-Oliveira, R. Anopheles (Nyssorhynchus) konderi Galvão and Damasceno: Neotype designation and resurrection from synonymy with Anopheles (Nyssorhynchus) oswaldoi (Peryassú) (Diptera: Culicidae). Proc. Entomol. Soc. Wash. 2004, 106, 118–132. [Google Scholar]
- Marrelli, M.T.; Honório, N.A.; Flores-Mendoza, C.; Lourenço-de-Oliveira, R.; Marinotti, O.; Kloetzel, J.K. Comparative susceptibility of two members of the Anopheles oswaldoi complex, An. oswaldoi and An. Konderi, to infection by Plasmodium vivax. Trans. R. Soc. Trop. Med. Hyg. 1999, 93, 381–384. [Google Scholar] [CrossRef]
- Saraiva, J.F.; Scarpassa, V.M. Anopheles (Nyssorhynchus) tadei: A new species of the Oswaldoi-konderi complex (Diptera, Anophelinae) and its morphological and molecular distinctions from An. konderi sensu stricto. Acta Trop. 2021, 221, 106004. [Google Scholar] [CrossRef]
- Marrelli, M.T.; Malafronte, R.S.; Flores-Mendoza, C.; Lourenço-de-Oliveira, R.; Kloetzel, J.K.; Marinotti, O. Sequence analysis of the second internal transcribed spacer of ribosomal DNA in Anopheles oswaldoi (Diptera: Culicidae). J. Med. Entomol. 1999, 36, 679–684. [Google Scholar] [CrossRef]
- Scarpassa, V.M.; Conn, J.E. Molecular differentiation in natural populations of Anopheles oswaldoi sensu lato (Diptera: Culicidae) from the Brazilian Amazon; using sequences of the COI gene from mitochondrial DNA. Genet. Mol. Res. 2006, 5, 493–502. [Google Scholar]
- Sallum, M.A.; Marrelli, M.T.; Nagaki, S.S.; Laporta, G.Z.; Dos Santos, C.L. Insight into Anopheles (Nyssorhynchus) (Diptera: Culicidae) species from Brazil. J. Med. Entomol. 2008, 45, 970–981. [Google Scholar] [CrossRef]
- Dos Santos, M.M.M.; Sucupira, I.M.C.; Dos Santos, T.V.; Dos Santos, A.C.F.; Lacerda, R.N.D.L.; Póvoa, M.M. Morphological identification of species of the Nuneztovari Complex of Anopheles (Diptera: Culicidae) from an area affected by a Brazilian hydroelectric plant. Zootaxa 2019, 4565, 235–244. [Google Scholar] [CrossRef]
- De Azevedo, T.S.; Lorenz, C.; Chiaravalloti-Neto, F.; Sallum, M.A.M. Kerteszia cruzii and extra-Amazonian malaria in Brazil: Challenges due to climate change in the Atlantic Forest. Infect. Genet. Evol. 2020, 85, 104456. [Google Scholar] [CrossRef] [PubMed]
- Guimarães, A.E.; Lopes, C.M.; de Mello, R.P.; Alencar, J. Ecologia de mosquitos (Diptera;Culicidae) em áreas do Parque Nacional do Iguaçu, Brasil: 1 Distribuição por hábitat [Mosquito (Diptera;Culicidae) ecology in the Iguaçu National Park, Brazil: 1 Habitat distribution]. Cad. Saude Publica 2003, 19, 1107–1116. [Google Scholar] [CrossRef]
- Hutchings, R.S.; Sallum, M.A.; Ferreira, R.L.; Hutchings, R.W. Mosquitoes of the Jaú National Park and their potential importance in Brazilian Amazonia. Med. Vet. Entomol. 2005, 19, 428–441. [Google Scholar] [CrossRef] [PubMed]
- Deane, L.M.; Deane, M.P.; Ferreira Neto, J.; Almeida, F.B. On the transmission of simian malaria in Brazil. Rev. Inst. Med. Trop. São Paulo 1971, 15, 311–319. [Google Scholar]
- Harrison, B.A.; Ruíz-López, F.; Calderón, G.; Savage, H.M.; Pecor, J.E.; Wilkerson, R.C. Anopheles (Kerteszia) lepidotus (Diptera: Culicidae), not the malaria vector we thought it was: Revised male and female morphology, larva, pupa and male genitalia characters and molecular verification. Zootaxa 2012, 3218, 1–17. [Google Scholar] [CrossRef]
- Escovar, J.E.; González, R.; Quiñones, M.L.; Wilkerson, R.C.; Ruiz, F.; Harrison, B.A. Morphology of the larvae; male genitalia and DNA sequences of Anopheles (Kerteszia) pholidotus (Diptera: Culicidae) from Colombia. Mem. Inst. Oswaldo Cruz 2014, 109, 473–479. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sallum, M.A.M.; Wilkerson, R.C.; Forattini, O.P. Taxonomic study of species formerly identified as Anopheles mediopunctatus and resurrection of An. costai (Diptera: Culicidae). J. Med. Entomol. 1999, 36, 282–300. [Google Scholar] [CrossRef] [PubMed]
- Wilkerson, R.C.; Sallum, M.A. Anopheles (Anopheles) forattinii: A new species in Series Arribalzagia (Diptera: Culicidae). J. Med. Entomol. 1999, 36, 345–354. [Google Scholar] [CrossRef]
- De Carvalho, G.C.; Ceretti, W., Jr.; Natal, D.; Medeiros-Sousa, A.R.; Urbinatti, P.R.; de Paula, M.B.; Fernandes, A.; Marrelli, M.T. Occurrence of Anopheles (Anopheles) fluminensis root in the city of São Paulo, Brazil. J. Vector Borne Dis. 2013, 50, 65–67. [Google Scholar] [PubMed]
- Lopes, J.; Lozovei, A.L. Ecologia de mosquitos (Diptera: Culicidae) em criadouros naturais e artificiais de área rural do Norte do Estado do Paraná, Brasil. I—Coletas ao longo do leito do ribeirão. Rev. Saúde Pública 1995, 29, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Neves, A.; Urbinatti, P.R.; dos Santos Malafronte, R.; Fernandes, A.; da Silva Paganini, W.; Natal, D. Malaria outside the Amazon region: Natural Plasmodium infection in anophelines collected near an indigenous village in the Vale do Rio Branco, Itanhaém, SP, Brazil. Acta Trop. 2013, 125, 102–106. [Google Scholar] [CrossRef] [PubMed]
- De Barros, F.S.; de Aguiar, D.B.; Rosa-Freitas, M.G.; Luitgards-Moura, J.F.; Gurgel Hda, C.; Honório, N.A.; de Arruda, M.E.; Tsouris, P.; Vasconcelos, S.D. Distribution summaries of malaria vectors in the northern Brazilian Amazon. J. Vector Ecol. 2007, 32, 161–167. [Google Scholar] [CrossRef]


| Species | Status Plasmodium Vector [Reference] | References for Species Found in This Study |
|---|---|---|
| An. mattogrossensis | Its role as Plasmodium vector is poorly known in Brazil [57] | |
| An. medialis | Its role as Plasmodium vector is poorly known in Brazil [58,59] | |
| Ke. lepidota | It is a Plasmodium vector in Colombia [60] | |
| Ke. neivai | It is a Plasmodium vector in localities on Pacific Coast, Colombia [61] | |
| Ny. darlingi | It is the dominant Plasmodium vector in several regions of the Brazilian Amazon [62] | |
| Ny. dunhami | It has been found naturally Plasmodium infected in Iquitos, Peru [63] | |
| Ny. goeldii | It has been found naturally Plasmodium infected in locations across the Brazilian Amazon [62] | |
| Ny. nuneztovari | It is a primary Plasmodium vector in Venezuela and Colombia [64,65] | |
| Ny. evansae | Unknown [66] | |
| Ny. oswaldoi s.s. | Unknown [29,67] | |
| Ny. oswaldoi B | Potential Plasmodium vector in Putumayo, Colombia [68] | |
| Ny. konderi C | Unknown | [13] |
| Ny. konderi s.l. | Unknown | [13,35] |
| Ny. nuneztovari s.l. | Nyssorhynchus nuneztovari A is a local Plasmodium vector in localities across the Brazilian Amazon [58,69,70,71] | |
| Ny. oswaldoi A | Potential Plasmodium vector in Acre [72] | |
| Ny. tadei | Local vector of Plasmodium across the Brazilian Amazon [4] | |
| Ny. triannulatus | It was found naturally Plasmodium infected in localities across the Brazilian Amazon [58,62] | |
| An. aff. guarao | Unknown | - |
| An. aff. guarao | Unknown | - |
| An. near costai G1 | Unknown | [13] |
| An. near costai G4 | Unknown | [13] |
| An. near fluminensis G1 | Unknown | [13] |
| Ke. neivai A | Unknown | - |
| Ke. neivai B | Unknown | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silva-do-Nascimento, T.F.; Sánchez-Ribas, J.; Oliveira, T.M.P.; Bourke, B.P.; Oliveira-Ferreira, J.; Rosa-Freitas, M.G.; Lourenço-de-Oliveira, R.; Marinho-e-Silva, M.; Neves, M.S.A.S.; Conn, J.E.; et al. Molecular Analysis Reveals a High Diversity of Anopheline Mosquitoes in Yanomami Lands and the Pantanal Region of Brazil. Genes 2021, 12, 1995. https://doi.org/10.3390/genes12121995
Silva-do-Nascimento TF, Sánchez-Ribas J, Oliveira TMP, Bourke BP, Oliveira-Ferreira J, Rosa-Freitas MG, Lourenço-de-Oliveira R, Marinho-e-Silva M, Neves MSAS, Conn JE, et al. Molecular Analysis Reveals a High Diversity of Anopheline Mosquitoes in Yanomami Lands and the Pantanal Region of Brazil. Genes. 2021; 12(12):1995. https://doi.org/10.3390/genes12121995
Chicago/Turabian StyleSilva-do-Nascimento, Teresa Fernandes, Jordi Sánchez-Ribas, Tatiane M. P. Oliveira, Brian Patrick Bourke, Joseli Oliveira-Ferreira, Maria Goreti Rosa-Freitas, Ricardo Lourenço-de-Oliveira, Mariana Marinho-e-Silva, Maycon Sebastião Alberto Santos Neves, Jan E. Conn, and et al. 2021. "Molecular Analysis Reveals a High Diversity of Anopheline Mosquitoes in Yanomami Lands and the Pantanal Region of Brazil" Genes 12, no. 12: 1995. https://doi.org/10.3390/genes12121995
APA StyleSilva-do-Nascimento, T. F., Sánchez-Ribas, J., Oliveira, T. M. P., Bourke, B. P., Oliveira-Ferreira, J., Rosa-Freitas, M. G., Lourenço-de-Oliveira, R., Marinho-e-Silva, M., Neves, M. S. A. S., Conn, J. E., & Sallum, M. A. M. (2021). Molecular Analysis Reveals a High Diversity of Anopheline Mosquitoes in Yanomami Lands and the Pantanal Region of Brazil. Genes, 12(12), 1995. https://doi.org/10.3390/genes12121995








