The Diversity of Intermediate Filaments in Astrocytes
Abstract
1. Introduction
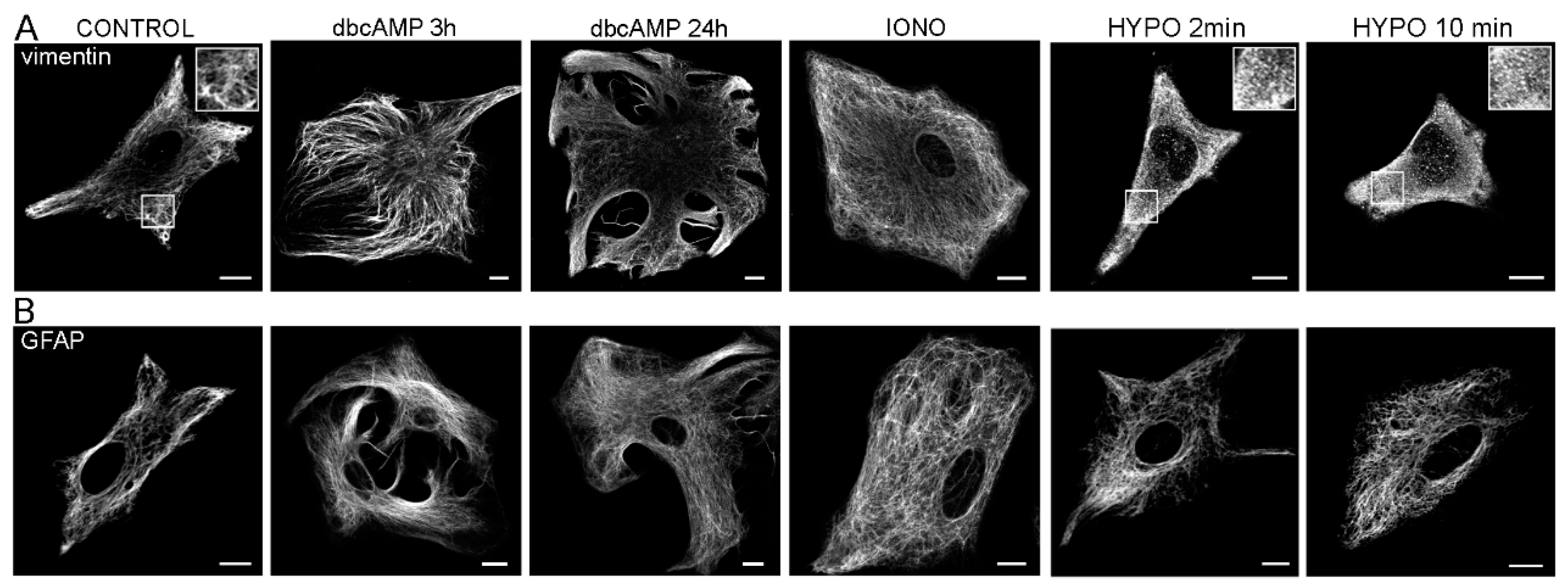
2. Glial Fibrillary Acidic Protein and Vimentin
2.1. Structure and Expression of Glial Fibrillary Acidic Protein in Astrocytes
2.2. Structure and Expression of Vimentin in Astrocytes
2.3. Reactive Gliosis
3. Nestin
3.1. Structure of Nestin
3.2. Expression and Function of Nestin in Astrocytes
4. Synemin
4.1. Structure and Expression of Synemin
4.2. Subcellular Localization of Synemin
4.3. Synemin in Reactive Astrocytes
5. Lamins
Lamins and Diseases
6. Cytolinkers and Related Proteins
6.1. Structure and Expression in Astrocytes
6.2. Plectin
7. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Zorec, R.; Araque, A.; Carmignoto, G.; Haydon, P.G.; Verkhratsky, A.; Parpura, V. Astroglial excitability and gliotransmission: An appraisal of Ca2+ as a signalling route. ASN Neuro 2012, 4. [Google Scholar] [CrossRef]
- Glickstein, M. Golgi and Cajal: The neuron doctrine and the 100th anniversary of the 1906 Nobel Prize. Curr. Biol. 2006, 16, R147–R151. [Google Scholar] [CrossRef]
- Eng, L.F.; Vanderhaeghen, J.J.; Bignami, A.; Gerstl, B. An acidic protein isolated from fibrous astrocytes. Brain Res. 1971, 28, 351–354. [Google Scholar] [CrossRef]
- Barres, B.A.; Koroshetz, W.J.; Chun, L.L.; Corey, D.P. Ion channel expression by white matter glia: The type-1 astrocyte. Neuron 1990, 5, 527–544. [Google Scholar] [CrossRef]
- Cornell-Bell, A.H.; Finkbeiner, S.M.; Cooper, M.S.; Smith, S.J. Glutamate induces calcium waves in cultured astrocytes: Long-range glial signaling. Science 1990, 247, 470–473. [Google Scholar] [CrossRef]
- Charles, A.C.; Merrill, J.E.; Dirksen, E.R.; Sanderson, M.J. Intercellular signaling in glial cells: Calcium waves and oscillations in response to mechanical stimulation and glutamate. Neuron 1991, 6, 983–992. [Google Scholar] [CrossRef]
- Dani, J.W.; Chernjavsky, A.; Smith, S.J. Neuronal activity triggers calcium waves in hippocampal astrocyte networks. Neuron 1992, 8, 429–440. [Google Scholar] [CrossRef]
- Nedergaard, M. Direct signaling from astrocytes to neurons in cultures of mammalian brain cells. Science 1994, 263, 1768–1771. [Google Scholar] [CrossRef]
- Parpura, V.; Basarsky, T.A.; Liu, F.; Jeftinija, K.; Jeftinija, S.; Haydon, P.G. Glutamate-mediated astrocyte-neuron signalling. Nature 1994, 369, 744–747. [Google Scholar] [CrossRef]
- Araque, A.; Parpura, V.; Sanzgiri, R.P.; Haydon, P.G. Tripartite synapses: Glia, the unacknowledged partner. Trends Neurosci. 1999, 22, 208–215. [Google Scholar] [CrossRef]
- Potokar, M.; Vardjan, N.; Stenovec, M.; Gabrijel, M.; Trkov, S.; Jorgačevski, J.; Kreft, M.; Zorec, R. Astrocytic vesicle mobility in health and disease. Int. J. Mol. Sci. 2013, 14, 11238–11258. [Google Scholar] [CrossRef] [PubMed]
- Potokar, M.; Stenovec, M.; Gabrijel, M.; Li, L.; Kreft, M.; Grilc, S.; Pekny, M.; Zorec, R. Intermediate filaments attenuate stimulation-dependent mobility of endosomes/lysosomes in astrocytes. Glia 2010, 58, 1208–1219. [Google Scholar] [CrossRef] [PubMed]
- Potokar, M.; Kreft, M.; Li, L.; Daniel Andersson, J.; Pangrsic, T.; Chowdhury, H.H.; Pekny, M.; Zorec, R. Cytoskeleton and vesicle mobility in astrocytes. Traffic 2007, 8, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Margiotta, A.; Bucci, C. Role of intermediate filaments in vesicular traffic. Cells 2016, 5, 20. [Google Scholar] [CrossRef]
- Kimelberg, H.K. The problem of astrocyte identity. Neurochem. Int. 2004, 45, 191–202. [Google Scholar] [CrossRef]
- Eriksson, J.E.; Dechat, T.; Grin, B.; Helfand, B.; Mendez, M.; Pallari, H.M.; Goldman, R.D. Introducing intermediate filaments: From discovery to disease. J. Clin. Investig. 2009, 119, 1763–1771. [Google Scholar] [CrossRef]
- Hesse, M.; Magin, T.M.; Weber, K. Genes for intermediate filament proteins and the draft sequence of the human genome: Novel keratin genes and a surprisingly high number of pseudogenes related to keratin genes 8 and 18. J. Cell Sci. 2001, 114, 2569–2575. [Google Scholar]
- Hol, E.M.; Pekny, M. Glial fibrillary acidic protein (GFAP) and the astrocyte intermediate filament system in diseases of the central nervous system. Curr. Opin. Cell Biol. 2015, 32, 121–130. [Google Scholar] [CrossRef]
- Sofroniew, M.V.; Vinters, H.V. Astrocytes: Biology and pathology. Acta Neuropathol. 2010, 119, 7–35. [Google Scholar] [CrossRef]
- Fraser, R.D.; Macrae, T.P. The molecular configuration of alpha-keratin. J. Mol. Biol. 1961, 3, 640–647. [Google Scholar] [CrossRef]
- Goldman, R.D.; Follett, E.A. Birefringent filamentous organelle in BHK-21 cells and its possible role in cell spreading and motility. Science 1970, 169, 286–288. [Google Scholar] [CrossRef]
- Goldman, R.D. The role of three cytoplasmic fibers in BHK-21 cell motility. I. Microtubules and the effects of colchicine. J. Cell Biol. 1971, 51, 752–762. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, H.; Bischoff, R.; Holtzer, H. Mitosis and intermediate-sized filaments in developing skeletal muscle. J. Cell Biol. 1968, 38, 538–555. [Google Scholar] [CrossRef]
- Starger, J.M.; Goldman, R.D. Isolation and preliminary characterization of 10-nm filaments from baby hamster kidney (BHK-21) cells. Proc. Natl. Acad. Sci. USA 1977, 74, 2422–2426. [Google Scholar] [CrossRef] [PubMed]
- Starger, J.M.; Brown, W.E.; Goldman, A.E.; Goldman, R.D. Biochemical and immunological analysis of rapidly purified 10-nm filaments from baby hamster kidney (BHK-21) cells. J. Cell Biol. 1978, 78, 93–109. [Google Scholar] [CrossRef] [PubMed]
- Pruss, R.M.; Mirsky, R.; Raff, M.C.; Thorpe, R.; Dowding, A.J.; Anderton, B.H. All classes of intermediate filaments share a common antigenic determinant defined by a monoclonal antibody. Cell 1981, 27, 419–428. [Google Scholar] [CrossRef]
- Uyeda, C.T.; Eng, L.F.; Bignami, A. Immunological study of the glial fibrillary acidic protein. Brain Res. 1972, 37, 81–89. [Google Scholar] [CrossRef]
- Weber, K.; Geisler, N. Intermediate filaments: Structural conservation and divergence. Ann. N. Y. Acad. Sci. 1985, 455, 126–143. [Google Scholar] [CrossRef]
- Lazarides, E. Intermediate filaments as mechanical integrators of cellular space. Nature 1980, 283, 249–256. [Google Scholar] [CrossRef]
- Dahlstrand, J.; Zimmerman, L.B.; McKay, R.D.; Lendahl, U. Characterization of the human nestin gene reveals a close evolutionary relationship to neurofilaments. J. Cell Sci. 1992, 103 Pt 2, 589–597. [Google Scholar]
- Rutka, J.T.; Giblin, J.R.; Balkissoon, R.; Wen, D.; Myatt, C.A.; McCulloch, J.R.; Rosenblum, M.L. Characterization of fetal human brain cultures. Development of a potential model for selectively purifying human glial cells in culture. Dev. Neurosci. 1987, 9, 154–173. [Google Scholar] [CrossRef]
- Pekny, M.; Pekna, M. Astrocyte intermediate filaments in CNS pathologies and regeneration. J. Pathol. 2004, 204, 428–437. [Google Scholar] [CrossRef]
- Budka, H. Non-glial specificities of immunocytochemistry for the glial fibrillary acidic protein (GFAP). Triple expression of GFAP, vimentin and cytokeratins in papillary meningioma and metastasizing renal carcinoma. Acta Neuropathol. 1986, 72, 43–54. [Google Scholar] [CrossRef]
- Roelofs, R.F.; Fischer, D.F.; Houtman, S.H.; Sluijs, J.A.; Van Haren, W.; Van Leeuwen, F.W.; Hol, E.M. Adult human subventricular, subgranular, and subpial zones contain astrocytes with a specialized intermediate filament cytoskeleton. Glia 2005, 52, 289–300. [Google Scholar] [CrossRef]
- van den Berge, S.A.; Middeldorp, J.; Zhang, C.E.; Curtis, M.A.; Leonard, B.W.; Mastroeni, D.; Voorn, P.; van de Berg, W.D.; Huitinga, I.; Hol, E.M. Longterm quiescent cells in the aged human subventricular neurogenic system specifically express GFAP-delta. Aging Cell 2010, 9, 313–326. [Google Scholar] [CrossRef]
- Cahoy, J.D.; Emery, B.; Kaushal, A.; Foo, L.C.; Zamanian, J.L.; Christopherson, K.S.; Xing, Y.; Lubischer, J.L.; Krieg, P.A.; Krupenko, S.A.; et al. A transcriptome database for astrocytes, neurons, and oligodendrocytes: A new resource for understanding brain development and function. J. Neurosci. 2008, 28, 264–278. [Google Scholar] [CrossRef]
- Middeldorp, J.; Hol, E.M. GFAP in health and disease. Prog. Neurobiol. 2011, 93, 421–443. [Google Scholar] [CrossRef]
- Zoltewicz, J.S.; Scharf, D.; Yang, B.; Chawla, A.; Newsom, K.J.; Fang, L. Characterization of antibodies that detect human GFAP after traumatic brain injury. Biomark. Insights 2012, 7, 71–79. [Google Scholar] [CrossRef]
- Reeves, S.A.; Helman, L.J.; Allison, A.; Israel, M.A. Molecular cloning and primary structure of human glial fibrillary acidic protein. Proc. Natl. Acad. Sci. USA 1989, 86, 5178–5182. [Google Scholar] [CrossRef]
- Chen, W.J.; Liem, R.K. The endless story of the glial fibrillary acidic protein. J. Cell Sci. 1994, 107 Pt 8, 2299–2311. [Google Scholar]
- Eliasson, C.; Sahlgren, C.; Berthold, C.H.; Stakeberg, J.; Celis, J.E.; Betsholtz, C.; Eriksson, J.E.; Pekny, M. Intermediate filament protein partnership in astrocytes. J. Biol. Chem. 1999, 274, 23996–24006. [Google Scholar] [CrossRef] [PubMed]
- Potokar, M.; Stenovec, M.; Kreft, M.; Kreft, M.; Zorec, R. Stimulation inhibits the mobility of recycling peptidergic vesicles in astrocytes. Glia 2008, 56, 135–144. [Google Scholar] [CrossRef] [PubMed]
- Vardjan, N.; Gabrijel, M.; Potokar, M.; Svajger, U.; Kreft, M.; Jeras, M.; de Pablo, Y.; Faiz, M.; Pekny, M.; Zorec, R. IFN-γ-induced increase in the mobility of MHC class II compartments in astrocytes depends on intermediate filaments. J. Neuroinflamm. 2012, 9, 144. [Google Scholar] [CrossRef] [PubMed]
- De Pascalis, C.; Pérez-González, C.; Seetharaman, S.; Boëda, B.; Vianay, B.; Burute, M.; Leduc, C.; Borghi, N.; Trepat, X.; Etienne-Manneville, S. Intermediate filaments control collective migration by restricting traction forces and sustaining cell-cell contacts. J. Cell Biol. 2018, 217, 3031–3044. [Google Scholar] [CrossRef]
- Ding, M.; Eliasson, C.; Betsholtz, C.; Hamberger, A.; Pekny, M. Altered taurine release following hypotonic stress in astrocytes from mice deficient for GFAP and vimentin. Brain Res. Mol. Brain Res. 1998, 62, 77–81. [Google Scholar] [CrossRef]
- Franke, W.W.; Schmid, E.; Osborn, M.; Weber, K. Different intermediate-sized filaments distinguished by immunofluorescence microscopy. Proc. Natl. Acad. Sci. USA 1978, 75, 5034–5038. [Google Scholar] [CrossRef]
- Wilhelmsson, U.; Li, L.; Pekna, M.; Berthold, C.; Blom, S.; Eliasson, C.; Renner, O.; Bushong, E.; Ellisman, M.; Morgan, T.; et al. Absence of glial fibrillary acidic protein and vimentin prevents hypertrophy of astrocytic processes and improves post-traumatic regeneration. J. Neurosci. 2004, 24, 5016–5021. [Google Scholar] [CrossRef]
- Pekny, M.; Johansson, C.B.; Eliasson, C.; Stakeberg, J.; Wallén, A.; Perlmann, T.; Lendahl, U.; Betsholtz, C.; Berthold, C.H.; Frisén, J. Abnormal reaction to central nervous system injury in mice lacking glial fibrillary acidic protein and vimentin. J. Cell Biol. 1999, 145, 503–514. [Google Scholar] [CrossRef]
- de Pablo, Y.; Marasek, P.; Pozo-Rodrigálvarez, A.; Wilhelmsson, U.; Inagaki, M.; Pekna, M.; Pekny, M. Vimentin phosphorylation is required for normal cell division of immature astrocytes. Cells 2019, 8, 1016. [Google Scholar] [CrossRef]
- Hockfield, S.; McKay, R.D. Identification of major cell classes in the developing mammalian nervous system. J. Neurosci. 1985, 5, 3310–3328. [Google Scholar] [CrossRef]
- Wilhelmsson, U.; Lebkuechner, I.; Leke, R.; Marasek, P.; Yang, X.; Antfolk, D.; Chen, M.; Mohseni, P.; Lasič, E.; Bobnar, S.T.; et al. Nestin regulates neurogenesis in mice through notch signaling from astrocytes to neural stem cells. Cereb. Cortex 2019, 29, 4050–4066. [Google Scholar] [CrossRef]
- Granger, B.L.; Lazarides, E. Synemin: A new high molecular weight protein associated with desmin and vimentin filaments in muscle. Cell 1980, 22, 727–738. [Google Scholar] [CrossRef]
- Mizuno, Y.; Thompson, T.G.; Guyon, J.R.; Lidov, H.G.; Brosius, M.; Imamura, M.; Ozawa, E.; Watkins, S.C.; Kunkel, L.M. Desmuslin, an intermediate filament protein that interacts with alpha -dystrobrevin and desmin. Proc. Natl. Acad. Sci. USA 2001, 98, 6156–6161. [Google Scholar] [CrossRef] [PubMed]
- Titeux, M.; Brocheriou, V.; Xue, Z.; Gao, J.; Pellissier, J.F.; Guicheney, P.; Paulin, D.; Li, Z. Human synemin gene generates splice variants encoding two distinct intermediate filament proteins. Eur. J. Biochem. 2001, 268, 6435–6449. [Google Scholar] [CrossRef]
- Jing, R.; Pizzolato, G.; Robson, R.M.; Gabbiani, G.; Skalli, O. Intermediate filament protein synemin is present in human reactive and malignant astrocytes and associates with ruffled membranes in astrocytoma cells. Glia 2005, 50, 107–120. [Google Scholar] [CrossRef]
- Kaufmann, S.H.; Gibson, W.; Shaper, J.H. Characterization of the major polypeptides of the rat liver nuclear envelope. J. Biol. Chem. 1983, 258, 2710–2719. [Google Scholar] [PubMed]
- Shumaker, D.K.; Solimando, L.; Sengupta, K.; Shimi, T.; Adam, S.A.; Grunwald, A.; Strelkov, S.V.; Aebi, U.; Cardoso, M.C.; Goldman, R.D. The highly conserved nuclear lamin Ig-fold binds to PCNA: Its role in DNA replication. J. Cell Biol. 2008, 181, 269–280. [Google Scholar] [CrossRef] [PubMed]
- Koliou, X.; Fedonidis, C.; Kalpachidou, T.; Mangoura, D. Nuclear import mechanism of neurofibromin for localization on the spindle and function in chromosome congression. J. Neurochem. 2016, 136, 78–91. [Google Scholar] [CrossRef]
- Marín, M.P.; Tomas, M.; Esteban-Pretel, G.; Megías, L.; López-Iglesias, C.; Egea, G.; Renau-Piqueras, J. Chronic ethanol exposure induces alterations in the nucleocytoplasmic transport in growing astrocytes. J. Neurochem. 2008, 106, 1914–1928. [Google Scholar] [CrossRef] [PubMed]
- van Bodegraven, E.J.; van Asperen, J.V.; Robe, P.A.J.; Hol, E.M. Importance of GFAP isoform-specific analyses in astrocytoma. Glia 2019, 67, 1417–1433. [Google Scholar] [CrossRef] [PubMed]
- Perng, M.D.; Wen, S.F.; Gibbon, T.; Middeldorp, J.; Sluijs, J.; Hol, E.M.; Quinlan, R.A. Glial fibrillary acidic protein filaments can tolerate the incorporation of assembly-compromised GFAP-delta, but with consequences for filament organization and alphaB-crystallin association. Mol. Biol. Cell 2008, 19, 4521–4533. [Google Scholar] [CrossRef]
- Moeton, M.; Kanski, R.; Stassen, O.M.; Sluijs, J.A.; Geerts, D.; van Tijn, P.; Wiche, G.; van Strien, M.E.; Hol, E.M. Silencing GFAP isoforms in astrocytoma cells disturbs laminin-dependent motility and cell adhesion. FASEB J. 2014, 28, 2942–2954. [Google Scholar] [CrossRef] [PubMed]
- Sosunov, A.A.; McKhann, G.M., 2nd; Goldman, J.E. The origin of Rosenthal fibers and their contributions to astrocyte pathology in Alexander disease. Acta Neuropathol. Commun. 2017, 5, 27. [Google Scholar] [CrossRef]
- Tian, R.; Gregor, M.; Wiche, G.; Goldman, J.E. Plectin regulates the organization of glial fibrillary acidic protein in Alexander disease. Am. J. Pathol. 2006, 168, 888–897. [Google Scholar] [CrossRef] [PubMed]
- Pekny, T.; Faiz, M.; Wilhelmsson, U.; Curtis, M.A.; Matej, R.; Skalli, O.; Pekny, M. Synemin is expressed in reactive astrocytes and Rosenthal fibers in Alexander disease. APMIS 2014, 122, 76–80. [Google Scholar] [CrossRef] [PubMed]
- Heaven, M.R.; Flint, D.; Randall, S.M.; Sosunov, A.A.; Wilson, L.; Barnes, S.; Goldman, J.E.; Muddiman, D.C.; Brenner, M. Composition of Rosenthal fibers, the protein aggregate hallmark of Alexander disease. J. Proteome Res. 2016, 15, 2265–2282. [Google Scholar] [CrossRef] [PubMed]
- Quinlan, R.A.; Franke, W.W. Molecular interactions in intermediate-sized filaments revealed by chemical cross-linking. Heteropolymers of vimentin and glial filament protein in cultured human glioma cells. Eur. J. Biochem. 1983, 132, 477–484. [Google Scholar] [CrossRef]
- Jing, R.; Wilhelmsson, U.; Goodwill, W.; Li, L.; Pan, Y.; Pekny, M.; Skalli, O. Synemin is expressed in reactive astrocytes in neurotrauma and interacts differentially with vimentin and GFAP intermediate filament networks. J. Cell Sci. 2007, 120, 1267–1277. [Google Scholar] [CrossRef] [PubMed]
- Sultana, S.; Sernett, S.W.; Bellin, R.M.; Robson, R.M.; Skalli, O. Intermediate filament protein synemin is transiently expressed in a subset of astrocytes during development. Glia 2000, 30, 143–153. [Google Scholar] [CrossRef]
- Viedma-Poyatos, Á.; de Pablo, Y.; Pekny, M.; Pérez-Sala, D. The cysteine residue of glial fibrillary acidic protein is a critical target for lipoxidation and required for efficient network organization. Free Radi. Biol. Med. 2018, 120, 380–394. [Google Scholar] [CrossRef]
- Ciesielski-Treska, J.; Goetschy, J.F.; Ulrich, G.; Aunis, D. Acquisition of vimentin in astrocytes cultured from postnatal rat brain. J. Neurocytol. 1988, 17, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Sharp, G.; Osborn, M.; Weber, K. Occurrence of two different intermediate filament proteins in the same filament in situ within a human glioma cell line. An immunoelectron microscopical study. Exp. Cell Res. 1982, 141, 385–395. [Google Scholar] [CrossRef]
- Shaw, G.; Osborn, M.; Weber, K. An immunofluorescence microscopical study of the neurofilament triplet proteins, vimentin and glial fibrillary acidic protein within the adult rat brain. Eur. J. Cell Biol. 1981, 26, 68–82. [Google Scholar]
- Wang, E.; Cairncross, J.G.; Liem, R.K. Identification of glial filament protein and vimentin in the same intermediate filament system in human glioma cells. Proc. Natl. Acad. Sci. USA 1984, 81, 2102–2106. [Google Scholar] [CrossRef] [PubMed]
- Schnitzer, J.; Franke, W.W.; Schachner, M. Immunocytochemical demonstration of vimentin in astrocytes and ependymal cells of developing and adult mouse nervous system. J. Cell Biol. 1981, 90, 435–447. [Google Scholar] [CrossRef] [PubMed]
- Bovolenta, P.; Liem, R.K.; Mason, C.A. Development of cerebellar astroglia: Transitions in form and cytoskeletal content. Dev. Biol. 1984, 102, 248–259. [Google Scholar] [CrossRef]
- Ivaska, J.; Pallari, H.M.; Nevo, J.; Eriksson, J.E. Novel functions of vimentin in cell adhesion, migration, and signaling. Exp. Cell Res. 2007, 313, 2050–2062. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, J.E.; Opal, P.; Goldman, R.D. Intermediate filament dynamics. Curr. Opin. Cell Biol. 1992, 4, 99–104. [Google Scholar] [CrossRef]
- Eriksson, J.; Brautigan, D.; Vallee, R.; Olmsted, J.; Fujiki, H.; Goldman, R. Cytoskeletal integrity in interphase cells requires protein phosphatase activity. Proc. Natl. Acad. Sci. USA 1992, 89, 11093–11097. [Google Scholar] [CrossRef]
- Eriksson, J.E.; He, T.; Trejo-Skalli, A.V.; Härmälä-Braskén, A.S.; Hellman, J.; Chou, Y.H.; Goldman, R.D. Specific in vivo phosphorylation sites determine the assembly dynamics of vimentin intermediate filaments. J. Cell Sci. 2004, 117, 919–932. [Google Scholar] [CrossRef]
- Wang, K.; Bekar, L.K.; Furber, K.; Walz, W. Vimentin-expressing proximal reactive astrocytes correlate with migration rather than proliferation following focal brain injury. Brain Res. 2004, 1024, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Tzivion, G.; Luo, Z.J.; Avruch, J. Calyculin A-induced vimentin phosphorylation sequesters 14-3-3 and displaces other 14-3-3 partners in vivo. J. Biol. Chem. 2000, 275, 29772–29778. [Google Scholar] [CrossRef] [PubMed]
- Muslin, A.J.; Lau, J.M. Differential functions of 14-3-3 isoforms in vertebrate development. Curr. Top. Dev. Biol. 2005, 65, 211–228. [Google Scholar] [CrossRef]
- Potokar, M.; Stenovec, M.; Jorgačevski, J.; Holen, T.; Kreft, M.; Ottersen, O.P.; Zorec, R. Regulation of AQP4 surface expression via vesicle mobility in astrocytes. Glia 2013, 61, 917–928. [Google Scholar] [CrossRef]
- Kamat, P.K.; Swarnkar, S.; Rai, S.; Kumar, V.; Tyagi, N. Astrocyte mediated MMP-9 activation in the synapse dysfunction: An implication in Alzheimer disease. Ther. Targets Neurol. Dis. 2015, 1. [Google Scholar] [CrossRef]
- Seifert, G.; Schilling, K.; Steinhäuser, C. Astrocyte dysfunction in neurological disorders: A molecular perspective. Nat. Rev. Neurosci. 2006, 7, 194–206. [Google Scholar] [CrossRef]
- Bush, T.G.; Puvanachandra, N.; Horner, C.H.; Polito, A.; Ostenfeld, T.; Svendsen, C.N.; Mucke, L.; Johnson, M.H.; Sofroniew, M.V. Leukocyte infiltration, neuronal degeneration, and neurite outgrowth after ablation of scar-forming, reactive astrocytes in adult transgenic mice. Neuron 1999, 23, 297–308. [Google Scholar] [CrossRef]
- Okada, S.; Nakamura, M.; Katoh, H.; Miyao, T.; Shimazaki, T.; Ishii, K.; Yamane, J.; Yoshimura, A.; Iwamoto, Y.; Toyama, Y.; et al. Conditional ablation of Stat3 or Socs3 discloses a dual role for reactive astrocytes after spinal cord injury. Nat. Med. 2006, 12, 829–834. [Google Scholar] [CrossRef]
- Chung, W.S.; Welsh, C.A.; Barres, B.A.; Stevens, B. Do glia drive synaptic and cognitive impairment in disease? Nat. Neurosci. 2015, 18, 1539–1545. [Google Scholar] [CrossRef]
- Clarke, L.E.; Barres, B.A. Emerging roles of astrocytes in neural circuit development. Nat. Rev. Neurosci. 2013, 14, 311–321. [Google Scholar] [CrossRef]
- Faiz, M.; Sachewsky, N.; Gascón, S.; Bang, K.W.A.; Morshead, C.M.; Nagy, A. Adult neural stem cells from the subventricular zone give rise to reactive astrocytes in the cortex after stroke. Cell Stem Cell 2015, 17, 624–634. [Google Scholar] [CrossRef]
- Honsa, P.; Valny, M.; Kriska, J.; Matuskova, H.; Harantova, L.; Kirdajova, D.; Valihrach, L.; Androvic, P.; Kubista, M.; Anderova, M. Generation of reactive astrocytes from NG2 cells is regulated by sonic hedgehog. Glia 2016, 64, 1518–1531. [Google Scholar] [CrossRef] [PubMed]
- Hackett, A.R.; Yahn, S.L.; Lyapichev, K.; Dajnoki, A.; Lee, D.H.; Rodriguez, M.; Cammer, N.; Pak, J.; Mehta, S.T.; Bodamer, O.; et al. Injury type-dependent differentiation of NG2 glia into heterogeneous astrocytes. Exp. Neurol. 2018, 308, 72–79. [Google Scholar] [CrossRef]
- Zhu, X.; Hill, R.A.; Dietrich, D.; Komitova, M.; Suzuki, R.; Nishiyama, A. Age-dependent fate and lineage restriction of single NG2 cells. Development 2011, 138, 745–753. [Google Scholar] [CrossRef] [PubMed]
- Morita, M.; Saruta, C.; Kozuka, N.; Okubo, Y.; Itakura, M.; Takahashi, M.; Kudo, Y. Dual regulation of astrocyte gap junction hemichannels by growth factors and a pro-inflammatory cytokine via the mitogen-activated protein kinase cascade. Glia 2007, 55, 508–515. [Google Scholar] [CrossRef] [PubMed]
- Morita, M.; Higuchi, C.; Moto, T.; Kozuka, N.; Susuki, J.; Itofusa, R.; Yamashita, J.; Kudo, Y. Dual regulation of calcium oscillation in astrocytes by growth factors and pro-inflammatory cytokines via the mitogen-activated protein kinase cascade. J. Neurosci. 2003, 23, 10944–10952. [Google Scholar] [CrossRef] [PubMed]
- Zamanian, J.L.; Xu, L.; Foo, L.C.; Nouri, N.; Zhou, L.; Giffard, R.G.; Barres, B.A. Genomic analysis of reactive astrogliosis. J. Neurosci. 2012, 32, 6391–6410. [Google Scholar] [CrossRef]
- Liddelow, S.A.; Guttenplan, K.A.; Clarke, L.E.; Bennett, F.C.; Bohlen, C.J.; Schirmer, L.; Bennett, M.L.; Münch, A.E.; Chung, W.S.; Peterson, T.C.; et al. Neurotoxic reactive astrocytes are induced by activated microglia. Nature 2017, 541, 481–487. [Google Scholar] [CrossRef]
- Fuchs, P.; Spazierer, D.; Wiche, G. Plectin rodless isoform expression and its detection in mouse brain. Cell Mol. Neurobiol. 2005, 25, 1141–1150. [Google Scholar] [CrossRef]
- Pekny, M.; Wilhelmsson, U.; Pekna, M. The dual role of astrocyte activation and reactive gliosis. Neurosci. Lett. 2014, 565, 30–38. [Google Scholar] [CrossRef]
- Pekny, M.; Pekna, M. Astrocyte reactivity and reactive astrogliosis: Costs and benefits. Physiol. Rev. 2014, 94, 1077–1098. [Google Scholar] [CrossRef] [PubMed]
- Pekny, M.; Levéen, P.; Pekna, M.; Eliasson, C.; Berthold, C.H.; Westermark, B.; Betsholtz, C. Mice lacking glial fibrillary acidic protein display astrocytes devoid of intermediate filaments but develop and reproduce normally. EMBO J. 1995, 14, 1590–1598. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Lundkvist, A.; Andersson, D.; Wilhelmsson, U.; Nagai, N.; Pardo, A.C.; Nodin, C.; Ståhlberg, A.; Aprico, K.; Larsson, K.; et al. Protective role of reactive astrocytes in brain ischemia. J. Cereb. Blood Flow Metab. 2008, 28, 468–481. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.; Malouf, A.T.; Messing, A.; Silver, J. Glial fibrillary acidic protein is necessary for mature astrocytes to react to β-amyloid. Glia 1999, 25, 390–403. [Google Scholar] [CrossRef]
- Liedtke, W.; Edelmann, W.; Chiu, F.C.; Kucherlapati, R.; Raine, C.S. Experimental autoimmune encephalomyelitis in mice lacking glial fibrillary acidic protein is characterized by a more severe clinical course and an infiltrative central nervous system lesion. Am. J. Pathol. 1998, 152, 251–259. [Google Scholar]
- Stenzel, W.; Soltek, S.; Schlüter, D.; Deckert, M. The intermediate filament GFAP is important for the control of experimental murine Staphylococcus aureus-induced brain abscess and Toxoplasma encephalitis. J. Neuropathol. Exp. Neurol. 2004, 63, 631–640. [Google Scholar] [CrossRef]
- Lendahl, U.; Zimmerman, L.B.; McKay, R.D. CNS stem cells express a new class of intermediate filament protein. Cell 1990, 60, 585–595. [Google Scholar] [CrossRef]
- Frederiksen, K.; McKay, R.D. Proliferation and differentiation of rat neuroepithelial precursor cells in vivo. J. Neurosci. 1988, 8, 1144–1151. [Google Scholar] [CrossRef]
- Dahlstrand, J.; Lardelli, M.; Lendahl, U. Nestin mRNA expression correlates with the central nervous system progenitor cell state in many, but not all, regions of developing central nervous system. Brain Res. Dev. Brain Res. 1995, 84, 109–129. [Google Scholar] [CrossRef]
- Arai, H.; Ikota, H.; Sugawara, K.; Nobusawa, S.; Hirato, J.; Nakazato, Y. Nestin expression in brain tumors: Its utility for pathological diagnosis and correlation with the prognosis of high-grade gliomas. Brain Tumor Pathol. 2012, 29, 160–167. [Google Scholar] [CrossRef]
- Dahlstrand, J.; Collins, V.P.; Lendahl, U. Expression of the class VI intermediate filament nestin in human central nervous system tumors. Cancer Res. 1992, 52, 5334–5341. [Google Scholar]
- Tohyama, T.; Lee, V.M.; Rorke, L.B.; Marvin, M.; McKay, R.D.; Trojanowski, J.Q. Nestin expression in embryonic human neuroepithelium and in human neuroepithelial tumor cells. Lab. Investig. 1992, 66, 303–313. [Google Scholar] [PubMed]
- Shimizu, T.; Sugawara, K.; Tosaka, M.; Imai, H.; Hoya, K.; Takeuchi, T.; Sasaki, T.; Saito, N. Nestin expression in vascular malformations: A novel marker for proliferative endothelium. Neurol. Med. Chir. 2006, 46, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Pallari, H.M.; Lindqvist, J.; Torvaldson, E.; Ferraris, S.E.; He, T.; Sahlgren, C.; Eriksson, J.E. Nestin as a regulator of Cdk5 in differentiating myoblasts. Mol. Biol. Cell 2011, 22, 1539–1549. [Google Scholar] [CrossRef] [PubMed]
- Takamori, Y.; Mori, T.; Wakabayashi, T.; Nagasaka, Y.; Matsuzaki, T.; Yamada, H. Nestin-positive microglia in adult rat cerebral cortex. Brain Res. 2009, 1270, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Zerlin, M.; Levison, S.W.; Goldman, J.E. Early patterns of migration, morphogenesis, and intermediate filament expression of subventricular zone cells in the postnatal rat forebrain. J. Neurosci. 1995, 15, 7238–7249. [Google Scholar] [CrossRef]
- Middeldorp, J.; Kamphuis, W.; Sluijs, J.A.; Achoui, D.; Leenaars, C.H.; Feenstra, M.G.; van Tijn, P.; Fischer, D.F.; Berkers, C.; Ovaa, H.; et al. Intermediate filament transcription in astrocytes is repressed by proteasome inhibition. FASEB J. 2009, 23, 2710–2726. [Google Scholar] [CrossRef]
- Frisén, J.; Johansson, C.B.; Török, C.; Risling, M.; Lendahl, U. Rapid, widespread, and longlasting induction of nestin contributes to the generation of glial scar tissue after CNS injury. J. Cell Biol. 1995, 131, 453–464. [Google Scholar] [CrossRef]
- Clarke, S.R.; Shetty, A.K.; Bradley, J.L.; Turner, D.A. Reactive astrocytes express the embryonic intermediate neurofilament nestin. Neuroreport 1994, 5, 1885–1888. [Google Scholar] [CrossRef]
- Bellin, R.M.; Sernett, S.W.; Becker, B.; Ip, W.; Huiatt, T.W.; Robson, R.M. Molecular characteristics and interactions of the intermediate filament protein synemin. Interactions with alpha-actinin may anchor synemin-containing heterofilaments. J. Biol. Chem. 1999, 274, 29493–29499. [Google Scholar] [CrossRef][Green Version]
- Steinert, P.M.; Chou, Y.H.; Prahlad, V.; Parry, D.A.; Marekov, L.N.; Wu, K.C.; Jang, S.I.; Goldman, R.D. A high molecular weight intermediate filament-associated protein in BHK-21 cells is nestin, a type VI intermediate filament protein. Limited co-assembly in vitro to form heteropolymers with type III vimentin and type IV alpha-internexin. J. Biol. Chem. 1999, 274, 9881–9890. [Google Scholar] [CrossRef]
- Moeton, M.; Stassen, O.M.; Sluijs, J.A.; van der Meer, V.W.; Kluivers, L.J.; van Hoorn, H.; Schmidt, T.; Reits, E.A.; van Strien, M.E.; Hol, E.M. GFAP isoforms control intermediate filament network dynamics, cell morphology, and focal adhesions. Cell. Mol. Life Sci. 2016, 73, 4101–4120. [Google Scholar] [CrossRef]
- Herrmann, H.; Häner, M.; Brettel, M.; Müller, S.A.; Goldie, K.N.; Fedtke, B.; Lustig, A.; Franke, W.W.; Aebi, U. Structure and assembly properties of the intermediate filament protein vimentin: The role of its head, rod and tail domains. J. Mol. Biol. 1996, 264, 933–953. [Google Scholar] [CrossRef]
- Hatzfeld, M.; Burba, M. Function of type I and type II keratin head domains: Their role in dimer, tetramer and filament formation. J. Cell Sci. 1994, 107 Pt 7, 1959–1972. [Google Scholar]
- Herrmann, H.; Aebi, U. Intermediate filaments and their associates: Multi-talented structural elements specifying cytoarchitecture and cytodynamics. Curr. Opin. Cell Biol. 2000, 12, 79–90. [Google Scholar] [CrossRef]
- Herrmann, H.; Aebi, U. Intermediate filaments: Molecular structure, assembly mechanism, and integration into functionally distinct intracellular Scaffolds. Annu. Rev. Biochem. 2004, 73, 749–789. [Google Scholar] [CrossRef]
- Bernal, A.; Arranz, L. Nestin-expressing progenitor cells: Function, identity and therapeutic implications. Cell. Mol. Life Sci. 2018, 75, 2177–2195. [Google Scholar] [CrossRef]
- Hemmati-Brivanlou, A.; Mann, R.W.; Harland, R.M. A protein expressed in the growth cones of embryonic vertebrate neurons defines a new class of intermediate filament protein. Neuron 1992, 9, 417–428. [Google Scholar] [CrossRef]
- Napier, A.; Yuan, A.; Cole, G.J. Characterization of the chicken transitin gene reveals a strong relationship to the nestin intermediate filament class. J. Mol. Neurosci. 1999, 12, 11–22. [Google Scholar] [CrossRef]
- Guérette, D.; Khan, P.A.; Savard, P.E.; Vincent, M. Molecular evolution of type VI intermediate filament proteins. BMC Evol. Biol. 2007, 7, 164. [Google Scholar] [CrossRef]
- Thomsen, R.; Pallesen, J.; Daugaard, T.F.; Børglum, A.D.; Nielsen, A.L. Genome wide assessment of mRNA in astrocyte protrusions by direct RNA sequencing reveals mRNA localization for the intermediate filament protein nestin. Glia 2013, 61, 1922–1937. [Google Scholar] [CrossRef]
- de Pablo, Y.; Chen, M.; Möllerström, E.; Pekna, M.; Pekny, M. Drugs targeting intermediate filaments can improve neurosupportive properties of astrocytes. Brain Res. Bull. 2018, 136, 130–138. [Google Scholar] [CrossRef]
- Bigas, A.; Espinosa, L. The multiple usages of Notch signaling in development, cell differentiation and cancer. Curr. Opin. Cell Biol. 2018, 55, 1–7. [Google Scholar] [CrossRef]
- Irvin, D.K.; Zurcher, S.D.; Nguyen, T.; Weinmaster, G.; Kornblum, H.I. Expression patterns of Notch1, Notch2, and Notch3 suggest multiple functional roles for the Notch-DSL signaling system during brain development. J. Comp. Neurol. 2001, 436, 167–181. [Google Scholar] [CrossRef]
- Marumo, T.; Takagi, Y.; Muraki, K.; Hashimoto, N.; Miyamoto, S.; Tanigaki, K. Notch signaling regulates nucleocytoplasmic Olig2 translocation in reactive astrocytes differentiation after ischemic stroke. Neurosci. Res. 2013, 75, 204–209. [Google Scholar] [CrossRef]
- Benner, E.J.; Luciano, D.; Jo, R.; Abdi, K.; Paez-Gonzalez, P.; Sheng, H.; Warner, D.S.; Liu, C.; Eroglu, C.; Kuo, C.T. Protective astrogenesis from the SVZ niche after injury is controlled by Notch modulator Thbs4. Nature 2013, 497, 369–373. [Google Scholar] [CrossRef]
- Lasič, E.; Trkov Bobnar, S.; Wilhelmsson, U.; de Pablo, Y.; Pekny, M.; Zorec, R.; Stenovec, M. Nestin affects fusion pore dynamics in mouse astrocytes. Acta Physiol. (Oxf.) 2020, 228, e13399. [Google Scholar] [CrossRef]
- Wilhelmsson, U.; Faiz, M.; de Pablo, Y.; Sjöqvist, M.; Andersson, D.; Widestrand, A.; Potokar, M.; Stenovec, M.; Smith, P.L.; Shinjyo, N.; et al. Astrocytes negatively regulate neurogenesis through the Jagged1-mediated Notch pathway. Stem Cells 2012, 30, 2320–2329. [Google Scholar] [CrossRef]
- Wilhelmsson, U.; Kalm, M.; Pekna, M.; Pekny, M. Nestin null mice show improved reversal place learning. Neurochem. Res. 2020, 45, 215–220. [Google Scholar] [CrossRef]
- Chou, Y.H.; Khuon, S.; Herrmann, H.; Goldman, R.D. Nestin promotes the phosphorylation-dependent disassembly of vimentin intermediate filaments during mitosis. Mol. Biol. Cell 2003, 14, 1468–1478. [Google Scholar] [CrossRef]
- Leduc, C.; Etienne-Manneville, S. Regulation of microtubule-associated motors drives intermediate filament network polarization. J. Cell Biol. 2017, 216, 1689–1703. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, Y.; Boëda, B.; Etienne-Manneville, S. APC binds intermediate filaments and is required for their reorganization during cell migration. J. Cell Biol. 2013, 200, 249–258. [Google Scholar] [CrossRef] [PubMed]
- Xue, Z.G.; Cheraud, Y.; Brocheriou, V.; Izmiryan, A.; Titeux, M.; Paulin, D.; Li, Z. The mouse synemin gene encodes three intermediate filament proteins generated by alternative exon usage and different open reading frames. Exp. Cell Res. 2004, 298, 431–444. [Google Scholar] [CrossRef]
- Izmiryan, A.; Cheraud, Y.; Khanamiryan, L.; Leterrier, J.F.; Federici, T.; Peltekian, E.; Moura-Neto, V.; Paulin, D.; Li, Z.; Xue, Z.G. Different expression of synemin isoforms in glia and neurons during nervous system development. Glia 2006, 54, 204–213. [Google Scholar] [CrossRef]
- Hirako, Y.; Yamakawa, H.; Tsujimura, Y.; Nishizawa, Y.; Okumura, M.; Usukura, J.; Matsumoto, H.; Jackson, K.W.; Owaribe, K.; Ohara, O. Characterization of mammalian synemin, an intermediate filament protein present in all four classes of muscle cells and some neuroglial cells: Co-localization and interaction with type III intermediate filament proteins and keratins. Cell Tissue Res. 2003, 313, 195–207. [Google Scholar] [CrossRef] [PubMed]
- Luna, G.; Lewis, G.P.; Banna, C.D.; Skalli, O.; Fisher, S.K. Expression profiles of nestin and synemin in reactive astrocytes and Müller cells following retinal injury: A comparison with glial fibrillar acidic protein and vimentin. Mol. Vis. 2010, 16, 2511–2523. [Google Scholar] [PubMed]
- de Souza Martins, S.C.; Agbulut, O.; Diguet, N.; Larcher, J.C.; Paulsen, B.S.; Rehen, S.K.; Moura-Neto, V.; Paulin, D.; Li, Z.; Xue, Z. Dynamic expression of synemin isoforms in mouse embryonic stem cells and neural derivatives. BMC Cell Biol. 2011, 12, 51. [Google Scholar] [CrossRef] [PubMed]
- Bellin, R.M.; Huiatt, T.W.; Critchley, D.R.; Robson, R.M. Synemin may function to directly link muscle cell intermediate filaments to both myofibrillar Z-lines and costameres. J. Biol. Chem. 2001, 276, 32330–32337. [Google Scholar] [CrossRef]
- Fawcett, D.W. On the occurrence of a fibrous lamina on the inner aspect of the nuclear envelope in certain cells of vertebrates. Am. J. Anat. 1966, 119, 129–145. [Google Scholar] [CrossRef]
- Herrmann, H.; Strelkov, S.V. History and phylogeny of intermediate filaments: Now in insects. BMC Biol. 2011, 9, 16. [Google Scholar] [CrossRef]
- de Leeuw, R.; Gruenbaum, Y.; Medalia, O. Nuclear Lamins: Thin Filaments with Major Functions. Trends Cell Biol. 2018, 28, 34–45. [Google Scholar] [CrossRef] [PubMed]
- Dechat, T.; Adam, S.A.; Taimen, P.; Shimi, T.; Goldman, R.D. Nuclear lamins. Cold Spring Harbor Perspect. Biol. 2010, 2, a000547. [Google Scholar] [CrossRef] [PubMed]
- Aaronson, R.P.; Blobel, G. Isolation of nuclear pore complexes in association with a lamina. Proc. Natl. Acad. Sci. USA 1975, 72, 1007–1011. [Google Scholar] [CrossRef]
- Gerace, L.; Blobel, G. The nuclear envelope lamina is reversibly depolymerized during mitosis. Cell 1980, 19, 277–287. [Google Scholar] [CrossRef]
- Goldman, A.E.; Maul, G.; Steinert, P.M.; Yang, H.Y.; Goldman, R.D. Keratin-like proteins that coisolate with intermediate filaments of BHK-21 cells are nuclear lamins. Proc. Natl. Acad. Sci. USA 1986, 83, 3839–3843. [Google Scholar] [CrossRef]
- McKeon, F.D.; Kirschner, M.W.; Caput, D. Homologies in both primary and secondary structure between nuclear envelope and intermediate filament proteins. Nature 1986, 319, 463–468. [Google Scholar] [CrossRef]
- Gray, E.G.; Guillery, R.W. On nuclear structure in the ventral nerve cord of the leech Hirudo medicinalis. Zellforsch. Mikrosk. Anat. 1963, 59, 738–745. [Google Scholar] [CrossRef]
- Krohne, G.; Benavente, R. The nuclear lamins. A multigene family of proteins in evolution and differentiation. Exp. Cell Res. 1986, 162, 1–10. [Google Scholar] [CrossRef]
- Worman, H.J.; Lazaridis, I.; Georgatos, S.D. Nuclear lamina heterogeneity in mammalian cells. Differential expression of the major lamins and variations in lamin B phosphorylation. J. Biol. Chem. 1988, 263, 12135–12141. [Google Scholar]
- Toesca, A. Distribution and developmental expression of lamin-like immunoreactivity in the central nervous system. Cell Biol. Int. Rep. 1992, 16, 165–174. [Google Scholar] [CrossRef]
- Röber, R.A.; Weber, K.; Osborn, M. Differential timing of nuclear lamin A/C expression in the various organs of the mouse embryo and the young animal: A developmental study. Development 1989, 105, 365–378. [Google Scholar] [PubMed]
- Altman, J.; Das, G.D. Autoradiographic and histological studies of postnatal neurogenesis. I. A longitudinal investigation of the kinetics, migration and transformation of cells incorporating tritiated thymidine in neonate rats, with special reference to postnatal neurogenesis in some brain regions. J. Comp. Neurol. 1966, 126, 337–389. [Google Scholar] [CrossRef] [PubMed]
- Mahajani, S.; Giacomini, C.; Marinaro, F.; De Pietri Tonelli, D.; Contestabile, A.; Gasparini, L. Lamin B1 levels modulate differentiation into neurons during embryonic corticogenesis. Sci. Rep. 2017, 7, 4897. [Google Scholar] [CrossRef]
- Jung, H.J.; Coffinier, C.; Choe, Y.; Beigneux, A.P.; Davies, B.S.; Yang, S.H.; Barnes, R.H.; Hong, J.; Sun, T.; Pleasure, S.J.; et al. Regulation of prelamin A but not lamin C by miR-9, a brain-specific microRNA. Proc. Natl. Acad. Sci. USA 2012, 109, E423–E431. [Google Scholar] [CrossRef]
- Takamori, Y.; Hirahara, Y.; Wakabayashi, T.; Mori, T.; Koike, T.; Kataoka, Y.; Tamura, Y.; Kurebayashi, S.; Kurokawa, K.; Yamada, H. Differential expression of nuclear lamin subtypes in the neural cells of the adult rat cerebral cortex. IBRO Rep. 2018, 5, 99–109. [Google Scholar] [CrossRef]
- Dubińska-Magiera, M.; Kozioł, K.; Machowska, M.; Piekarowicz, K.; Filipczak, D.; Rzepecki, R. Emerin is required for proper nucleus reassembly after mitosis: Implications for new pathogenetic mechanisms for laminopathies detected in EDMD1 patients. Cells 2019, 8, 240. [Google Scholar] [CrossRef]
- Peric-Hupkes, D.; Meuleman, W.; Pagie, L.; Bruggeman, S.W.; Solovei, I.; Brugman, W.; Gräf, S.; Flicek, P.; Kerkhoven, R.M.; van Lohuizen, M.; et al. Molecular maps of the reorganization of genome-nuclear lamina interactions during differentiation. Mol. Cell 2010, 38, 603–613. [Google Scholar] [CrossRef]
- Crisp, M.; Liu, Q.; Roux, K.; Rattner, J.B.; Shanahan, C.; Burke, B.; Stahl, P.D.; Hodzic, D. Coupling of the nucleus and cytoplasm: Role of the LINC complex. J. Cell Biol. 2006, 172, 41–53. [Google Scholar] [CrossRef]
- Tzur, Y.B.; Wilson, K.L.; Gruenbaum, Y. SUN-domain proteins: ‘Velcro’ that links the nucleoskeleton to the cytoskeleton. Nat. Rev. Mol. Cell Biol. 2006, 7, 782–788. [Google Scholar] [CrossRef]
- Bione, S.; Maestrini, E.; Rivella, S.; Mancini, M.; Regis, S.; Romeo, G.; Toniolo, D. Identification of a novel X-linked gene responsible for Emery-Dreifuss muscular dystrophy. Nat. Genet. 1994, 8, 323–327. [Google Scholar] [CrossRef]
- Bonne, G.; Di Barletta, M.R.; Varnous, S.; Bécane, H.M.; Hammouda, E.H.; Merlini, L.; Muntoni, F.; Greenberg, C.R.; Gary, F.; Urtizberea, J.A.; et al. Mutations in the gene encoding lamin A/C cause autosomal dominant Emery-Dreifuss muscular dystrophy. Nat. Genet. 1999, 21, 285–288. [Google Scholar] [CrossRef] [PubMed]
- Deal, S.L.; Yamamoto, S. Unweaving the role of nuclear Lamins in neural circuit integrity. Cell Stress 2018, 2, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, K.H.; Kennedy, B.K. When lamins go bad: Nuclear structure and disease. Cell 2013, 152, 1365–1375. [Google Scholar] [CrossRef] [PubMed]
- De Sandre-Giovannoli, A.; Chaouch, M.; Kozlov, S.; Vallat, J.M.; Tazir, M.; Kassouri, N.; Szepetowski, P.; Hammadouche, T.; Vandenberghe, A.; Stewart, C.L.; et al. Homozygous defects in LMNA, encoding lamin A/C nuclear-envelope proteins, cause autosomal recessive axonal neuropathy in human (Charcot-Marie-Tooth disorder type 2) and mouse. Am. J. Hum. Genet. 2002, 70, 726–736. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, T.; Escalante-Alcalde, D.; Bhatt, H.; Anver, M.; Bhat, N.; Nagashima, K.; Stewart, C.L.; Burke, B. Loss of A-type lamin expression compromises nuclear envelope integrity leading to muscular dystrophy. J. Cell Biol. 1999, 147, 913–920. [Google Scholar] [CrossRef]
- Arocena, D.G.; Iwahashi, C.K.; Won, N.; Beilina, A.; Ludwig, A.L.; Tassone, F.; Schwartz, P.H.; Hagerman, P.J. Induction of inclusion formation and disruption of lamin A/C structure by premutation CGG-repeat RNA in human cultured neural cells. Hum. Mol. Genet. 2005, 14, 3661–3671. [Google Scholar] [CrossRef]
- Hagerman, R.J.; Hagerman, P. Fragile X-associated tremor/ataxia syndrome—Features, mechanisms and management. Nat. Rev. Neurol. 2016, 12, 403–412. [Google Scholar] [CrossRef]
- Iwahashi, C.K.; Yasui, D.H.; An, H.J.; Greco, C.M.; Tassone, F.; Nannen, K.; Babineau, B.; Lebrilla, C.B.; Hagerman, R.J.; Hagerman, P.J. Protein composition of the intranuclear inclusions of FXTAS. Brain 2006, 129, 256–271. [Google Scholar] [CrossRef]
- Hagerman, R.J.; Coffey, S.M.; Maselli, R.; Soontarapornchai, K.; Brunberg, J.A.; Leehey, M.A.; Zhang, L.; Gane, L.W.; Fenton-Farrell, G.; Tassone, F.; et al. Neuropathy as a presenting feature in fragile X-associated tremor/ataxia syndrome. Am. J. Med. Genet. A 2007, 143A, 2256–2260. [Google Scholar] [CrossRef]
- Naidoo, M.; Anthony, K. Dystrophin Dp71 and the neuropathophysiology of Duchenne muscular dystrophy. Mol. Neurobiol. 2020, 57, 1748–1767. [Google Scholar] [CrossRef]
- Ruggieri, S.; De Giorgis, M.; Annese, T.; Tamma, R.; Notarangelo, A.; Marzullo, A.; Senetta, R.; Cassoni, P.; Notarangelo, M.; Ribatti, D.; et al. Dp71 Expression in Human Glioblastoma. Int. J. Mol. Sci. 2019, 20, 5429. [Google Scholar] [CrossRef]
- Ratner, N.; Miller, S.J. A RASopathy gene commonly mutated in cancer: The neurofibromatosis type 1 tumour suppressor. Nat. Rev. Cancer 2015, 15, 290–301. [Google Scholar] [CrossRef]
- Pamenter, M.E.; Perkins, G.A.; McGinness, A.K.; Gu, X.Q.; Ellisman, M.H.; Haddad, G.G. Autophagy and apoptosis are differentially induced in neurons and astrocytes treated with an in vitro mimic of the ischemic penumbra. PLoS ONE 2012, 7, e51469. [Google Scholar] [CrossRef] [PubMed]
- Bigley, T.M.; Reitsma, J.M.; Terhune, S.S. Antagonistic Relationship between Human Cytomegalovirus pUL27 and pUL97 Activities during Infection. J. Virol. 2015, 89, 10230–10246. [Google Scholar] [CrossRef] [PubMed]
- Trias, E.; Beilby, P.R.; Kovacs, M.; Ibarburu, S.; Varela, V.; Barreto-Núñez, R.; Bradford, S.C.; Beckman, J.S.; Barbeito, L. Emergence of Microglia Bearing Senescence Markers During Paralysis Progression in a Rat Model of Inherited ALS. Front. Aging Neurosci. 2019, 11, 42. [Google Scholar] [CrossRef] [PubMed]
- Ruhrberg, C.; Watt, F.M. The plakin family: Versatile organizers of cytoskeletal architecture. Curr. Opin. Genet. Dev. 1997, 7, 392–397. [Google Scholar] [CrossRef]
- Jefferson, J.J.; Leung, C.L.; Liem, R.K. Plakins: Goliaths that link cell junctions and the cytoskeleton. Nat. Rev. Mol. Cell Biol. 2004, 5, 542–553. [Google Scholar] [CrossRef]
- Byers, T.J.; Beggs, A.H.; McNally, E.M.; Kunkel, L.M. Novel actin crosslinker superfamily member identified by a two step degenerate PCR procedure. FEBS Lett. 1995, 368, 500–504. [Google Scholar] [CrossRef]
- Okuda, T.; Matsuda, S.; Nakatsugawa, S.; Ichigotani, Y.; Iwahashi, N.; Takahashi, M.; Ishigaki, T.; Hamaguchi, M. Molecular cloning of macrophin, a human homologue of Drosophila kakapo with a close structural similarity to plectin and dystrophin. Biochem. Biophys. Res. Commun. 1999, 264, 568–574. [Google Scholar] [CrossRef]
- Sun, Y.; Zhang, J.; Kraeft, S.K.; Auclair, D.; Chang, M.S.; Liu, Y.; Sutherland, R.; Salgia, R.; Griffin, J.D.; Ferland, L.H.; et al. Molecular cloning and characterization of human trabeculin-alpha, a giant protein defining a new family of actin-binding proteins. J. Biol. Chem. 1999, 274, 33522–33530. [Google Scholar] [CrossRef]
- Sonnenberg, A.; Liem, R.K. Plakins in development and disease. Exp. Cell Res. 2007, 313, 2189–2203. [Google Scholar] [CrossRef] [PubMed]
- Errante, L.D.; Wiche, G.; Shaw, G. Distribution of plectin, an intermediate filament-associated protein, in the adult rat central nervous system. J. Neurosci. Res. 1994, 37, 515–528. [Google Scholar] [CrossRef] [PubMed]
- Kálmán, M.; Szabó, A. Plectin immunopositivity appears in the astrocytes in the white matter but not in the gray matter after stab wounds. Brain Res. 2000, 857, 291–294. [Google Scholar] [CrossRef]
- Matsunaga, T.; Kanzaki, J.; Hosoda, Y. Ultrastructure of astrocytes in the transitional region of the human eighth cranial nerve. Hear. Res. 1994, 76, 118–126. [Google Scholar] [CrossRef]
- Wegiel, J.; Wisniewski, H.M. Rosenthal fibers, eosinophilic inclusions, and anchorage densities with desmosome-like structures in astrocytes in Alzheimer’s disease. Acta Neuropathol. 1994, 87, 355–361. [Google Scholar] [CrossRef]
- Dubash, A.D.; Green, K.J. Desmosomes. Curr. Biol. 2011, 21, R529–R531. [Google Scholar] [CrossRef]
- Walko, G.; Castanon, M.J.; Wiche, G. Molecular architecture and function of the hemidesmosome. Cell Tissue Res. 2015, 360, 529–544. [Google Scholar] [CrossRef]
- Wagner, S.; Tagaya, M.; Koziol, J.A.; Quaranta, V.; del Zoppo, G.J. Rapid disruption of an astrocyte interaction with the extracellular matrix mediated by integrin alpha 6 beta 4 during focal cerebral ischemia/reperfusion. Stroke 1997, 28, 858–865. [Google Scholar] [CrossRef]
- Mueller, S.; Klaus-Kovtun, V.; Stanley, J.R. A 230-kD basic protein is the major bullous pemphigoid antigen. J. Investig. Dermatol. 1989, 92, 33–38. [Google Scholar] [CrossRef]
- Curry, N.; Ghezali, G.; Kaminski Schierle, G.S.; Rouach, N.; Kaminski, C.F. Correlative STED and Atomic Force Microscopy on Live Astrocytes Reveals Plasticity of Cytoskeletal Structure and Membrane Physical Properties during Polarized Migration. Front. Cell. Neurosci. 2017, 11, 104. [Google Scholar] [CrossRef]
- Moffat, J.J.; Ka, M.; Jung, E.M.; Smith, A.L.; Kim, W.Y. The role of MACF1 in nervous system development and maintenance. Semin. Cell Dev. Biol. 2017, 69, 9–17. [Google Scholar] [CrossRef]
- Afghani, N.; Mehta, T.; Wang, J.; Tang, N.; Skalli, O.; Quick, Q.A. Microtubule actin cross-linking factor 1, a novel target in glioblastoma. Int. J. Oncol. 2017, 50, 310–316. [Google Scholar] [CrossRef] [PubMed]
- Pytela, R.; Wiche, G. High molecular weight polypeptides (270,000-340,000) from cultured cells are related to hog brain microtubule-associated proteins but copurify with intermediate filaments. Proc. Natl. Acad. Sci. USA 1980, 77, 4808–4812. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, P.; Zörer, M.; Reipert, S.; Rezniczek, G.A.; Propst, F.; Walko, G.; Fischer, I.; Bauer, J.; Leschnik, M.W.; Lüscher, B.; et al. Targeted inactivation of a developmentally regulated neural plectin isoform (plectin 1c) in mice leads to reduced motor nerve conduction velocity. J. Biol. Chem. 2009, 284, 26502–26509. [Google Scholar] [CrossRef]
- Castañón, M.J.; Walko, G.; Winter, L.; Wiche, G. Plectin-intermediate filament partnership in skin, skeletal muscle, and peripheral nerve. Histochem. Cell Biol. 2013, 140, 33–53. [Google Scholar] [CrossRef] [PubMed]
- Wiche, G.; Krepler, R.; Artlieb, U.; Pytela, R.; Denk, H. Occurrence and immunolocalization of plectin in tissues. J. Cell Biol. 1983, 97, 887–901. [Google Scholar] [CrossRef]
- Winter, L.; Wiche, G. The many faces of plectin and plectinopathies: Pathology and mechanisms. Acta Neuropathol. 2013, 125, 77–93. [Google Scholar] [CrossRef]
- Fuchs, P.; Zörer, M.; Rezniczek, G.A.; Spazierer, D.; Oehler, S.; Castañón, M.J.; Hauptmann, R.; Wiche, G. Unusual 5′ transcript complexity of plectin isoforms: Novel tissue-specific exons modulate actin binding activity. Hum. Mol. Genet. 1999, 8, 2461–2472. [Google Scholar] [CrossRef]
- Elliott, C.E.; Becker, B.; Oehler, S.; Castanon, M.J.; Hauptmann, R.; Wiche, G. Plectin transcript diversity: Identification and tissue distribution of variants with distinct first coding exons and rodless isoforms. Genomics 1997, 42, 115–125. [Google Scholar] [CrossRef]
- Sevcik, J.; Urbanikova, L.; Kost’an, J.; Janda, L.; Wiche, G. Actin-binding domain of mouse plectin. Crystal structure and binding to vimentin. Eur. J. Biochem. 2004, 271, 1873–1884. [Google Scholar] [CrossRef]
- Foisner, R.; Traub, P.; Wiche, G. Protein kinase A- and protein kinase C-regulated interaction of plectin with lamin B and vimentin. Proc. Natl. Acad. Sci. USA 1991, 88, 3812–3816. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, H.; Wiche, G. Specific in situ phosphorylation of plectin in detergent-resistant cytoskeletons from cultured Chinese hamster ovary cells. J. Biol. Chem. 1983, 258, 14610–14618. [Google Scholar] [PubMed]
- Andrä, K.; Nikolic, B.; Stöcher, M.; Drenckhahn, D.; Wiche, G. Not just scaffolding: Plectin regulates actin dynamics in cultured cells. Genes Dev. 1998, 12, 3442–3451. [Google Scholar] [CrossRef] [PubMed]
- Anton-Lamprecht, I. Genetically induced abnormalities of epidermal differentiation and ultrastructure in ichthyoses and epidermolyses: Pathogenesis, heterogeneity, fetal manifestation, and prenatal diagnosis. J. Investig. Dermatol. 1983, 81, 149s–156s. [Google Scholar] [CrossRef]
- Fuchs, E. Intermediate filaments and disease: Mutations that cripple cell strength. J. Cell Biol. 1994, 125, 511–516. [Google Scholar] [CrossRef]
- Gostyńska, K.B.; Nijenhuis, M.; Lemmink, H.; Pas, H.H.; Pasmooij, A.M.; Lang, K.K.; Castañón, M.J.; Wiche, G.; Jonkman, M.F. Mutation in exon 1a of PLEC, leading to disruption of plectin isoform 1a, causes autosomal-recessive skin-only epidermolysis bullosa simplex. Hum. Mol. Genet. 2015, 24, 3155–3162. [Google Scholar] [CrossRef]
- Winter, L.; Türk, M.; Harter, P.N.; Mittelbronn, M.; Kornblum, C.; Norwood, F.; Jungbluth, H.; Thiel, C.T.; Schlötzer-Schrehardt, U.; Schröder, R. Downstream effects of plectin mutations in epidermolysis bullosa simplex with muscular dystrophy. Acta Neuropathol. Commun. 2016, 4, 44. [Google Scholar] [CrossRef]
- Valencia, R.G.; Walko, G.; Janda, L.; Novacek, J.; Mihailovska, E.; Reipert, S.; Andrä-Marobela, K.; Wiche, G. Intermediate filament-associated cytolinker plectin 1c destabilizes microtubules in keratinocytes. Mol. Biol. Cell 2013, 24, 768–784. [Google Scholar] [CrossRef]
| IF Protein | Type | Isoforms in Astrocytes | Size (kDa) | Function in Astrocytes |
|---|---|---|---|---|
| Glial fibrillary acidic protein (GFAP) | III | α, β, γ, δ (ε), ξ, κ, Δ135, Δ164, Δexon6, Δexon7 [18,37] | 38–50 [38] | Resistance to mechanical stress, cell migration and motility, mitosis, myelinization, maintenance and permeability of the blood–brain barrier, neurogenesis, chaperone-mediated autophagy, vesicle mobility, glial scar formation, response to hypoosmotic stress [12,13,32,37,41,42,43,44,45] |
| Vimentin | III | No splice variants reported in astrocytes | 57 [46] | Mechanical integrity of cells and tissues, neurogenesis, glial scar formation, vesicle trafficking, cell morphology, cell motility, cell division, response to hypoosmotic stress [12,13,41,43,44,45,47,48,49] |
| Nestin | VI | No splice variants reported in astrocytes | 240 [50] | Shape of protrusions, neurogenesis, cell motility [44,51] |
| Synemin | VI | α, β (human), H, M, L (mouse) | 230 [52], 180 H (α),150 M (β), 41 L [53,54] | Astrocytoma motility [55] |
| Lamin | V | A, B1, B2, C | 60–70 kDa [56] | Control of gene expression [57], transport of neurofibromin to the nucleus [58], nucleocytoplasmic transport [59] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Potokar, M.; Morita, M.; Wiche, G.; Jorgačevski, J. The Diversity of Intermediate Filaments in Astrocytes. Cells 2020, 9, 1604. https://doi.org/10.3390/cells9071604
Potokar M, Morita M, Wiche G, Jorgačevski J. The Diversity of Intermediate Filaments in Astrocytes. Cells. 2020; 9(7):1604. https://doi.org/10.3390/cells9071604
Chicago/Turabian StylePotokar, Maja, Mitsuhiro Morita, Gerhard Wiche, and Jernej Jorgačevski. 2020. "The Diversity of Intermediate Filaments in Astrocytes" Cells 9, no. 7: 1604. https://doi.org/10.3390/cells9071604
APA StylePotokar, M., Morita, M., Wiche, G., & Jorgačevski, J. (2020). The Diversity of Intermediate Filaments in Astrocytes. Cells, 9(7), 1604. https://doi.org/10.3390/cells9071604






