Transmission of Functional, Wild-Type Mitochondria and the Fittest mtDNA to the Next Generation: Bottleneck Phenomenon, Balbiani Body, and Mitophagy
Abstract
1. Introduction
2. Categories (Types) of Animal Ovaries
3. Selection of Highly Active (“Healthy”) Mitochondria and the Balbiani Body—The High Energy Pathway
- The early germline cells receive mitochondria in various functional states. They are not selected but partitioned to the individual germline cells stochastically as assumed in the bottleneck phenomenon.
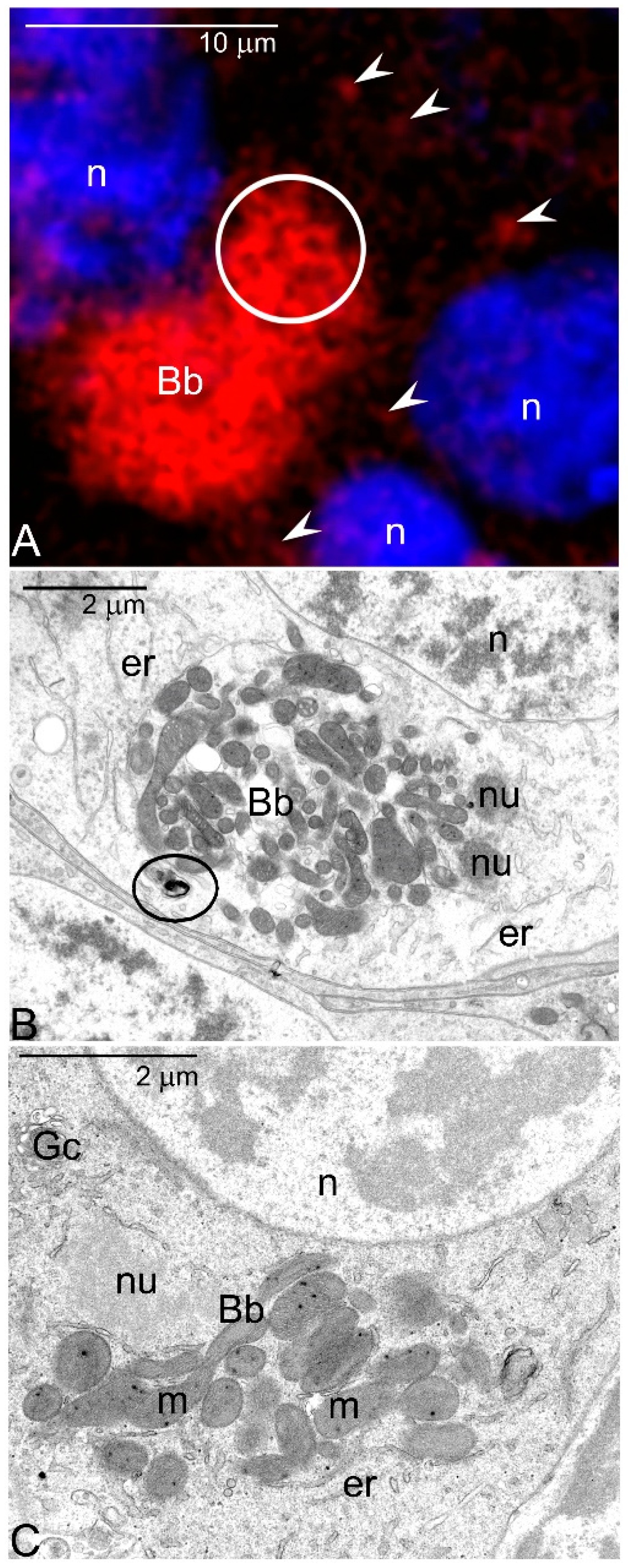
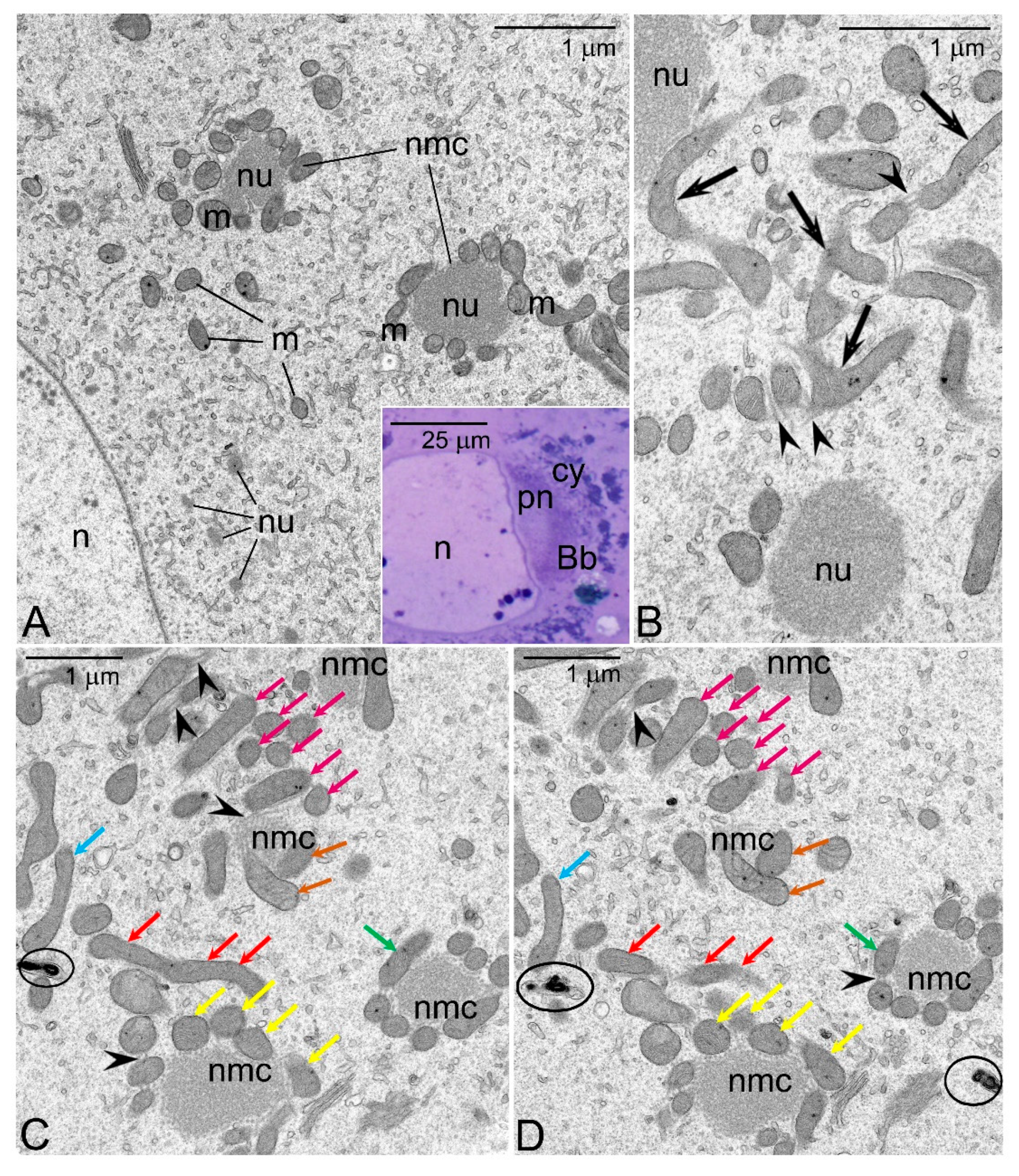
- As oogenesis progresses, the mitochondria present in each germline cell gather next to the nucleus and form the Bb. Within the Bb, mitochondria multiply and fuse forming an extensive local network.
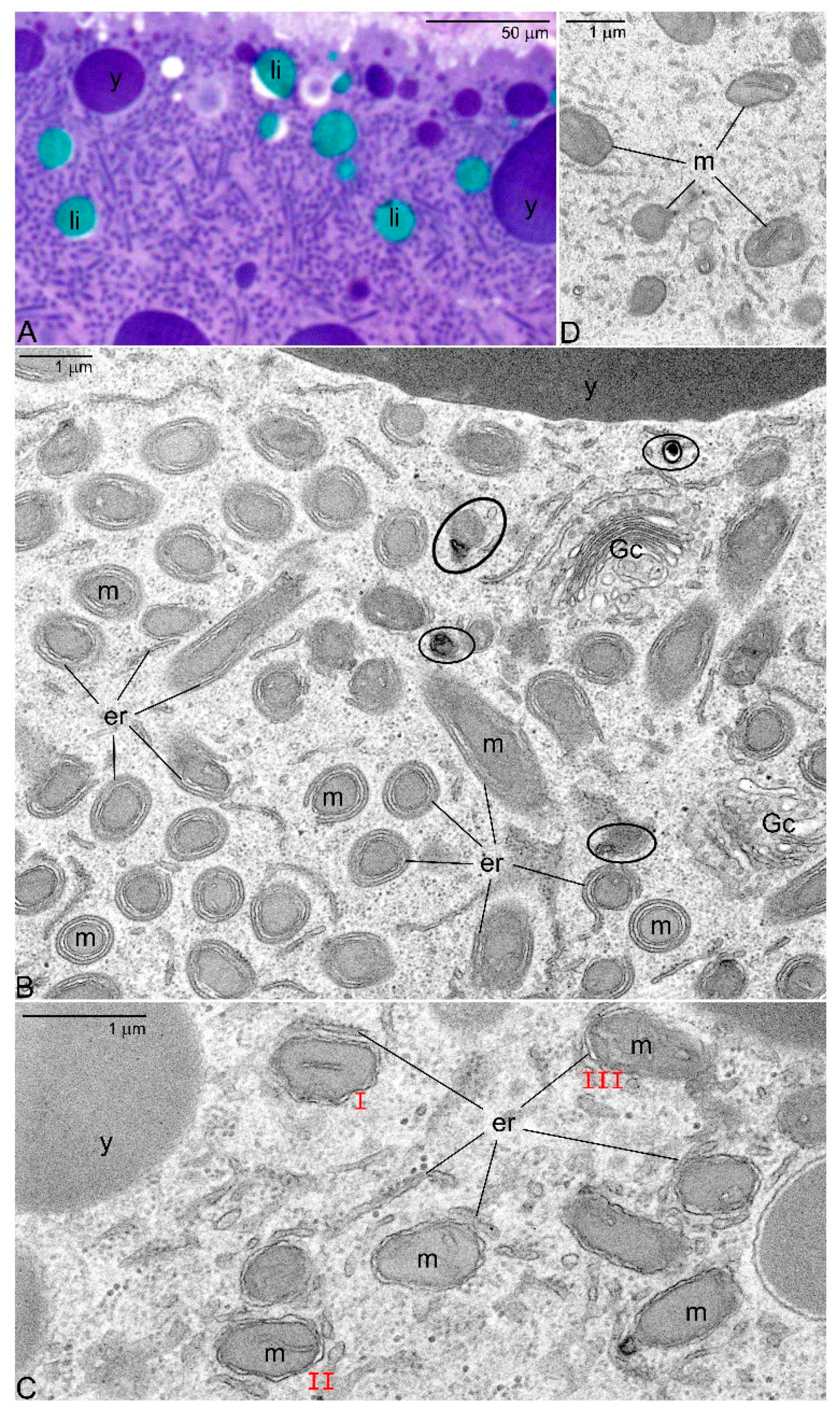
- Dysfunctional mitochondrial units (containing mutated mtDNA) are separated from the network and eliminated by mitophagy.
- After dispersal of the Bb selected (healthy) mitochondria populate the ooplasm.
- Labelling with various antibodies and fluorescent probes showed that in the oocytes of a sea urchin, Paracentrotus lividus highly active mitochondria accumulate next to the germinal vesicle and here colocalize with vesicular acidic organelles (autophagosomes, autolysosomes) [35,36]. Thus, the arrangement of organelles in sea urchin oocytes mimics those in Thermobia.
- Autoradiographic studies revealed that in Xenopus Bb mtDNA is replicated synchronously “in neighboring mitochondria” [32] implying that in this species several smaller mitochondrial networks may coexist within single Bb.
- In murine oocytes the number of mitochondria remaining in physical contact has been estimated to increase from 21–58% during Bb formation [37]. Moreover, reevaluation of microphotographs published elsewhere (e.g., Figure 5B,F in [37]; 1E in [38]; 7B in [39]) suggests that the Bb mitochondria are also interconnected in mammals.
- It has been recently shown, using an allele-specific FISH approach, that mitochondrial fragmentation is responsible for the removal of mutated mtDNA in the female germline cells of Drosophila [40].
4. Silencing the Mitochondrial Activity—Low Energy Pathway
5. Can High Energy and Low Energy Pathways Coexist in Related Animal Lineages?
6. Conclusions and Future Perspectives
- On the basis of our EM studies, we suggest that the “high energy” and “low energy” pathways may operate even in closely related lineages of a given taxon. This, in turn, implies that these pathways are not evolutionary conserved and are rather related to morpho-functional characteristics of the ovaries as for instance: presence/absence of the nurse cells associated with the oocyte or presence of the Bb in the ooplasm.
- It is well known that in the ovaries of both vertebrates and invertebrates, a relatively large fraction of young germline cells is eliminated via apoptosis [53,54,55,56]. We believe, in this context that the selection of the mitochondria in oocytes is a two-step process: The first step operates at the cellular level and involves selection of mitochondria and mtDNA variants. In the second, all the germline cells that still (after the first step) contain large number of mutated mtDNA are eliminated via apoptosis. This idea is in line with our EM analyses showing that in apoptotic oocytes of Thermobia, the Bbs mitochondria reveal highly altered morphology (Figure 1C).
- The mechanisms responsible for mitochondrial inheritance obviously need further studies. There are at least two fundamental questions that should be answered:
- How replication of mtDNA (and consequent multiplication of mitochondria) located within the Bb is initiated? Is nuage material implicated in this initiation?
- Is recruitment of highly functional and healthy mitochondria to the Bb guaranteed by cytoskeleton (microtubules) as suggested by Milani [8] or it is an obvious consequence of a bottleneck phenomenon?
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Westermann, B. Mitochondrial fusion and fission in cell life and death. Nat. Rev. Mol. Cell Biol. 2010, 11, 872–884. [Google Scholar] [CrossRef] [PubMed]
- Youle, R.J.; van der Bliek, A.M. Mitochondrial fission, fusion, and stress. Science 2012, 337, 1062–1065. [Google Scholar] [CrossRef] [PubMed]
- Mitra, K. Mitochondrial fission-fusion as an emerging key regulator of cell proliferation and differentiation. Bioessays 2013, 35, 955–964. [Google Scholar] [CrossRef] [PubMed]
- Friedman, J.R.; Nunnari, J. Mitochondrial form and function. Nature 2014, 505, 335–343. [Google Scholar] [CrossRef] [PubMed]
- Van der Bliek, A.M.; Shen, Q.; Kawajiri, S. Mechanisms of mitochondrial fission and fusion. Cold Spring Harb. Perspect. Biol. 2013, 5, a011072. [Google Scholar] [CrossRef] [PubMed]
- Ghiselli, F.; Maurizii, M.G.; Reunov, A.; Ariño-Bassols, H.; Cifaldi, C.; Pecci, A.; Alexandrova, Y.; Bettini, S.; Passamonti, M.; Franceschini, V.; et al. Natural heteroplasmy and mitochondrial inheritance in bivalve molluscs. Integr. Comp. Biol. 2019, 59, 1016–1032. [Google Scholar] [CrossRef] [PubMed]
- Milani, L.; Ghiselli, F. Mitochondrial activity in gametes and transmission of viable mtDNA. Biol. Direct. 2015, 10, 22. [Google Scholar] [CrossRef]
- Milani, L. Mitochondrial membrane potential: A trait involved in organelle inheritance? Biol. Lett. 2015, 11, 20150732. [Google Scholar] [CrossRef]
- Buning, J. The Insect Ovary. Ultrastructure, Previtellogenic Growth and Evolution; Chapman & Hall: London, UK, 1994. [Google Scholar]
- Pepling, M.E.; de Cuevas, M.; Spradling, A.C. Germline cysts: A conserved phase of germ cell development? Trends Cell Biol. 1999, 9, 257–262. [Google Scholar] [CrossRef]
- Bilinski, S.M.; Kubiak, J.Z.; Kloc, M. Asymmetric Divisions in Oogenesis. In Asymmetric Cell Division in Development, Differentiation and Cancer. Results and Problems in Cell Differerentiation; Tassan, J.-P., Kubiak, J.Z., Eds.; Springer: Cham, Switzerland, 2017; pp. 211–228. [Google Scholar]
- Urbisz, A.Z.; Chajec, L.; Brąszewska-Zalewska, A.; Kubrakiewicz, J.; Swiatek, P. Ovaries of the white worm (Enchytraeus albidus, Annelida, Clitellata) are composed of 16-celled meroistic germ-line cysts. Dev. Biol. 2017, 426, 28–42. [Google Scholar] [CrossRef]
- Jaglarz, M.K.; Kubrakiewicz, J.; Bilinski, S.M. The ovary structure and oogenesis in the basal crustaceans and hexapods. Possible phylogenetic significance. Arthropod Struct. Dev. 2014, 43, 349–360. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Franyutti, A.; Uribe Aranzábal, M.C.; Guillette, L.J., Jr. Oogenesis in the viviparous matrotrophic lizard Mabuya brachypoda. J. Morphol. 2005, 265, 152–164. [Google Scholar] [CrossRef] [PubMed]
- Lei, L.; Spradling, A.C. Mouse oocytes differentiate through organelle enrichment from sister cyst germ cells. Science 2016, 352, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Ikami, K.; Nuzhat, N.; Lei, L. Organelle transport during mouse oocyte differentiation in germline cysts. Curr. Opin. Cell Biol. 2017, 44, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Fan, W.; Waymire, K.G.; Narula, N.; Li, P.; Rocher, C.; Coskun, P.E.; Vannan, M.A.; Narula, J.; Macgregor, G.R.; Wallace, D.C. A mouse model of mitochondrial disease reveals germline selection against severe mtDNA mutations. Science 2008, 319, 958–962. [Google Scholar] [CrossRef] [PubMed]
- Stewart, J.B.; Freyer, C.; Elson, J.L.; Wredenberg, A.; Cansu, Z.; Trifunovic, A.; Larsson, N.G. Strong purifying selection in transmission of mammalian mitochondrial DNA. PLoS Biol. 2008, 6, e10. [Google Scholar] [CrossRef]
- Hill, J.H.; Chen, Z.; Xu, H. Selective propagation of functional mitochondrial DNA during oogenesis restricts the transmission of a deleterious mitochondrial variant. Nat. Genet. 2014, 46, 389–392. [Google Scholar] [CrossRef]
- Cox, R.T.; Spradling, A.C. A Balbiani body and the fusome mediate mitochondrial inheritance during Drosophila oogenesis. Development 2003, 130, 1579–1590. [Google Scholar] [CrossRef]
- Zhou, R.R.; Wang, B.; Wang, J.; Schatten, H.; Zhang, Y.Z. Is the mitochondrial cloud the selection machinery for preferentially transmitting wild-type mtDNA between generations? Rewinding Müller’s ratchet efficiently. Curr. Genet. 2010, 56, 101–107. [Google Scholar] [CrossRef]
- Kloc, M.; Bilinski, S.; Etkin, L.D. The Balbiani body and germ cell determinants: 150 years later. Curr. Top. Dev. Biol. 2004, 59, 1–36. [Google Scholar]
- Kloc, M.; Jedrzejowska, I.; Tworzydlo, W.; Bilinski, S.M. Balbiani body, nuage and sponge bodies-germ plasm pathway players. Arthropod Struct. Dev. 2014, 43, 341–438. [Google Scholar] [CrossRef] [PubMed]
- Heasman, J.; Quarmby, J.; Wylie, C.C. The mitochondrial cloud of Xenopus oocytes: The source of germinal granule material. Dev. Biol. 1984, 105, 458–469. [Google Scholar] [CrossRef]
- Marlow, F.L.; Mullins, M.C. Bucky ball functions in Balbiani body assembly and animal-vegetal polarity in the oocyte and follicle cell layer in zebrafish. Dev. Biol. 2008, 321, 40–50. [Google Scholar] [CrossRef]
- Mahowald, A.P. Assembly of the Drosophila germ plasm. Int. Rev. Cytol. 2001, 203, 187–213. [Google Scholar] [PubMed]
- Jaglarz, M.K.; Kloc, M.; Jankowska, W.; Szymanska, B.; Bilinski, S.M. Nuage morphogenesis becomes more complex: Two translocation pathways and two forms of nuage coexist in Drosophila germline syncytia. Cell Tissue Res. 2011, 344, 169–181. [Google Scholar] [CrossRef] [PubMed]
- Voronina, E.; Seydoux, G.; Sassone-Corsi, P.; Nagamori, I. RNA granules in germ cells. Cold Spring Harb. Perspect. Biol. 2011, 3, a002774. [Google Scholar] [CrossRef]
- Wilding, M.; Carotenuto, R.; Infante, V.; Dale, B.; Marino, M.; Di Matteo, L.; Campanella, C. Confocal microscopy analysis of the activity of mitochondria contained within the ‘mitochondrial cloud’ during oogenesis in Xenopus laevis. Zygote 2001, 9, 347–352. [Google Scholar] [CrossRef]
- Zhang, Y.Z.; Ouyang, Y.C.; Hou, Y.; Schatten, H.; Chen, D.Y.; Sun, Q.Y. Mitochondrial behavior during oogenesis in zebrafish: A confocal microscopy analysis. Dev. Growth Differ. 2008, 50, 189–201. [Google Scholar] [CrossRef]
- Tworzydlo, W.; Kisiel, E.; Jankowska, W.; Witwicka, A.; Bilinski, S.M. Exclusion of dysfunctional mitochondria from Balbiani body during early oogenesis of Thermobia. Cell Tissue Res. 2016, 366, 191–201. [Google Scholar] [CrossRef]
- Tourte, M.; Mignotte, F.; Mounolou, J.C. Organization and replication activity of the mitochondrial mass of oogonia and previtellogenic oocytes in Xenopus laevis. Dev. Growth Differ. 1981, 23, 9–21. [Google Scholar] [CrossRef]
- Bilinski, S.M.; Jaglarz, M.K.; Tworzydlo, W. Organelle assemblages implicated in the transfer of oocyte components to the embryo: An insect perspective. Curr. Opin. Insect Sci. 2019, 31, 1–7. [Google Scholar] [CrossRef]
- Bilinski, S.M.; Kloc, M.; Tworzydlo, W. Selection of mitochondria in female germline cells: Is Balbiani body implicated in this process? J. Assist. Reprod. Genet. 2017, 34, 1405–1412. [Google Scholar] [CrossRef]
- Agnello, M.; Chiarelli, R.; Martino, C.; Bosco, L.; Roccheri, M.C. Autophagy is required for sea urchin oogenesis and early development. Zygote 2016, 24, 918–926. [Google Scholar] [CrossRef]
- Agnello, M.; Roccheri, M.C.; Morici, G.; Rinaldi, A.M. Mitochondria during sea urchin oogenesis. Zygote 2017, 25, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Pepling, M.E.; Spradling, A.C. Mouse ovarian germ cell cysts undergo programmed breakdown to form primordial follicles. Dev. Biol. 2001, 234, 339–351. [Google Scholar] [CrossRef] [PubMed]
- Pepling, M.E.; Wilhelm, J.E.; O’Hara, A.L.; Gephardt, G.W.; Spradling, A.C. Mouse oocytes within germ cell cysts and primordial follicles contain a Balbiani body. Proc. Natl. Acad. Sci. USA 2007, 104, 187–192. [Google Scholar] [CrossRef] [PubMed]
- Kloc, M.; Jaglarz, M.; Dougherty, M.; Stewart, M.D.; Nel-Themaat, L.; Bilinski, S. Mouse early oocytes are transiently polar: Three-dimensional and ultrastructural analysis. Exp. Cell Res. 2008, 314, 3245–3254. [Google Scholar] [CrossRef]
- Lieber, T.; Jeedigunta, S.P.; Palozzi, J.M.; Lehmann, R.; Hurd, T.R. Mitochondrial fragmentation drives selective removal of deleterious mtDNA in the germline. Nature 2019, 570, 380–384. [Google Scholar] [CrossRef]
- Van Blerkom, J.; Davis, P.; Mathwig, V.; Alexander, S. Domains of high-polarized and low-polarized mitochondria may occur in mouse and human oocytes and early embryos. Hum. Reprod. 2002, 17, 393–406. [Google Scholar] [CrossRef]
- Dumollard, R.; Duchen, M.; Sardet, C. Calcium signals and mitochondria at fertilisation. Semin. Cell Dev. Biol. 2006, 17, 314–323. [Google Scholar] [CrossRef]
- Kogo, N.; Tazaki, A.; Kashimno, Y.; Morichika, K.; Orii, H.; Mochii, M.; Watanabe, K. Germ-line mitochondria exhibit suppressed respiratory activity to support their accurate transmission to the next generation. Dev. Biol. 2011, 349, 462–469. [Google Scholar] [CrossRef] [PubMed]
- Faron, J.; Bernas, T.; Sas-Nowosielska, H.; Klag, J. Analysis of the behavior of mitochondria in the ovaries of the earthworm Dendrobaena veneta Rosa 1839. PLoS ONE 2015, 10, e0117187. [Google Scholar] [CrossRef]
- Allen, C.A.; Van der Giezen, M.; Allen, J.F. Origin, function, and transmission of mitochondria. In Origin of Mitochondria and Hydrogenosomes; Martin, W.F., Müller, M., Eds.; Springer: Berlin/Heidelberg, Germany, 2007; pp. 39–56. [Google Scholar]
- St John, J.C.; Facucho-Oliveira, J.; Jiang, Y.; Kelly, R.; Salah, R. Mitochondrial DNA transmission, replication and inheritance: A journey from the gamete through the embryo and into offspring and embryonic stem cells. Hum. Reprod. Update 2010, 16, 488–509. [Google Scholar] [CrossRef]
- Tworzydlo, W.; Bilinski, S.M. Structure of ovaries and oogenesis in dermapterans. I. Origin and functioning of the ovarian follicles. Arthropod Struct. Dev. 2008, 37, 310–320. [Google Scholar] [CrossRef]
- Tworzydlo, W.; Bilinski, S.M.; Kocarek, P.; Haas, F. Ovaries and germline cysts and their evolution in Dermaptera (Insecta). Arthropod Struct. Dev. 2010, 39, 360–368. [Google Scholar] [CrossRef]
- Hamasaki, M.; Furuta, N.; Matsuda, A.; Nezu, A.; Yamamoto, A.; Fujita, N.; Oomori, H.; Noda, T.; Haraguchi, T.; Hiraoka, Y.; et al. Autophagosomes form at ER-mitochondria contact sites. Nature 2013, 495, 389–393. [Google Scholar] [CrossRef]
- Bockler, S.; Westermann, B. ER-mitochondria contacts as sites of mitophagosome formation. Autophagy 2014, 10, 1346–1347. [Google Scholar] [CrossRef] [PubMed]
- Bockler, S.; Westermann, B. Mitochondrial ER contacts are crucial for mitophagy in yeast. Dev. Cell 2014, 28, 450–458. [Google Scholar] [CrossRef] [PubMed]
- Zachari, M.; Gudmundsson, S.R.; Li, Z.; Manifava, M.; Shah, R.; Smith, M.; Stronge, J.; Karanasios, E.; Piunti, C.; Kishi-Itakura, C.; et al. Selective autophagy of mitochondria on a ubiquitin-endoplasmic-reticulum platform. Dev. Cell 2019, 50, 627–643. [Google Scholar] [CrossRef]
- Ghafari, F.; Gutierrez, C.G.; Hartshorne, G.M. Apoptosis in mouse fetal and neonatal oocytes during meiotic prophase one. BMC Dev. Biol. 2007, 7, 87. [Google Scholar] [CrossRef]
- Tiwari, M.; Prasad, S.; Tripathi, A.; Pandey, A.N.; Ali, I.; Singh, A.K.; Shrivastav, T.G.; Chaube, S.K. Apoptosis in mammalian oocytes: A review. Apoptosis 2015, 20, 1019–1025. [Google Scholar] [CrossRef] [PubMed]
- Buszczak, M.; Cooley, L. Eggs to die for: Cell death during Drosophila oogenesis. Cell Death Differ. 2000, 7, 1071–1074. [Google Scholar] [CrossRef] [PubMed]
- Peterson, J.S.; Barkett, M.; McCall, K. Stage-specific regulation of caspase activity in Drosophila oogenesis. Dev. Biol. 2003, 260, 113–123. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tworzydlo, W.; Sekula, M.; Bilinski, S.M. Transmission of Functional, Wild-Type Mitochondria and the Fittest mtDNA to the Next Generation: Bottleneck Phenomenon, Balbiani Body, and Mitophagy. Genes 2020, 11, 104. https://doi.org/10.3390/genes11010104
Tworzydlo W, Sekula M, Bilinski SM. Transmission of Functional, Wild-Type Mitochondria and the Fittest mtDNA to the Next Generation: Bottleneck Phenomenon, Balbiani Body, and Mitophagy. Genes. 2020; 11(1):104. https://doi.org/10.3390/genes11010104
Chicago/Turabian StyleTworzydlo, Waclaw, Malgorzata Sekula, and Szczepan M. Bilinski. 2020. "Transmission of Functional, Wild-Type Mitochondria and the Fittest mtDNA to the Next Generation: Bottleneck Phenomenon, Balbiani Body, and Mitophagy" Genes 11, no. 1: 104. https://doi.org/10.3390/genes11010104
APA StyleTworzydlo, W., Sekula, M., & Bilinski, S. M. (2020). Transmission of Functional, Wild-Type Mitochondria and the Fittest mtDNA to the Next Generation: Bottleneck Phenomenon, Balbiani Body, and Mitophagy. Genes, 11(1), 104. https://doi.org/10.3390/genes11010104



