High Throughput strategies Aimed at Closing the GAP in Our Knowledge of Rho GTPase Signaling
Abstract
1. Introduction
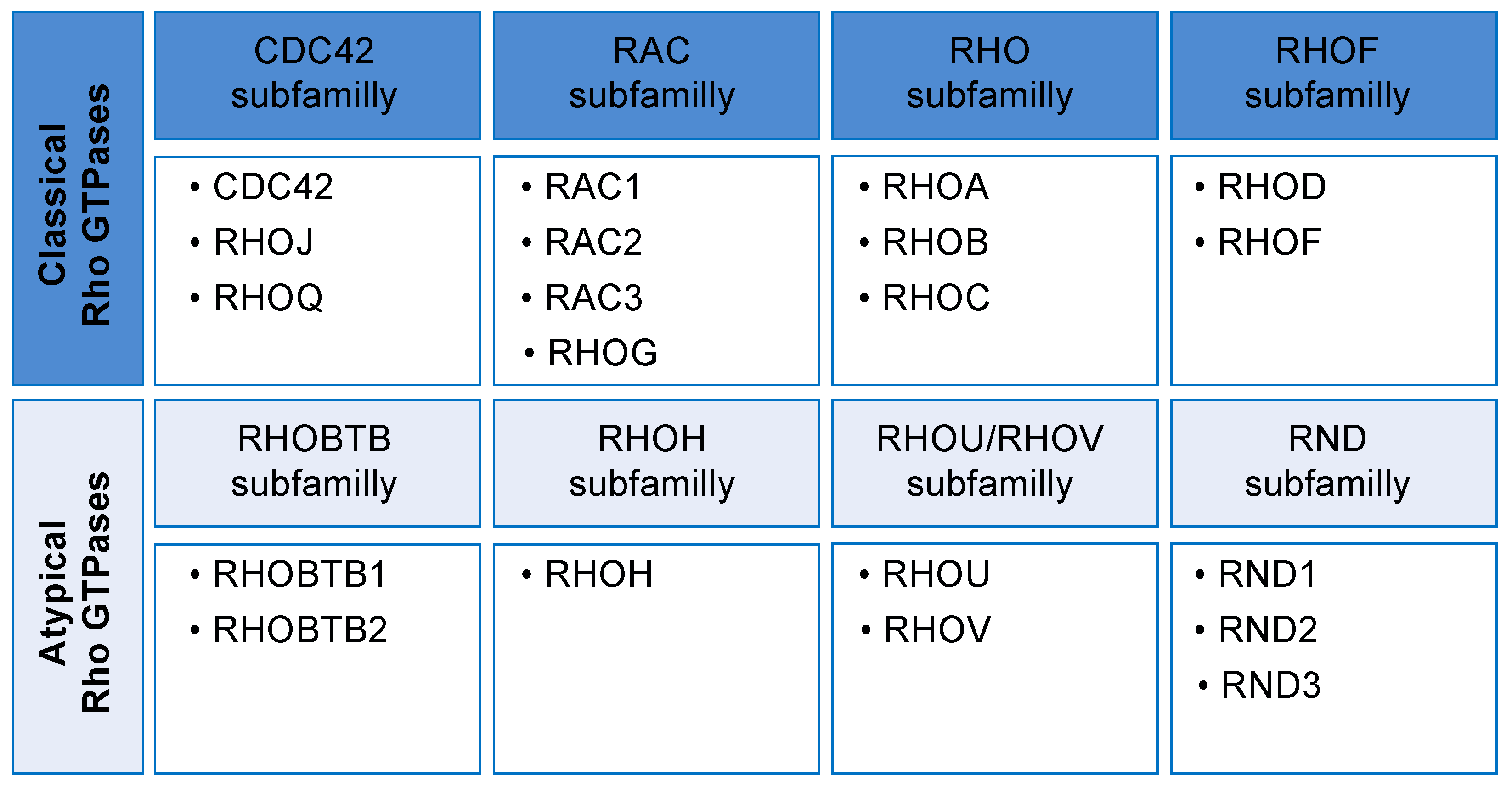
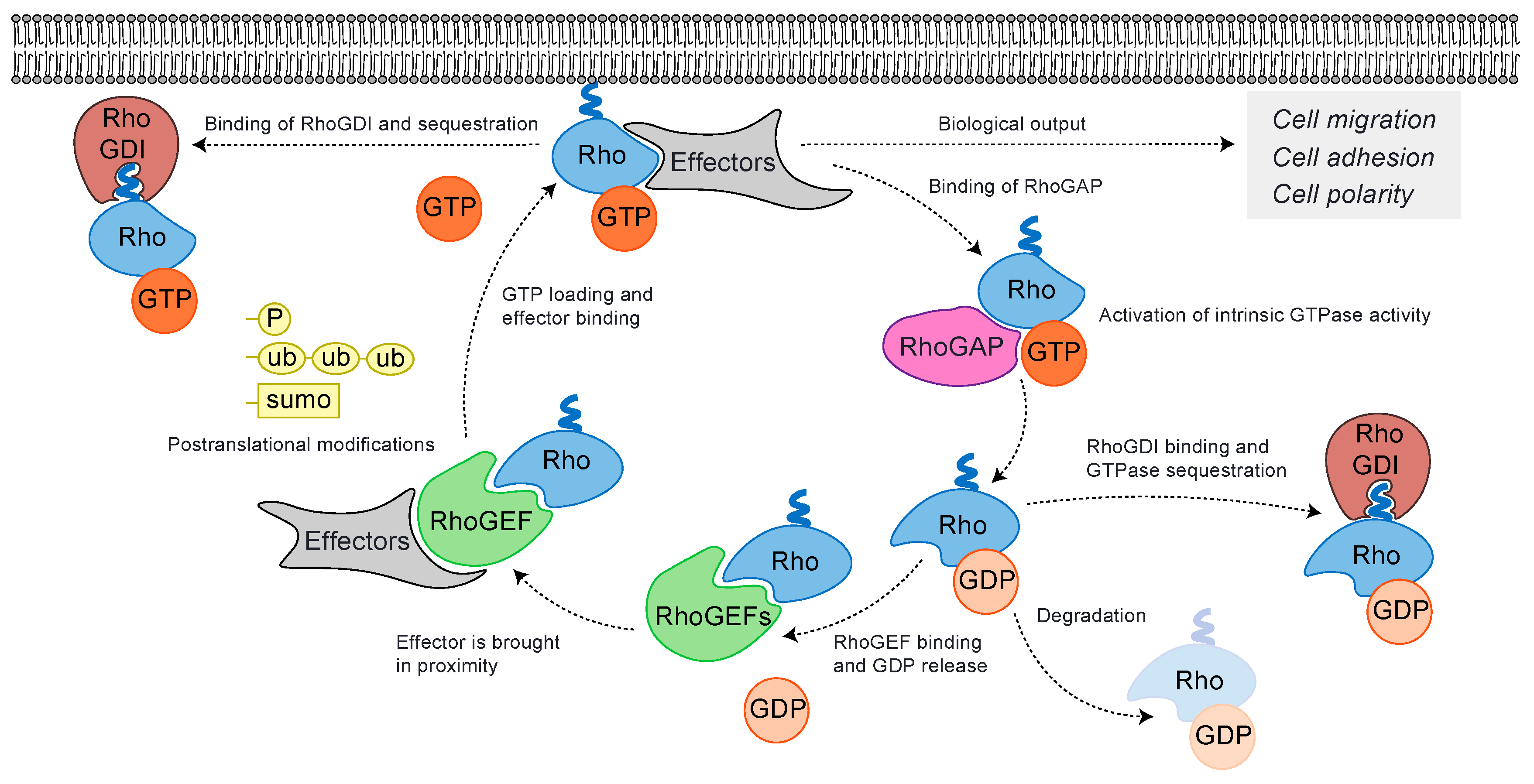
2. Rho GTPases Cycle and Their Regulation
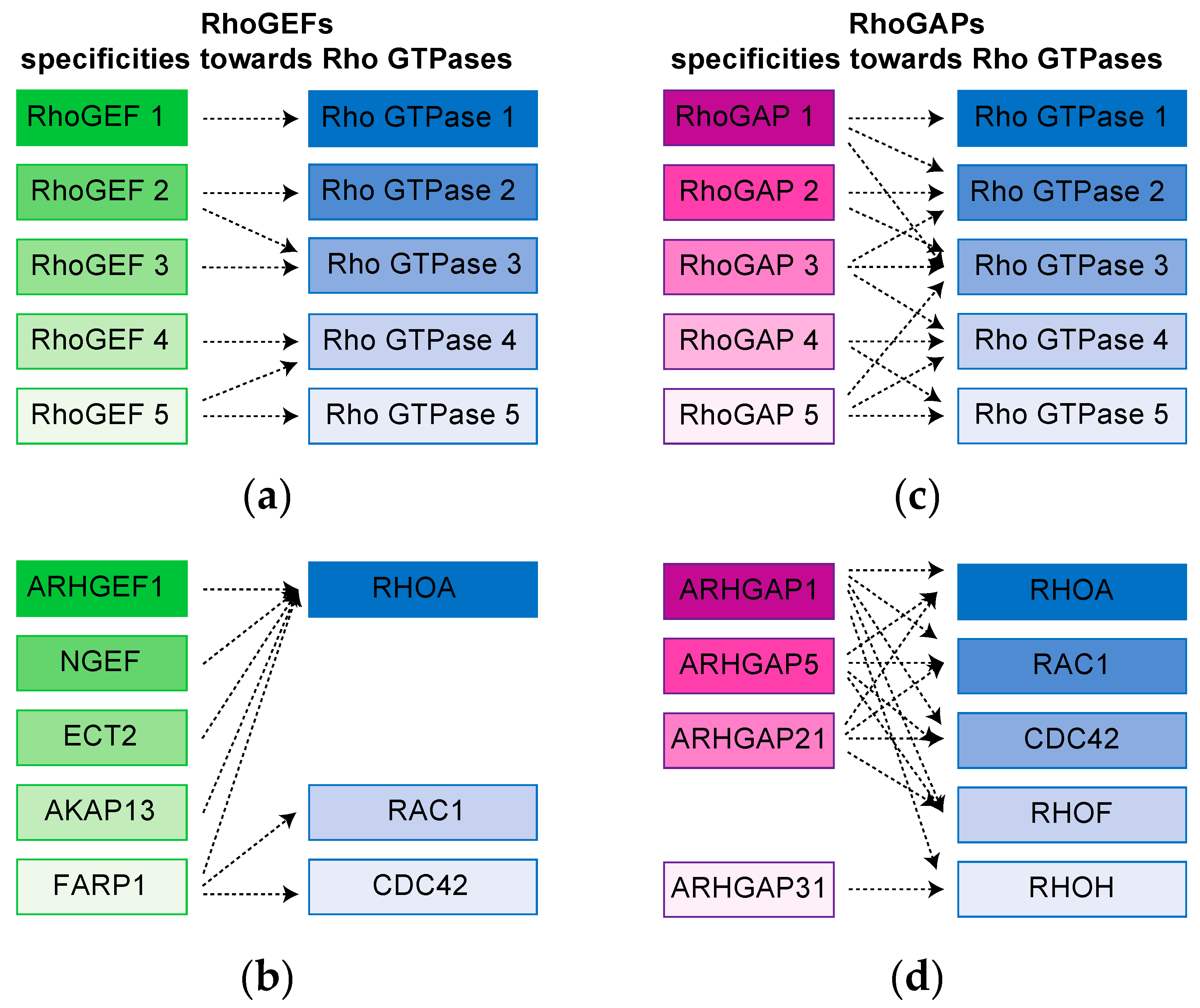
3. Complex Rho GTPases signaling Hubs Are Revealed by proteomic Approaches
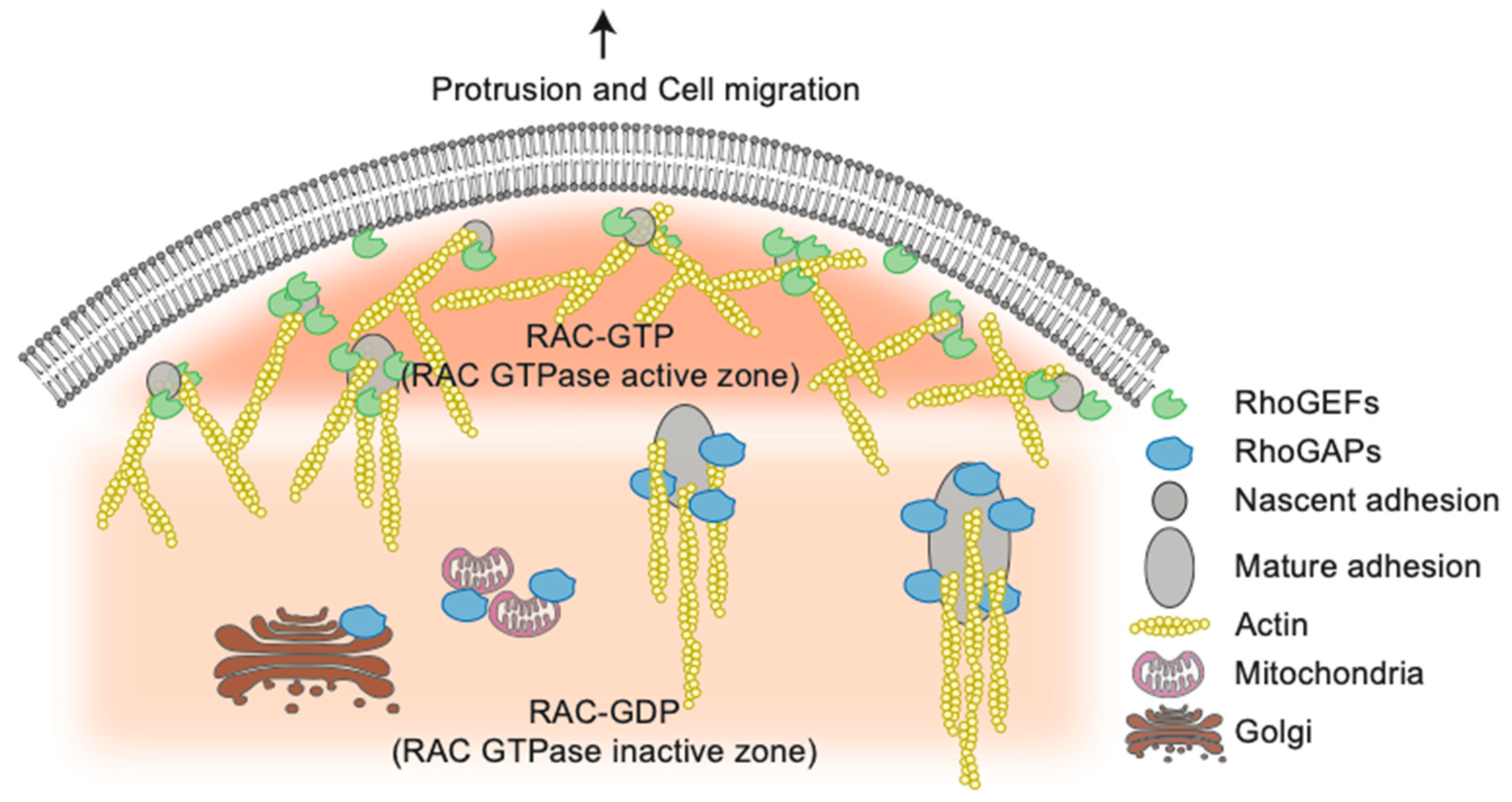
4. High throughput screening Strategies Uncover New Functions for Rho GTPases Network Components
5. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Heasman, S.J.; Ridley, A.J. Mammalian Rho GTPases: New insights into their functions from in vivo studies. Nat. Rev. Mol. Cell Biol. 2008, 9, 690–701. [Google Scholar] [CrossRef]
- Van Aelst, L.; Symons, M. Role of Rho family GTPases in epithelial morphogenesis. Genes Dev. 2002, 16, 1032–1054. [Google Scholar] [CrossRef]
- Porter, A.P.; Papaioannou, A.; Malliri, A. Deregulation of Rho GTPases in cancer. Small GTPases 2016, 7, 123–138. [Google Scholar] [CrossRef]
- Wennerberg, K.; Rossman, K.L.; Der, C.J. The Ras superfamily at a glance. J. Cell Sci. 2005, 118, 843–846. [Google Scholar] [CrossRef] [PubMed]
- Rojas, A.M.; Fuentes, G.; Rausell, A.; Valencia, A. The Ras protein superfamily: Evolutionary tree and role of conserved amino acids. J. Cell Biol. 2012, 196, 189–201. [Google Scholar] [CrossRef] [PubMed]
- Goitre, L.; Trapani, E.; Trabalzini, L.; Retta, S.F. The ras superfamily of small GTPases: The unlocked secrets. Methods Mol. Biol. 2014, 1120, 1–18. [Google Scholar] [PubMed]
- Tapon, N.; Hall, A. Rho, Rac and Cdc42 GTPases regulate the organization of the actin cytoskeleton. Curr. Opin. Cell Biol. 1997, 9, 86–92. [Google Scholar] [CrossRef]
- Aspenström, P.; Ruusala, A.; Pacholsky, D. Taking Rho GTPases to the next level: The cellular functions of atypical Rho GTPases. Exp. Cell Res. 2007, 313, 3673–3679. [Google Scholar] [CrossRef] [PubMed]
- Saras, J.; Wollberg, P.; Aspenström, P. Wrch1 is a GTPase-deficient Cdc42-like protein with unusual binding characteristics and cellular effects. Exp. Cell Res. 2004, 299, 356–369. [Google Scholar] [CrossRef]
- Shutes, A.; Berzat, A.C.; Cox, A.D.; Der, C.J. Atypical Mechanism of Regulation of the Wrch-1 Rho Family Small GTPase. Curr. Boil. 2004, 14, 2052–2056. [Google Scholar] [CrossRef]
- Hodge, R.G.; Ridley, A.J. Regulation and functions of RhoU and RhoV. Small GTPases 2017, 11, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Schiavone, D.; Dewilde, S.; Vallania, F.; Turkson, J.; Di Cunto, F.; Poli, V. The RhoU/Wrch1 Rho GTPase gene is a common transcriptional target of both the gp130/STAT3 and Wnt-1 pathways. Biochem. J. 2009, 421, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Na, J.; Shin, J.Y.; Jeong, H.; Lee, J.Y.; Kim, B.J.; Kim, W.S.; Yune, T.; Ju, B.-G. JMJD3 and NF-κB-dependent activation of Notch1 gene is required for keratinocyte migration during skin wound healing. Sci. Rep. 2017, 7, 6494. [Google Scholar] [CrossRef]
- Loebel, D.A.F.; Studdert, J.B.; Power, M.; Radziewic, T.; Jones, V.; Coultas, L.; Jackson, Y.; Rao, R.S.; Steiner, K.; Fossat, N.; et al. Rhou maintains the epithelial architecture and facilitates differentiation of the foregut endoderm. Development 2011, 138, 4511–4522. [Google Scholar] [CrossRef] [PubMed]
- Dickover, M.; Hegarty, J.M.; Ly, K.; López, D.; Yang, H.; Zhang, R.; Tedeschi, N.; Hsiai, T.K.; Chi, N.C. The atypical Rho GTPase, RhoU, regulates cell-adhesion molecules during cardiac morphogenesis. Dev. Boil. 2014, 389, 182–191. [Google Scholar] [CrossRef] [PubMed]
- Tao, W.; Pennica, D.; Xu, L.; Kalejta, R.F.; Levine, A.J. Wrch-1, a novel member of the Rho gene family that is regulated by Wnt-1. Genome Res. 2001, 15, 1796–1807. [Google Scholar] [CrossRef]
- Chardin, P. Function and regulation of Rnd proteins. Nat. Rev. Mol. Cell Boil. 2006, 7, 54–62. [Google Scholar] [CrossRef]
- Fueller, F.; Kubatzky, K.F. The small GTPase RhoH is an atypical regulator of haematopoietic cells. Cell Commun. Signal. 2008, 6, 6. [Google Scholar] [CrossRef]
- Li, X.; Bu, X.; Lu, B.; Avraham, H.; Flavell, R.A.; Lim, B. The Hematopoiesis-Specific GTP-Binding Protein RhoH Is GTPase Deficient and Modulates Activities of Other Rho GTPases by an Inhibitory Function. Mol. Cell. Boil. 2002, 22, 1158–1171. [Google Scholar] [CrossRef]
- Gu, Y.; Jasti, A.C.; Jansen, M.; Siefring, J.E. RhoH, a hematopoietic-specific Rho GTPase, regulates proliferation, survival, migration, and engraftment of hematopoietic progenitor cells. Blood 2005, 105, 1467–1475. [Google Scholar] [CrossRef]
- Tajadura-Ortega, V.; Garg, R.; Allen, R.; Owczarek, C.; Bright, M.D.; Kean, S.; Mohd-Noor, A.; Grigoriadis, A.; Elston, T.C.; Hahn, K.M.; et al. An RNAi screen of Rho signalling networks identifies RhoH as a regulator of Rac1 in prostate cancer cell migration. BMC Boil. 2018, 16, 29. [Google Scholar] [CrossRef] [PubMed]
- Wennerberg, K.; Forget, M.-A.; Ellerbroek, S.M.; Arthur, W.T.; Burridge, K.; Settleman, J.; Der, C.J.; Hansen, S.H. Rnd Proteins Function as RhoA Antagonists by Activating p190 RhoGAP. Curr. Boil. 2003, 13, 1106–1115. [Google Scholar] [CrossRef]
- Ramos, S.; Khademi, F.; Somesh, B.P.; Rivero, F. Genomic organization and expression profile of the small GTPases of the RhoBTB family in human and mouse. Gene 2002, 298, 147–157. [Google Scholar] [CrossRef]
- Berthold, J.; Schenková, K.; Ramos, S.; Miura, Y.; Furukawa, M.; Aspenström, P.; Rivero, F. Characterization of RhoBTB-dependent Cul3 ubiquitin ligase complexes—Evidence for an autoregulatory mechanism. Exp. Cell Res. 2008, 314, 3453–3465. [Google Scholar] [CrossRef] [PubMed]
- Manjarrez, J.R.; Sun, L.; Prince, T.; Matts, R.L. Hsp90-Dependent Assembly of the DBC2/RhoBTB2-Cullin3 E3-Ligase Complex. PLoS ONE 2014, 9, e90054. [Google Scholar] [CrossRef] [PubMed]
- Ji, W.; Rivero, F. Atypical Rho GTPases of the RhoBTB Subfamily: Roles in Vesicle Trafficking and Tumorigenesis. Cells 2016, 5, 28. [Google Scholar] [CrossRef]
- Fransson, A.; Ruusala, A.; Aspenström, P. Atypical Rho GTPases Have Roles in Mitochondrial Homeostasis and Apoptosis. J. Boil. Chem. 2002, 278, 6495–6502. [Google Scholar] [CrossRef]
- Tang, B.L. MIRO GTPases in Mitochondrial Transport, Homeostasis and Pathology. Cells 2015, 5, 1. [Google Scholar] [CrossRef]
- Cherfils, J.; Zeghouf, M. Regulation of Small GTPases by GEFs, GAPs, and GDIs. Physiol. Rev. 2013, 93, 269–309. [Google Scholar] [CrossRef]
- Laurin, M.; Coté, J.-F. Insights into the biological functions of Dock family guanine nucleotide exchange factors. Genes Dev. 2014, 28, 533–547. [Google Scholar] [CrossRef]
- Fort, P.; Blangy, A. The Evolutionary Landscape of Dbl-Like RhoGEF Families: Adapting Eukaryotic Cells to Environmental Signals. Genome Boil. Evol. 2017, 9, 1471–1486. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Zhang, Z.; Roe, S.M.; Marshall, C.J.; Barford, D. Activation of Rho GTPases by DOCK Exchange Factors Is Mediated by a Nucleotide Sensor. Science 2009, 325, 1398–1402. [Google Scholar] [CrossRef] [PubMed]
- Rossman, K.L.; Der, C.J.; Sondek, J. GEF means go: Turning on RHO GTPases with guanine nucleotide-exchange factors. Nat. Rev. Mol. Cell Boil. 2005, 6, 167–180. [Google Scholar] [CrossRef] [PubMed]
- Worthylake, D.K.; Rossman, K.L.; Sondek, J. Crystal structure of Rac1 in complex with the guanine nucleotide exchange region of Tiam1. Nature 2000, 408, 682–688. [Google Scholar] [CrossRef] [PubMed]
- Cook, D.R.; Rossman, K.L.; Der, C.J. Rho guanine nucleotide exchange factors: Regulators of Rho GTPase activity in development and disease. Oncogene 2013, 33, 4021–4035. [Google Scholar] [CrossRef]
- Vetter, I.R.; Wittinghofer, A. The Guanine Nucleotide-Binding Switch in Three Dimensions. Science 2001, 294, 1299–1304. [Google Scholar] [CrossRef]
- Tcherkezian, J.; Lamarche-Vane, N. Current knowledge of the large RhoGAP family of proteins. Boil. Cell 2007, 99, 67–86. [Google Scholar] [CrossRef]
- Wennerberg, K.; Der, C.J. Rho-family GTPases: It’s not only Rac and Rho (and I like it). J. Cell Sci. 2004, 117, 1301–1312. [Google Scholar] [CrossRef]
- Michaelson, D.; Silletti, J.; Murphy, G.; D’Eustachio, P.; Rush, M.; Philips, M.R. Differential Localization of Rho Gtpases in Live Cells. J. Cell Boil. 2001, 152, 111–126. [Google Scholar] [CrossRef]
- Mitin, N.; Roberts, P.J.; Chenette, E.; Der, C.J. Posttranslational Lipid Modification of Rho Family Small GTPases. Adv. Struct. Saf. Stud. 2011, 827, 87–95. [Google Scholar] [CrossRef]
- DerMardirossian, C.; Bokoch, G.M. GDIs: Central regulatory molecules in Rho GTPase activation. Trends Cell Boil. 2005, 15, 356–363. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, G.R.; Nassar, N.; Cerione, R.A. Structure of the Rho Family GTP-Binding Protein Cdc42 in Complex with the Multifunctional Regulator RhoGDI. Cell 2000, 100, 345–356. [Google Scholar] [CrossRef]
- Longenecker, K.; Read, P.; Derewenda, U.; Dauter, Z.; Liu, X.; Garrard, S.; Walker, L.; Somlyo, A.V.; Nakamoto, R.K.; Somlyo, A.P.; et al. How RhoGDI binds Rho. Acta Crystallogr. Sect. D Boil. Crystallogr. 1999, 55, 1503–1515. [Google Scholar] [CrossRef]
- Grizot, S.; Fauré, J.; Fieschi, F.; Vignais, P.V.; Dagher, M.-C.; Pebay-Peyroula, E. Crystal Structure of the Rac1−RhoGDI Complex Involved in NADPH Oxidase Activation. Biochemistry 2001, 40, 10007–10013. [Google Scholar] [CrossRef] [PubMed]
- Scheffzek, K.; Stephan, I.; Jensen, O.N.; Illenberger, D.; Gierschik, P. The Rac-RhoGDI complex and the structural basis for the regulation of Rho proteins by RhoGDI. Nat. Genet. 2000, 7, 122–126. [Google Scholar] [CrossRef]
- Gosser, Y.Q.; Nomanbhoy, T.K.; Aghazadeh, B.; Manor, D.; Combs, C.; Cerione, R.A.; Rosen, M.K. C-terminal binding domain of Rho GDP-dissociation inhibitor directs N-terminal inhibitory peptide to GTPases. Nature 1997, 387, 814–819. [Google Scholar] [CrossRef]
- Keep, N.H.; Barnes, M.; Barsukov, I.; Badii, R.; Lian, L.-Y.; Segal, A.W.; Moody, P.C.E.; Roberts, G. A modulator of rho family G proteins, rhoGDI, binds these G proteins via an immunoglobulin-like domain and a flexible N-terminal arm. Structure 1997, 5, 623–633. [Google Scholar] [CrossRef]
- Garcia-Mata, R.; Boulter, E.; Burridge, K. The ‘invisible hand’: Regulation of RHO GTPases by RHOGDIs. Nat. Rev. Mol. Cell Boil. 2011, 12, 493–504. [Google Scholar] [CrossRef] [PubMed]
- Boulter, E.; Garcia-Mata, R.; Guilluy, C.; Dubash, A.; Rossi, G.; Brennwald, P.J.; Burridge, K. Regulation of Rho GTPase crosstalk, degradation and activity by RhoGDI1. Nature 2010, 12, 477–483. [Google Scholar] [CrossRef]
- Hancock, J.F.; Hall, A. A novel role for RhoGDI as an inhibitor of GAP proteins. EMBO J. 1993, 12, 1915–1921. [Google Scholar] [CrossRef]
- Hart, M.; Maru, Y.; Leonard, D.; Witte, O.; Evans, T.; Cerione, R. A GDP dissociation inhibitor that serves as a GTPase inhibitor for the Ras-like protein CDC42Hs. Science 1992, 258, 812–815. [Google Scholar] [CrossRef] [PubMed]
- Nomanbhoy, T.K.; Cerione, R.A. Characterization of the Interaction between RhoGDI and Cdc42Hs Using Fluorescence Spectroscopy. J. Boil. Chem. 1996, 271, 10004–10009. [Google Scholar] [CrossRef] [PubMed]
- Tnimov, Z.; Guo, Z.; Gambin, Y.; Nguyen, U.T.T.; Wu, Y.; Abankwa, D.; Stigter, A.; Collins, B.M.; Waldmann, H.; Goody, R.S.; et al. Quantitative Analysis of Prenylated RhoA Interaction with Its Chaperone, RhoGDI*. J. Boil. Chem. 2012, 287, 26549–26562. [Google Scholar] [CrossRef]
- DerMardirossian, C.; Schnelzer, A.; Bokoch, G.M. Phosphorylation of RhoGDI by Pak1 Mediates Dissociation of Rac GTPase. Mol. Cell 2004, 15, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, T.; Kato, M.; Takai, Y. Consequences of weak interaction of rho GDI with the GTP-bound forms of rho p21 and rac p21. J. Boil. Chem. 1993, 268, 23959–23963. [Google Scholar]
- Chuang, T.H.; Xu, X.; Knaus, U.G.; Hart, M.J.; Bokoch, G.M. GDP dissociation inhibitor prevents intrinsic and GTPase activating protein-stimulated GTP hydrolysis by the Rac GTP-binding protein. J. Boil. Chem. 1993, 268, 775–778. [Google Scholar]
- Abo, A.; Webb, M.; Grogan, A.; Segal, A.W. Activation of NADPH oxidase involves the dissociation of p21rac from its inhibitory GDP/GTP exchange protein (rhoGDI) followed by its translocation to the plasma membrane. Biochem. J. 1994, 298, 585–591. [Google Scholar] [CrossRef]
- Perry, J.A.; Maddox, A.S. Uncovering the secret life of Rho GTPases. eLife 2019, 8, e53276. [Google Scholar] [CrossRef]
- Golding, A.E.; Visco, I.; Bieling, P.; Bement, W.M. Extraction of active RhoGTPases by RhoGDI regulates spatiotemporal patterning of RhoGTPases. eLife 2019, 8, e50471. [Google Scholar] [CrossRef]
- Hodge, R.G.; Ridley, A.J. Regulating Rho GTPases and their regulators. Nat. Rev. Mol. Cell Boil. 2016, 17, 496–510. [Google Scholar] [CrossRef] [PubMed]
- Paul, F.; Zauber, H.; Von Berg, L.; Rocks, O.; Daumke, O.; Selbach, M. Quantitative GTPase Affinity Purification Identifies Rho Family Protein Interaction Partners. Mol. Cell. Proteom. 2016, 16, 73–85. [Google Scholar] [CrossRef]
- Roux, K.J.; Kim, D.I.; Raida, M.; Burke, B. A promiscuous biotin ligase fusion protein identifies proximal and interacting proteins in mammalian cells. J. Cell Boil. 2012, 196, 801–810. [Google Scholar] [CrossRef]
- Gingras, A.-C.; Abe, K.T.; Raught, B. Getting to know the neighborhood: Using proximity-dependent biotinylation to characterize protein complexes and map organelles. Curr. Opin. Chem. Boil. 2019, 48, 44–54. [Google Scholar] [CrossRef]
- Bagci, H.; Sriskandarajah, N.; Robert, A.; Boulais, J.; Elkholi, I.E.; Tran, V.; Lin, Z.-Y.; Thibault, M.-P.; Dubé, N.; Faubert, D.; et al. Mapping the proximity interaction network of the Rho-family GTPases reveals signalling pathways and regulatory mechanisms. Nature 2019, 22, 120–134. [Google Scholar] [CrossRef]
- Amin, E.; Jaiswal, M.; Derewenda, U.; Reis, K.; Nouri, K.; Koessmeier, K.T.; Aspenström, P.; Somlyo, A.V.; Dvorsky, R.; Ahmadian, M.R. Deciphering the Molecular and Functional Basis of RHOGAP Family Proteins. J. Boil. Chem. 2016, 291, 20353–20371. [Google Scholar] [CrossRef]
- Pertz, O. Spatio-temporal Rho GTPase signaling - where are we now? J. Cell Sci. 2010, 123, 1841–1850. [Google Scholar] [CrossRef] [PubMed]
- Benink, H.A.; Bement, W.M. Concentric zones of active RhoA and Cdc42 around single cell wounds. J. Cell Boil. 2005, 168, 429–439. [Google Scholar] [CrossRef] [PubMed]
- Machacek, M.; Hodgson, L.; Welch, C.; Elliott, H.; Pertz, O.; Nalbant, P.; Abell, A.; Johnson, G.L.; Hahn, K.M.; Danuser, G. Coordination of Rho GTPase activities during cell protrusion. Nature 2009, 461, 99–103. [Google Scholar] [CrossRef] [PubMed]
- Bravo-Cordero, J.J.; Oser, M.; Chen, X.; Eddy, R.; Hodgson, L.; Condeelis, J. A Novel Spatiotemporal RhoC Activation Pathway Locally Regulates Cofilin Activity at Invadopodia. Curr. Boil. 2011, 21, 635–644. [Google Scholar] [CrossRef] [PubMed]
- Fritz, R.D.; Letzelter, M.; Reimann, A.; Martin, K.; Fusco, L.; Ritsma, L.; Ponsioen, B.; Fluri, E.; Schulte-Merker, S.; Van Rheenen, J.; et al. A Versatile Toolkit to Produce Sensitive FRET Biosensors to Visualize Signaling in Time and Space. Sci. Signal. 2013, 6, rs12. [Google Scholar] [CrossRef] [PubMed]
- Graessl, M.; Koch, J.; Calderon, A.; Kamps, D.; Banerjee, S.; Mazel, T.; Schulze, N.; Jungkurth, J.K.; Patwardhan, R.; Solouk, D.; et al. An excitable Rho GTPase signaling network generates dynamic subcellular contraction patterns. J. Cell Boil. 2017, 216, 4271–4285. [Google Scholar] [CrossRef] [PubMed]
- Hoppe, A.D.; Swanson, J.A. Cdc42, Rac1, and Rac2 Display Distinct Patterns of Activation during Phagocytosis. Mol. Boil. Cell 2004, 15, 3509–3519. [Google Scholar] [CrossRef] [PubMed]
- Müller, P.M.; Rademacher, J.; Bagshaw, R.D.; Wortmann, C.; Barth, C.; Van Unen, J.; Alp, K.M.; Giudice, G.; Eccles, R.L.; Heinrich, L.E.; et al. Systems analysis of RhoGEF and RhoGAP regulatory proteins reveals spatially organized RAC1 signalling from integrin adhesions. Nature 2020, 22, 498–511. [Google Scholar] [CrossRef] [PubMed]
- Martin, K.; Reimann, A.; Fritz, R.D.; Ryu, H.; Jeon, N.L.; Pertz, O. Spatio-temporal co-ordination of RhoA, Rac1 and Cdc42 activation during prototypical edge protrusion and retraction dynamics. Sci. Rep. 2016, 6, 21901. [Google Scholar] [CrossRef]
- Yang, J.; Fan, J.; Li, Y.; Li, F.; Chen, P.; Fan, Y.; Xia, X.; Wong, S.T. Genome-Wide RNAi Screening Identifies Genes Inhibiting the Migration of Glioblastoma Cells. PLoS ONE 2013, 8, e61915. [Google Scholar] [CrossRef]
- Rohn, J.L.; Sims, D.; Liu, T.; Fedorova, M.; Schöck, F.; Dopie, J.; Vartiainen, M.K.; Kiger, A.A.; Perrimon, N.; Baum, B. Comparative RNAi screening identifies a conserved core metazoan actinome by phenotype. J. Cell Boil. 2011, 194, 789–805. [Google Scholar] [CrossRef]
- Bai, S.W.; Herrera-Abreu, M.T.; Rohn, J.L.; Racine, V.; Tajadura-Ortega, V.; Suryavanshi, N.; Bechtel, S.; Wiemann, S.; Baum, B.; Ridley, A.J. Identification and characterization of a set of conserved and new regulators of cytoskeletal organization, cell morphology and migration. BMC Boil. 2011, 9, 54. [Google Scholar] [CrossRef]
- Smolen, G.A.; Zhang, J.; Zubrowski, M.J.; Edelman, E.J.; Luo, B.; Yu, M.; Ng, L.W.; Scherber, C.M.; Schott, B.J.; Ramaswamy, S.; et al. A genome-wide RNAi screen identifies multiple RSK-dependent regulators of cell migration. Genome Res. 2010, 24, 2654–2665. [Google Scholar] [CrossRef]
- Simpson, K.J.; Selfors, L.M.; Bui, J.; Reynolds, A.; Leake, D.; Khvorova, A.; Brugge, J.S. Identification of genes that regulate epithelial cell migration using an siRNA screening approach. Nature 2008, 10, 1027–1038. [Google Scholar] [CrossRef]
- Gadéa, G.; Sanz-Moreno, V.; Self, A.; Godi, A.; Marshall, C.J. DOCK10-Mediated Cdc42 Activation Is Necessary for Amoeboid Invasion of Melanoma Cells. Curr. Boil. 2008, 18, 1456–1465. [Google Scholar] [CrossRef]
- Sanz-Moreno, V.; Gadéa, G.; Ahn, J.; Paterson, H.; Marra, P.; Pinner, S.; Sahai, E.; Marshall, C.J. Rac Activation and Inactivation Control Plasticity of Tumor Cell Movement. Cell 2008, 135, 510–523. [Google Scholar] [CrossRef]
- Sahai, E.; Marshall, C.J. Differing modes of tumour cell invasion have distinct requirements for Rho/ROCK signalling and extracellular proteolysis. Nature 2003, 5, 711–719. [Google Scholar] [CrossRef]
- Friedl, P.; Wolf, K. Tumour-cell invasion and migration: Diversity and escape mechanisms. Nat. Rev. Cancer 2003, 3, 362–374. [Google Scholar] [CrossRef] [PubMed]
- Fingleton, B. MMPs as therapeutic targets-Still a viable option? In Seminars in Cell & Developmental Biology; Academic Press: Cambridge, MA, USA, 2008. [Google Scholar]
- Fields, G.B. The Rebirth of Matrix Metalloproteinase Inhibitors: Moving Beyond the Dogma. Cells 2019, 8, 984. [Google Scholar] [CrossRef] [PubMed]
- Pascual-Vargas, P.; Cooper, S.; Sero, J.E.; Bousgouni, V.; Arias-Garcia, M.; Bakal, C. RNAi screens for Rho GTPase regulators of cell shape and YAP/TAZ localisation in triple negative breast cancer. Sci. Data 2017, 4, 170018. [Google Scholar] [CrossRef] [PubMed]
- Kang, N.; Matsui, T.S.; Liu, S.; Fujiwara, S.; Deguchi, S. Comprehensive analysis on the whole Rho-GAP family reveals that ARHGAP4 suppresses EMT in epithelial cells under negative regulation by Septin9. FASEB J. 2020. [Google Scholar] [CrossRef] [PubMed]
- Amado-Azevedo, J.; De Menezes, R.X.; Amerongen, G.P.V.N.; Van Hinsbergh, V.W.M.; Hordijk, P. A functional siRNA screen identifies RhoGTPase-associated genes involved in thrombin-induced endothelial permeability. PLoS ONE 2018, 13, e0201231. [Google Scholar] [CrossRef]
- Zaritsky, A.; Tseng, Y.-Y.; Rabadan, M.A.; Krishna, S.; Overholtzer, M.; Danuser, G.; Hall, A. Diverse roles of guanine nucleotide exchange factors in regulating collective cell migration. J. Cell Boil. 2017, 216, 1543–1556. [Google Scholar] [CrossRef]
- Duquette, P.M.; Lamarche-Vane, N. Rho GTPases in embryonic development. Small GTPases 2014, 5, 1–9. [Google Scholar] [CrossRef]
- Luxenburg, C.; Zaidel-Bar, R. From cell shape to cell fate via the cytoskeleton—Insights from the epidermis. Exp. Cell Res. 2019, 378, 232–237. [Google Scholar] [CrossRef]
- Niessen, M.T.; Iden, S.; Niessen, C.M. The in vivo function of mammalian cell and tissue polarity regulators—How to shape and maintain the epidermal barrier. J. Cell Sci. 2012, 125, 3501–3510. [Google Scholar] [CrossRef] [PubMed]
- Simpson, C.L.; Patel, D.M.; Green, K.J. Deconstructing the skin: Cytoarchitectural determinants of epidermal morphogenesis. Nat. Rev. Mol. Cell Boil. 2011, 12, 565–580. [Google Scholar] [CrossRef] [PubMed]
- Beronja, S.; Livshits, G.; Williams, S.E.; Fuchs, E. Rapid functional dissection of genetic networks via tissue-specific transduction and RNAi in mouse embryos. Nat. Med. 2010, 16, 821. [Google Scholar] [CrossRef] [PubMed]
- Schramek, D.; Sendoel, A.; Segal, J.P.; Beronja, S.; Heller, E.; Oristian, D.; Reva, B.; Fuchs, E. Direct in Vivo RNAi Screen Unveils Myosin IIa as a Tumor Suppressor of Squamous Cell Carcinomas. Science 2014, 343, 309–313. [Google Scholar] [CrossRef] [PubMed]
- Loganathan, S.K.; Schleicher, K.; Malik, A.; Quevedo, R.; Langille, E.; Teng, K.; Oh, R.H.; Rathod, B.; Tsai, R.; Samavarchi-Tehrani, P.; et al. Rare driver mutations in head and neck squamous cell carcinomas converge on NOTCH signaling. Science 2020, 367, 1264–1269. [Google Scholar] [CrossRef]
- Laurin, M.; Gomez, N.C.; Levorse, J.; Sendoel, A.; Sribour, M.; Fuchs, E. An RNAi screen unravels the complexities of Rho GTPase networks in skin morphogenesis. eLife 2019, 8. [Google Scholar] [CrossRef]
- Sendoel, A.; Dunn, J.G.; Rodriguez, E.H.; Naik, S.; Gomez, N.C.; Hurwitz, B.; Levorse, J.; Dill, B.D.; Schramek, D.; Molina, H.; et al. Translation from unconventional 5′ start sites drives tumour initiation. Nature 2017, 541, 494–499. [Google Scholar] [CrossRef]
- Asare, A.; Levorse, J.; Fuchs, E. Coupling organelle inheritance with mitosis to balance growth and differentiation. Science 2017, 355, eaah4701. [Google Scholar] [CrossRef]
- Ge, Y.; Zhang, L.; Nikolova, M.; Reva, B.; Fuchs, E. Strand-specific in vivo screen of cancer-associated miRNAs unveils a role for miR-21∗ in SCC progression. Nature 2015, 18, 111–121. [Google Scholar] [CrossRef]
- Beronja, S.; Janki, P.; Heller, E.; Lien, W.-H.; Keyes, B.E.; Oshimori, N.; Fuchs, E. RNAi screens in mice identify physiological regulators of oncogenic growth. Nature 2013, 501, 185–190. [Google Scholar] [CrossRef]
- Sennett, R.; Wang, Z.; Rezza, A.; Grisanti, L.; Roitershtein, N.; Sicchio, C.; Mok, K.W.; Heitman, N.; Clavel, C.; Ma’Ayan, A.; et al. An Integrated Transcriptome Atlas of Embryonic Hair Follicle Progenitors, Their Niche, and the Developing Skin. Dev. Cell 2015, 34, 577–591. [Google Scholar] [CrossRef] [PubMed]
- Rezza, A.; Wang, Z.; Sennett, R.; Qiao, W.; Wang, N.; Heitman, N.; Mok, K.W.; Clavel, C.; Yi, R.; Zandstra, P.; et al. Signaling Networks among Stem Cell Precursors, Transit-Amplifying Progenitors, and their Niche in Developing Hair Follicles. Cell Rep. 2016, 14, 3001–3018. [Google Scholar] [CrossRef] [PubMed]
- Devenport, D.; Fuchs, E. Planar polarization in embryonic epidermis orchestrates global asymmetric morphogenesis of hair follicles. Nature 2008, 10, 1257–1268. [Google Scholar] [CrossRef] [PubMed]
- Luxenburg, C.; Heller, E.; Pasolli, H.A.; Chai, S.; Nikolova, M.; Stokes, N.; Fuchs, E. Wdr1-mediated cell shape dynamics and cortical tension are essential for epidermal planar cell polarity. Nature 2015, 17, 592–604. [Google Scholar] [CrossRef] [PubMed]
- Ahtiainen, L.; Lefebvre, S.; Lindfors, P.H.; Renvoisé, E.; Shirokova, V.; Vartiainen, M.K.; Thesleff, I.; Mikkola, M.L. Directional Cell Migration, but Not Proliferation, Drives Hair Placode Morphogenesis. Dev. Cell 2014, 28, 588–602. [Google Scholar] [CrossRef] [PubMed]
- Le, H.; Ghatak, S.; Yeung, C.-Y.C.; Tellkamp, F.; Günschmann, C.; Dieterich, C.; Yeroslaviz, A.; Habermann, B.; Pombo, A.; Niessen, C.M.; et al. Mechanical regulation of transcription controls Polycomb-mediated gene silencing during lineage commitment. Nature 2016, 18, 864–875. [Google Scholar] [CrossRef]
- Aw, W.Y.; Heck, B.W.; Joyce, B.; Devenport, D. Transient Tissue-Scale Deformation Coordinates Alignment of Planar Cell Polarity Junctions in the Mammalian Skin. Curr. Boil. 2016, 26, 2090–2100. [Google Scholar] [CrossRef]
- Rhee, H.-W.; Zou, P.; Udeshi, N.D.; Martell, J.D.; Mootha, V.K.; Carr, S.A.; Ting, A.Y. Proteomic Mapping of Mitochondria in Living Cells via Spatially Restricted Enzymatic Tagging. Science 2013, 339, 1328–1331. [Google Scholar] [CrossRef]
- Branon, T.C.; Bosch, J.A.; Sanchez, A.D.; Udeshi, N.D.; Svinkina, T.; Carr, S.A.; Feldman, J.L.; Perrimon, N.; Ting, A.Y. Efficient proximity labeling in living cells and organisms with TurboID. Nat. Biotechnol. 2018, 36, 880–887. [Google Scholar] [CrossRef]
- Doerr, A. Proximity labeling with TurboID. Nat. Methods 2018, 15, 764. [Google Scholar] [CrossRef]
- Vigil, M.; Cherfils, J.; Rossman, K.L.; Der, C.J. Ras superfamily GEFs and GAPs: Validated and tractable targets for cancer therapy? Nat. Rev. Cancer 2010, 10, 842–857. [Google Scholar] [CrossRef] [PubMed]
- Barrio-Real, L.; Kazanietz, M.G. Rho GEFs and Cancer: Linking Gene Expression and Metastatic Dissemination. Sci. Signal. 2012, 5, pe43. [Google Scholar] [CrossRef] [PubMed]
- Fritz, G.; Brachetti, C.; Bahlmann, F.; Schmidt, M.; Kaina, B. Rho GTPases in human breast tumours: Expression and mutation analyses and correlation with clinical parameters. Br. J. Cancer 2002, 87, 635–644. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wang, Y.; Zhang, Y.; Miao, Y.; Zhao, Y.; Zhang, P.-X.; Jiang, G.; Zhang, J.-Y.; Han, Y.; Lin, X.-Y.; et al. Abnormal expression of p120-catenin, E-cadherin, and small GTPases is significantly associated with malignant phenotype of human lung cancer. Lung Cancer 2009, 63, 375–382. [Google Scholar] [CrossRef]
- Arias-Romero, L.E.; Chernoff, J. Targeting Cdc42 in cancer. Expert Opin. Ther. Targets 2013, 17, 1263–1273. [Google Scholar] [CrossRef]
- Ji, J.; Feng, X.; Shi, M.; Cai, Q.; Yu, Y.; Zhu, Z.; Zhang, J. Rac1 is correlated with aggressiveness and a potential therapeutic target for gastric cancer. Int. J. Oncol. 2015, 46, 1343–1353. [Google Scholar] [CrossRef]
- Fritz, G.; Just, I.; Kaina, B. Rho GTPases are over-expressed in human tumors. Int. J. Cancer 1999, 81, 682–687. [Google Scholar] [CrossRef]
- Pan, Y.; Bi, F.; Liu, N.; Xue, Y.; Yao, X.; Zheng, Y.; Fan, D. Expression of seven main Rho family members in gastric carcinoma. Biochem. Biophys. Res. Commun. 2004, 315, 686–691. [Google Scholar] [CrossRef]
- Kamai, T.; Yamanishi, T.; Shirataki, H.; Takagi, K.; Asami, H.; Ito, Y.; Yoshida, K.-I. Overexpression of RhoA, Rac1, and Cdc42 GTPases Is Associated with Progression in Testicular Cancer. Clin. Cancer Res. 2004, 10, 4799–4805. [Google Scholar] [CrossRef]
- Mazieres, J.; Antonia, T.; Daste, G.; Muro-Cacho, C.; Berchery, D.; Tillement, V.; Pradines, A.; Sebti, S.; Favre, G. Loss of RhoB expression in human lung cancer progression. Clin. Cancer Res. 2004, 10, 2742–2750. [Google Scholar] [CrossRef]
- Engers, R.; Ziegler, S.; Mueller, M.; Walter, A.; Willers, R.; E Gabbert, H. Prognostic relevance of increased Rac GTPase expression in prostate carcinomas. Endocr. Relat. Cancer 2007, 14, 245–256. [Google Scholar] [CrossRef] [PubMed]
- Sato, N.; Fukui, T.; Taniguchi, T.; Yokoyama, T.; Kondo, M.; Nagasaka, T.; Goto, Y.; Gao, W.; Ueda, Y.; Yokoi, K.; et al. RhoB is frequently downregulated in non-small-cell lung cancer and resides in the 2p24 homozygous deletion region of a lung cancer cell line. Int. J. Cancer 2006, 120, 543–551. [Google Scholar] [CrossRef] [PubMed]
- Tucci, M.; Lucarini, G.; Brancorsini, D.; Zizzi, A.; Pugnaloni, A.; Giacchetti, A.; Ricotti, G.; Biagini, G. Involvement of E-cadherin, β-catenin, Cdc42 and CXCR4 in the progression and prognosis of cutaneous melanoma. Br. J. Dermatol. 2007, 157, 1212–1216. [Google Scholar] [CrossRef] [PubMed]
- Faried, A.; Faried, L.S.; Usman, N.; Kato, H.; Kuwano, H. Clinical and Prognostic Significance of RhoA and RhoC Gene Expression in Esophageal Squamous Cell Carcinoma. Ann. Surg. Oncol. 2007, 14, 3593–3601. [Google Scholar] [CrossRef]
- Del Pulgar, T.G.; Valdés-Mora, F.; Bandrés, E.; Pérez-Palacios, R.; Espina, C.; Cejas, P.; García-Cabezas, M.Á.; Nistal, M.; Casado, E.; González-Barón, M.; et al. Cdc42 is highly expressed in colorectal adenocarcinoma and downregulates ID4 through an epigenetic mechanism. Int. J. Oncol. 2008, 33, 185–193. [Google Scholar] [CrossRef]
- Krauthammer, M.; Kong, Y.; Ha, B.H.; Evans, P.; Bacchiocchi, A.; McCusker, J.P.; Cheng, E.; Davis, M.J.; Goh, G.; Choi, M.; et al. Exome sequencing identifies recurrent somatic RAC1 mutations in melanoma. Nat. Genet. 2012, 44, 1006–1014. [Google Scholar] [CrossRef]
- Davis, M.J.; Ha, B.H.; Holman, E.; Halaban, R.; Schlessinger, J.; Boggon, T.J. RAC1P29S is a spontaneously activating cancer-associated GTPase. Proc. Natl. Acad. Sci. USA 2013, 110, 912–917. [Google Scholar] [CrossRef]
- Kawazu, M.; Ueno, T.; Kontani, K.; Ogita, Y.; Ando, M.; Fukumura, K.; Yamato, A.; Soda, M.; Takeuchi, K.; Miki, Y.; et al. Transforming mutations of RAC guanosine triphosphatases in human cancers. Proc. Natl. Acad. Sci. USA 2013, 110, 3029–3034. [Google Scholar] [CrossRef]
- Kakiuchi, M.; Nishizawa, T.; Ueda, H.; Gotoh, K.; Tanaka, A.; Hayashi, A.; Yamamoto, S.; Tatsuno, K.; Katoh, H.; Watanabe, Y.; et al. Recurrent gain-of-function mutations of RHOA in diffuse-type gastric carcinoma. Nat. Genet. 2014, 46, 583–587. [Google Scholar] [CrossRef]
- Manso, R.; Sánchez-Beato, M.; Monsalvo, S.; Gómez, S.; Cereceda, L.; Llamas, P.; Rojo, F.; Mollejo, M.; Menárguez, J.; Alves, J.; et al. The RHOA G17V gene mutation occurs frequently in peripheral T-cell lymphoma and is associated with a characteristic molecular signature. Blood 2014, 123, 2893–2894. [Google Scholar] [CrossRef]
- Mar, V.J.; Wong, S.Q.; Logan, A.; Nguyen, T.; Cebon, J.; Kelly, J.W.; Wolfe, R.; Dobrovic, A.; McLean, C.; McArthur, G. Clinical and pathological associations of the activatingRAC1P29S mutation in primary cutaneous melanoma. Pigment. Cell Melanoma Res. 2014, 27, 1117–1125. [Google Scholar] [CrossRef] [PubMed]
- Palomero, T.; Couronné, L.; Khiabanian, H.; Kim, M.-Y.; Ambesi-Impiombato, A.; Perez-Garcia, A.; Carpenter, Z.; Abate, F.; Allegretta, M.; Haydu, J.E.; et al. Recurrent mutations in epigenetic regulators, RHOA and FYN kinase in peripheral T cell lymphomas. Nat. Genet. 2014, 46, 166–170. [Google Scholar] [CrossRef] [PubMed]
- Yoo, H.Y.; Sung, M.; Lee, S.H.; Kim, S.; Lee, H.; Park, S.; Kim, S.C.; Lee, B.; Rho, K.; Lee, W.T.; et al. A recurrent inactivating mutation in RHOA GTPase in angioimmunoblastic T cell lymphoma. Nat. Genet. 2014, 46, 371–375. [Google Scholar] [CrossRef] [PubMed]
- Vu, H.L.; Rosenbaum, S.; Purwin, T.J.; Davies, M.A.; Aplin, A.E. RAC1 P29S regulates PD-L1 expression in melanoma. Pigment. Cell Melanoma Res. 2015, 28, 590–598. [Google Scholar] [CrossRef] [PubMed]
- Sahai, E.; Alberts, A.S.; Treisman, R. RhoA effector mutants reveal distinct effector pathways for cytoskeletal reorganization, SRF activation and transformation. EMBO J. 1998, 17, 1350–1361. [Google Scholar] [CrossRef]
| Approach | Goal | Cell Types | Reference |
|---|---|---|---|
| qGAP | Identify Rho GTPases binding partners | HEK293 Mouse brain lysate | [61] |
| BioID | Define Rho family proximity interactome | HEK293 HeLa | [64] |
| Flag-IP | Define RhoGEFs and RhoGAPs interactome | HEK293 | [73] |
| In vitro siRNA screen | Identify RhoGEFs required for amoeboid movement in melanoma cells | A375M2 | [80] |
| In vitro siRNA screen | Identify RhoGEFs that regulate amoeboid and mesenchymal features | A375M2 | [81] |
| In vitro siRNA screen | Investigate the role of Rho GTPases network components during prostate cancer cell migration | PC3 | [21] |
| In vitro siRNA screen | Identify RhoGEFs and RhoGAPs that regulate breast cancer cell morphology | LM2 MDA-MB-231 | [86] |
| In vitro siRNA screen | Identify RhoGAPs that contribute to EMT | MCF10A | [87] |
| In vitro siRNA screen | Identify regulators of the endothelial barrier among Rho GTPases network components. | HUVEC | [88] |
| In vitro shRNA screen | Identify RhoGEFs regulating collective migration | 16HBE14o | [89] |
| In vivo shRNA morphogenesis screen | Identify regulators of skin morphogenesis among Rho GTPases network components | Mouse embryos Primary mouse keratinocytes | [97] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dahmene, M.; Quirion, L.; Laurin, M. High Throughput strategies Aimed at Closing the GAP in Our Knowledge of Rho GTPase Signaling. Cells 2020, 9, 1430. https://doi.org/10.3390/cells9061430
Dahmene M, Quirion L, Laurin M. High Throughput strategies Aimed at Closing the GAP in Our Knowledge of Rho GTPase Signaling. Cells. 2020; 9(6):1430. https://doi.org/10.3390/cells9061430
Chicago/Turabian StyleDahmene, Manel, Laura Quirion, and Mélanie Laurin. 2020. "High Throughput strategies Aimed at Closing the GAP in Our Knowledge of Rho GTPase Signaling" Cells 9, no. 6: 1430. https://doi.org/10.3390/cells9061430
APA StyleDahmene, M., Quirion, L., & Laurin, M. (2020). High Throughput strategies Aimed at Closing the GAP in Our Knowledge of Rho GTPase Signaling. Cells, 9(6), 1430. https://doi.org/10.3390/cells9061430





