The Potential Role of Activating the ATP-Sensitive Potassium Channel in the Treatment of Hyperphagic Obesity
Abstract
1. Introduction
2. Methods
3. Results
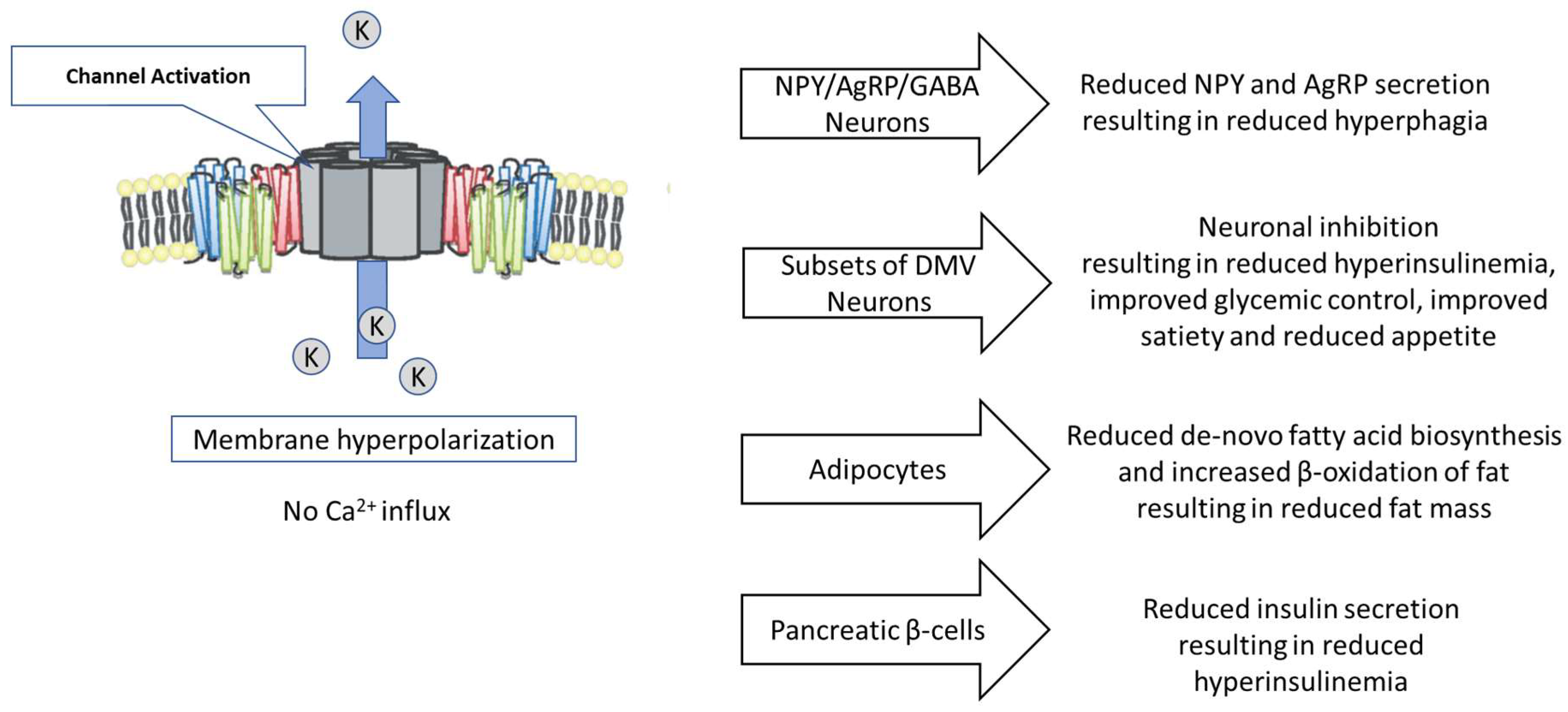
3.1. The Structure of and Genes Encoding the KATP Channel
3.2. The Role of the KATP Channel in the Central Regulation of Appetite by the Arcuate Nucleus
3.3. The Role of the KATP Channel in the Regulation of Neuronal Function in the Dorsal Motor Nucleus of the Vagus
3.4. The Role of The KATP Channel in the Regulation of Adipocyte Metabolism and Fat Mass
3.5. The Role of the KATP Channel in Reducing Hyperinsulinemia
3.6. Insulin Resistance and Hyperinsulinemia
3.7. The Use of KATP Channel Activators in Animal Models of Hyperphagic Obesity
3.7.1. Magel2 Knockout Mice—A Model of Prader–Willi Syndrome
3.7.2. Zucker Fatty Rat—A Model of LepR Deficiency
3.7.3. Zucker Diabetic Fatty Rat—A Model of LepR Deficiency
3.7.4. db/db Mouse—A Model of LepR Deficiency
3.7.5. Otsuka Long Evans Tokushima Fatty Rat—A Model of CCK1 Receptor Deficiency
3.7.6. Hypothalamic Injured Rat—A Model of Hyperinsulinemia Driven Hyperphagic Obesity
3.7.7. Hypothalamic Injured Chicken—A Model of Hyperinsulinemia Driven Hyperphagic Obesity
3.7.8. Streptozotocin Induced Diabetic Rat
3.7.9. High-Fat Diet-Induced Obese Mouse
3.7.10. Tabular Summary of Animal Model Results
3.8. Experience with the Diazoxide Choline Controlled-Release Tablet (DCCR) in Prader–Willi Syndrome
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dubern, B.; Clément, K. Leptin and leptin receptor-related monogenic obesity. Biochimie 2012, 94, 2111–2115. [Google Scholar] [CrossRef]
- Butler, M.G.; Miller, J.L.; Forster, J.L. Prader-Willi Syndrome—Clinical Genetics, Diagnosis and Treatment Approaches: An Update. Curr. Pediatr. Rev. 2019, 15, 207–244. [Google Scholar] [CrossRef]
- Lustig, R.H. Hypothalamic obesity: Causes, consequences, treatment. Pediatr. Endocrinol. Rev. 2008, 6, 220–227. [Google Scholar]
- Foster, M.N.; Coetzee, W. KATP Channels in the Cardiovascular System. Physiol. Rev. 2016, 96, 177–252. [Google Scholar] [CrossRef]
- Baquero, A.F.; De Solis, A.J.; Lindsley, S.R.; Kirigiti, M.A.; Smith, M.S.; Cowley, M.; Zeltser, L.; Grove, K.L. Developmental switch of leptin signaling in arcuate nucleus neurons. J. Neurosci. 2014, 34, 9982–9994. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Moustaid-Moussa, N.; Wilkison, W.O.; Zemel, M. Role of the sulfonylurea receptor in regulating human adipocyte metabolism. FASEB J. 1999, 13, 1833–1838. [Google Scholar] [CrossRef] [PubMed]
- Krashes, M.J.; Shah, B.P.; Koda, S.; Lowell, B.B. Rapid versus delayed stimulation of feeding by the endogenously released AgRP neuron mediators GABA, NPY, and AgRP. Cell Metab. 2013, 18, 588–595. [Google Scholar] [CrossRef] [PubMed]
- Cansell, C.; Denis, R.; Joly-Amado, A.; Castel, J.; Luquet, S.H. Arcuate AgRP neurons and the regulation of energy balance. Front. Endocrinol. 2012, 3. [Google Scholar] [CrossRef] [PubMed]
- Cyr, N.E.; Toorie, A.M.; Steger, J.S.; Sochat, M.M.; Hyner, S.; Perelló, M.; Stuart, R.; Nillni, E.A. Mechanisms by which the orexigen NPY regulates anorexigenic α-MSH and TRH. Am. J. Physiol. Metab. 2013, 304, E640–E650. [Google Scholar] [CrossRef]
- Bouret, S.G. Organizational actions of metabolic hormones. Front. Neuroendocr. 2013, 34, 18–26. [Google Scholar] [CrossRef]
- Beck, B. Neuropeptide Y in normal eating and in genetic and dietary-induced obesity. Philos. Trans. R. Soc. B Boil. Sci. 2006, 361, 1159–1185. [Google Scholar] [CrossRef] [PubMed]
- Luquet, S.H.; Perez, F.A.; Hnasko, T.S.; Palmiter, R.D. NPY/AgRP Neurons Are Essential for Feeding in Adult Mice but Can Be Ablated in Neonates. Science 2005, 310, 683–685. [Google Scholar] [CrossRef] [PubMed]
- Baver, S.B.; Hope, K.; Guyot, S.; Bjørbaek, C.; Kaczorowski, C.; O’Connell, K.M.S. Leptin modulates the intrinsic excitability of AgRP/NPY neurons in the arcuate nucleus of the hypothalamus. J. Neurosci. 2014, 34, 5486–5496. [Google Scholar] [CrossRef]
- Spanswick, D.; Smith, M.A.; Groppi, V.E.; Logan, S.D.; Ashford, M. Leptin inhibits hypothalamic neurons by activation of ATP-sensitive potassium channels. Nature 1997, 390, 521–525. [Google Scholar] [CrossRef] [PubMed]
- Top, M.V.D.; Lee, K.; Whyment, A.D.; Blanks, A.M.; Spanswick, D.C. Orexigen-sensitive NPY/AgRP pacemaker neurons in the hypothalamic arcuate nucleus. Nat. Neurosci. 2004, 7, 493–494. [Google Scholar] [CrossRef] [PubMed]
- Ramamoorthy, P.; Wang, Q.; Whim, M.D. Cell Type-Dependent Trafficking of Neuropeptide Y-Containing Dense Core Granules in CNS Neurons. J. Neurosci. 2011, 31, 14783–14788. [Google Scholar] [CrossRef]
- Stricker-Krongrad, A.; Barbanel, G.; Beck, B.; Burlet, A.; Nicolas, J.; Burlet, C. K+-stimulated neuropeptide Y release into the paraventricular nucleus and relation to feeding behavior in free-moving rats. Neuropeptides 1993, 24, 307–312. [Google Scholar] [CrossRef]
- Top, M.V.D.; Lyons, D.; Lee, K.; Coderre, E.; Renaud, L.; Spanswick, D.C. Pharmacological and molecular characterization of ATP-sensitive K+ conductances in CART and NPY/AgRP expressing neurons of the hypothalamic arcuate nucleus. Neuroscience 2007, 144, 815–824. [Google Scholar] [CrossRef]
- Laughton, W.B.; Powley, T.L. Localization of efferent function in the dorsal motor nucleus of the vagus. Am. J. Physiol. Integr. Comp. Physiol. 1987, 252, R13–R25. [Google Scholar] [CrossRef]
- Berthoud, H.-R. The vagus nerve, food intake and obesity. Regul. Pept. 2008, 149, 15–25. [Google Scholar] [CrossRef]
- Saper, C.B.; Loewy, A.; Swanson, L.; Cowan, W. Direct hypothalamo-autonomic connections. Brain Res. 1976, 117, 305–312. [Google Scholar] [CrossRef]
- Swanson, L.W.; Sawchenko, P.E. Parventricular nucleus: A site for the integration of neuroendocrine and autonomic mechanisms. Neuroendocrinology 1980, 31, 410–417. [Google Scholar] [CrossRef] [PubMed]
- Zsombok, A.; Smith, B. Plasticity of central autonomic neural circuits in diabetes. Biochim. Biophys. Acta 2008, 1792, 423–431. [Google Scholar] [CrossRef] [PubMed]
- Berglund, E.D.; Liu, T.; Kong, X.; Sohn, J.-W.; Vong, L.; Deng, Z.; Lee, C.E.; Lee, S.; Williams, K.W.; Olson, D.P.; et al. Melanocortin 4 receptors in autonomic neurons regulate thermogenesis and glycemia. Nat. Neurosci. 2014, 17, 911–913. [Google Scholar] [CrossRef] [PubMed]
- Sohn, J.-W.; Harris, L.E.; Berglund, E.D.; Liu, T.; Vong, L.; Lowell, B.B.; Balthasar, N.; Williams, K.W.; Elmquist, J.K. Melanocortin 4 receptors reciprocally regulate sympathetic and parasympathetic – preganglionic neurons. Cell 2013, 152, 612–619. [Google Scholar] [CrossRef]
- Hjelland, I.; Oveland, N.P.; Leversen, K.; Berstad, A.; Hausken, T. Insulin-Induced Hypoglycemia Stimulates Gastric Vagal Activity and Motor Function without Increasing Cardiac Vagal Activity. Digestion 2005, 72, 43–48. [Google Scholar] [CrossRef]
- Maeda, C.Y.; Fernandes, T.G.; Lulhier, F.; Irigoyen, M.C.C. Streptozotocin diabetes modifies arterial pressure and baroreflex sensitivity in rats. Braz. J. Med. Biol. Res. 1995, 28, 497–501. [Google Scholar]
- Plum, L.; Schubert, M.; Brüning, J.C. The role of insulin receptor signaling in the brain. Trends Endocrinol. Metab. 2005, 16, 59–65. [Google Scholar] [CrossRef]
- Plum, L.; Belgardt, B.F.; Brüning, J.C. Central insulin action in energy and glucose homeostasis. J. Clin. Investig. 2006, 116, 1761–1766. [Google Scholar] [CrossRef]
- Lam, T.K.T.; Pocai, A.; Gutierrez-Juarez, R.; Obici, S.; Bryan, J.; Aguilar-Bryan, L.; Schwartz, G.J.; Rossetti, L. Hypothalamic sensing of circulating fatty acids is required for glucose homeostasis. Nat. Med. 2005, 11, 320–327. [Google Scholar] [CrossRef]
- Obici, S.; Feng, Z.; Karkanias, G.; Baskin, D.G.; Rossetti, L. Decreasing hypothalamic insulin receptors causes hyperphagia and insulin resistance in rats. Nat. Neurosci. 2002, 5, 566–572. [Google Scholar] [CrossRef] [PubMed]
- Obici, S.; Zhang, B.B.; Karkanias, G.; Rossetti, L. Hypothalamic insulin signaling is required for inhibition of glucose production. Nat. Med. 2002, 8, 1376–1382. [Google Scholar] [CrossRef]
- Huang, H.-N.; Lu, P.-J.; Lo, W.-C.; Lin, C.-H.; Hsiao, M.; Tseng, C.J. In Situ Akt Phosphorylation in the Nucleus Tractus Solitarii Is Involved in Central Control of Blood Pressure and Heart Rate. Circulation 2004, 110, 2476–2483. [Google Scholar] [CrossRef] [PubMed]
- Krowicki, Z.; Nathan, N.A.; Hornby, P.J. Gastric motor and cardiovascular effects of insulin in dorsal vagal complex of the rat. Am. J. Physiol. Content 1998, 275, G964–G972. [Google Scholar] [CrossRef] [PubMed]
- Blake, C.B.; Smith, B. Insulin reduces excitation in gastric-related neurons of the dorsal motor nucleus of the vagus. Am. J. Physiol. Integr. Comp. Physiol. 2012, 303, R807–R814. [Google Scholar] [CrossRef] [PubMed]
- Pocai, A.; Lam, T.K.T.; Gutierrez-Juarez, R.; Obici, S.; Schwartz, G.J.; Bryan, J.; Aguilar-Bryan, L.; Rossetti, L. Hypothalamic KATP channels control hepatic glucose production. Nature 2005, 434, 1026–1031. [Google Scholar] [CrossRef] [PubMed]
- Grill, H.J.; Schwartz, M.W.; Kaplan, J.M.; Foxhall, J.S.; Breininger, J.; Baskin, D.G. Evidence that the caudal brainstem is a target for the inhibitory effect of leptin on food intake. Endocrinology 2002, 143, 239–246. [Google Scholar] [CrossRef]
- Williams, K.W.; Zsombok, A.; Smith, B. Rapid inhibition of neurons in the dorsal motor nucleus of the vagus by leptin. Endocrinology 2006, 148, 1868–1881. [Google Scholar] [CrossRef]
- Rogers, R.C.; Hermann, G.E. Oxytocin, oxytocin antagonist, TRH, and hypothalamic paraventricular nucleus stimulation effects on gastric motility. Peptides 1987, 8, 505–513. [Google Scholar] [CrossRef]
- Abrahamsson, H.; Jansson, G. Elicitation of Reflex Vagal Relaxation of the Stomach from Pharynx and Esophagus in the Cat. Acta Physiol. Scand. 1969, 77, 172–178. [Google Scholar] [CrossRef]
- Ferreira, M.; Browning, K.N.; Sahibzada, N.; Verbalis, J.G.; Gillis, R.A.; Travagli, R.A. Glucose effects on gastric motility and tone evoked from the rat dorsal vagal complex. J. Physiol. 2001, 536, 141–152. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, M.W.; Porte, D. Diabetes, Obesity, and the Brain. Science 2005, 307, 375–379. [Google Scholar] [CrossRef] [PubMed]
- Bogacka, I.; Roane, D.S.; Xi, X.; Zhou, J.; Li, B.; Ryan, D.; Martin, R.J. Expression Levels of Genes Likely Involved in Glucose-sensing in the Obese Zucker Rat Brain. Nutr. Neurosci. 2004, 7, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Alemzadeh, R.; Karlstad, M.; Tushaus, K.; Buchholz, M. Diazoxide enhances basal metabolic rate and fat oxidation in obese Zucker rats. Metabolism 2008, 57, 1597–1607. [Google Scholar] [CrossRef] [PubMed]
- Alemzadeh, R.; Tushaus, K. Diazoxide attenuates insulin secretion and hepatic lipogenesis in zucker diabetic fatty rats. Med. Sci. Monit. 2005, 11, BR439–BR448. [Google Scholar]
- Komatsu, M.; Takei, M.; Ishii, H.; Sato, Y. Glucose-stimulated insulin secretion: A newer perspective. J. Diabetes Investig. 2013, 4, 511–516. [Google Scholar] [CrossRef]
- Page, M.M.; Johnson, J.D. Mild Suppression of Hyperinsulinemia to Treat Obesity and Insulin Resistance. Trends Endocrinol. Metab. 2018, 29, 389–399. [Google Scholar] [CrossRef]
- Templeman, N.M.; Skovsø, S.; Page, M.M.; E Lim, G.; Johnson, J.D. A causal role for hyperinsulinemia in obesity. J. Endocrinol. 2017, 232, R173–R183. [Google Scholar] [CrossRef]
- Gagnon, A.; Sorisky, A. The effect of glucose concentration on insulin-induced 3T3-L1 adipose cell differentiation. Obes. Res. 1998, 6, 157–163. [Google Scholar] [CrossRef]
- Nielsen, T.S.; Jessen, N.; Jørgensen, J.O.L.; Moller, N.; Lund, S. Dissecting adipose tissue lipolysis: Molecular regulation and implications for metabolic disease. J. Mol. Endocrinol. 2014, 52, R199–R222. [Google Scholar] [CrossRef]
- Czech, M.P.; Tencerova, M.; Pedersen, D.J.; Aouadi, M. Insulin signalling mechanisms for triacylglycerol storage. Diabetologia 2013, 56, 949–964. [Google Scholar] [CrossRef] [PubMed]
- Cristancho, A.G.; Lazar, M.A. Forming functional fat: A growing understanding of adipocyte differentiation. Nat. Rev. Mol. Cell Boil. 2011, 12, 722–734. [Google Scholar] [CrossRef] [PubMed]
- Corkey, B. Banting Lecture 2011: Hyperinsulinemia: Cause or Consequence? Diabetes 2011, 61, 4–13. [Google Scholar] [CrossRef] [PubMed]
- Mehran, A.E.; Templeman, N.M.; Brigidi, G.S.; Lim, G.E.; Chu, K.-Y.; Hu, X.; Botezelli, J.D.; Asadi, A.; Hoffman, B.; Kieffer, T.J.; et al. Hyperinsulinemia Drives Diet-Induced Obesity Independently of Brain Insulin Production. Cell Metab. 2012, 16, 723–737. [Google Scholar] [CrossRef]
- D’Souza, A.M.; Johnson, J.D.; Clee, S.M.; Kieffer, T.J. Suppressing hyperinsulinemia prevents obesity but causes rapid onset of diabetes in leptin-deficient Lepob/ob mice. Mol. Metab. 2016, 5, 1103–1112. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Charukeshi, C.P.; Pippin, J.J. Leptin- and Leptin Receptor-Deficient Rodent Models: Relevance for Human Type 2 Diabetes. Curr. Diabetes Rev. 2014, 10, 131–145. [Google Scholar] [CrossRef]
- Gray, S.L.; Donald, C.; Jetha, A.; Covey, S.D.; Kieffer, T.J. Hyperinsulinemia Precedes Insulin Resistance in Mice Lacking Pancreatic β-Cell Leptin Signaling. Endocrinology 2010, 151, 4178–4186. [Google Scholar] [CrossRef]
- Hales, C.; Kennedy, G.; Byrne, C.D.; Brindle, N.P.J.; Wang, T.W.M.; Zorzano, A.; Balon, T.W.; Brady, L.J.; Rivera, P.; Garetto, L.P.; et al. Plasma glucose, non-esterified fatty acid and insulin concentrations in hypothalamic-hyperphagic rats. Biochem. J. 1964, 90, 620–624. [Google Scholar] [CrossRef]
- York, D.A.; Bray, G.A. Dependence of Hypothalamic Obesity on Insulin, the Pituitary and the Adrenal Gland11. Endocrinology 1972, 90, 885–894. [Google Scholar] [CrossRef]
- Han, P.; Frohman, L. Hyperinsulinemia in tube-fed hypophysectomized rats bearing hypothalamic lesions. Am. J. Physiol. Content 1970, 219, 1632–1636. [Google Scholar] [CrossRef]
- Goldman, J.; Bernardis, L.; Frohman, L. Food intake in hypothalamic obesity. Am. J. Physiol. Content 1974, 227, 88–91. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.; Olefsky, J.M. Effect of experimental hyperinsulinemia on insulin binding and glucose transport in isolated rat adipocytes. Am. J. Physiol. Metab. 1978, 235, E53. [Google Scholar] [CrossRef]
- Rizza, R.A.; Mandarino, L.J.; Genest, J.; Baker, B.A.; Gerich, J.E. Production of insulin resistance by hyperinsulinemia in man. Diabetologia 1985, 28, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Shanik, M.H.; Xu, Y.; Škrha, J.; Dankner, R.; Zick, Y.; Roth, J. Insulin Resistance and Hyperinsulinemia: Is hyperinsulinemia the cart or the horse? Diabetes Care 2008, 31, S262–S268. [Google Scholar] [CrossRef] [PubMed]
- Ratzmann, K.P.; Ruhnke, R.; Kohnert, K.D. Effect of pharmacological suppression of insulin secretion on tissue sensitivity to insulin in subjects with moderate obesity. Int. J. Obes. 1983, 7, 453–458. [Google Scholar]
- Wigand, J.; Blackard, W. Downregulation of insulin receptors in obese men. Diabetes 1979, 28, 287–291. [Google Scholar] [CrossRef]
- Alemzadeh, R.; Zhang, J.; Tushaus, K.; Koontz, J. Diazoxide enhances adipose tissue protein kinase B activation and glucose transporter-4 expression in obese Zucker rats. Med. Sci. Monit. 2004, 10, BR53–BR60. [Google Scholar]
- Alemzadeh, R.; Tushaus, K.M. Modulation of Adipoinsular Axis in Prediabetic Zucker Diabetic Fatty Rats by Diazoxide. Endocrinology 2004, 145, 5476–5484. [Google Scholar] [CrossRef]
- Škrha, J.; Svacina, S.; Šrámková, J.; Páv, J. Use of euglycaemic clamping in evaluation of diazoxide treatment of insulinoma. Eur. J. Clin. Pharmacol. 1989, 36, 199–201. [Google Scholar] [CrossRef]
- Lutz, T.A.; Woods, S.C. Overview of animal models of obesity. Curr. Protoc. Pharmacol. 2012, 58, 5–61. [Google Scholar] [CrossRef]
- Dube, M.G.; Kalra, S.P.; Kalra, P.S. Low abundance of NPY in the hypothalamus can produce hyperphagia and obesity. Peptides 2007, 28, 475–479. [Google Scholar] [CrossRef] [PubMed]
- Bischof, J.M.; Wevrick, R. Chronic diazoxide treatment decreases fat mass and improves endurance capacity in an obese mouse model of Prader-Willi syndrome. Mol. Genet. Metab. 2018, 123, 511–517. [Google Scholar] [CrossRef] [PubMed]
- Knani, I.; Earley, B.J.; Udi, S.; Nemirovski, A.; Hadar, R.; Gammal, A.; Cinar, R.; Hirsch, H.J.; Pollak, Y.; Gross, I.; et al. Targeting the endocannabinoid/CB1 receptor system for treating obesity in Prader-Willi syndrome. Mol. Metab. 2016, 5, 1187–1199. [Google Scholar] [CrossRef] [PubMed]
- Dryden, S.; Pickavance, L.; Frankish, H.M.; Williams, G. Increased neuropeptide Y secretion in the hypothalamic paraventricular nucleus of obese (fa/fa) Zucker rats. Brain Res. 1995, 690, 185–188. [Google Scholar] [CrossRef]
- Alemzadeh, R.; Slonim, A.E.; Zdanowicz, M.M.; Maturo, J. Modification of insulin resistance by diazoxide in obese Zucker rats. Endocrinology 1993, 133, 705–712. [Google Scholar] [CrossRef] [PubMed]
- Alemzadeh, R.; Jacobs, W.; Pitukcheewanont, P. Antiobesity effect of diazoxide in obese zucker rats. Metabolism 1996, 45, 334–341. [Google Scholar] [CrossRef]
- Alemzadeh, R.; Holshouser, S. Effect of diazoxide on brain capillary insulin receptor binding and food intake in hyperphagic obese Zucker rats. Endocrinology 1999, 140, 3197–3202. [Google Scholar] [CrossRef]
- Standridge, M.; Alemzadeh, R.; Zemel, M.; Koontz, J.; Moustaid-Moussa, N. Diazoxide down-regulates leptin and lipid metabolizing enzymes in adipose tissue of Zucker rats. FASEB J. 2000, 14, 455–460. [Google Scholar] [CrossRef]
- Hensley, I.; E Lawler, J.; Alemzadeh, R.; Holshouser, S.J. Diazoxide effects on hypothalamic and extra-hypothalamic NPY content in Zucker rats. Peptides 2001, 22, 899–908. [Google Scholar] [CrossRef]
- De Luca, C.; Kowalski, T.J.; Zhang, Y.; Elmquist, J.K.; Lee, C.; Kilimann, M.W.; Ludwig, T.; Liu, S.-M.; Chua, S.C. Complete rescue of obesity, diabetes, and infertility in db/db mice by neuron-specific LEPR-B transgenes. J. Clin. Investig. 2005, 115, 3484–3493. [Google Scholar] [CrossRef]
- Lee, S. Effects of diazoxide on insulin secretion and metabolic efficiency in the db/db mouse. Life Sci. 1981, 28, 1829–1840. [Google Scholar] [CrossRef]
- Bi, S.; Moran, T.H. Obesity in the Otsuka Long Evans Tokushima Fatty Rat: Mechanisms and Discoveries. Front. Nutr. 2016, 3. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Bu, S.; Yu, Y.; Ghatnekar, G.; Wang, M.; Chen, L.; Bu, M.; Yang, L.; Zhu, B.; Feng, Z.; et al. Diazoxide prevents abdominal adiposity and fatty liver in obese OLETF rats at prediabetic stage. J. Diabetes Complicat. 2008, 22, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Larue-Achagiotis, C.; Le Magnen, J. Effects of a Diazoxide inhibition of insulin release on food intake of normal and hyperphagic hypothalamic rats. Pharmacol. Biochem. Behav. 1978, 9, 717–720. [Google Scholar] [CrossRef]
- Sonoda, T. Hyperinsulinemia and its role in maintaining the hypothalamic hyperphagia in chickens. Physiol. Behav. 1983, 30, 325–329. [Google Scholar] [CrossRef]
- Matsuda, M.; Kawasaki, F.; Mikami, Y.; Takeuchi, Y.; Saito, M.; Eto, M.; Kaku, K. Rescue of beta-cell exhaustion by diazoxide after the development of diabetes mellitus in rats with streptozotocin-induced diabetes. Eur. J. Pharmacol. 2002, 453, 141–148. [Google Scholar] [CrossRef]
- Surwit, R.S.; Dixon, T.M.; Petro, A.E.; Daniel, K.W.; Collins, S. Diazoxide restores β3-adrenergic receptor function in diet-induced obesity and diabetes. Endocrinology 2000, 141, 3630–3637. [Google Scholar] [CrossRef][Green Version]
- McCandless, S.E.; Committee on Genetics American Academy of Pediatrics. Clinical Report-Health supervision for children with Prader-Willi syndrome. Pediatrics 2011, 127, 195–204. [Google Scholar] [CrossRef]
- Miller, J.L.; Lynn, C.H.; Driscoll, D.C.; Goldstone, A.P.; Gold, J.-A.; Kimonis, V.; Dykens, E.; Butler, M.G.; Shuster, J.J.; Driscoll, D.J. Nutritional phases in Prader-Willi syndrome. Am. J. Med. Genet. Part. A 2011, 155, 1040–1049. [Google Scholar] [CrossRef]
- Hedgeman, E.; Ulrichsen, S.P.; Carter, S.; Kreher, N.C.; Malobisky, K.P.; Braun, M.M.; Fryzek, J.; Olsen, M.S. Long-term health outcomes in patients with Prader–Willi Syndrome: A nationwide cohort study in Denmark. Int. J. Obes. 2017, 41, 1531–1538. [Google Scholar] [CrossRef]
- Summary of the Impact of PWS on Individuals and Their Families and Views on Treatments: Results of an International Online Survey. Available online: https://www.fpwr.org/pws-patient-voices (accessed on 19 April 2020).
- Kimonis, V.E.; Surampalli, A.; Wencel, M.; Gold, J.-A.; Cowen, N.M. A randomized pilot efficacy and safety trial of diazoxide choline controlled-release in patients with Prader-Willi syndrome. PLoS ONE 2019, 14, e0221615. [Google Scholar] [CrossRef] [PubMed]
| Model | Energy Intake | Weight | Body Fat | Glycemic Control | Circulating Lipids | Hepatic Lipids |
|---|---|---|---|---|---|---|
| Magel2 mouse | NM | Weight loss | Loss of body fat | Improved | NM | NM |
| ZF rat | Reduced | Reduced rate of gain | NM | Improved | Improved | NM |
| ZDF rat | Reduced | Reduced rate of gain | NM | Improved | Improved | Improved |
| db/db mouse | Reduced | NM | NM | NM | NM | NM |
| OTLEF rat | Reduced | Reduced rate of gain | NM | Improved | Improved | Improved |
| Hypothalamic injury rat | Reduced | NM | NM | NM | NM | NM |
| Hypothalamic injury chicken | Reduced | NM | NM | NM | NM | NM |
| Streptozotocin diabetic rat | NM | Reduced rate of gain | NM | NM | NM | NM |
| HFD obese mouse | Reduced | Weight loss or reduced rate of gain | Loss of body fat | Improved | Improved | NM |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cowen, N.; Bhatnagar, A. The Potential Role of Activating the ATP-Sensitive Potassium Channel in the Treatment of Hyperphagic Obesity. Genes 2020, 11, 450. https://doi.org/10.3390/genes11040450
Cowen N, Bhatnagar A. The Potential Role of Activating the ATP-Sensitive Potassium Channel in the Treatment of Hyperphagic Obesity. Genes. 2020; 11(4):450. https://doi.org/10.3390/genes11040450
Chicago/Turabian StyleCowen, Neil, and Anish Bhatnagar. 2020. "The Potential Role of Activating the ATP-Sensitive Potassium Channel in the Treatment of Hyperphagic Obesity" Genes 11, no. 4: 450. https://doi.org/10.3390/genes11040450
APA StyleCowen, N., & Bhatnagar, A. (2020). The Potential Role of Activating the ATP-Sensitive Potassium Channel in the Treatment of Hyperphagic Obesity. Genes, 11(4), 450. https://doi.org/10.3390/genes11040450




