Matrix Metalloproteinases as Potential Biomarkers and Therapeutic Targets in Liver Diseases
Abstract
1. Introduction
2. Pathogenesis of Liver Diseases
3. Matrix Metalloproteinases (MMPs) and Their Role in Liver Disease
4. MMPs as Therapeutic Targets
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Junqueira, L.C.; Carneiro, J.; Wisse, E. Functionele Histologie; Elsevier Gezondheidszorg: Amsterdam, The Netherlands, 2010. [Google Scholar]
- Saile, B.; Ramadori, G. Inflammation, damage repair and liver fibrosis—Role of cytokines and different cell types. Z Gastroenterol. 2007, 45, 77–86. [Google Scholar] [CrossRef] [PubMed]
- Bedossa, P.; Paradis, V. Liver extracellular matrix in health and disease. J. Pathol. 2003, 200, 504–515. [Google Scholar] [CrossRef] [PubMed]
- Duarte, S.; Baber, J.; Fujii, T.; Coito, A.J. Matrix metalloproteinases in liver injury, repair and fibrosis. Matrix Biol. 2015, 44–46, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Omar, R.; Yang, J.; Liu, H.; Davies, N.M.; Gong, Y.-W.; Omar, R.; Yang, J.; Liu, H.; Davies, N.M.; Gong, Y.-W. Hepatic Stellate Cells in Liver Fibrosis and siRNA-Based Therapy. Rev Physiol Biochem Pharmacol. 2016, 172, 1–37. [Google Scholar]
- Puche, J.E.; Saiman, Y.; Friedman, S.L. Hepatic Stellate Cells and Liver Fibrosis. Compr. Physiol. 2013, 3, 1473–1492. [Google Scholar]
- Roderfeld, M. Matrix metalloproteinase functions in hepatic injury and fibrosis. Matrix Biol. 2018, 68–69, 452–462. [Google Scholar] [CrossRef]
- Naim, A.; Pan, Q.; Baig, M.S. Matrix Metalloproteinases (MMPs) in Liver Diseases. J. Clin. Exp. Hepatol. 2017, 7, 367–372. [Google Scholar] [CrossRef]
- Wilde, B.; Katsounas, A. Immune Dysfunction and Albumin-Related Immunity in Liver Cirrhosis. Mediat. Inflamm. 2019, 2019, 7537649. [Google Scholar] [CrossRef]
- Wiegand, J.; Berg, T. The Etiology, Diagnosis and Prevention of Liver Cirrhosis. Dtsch. Aerztebl. Int. 2013, 110, 85–91. [Google Scholar] [CrossRef]
- Martínez, A.K.; Maroni, L.; Marzioni, M.; Ahmed, S.T.; Milad, M.; Ray, D.; Alpini, G.; Glaser, S.S. Mouse Models of Liver Fibrosis Mimic Human Liver Fibrosis of Different Etiologies. Curr. Pathobiol. Rep. 2014, 2, 143–153. [Google Scholar] [CrossRef]
- Sharma, P.; Arora, A. Clinical presentation of alcoholic liver disease and non-alcoholic fatty liver disease: Spectrum and diagnosis. Transl. Gastroenterol. Hepatol. 2020, 5, 19. [Google Scholar] [CrossRef]
- Böttcher, K.; Pinzani, M. Pathophysiology of liver fibrosis and the methodological barriers to the development of anti-fibrogenic agents. Adv. Drug Deliv. Rev. 2017, 121, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Tsuchida, T.; Friedman, S.L. Mechanisms of hepatic stellate cell activation. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 397–411. [Google Scholar] [CrossRef] [PubMed]
- Arriazu, E.; De Galarreta, M.R.; Cubero, F.J.; Varela-Rey, M.; De Obanos, M.P.P.; Leung, T.M.; Lopategi, A.; Benedicto, A.; Abraham-Enachescu, I.; Nieto, N. Extracellular Matrix and Liver Disease. Antioxid. Redox Signal. 2014, 21, 1078–1097. [Google Scholar] [CrossRef] [PubMed]
- Abdu, S.B.; Al-, F.M. Portal Fibroblast Role in Liver Fibrosis in Rats. Int. J. Pharmacol. 2018, 14, 717–726. [Google Scholar] [CrossRef]
- Karin, D.; Koyama, Y.; Brenner, D.A.; Kisseleva, T. The characteristics of activated portal fibroblasts/myofibroblasts in liver fibrosis. Differentiation 2016, 92, 84–92. [Google Scholar] [CrossRef]
- Schuppan, D.; Afdhal, N.H. Liver Cirrhosis. Lancet 2008, 371, 838–851. [Google Scholar] [CrossRef]
- Schilter, H.; Findlay, A.D.; Perryman, L.; Yow, T.T.; Moses, J.; Zahoor, A.; Turner, C.I.; Deodhar, M.; Foot, J.S.; Zhou, W. The lysyl oxidase like 2/3 enzymatic inhibitor, PXS-5153A, reduces crosslinks and ameliorates fibrosis. J. Cell. Mol. Med. 2019, 23, 1759–1770. [Google Scholar] [CrossRef]
- Sprengers, D.; Janssen, H.L.A. Immunomodulatory therapy for chronic hepatitis B virus infection. Fundam. Clin. Pharmacol. 2005, 19, 17–26. [Google Scholar] [CrossRef]
- Forde, K.A.; Reddy, K.R. Hepatitis C Virus Infection and Immunomodulatory Therapies. Clin. Liver Dis. 2009, 13, 391–401. [Google Scholar] [CrossRef]
- Blachier, M.; Leleu, H.; Peck-Radosavljevic, M.; Valla, D.; Roudot-Thoraval, F. The burden of liver disease in Europe: A review of available epidemiological data. J. Hepatol. 2013, 58, 593–608. [Google Scholar] [CrossRef] [PubMed]
- Bansal, R.; Nagórniewicz, B.; Prakash, J. Clinical Advancements in the Targeted Therapies against Liver Fibrosis. Mediat. Inflamm. 2016, 2016, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, D.; Ditah, I.C.; Saeian, K.; Lalehzari, M.; Aronsohn, A.; Gorospe, E.C.; Charlton, M. Changes in the Prevalence of Hepatitis C Virus Infection, Nonalcoholic Steatohepatitis, and Alcoholic Liver Disease Among Patients with Cirrhosis or Liver Failure on the Waitlist for Liver Transplantation. Gastroenterology 2017, 152, 1090–1099. [Google Scholar] [CrossRef] [PubMed]
- Du, C.; Jiang, M.; Wei, X.; Qin, J.; Xu, H.; Wang, Y.; Zhang, Y.; Zhou, D.; Xue, H.; Zheng, S.; et al. Transplantation of human matrix metalloproteinase-1 gene-modified bone marrow-derived mesenchymal stem cell attenuates CCL4-induced liver fibrosis in rats. Int. J. Mol. Med. 2018, 41, 3175–3184. [Google Scholar] [CrossRef]
- Fonovic, M.; Turk, B. Cysteine cathepsins and extracellular matrix degradation. Biochim. Biophys. Acta 2014, 1840, 2560–2570. [Google Scholar] [CrossRef]
- Binder, M.J.; McCoombe, S.; Williams, E.D.; McCulloch, D.; Ward, A. The extracellular matrix in cancer progression: Role of hyalectan proteoglycans and ADAMTS enzymes. Cancer Lett. 2017, 385, 55–64. [Google Scholar] [CrossRef]
- Lu, P.; Takai, K.; Weaver, V.M.; Werb, Z. Extracellular Matrix Degradation and Remodeling in Development and Disease. Cold Spring Harb. Perspect. Biol. 2011, 3, a005058. [Google Scholar] [CrossRef]
- Sternlicht, M.D.; Werb, Z. How Matrix Metalloproteinases Regulate Cell Behavior. Annu. Rev. Cell Dev. Biol. 2001, 17, 463–516. [Google Scholar] [CrossRef]
- Flannery, C.R. MMPs and ADAMTSs: Functional studies. Front. Biosci. 2006, 11, 544569. [Google Scholar] [CrossRef]
- Gross, J.; Lapière, C.M. Collagenolytic Activity in Amphibian Tissues: A Tissue Culture Assay. Proc. Natl. Acad. Sci. USA 1962, 48, 1014–1022. [Google Scholar] [CrossRef]
- Löffek, S.; Schilling, O.; Franzke, C.W. Series ‘matrix metalloproteinases in lung health and disease’ edited by J. Müller-Quernheim and O. Eickelberg number 1 in this series: Biological role of matrix metalloproteinases: A critical balance. Eur. Respir. J. 2011, 38, 191–208. [Google Scholar] [CrossRef] [PubMed]
- Chuang, H.-M.; Chen, Y.-S.; Harn, H.-J. The Versatile Role of Matrix Metalloproteinase for the Diverse Results of Fibrosis Treatment. Molecules 2019, 24, 4188. [Google Scholar] [CrossRef] [PubMed]
- Madzharova, E.; Kastl, P.; Sabino, F.M.R.; Keller, U.A.D. Post-Translational Modification-Dependent Activity of Matrix Metalloproteinases. Int. J. Mol. Sci. 2019, 20, 3077. [Google Scholar] [CrossRef] [PubMed]
- Cui, N.; Hu, M.; Khalil, R.A. Biochemical and Biological Attributes of Matrix Metalloproteinases. Prog Mol Biol Transl Sci. 2017, 147, 1–73. [Google Scholar] [PubMed]
- Consolo, M.; Amoroso, A.; Spandidos, D.A.; Mazzarino, M.C. Matrix metalloproteinases and their inhibitors as markers of inflammation and fibrosis in chronic liver disease (Review). Int. J. Mol. Med. 2009, 24, 143–152. [Google Scholar] [PubMed]
- Iredale, J. Tissue inhibitors of metalloproteinases in liver fibrosis. Int. J. Biochem. Cell Biol. 1997, 29, 43–54. [Google Scholar] [CrossRef]
- Roeb, E. Matrix metalloproteinases and liver fibrosis (translational aspects). Matrix Biol. 2018, 68–69, 463–473. [Google Scholar] [CrossRef]
- Murawaki, Y.; Ikuta, Y.; Idobe, Y.; Kawasaki, H. Serum matrix metalloproteinase-1 in patients with chronic viral hepatitis. J. Gastroenterol. Hepatol. 2002, 14, 138–145. [Google Scholar] [CrossRef]
- Attallah, A.; El-Far, M.; Malak, C.A.A.; Omran, M.M.; Farid, K.; Hussien, M.A.; Albannan, M.; Attallah, A.; Elbendary, M.S.; Elbesh, D.A.; et al. Fibro-check: A combination of direct and indirect markers for liver fibrosis staging in chronic hepatitis C patients. Ann. Hepatol. 2015, 14, 225–233. [Google Scholar] [CrossRef]
- Ando, W.; Yokomori, H.; Tsutsui, N.; Yamanouchi, E.; Suzuki, Y.; Oda, M.; Inagaki, Y.; Otori, K.; Okazaki, I. Serum matrix metalloproteinase-1 level represents disease activity as opposed to fibrosis in patients with histologically proven nonalcoholic steatohepatitis. Clin. Mol. Hepatol. 2018, 24, 61–76. [Google Scholar] [CrossRef]
- Zhou, Z.; Ma, X.; Wang, F.-M.; Sun, L.; Zhang, G. A Matrix Metalloproteinase-1 Polymorphism, MMP1–1607 (1G>2G), Is Associated with Increased Cancer Risk: A Meta-Analysis Including 21,327 Patients. Dis. Markers 2018, 2018, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Iimuro, Y.; Nishio, T.; Morimoto, T.; Nitta, T.; Stefanovic, B.; Choi, S.K.; Brenner, D.A.; Yamaoka, Y. Delivery of matrix metalloproteinase-1 attenuates established liver fibrosis in the rat. Gastroenterol. 2003, 124, 445–458. [Google Scholar] [CrossRef] [PubMed]
- Takahara, T.; Furui, K.; Yata, Y.; Jin, B.; Zhang, L.P.; Nambu, S.; Sato, H.; Seiki, M.; Watanabe, A. Dual expression of matrix metalloproteinase-2 and membrane-type 1-matrix metalloproteinase in fibrotic human livers. Hepatology 1997, 26, 1521–1599. [Google Scholar] [CrossRef] [PubMed]
- Lichtinghagen, R.; Bahr, M.J.; Wehmeier, M.; Michels, D.; Haberkorn, C.I.; Arndt, B.; Flemming, P.; Manns, M.P.; Boeker, K.H.W. Expression and coordinated regulation of matrix metalloproteinases in chronic hepatitis C and hepatitis C virus-induced liver cirrhosis. Clin. Sci. 2003, 105, 373–382. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.-C.; Chuang, J.-H.; Chou, M.-H.; Wu, C.-L.; Chen, C.-M.; Wang, C.-C.; Chen, Y.-S.; Chen, C.-L.; Tai, M.-H. Matrilysin (MMP-7) is a major matrix metalloproteinase upregulated in biliary atresia-associated liver fibrosis. Mod. Pathol. 2005, 18, 941–950. [Google Scholar] [CrossRef]
- Cursio, R.; Mari, B.; Louis, K.; Rostagno, P.; Saint-Paul, M.-C.; Giudicelli, J.; Bottero, V.; Anglard, P.; Yiotakis, A.; Dive, V.; et al. Rat liver injury following normothermic ischemia is prevented by a phosphinic matrix metalloproteinase inhibitor. FASEB J. 2002, 16, 93–95. [Google Scholar] [CrossRef]
- Prystupa, A.; Boguszewska-Czubara, A.; Bojarska-Junak, A.; Toruń-Jurkowska, A.; Roliński, J.; Załuska, W.; Załuska, W. Activity of MMP-2, MMP-8 and MMP-9 in serum as a marker of progression of alcoholic liver disease in people from Lublin Region, eastern Poland. Ann. Agric. Environ. Med. 2015, 22, 325–328. [Google Scholar] [CrossRef]
- Radbill, B.D.; Gupta, R.; Ramirez, M.C.M.; DiFeo, A.; Martignetti, J.A.; Alvarez, C.E.; Friedman, S.L.; Narla, G.; Vrabie, R.; Bowles, R.; et al. Loss of Matrix Metalloproteinase-2 Amplifies Murine Toxin-Induced Liver Fibrosis by Upregulating Collagen I Expression. Dig. Dis. Sci. 2011, 56, 406–416. [Google Scholar] [CrossRef]
- Onozuka, I.; Kakinuma, S.; Kamiya, A.; Miyoshi, M.; Sakamoto, N.; Kiyohashi, K.; Watanabe, T.; Funaoka, Y.; Ueyama, M.; Nakagawa, M.; et al. Cholestatic liver fibrosis and toxin-induced fibrosis are exacerbated in matrix metalloproteinase-2 deficient mice. Biochem. Biophys. Res. Commun. 2011, 406, 134–140. [Google Scholar] [CrossRef]
- Bodey, B.; Bodey, B., Jr.; Siegel, S.E.; Kaiser, H.E. Immunocytochemical detection of MMP-3 and -10 expression in hepatocellular carcinomas. Anticancer. Res. 2000, 20, 4585–4590. [Google Scholar]
- Okamoto, K.; Mandai, M.; Mimura, K.; Murawaki, Y.; Yuasa, I. The association of MMP-1, -3 and -9 genotypes with the prognosis of HCV-related hepatocellular carcinoma patients. Res. Commun. Mol. Pathol. Pharmacol. 2005, 117-118, 77–89. [Google Scholar] [PubMed]
- Okamoto, K.; Mimura, K.; Murawaki, Y.; Yuasa, I. Association of functional gene polymorphisms of matrix metalloproteinase (MMP)-1, MMP-3 and MMP-9 with the progression of chronic liver disease. J. Gastroenterol. Hepatol. 2005, 20, 1102–1108. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, K.; Ishida, C.; Ikebuchi, Y.; Mandai, M.; Mimura, K.; Murawaki, Y.; Yuasa, I. The genotypes of IL-1 beta and MMP-3 are associated with the prognosis of HCV-related hepatocellular carcinoma. Intern. Med. 2010, 49, 887–895. [Google Scholar] [CrossRef] [PubMed]
- Juran, B.D.; Atkinson, E.J.; Schlicht, E.M.; Larson, J.J.; Ellinghaus, D.; Franke, A.; Lazaridis, K.N. Genetic polymorphisms of matrix metalloproteinase 3 in primary sclerosing cholangitis. Liver Int. 2010, 31, 785–791. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Dai, C.; Zhu, H.; Chen, S.; Wu, Y.; Li, Q.; Zeng, X.; Wang, W.; Zuo, J.; Zhou, M.; et al. Cyclophilin A promotes human hepatocellular carcinoma cell metastasis via regulation of MMP3 and MMP9. Mol. Cell. Biochem. 2011, 357, 387–395. [Google Scholar] [CrossRef]
- Jeong, W.-I.; Do, S.-H.; Jeong, D.-H.; Hong, I.-H.; Park, J.-K.; Ran, K.-M.; Yang, H.-J.; Yuan, D.-W.; Kim, S.-B.; Cha, M.-S.; et al. Kinetics of MMP-1 and MMP-3 produced by mast cells and macrophages in liver fibrogenesis of rat. Anticancer. Res. 2006, 26, 3517–3526. [Google Scholar]
- Kerola, A.; Lampela, H.; Lohi, J.; Heikkilä, P.; Mutanen, A.; Hagström, J.; Tervahartiala, T.; Sorsa, T.; Haglund, C.; Jalanko, H.; et al. Increased MMP-7 expression in biliary epithelium and serum underpins native liver fibrosis after successful portoenterostomy in biliary atresia. J. Pathol. Clin. Res. 2016, 2, 187–198. [Google Scholar] [CrossRef]
- Zeng, Z.; Shu, W.-P.; Cohen, A.M.; Guillem, J.G. Matrix metalloproteinase-7 expression in colorectal cancer liver metastases: Evidence for involvement of MMP-7 activation in human cancer metastases. Clin. Cancer Res. 2002, 8, 144–148. [Google Scholar]
- Chiu, C.-C.; Sheu, J.-C.; Chen, C.-H.; Lee, C.-Z.; Chiou, L.-L.; Chou, S.-H.; Huang, G.-T.; Lee, H.-S. Global gene expression profiling reveals a key role of CD44 in hepatic oval-cell reaction after 2-AAF/CCl4 injury in rodents. Histochem. Cell Boil. 2009, 132, 479–489. [Google Scholar] [CrossRef]
- Harty, M.W.; Huddleston, H.M.; Papa, E.F.; Puthawala, T.; Tracy, A.P.; Ramm, G.A.; Gehring, S.; Gregory, S.H.; Tracy, T.F. Repair after cholestatic liver injury correlates with neutrophil infiltration and matrix metalloproteinase 8 activity. Surgery 2005, 138, 313–320. [Google Scholar] [CrossRef]
- Liu, J.; Li, J.; Fu, W.; Tang, J.; Feng, X.; Chen, J.; Liang, Y.; Jin, R.; Xie, A.; Cai, X. Adenoviral delivery of truncated MMP-8 fused with the hepatocyte growth factor mutant 1K1 ameliorates liver cirrhosis and promotes hepatocyte proliferation. Drug Des. Dev. Ther. 2015, 9, 5655–5667. [Google Scholar] [CrossRef] [PubMed]
- Siller-López, F.; Sandoval, A.; Salgado, S.; Salazar, A.; Bueno, M.; García, J.; Cañaveral, I.H.; Gálvez, J.; Hernandez, I.; Ramos, M. Treatment with human metalloproteinase-8 gene delivery ameliorates experimental rat liver cirrhosis. Gastroenterology 2004, 126, 1122–1133. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Bañuelos, J.; Siller-Lopez, F.; Miranda, A.; Aguilar, L.K.; Aguilar-Cordova, E.; Armendariz-Borunda, J. Cirrhotic rat livers with extensive fibrosis can be safely transduced with clinical-grade adenoviral vectors. Evidence of cirrhosis reversion. Gene Ther. 2002, 9, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Van Lint, P.; Wielockx, B.; Puimège, L.; Noel, A.; Lopez-Otin, C.; Libert, C. Resistance of collagenase-2 (matrix metalloproteinase-8)-deficient mice to TNF-induced lethal hepatitis. J. Immunol. 2005, 175, 7642–7649. [Google Scholar] [CrossRef]
- Qin, G.; Luo, M.; Chen, J.; Dang, Y.; Chen, G.; Li, L.; Zeng, J.; Lu, Y.; Yang, J. Reciprocal activation between MMP-8 and TGF-β1 stimulates EMT and malignant progression of hepatocellular carcinoma. Cancer Lett. 2016, 374, 85–95. [Google Scholar] [CrossRef]
- Moore, C.; Shen, X.-D.; Gao, F.; Busuttil, R.W.; Coito, A.J. Fibronectin-α4β1 Integrin Interactions Regulate Metalloproteinase-9 Expression in Steatotic Liver Ischemia and Reperfusion Injury. Am. J. Pathol. 2007, 170, 567–577. [Google Scholar] [CrossRef]
- Hamada, T.; Duarte, S.; Tsuchihashi, S.; Busuttil, R.W.; Coito, A.J. Inducible Nitric Oxide Synthase Deficiency Impairs Matrix Metalloproteinase-9 Activity and Disrupts Leukocyte Migration in Hepatic Ischemia/Reperfusion Injury. Am. J. Pathol. 2009, 174, 2265–2277. [Google Scholar] [CrossRef]
- Hamada, T.; Fondevila, C.; Busuttil, R.W.; Coito, A.J. Metalloproteinase-9 deficiency protects against hepatic ischemia/reperfusion injury. Hepatology 2007, 47, 186–198. [Google Scholar] [CrossRef]
- Kato, H.; Kuriyama, N.; Duarte, S.; Clavien, P.-A.; Busuttil, R.W.; Coito, A.J. MMP-9 deficiency shelters endothelial PECAM-1 expression and enhances regeneration of steatotic livers after ischemia and reperfusion injury. J. Hepatol. 2014, 60, 1032–1039. [Google Scholar] [CrossRef]
- Wang, X.; Maretti-Mira, A.C.; Wang, L.; Deleve, L.D. Liver-Selective MMP-9 Inhibition in the Rat Eliminates Ischemia-Reperfusion Injury and Accelerates Liver Regeneration. Hepatology 2019, 69, 314–328. [Google Scholar] [CrossRef]
- Arii, S.; Mise, M.; Harada, T.; Furutani, M.; Ishigami, S.; Niwano, M.; Mizumoto, M.; Fukumoto, M.; Imamura, M. Overexpression of matrix metalloproteinase 9 gene in hepatocellular carcinoma with invasive potential. Hepatology 1996, 24, 316–322. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.-H.; Han, X.-C.; Jia, M.-K.; Jiang, W.-D.; Wang, M.; Zhang, H.; Han, G.; Jiang, Y. Expressions of inducible nitric oxide synthase and matrix metalloproteinase-9 and their effects on angiogenesis and progression of hepatocellular carcinoma. World J. Gastroenterol. 2005, 11, 5931–5937. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Murphy, F.R.; Gehdu, N.; Zhang, J.; Iredale, J.P.; Benyon, R.C. Engagement of alphavbeta3 integrin regulates proliferation and apoptosis of hepatic stellate cells. J. Biol. Chem. 2004, 279, 23996–24006. [Google Scholar] [CrossRef] [PubMed]
- Nart, D.; Karasu, Z.; Yaman, B.; Yilmaz, F.; Zeytunlu, M.; Kılıç, M. Expression of MMP-9 in predicting prognosis of hepatocellular carcinoma after liver transplantation. Liver Transplant. 2010, 16, 621–630. [Google Scholar] [CrossRef]
- Olle, E.W.; Ren, X.; McClintock, S.D.; Warner, R.L.; Deogracias, M.P.; Johnson, K.J.; Colletti, L.M. Matrix metalloproteinase-9 is an important factor in hepatic regeneration after partial hepatectomy in mice. Hepatology 2006, 44, 540–549. [Google Scholar] [CrossRef]
- Nguyen, J.H.; Yamamoto, S.; Steers, J.; Sevlever, D.; Lin, W.; Shimojima, N.; Castanedes-Casey, M.; Genco, P.; Golde, T.; Richelson, E.; et al. Matrix metalloproteinase-9 contributes to brain extravasation and edema in fulminant hepatic failure mice. J. Hepatol. 2006, 44, 1105–1114. [Google Scholar] [CrossRef]
- Hori, T.; Uemoto, S.; Walden, L.B.; Chen, F.; Baine, A.-M.T.; Hata, T.; Kogure, T.; Nguyen, J.H. Matrix metalloproteinase-9 as a therapeutic target for the progression of fulminant liver failure with hepatic encephalopathy: A pilot study in mice. Hepatol. Res. 2013, 44, 651–662. [Google Scholar] [CrossRef]
- Garcia-Irigoyen, O.; Carotti, S.; Latasa, M.U.; Uriarte, I.; Fernandez-Barrena, M.G.; Elizalde, M.; Urtasun, R.; Gentilucci, U.V.; Morini, S.; Bañales, J.; et al. Matrix metalloproteinase-10 expression is induced during hepatic injury and plays a fundamental role in liver tissue repair. Liver Int. 2014, 34, e257–e270. [Google Scholar] [CrossRef]
- García-Irigoyen, O.; Latasa, M.U.; Carotti, S.; Uriarte, I.; Elizalde, M.; Urtasun, R.; Vespasiani-Gentilucci, U.; Morini, S.; Benito, P.; Ladero, J.M.; et al. Matrix Metalloproteinase 10 Contributes To Hepatocarcinogenesis in a Novel Crosstalk With the Stromal Derived Factor 1/C-X-C Chemokine Receptor 4 Axis. Hepatology 2015, 62, 166–178. [Google Scholar]
- Bi, Q.; Tang, S.; Xia, L.; Du, R.; Fan, R.; Gao, L.; Jin, J.; Liang, S.; Chen, Z.; Xu, G.; et al. Ectopic Expression of MiR-125a Inhibits the Proliferation and Metastasis of Hepatocellular Carcinoma by Targeting MMP11 and VEGF. PLoS ONE 2012, 7, e40169. [Google Scholar] [CrossRef]
- Wang, B.; Hsu, C.-J.; Lee, H.-L.; Chou, C.-H.; Su, C.-M.; Yang, S.-F.; Tang, C.-H. Impact of matrix metalloproteinase-11 gene polymorphisms upon the development and progression of hepatocellular carcinoma. Int. J. Med Sci. 2018, 15, 653–658. [Google Scholar] [CrossRef] [PubMed]
- Dali-Youcef, N.; Hnia, K.; Blaise, S.; Messaddeq, N.; Blanc, S.; Postic, C.; Valet, P.; Tomasetto, C.; Rio, M.-C. Matrix metalloproteinase 11 protects from diabesity and promotes metabolic switch. Sci. Rep. 2016, 6, 25140. [Google Scholar] [CrossRef] [PubMed]
- Madala, S.K.; Pesce, J.T.; Ramalingam, T.R.; Wilson, M.S.; Minnicozzi, S.; Cheever, A.W.; Thompson, R.W.; Mentink-Kane, M.M.; A Wynn, T. Matrix metalloproteinase 12-deficiency augments extracellular matrix degrading metalloproteinases and attenuates IL-13-dependent fibrosis. J. Immunol. 2010, 184, 3955–3963. [Google Scholar] [CrossRef] [PubMed]
- Ng, K.T.-P.; Qi, X.; Kong, K.-L.; Cheung, B.Y.-Y.; Lo, C.-M.; Poon, R.T.P.; Fan, S.T.; Man, K. Overexpression of matrix metalloproteinase-12 (MMP-12) correlates with poor prognosis of hepatocellular carcinoma. Eur. J. Cancer 2011, 47, 2299–2305. [Google Scholar] [CrossRef]
- Pellicoro, A.; Aucott, R.L.; Ramachandran, P.; Robson, A.J.; Fallowfield, J.; Snowdon, V.K.; Hartland, S.N.; Vernon, M.; Duffield, J.S.; Benyon, R.C.; et al. Elastin accumulation is regulated at the level of degradation by macrophage metalloelastase (MMP-12) during experimental liver fibrosis. Hepatology 2012, 55, 1965–1975. [Google Scholar] [CrossRef]
- Yata, Y.; Takahara, T.; Furui, K.; Zhang, L.P.; Watanabe, A. Expression of matrix metalloproteinase-13 and tissue inhibitor of metalloproteinase-1 in acute liver injury. J. Hepatol. 1999, 30, 419–424. [Google Scholar] [CrossRef]
- Uchinami, H.; Seki, E.; Brenner, D.A.; D’Armiento, J. Loss of MMP 13 attenuates murine hepatic injury and fibrosis during cholestasis. Hepatology 2006, 44, 420–429. [Google Scholar] [CrossRef]
- George, J.; Tsutsumi, M.; Tsuchishima, M. MMP-13 deletion decreases profibrogenic molecules and attenuates N-nitrosodimethylamine-induced liver injury and fibrosis in mice. J. Cell. Mol. Med. 2017, 21, 3821–3835. [Google Scholar] [CrossRef]
- Hironaka, K.; Sakaida, I.; Matsumura, Y.; Kaino, S.; Miyamoto, K.; Okita, K. Enhanced Interstitial Collagenase (Matrix Metalloproteinase-13) Production of Kupffer Cell by Gadolinium Chloride Prevents Pig Serum-Induced Rat Liver Fibrosis. Biochem. Biophys. Res. Commun. 2000, 267, 290–295. [Google Scholar] [CrossRef]
- Fallowfield, J.; Mizuno, M.; Kendall, T.; Constandinou, C.M.; Benyon, R.C.; Duffield, J.S.; Iredale, J.P. Scar-associated macrophages are a major source of hepatic matrix metalloproteinase-13 and facilitate the resolution of murine hepatic fibrosis. J. Immunol. 2007, 178, 5288–5295. [Google Scholar] [CrossRef]
- Calabro, S.R.; Maczurek, A.E.; Morgan, A.J.; Tu, T.; Wen, V.W.; Yee, C.; Mridha, A.; Lee, M.; D’Avigdor, W.; Locarnini, S.A.; et al. Hepatocyte Produced Matrix Metalloproteinases Are Regulated by CD147 in Liver Fibrogenesis. PLoS ONE 2014, 9, e90571. [Google Scholar] [CrossRef] [PubMed]
- Endo, H.; Niioka, M.; Sugioka, Y.; Itoh, J.; Kameyama, K.; Okazaki, I.; Ala-Aho, R.; Kähäri, V.; Watanabe, T. Matrix Metalloproteinase-13 Promotes Recovery from Experimental Liver Cirrhosis in Rats. Pathobiology 2011, 78, 239–252. [Google Scholar] [CrossRef]
- Załuska, W.; Prystupa, A.; Szpetnar, M.; Boguszewska-Czubara, A.; Grzybowski, A.; Sak, J. Activity of MMP1 and MMP13 and Amino Acid Metabolism in Patients with Alcoholic Liver Cirrhosis. Med Sci. Monit. 2015, 21, 1008–1014. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jin, D.; Tao, J.; Li, D.; Wang, Y.; Li, L.; Hu, Z.; Zhou, Z.; Chang, X.; Qu, C.; Zhang, H. Golgi protein 73 activation of MMP-13 promotes hepatocellular carcinoma cell invasion. Oncotarget 2015, 6, 33523–33533. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.A.; Yan, H.Q.; Wang, F.; Wang, Y.Y.; Jiang, Y.N.; Wang, Y.N.; Gao, F.G. TIPE2 inhibits TNF-α-induced hepatocellular carcinoma cell metastasis via Erk1/2 downregulation and NF-κB activation. Int. J. Oncol. 2015, 46, 254–264. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Hovell, C.J.; Pawley, S.; Hutchings, M.I.; Arthur, M.J.P.; Iredale, J.P.; Benyon, R. Expression of matrix metalloproteinase-2 and -14 persists during early resolution of experimental liver fibrosis and might contribute to fibrolysis. Liver Int. 2004, 24, 492–501. [Google Scholar] [CrossRef]
- Duarte, S.; Shen, X.-D.; Fondevila, C.; Busuttil, R.W.; Coito, A.J. Fibronectin-α4β1 interactions in hepatic cold ischemia and reperfusion injury: Regulation of MMP-9 and MT1-MMP via the p38 MAPK pathway. Am. J. Transplant. 2012, 12, 2689–2699. [Google Scholar] [CrossRef]
- Harada, T.; Arii, S.; Mise, M.; Imamura, T.; Higashitsuji, H.; Furutani, M.; Niwano, M.; Ishigami, S.-I.; Fukumoto, M.; Seiki, M.; et al. Membrane-type matrix metalloproteinase-1(MT1-MMP) gene is overexpressed in highly invasive hepatocellular carcinomas. J. Hepatol. 1998, 28, 231–239. [Google Scholar] [CrossRef]
- Ip, Y.-C.; Cheung, S.T.; Leung, K.-L.; Fan, S.-T. Mechanism of metastasis by membrane type 1-matrix metalloproteinase in hepatocellular carcinoma. World J. Gastroenterol. 2005, 11, 6269–6276. [Google Scholar] [CrossRef]
- Mohammed, F.F.; Pennington, C.J.; Kassiri, Z.; Rubin, J.S.; Soloway, P.; Ruther, U.; Edwards, D.R.; Khokha, R. Metalloproteinase inhibitor TIMP-1 affects hepatocyte cell cycle via HGF activation in murine liver regeneration. Hepatology 2005, 41, 857–867. [Google Scholar] [CrossRef]
- Butler, G.S.; Will, H.; Atkinson, S.J.; Murphy, G. Membrane-Type-2 Matrix Metalloproteinase Can Initiate the Processing of Progelatinase A and is Regulated by the Tissue Inhibitors of Metalloproteinases. Eur. J. Biochem. 1997, 244, 653–657. [Google Scholar] [CrossRef] [PubMed]
- Tsai, H.-F.; Trubelja, A.; Shen, A.Q.; Bao, G. Tumour-on-a-chip: Microfluidic models of tumour morphology, growth and microenvironment. J. R. Soc. Interface 2017, 14, 20170137. [Google Scholar] [CrossRef] [PubMed]
- Arai, I.; Nagano, H.; Kondo, M.; Yamamoto, H.; Hiraoka, N.; Sugita, Y.; Ota, H.; Yoshioka, S.; Nakamura, M.; Wada, H.; et al. Overexpression of MT3-MMP in hepatocellular carcinoma correlates with capsular invasion. Hepatogastroenterology 2007, 54, 167–171. [Google Scholar] [PubMed]
- Jiroušková, M.; Zbodakova, O.; Gregor, M.; Chalupský, K.; Sarnova, L.; Hajdúch, M.; Ehrmann, J.; Jirkovska, M.; Sedlacek, R. Hepatoprotective Effect of MMP-19 Deficiency in a Mouse Model of Chronic Liver Fibrosis. PLoS ONE 2012, 7, e46271. [Google Scholar] [CrossRef] [PubMed]
- Stoyanov, E.; Ludwig, G.; Mizrahi, L.; Olam, D.; Schnitzer-Perlman, T.; Tasika, E.; Sass, G.; Tiegs, G.; Jiang, Y.; Nie, T.; et al. Chronic liver inflammation modifies DNA methylation at the precancerous stage of murine hepatocarcinogenesis. Oncotarget 2015, 6, 11047–11060. [Google Scholar] [CrossRef]
- Qi, F.; Song, J.; Yang, H.; Gao, W.; Liu, N.-A.; Zhang, B.; Lin, S. Mmp23b promotes liver development and hepatocyte proliferation through the tumor necrosis factor pathway in zebrafish. Hepatology 2010, 52, 2158–2166. [Google Scholar] [CrossRef]
- Soria-Valles, C.; Gutiérrez-Fernández, A.; Osorio, F.G.; Carrero, D.; Ferrando, A.A.; Colado, E.; Fernández-García, M.S.; Bonzon-Kulichenko, E.; Vázquez, J.; Fueyo, A.; et al. MMP-25 Metalloprotease Regulates Innate Immune Response through NF-κB Signaling. J. Immunol. 2016, 197, 296–302. [Google Scholar] [CrossRef]
- Rodgers, U.R.; Kevorkian, L.; Surridge, A.K.; Waters, J.G.; Swingler, T.E.; Culley, K.; Illman, S.; Lohi, J.; Parker, A.E.; Clark, I.M. Expression and function of matrix metalloproteinase (MMP)-28. Matrix Biol. 2009, 28, 263–272. [Google Scholar] [CrossRef]
- Zhou, J.; Zheng, X.; Feng, M.; Mo, Z.; Shan, Y.; Wang, Y.; Jin, J. Upregulated MMP28 in Hepatocellular Carcinoma Promotes Metastasis via Notch3 Signaling and Predicts Unfavorable Prognosis. Int. J. Boil. Sci. 2019, 15, 812–825. [Google Scholar] [CrossRef]
- Ma, Y.; Brás, L.E.D.C.; Toba, H.; Iyer, R.P.; Hall, M.E.; Winniford, M.D.; Lange, R.A.; Tyagi, S.C.; Lindsey, M.L. Myofibroblasts and the extracellular matrix network in post-myocardial infarction cardiac remodeling. Pflugers Arch. 2014, 466, 1113–1127. [Google Scholar] [CrossRef]
- Lichtinghagen, R.; Steinmetz, C.; Pehle, B.; Breitenstein, K.; Seifert, T.; Boeker, K.H.W. Matrix metalloproteinases in liver and serum in chronic active hepatitis C and HCV-induced cirrhosis. Hepatol. Res. 1999, 14, 119–134. [Google Scholar] [CrossRef]
- Iimuro, Y.; Brenner, D.A. Matrix Metalloproteinase Gene Delivery for Liver Fibrosis. Pharm. Res. 2007, 25, 249–258. [Google Scholar] [CrossRef] [PubMed]
- Fields, G.B. Interstitial Collagen Catabolism*. J. Biol. Chem. 2013, 288, 8785–8793. [Google Scholar] [CrossRef] [PubMed]
- Lauer-Fields, J.L.; Fields, G.B. Triple-helical peptide analysis of collagenolytic protease activity. Biol. Chem. 2002, 383, 1095–1105. [Google Scholar] [CrossRef] [PubMed]
- Powell, B.; Malaspina, D.C.; Szleifer, I.; Dhaher, Y. Effect of collagenase–gelatinase ratio on the mechanical properties of a collagen fibril: A combined Monte Carlo–molecular dynamics study. Biomech. Model. Mechanobiol. 2019, 18, 1809–1819. [Google Scholar] [CrossRef] [PubMed]
- Giannelli, G.; Bergamini, C.; Marinosci, F.; Fransvea, E.; Quaranta, M.; Lupo, L.; Schiraldi, O.; Antonaci, S. Clinical role of MMP-2/TIMP-2 imbalance in hepatocellular carcinoma. Int. J. Cancer 2002, 97, 425–431. [Google Scholar] [CrossRef]
- Ito, A.; Mukaiyama, A.; Itoh, Y.; Nagase, H.; Thøgersen, I.B.; Enghild, J.J.; Sasaguri, Y.; Mori, Y. Degradation of Interleukin 1β by Matrix Metalloproteinases. J. Biol. Chem. 1996, 271, 14657–14660. [Google Scholar] [CrossRef]
- Wu, J.-F.; Jeng, Y.-M.; Chen, H.-L.; Ni, Y.-H.; Hsu, H.-Y.; Chang, M.-H. Quantification of Serum Matrix Metallopeptide 7 Levels May Assist in the Diagnosis and Predict the Outcome for Patients with Biliary Atresia. J. Pediatr. 2019, 208, 30–37.e1. [Google Scholar] [CrossRef]
- Harpavat, S. MMP-7: The Next Best Serum Biomarker for Biliary Atresia? J. Pediatr. 2019, 208, 8–9. [Google Scholar] [CrossRef]
- Giannandrea, M.; Parks, W.C. Diverse functions of matrix metalloproteinases during fibrosis. Dis. Model. Mech. 2014, 7, 193–203. [Google Scholar] [CrossRef]
- Lachowski, D.; Cortes, E.; Rice, A.; Pinato, D.; Rombouts, K.; Hernandez, A.D.R. Matrix stiffness modulates the activity of MMP-9 and TIMP-1 in hepatic stellate cells to perpetuate fibrosis. Sci. Rep. 2019, 9, 7299. [Google Scholar] [CrossRef] [PubMed]
- Itoh, Y. Membrane-type matrix metalloproteinases: Their functions and regulations. Matrix Biol. 2015, 44, 207–223. [Google Scholar] [CrossRef] [PubMed]
- Scheau, C.; Badarau, I.A.; Costache, R.S.; Caruntu, C.; Mihai, G.L.; Didilescu, A.C.; Constantin, C.; Neagu, M. The Role of Matrix Metalloproteinases in the Epithelial-Mesenchymal Transition of Hepatocellular Carcinoma. Anal. Cell. Pathol. 2019, 2019, 9423907. [Google Scholar] [CrossRef] [PubMed]
- Fellows, C.R.; Matta, C.; Zakany, R.; Khan, I.M.; Mobasheri, A. Adipose, Bone Marrow and Synovial Joint-Derived Mesenchymal Stem Cells for Cartilage Repair. Front. Genet. 2016, 7, 1920. [Google Scholar] [CrossRef]
- Eom, Y.W.; Shim, K.Y.; Baik, S.K. Mesenchymal stem cell therapy for liver fibrosis. Korean J. Intern. Med. 2015, 30, 580–589. [Google Scholar] [CrossRef]
- Itaba, N.; Kono, Y.; Watanabe, K.; Yokobata, T.; Oka, H.; Osaki, M.; Kakuta, H.; Morimoto, M.; Shiota, G. Reversal of established liver fibrosis by IC-2-engineered mesenchymal stem cell sheets. Sci. Rep. 2019, 9, 6841. [Google Scholar] [CrossRef]
- Liu, T.; Wang, P.; Cong, M.; Zhang, D.; Liu, L.; Li, H.; Zhai, Q.; Li, Z.; Jia, J.; You, H. Matrix metalloproteinase-1 induction by diethyldithiocarbamate is regulated via Akt and ERK/miR222/ETS-1 pathways in hepatic stellate cells. Biosci. Rep. 2016, 36, e00371. [Google Scholar] [CrossRef]
- Li, Y.; Liu, F.; Ding, F.; Chen, P.; Tang, M. Inhibition of liver fibrosis using vitamin A-coupled liposomes to deliver matrix metalloproteinase-2 siRNA in vitro. Mol. Med. Rep. 2015, 12, 3453–3461. [Google Scholar] [CrossRef]
- Lu, C.; Zou, Y.; Liu, Y.; Niu, Y. Rosmarinic acid counteracts activation of hepatic stellate cells via inhibiting the ROS-dependent MMP-2 activity: Involvement of Nrf2 antioxidant system. Toxicol. Appl. Pharmacol. 2017, 318, 69–78. [Google Scholar] [CrossRef]
- Roderfeld, M.; Weiskirchen, R.; Wagner, S.; Berres, M.-L.; Henkel, C.; Grötzinger, J.; Gressner, A.M.; Matern, S.; Roeb, E. Inhibition of hepatic fibrogenesis by matrix metalloproteinase-9 mutants in mice. FASEB J. 2006, 20, 444–454. [Google Scholar] [CrossRef]
- Fields, G.B. The Rebirth of Matrix Metalloproteinase Inhibitors: Moving Beyond the Dogma. Cells 2019, 8, 984. [Google Scholar] [CrossRef] [PubMed]
- Freitas-Rodríguez, S.; Folgueras, A.; Lopez-Otin, C. The role of matrix metalloproteinases in aging: Tissue remodeling and beyond. Biochim. Biophys. Acta Mol. Cell Res. 2017, 1864, 2015–2025. [Google Scholar] [CrossRef] [PubMed]
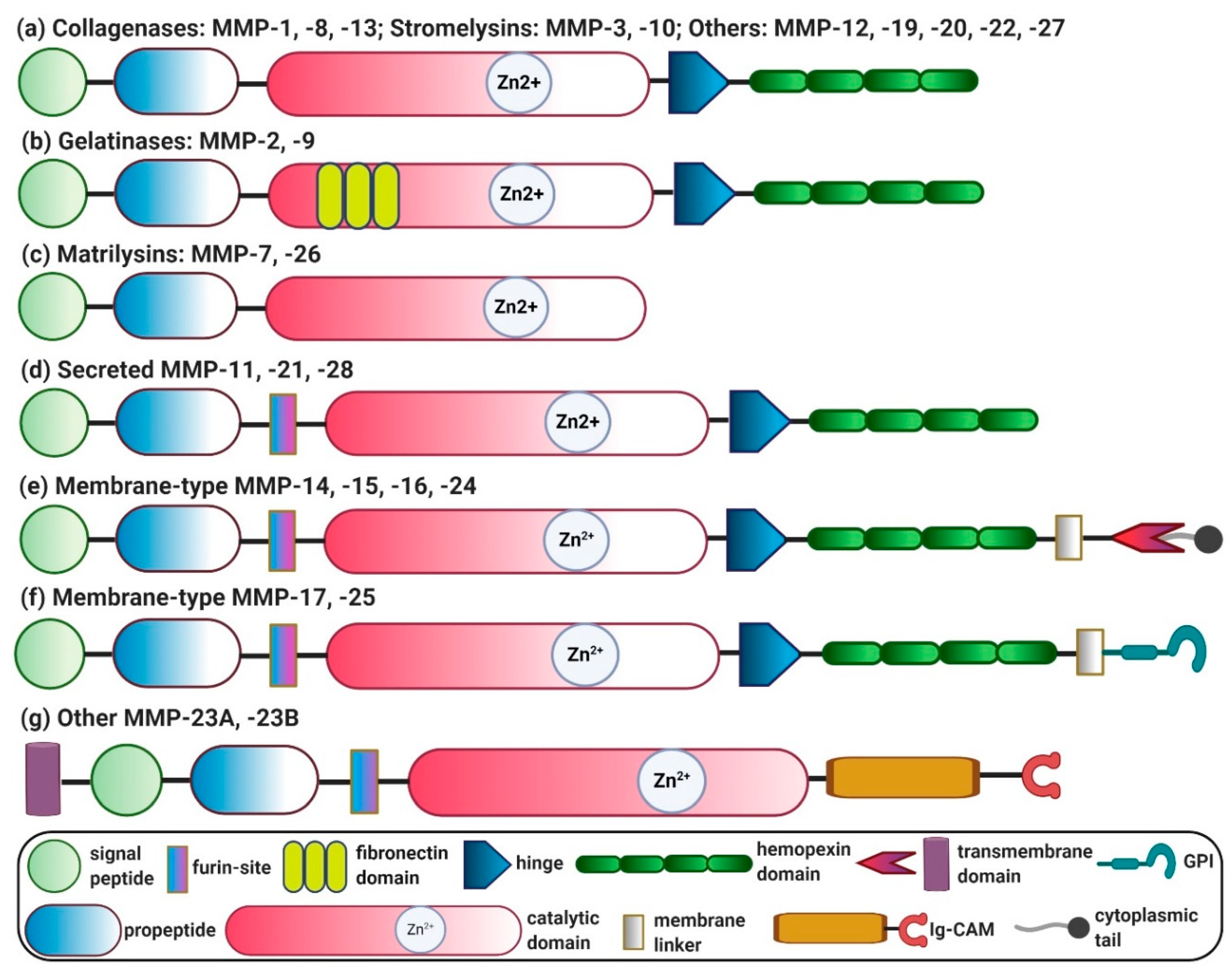
| MMP | Group | Role in Liver Diseases | Reference |
|---|---|---|---|
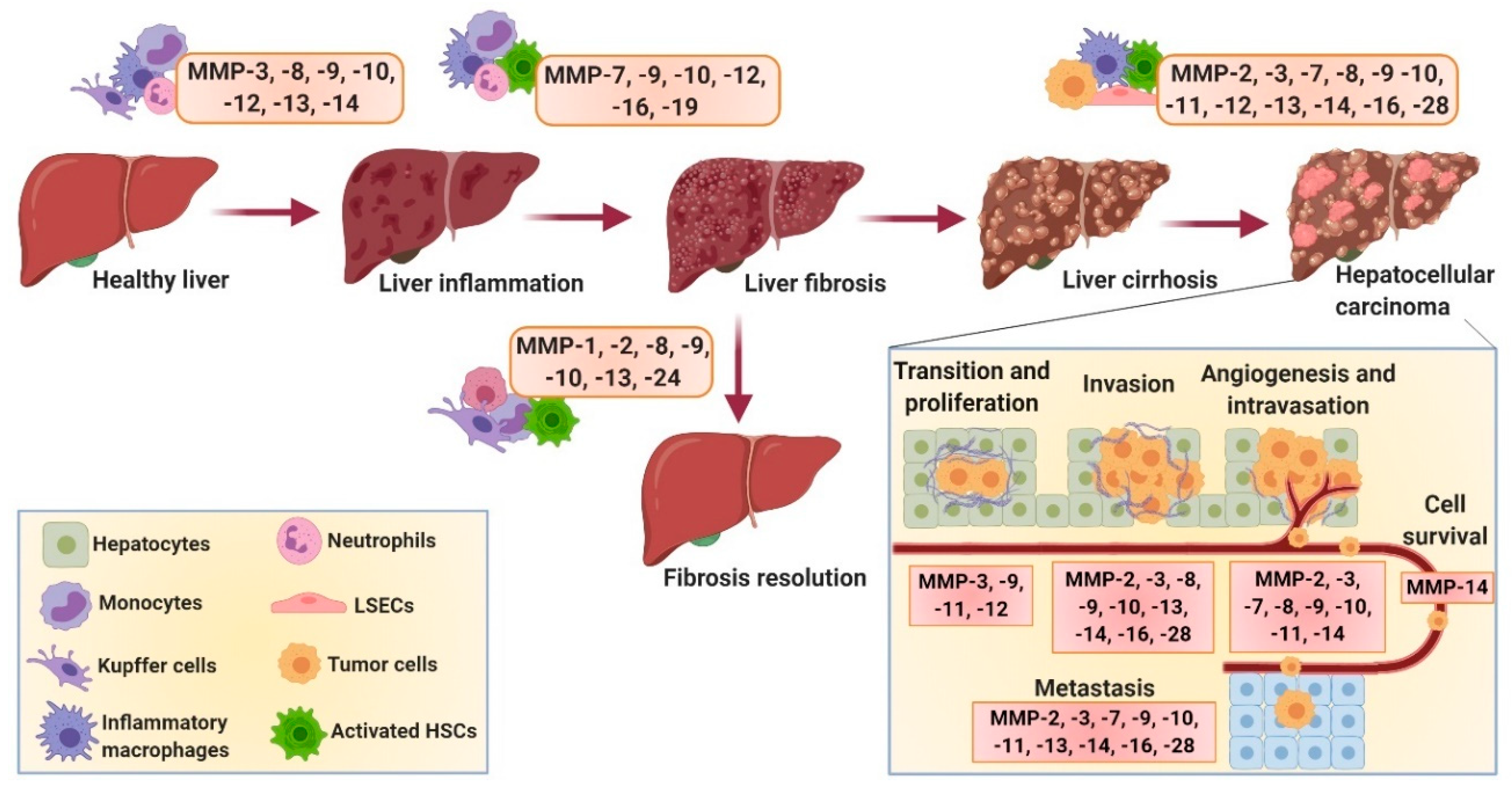
| MMP-1 | Collagenase-1 | Decreased expression is associated with disease severity in patients with chronic viral hepatitis and NASH; polymorphism is associated with HCC; induced expression attenuates liver fibrosis. | [25,39,40,41,42,43] |
| MMP-2 | Gelatinase-A | Increased expression in chronic viral hepatitis, liver fibrosis, alcoholic liver cirrhosis (all stages), IRI, BA and HCC (and metastasis); depletion/inhibition exacerbates fibrosis in CCl4-, toxin-, cholestasis- and IRI-induced fibrosis. | [44,45,46,47,48,49,50] |
| MMP-3 | Stromelysin-1 | Increased expression during liver inflammation, IRI and HCC (and metastasis); polymorphism is associated with progressive liver disease, PSC and HCC; inhibition/depletion inhibits liver injury. | [47,51,52,53,54,55,56,57] |
| MMP-7 | Matrilysin-7 | Increased expression in BA-associated fibrosis, chronic hepatitis C, HCC and colorectal cancer liver metastasis; regulates oval-cell-mediated liver regeneration, together with CD44, during liver injury. | [46,58,59,60] |
| MMP-8 | Collagenase-2 | Increased expression in alcoholic liver cirrhosis (stage C); promotes HCC invasion and metastasis; involved in cholestatic liver injury repair; overexpression ameliorates CCl4-, TAA- and BDL-induced fibrosis; depletion protects against TNF-induced hepatitis. | [48,61,62,63,64,65,66] |
| MMP-9 | Gelatinase-B | Increased expression in IRI, liver fibrogenesis, alcoholic liver cirrhosis (stage C), and HCC; induces HSCs apoptosis; promotes tumor invasion and metastasis; promotes fibrosis resolution; contributes to loss of BBB integrity and edema during ALF/FLF; depletion/inhibition ameliorates IRI, accelerates liver regeneration and improves early-stage brain injuries in ALF/FLF. | [48,67,68,69,70,71,72,73,74,75,76,77,78] |
| MMP-10 | Stromelysin-2 | Strongly expressed in acute liver injury, cirrhosis and HCC (and metastasis); deficiency impairs wound healing during cholestatic liver injury and after partial hepatectomy; contributes to HCC progression and metastasis. | [51,79,80] |
| MMP-11 | Stromelysin-3 | Genetic variants associated with HCC progression; regulates HCC proliferation and metastasis; protects against diabesity and hepatic steatosis; promotes liver regeneration in IRI. | [47,81,82,83] |
| MMP-12 | Metalloelastase | Increased expression in schistosomiasis, cirrhosis, IRI and HCC; regulates elastin degradation in fibrosis; mediates wound healing in IRI; inhibits MMPs (MMP-2, -9 and -13), and aggravates fibrosis. | [47,84,85,86] |
| MMP-13 | Collagenase-3 | Increased in HCC and positively correlated with HCC progression and metastasis; promotes inflammation and fibrogenesis during acute liver injury; deficiency/depletion attenuates cholestasis-induced liver inflammation and fibrogenesis; macrophage-secreted MMP-13 mediates fibrosis resolution; overexpression enhances recovery and hepatic repair; overexpressed in alcoholic liver cirrhosis (all stages). | [47,87,88,89,90,91,92,93,94,95,96] |
| MMP-14 | MT1-MMP | Highly expressed in HCC and positively correlated with invasion, metastasis and HCC recurrence; activates MMP-2 and -13; stimulates recruitment of macrophages and leukocytes in IRI. | [44,97,98,99,100] |
| MMP-15 | MT2-MMP | Expression downregulated during liver regeneration; activates MMP-2 and MMP-13. | [101,102] |
| MMP-16 | MT3-MMP | Expressed in hepatitis, cirrhosis and HCC (and metastasis). | [103,104] |
| MMP-19 | RASI-1 | Depletion reduces fibrosis and promotes regeneration. | [105] |
| MMP-23 | Other | Involved in inflammation-driven hepatocarcinogenesis; promotes hepatocyte proliferation and regeneration. | [106,107] |
| MMP-24 | MT5-MMP | Upregulation during liver regeneration. | [101] |
| MMP-25 | MT6-MMP | Implicated in tumor invasion. | [108] |
| MMP-28 | Secreted MMP (epilysin) | Upregulated during alcohol-mediated hepatocyte damage; upregulated and associated with migration, invasion and poor prognosis of HCC. | [109,110] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Geervliet, E.; Bansal, R. Matrix Metalloproteinases as Potential Biomarkers and Therapeutic Targets in Liver Diseases. Cells 2020, 9, 1212. https://doi.org/10.3390/cells9051212
Geervliet E, Bansal R. Matrix Metalloproteinases as Potential Biomarkers and Therapeutic Targets in Liver Diseases. Cells. 2020; 9(5):1212. https://doi.org/10.3390/cells9051212
Chicago/Turabian StyleGeervliet, Eline, and Ruchi Bansal. 2020. "Matrix Metalloproteinases as Potential Biomarkers and Therapeutic Targets in Liver Diseases" Cells 9, no. 5: 1212. https://doi.org/10.3390/cells9051212
APA StyleGeervliet, E., & Bansal, R. (2020). Matrix Metalloproteinases as Potential Biomarkers and Therapeutic Targets in Liver Diseases. Cells, 9(5), 1212. https://doi.org/10.3390/cells9051212






