Detection of Transgenes in Gene Delivery Model Mice by Adenoviral Vector Using ddPCR
Abstract
1. Introduction
2. Materials and Methods
2.1. Cloning of Recombinant Adenoviral Vectors Containing the mCherry Gene
2.2. Animal Experiments
2.2.1. Acute Experiments
2.2.2. Chronic Experiments
2.3. Confirmation of Gene and Protein Expression by Infection of rAdV Vectors In Vivo
2.4. DNA Extraction and Detection of Transgenes Using Three Different PCR Methods
2.5. Semi-Quantitative PCR (sqPCR)
2.6. Real-Time Quantitative PCR (qPCR)
2.7. Droplet Digital PCR (ddPCR)
2.8. Statistics
3. Results
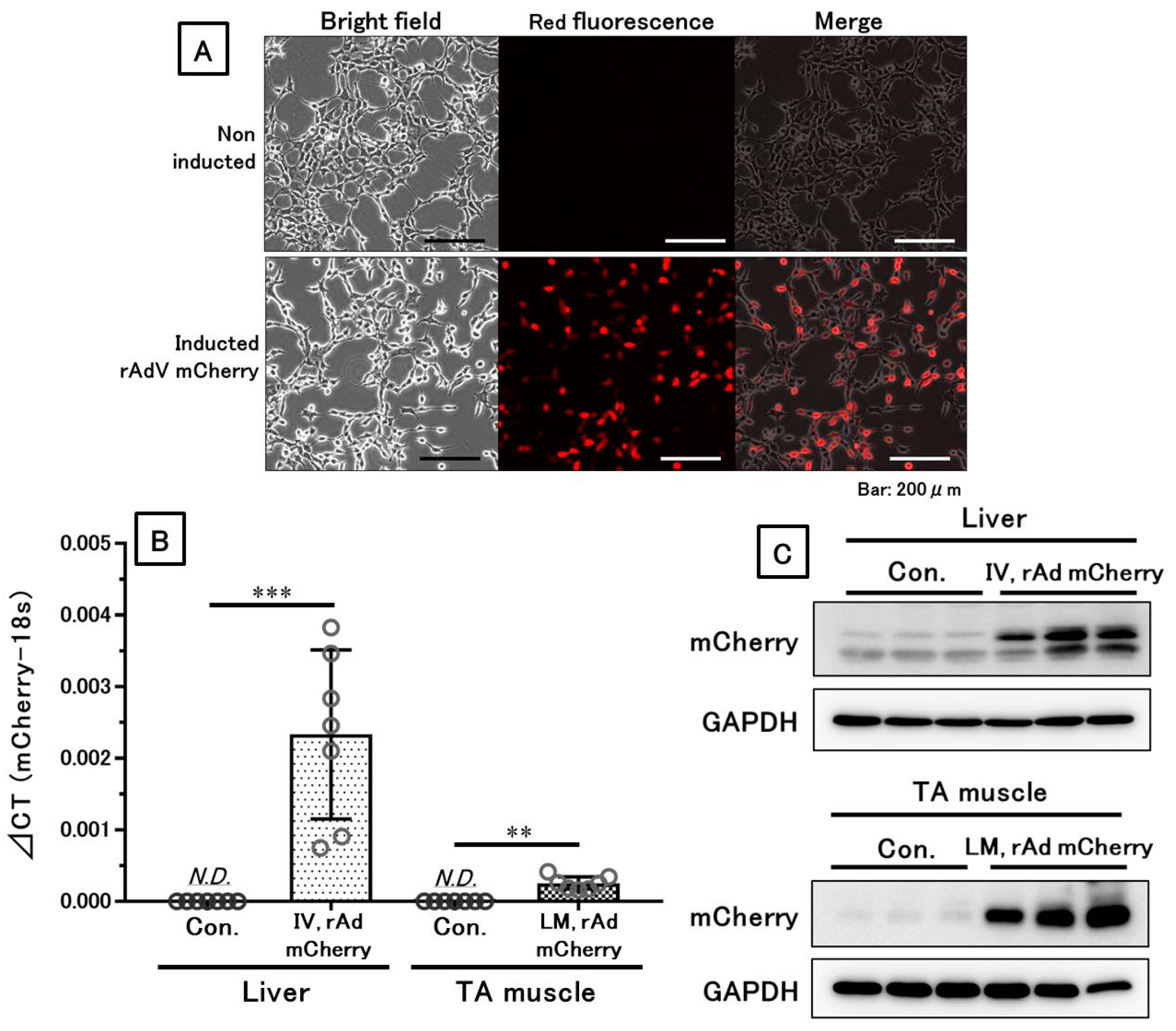
3.1. The mCherry Gene and Protein Were Sufficiently Expressed Both In Vitro and In Vivo
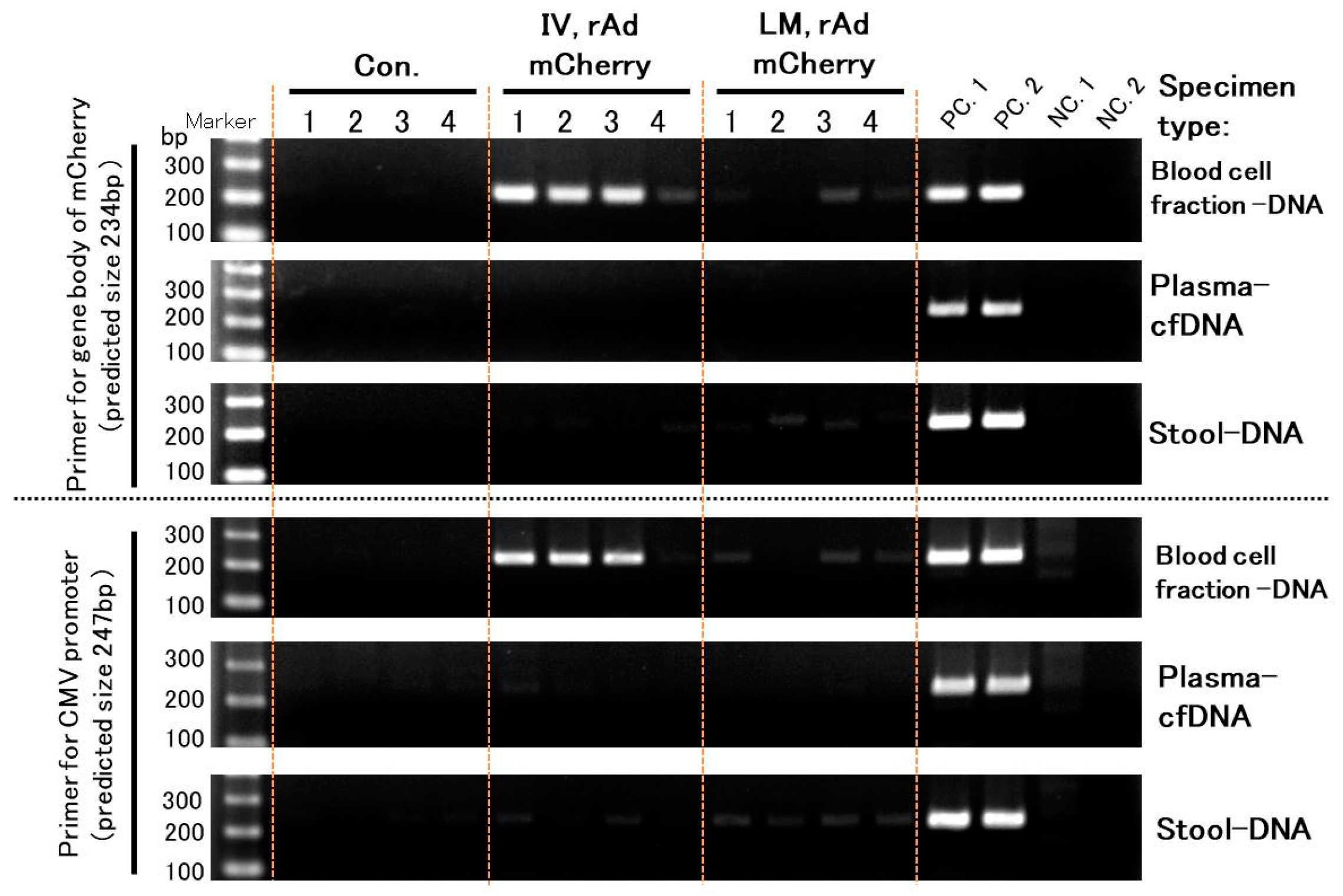
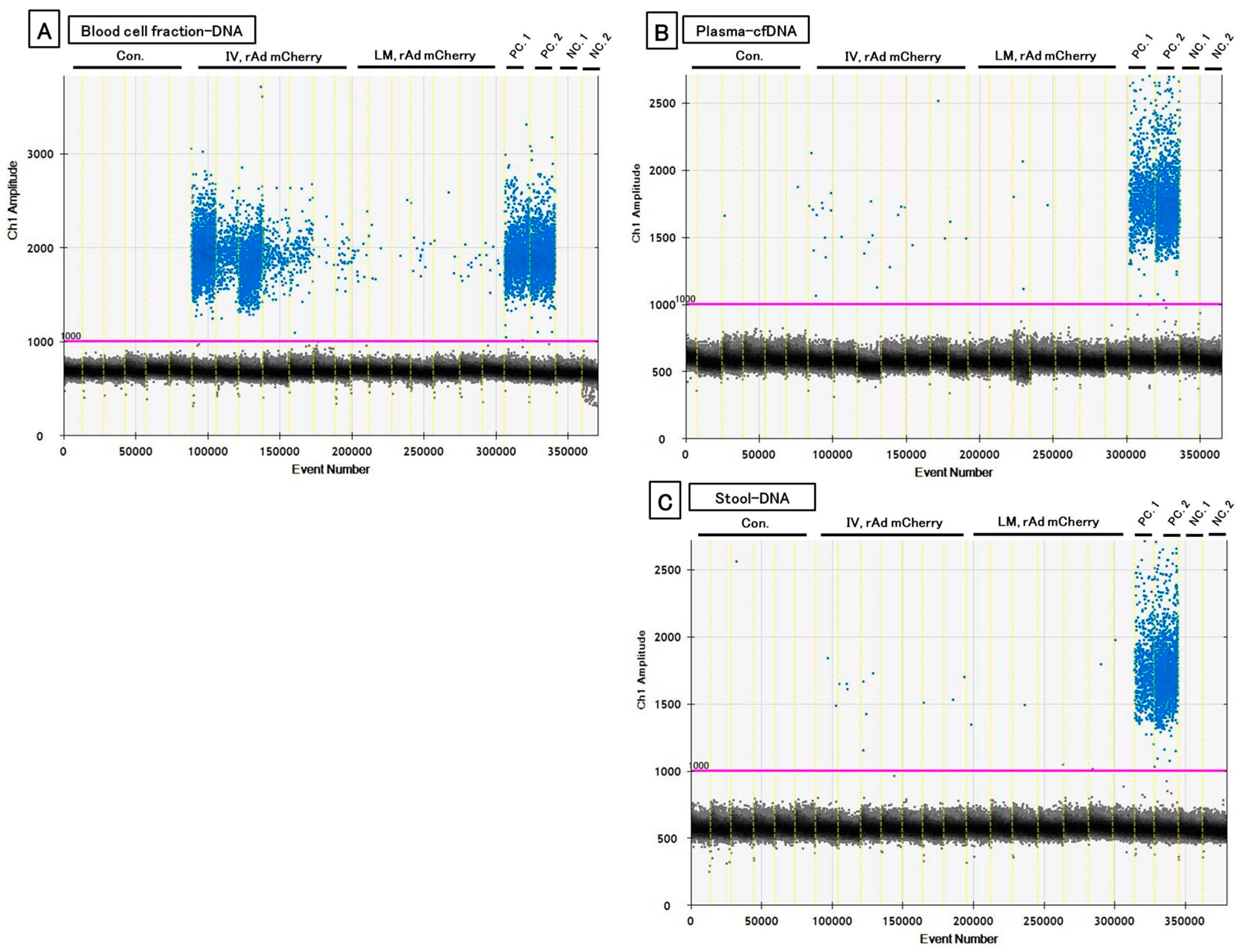
3.2. The Three PCR Methods Showed Each Characteristic and Could Detect Transgene Fragments in Acute Experiments
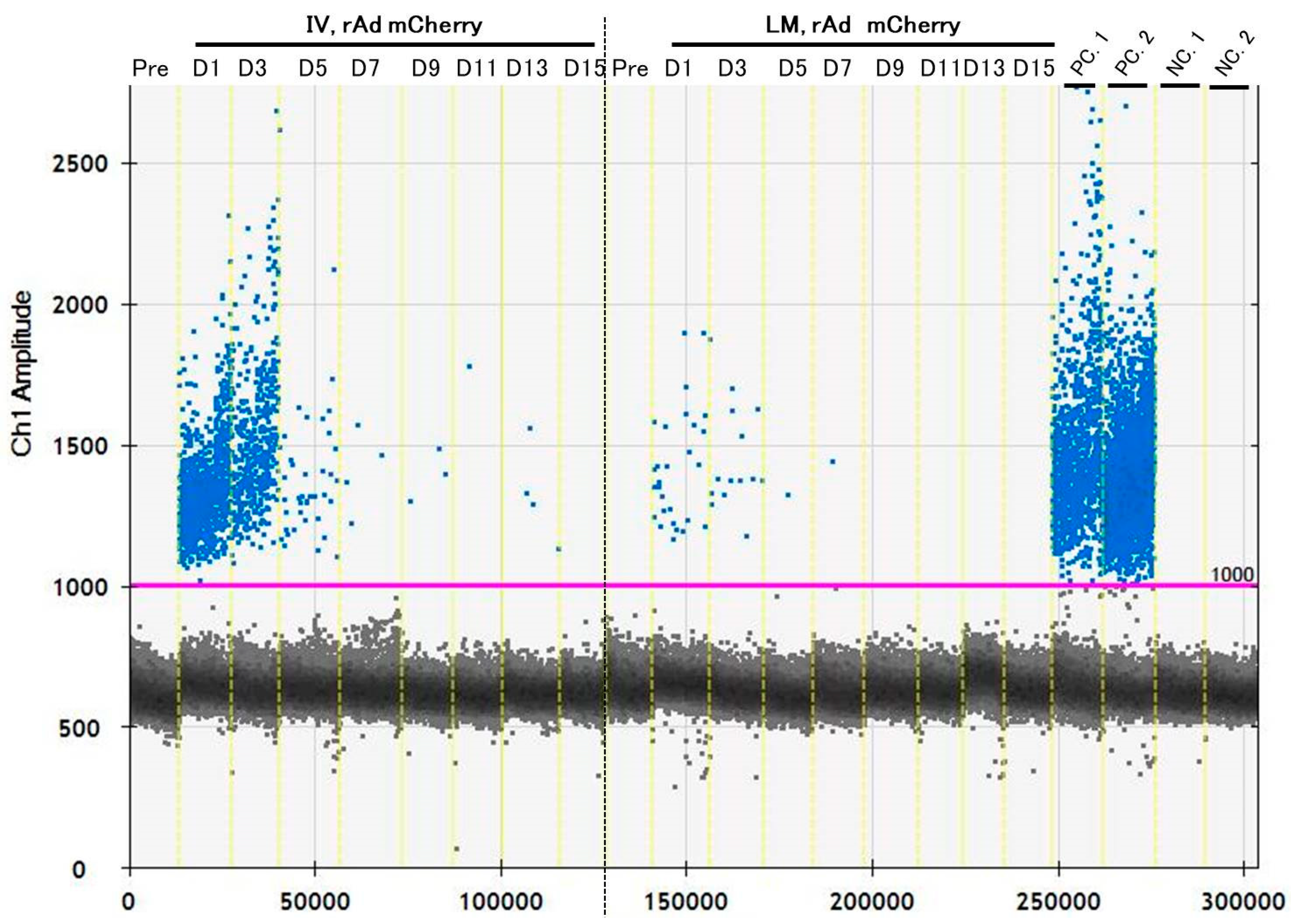
3.3. ddPCR on Chronic Experiments Showed a Possibility of Detecting Transgenes Repeatedly
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Japan Anti-Doping Agency (JADA). What Is Anti-Doping? Available online: https://www.playtruejapan.org/about/ (accessed on 12 March 2019).
- The World Anti-Doping Agency (WADA). Who We Are? Available online: https://www.wada-ama.org/en/who-we-are (accessed on 12 March 2019).
- The World Anti-Doping Agency (WADA). Anti-Doping Textbook; WADA: Montreal, QC, Canada, 2016. [Google Scholar]
- The World Anti-Doping Agency (WADA). World Anti-Doping Code with International Standard, Prohibited List; WADA: Montreal, QC, Canada, 2018. [Google Scholar]
- Duan, D. Systemic AAV Micro-dystrophin Gene Therapy for Duchenne Muscular Dystrophy. Mol. Ther. 2018, 26, 2337–2356. [Google Scholar] [CrossRef] [PubMed]
- High, K.A.; Anguela, X.M. Adeno-associated viral vectors for the treatment of hemophilia. Hum. Mol. Genet. 2016, 25, R36–R41. [Google Scholar] [CrossRef] [PubMed]
- Naso, M.F.; Tomkowicz, B.; Perry, W.L.; Strohl, W.R. Adeno-Associated Virus (AAV) as a Vector for Gene Therapy. BioDrugs 2017, 31, 317–334. [Google Scholar] [CrossRef] [PubMed]
- Kumaran, N.; Moore, A.T.; Weleber, R.G.; Michaelides, M. Leber congenital amaurosis/early-onset severe retinal dystrophy: Clinical features, molecular genetics and therapeutic interventions. Br. J. Ophthalmol. 2017, 101, 1147–1154. [Google Scholar] [CrossRef] [PubMed]
- Sharif, W.; Sharif, Z. Leber’s congenital amaurosis and the role of gene therapy in congenital retinal disorders. Int. J. Ophthalmol. 2017. [Google Scholar] [CrossRef]
- Wold, W.; Toth, K. Adenovirus Vectors for Gene Therapy, Vaccination and Cancer Gene Therapy. Curr. Gene Ther. 2014, 13, 421–433. [Google Scholar] [CrossRef]
- Lee, C.S.; Bishop, E.S.; Zhang, R.; Yu, X.; Farina, E.M.; Yan, S.; Zhao, C.; Zeng, Z.; Shu, Y.; Wu, X.; et al. Adenovirus-mediated gene delivery: Potential applications for gene and cell-based therapies in the new era of personalized medicine. Genes Dis. 2017, 4, 43–63. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Du, Z.; Wang, X.; Li, X. Treatment of Uterine Sarcoma with rAd-p53 (Gendicine) Followed by Chemotherapy: Clinical Study of TP53 Gene Therapy. Hum. Gene Ther. 2018, 29, 242–250. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.W.; Li, L.; Li, D.; Liu, J.; Li, X.; Li, W.; Xu, X.; Zhang, M.J.; Chandler, L.A.; Lin, H.; et al. The First Approved Gene Therapy Product for Cancer Ad- p53 (Gendicine): 12 Years in the Clinic. Hum. Gene Ther. 2018, 29, 160–179. [Google Scholar] [CrossRef] [PubMed]
- Liang, M. Oncorine, The World First Oncolytic Virus Medicine and its Update in China. Curr. Cancer Drug Targets 2018, 18, 29189159. [Google Scholar] [CrossRef] [PubMed]
- The Journal of Gene Medicine. Charts and Tables, Vectors. Available online: http://www.abedia.com/wiley/vectors.php (accessed on 11 March 2019).
- Hung, Y.P.; Albeck, J.G.; Tantama, M.; Yellen, G. Imaging cytosolic NADH-NAD + redox state with a genetically encoded fluorescent biosensor. Cell Metab. 2011, 14, 545–554. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, Y.; Yahagi, N.; Nakagawa, Y.; Matsuzaka, T.; Shimizu, R.; Sekiya, M.; Iizuka, Y.; Ohashi, K.; Gotoda, T.; Yamamoto, M.; et al. In vivo promoter analysis on refeeding response of hepatic sterol regulatory element-binding protein-1c expression. Biochem. Biophys. Res. Commun. 2007, 363, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, Y.; Yahagi, N.; Aita, Y.; Murayama, Y.; Sawada, Y.; Piao, X.; Toya, N.; Oya, Y.; Shikama, A.; Takarada, A.; et al. KLF15 Enables Rapid Switching between Lipogenesis and Gluconeogenesis during Fasting. Cell Rep. 2016, 16, 2373–2386. [Google Scholar] [CrossRef] [PubMed]
- Sweeney, J.; Hennessey, J. Evaluation of accuracy and precision of adenovirus absorptivity at 260 nm under conditions of complete DNA disruption. Virology 2002, 295, 284–288. [Google Scholar] [CrossRef] [PubMed]
- Tozaki, T.; Gamo, S.; Takasu, M.; Kikuchi, M.; Kakoi, H.; Hirota, K.I.; Kusano, K.; Nagata, S.I. Digital PCR detection of plasmid DNA administered to the skeletal muscle of a microminipig: a model case study for gene doping detection. BMC Res. Notes 2018, 11, 708. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.J.; Xu, J.F.; Shen, Y.W.; Ma, S.J.; Zhang, T.T.; Meng, Q.L.; Lan, W.J.; Zhang, C.; Liu, X.M. Detection of exogenous gene doping of IGF-I by a real-time quantitative PCR assay. Biotechnol. Appl. Biochem. 2016, 64, 549–554. [Google Scholar] [CrossRef] [PubMed]
- Brzeziańska, E.; Domańska, D.; Jegier, A. Gene doping in sport—Perspectives and risks. Biol. Sport 2014, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Takemasa, T.; Yakushiji, N.; Kikuchi, D.M.; Deocaris, C. Fundamental study of detection of muscle hypertrophy-oriented gene doping by myostatin knock down using RNA interference. J. Sports Sci. Med. 2012, 11, 294–303. [Google Scholar]
- Neuberger, E.W.I.; Simon, P. Gene and Cell Doping: The New Frontier - Beyond Myth or Reality. Med. Sports Sci. 2017, 91–106. [Google Scholar] [CrossRef]
- Yachie, N.; Takahashi, K.; Katayama, T.; Sakurada, T.; Kanda, G.N.; Takagi, E.; Hirose, T.; Katsura, T.; Moriya, T.; Kitano, H.; et al. Robotic crowd biology with Maholo LabDroids. Nat. Biotechnol. 2017, 35, 310–312. [Google Scholar] [CrossRef] [PubMed]

| Vector | Gene Therapy Clinical Trials | |
|---|---|---|
| Number | % | |
| Adenovirus | 541 | 18.5 |
| Retrovirus | 514 | 17.5 |
| Naked/Plasmid DNA | 452 | 15.4 |
| Lentivirus | 278 | 9.5 |
| Adeno-associated virus | 238 | 8.1 |
| Vaccinia virus | 133 | 4.5 |
| Lipofection | 119 | 4.1 |
| Others | 774 | 26.4 |
| Total | 2930 | 100 |
| Methods | Targets | Sequences | Predicted Size (bp) | |
|---|---|---|---|---|
| sqPCR | mCherry gene body | Forward | CACGAGTTCGAGATCGAGGG | 234 |
| Reverse | GCCGTCCTCGAAGTTCATCA | |||
| sqPCR | CMV promoter | Forward | CACGCCCATTGATGTACTGC | 247 |
| Reverse | ACGCCAATAGGGACTTTCCA | |||
| qPCR, ddPCR: Taq man probe assay | mCherry gene body | Forward | GGCACCAACTTCCCCTCC | 115 |
| Probe | 56FAM/CATGGTCTT/ZEN/CTTCTGCAT/3IABkFQ | |||
| Reverse | TCTGCTTGATCTCGCCCTTC | |||
| qPCR: SYBR green assay | 18s rRNA | Forward | AGTCCCTGCCCTTTGTACACA | 70 |
| Reverse | CGATCCGAGGGCCTCACTA | |||
| Group | Mouse No. | Copy/μL of Transgene | ||
|---|---|---|---|---|
| Blood Cell Fraction-DNA | Plasma-cfDNA | Stool-DNA | ||
| Con. | 1 | 0.0 | 0.0 | 0.0 |
| 2 | 0.0 | 0.0 | 0.0 | |
| 3 | 0.0 | 0.0 | 0.0 | |
| 4 | 0.0 | 0.0 | 0.0 | |
| 5 | 0.0 | 0.0 | 0.0 | |
| 6 | 0.0 | 0.0 | 0.0 | |
| Median | 0.0 | 0.0 | 0.0 | |
| IV, rAdV mCherry | 1 | 2528.2 | 33.4 | 3.7 |
| 2 | 390.3 | 3.3 | 0.0 | |
| 3 | 1808.4 | 8.5 | 1.6 | |
| 4 | 217.9 | 5.1 | 0.0 | |
| 5 | 230.4 | 4.9 | 0.9 | |
| 6 | 26.2 | 5.5 | 0.8 | |
| 7 | 63.1 | 4.0 | 2.6 | |
| Median | 230.4 a, b | 5.1 a, b | 0.9 a, b | |
| LM, rAdV mCherry | 1 | 56.2 | 0.0 | 1.8 |
| 2 | 11.8 | 0.0 | 0.0 | |
| 3 | 16.5 | 0.0 | 0.0 | |
| 4 | 30.7 | 0.0 | 0.0 | |
| 5 | 15.9 | 0.0 | 0.0 | |
| 6 | 31.7 | 0.0 | 0.0 | |
| 7 | 28.3 | 0.0 | 0.0 | |
| Median | 28.3 c | 0.0 | 0.0 | |
| Group | Mouse No. | Copy/μL of Transgene | ||
|---|---|---|---|---|
| Blood Cell Fraction-DNA | Plasma-cfDNA | Stool-DNA | ||
| Con. | 1 | 0.0 | 0.0 | 0.0 |
| 2 | 0.0 | 0.0 | 0.0 | |
| 3 | 0.0 | 0.8 | 0.7 | |
| 4 | 0.0 | 0.0 | 0.0 | |
| 5 | 0.6 | 0.0 | 0.0 | |
| 6 | 0.0 | 0.8 | 0.0 | |
| Median | 0.0 | 0.0 | 0.0 | |
| IV, rAdV mCherry | 1 | 4460.0 | 19.2 | 2.3 |
| 2 | 620.7 | 0.7 | 2.2 | |
| 3 | 2873.3 | 6.0 | 5.5 | |
| 4 | 276.7 | 4.3 | 0.0 | |
| 5 | 190.0 | 2.3 | 0.0 | |
| 6 | 13.7 | 1.9 | 0.8 | |
| 7 | 42.7 | 2.5 | 1.5 | |
| Median | 276.7 a, b | 2.5 a, b | 1.5 a | |
| LM, rAdV mCherry | 1 | 34.0 | 0.0 | 0.7 |
| 2 | 5.5 | 0.0 | 0.0 | |
| 3 | 4.5 | 3.0 | 0.7 | |
| 4 | 12.9 | 1.4 | 0.0 | |
| 5 | 3.8 | 0.0 | 0.7 | |
| 6 | 16.5 | 0.0 | 1.4 | |
| 7 | 11.5 | 0.0 | 1.5 | |
| Median | 11.5 c | 0.0 | 0.7 d | |
| Mouse No. | Copy/μL of Transgene | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Pre | 1 day | 3 days | 5 days | 7 days | 9 days | 11 days | 13 days | 15 days | ||
| IV, rAdV mCherry | 1 | 0.0 | 817.0 | 1398.0 | 48.0 | 4.3 | 1.4 | 0.0 | 0.8 | 0.0 |
| 2 | 0.0 | 890.0 | 209.0 | 25.0 | 2.4 | 0.0 | 0.7 | 3.4 | 0.7 | |
| 3 | 0.8 | 1108.0 | 796.0 | 10.3 | 0.9 | 0.0 | 0.0 | 10.4 | 0.0 | |
| 4 | 2.0 | 1422.0 | 298.0 | 13.7 | 1.4 | 2.7 | 1.7 | 0.0 | 0.0 | |
| 5 | 0.7 | 1900.0 | 1132.0 | 62.0 | 10.5 | 1.4 | 1.6 | 1.5 | 0.8 | |
| 6 | 0.0 | 4800.0 | 1261.0 | 39.0 | 7.1 | 2.0 | 0.7 | 0.9 | 1.5 | |
| Median | 0.4 | 1265.0 | 964.0 | 32.0 | 3.4 | 1.4 | 0.7 | 1.2 | 0.4 | |
| p- values vs Pre | 0.0002 | 0.0004 | 0.0063 | 0.0911 | 0.4181 | 0.5886 | 0.2992 | 0.6177 | ||
| LM, rAdV mCherry | 1 | 0.0 | 70.0 | 66.0 | 7.3 | 0.7 | 0.8 | 0.0 | 0.0 | 0.0 |
| 2 | 0.9 | 149.0 | 24.0 | 5.0 | 1.6 | 0.7 | 0.0 | 0.0 | 0.0 | |
| 3 | 0.0 | 19.0 | 7.2 | 4.2 | 0.7 | 0.0 | 0.0 | 0.0 | 0.0 | |
| 4 | 0.0 | 10.4 | 4.6 | 0.8 | 0.8 | 0.7 | 0.0 | 0.0 | 0.0 | |
| 5 | 0.0 | 8.6 | 9.0 | 0.0 | 0.8 | 0.8 | 0.0 | 1.5 | 0.0 | |
| 6 | 0.0 | 5.2 | 27.0 | 4.1 | 0.0 | 0.7 | 0.0 | 0.0 | 0.0 | |
| Median | 0.0 | 14.7 | 16.5 | 4.2 | 0.8 | 0.7 | 0.0 | 0.0 | 0.0 | |
| p- values vs Pre | 0.0012 | 0.0012 | 0.0666 | 0.2576 | 0.2902 | 0.5904 | 0.7753 | 0.5907 | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sugasawa, T.; Aoki, K.; Watanabe, K.; Yanazawa, K.; Natsume, T.; Takemasa, T.; Yamaguchi, K.; Takeuchi, Y.; Aita, Y.; Yahagi, N.; et al. Detection of Transgenes in Gene Delivery Model Mice by Adenoviral Vector Using ddPCR. Genes 2019, 10, 436. https://doi.org/10.3390/genes10060436
Sugasawa T, Aoki K, Watanabe K, Yanazawa K, Natsume T, Takemasa T, Yamaguchi K, Takeuchi Y, Aita Y, Yahagi N, et al. Detection of Transgenes in Gene Delivery Model Mice by Adenoviral Vector Using ddPCR. Genes. 2019; 10(6):436. https://doi.org/10.3390/genes10060436
Chicago/Turabian StyleSugasawa, Takehito, Kai Aoki, Koichi Watanabe, Koki Yanazawa, Tohru Natsume, Tohru Takemasa, Kaori Yamaguchi, Yoshinori Takeuchi, Yuichi Aita, Naoya Yahagi, and et al. 2019. "Detection of Transgenes in Gene Delivery Model Mice by Adenoviral Vector Using ddPCR" Genes 10, no. 6: 436. https://doi.org/10.3390/genes10060436
APA StyleSugasawa, T., Aoki, K., Watanabe, K., Yanazawa, K., Natsume, T., Takemasa, T., Yamaguchi, K., Takeuchi, Y., Aita, Y., Yahagi, N., Yoshida, Y., Tokinoya, K., Sekine, N., Takeuchi, K., Ueda, H., Kawakami, Y., Shimizu, S., & Takekoshi, K. (2019). Detection of Transgenes in Gene Delivery Model Mice by Adenoviral Vector Using ddPCR. Genes, 10(6), 436. https://doi.org/10.3390/genes10060436






