Comparative Genomics of the Transport Proteins of Ten Lactobacillus Strains
Abstract
1. Introduction
2. Materials and Methods
2.1. Genome-BLAST of the Ten Lactobacillus Proteomes
2.2. Examination of Distant Transport Protein Homologues in Lactobacillus spp.
2.3. Identification of Substrates Transported
2.4. Analysis of Multicomponent Systems
3. Results
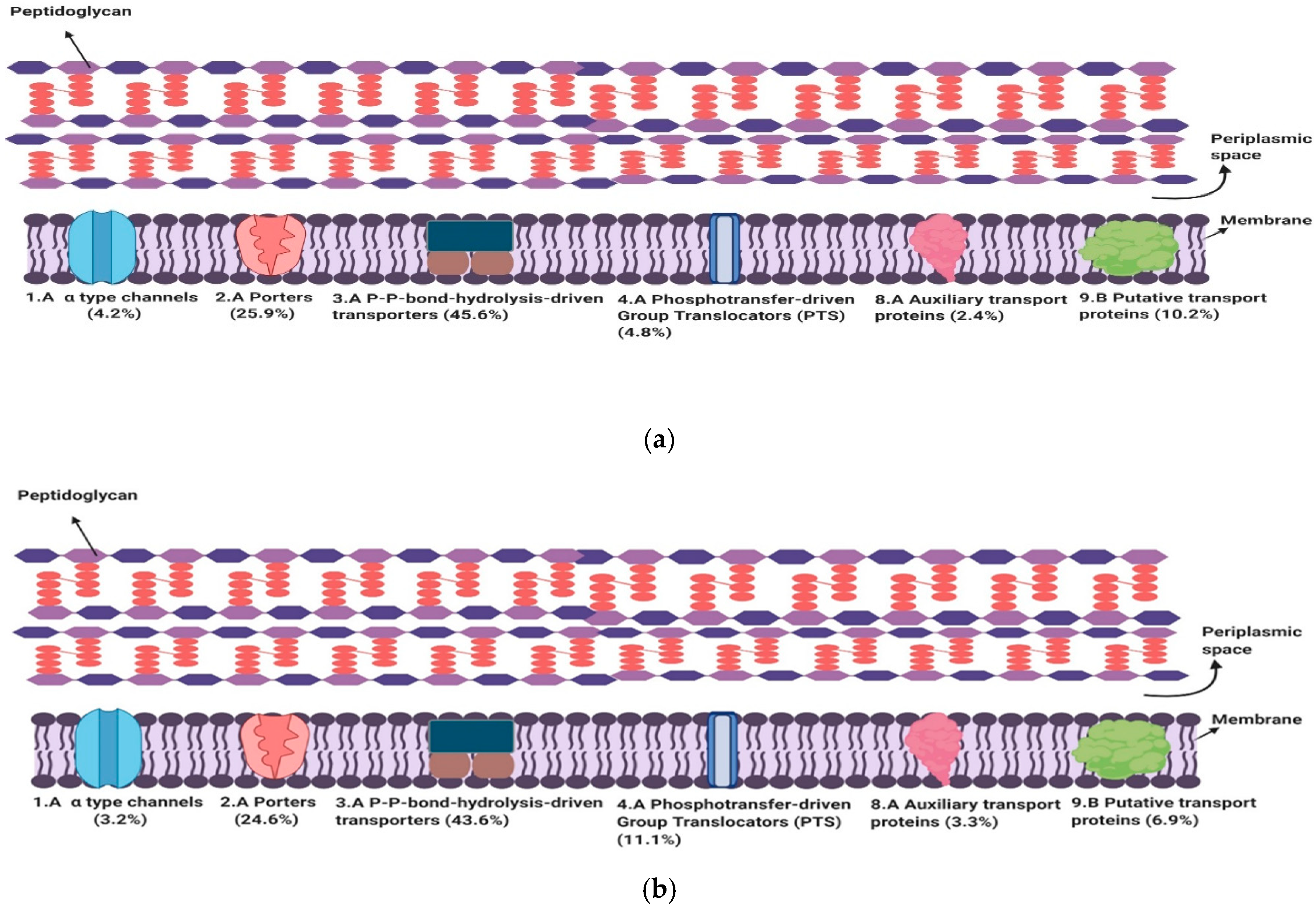
3.1. Subclasses of Transport Proteins
3.2. Channel Proteins (TC Subclass 1.A)
3.3. Pore-Forming Toxins (TC Subclass 1.C)
3.4. Holins (TC Subclass 1.E)
3.5. Bacterial Micro/NanoCompartment Shell Protein Pores (1.S)
3.6. Secondary Carriers (TC Subfamily 2.A)
3.7. Primary Active Transporters (TC Subclass 3.A)
3.8. Decarboxylation-Driven Transporters (TC Subclass 3.B)
3.9. Oxidoreduction Driven Transporters (TC Subclass 3.D)
3.10. Possible Group Translocators (TC Class 4)
3.11. Transmembrane Electron Carriers (TC Class 5)
3.12. Auxiliary Transporters (TC Subclass 8.A)
3.13. Incompletely Characterized Transport Systems (TC Class 9)
3.14. Differences in Substrates Transported by PAP and OP Strains
3.15. Major Families/Superfamilies Found in Lactobacillus Strains
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Walter, J. Ecological role of lactobacilli in the gastrointestinal tract: Implications for fundamental and biomedical research. Appl. Environ. Microbiol. 2008, 74, 4985–4996. [Google Scholar] [CrossRef] [PubMed]
- Campana, R.; van Hemert, S.; Baffone, W. Strain-specific probiotic properties of lactic acid bacteria and their interference with human intestinal pathogens invasion. Gut Pathog. 2017, 9, 12. [Google Scholar] [CrossRef]
- Rossi, F.; Amadoro, C.; Colavita, G. Members of the Lactobacillus Genus Complex (LGC) as Opportunistic Pathogens: A Review. Microorganisms 2019, 7, 126. [Google Scholar] [CrossRef] [PubMed]
- Martín, R.; Miquel, S.; Ulmer, J.; Kechaou, N.; Langella, P.; Bermúdez-Humarán, L.G. Role of commensal and probiotic bacteria in human health: A focus on inflammatory bowel disease. Microb. Cell Fact. 2013, 12, 71. [Google Scholar] [CrossRef] [PubMed]
- Baron, S. Medical Microbiology; University of Texas Medical Branch: Galveston, TX, USA, 1996; ISBN 9780963117212. [Google Scholar]
- Salvetti, E.; Torriani, S.; Felis, G.E. The Genus Lactobacillus: A Taxonomic Update. Probiotics Antimicrob. Proteins 2012, 4, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, S.; Demain, A.L. Metabolic regulation and overproduction of primary metabolites. Microb. Biotechnol. 2008, 1, 283–319. [Google Scholar] [CrossRef] [PubMed]
- Wells, J.M. Immunomodulatory mechanisms of lactobacilli. Microb. Cell Fact. 2011, 10 (Suppl. 1), S17. [Google Scholar] [CrossRef]
- Fijan, S. Microorganisms with claimed probiotic properties: An overview of recent literature. Int. J. Environ. Res. Public Health 2014, 11, 4745–4767. [Google Scholar] [CrossRef]
- Chen, C.C.; Lai, C.C.; Huang, H.L.; Huang, W.Y.; Toh, H.S.; Weng, T.C.; Chuang, Y.C.; Lu, Y.C.; Tang, H.J. Antimicrobial Activity of Lactobacillus Species against Carbapenem-Resistant Enterobacteriaceae. Front. Microbiol. 2019, 10, 789. [Google Scholar] [CrossRef]
- Gaspar, C.; Donders, G.G.; Palmeira-de-Oliveira, R.; Queiroz, J.A.; Tomaz, C.; Martinez-de-Oliveira, J.; Palmeira-de-Oliveira, A. Bacteriocin production of the probiotic Lactobacillus acidophilus KS400. AMB Express 2018, 8, 153. [Google Scholar] [CrossRef]
- Azad, M.A.K.; Sarker, M.; Li, T.; Yin, J. Probiotic Species in the Modulation of Gut Microbiota: An Overview. Biomed Res. Int. 2018, 2018, 9478630. [Google Scholar] [CrossRef]
- Ding, Y.H.; Qian, L.Y.; Pang, J.; Lin, J.Y.; Xu, Q.; Wang, L.H.; Huang, D.S.; Zou, H. The regulation of immune cells by Lactobacilli: A potential therapeutic target for anti-atherosclerosis therapy. Oncotarget 2017, 8, 59915–59928. [Google Scholar] [CrossRef]
- Yan, F.; Polk, D.B. Probiotics: Progress toward novel therapies for intestinal diseases. Curr. Opin. Gastroenterol. 2010, 26, 95–101. [Google Scholar] [CrossRef]
- Cannon, J.P.; Lee, T.A.; Bolanos, J.T.; Danziger, L.H. Pathogenic relevance of Lactobacillus: A retrospective review of over 200 cases. Eur. J. Clin. Microbiol. Infect. Dis. 2005, 24, 31–40. [Google Scholar] [CrossRef]
- Harty, D.W.; Oakey, H.J.; Patrikakis, M.; Hume, E.B.; Knox, K.W. Pathogenic potential of lactobacilli. Int. J. Food Microbiol. 1994, 24, 179–189. [Google Scholar] [CrossRef]
- Tang, F.; Saier, M.H., Jr. Transport proteins promoting Escherichia coli pathogenesis. Microb. Pathog. 2014, 71, 41–55. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Do, J.; Zafar, H.; Saier, M.H., Jr. Comparative genomics of transport proteins in probiotic and pathogenic Escherichia coli and Salmonella enterica strains. Microb. Pathog. 2017, 107, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Zafar, H.; Saier, M.H., Jr. Comparative genomics of transport proteins in seven Bacteroides species. PLoS ONE 2018, 13, e0208151. [Google Scholar] [CrossRef] [PubMed]
- Altermann, E.; Russell, W.M.; Azcarate-Peril, M.A.; Barrangou, R.; Buck, B.L.; McAuliffe, O.; Souther, N.; Dobson, A.; Duong, T.; Callanan, M.; et al. Complete genome sequence of the probiotic lactic acid bacterium Lactobacillus acidophilus NCFM. Proc. Natl. Acad. Sci. USA 2005, 102, 3906–3912. [Google Scholar] [CrossRef] [PubMed]
- Makarova, K.; Slesarev, A.; Wolf, Y.; Sorokin, A.; Mirkin, B.; Koonin, E.; Pavlov, A.; Pavlova, N.; Karamychev, V.; Polouchine, N.; et al. Comparative genomics of the lactic acid bacteria. Proc. Natl. Acad. Sci. USA 2006, 103, 15611–15616. [Google Scholar] [CrossRef] [PubMed]
- Ojala, T.; Kuparinen, V.; Koskinen, J.P.; Alatalo, E.; Holm, L.; Auvinen, P.; Edelman, S.; Westerlund-Wikström, B.; Korhonen, T.K.; Paulin, L.; et al. Genome sequence of Lactobacillus crispatus ST1. J. Bacteriol. 2010, 192, 3547–3548. [Google Scholar] [CrossRef] [PubMed]
- van de Guchte, M.; Penaud, S.; Grimaldi, C.; Barbe, V.; Bryson, K.; Nicolas, P.; Robert, C.; Oztas, S.; Mangenot, S.; Couloux, A.; et al. The complete genome sequence of Lactobacillus bulgaricus reveals extensive and ongoing reductive evolution. Proc. Natl. Acad. Sci. USA 2006, 103, 9274–9279. [Google Scholar] [CrossRef] [PubMed]
- Toh, H.; Oshima, K.; Nakano, A.; Takahata, M.; Murakami, M.; Takaki, T.; Nishiyama, H.; Igimi, S.; Hattori, M.; Morita, H. Genomic adaptation of the Lactobacillus casei group. PLoS ONE 2013, 8, e75073. [Google Scholar] [CrossRef] [PubMed]
- Siezen, R.J.; Francke, C.; Renckens, B.; Boekhorst, J.; Wels, M.; Kleerebezem, M.; van Hijum, S.A.F.T. Complete resequencing and reannotation of the Lactobacillus plantarum WCFS1 genome. J. Bacteriol. 2012, 194, 195–196. [Google Scholar] [CrossRef]
- Kankainen, M.; Paulin, L.; Tynkkynen, S.; von Ossowski, I.; Reunanen, J.; Partanen, P.; Satokari, R.; Vesterlund, S.; Hendrickx, A.P.A.; Lebeer, S.; et al. Comparative genomic analysis of Lactobacillus rhamnosus GG reveals pili containing a human- mucus binding protein. Proc. Natl. Acad. Sci. USA 2009, 106, 17193–17198. [Google Scholar] [CrossRef]
- Forde, B.M.; Neville, B.A.; O’Donnell, M.M.; Riboulet-Bisson, E.; Claesson, M.J.; Coghlan, A.; Ross, R.P.; O’Toole, P.W. Genome sequences and comparative genomics of two Lactobacillus ruminis strains from the bovine and human intestinal tracts. Microb. Cell Fact. 2011, 10 (Suppl. 1), S13. [Google Scholar] [CrossRef]
- Reddy, V.S.; Saier, M.H., Jr. BioV Suite--a collection of programs for the study of transport protein evolution. FEBS J. 2012, 279, 2036–2046. [Google Scholar] [CrossRef]
- Ikeda, M.; Arai, M.; Lao, D.M.; Shimizu, T. Transmembrane topology prediction methods: A re-assessment and improvement by a consensus method using a dataset of experimentally-characterized transmembrane topologies. In Silico Biol. 2002, 2, 19–33. [Google Scholar]
- Zhai, Y.; Saier, M.H., Jr. A web-based program (WHAT) for the simultaneous prediction of hydropathy, amphipathicity, secondary structure and transmembrane topology for a single protein sequence. J. Mol. Microbiol. Biotechnol. 2001, 3, 501–502. [Google Scholar]
- Ito, M.; Hicks, D.B.; Henkin, T.M.; Guffanti, A.A.; Powers, B.D.; Zvi, L.; Uematsu, K.; Krulwich, T.A. MotPS is the stator-force generator for motility of alkaliphilic Bacillus, and its homologue is a second functional Mot in Bacillus subtilis. Mol. Microbiol. 2004, 53, 1035–1049. [Google Scholar] [CrossRef]
- Neville, B.A.; Forde, B.M.; Claesson, M.J.; Darby, T.; Coghlan, A.; Nally, K.; Ross, R.P.; O’Toole, P.W. Characterization of pro-inflammatory flagellin proteins produced by Lactobacillus ruminis and related motile Lactobacilli. PLoS ONE 2012, 7, e40592. [Google Scholar] [CrossRef] [PubMed]
- Payandeh, J.; Pfoh, R.; Pai, E.F. The structure and regulation of magnesium selective ion channels. Biochim. Biophys. Acta 2013, 1828, 2778–2792. [Google Scholar] [CrossRef] [PubMed]
- Pohland, A.-C.; Schneider, D. Mg2+ homeostasis and transport in cyanobacteria—At the crossroads of bacterial and chloroplast Mg2+ import. Biol. Chem. 2019, 400, 1289–1301. [Google Scholar] [CrossRef] [PubMed]
- Stockbridge, R.B.; Robertson, J.L.; Kolmakova-Partensky, L.; Miller, C. A family of fluoride-specific ion channels with dual-topology architecture. Elife 2013, 2, e01084. [Google Scholar] [CrossRef] [PubMed]
- Baida, G.E.; Kuzmin, N.P. Cloning and primary structure of a new hemolysin gene from Bacillus cereus. Biochim. Biophys. Acta 1995, 1264, 151–154. [Google Scholar] [CrossRef]
- Baida, G.E.; Kuzmin, N.P. Mechanism of action of hemolysin III from Bacillus cereus. Biochim. Biophys. Acta 1996, 1284, 122–124. [Google Scholar] [CrossRef]
- Ríos Colombo, N.S.; Chalón, M.C.; Navarro, S.A.; Bellomio, A. Pediocin-like bacteriocins: New perspectives on mechanism of action and immunity. Curr. Genet. 2018, 64, 345–351. [Google Scholar] [CrossRef]
- Ahn, S.-J.; Burne, R.A. The atlA operon of Streptococcus mutans: Role in autolysin maturation and cell surface biogenesis. J. Bacteriol. 2006, 188, 6877–6888. [Google Scholar] [CrossRef]
- Oppegård, C.; Rogne, P.; Emanuelsen, L.; Kristiansen, P.E.; Fimland, G.; Nissen-Meyer, J. The two-peptide class II bacteriocins: Structure, production, and mode of action. J. Mol. Microbiol. Biotechnol. 2007, 13, 210–219. [Google Scholar] [CrossRef]
- Sharma, A.; Srivastava, S. Anti-Candida activity of two-peptide bacteriocins, plantaricins (Pln E/F and J/K) and their mode of action. Fungal Biol. 2014, 118, 264–275. [Google Scholar] [CrossRef]
- Hanny, E.L.L.; Mustopa, A.Z.; Budiarti, S.; Darusman, H.S.; Ningrum, R.A. Fatimah Efficacy, toxicity study and antioxidant properties of plantaricin E and F recombinants against enteropathogenic Escherichia coli K1.1 (EPEC K1.1). Mol. Biol. Rep. 2019, 46, 6501–6512. [Google Scholar] [CrossRef] [PubMed]
- Maqueda, M.; Sánchez-Hidalgo, M.; Fernández, M.; Montalbán-López, M.; Valdivia, E.; Martínez-Bueno, M. Genetic features of circular bacteriocins produced by Gram-positive bacteria. FEMS Microbiol. Rev. 2008, 32, 2–22. [Google Scholar] [CrossRef] [PubMed]
- Saier, M.H., Jr.; Reddy, B.L. Holins in bacteria, eukaryotes, and archaea: Multifunctional xenologues with potential biotechnological and biomedical applications. J. Bacteriol. 2015, 197, 7–17. [Google Scholar] [CrossRef]
- Ahn, S.-J.; Qu, M.-D.; Roberts, E.; Burne, R.A.; Rice, K.C. Identification of the Streptococcus mutans LytST two-component regulon reveals its contribution to oxidative stress tolerance. BMC Microbiol. 2012, 12, 187. [Google Scholar] [CrossRef]
- Bayles, K.W. Are the molecular strategies that control apoptosis conserved in bacteria? Trends Microbiol. 2003, 11, 306–311. [Google Scholar] [CrossRef]
- Brunskill, E.W.; Bayles, K.W. Identification of LytSR-regulated genes from Staphylococcus aureus. J. Bacteriol. 1996, 178, 5810–5812. [Google Scholar] [CrossRef]
- Tanouchi, Y.; Lee, A.J.; Meredith, H.; You, L. Programmed cell death in bacteria and implications for antibiotic therapy. Trends Microbiol. 2013, 21, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Chun, S.; Bobik, T.A.; Houk, K.N.; Yeates, T.O. Molecular Dynamics Simulations of Selective Metabolite Transport across the Propanediol Bacterial Microcompartment Shell. J. Phys. Chem. B 2017, 121, 8149–8154. [Google Scholar] [CrossRef]
- Crowley, C.S.; Sawaya, M.R.; Bobik, T.A.; Yeates, T.O. Structure of the PduU shell protein from the Pdu microcompartment of Salmonella. Structure 2008, 16, 1324–1332. [Google Scholar] [CrossRef]
- Fàbrega, A.; Martin, R.G.; Rosner, J.L.; Tavio, M.M.; Vila, J. Constitutive SoxS expression in a fluoroquinolone-resistant strain with a truncated SoxR protein and identification of a new member of the marA-soxS-rob regulon, mdtG. Antimicrob. Agents Chemother. 2010, 54, 1218–1225. [Google Scholar] [CrossRef]
- Clancy, J.; Petitpas, J.; Dib-Hajj, F.; Yuan, W.; Cronan, M.; Kamath, A.V.; Bergeron, J.; Retsema, J.A. Molecular cloning and functional analysis of a novel macrolide-resistance determinant, mefA, from Streptococcus pyogenes. Mol. Microbiol. 1996, 22, 867–879. [Google Scholar] [CrossRef] [PubMed]
- Santagati, M.; Iannelli, F.; Cascone, C.; Campanile, F.; Oggioni, M.R.; Stefani, S.; Pozzi, G. The novel conjugative transposon tn1207.3 carries the macrolide efflux gene mef(A) in Streptococcus pyogenes. Microb. Drug Resist. 2003, 9, 243–247. [Google Scholar] [CrossRef] [PubMed]
- Schweikhard, E.S.; Ziegler, C.M. Amino acid secondary transporters: Toward a common transport mechanism. Curr. Top. Membr. 2012, 70, 1–28. [Google Scholar] [PubMed]
- Burkovski, A.; Krämer, R. Bacterial amino acid transport proteins: Occurrence, functions, and significance for biotechnological applications. Appl. Microbiol. Biotechnol. 2002, 58, 265–274. [Google Scholar] [PubMed]
- Buyuktimkin, B.; Zafar, H.; Saier, M.H., Jr. Comparative genomics of the transportome of Ten Treponema species. Microb. Pathog. 2019, 132, 87–99. [Google Scholar] [CrossRef]
- Teh, A.H.T.; Lee, S.M.; Dykes, G.A. Identification of potential Campylobacter jejuni genes involved in biofilm formation by EZ-Tn5 Transposome mutagenesis. BMC Res. Notes 2017, 10, 182. [Google Scholar] [CrossRef]
- Kermani, A.A.; Macdonald, C.B.; Gundepudi, R.; Stockbridge, R.B. Guanidinium export is the primal function of SMR family transporters. Proc. Natl. Acad. Sci. USA 2018, 115, 3060–3065. [Google Scholar] [CrossRef]
- Ioannou, C.J.; Hanlon, G.W.; Denyer, S.P. Action of disinfectant quaternary ammonium compounds against Staphylococcus aureus. Antimicrob. Agents Chemother. 2007, 51, 296–306. [Google Scholar] [CrossRef]
- Hvorup, R.N.; Winnen, B.; Chang, A.B.; Jiang, Y.; Zhou, X.-F.; Saier, M.H., Jr. The multidrug/oligosaccharidyl-lipid/polysaccharide (MOP) exporter superfamily. Eur. J. Biochem. 2003, 270, 799–813. [Google Scholar] [CrossRef]
- Tocci, N.; Iannelli, F.; Bidossi, A.; Ciusa, M.L.; Decorosi, F.; Viti, C.; Pozzi, G.; Ricci, S.; Oggioni, M.R. Functional analysis of pneumococcal drug efflux pumps associates the MATE DinF transporter with quinolone susceptibility. Antimicrob. Agents Chemother. 2013, 57, 248–253. [Google Scholar] [CrossRef]
- Kaatz, G.W.; DeMarco, C.E.; Seo, S.M. MepR, a repressor of the Staphylococcus aureus MATE family multidrug efflux pump MepA, is a substrate-responsive regulatory protein. Antimicrob. Agents Chemother. 2006, 50, 1276–1281. [Google Scholar] [CrossRef] [PubMed]
- Garvis, S.; Mei, J.-M.; Ruiz-Albert, J.; Holden, D.W. Staphylococcus aureus svrA: A gene required for virulence and expression of the agr locus. Microbiology 2002, 148, 3235–3243. [Google Scholar] [CrossRef] [PubMed]
- Kaatz, G.W.; McAleese, F.; Seo, S.M. Multidrug resistance in Staphylococcus aureus due to overexpression of a novel multidrug and toxin extrusion (MATE) transport protein. Antimicrob. Agents Chemother. 2005, 49, 1857–1864. [Google Scholar] [CrossRef] [PubMed]
- Davidson, A.L.; Maloney, P.C. ABC transporters: How small machines do a big job. Trends Microbiol. 2007, 15, 448–455. [Google Scholar] [CrossRef] [PubMed]
- Paulsen, I.T.; Nguyen, L.; Sliwinski, M.K.; Rabus, R.; Saier, M.H., Jr. Microbial genome analyses: Comparative transport capabilities in eighteen prokaryotes. J. Mol. Biol. 2000, 301, 75–100. [Google Scholar] [CrossRef]
- Gibson, G.R.; Beatty, E.R.; Wang, X.; Cummings, J.H. Selective stimulation of bifidobacteria in the human colon by oligofructose and inulin. Gastroenterology 1995, 108, 975–982. [Google Scholar] [CrossRef]
- Basavanna, S.; Khandavilli, S.; Yuste, J.; Cohen, J.M.; Hosie, A.H.F.; Webb, A.J.; Thomas, G.H.; Brown, J.S. Screening of Streptococcus pneumoniae ABC transporter mutants demonstrates that LivJHMGF, a branched-chain amino acid ABC transporter, is necessary for disease pathogenesis. Infect. Immun. 2009, 77, 3412–3423. [Google Scholar] [CrossRef]
- Wortham, B.W.; Patel, C.N.; Oliveira, M.A. Polyamines in bacteria: Pleiotropic effects yet specific mechanisms. Adv. Exp. Med. Biol. 2007, 603, 106–115. [Google Scholar]
- Abrahams, J.P.; Leslie, A.G.; Lutter, R.; Walker, J.E. Structure at 2.8 A resolution of F1-ATPase from bovine heart mitochondria. Nature 1994, 370, 621–628. [Google Scholar] [CrossRef]
- Balsera, M.; Buey, R.M.; Li, X.-D. Quaternary structure of the oxaloacetate decarboxylase membrane complex and mechanistic relationships to pyruvate carboxylases. J. Biol. Chem. 2011, 286, 9457–9467. [Google Scholar] [CrossRef]
- Kleerebezem, M.; Hols, P.; Bernard, E.; Rolain, T.; Zhou, M.; Siezen, R.J.; Bron, P.A. The extracellular biology of the lactobacilli. FEMS Microbiol. Rev. 2010, 34, 199–230. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; van der Veen, S.; Nakajima, H.; Abee, T. Effect of respiration and manganese on oxidative stress resistance of Lactobacillus plantarum WCFS1. Microbiology 2012, 158, 293–300. [Google Scholar] [CrossRef]
- Morita, H.; Toh, H.; Fukuda, S.; Horikawa, H.; Oshima, K.; Suzuki, T.; Murakami, M.; Hisamatsu, S.; Kato, Y.; Takizawa, T.; et al. Comparative genome analysis of Lactobacillus reuteri and Lactobacillus fermentum reveal a genomic island for reuterin and cobalamin production. DNA Res. 2008, 15, 151–161. [Google Scholar] [CrossRef] [PubMed]
- Black, P.N.; DiRusso, C.C. Vectorial acylation: Linking fatty acid transport and activation to metabolic trafficking. Novartis Found. Symp. 2007, 286, 127–203, discussion 138–141, 162–163, 196–203. [Google Scholar]
- ZJia, Z.; Pei, Z.; Maiguel, D.; Toomer, C.J.; Watkins, P.A. The fatty acid transport protein (FATP) family: Very long chain acyl-CoA synthetases or solute carriers? J. Mol. Neurosci. 2007, 33, 25–31. [Google Scholar]
- Davis, J.K. Combining polysaccharide biosynthesis and transport in a single enzyme: Dual-function cell wall glycan synthases. Front. Plant Sci. 2012, 3, 138. [Google Scholar] [CrossRef]
- Ernst, C.M.; Peschel, A. Broad-spectrum antimicrobial peptide resistance by MprF-mediated aminoacylation and flipping of phospholipids. Mol. Microbiol. 2011, 80, 290–299. [Google Scholar] [CrossRef]
- Brooijmans, R.; de Vos, W.M.; Hugenholtz, J. Electron transport chains of lactic acid bacteria—Walking on crutches is part of their lifestyle. F1000 Biol. Rep. 2009, 1, 34. [Google Scholar] [CrossRef]
- Akiyama, Y. Quality control of cytoplasmic membrane proteins in Escherichia coli. J. Biochem. 2009, 146, 449–454. [Google Scholar] [CrossRef] [PubMed]
- Coulter, S.N.; Schwan, W.R.; Ng, E.Y.; Langhorne, M.H.; Ritchie, H.D.; Westbrock-Wadman, S.; Hufnagle, W.O.; Folger, K.R.; Bayer, A.S.; Stover, C.K. Staphylococcus aureus genetic loci impacting growth and survival in multiple infection environments. Mol. Microbiol. 1998, 30, 393–404. [Google Scholar] [CrossRef]
- Steeb, B.; Claudi, B.; Burton, N.A.; Tienz, P.; Schmidt, A.; Farhan, H.; Mazé, A.; Bumann, D. Parallel exploitation of diverse host nutrients enhances Salmonella virulence. PLoS Pathog. 2013, 9, e1003301. [Google Scholar] [CrossRef] [PubMed]
- Costliow, Z.A.; Degnan, P.H. Thiamine Acquisition Strategies Impact Metabolism and Competition in the Gut Microbe Bacteroides thetaiotaomicron. mSystems 2017, 2, e00116-17. [Google Scholar] [CrossRef] [PubMed]
- Paulsen, I.T.; Sliwinski, M.K.; Saier, M.H., Jr. Microbial genome analyses: Global comparisons of transport capabilities based on phylogenies, bioenergetics, and substrate specificities. J. Mol. Biol. 1998, 277, 573–592. [Google Scholar] [CrossRef] [PubMed]
- Duar, R.M.; Lin, X.B.; Zheng, J.; Martino, M.E.; Grenier, T.; Perez-Munoz, M.E.; Leulier, F.; Ganzle, M.; Walter, J. Lifestyles in transition: Evolution and natural history of the genus Lactobacillus. FEMS Microbiol. Rev. 2017, 41, S27–S48. [Google Scholar] [CrossRef]
- Castro-González, J.M.; Castro, P.; Sandoval, H.; Castro-Sandoval, D. Probiotic Lactobacilli Precautions. Front. Microbiol. 2019, 10, 375. [Google Scholar] [CrossRef] [PubMed]
- de Jong, A.; van Hijum, S.A.F.T.; Bijlsma, J.J.E.; Kok, J.; Kuipers, O.P. BAGEL: A web-based bacteriocin genome mining tool. Nucleic Acids Res. 2006, 34, W273–W279. [Google Scholar] [CrossRef]
- Leyva Salas, M.; Thierry, A.; Lemaître, M.; Garric, G.; Harel-Oger, M.; Chatel, M.; Lê, S.; Mounier, J.; Valence, F.; Coton, E. Antifungal Activity of Lactic Acid Bacteria Combinations in Dairy Mimicking Models and Their Potential as Bioprotective Cultures in Pilot Scale Applications. Front. Microbiol. 2018, 9, 1787. [Google Scholar] [CrossRef]
- Karami, S.; Roayaei, M.; Zahedi, E.; Bahmani, M.; Mahmoodnia, L.; Hamzavi, H.; Rafieian-Kopaei, M. Antifungal effects of Lactobacillus species isolated from local dairy products. Int. J. Pharm. Investig. 2017, 7, 77–81. [Google Scholar]
- Gerbaldo, G.A.; Barberis, C.; Pascual, L.; Dalcero, A.; Barberis, L. Antifungal activity of two Lactobacillus strains with potential probiotic properties. FEMS Microbiol. Lett. 2012, 332, 27–33. [Google Scholar] [CrossRef]
- Kim, S.W.; Ha, Y.J.; Bang, K.H.; Lee, S.; Yeo, J.-H.; Yang, H.-S.; Kim, T.-W.; Lee, K.P.; Bang, W.Y. Potential of Bacteriocins from Lactobacillus taiwanensis for Producing Bacterial Ghosts as a Next Generation Vaccine. Toxins 2020, 12, 432. [Google Scholar] [CrossRef]
- Lorca, G.L.; Barabote, R.D.; Zlotopolski, V.; Tran, C.; Winnen, B.; Hvorup, R.N.; Stonestrom, A.J.; Nguyen, E.; Huang, L.-W.; Kim, D.S.; et al. Transport capabilities of eleven gram-positive bacteria: Comparative genomic analyses. Biochim. Biophys. Acta 2007, 1768, 1342–1366. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Hagelsieb, G.; Vitug, B.; Medrano-Soto, A.; Saier, M.H., Jr. The Membrane Attack Complex/Perforin Superfamily. J. Mol. Microbiol. Biotechnol. 2017, 27, 252–267. [Google Scholar] [CrossRef] [PubMed]
| Strain | Abbreviation | Accession # | Genome Size (Mbp) | Total # of Proteins Identified | Transport Proteins (% of Total) | Location in Host | Relationship with Host |
|---|---|---|---|---|---|---|---|
| Lactobacillus acidophilus NCFM | Lac | NC_006814.3 | 1.99 | 303 | 17.1 | GIT, oral cavity, vagina | Probiotic; may cause endocarditis |
| Lactobacillus brevis ATCC 367 | Lbr | NC_008497.1 | 2.29 | 327 | 15.4 | GIT, vagina | Probiotic; non-pathogenic |
| Lactobacillus delbrueckii subsp. bulgaricus ATCC 11842 | Lbu | NC_008054.1 | 1.87 | 246 | 15.7 | GIT | Probiotic; non-pathogenic |
| Lactobacillus crispatus ST1 | Lcr | NC_014106.1 | 2.04 | 328 | 17.8 | GIT, vagina | Probiotic; non-pathogenic |
| Lactobacillus gasseri ATCC | Lga | NC_008530.1 | 1.89 | 286 | 16.3 | GIT, vagina | Probiotic; non-pathogenic |
| Lactobacillus paracasei JCM 8130 | Lpa | NZ_AP012541.1 | 3.00 | 458 | 16.6 | GIT | Probiotic and pathogenic |
| Lactobacillus plantarum WCFS1 | Lpl | NC_004567.2 | 3.31 | 434 | 14.5 | GIT | Probiotic and pathogenic |
| Lactobacillus reuteri DSM 20016 | Lre | NC_009513.1 | 2.00 | 281 | 14.9 | GIT, urinary tract, skin | Probiotic; non-pathogenic |
| Lactobacillus rhamnosus GG | Lrh | NC_013198.1 | 3.01 | 407 | 14.7 | GIT, vagina | Probiotic and pathogenic |
| Lactobacillus ruminis ATCC 27782 | Lru | NC_015975.1 | 2.07 | 289 | 15.8 | GIT | Probiotic; non-pathogenic |
| TC Subclass and Description | Lac | Lbr | Lbu | Lcr | Lga | Lpa | Lpl | Lre | Lrh | Lru | Lac | Lbr | Lbu | Lcr | Lga | Lpa | Lpl | Lre | Lrh | Lru |
| 1.A, α-type channels | 12 | 13 | 11 | 13 | 10 | 14 | 14 | 13 | 11 | 14 | 3.9 | 4.0 | 4.5 | 4.0 | 3.5 | 3.1 | 3.2 | 4.6 | 2.7 | 4.8 |
| 1.B, β-barrel porins | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 1.C, Pore-forming toxins | 7 | 3 | 4 | 5 | 4 | 4 | 7 | 3 | 3 | 4 | 2.3 | 0.9 | 1.6 | 1.5 | 1.4 | 0.9 | 1.6 | 1.1 | 0.7 | 1.4 |
| 1.E, Holins | 1 | 5 | 0 | 3 | 3 | 6 | 8 | 4 | 5 | 3 | 0.3 | 1.5 | 0 | 0.9 | 1.1 | 1.3 | 1.9 | 1.4 | 1.2 | 1.0 |
| 1.S: Bacterial Micro/Nano Compartment Shell Protein Pores | 0 | 3 | 0 | 0 | 0 | 0 | 0 | 3 | 0 | 0 | 0 | 0.9 | 0 | 0 | 0 | 0 | 0 | 1.1 | 0 | 0 |
| 2.A, Porters (uniporters, symporters, antiporters) | 79 | 114 | 51 | 68 | 66 | 94 | 130 | 100 | 89 | 59 | 26.0 | 34.8 | 20.8 | 20.7 | 23.1 | 20.5 | 30.0 | 35.6 | 21.9 | 20.4 |
| 3.A, P-P-bond-hydrolysis-driven transporters | 149 | 128 | 137 | 161 | 123 | 201 | 170 | 101 | 172 | 148 | 49.2 | 39.1 | 55.7 | 49.1 | 43.0 | 43.8 | 39.2 | 35.9 | 42.3 | 51.0 |
| 3.B, Decarboxylation-driven transporters | 0 | 0 | 1 | 2 | 0 | 3 | 0 | 0 | 3 | 0 | 0 | 0 | 0.4 | 0.6 | 0 | 0.7 | 0 | 0 | 0.7 | 0 |
| 3.D, Oxidoreduction-driven transporters | 2 | 3 | 0 | 4 | 4 | 3 | 3 | 5 | 0 | 3 | 0.7 | 0.9 | 0 | 1.2 | 1.4 | 0.7 | 0.7 | 1.7 | 0 | 1.0 |
| 4.A Phosphotransfer-driven Group Translocators (PTS) | 23 | 3 | 7 | 30 | 31 | 61 | 44 | 2 | 54 | 13 | 7.6 | 0.9 | 2.8 | 9.2 | 10.8 | 13.3 | 10.1 | 0.7 | 13.3 | 4.5 |
| 4.B, Nicotinamide ribonucleoside uptake transporters | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0.3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 4.C, Acyl-CoA ligase-coupled transporters | 0 | 1 | 0 | 1 | 1 | 1 | 0 | 0 | 0 | 0, | 0 | 0.3 | 0 | 0.3 | 0.35 | 0.2 | 0 | 0 | 0 | 0 |
| 4.D, Polysaccharide synthase exporters | 2 | 4 | 1 | 2 | 2 | 4 | 4 | 1 | 2 | 3 | 0.7 | 1.3 | 0.4 | 0.6 | 0.65 | 0.9 | 0.9 | 0.4 | 0.5 | 1.0 |
| 4.F: Choline/Ethanolamine Phosphotransferase 1 (CEPT1) | 0 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 0.3 | 0.4 | 0.3 | 0.3 | 0.2 | 0 | 0.4 | 0 | 0.4 |
| 4.H: Lysylphosphatidylglycerol Synthase/Flippases | 0 | 1 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 1 | 0 | 0.3 | 0 | 0 | 0 | 0.2 | 0.2 | 0.4 | 0.2 | 0.4 |
| 5.A, Transmembrane two-electron transfer carriers | 0 | 0 | 0 | 0 | 0 | 0 | 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0.7 | 0 | 0 | 0 |
| 5.B: Transmembrane 1-electron transfer carriers | 0 | 3 | 1 | 3 | 3 | 11 | 8 | 4 | 12 | 1 | 0 | 0.9 | 0.4 | 0.9 | 1.1 | 2.4 | 1.9 | 1.4 | 3.0 | 0.4 |
| 8.A, Auxiliary transport proteins | 9 | 11 | 5 | 6 | 7 | 17 | 11 | 9 | 16 | 4 | 3.0 | 3.4 | 2.0 | 1.8 | 2.5 | 3.7 | 2.5 | 3.2 | 3.9 | 1.4 |
| 9.A, Recognized transporters of unknown biochemical mechanism | 2 | 1 | 2 | 1 | 1 | 3 | 4 | 3 | 5 | 2 | 0.7 | 0.3 | 0.8 | 0.3 | 0.3 | 0.7 | 0.9 | 1.1 | 1.2 | 0.8 |
| 9.B, Putative transport proteins | 17 | 32 | 25 | 28 | 30 | 34 | 27 | 31 | 34 | 33 | 5.6 | 9.8 | 10.2 | 8.5 | 10.5 | 7.4 | 6.2 | 11.0 | 8.4 | 11.4 |
| Total | 303 | 327 | 246 | 328 | 286 | 458 | 434 | 281 | 407 | 289 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| TCID | Family | Function | Lac | Lbr | Lbu | Lcr | Lga | Lpa | Lpl | Lre | Lrh | Lru | Type of Strains |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1.C.24.1.1 | Pediocin Family | Pore formation | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | OP |
| 1.C.29.1.1 | Plantaricin EF Family | Pore formation | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | PAP |
| 1.C.30.1.1 | Plantaricin JK Family | Pore formation | 0 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | PAP and OP |
| 1.C.30.1.3 | Plantaricin JK Family | Pore formation | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | PAP |
| 1.C.75.1.7 | Serratia-type Pore-forming Toxin (S-PFT) Family | Pore formation | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | PAP |
| 1.C.82.1.1 | Pore-forming Amphipathic Helical Peptide (HP2-20) Family | Pore formation | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | PAP and OP |
| 1.C.83.1.2 | Gassericin Family | Pore formation | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | PAP |
| 1.C.105.2.9 | Bacillus thuringiensis Vegetative Insecticidal Protein-3 (Vip3) Family | Pore formation | 1 | 0 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | PAP and OP |
| 1.C.109.1.5 | Bacterial Hemolysin A Family | Pore formation | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | PAP and OP |
| 1.C.113.1.1 | Hly III Family | Pore formation | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | PAP and OP |
| 1.C.126.1.2 | HlyC Family of Haemolysins | Pore formation | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | OP |
| 1.C.126.1.3 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | OP |
| Substrate Category | Lac | Lbr | Lbu | Lcr | Lga | Lpa | Lpl | Lre | Lrh | Lru |
| Inorganic Anions | 7 | 8 | 3 | 6 | 4 | 8 | 5 | 6 | 7 | 9 |
| Inorganic Cations | 30 | 43 | 27 | 39 | 25 | 38 | 54 | 38 | 31 | 27 |
| Amines | 4 | 5 | 4 | 4 | 4 | 4 | 5 | 4 | 4 | 4 |
| Amino acids | 36 | 31 | 30 | 35 | 37 | 43 | 47 | 39 | 38 | 35 |
| Non-selective | 44 | 52 | 39 | 45 | 46 | 75 | 77 | 36 | 57 | 44 |
| Drugs | 12 | 21 | 13 | 14 | 16 | 20 | 25 | 8 | 19 | 11 |
| Nucleobases, Nucleosides, Nucleotides | 26 | 22 | 13 | 24 | 17 | 14 | 19 | 18 | 13 | 10 |
| Proteins, Peptides | 30 | 27 | 24 | 29 | 19 | 45 | 35 | 20 | 37 | 36 |
| Sugars and Sugar Derivatives | 30 | 8 | 13 | 43 | 33 | 61 | 56 | 12 | 63 | 25 |
| Lipids | 4 | 2 | 3 | 4 | 4 | 3 | 2 | 2 | 3 | 2 |
| Vitamins | 7 | 13 | 11 | 10 | 7 | 16 | 14 | 11 | 15 | 12 |
| Unknown | 73 | 95 | 66 | 75 | 74 | 131 | 95 | 87 | 120 | 74 |
| Total | 303 | 327 | 246 | 328 | 286 | 458 | 434 | 281 | 407 | 289 |
| Family name, abbreviation, and TC# | Lac | Lbr | Lbu | Lcr | Lga | Lpa | Lpl | Lrh | Lre | Lru | Total # | Lac | Lbr | Lbu | Lcr | Lga | Lpa | Lpl | Lrh | Lre | Lru | Average% |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| The Major Facilitator Superfamily (MFS) (TC#2.A.1) | 12 | 32 | 10 | 13 | 15 | 25 | 38 | 30 | 23 | 11 | 209 | 3.9 | 9.8 | 4.1 | 3.9 | 5.2 | 5.5 | 8.8 | 7.3 | 8.2 | 3.8 | 6.1 |
| The Amino Acid-Polyamine-Organocation (APC) Superfamily (TC#2.A.3) | 15 | 12 | 4 | 15 | 13 | 10 | 12 | 13 | 20 | 6 | 120 | 4.9 | 3.6 | 1.6 | 4.6 | 4.5 | 2.2 | 2.8 | 3.2 | 7.1 | 2.1 | 3.7 |
| The Drug/Metabolite Transporter (DMT) Superfamily (TC#2.A.7) | 5 | 6 | 3 | 3 | 4 | 4 | 10 | 4 | 6 | 1 | 46 | 1.7 | 1.8 | 1.2 | 0.9 | 1.4 | 0.8 | 2.3 | 1.0 | 2.1 | 0.3 | 1.3 |
| The ATP-binding Cassette (ABC) Superfamily (TC#3.A.1) | 103 | 86 | 102 | 110 | 82 | 160 | 127 | 136 | 62 | 93 | 1061 | 33.9 | 26.2 | 41.4 | 33.5 | 28.7 | 34.9 | 29.2 | 33.4 | 22.0 | 32.1 | 31.6 |
| Totals | 135 | 136 | 119 | 141 | 114 | 199 | 187 | 183 | 111 | 111 | 1436/3359 | 44.4 | 41.4 | 48.3 | 42.9 | 39.8 | 43.4 | 43.1 | 44.9 | 39.4 | 38.3 | 42.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zafar, H.; Saier, M.H., Jr. Comparative Genomics of the Transport Proteins of Ten Lactobacillus Strains. Genes 2020, 11, 1234. https://doi.org/10.3390/genes11101234
Zafar H, Saier MH Jr. Comparative Genomics of the Transport Proteins of Ten Lactobacillus Strains. Genes. 2020; 11(10):1234. https://doi.org/10.3390/genes11101234
Chicago/Turabian StyleZafar, Hassan, and Milton H. Saier, Jr. 2020. "Comparative Genomics of the Transport Proteins of Ten Lactobacillus Strains" Genes 11, no. 10: 1234. https://doi.org/10.3390/genes11101234
APA StyleZafar, H., & Saier, M. H., Jr. (2020). Comparative Genomics of the Transport Proteins of Ten Lactobacillus Strains. Genes, 11(10), 1234. https://doi.org/10.3390/genes11101234





