Soluble CD5 and CD6: Lymphocytic Class I Scavenger Receptors as Immunotherapeutic Agents
Abstract
1. Introduction
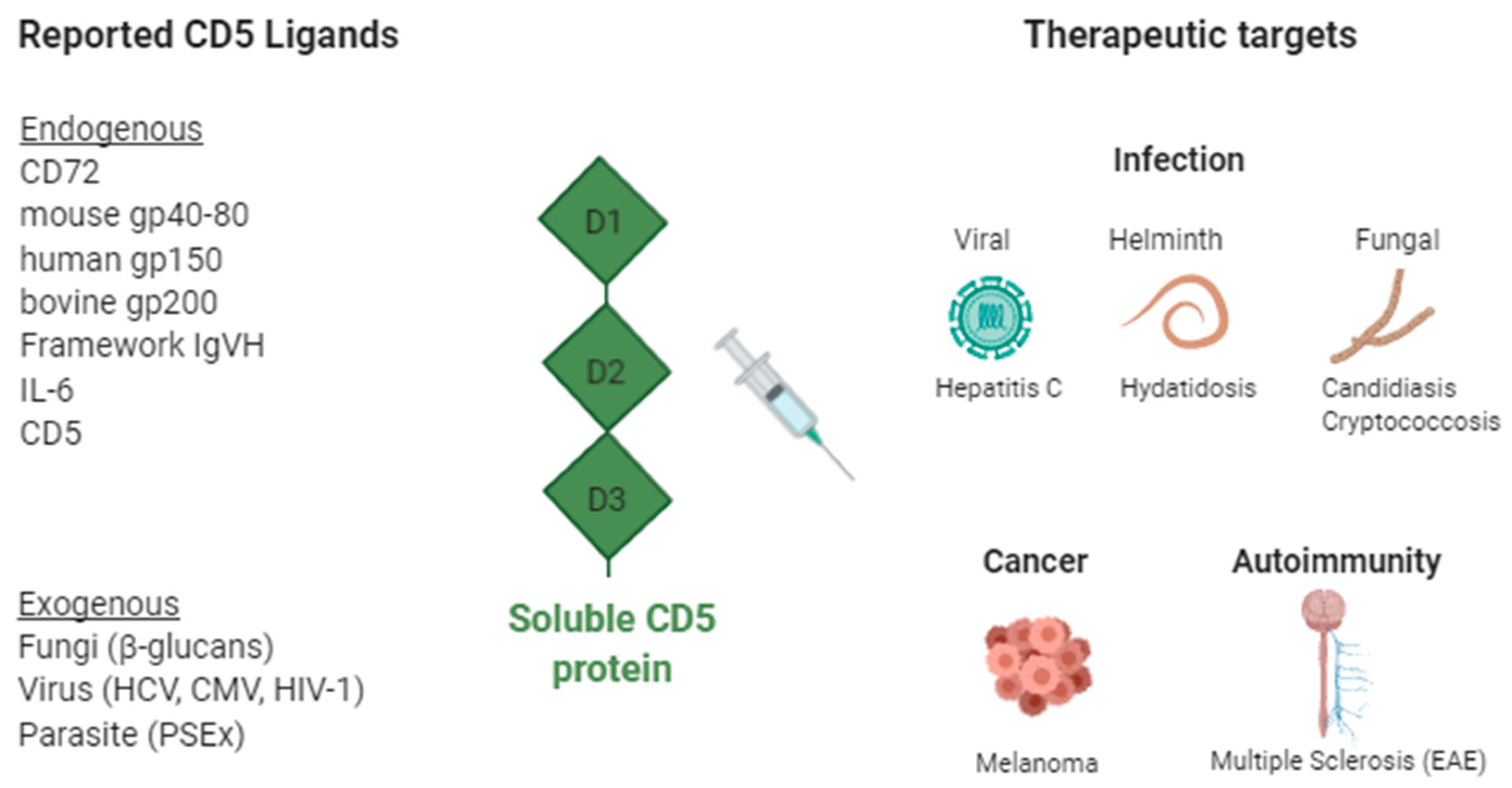
2. The Lymphocyte Receptor CD5
2.1. Soluble CD5 as Therapeutic Agent in Infection
2.2. Soluble CD5 as Therapeutic Agent in Cancer
2.3. Soluble CD5 as Therapeutic Agent in Autoimmunity
3. The Lymphocyte Receptor CD6
3.1. Soluble CD6 as Therapeutic Agent in Infection
3.2. Soluble CD6 as Therapeutic Agent in Cancer
3.3. Soluble CD6 as Therapeutic Agent in Autoimmunity
4. Concluding Remarks
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Martinez, V.G.; Moestrup, S.K.; Holmskov, U.; Mollenhauer, J.; Lozano, F. The conserved scavenger receptor cysteine-rich superfamily in therapy and diagnosis. Pharmacol. Rev. 2011, 63, 967–1000. [Google Scholar] [CrossRef]
- Pombinho, R.; Sousa, S.; Cabanes, D. Scavenger Receptors: Promiscuous Players during Microbial Pathogenesis. Crit. Rev. Microbiol. 2018, 44, 685–700. [Google Scholar] [CrossRef]
- PrabhuDas, M.R.; Baldwin, C.L.; Bollyky, P.L.; Bowdish, D.M.E.; Drickamer, K.; Febbraio, M.; Herz, J.; Kobzik, L.; Krieger, M.; Loike, J.; et al. A Consensus Definitive Classification of Scavenger Receptors and Their Roles in Health and Disease. J. Immunol. 2017, 198, 3775–3789. [Google Scholar] [CrossRef]
- Sarrias, M.R.; Grønlund, J.; Padilla, O.; Madsen, J.; Holmskov, U.; Lozano, F. The Scavenger Receptor Cysteine-Rich (SRCR) domain: An ancient and highly conserved protein module of the innate immune system. Crit. Rev. Immunol. 2004, 24, 1–37. [Google Scholar] [CrossRef]
- Gimferrer, I.; Farnós, M.; Calvo, M.; Mittelbrunn, M.; Enrich, C.; Sánchez-Madrid, F.; Vives, J.; Lozano, F. The accessory molecules CD5 and CD6 associate on the membrane of lymphoid T cells. J. Biol. Chem. 2003, 278, 8564–8571. [Google Scholar] [CrossRef]
- Beyers, A.D.; Spruyt, L.L.; Williams, A.F. Multimolecular associations of the T-cell antigen receptor. Trends Cell Biol. 1992, 2, 253–255. [Google Scholar] [CrossRef]
- Lankester, A.C.; van Schijndel, G.M.; Cordell, J.L.; van Noesel, C.J.; van Lier, R.A. CD5 is associated with the human B cell antigen receptor complex. Eur. J. Immunol. 1994, 24, 812–816. [Google Scholar] [CrossRef]
- Gimferrer, I.; Calvo, M.; Mittelbrunn, M.; Farnós, M.; Sarrias, M.R.; Enrich, C.; Vives, J.; Sánchez-Madrid, F.; Lozano, F. Relevance of CD6-Mediated Interactions in T Cell Activation and Proliferation. J. Immunol. 2004, 173, 2262–2270. [Google Scholar] [CrossRef]
- Cho, J.H.; Sprent, J. TCR tuning of T cell subsets. Immunol. Rev. 2018, 283, 129–137. [Google Scholar] [CrossRef]
- Alizadeh, M.; Safarzadeh, A.; Hoseini, S.A.; Piryaei, R.; Mansoori, B.; Hajiasgharzadeh, K.; Baghbanzadeh, A.; Baradaran, B. The potentials of immune checkpoints for the treatment of blood malignancies. Crit. Rev. Oncol. Hematol. 2020, 153, 103031. [Google Scholar] [CrossRef]
- Mourglia-Ettlin, G.; Miles, S.; Velasco-De-Andrés, M.; Armiger-Borràs, N.; Cucher, M.; Dematteis, S.; Lozano, F. The ectodomains of the lymphocyte scavenger receptors CD5 and CD6 interact with tegumental antigens from Echinococcus granulosus sensu lato and protect mice against secondary cystic echinococcosis. PLoS Negl. Trop. Dis. 2018, 12, e0006891. [Google Scholar] [CrossRef]
- Vera, J.; Fenutria, R.; Canadas, O.; Figueras, M.; Mota, R.; Sarrias, M.-R.; Williams, D.L.; Casals, C.; Yelamos, J.; Lozano, F. The CD5 ectodomain interacts with conserved fungal cell wall components and protects from zymosan-induced septic shock-like syndrome. Proc. Natl. Acad. Sci. USA 2009, 106, 1506–1511. [Google Scholar] [CrossRef]
- Sarrias, M.-R.; Farnos, M.; Mota, R.; Sanchez-Barbero, F.; Ibanez, A.; Gimferrer, I.; Vera, J.; Fenutria, R.; Casals, C.; Yelamos, J.; et al. CD6 binds to pathogen-associated molecular patterns and protects from LPS-induced septic shock. Proc. Natl. Acad. Sci. USA 2007, 104, 11724–11729. [Google Scholar] [CrossRef]
- Martínez-Florensa, M.; Consuegra-Fernández, M.; Martínez, V.G.; Cañadas, O.; Armiger-Borràs, N.; Bonet-Roselló, L.; Farrán, A.; Vila, J.; Casals, C.; Lozano, F. Targeting of key pathogenic factors from gram-positive bacteria by the soluble ectodomain of the scavenger-like lymphocyte receptor CD6. J. Infect. Dis. 2014, 209, 1077–1086. [Google Scholar] [CrossRef][Green Version]
- Lenz, L.L. CD5 sweetens lymphocyte responses. Proc. Natl. Acad. Sci. USA 2009, 106, 1303–1304. [Google Scholar] [CrossRef]
- Bernard, A.; Boumsell, L.; Dausset, J.; Milstein, C.; Schlossman, S.F. Leucocyte Typing; Bernard, A., Boumsell, L., Dausset, J., Milstein, C., Schlossman, S.F., Eds.; Springer: Berlin/Heidelberg, Germany, 1984; ISBN 978-3-642-68859-1. [Google Scholar]
- Tarakhovsky, A.; Kanner, S.B.; Hombach, J.; Ledbetter, J.A.; Müller, W.; Killeen, N.; Rajewsky, K. A role for CD5 in TCR-mediated signal transduction and thymocyte selection. Science 1995, 269, 535–537. [Google Scholar] [CrossRef]
- Enyindah-Asonye, G.; Li, Y.; Ruth, J.H.; Spassov, D.S.; Hebron, K.E.; Zijlstra, A.; Moasser, M.M.; Wang, B.; Singer, N.G.; Cui, H.; et al. CD318 is a ligand for CD6. Proc. Natl. Acad. Sci. USA 2017, 114, E6912–E6921. [Google Scholar] [CrossRef]
- Orta-Mascaró, M.; Consuegra-Fernández, M.; Carreras, E.; Roncagalli, R.; Carreras-Sureda, A.; Alvarez, P.; Girard, L.; Simões, I.; Martínez-Florensa, M.; Aranda, F.; et al. CD6 modulates thymocyte selection and peripheral T cell homeostasis. J. Exp. Med. 2016, 213, 1387–1397. [Google Scholar] [CrossRef]
- Reinherz, E.L.; Kung, P.C.; Goldstein, G.; Schlossman, S.F. A monoclonal antibody with selective reactivity with functionally mature human thymocytes and all peripheral human T cells. J. Immunol. 1979, 123, 1312–1317. [Google Scholar]
- Ledbetter, J.A.; Rouse, R.V.; Micklem, H.S.; Herzenberg, L.A. T cell subsets defined by expression of Lyt-1,2,3 and Thy-1 antigens. Two-parameter immunofluorescence and cytotoxicity analysis with monoclonal antibodies modifies current views. J. Exp. Med. 1980, 152, 280–295. [Google Scholar] [CrossRef]
- Berland, R.; Wortis, H.H. Origins and functions of B-1 cells with notes on the role of CD5. Annu. Rev. Immunol 2002, 20, 253–300. [Google Scholar] [CrossRef] [PubMed]
- Yanaba, K.; Bouaziz, J.-D.; Matsushita, T.; Tsubata, T.; Tedder, T.F. The Development and Function of Regulatory B Cells Expressing IL-10 (B10 Cells) Requires Antigen Receptor Diversity and TLR Signals. J. Immunol. 2009, 182, 7459–7472. [Google Scholar] [CrossRef] [PubMed]
- Borrello, M.A.; Palis, J.; Phipps, R.P. The relationship of CD5+ B lymphocytes to macrophages: Insights from normal biphenotypic B/macrophage cells. Int. Rev. Immunol. 2001, 20, 137–155. [Google Scholar] [CrossRef] [PubMed]
- Moreau, M.F.; Thibaud, J.L.; Miled, L.B.; Chaussepied, M.; Baumgartner, M.; Davis, W.C.; Minoprio, P.; Langsley, G. Theileria annulata in CD5(+) macrophages and B1 B cells. Infect. Immun. 1999, 67, 6678–6682. [Google Scholar] [CrossRef]
- Gogolin-Ewens, K.; Meeusen, E.; Lee, C.-S.; Brandon, M. Expression of CD5, a lymphocyte surface antigen on the endothelium of blood vessels. Eur. J. Immunol. 1989, 19, 935–938. [Google Scholar] [CrossRef]
- De Bernardis, F.; Lucciarini, R.; Boccanera, M.; Amantini, C.; Arancia, S.; Morrone, S.; Mosca, M.; Cassone, A.; Santoni, G. Phenotypic and Functional Characterization of Vaginal Dendritic Cells in a Rat Model of Candida albicans Vaginitis. Infect. Immun. 2006, 74, 4282–4294. [Google Scholar] [CrossRef]
- Korenfeld, D.; Gorvel, L.; Munk, A.; Man, J.; Schaffer, A.; Tung, T.; Mann, C.; Klechevsky, E. A type of human skin dendritic cell marked by CD5 is associated with the development of inflammatory skin disease. JCI Insight 2017, 2, e96101. [Google Scholar] [CrossRef]
- Li, H.; Burgueño-Bucio, E.; Xu, S.; Das, S.; Olguin-Alor, R.; Elmets, C.A.; Athar, M.; Raman, C.; Soldevila, G.; Xu, H. CD5 on dendritic cells regulates CD4+ and CD8+ T cell activation and induction of immune responses. PLoS ONE 2019, 14, e0222301. [Google Scholar] [CrossRef]
- Ordoñez-Rueda, D.; Lozano, F.; Sarukhan, A.; Raman, C.; Garcia-Zepeda, E.A.; Soldevila, G. Increased numbers of thymic and peripheral CD4 + CD25 + Foxp3 + cells in the absence of CD5 signaling. Eur. J. Immunol. 2009, 39, 2233–2247. [Google Scholar] [CrossRef]
- Lozano, F.; Simarro, M.; Calvo, J.; Vila, J.M.; Padilla, O.; Bowen, M.A.; Campbell, K.S. CD5 signal transduction: Positive or negative modulation of antigen receptor signaling. Crit. Rev. Immunol. 2000, 20, 347–358. [Google Scholar] [CrossRef]
- Soldevila, G.; Raman, C.; Lozano, F. The immunomodulatory properties of the CD5 lymphocyte receptor in health and disease. Curr. Opin. Immunol. 2011, 23, 310–318. [Google Scholar] [CrossRef] [PubMed]
- Freitas, C.; Johnson, D.; Weber, K. T Cell Calcium Signaling Regulation by the Co-Receptor CD5. Int. J. Mol. Sci. 2018, 19, 1295. [Google Scholar] [CrossRef] [PubMed]
- Voisinne, G.; Gonzalez de Peredo, A.; Roncagalli, R. CD5, an Undercover Regulator of TCR Signaling. Front. Immunol. 2018, 9, 2900. [Google Scholar] [CrossRef] [PubMed]
- Burgueño-Bucio, E.; Mier-Aguilar, C.A.; Soldevila, G. The multiple faces of CD5. J. Leukoc. Biol. 2019, 105, 891–904. [Google Scholar] [CrossRef] [PubMed]
- Bhandoola, A.; Sambandam, A.; Allman, D.; Meraz, A.; Schwarz, B. Early T Lineage Progenitors: New Insights, but Old Questions Remain. J. Immunol. 2003, 171, 5653–5658. [Google Scholar] [CrossRef]
- Calvo, J.; Places, L.; Espinosa, G.; Padilla, O.; Vilà, J.M.; Villamor, N.; Ingelmo, M.; Gallart, T.; Vives, J.; Font, J.; et al. Identification of a natural soluble form of human CD5. Tissue Antigens 1999, 54, 128–137. [Google Scholar] [CrossRef]
- Ramos-Casals, M.; Font, J.; García-Carrasco, M.; Calvo, J.; Places, L.; Padilla, O.; Cervera, R.; Bowen, M.A.; Lozano, F.; Ingelmo, M. High circulating levels of soluble scavenger receptors (sCD5 and sCD6) in patients with primary Sjögren’s syndrome. Rheumatology 2001, 40, 1056–1059. [Google Scholar] [CrossRef]
- Aibar, J.; Martínez-Florensa, M.; Castro, P.; Carrasco, E.; Escoda-Ferran, C.; Fernández, S.; Butjosa, M.; Hernández, C.; Rinaudo, M.; Lozano, F.; et al. Pattern of soluble CD5 and CD6 lymphocyte receptors in critically ill patients with septic syndromes. J. Crit. Care 2015, 30, 914–919. [Google Scholar] [CrossRef]
- Jamin, C.; Magadur, G.; Lamour, A.; Mackenzie, L.; Lydyard, P.; Katsikis, P.; Youinou, P. Cell-free CD5 in patients with rheumatic diseases. Immunol. Lett. 1992, 31, 79–83. [Google Scholar] [CrossRef]
- Noh, G.W.; Lee, K.Y. Circulating Soluble CD5 in Atopic Dermatitis. Mol. Cells 1998, 8, 618–622. [Google Scholar]
- Van de Velde, H.; von Hoegen, I.; Luo, W.; Parnes, J.R.; Thielemans, K. The B-cell surface protein CD72/Lyb-2 is the ligand for CDS. Nature 1991, 351, 662–665. [Google Scholar] [CrossRef] [PubMed]
- Pospisil, R.; Silverman, G.J.; Marti, G.E.; Aruffo, A.; Bowen, M.A.; Mage, R.G. CD5 is A potential selecting ligand for B-cell surface immunoglobulin: A possible role in maintenance and selective expansion of normal and malignant B cells. Leuk. Lymphoma 2000, 36, 353–365. [Google Scholar] [CrossRef] [PubMed]
- Haas, K.M.; Estes, D.M. The identification and characterization of a ligand for bovine CD5. J. Immunol. 2001, 166, 3158–3166. [Google Scholar] [CrossRef] [PubMed]
- Biancone, L.; Bowen, M.A.; Lim, A.; Aruffo, A.; Andres, G.; Stamenkovic, I. Identification of a novel inducible cell-surface ligand of CD5 on activated lymphocytes. J. Exp. Med. 1996, 184, 811–819. [Google Scholar] [CrossRef]
- Bikah, G.; Carey, J.; Ciallella, J.R.; Tarakhovsky, A.; Bondada, S. CD5-mediated negative regulation of antigen receptor-induced growth signals in B-1 B cells. Science 1996, 274, 1906–1909. [Google Scholar] [CrossRef]
- Calvo, J.; Places, L.; Padilla, O.; Vilà, J.M.; Vives, J.; Bowen, M.A.; Lozano, F. Interaction of recombinant and natural soluble CD5 forms with an alternative cell surface ligand. Eur. J. Immunol. 1999, 29, 2119–2129. [Google Scholar] [CrossRef]
- Masuda, K.; Kishimoto, T. CD5: A New Partner for IL-6. Immunity 2016, 44, 720–722. [Google Scholar] [CrossRef]
- Zhang, C.; Xin, H.; Zhang, W.; Yazaki, P.J.; Zhang, Z.; Le, K.; Li, W.; Lee, H.; Kwak, L.; Forman, S.; et al. CD5 Binds to Interleukin-6 and Induces a Feed-Forward Loop with the Transcription Factor STAT3 in B Cells to Promote Cancer. Immunity 2016, 44, 913–923. [Google Scholar] [CrossRef]
- Brown, M.H.; Lacey, E. A Ligand for CD5 Is CD5. J. Immunol. 2010, 185, 6068–6074. [Google Scholar] [CrossRef]
- Adams, E.L.; Rice, P.J.; Graves, B.; Ensley, H.E.; Yu, H.; Brown, G.D.; Gordon, S.; Monteiro, M.A.; Papp-Szabo, E.; Lowman, D.W.; et al. Differential high-affinity interaction of dectin-1 with natural or synthetic glucans is dependent upon primary structure and is influenced by polymer chain length and side-chain branching. J. Pharmacol. Exp. Ther. 2008, 325, 115–123. [Google Scholar] [CrossRef]
- Miles, S.; Velasco-de-Andrés, M.; Lozano, F.; Mourglia-Ettlin, G. Interactome analysis of CD5 and CD6 ectodomains with tegumental antigens from the helminth parasite Echinococcus granulosus sensu lato. Int. J. Biol. Macromol. 2020, 164, 3718–3728. [Google Scholar] [CrossRef] [PubMed]
- Sarhan, M.A.; Pham, T.N.Q.; Chen, A.Y.; Michalak, T.I. Hepatitis C virus infection of human T lymphocytes is mediated by CD5. J. Virol. 2012, 86, 3723–3735. [Google Scholar] [CrossRef] [PubMed]
- Velasco-de-Andrés, M.; Català, C.; Casadó-Llombart, S.; Simões, I.; Zaragoza, O.; Carreras, E.; Lozano, F. The lymphocyte scavenger receptor CD5 plays a nonredundant role in fungal infection. Cell. Mol. Immunol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Velasco-de Andrés, M.; Català, C.; Casadó-Llombart, S.; Martínez-Florensa, M.; Simões, I.; García-Luna, J.; Mourglia-Ettlin, G.; Zaragoza, Ó.; Carreras, E.; Lozano, F. The lymphocytic scavenger receptor CD5 shows therapeutic potential in mouse models of fungal infection. Antimicrob. Agents Chemother. 2020, 65, e01103–e01120. [Google Scholar] [CrossRef] [PubMed]
- Sarhan, M.A.; Chen, A.Y.; Michalak, T.I. Differential Expression of Candidate Virus Receptors in Human T Lymphocytes Prone or Resistant to Infection with Patient-Derived Hepatitis C Virus. PLoS ONE 2013, 8, e62159. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Lv, J.; Tu, Z.; Hu, X.; Yan, H.; Pan, Y.; Xu, D.; Lian, Z.; Chi, X.; Niu, J. Antiviral treatment improves disrupted peripheral B lymphocyte homeostasis in chronic hepatitis B virus-infected patients. Exp. Biol. Med. 2013, 238, 1275–1283. [Google Scholar] [CrossRef]
- Penney, S.J.; Gallant, M.E.; Grant, M.D. Greater frequency of CD5-negative CD8+ T cells against human immunodeficiency virus type 1 than other viruses is consistent with adaptation to antigenic variation. AIDS Res. Ther. 2014, 11, 30. [Google Scholar] [CrossRef]
- Tumas, D.B.; Hines, M.T.; Perryman, L.E.; Davis, W.C.; McGuire, T.C. Corticosteroid Immunosuppression and Monoclonal Antibody-mediated CD5+ T Lymphocyte Depletion in Normal and Equine Infectious Anaemia Virus-carrier Horses. J. Gen. Virol. 1994, 75, 959–968. [Google Scholar] [CrossRef]
- Karandikar, N.J.; Kroft, S.H.; Yegappan, S.; Rogers, B.B.; Aquino, V.M.; Lee, K.-M.; Kumar, V.; Guenaga, F.J.; Jaffe, E.S.; Douek, D.C.; et al. Unusual immunophenotype of CD8+ T cells in familial hemophagocytic lymphohistiocytosis. Blood 2004, 104, 2007–2009. [Google Scholar] [CrossRef]
- Dorothée, G.; Vergnon, I.; El Hage, F.; Chansac, B.L.M.; Ferrand, V.; Lécluse, Y.; Opolon, P.; Chouaib, S.; Bismuth, G.; Mami-Chouaib, F. In Situ Sensory Adaptation of Tumor-Infiltrating T Lymphocytes to Peptide-MHC Levels Elicits Strong Antitumor Reactivity. J. Immunol. 2005, 174, 6888–6897. [Google Scholar] [CrossRef]
- Potrony, M.; Carreras, E.; Aranda, F.; Zimmer, L.; Puig-Butille, J.-A.; Tell-Martí, G.; Armiger, N.; Sucker, A.; Giménez-Xavier, P.; Martínez-Florensa, M.; et al. Inherited functional variants of the lymphocyte receptor CD5 influence melanoma survival. Int. J. Cancer 2016, 139, 1297–1302. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Manuel, A.; Jantus-Lewintre, E.; Simões, I.; Aranda, F.; Calabuig-Fariñas, S.; Carreras, E.; Zúñiga, S.; Saenger, Y.; Rosell, R.; Camps, C.; et al. CD5 and CD6 as immunoregulatory biomarkers in non-small cell lung cancer. Transl. Lung Cancer Res. 2020, 9, 1074–1083. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Shapiro, B.; Vucic, E.A.; Vogt, S.; Bar-Sagi, D. Tumor cell-derived IL1β promotes desmoplasia and immune suppression in pancreatic cancer. Cancer Res. 2020, 80, 1088–1101. [Google Scholar] [CrossRef] [PubMed]
- Roudafshani, Z.; Jazayeri, M.H.; Mahmoudi, A.R.; Nedaeinia, R.; Safari, E.; Jazayeri, A. Evaluation of the frequency of CD5+ B cells as natural immunoglobulin M producers and circulating soluble CD5 in patients with bladder cancer. Mol. Biol. Rep. 2019, 46, 6405–6411. [Google Scholar] [CrossRef] [PubMed]
- Norouzian, M.; Mehdipour, F.; Balouchi Anaraki, S.; Ashraf, M.J.; Khademi, B.; Ghaderi, A. Atypical Memory and Regulatory B Cell Subsets in Tumor Draining Lymph Nodes of Head and Neck Squamous Cell Carcinoma Correlate with Good Prognostic Factors. Head Neck Pathol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Tabbekh, M.; Franciszkiewicz, K.; Haouas, H.; Lécluse, Y.; Benihoud, K.; Raman, C.; Mami-Chouaib, F. Rescue of Tumor-Infiltrating Lymphocytes from Activation-Induced Cell Death Enhances the Antitumor CTL Response in CD5-Deficient Mice. J. Immunol. 2011, 187, 102–109. [Google Scholar] [CrossRef]
- Alotaibi, F.; Rytelewski, M.; Figueredo, R.; Zareardalan, R.; Zhang, M.; Ferguson, P.J.; Maleki Vareki, S.; Najajreh, Y.; El-Hajjar, M.; Zheng, X.; et al. CD5 blockade enhances ex vivo CD8+ T cell activation and tumour cell cytotoxicity. Eur. J. Immunol. 2020, 50, 695–704. [Google Scholar] [CrossRef]
- Fenutría, R.; Martinez, V.G.; Simões, I.; Postigo, J.; Gil, V.; Martínez-Florensa, M.; Sintes, J.; Naves, R.; Cashman, K.S.; Alberola-Ila, J.; et al. Transgenic expression of soluble human CD5 enhances experimentally-induced autoimmune and anti-tumoral immune responses. PLoS ONE 2014, 9, e84895. [Google Scholar] [CrossRef]
- Simões, I.T.; Aranda, F.; Carreras, E.; Andrés, M.V.; Casadó-Llombart, S.; Martinez, V.G.; Lozano, F. Immunomodulatory effects of soluble CD5 on experimental tumor models. Oncotarget 2017, 8, 108156–108169. [Google Scholar] [CrossRef]
- Cifaldi, L.; Prencipe, G.; Caiello, I.; Bracaglia, C.; Locatelli, F.; De Benedetti, F.; Strippoli, R. Inhibition of Natural Killer Cell Cytotoxicity by Interleukin-6: Implications for the Pathogenesis of Macrophage Activation Syndrome. Arthritis Rheumatol. 2015, 67, 3037–3046. [Google Scholar] [CrossRef]
- Rautela, J.; Huntington, N.D. IL-15 signaling in NK cell cancer immunotherapy. Curr. Opin. Immunol. 2017, 44, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Jaseb, K.; Purrahman, D.; Shahrabi, S.; Ghanavat, M.; Rezaeean, H.; Saki, N. Prognostic significance of aberrant CD5 expression in B-cell leukemia. Oncol. Rev. 2019, 13, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Gary-Gouy, H.; Sainz-Perez, A.; Marteau, J.-B.; Marfaing-Koka, A.; Delic, J.; Merle-Beral, H.; Galanaud, P.; Dalloul, A. Natural Phosphorylation of CD5 in Chronic Lymphocytic Leukemia B Cells and Analysis of CD5-Regulated Genes in a B Cell Line Suggest a Role for CD5 in Malignant Phenotype. J. Immunol. 2007, 179, 4335–4344. [Google Scholar] [CrossRef] [PubMed]
- Dallman, M.J.; Thomas, M.L.; Green, J.R. MRC OX-19: A monoclonal antibody that labels rat T lymphocytes and augments in vitro proliferative responses. Eur. J. Immunol. 1984, 14, 260–267. [Google Scholar] [CrossRef]
- Like, A.A.; Biron, C.A.; Weringer, E.J.; Byman, K.; Sroczynski, E.; Guberski, D.L. Prevention of diabetes in biobreeding/worcester rats with monoclonal antibodies that recognize T lymphocytes or natural killer cells. J. Exp. Med. 1986, 164, 1145–1159. [Google Scholar] [CrossRef]
- Ellerman, K.E.; Richards, C.A.; Guberski, D.L.; Shek, W.R.; Like, A.A. Kilham rat virus triggers T-cell-dependent autoimmune diabetes in multiple strains of rat. Diabetes 1996, 45, 557–562. [Google Scholar] [CrossRef]
- Ikezumi, Y.; Kawachi, H.; Toyabe, S.; Uchiyama, M.; Shimizu, F. An anti-CD5 monoclonal antibody ameliorates proteinuria and glomerular lesions in rat mesangioproliferative glomerulonephritis. Kidney Int. 2000, 58, 100–114. [Google Scholar] [CrossRef]
- Strigård, K.; Olsson, T.; Larsson, P.; Holmdahl, R.; Klareskog, L. Modulation of experimental allergic neuritis in rats by in vivo treatment with monoclonal anti T cell antibodies. J. Neurol. Sci. 1988, 83, 283–291. [Google Scholar] [CrossRef]
- Gary-Gouy, H.; Harriague, J.; Bismuth, G.; Platzer, C.; Schmitt, C.; Dalloul, A.H. Human CD5 promotes B-cell survival through stimulation of autocrine IL-10 production. Blood 2002, 100, 4537–4543. [Google Scholar] [CrossRef]
- Blaize, G.; Daniels-Treffandier, H.; Aloulou, M.; Rouquié, N.; Yang, C.; Marcellin, M.; Gador, M.; Benamar, M.; Ducatez, M.; Song, K.D.; et al. CD5 signalosome coordinates antagonist TCR signals to control the generation of Treg cells induced by foreign antigens. Proc. Natl. Acad. Sci. USA 2020, 117, 12969–12979. [Google Scholar] [CrossRef]
- Bajwa, R.; Cheema, A.; Khan, T.; Amirpour, A.; Paul, A.; Chaughtai, S.; Patel, S.; Patel, T.; Bramson, J.; Gupta, V.; et al. Adverse Effects of Immune Checkpoint Inhibitors (Programmed Death-1 Inhibitors and Cytotoxic T-Lymphocyte-Associated Protein-4 Inhibitors): Results of a Retrospective Study. J. Clin. Med. Res. 2019, 11, 225–236. [Google Scholar] [CrossRef] [PubMed]
- Axtell, R.C.; Webb, M.S.; Barnum, S.R.; Raman, C. Cutting Edge: Critical Role for CD5 in Experimental Autoimmune Encephalomyelitis: Inhibition of Engagement Reverses Disease in Mice. J. Immunol. 2004, 173, 2928–2932. [Google Scholar] [CrossRef] [PubMed]
- Kamoun, M.; Kadin, M.E.; Martin, P.J.; Nettleton, J.; Hansen, J.A. A novel human T cell antigen preferentially expressed on mature T cells and shared by both well and poorly differentiated B cell leukemias and lymphomas. J. Immunol. 1981, 127, 987–991. [Google Scholar] [PubMed]
- Braun, M.; Müller, B.; Ter Meer, D.; Raffegerst, S.; Simm, B.; Wilde, S.; Spranger, S.; Ellwart, J.; Mosetter, B.; Umansky, L.; et al. The CD6 scavenger receptor is differentially expressed on a CD56 dim natural killer cell subpopulation and contributes to natural killer-derived cytokine and chemokine secretion. J. Innate Immun. 2011, 3, 420–434. [Google Scholar] [CrossRef] [PubMed]
- Cortés, F.; Deschaseaux, F.; Uchida, N.; Labastie, M.C.; Friera, A.M.; He, D.; Charbord, P.; Péault, B. HCA, an immunoglobulin-like adhesion molecule present on the earliest human hematopoietic precursor cells, is also expressed by stromal cells in blood-forming tissues. Blood 1999, 93, 826–837. [Google Scholar] [CrossRef]
- Konno, A.; Ahn, J.S.; Kitamura, H.; Hamilton, M.J.; Gebe, J.A.; Aruffo, A.; Davis, W.C. Tissue distribution of CD6 and CD6 ligand in cattle: Expression of the CD6 ligand (CD166) in the autonomic nervous system of cattle and the human. J. Leukoc. Biol. 2001, 69, 944–950. [Google Scholar]
- Consuegra-Fernández, M.; Lin, F.; Fox, D.A.; Lozano, F. Clinical and experimental evidence for targeting CD6 in immune-based disorders. Autoimmun. Rev. 2018, 17, 493–503. [Google Scholar] [CrossRef]
- Gonçalves, C.M.; Henriques, S.N.; Santos, R.F.; Carmo, A.M. CD6, a rheostat-type signalosome that tunes T cell activation. Front. Immunol. 2018, 9. [Google Scholar] [CrossRef]
- Mori, D.; Grégoire, C.; Voisinne, G.; Celis-Gutierrez, J.; Aussel, R.; Girard, L.; Camus, M.; Marcellin, M.; Argenty, J.; Burlet-Schiltz, O.; et al. The T cell CD6 receptor operates a multitask signalosome with opposite functions in T cell activation. J. Exp. Med. 2021, 218, e20201011. [Google Scholar] [CrossRef]
- Carrasco, E.; Escoda-Ferran, C.; Climent, N.; Miró-Julià, C.; Simões, I.T.; Martínez-Florensa, M.; Sarukhan, A.; Carreras, E.; Lozano, F. Human CD6 Down-Modulation following T-Cell Activation Compromises Lymphocyte Survival and Proliferative Responses. Front. Immunol. 2017, 8, 769. [Google Scholar] [CrossRef]
- Bowen, M.A.; Patel, D.D.; Li, X.; Modrell, B.; Malacko, A.R.; Wang, W.C.; Marquardt, H.; Neubauer, M.; Pesando, J.M.; Francke, U.; et al. Cloning, mapping, and characterization of activated leukocyte-cell adhesion molecule (ALCAM), a CD6 ligand. J. Exp. Med. 1995, 181, 2213–2220. [Google Scholar] [CrossRef] [PubMed]
- Hassan, N.J.; Barclay, A.N.; Brown, M.H. Frontline: Optimal T cell activation requires the engagement of CD6 and CD166. Eur. J. Immunol. 2004, 34, 930–940. [Google Scholar] [CrossRef] [PubMed]
- Escoda-Ferran, C.; Carrasco, E.; Caballero-Baños, M.; Miró-Julià, C.; Martínez-Florensa, M.; Consuegra-Fernández, M.; Martínez, V.G.; Liu, F.-T.; Lozano, F. Modulation of CD6 function through interaction with Galectin-1 and -3. FEBS Lett. 2014, 588, 2805–2813. [Google Scholar] [CrossRef] [PubMed]
- Tobias, P.S.; Soldau, K.; Gegner, J.A.; Mintz, D.; Ulevitch, R.J. Lipopolysaccharide binding protein-mediated complexation of lipopolysaccharide with soluble CD14. J. Biol. Chem. 1995, 270, 10482–10488. [Google Scholar] [CrossRef]
- Carrasco, E.; Escoda, C.; Alvarez-Fernández, C.; Sanchez-Palomino, S.; Carreras, E.; Gatell, J.M.; Gallart, T.; García, F.; Climent, N.; Lozano, F. A Role for Scavenger-like Lymphocyte Receptor CD6 in HIV-1 Viral Infection. AIDS Res. Hum. Retrovir. 2014, 30, A49–A50. [Google Scholar] [CrossRef]
- Jacque, B.; Stephan, K.; Smirnova, I.; Kim, B.; Gilling, D.; Poltorak, A. Mice expressing high levels of soluble CD14 retain LPS in the circulation and are resistant to LPS-induced lethality. Eur. J. Immunol. 2006, 36, 3007–3016. [Google Scholar] [CrossRef]
- Dejager, L.; Pinheiro, I.; Dejonckheere, E.; Libert, C. Cecal ligation and puncture: The gold standard model for polymicrobial sepsis? Trends Microbiol. 2011, 19, 198–208. [Google Scholar] [CrossRef]
- Martínez-Florensa, M.; Consuegra-Fernández, M.; Aranda, F.; Armiger-Borràs, N.; Di Scala, M.; Carrasco, E.; Pachón, J.; Vila, J.; González-Aseguinolaza, G.; Lozano, F. Protective Effects of Human and Mouse Soluble Scavenger-Like CD6 Lymphocyte Receptor in a Lethal Model of Polymicrobial Sepsis. Antimicrob. Agents Chemother. 2017, 61, e01391–e14016. [Google Scholar] [CrossRef]
- Martínez-Florensa, M.; Català, C.; Velasco-de Andrés, M.; Cañadas, O.; Fraile-Ágreda, V.; Casadó-Llombart, S.; Armiger-Borràs, N.; Consuegra-Fernández, M.; Casals, C.; Lozano, F. Conserved Bacterial-Binding Peptides of the Scavenger-Like Human Lymphocyte Receptor CD6 Protect From Mouse Experimental Sepsis. Front. Immunol. 2018, 9, 627. [Google Scholar] [CrossRef]
- Bikker, F.J.; Ligtenberg, A.J.M.; End, C.; Renner, M.; Blaich, S.; Lyer, S.; Wittig, R.; van’t Hof, W.; Veerman, E.C.I.; Nazmi, K.; et al. Bacteria Binding by DMBT1/SAG/gp-340 Is Confined to the VEVL XXXX W Motif in Its Scavenger Receptor Cysteine-rich Domains. J. Biol. Chem. 2004, 279, 47699–47703. [Google Scholar] [CrossRef]
- Zimmermann, M.; Busch, K.; Kuhn, S.; Zeppezauer, M. Endotoxin adsorbent based on immobilized human serum albumin. Clin. Chem. Lab. Med. 1999, 37, 373–379. [Google Scholar] [CrossRef] [PubMed]
- Sembries, S.; Pahl, H.; Stilgenbauer, S.; Döhner, H.; Schriever, F. Reduced expression of adhesion molecules and cell signaling receptors by chronic lymphocytic leukemia cells with 11q deletion. Blood 1999, 93, 624–631. [Google Scholar] [CrossRef] [PubMed]
- Osorio, L.M.; Jondal, M.; Aguilar-Santelises, M. Regulation of B-CLL apoptosis through membrane receptors and Bcl-2 family proteins. Leuk. Lymphoma 1998, 30, 247–256. [Google Scholar] [CrossRef] [PubMed]
- Swart, G.W.M. Activated leukocyte cell adhesion molecule (CD166/ALCAM): Developmental and mechanistic aspects of cell clustering and cell migration. Eur. J. Cell Biol. 2002, 81, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Cayrol, R.; Wosik, K.; Berard, J.L.; Dodelet-Devillers, A.; Ifergan, I.; Kebir, H.; Haqqani, A.S.; Kreymborg, K.; Krug, S.; Moumdjian, R.; et al. Activated leukocyte cell adhesion molecule promotes leukocyte trafficking into the central nervous system. Nat. Immunol. 2008, 9, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Weidle, U.H.; Eggle, D.; Klostermann, S.; Swart, G.W.M. ALCAM/CD166: Cancer-related issues. Cancer Genom. Proteom. 2010, 7, 231–244. [Google Scholar]
- Darvishi, B.; Boroumandieh, S.; Majidzadeh-A, K.; Salehi, M.; Jafari, F.; Farahmand, L. The role of activated leukocyte cell adhesion molecule (ALCAM) in cancer progression, invasion, metastasis and recurrence: A novel cancer stem cell marker and tumor-specific prognostic marker. Exp. Mol. Pathol. 2020, 115, 104443. [Google Scholar] [CrossRef]
- Kim, Y.S.; Kim, M.N.; Lee, K.E.; Hong, J.Y.; Oh, M.S.; Kim, S.Y.; Kim, K.W.; Sohn, M.H. Activated leucocyte cell adhesion molecule (ALCAM/CD166) regulates T cell responses in a murine model of food allergy. Clin. Exp. Immunol. 2018, 192, 151–164. [Google Scholar] [CrossRef]
- Simões, I.T.; Aranda, F.; Casadó-Llombart, S.; Velasco-de Andrés, M.; Català, C.; Álvarez, P.; Consuegra-Fernández, M.; Orta-Mascaró, M.; Merino, R.; Merino, J.; et al. Multifaceted effects of soluble human CD6 in experimental cancer models. J. Immunother. Cancer 2020, 8, e000172. [Google Scholar] [CrossRef]
- Kofler, D.M.; Farkas, A.; von Bergwelt-Baildon, M.; Hafler, D.A. The Link Between CD6 and Autoimmunity: Genetic and Cellular Associations. Curr. Drug Targets 2016, 17, 651–665. [Google Scholar] [CrossRef]
- Wagner, M.; Bilinska, M.; Pokryszko-Dragan, A.; Sobczynski, M.; Cyrul, M.; Kusnierczyk, P.; Jasek, M. ALCAM and CD6-multiple sclerosis risk factors. J. Neuroimmunol. 2014, 276, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Singer, N.G.; Whitbred, J.; Bowen, M.A.; Fox, D.A.; Lin, F. CD6 as a potential target for treating multiple sclerosis. Proc. Natl. Acad. Sci. USA 2017, 114, 2687–2692. [Google Scholar] [CrossRef] [PubMed]
- Consuegra-Fernández, M.; Martínez-Florensa, M.; Aranda, F.; de Salort, J.; Armiger-Borràs, N.; Lozano, T.; Casares, N.; Lasarte, J.J.; Engel, P.; Lozano, F. Relevance of CD6-mediated interactions in the regulation of peripheral T-cell responses and tolerance. Front. Immunol. 2017, 8, 594. [Google Scholar] [CrossRef] [PubMed]
- Consuegra-Fernández, M.; Julià, M.; Martínez-Florensa, M.; Aranda, F.; Català, C.; Armiger-Borràs, N.; Arias, M.T.; Santiago, F.; Guilabert, A.; Esteve, A.; et al. Genetic and experimental evidence for the involvement of the CD6 lymphocyte receptor in psoriasis. Cell. Mol. Immunol. 2018, 15, 898–906. [Google Scholar] [CrossRef]
- Zhang, L.; Li, Y.; Qiu, W.; Bell, B.A.; Dvorina, N.; Baldwin, W.M.; Singer, N.; Kern, T.; Caspi, R.R.; Fox, D.A.; et al. Targeting CD6 for the treatment of experimental autoimmune uveitis. J. Autoimmun. 2018, 90, 84–93. [Google Scholar] [CrossRef]
- Li, Y.; Ruth, J.H.; Rasmussen, S.M.; Athukorala, K.S.; Weber, D.P.; Amin, M.A.; Campbell, P.L.; Singer, N.G.; Fox, D.A.; Lin, F. Attenuation of Murine Collagen-Induced Arthritis by Targeting CD6. Arthritis Rheumatol. 2020, 72, 1505–1513. [Google Scholar] [CrossRef]
- Levesque, M.C.; Heinly, C.S.; Whichard, L.P.; Patel, D.D. Cytokine-regulated expression of activated leukocyte cell adhesion molecule (CD166) on monocyte-lineage cells and in rheumatoid arthritis synovium. Arthritis Rheum. 1998, 41, 2221–2229. [Google Scholar] [CrossRef]
- Hernández, P.; Moreno, E.; Aira, L.E.; Rodríguez, P.C. Therapeutic Targeting of CD6 in Autoimmune Diseases: A Review of Cuban Clinical Studies with the Antibodies IOR-T1 and Itolizumab. Curr. Drug Targets 2016, 17, 666–677. [Google Scholar]
- Jayaraman, K. Biocon’s first-in-class anti-CD6 mAb reaches the market. Nat. Biotechnol. 2013, 31, 1062–1063. [Google Scholar] [CrossRef]
- Dogra, S.; Shabeer, D.; Rajagopalan, M. Anti-CD6 mAbs for the treatment of psoriasis. Expert Opin. Biol. Ther. 2020, 20, 1215–1222. [Google Scholar] [CrossRef]
- Krupashankar, D.S.; Dogra, S.; Kura, M.; Saraswat, A.; Budamakuntla, L.; Sumathy, T.K.; Shah, R.; Gopal, M.G.; Narayana Rao, T.; Srinivas, C.R.; et al. Efficacy and safety of itolizumab, a novel anti-CD6 monoclonal antibody, in patients with moderate to severe chronic plaque psoriasis: Results of a double-blind, randomized, placebo-controlled, phase-III study. J. Am. Acad. Dermatol. 2014, 71, 484–492. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Velasco-de Andrés, M.; Casadó-Llombart, S.; Català, C.; Leyton-Pereira, A.; Lozano, F.; Aranda, F. Soluble CD5 and CD6: Lymphocytic Class I Scavenger Receptors as Immunotherapeutic Agents. Cells 2020, 9, 2589. https://doi.org/10.3390/cells9122589
Velasco-de Andrés M, Casadó-Llombart S, Català C, Leyton-Pereira A, Lozano F, Aranda F. Soluble CD5 and CD6: Lymphocytic Class I Scavenger Receptors as Immunotherapeutic Agents. Cells. 2020; 9(12):2589. https://doi.org/10.3390/cells9122589
Chicago/Turabian StyleVelasco-de Andrés, María, Sergi Casadó-Llombart, Cristina Català, Alejandra Leyton-Pereira, Francisco Lozano, and Fernando Aranda. 2020. "Soluble CD5 and CD6: Lymphocytic Class I Scavenger Receptors as Immunotherapeutic Agents" Cells 9, no. 12: 2589. https://doi.org/10.3390/cells9122589
APA StyleVelasco-de Andrés, M., Casadó-Llombart, S., Català, C., Leyton-Pereira, A., Lozano, F., & Aranda, F. (2020). Soluble CD5 and CD6: Lymphocytic Class I Scavenger Receptors as Immunotherapeutic Agents. Cells, 9(12), 2589. https://doi.org/10.3390/cells9122589





