Current Status and Future Prospects of Perinatal Stem Cells
Abstract
1. Introduction
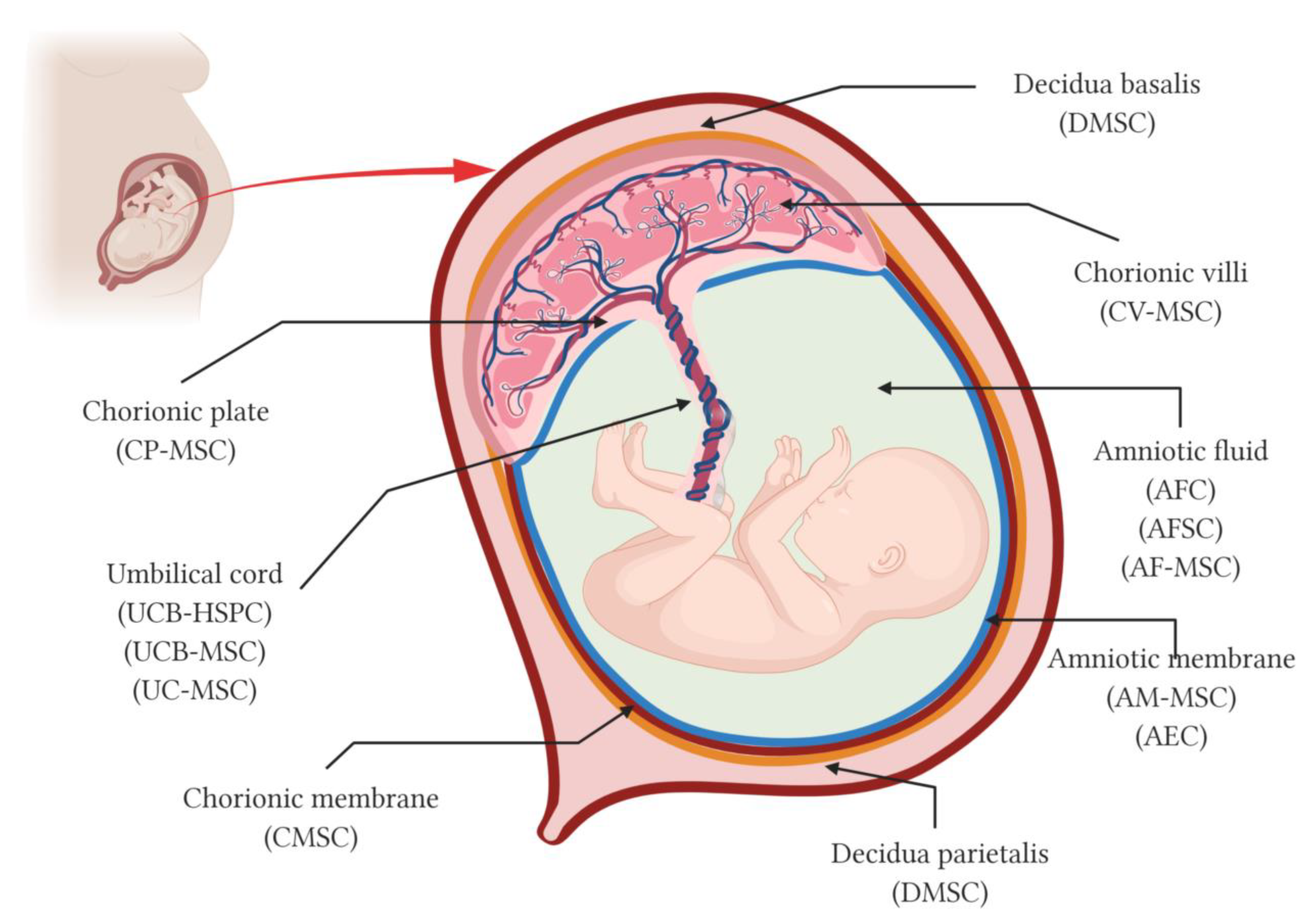
2. The Placenta and Its Fetal Adnexa as a Source of Stem Cells
2.1. Amniotic Fluid
2.2. Amniotic Membrane
2.3. Chorionic Membrane
2.4. Chorionic Plate
2.5. Chorionic Villi
2.6. Umbilical Cord
2.7. Decidua
3. Immunological Properties of Perinatal Stem Cells
4. Biobanking of Perinatal Stem Cells and Tissues
5. Clinical Applications of Perinatal Stem Cells
6. Clinical Use of Perinatal Stem Cells in the Treatment of COVID-19 Pneumonia
7. Nanotechnology for Perinatal-Derived Stromal Cells
8. Future Directions and New Prospects
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Parolini, O.; Alviano, F.; Bagnara, G.P.; Bilic, G.; Buhring, H.J.; Evangelista, M.; Hennerbichler, S.; Liu, B.; Magatti, M.; Mao, N.; et al. Concise review: Isolation and characterization of cells from human term placenta: Outcome of the first international Workshop on Placenta Derived Stem Cells. Stem Cells 2008, 26, 300–311. [Google Scholar] [CrossRef]
- Silini, A.R.; Masserdotti, A.; Papait, A.; Parolini, O. Shaping the Future of Perinatal Cells: Lessons From the Past and Interpretations of the Present. Front. Bioeng. Biotechnol. 2019, 7, 75. [Google Scholar] [CrossRef]
- Silini, A.R.; Di Pietro, P.; Lang, I.; Alviano, F.; Banerjee, A.; Basile, M.; Borutinskaitė, V.V.; Eissner, G.; Gellhaus, A.; Giebel, B.; et al. Perinatal derivatives: Where do we stand? A roadmap of the human placenta and consensus for tissue and cell nomenclature. Front. Bioeng. Biotechnol. 2020. Provisionally accepted. [Google Scholar]
- Hoehn, H.; Bryant, E.M.; Fantel, A.G.; Martin, G.M. Cultivated cells from diagnostic amniocentesis in second trimester pregnancies. III. The fetal urine as a potential source of clonable cells. Humangenetik 1975, 29, 285–290. [Google Scholar] [CrossRef]
- Moraghebi, R.; Kirkeby, A.; Chaves, P.; Ronn, R.E.; Sitnicka, E.; Parmar, M.; Larsson, M.; Herbst, A.; Woods, N.B. Term amniotic fluid: An unexploited reserve of mesenchymal stromal cells for reprogramming and potential cell therapy applications. Stem Cell Res. Ther. 2017, 8, 190. [Google Scholar] [CrossRef]
- Prusa, A.R.; Marton, E.; Rosner, M.; Bernaschek, G.; Hengstschlager, M. Oct-4-expressing cells in human amniotic fluid: A new source for stem cell research? Hum. Reprod. 2003, 18, 1489–1493. [Google Scholar] [CrossRef]
- Tsai, M.S.; Lee, J.L.; Chang, Y.J.; Hwang, S.M. Isolation of human multipotent mesenchymal stem cells from second-trimester amniotic fluid using a novel two-stage culture protocol. Hum. Reprod. 2004, 19, 1450–1456. [Google Scholar] [CrossRef]
- Bossolasco, P.; Montemurro, T.; Cova, L.; Zangrossi, S.; Calzarossa, C.; Buiatiotis, S.; Soligo, D.; Bosari, S.; Silani, V.; Deliliers, G.L.; et al. Molecular and phenotypic characterization of human amniotic fluid cells and their differentiation potential. Cell Res. 2006, 16, 329–336. [Google Scholar] [CrossRef]
- Loukogeorgakis, S.P.; De Coppi, P. Concise Review: Amniotic Fluid Stem Cells: The Known, the Unknown, and Potential Regenerative Medicine Applications. Stem Cells 2017, 35, 1663–1673. [Google Scholar] [CrossRef]
- Moschidou, D.; Mukherjee, S.; Blundell, M.P.; Jones, G.N.; Atala, A.J.; Thrasher, A.J.; Fisk, N.M.; De Coppi, P.; Guillot, P.V. Human mid-trimester amniotic fluid stem cells cultured under embryonic stem cell conditions with valproic acid acquire pluripotent characteristics. Stem Cells Dev. 2013, 22, 444–458. [Google Scholar] [CrossRef]
- Roubelakis, M.G.; Pappa, K.I.; Bitsika, V.; Zagoura, D.; Vlahou, A.; Papadaki, H.A.; Antsaklis, A.; Anagnou, N.P. Molecular and proteomic characterization of human mesenchymal stem cells derived from amniotic fluid: Comparison to bone marrow mesenchymal stem cells. Stem Cells Dev. 2007, 16, 931–952. [Google Scholar] [CrossRef]
- Tsai, M.S.; Hwang, S.M.; Tsai, Y.L.; Cheng, F.C.; Lee, J.L.; Chang, Y.J. Clonal amniotic fluid-derived stem cells express characteristics of both mesenchymal and neural stem cells. Biol. Reprod. 2006, 74, 545–551. [Google Scholar] [CrossRef]
- Zagoura, D.S.; Trohatou, O.; Bitsika, V.; Makridakis, M.; Pappa, K.I.; Vlahou, A.; Roubelakis, M.G.; Anagnou, N.P. AF-MSCs fate can be regulated by culture conditions. Cell Death Dis. 2013, 4, e571. [Google Scholar] [CrossRef]
- Savickiene, J.; Treigyte, G.; Baronaite, S.; Valiuliene, G.; Kaupinis, A.; Valius, M.; Arlauskiene, A.; Navakauskiene, R. Human Amniotic Fluid Mesenchymal Stem Cells from Second- and Third-Trimester Amniocentesis: Differentiation Potential, Molecular Signature, and Proteome Analysis. Stem Cells Int. 2015, 2015, 319238. [Google Scholar] [CrossRef]
- De Coppi, P.; Bartsch, G., Jr.; Siddiqui, M.M.; Xu, T.; Santos, C.C.; Perin, L.; Mostoslavsky, G.; Serre, A.C.; Snyder, E.Y.; Yoo, J.J.; et al. Isolation of amniotic stem cell lines with potential for therapy. Nat. Biotechnol. 2007, 25, 100–106. [Google Scholar] [CrossRef]
- Rosner, M.; Schipany, K.; Shanmugasundaram, B.; Lubec, G.; Hengstschlager, M. Amniotic fluid stem cells: Future perspectives. Stem Cells Int. 2012, 2012, 741810. [Google Scholar] [CrossRef]
- Srivastava, M.; Ahlawat, N.; Srivastava, A. Amniotic Fluid Stem Cells: A New Era in Regenerative Medicine. J. Obstet. Gynecol. India 2018, 68, 15–19. [Google Scholar] [CrossRef]
- Moore, R.; Silver, R.; Moore, J. Physiological apoptotic agents have different effects upon human amnion epithelial and mesenchymal cells. Placenta 2003, 24, 173–180. [Google Scholar] [CrossRef]
- Casey, M.L.; Macdonald, P.C. Interstitial collagen synthesis and processing in human amnion: A property of the mesenchymal cells. Biol. Reprod. 1996, 55, 1253–1260. [Google Scholar] [CrossRef]
- Kim, J.; Kang, H.M.; Kim, H.; Kim, M.R.; Kwon, H.C.; Gye, M.C.; Kang, S.G.; Yang, H.S.; You, J. Ex vivo characteristics of human amniotic membrane-derived stem cells. Cloning Stem Cells 2007, 9, 581–594. [Google Scholar] [CrossRef]
- Manuelpillai, U.; Moodley, Y.; Borlongan, C.V.; Parolini, O. Amniotic membrane and amniotic cells: Potential therapeutic tools to combat tissue inflammation and fibrosis? Placenta 2011, 32 (Suppl. 4), S320–S325. [Google Scholar] [CrossRef]
- Cargnoni, A.; Gibelli, L.; Tosini, A.; Signoroni, P.B.; Nassuato, C.; Arienti, D.; Lombardi, G.; Albertini, A.; Wengler, G.S.; Parolini, O. Transplantation of allogeneic and xenogeneic placenta-derived cells reduces bleomycin-induced lung fibrosis. Cell Transplant. 2009, 18, 405–422. [Google Scholar] [CrossRef]
- Liu, D.; Hui, H.; Chai, X.; Wang, B.; Qiu, J. Construction of tissue-engineered cartilage using human placenta-derived stem cells. Sci. China Life Sci. 2010, 53, 207–214. [Google Scholar] [CrossRef]
- Portmann-Lanz, C.B.; Schoeberlein, A.; Huber, A.; Sager, R.; Malek, A.; Holzgreve, W.; Surbek, D.V. Placental mesenchymal stem cells as potential autologous graft for pre- and perinatal neuroregeneration. Am. J. Obstet. Gynecol. 2006, 194, 664–673. [Google Scholar] [CrossRef]
- Rylova, Y.V.; Milovanova, N.V.; Gordeeva, M.N.; Savilova, A.M. Characteristics of Multipotent Mesenchymal Stromal Cells from Human Terminal Placenta. Bull. Exp. Biol. Med. 2015, 159, 253–257. [Google Scholar] [CrossRef]
- Miki, T.; Mitamura, K.; Ross, M.A.; Stolz, D.B.; Strom, S.C. Identification of stem cell marker-positive cells by immunofluorescence in term human amnion. J. Reprod. Immunol. 2007, 75, 91–96. [Google Scholar] [CrossRef]
- Garcia-Castro, I.L.; Garcia-Lopez, G.; Avila-Gonzalez, D.; Flores-Herrera, H.; Molina-Hernandez, A.; Portillo, W.; Ramon-Gallegos, E.; Diaz, N.F. Markers of Pluripotency in Human Amniotic Epithelial Cells and Their Differentiation to Progenitor of Cortical Neurons. PLoS ONE 2015, 10, e0146082. [Google Scholar] [CrossRef]
- Maymo, J.L.; Riedel, R.; Perez-Perez, A.; Magatti, M.; Maskin, B.; Duenas, J.L.; Parolini, O.; Sanchez-Margalet, V.; Varone, C.L. Proliferation and survival of human amniotic epithelial cells during their hepatic differentiation. PLoS ONE 2018, 13, e0191489. [Google Scholar] [CrossRef]
- Ilancheran, S.; Michalska, A.; Peh, G.; Wallace, E.M.; Pera, M.; Manuelpillai, U. Stem cells derived from human fetal membranes display multilineage differentiation potential. Biol. Reprod. 2007, 77, 577–588. [Google Scholar] [CrossRef]
- Miki, T.; Marongiu, F.; Ellis, E.; Strom, C.S. Isolation of amniotic epithelial stem cells. Curr. Protoc. Stem Cell Biol. 2007, 12, 1E-3. [Google Scholar] [CrossRef]
- Strom, S.C.; Gramignoli, R. Human amnion epithelial cells expressing HLA-G as novel cell-based treatment for liver disease. Hum. Immunol. 2016, 77, 734–739. [Google Scholar] [CrossRef]
- Magatti, M.; Vertua, E.; Cargnoni, A.; Silini, A.; Parolini, O. The Immunomodulatory Properties of Amniotic Cells: The Two Sides of the Coin. Cell Transpl. 2018, 27, 31–44. [Google Scholar] [CrossRef]
- Nakamura, T.; Inatomi, T.; Sotozono, C.; Ang, L.P.; Koizumi, N.; Yokoi, N.; Kinoshita, S. Transplantation of autologous serum-derived cultivated corneal epithelial equivalents for the treatment of severe ocular surface disease. Ophthalmology 2006, 113, 1765–1772. [Google Scholar] [CrossRef]
- Takahashi, S.; Ohsugi, K.; Yamamoto, T.; Shiomi, M.; Sakuragawa, N. A novel approach to ex vivo gene therapy for familial hypercholesterolemia using human amniotic epithelial cells as a transgene carrier. Tohoku J. Exp. Med. 2001, 193, 279–292. [Google Scholar] [CrossRef][Green Version]
- Fujimoto, K.L.; Miki, T.; Liu, L.J.; Hashizume, R.; Strom, S.C.; Wagner, W.R.; Keller, B.B.; Tobita, K. Naive rat amnion-derived cell transplantation improved left ventricular function and reduced myocardial scar of postinfarcted heart. Cell Transpl. 2009, 18, 477–486. [Google Scholar] [CrossRef]
- Manuelpillai, U.; Tchongue, J.; Lourensz, D.; Vaghjiani, V.; Samuel, C.S.; Liu, A.; Williams, E.D.; Sievert, W. Transplantation of human amnion epithelial cells reduces hepatic fibrosis in immunocompetent CCl(4)-treated mice. Cell Transpl. 2010, 19, 1157–1168. [Google Scholar] [CrossRef]
- Kawamichi, Y.; Cui, C.H.; Toyoda, M.; Makino, H.; Horie, A.; Takahashi, Y.; Matsumoto, K.; Saito, H.; Ohta, H.; Saito, K.; et al. Cells of extraembryonic mesodermal origin confer human dystrophin in the mdx model of Duchenne muscular dystrophy. J. Cell Physiol. 2010, 223, 695–702. [Google Scholar] [CrossRef]
- Sankar, V.; Muthusamy, R. Role of human amniotic epithelial cell transplantation in spinal cord injury repair research. Neuroscience 2003, 118, 11–17. [Google Scholar] [CrossRef]
- Park, S.; Kim, E.; Koh, S.E.; Maeng, S.; Lee, W.D.; Lim, J.; Shim, I.; Lee, Y.J. Dopaminergic differentiation of neural progenitors derived from placental mesenchymal stem cells in the brains of Parkinson’s disease model rats and alleviation of asymmetric rotational behavior. Brain Res. 2012, 1466, 158–166. [Google Scholar] [CrossRef]
- Chen, Z.; Tortella, F.C.; Dave, J.R.; Marshall, V.S.; Clarke, D.L.; Sing, G.; Du, F.; Lu, X.C. Human amnion-derived multipotent progenitor cell treatment alleviates traumatic brain injury-induced axonal degeneration. J. Neurotrauma 2009, 26, 1987–1997. [Google Scholar] [CrossRef]
- Fisher-Shoval, Y.; Barhum, Y.; Sadan, O.; Yust-Katz, S.; Ben-Zur, T.; Lev, N.; Benkler, C.; Hod, M.; Melamed, E.; Offen, D. Transplantation of placenta-derived mesenchymal stem cells in the EAE mouse model of MS. J. Mol. Neurosci. 2012, 48, 176–184. [Google Scholar] [CrossRef]
- Niknejad, H.; Peirovi, H.; Jorjani, M.; Ahmadiani, A.; Ghanavi, J.; Seifalian, A.M. Properties of the amniotic membrane for potential use in tissue engineering. Eur. Cell Mater. 2008, 15, 88–99. [Google Scholar] [CrossRef]
- Song, M.; Wang, W.; Ye, Q.; Bu, S.; Shen, Z.; Zhu, Y. The repairing of full-thickness skin deficiency and its biological mechanism using decellularized human amniotic membrane as the wound dressing. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 77, 739–747. [Google Scholar] [CrossRef]
- Chen, X.; Zhou, Y. Preventive effects of transplantation of oral mucosal epithelial cells seeded on a decellularized amniotic membrane in a model of intrauterine adhesion. Int. J. Clin. Exp. Pathol. 2018, 11, 1510–1519. [Google Scholar]
- Fenelon, M.; Catros, S.; Fricain, J.C. What is the benefit of using amniotic membrane in oral surgery? A comprehensive review of clinical studies. Clin. Oral Investig. 2018, 22, 1881–1891. [Google Scholar] [CrossRef]
- Jirsova, K.; Jones, G.L.A. Amniotic membrane in ophthalmology: Properties, preparation, storage and indications for grafting-a review. Cell Tissue Bank. 2017, 18, 193–204. [Google Scholar] [CrossRef]
- Shi, P.; Gao, M.; Shen, Q.; Hou, L.; Zhu, Y.; Wang, J. Biocompatible surgical meshes based on decellularized human amniotic membrane. Mater. Sci. Eng. C 2015, 54, 112–119. [Google Scholar] [CrossRef]
- Soncini, M.; Vertua, E.; Gibelli, L.; Zorzi, F.; Denegri, M.; Albertini, A.; Wengler, G.S.; Parolini, O. Isolation and characterization of mesenchymal cells from human fetal membranes. J. Tissue Eng. Regen. Med. 2007, 1, 296–305. [Google Scholar] [CrossRef]
- Araujo, A.B.; Salton, G.D.; Furlan, J.M.; Schneider, N.; Angeli, M.H.; Laureano, A.M.; Silla, L.; Passos, E.P.; Paz, A.H. Comparison of human mesenchymal stromal cells from four neonatal tissues: Amniotic membrane, chorionic membrane, placental decidua and umbilical cord. Cytotherapy 2017, 19, 577–585. [Google Scholar] [CrossRef]
- Bailo, M.; Soncini, M.; Vertua, E.; Signoroni, P.B.; Sanzone, S.; Lombardi, G.; Arienti, D.; Calamani, F.; Zatti, D.; Paul, P.; et al. Engraftment potential of human amnion and chorion cells derived from term placenta. Transplantation 2004, 78, 1439–1448. [Google Scholar] [CrossRef]
- Battula, V.L.; Bareiss, P.M.; Treml, S.; Conrad, S.; Albert, I.; Hojak, S.; Abele, H.; Schewe, B.; Just, L.; Skutella, T.; et al. Human placenta and bone marrow derived MSC cultured in serum-free, b-FGF-containing medium express cell surface frizzled-9 and SSEA-4 and give rise to multilineage differentiation. Differentiation 2007, 75, 279–291. [Google Scholar] [CrossRef]
- Deus, I.A.; Mano, J.F.; Custódio, C.A. Perinatal tissues and cells in tissue engineering and regenerative medicine. Acta Biomater. 2020, 110, 1–14. [Google Scholar] [CrossRef]
- Huang, Q.; Yang, Y.; Luo, C.; Wen, Y.; Liu, R.; Li, S.; Chen, T.; Sun, H.; Tang, L. An efficient protocol to generate placental chorionic plate-derived mesenchymal stem cells with superior proliferative and immunomodulatory properties. Stem Cell Res. Ther. 2019, 10, 301. [Google Scholar] [CrossRef]
- Kim, M.J.; Shin, K.S.; Jeon, J.H.; Lee, D.R.; Shim, S.H.; Kim, J.K.; Cha, D.H.; Yoon, T.K.; Kim, G.J. Human chorionic-plate-derived mesenchymal stem cells and Wharton’s jelly-derived mesenchymal stem cells: A comparative analysis of their potential as placenta-derived stem cells. Cell Tissue Res. 2011, 346, 53–64. [Google Scholar] [CrossRef]
- Ma, J.; Wu, J.; Han, L.; Jiang, X.; Yan, L.; Hao, J.; Wang, H. Comparative analysis of mesenchymal stem cells derived from amniotic membrane, umbilical cord, and chorionic plate under serum-free condition. Stem Cell Res. Ther. 2019, 10, 19. [Google Scholar] [CrossRef]
- Lee, M.J.; Jung, J.; Na, K.H.; Moon, J.S.; Lee, H.J.; Kim, J.H.; Kim, G.I.; Kwon, S.W.; Hwang, S.G.; Kim, G.J. Anti-fibrotic effect of chorionic plate-derived mesenchymal stem cells isolated from human placenta in a rat model of CCl(4)-injured liver: Potential application to the treatment of hepatic diseases. J. Cell Biochem. 2010, 111, 1453–1463. [Google Scholar] [CrossRef]
- Lee, Y.B.; Choi, J.H.; Kim, E.N.; Seok, J.; Lee, H.J.; Yoon, J.H.; Kim, G.J. Human Chorionic Plate-Derived Mesenchymal Stem Cells Restore Hepatic Lipid Metabolism in a Rat Model of Bile Duct Ligation. Stem Cells Int. 2017, 2017, 5180579. [Google Scholar] [CrossRef]
- Chung, S.; Rho, S.; Kim, G.; Kim, S.R.; Baek, K.H.; Kang, M.; Lew, H. Human umbilical cord blood mononuclear cells and chorionic plate-derived mesenchymal stem cells promote axon survival in a rat model of optic nerve crush injury. Int. J. Mol. Med. 2016, 37, 1170–1180. [Google Scholar] [CrossRef]
- Li, J.; Yu, Q.; Huang, H.; Deng, W.; Cao, X.; Adu-Frimpong, M.; Yu, J.; Xu, X. Human chorionic plate-derived mesenchymal stem cells transplantation restores ovarian function in a chemotherapy-induced mouse model of premature ovarian failure. Stem Cell Res. Ther. 2018, 9, 81. [Google Scholar] [CrossRef]
- Zhang, X.; Mitsuru, A.; Igura, K.; Takahashi, K.; Ichinose, S.; Yamaguchi, S.; Takahashi, T.A. Mesenchymal progenitor cells derived from chorionic villi of human placenta for cartilage tissue engineering. Biochem. Biophys. Res. Commun. 2006, 340, 944–952. [Google Scholar] [CrossRef]
- Heazlewood, C.F.; Sherrell, H.; Ryan, J.; Atkinson, K.; Wells, C.A.; Fisk, N.M. High incidence of contaminating maternal cell overgrowth in human placental mesenchymal stem/stromal cell cultures: A systematic review. Stem Cells Transl. Med. 2014, 3, 1305–1311. [Google Scholar] [CrossRef]
- Ventura Ferreira, M.S.; Bienert, M.; Muller, K.; Rath, B.; Goecke, T.; Oplander, C.; Braunschweig, T.; Mela, P.; Brummendorf, T.H.; Beier, F.; et al. Comprehensive characterization of chorionic villi-derived mesenchymal stromal cells from human placenta. Stem Cell Res. Ther. 2018, 9, 28. [Google Scholar] [CrossRef]
- Katsiani, E.; Garas, A.; Skentou, C.; Tsezou, A.; Messini, C.I.; Dafopoulos, K.; Daponte, A.; Messinis, I.E. Chorionic villi derived mesenchymal like stem cells and expression of embryonic stem cells markers during long-term culturing. Cell Tissue Bank. 2016, 17, 517–529. [Google Scholar] [CrossRef]
- Liu, H.; Murthi, P.; Qin, S.; Kusuma, G.D.; Borg, A.J.; Knofler, M.; Haslinger, P.; Manuelpillai, U.; Pertile, M.D. A novel combination of homeobox genes is expressed in mesenchymal chorionic stem/stromal cells in first trimester and term pregnancies. Reprod. Sci. 2014, 21, 1382–1394. [Google Scholar] [CrossRef]
- Chien, C.C.; Yen, B.L.; Lee, F.K.; Lai, T.H.; Chen, Y.C.; Chan, S.H.; Huang, H.I. In vitro differentiation of human placenta-derived multipotent cells into hepatocyte-like cells. Stem Cells 2006, 24, 1759–1768. [Google Scholar] [CrossRef]
- Basmaeil, Y.S.; Al Subayyil, A.M.; Khatlani, T.; Bahattab, E.; Al-Alwan, M.; Abomaray, F.M.; Kalionis, B.; Alshabibi, M.A.; AlAskar, A.S.; Abumaree, M.H. Human chorionic villous mesenchymal stem/stromal cells protect endothelial cells from injury induced by high level of glucose. Stem Cell Res. Ther. 2018, 9, 238. [Google Scholar] [CrossRef]
- Alshareeda, A.T.; Rakha, E.; Alghwainem, A.; Alrfaei, B.; Alsowayan, B.; Albugami, A.; Alsubayyil, A.M.; Abomraee, M.; Mohd Zin, N.K. The effect of human placental chorionic villi derived mesenchymal stem cell on triple-negative breast cancer hallmarks. PLoS ONE 2018, 13, e0207593. [Google Scholar] [CrossRef]
- Knudtzon, S. In vitro growth of granulocytic colonies from circulating cells in human cord blood. Blood 1974, 43, 357–361. [Google Scholar] [CrossRef]
- Broxmeyer, H.E.; Douglas, G.W.; Hangoc, G.; Cooper, S.; Bard, J.; English, D.; Arny, M.; Thomas, L.; Boyse, E.A. Human umbilical cord blood as a potential source of transplantable hematopoietic stem/progenitor cells. Proc. Natl. Acad. Sci. USA 1989, 86, 3828–3832. [Google Scholar] [CrossRef]
- Gluckman, E.; Broxmeyer, H.A.; Auerbach, A.D.; Friedman, H.S.; Douglas, G.W.; Devergie, A.; Esperou, H.; Thierry, D.; Socie, G.; Lehn, P.; et al. Hematopoietic reconstitution in a patient with Fanconi’s anemia by means of umbilical-cord blood from an HLA-identical sibling. N. Engl. J. Med. 1989, 321, 1174–1178. [Google Scholar] [CrossRef]
- Brown, K.S.; Rao, M.S.; Brown, H.L. The Future State of Newborn Stem Cell Banking. J. Clin. Med. 2019, 8, 117. [Google Scholar] [CrossRef]
- Bornstein, R.; Flores, A.I.; Montalban, M.A.; del Rey, M.J.; de la Serna, J.; Gilsanz, F. A modified cord blood collection method achieves sufficient cell levels for transplantation in most adult patients. Stem Cells 2005, 23, 324–334. [Google Scholar] [CrossRef]
- Ballen, K.K.; Gluckman, E.; Broxmeyer, H.E. Umbilical cord blood transplantation: The first 25 years and beyond. Blood 2013, 122, 491–498. [Google Scholar] [CrossRef]
- Rebulla, P.; Querol, S.; Madrigal, A. Umbilical Cord Blood as a Source of Novel Reagents and Therapeutics. In Perinatal Stem Cells; Han, Z., Takahashi, T., Han, Z., Li, Z., Eds.; Springer: Singapore, 2019; pp. 75–81. [Google Scholar]
- Zheng, C.C.; Zhu, X.Y.; Tang, B.L.; Zhang, X.H.; Zhang, L.; Geng, L.Q.; Liu, H.L.; Sun, Z.M. Double vs. single cord blood transplantation in adolescent and adult hematological malignancies with heavier body weight (>/=50 kg). Hematology 2018, 23, 96–104. [Google Scholar] [CrossRef]
- Mousavi, S.H.; Abroun, S.; Soleimani, M.; Mowla, S.J. 3-Dimensional nano-fibre scaffold for ex vivo expansion of cord blood haematopoietic stem cells. Artif. Cells Nanomed. Biotechnol. 2017, 46, 740–748. [Google Scholar] [CrossRef]
- Flores, A.I.; McKenna, D.H.; Montalban, M.A.; De la Cruz, J.; Wagner, J.E.; Bornstein, R. Consistency of the initial cell acquisition procedure is critical to the standardization of CD34+ cell enumeration by flow cytometry: Results of a pairwise analysis of umbilical cord blood units and cryopreserved aliquots. Transfusion 2009, 49, 636–647. [Google Scholar] [CrossRef]
- Aljitawi, O.S.; Paul, S.; Ganguly, A.; Lin, T.L.; Ganguly, S.; Vielhauer, G.; Capitano, M.L.; Cantilena, A.; Lipe, B.; Mahnken, J.D.; et al. Erythropoietin modulation is associated with improved homing and engraftment after umbilical cord blood transplantation. Blood 2016, 128, 3000–3010. [Google Scholar] [CrossRef]
- Battiwalla, M.; Hematti, P. Mesenchymal stem cells in hematopoietic stem cell transplantation. Cytotherapy 2009, 11, 503–515. [Google Scholar] [CrossRef] [PubMed]
- Pipes, B.; Tsang, T.; Peng, S.-X.; Fiederlein, R.; Graham, M.; Harris, D. Telomere length changes after umbilical cord blood transplant. Transfusion 2006, 46, 1038–1043. [Google Scholar] [CrossRef]
- Hordyjewska, A.; Popiołek, Ł.; Horecka, A. Characteristics of hematopoietic stem cells of umbilical cord blood. Cytotechnology 2015, 67, 387–396. [Google Scholar] [CrossRef] [PubMed]
- Kuchma, M.D.; Kyryk, V.M.; Svitina, H.M.; Shablii, Y.M.; Lukash, L.L.; Lobyntseva, G.S.; Shablii, V.A. Comparative Analysis of the Hematopoietic Progenitor Cells from Placenta, Cord Blood, and Fetal Liver, Based on Their Immunophenotype. BioMed Res. Int. 2015, 2015, 418752. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Sun, Z.; Lv, G.; Guo, X.; Zhang, Y.; Yu, W.; Wang, W.; Ma, X. Microencapsulated UCB cells repair hepatic injure by intraperitoneal transplantation. Cytotherapy 2009, 11, 1032–1040. [Google Scholar] [CrossRef] [PubMed]
- Vendrame, M.; Gemma, C.; de Mesquita, D.; Collier, L.; Bickford, P.C.; Sanberg, C.D.; Sanberg, P.R.; Pennypacker, K.R.; Willing, A.E. Anti-inflammatory effects of human cord blood cells in a rat model of stroke. Stem Cells Dev. 2005, 14, 595–604. [Google Scholar] [CrossRef] [PubMed]
- Burkhart, H.M.; Qureshi, M.Y.; Peral, S.C.; O’Leary, P.W.; Olson, T.M.; Cetta, F.; Nelson, T.J.; Wanek Program Clinical Pipeline Group. Regenerative therapy for hypoplastic left heart syndrome: First report of intraoperative intramyocardial injection of autologous umbilical-cord blood-derived cells. J. Thorac. Cardiovasc. Surg. 2015, 149, e35–e37. [Google Scholar] [CrossRef]
- Haller, M.J.; Viener, H.L.; Wasserfall, C.; Brusko, T.; Atkinson, M.A.; Schatz, D.A. Autologous umbilical cord blood infusion for type 1 diabetes. Exp. Hematol. 2008, 36, 710–715. [Google Scholar] [CrossRef][Green Version]
- Penny, T.R.; Sutherland, A.E.; Mihelakis, J.G.; Paton, M.C.B.; Pham, Y.; Lee, J.; Jones, N.M.; Jenkin, G.; Fahey, M.C.; Miller, S.L.; et al. Human Umbilical Cord Therapy Improves Long-Term Behavioral Outcomes Following Neonatal Hypoxic Ischemic Brain Injury. Front. Physiol. 2019, 10, 283. [Google Scholar] [CrossRef]
- Giannaccare, G.; Carnevali, A.; Senni, C.; Logozzo, L.; Scorcia, V. Umbilical Cord Blood and Serum for the Treatment of Ocular Diseases: A Comprehensive Review. Ophthalmol. Ther. 2020, 9, 235–248. [Google Scholar] [CrossRef]
- Mareschi, K.; Biasin, E.; Piacibello, W.; Aglietta, M.; Madon, E.; Fagioli, F. Isolation of human mesenchymal stem cells: Bone marrow versus umbilical cord blood. Haematologica 2001, 86, 1099–1100. [Google Scholar]
- Wexler, S.A.; Donaldson, C.; Denning-Kendall, P.; Rice, C.; Bradley, B.; Hows, J.M. Adult bone marrow is a rich source of human mesenchymal ‘stem’ cells but umbilical cord and mobilized adult blood are not. Br. J. Haematol. 2003, 121, 368–374. [Google Scholar] [CrossRef]
- Yang, S.E.; Ha, C.W.; Jung, M.; Jin, H.J.; Lee, M.; Song, H.; Choi, S.; Oh, W.; Yang, Y.S. Mesenchymal stem/progenitor cells developed in cultures from UC blood. Cytotherapy 2004, 6, 476–486. [Google Scholar] [CrossRef]
- Amati, E.; Sella, S.; Perbellini, O.; Alghisi, A.; Bernardi, M.; Chieregato, K.; Lievore, C.; Peserico, D.; Rigno, M.; Zilio, A.; et al. Generation of mesenchymal stromal cells from cord blood: Evaluation of in vitro quality parameters prior to clinical use. Stem Cell Res. Ther. 2017, 8, 14. [Google Scholar] [CrossRef] [PubMed]
- Yoshioka, S.; Miura, Y.; Iwasa, M.; Fujishiro, A.; Yao, H.; Miura, M.; Fukuoka, M.; Nakagawa, Y.; Yokota, A.; Hirai, H.; et al. Isolation of mesenchymal stromal/stem cells from small-volume umbilical cord blood units that do not qualify for the banking system. Int. J. Hematol. 2015, 102, 218–229. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Yue, A.; Ruan, Z.; Yin, Y.; Wang, R.; Ren, Y.; Zhu, L. Human umbilical cord-derived mesenchymal stem cells do not undergo malignant transformation during long-term culturing in serum-free medium. PLoS ONE 2014, 9, e98565. [Google Scholar] [CrossRef]
- Batsali, A.K.; Kastrinaki, M.C.; Papadaki, H.A.; Pontikoglou, C. Mesenchymal stem cells derived from Wharton’s Jelly of the umbilical cord: Biological properties and emerging clinical applications. Curr. Stem Cell Res. Ther. 2013, 8, 144–155. [Google Scholar] [CrossRef]
- El Omar, R.; Beroud, J.; Stoltz, J.F.; Menu, P.; Velot, E.; Decot, V. Umbilical cord mesenchymal stem cells: The new gold standard for mesenchymal stem cell-based therapies? Tissue Eng. Part B Rev. 2014, 20, 523–544. [Google Scholar] [CrossRef]
- Arutyunyan, I.; Elchaninov, A.; Makarov, A.; Fatkhudinov, T. Umbilical Cord as Prospective Source for Mesenchymal Stem Cell-Based Therapy. Stem Cells Int. 2016, 2016, 6901286. [Google Scholar] [CrossRef]
- Greco, S.J.; Liu, K.; Rameshwar, P. Functional similarities among genes regulated by OCT4 in human mesenchymal and embryonic stem cells. Stem Cells 2007, 25, 3143–3154. [Google Scholar] [CrossRef]
- He, H.; Nagamura-Inoue, T.; Tsunoda, H.; Yuzawa, M.; Yamamoto, Y.; Yorozu, P.; Agata, H.; Tojo, A. Stage-specific embryonic antigen 4 in Wharton’s jelly-derived mesenchymal stem cells is not a marker for proliferation and multipotency. Tissue Eng. Part A 2014, 20, 1314–1324. [Google Scholar] [CrossRef]
- Baksh, D.; Yao, R.; Tuan, R.S. Comparison of proliferative and multilineage differentiation potential of human mesenchymal stem cells derived from umbilical cord and bone marrow. Stem Cells 2007, 25, 1384–1392. [Google Scholar] [CrossRef]
- Karahuseyinoglu, S.; Cinar, O.; Kilic, E.; Kara, F.; Akay, G.G.; Demiralp, D.O.; Tukun, A.; Uckan, D.; Can, A. Biology of stem cells in human umbilical cord stroma: In situ and in vitro surveys. Stem Cells 2007, 25, 319–331. [Google Scholar] [CrossRef]
- Conconi, M.T.; Burra, P.; Di Liddo, R.; Calore, C.; Turetta, M.; Bellini, S.; Bo, P.; Nussdorfer, G.G.; Parnigotto, P.P. CD105(+) cells from Wharton’s jelly show in vitro and in vivo myogenic differentiative potential. Int. J. Mol. Med. 2006, 18, 1089–1096. [Google Scholar] [CrossRef] [PubMed]
- Kadivar, M.; Khatami, S.; Mortazavi, Y.; Shokrgozar, M.A.; Taghikhani, M.; Soleimani, M. In vitro cardiomyogenic potential of human umbilical vein-derived mesenchymal stem cells. Biochem. Biophys. Res. Commun. 2006, 340, 639–647. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.S.; Cheng, Y.C.; Lin, M.Y.; Cheng, H.; Chu, P.M.; Chou, S.C.; Shih, Y.H.; Ko, M.H.; Sung, M.S. Conversion of human umbilical cord mesenchymal stem cells in Wharton’s jelly to dopaminergic neurons in vitro: Potential therapeutic application for Parkinsonism. Stem Cells 2006, 24, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.S.; Shih, Y.T.; Cheng, Y.C.; Min, M.Y. Transformation of human umbilical mesenchymal cells into neurons in vitro. J. Biomed. Sci. 2004, 11, 652–660. [Google Scholar] [CrossRef]
- Campard, D.; Lysy, P.A.; Najimi, M.; Sokal, E.M. Native umbilical cord matrix stem cells express hepatic markers and differentiate into hepatocyte-like cells. Gastroenterology 2008, 134, 833–848. [Google Scholar] [CrossRef]
- Wu, L.F.; Wang, N.N.; Liu, Y.S.; Wei, X. Differentiation of Wharton’s jelly primitive stromal cells into insulin-producing cells in comparison with bone marrow mesenchymal stem cells. Tissue Eng. Part A 2009, 15, 2865–2873. [Google Scholar] [CrossRef]
- Wu, K.H.; Zhou, B.; Lu, S.H.; Feng, B.; Yang, S.G.; Du, W.T.; Gu, D.S.; Han, Z.C.; Liu, Y.L. In vitro and in vivo differentiation of human umbilical cord derived stem cells into endothelial cells. J. Cell Biochem. 2007, 100, 608–616. [Google Scholar] [CrossRef]
- Latifpour, M.; Shakiba, Y.; Amidi, F.; Mazaheri, Z.; Sobhani, A. Differentiation of human umbilical cord matrix-derived mesenchymal stem cells into germ-like cells. Avicenna J. Med. Biotechnol. 2014, 6, 218–227. [Google Scholar]
- Nasadyuk, C.M. Umbilical cord stem cells: Biological characteristics, approaches to banking and clinical application. Cell Organ Transplantol. 2016, 4, 230–235. [Google Scholar] [CrossRef]
- Macias, M.I.; Grande, J.; Moreno, A.; Dominguez, I.; Bornstein, R.; Flores, A.I. Isolation and characterization of true mesenchymal stem cells derived from human term decidua capable of multilineage differentiation into all 3 embryonic layers. Am. J. Obstet. Gynecol. 2010, 203, 495.e9–495.e23. [Google Scholar] [CrossRef]
- Abomaray, F.M.; Al Jumah, M.A.; Alsaad, K.O.; Jawdat, D.; Al Khaldi, A.; AlAskar, A.S.; Al Harthy, S.; Al Subayyil, A.M.; Khatlani, T.; Alawad, A.O.; et al. Phenotypic and Functional Characterization of Mesenchymal Stem/Multipotent Stromal Cells from Decidua Basalis of Human Term Placenta. Stem Cells Int. 2016, 2016, 5184601. [Google Scholar] [CrossRef] [PubMed]
- Barlow, S.; Brooke, G.; Chatterjee, K.; Price, G.; Pelekanos, R.; Rossetti, T.; Doody, M.; Venter, D.; Pain, S.; Gilshenan, K.; et al. Comparison of human placenta- and bone marrow-derived multipotent mesenchymal stem cells. Stem Cells Dev. 2008, 17, 1095–1107. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Yue, A.; Ruan, Z.; Yin, Y.; Wang, R.; Ren, Y.; Zhu, L. Comparison of biological characteristics of mesenchymal stem cells derived from maternal-origin placenta and Wharton’s jelly. Stem Cell Res. Ther. 2015, 6, 228. [Google Scholar] [CrossRef] [PubMed]
- Cerrada, A.; de la Torre, P.; Grande, J.; Haller, T.; Flores, A.I.; Perez-Gil, J. Human decidua-derived mesenchymal stem cells differentiate into functional alveolar type II-like cells that synthesize and secrete pulmonary surfactant complexes. PLoS ONE 2014, 9, e110195. [Google Scholar] [CrossRef]
- Bornstein, R.; Macias, M.I.; de la Torre, P.; Grande, J.; Flores, A.I. Human decidua-derived mesenchymal stromal cells differentiate into hepatic-like cells and form functional three-dimensional structures. Cytotherapy 2012, 14, 1182–1192. [Google Scholar] [CrossRef]
- Vegh, I.; Grau, M.; Gracia, M.; Grande, J.; de la Torre, P.; Flores, A.I. Decidua mesenchymal stem cells migrated toward mammary tumors in vitro and in vivo affecting tumor growth and tumor development. Cancer Gene Ther. 2013, 20, 8–16. [Google Scholar] [CrossRef]
- Bravo, B.; Gallego, M.I.; Flores, A.I.; Bornstein, R.; Puente-Bedia, A.; Hernandez, J.; de la Torre, P.; Garcia-Zaragoza, E.; Perez-Tavarez, R.; Grande, J.; et al. Restrained Th17 response and myeloid cell infiltration into the central nervous system by human decidua-derived mesenchymal stem cells during experimental autoimmune encephalomyelitis. Stem Cell Res. Ther. 2016, 7, 43. [Google Scholar] [CrossRef]
- Basmaeil, Y.S.; Bahattab, E.; Alshabibi, M.A.; Abomaray, F.M.; Abumaree, M.; Khatlani, T. Human Decidua Basalis mesenchymal stem/stromal cells reverse the damaging effects of high level of glucose on endothelial cells in vitro. J. Cell Mol. Med. 2020. [Google Scholar] [CrossRef]
- Chatterjee, P.; Chiasson, V.L.; Pinzur, L.; Raveh, S.; Abraham, E.; Jones, K.A.; Bounds, K.R.; Ofir, R.; Flaishon, L.; Chajut, A.; et al. Human placenta-derived stromal cells decrease inflammation, placental injury and blood pressure in hypertensive pregnant mice. Clin. Sci. (Lond.) 2016, 130, 513–523. [Google Scholar] [CrossRef]
- Abumaree, M.H.; Abomaray, F.M.; Alshabibi, M.A.; AlAskar, A.S.; Kalionis, B. Immunomodulatory properties of human placental mesenchymal stem/stromal cells. Placenta 2017, 59, 87–95. [Google Scholar] [CrossRef]
- Wassmer, C.H.; Berishvili, E. Immunomodulatory Properties of Amniotic Membrane Derivatives and Their Potential in Regenerative Medicine. Curr. Diabetes Rep. 2020, 20, 31. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.J.; Wang, C.J.; Chang, C.J.; Hu, H.I.; Hsu, P.J.; Wu, Y.C.; Bai, C.H.; Sytwu, H.K.; Yen, B.L. Surface expression of HLA-G is involved in mediating immunomodulatory effects of placenta-derived multipotent cells (PDMCs) towards natural killer lymphocytes. Cell Transplant. 2011, 20, 1721–1730. [Google Scholar] [CrossRef] [PubMed]
- Magatti, M.; Caruso, M.; De Munari, S.; Vertua, E.; De, D.; Manuelpillai, U.; Parolini, O. Human Amniotic Membrane-Derived Mesenchymal and Epithelial Cells Exert Different Effects on Monocyte-Derived Dendritic Cell Differentiation and Function. Cell Transplant. 2015, 24, 1733–1752. [Google Scholar] [CrossRef] [PubMed]
- Broxmeyer, H.E.; Lee, M.R.; Hangoc, G.; Cooper, S.; Prasain, N.; Kim, Y.J.; Mallett, C.; Ye, Z.; Witting, S.; Cornetta, K.; et al. Hematopoietic stem/progenitor cells, generation of induced pluripotent stem cells, and isolation of endothelial progenitors from 21- to 23.5-year cryopreserved cord blood. Blood 2011, 117, 4773–4777. [Google Scholar] [CrossRef] [PubMed]
- Badowski, M.S.; Harris, D.T. Collection, processing, and banking of umbilical cord blood stem cells for transplantation and regenerative medicine. Methods Mol. Biol. 2012, 879, 279–290. [Google Scholar]
- Parent’s Guide to Cord Blood Foundation. Available online: https://parentsguidecordblood.org/en (accessed on 15 November 2020).
- Antoniadou, E.; David, A.L. Placental stem cells. Best Pract. Res. Clin. Obstet. Gynaecol. 2016, 31, 13–29. [Google Scholar] [CrossRef]
- Amniotic Membrane of the Placenta—Part 2. Available online: https://parentsguidecordblood.org/en/news/amniotic-membrane-placenta-part-2 (accessed on 15 November 2020).
- National Health Service Blood and Transplant in UK. Available online: https://www.nhsbt.nhs.uk/what-we-do/transplantation-services/tissue-and-eye-services/tissue-donation/become-a-donor/living-amniotic-membraneplacenta-donation-programme/ (accessed on 15 November 2020).
- Donate Life America in USA. Available online: https://www.donatelife.net/types-of-donation/birth-tissue/ (accessed on 15 November 2020).
- Yoshizawa, R.S. Review: Public perspectives on the utilization of human placentas in scientific research and medicine. Placenta 2013, 34, 9–13. [Google Scholar] [CrossRef]
- Pogozhykh, O.; Prokopyuk, V.; Figueiredo, C.; Pogozhykh, D. Placenta and Placental Derivatives in Regenerative Therapies: Experimental Studies, History, and Prospects. Stem Cells Int. 2018, 2018, 4837930. [Google Scholar] [CrossRef]
- Pogozhykh, D.; Pogozhykh, O.; Prokopyuk, V.; Kuleshova, L.; Goltsev, A.; Blasczyk, R.; Mueller, T. Influence of temperature fluctuations during cryopreservation on vital parameters, differentiation potential, and transgene expression of placental multipotent stromal cells. Stem Cell Res. Ther. 2017, 8, 66. [Google Scholar] [CrossRef]
- Arutyunyan, I.; Fatkhudinov, T.; Sukhikh, G. Umbilical cord tissue cryopreservation: A short review. Stem Cell Res. Ther. 2018, 9, 236. [Google Scholar] [CrossRef]
- De la Torre, P.; Pérez-Lorenzo, M.J.; Flores, A.I. Human Placenta-Derived Mesenchymal Stromal Cells: A Review from Basic Research to Clinical Application. In Stromal Cells—Structure, Function, and Therapeutic Implications; Valarmathi, M.T., Ed.; IntechOpen: Rijeka, Italy, 2018. [Google Scholar]
- Couto, P.S.; Bersenev, A.; Verter, F. The first decade of advanced cell therapy clinical trials using perinatal cells (2005–2015). Regen. Med. 2017, 12, 953–968. [Google Scholar] [CrossRef] [PubMed]
- Ballen, K. Update on umbilical cord blood transplantation. F1000Research 2017, 6, 1556. [Google Scholar] [CrossRef] [PubMed]
- Feng, M.; Lu, A.; Gao, H.; Qian, C.; Zhang, J.; Lin, T.; Zhao, Y. Safety of Allogeneic Umbilical Cord Blood Stem Cells Therapy in Patients with Severe Cerebral Palsy: A Retrospective Study. Stem Cells Int. 2015, 2015, 325652. [Google Scholar] [CrossRef] [PubMed]
- Dawson, G.; Sun, J.M.; Davlantis, K.S.; Murias, M.; Franz, L.; Troy, J.; Simmons, R.; Sabatos-DeVito, M.; Durham, R.; Kurtzberg, J. Autologous Cord Blood Infusions Are Safe and Feasible in Young Children with Autism Spectrum Disorder: Results of a Single-Center Phase I Open-Label Trial. Stem Cells Transl. Med. 2017, 6, 1332–1339. [Google Scholar] [CrossRef]
- Cotten, C.M.; Murtha, A.P.; Goldberg, R.N.; Grotegut, C.A.; Smith, P.B.; Goldstein, R.F.; Fisher, K.A.; Gustafson, K.E.; Waters-Pick, B.; Swamy, G.K.; et al. Feasibility of autologous cord blood cells for infants with hypoxic-ischemic encephalopathy. J. Pediatr. 2014, 164, 973–979.e1. [Google Scholar] [CrossRef]
- Sarin, K.; Chauhan, S.; Bisoi, A.K.; Hazarika, A.; Malhotra, N.; Manek, P. Use of autologous umbilical cord blood transfusion in neonates undergoing surgical correction of congenital cardiac defects: A pilot study. Ann. Card Anaesth. 2018, 21, 270–274. [Google Scholar] [CrossRef]
- Laskowitz, D.T.; Bennett, E.R.; Durham, R.J.; Volpi, J.J.; Wiese, J.R.; Frankel, M.; Shpall, E.; Wilson, J.M.; Troy, J.; Kurtzberg, J. Allogeneic Umbilical Cord Blood Infusion for Adults with Ischemic Stroke: Clinical Outcomes from a Phase I Safety Study. Stem Cells Transl. Med. 2018, 7, 521–529. [Google Scholar] [CrossRef]
- He, B.; Li, X.; Yu, H.; Zhou, Z. Therapeutic potential of umbilical cord blood cells for type 1 diabetes mellitus. J. Diabetes 2015, 7, 762–773. [Google Scholar] [CrossRef]
- Rizk, M.; Aziz, J.; Shorr, R.; Allan, D.S. Cell-Based Therapy Using Umbilical Cord Blood for Novel Indications in Regenerative Therapy and Immune Modulation: An Updated Systematic Scoping Review of the Literature. Biol. Blood Marrow Transplant. 2017, 23, 1607–1613. [Google Scholar] [CrossRef]
- Couto, P.S.; Shatirishvili, G.; Bersenev, A.; Verter, F. First decade of clinical trials and published studies with mesenchymal stromal cells from umbilical cord tissue. RegenMed 2019, 14, 309–319. [Google Scholar] [CrossRef]
- Recruiting Trials: Umbilical Cord Tissue. Available online: https://parentsguidecordblood.org/en/trials-umbilical-cord-tissue (accessed on 15 November 2020).
- Can, A.; Celikkan, F.T.; Cinar, O. Umbilical cord mesenchymal stromal cell transplantations: A systemic analysis of clinical trials. Cytotherapy 2017, 19, 1351–1382. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Tang, F.; Xiao, Z.; Han, G.; Wang, N.; Yin, N.; Chen, B.; Jiang, X.; Yun, C.; Han, W.; et al. Clinical Study of NeuroRegen Scaffold Combined With Human Mesenchymal Stem Cells for the Repair of Chronic Complete Spinal Cord Injury. Cell Transpl. 2017, 26, 891–900. [Google Scholar] [CrossRef] [PubMed]
- Hou, Z.L.; Liu, Y.; Mao, X.H.; Wei, C.Y.; Meng, M.Y.; Liu, Y.H.; Zhuyun Yang, Z.; Zhu, H.; Short, M.; Bernard, C.; et al. Transplantation of umbilical cord and bone marrow-derived mesenchymal stem cells in a patient with relapsing-remitting multiple sclerosis. Cell Adhes. Migr. 2013, 7, 404–407. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Xi, H.; Huang, H.; Zhang, F.; Liu, Y.; Chen, D.; Xiao, J. Multiple cell transplantation based on an intraparenchymal approach for patients with chronic phase stroke. Cell Transplant. 2013, 22 (Suppl. 1), 83–91. [Google Scholar] [CrossRef]
- Jiang, Y.; Zhu, W.; Zhu, J.; Wu, L.; Xu, G.; Liu, X. Feasibility of delivering mesenchymal stem cells via catheter to the proximal end of the lesion artery in patients with stroke in the territory of the middle cerebral artery. Cell Transplant. 2013, 22, 2291–2298. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Cheng, H.; Dai, G.; Wang, X.; Hua, R.; Liu, X.; Wang, P.; Chen, G.; Yue, W.; An, Y. Umbilical cord mesenchymal stem cell transplantation significantly improves neurological function in patients with sequelae of traumatic brain injury. Brain Res. 2013, 1532, 76–84. [Google Scholar] [CrossRef]
- Miao, X.; Wu, X.; Shi, W. Umbilical cord mesenchymal stem cells in neurological disorders: A clinical study. Indian J. Biochem. Biophys. 2015, 52, 140–146. [Google Scholar]
- Lv, Y.T.; Zhang, Y.; Liu, M.; Qiuwaxi, J.N.; Ashwood, P.; Cho, S.C.; Huan, Y.; Ge, R.C.; Chen, X.W.; Wang, Z.J.; et al. Transplantation of human cord blood mononuclear cells and umbilical cord-derived mesenchymal stem cells in autism. J. Transl. Med. 2013, 11, 196. [Google Scholar] [CrossRef]
- Wang, X.; Hu, H.; Hua, R.; Yang, J.; Zheng, P.; Niu, X.; Cheng, H.; Dai, G.; Liu, X.; Zhang, Z.; et al. Effect of umbilical cord mesenchymal stromal cells on motor functions of identical twins with cerebral palsy: Pilot study on the correlation of efficacy and hereditary factors. Cytotherapy 2015, 17, 224–231. [Google Scholar] [CrossRef]
- Xie, B.; Gu, P.; Wang, W.; Dong, C.; Zhang, L.; Zhang, J.; Liu, H.; Qiu, F.; Han, R.; Zhang, Z.; et al. Therapeutic effects of human umbilical cord mesenchymal stem cells transplantation on hypoxic ischemic encephalopathy. Am. J. Transl. Res. 2016, 8, 3241–3250. [Google Scholar]
- He, Y.; Jin, X.; Wang, J.; Meng, M.; Hou, Z.; Tian, W.; Li, Y.; Wang, W.; Wei, Y.; Wang, Y.; et al. Umbilical cord-derived mesenchymal stem cell transplantation for treating elderly vascular dementia. Cell Tissue Bank. 2017, 18, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Bilal, M.; Haseeb, A.; Sher Khan, M.A. Intracoronary infusion of Wharton’s jelly-derived mesenchymal stem cells: A novel treatment in patients of acute myocardial infarction. J. Pak. Med. Assoc. 2015, 65, 1369. [Google Scholar] [PubMed]
- Zhao, X.F.; Xu, Y.; Zhu, Z.Y.; Gao, C.Y.; Shi, Y.N. Clinical observation of umbilical cord mesenchymal stem cell treatment of severe systolic heart failure. Genet. Mol. Res. 2015, 14, 3010–3017. [Google Scholar] [CrossRef]
- Xue, H.L.; Zeng, W.Z.; Wu, X.L.; Jiang, M.D.; Zheng, S.M.; Zhang, Y.; Li, H.Y. Clinical therapeutic effects of human umbilical cord-derived mesenchymal stem cells transplantation in the treatment of end-stage liver disease. Transplant. Proc. 2015, 47, 412–418. [Google Scholar] [CrossRef] [PubMed]
- Shi, M.; Zhang, Z.; Xu, R.; Lin, H.; Fu, J.; Zou, Z.; Zhang, A.; Shi, J.; Chen, L.; Lv, S.; et al. Human mesenchymal stem cell transfusion is safe and improves liver function in acute-on-chronic liver failure patients. Stem Cells Transl. Med. 2012, 1, 725–731. [Google Scholar] [CrossRef]
- Shi, M.; Liu, Z.; Wang, Y.; Xu, R.; Sun, Y.; Zhang, M.; Yu, X.; Wang, H.; Meng, L.; Su, H.; et al. A Pilot Study of Mesenchymal Stem Cell Therapy for Acute Liver Allograft Rejection. Stem Cells Transl. Med. 2017, 6, 2053–2061. [Google Scholar] [CrossRef]
- Gao, L.; Zhang, Y.; Hu, B.; Liu, J.; Kong, P.; Lou, S.; Su, Y.; Yang, T.; Li, H.; Liu, Y.; et al. Phase II Multicenter, Randomized, Double-Blind Controlled Study of Efficacy and Safety of Umbilical Cord-Derived Mesenchymal Stromal Cells in the Prophylaxis of Chronic Graft-Versus-Host Disease After HLA-Haploidentical Stem-Cell Transplantation. J. Clin. Oncol. 2016, 34, 2843–2850. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, Z.; Cao, Y.; Xu, L.; Li, X.; Liu, P.; Yan, P.; Liu, Z.; Zhao, D.; Wang, J.; et al. Cotransplantation of haploidentical hematopoietic and umbilical cord mesenchymal stem cells with a myeloablative regimen for refractory/relapsed hematologic malignancy. Ann. Hematol. 2013, 92, 1675–1684. [Google Scholar] [CrossRef]
- Zhu, L.; Wang, Z.; Zheng, X.; Ding, L.; Han, D.; Yan, H.; Guo, Z.; Wang, H. Haploidentical hematopoietic stem cell transplant with umbilical cord-derived multipotent mesenchymal cell infusion for the treatment of high-risk acute leukemia in children. Leuk Lymphoma 2015, 56, 1346–1352. [Google Scholar] [CrossRef]
- Wang, H.; Wang, Z.; Zheng, X.; Ding, L.; Zhu, L.; Yan, H.; Guo, Z. Hematopoietic stem cell transplantation with umbilical cord multipotent stromal cell infusion for the treatment of aplastic anemia--a single-center experience. Cytotherapy 2013, 15, 1118–1125. [Google Scholar] [CrossRef]
- Wang, X.; Yin, X.; Sun, W.; Bai, J.; Shen, Y.; Ao, Q.; Gu, Y.; Liu, Y. Intravenous infusion umbilical cord-derived mesenchymal stem cell in primary immune thrombocytopenia: A two-year follow-up. Exp. Ther. Med. 2017, 13, 2255–2258. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wang, L.; Cong, X.; Liu, G.; Zhou, J.; Bai, B.; Li, Y.; Bai, W.; Li, M.; Ji, H.; et al. Human umbilical cord mesenchymal stem cell therapy for patients with active rheumatoid arthritis: Safety and efficacy. Stem Cells Dev. 2013, 22, 3192–3202. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Zhao, G.; Zhang, L.; Qiao, C.; Di, A.; Gao, H.; Xu, H. Safety and therapeutic effect of mesenchymal stem cell infusion on moderate to severe ulcerative colitis. Exp. Ther. Med. 2016, 12, 2983–2989. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Li, J.; Zhang, Y.; Zhang, M.; Chen, J.; Li, X.; Hu, X.; Jiang, S.; Shi, S.; Sun, L. Umbilical cord mesenchymal stem cell transplantation in active and refractory systemic lupus erythematosus: A multicenter clinical study. Arthritis Res. Ther. 2014, 16, R79. [Google Scholar] [CrossRef]
- Zhang, Z.; Fu, J.; Xu, X.; Wang, S.; Xu, R.; Zhao, M.; Nie, W.; Wang, X.; Zhang, J.; Li, T.; et al. Safety and immunological responses to human mesenchymal stem cell therapy in difficult-to-treat HIV-1-infected patients. AIDS 2013, 27, 1283–1293. [Google Scholar] [CrossRef]
- Zhang, C.; Yin, X.; Zhang, J.; Ao, Q.; Gu, Y.; Liu, Y. Clinical observation of umbilical cord mesenchymal stem cell treatment of severe idiopathic pulmonary fibrosis: A case report. Exp. Ther. Med. 2017, 13, 1922–1926. [Google Scholar] [CrossRef]
- Nguyen, L.T.; Trieu, T.T.H.; Bui, H.T.H.; Hoang, V.T.; Nguyen, A.T.T.; Trinh, N.T.H.; Nguyen, K.T.; Hoang, D.M. Allogeneic administration of human umbilical cord-derived mesenchymal stem/stromal cells for bronchopulmonary dysplasia: Preliminary outcomes in four Vietnamese infants. J. Transl. Med. 2020, 18, 398. [Google Scholar] [CrossRef]
- Liu, X.; Zheng, P.; Wang, X.; Dai, G.; Cheng, H.; Zhang, Z.; Hua, R.; Niu, X.; Shi, J.; An, Y. A preliminary evaluation of efficacy and safety of Wharton’s jelly mesenchymal stem cell transplantation in patients with type 2 diabetes mellitus. Stem Cell Res. Ther. 2014, 5, 57. [Google Scholar] [CrossRef]
- Hu, J.; Yu, X.; Wang, Z.; Wang, F.; Wang, L.; Gao, H.; Chen, Y.; Zhao, W.; Jia, Z.; Yan, S.; et al. Long term effects of the implantation of Wharton’s jelly-derived mesenchymal stem cells from the umbilical cord for newly-onset type 1 diabetes mellitus. Endocr. J. 2013, 60, 347–357. [Google Scholar] [CrossRef]
- Özmert, E.; Arslan, U. Management of retinitis pigmentosa by Wharton’s jelly-derived mesenchymal stem cells: Prospective analysis of 1-year results. Stem Cell Res. Ther. 2020, 11, 353. [Google Scholar] [CrossRef]
- Li, P.; Cui, K.; Zhang, B.; Wang, Z.; Shen, Y.; Wang, X.; Zhang, J.; Tong, F.; Li, S. Transplantation of human umbilical cord-derived mesenchymal stems cells for the treatment of Becker muscular dystrophy in affected pedigree members. Int. J. Mol. Med. 2015, 35, 1051–1057. [Google Scholar] [CrossRef] [PubMed]
- Qu, Z.; Fang, G.; Cui, Z.; Liu, Y. Cell therapy for bone nonunion: A retrospective study. Minerva Med. 2015, 106, 315–321. [Google Scholar] [PubMed]
- Dilogo, I.H.; Primaputra, M.R.A.; Pawitan, J.A.; Liem, I.K. Modified Masquelet technique using allogeneic umbilical cord-derived mesenchymal stem cells for infected non-union femoral shaft fracture with a 12 cm bone defect: A case report. Int. J. Surg. Case Rep. 2017, 34, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Rajput, B.S.; Chakrabarti, S.K.; Dongare, V.S.; Ramirez, C.M.; Deb, K.D. Human Umbilical Cord Mesenchymal Stem Cells in the Treatment of Duchenne Muscular Dystrophy: Safety and Feasibility Study in India. J. Stem Cells 2015, 10, 141–156. [Google Scholar] [PubMed]
- Chen, C.; Qu, Z.; Yin, X.; Shang, C.; Ao, Q.; Gu, Y.; Liu, Y. Efficacy of umbilical cord-derived mesenchymal stem cell-based therapy for osteonecrosis of the femoral head: A three-year follow-up study. Mol. Med. Rep. 2016, 14, 4209–4215. [Google Scholar] [CrossRef] [PubMed]
- Song, J.S.; Hong, K.T.; Kim, N.M.; Jung, J.Y.; Park, H.S.; Lee, S.H.; Cho, Y.J.; Kim, S.J. Implantation of allogenic umbilical cord blood-derived mesenchymal stem cells improves knee osteoarthritis outcomes: Two-year follow-up. Regen. Ther. 2020, 14, 32–39. [Google Scholar] [CrossRef]
- Chen, H.; Niu, J.W.; Ning, H.M.; Pan, X.; Li, X.B.; Li, Y.; Wang, D.H.; Hu, L.D.; Sheng, H.X.; Xu, M.; et al. Treatment of Psoriasis with Mesenchymal Stem Cells. Am. J. Med. 2016, 129, e13–e14. [Google Scholar] [CrossRef]
- Fan, D.; Wu, S.; Ye, S.; Wang, W.; Guo, X.; Liu, Z. Umbilical cord mesenchyme stem cell local intramuscular injection for treatment of uterine niche: Protocol for a prospective, randomized, double-blinded, placebo-controlled clinical trial. Medicine (Baltimore) 2017, 96, e8480. [Google Scholar] [CrossRef]
- Gupta, A.; Madhavan, M.V.; Sehgal, K.; Nair, N.; Mahajan, S.; Sehrawat, T.S.; Bikdeli, B.; Ahluwalia, N.; Ausiello, J.C.; Wan, E.Y.; et al. Extrapulmonary manifestations of COVID-19. Nat. Med. 2020, 26, 1017–1032. [Google Scholar] [CrossRef]
- Berishvili, E.; Kaiser, L.; Cohen, M.; Berney, T.; Scholz, H.; Floisand, Y.; Mattsson, J. Treatment of COVID-19 Pneumonia: The Case for Placenta-derived Cell Therapy. Stem Cell Rev. Rep. 2020. [Google Scholar] [CrossRef]
- Merad, M.; Martin, J.C. Pathological inflammation in patients with COVID-19: A key role for monocytes and macrophages. Nat. Rev. Immunol. 2020, 20, 355–362. [Google Scholar] [CrossRef]
- Theoharides, T.C.; Conti, P. Dexamethasone for COVID-19? Not so fast. J. Biol. Regul. Homeost Agents 2020, 34, 1241–1243. [Google Scholar]
- Della-Torre, E.; Campochiaro, C.; Cavalli, G.; De Luca, G.; Napolitano, A.; La Marca, S.; Boffini, N.; Da Prat, V.; Di Terlizzi, G.; Lanzillotta, M.; et al. Interleukin-6 blockade with sarilumab in severe COVID-19 pneumonia with systemic hyperinflammation: An open-label cohort study. Ann. Rheum. Dis. 2020, 79, 1277–1285. [Google Scholar] [CrossRef]
- Yadav, P.; Vats, R.; Bano, A.; Bhardwaj, R. Mesenchymal stem cell immunomodulation and regeneration therapeutics as an ameliorative approach for COVID-19 pandemics. Life Sci. 2020, 263, 118588. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.W.; Gupta, N.; Serikov, V.; Matthay, M.A. Potential application of mesenchymal stem cells in acute lung injury. Expert Opin. Biol. Ther. 2009, 9, 1259–1270. [Google Scholar] [CrossRef] [PubMed]
- Herold, S.; Mayer, K.; Lohmeyer, J. Acute lung injury: How macrophages orchestrate resolution of inflammation and tissue repair. Front. Immunol. 2011, 2, 65. [Google Scholar] [CrossRef] [PubMed]
- Abumaree, M.H.; Al Jumah, M.A.; Kalionis, B.; Jawdat, D.; Al Khaldi, A.; Abomaray, F.M.; Fatani, A.S.; Chamley, L.W.; Knawy, B.A. Human placental mesenchymal stem cells (pMSCs) play a role as immune suppressive cells by shifting macrophage differentiation from inflammatory M1 to anti-inflammatory M2 macrophages. Stem Cell Rev. Rep. 2013, 9, 620–641. [Google Scholar] [CrossRef]
- Shetty, A.K. Mesenchymal Stem Cell Infusion Shows Promise for Combating Coronavirus (COVID-19)-Induced Pneumonia. Aging Dis. 2020, 11, 462–464. [Google Scholar] [CrossRef]
- Muraca, M.; Pessina, A.; Pozzobon, M.; Dominici, M.; Galderisi, U.; Lazzari, L.; Parolini, O.; Lucarelli, E.; Perilongo, G.; Baraldi, E. Mesenchymal stromal cells and their secreted extracellular vesicles as therapeutic tools for COVID-19 pneumonia? J. Control Release 2020, 325, 135–140. [Google Scholar] [CrossRef]
- Le Thi Bich, P.; Nguyen Thi, H.; Dang Ngo Chau, H.; Phan Van, T.; Do, Q.; Dong Khac, H.; Le Van, D.; Nguyen Huy, L.; Mai Cong, K.; Ta Ba, T.; et al. Allogeneic umbilical cord-derived mesenchymal stem cell transplantation for treating chronic obstructive pulmonary disease: A pilot clinical study. Stem Cell Res. Ther. 2020, 11, 60. [Google Scholar] [CrossRef]
- Meng, F.; Xu, R.; Wang, S.; Xu, Z.; Zhang, C.; Li, Y.; Yang, T.; Shi, L.; Fu, J.; Jiang, T.; et al. Human umbilical cord-derived mesenchymal stem cell therapy in patients with COVID-19: A phase 1 clinical trial. Signal Transduct. Target. Ther. 2020, 5, 172. [Google Scholar] [CrossRef]
- Shu, L.; Niu, C.; Li, R.; Huang, T.; Wang, Y.; Huang, M.; Ji, N.; Zheng, Y.; Chen, X.; Shi, L.; et al. Treatment of severe COVID-19 with human umbilical cord mesenchymal stem cells. Stem Cell Res. Ther. 2020, 11, 361. [Google Scholar] [CrossRef] [PubMed]
- De la Torre, P.; Flores, A.I. Nanotechnology and Mesenchymal Stem Cells for Regenerative Medicine. Glob. J. Nanomed. 2017, 1, 555559. [Google Scholar] [CrossRef]
- Yi, D.K.; Nanda, S.S.; Kim, K.; Tamil Selvan, S. Recent progress in nanotechnology for stem cell differentiation, labeling, tracking and therapy. J. Mater. Chem. B 2017, 5, 9429–9451. [Google Scholar] [CrossRef] [PubMed]
- De la Torre, P.; Perez-Lorenzo, M.J.; Alcazar-Garrido, A.; Flores, A.I. Cell-Based Nanoparticles Delivery Systems for Targeted Cancer Therapy: Lessons from Anti-Angiogenesis Treatments. Molecules 2020, 25, 715. [Google Scholar] [CrossRef] [PubMed]
- Orza, A.; Soritau, O.; Olenic, L.; Diudea, M.; Florea, A.; Rus Ciuca, D.; Mihu, C.; Casciano, D.; Biris, A.S. Electrically conductive gold-coated collagen nanofibers for placental-derived mesenchymal stem cells enhanced differentiation and proliferation. ACS Nano 2011, 5, 4490–4503. [Google Scholar] [CrossRef]
- Kim, T.; Sridharan, I.; Zhu, B.; Orgel, J.; Wang, R. Effect of CNT on collagen fiber structure, stiffness assembly kinetics and stem cell differentiation. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 49, 281–289. [Google Scholar] [CrossRef]
- Muduli, S.; Lee, H.H.; Yang, J.S.; Chen, T.Y.; Higuchi, A.; Kumar, S.S.; Alarfaj, A.A.; Munusamy, M.A.; Benelli, G.; Murugan, K.; et al. Proliferation and osteogenic differentiation of amniotic fluid-derived stem cells. J. Mater. Chem. B 2017, 5, 5345–5354. [Google Scholar] [CrossRef]
- Kargozar, S.; Mozafari, M.; Hashemian, S.J.; Brouki Milan, P.; Hamzehlou, S.; Soleimani, M.; Joghataei, M.T.; Gholipourmalekabadi, M.; Korourian, A.; Mousavizadeh, K.; et al. Osteogenic potential of stem cells-seeded bioactive nanocomposite scaffolds: A comparative study between human mesenchymal stem cells derived from bone, umbilical cord Wharton’s jelly, and adipose tissue. J. Biomed. Mater. Res. Part B Appl. Biomater. 2018, 106, 61–72. [Google Scholar] [CrossRef]
- Paris, J.L.; de la Torre, P.; Manzano, M.; Cabanas, M.V.; Flores, A.I.; Vallet-Regi, M. Decidua-derived mesenchymal stem cells as carriers of mesoporous silica nanoparticles. In vitro and in vivo evaluation on mammary tumors. Acta Biomater. 2016, 33, 275–282. [Google Scholar] [CrossRef]
- Paris, J.L.; de la Torre, P.; Victoria Cabanas, M.; Manzano, M.; Grau, M.; Flores, A.I.; Vallet-Regi, M. Vectorization of ultrasound-responsive nanoparticles in placental mesenchymal stem cells for cancer therapy. Nanoscale 2017, 9, 5528–5537. [Google Scholar] [CrossRef] [PubMed]
- Paris, J.L.; de la Torre, P.; Cabanas, M.V.; Manzano, M.; Flores, A.I.; Vallet-Regi, M. Suicide-gene transfection of tumor-tropic placental stem cells employing ultrasound-responsive nanoparticles. Acta Biomater. 2019, 83, 372–378. [Google Scholar] [CrossRef] [PubMed]
- Bonomi, A.; Silini, A.; Vertua, E.; Signoroni, P.B.; Cocce, V.; Cavicchini, L.; Sisto, F.; Alessandri, G.; Pessina, A.; Parolini, O. Human amniotic mesenchymal stromal cells (hAMSCs) as potential vehicles for drug delivery in cancer therapy: An in vitro study. Stem Cell Res. Ther. 2015, 6, 155. [Google Scholar] [CrossRef] [PubMed]
- Su, L.J.; Wu, M.S.; Hui, Y.Y.; Chang, B.M.; Pan, L.; Hsu, P.C.; Chen, Y.T.; Ho, H.N.; Huang, Y.H.; Ling, T.Y.; et al. Fluorescent nanodiamonds enable quantitative tracking of human mesenchymal stem cells in miniature pigs. Sci. Rep. 2017, 7, 45607. [Google Scholar] [CrossRef]
- Hsu, F.T.; Wei, Z.H.; Hsuan, Y.C.; Lin, W.; Su, Y.C.; Liao, C.H.; Hsieh, C.L. MRI tracking of polyethylene glycol-coated superparamagnetic iron oxide-labelled placenta-derived mesenchymal stem cells toward glioblastoma stem-like cells in a mouse model. Artif. Cells Nanomed. Biotechnol. 2018, 46 (Suppl. 3), S448–S459. [Google Scholar] [CrossRef]
- Dimmeler, S.; Ding, S.; Rando, T.A.; Trounson, A. Translational strategies and challenges in regenerative medicine. Nat. Med. 2014, 20, 814–821. [Google Scholar] [CrossRef]
- Pittenger, M.F.; Discher, D.E.; Peault, B.M.; Phinney, D.G.; Hare, J.M.; Caplan, A.I. Mesenchymal stem cell perspective: Cell biology to clinical progress. NPJ Regen. Med. 2019, 4, 22. [Google Scholar] [CrossRef]
| Public Banks | Family Banks | |
|---|---|---|
| America | 44 | 103 |
| Europe | 69 | 138 |
| Asia | 38 | 119 |
| Africa | 0 | 17 |
| Oceania | 3 | 2 |
| Total number of banks | 154 | 379 |
| Disorders | Disease Treated | Reference |
|---|---|---|
| Neurologic | Spinal cord injury | [149] |
| Multiple Sclerosis | [150] | |
| Stroke | [151,152] | |
| Traumatic brain injury | [153] | |
| Amyotrophic lateral sclerosis | [154] | |
| Autism | [155] | |
| Cerebral palsy | [156] | |
| Hypoxic ischemic encephalopathy | [157] | |
| Vascular dementia | [158] | |
| Cardiovascular | Acute myocardial infarction | [159] |
| Systolic heart failure | [160] | |
| Hepatic | Liver failure | [161,162] |
| Transplant rejection | [163] | |
| Hematologic | Graft versus host disease (acute, chronic) | [164,165] |
| Leukemia | [166] | |
| Aplastic anemia | [167] | |
| Thrombocytopenia | [168] | |
| Immunologic | Rheumatoid arthritis | [169] |
| Ulcerative colitis | [170] | |
| Systemic lupus erythematosus | [171] | |
| HIV infection | [172] | |
| Pulmonary | Severe idiopathic pulmonary fibrosis | [173] |
| Bronchopulmonary dysplasia | [174] | |
| Endocrine | Diabetes (Type I, Type II) | [175] |
| Diabetic foot ulcer | [176] | |
| Ophthalmologic | Retinitis pigmentosa | [177] |
| Musculoskeletal disorders | Becker muscular dystrophy | [178] |
| Bone nonunion (fractured, infected) | [179,180] | |
| Duchenne muscular dystrophy | [181] | |
| Osteonecrosis of femoral head | [182] | |
| Cartilage regeneration | [183] | |
| Dermatologic | Psoriasis vulgaris | [184] |
| Cesarean scar defect | [185] |
| NCT Study | Status/No. Patients | Treatment | Study Type | Start Date | Location |
|---|---|---|---|---|---|
| NCT04366271 | Recruiting/106 | CT: UC-MSC Control: Standard care | Phase 2 | 7 May 2020 | Spain |
| NCT04273646 | Not yet recruiting/48 | CT: UC-MSC Control: Placebo | N/A | 20 April 2020 | China |
| NCT04288102 | Completed/100 | CT: UC-MSC CG: Saline + HSA | Phase 1/2 | 5 March 2020 | China |
| NCT04333368 | Recruiting/40 | CT: WJ-MSC Control: Saline | Phase 1/2 | 6 April 2020 | France |
| NCT04494386 | Recruiting/60 | CT: UC-MSC Control: Placebo | Phase 1/2 | 23 July 2020 | United States |
| NCT04490486 | Not yet recruiting/21 | CT: UC-MSC Control: Placebo | Phase 1 | 1 March 2021 | United States |
| NCT04457609 | Recruiting/40 | CT: Standard care + UC-MSC Control: Standard care | Phase 1 | 7 July, 2020 | Indonesia |
| NCT04355728 | Active, not recruiting/24 | CT: UC-MSC Control: Vehicle + Heparin | Phase 1/2 | 25 April 2020 | United States |
| NCT04461925 | Recruiting/40 | CT: Standard care + P-MSC or UC-MSC Control: Standard care | Phase 1/2 | 2 May 2020 | Ukraine |
| NCT04429763 | Not yet recruiting/30 | CT: UC-MSC Control: Placebo | Phase 2 | July 2020 | Colombia |
| NCT04293692 | Withdrawn | CT: UC-MSC Control: Placebo | N/A | 24 February 2020 | China |
| NCT04452097 | Not yet recruiting/9 | CT: UC-MSC + Standard care | Phase 1 | 1 December 2020 | United States |
| NCT03042143 | Recruiting/75 | CT: UC-MSC Control: Placebo | Phase 1/2 | 7 January 2019 | United Kingdom |
| NCT04456361 | Active, not recruiting/9 | CT: WJ-MSC | Early Phase 1 | 16 April 2020 | Mexico |
| NCT04269525 | Recruiting/16 | CT: UC-MSC | Phase 2 | 6 February 2020 | China |
| NCT04565665 | Recruiting/70 | CT: UCB-MSC | Phase 1 | 29 July 2020 | United States |
| NCT04437823 | Recruiting/20 | CT: UC-MSC + standard care Control: standard care | Phase 2 | 1 June 2020 | Pakistan |
| NCT04371601 | Active, not recruiting/60 | CT: UC-MSC + standard care Control: Standard care | Early Phase 1 | 1 March 2020 | China |
| NCT04573270 | Completed/40 | CT: UC-MSC Control: Placebo | Phase 1 | 24 April 2020 | United States |
| NCT04313322 | Recruiting/5 | CT: WJ-MSC | Phase 1 | 16 March 2020 | Jordan |
| NCT04390152 | Not yet recruiting/40 | CT: UC-MSC + standard care Control: Standard care | Phase 1/2 | September 2020 | Colombia |
| NCT04390139 | Recruiting/30 | CT: WJ-MSC Control: Placebo | Phase 1/2 | 13 May 2020 | Spain |
| NCT04339660 | Recruiting/30 | CT: WJ-MSC Control: Saline | Phase 1/2 | 9 April 2020 | China |
| NCT04389450 | Recruiting/140 | CT: PLX-PAD Control: Saline | Phase 2 | 1 October 2020 | United States |
| NCT04451291 | Not yet recruiting/20 | CT: DSC | N/A | 24 August 2020 | Canada |
| NCT04614025 | Recruiting/40 | CT: PLX-PAD + standard care Control: Standard care | Phase 2 | 3 November 2020 | Germany/ Israel |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Torre, P.d.l.; Flores, A.I. Current Status and Future Prospects of Perinatal Stem Cells. Genes 2021, 12, 6. https://doi.org/10.3390/genes12010006
Torre Pdl, Flores AI. Current Status and Future Prospects of Perinatal Stem Cells. Genes. 2021; 12(1):6. https://doi.org/10.3390/genes12010006
Chicago/Turabian StyleTorre, Paz de la, and Ana I. Flores. 2021. "Current Status and Future Prospects of Perinatal Stem Cells" Genes 12, no. 1: 6. https://doi.org/10.3390/genes12010006
APA StyleTorre, P. d. l., & Flores, A. I. (2021). Current Status and Future Prospects of Perinatal Stem Cells. Genes, 12(1), 6. https://doi.org/10.3390/genes12010006






