Differentiation of Human Mesenchymal Stem Cells from Wharton’s Jelly Towards Neural Stem Cells Using a Feasible and Repeatable Protocol
Abstract
:1. Introduction
2. Materials and Methods
2.1. Isolation and Culture of MSCs from Human Wharton’s Jelly of the Umbilical Cord (hWJ-MSCs) Using Tissue Explants Method
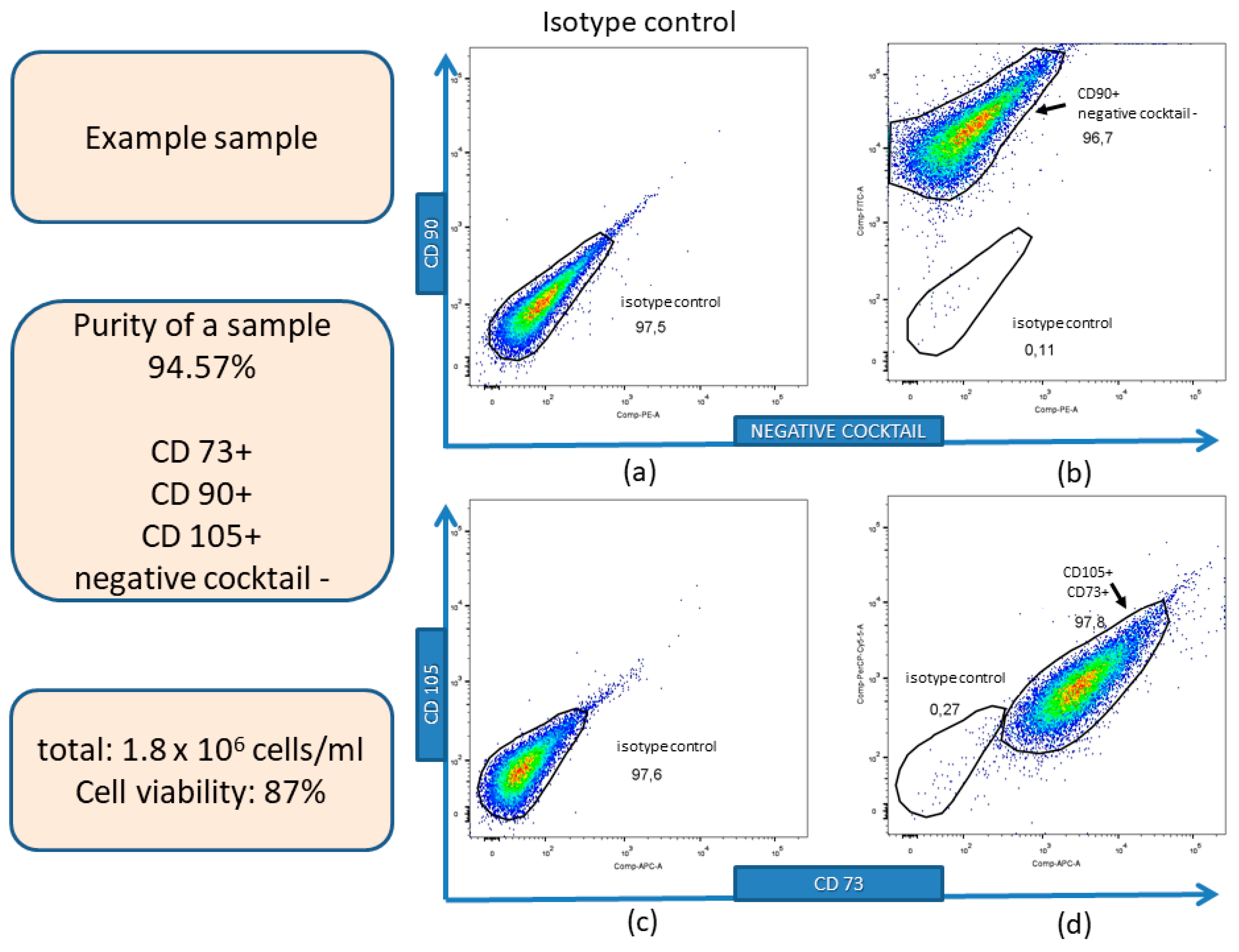
2.2. Immunophenotypic Characterization of hWJ-MSCs
2.3. Neural Induction of hWJ-MSCs
2.4. Microscopic Characterization of Differentiated Cells (hWJ-NSCs—Human Wharton’s Jelly Derived Neural Stem Cells)
2.5. Immunocytochemistry Analysis
2.6. Immunophenotypic Characterization of Differentiated Cells (hWJ-NSCs)
2.7. Quantitative Reverse Transcription-Polymerase Chain Reaction (qRT-PCR)
3. Results
3.1. Isolation, Expansion, and Immunophenotyping Characterization of hWJ-MSCs
3.2. Neural Induction of hWJ-MSCs
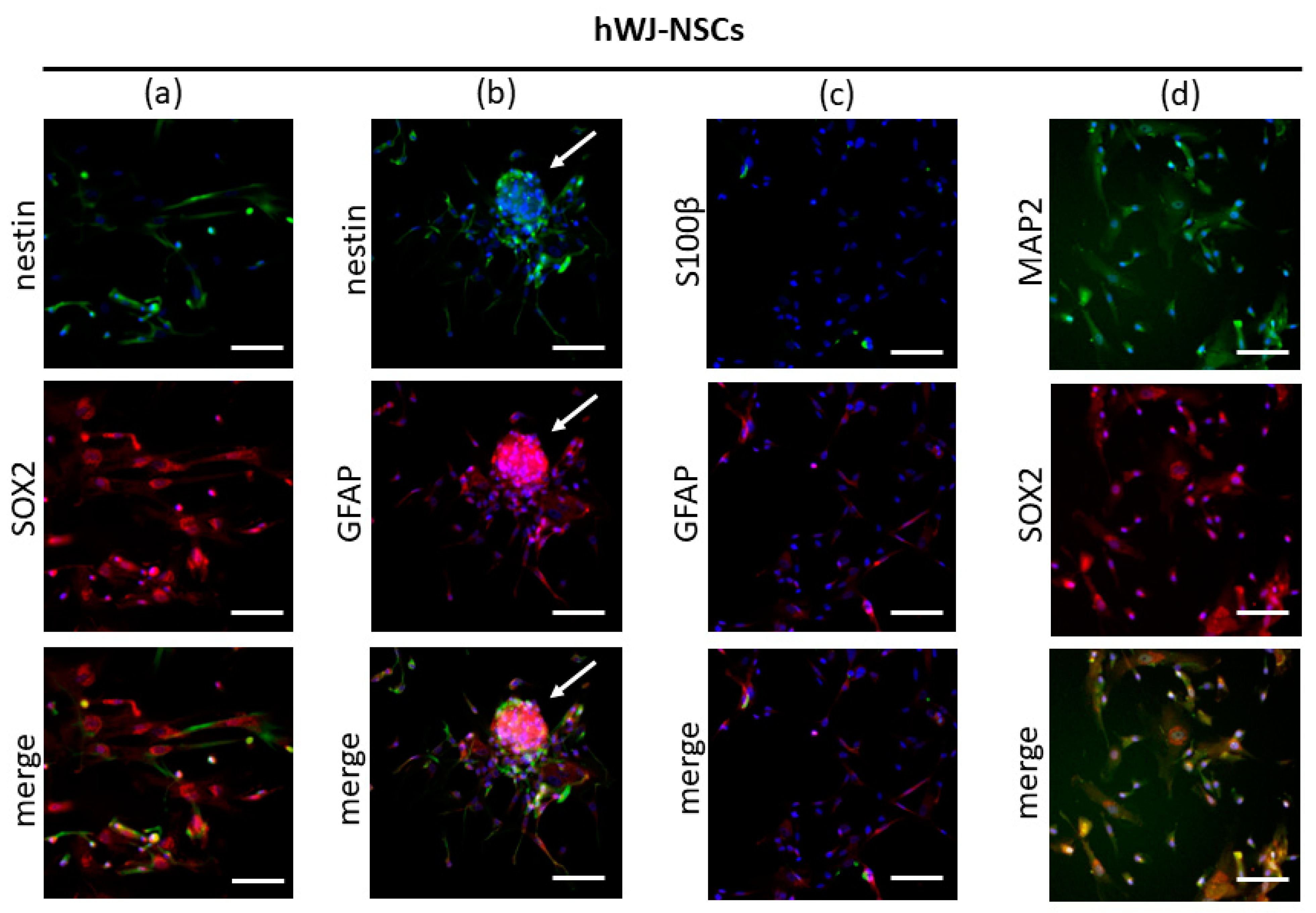
3.3. Characterization of hWJ-NSCs
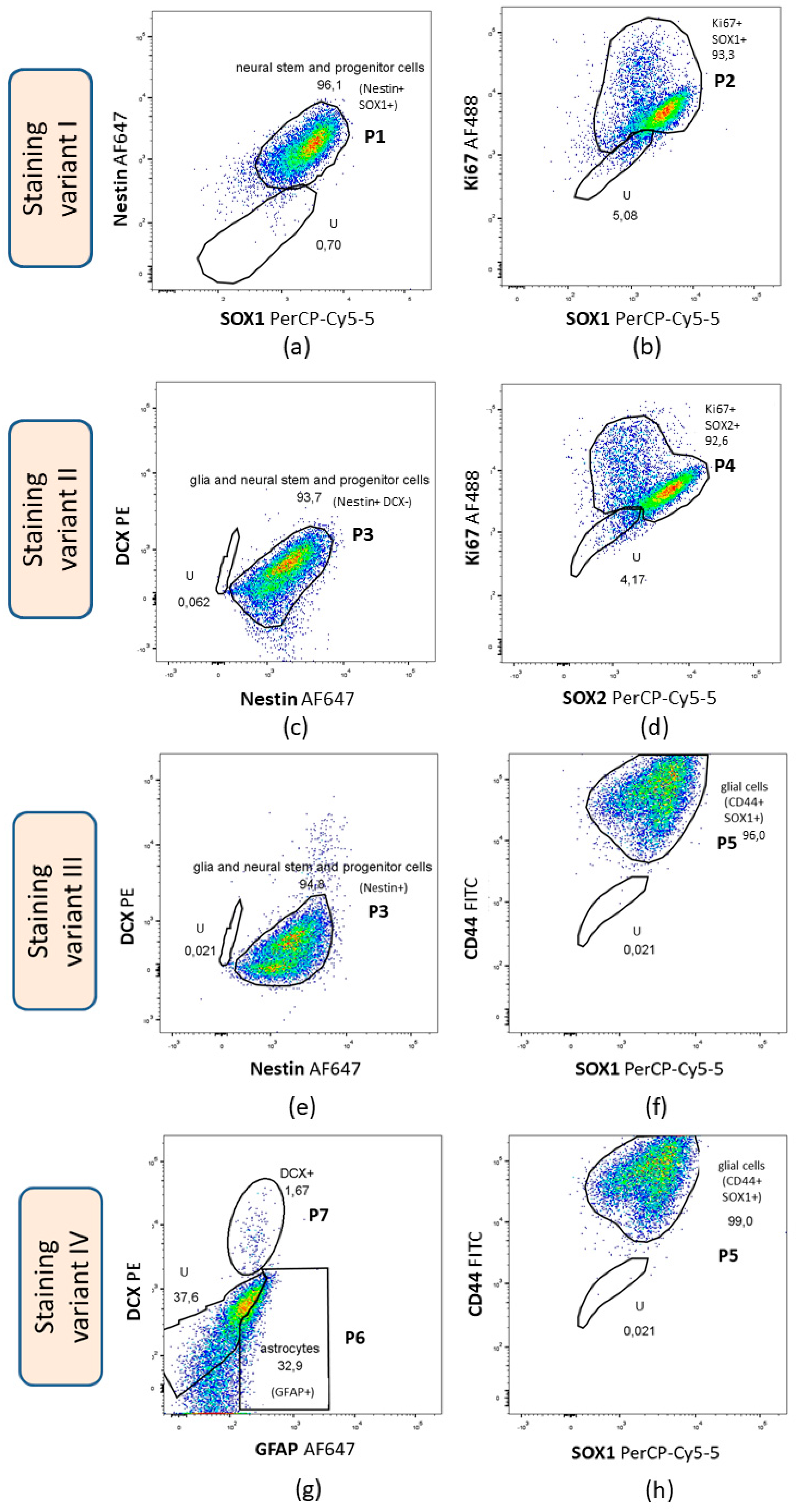
3.3.1. Flow Cytometry
3.3.2. Immunocytochemistry
3.4. Quantitative Reverse Transcription Polymerase Chain Reaction (qRT-PCR)
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sakthiswary, R.; Raymond, A.A. Stem cell therapy in neurodegenerative diseases: From principles to practice. Neural Regen. Res. 2012, 7, 1822–1831. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.U.; Lee, H.J.; Kim, Y.B. Neural stem cell-based treatment for neurodegenerative diseases. Neuropathology 2013, 33, 491–504. [Google Scholar] [CrossRef] [PubMed]
- Marsh, S.E.; Blurton-Jones, M. Neural stem cell therapy for neurodegenerative disorders: The role of neurotrophic support. Neurochem. Int. 2017, 106, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Dantuma, E.; Merchant, S.; Sugaya, K. Stem cells for the treatment of neurodegenerative diseases. Stem Cell Res. Ther. 2010, 1, 37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kwak, K.A.; Lee, S.P.; Yang, J.Y.; Park, Y.S. Current Perspectives regarding Stem Cell-Based Therapy for Alzheimer’s Disease. Stem Cells Int. 2018, 2018, 6392986. [Google Scholar] [CrossRef] [Green Version]
- Bydon, M.; Dietz, A.B.; Goncalves, S.; Moinuddin, F.M.; Alvi, M.A.; Goyal, A.; Yolcu, Y.; Hunt, C.L.; Garlanger, K.L.; Del Fabro, A.S.; et al. CELLTOP Clinical Trial: First Report From a Phase 1 Trial of Autologous Adipose Tissue-Derived Mesenchymal Stem Cells in the Treatment of Paralysis Due to Traumatic Spinal Cord Injury. Mayo Clin. Proc. 2020, 95, 406–414. [Google Scholar] [CrossRef] [Green Version]
- Low, C.B.; Liou, Y.C.; Tang, B.L. Neural differentiation and potential use of stem cells from the human umbilical cord for central nervous system transplantation therapy. J. Neurosci. Res. 2008, 86, 1670–1679. [Google Scholar] [CrossRef]
- Tang, Y.; Yu, P.; Cheng, L. Current progress in the derivation and therapeutic application of neural stem cells. Cell Death Dis. 2017, 8, e3108. [Google Scholar] [CrossRef] [Green Version]
- Casarosa, S.; Bozzi, Y.; Conti, L. Neural stem cells: Ready for therapeutic applications? Mol. Cell. Ther. 2014, 2, 31. [Google Scholar] [CrossRef] [Green Version]
- Barreau, K.; Lépinoux-Chambaud, C.; Eyer, J. Review of Clinical Trials Using Neural Stem Cells. JSM Biotechnol. Biomed. Eng. 2016, 3, 1057. [Google Scholar]
- Thonhoff, J.R.; Ojeda, L.; Wu, P. Stem cell-derived motor neurons: Applications and challenges in amyotrophic lateral sclerosis. Curr. Stem Cell Res. Ther. 2009, 4, 178–199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiao, J.; Yang, R.; Biswas, S.; Zhu, Y.; Qin, X.; Zhang, M.; Zhai, L.; Luo, Y.; He, X.; Mao, C.; et al. Neural Stem Cell-Based Regenerative Approaches for the Treatment of Multiple Sclerosis. Mol. Neurobiol. 2018, 55, 3152–3171. [Google Scholar] [CrossRef] [PubMed]
- Blurton-Jones, M.; Spencer, B.; Michael, S.; Castello, N.A.; Agazaryan, A.A.; Davis, J.L.; Müller, F.J.; Loring, J.F.; Masliah, E.; LaFerla, F.M. Neural stem cells genetically-modified to express neprilysin reduce pathology in Alzheimer transgenic models. Stem Cell Res. Ther. 2014, 5, 46. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Byrne, J.A. Developing neural stem cell-based treatments for neurodegenerative diseases. Stem Cell Res. Ther. 2014, 5, 72. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Portnow, J.; Synold, T.W.; Badie, B.; Tirughana, R.; Lacey, S.F.; D’Apuzzo, M.; Metz, M.Z.; Najbauer, J.; Bedell, V.; Vo, T.; et al. Neural Stem Cell-Based Anticancer Gene Therapy: A First-in-Human Study in Recurrent High-Grade Glioma Patients. Clin. Cancer Res. 2017, 23, 2951–2960. [Google Scholar] [CrossRef] [Green Version]
- Gage, F.H.; Temple, S. Neural stem cells: Generating and regenerating the brain. Neuron 2013, 80, 588–601. [Google Scholar] [CrossRef] [Green Version]
- Teng, Y.D.; Santos, F.N.C.; Black, P.M.; Konya, D.; Park, K.I.; Sidman, R.L.; Snyder, E.Y. Neural Stem Cells. In Principles of Regenerative Medicine; Atala, A., Thomson, J., Lanza, R., Nerem, R.M., Eds.; Academic Press: Cambridge, MA, USA, 2008; pp. 300–317. [Google Scholar]
- Martínez-Cerdeño, V.; Noctor, S.C. Neural Progenitor Cell Terminology. Front. Neuroanat. 2018, 12, 104. [Google Scholar] [CrossRef]
- Lovinger, D.M. Communication networks in the brain: Neurons, receptors, neurotransmitters, and alcohol. Alcohol Res. Health 2008, 31, 196–214. [Google Scholar]
- Barres, B.A. The mystery and magic of glia: A perspective on their roles in health and disease. Neuron 2008, 60, 430–440. [Google Scholar] [CrossRef] [Green Version]
- Domingues, H.S.; Portugal, C.C.; Socodato, R.; Relvas, J.B. Oligodendrocyte, Astrocyte, and Microglia Crosstalk in Myelin Development, Damage, and Repair. Front. Cell Dev. Biol. 2016, 4, 71. [Google Scholar] [CrossRef]
- Zhang, J.; Jiao, J. Molecular Biomarkers for Embryonic and Adult Neural Stem Cell and Neurogenesis. Biomed. Res. Int. 2015, 2015, 727542. [Google Scholar] [CrossRef] [Green Version]
- Oikari, L.E.; Okolicsanyi, R.K.; Griffiths, L.R.; Haupt, L.M. Data defining markers of human neural stem cell lineage potential. Data Brief. 2016, 7, 206–215. [Google Scholar] [CrossRef] [Green Version]
- Bernal, A.; Arranz, L. Nestin-expressing progenitor cells: Function, identity and therapeutic implications. Cell. Mol. Life Sci. 2018, 75, 2177–2195. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Osumi, N.; Shinohara, H.; Numayama-Tsuruta, K.; Maekawa, M. Concise review: Pax6 transcription factor contributes to both embryonic and adult neurogenesis as a multifunctional regulator. Stem Cells 2008, 26, 1663–1672. [Google Scholar] [CrossRef]
- Aubert, J.; Stavridis, M.P.; Tweedie, S.; O’Reilly, M.; Vierlinger, K.; Li, M.; Ghazal, P.; Pratt, T.; Mason, J.O.; Roy, D.; et al. Screening for mammalian neural genes via fluorescence-activated cell sorter purification of neural precursors from Sox1-gfp knock-in mice. Proc. Natl. Acad. Sci. USA 2003, 100 (Suppl. 1), 11836–11841. [Google Scholar] [CrossRef] [Green Version]
- Zhang, S.; Cui, W. Sox2, a key factor in the regulation of pluripotency and neural differentiation. World J. Stem Cells 2014, 6, 305–311. [Google Scholar] [CrossRef] [Green Version]
- Tantrawatpan, C.; Manochantr, S.; Kheolamai, P.; U-Pratya, Y.; Supokawej, A.; Issaragrisil, S. Pluripotent gene expression in mesenchymal stem cells from human umbilical cord Wharton’s jelly and their differentiation potential to neural-like cells. J. Med. Assoc. Thai 2013, 96, 1208–1217. [Google Scholar]
- Gascon, E.; Vutskits, L.; Kiss, J.Z. The role of PSA-NCAM in adult neurogenesis. Adv. Exp. Med. Biol. 2010, 663, 127–136. [Google Scholar] [CrossRef]
- Ayanlaja, A.A.; Xiong, Y.; Gao, Y.; Ji, G.; Tang, C.; Abdikani Abdullah, Z.; Gao, D. Distinct Features of Doublecortin as a Marker of Neuronal Migration and Its Implications in Cancer Cell Mobility. Front. Mol. Neurosci. 2017, 10, 199. [Google Scholar] [CrossRef] [Green Version]
- Menezes, J.R.; Luskin, M.B. Expression of neuron-specific tubulin defines a novel population in the proliferative layers of the developing telencephalon. J. Neurosci. 1994, 14, 5399–5416. [Google Scholar] [CrossRef]
- Sánchez, C.; Díaz-Nido, J.; Avila, J. Phosphorylation of microtubule-associated protein 2 (MAP2) and its relevance for the regulation of the neuronal cytoskeleton function. Prog. Neurobiol. 2000, 61, 133–168. [Google Scholar] [CrossRef]
- Boggs, J.M. Myelin basic protein: A multifunctional protein. Cell. Mol. Life Sci. 2006, 63, 1945–1961. [Google Scholar] [CrossRef] [PubMed]
- Bradl, M.; Lassmann, H. Oligodendrocytes: Biology and pathology. Acta Neuropathol. 2010, 119, 37–53. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, J.; Delli-Bovi, L.C.; Hecht, J.; Folkerth, R.; Sheen, V.L. Generation of neural stem cells from discarded human fetal cortical tissue. J. Vis. Exp. 2011, 51, e2681. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duan, X.; Kang, E.; Liu, C.Y.; Ming, G.L.; Song, H. Development of neural stem cell in the adult brain. Curr. Opin. Neurobiol. 2008, 18, 108–115. [Google Scholar] [CrossRef] [Green Version]
- Yan, Y.; Shin, S.; Jha, B.S.; Liu, Q.; Sheng, J.; Li, F.; Zhan, M.; Davis, J.; Bharti, K.; Zeng, X.; et al. Efficient and rapid derivation of primitive neural stem cells and generation of brain subtype neurons from human pluripotent stem cells. Stem Cells Transl. Med. 2013, 2, 862–870. [Google Scholar] [CrossRef]
- Yu, K.R.; Shin, J.H.; Kim, J.J.; Koog, M.G.; Lee, J.Y.; Choi, S.W.; Kim, H.S.; Seo, Y.; Lee, S.; Shin, T.H.; et al. Rapid and Efficient Direct Conversion of Human Adult Somatic Cells into Neural Stem Cells by HMGA2/let-7b. Cell Rep. 2015, 10, 441–452. [Google Scholar] [CrossRef]
- Löhle, M.; Hermann, A.; Glass, H.; Kempe, A.; Schwarz, S.C.; Kim, J.B.; Poulet, C.; Ravens, U.; Schwarz, J.; Schöler, H.R.; et al. Differentiation efficiency of induced pluripotent stem cells depends on the number of reprogramming factors. Stem Cells 2012, 30, 570–579. [Google Scholar] [CrossRef]
- Fu, L.; Zhu, L.; Huang, Y.; Lee, T.D.; Forman, S.J.; Shih, C.C. Derivation of neural stem cells from mesenchymal stemcells: Evidence for a bipotential stem cell population. Stem Cells Dev. 2008, 17, 1109–1121. [Google Scholar] [CrossRef] [Green Version]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.; Keating, A.; Prockop, D.; Horwitz, E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef]
- Bajek, A.; Olkowska, J.; Drewa, T. Mesenchymal stem cells as a therapeutic tool in tissue and organ regeneration. Postepy Hig. Med. Dosw. (Online) 2011, 65, 124–132. [Google Scholar] [CrossRef] [PubMed]
- Pittenger, M.F.; Mackay, A.M.; Beck, S.C.; Jaiswal, R.K.; Douglas, R.; Mosca, J.D.; Moorman, M.A.; Simonetti, D.W.; Craig, S.; Marshak, D.R. Multilineage potential of adult human mesenchymal stem cells. Science 1999, 284, 143–147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zuk, P.A.; Zhu, M.; Ashjian, P.; De Ugarte, D.A.; Huang, J.I.; Mizuno, H.; Alfonso, Z.C.; Fraser, J.K.; Benhaim, P.; Hedrick, M.H. Human adipose tissue is a source of multipotent stem cells. Mol. Biol. Cell 2002, 13, 4279–4295. [Google Scholar] [CrossRef] [PubMed]
- Miao, Z.; Jin, J.; Chen, L.; Zhu, J.; Huang, W.; Zhao, J.; Qian, H.; Zhang, X. Isolation of mesenchymal stem cells from human placenta: Comparison with human bone marrow mesenchymal stem cells. Cell Biol. Int. 2006, 30, 681–687. [Google Scholar] [CrossRef] [PubMed]
- Int Anker, P.S.; Scherjon, S.A.; Kleijburg-van der Keur, C.; Noort, W.A.; Claas, F.H.; Willemze, R.; Fibbe, W.E.; Kanhai, H.H. Amniotic fluid as a novel source of mesenchymal stem cells for therapeutic transplantation. Blood 2003, 102, 1548–1549. [Google Scholar] [CrossRef]
- Arutyunyan, I.; Elchaninov, A.; Makarov, A.; Fatkhudinov, T. Umbilical Cord as Prospective Source for Mesenchymal Stem Cell-Based Therapy. Stem Cells Int. 2016, 2016, 6901286. [Google Scholar] [CrossRef] [Green Version]
- Divya, M.S.; Roshin, G.E.; Divya, T.S.; Rasheed, V.A.; Santhoshkumar, T.R.; Elizabeth, K.E.; James, J.; Pillai, R.M. Umbilical cord blood-derived mesenchymal stem cells consist of a unique population of progenitors co-expressing mesenchymal stem cell and neuronal markers capable of instantaneous neuronal differentiation. Stem Cell Res. Ther. 2012, 3, 57. [Google Scholar] [CrossRef] [Green Version]
- Hass, R.; Kasper, C.; Böhm, S.; Jacobs, R. Different populations and sources of human mesenchymal stem cells (MSC): A comparison of adult and neonatal tissue-derived MSC. Cell Commun. Signal. 2011, 9, 12. [Google Scholar] [CrossRef] [Green Version]
- Elahi, K.C.; Klein, G.; Avci-Adali, M.; Sievert, K.D.; MacNeil, S.; Aicher, W.K. Human Mesenchymal Stromal Cells from Different Sources Diverge in Their Expression of Cell Surface Proteins and Display Distinct Differentiation Patterns. Stem Cells Int. 2016, 2016, 5646384. [Google Scholar] [CrossRef] [Green Version]
- Kern, S.; Eichler, H.; Stoeve, J.; Klüter, H.; Bieback, K. Comparative analysis of mesenchymal stem cells from bone marrow, umbilical cord blood, or adipose tissue. Stem Cells 2006, 24, 1294–1301. [Google Scholar] [CrossRef]
- Kozlowska, U.; Krawczenko, A.; Futoma, K.; Jurek, T.; Rorat, M.; Patrzalek, D.; Klimczak, A. Similarities and differences between mesenchymal stem/progenitor cells derived from various human tissues. World J. Stem Cells 2019, 11, 347–374. [Google Scholar] [CrossRef] [PubMed]
- Jackson, L.; Jones, D.R.; Scotting, P.; Sottile, V. Adult mesenchymal stem cells: Differentiation potential and therapeutic applications. J. Postgrad. Med. 2007, 53, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Gimble, J.M.; Guilak, F.; Nuttall, M.E.; Sathishkumar, S.; Vidal, M.; Bunnell, B.A. In vitro Differentiation Potential of Mesenchymal Stem Cells. Transfus. Med. Hemother. 2008, 35, 228–238. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alexanian, A.R. An efficient method for generation of neural-like cells from adult human bone marrow-derived mesenchymal stem cells. Regen. Med. 2010, 5, 891–900. [Google Scholar] [CrossRef]
- Ma, K.; Fox, L.; Shi, G.; Shen, J.; Liu, Q.; Pappas, J.D.; Cheng, J.; Qu, T. Generation of neural stem cell-like cells from bone marrow-derived human mesenchymal stem cells. Neurol. Res. 2011, 33, 1083–1093. [Google Scholar] [CrossRef] [Green Version]
- Balasubramanian, S.; Thej, C.; Venugopal, P.; Priya, N.; Zakaria, Z.; Sundarraj, S.; Majumdar, A.S. Higher propensity of Wharton’s jelly derived mesenchymal stromal cells towards neuronal lineage in comparison to those derived from adipose and bone marrow. Cell Biol. Int. 2013, 37, 507–515. [Google Scholar] [CrossRef]
- Montanucci, P.; Basta, G.; Pescara, T.; Pennoni, I.; Di Giovanni, F.; Calafiore, R. New simple and rapid method for purification of mesenchymal stem cells from the human umbilical cord Wharton jelly. Tissue Eng. Part A 2011, 17, 2651–2661. [Google Scholar] [CrossRef] [PubMed]
- Anzalone, R.; Timoneri, F.; Rocca, G.L.; Iacono, M.L.; Amico, G.; Corsello, T.; Conaldi, P.G.; Zummo, G.; Farina, F. Isolation and phenotypical characterization of mesenchymal stem cells from the Wharton’s jelly of preterm human umbilical cord. Ital. J. Anat. Embryol. 2015, 119, 11. [Google Scholar]
- Hsieh, J.Y.; Wang, H.W.; Chang, S.J.; Liao, K.H.; Lee, I.H.; Lin, W.S.; Wu, C.H.; Lin, W.Y.; Cheng, S.M. Mesenchymal stem cells from human umbilical cord express preferentially secreted factors related to neuroprotection, neurogenesis, and angiogenesis. PLoS ONE 2013, 8, e72604. [Google Scholar] [CrossRef] [Green Version]
- Messerli, M.; Wagner, A.; Sager, R.; Mueller, M.; Baumann, M.; Surbek, D.V.; Schoeberlein, A. Stem cells from umbilical cord Wharton’s jelly from preterm birth have neuroglial differentiation potential. Reprod. Sci. 2013, 20, 1455–1464. [Google Scholar] [CrossRef]
- Leite, C.; Silva, N.T.; Mendes, S.; Ribeiro, A.; de Faria, J.P.; Lourenço, T.; dos Santos, F.; Andrade, P.Z.; Cardoso, C.M.; Vieira, M.; et al. Differentiation of human umbilical cord matrix mesenchymal stem cells into neural-like progenitor cells and maturation into an oligodendroglial-like lineage. PLoS ONE 2014, 9, e111059. [Google Scholar] [CrossRef] [PubMed]
- Jensen, J.B.; Parmar, M. Strengths and limitations of the neurosphere culture system. Mol. Neurobiol. 2006, 34, 153–161. [Google Scholar] [CrossRef]
- Chen, S.; Zhang, W.; Wang, J.M.; Duan, H.T.; Kong, J.H.; Wang, Y.X.; Dong, M.; Bi, X.; Song, J. Differentiation of isolated human umbilical cord mesenchymal stem cells into neural stem cells. Int. J. Ophthalmol. 2016, 9, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, K.E.; Weiss, M.L.; Mitchell, B.M.; Martin, P.; Davis, D.; Morales, L.; Helwig, B.; Beerenstrauch, M.; Abou-Easa, K.; Hildreth, T.; et al. Matrix cells from Wharton’s jelly form neurons and glia. Stem Cells 2003, 21, 50–60. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Ye, Z.; Wang, Y.; Deng, Y.; Zhou, G. Induction-dependent neural marker expression and electrophysiological characteristics of bone marrow mesenchymal stem cells that naturally express high levels of nestin. Chin. Sci. Bull. 2011, 56, 640–646. [Google Scholar] [CrossRef] [Green Version]
- Rafieemehr, H.; Kheirandish, M.; Soleimani, M. Improving the neuronal differentiation efficiency of umbilical cord blood-derived mesenchymal stem cells cultivated under appropriate conditions. Iran. J. Basic Med. Sci. 2015, 18, 1100–1106. [Google Scholar]
- Shahbazi, A.; Safa, M.; Alikarami, F.; Kargozar, S.; Asadi, M.H.; Joghataei, M.T.; Soleimani, M. Rapid Induction of Neural Differentiation in Human Umbilical Cord Matrix Mesenchymal Stem Cells by cAMP-elevating Agents. Int. J. Mol. Cell. Med. 2016, 5, 167–177. [Google Scholar]
- Ge, W.; Ren, C.; Duan, X.; Geng, D.; Zhang, C.; Liu, X.; Chen, H.; Wan, M.; Geng, R. Differentiation of mesenchymal stem cells into neural stem cells using cerebrospinal fluid. Cell Biochem. Biophys. 2015, 71, 449–455. [Google Scholar] [CrossRef]
- Guo, L.; Wang, L.; Yun-Peng, S.; Zhou, J.J.; Zhao, Z.; Li, D.P. Resveratrol Induces Differentiation of Human Umbilical Cord Mesenchymal Stem Cells into Neuron-Like Cells. Stem Cells Int. 2017, 2017, 1651325. [Google Scholar] [CrossRef]
- Sun, Y.; Pollard, S.; Conti, L.; Toselli, M.; Biella, G.; Parkin, G.; Willatt, L.; Falk, A.; Cattaneo, E.; Smith, A. Long-term tripotent differentiation capacity of human neural stem (NS) cells in adherent culture. Mol. Cell. Neurosci. 2008, 38, 245–258. [Google Scholar] [CrossRef]
- Park, J.; Lee, N.; Lee, J.; Choe, E.K.; Kim, M.K.; Byun, M.S.; Chon, M.W.; Kim, S.W.; Lee, C.J.; Kim, J.H.; et al. Small molecule-based lineage switch of human adipose-derived stem cells into neural stem cells and functional GABAergic neurons. Sci. Rep. 2017, 7, 10166. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chomczynski, P.; Sacchi, N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal. Biochem. 1987, 162, 156–159. [Google Scholar] [CrossRef]
- Griggs, L.A.; Hassan, N.T.; Malik, R.S.; Griffin, B.P.; Martinez, B.A.; Elmore, L.W.; Lemmon, C.A. Fibronectin fibrils regulate TGF-β1-induced Epithelial-Mesenchymal Transition. Matrix Biol. 2017, 60–61, 157–175. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.D.; Dusevich, V.; Feng, J.Q.; Manolagas, S.C.; Jilka, R.L. Extracellular matrix made by bone marrow cells facilitates expansion of marrow-derived mesenchymal progenitor cells and prevents their differentiation into osteoblasts. J. Bone Miner. Res. 2007, 22, 1943–1956. [Google Scholar] [CrossRef]
- Zeng, X.; Ma, Y.H.; Chen, Y.F.; Qiu, X.C.; Wu, J.L.; Ling, E.A.; Zeng, Y.S. Autocrine fibronectin from differentiating mesenchymal stem cells induces the neurite elongation in vitro and promotes nerve fiber regeneration in transected spinal cord injury. J. Biomed. Mater. Res. A 2016, 104, 1902–1911. [Google Scholar] [CrossRef] [Green Version]
- Bae, K.S.; Park, J.B.; Kim, H.S.; Kim, D.S.; Park, D.J.; Kang, S.J. Neuron-like differentiation of bone marrow-derived mesenchymal stem cells. Yonsei Med. J. 2011, 52, 401–412. [Google Scholar] [CrossRef] [Green Version]
- Zeng, R.; Wang, L.W.; Hu, Z.B.; Guo, W.T.; Wei, J.S.; Lin, H.; Sun, X.; Chen, L.X.; Yang, L.J. Differentiation of human bone marrow mesenchymal stem cells into neuron-like cells in vitro. Spine (Phila Pa 1976) 2011, 36, 997–1005. [Google Scholar] [CrossRef] [Green Version]
- Luo, H.; Xu, C.; Liu, Z.; Yang, L.; Hong, Y.; Liu, G.; Zhong, H.; Cai, X.; Lin, X.; Chen, X.; et al. Neural differentiation of bone marrow mesenchymal stem cells with human brain-derived neurotrophic factor gene-modified in functionalized self-assembling peptide hydrogel in vitro. J. Cell. Biochem. 2019, 120, 2828–2835. [Google Scholar] [CrossRef]
- Anghileri, E.; Marconi, S.; Pignatelli, A.; Cifelli, P.; Galié, M.; Sbarbati, A.; Krampera, M.; Belluzzi, O.; Bonetti, B. Neuronal differentiation potential of human adipose-derived mesenchymal stem cells. Stem Cells Dev. 2008, 17, 909–916. [Google Scholar] [CrossRef]
- Mostafavi, F.S.; Razavi, S.; Mardani, M.; Esfandiari, E.; Esfahani, H.Z.; Kazemi, M. Comparative Study of Microtubule-associated Protein-2 and Glial Fibrillary Acidic Proteins during Neural Induction of Human Bone Marrow Mesenchymal Stem Cells and Adipose-Derived Stem Cells. Int. J. Prev. Med. 2014, 5, 584–595. [Google Scholar]
- Qin, Y.; Zhou, C.; Wang, N.; Yang, H.; Gao, W.Q. Conversion of Adipose Tissue-Derived Mesenchymal Stem Cells to Neural Stem Cell-Like Cells by a Single Transcription Factor, Sox2. Cell. Reprogram. 2015, 17, 221–226. [Google Scholar] [CrossRef] [PubMed]
- Moon, M.Y.; Kim, H.J.; Choi, B.Y.; Sohn, M.; Chung, T.N.; Suh, S.W. Zinc Promotes Adipose-Derived Mesenchymal Stem Cell Proliferation and Differentiation towards a Neuronal Fate. Stem Cells Int. 2018, 2018, 5736535. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yan, Z.J.; Hu, Y.Q.; Zhang, H.T.; Zhang, P.; Xiao, Z.Y.; Sun, X.L.; Cai, Y.Q.; Hu, C.C.; Xu, R.X. Comparison of the neural differentiation potential of human mesenchymal stem cells from amniotic fluid and adult bone marrow. Cell. Mol. Neurobiol. 2013, 33, 465–475. [Google Scholar] [CrossRef]
- Li, M.; Zhao, W.; Gao, Y.; Hao, P.; Shang, J.; Duan, H.; Yang, Z.; Li, X. Differentiation of Bone Marrow Mesenchymal Stem Cells into Neural Lineage Cells Induced by bFGF-Chitosan Controlled Release System. Biomed. Res. Int. 2019, 2019, 5086297. [Google Scholar] [CrossRef]
- Hermann, A.; Gastl, R.; Liebau, S.; Popa, M.O.; Fiedler, J.; Boehm, B.O.; Maisel, M.; Lerche, H.; Schwarz, J.; Brenner, R.; et al. Efficient generation of neural stem cell-like cells from adult human bone marrow stromal cells. J. Cell Sci. 2004, 117, 4411–4422. [Google Scholar] [CrossRef] [Green Version]
- Park, S.B.; Seo, K.W.; So, A.Y.; Seo, M.S.; Yu, K.R.; Kang, S.K.; Kang, K.S. SOX2 has a crucial role in the lineage determination and proliferation of mesenchymal stem cells through Dickkopf-1 and c-MYC. Cell Death Differ. 2012, 19, 534–545. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Izadpanah, R.; Trygg, C.; Patel, B.; Kriedt, C.; Dufour, J.; Gimble, J.M.; Bunnell, B.A. Biologic properties of mesenchymal stem cells derived from bone marrow and adipose tissue. J. Cell. Biochem. 2006, 99, 1285–1297. [Google Scholar] [CrossRef] [Green Version]
- Jang, S.; Cho, H.H.; Cho, Y.B.; Park, J.S.; Jeong, H.S. Functional neural differentiation of human adipose tissue-derived stem cells using bFGF and forskolin. BMC Cell. Biol. 2010, 11, 25. [Google Scholar] [CrossRef] [Green Version]
- Tondreau, T.; Lagneaux, L.; Dejeneffe, M.; Massy, M.; Mortier, C.; Delforge, A.; Bron, D. Bone marrow-derived mesenchymal stem cells already express specific neural proteins before any differentiation. Differentiation 2004, 72, 319–326. [Google Scholar] [CrossRef]
- Ying, Q.L.; Stavridis, M.; Griffiths, D.; Li, M.; Smith, A. Conversion of embryonic stem cells into neuroectodermal precursors in adherent monoculture. Nat. Biotechnol. 2003, 21, 183–186. [Google Scholar] [CrossRef]
- Conti, L.; Pollard, S.M.; Gorba, T.; Reitano, E.; Toselli, M.; Biella, G.; Sun, Y.; Sanzone, S.; Ying, Q.L.; Cattaneo, E.; et al. Niche-independent symmetrical self-renewal of a mammalian tissue stem cell. PLoS Biol. 2005, 3, e283. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pollard, S.M.; Conti, L.; Sun, Y.; Goffredo, D.; Smith, A. Adherent neural stem (NS) cells from fetal and adult forebrain. Cereb. Cortex 2006, 16 (Suppl. 1), i112–i120. [Google Scholar] [CrossRef] [PubMed]
- Neuhuber, B.; Gallo, G.; Howard, L.; Kostura, L.; Mackay, A.; Fischer, I. Reevaluation of in vitro differentiation protocols for bone marrow stromal cells: Disruption of actin cytoskeleton induces rapid morphological changes and mimics neuronal phenotype. J. Neurosci. Res. 2004, 77, 192–204. [Google Scholar] [CrossRef] [PubMed]
- Ciccolini, F.; Mandl, C.; Hölzl-Wenig, G.; Kehlenbach, A.; Hellwig, A. Prospective isolation of late development multipotent precursors whose migration is promoted by EGFR. Dev. Biol. 2005, 284, 112–125. [Google Scholar] [CrossRef] [Green Version]
- Hernández, R.; Jiménez-Luna, C.; Perales-Adán, J.; Perazzoli, G.; Melguizo, C.; Prados, J. Differentiation of Human Mesenchymal Stem Cells towards Neuronal Lineage: Clinical Trials in Nervous System Disorders. Biomol. Ther. 2020, 28, 34–44. [Google Scholar] [CrossRef]
- Han, Z.C.; Du, W.J.; Han, Z.B.; Liang, L. New insights into the heterogeneity and functional diversity of human mesenchymal stem cells. Biomed. Mater. Eng. 2017, 28, S29–S45. [Google Scholar] [CrossRef] [PubMed]
- Urrutia, D.N.; Caviedes, P.; Mardones, R.; Minguell, J.J.; Vega-Letter, A.M.; Jofre, C.M. Comparative study of the neural differentiation capacity of mesenchymal stromal cells from different tissue sources: An approach for their use in neural regeneration therapies. PLoS ONE 2019, 14, e0213032. [Google Scholar] [CrossRef] [PubMed]
- Raponi, E.; Agenes, F.; Delphin, C.; Assard, N.; Baudier, J.; Legraverend, C.; Deloulme, J.C. S100B expression defines a state in which GFAP-expressing cells lose their neural stem cell potential and acquire a more mature developmental stage. Glia 2007, 55, 165–177. [Google Scholar] [CrossRef] [Green Version]
- Saulite, L.; Vavers, E.; Zvejniece, L.; Dambrova, M.; Riekstina, U. The Differentiation of Skin Mesenchymal Stem Cells Towards a Schwann Cell Phenotype: Impact of Sigma-1 Receptor Activation. Mol. Neurobiol. 2018, 55, 2840–2850. [Google Scholar] [CrossRef]
- Eng, L.F.; Ghirnikar, R.S.; Lee, Y.L. Glial fibrillary acidic protein: GFAP-thirty-one years (1969-2000). Neurochem. Res. 2000, 25, 1439–1451. [Google Scholar] [CrossRef]
- van Strien, M.E.; Sluijs, J.A.; Reynolds, B.A.; Steindler, D.A.; Aronica, E.; Hol, E.M. Isolation of neural progenitor cells from the human adult subventricular zone based on expression of the cell surface marker CD271. Stem Cells Transl. Med. 2014, 3, 470–480. [Google Scholar] [CrossRef] [PubMed]
- D’Andrea, M.R.; Howanski, R.J.; Saller, C.F. MAP2 IHC detection: A marker of antigenicity in CNS tissues. Biotech. Histochem. 2017, 92, 363–373. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Han, S.S.; Wu, Y.; Tuohy, T.M.; Xue, H.; Cai, J.; Back, S.A.; Sherman, L.S.; Fischer, I.; Rao, M.S. CD44 expression identifies astrocyte-restricted precursor cells. Dev. Biol. 2004, 276, 31–46. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Su, W.; Foster, S.C.; Xing, R.; Feistel, K.; Olsen, R.H.; Acevedo, S.F.; Raber, J.; Sherman, L.S. CD44 Transmembrane Receptor and Hyaluronan Regulate Adult Hippocampal Neural Stem Cell Quiescence and Differentiation. J. Biol. Chem. 2017, 292, 4434–4445. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- L Ramos, T.; Sánchez-Abarca, L.I.; Muntión, S.; Preciado, S.; Puig, N.; López-Ruano, G.; Hernández-Hernández, Á.; Redondo, A.; Ortega, R.; Rodríguez, C.; et al. MSC surface markers (CD44, CD73, and CD90) can identify human MSC-derived extracellular vesicles by conventional flow cytometry. Cell Commun. Signal. 2016, 14, 2. [Google Scholar] [CrossRef] [Green Version]
- Weible, M.W.; Chan-Ling, T. Phenotypic characterization of neural stem cells from human fetal spinal cord: Synergistic effect of LIF and BMP4 to generate astrocytes. Glia 2007, 55, 1156–1168. [Google Scholar] [CrossRef]
- Behnan, J.; Stangeland, B.; Langella, T.; Finocchiaro, G.; Tringali, G.; Meling, T.R.; Murrell, W. Identification and characterization of a new source of adult human neural progenitors. Cell Death Dis. 2017, 8, e2991. [Google Scholar] [CrossRef] [Green Version]
- Zheng, Y.; Huang, C.; Liu, F.; Lin, H.; Yang, X.; Zhang, Z. Comparison of the neuronal differentiation abilities of bone marrow-derived and adipose tissue-derived mesenchymal stem cells. Mol. Med. Rep. 2017, 16, 3877–3886. [Google Scholar] [CrossRef] [Green Version]
- Mohammad, M.H.; Al-Shammari, A.M.; Al-Juboory, A.A.; Yaseen, N.Y. Characterization of neural stemness status through the neurogenesis process for bone marrow mesenchymal stem cells. Stem Cells Cloning 2016, 9, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Perl, K.; Ushakov, K.; Pozniak, Y.; Yizhar-Barnea, O.; Bhonker, Y.; Shivatzki, S.; Geiger, T.; Avraham, K.B.; Shamir, R. Reduced changes in protein compared to mRNA levels across non-proliferating tissues. BMC Genom. 2017, 18, 305. [Google Scholar] [CrossRef]
- Brockmann, R.; Beyer, A.; Heinisch, J.J.; Wilhelm, T. Posttranscriptional expression regulation: What determines translation rates? PLoS Comput. Biol. 2007, 3, e57. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.; Beyer, A.; Aebersold, R. On the Dependency of Cellular Protein Levels on mRNA Abundance. Cell 2016, 165, 535–550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brauer, M.J.; Huttenhower, C.; Airoldi, E.M.; Rosenstein, R.; Matese, J.C.; Gresham, D.; Boer, V.M.; Troyanskaya, O.G.; Botstein, D. Coordination of growth rate, cell cycle, stress response, and metabolic activity in yeast. Mol. Biol. Cell 2008, 19, 352–367. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kellerman, K.A.; Mattson, D.M.; Duncan, I. Mutations affecting the stability of the fushi tarazu protein of Drosophila. Genes Dev. 1990, 4, 1936–1950. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matic, I.; Antunovic, M.; Brkic, S.; Josipovic, P.; Mihalic, K.C.; Karlak, I.; Ivkovic, A.; Marijanovic, I. Expression of OCT-4 and SOX-2 in Bone Marrow-Derived Human Mesenchymal Stem Cells during Osteogenic Differentiation. Open Access Maced. J. Med. Sci. 2016, 4, 9–16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hosseini, S.M.; Vasaghi, A.; Nakhlparvar, N.; Roshanravan, R.; Talaei-Khozani, T.; Razi, Z. Differentiation of Wharton’s jelly mesenchymal stem cells into neurons in alginate scaffold. Neural Regen. Res. 2015, 10, 1312–1316. [Google Scholar] [CrossRef]



| Cell Type | Specific Marker | Description |
|---|---|---|
| NSCs | nestin | class VI cytoskeletal intermediate filament protein, essential for stem cell survival, self-renewal and proliferation, a pivotal regulator of cell differentiation and migration, can also be expressed in glial cells [24] |
| PAX6 | nuclear transcription factor, controls NSCs proliferation and plays a crucial role in neuronal fate determination [25] | |
| SOX1 | transcription factor from Sox family, the earliest marker of neural fate decision [26] | |
| SOX2 | transcription factor from Sox family, pluripotency marker, regulates neural differentiation [27], can be expressed on MSCs isolated from human Wharton’s jelly [28] | |
| NPCs | GFAP | intermediate filament protein that serves as a holder of astrocyte mechanical strength, involved in astrocyte communication, expressed also in NPCs [22] |
| PSA-NCAM | cell adhesion molecule involved in regulating several steps of adult neurogenesis [29] | |
| SOX1 | see above | |
| SOX2 | see above | |
| nestin | see above | |
| immature neurons | DCX | protein that facilitates microtubule polymerization, a marker of migrating neuroblasts and immature neurons [30] |
| βIII tubulin | microtubule protein, expressed in immature neurons and differentiated neurons [31] | |
| neurons | MAP2 | protein involved in the growth and assembly of microtubules, required for neurite formation in neurons [32] |
| astrocytes | GFAP | see above |
| S100β | calcium-binding protein β, involved in the cell cycle and differentiation [22] | |
| oligodendrocytes | MBP | protein involved in the myelination of neurons in the CNS [33] |
| Olig1, Olig2 | oligodendrocyte transcription factors [22] | |
| GalC | galactosylceramide expressed in differentiating oligodendrocyte precursor cells and mature oligodendrocytes [34] |
| Positive Markers | Negative Markers |
|---|---|
| CD90 CD105 CD73 | CD34 CD11b CD19 CD45 HLA-DR |
| Specific Marker | Antibody | Concentration | Company (Cat. No) |
|---|---|---|---|
| HSCs marker | mouse anti-human CD45 | 1:500 | BD Pharmingen (San Jose, CA, USA) (Cat. No 555480) |
| MSCs marker | mouse anti-human CD90 | 1:500 | BD Pharmingen (San Jose, CA, USA) (Cat. No 555593) |
| proliferation marker | mouse anti-human Ki67 | 1:500 | Merck (Darmstadt, Germany) (Cat. No MAB4190) |
| NSCs/NPCs marker | mouse anti-human nestin | 1:250 | Merck (Darmstadt, Germany) (Cat. No MAB5326) |
| neuronal marker | mouse anti-human MAP2 | 1:250 | Invitrogen (Carlsbad, CA, USA) (Cat. No MA1-25044) |
| astrocyte marker | mouse anti-human S100β | 1:250 | Invitrogen (Carlsbad, CA, USA) (Cat. No MA1-25005) |
| NSCs/NPCs marker | rabbit anti-human SOX2 | 1:250 | Invitrogen (Carlsbad, CA, USA) (Cat. No PA1-16968) |
| NPCs, astrocyte marker | rabbit anti-human GFAP | 1:250 | Invitrogen (Carlsbad, CA, USA) (Cat. No PA1-10019) |
| AF488 goat anti-mouse IgG1 | 1:500 | Invitrogen (Carlsbad, CA, USA) (Cat. No 845809) | |
| AF555 goat anti-mouse IgG1 | 1:500 | Invitrogen (Carlsbad, CA, USA) (Cat. No 982413) | |
| AF568 goat anti-rabbit IgG (H+L) | 1:500 | Invitrogen (Carlsbad, CA, USA) (Cat. No 948491) |
| Staining Variant | Antigens Stained |
|---|---|
| I | Nestin, SOX1, Ki67 |
| II | Nestin, DCX, Ki67, SOX2 |
| III | DCX, nestin, SOX1, CD44 |
| IV | DCX, GFAP, SOX1, CD44 |
| Gene Name | Primer | Sequence |
|---|---|---|
| β-actin | Forward | CAGAAGGATTCCTATGTGGGC |
| Reverse | GAGGGCATACCCCTCGTAGAT | |
| fibronectin | Forward Reverse | GAGATCAGTGGGATAAGCAGCA CCTCTTCATGACGCTTGTGGA |
| SOX2 | Forward Reverse | CAGGAGAACCCCAAGATGC GCAGCCGCTTAGCCTCG |
| nestin | Forward Reverse | CAGCTGGCGCACCTCAAGATG AGGGAAGTTGGGCTCAGGACTGG |
| MAP2 | Forward Reverse | GGGCCTTTTCTTTGAAATCTAGTTT CAAATGTGGCTCTCTGAAGAACA |
| GFAP | Forward Reverse | CTGTTGCCAGAGATGGAGGTT TCATCGCTCAGGAGGTCCTT |
| MBP | Forward Reverse | CTGGGCAGCTGTTAGAGTCC TGGAGCAAAGGTTTGGTGTC |
| Population Number | Population Name | Phenotype | Staining Variant |
|---|---|---|---|
| P1 | neural stem and progenitor cells | Nestin+ SOX1+ | I |
| P2 | Ki67+ SOX1+ | Ki67+ SOX1+ | |
| P3 | glia and neural stem and progenitor cells | Nestin+ DCX- | II, III |
| P4 | Ki67+ SOX2+ | Ki67+ SOX2+ | II |
| P5 | glial cells | CD44+ SOX1+ | III, IV |
| P6 | astrocytes | GFAP+ DCX- | IV |
| P7 | DCX+ | DCX+ |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kruminis-Kaszkiel, E.; Osowski, A.; Bejer-Oleńska, E.; Dziekoński, M.; Wojtkiewicz, J. Differentiation of Human Mesenchymal Stem Cells from Wharton’s Jelly Towards Neural Stem Cells Using a Feasible and Repeatable Protocol. Cells 2020, 9, 739. https://doi.org/10.3390/cells9030739
Kruminis-Kaszkiel E, Osowski A, Bejer-Oleńska E, Dziekoński M, Wojtkiewicz J. Differentiation of Human Mesenchymal Stem Cells from Wharton’s Jelly Towards Neural Stem Cells Using a Feasible and Repeatable Protocol. Cells. 2020; 9(3):739. https://doi.org/10.3390/cells9030739
Chicago/Turabian StyleKruminis-Kaszkiel, Ewa, Adam Osowski, Ewa Bejer-Oleńska, Mariusz Dziekoński, and Joanna Wojtkiewicz. 2020. "Differentiation of Human Mesenchymal Stem Cells from Wharton’s Jelly Towards Neural Stem Cells Using a Feasible and Repeatable Protocol" Cells 9, no. 3: 739. https://doi.org/10.3390/cells9030739
APA StyleKruminis-Kaszkiel, E., Osowski, A., Bejer-Oleńska, E., Dziekoński, M., & Wojtkiewicz, J. (2020). Differentiation of Human Mesenchymal Stem Cells from Wharton’s Jelly Towards Neural Stem Cells Using a Feasible and Repeatable Protocol. Cells, 9(3), 739. https://doi.org/10.3390/cells9030739





