Calcium- and Voltage-Dependent Dual Gating ANO1 is an Intrinsic Determinant of Repolarization in Rod Bipolar Cells of the Mouse Retina
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Standards
2.2. Preparation of Animals and Tissue Samples
2.3. Preparation of Solutions and Drugs
2.4. Data Analysis
3. Results
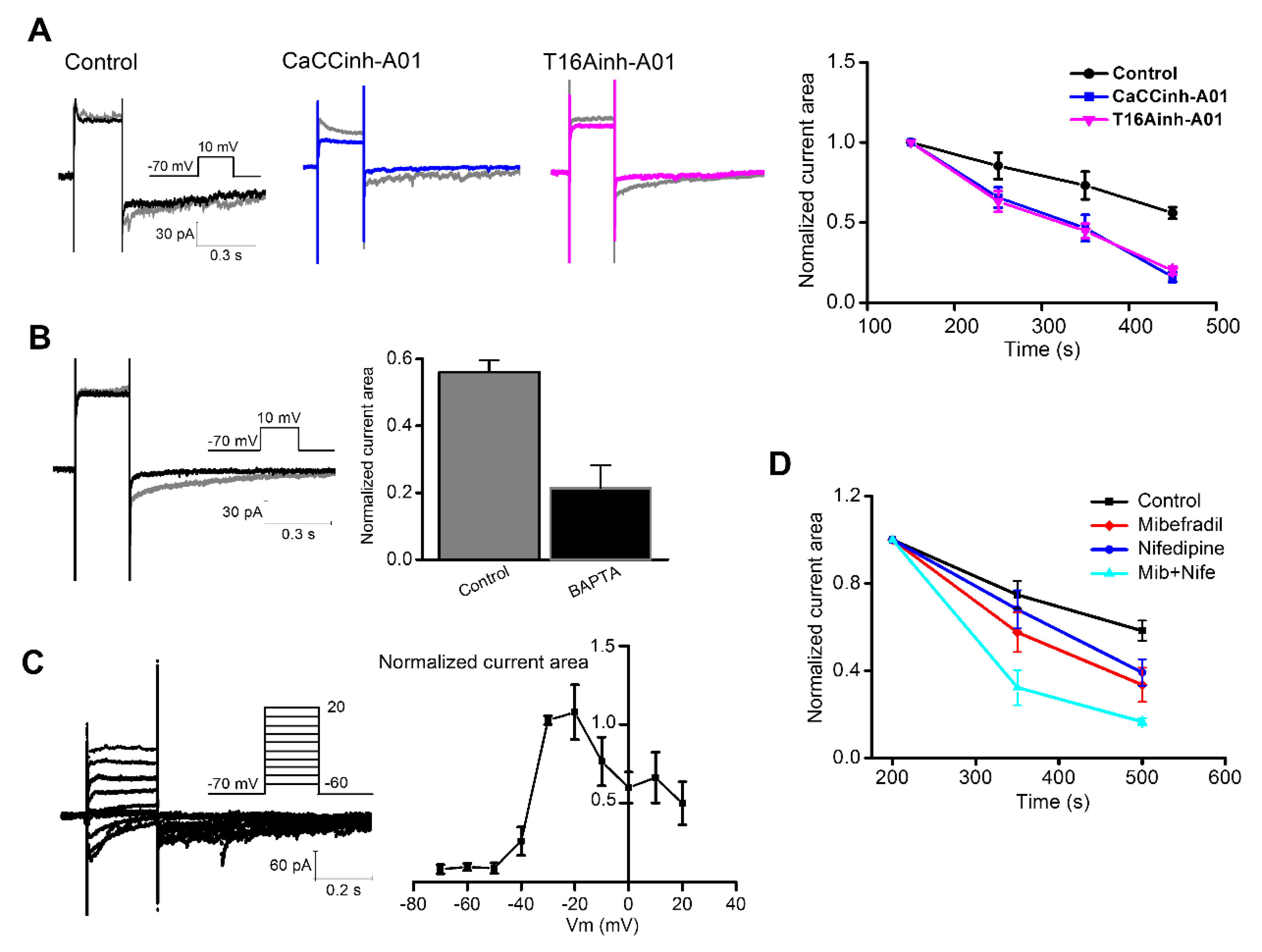
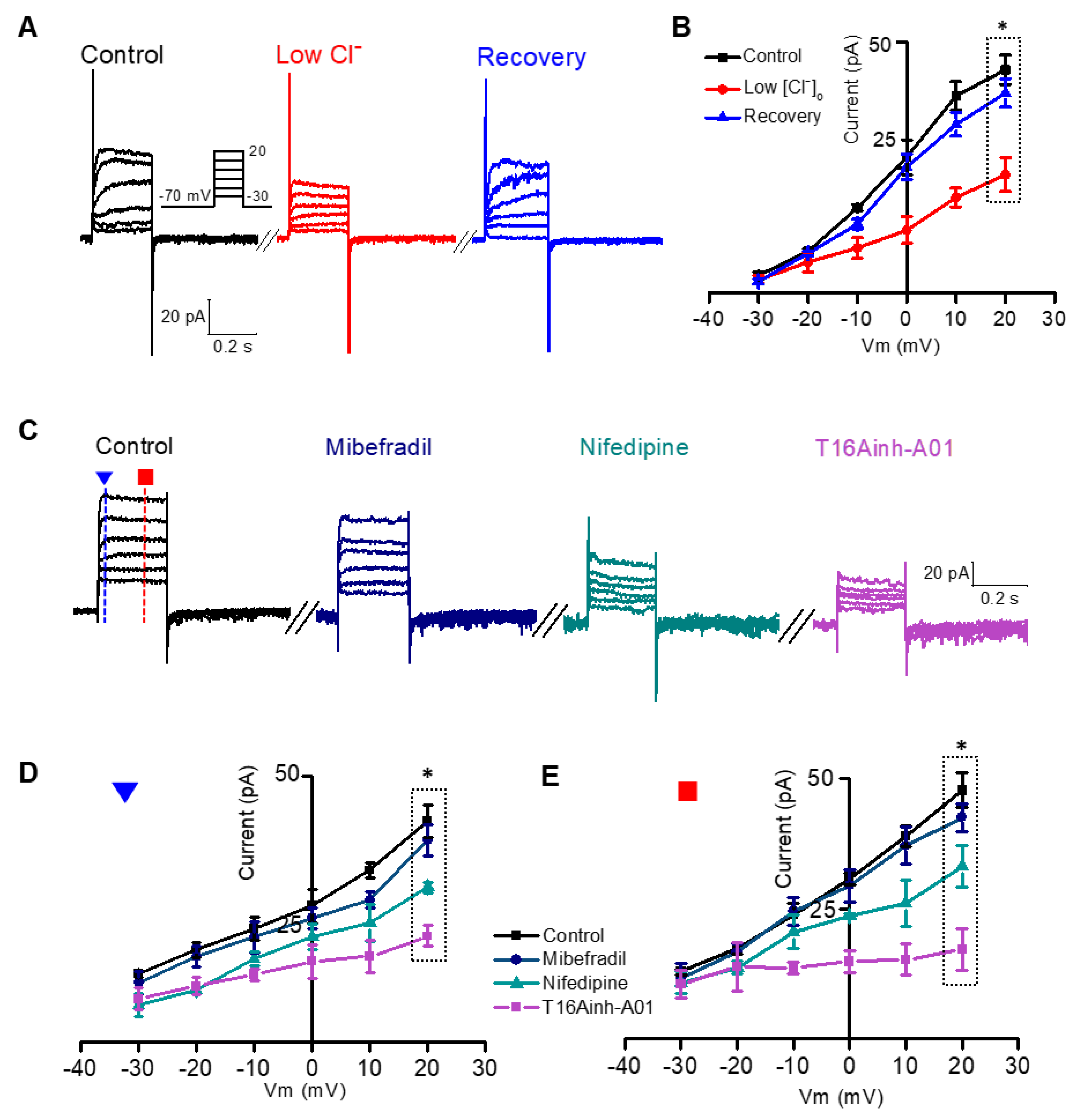
3.1. Relationship Between Ca2+-Dependent Characteristics of the ANO1 Current and VGCC
3.2. Isolation of the Voltage-Dependent and Outward Component of the ANO1 Current
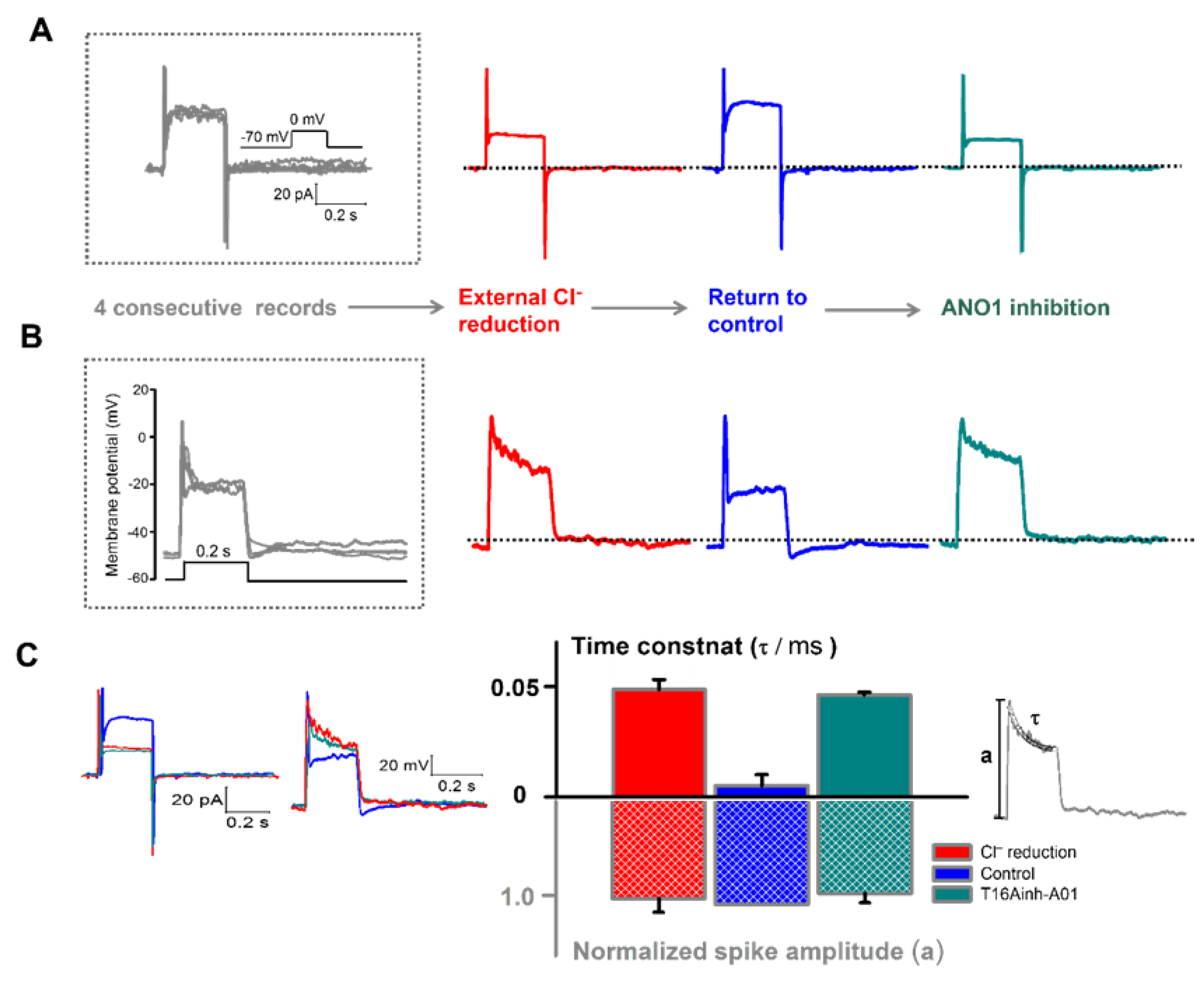
3.3. Involvement of the Voltage-Dependent Component of ANO1 in the Rapid Decay of the Initial Phase of Waveform
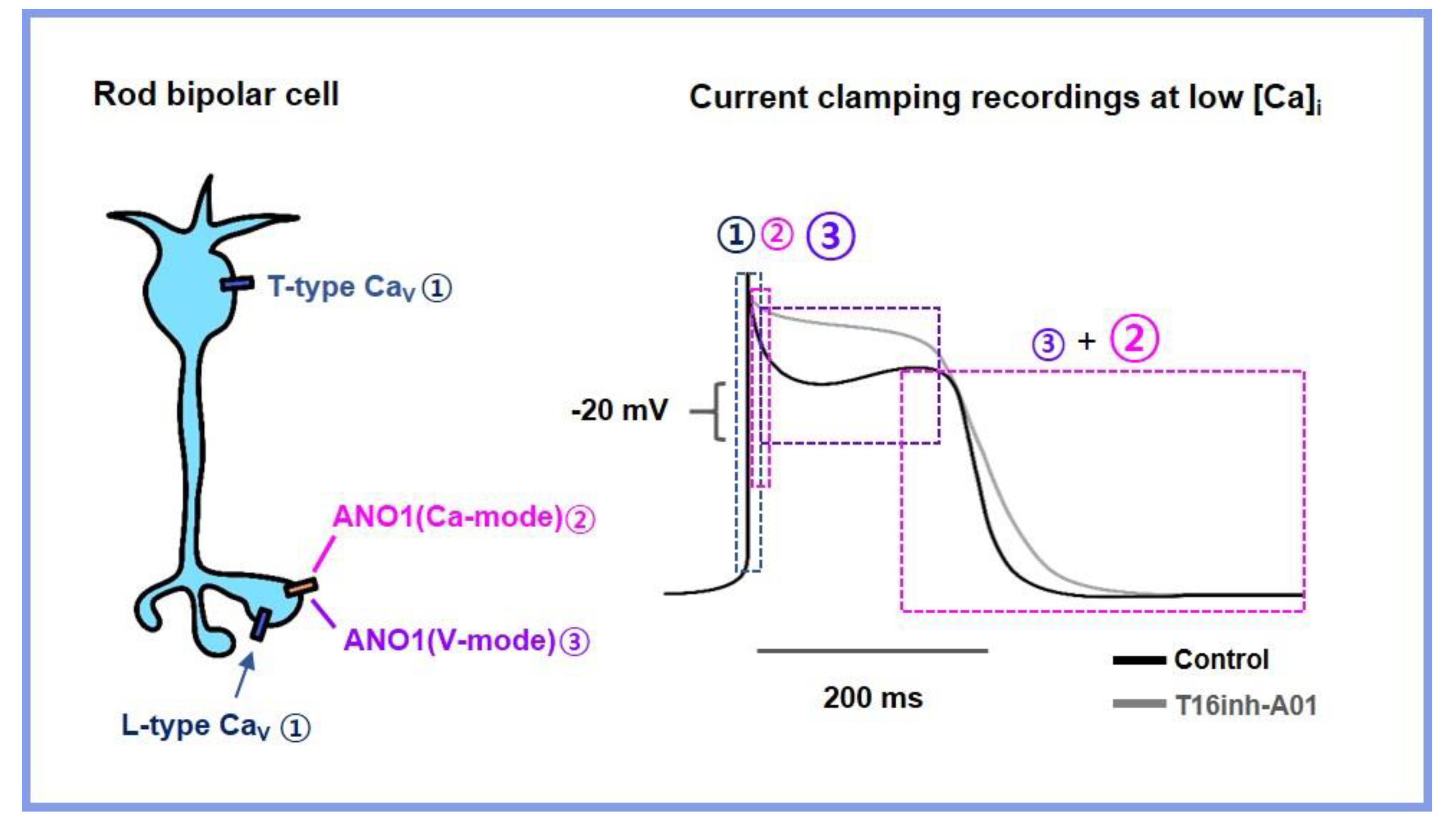
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Masland, R.H. Processing and encoding of visual information in the retina. Curr. Opin. Neurobiol. 1996, 6, 467–474. [Google Scholar] [CrossRef]
- Boycott, B.; Wässle, H. Parallel processing in the mammalian retina: The Proctor Lecture. Invest. Ophthalmol. Vis. Sci. 1999, 40, 1313–1327. [Google Scholar] [PubMed]
- Wässle, H. Parallel processing in the mammalian retina. Nat. Rev. Neurosci. 2004, 5, 747–757. [Google Scholar] [CrossRef] [PubMed]
- Dowling, J.E.; Werblin, F.S. Organization of retina of the mudpuppy, Necturus maculosus. I. Synaptic structure. J. Neurophysiol. 1969, 32, 315–338. [Google Scholar] [CrossRef]
- Saito, T.; Kondo, H.; Toyoda, J.I. Ionic mechanisms of two types of on-center bipolar cells in the carp retina. I. The responses to central illumination. J. Gen. Physiol. 1979, 73, 73–90. [Google Scholar] [CrossRef]
- Ashmore, J.F.; Falk, G. Responses of rod bipolar cells in the dark-adapted retina of the dogfish, Scyliorhinus canicula. J. Physiol. 1980, 300, 115–150. [Google Scholar] [CrossRef]
- Duvvuri, U.; Shiwarski, D.J.; Xiao, D.; Bertrand, C.; Huang, X.; Edinger, R.S.; Rock, J.R.; Harfe, B.D.; Henson, B.J.; Kunzelmann, K.; et al. TMEM16A induces MAPK and contributes directly to tumorigenesis and cancer progression. Cancer Res. 2012, 72, 3270–3281. [Google Scholar] [CrossRef]
- Large, W.A.; Wang, Q. Characteristics and physiological role of the Ca(2+)-activated Cl- conductance in smooth muscle. Am. J. Physiol. 1996, 271, 435–454. [Google Scholar] [CrossRef]
- Romanenko, V.G.; Catalan, M.A.; Brown, D.A.; Putzier, I.; Hartzell, H.C.; Marmorstein, A.D.; Gonzalez-Begne, M.; Rock, J.R.; Harfe, B.D.; Melvin, J.E. Tmem16A encodes the Ca2+-activated Cl- channel in mouse submandibular salivary gland acinar cells. J. Biol. Chem. 2010, 285, 12990–13001. [Google Scholar] [CrossRef]
- Hartzell, C.; Putzier, I.; Arreola, J. Calcium-activated chloride channels. Annu. Rev. Physiol. 2005, 67, 719–758. [Google Scholar] [CrossRef]
- Melvin, J.E.; Yule, D.; Shuttleworth, T.; Begenisich, T. Regulation of fluid and electrolyte secretion in salivary gland acinar cells. Annu. Rev. Physiol. 2005, 67, 445–469. [Google Scholar] [CrossRef] [PubMed]
- Ha, G.E.; Cheong, E. Calcium-activated chloride channels: A new target to control the spiking pattern of neurons. BMB Rep. 2017, 50, 109–110. [Google Scholar] [CrossRef] [PubMed]
- Jeon, J.H.; Paik, S.S.; Chun, M.H.; Oh, U.; Kim, I.B. Presynaptic Localization and Possible Function of Calcium-Activated Chloride Channel Anoctamin 1 in the Mammalian Retina. PLoS ONE 2013, 8, e67989. [Google Scholar] [CrossRef] [PubMed]
- Lalonde, M.R.; Kelly, M.E.; Barnes, S. Calcium-activated chloride channels in the retina. Channels (Austin) 2008, 2, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Caputo, A.; Piano, I.; Demontis, G.C.; Bacchi, N.; Casarosa, S.; Della Santina, L.; Gargini, C. TMEM16A is associated with voltage-gated calcium channels in mouse retina and its function is disrupted upon mutation of the auxiliary α2δ4 subunit. Front. Cell. Neurosci. 2015, 9, 422. [Google Scholar] [CrossRef] [PubMed]
- Stohr, H.; Heisig, J.B.; Benz, P.M.; Schoberl, S.; Milenkovic, V.M.; Strauss, O.; Aartsen, W.M.; Wijnholds, J.; Weber, B.H.; Schulz, H.L. TMEM16B, a novel protein with calcium-dependent chloride channel activity, associates with a presynaptic protein complex in photoreceptor terminals. J. Neurosci. 2009, 29, 6809–6818. [Google Scholar] [CrossRef] [PubMed]
- Barnes, S.; Hille, B. Ionic channels of the inner segment of tiger salamander cone photoreceptors. J. Gen. Physiol. 1989, 94, 719–743. [Google Scholar] [CrossRef]
- Endeman, D.; Fahrenfort, I.; Sjoerdsma, T.; Steijaert, M.; Ten Eikelder, H.; Kamermans, M. Chloride currents in cones modify feedback from horizontal cells to cones in goldfish retina. J. Physiol. 2012, 590, 5581–5595. [Google Scholar] [CrossRef]
- Kraaij, D.A.; Spekreijse, H.; Kamermans, M. The nature of surround-induced depolarizing responses in goldfish cones. J. Gen. Physiol. 2000, 115, 3–16. [Google Scholar] [CrossRef]
- Okada, T.; Horiguchi, H.; Tachibana, M. Ca(2+)-dependent Cl- current at the presynaptic terminals of goldfish retinal bipolar cells. Neurosci. Res. 1995, 23, 297–303. [Google Scholar] [CrossRef]
- Stephan, A.B.; Shum, E.Y.; Hirsh, S.; Cygnar, K.D.; Reisert, J.; Zhao, H. ANO2 is the cilial calcium-activated chloride channel that may mediate olfactory amplification. Proc. Natl. Acad. Sci. USA 2009, 106, 11776. [Google Scholar] [CrossRef] [PubMed]
- Billig, G.M.; Pál, B.; Fidzinski, P.; Jentsch, T.J. Ca2+-activated Cl− currents are dispensable for olfaction. Nat. Neurosci. 2011, 14, 763–769. [Google Scholar] [CrossRef] [PubMed]
- Amjad, A.; Hernandez-Clavijo, A.; Pifferi, S.; Maurya, D.K.; Boccaccio, A.; Franzot, J.; Rock, J.; Menini, A. Conditional knockout of TMEM16A/anoctamin1 abolishes the calcium-activated chloride current in mouse vomeronasal sensory neurons. J. Gen. Physiol. 2015, 145, 285–301. [Google Scholar] [CrossRef] [PubMed]
- Dauner, K.; Möbus, C.; Frings, S.; Möhrlen, F. Targeted expression of anoctamin calcium-activated chloride channels in rod photoreceptor terminals of the rodent retina. Invest. Ophthalmol. Vis. Sci. 2013, 54, 3126–3136. [Google Scholar] [CrossRef]
- Jeon, J.H.; Park, J.W.; Lee, J.W.; Jeong, S.W.; Yeo, S.W.; Kim, I.B. Expression and immunohistochemical localization of TMEM16A/anoctamin 1, a calcium-activated chloride channel in the mouse cochlea. Cell. Tissue. Res. 2011, 345, 223–230. [Google Scholar] [CrossRef]
- Zhang, X.-D.; Lee, J.-H.; Lv, P.; Chen, W.C.; Kim, H.J.; Wei, D.; Wang, W.; Sihn, C.-R.; Doyle, K.J.; Rock, J.R.; et al. Etiology of distinct membrane excitability in pre- and posthearing auditory neurons relies on activity of Cl- channel TMEM16A. Proc. Natl. Acad. Sci. USA 2015, 112, 2575–2580. [Google Scholar] [CrossRef]
- Cherkashin, A.P.; Kolesnikova, A.S.; Tarasov, M.V.; Romanov, R.A.; Rogachevskaja, O.A.; Bystrova, M.F.; Kolesnikov, S.S. Expression of calcium-activated chloride channels Ano1 and Ano2 in mouse taste cells. Pflugers. Arch. 2016, 468, 305–319. [Google Scholar] [CrossRef]
- Cho, H.; Yang, Y.D.; Lee, J.; Lee, B.; Kim, T.; Jang, Y.; Back, S.K.; Na, H.S.; Harfe, B.D.; Wang, F.; et al. The calcium-activated chloride channel anoctamin 1 acts as a heat sensor in nociceptive neurons. Nat. Neurosci. 2012, 15, 1015–1021. [Google Scholar] [CrossRef]
- Huang, W.C.; Xiao, S.; Huang, F.; Harfe, B.D.; Jan, Y.N.; Jan, L.Y. Calcium-activated chloride channels (CaCCs) regulate action potential and synaptic response in hippocampal neurons. Neuron 2012, 74, 179–192. [Google Scholar] [CrossRef]
- Ha, G.E.; Lee, J.; Kwak, H.; Song, K.; Kwon, J.; Jung, S.-Y.; Hong, J.; Chang, G.-E.; Hwang, E.M.; Shin, H.-S.; et al. The Ca2+-activated chloride channel anoctamin-2 mediates spike-frequency adaptation and regulates sensory transmission in thalamocortical neurons. Nat. Commun. 2016, 7, 13791. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, Z.; Xiao, S.; Tien, J.; Le, S.; Le, T.; Jan, L.Y.; Yang, H. Inferior Olivary TMEM16B Mediates Cerebellar Motor Learning. Neuron 2017, 95, 1103–1111. [Google Scholar] [CrossRef] [PubMed]
- Neureither, F.; Ziegler, K.; Pitzer, C.; Frings, S.; Möhrlen, F. Impaired Motor Coordination and Learning in Mice Lacking Anoctamin 2 Calcium-Gated Chloride Channels. Cerebellum (London, England) 2017, 16, 929–937. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Schmelzeisen, S.; Parthier, D.; Frings, S.; Möhrlen, F. Anoctamin Calcium-Activated Chloride Channels May Modulate Inhibitory Transmission in the Cerebellar Cortex. PLoS ONE 2015, 10, e0142160. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.J.; Jeon, J.H.; Chun, D.I.; Yeo, S.W.; Kim, I.-B. Anoctamin 1 expression in the mouse auditory brainstem. Cell. Tissue. Res. 2014, 357, 563–569. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.S.; Jeon, J.H.; Lee, S.H.; Paik, S.S.; Kim, I.-B. Immunohistochemical Localization of Anoctamin 1 in the Mouse Cerebellum. Appl. Microsc. 2018, 48, 110–116. [Google Scholar] [CrossRef]
- Xiao, Q.; Yu, K.; Perez-Cornejo, P.; Cui, Y.; Arreola, J.; Hartzell, H.C. Voltage- and calcium-dependent gating of TMEM16A/Ano1 chloride channels are physically coupled by the first intracellular loop. Proc. Natl. Acad. Sci. USA 2011, 108, 8891. [Google Scholar] [CrossRef]
- Cruz-Rangel, S.; De Jesus-Perez, J.J.; Contreras-Vite, J.A.; Perez-Cornejo, P.; Hartzell, H.C.; Arreola, J. Gating modes of calcium-activated chloride channels TMEM16A and TMEM16B. J. Physiol. 2015, 593, 5283–5298. [Google Scholar] [CrossRef]
- Contreras-Vite, J.A.; Cruz-Rangel, S.; De Jesus-Perez, J.J.; Figueroa, I.A.; Rodriguez-Menchaca, A.A.; Perez-Cornejo, P.; Hartzell, H.C.; Arreola, J. Revealing the activation pathway for TMEM16A chloride channels from macroscopic currents and kinetic models. Pflugers. Arch. 2016, 468, 1241–1257. [Google Scholar] [CrossRef]
- Yang, Y.D.; Cho, H.; Koo, J.Y.; Tak, M.H.; Cho, Y.; Shim, W.S.; Park, S.P.; Lee, J.; Lee, B.; Kim, B.M.; et al. TMEM16A confers receptor-activated calcium-dependent chloride conductance. Nature 2008, 455, 1210–1215. [Google Scholar] [CrossRef]
- Zhou, Z.-Y.; Wan, Q.-F.; Thakur, P.; Heidelberger, R. Capacitance Measurements in the Mouse Rod Bipolar Cell Identify a Pool of Releasable Synaptic Vesicles. J. Neurophysiol. 2006, 96, 2539–2548. [Google Scholar] [CrossRef]
- Namkung, W.; Phuan, P.W.; Verkman, A.S. TMEM16A inhibitors reveal TMEM16A as a minor component of calcium-activated chloride channel conductance in airway and intestinal epithelial cells. J. Biol. Chem. 2011, 286, 2365–2374. [Google Scholar] [CrossRef] [PubMed]
- Seo, Y.; Lee, H.K.; Park, J.; Jeon, D.-K.; Jo, S.; Jo, M.; Namkung, W. Ani9, A Novel Potent Small-Molecule ANO1 Inhibitor with Negligible Effect on ANO2. PLoS ONE 2016, 11, e0155771. [Google Scholar] [CrossRef] [PubMed]
- de la Villa, P.; Vaquero, C.F.; Kaneko, A. Two types of calcium currents of the mouse bipolar cells recorded in the retinal slice preparation. Eur. J. Neurosci. 1998, 10, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, A.; Pinto, L.H.; Tachibana, M. Transient calcium current of retinal bipolar cells of the mouse. J. Physiol. 1989, 410, 613–629. [Google Scholar] [CrossRef]
- Takayama, Y.; Uta, D.; Furue, H.; Tominaga, M. Pain-enhancing mechanism through interaction between TRPV1 and anoctamin 1 in sensory neurons. Proc. Natl. Acad. Sci. USA 2015, 112, 5213–5218. [Google Scholar] [CrossRef]
- Keckeis, S.; Reichhart, N.; Roubeix, C.; Strauss, O. Anoctamin2 (TMEM16B) forms the Ca(2+)-activated Cl(-) channel in the retinal pigment epithelium. Exp. Eye. Res. 2017, 154, 139–150. [Google Scholar] [CrossRef]
- Protti, D.A.; Llano, I. Calcium Currents and Calcium Signaling in Rod Bipolar Cells of Rat Retinal Slices. J. Neurosci. 1998, 18, 3715. [Google Scholar] [CrossRef]
- Wan, Q.-F.; Heidelberger, R. Synaptic release at mammalian bipolar cell terminals. Vis. Neurosci. 2011, 28, 109–119. [Google Scholar] [CrossRef]
- Mercer, A.J.; Rabl, K.; Riccardi, G.E.; Brecha, N.C.; Stella, S.L., Jr.; Thoreson, W.B. Location of release sites and calcium-activated chloride channels relative to calcium channels at the photoreceptor ribbon synapse. J. Neurophysiol. 2011, 105, 321–335. [Google Scholar] [CrossRef]
- Pan, Z.-H.; Hu, H.-J.; Perring, P.; Andrade, R. T-Type Ca2+ Channels Mediate Neurotransmitter Release in Retinal Bipolar Cells. Neuron 2001, 32, 89–98. [Google Scholar] [CrossRef]
- Yu, K.; Duran, C.; Qu, Z.; Cui, Y.-Y.; Hartzell, H.C. Explaining calcium-dependent gating of anoctamin-1 chloride channels requires a revised topology. Circ. Res. 2012, 110, 990–999. [Google Scholar] [CrossRef] [PubMed]
- Manoury, B.; Tamuleviciute, A.; Tammaro, P. TMEM16A/Anoctamin 1 protein mediates calcium-activated chloride currents in pulmonary arterial smooth muscle cells. J. Physiol. 2010, 588, 2305–2314. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Birnbaumer, L.; Singh, B.B. TRPC1 regulates calcium-activated chloride channels in salivary gland cells. J. Cell. Physiol. 2015, 230, 2848–2856. [Google Scholar] [CrossRef] [PubMed]
- Peters, C.J.; Gilchrist, J.M.; Tien, J.; Bethel, N.P.; Qi, L.; Chen, T.; Wang, L.; Jan, Y.N.; Grabe, M.; Jan, L.Y. The Sixth Transmembrane Segment Is a Major Gating Component of the TMEM16A Calcium-Activated Chloride Channel. Neuron 2018, 97, 1063–1077. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Simms, J.; Peters, C.J.; Tynan-La Fontaine, M.; Li, K.; Gill, T.M.; Jan, Y.N.; Jan, L.Y. TMEM16B Calcium-Activated Chloride Channels Regulate Action Potential Firing in Lateral Septum and Aggression in Male Mice. J. Neurosci. 2019, 39, 7102–7117. [Google Scholar] [CrossRef] [PubMed]
- Lipin, M.Y.; Vigh, J. Calcium spike-mediated digital signaling increases glutamate output at the visual threshold of retinal bipolar cells. J. Neurophysiol. 2014, 113, 550–566. [Google Scholar] [CrossRef]
- Protti, D.A.; Flores-Herr, N.; von Gersdorff, H. Light Evokes Ca2+ Spikes in the Axon Terminal of a Retinal Bipolar Cell. Neuron 2000, 25, 215–227. [Google Scholar] [CrossRef]
- Vergara, C.; Latorre, R.; Marrion, N.V.; Adelman, J.P. Calcium-activated potassium channels. Curr. Opin. Neurobiol. 1998, 8, 321–329. [Google Scholar] [CrossRef]
- Faber, E.S.L.; Sah, P. Calcium-Activated Potassium Channels: Multiple Contributions to Neuronal Function. Neuroscientist 2003, 9, 181–194. [Google Scholar] [CrossRef]
- Grimes, W.N.; Li, W.; Chávez, A.E.; Diamond, J.S. BK channels modulate pre- and postsynaptic signaling at reciprocal synapses in retina. Nat. Neurosci. 2009, 12, 585–592. [Google Scholar] [CrossRef]
- Tanimoto, N.; Sothilingam, V.; Euler, T.; Ruth, P.; Seeliger, M.W.; Schubert, T. BK Channels Mediate Pathway-Specific Modulation of Visual Signals in the in vivo Mouse Retina. J. Neurosci. 2012, 32, 4861. [Google Scholar] [CrossRef] [PubMed]
- Klocker, N.; Oliver, D.; Ruppersberg, J.P.; Knaus, H.G.; Fakler, B. Developmental expression of the small-conductance Ca(2+)-activated potassium channel SK2 in the rat retina. Mol. Cell. Neurosci. 2001, 17, 514–520. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paik, S.-S.; Park, Y.S.; Kim, I.-B. Calcium- and Voltage-Dependent Dual Gating ANO1 is an Intrinsic Determinant of Repolarization in Rod Bipolar Cells of the Mouse Retina. Cells 2020, 9, 543. https://doi.org/10.3390/cells9030543
Paik S-S, Park YS, Kim I-B. Calcium- and Voltage-Dependent Dual Gating ANO1 is an Intrinsic Determinant of Repolarization in Rod Bipolar Cells of the Mouse Retina. Cells. 2020; 9(3):543. https://doi.org/10.3390/cells9030543
Chicago/Turabian StylePaik, Sun-Sook, Yong Soo Park, and In-Beom Kim. 2020. "Calcium- and Voltage-Dependent Dual Gating ANO1 is an Intrinsic Determinant of Repolarization in Rod Bipolar Cells of the Mouse Retina" Cells 9, no. 3: 543. https://doi.org/10.3390/cells9030543
APA StylePaik, S.-S., Park, Y. S., & Kim, I.-B. (2020). Calcium- and Voltage-Dependent Dual Gating ANO1 is an Intrinsic Determinant of Repolarization in Rod Bipolar Cells of the Mouse Retina. Cells, 9(3), 543. https://doi.org/10.3390/cells9030543





