Expression Analysis of XTH in Stem Swelling of Stem Mustard and Selection of Reference Genes
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant RNA Extraction and cDNA Synthesis
2.2. Selection of Reference Genes, Primer Design, and Cloning
2.3. Real-Time Quantitative PCR Amplification (qPCR)
2.4. Data Analysis and Evaluation of Stability of Reference Genes
2.5. Identification of XTH Family Genes in B. juncea
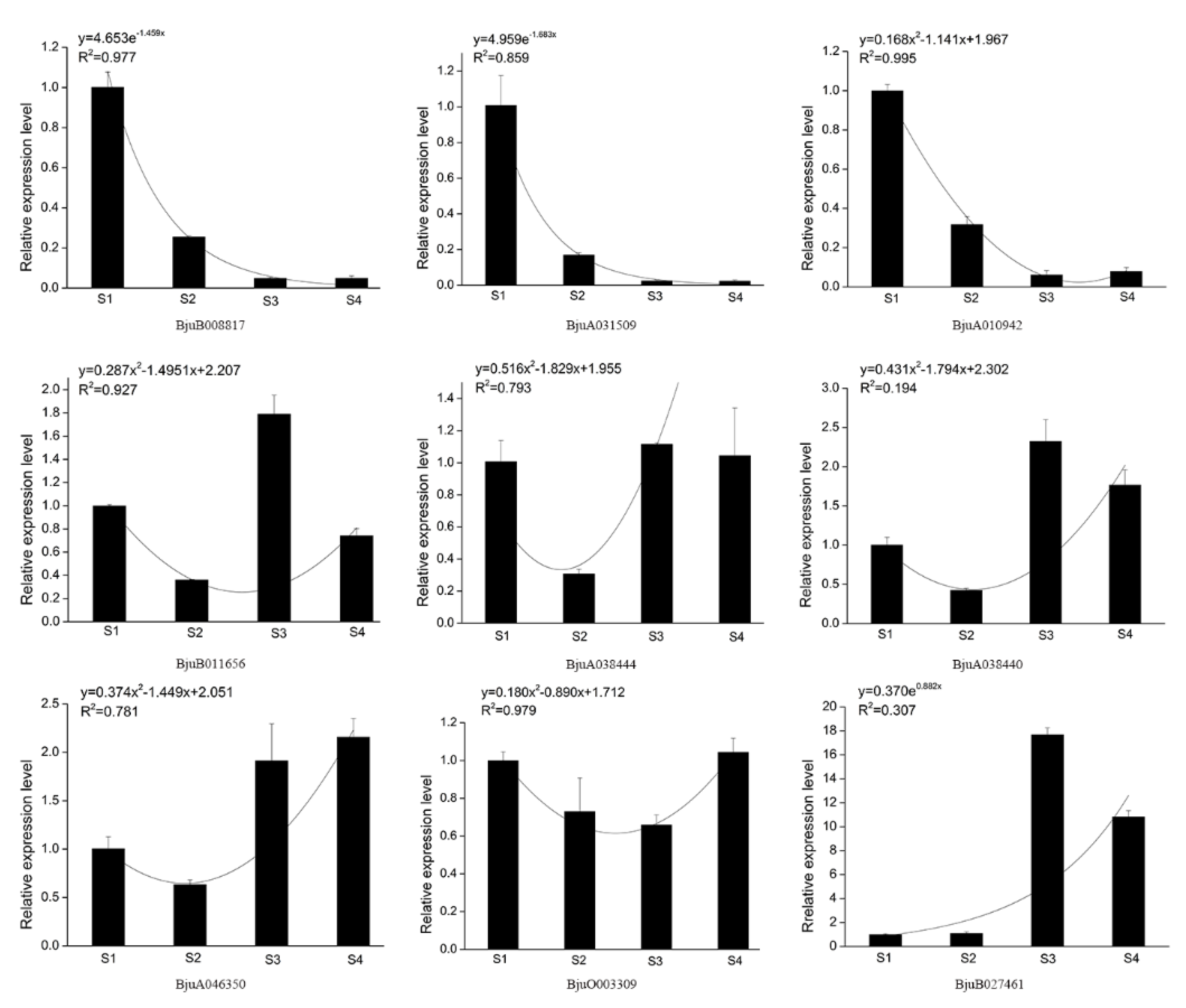
2.6. Expression Analysis and Validation of XTH Using qPCR
3. Results
3.1. Specificity and Efficiency in Amplification of Candidate Reference Genes
3.2. Evaluation of Expression Stability of Reference Genes
3.2.1. Cq Value of Candidate Reference Genes
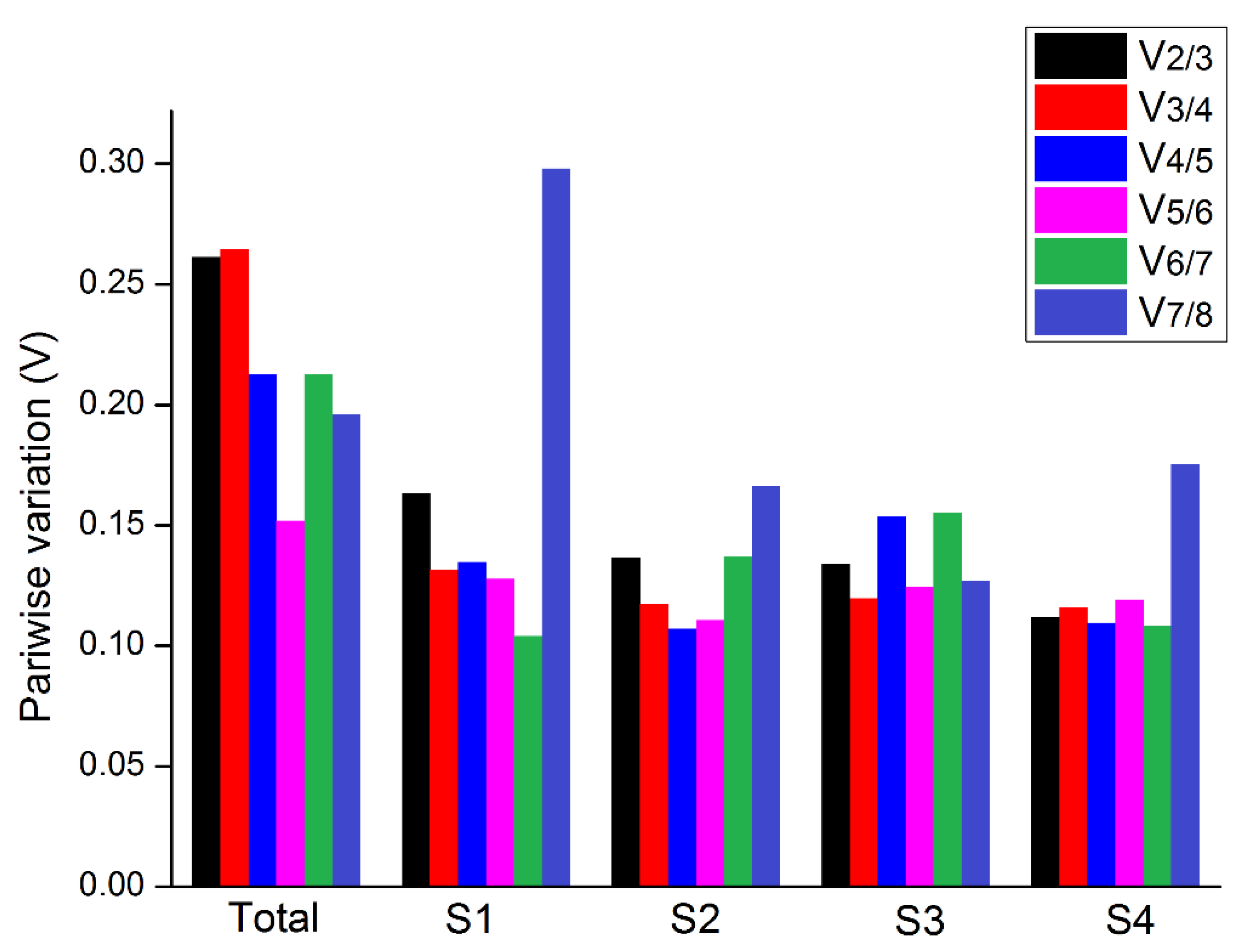
3.2.2. geNorm Software Analysis
3.2.3. NormFinder Software Analysis
3.2.4. BestKeeper Software Analysis
3.2.5. RefFinder Software Analysis
3.3. Determination of the Optimal Number of Reference Genes
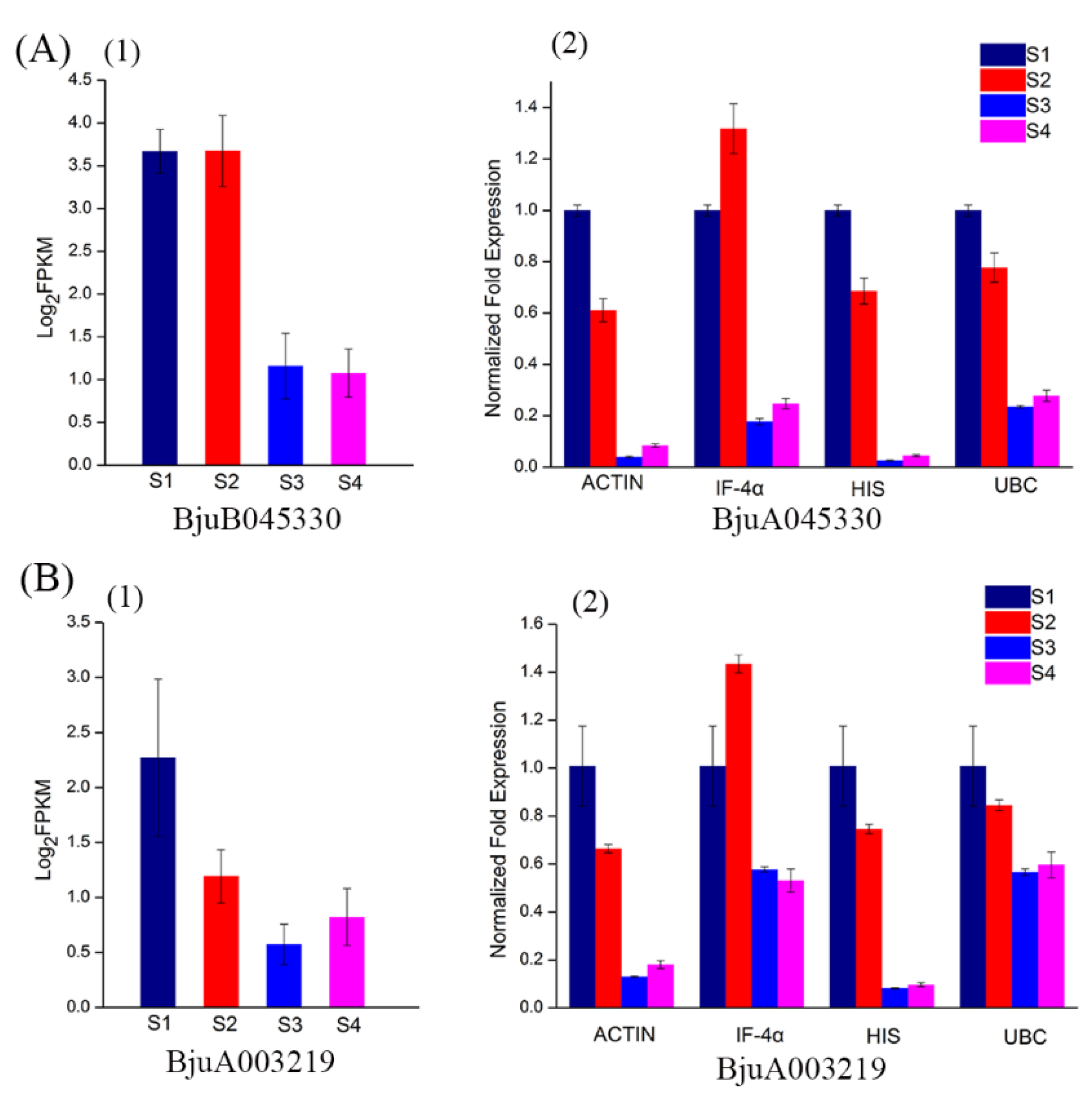
3.4. Validation of Reference Genes Using qPCR
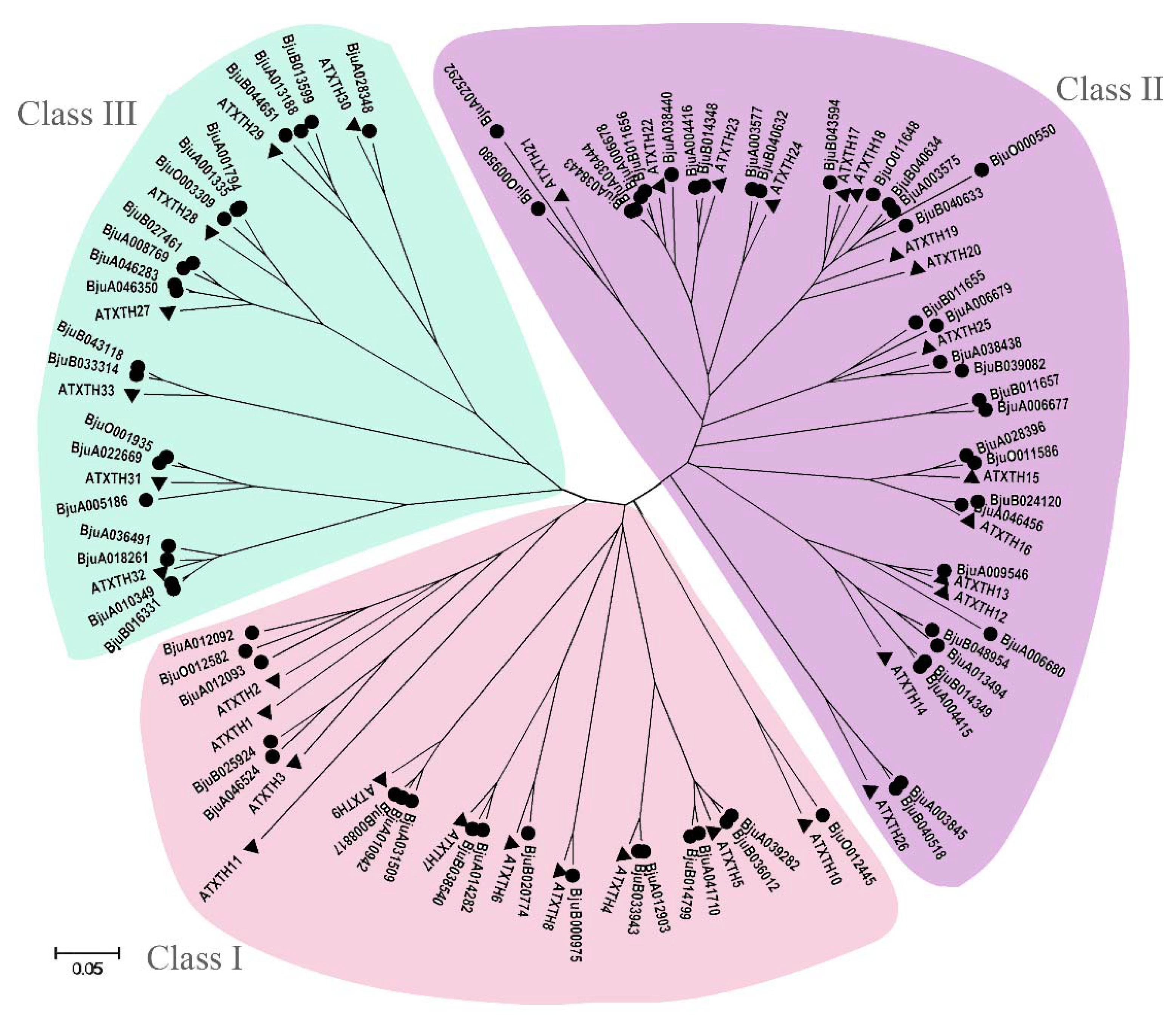
3.5. Identification and Phylogenetic Analysis of XTH Gene Family in B. juncea
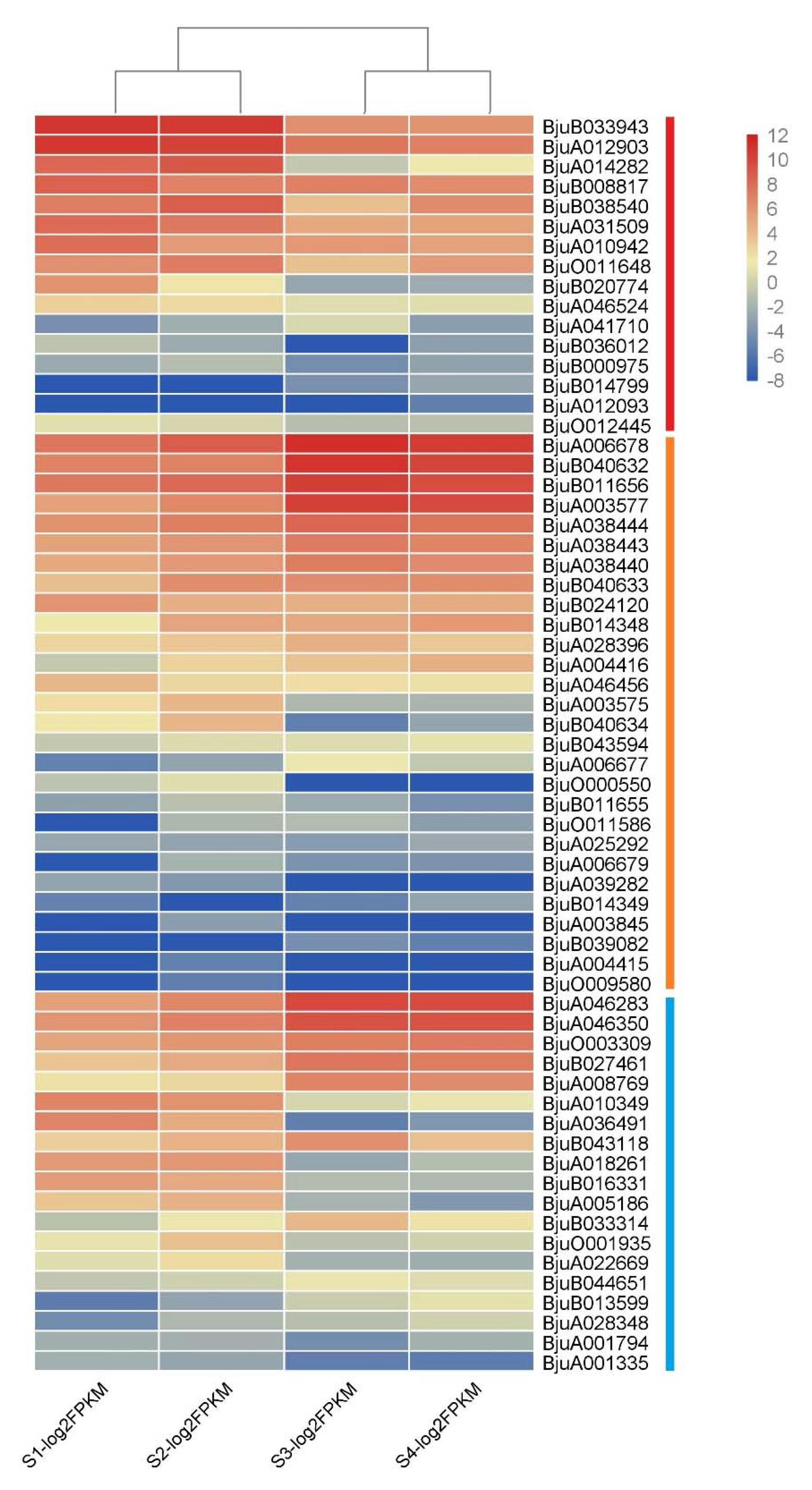
3.6. Gene Expression Analysis of Mustard XTH Family
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Zhang, Y.; Han, X.; Chen, S.; Zheng, L.; He, X.; Liu, M.; Qiao, G.; Wang, Y.; Zhuo, R. Selection of suitable reference genes for quantitative real-time PCR gene expression analysis in Salix matsudana under different abiotic stresses. Sci. Rep. 2017, 7, 40290. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Qi, X.; Yan, H.; Huang, L.; Nie, G.; Zhang, X. Reference gene selection for quantitative real-time reverse-transcriptase PCR in annual ryegrass (Lolium multiflorum) subjected to various abiotic stresses. Molecules 2018, 23, 172. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Q.; Wang, F.; Li, M.Y.; Ma, J.; Tan, G.F.; Xiong, A.S. Selection of suitable reference genes for qPCR normalization under abiotic stresses in Oenanthe javanica (BI.) DC. PLoS ONE 2014, 9, e92262. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Peng, X.; Liu, Y.; Li, Y.; Luo, Y.; Wang, X.; Tang, H. Evaluation of suitable reference genes for qRT-PCR normalization in strawberry (Fragaria × ananassa) under different experimental conditions. BMC Mol. Biol. 2018, 19, 8. [Google Scholar] [CrossRef] [PubMed]
- Yperman, J.; Visscher, G.D.; Holvoet, P.; Flameng, W. Beta-actin cannot be used as a control for gene expression in ovine interstitial cells derived from heart valves. J. Heart Valve Dis. 2004, 13, 848–853. [Google Scholar] [CrossRef]
- Guo, R.; Guo, H.; Zhang, Q.; Guo, M.; Xu, Y.; Zeng, M.; Lv, P.; Chen, X.; Yang, M. Evaluation of reference genes for RT-qPCR analysis in wild and cultivated Cannabis. Biosci. Biotechnol. Biochem. 2018, 82, 1902–1910. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Wu, Z.; Jiang, F. Selection and validation of garlic reference genes for quantitative real-time PCR normalization. Plant Cell Tiss. Organ Cult. 2015, 122, 435–444. [Google Scholar] [CrossRef]
- Andrade, L.M.; Brito, M.; Junior, R.P.; Marchiori, P.E.R.; Martins, P.M.; Ribeiro, R.V.; Creste, S. Reference genes for normalization of qPCR assays in sugarcane plants under water deficit. Plant Methods 2017, 13, 28. [Google Scholar] [CrossRef] [PubMed]
- Sudhakar Reddy, P.; Srinivas Reddy, D.; Sivasakthi, K.; Bhatnagar-Mathur, P.; Vadez, V.; Sharma, K.K. Evaluation of Sorghum [Sorghum bicolor (L.)] Reference genes in various tissues and under abiotic stress conditions for quantitative real-time PCR data normalization. Front. Plant Sci. 2016, 7, 529. [Google Scholar] [CrossRef]
- Vissenberg, K.; Oyama, M.; Osato, Y.; Yokoyama, R.; Verbelen, J.P.; Nishitani, K. Differential expression of AtXTH17, AtXTH18, AtXTH19 and AtXTH20 genes in Arabidopsis roots. Physiological roles in specification in cell wall construction. Plant Cell Physiol. 2005, 46, 192–200. [Google Scholar] [CrossRef]
- Catalá, C.; Rose, J.K.; Bennett, A.B. Auxin regulation and spatial localization of an endo-1,4-beta-D-glucanase and a xyloglucan endotransglycosylase in expanding tomato hypocotyls. Plant J. 1997, 12, 417–426. [Google Scholar] [CrossRef] [PubMed]
- Hyodo, H.; Yamakawa, S.; Takeda, Y.; Tsuduki, M.; Yokota, A.; Nishitani, K.; Kouchi, T. Active gene expression of a xyloglucan endotransglucosylase/hydrolase gene, XTH9, in inflorescence apices is related to cell elongation in Arabidopsis thaliana. Plant Mol. Biol. 2003, 54, 473–482. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.; Zhang, Z.; Rao, J.; Huber, D.J.; Lv, J.; Hou, Y.; Song, K. Identification of xyloglucan endotransglucosylase/hydrolase genes (XTHs) and their expression in persimmon fruit as influenced by 1-methylcyclopropene and gibberellic acid during storage at ambient temperature. Food Chem. 2013, 138, 471–477. [Google Scholar] [CrossRef]
- Shi, H.; Wang, L.L.; Sun, L.T.; Dong, L.L.; Liu, B.; Chen, L.P. Cell division and endoreduplication play important roles in stem swelling of tuber mustard (Brassica juncea Coss. var. tumida Tsen et Lee). Plant Biol. 2012, 14, 956–963. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.K.; Kim, J.E.; Park, J.A.; Eom, T.J.; Kim, W.T. Constitutive expression of abiotic stress-inducible hot pepper CaXTH3, which encodes a xyloglucan endotransglucosylase/hydrolase homolog, improves drought and salt tolerance in transgenic Arabidopsis plants. FEBS Lett. 2006, 580, 3136–3144. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.Y.; Seo, Y.S.; Kim, S.J.; Kim, W.T.; Shin, J.S. Constitutive expression of CaXTH3, ahotpepperxyloglucan endotransglucosylase/hydrolase, enhanced tolerance to salt and drought stresses without phenotypic defects in tomato plants (Solanum lycopersicum cv. Dotaerang). Plant Cell Rep. 2011, 30, 867–877. [Google Scholar] [CrossRef]
- Han, Y.; Wang, W.; Sun, J.; Ding, M.; Zhao, R.; Deng, S.; Wang, F.; Hu, Y.; Wang, Y.; Lu, Y.; et al. Populus euphratica XTH overexpression enhances salinity tolerance by the development of leaf succulence in transgenic tobacco plants. J. Exp. Bot. 2013, 64, 4225–4238. [Google Scholar] [CrossRef]
- Vandesompele, J.; De Preter, K.; Pattyn, F.; Poppe, B.; Van Roy, N.; De Paepe, A.; Speleman, F. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. 2002, 15. [Google Scholar] [CrossRef]
- Andersen, C.L.; Jensen, J.L.; Orntoft, T.F. Normalization of real-time quantitative reverse transcription-PCR data: A model-based variance estimation approach to identify genes suited for normalization, applied to bladder and colon cancer data sets. Cancer Res. 2004, 64, 5245–5250. [Google Scholar] [CrossRef]
- Pfaff, M.W.; Tichopad, A.; Prgomet, C.; Neuvians, T.P. Determination of stable housekeeping genes, differentially regulated target genes and sample integrity, BestKeeper–Excel–based tool using pair-wise correlations. Biotechnol. Lett. 2004, 26, 509–515. [Google Scholar] [CrossRef]
- Xie, F.L.; Xiao, P.; Chen, D.; Xu, L.; Zhang, B. miRDeepFinder: A miRNA analysis tool for deep sequencing of plant small RNAs. Plant Mol. Biol. 2012, 80, 75–84. [Google Scholar] [CrossRef]
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001, 29, e45. [Google Scholar] [CrossRef] [PubMed]
- Ramakers, C.; Ruijter, J.M.; Deprez, R.H.; Moorman, A.F. Assumption-free analysis of quantitative real-time polymerase chain reaction (PCR) data. Neurosci. Lett. 2003, 339, 62–66. [Google Scholar] [CrossRef]
- Lutova, L.A.; Dolgikh, E.A.; Dodueva, I.E.; Osipova, M.A.; Il’ina, E.L. Investigation of systemic control of plant cell division and differentiation in the model of tumor growth in radish. Genetika 2012, 44, 1075–1083. [Google Scholar] [CrossRef]
- Li, Y.; Yu, J.Q.; Ye, Q.J.; Zhu, Z.J.; Guo, Z.J. Expression of CycD3 is transiently increased by pollination and N-(2-chloro-4-pyridyl)-N’-phenylurea in ovaries of Lagenaria leucantha. J. Exp. Bot. 2003, 54, 1245–1251. [Google Scholar] [CrossRef] [PubMed]
- Mallona, I.; Lischewski, S.; Weiss, J.; Hause, B.; Egea-Cortines, M. Validation of reference genes for quantitative real-time PCR during leaf and flower development in Petunia hybrida. BMC Plant Biol. 2010, 10, 4. [Google Scholar] [CrossRef]
- Kozera, B.; Rapacz, M. Reference genes in real-time PCR. J. Appl. Genet. 2013, 54, 391–406. [Google Scholar] [CrossRef]
- Nicot, N.; Hausman, J.F.; Hoffmann, L.; Evers, D. Housekeeping gene selection for real-time RT-PCR normalization in potato during biotic and abiotic. J. Exp. Bot. 2005, 56, 2907–2914. [Google Scholar] [CrossRef]
- Wu, J.; Zhang, H.; Liu, L.; Li, W.; Wei, Y.; Shi, S. Validation of reference genes for RT-qPCR studies of gene expression in preharvest and postharvest longan fruits under different experimental conditions. Front. Plant Sci. 2016, 7, 780. [Google Scholar] [CrossRef]
- Tian, C.; Jiang, Q.; Wang, F.; Wang, G.L.; Xu, Z.S.; Xiong, A.S. Selection of suitable reference genes for qPCR normalization under abiotic stresses and hormone stimuli in carrot leaves. PLoS ONE 2015, 10, e0117569. [Google Scholar] [CrossRef]
- Shivhare, R.; Lata, C. Selection of suitable reference genes for assessing gene expression in pearl millet under different abiotic stresses and their combinations. Sci. Rep. 2016, 6, 23036. [Google Scholar] [CrossRef] [PubMed]
- Løvdal, T.; Lillo, C. Reference gene selection for quantitative real-time PCR normalization in tomato subjected to nitrogen, cold, and light stress. Anal. Biochem. 2009, 387, 238–242. [Google Scholar] [CrossRef] [PubMed]
- Jain, M.; Nijhawan, A.; Tyagi, A.K.; Khurana, J.P. Validation of housekeeping genes as internal control for studying gene expression in rice by quantitative real-time PCR. Biochem. Biophys. Res. Commun. 2006, 345, 646–651. [Google Scholar] [CrossRef] [PubMed]
- Li, M.Y.; Wang, F.; Jiang, Q.; Wang, G.L.; Tian, C.; Xiong, A.S. Validation and comparison of reference genes for qPCR normalization of celery (Apium graveolens) at different development stages. Front. Plant Sci. 2016, 7, 313. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.J.; Tian, C.; Jiang, Q.; Li, X.H.; Zhuang, J. Selection of suitable reference genes for qRT-PCR normalization during leaf development and hormonal stimuli in tea plant (Camellia sinensis). Sci. Rep. 2016, 6, 19748. [Google Scholar] [CrossRef] [PubMed]
- Obrero, A.; Die, J.V.; Román, B.; Gómez, P.; Nadal, S.; González-Verdejo, C.I. Selection of reference genes for gene expression studies in zucchini (Cucurbita pepo) using qPCR. J. Agric. Food Chem. 2011, 59, 5402–5411. [Google Scholar] [CrossRef] [PubMed]
- Behar, H.; Graham, S.W.; Brumer, H. Comprehensive cross-genome survey and phylogeny of glycoside hydrolase family 16 members reveals the evolutionary origin of EG16 and XTH proteins in plant lineages. Plant J. 2018, 95, 1114–1128. [Google Scholar] [CrossRef] [PubMed]
- Rose, J.K.; Braam, J.; Fry, S.C.; Nishitani, K. The XTH family of enzymes involved in xyloglucan endotransglucosylation and endohydrolysis: Current perspectives and a new unifying nomenclature. Plant Cell Physiol. 2002, 43, 1421–1435. [Google Scholar] [CrossRef]
- Yokoyama, R.; Rose, J.K.; Nishitani, K. A surprising diversity and abundance of xyloglucan endotransglucosylase/hydrolases in rice. Classification and expression analysis. Plant Physiol. 2004, 134, 1088–1099. [Google Scholar] [CrossRef]
- Song, L.; Valliyodan, B.; Prince, S.; Wan, J.; Nguyen, H.T. Characterization of the XTH gene family: New insight to the roles in soybean flooding tolerance. Int. J. Mol. Sci. 2018, 19, 2705. [Google Scholar] [CrossRef]
- Wang, M.; Xu, Z.; Ding, A.; Kong, Y. Genome-wide identification and expression profiling analysis of the xyloglucan endotransglucosylase/hydrolase gene family in tobacco (Nicotiana tabacum L.). Genes 2018, 9, 273. [Google Scholar] [CrossRef] [PubMed]
- Lysak, M.A.; Koch, M.A.; Pecinka, A.; Schubert, I. Chromosome triplication found across the tribe Brassiceae. Genome Res. 2005, 15, 516–525. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, H.; Wang, J.; Sun, R.; Wu, J.; Liu, S.; Bai, Y.; Mun, J.H.; Bancroft, I.; Cheng, F.; et al. The genome of the mesopolyploid crop species Brassica rapa. Nat. Genet. 2011, 43, 1035–1039. [Google Scholar] [CrossRef] [PubMed]
- Nagaharu, U. Genome analysis in Brassica with special reference to the experimental formation of B. napus and peculiar mode of fertilization. Jpn. J. Bot. 1935, 7, 389–452. [Google Scholar]
- Chen, A.H.; Chai, Y.R.; Li, J.N.; Chen, L. Molecular cloning of two genes encoding cinnamate 4-hydroxylase (C4H) from oilseed rape (Brassica napus). J. Biochem. Mol. Biol. 2007, 40, 247–260. [Google Scholar] [CrossRef] [PubMed]
- Shopan, J.; Mou, H.; Zhang, L.; Zhang, C.; Ma, W.; Walsh, J.A.; Hu, Z.; Yang, J.; Zhang, M. Eukaryotic translation initiation factor 2B-beta (eIF2Bβ), a new class of plant virus resistance gene. Plant J. 2017, 90, 929–940. [Google Scholar] [CrossRef] [PubMed]
- Van Sandt, V.S.; Suslov, D.; Verbelen, J.P.; Vissenberg, K. Xyloglucan endotransglucosylase activity loosens a plant cell wall. Ann. Bot. 2007, 100, 1467–1473. [Google Scholar] [CrossRef]
- Kaku, T.; Tabuchi, A.; Wakabayashi, K.; Hoson, T. Xyloglucan oligosaccharides cause cell wall loosening by enhancing xyloglucan endotransglucosylase/hydrolase activity in azuki bean epicotyls. Plant Cell Physiol. 2004, 45, 77–82. [Google Scholar] [CrossRef]
- Witasari, L.D.; Huang, F.C.; Hoffmann, T.; Rozhon, W.; Fry, S.C.; Schwab, W. Higher expression of the strawberry xyloglucan endotransglucosylase/hydrolase genes FvXTH9 and FvXTH6 accelerates fruit ripening. Plant J. 2019, 100, 1237–1253. [Google Scholar] [CrossRef]
- Lee, J.; Burns, T.H.; Light, G.; Sun, Y.; Fokar, M.; Kasukabe, Y.; Fujisawa, K.; Maekawa, Y.; Allen, R.D. Xyloglucan endotransglycosylase/hydrolase genes in cotton and their role in fiber elongation. Planta 2010, 232, 1191–1205. [Google Scholar] [CrossRef]
- Miedes, E.; Suslov, D.; Vandenbussche, F.; Kenobi, K.; Ivakov, A.; Van Der Straeten, D.; Lorences, E.P.; Mellerowicz, E.J.; Verbelen, J.P.; Vissenberg, K. Xyloglucan endotransglucosylase/hydrolase (XTH) overexpression affects growth and cell wall mechanics in etiolated Arabidopsis hypocotyls. J. Exp. Bot. 2013, 64, 2481–2497. [Google Scholar] [CrossRef] [PubMed]
- Becnel, J.; Natarajan, M.; Kipp, A.; Braam, J. Developmental expression patterns of Arabidopsis XTH genes reported by transgenes and genevestigator. Plant Mol. Biol. 2006, 61, 451–467. [Google Scholar] [CrossRef] [PubMed]

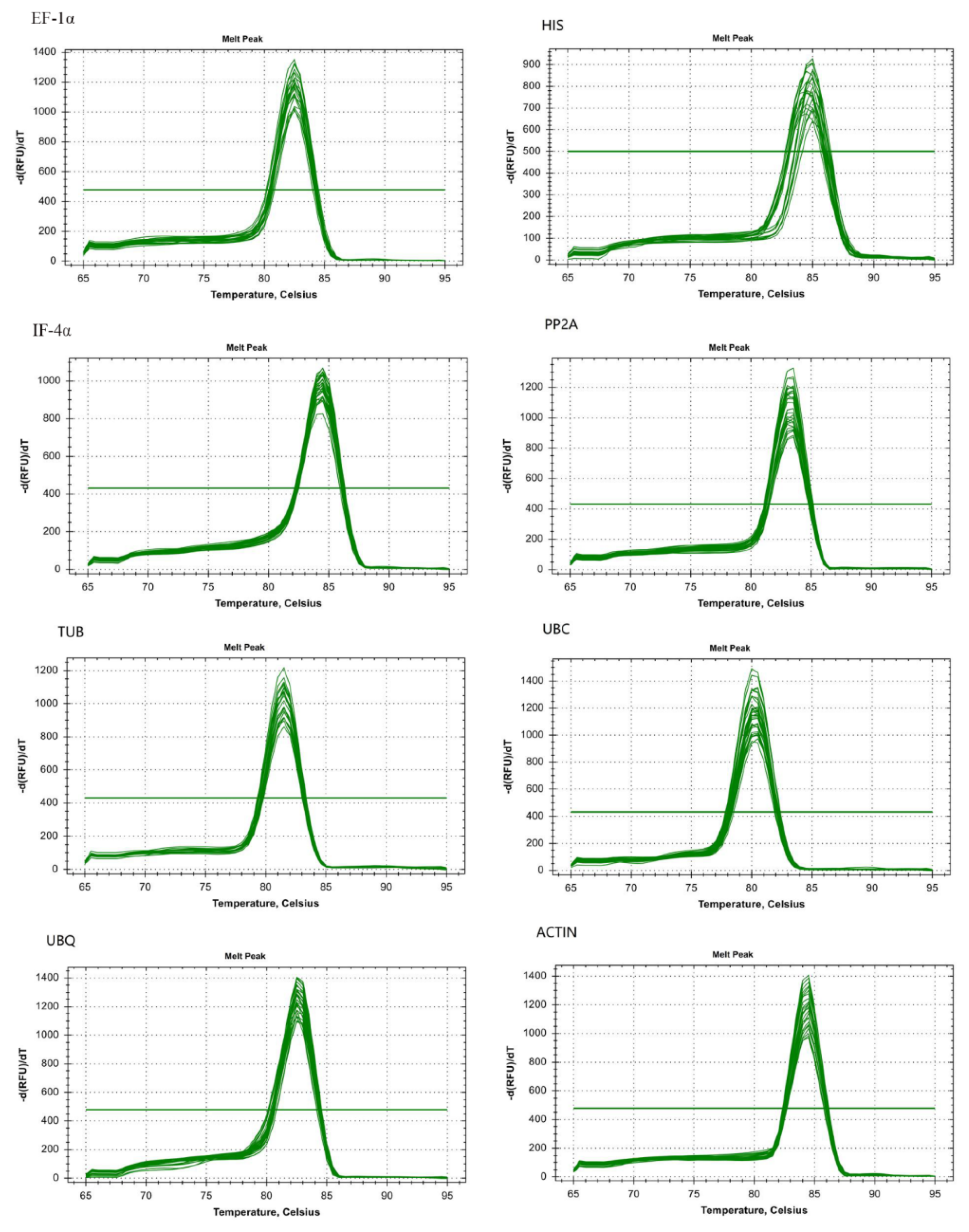
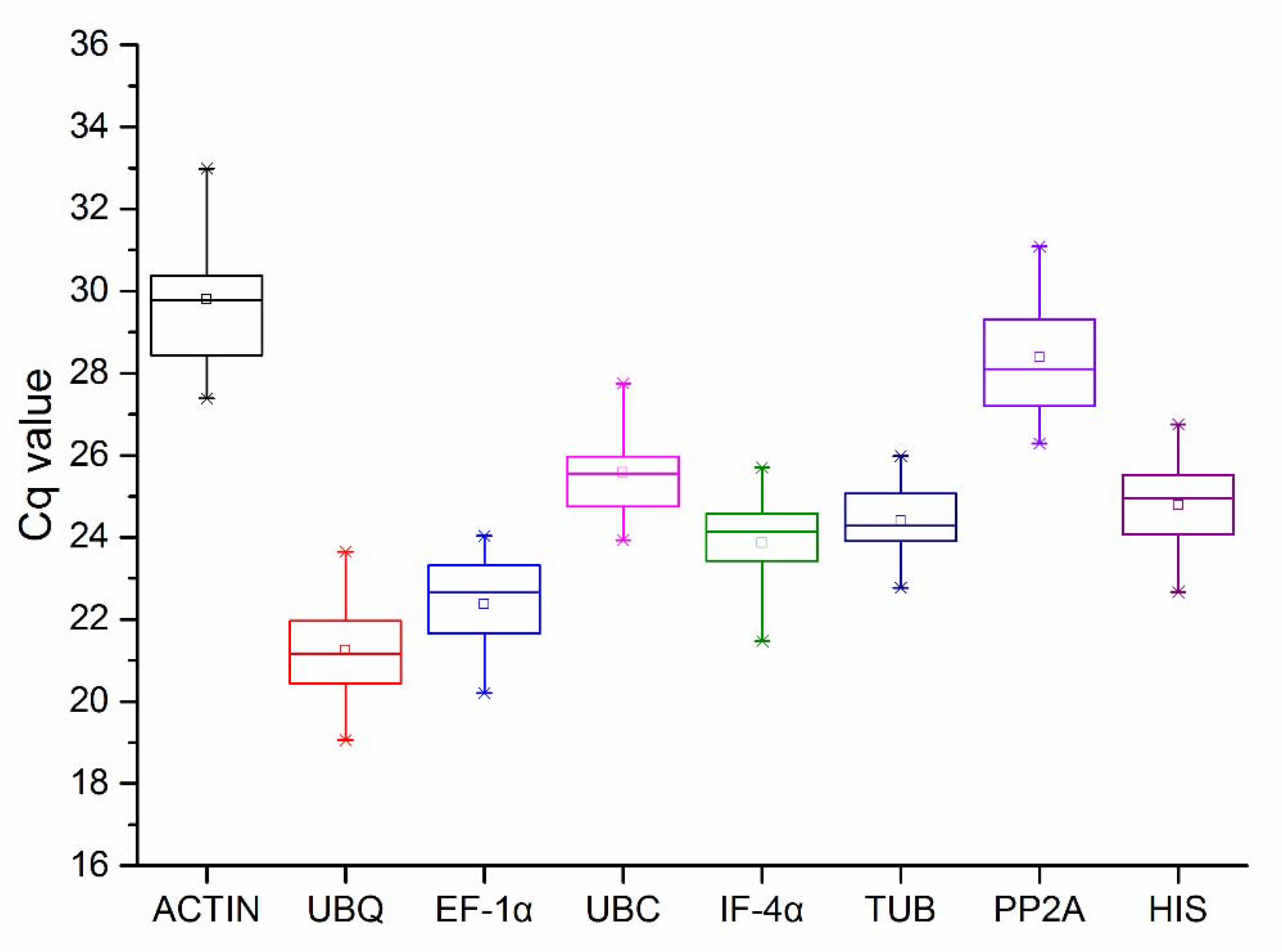
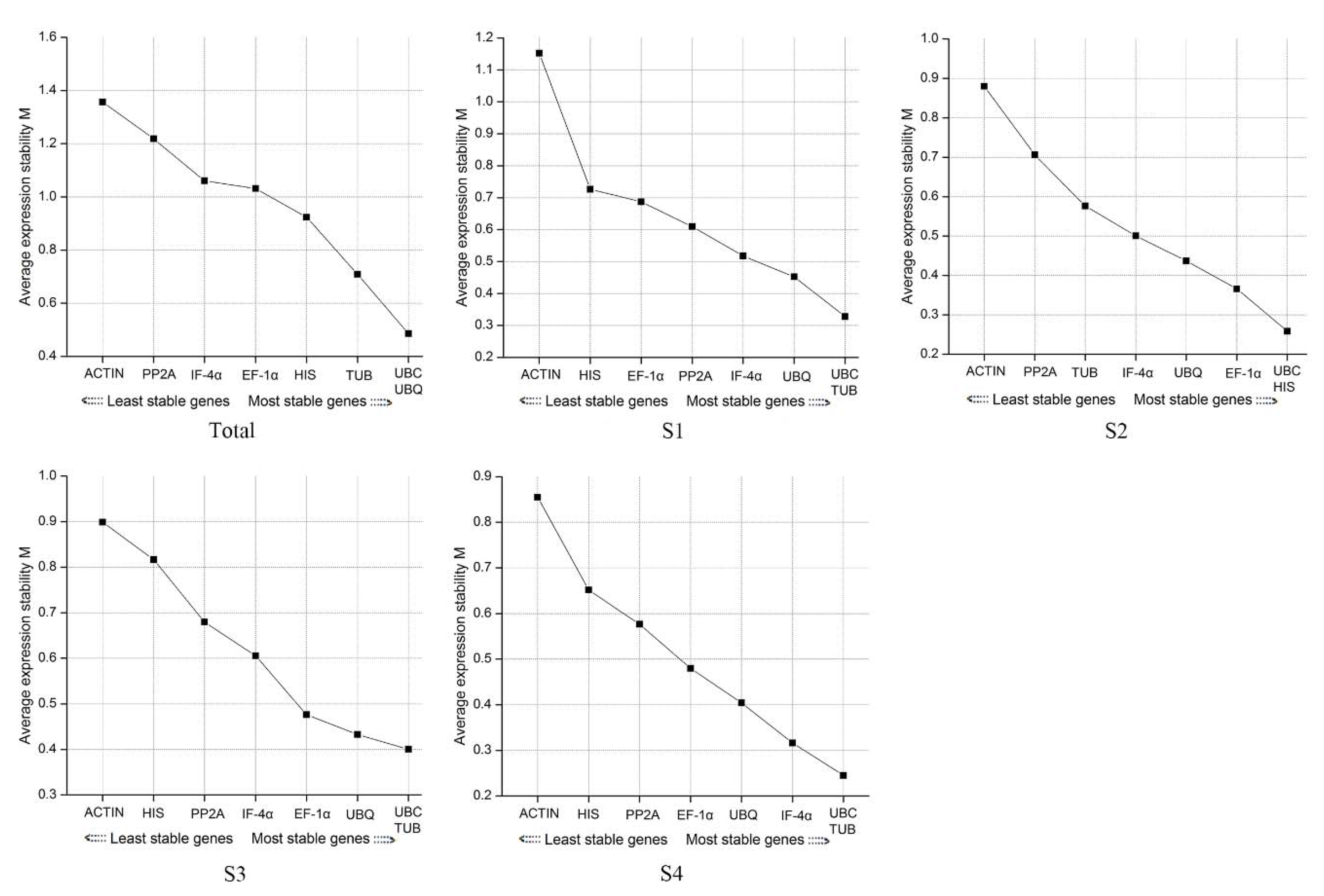
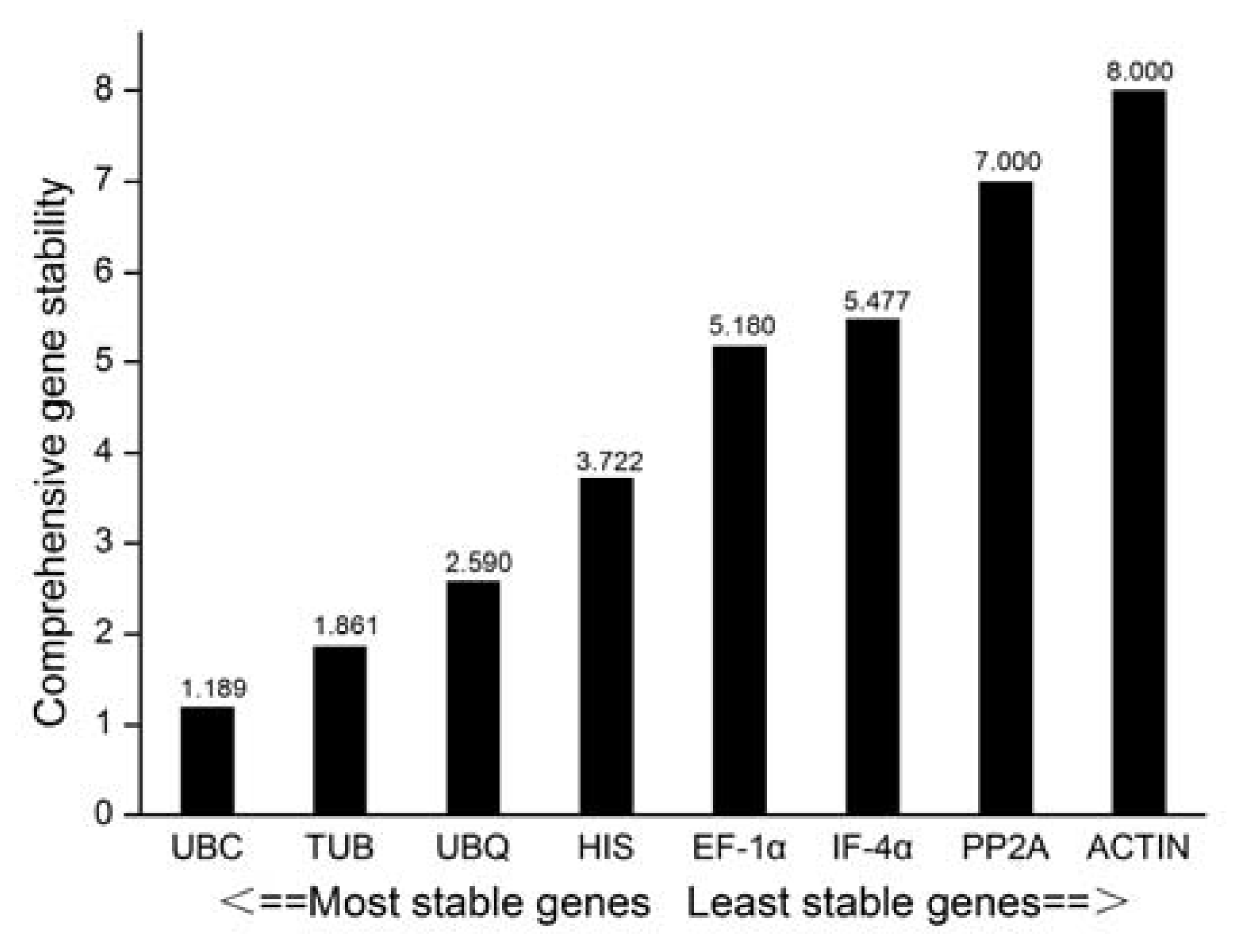
| Gene Symbol | Gene Name | Primer Sequence (5’-3’) | Amplicon Size (bp) | Correlation Coefficient |
|---|---|---|---|---|
| EF-1α | Elongation factor -1α gene | CGTCTGCTTAGTGAACCTGCTC/ GAAGGAGCGAATGTCACAACCA | 112 | 0.996 |
| HIS | Histone | AGGGAAAGCCGCTCCACTTC/ TCGTAACCCTCTTGGCGTGAAT | 152 | 0.996 |
| IF-4α | Eukaryotic initiation factor 4 alpha | CAAGCCGAGCCTGCGATCAT/ TCGTGTTCTGGTCCATGTCTCC | 121 | 0.996 |
| PP2A | Protein phosphatase 2A | AAGCCGAGCCTTCCATCATAGC/ ACCACCACCACCACTCATTGC | 197 | 0.992 |
| TUB | Tubulin gene | GCGTCTTGTCCGTGAGATTGC/ GCCGAGATGAGGTGGTTAAGGT | 109 | 0.994 |
| UBC | Ubiquitin C gene | GCCATCACTCAGAGCGTCATCT/ AAGGAGACTGTGTAGGACCAAGAA | 100 | 0.999 |
| UBQ | Polyubiquitin 10 gene | TGCGTCTACCACTTCAGGATGT/ TCTGCTGGTCTGGAGGAATGC | 145 | 0.992 |
| ACTIN | Actin gene | ATCGTCTGTGACAACGGTAC/ ATGGAGGGTGATGAGATTCAGC | 267 | 0.992 |
| Group | Rank | geNorm | NormFinder | BestKeeper | ||||
|---|---|---|---|---|---|---|---|---|
| Gene | Stability | Gene Name | Stability Value | Gene | SD [±Cq] | CV [%Cq] | ||
| Total | 1 | UBC | 0.49 | UBC | 0.31 | TUB | 0.67 | 2.73 |
| 2 | UBQ | 0.49 | TUB | 0.39 | UBC | 0.77 | 3.02 | |
| 3 | TUB | 0.71 | UBQ | 0.48 | UBQ | 0.8 | 3.74 | |
| 4 | HIS | 0.92 | HIS | 0.59 | ACTIN | 0.86 | 2.91 | |
| 5 | EF-1α | 1.03 | IF-4α | 0.68 | HIS | 0.87 | 3.51 | |
| 6 | IF-4α | 1.06 | EF-1α | 0.7 | EF-1α | 0.9 | 4.01 | |
| 7 | PP2A | 1.22 | PP2A | 0.97 | IF-4α | 0.92 | 3.83 | |
| 8 | ACTIN | 1.36 | ACTIN | 1.06 | PP2A | 1 | 3.53 | |
| S1 | 1 | UBC | 0.33 | UBC | 0.11 | EF-1α | 0.28 | 1.26 |
| 2 | TUB | 0.33 | IF-4α | 0.17 | IF-4α | 0.39 | 1.61 | |
| 3 | UBQ | 0.45 | TUB | 0.26 | HIS | 0.53 | 2.13 | |
| 4 | IF-4α | 0.52 | UBQ | 0.45 | PP2A | 0.66 | 2.26 | |
| 5 | PP2A | 0.61 | EF-1α | 0.45 | UBC | 0.71 | 2.67 | |
| 6 | EF-1α | 0.69 | PP2A | 0.45 | TUB | 0.81 | 3.29 | |
| 7 | HIS | 0.73 | HIS | 0.56 | UBQ | 0.9 | 4.01 | |
| 8 | ACTIN | 1.15 | ACTIN | 1.65 | ACTIN | 1.83 | 6.05 | |
| S2 | 1 | UBC | 0.26 | IF-4α | 0.19 | TUB | 0.25 | 1.02 |
| 2 | HIS | 0.26 | UBC | 0.23 | IF-4α | 0.62 | 2.78 | |
| 3 | EF-1α | 0.37 | UBQ | 0.25 | ACTIN | 0.7 | 2.39 | |
| 4 | UBQ | 0.44 | EF-1α | 0.35 | UBQ | 0.86 | 4.09 | |
| 5 | IF-4α | 0.5 | TUB | 0.39 | UBC | 0.93 | 3.75 | |
| 6 | TUB | 0.58 | HIS | 0.42 | EF-1α | 1.05 | 4.9 | |
| 7 | PP2A | 0.71 | PP2A | 0.62 | HIS | 1.05 | 4.37 | |
| 8 | ACTIN | 0.88 | ACTIN | 0.93 | PP2A | 1.14 | 3.89 | |
| S3 | 1 | UBC | 0.4 | EF-1α | 0.11 | EF-1α | 0.23 | 0.96 |
| 2 | TUB | 0.4 | UBC | 0.27 | UBC | 0.36 | 1.4 | |
| 3 | UBQ | 0.43 | PP2A | 0.33 | PP2A | 0.5 | 1.85 | |
| 4 | EF-1α | 0.48 | IF-4α | 0.38 | UBQ | 0.51 | 2.39 | |
| 5 | IF-4α | 0.61 | UBQ | 0.46 | TUB | 0.6 | 2.37 | |
| 6 | PP2A | 0.68 | TUB | 0.55 | IF-4α | 0.62 | 2.54 | |
| 7 | HIS | 0.82 | HIS | 0.68 | HIS | 0.73 | 2.84 | |
| 8 | ACTIN | 0.9 | ACTIN | 0.68 | ACTIN | 0.84 | 2.84 | |
| S4 | 1 | UBC | 0.25 | UBC | 0.08 | EF-1α | 0.38 | 1.68 |
| 2 | TUB | 0.25 | TUB | 0.23 | PP2A | 0.57 | 2.09 | |
| 3 | IF-4α | 0.32 | EF-1α | 0.27 | IF-4α | 0.58 | 2.35 | |
| 4 | UBQ | 0.4 | IF-4α | 0.32 | UBC | 0.6 | 2.39 | |
| 5 | EF-1α | 0.48 | PP2A | 0.37 | TUB | 0.68 | 2.81 | |
| 6 | PP2A | 0.58 | UBQ | 0.37 | UBQ | 0.83 | 3.99 | |
| 7 | HIS | 0.65 | HIS | 0.55 | HIS | 0.94 | 3.83 | |
| 8 | ACTIN | 0.86 | ACTIN | 0.96 | ACTIN | 1.05 | 3.61 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, M.; Xie, F.; He, Q.; Li, J.; Liu, J.; Sun, B.; Luo, Y.; Zhang, Y.; Chen, Q.; Zhang, F.; et al. Expression Analysis of XTH in Stem Swelling of Stem Mustard and Selection of Reference Genes. Genes 2020, 11, 113. https://doi.org/10.3390/genes11010113
Li M, Xie F, He Q, Li J, Liu J, Sun B, Luo Y, Zhang Y, Chen Q, Zhang F, et al. Expression Analysis of XTH in Stem Swelling of Stem Mustard and Selection of Reference Genes. Genes. 2020; 11(1):113. https://doi.org/10.3390/genes11010113
Chicago/Turabian StyleLi, Mengyao, Fangjie Xie, Qi He, Jie Li, Jiali Liu, Bo Sun, Ya Luo, Yong Zhang, Qing Chen, Fen Zhang, and et al. 2020. "Expression Analysis of XTH in Stem Swelling of Stem Mustard and Selection of Reference Genes" Genes 11, no. 1: 113. https://doi.org/10.3390/genes11010113
APA StyleLi, M., Xie, F., He, Q., Li, J., Liu, J., Sun, B., Luo, Y., Zhang, Y., Chen, Q., Zhang, F., Gong, R., Wang, Y., Wang, X., & Tang, H. (2020). Expression Analysis of XTH in Stem Swelling of Stem Mustard and Selection of Reference Genes. Genes, 11(1), 113. https://doi.org/10.3390/genes11010113








