Ribosomopathies: New Therapeutic Perspectives
Abstract
1. Introduction
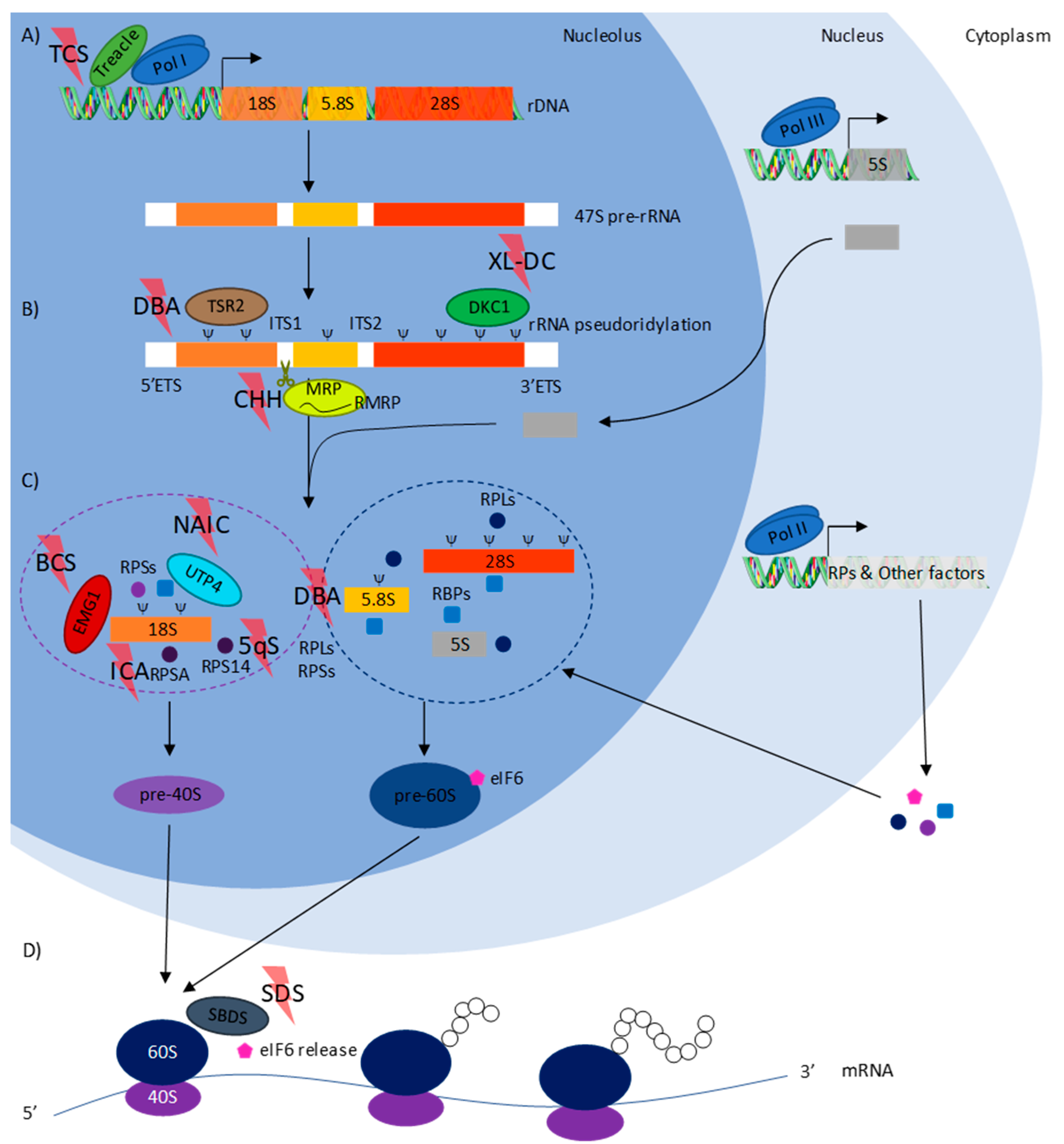
2. The Main Mechanisms of Ribosomopathies
3. Ribosomopathies with Specific Corrections
3.1. Diamond–Blackfan Anemia (DBA)
3.2. Current Treatment
3.3. Therapeutic Perspectives
3.4. Dyskeratosis Congenita (DC)
3.5. Current Treatment
3.6. Therapeutic Perspectives
3.7. Treacher Collins Syndrome (TCS)
3.8. Current Treatment
3.9. Therapeutic Perspectives
3.10. Cartilage Hair Hypoplasia (CHH)
3.11. Current Treatment
3.12. Therapeutic Perspectives
3.13. Shwachman–Diamond Syndrome (SDS)
3.14. Current Treatment
3.15. Therapeutic Perspectives
3.16. 5q-Syndrome (5q)
3.17. Current Treatment
3.18. Therapeutic Perspectives
4. Ribosomopathies Treated with Generic Guidelines
4.1. North American Indian Childhood Cirrhosis (NAIC)
4.2. Isolated Congenital Asplenia (ICA)
4.3. Neurodevelopmental Disorders (ND)
5. Ribosomopathies that Remain Untreated
Bowen-Conradi Syndrome (BCS)
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Khatter, H.; Myasnikov, A.G.; Natchiar, S.K.; Klaholz, B.P. Structure of the human 80S ribosome. Nature 2015, 520, 640–645. [Google Scholar] [CrossRef]
- Rodnina, M.V. The ribosome in action: Tuning of translational efficiency and protein folding: The Ribosome in Action. Protein Sci. 2016, 25, 1390–1406. [Google Scholar] [CrossRef]
- Kater, L.; Thoms, M.; Barrio-Garcia, C.; Cheng, J.; Ismail, S.; Ahmed, Y.L.; Bange, G.; Kressler, D.; Berninghausen, O.; Sinning, I.; et al. Visualizing the Assembly Pathway of Nucleolar Pre-60S Ribosomes. Cell 2017, 171, 1599–1610. [Google Scholar] [CrossRef]
- Aubert, M.; O’Donohue, M.F.; Lebaron, S.; Gleizes, P.E. Pre-Ribosomal RNA Processing in Human Cells: From Mechanisms to Congenital Diseases. Biomolecules 2018, 8, 123. [Google Scholar] [CrossRef]
- Prattes, M.; Lo, Y.H.; Bergler, H.; Stanley, R.E. Shaping the Nascent Ribosome: AAA-ATPases in Eukaryotic Ribosome Biogenesis. Biomolecules 2019, 9, 715. [Google Scholar] [CrossRef]
- Yelick, P.C.; Trainor, P.A. Ribosomopathies: Global process, tissue specific defects. Rare Dis. 2015, 3, e1025185. [Google Scholar] [CrossRef]
- Danilova, N.; Gazda, H.T. Ribosomopathies: How a common root can cause a tree of pathologies. Dis. Model. Mech. 2015, 8, 1013–1026. [Google Scholar] [CrossRef]
- Le Caignec, C.; Ory, B.; Lamoureux, F.; O’Donohue, M.F.; Orgebin, E.; Lindenbaum, P.; Téletchéa, S.; Saby, M.; Hurst, A.; Nelson, K.; et al. RPL13 Variants Cause Spondyloepimetaphyseal Dysplasia with Severe Short Stature. Am. J. Hum. Genet. 2019, 105, 1040–1047. [Google Scholar] [CrossRef]
- Sulima, S.O.; Hofman, I.J.F.; De Keersmaecker, K.; Dinman, J.D. How Ribosomes Translate Cancer. Cancer Discov. 2017, 7, 1069–1087. [Google Scholar] [CrossRef]
- Sulima, S.; Kampen, K.; De Keersmaecker, K. Cancer Biogenesis in Ribosomopathies. Cells 2019, 8, 229. [Google Scholar] [CrossRef]
- Pelletier, J.; Thomas, G.; Volarević, S. Ribosome biogenesis in cancer: New players and therapeutic avenues. Nat. Rev. Cancer 2018, 18, 51–63. [Google Scholar] [CrossRef]
- Aspesi, A.; Ellis, S.R. Rare ribosomopathies: Insights into mechanisms of cancer. Nat. Rev. Cancer 2019, 19, 228–238. [Google Scholar] [CrossRef]
- Bellodi, C.; Krasnykh, O.; Haynes, N.; Theodoropoulou, M.; Peng, G.; Montanaro, L.; Ruggero, D. Loss of function of the tumor suppressor DKC1 perturbs p27 translation control and contributes to pituitary tumorigenesis. Cancer Res. 2010, 70, 6026–6035. [Google Scholar] [CrossRef]
- Montanaro, L.; Calienni, M.; Bertoni, S.; Rocchi, L.; Sansone, P.; Storci, G.; Santini, D.; Ceccarelli, C.; Taffurelli, M.; Carnicelli, D.; et al. Novel dyskerin-mediated mechanism of p53 inactivation through defective mRNA translation. Cancer Res. 2010, 70, 4767–4777. [Google Scholar] [CrossRef]
- Luzzatto, L.; Karadimitris, A. Dyskeratosis and ribosomal rebellion. Nat. Genet. 1998, 19, 6–7. [Google Scholar] [CrossRef]
- Farley-Barnes, K.I.; Ogawa, L.M.; Baserga, S.J. Ribosomopathies: Old Concepts, New Controversies. Trends Genet. 2019, 35, 754–767. [Google Scholar] [CrossRef]
- Mills, E.W.; Green, R. Ribosomopathies: There’s strength in numbers. Science 2017, 358, eaan2755. [Google Scholar] [CrossRef]
- Khajuria, R.K.; Munschauer, M.; Ulirsch, J.C.; Fiorini, C.; Ludwig, L.S.; McFarland, S.K.; Abdulhay, N.J.; Specht, H.; Keshishian, H.; Mani, D.R.; et al. Ribosome Levels Selectively Regulate Translation and Lineage Commitment in Human Hematopoiesis. Cell 2018, 173, 90–103. [Google Scholar] [CrossRef]
- Shi, Z.; Fujii, K.; Kovary, K.M.; Genuth, N.R.; Röst, H.L.; Teruel, M.N.; Barna, M. Heterogeneous Ribosomes Preferentially Translate Distinct Subpools of mRNAs Genome-wide. Mol. Cell 2017, 67, 71–83. [Google Scholar] [CrossRef]
- Cheng, Z.; Mugler, C.F.; Keskin, A.; Hodapp, S.; Chan, L.Y.L.; Weis, K.; Mertins, P.; Regev, A.; Jovanovic, M.; Brar, G.A. Small and Large Ribosomal Subunit Deficiencies Lead to Distinct Gene Expression Signatures that Reflect Cellular Growth Rate. Mol. Cell 2019, 73, 36–47. [Google Scholar] [CrossRef]
- Vlachos, A.; Ball, S.; Dahl, N.; Alter, B.P.; Sheth, S.; Ramenghi, U.; Meerpohl, J.; Karlsson, S.; Liu, J.M.; Leblanc, T.; et al. Diagnosing and treating Diamond Blackfan anaemia: Results of an international clinical consensus conference. Br. J. Haematol. 2008, 142, 859–876. [Google Scholar] [CrossRef] [PubMed]
- Lipton, J.M.; Atsidaftos, E.; Zyskind, I.; Vlachos, A. Improving clinical care and elucidating the pathophysiology of Diamond Blackfan anemia: An update from the Diamond Blackfan Anemia Registry. Pediatric Blood Cancer 2006, 46, 558–564. [Google Scholar] [CrossRef] [PubMed]
- Draptchinskaia, N.; Gustavsson, P.; Andersson, B.; Pettersson, M.; Willig, T.N.; Dianzani, I.; Ball, S.; Tchernia, G.; Klar, J.; Matsson, H.; et al. The gene encoding ribosomal protein S19 is mutated in Diamond-Blackfan anaemia. Nat. Genet. 1999, 21, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Gazda, H.T.; Grabowska, A.; Merida-Long, L.B.; Latawiec, E.; Schneider, H.E.; Lipton, J.M.; Vlachos, A.; Atsidaftos, E.; Ball, S.E.; Orfali, K.A.; et al. Ribosomal Protein S24 Gene Is Mutated in Diamond-Blackfan Anemia. Am. J. Hum. Genet. 2006, 79, 1110–1118. [Google Scholar] [CrossRef] [PubMed]
- Cmejla, R.; Cmejlova, J.; Handrkova, H.; Petrak, J.; Pospisilova, D. Ribosomal protein S17 gene (RPS17) is mutated in Diamond-Blackfan anemia. Hum. Mutat. 2007, 28, 1178–1182. [Google Scholar] [CrossRef]
- Farrar, J.E.; Nater, M.; Caywood, E.; McDevitt, M.A.; Kowalski, J.; Takemoto, C.M.; Talbot, C.C.; Meltzer, P.; Esposito, D.; Beggs, A.H.; et al. Abnormalities of the large ribosomal subunit protein, Rpl35a, in Diamond-Blackfan anemia. Blood 2008, 112, 1582–1592. [Google Scholar] [CrossRef]
- Gazda, H.T.; Sheen, M.R.; Vlachos, A.; Choesmel, V.; O’Donohue, M.F.; Schneider, H.; Darras, N.; Hasman, C.; Sieff, C.A.; Newburger, P.E.; et al. Ribosomal Protein L5 and L11 Mutations Are Associated with Cleft Palate and Abnormal Thumbs in Diamond-Blackfan Anemia Patients. Am. J. Hum. Genet. 2008, 83, 769–780. [Google Scholar] [CrossRef]
- Doherty, L.; Sheen, M.R.; Vlachos, A.; Choesmel, V.; O’Donohue, M.F.; Clinton, C.; Schneider, H.E.; Sieff, C.A.; Newburger, P.E.; Ball, S.E.; et al. Ribosomal Protein Genes RPS10 and RPS26 Are Commonly Mutated in Diamond-Blackfan Anemia. Am. J. Hum. Genet. 2010, 86, 222–228. [Google Scholar] [CrossRef]
- Gazda, H.T.; Preti, M.; Sheen, M.R.; O’Donohue, M.F.; Vlachos, A.; Davies, S.M.; Kattamis, A.; Doherty, L.; Landowski, M.; Buros, C.; et al. Frameshift mutation in p53 regulator RPL26 is associated with multiple physical abnormalities and a specific pre-ribosomal RNA processing defect in diamond-blackfan anemia. Hum. Mutat. 2012, 33, 1037–1044. [Google Scholar] [CrossRef]
- Landowski, M.; O’Donohue, M.F.; Buros, C.; Ghazvinian, R.; Montel-Lehry, N.; Vlachos, A.; Sieff, C.A.; Newburger, P.E.; Niewiadomska, E.; Matysiak, M.; et al. Novel deletion of RPL15 identified by array-comparative genomic hybridization in Diamond–Blackfan anemia. Hum. Genet. 2013, 132, 1265–1274. [Google Scholar] [CrossRef]
- Mirabello, L.; Khincha, P.P.; Ellis, S.R.; Giri, N.; Brodie, S.; Chandrasekharappa, S.C.; Donovan, F.X.; Zhou, W.; Hicks, B.D.; Boland, J.F.; et al. Novel and known ribosomal causes of Diamond-Blackfan anaemia identified through comprehensive genomic characterisation. J. Med. Genet. 2017, 54, 417–425. [Google Scholar] [CrossRef] [PubMed]
- Ulirsch, J.C.; Verboon, J.M.; Kazerounian, S.; Guo, M.H.; Yuan, D.; Ludwig, L.S.; Handsaker, R.E.; Abdulhay, N.J.; Fiorini, C.; Genovese, G.; et al. The Genetic Landscape of Diamond-Blackfan Anemia. Am. J. Hum. Genet. 2018, 103, 930–947. [Google Scholar] [CrossRef]
- Lezzerini, M.; Penzo, M.; O’Donohue, M.F.; Marques dos Santos Vieira, C.; Saby, M.; Elfrink, H.L.; Diets, I.J.; Hesse, A.M.; Couté, Y.; Gastou, M.; et al. Ribosomal protein gene RPL9 variants can differentially impair ribosome function and cellular metabolism. Nucleic Acids Res. 2020, 48, 770–787. [Google Scholar] [CrossRef]
- Farrar, J.E.; Vlachos, A.; Atsidaftos, E.; Carlson-Donohoe, H.; Markello, T.C.; Arceci, R.J.; Ellis, S.R.; Lipton, J.M.; Bodine, D.M. Ribosomal protein gene deletions in Diamond-Blackfan anemia. Blood 2011, 118, 6943–6951. [Google Scholar] [CrossRef]
- Kuramitsu, M.; Sato-Otsubo, A.; Morio, T.; Takagi, M.; Toki, T.; Terui, K.; Wang, R.; Kanno, H.; Ohga, S.; Ohara, A.; et al. Extensive gene deletions in Japanese patients with Diamond-Blackfan anemia. Blood 2012, 119, 2376–2384. [Google Scholar] [CrossRef]
- Quarello, P.; Garelli, E.; Brusco, A.; Carando, A.; Mancini, C.; Pappi, P.; Vinti, L.; Svahn, J.; Dianzani, I.; Ramenghi, U. High frequency of ribosomal protein gene deletions in Italian Diamond-Blackfan anemia patients detected by multiplex ligation-dependent probe amplification assay. Haematologica 2012, 97, 1813–1817. [Google Scholar] [CrossRef]
- Idol, R.A.; Robledo, S.; Du, H.Y.; Crimmins, D.L.; Wilson, D.B.; Ladenson, J.H.; Bessler, M.; Mason, P.J. Cells depleted for RPS19, a protein associated with Diamond Blackfan Anemia, show defects in 18S ribosomal RNA synthesis and small ribosomal subunit production. Blood Cells Mol. Dis. 2007, 39, 35–43. [Google Scholar] [CrossRef]
- Flygare, J.; Aspesi, A.; Bailey, J.C.; Miyake, K.; Caffrey, J.M.; Karlsson, S.; Ellis, S.R. Human RPS19, the gene mutated in Diamond-Blackfan anemia, encodes a ribosomal protein required for the maturation of 40S ribosomal subunits. Blood 2007, 109, 980–986. [Google Scholar] [CrossRef]
- Choesmel, V.; Bacqueville, D.; Rouquette, J.; Noaillac-Depeyre, J.; Fribourg, S.; Crétien, A.; Leblanc, T.; Tchernia, G.; Da Costa, L.; Gleizes, P.E. Impaired ribosome biogenesis in Diamond-Blackfan anemia. Blood 2007, 109, 1275–1283. [Google Scholar] [CrossRef]
- Choesmel, V.; Fribourg, S.; Aguissa-Touré, A.H.; Pinaud, N.; Legrand, P.; Gazda, H.T.; Gleizes, P.E. Mutation of ribosomal protein RPS24 in Diamond-Blackfan anemia results in a ribosome biogenesis disorder. Hum. Mol. Genet. 2008, 17, 1253–1263. [Google Scholar] [CrossRef]
- Sankaran, V.G.; Ghazvinian, R.; Do, R.; Thiru, P.; Vergilio, J.A.; Beggs, A.H.; Sieff, C.A.; Orkin, S.H.; Nathan, D.G.; Lander, E.S.; et al. Exome sequencing identifies GATA1 mutations resulting in Diamond-Blackfan anemia. J. Clin. Investig. 2012, 122, 2439–2443. [Google Scholar] [CrossRef] [PubMed]
- Willig, T.N.; Niemeyer, C.M.; Leblanc, T.; Tiemann, C.; Robert, A.; Budde, J.; Lambiliotte, A.; Kohne, E.; Souillet, G.; Eber, S.; et al. Identification of new prognosis factors from the clinical and epidemiologic analysis of a registry of 229 Diamond-Blackfan anemia patients. DBA group of Société d’Hématologie et d’Immunologie Pédiatrique (SHIP), Gesellshaft für Pädiatrische Onkologie und Hämatologie (GPOH), and the European Society for Pediatric Hematology and Immunology (ESPHI). Pediatric Res. 1999, 46, 553–561. [Google Scholar] [CrossRef]
- Behfar, M.; Koochakzadeh, L.; Yazdanian, N.; Salajegheh, P.; Rostami, T.; Khodayari-Namini, N.; Ghavamzadeh, A.; Hamidieh, A.A. Outcome of allogeneic hematopoietic stem cell transplantation on diamond-blackfan anemia using busulfan-based myeloablative regimen. Turk. J. Pediatrics 2019, 61, 407. [Google Scholar] [CrossRef] [PubMed]
- Payne, E.M.; Virgilio, M.; Narla, A.; Sun, H.; Levine, M.; Paw, B.H.; Berliner, N.; Look, A.T.; Ebert, B.L.; Khanna-Gupta, A. L-leucine improves the anemia and developmental defects associated with Diamond-Blackfan anemia and del(5q) MDS by activating the mTOR pathway. Blood 2012, 120, 2214–2224. [Google Scholar] [CrossRef]
- Jaako, P.; Debnath, S.; Olsson, K.; Bryder, D.; Flygare, J.; Karlsson, S. Dietary L-leucine improves the anemia in a mouse model for Diamond-Blackfan anemia. Blood 2012, 120, 2225–2228. [Google Scholar] [CrossRef]
- Roux, P.P.; Topisirovic, I. Signaling Pathways Involved in the Regulation of mRNA Translation. Mol. Cell. Biol. 2018, 38. [Google Scholar] [CrossRef]
- Dussiot, M.; Maciel, T.T.; Fricot, A.; Chartier, C.; Negre, O.; Veiga, J.; Grapton, D.; Paubelle, E.; Payen, E.; Beuzard, Y.; et al. An activin receptor IIA ligand trap corrects ineffective erythropoiesis in β-thalassemia. Nat. Med. 2014, 20, 398–407. [Google Scholar] [CrossRef]
- Ear, J.; Huang, H.; Wilson, T.; Tehrani, Z.; Lindgren, A.; Sung, V.; Laadem, A.; Daniel, T.O.; Chopra, R.; Lin, S. RAP-011 improves erythropoiesis in zebrafish model of Diamond-Blackfan anemia through antagonizing lefty1. Blood 2015, 126, 880–890. [Google Scholar] [CrossRef]
- Bewersdorf, J.P.; Zeidan, A.M. Transforming growth factor (TGF)-β pathway as a therapeutic target in lower risk myelodysplastic syndromes. Leukemia 2019, 33, 1303–1312. [Google Scholar] [CrossRef]
- Macari, E.R.; Taylor, A.M.; Raiser, D.; Siva, K.; McGrath, K.; Humphries, J.M.; Flygare, J.; Ebert, B.L.; Zon, L.I. Calmodulin Inhibition Rescues DBA Models with Ribosomal Protein Deficiency through Reduction of RSK Signaling. Blood 2016, 128, 332. [Google Scholar] [CrossRef]
- Doulatov, S.; Vo, L.T.; Macari, E.R.; Wahlster, L.; Kinney, M.A.; Taylor, A.M.; Barragan, J.; Gupta, M.; McGrath, K.; Lee, H.Y.; et al. Drug discovery for Diamond-Blackfan anemia using reprogrammed hematopoietic progenitors. Sci. Transl. Med. 2017, 9. [Google Scholar] [CrossRef]
- Heijnen, H.F.; van Wijk, R.; Pereboom, T.C.; Goos, Y.J.; Seinen, C.W.; van Oirschot, B.A.; van Dooren, R.; Gastou, M.; Giles, R.H.; van Solinge, W.; et al. Ribosomal protein mutations induce autophagy through S6 kinase inhibition of the insulin pathway. PLoS Genet. 2014, 10, e1004371. [Google Scholar] [CrossRef]
- Winkler, T.; Townsley, D.M.; Desmond, R.; Dumitriu, B.; Grasmeder, S.; Young, N.; Dunbar, C.E. Successful Treatment of a Diamond-Blackfan Anemia (DBA) Patient with Eltrombopag. Blood 2016, 128, 2682. [Google Scholar] [CrossRef]
- Ghanima, W.; Cooper, N.; Rodeghiero, F.; Godeau, B.; Bussel, J.B. Thrombopoietin receptor agonists: Ten years later. Haematologica 2019, 104, 1112–1123. [Google Scholar] [CrossRef]
- Chai, K.Y.; Quijano, C.J.; Chiruka, S. Danazol: An Effective and Underutilised Treatment Option in Diamond-Blackfan Anaemia. Case Rep. Hematol. 2019, 2019, 4684156. [Google Scholar] [CrossRef]
- Shahidi, N.T.; Diamond, L.K. Testosterone-induced remission in aplastic anemia of both acquired and congenital types. Further observations in 24 cases. N. Engl. J. Med. 1961, 264, 953–967. [Google Scholar] [CrossRef]
- Seip, M. Malignant tumors in two patients with Diamond-Blackfan anemia treated with corticosteroids and androgens. Pediatric Hematol. Oncol. 1994, 11, 423–426. [Google Scholar] [CrossRef]
- Knight, S.; Vulliamy, T.; Copplestone, A.; Gluckman, E.; Mason, P.; Dokal, I. Dyskeratosis Congenita (DC) Registry: Identification of new features of DC. Br. J. Haematol. 1998, 103, 990–996. [Google Scholar] [CrossRef]
- Armanios, M.; Chen, J.L.; Chang, Y.P.C.; Brodsky, R.A.; Hawkins, A.; Griffin, C.A.; Eshleman, J.R.; Cohen, A.R.; Chakravarti, A.; Hamosh, A.; et al. Haploinsufficiency of telomerase reverse transcriptase leads to anticipation in autosomal dominant dyskeratosis congenita. Proc. Natl. Acad. Sci. USA 2005, 102, 15960–15964. [Google Scholar] [CrossRef]
- Vulliamy, T.J.; Walne, A.; Baskaradas, A.; Mason, P.J.; Marrone, A.; Dokal, I. Mutations in the reverse transcriptase component of telomerase (TERT) in patients with bone marrow failure. Blood Cells Mol. Dis. 2005, 34, 257–263. [Google Scholar] [CrossRef]
- Vulliamy, T.; Marrone, A.; Goldman, F.; Dearlove, A.; Bessler, M.; Mason, P.J.; Dokal, I. The RNA component of telomerase is mutated in autosomal dominant dyskeratosis congenita. Nature 2001, 413, 432–435. [Google Scholar] [CrossRef] [PubMed]
- Kirwan, M.; Dokal, I. Dyskeratosis congenita: A genetic disorder of many faces. Clin. Genet. 2008, 73, 103–112. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, S. Evaluation and Management of Hematopoietic Failure in Dyskeratosis Congenita. Hematol. Oncol. Clin. N. Am. 2018, 32, 669–685. [Google Scholar] [CrossRef] [PubMed]
- Tollervey, D.; Kiss, T. Function and synthesis of small nucleolar RNAs. Curr. Opin. Cell Biol. 1997, 9, 337–342. [Google Scholar] [CrossRef]
- Filipowicz, W.; Pogacić, V. Biogenesis of small nucleolar ribonucleoproteins. Curr. Opin. Cell Biol. 2002, 14, 319–327. [Google Scholar] [CrossRef]
- Yoon, A.; Peng, G.; Brandenburger, Y.; Brandenburg, Y.; Zollo, O.; Xu, W.; Rego, E.; Ruggero, D. Impaired control of IRES-mediated translation in X-linked dyskeratosis congenita. Science 2006, 312, 902–906. [Google Scholar] [CrossRef]
- Thumati, N.R.; Zeng, X.L.; Au, H.H.T.; Jang, C.J.; Jan, E.; Wong, J.M.Y. Severity of X-linked Dyskeratosis Congenita (DKCX) Cellular Defects Is not Directly Related to Dyskerin (DKC1) Activity in Ribosomal RNA Biogenesis or mRNA Translation. Hum. Mutat. 2013, 34, 1698–1707. [Google Scholar] [CrossRef]
- Niewisch, M.R.; Savage, S.A. An update on the biology and management of dyskeratosis congenita and related telomere biology disorders. Expert. Rev. Hematol. 2019, 12, 1037–1052. [Google Scholar] [CrossRef]
- Olnes, M.J.; Scheinberg, P.; Calvo, K.R.; Desmond, R.; Tang, Y.; Dumitriu, B.; Parikh, A.R.; Soto, S.; Biancotto, A.; Feng, X.; et al. Eltrombopag and improved hematopoiesis in refractory aplastic anemia. N. Engl. J. Med. 2012, 367, 11–19. [Google Scholar] [CrossRef]
- Islam, A.; Rafiq, S.; Kirwan, M.; Walne, A.; Cavenagh, J.; Vulliamy, T.; Dokal, I. Haematological recovery in dyskeratosis congenita patients treated with danazol. Br. J. Haematol. 2013, 162, 854–856. [Google Scholar] [CrossRef]
- Khincha, P.P.; Wentzensen, I.M.; Giri, N.; Alter, B.P.; Savage, S.A. Response to androgen therapy in patients with dyskeratosis congenita. Br. J. Haematol. 2014, 165, 349–357. [Google Scholar] [CrossRef]
- Calado, R.T.; Yewdell, W.T.; Wilkerson, K.L.; Regal, J.A.; Kajigaya, S.; Stratakis, C.A.; Young, N.S. Sex hormones, acting on the TERT gene, increase telomerase activity in human primary hematopoietic cells. Blood 2009, 114, 2236–2243. [Google Scholar] [CrossRef]
- Townsley, D.M.; Dumitriu, B.; Liu, D.; Biancotto, A.; Weinstein, B.; Chen, C.; Hardy, N.; Mihalek, A.D.; Lingala, S.; Kim, Y.J.; et al. Danazol Treatment for Telomere Diseases. N. Engl. J. Med. 2016, 374, 1922–1931. [Google Scholar] [CrossRef]
- Dixon, M.J. Treacher Collins syndrome. Hum. Mol. Genet. 1996, 5, 1391–1393. [Google Scholar] [CrossRef]
- Dixon, J.; Jones, N.C.; Sandell, L.L.; Jayasinghe, S.M.; Crane, J.; Rey, J.P.; Dixon, M.J.; Trainor, P.A. Tcof1/Treacle is required for neural crest cell formation and proliferation deficiencies that cause craniofacial abnormalities. Proc. Natl. Acad. Sci. USA 2006, 103, 13403–13408. [Google Scholar] [CrossRef]
- Marsh, K.L.; Dixon, J.; Dixon, M.J. Mutations in the Treacher Collins Syndrome Gene Lead to Mislocalization of the Nucleolar Protein Treacle. Hum. Mol. Genet. 1998, 7, 1795–1800. [Google Scholar] [CrossRef]
- Winokur, S.T.; Shiang, R. The Treacher Collins syndrome (TCOF1) gene product, treacle, is targeted to the nucleolus by signals in its C-terminus. Hum. Mol. Genet. 1998, 7, 1947–1952. [Google Scholar] [CrossRef]
- Lin, C.I.; Yeh, N.H. Treacle recruits RNA polymerase I complex to the nucleolus that is independent of UBF. Biochem. Biophys. Res. Commun. 2009, 386, 396–401. [Google Scholar] [CrossRef]
- Dauwerse, J.G.; Dixon, J.; Seland, S.; Ruivenkamp, C.A.L.; van Haeringen, A.; Hoefsloot, L.H.; Peters, D.J.M.; Boers, A.C.; Daumer-Haas, C.; Maiwald, R.; et al. Mutations in genes encoding subunits of RNA polymerases I and III cause Treacher Collins syndrome. Nat. Genet. 2011, 43, 20–22. [Google Scholar] [CrossRef]
- Vincent, M.; Geneviève, D.; Ostertag, A.; Marlin, S.; Lacombe, D.; Martin-Coignard, D.; Coubes, C.; David, A.; Lyonnet, S.; Vilain, C.; et al. Treacher Collins syndrome: A clinical and molecular study based on a large series of patients. Genet. Med. 2016, 18, 49–56. [Google Scholar] [CrossRef]
- Plomp, R.G.; van Lieshout, M.J.S.; Joosten, K.F.M.; Wolvius, E.B.; van der Schroeff, M.P.; Versnel, S.L.; Poublon, R.M.L.; Mathijssen, I.M.J. Treacher Collins Syndrome: A Systematic Review of Evidence-Based Treatment and Recommendations. Plast. Reconstr. Surg. 2016, 137, 191–204. [Google Scholar] [CrossRef]
- Weiss, C.; Uziel, O.; Wolach, O.; Nordenberg, J.; Beery, E.; Bulvick, S.; Kanfer, G.; Cohen, O.; Ram, R.; Bakhanashvili, M.; et al. Differential downregulation of telomerase activity by bortezomib in multiple myeloma cells-multiple regulatory pathways in vitro and ex vivo. Br. J. Cancer 2012, 107, 1844–1852. [Google Scholar] [CrossRef]
- Shalem-Cohavi, N.; Beery, E.; Nordenberg, J.; Rozovski, U.; Raanani, P.; Lahav, M.; Uziel, O. The Effects of Proteasome Inhibitors on Telomerase Activity and Regulation in Multiple Myeloma Cells. Int. J. Mol. Sci. 2019, 20, 2509. [Google Scholar] [CrossRef]
- Rosas, M.G.; Lorenzatti, A.; Porcel de Peralta, M.S.; Calcaterra, N.B.; Coux, G. Proteasomal inhibition attenuates craniofacial malformations in a zebrafish model of Treacher Collins Syndrome. Biochem. Pharmacol. 2019, 163, 362–370. [Google Scholar] [CrossRef]
- De Peralta, M.S.P.; Mouguelar, V.S.; Sdrigotti, M.A.; Ishiy, F.A.A.; Fanganiello, R.D.; Passos-Bueno, M.R.; Coux, G.; Calcaterra, N.B. Cnbp ameliorates Treacher Collins Syndrome craniofacial anomalies through a pathway that involves redox-responsive genes. Cell Death Dis. 2016, 7, e2397. [Google Scholar] [CrossRef]
- Mäkitie, O.; Kaitila, I. Cartilage-hair hypoplasia—Clinical manifestations in 108 Finnish patients. Eur. J. Pediatric 1993, 152, 211–217. [Google Scholar] [CrossRef]
- Sulisalo, T.; Francomano, C.A.; Sistonen, P.; Maher, J.F.; McKusick, V.A.; de la Chapelle, A.; Kaitila, I. High-Resolution Genetic Mapping of the Cartilage-Hair Hypoplasia (CHH) Gene in Amish and Finnish Families. Genomics 1994, 20, 347–353. [Google Scholar] [CrossRef]
- Mäkitie, O. Cartilage-hair hypoplasia in Finland: Epidemiological and genetic aspects of 107 patients. J. Med. Genet. 1992, 29, 652–655. [Google Scholar] [CrossRef]
- Ridanpää, M.; van Eenennaam, H.; Pelin, K.; Chadwick, R.; Johnson, C.; Yuan, B.; vanVenrooij, W.; Pruijn, G.; Salmela, R.; Rockas, S.; et al. Mutations in the RNA Component of RNase MRP Cause a Pleiotropic Human Disease, Cartilage-Hair Hypoplasia. Cell 2001, 104, 195–203. [Google Scholar] [CrossRef]
- Nakashima, E.; Tran, J.R.; Welting, T.J.M.; Pruijn, G.J.M.; Hirose, Y.; Nishimura, G.; Ohashi, H.; Schurman, S.H.; Cheng, J.; Candotti, F.; et al. Cartilage hair hypoplasia mutations that lead to RMRP promoter inefficiency or RNA transcript instability. Am. J. Med. Genet. A 2007, 143A, 2675–2681. [Google Scholar] [CrossRef]
- Steinbusch, M.M.F.; Caron, M.M.J.; Surtel, D.A.M.; Friedrich, F.; Lausch, E.; Pruijn, G.J.M.; Verhesen, W.; Schroen, B.L.M.; van Rhijn, L.W.; Zabel, B.; et al. Expression of RMRP RNA is regulated in chondrocyte hypertrophy and determines chondrogenic differentiation. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef]
- Sun, X.; Zhang, R.; Liu, M.; Chen, H.; Chen, L.; Luo, F.; Zhang, D.; Huang, J.; Li, F.; Ni, Z.; et al. Rmrp Mutation Disrupts Chondrogenesis and Bone Ossification in Zebrafish Model of Cartilage-Hair Hypoplasia via Enhanced Wnt/β-Catenin Signaling. J. Bone Miner. Res. 2019, 34, 2101–2116. [Google Scholar] [CrossRef]
- Ammann, R.A.; Duppenthaler, A.; Bux, J.; Aebi, C. Granulocyte colony-stimulating factor-responsive chronic neutropenia in cartilage-hair hypoplasia. J. Pediatric Hematol. Oncol. 2004, 26, 379–381. [Google Scholar] [CrossRef]
- Guggenheim, R.; Somech, R.; Grunebaum, E.; Atkinson, A.; Roifman, C.M. Bone marrow transplantation for cartilage-hair-hypoplasia. Bone Marrow Transplant. 2006, 38, 751–756. [Google Scholar] [CrossRef][Green Version]
- Obara-Moszynska, M.; Wielanowska, W.; Rojek, A.; Wolnik-Brzozowska, D.; Niedziela, M. Treatment of cartilage-hair hypoplasia with recombinant human growth hormone: RhGH for cartilage-hair hypoplasia. Pediatric Int. 2013, 55, e162–e164. [Google Scholar] [CrossRef]
- Bocca, G.; Weemaes, C.M.; van der Burgt, I.; Otten, B.J. Growth hormone treatment in cartilage-hair hypoplasia: Effects on growth and the immune system. J. Pediatric Endocrinol. Metab. 2004, 17, 47–54. [Google Scholar] [CrossRef]
- Rothbaum, R.; Perrault, J.; Vlachos, A.; Cipolli, M.; Alter, B.P.; Burroughs, S.; Durie, P.; Elghetany, M.T.; Grand, R.; Hubbard, V.; et al. Shwachman-Diamond syndrome: Report from an international conference. J. Pediatric 2002, 141, 266–270. [Google Scholar] [CrossRef]
- Boocock, G.R.B.; Morrison, J.A.; Popovic, M.; Richards, N.; Ellis, L.; Durie, P.R.; Rommens, J.M. Mutations in SBDS are associated with Shwachman-Diamond syndrome. Nat. Genet. 2003, 33, 97–101. [Google Scholar] [CrossRef]
- Finch, A.J.; Hilcenko, C.; Basse, N.; Drynan, L.F.; Goyenechea, B.; Menne, T.F.; González Fernández, A.; Simpson, P.; D’Santos, C.S.; Arends, M.J.; et al. Uncoupling of GTP hydrolysis from eIF6 release on the ribosome causes Shwachman-Diamond syndrome. Genes Dev. 2011, 25, 917–929. [Google Scholar] [CrossRef]
- Weis, F.; Giudice, E.; Churcher, M.; Jin, L.; Hilcenko, C.; Wong, C.C.; Traynor, D.; Kay, R.R.; Warren, A.J. Mechanism of eIF6 release from the nascent 60S ribosomal subunit. Nat. Struct. Mol. Biol. 2015, 22, 914–919. [Google Scholar] [CrossRef]
- Tummala, H.; Walne, A.J.; Williams, M.; Bockett, N.; Collopy, L.; Cardoso, S.; Ellison, A.; Wynn, R.; Leblanc, T.; Fitzgibbon, J.; et al. DNAJC21 Mutations Link a Cancer-Prone Bone Marrow Failure Syndrome to Corruption in 60S Ribosome Subunit Maturation. Am. J. Hum. Genet. 2016, 99, 115–124. [Google Scholar] [CrossRef]
- Stepensky, P.; Chacón-Flores, M.; Kim, K.H.; Abuzaitoun, O.; Bautista-Santos, A.; Simanovsky, N.; Siliqi, D.; Altamura, D.; Méndez-Godoy, A.; Gijsbers, A.; et al. Mutations in EFL1, an SBDS partner, are associated with infantile pancytopenia, exocrine pancreatic insufficiency and skeletal anomalies in aShwachman-Diamond like syndrome. J. Med. Genet. 2017, 54, 558–566. [Google Scholar] [CrossRef]
- Carapito, R.; Konantz, M.; Paillard, C.; Miao, Z.; Pichot, A.; Leduc, M.S.; Yang, Y.; Bergstrom, K.L.; Mahoney, D.H.; Shardy, D.L.; et al. Mutations in signal recognition particle SRP54 cause syndromic neutropenia with Shwachman-Diamond-like features. J. Clin. Investig. 2017, 127, 4090–4103. [Google Scholar] [CrossRef]
- Bezzerri, V.; Bardelli, D.; Morini, J.; Vella, A.; Cesaro, S.; Sorio, C.; Biondi, A.; Danesino, C.; Farruggia, P.; Assael, B.M.; et al. Ataluren-driven restoration of Shwachman-Bodian-Diamond syndrome protein function in Shwachman-Diamond syndrome bone marrow cells. Am. J. Hematol. 2018, 93, 527–536. [Google Scholar] [CrossRef]
- Dror, Y.; Donadieu, J.; Koglmeier, J.; Dodge, J.; Toiviainen-Salo, S.; Makitie, O.; Kerr, E.; Zeidler, C.; Shimamura, A.; Shah, N.; et al. Draft consensus guidelines for diagnosis and treatment of Shwachman-Diamond syndrome. Ann. N. Y. Acad. Sci. 2011, 1242, 40–55. [Google Scholar] [CrossRef]
- Joyce, C.E.; Saadatpour, A.; Ruiz-Gutierrez, M.; Bolukbasi, O.V.; Jiang, L.; Thomas, D.D.; Young, S.; Hofmann, I.; Sieff, C.A.; Myers, K.C.; et al. TGF-β signaling underlies hematopoietic dysfunction and bone marrow failure in Shwachman-Diamond syndrome. J. Clin. Investig. 2019, 129, 3821–3826. [Google Scholar] [CrossRef]
- Ebert, B.L.; Pretz, J.; Bosco, J.; Chang, C.Y.; Tamayo, P.; Galili, N.; Raza, A.; Root, D.E.; Attar, E.; Ellis, S.R.; et al. Identification of RPS14 as a 5q- syndrome gene by RNA interference screen. Nature 2008, 451, 335–339. [Google Scholar] [CrossRef]
- Fenaux, P.; Kelaidi, C. Treatment of the 5q– Syndrome. Hematology 2006, 2006, 192–198. [Google Scholar] [CrossRef]
- Komrokji, R.S.; Padron, E.; Ebert, B.L.; List, A.F. Deletion 5q MDS: Molecular and therapeutic implications. Best Pract. Res. Clin. Haematol. 2013, 26, 365–375. [Google Scholar] [CrossRef]
- Wei, S.; Chen, X.; McGraw, K.; Zhang, L.; Komrokji, R.; Clark, J.; Caceres, G.; Billingsley, D.; Sokol, L.; Lancet, J.; et al. Lenalidomide Promotes p53 Degradation by Inhibiting MDM2 Auto-ubiquitination in Myelodysplastic Syndrome with Chromosome 5q Deletion. Oncogene 2013, 32, 1110–1120. [Google Scholar] [CrossRef]
- Cortes, J.; Kantarjian, H.; Ball, E.D.; Dipersio, J.; Kolitz, J.E.; Fernandez, H.F.; Goodman, M.; Borthakur, G.; Baer, M.R.; Wetzler, M. Phase 2 randomized study of p53 antisense oligonucleotide (cenersen) plus idarubicin with or without cytarabine in refractory and relapsed acute myeloid leukemia. Cancer 2012, 118, 418–427. [Google Scholar] [CrossRef] [PubMed]
- Bishop, M.R.; Iversen, P.L.; Bayever, E.; Sharp, J.G.; Greiner, T.C.; Copple, B.L.; Ruddon, R.; Zon, G.; Spinolo, J.; Arneson, M.; et al. Phase I trial of an antisense oligonucleotide OL(1)p53 in hematologic malignancies. J. Clin. Oncol. 1996, 14, 1320–1326. [Google Scholar] [CrossRef] [PubMed]
- Lanasa, M.C.; Davis, P.H.; Datto, M.; Li, Z.; Gockerman, J.P.; Moore, J.O.; DeCastro, C.M.; Friedman, D.R.; Diehl, L.F.; Rehder, C.; et al. Phase II study of cenersen, an antisense inhibitor of p53, in combination with fludarabine, cyclophosphamide and rituximab for high-risk chronic lymphocytic leukemia. Leuk. Lymphoma 2012, 53, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Alachkar, H.; Xie, Z.; Marcucci, G.; Chan, K.K. Determination of cellular uptake and intracellular levels of Cenersen (Aezea(®), EL625), a p53 antisense oligonucleotide in acute myeloid leukemia cells. J. Pharm. Biomed. Anal. 2012, 71, 228–232. [Google Scholar] [CrossRef]
- Caceres, G.; McGraw, K.; Yip, B.H.; Pellagatti, A.; Johnson, J.; Zhang, L.; Liu, K.; Zhang, L.M.; Fulp, W.J.; Lee, J.H.; et al. TP53 suppression promotes erythropoiesis in del(5q) MDS, suggesting a targeted therapeutic strategy in lenalidomide-resistant patients. Proc. Natl. Acad. Sci. USA 2013, 110, 16127–16132. [Google Scholar] [CrossRef]
- Drouin, E.; Russo, P.; Tuchweber, B.; Mitchell, G.; Rasquin-Weber, A. North American Indian cirrhosis in children: A review of 30 cases. J. Pediatric Gastroenterol. Nutr. 2000, 31, 395–404. [Google Scholar] [CrossRef]
- Chagnon, P.; Michaud, J.; Mitchell, G.; Mercier, J.; Marion, J.F.; Drouin, E.; Rasquin-Weber, A.; Hudson, T.J.; Richter, A. A Missense Mutation (R565W) in Cirhin (FLJ14728) in North American Indian Childhood Cirrhosis. Am. J. Hum. Genet. 2002, 71, 1443–1449. [Google Scholar] [CrossRef]
- Barandun, J.; Hunziker, M.; Klinge, S. Assembly and structure of the SSU processome-a nucleolar precursor of the small ribosomal subunit. Curr. Opin. Struct. Biol. 2018, 49, 85–93. [Google Scholar] [CrossRef]
- Mahlaoui, N.; Minard-Colin, V.; Picard, C.; Bolze, A.; Ku, C.L.; Tournilhac, O.; Gilbert-Dussardier, B.; Pautard, B.; Durand, P.; Devictor, D.; et al. Isolated congenital asplenia: A French nationwide retrospective survey of 20 cases. J. Pediatric 2011, 158, 142–148. [Google Scholar] [CrossRef]
- Bolze, A.; Mahlaoui, N.; Byun, M.; Turner, B.; Trede, N.; Ellis, S.R.; Abhyankar, A.; Itan, Y.; Patin, E.; Brebner, S.; et al. Ribosomal Protein SA Haploinsufficiency in Humans with Isolated Congenital Asplenia. Science 2013, 340, 976–978. [Google Scholar] [CrossRef]
- Davies, J.M.; Lewis, M.P.N.; Wimperis, J.; Rafi, I.; Ladhani, S.; Bolton-Maggs, P.H.B. Review of guidelines for the prevention and treatment of infection in patients with an absent or dysfunctional spleen: Prepared on behalf of the British Committee for Standards in Haematology by a Working Party of the Haemato-Oncology Task Force. Br. J. Haematol. 2011, 155, 308–317. [Google Scholar] [CrossRef] [PubMed]
- Ernst, C. Proliferation and Differentiation Deficits are a Major Convergence Point for Neurodevelopmental Disorders. Trends Neurosci. 2016, 39, 290–299. [Google Scholar] [CrossRef] [PubMed]
- Thapar, A.; Cooper, M.; Rutter, M. Neurodevelopmental disorders. Lancet Psychiatry 2017, 4, 339–346. [Google Scholar] [CrossRef]
- Hetman, M.; Slomnicki, L.P. Ribosomal biogenesis as an emerging target of neurodevelopmental pathologies. J. Neurochem. 2019, 148, 325–347. [Google Scholar] [CrossRef]
- Klauck, S.M.; Felder, B.; Kolb-Kokocinski, A.; Schuster, C.; Chiocchetti, A.; Schupp, I.; Wellenreuther, R.; Schmötzer, G.; Poustka, F.; Breitenbach-Koller, L.; et al. Mutations in the ribosomal protein gene RPL10 suggest a novel modulating disease mechanism for autism. Mol. Psychiatry 2006, 11, 1073–1084. [Google Scholar] [CrossRef] [PubMed]
- Zanni, G.; Kalscheuer, V.M.; Friedrich, A.; Barresi, S.; Alfieri, P.; Di Capua, M.; Haas, S.A.; Piccini, G.; Karl, T.; Klauck, S.M.; et al. A Novel Mutation in RPL10 (Ribosomal Protein L10) Causes X-Linked Intellectual Disability, Cerebellar Hypoplasia, and Spondylo-Epiphyseal Dysplasia. Hum. Mutat. 2015, 36, 1155–1158. [Google Scholar] [CrossRef]
- Brooks, S.S.; Wall, A.L.; Golzio, C.; Reid, D.W.; Kondyles, A.; Willer, J.R.; Botti, C.; Nicchitta, C.V.; Katsanis, N.; Davis, E.E. A novel ribosomopathy caused by dysfunction of RPL10 disrupts neurodevelopment and causes X-linked microcephaly in humans. Genetics 2014, 198, 723–733. [Google Scholar] [CrossRef]
- Paolini, N.A.; Attwood, M.; Sondalle, S.B.; Vieira, C.M.D.S.; van Adrichem, A.M.; di Summa, F.M.; O’Donohue, M.F.; Gleizes, P.E.; Rachuri, S.; Briggs, J.W.; et al. A Ribosomopathy Reveals Decoding Defective Ribosomes Driving Human Dysmorphism. Am. J. Hum. Genet. 2017, 100, 506–522. [Google Scholar] [CrossRef]
- Butterfield, R.J.; Stevenson, T.J.; Xing, L.; Newcomb, T.M.; Nelson, B.; Zeng, W.; Li, X.; Lu, H.M.; Lu, H.; Farwell Gonzalez, K.D.; et al. Congenital lethal motor neuron disease with a novel defect in ribosome biogenesis. Neurology 2014, 82, 1322–1330. [Google Scholar] [CrossRef]
- Bernabò, P.; Tebaldi, T.; Groen, E.J.N.; Lane, F.M.; Perenthaler, E.; Mattedi, F.; Newbery, H.J.; Zhou, H.; Zuccotti, P.; Potrich, V.; et al. In Vivo Translatome Profiling in Spinal Muscular Atrophy Reveals a Role for SMN Protein in Ribosome Biology. Cell Rep. 2017, 21, 953–965. [Google Scholar] [CrossRef]
- Gillespie, A.; Gabunilas, J.; Jen, J.C.; Chanfreau, G.F. Mutations of EXOSC3/Rrp40p associated with neurological diseases impact ribosomal RNA processing functions of the exosome in S. cerevisiae. RNA 2017, 23, 466–472. [Google Scholar] [CrossRef] [PubMed]
- Edvardson, S.; Nicolae, C.M.; Agrawal, P.B.; Mignot, C.; Payne, K.; Prasad, A.N.; Prasad, C.; Sadler, L.; Nava, C.; Mullen, T.E.; et al. Heterozygous De Novo UBTF Gain-of-Function Variant Is Associated with Neurodegeneration in Childhood. Am. J. Hum. Genet. 2017, 101, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Toro, C.; Hori, R.T.; Malicdan, M.C.V.; Tifft, C.J.; Goldstein, A.; Gahl, W.A.; Adams, D.R.; Fauni, H.B.; Wolfe, L.A.; Xiao, J.; et al. A recurrent de novo missense mutation in UBTF causes developmental neuroregression. Hum. Mol. Genet. 2018, 27, 691–705. [Google Scholar] [CrossRef] [PubMed]
- Lowry, R.B.; Innes, A.M.; Bernier, F.P.; McLeod, D.R.; Greenberg, C.R.; Chudley, A.E.; Chodirker, B.; Marles, S.L.; Crumley, M.J.; Loredo-Osti, J.C.; et al. Bowen-Conradi syndrome: A clinical and genetic study. Am. J. Med. Genet. A 2003, 120A, 423–428. [Google Scholar] [CrossRef]
- Warda, A.S.; Freytag, B.; Haag, S.; Sloan, K.E.; Görlich, D.; Bohnsack, M.T. Effects of the Bowen-Conradi syndrome mutation in EMG1 on its nuclear import, stability and nucleolar recruitment. Hum. Mol. Genet. 2016, 25, 5353–5364. [Google Scholar] [CrossRef]
- Hamaguchi, I.; Ooka, A.; Brun, A.; Richter, J.; Dahl, N.; Karlsson, S. Gene transfer improves erythroid development in ribosomal protein S19-deficient Diamond-Blackfan anemia. Blood 2002, 100, 2724–2731. [Google Scholar] [CrossRef]
- Jaako, P.; Debnath, S.; Olsson, K.; Modlich, U.; Rothe, M.; Schambach, A.; Flygare, J.; Karlsson, S. Gene therapy cures the anemia and lethal bone marrow failure in a mouse model of RPS19-deficient Diamond-Blackfan anemia. Haematologica 2014, 99, 1792–1798. [Google Scholar] [CrossRef]
- Debnath, S.; Jaako, P.; Siva, K.; Rothe, M.; Chen, J.; Dahl, M.; Gaspar, H.B.; Flygare, J.; Schambach, A.; Karlsson, S. Lentiviral Vectors with Cellular Promoters Correct Anemia and Lethal Bone Marrow Failure in a Mouse Model for Diamond-Blackfan Anemia. Mol. Ther. 2017, 25, 1805–1814. [Google Scholar] [CrossRef]
- Aspesi, A.; Monteleone, V.; Betti, M.; Actis, C.; Morleo, G.; Sculco, M.; Guarrera, S.; Wlodarski, M.W.; Ramenghi, U.; Santoro, C.; et al. Author Correction: Lymphoblastoid cell lines from Diamond Blackfan anaemia patients exhibit a full ribosomal stress phenotype that is rescued by gene therapy. Sci. Rep. 2018, 8, 17227. [Google Scholar] [CrossRef]
- Bueren, J.A.; Quintana-Bustamante, O.; Almarza, E.; Navarro, S.; Río, P.; Segovia, J.C.; Guenechea, G. Advances in the gene therapy of monogenic blood cell diseases. Clin. Genet. 2020, 97, 89–102. [Google Scholar] [CrossRef]
- Amoasii, L.; Hildyard, J.C.W.; Li, H.; Sanchez-Ortiz, E.; Mireault, A.; Caballero, D.; Harron, R.; Stathopoulou, T.R.; Massey, C.; Shelton, J.M.; et al. Gene editing restores dystrophin expression in a canine model of Duchenne muscular dystrophy. Science 2018, 362, 86–91. [Google Scholar] [CrossRef]
- Niggemann, P.; György, B.; Chen, Z.Y. Genome and base editing for genetic hearing loss. Hear. Res. 2020, 394, 107958. [Google Scholar] [CrossRef] [PubMed]
- Aspesi, A.; Borsotti, C.; Follenzi, A. Emerging Therapeutic Approaches for Diamond Blackfan Anemia. Curr. Gene Ther. 2018, 18, 327–335. [Google Scholar] [CrossRef] [PubMed]
- Genuth, N.R.; Barna, M. Heterogeneity and specialized functions of translation machinery: From genes to organisms. Nat. Rev. Genet. 2018, 19, 431–452. [Google Scholar] [CrossRef] [PubMed]
- Tahmasebi, S.; Khoutorsky, A.; Mathews, M.B.; Sonenberg, N. Translation deregulation in human disease. Nat. Rev. Mol. Cell Biol. 2018, 19, 791–807. [Google Scholar] [CrossRef] [PubMed]

| Name | OMIM | Mutations | Phenotype | Current Treatment | Therapeutic Perspectives | |
|---|---|---|---|---|---|---|
| Diamond–Blackfan anemia | 105650 | •RPS7 | •RPL10A | •Anemia | •Chronic transfusions | •Danazol |
| •RPS10 | •RPL11 | •Growth retardation | •Steroids | •L-leucine | ||
| •RPS17 | •RPL15 | •Other defects ~30–50% | or | •Sotatercept | ||
| •RPS19 | •RPL18 | •Craniofacial | • Hematopoietic Stem Cell Transplantation (HSCT) | •Trifluoperazine | ||
| •RPS24 | •RPL19 | •Upper limb | •SMER28 | |||
| •RPS26 | •RPL26 | •Heart malformations | •Eltrombopag | |||
| •RPLP0 | •RPL34 | •Urinary system malformations | ||||
| •RPL3 | •RPL35 | |||||
| •RPL5 | •RPL35A | |||||
| •RPL9 | •TSR2 | |||||
| •RPL10 | ||||||
| X-linked dyskeratosis congenita | 305000 | •DKC1 | •Skin pigmentation | •Chronic transfusions | •Eltrombopag | |
| •Nail dystrophy | •HSCT | •Danazol | ||||
| •Leucoplakia | •Regular examinations in | |||||
| •Cytopenia | •Immunology | |||||
| •Other defect >30% | •Dermatology | |||||
| •Epiphora | •Neurology | |||||
| •Learning difficulties/mental retardation | •Ophthalmology | |||||
| •Pulmonary disease | •Otolaryngology | |||||
| •Hyperhidrosis | •Dental | |||||
| •Extensive dental carries/loss | •Cardiology | |||||
| •Short stature | •Pulmonary | |||||
| •Hair loss/grey hair or sparse eyelashes | ||||||
| •Esophageal stricture | ||||||
| •Hypogonadism/undescended testes | ||||||
| •Urethral stricture/phimosis | ||||||
| •Malignancy | ||||||
| •Liver cirrhosis/adenoma | ||||||
| •Abnormal bone trabeculation/osteoporosis | ||||||
| Treacher Collins syndrome | 154500 | •Defects of craniofacial development | •Reconstructive surgery | •MG132 or Bortezomib | ||
| •TCOF1 | •Conductive hearing loss | •Speech therapy | ||||
| •POLR1C | •Palpebral fissures’ lateral downward sloping | |||||
| •POLR1D | •Mandible hypoplasia | |||||
| •Cleft palate | ||||||
| Cartilage hair hypoplasia | 250250 | •RMRP | •Short stature | •Granulocyte Colony-Stimulating Factor (GCSF) | •Recombinant growth hormone | |
| •Hypoplastic hair | •HSCT | •XAV939 | ||||
| •Ligamentous laxity | ||||||
| •Defective immunity | ||||||
| •Hypoplastic anemia | ||||||
| •Neuronal dysplasia of the intestine | ||||||
| Shwachman–Diamond syndrome | 260400 | •Neutropenia | •Chronic transfusions | •Transforming Growth Factor beta (TGF-β) | ||
| •SBDS | •Anemia | •Androgens | •Ataluren | |||
| •DNAJC21 | •Thrombocytopenia | •HSCT | ||||
| •EFL1 | •Bone marrow hypoplasia | •Reconstructive surgery | ||||
| •SRP54 | •Skeletal abnormalities | •Pancreatic enzymes | ||||
| •Pancreatic defects | •Vitamin supplements | |||||
| •Dietary advice and surveillance | ||||||
| 5q-syndrome | 153550 | •RPS14 | •Macrocytic anemia | •Red blood cell transfusions | •Cenersen | |
| •Erythroid hypoplasia | •Recombinant erythropoietin | •L-leucine | ||||
| •Hypolobated micromegakaryocytes | •Thalidomide | |||||
| •Retinoids | ||||||
| •Chemotherapy | ||||||
| •Hypomethylating agents | ||||||
| •Bone marrow transplantation | ||||||
| North American Indian | 604901 | •UTP4 | •Natal transient jaundice | •Liver transplantation | ||
| childhood cirrhosis | •Biliary cirrhosis | |||||
| •Portal hypertension | ||||||
| Isolated congenital asplenia | 271400 | •RPSA | •Absence of spleen | •Anti-infection and antibiotic prophylaxis | ||
| •Vaccination | ||||||
| •Pneumococcal | ||||||
| •Haemophilus influenzae type b | ||||||
| •Meningococcal | ||||||
| •Influenza | ||||||
| Neurodevelopmental disorders | •RPL10 | •POLR1A | •Neural tube defects | •Mental health medication | ||
| •RPS3 | •ERCC6 | •Microcephaly | •Speech therapy | |||
| •LAS1L | •CSB | •Autism | •Pediatrics | |||
| •SMN1 | •ERCC8 | •Schizophrenia | •Educational help | |||
| •EXOSC3 | •CSA | •Epilepsy | ||||
| •UBTF | •Cerebral palsy/periventricular leukomalacia | |||||
| Bowen–Conradi syndrome | 211180 | •EMG1 | •Psychomotor defects | |||
| •Growth retardation | ||||||
| •Microcephaly | ||||||
| •Micrognathia | ||||||
| •Congenital vertical talus | ||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Orgebin, E.; Lamoureux, F.; Isidor, B.; Charrier, C.; Ory, B.; Lézot, F.; Baud’huin, M. Ribosomopathies: New Therapeutic Perspectives. Cells 2020, 9, 2080. https://doi.org/10.3390/cells9092080
Orgebin E, Lamoureux F, Isidor B, Charrier C, Ory B, Lézot F, Baud’huin M. Ribosomopathies: New Therapeutic Perspectives. Cells. 2020; 9(9):2080. https://doi.org/10.3390/cells9092080
Chicago/Turabian StyleOrgebin, Emilien, François Lamoureux, Bertrand Isidor, Céline Charrier, Benjamin Ory, Frédéric Lézot, and Marc Baud’huin. 2020. "Ribosomopathies: New Therapeutic Perspectives" Cells 9, no. 9: 2080. https://doi.org/10.3390/cells9092080
APA StyleOrgebin, E., Lamoureux, F., Isidor, B., Charrier, C., Ory, B., Lézot, F., & Baud’huin, M. (2020). Ribosomopathies: New Therapeutic Perspectives. Cells, 9(9), 2080. https://doi.org/10.3390/cells9092080







