Role of GPER-Mediated Signaling in Testicular Functions and Tumorigenesis
Abstract
:1. Introduction
2. GPER Role in Testicular Interstitial Compartment
2.1. GPER in Leydig Cells
2.2. GPER in Telocytes
3. GPER Role in Testicular Tubular Compartment
3.1. GPER in Sertoli Cells
3.2. GPER in Peritubular Cells
3.3. GPER in Germ Cells
4. GPER Role in Epididymis
5. GPER Role in Testicular Tumors
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AC | adenylyl cyclase; |
| AGO2 | argonaute 2; |
| Akt | protein kinase b [PKB]; |
| ArKO | aromatase knockout; |
| BAX | BCL2 associated X protein; |
| BCL2 | B-cell lymphoma 2; |
| BCL2L2 | BCL2 like protein 2; |
| BPA | bisphenol a; |
| cAMP | cyclic adenosine monophosphate; |
| CDKN1B | cyclin dependent kinase inhibitor 1b; |
| CA1 | cyclin A1; |
| CB1 | cyclin B1; |
| CD1 | cyclin D1; |
| cGMP | cyclic guanosine monophosphate; |
| CIS | germ cell neoplasia in situ; |
| CREB | cAMP response element-binding protein; |
| DICER | dicer 1, ribonuclease III; |
| DPN | 2,3-bis[4-Hydroxyphenyl]-propionitrile; |
| DROSHA | drosha ribonuclease III; |
| E2 | 17beta-estradiol; |
| EGFR | epidermal growth factor receptor; |
| ERα | estrogen receptor α; |
| ERβ | estrogen receptor β; |
| ERs | estrogen receptors; |
| ERE | estrogen response element; |
| ERK | extracellular signal-regulated kinase; |
| ERKO | estrogen receptor knockout; |
| ERRβ | estrogen-related receptor β; |
| ERRs | estrogen-related receptors; |
| EXPO5 | exportin-5; |
| FSH | follicle stimulatimg hormone; |
| G15 | [3aS,4R,9bR]-4-[6-bromo-1,3-benzodioxol-5-yl]-3a,4,5,9b-tetrahydro-3H-cyclopenta[c]quinolone; |
| G1 | [±]-1-[[3aR*,4S*,9bS*]-4-[6-Bromo-1,3-benzodioxol-5-yl]-3a,4,5,9b-tetrahydro-3H-cyclopenta[c]quinolin-8-yl]-ethanone; |
| GDNF | glial cell-derived neurotrophic factor; |
| GPCR | G protein-coupled receptor; |
| GPER | G protein-coupled estrogen receptor 1; |
| HMGCR | hydroxymethylglutaril-coa reductase; |
| HMGCS | hydroxymethylglutaril-coa synthase; |
| 3β-HSD | 3β-hydroxysteroid dehydrogenase; |
| HSL | hormone sensitive lipase; |
| LC3 | microtubule-associated protein 1A/1B-light chain; |
| LCT | leydig cell tumor; |
| LHR | luteotropin receptor; |
| MAPK | mitogen-activated protein kinase; |
| mTOR | mammalian target of rapamycin; |
| mTORC1 | mammalian target of rapamycin complex 1; |
| NFkB | nuclear factor kappa-light-chain-enhancer of activated B cells; |
| p70SK6 | ribosomal protein s6 kinase beta-1; |
| PARP1 | poly [ADP-ribose] polymerase 1; |
| PCNA | proliferating cell nuclear antigen; |
| PDAC | pancreatic ductal adenocarcinoma; |
| PDGFR | platelet-derived growth factor receptor; |
| PD-L1 | programmed death ligand 1; |
| PI3K | phosphoinositide 3-kinase; |
| PKA | protein kinase a; |
| PKG | cGMP-dependent protein kinase; |
| PLC | phospholipase c; |
| PLIN | perilipin; |
| PPARα | peroxisome proliferator-activated receptor α; |
| PPARβ | peroxisome proliferator-activated receptor β; |
| PPARγ | peroxisome proliferator-activated receptor γ; |
| PPARs | peroxisome proliferator-activated receptors; |
| PPT | 4,4′,4″-[4-Propyl-[1H] -pyrazole-1,3,5-triyl] trisphenol; |
| Rb | retinoblastoma protein; |
| RS | round spermatids; |
| Skp2 | s-phase kinase-associated protein 2; |
| Src | proto-oncogene tyrosine-protein kinase; |
| StAR | acute steroidogenic regulatory protein; |
| TBBPA | tetrabromobisphenol A; |
| TCBPA | tetrachlorobisphenol A; |
| TGTCs | testicular germ cell tumors; |
| TORC1 | mammalian target of rapamycin complex 1; |
| TSPO | translocator protein; |
| VEGF | vascular endothelium growth factor; |
References
- Weinbauer, G.; Luetjens, C.M.; Simoni, M.; Nieschlag, E. Physiology of Testicular Function. Andrology; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2010; pp. 11–59. ISBN 978-3-540-78354-1. [Google Scholar] [CrossRef]
- Liu, Y.; Liang, Y.; Wang, S.; Tarique, I.; Vistro, W.A.; Zhang, H.; Haseeb, A.; Gandahi, N.S.; Iqbal, A.; An, T.; et al. Identification and characterization of telocytes in rat testis. Aging 2019, 11, 5757–5768. [Google Scholar] [CrossRef] [PubMed]
- Akingbemi, B.T. Estrogen regulation of testicular function. Reprod. Biol. Endocrinol. 2005, 3, 51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carreau, S.; Hess, R.A. Oestrogens and spermatogenesis. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2010, 365, 1517–1535. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carreau, S.; Bouraima-Lelong, H.; Delalande, C. Estrogen, a female hormone involved in spermatogenesis. Adv. Med. Sci-Poland 2012, 57, 31–36. [Google Scholar] [CrossRef]
- Nilsson, S.; Makela, S.; Treuter, E.; Tujague, M.; Thomsen, J.; Andersson, G.; Enmark, E.; Pettersson, K.; Warner, M.; Gustafsson, J.A. Mechanisms of estrogen action. Physiol. Rev. 2001, 81, 1535–1565. [Google Scholar] [CrossRef]
- McDonnell, D.P.; Norris, J.D. Connections and regulation of the human estrogen receptor. Science 2002, 296, 1642–1644. [Google Scholar] [CrossRef]
- Hammes, S.R.; Levin, E.R. Extranuclear steroid receptors: Nature and actions. Endocr. Rev. 2007, 28, 726–741. [Google Scholar] [CrossRef]
- Kelly, M.J.; Levin, E.R. Rapid actions of plasma membrane estrogen receptors. Trends Endocrin. Met. 2001, 12, 152–156. [Google Scholar] [CrossRef]
- Levin, E.R. Cellular functions of plasma membrane estrogen receptors. Steroids 2002, 67, 471–475. [Google Scholar] [CrossRef]
- Pedram, A.; Razandi, M.; Aitkenhead, M.; Hughes, C.C.W.; Levin, E.R. Integration of the non-genomic and genomic actions of estrogen-Membrane-initiated signaling by steroid to transcription and cell biology. J. Biol. Chem. 2002, 277, 50768–50775. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Katzenellenbogen, B.S.; Katzenellenbogen, J.A. Estrogen receptor transcription and transactivation Estrogen receptor alpha and estrogen receptor beta: Regulation by selective estrogen receptor modulators and importance in breast cancer. Breast Cancer Res. 2000, 2, 335–344. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lazari, M.F.M.; Lucas, T.F.G.; Yasuhara, F.; Gomes, G.R.O.; Siu, E.R.; Royer, C.; Fernandes, S.A.F.; Porto, C.S. Estrogen receptors and function in the male reproductive system. Arq. Bras. Endocrinol. Metab. 2009, 53, 923–933. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Z.Y.; Zhang, X.T.; Shen, P.; Loggie, B.W.; Chang, Y.C.; Deuel, T.F. Identification, cloning, and expression of human estrogen receptor-alpha 36, a novel variant of human estrogen receptor-alpha 66. Biochem. Bioph. Res Commun. 2005, 336, 1023–1027. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.Y.; Zhang, X.T.; Shen, P.; Loggie, B.W.; Chang, Y.; Deuel, T.F. A variant of estrogen receptor-alpha, hER-alpha 36: Transduction of estrogen- and antiestrogen-dependent membrane-initiated mitogenic signaling. Proc. Natl. Acad. Sci. USA 2006, 103, 9063–9068. [Google Scholar] [CrossRef] [Green Version]
- Li, L.; Haynes, M.P.; Bender, J.R. Plasma membrane localization and function of the estrogen receptor alpha variant (ER46) in human endothelial cells. Proc. Natl. Acad. Sci. USA 2003, 100, 4807–4812. [Google Scholar] [CrossRef] [Green Version]
- Revankar, C.M.; Cimino, D.F.; Sklar, L.A.; Arterburn, J.B.; Prossnitz, E.R. A transmembrane intracellular estrogen receptor mediates rapid cell signaling. Science 2005, 307, 1625–1630. [Google Scholar] [CrossRef] [Green Version]
- Levin, E.R. G protein-coupled receptor 30: Estrogen receptor or collaborator? Endocrinology 2009, 150, 1563–1565. [Google Scholar] [CrossRef] [Green Version]
- Revankar, C.M.; Mitchell, H.D.; Field, A.S.; Burai, R.; Corona, C.; Ramesh, C.; Sklar, L.A.; Arterburn, J.B.; Prossnitz, E.R. Synthetic estrogen derivatives demonstrate the functionality of intracellular GPR30. ACS Chem. Biol. 2007, 2, 536–544. [Google Scholar] [CrossRef]
- Thomas, P.; Dong, J. Binding and activation of the seven-transmembrane estrogen receptor GPR30 by environmental estrogens: A potential novel mechanism of endocrine disruption. J. Steroid Biochem. Mol. Biol. 2006, 102, 175–179. [Google Scholar] [CrossRef]
- Bologa, C.G.; Revankar, C.M.; Young, S.M.; Edwards, B.S.; Arterburn, J.B.; Kiselyov, A.S.; Parker, M.A.; Tkachenko, S.E.; Savchuck, N.P.; Sklar, L.A.; et al. Virtual and biomolecular screening converge on a selective agonist for GPR30. Nat. Chem. Biol. 2006, 2, 207–212. [Google Scholar] [CrossRef]
- Filardo, E.J.; Thomas, P. Minireview: G protein-coupled estrogen receptor-1, GPER-1: Its mechanism of action and role in female reproductive cancer, renal and vascular physiology. Endocrinology 2012, 153, 2953–2962. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Filardo, E.J.; Quinn, J.A.; Bland, K.I.; Frackelton, A.R., Jr. Estrogen-induced activation of Erk-1 and Erk-2 requires the G protein-coupled receptor homolog, GPR30, and occurs via trans-activation of the epidermal growth factor receptor through release of HB-EGF. Mol. Endocrinol. 2000, 14, 1649–1660. [Google Scholar] [CrossRef] [PubMed]
- Filardo, E.J. Epidermal growth factor receptor (EGFR) transactivation by estrogen via the G-protein-coupled receptor, GPR30: A novel signaling pathway with potential significance for breast cancer. J. Steroid Biochem. Mol. Biol. 2002, 80, 231–238. [Google Scholar] [CrossRef]
- Prossnitz, E.R.; Barton, M. Signaling, physiological functions and clinical relevance of the G protein-coupled estrogen receptor GPER. Prostag. Other Lipid Mediat. 2009, 89, 89–97. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prossnitz, E.R.; Maggiolini, M. Mechanisms of estrogen signaling and gene expression via GPR30. Mol. Cell. Endocrinol. 2009, 308, 32–38. [Google Scholar] [CrossRef] [Green Version]
- Prossnitz, E.R.; Barton, M. The G-protein-coupled estrogen receptor GPER in health and disease. Nat. Rev. Endocrinol. 2011, 7, 715–726. [Google Scholar] [CrossRef] [Green Version]
- Prenzel, N.; Zwick, E.; Daub, H.; Leserer, M.; Abraham, R.; Wallasch, C.; Ullrich, A. EGF receptor transactivation by G-protein-coupled receptors requires metalloproteinase cleavage of proHB-EGF. Nature 1999, 402, 884–888. [Google Scholar] [CrossRef]
- Filardo, E.J.; Quinn, J.A.; Frackelton, A.R., Jr.; Bland, K.I. Estrogen action via the G protein-coupled receptor, GPR30: Stimulation of adenylyl cyclase and cAMP-mediated attenuation of the epidermal growth factor receptor-to-MAPK signaling axis. Mol. Endocrinol. 2002, 16, 70–84. [Google Scholar] [CrossRef]
- Chimento, A.; Sirianni, R.; Zolea, F.; Bois, C.; Delalande, C.; Ando, S.; Maggiolini, M.; Aquila, S.; Carreau, S.; Pezzi, V. Gper and ESRs are expressed in rat round spermatids and mediate oestrogen-dependent rapid pathways modulating expression of cyclin B1 and Bax. Int. J. Androl. 2011, 34, 420–429. [Google Scholar] [CrossRef]
- Chimento, A.; Sirianni, R.; Delalande, C.; Silandre, D.; Bois, C.; Ando, S.; Maggiolini, M.; Carreau, S.; Pezzi, V. 17 beta-estradiol activates rapid signaling pathways involved in rat pachytene spermatocytes apoptosis through GPR30 and ER alpha. Mol. Cell. Endocrinol. 2010, 320, 136–144. [Google Scholar] [CrossRef]
- Chimento, A.; Sirianni, R.; Casaburi, I.; Ruggiero, C.; Maggiolini, M.; Ando, S.; Pezzi, V. 17beta-Estradiol activates GPER- and ESR1-dependent pathways inducing apoptosis in GC-2 cells, a mouse spermatocyte-derived cell line. Mol. Cell. Endocrinol. 2012, 355, 49–59. [Google Scholar] [CrossRef] [PubMed]
- Lucas, T.F.; Royer, C.; Siu, E.R.; Lazari, M.F.; Porto, C.S. Expression and signaling of G protein-coupled estrogen receptor 1 (GPER) in rat sertoli cells. Biol. Reprod. 2010, 83, 307–317. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qian, H.; Xuan, J.; Liu, Y.; Shi, G. Function of G-Protein-Coupled Estrogen Receptor-1 in Reproductive System Tumors. J. Immunol. Res. 2016, 2016, 7128702. [Google Scholar] [CrossRef] [Green Version]
- Chimento, A.; Sirianni, R.; Casaburi, I.; Pezzi, V. GPER Signaling in Spermatogenesis and Testicular Tumors. Front. Endocrinol. 2014, 5, 30. [Google Scholar] [CrossRef] [Green Version]
- Romano, S.N.; Gorelick, D.A. Crosstalk between nuclear and G protein-coupled estrogen receptors. Gen. Comp. Endocrinol. 2018, 261, 190–197. [Google Scholar] [CrossRef]
- Skinner, M.K.; Norton, J.N.; Mullaney, B.P.; Rosselli, M.; Whaley, P.D.; Anthony, C.T. Cell-cell interactions and the regulation of testis function. Ann. N. Y. Acad. Sci. 1991, 637, 354–363. [Google Scholar] [CrossRef] [PubMed]
- Kotula-Balak, M.; Pawlicki, P.; Milon, A.; Tworzydlo, W.; Sekula, M.; Pacwa, A.; Gorowska-Wojtowicz, E.; Bilinska, B.; Pawlicka, B.; Wiater, J.; et al. The role of G-protein-coupled membrane estrogen receptor in mouse Leydig cell function-in vivo and in vitro evaluation. Cell Tissue Res. 2018, 374, 389–412. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sewer, M.B.; Li, D. Regulation of steroid hormone biosynthesis by the cytoskeleton. Lipids 2008, 43, 1109–1115. [Google Scholar] [CrossRef] [Green Version]
- Vaucher, L.; Funaro, M.G.; Mehta, A.; Mielnik, A.; Bolyakov, A.; Prossnitz, E.R.; Schlegel, P.N.; Paduch, D.A. Activation of GPER-1 estradiol receptor downregulates production of testosterone in isolated rat Leydig cells and adult human testis. PLoS ONE 2014, 9, e92425. [Google Scholar] [CrossRef]
- Sharma, G.; Prossnitz, E.R. GPER/GPR30 Knockout Mice: Effects of GPER on Metabolism. Methods Mol. Biol. 2016, 1366, 489–502. [Google Scholar] [CrossRef] [Green Version]
- Ascoli, M. Effects of hypocholesterolemia and chronic hormonal stimulation on sterol and steroid metabolism in a Leydig cell tumor. J. Lipid Res. 1981, 22, 1247–1253. [Google Scholar]
- Milon, A.; Kaczmarczyk, M.; Pawlicki, P.; Bilinska, B.; Duliban, M.; Gorowska-Wojtowicz, E.; Tworzydlo, W.; Kotula-Balak, M. Do estrogens regulate lipid status in testicular steroidogenic Leydig cell? Acta Histochem. 2019, 121, 611–618. [Google Scholar] [CrossRef]
- Yang, P.L.; Hsu, T.H.; Wang, C.W.; Chen, R.H. Lipid droplets maintain lipid homeostasis during anaphase for efficient cell separation in budding yeast. Mol. Biol. Cell. 2016, 27, 2368–2380. [Google Scholar] [CrossRef] [Green Version]
- Gorowska-Wojtowicz, E.; Dutka, P.; Kudrycka, M.; Pawlicki, P.; Milon, A.; Plachno, B.J.; Tworzydlo, W.; Pardyak, L.; Kaminska, A.; Hejmej, A.; et al. Regulation of steroidogenic function of mouse Leydig cells: G-coupled membrane estrogen receptor and peroxisome proliferator-activated receptor partnership. J. Physiol. Pharmacol. 2018, 69. [Google Scholar] [CrossRef]
- Kotula-Balak, M.; Duliban, M.; Pawlicki, P.; Tuz, R.; Bilinska, B.; Plachno, B.J.; Arent, Z.J.; Krakowska, I.; Tarasiuk, K. The meaning of non-classical estrogen receptors and peroxisome proliferator-activated receptor for boar Leydig cell of immature testis. Acta Histochem. 2020, 122, 151526. [Google Scholar] [CrossRef]
- Vidal-Gomez, X.; Perez-Cremades, D.; Mompeon, A.; Dantas, A.P.; Novella, S.; Hermenegildo, C. MicroRNA as Crucial Regulators of Gene Expression in Estradiol-Treated Human Endothelial Cells. Cell. Physiol. Biochem. 2018, 45, 1878–1892. [Google Scholar] [CrossRef]
- Gao, G.Z.; Zhao, Y.; Li, H.X.; Li, W. Bisphenol A-elicited miR-146a-5p impairs murine testicular steroidogenesis through negative regulation of Mta3 signaling. Biochem. Biophys. Res. Commun. 2018, 501, 478–485. [Google Scholar] [CrossRef]
- Pawlicki, P.; Duliban, M.; Tuz, R.; Ptak, A.; Milon, A.; Gorowska-Wojtowicz, E.; Tworzydlo, W.; Plachno, B.J.; Bilinska, B.; Knapczyk-Stwora, K.; et al. Do G-protein coupled estrogen receptor and bisphenol A analogs influence on Leydig cell epigenetic regulation in immature boar testis ex vivo? Anim. Reprod. Sci. 2019, 207, 21–35. [Google Scholar] [CrossRef]
- Hasirci, E.; Turunc, T.; Bal, N.; Goren, M.R.; Celik, H.; Kervancioglu, E.; Dirim, A.; Tekindal, M.A.; Ozkardes, H. Distribution and number of Cajal-like cells in testis tissue with azoospermia. Kaohsiung J. Med. Sci. 2017, 33, 181–186. [Google Scholar] [CrossRef]
- Cretoiu, S.M.; Popescu, L.M. Telocytes revisited. Biomol. Concepts 2014, 5, 353–369. [Google Scholar] [CrossRef]
- Pawlicki, P.; Hejmej, A.; Milon, A.; Lustofin, K.; Plachno, B.J.; Tworzydlo, W.; Gorowska-Wojtowicz, E.; Pawlicka, B.; Kotula-Balak, M.; Bilinska, B. Telocytes in the mouse testicular interstitium: Implications of G-protein-coupled estrogen receptor (GPER) and estrogen-related receptor (ERR) in the regulation of mouse testicular interstitial cells. Protoplasma 2019, 256, 393–408. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Milon, A.; Pawlicki, P.; Rak, A.; Mlyczynska, E.; Plachno, B.J.; Tworzydlo, W.; Gorowska-Wojtowicz, E.; Bilinska, B.; Kotula-Balak, M. Telocytes are localized to testis of the bank vole (Myodes glareolus) and are affected by lighting conditions and G-coupled membrane estrogen receptor (GPER) signaling. Gen. Comp. End. 2019, 271, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Holstein, A.F.; Maekawa, M.; Nagano, T.; Davidoff, M.S. Myofibroblasts in the lamina propria of human semi-niferous tubules are dynamic structures of heterogeneous phenotype. Arch. Histol. Cytol. 1996, 59, 109–125. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Albrecht, M.; Ramsch, R.; Kohn, F.M.; Schwarzer, J.U.; Mayerhofer, A. Isolation and cultivation of human testicular peritubular cells: A new model for the investigation of fibrotic processes in the human testis and male infertility. J. Clin. Endocrinol. Metab. 2006, 91, 1956–1960. [Google Scholar] [CrossRef] [Green Version]
- Schell, C.; Albrecht, M.; Mayer, C.; Schwarzer, J.U.; Frungieri, M.B.; Mayerhofer, A. Exploring human testicular peritubular cells: Identification of secretory products and regulation by tumor necrosis factor-alpha. Endocrinology 2008, 149, 1678–1686. [Google Scholar] [CrossRef] [PubMed]
- de Kretser, D.M.; Loveland, K.L.; Meinhardt, A.; Simorangkir, D.; Wreford, N. Spermatogenesis. Hum. Reprod. 1998, 13 (Suppl. S1), 1–8. [Google Scholar] [CrossRef] [Green Version]
- Jegou, B. The Sertoli-germ cell communication network in mammals. Int. Rev. Cytol. 1993, 147, 25–96. [Google Scholar]
- Meroni, S.B.; Galardo, M.N.; Rindone, G.; Gorga, A.; Riera, M.F.; Cigorraga, S.B. Molecular Mechanisms and Signaling Pathways Involved in Sertoli Cell Proliferation. Front. Endocrinol. 2019, 10, 224. [Google Scholar] [CrossRef]
- Papadopoulos, V.; Carreau, S.; Szerman-Joly, E.; Drosdowsky, M.A.; Dehennin, L.; Scholler, R. Rat testis 17 beta-estradiol: Identification by gas chromatography-mass spectrometry and age related cellular distribution. J. Steroid. Biochem. 1986, 24, 1211–1216. [Google Scholar] [CrossRef]
- Levallet, J.; Bilinska, B.; Mittre, H.; Genissel, C.; Fresnel, J.; Carreau, S. Expression and immunolocalization of functional cytochrome P450 aromatase in mature rat testicular cells. Biol. Reprod. 1998, 58, 919–926. [Google Scholar] [CrossRef]
- Bouraima-Lelong, H.; Vanneste, M.; Delalande, C.; Zanatta, L.; Wolczynski, S.; Carreau, S. Aromatase gene expression in immature rat Sertoli cells: Age-related changes in the FSH signalling pathway. Reprod. Fert. Dev. 2010, 22, 508–515. [Google Scholar] [CrossRef] [PubMed]
- Lucas, T.F.; Siu, E.R.; Esteves, C.A.; Monteiro, H.P.; Oliveira, C.A.; Porto, C.S.; Lazari, M.F. 17beta-estradiol induces the translocation of the estrogen receptors ESR1 and ESR2 to the cell membrane, MAPK3/1 phosphorylation and proliferation of cultured immature rat Sertoli cells. Biol. Reprod. 2008, 78, 101–114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lucas, T.F.G.; Lazari, M.F.M.; Porto, C.S. Differential role of the estrogen receptors ESR1 and ESR2 on the regulation of proteins involved with proliferation and differentiation of Sertoli cells from 15-day-old rats. Mol. Cell. Endocrinol. 2014, 382, 84–96. [Google Scholar] [CrossRef] [PubMed]
- Lucas, T.F.; Pimenta, M.T.; Pisolato, R.; Lazari, M.F.; Porto, C.S. 17beta-estradiol signaling and regulation of Sertoli cell function. Spermatogenesis 2011, 1, 318–324. [Google Scholar] [CrossRef] [Green Version]
- Royer, C.; Lucas, T.F.; Lazari, M.F.; Porto, C.S. 17Beta-estradiol signaling and regulation of proliferation and apoptosis of rat Sertoli cells. Biol. Reprod. 2012, 86, 108. [Google Scholar] [CrossRef]
- Yang, W.R.; Zhu, F.W.; Zhang, J.J.; Wang, Y.; Zhang, J.H.; Lu, C.; Wang, X.Z. PI3K/Akt Activated by GPR30 and Src Regulates 17beta-Estradiol-Induced Cultured Immature Boar Sertoli Cells Proliferation. Reprod. Sci. 2017, 24, 57–66. [Google Scholar] [CrossRef]
- Ge, L.C.; Chen, Z.J.; Liu, H.Y.; Zhang, K.S.; Liu, H.; Huang, H.B.; Zhang, G.; Wong, C.K.; Giesy, J.P.; Du, J.; et al. Involvement of activating ERK1/2 through G protein coupled receptor 30 and estrogen receptor alpha/beta in low doses of bisphenol A promoting growth of Sertoli TM4 cells. Toxicol. Lett. 2014, 226, 81–89. [Google Scholar] [CrossRef]
- Maekawa, M.; Kamimura, K.; Nagano, T. Peritubular myoid cells in the testis: Their structure and function. Arch. Histol. Cytol. 1996, 59, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Schlatt, S.; Weinbauer, G.F.; Arslan, M.; Nieschlag, E. Appearance of alpha-smooth muscle actin in peritubular cells of monkey testes is induced by androgens, modulated by follicle-stimulating hormone, and maintained after hormonal withdrawal. J. Androl. 1993, 14, 340–350. [Google Scholar] [CrossRef]
- Prabhushankar, R.; Krueger, C.; Manrique, C. Membrane estrogen receptors: Their role in blood pressure regulation and cardiovascular disease. Curr. Hypertens. Rep. 2014, 16, 408. [Google Scholar] [CrossRef]
- Haas, E.; Bhattacharya, I.; Brailoiu, E.; Damjanovic, M.; Brailoiu, G.C.; Gao, X.; Mueller-Guerre, L.; Marjon, N.A.; Gut, A.; Minotti, R.; et al. Regulatory role of G protein-coupled estrogen receptor for vascular function and obesity. Circ. Res. 2009, 104, 288–291. [Google Scholar] [CrossRef] [Green Version]
- Spinnler, K.; Kohn, F.M.; Schwarzer, U.; Mayerhofer, A. Glial cell line-derived neurotrophic factor is constitutively produced by human testicular peritubular cells and may contribute to the spermatogonial stem cell niche in man. Hum. Reprod. 2010, 25, 2181–2187. [Google Scholar] [CrossRef]
- Sandner, F.; Welter, H.; Schwarzer, J.U.; Kohn, F.M.; Urbanski, H.F.; Mayerhofer, A. Expression of the oestrogen receptor GPER by testicular peritubular cells is linked to sexual maturation and male fertility. Andrology 2014, 2, 695–701. [Google Scholar] [CrossRef] [Green Version]
- Hess, R.A.; Bunick, D.; Lee, K.H.; Bahr, J.; Taylor, J.A.; Korach, K.S.; Lubahn, D.B. A role for oestrogens in the male reproductive system. Nature 1997, 390, 509–512. [Google Scholar] [CrossRef] [Green Version]
- Carreau, S.; Delalande, C.; Silandre, D.; Bourguiba, S.; Lambard, S. Aromatase and estrogen receptors in male reproduction. Mol. Cell. Endocrinol. 2006, 246, 65–68. [Google Scholar] [CrossRef]
- Hewitt, S.C.; Harrell, J.C.; Korach, K.S. Lessons in estrogen biology from knockout and transgenic animals. Annu Rev Physiol 2005, 67, 285–308. [Google Scholar] [CrossRef] [Green Version]
- Murata, Y.; Robertson, K.M.; Jones, M.E.; Simpson, E.R. Effect of estrogen deficiency in the male: The ArKO mouse model. Mol. Cell. Endocrinol. 2002, 193, 7–12. [Google Scholar] [CrossRef]
- Honda, S.; Harada, N.; Ito, S.; Takagi, Y.; Maeda, S. Disruption of sexual behavior in male aromatase-deficient mice lacking exons 1 and 2 of the cyp19 gene. Biochem. Biophys. Res. Commun. 1998, 252, 445–449. [Google Scholar] [CrossRef]
- Robertson, K.M.; O’Donnell, L.; Jones, M.E.; Meachem, S.J.; Boon, W.C.; Fisher, C.R.; Graves, K.H.; McLachlan, R.I.; Simpson, E.R. Impairment of spermatogenesis in mice lacking a functional aromatase (cyp 19) gene. Proc. Natl. Acad. Sci. USA 1999, 96, 7986–7991. [Google Scholar] [CrossRef] [Green Version]
- Cobellis, G.; Pierantoni, R.; Minucci, S.; Pernas-Alonso, R.; Meccariello, R.; Fasano, S. c-fos activity in Rana esculenta testis: Seasonal and estradiol-induced changes. Endocrinology 1999, 140, 3238–3244. [Google Scholar] [CrossRef] [PubMed]
- Chieffi, P.; Colucci-D’Amato, G.L.; Staibano, S.; Franco, R.; Tramontano, D. Estradiol-induced mitogen-activated protein kinase (extracellular signal-regulated kinase 1 and 2) activity in the frog (Rana esculenta) testis. J. Endocrinol. 2000, 167, 77–84. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chieffi, P.; Colucci D’Amato, L.; Guarino, F.; Salvatore, G.; Angelini, F. 17 beta-estradiol induces spermatogonial proliferation through mitogen-activated protein kinase (extracellular signal-regulated kinase 1/2) activity in the lizard (Podarcis s. sicula). Mol. Reprod. Dev. 2002, 61, 218–225. [Google Scholar] [CrossRef] [PubMed]
- Sirianni, R.; Chimento, A.; Ruggiero, C.; De Luca, A.; Lappano, R.; Ando, S.; Maggiolini, M.; Pezzi, V. The novel estrogen receptor, G protein-coupled receptor 30, mediates the proliferative effects induced by 17beta-estradiol on mouse spermatogonial GC-1 cell line. Endocrinology 2008, 149, 5043–5051. [Google Scholar] [CrossRef] [Green Version]
- Sheng, Z.G.; Zhu, B.Z. Low concentrations of bisphenol A induce mouse spermatogonial cell proliferation by G protein-coupled receptor 30 and estrogen receptor-alpha. Environ. Heal. Perspect. 2011, 119, 1775–1780. [Google Scholar] [CrossRef]
- Sheng, Z.G.; Huang, W.; Liu, Y.X.; Zhu, B.Z. Bisphenol A at a low concentration boosts mouse spermatogonial cell proliferation by inducing the G protein-coupled receptor 30 expression. Toxicol. Appl. Pharmacol. 2013, 267, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, M.C.; Hess, R.A.; Goldberg, E.; Millan, J.L. Immortalized germ cells undergo meiosis in vitro. Proc. Natl. Acad. Sci. USA 1994, 91, 5533–5537. [Google Scholar] [CrossRef] [Green Version]
- Wang, C.; Zhang, J.; Li, Q.; Zhang, T.; Deng, Z.; Lian, J.; Jia, D.; Li, R.; Zheng, T.; Ding, X.; et al. Low concentration of BPA induces mice spermatocytes apoptosis via GPR30. Oncotarget 2017, 8, 49005–49015. [Google Scholar] [CrossRef] [Green Version]
- Cooper, T.G. Epididymis and sperm function. Andrology 1996, 28 (Suppl. S1), 57–59. [Google Scholar]
- Krejcirova, R.; Manasova, M.; Sommerova, V.; Langhamerova, E.; Rajmon, R.; Manaskova-Postlerova, P. G protein-coupled estrogen receptor (GPER) in adult boar testes, epididymis and spermatozoa during epididymal maturation. Int. J. Biol. Macromol. 2018, 116, 113–119. [Google Scholar] [CrossRef]
- Menad, R.; Fernini, M.; Smai, S.; Bonnet, X.; Gernigon-Spychalowicz, T.; Moudilou, E.; Khammar, F.; Exbrayat, J.M. GPER1 in sand rat epididymis: Effects of seasonal variations, castration and efferent ducts ligation. Anim. Reprod. Sci. 2017, 183, 9–20. [Google Scholar] [CrossRef]
- Hess, R.A.; Fernandes, S.A.; Gomes, G.R.; Oliveira, C.A.; Lazari, M.F.; Porto, C.S. Estrogen and its receptors in efferent ductules and epididymis. J. Androl. 2011, 32, 600–613. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Traverso, G.B.; Pearl, C.A. Immunolocalization of G protein-coupled estrogen receptor in the rat epididymis. Reprod. Biol. Edocrinol. 2015, 13, 48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ge, W.; Xiao, L.; Duan, H.; Jiang, Y.; Lv, J.; Ding, Z.; Hu, J.; Zhao, X.; Zhang, Y. Androgen receptor, aromatase, oestrogen receptor alpha/beta and G protein-coupled receptor 30 expression in the testes and epididymides of adult sheep. Reprod. Domest. Anim. 2020, 55, 460–468. [Google Scholar] [CrossRef]
- Antalikova, J.; Secova, P.; Horovska, L.; Krejcirova, R.; Simonik, O.; Jankovicova, J.; Bartokova, M.; Tumova, L.; Manaskova-Postlerova, P. Missing Information from the Estrogen Receptor Puzzle: Where Are They Localized in Bull Reproductive Tissues and Spermatozoa? Cells 2020, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kotula-Balak, M.; Hejmej, A.; Lydka, M.; Cierpich, A.; Bilinska, B. Detection of aromatase, androgen, and estrogen receptors in bank vole spermatozoa. Theriogenology 2012, 78, 385–392. [Google Scholar] [CrossRef]
- Rago, V.; Siciliano, L.; Aquila, S.; Carpino, A. Detection of estrogen receptors ER-alpha and ER-beta in human ejaculated immature spermatozoa with excess residual cytoplasm. Reprod. Biol. Endocrinol. 2006, 4, 36. [Google Scholar] [CrossRef] [Green Version]
- Gunawan, A.; Kaewmala, K.; Uddin, M.J.; Cinar, M.U.; Tesfaye, D.; Phatsara, C.; Tholen, E.; Looft, C.; Schellander, K. Association study and expression analysis of porcine ESR1 as a candidate gene for boar fertility and sperm quality. Anim. Reprod. Science 2011, 128, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Rago, V.; Aquila, S.; Panza, R.; Carpino, A. Cytochrome P450arom, androgen and estrogen receptors in pig sperm. Reprod. Biol Endocrinol. 2007, 5, 23. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luconi, M.; Francavilla, F.; Porazzi, I.; Macerola, B.; Forti, G.; Baldi, E. Human spermatozoa as a model for studying membrane receptors mediating rapid nongenomic effects of progesterone and estrogens. Steroids 2004, 69, 553–559. [Google Scholar] [CrossRef]
- Rago, V.; Giordano, F.; Brunelli, E.; Zito, D.; Aquila, S.; Carpino, A. Identification of G protein-coupled estrogen receptor in human and pig spermatozoa. J. Anat. 2014, 224, 732–736. [Google Scholar] [CrossRef] [Green Version]
- Gautier, C.; Barrier-Battut, I.; Guenon, I.; Goux, D.; Delalande, C.; Bouraima-Lelong, H. Implication of the estrogen receptors GPER, ESR1, ESR2 in post-testicular maturations of equine spermatozoa. Gen. Comp. Endocrinol. 2016, 233, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Arkoun, B.; Gautier, C.; Delalande, C.; Barrier-Battut, I.; Guenon, I.; Goux, D.; Bouraima-Lelong, H. Stallion spermatozoa: Putative target of estrogens; presence of the estrogen receptors ESR1, ESR2 and identification of the estrogen-membrane receptor GPER. Gen. Comp. Endocrinol. 2014, 200, 35–43. [Google Scholar] [CrossRef]
- Cornwall, G.A. Role of posttranslational protein modifications in epididymal sperm maturation and extracellular quality control. Adv. Exp. Med. Biol. 2014, 759, 159–180. [Google Scholar] [CrossRef]
- Dacheux, J.L.; Belleannee, C.; Guyonnet, B.; Labas, V.; Teixeira-Gomes, A.P.; Ecroyd, H.; Druart, X.; Gatti, J.L.; Dacheux, F. The contribution of proteomics to understanding epididymal maturation of mammalian spermatozoa. Syst. Biol. Reprod. Med. 2012, 58, 197–210. [Google Scholar] [CrossRef]
- Prossnitz, E.R.; Oprea, T.I.; Sklar, L.A.; Arterburn, J.B. The ins and outs of GPR30: A transmembrane estrogen receptor. J. Steroid Biochem. Mol. Biol. 2008, 109, 350–353. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schwartz, N.; Verma, A.; Bivens, C.B.; Schwartz, Z.; Boyan, B.D. Rapid steroid hormone actions via membrane receptors. Bioch. Biophys. Acta 2016, 1863, 2289–2298. [Google Scholar] [CrossRef]
- Boccellino, M.; Vanacore, D.; Zappavigna, S.; Cavaliere, C.; Rossetti, S.; D’Aniello, C.; Chieffi, P.; Amler, E.; Buonerba, C.; Di Lorenzo, G.; et al. Testicular cancer from diagnosis to epigenetic factors. Oncotarget 2017, 8, 104654–104663. [Google Scholar] [CrossRef]
- Batool, A.; Karimi, N.; Wu, X.N.; Chen, S.R.; Liu, Y.X. Testicular germ cell tumor: A comprehensive review. Cell. Mol. Life Sci. 2019, 76, 1713–1727. [Google Scholar] [CrossRef]
- Baroni, T.; Arato, I.; Mancuso, F.; Calafiore, R.; Luca, G. On the Origin of Testicular Germ Cell Tumors: From Gonocytes to Testicular Cancer. Front. Endocrinol. 2019, 10, 343. [Google Scholar] [CrossRef] [PubMed]
- Albers, P.; Albrecht, W.; Algaba, F.; Bokemeyer, C.; Cohn-Cedermark, G.; Fizazi, K.; Horwich, A.; Laguna, M.P.; Nicolai, N.; Oldenburg, J. Guidelines on Testicular Cancer: 2015 Update. Eur. Urol. 2015, 68, 1054–1068. [Google Scholar] [CrossRef]
- Facchini, G.; Rossetti, S.; Berretta, M.; Cavaliere, C.; D’Aniello, C.; Iovane, G.; Mollo, G.; Capasso, M.; Della Pepa, C.; Pesce, L.; et al. Prognostic and predictive factors in testicular cancer. Eur. Rev. Med. Pharmaco. Sci. 2019, 23, 3885–3891. [Google Scholar] [CrossRef]
- Feldman, P.S.; Kovacs, K.; Horvath, E.; Adelson, G.L. Malignant Leydig cell tumor: Clinical, histologic and electron microscopic features. Cancer 1982, 49, 714–721. [Google Scholar] [CrossRef]
- Muheilan, M.M.; Shomaf, M.; Tarawneh, E.; Murshidi, M.M.; Al-Sayyed, M.R.; Murshidi, M.M. Leydig cell tumor in grey zone: A case report. Int. J. Surg. Case Rep. 2017, 35, 12–16. [Google Scholar] [CrossRef]
- Kim, I.; Young, R.H.; Scully, R.E. Leydig cell tumors of the testis. A clinicopathological analysis of 40 cases and review of the literature. Am. J. Surg. Pathol. 1985, 9, 177–192. [Google Scholar] [CrossRef] [PubMed]
- Jou, P.; Maclennan, G.T. Leydig cell tumor of the testis. J. Urol. 2009, 181, 2299–2300. [Google Scholar] [CrossRef] [PubMed]
- Sawhney, P.; Giammona, C.J.; Meistrich, M.L.; Richburg, J.H. Cisplatin-induced long-term failure of spermatogenesis in adult C57/Bl/6J mice. J. Androl. 2005, 26, 136–145. [Google Scholar]
- Grem, J.L.; Robins, H.I.; Wilson, K.S.; Gilchrist, K.; Trump, D.L. Metastatic Leydig cell tumor of the testis. Report of three cases and review of the literature. Cancer 1986, 58, 2116–2119. [Google Scholar] [CrossRef]
- Franco, R.; Boscia, F.; Gigantino, V.; Marra, L.; Esposito, F.; Ferrara, D.; Pariante, P.; Botti, G.; Caraglia, M.; Minucci, S.; et al. GPR30 is overexpressed in post-puberal testicular germ cell tumors. Cancer Biol. Ther. 2011, 11, 609–613. [Google Scholar] [CrossRef] [Green Version]
- Rago, V.; Romeo, F.; Giordano, F.; Ferraro, A.; Ando, S.; Carpino, A. Identification of ERbeta1 and ERbeta2 in human seminoma, in embryonal carcinoma and in their adjacent intratubular germ cell neoplasia. Reprod. Biol. Endocrinol. 2009, 7, 56. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guido, C.; Panza, S.; Santoro, M.; Avena, P.; Panno, M.L.; Perrotta, I.; Giordano, F.; Casaburi, I.; Catalano, S.; De Amicis, F.; et al. Estrogen receptor beta (ERbeta) produces autophagy and necroptosis in human seminoma cell line through the binding of the Sp1 on the phosphatase and tensin homolog deleted from chromosome 10 (PTEN) promoter gene. Cell Cycle 2012, 11, 2911–2921. [Google Scholar] [CrossRef] [Green Version]
- Kinugawa, K.; Hyodo, F.; Matsuki, T.; Jo, Y.; Furukawa, Y.; Ueki, A.; Tanaka, H. Establishment and characterization of a new human testicular seminoma cell line, JKT-1. Int. J. Urol. 1998, 5, 282–287. [Google Scholar] [CrossRef] [PubMed]
- Bouskine, A.; Nebout, M.; Mograbi, B.; Brucker-Davis, F.; Roger, C.; Fenichel, P. Estrogens promote human testicular germ cell cancer through a membrane-mediated activation of extracellular regulated kinase and protein kinase A. Endocrinology 2008, 149, 565–573. [Google Scholar] [CrossRef] [PubMed]
- Bouskine, A.; Nebout, M.; Brucker-Davis, F.; Benahmed, M.; Fenichel, P. Low doses of bisphenol A promote human seminoma cell proliferation by activating PKA and PKG via a membrane G-protein-coupled estrogen receptor. Environ. Health Perspect. 2009, 117, 1053–1058. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chevalier, N.; Bouskine, A.; Fenichel, P. Bisphenol A promotes testicular seminoma cell proliferation through GPER/GPR30. Int. J. Cancer 2012, 130, 241–242. [Google Scholar] [CrossRef]
- Roger, C.; Lambard, S.; Bouskine, A.; Mograbi, B.; Chevallier, D.; Nebout, M.; Pointis, G.; Carreau, S.; Fenichel, P. Estrogen-induced growth inhibition of human seminoma cells expressing estrogen receptor beta and aromatase. J. Mol. Endocrinol. 2005, 35, 191–199. [Google Scholar] [CrossRef]
- Chevalier, N.; Paul-Bellon, R.; Camparo, P.; Michiels, J.F.; Chevallier, D.; Fenichel, P. Genetic variants of GPER/GPR30, a novel estrogen-related G protein receptor, are associated with human seminoma. Int. J. Mol. Sci. 2014, 15, 1574–1589. [Google Scholar] [CrossRef] [Green Version]
- Wallacides, A.; Chesnel, A.; Ajj, H.; Chillet, M.; Flament, S.; Dumond, H. Estrogens promote proliferation of the seminoma-like TCam-2 cell line through a GPER-dependent ERalpha36 induction. Mol. Cell. Endocrinol. 2012, 350, 61–71. [Google Scholar] [CrossRef]
- Boscia, F.; Passaro, C.; Gigantino, V.; Perdona, S.; Franco, R.; Portella, G.; Chieffi, S.; Chieffi, P. High Levels of Gpr30 Protein in Human Testicular Carcinoma In Situ and Seminomas Correlate with Low Levels of Estrogen Receptor-Beta and Indicate a Switch in Estrogen Responsiveness. J. Cell. Physiol. 2015, 230, 1290–1297. [Google Scholar] [CrossRef]
- Chimento, A.; Casaburi, I.; Bartucci, M.; Patrizii, M.; Dattilo, R.; Avena, P.; Ando, S.; Pezzi, V.; Sirianni, R. Selective GPER activation decreases proliferation and activates apoptosis in tumor Leydig cells. Cell Death Dis. 2013, 4, e747. [Google Scholar] [CrossRef] [Green Version]
- Ariazi, E.A.; Brailoiu, E.; Yerrum, S.; Shupp, H.A.; Slifker, M.J.; Cunliffe, H.E.; Black, M.A.; Donato, A.L.; Arterburn, J.B.; Oprea, T.I.; et al. The G protein-coupled receptor GPR30 inhibits proliferation of estrogen receptor-positive breast cancer cells. Cancer Res. 2010, 70, 1184–1194. [Google Scholar] [CrossRef] [Green Version]
- Chan, Q.K.; Lam, H.M.; Ng, C.F.; Lee, A.Y.; Chan, E.S.; Ng, H.K.; Ho, S.M.; Lau, K.M. Activation of GPR30 inhibits the growth of prostate cancer cells through sustained activation of Erk1/2, c-jun/c-fos-dependent upregulation of p21, and induction of G(2) cell-cycle arrest. Cell Death Diff. 2010, 17, 1511–1523. [Google Scholar] [CrossRef] [PubMed]
- Gorowska-Wojtowicz, E.; Duliban, M.; Kudrycka, M.; Dutka, P.; Pawlicki, P.; Milon, A.; Zarzycka, M.; Placha, W.; Kotula-Balak, M.; Ptak, A.; et al. Leydig cell tumorigenesis–implication of G-protein coupled membrane estrogen receptor, peroxisome proliferator-activated receptor and xenoestrogen exposure. In vivo and in vitro appraisal. Tissue Cell 2019, 61, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Kotula-Balak, M.; Gorowska-Wojtowicz, E.; Milon, A.; Pawlicki, P.; Tworzydlo, W.; Plachno, B.J.; Krakowska, I.; Hejmej, A.; Wolski, J.K.; Bilinska, B. Towards understanding leydigioma: Do G protein-coupled estrogen receptor and peroxisome proliferator-activated receptor regulate lipid metabolism and steroidogenesis in Leydig cell tumors? Protoplasma 2020. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Natale, C.A.; Li, J.; Zhang, J.; Dahal, A.; Dentchev, T.; Stanger, B.Z.; Ridky, T.W. Activation of G protein-coupled estrogen receptor signaling inhibits melanoma and improves response to immune checkpoint blockade. eLife 2018, 7. [Google Scholar] [CrossRef]
- Natale, C.A.; Li, J.; Pitarresi, J.R.; Norgard, R.J.; Dentchev, T.; Capell, B.C.; Seykora, J.T.; Stanger, B.Z.; Ridky, T.W. Pharmacologic Activation of the G Protein-Coupled Estrogen Receptor Inhibits Pancreatic Ductal Adenocarcinoma. Cell. Mol. Gastroenter. Hepatol. 2020. [Google Scholar] [CrossRef]
- Phase 1 Study to Determine the MTD, Safety, Tolerability, PK and Preliminary Anti-Tumor Effects of LNS8801alone and in Combination with Pembrolizumab. Available online: https://clinicaltrials.gov/ct2/show/NCT04130516 (accessed on 6 August 2020).
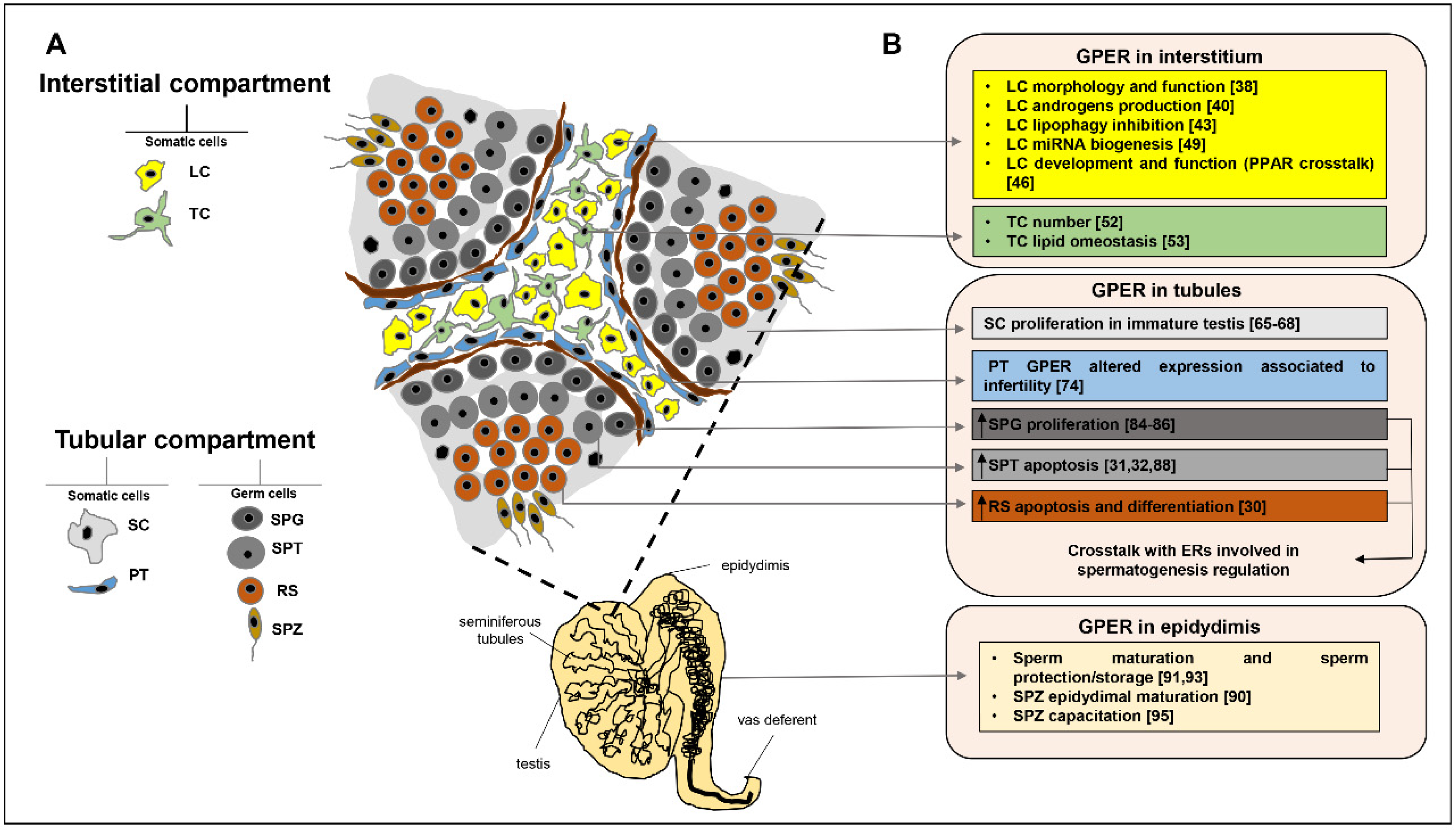
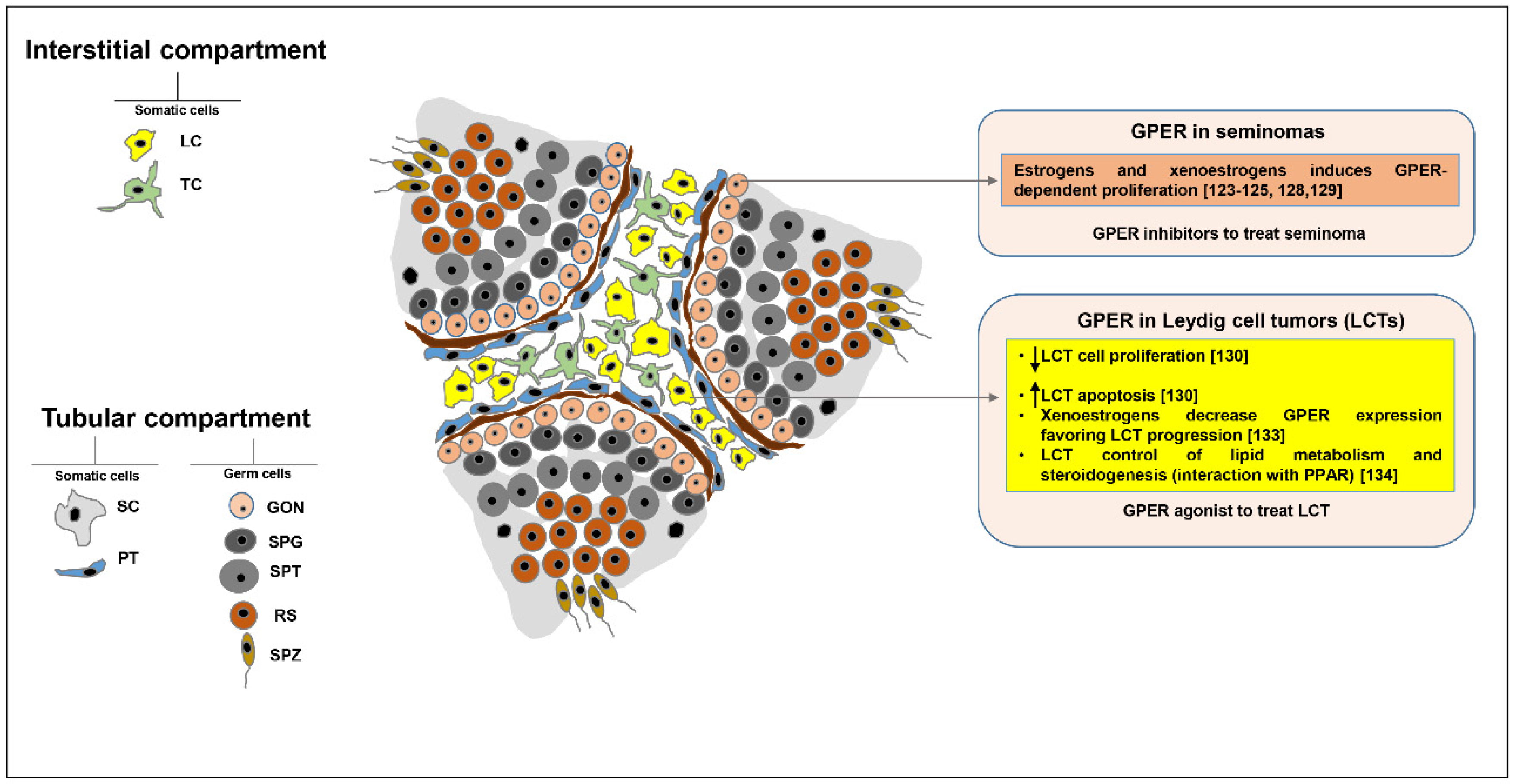
| Cell Types | Testicular Physiology | Testicular Tumors |
|---|---|---|
| SPG | GPER activation induces proliferation [84,85,86] | GPER is overexpressed in seminoma and embryonal carcinoma [119], (whereas ERα is missing [120]) GPER mediates estrogen and xenoestrogen-dependent proliferation in seminomas [123,124,125,128,129] |
| SPT | GPER activation induces apoptosis [31,32,88] | |
| RS | GPER activation induces apoptosis [30] | |
| LC | GPER is involved in: LC morphology and function [38] | LCT [where ERα is overexpressed]: GPER activation decrease cell proliferation and induces apoptosis [130] Xenoestrogens decrease GPER expression favoring LCT progression [133] GPER is involved in LCT control of lipid metabolism and steroidogenesis (interaction with PPAR) [134] |
| LC androgens production [40] | ||
| LC lipophagy inhibition [43] | ||
| LC miRNA biogenesis [49] | ||
| LC development and function [PPAR crosstalk] [46] | ||
| SC | GPER activation induces proliferation in immature testis [65,66,67,68] | Sertoli cell tumors: GPER is expressed but the role has been not identified. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chimento, A.; De Luca, A.; Nocito, M.C.; Avena, P.; La Padula, D.; Zavaglia, L.; Pezzi, V. Role of GPER-Mediated Signaling in Testicular Functions and Tumorigenesis. Cells 2020, 9, 2115. https://doi.org/10.3390/cells9092115
Chimento A, De Luca A, Nocito MC, Avena P, La Padula D, Zavaglia L, Pezzi V. Role of GPER-Mediated Signaling in Testicular Functions and Tumorigenesis. Cells. 2020; 9(9):2115. https://doi.org/10.3390/cells9092115
Chicago/Turabian StyleChimento, Adele, Arianna De Luca, Marta Claudia Nocito, Paola Avena, Davide La Padula, Lucia Zavaglia, and Vincenzo Pezzi. 2020. "Role of GPER-Mediated Signaling in Testicular Functions and Tumorigenesis" Cells 9, no. 9: 2115. https://doi.org/10.3390/cells9092115






