The Meniscus Tear: A Review of Stem Cell Therapies
Abstract
1. Introduction
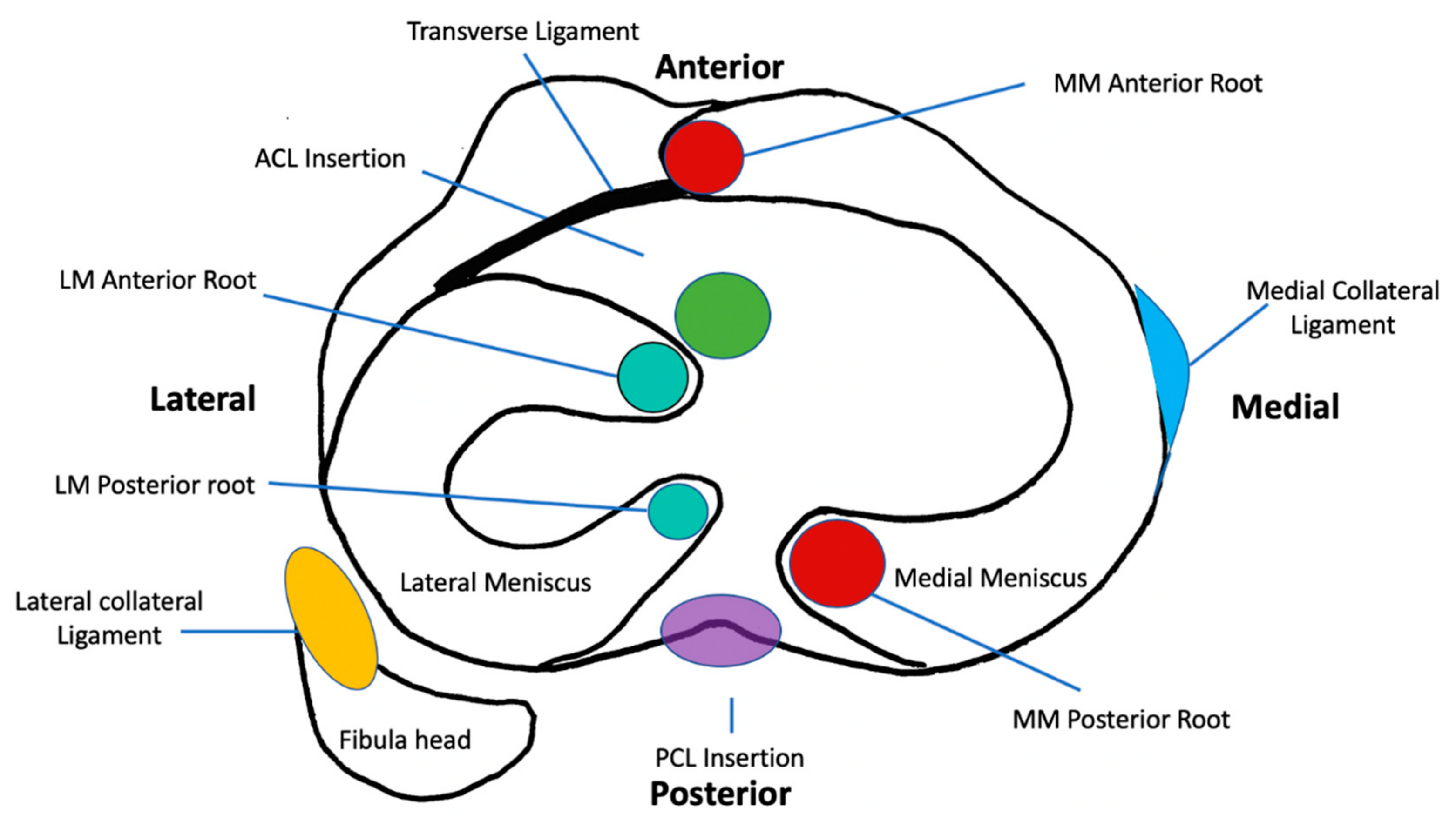
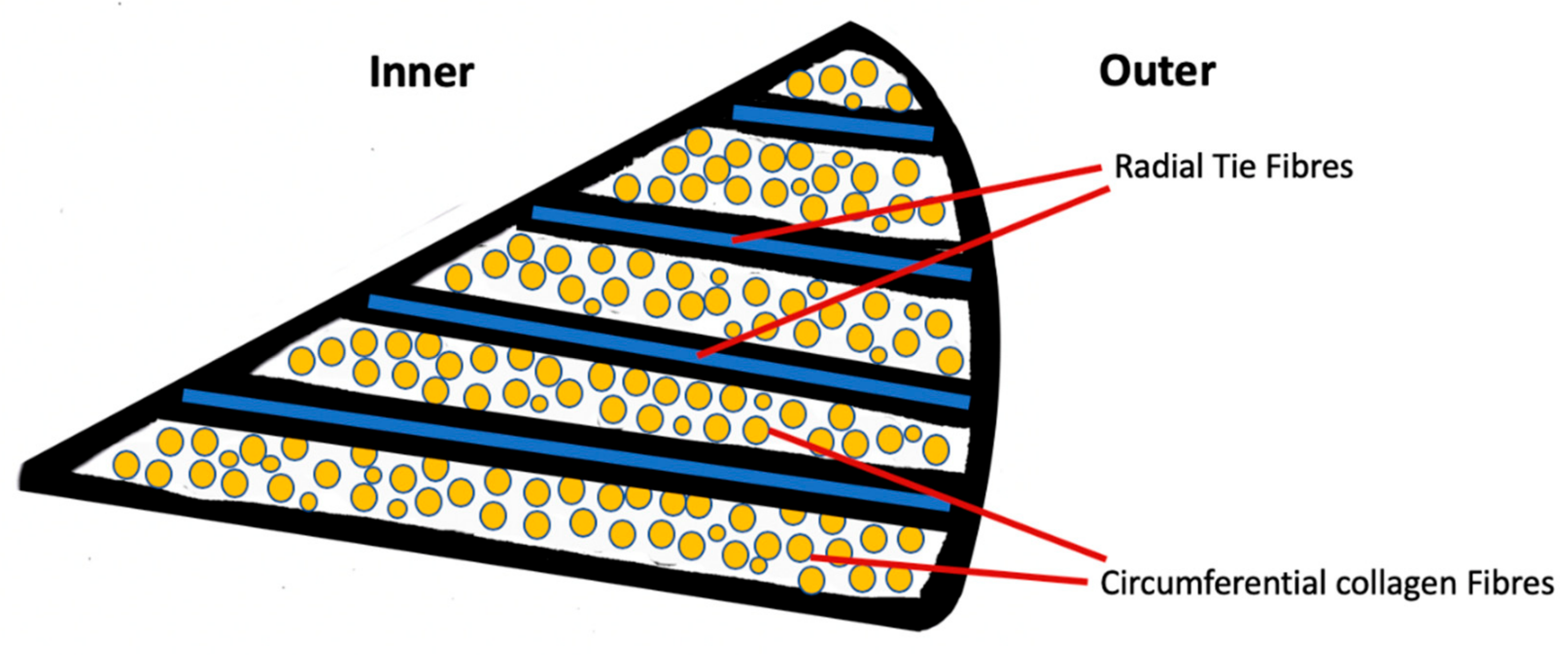
2. Anatomy of the Meniscus
3. Functions of the Meniscus
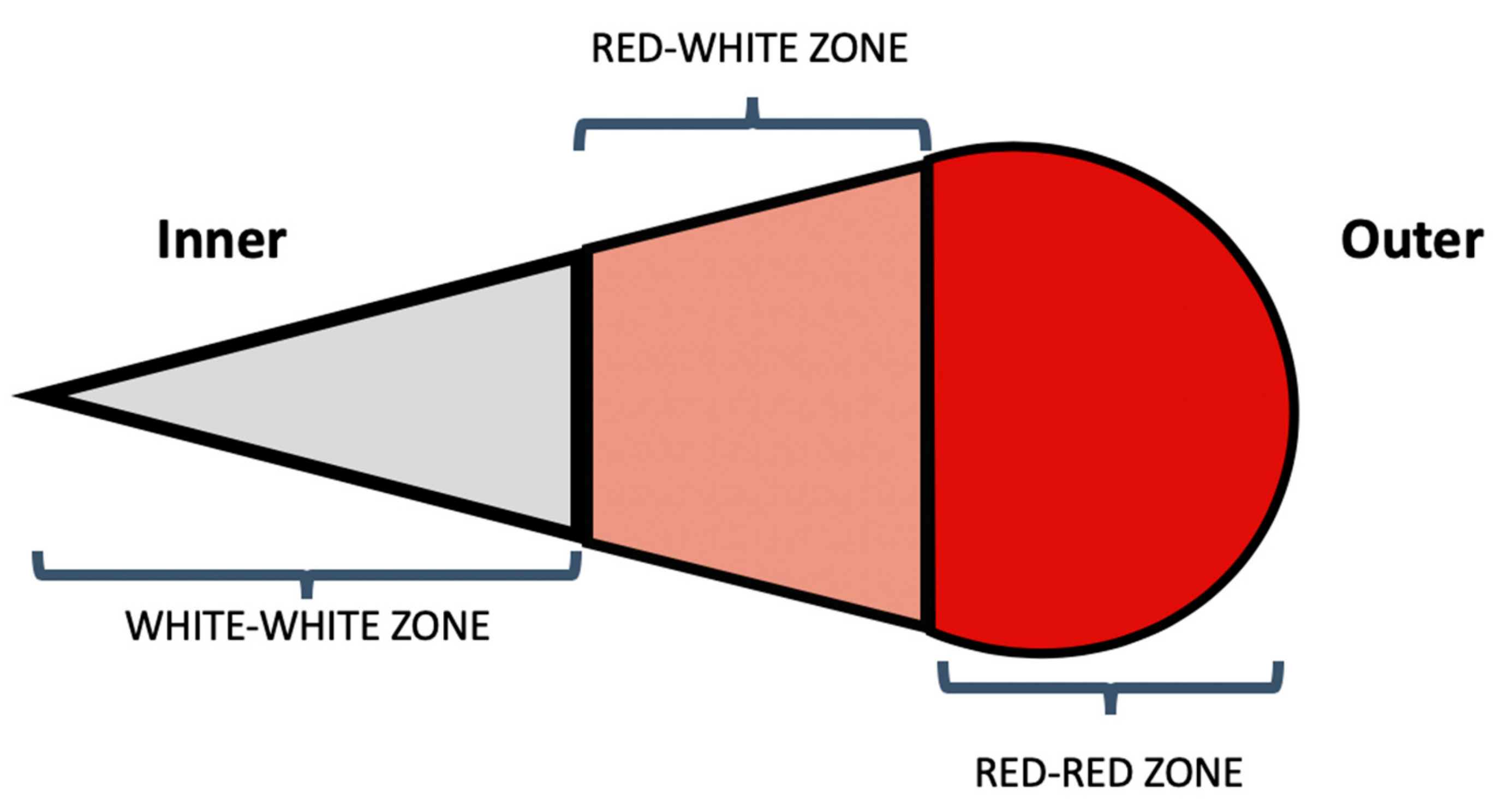
4. Meniscus Pathology
5. Types of Mesenchymal Stem Cells
6. Mechanism of Meniscal Repair
7. Pre-Clinical Studies
7.1. Stem Cell Injection
7.2. Tissue Engineering
8. Clinical Studies
8.1. Stem Cell Injection
8.2. Tissue Engineering
9. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- McDermott, I.D.; Amis, A.A. The consequences of meniscectomy. J. Bone Jt. Surg. 2006, 88, 1549–1556. [Google Scholar] [CrossRef] [PubMed]
- Fox, A.J.S.; Bedi, A.; Rodeo, S.A. The Basic Science of Human Knee Menisci: Structure, Composition, and Function. Sports Health 2012, 4, 340–351. [Google Scholar] [CrossRef] [PubMed]
- Arnoczky, S.P.; Warren, R.F. Microvasculature of the human meniscus. Am. J. Sports Med. 1982, 10, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Kawamura, S.; Lotito, K.; Rodeo, S.A. Biomechanics and healing response of the meniscus. Oper. Tech. Sports Med. 2003, 11, 68–76. [Google Scholar] [CrossRef]
- McNeill Love, R.J. Prognosis after removal of semilunar cartilages. Br. Med. J. 1923, 2, 324–326. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Seil, R.; Becker, R. Time for a paradigm change in meniscal repair: Save the meniscus! Knee Surgery, Sport. Traumatol. Arthrosc. 2016, 24, 1421–1423. [Google Scholar] [CrossRef]
- Zellner, J.; Pattappa, G.; Koch, M.; Lang, S.; Weber, J.; Pfeifer, C.G.; Mueller, M.B.; Kujat, R.; Nerlich, M.; Angele, P. Autologous mesenchymal stem cells or meniscal cells: What is the best cell source for regenerative meniscus treatment in an early osteoarthritis situation? Stem Cell Res. Ther. 2017, 8, 225. [Google Scholar] [CrossRef]
- Vaquero, J.; Forriol, F. Meniscus tear surgery and meniscus replacement. Muscles. Ligaments Tendons J. 2016, 6, 71–89. [Google Scholar] [CrossRef]
- Zaffagnini, S.; Grassi, A.; Marcheggiani Muccioli, G.M.; Bonanzinga, T.; Nitri, M.; Raggi, F.; Ravazzolo, G.; Marcacci, M. MRI evaluation of a collagen meniscus implant: A systematic review. Knee Surgery, Sport. Traumatol. Arthrosc. 2015, 23, 3228–3237. [Google Scholar] [CrossRef]
- Krych, A.J.; Lorich, D.G.; Kelly, B.T. Treatment of Focal Osteochondral Defects of the Acetabulum with Osteochondral Allograft Transplantation. Orthopedics 2011, 34, e307–e311. [Google Scholar] [CrossRef]
- Monllau, J.C.; Gelber, P.E.; Abat, F.; Pelfort, X.; Abad, R.; Hinarejos, P.; Tey, M. Outcome after partial medial meniscus substitution with the collagen meniscal implant at a minimum of 10 years’ follow-up. Arthroscopy 2011, 27, 933–943. [Google Scholar] [CrossRef] [PubMed]
- Makris, E.A.; Hadidi, P.; Athanasiou, K.A. The knee meniscus: Structure-function, pathophysiology, current repair techniques, and prospects for regeneration. Biomaterials 2011, 32, 7411–7431. [Google Scholar] [CrossRef] [PubMed]
- King, D. The healing of semilunar cartilages. 1936. Clin. Orthop. Relat. Res. 1990, 252, 4–7. [Google Scholar]
- Mauck, R.L.; Burdick, J.A. From Repair to Regeneration: Biomaterials to Reprogram the Meniscus Wound Microenvironment. Ann. Biomed. Eng. 2015, 43, 529–542. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Osawa, A.; Harner, C.D.; Gharaibeh, B.; Matsumoto, T.; Mifune, Y.; Kopf, S.; Ingham, S.J.M.; Schreiber, V.; Usas, A.; Huard, J. The use of blood vessel-derived stem cells for meniscal regeneration and repair. Med. Sci. Sports Exerc. 2013, 45, 813–823. [Google Scholar] [CrossRef] [PubMed]
- Kusayama, T.; Harner, C.D.; Carlin, G.J.; Xerogeanes, J.W.; Smith, B.A. Anatomical and biomechanical characteristics of human meniscofemoral ligaments. Knee Surgery, Sport. Traumatol. Arthrosc. 1994, 2, 234–237. [Google Scholar] [CrossRef] [PubMed]
- Laprade, C.M.; Ellman, M.B.; Rasmussen, M.T.; James, E.W.; Wijdicks, C.A.; Engebretsen, L.; Laprade, R.F. Anatomy of the anterior root attachments of the medial and lateral menisci: A quantitative analysis. Am. J. Sports Med. 2014, 42, 2386–2392. [Google Scholar] [CrossRef]
- Andrews, S.H.J.; Adesida, A.B.; Abusara, Z.; Shrive, N.G. Current concepts on structure–function relationships in the menisci. Connect. Tissue Res. 2017, 58, 271–281. [Google Scholar] [CrossRef]
- Andrews, S.H.J.; Ronsky, J.L.; Rattner, J.B.; Shrive, N.G.; Jamniczky, H.A. An evaluation of meniscal collagenous structure using optical projection tomography. BMC Med. Imaging 2013, 13, 21. [Google Scholar] [CrossRef]
- Melrose, J.; Smith, S.; Cake, M.; Read, R.; Whitelock, J. Comparative spatial and temporal localisation of perlecan, aggrecan and type I, II and IV collagen in the ovine meniscus: An ageing study. Histochem. Cell Biol. 2005, 124, 225–235. [Google Scholar] [CrossRef]
- Doral, M.N.; Bilge, O.; Huri, G.; Turhan, E.; Verdonk, R. Modern treatment of meniscal tears. EFORT Open Rev. 2018, 3, 260–268. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, M.; Evans, E.; Donthineni Rao, R.; Findlay, J.; Pepberton, D. Quantitative differences in the histology of the attachemnt zones of tghe mesiscal horns in the knee joint of man. J. Anat. 1991, 177, 127–134. [Google Scholar] [PubMed]
- Herwig, J.; Egner, E.; Buddecke, E. Chemical changes of human knee joint menisci in various stages of degeneration. Ann. Rheum. Dis. 1984, 43, 635–640. [Google Scholar] [CrossRef]
- Cheung, H.S. Distribution of type I, II, III and v in the pepsin solubilized collagens in bovine menisci. Connect. Tissue Res. 1987, 16, 343–356. [Google Scholar] [CrossRef]
- Nakano, T.; Dodd, C.M.; Scott, P.G. Glycosaminoglycans and proteoglycans from different zones of the porcine knee meniscus. J. Orthop. Res. 1997, 15, 213–220. [Google Scholar] [CrossRef]
- Swann, D.A.; Silver, F.H.; Slayter, H.S.; Stafford, W.; Shore, E. The molecular structure and lubricating activity of lubricin isolated from bovine and human synovial fluids. Biochem. J. 1985, 225, 195–201. [Google Scholar] [CrossRef]
- Peters, T.J.; Smillie, I.S. Studies on the chemical composition of the menisci of the knee joint with special reference to the horizontal cleavage lesion. Clin. Orthop. Relat. Res. 1972, 86, 245–252. [Google Scholar] [CrossRef]
- McDevitt, C.A.; Mukherjee, S.; Kambic, H.; Parker, R. Emerging concepts of the cell biology of the meniscus. Curr. Opin. Orthop. 2002, 13, 345–350. [Google Scholar] [CrossRef]
- Verdonk, P.C.M.; Forsyth, R.G.; Wang, J.; Almqvist, K.F.; Verdonk, R.; Veys, E.M.; Verbruggen, G. Characterisation of human knee meniscus cell phenotype. Osteoarthr. Cartil. 2005, 13, 548–560. [Google Scholar] [CrossRef]
- Le Graverand, M.P.H.; Ou, Y.C.; Schield-Yee, T.; Barclay, L.; Hart, D.; Natsume, T.; Rattner, J.B. The cells of the rabbit meniscus: Their arrangement, interrelationship, morphological variations and cytoarchitecture. J. Anat. 2001, 198, 525–535. [Google Scholar] [CrossRef]
- Van der Bracht, H.; Verdonk, R.; Verbruggen, D.; Elewaut, D.; Verdonk, P. Cell-based meniscus tissue engineering. In Topics in Tissue Engineering; Ashammakhi, N., Reis, R.L., Chiellini, E., Eds.; Biomaterials and Tissue Engineering Group (BTE): London, UK, 2007; Volume 3. [Google Scholar]
- Cameron, H.U.; Macnab, I. The structure of the meniscus of the human knee joint. Clin. Orthop. Relat. Res. 1972, 89, 215–219. [Google Scholar] [CrossRef]
- Proctor, C.S.; Schmidt, M.B.; Whipple, R.R.; Kelly, M.A.; Mow, V.C. Material properties of the normal medial bovine meniscus. J. Orthop. Res. 1989, 7, 771–782. [Google Scholar] [CrossRef]
- Voloshin, A.S.; Wosk, J. Shock absorption of meniscectomized and painful knees: A comparative in vivo study. J. Biomed. Eng. 1983, 5, 157–161. [Google Scholar] [CrossRef]
- Walker, P.S.; Erkman, M.J. The role of the menisci in force transmission across the knee. Clin. Orthop. 1975, 109, 184–192. [Google Scholar] [CrossRef]
- Hoshino, A.; Wallace, W.A. Impact-absorbing properties of the human knee. J. Bone Jt. Surg. 1987, 69, 807–811. [Google Scholar] [CrossRef]
- Markolf, K.L.; Mensch, J.S.; Amstutz, H.C. Stiffness and laxity of the knee—The contributions of the supporting structures. J. Bone Jt. Surg. Am. 1976, 58, 583–594. [Google Scholar] [CrossRef]
- Bird, M.D.T.; Sweet, M.B.E. Canals in the semilunar meniscus: Brief report. J. Bone Jt. Surg. 1988, 70, 839. [Google Scholar] [CrossRef]
- Arnoczky, S.P.; Warren, R.F.; Spivak, J.M. Meniscal repair using an exogenous fibrin clot. An experimental study in dogs. J. Bone Jt. Surg. 1988, 70, 1209–1217. [Google Scholar] [CrossRef]
- Skinner, H.B.; Barrack, R.L.; Cook, S.D. Age related decline in proprioception. Clin. Orthop. Relat. Res. 1984, 184, 208–211. [Google Scholar] [CrossRef]
- Reider, B.; Arcand, M.A.; Diehl, L.H.; Mroczek, K.; Abulencia, A.; Stroud, C.C.; Palm, M.; Gilbertson, J.; Staszak, P. Proprioception of the knee before and after anterior cruciate ligament reconstruction. Arthroscopy 2003, 19, 2–12. [Google Scholar] [CrossRef]
- Kennedy, J.C.; Alexander, I.J.; Hayes, K.C. Nerve supply of the human knee and its functional importance. Am. J. Sports Med. 1982, 10, 329–335. [Google Scholar] [CrossRef]
- Bhattacharyya, T.; Gale, D.; Dewire, P.; Totterman, S.; Gale, M.E.; McLaughlin, S.; Einhorn, T.A.; Felson, D.T. The clinical importance of meniscal tears demonstrated by magnetic resonance imaging in osteoarthritis of the knee. J. Bone Jt. Surg. 2003, 85, 4–9. [Google Scholar] [CrossRef]
- Link, T.M.; Steinbach, L.S.; Ghosh, S.; Ries, M.; Lu, Y.; Lane, N.; Majumdar, S. Osteoarthritis: MR imaging findings in different stages of disease and correlation with clinical findings. Radiology 2003, 226, 373–381. [Google Scholar] [CrossRef]
- Boks, S.S.; Vroegindeweij, D.; Koes, B.W.; Hunink, M.M.G.M.; Bierma-Zeinstra, S.M.A. Magnetic resonance imaging abnormalities in symptomatic and contralateral knees: Prevalence and associations with traumatic history in general practice. Am. J. Sports Med. 2006, 34, 1984–1991. [Google Scholar] [CrossRef]
- Ding, C.; Martel-Pelletier, J.; Pelletier, J.P.; Abram, F.; Raynauld, J.P.; Cicuttini, F.; Jones, G. Meniscal tear as an osteoarthritis risk factor in a largely non-osteoarthritic cohort: A cross-sectional study. J. Rheumatol. 2007, 34, 776–784. [Google Scholar]
- Noble, J.; Hamblen, D.L. The pathology of the degenerate meniscus lesion. J. Bone Jt. Surg. 1975, 57, 180–186. [Google Scholar] [CrossRef]
- Englund, M.; Roemer, F.W.; Hayashi, D.; Crema, M.D.; Guermazi, A. Meniscus pathology, osteoarthritis and the treatment controversy. Nat. Rev. Rheumatol. 2012, 22, 412–419. [Google Scholar] [CrossRef]
- Kenny, C. Radial displacement of the medial meniscus and Fairbank’s signs. Clin. Orthop. Relat. Res. 1997, 339, 163–173. [Google Scholar] [CrossRef]
- Rauscher, I.; Stahl, R.; Cheng, J.; Li, X.; Huber, M.B.; Luke, A.; Majumdar, S.; Link, T.M. Meniscal measurements of T1 p and T2 at MR imaging in healthy subjects and patients with osteoarthritis. Radiology 2008, 249, 591–600. [Google Scholar] [CrossRef]
- Hunter, D.J.; Buck, R.; Vignon, E.; Eckstein, F.; Brandt, K.; Mazzuca, S.A.; Wyman, B.T.; Otterness, I.; Hellio Le Graverand, M.P. Relation of regional articular cartilage morphometry and meniscal position by MRI to joint space width in knee radiographs. Osteoarthr. Cartil. 2009, 17, 1170–1176. [Google Scholar] [CrossRef][Green Version]
- Forriol, F. Growth factors in cartilage and meniscus repair. Injury 2009, 40, S12–S16. [Google Scholar] [CrossRef]
- Hunter, D.J.; Zhang, Y.Q.; Niu, J.B.; Tu, X.; Amin, S.; Clancy, M.; Guermazi, A.; Grigorian, M.; Gale, D.; Felson, D.T. The association of meniscal pathologic changes with cartilage loss in symptomatic knee osteoarthritis. Arthritis Rheum. 2006, 54, 795–801. [Google Scholar] [CrossRef]
- Berthiaume, M.J.; Raynauld, J.P.; Martel-Pelletier, J.; Labonté, F.; Beaudoin, G.; Bloch, D.A.; Choquette, D.; Haraoui, B.; Altman, R.D.; Hochberg, M.; et al. Meniscal tear and extrusion are strongly associated with progression of symptomatic knee osteoarthritis as assessed by quantitative magnetic resonance imaging. Ann. Rheum. Dis. 2005, 64, 556–563. [Google Scholar] [CrossRef]
- Kraus, V.B.; Feng, S.; Wang, S.C.; White, S.; Ainslie, M.; Brett, A.; Holmes, A.; Charles, H.C. Trabecular morphometry by fractal signature analysis is a novel marker of osteoarthritis progression. Arthritis Rheum. 2009, 60, 3711–3722. [Google Scholar] [CrossRef]
- Wolski, M.; Podsiadlo, P.; Stachowiak, G.W.; Lohmander, L.S.; Englund, M. Differences in trabecular bone texture between knees with and without radiographic osteoarthritis detected by directional fractal signature method. Osteoarthr. Cartil. 2010, 18, 684–690. [Google Scholar] [CrossRef]
- Hunter, D.J.; Zhang, Y.Q.; Tu, X.; LaValley, M.; Niu, J.B.; Amin, S.; Guermazi, A.; Genant, H.; Gale, D.; Felson, D.T. Change in joint space width: Hyaline articular cartilage loss or alteration in meniscus? Arthritis Rheum. 2006, 54, 2488–2495. [Google Scholar] [CrossRef]
- Englund, M.; Guermazi, A.; Roemer, F.W.; Yang, M.; Zhang, Y.; Nevitt, M.C.; Lynch, J.A.; Lewis, C.E.; Torner, J.; Felson, D.T. Meniscal pathology on MRI increases the risk for both incident and enlarging subchondral bone marrow lesions of the knee: The MOST study. Ann. Rheum. Dis. 2010, 69, 1796–1802. [Google Scholar] [CrossRef]
- Takahashi, K.; Yamanaka, S. Induction of Pluripotent Stem Cells from Mouse Embryonic and Adult Fibroblast Cultures by Defined Factors. Cell 2006, 25, 663–676. [Google Scholar] [CrossRef]
- Ullah, I.; Subbarao, R.B.; Rho, G.J. Human mesenchymal stem cells - current trends and future prospective. Biosci. Rep. 2015, 28, 35. [Google Scholar] [CrossRef]
- Fan, J.; Varshney, R.R.; Ren, L.; Cai, D.; Wang, D.A. Synovium-derived mesenchymal stem cells: A new cell source for musculoskeletal regeneration. Tissue Eng. 2009, 15, 75–86. [Google Scholar] [CrossRef]
- Friedenstein, A.J.; Gorskaja, U.F.; Kulagina, N.N. Fibroblast precursors in normal and irradiated mouse hematopoietic organs. Exp. Hematol. 1976, 4, 267–274. [Google Scholar]
- Fraser, J.K.; Wulur, I.; Alfonso, Z.; Hedrick, M.H. Fat tissue: An underappreciated source of stem cells for biotechnology. Trends Biotechnol. 2006, 24, 150–154. [Google Scholar] [CrossRef]
- Sakaguchi, Y.; Sekiya, I.; Yagishita, K.; Muneta, T. Comparison of human stem cells derived from various mesenchymal tissues: Superiority of synovium as a cell source. Arthritis Rheum. 2005, 52, 2521–2529. [Google Scholar] [CrossRef]
- Cao, C.; Dong, Y.; Dong, Y. Study on culture and in vitro osteogenesis of blood-derived human mesenchymal stem cells. Chin. J. Repar. Reconstruct. Surg. 2005, 19, 642–647. [Google Scholar]
- Yoshimura, H.; Muneta, T.; Nimura, A.; Yokoyama, A.; Koga, H.; Sekiya, I. Comparison of rat mesenchymal stem cells derived from bone marrow, synovium, periosteum, adipose tissue, and muscle. Cell Tissue Res. 2007, 327, 449–462. [Google Scholar] [CrossRef]
- Shirasawa, S.; Sekiya, I.; Sakaguchi, Y.; Yagishita, K.; Ichinose, S.; Muneta, T. In vitro chondrogenesis of human synovium-derived mesenchymal stem cells: Optimal condition and comparison with bone marrow-derived cells. J. Cell. Biochem. 2006, 97, 84–97. [Google Scholar] [CrossRef]
- Mochizuki, T.; Muneta, T.; Sakaguchi, Y.; Nimura, A.; Yokoyama, A.; Koga, H.; Sekiya, I. Higher chondrogenic potential of fibrous synovium- and adipose synovium-derived cells compared with subcutaneous fat-derived cells: Distinguishing properties of mesenchymal stem cells in humans. Arthritis Rheum. 2006, 54, 848–853. [Google Scholar] [CrossRef]
- Banfi, A.; Muraglia, A.; Dozin, B.; Mastrogiacomo, M.; Cancedda, R.; Quarto, R. Proliferation kinetics and differentiation potential of ex vivo expanded human bone marrow stromal cells: Implications for their use in cell therapy. Exp. Hematol. 2000, 28, 707–715. [Google Scholar] [CrossRef]
- Aust, L.; Devlin, B.; Foster, S.J.; Halvorsen, Y.D.C.; Hicok, K.; du Laney, T.; Sen, A.; Willingmyre, G.D.; Gimble, J.M. Yield of human adipose-derived adult stem cells from liposuction aspirates. Cytotherapy 2004, 6, 7–14. [Google Scholar] [CrossRef]
- Im, G.I.; Shin, Y.W.; Lee, K.B. Do adipose tissue-derived mesenchymal stem cells have the same osteogenic and chondrogenic potential as bone marrow-derived cells? Osteoarthr. Cartil. 2005, 13, 845–853. [Google Scholar] [CrossRef]
- De Albornoz, P.M.; Forriol, F. The meniscal healing process. Muscles Ligaments Tendons J. 2012, 2, 10–18. [Google Scholar] [PubMed]
- Guisasola, I.; Vaquero, J.; Forriol, F. Knee immobilization on meniscal healing after suture: An experimental study in sheep. Clin. Orthop. Relat. Res. 2002, 395, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Caplan, A.I.; Dennis, J.E. Mesenchymal stem cells as trophic mediators. J. Cell. Biochem. 2006, 98, 1076–1084. [Google Scholar] [CrossRef] [PubMed]
- Dehaven, K.E.; Lohrer, W.A.; Lovelock, J.E. Long-Term Results of Open Meniscal Repair. Am. J. Sports Med. 1995, 23, 524–530. [Google Scholar] [CrossRef]
- Matsukura, Y.; Muneta, T.; Tsuji, K.; Koga, H.; Sekiya, I. Mesenchymal stem cells in synovial fluid increase after meniscus injury. Clin. Orthop. Relat. Res. 2014, 472, 1357–1364. [Google Scholar] [CrossRef]
- King, T.V.; Vallee, B.L. Neovascularisation of the meniscus with angiogenin: An experimental study in rabbits. J. Bone Jt. Surg. 1991, 73, 587–590. [Google Scholar] [CrossRef]
- Zhongran, Z.; Kaiyuan, T.; Yinkan, X.; Wenming, Z.; Zhentian, L.; Shihuan, O. Treatment of longitudinal injuries in avascular area of meniscus in dogs by trepanation. Arthrosc. J. Arthrosc. Relat. Surg. 1988, 4, 151–159. [Google Scholar] [CrossRef]
- Arnoczky, S.P.; Warren, R.F. The microvasculature of the meniscus and its response to injury: An experimental study in the dog. Am. J. Sports Med. 1983, 11, 131–141. [Google Scholar] [CrossRef]
- Cabaud, H.E.; Rodkey, W.G.; Fitzwater, J.E. Medial meniscus repairs. An experimental and morphologic study. Am. J. Sports Med. 1981, 9, 129–134. [Google Scholar] [CrossRef]
- Heatley, F.W. The meniscus—Can it be repaired? An experimental investigation in rabbits. J. Bone Jt. Surg. 1980, 62, 397–402. [Google Scholar] [CrossRef]
- Hofer, H.R.; Tuan, R.S. Secreted trophic factors of mesenchymal stem cells support neurovascular and musculoskeletal therapies. Stem Cell Res. Ther. 2016, 7, 131. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, Y.; Fortier, L.A.; Mao, J.J.; Lee, C.H.; Goodale, M.B.; Koff, M.F.; Uppstrom, T.J.; Croen, B.; Wada, S.; Carballo, C.B.; et al. Long-term Evaluation of Meniscal Tissue Formation in 3-dimensional–Printed Scaffolds with Sequential Release of Connective Tissue Growth Factor and TGF-β3 in an Ovine Model. Am. J. Sports Med. 2019, 47, 2596–2607. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.H.; Rodeo, S.A.; Fortier, L.A.; Lu, C.; Erisken, C.; Mao, J.J. Protein-releasing polymeric scaffolds induce fibrochondrocytic differentiation of endogenous cells for knee meniscus regeneration in sheep. Sci. Transl. Med. 2014, 6, 266ra171. [Google Scholar] [CrossRef]
- Bray, R.C.; Smith, J.A.; Eng, M.K.; Leonard, C.A.; Sutherland, C.A.; Salo, P.T. Vascular response of the meniscus to injury: Effects of immobilization. J. Orthop. Res. 2001, 19, 384–390. [Google Scholar] [CrossRef]
- Dowdy, P.A.; Miniaci, A.; Arnoczky, S.P.; Fowler, P.J.; Boughner, D.R. The Effect of Cast Immobilization on Meniscal Healing: An Experimental Study in the Dog. Am. J. Sports Med. 1995, 23, 721–728. [Google Scholar] [CrossRef]
- McNulty, A.L.; Guilak, F. Mechanobiology of the meniscus. J. Biomech. 2015, 48, 1469–1478. [Google Scholar] [CrossRef]
- Koga, H.; Muneta, T.; Nagase, T.; Nimura, A.; Ju, Y.J.; Mochizuki, T.; Sekiya, I. Comparison of mesenchymal tissues-derived stem cells for in vivo chondrogenesis: Suitable conditions for cell therapy of cartilage defects in rabbit. Cell Tissue Res. 2008, 333, 207–215. [Google Scholar] [CrossRef]
- Nimura, A.; Muneta, T.; Koga, H.; Mochizuki, T.; Suzuki, K.; Makino, H.; Umezawa, A.; Sekiya, I. Increased proliferation of human synovial mesenchymal stem cells with autologous human serum: Comparisons with bone marrow mesenchymal stem cells and with fetal bovine serum. Arthritis Rheum. 2008, 58, 501–510. [Google Scholar] [CrossRef]
- Nakagawa, Y.; Muneta, T.; Kondo, S.; Mizuno, M.; Takakuda, K.; Ichinose, S.; Tabuchi, T.; Koga, H.; Tsuji, K.; Sekiya, I. Synovial mesenchymal stem cells promote healing after meniscal repair in microminipigs. Osteoarthr. Cartil. 2015, 23, 1007–1017. [Google Scholar] [CrossRef]
- Hatsushika, D.; Muneta, T.; Nakamura, T.; Horie, M.; Koga, H.; Nakagawa, Y.; Tsuji, K.; Hishikawa, S.; Kobayashi, E.; Sekiya, I. Repetitive allogeneic intraarticular injections of synovial mesenchymal stem cells promote meniscus regeneration in a porcine massive meniscus defect model. Osteoarthr. Cartil. 2014, 22, 941–950. [Google Scholar] [CrossRef]
- Ruiz-Ibán, M.N.; Díaz-Heredia, J.; García-Gómez, I.; Gonzalez-Lizán, F.; Elías-Martín, E.; Abraira, V. The effect of the addition of adipose-derived mesenchymal stem cells to a meniscal repair in the avascular zone: An experimental study in rabbits. Arthroscopy 2011, 27, 1688–1696. [Google Scholar] [CrossRef] [PubMed]
- Ferris, D.J.; Frisbie, D.D.; Kisiday, J.D.; Mcilwraith, C.W.; Hague, B.A.; Major, M.D.; Schneider, R.K.; Zubrod, C.J.; Kawcak, C.E.; Goodrich, L.R. Clinical outcome after intra-articular administration of bone marrow derived mesenchymal stem cells in 33 horses with stifle injury. Vet. Surg. 2014, 43, 255–265. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Hamid, M.; Hussein, M.R.; Ahmad, A.F.; Elgezawi, E.M. Enhancement of the repair of meniscal wounds in the red-white zone (middle third) by the injection of bone marrow cells in canine animal model. Int. J. Exp. Pathol. 2005, 86, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo Perea, S.; Lyons, L.P.; Nishimuta, J.F.; Weinberg, J.B.; McNulty, A.L. Evaluation of culture conditions for in vitro meniscus repair model systems using bone marrow-derived mesenchymal stem cells. Connect. Tissue Res. 2019, 29, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Thomas Vangsness, J.; Burke, W.S.; Narvy, S.J.; MacPhee, R.D.; Fedenko, A.N. Human knee synovial fluid cytokines correlated with grade of knee osteoarthritis: A pilot study. Bull. NYU Hosp. Jt. Dis. 2011, 69, 122–127. [Google Scholar]
- Hennerbichler, A.; Moutos, F.T.; Hennerbichler, D.; Weinberg, J.B.; Guilak, F. Interleukin-1 and tumor necrosis factor alpha inhibit repair of the porcine meniscus in vitro. Osteoarthr. Cartil. 2007, 15, 1053–1060. [Google Scholar] [CrossRef]
- McNulty, A.L.; Moutos, F.T.; Weinberg, J.B.; Guilak, F. Enhanced integrative repair of the porcine meniscus in vitro by inhibition of interleukin-1 or tumor necrosis factor α. Arthritis Rheum. 2007, 56, 3033–3042. [Google Scholar] [CrossRef]
- Upton, M.L.; Guilak, F.; Laursen, T.A.; Setton, L.A. Finite element modeling predictions of region-specific cell-matrix mechanics in the meniscus. Biomech. Model. Mechanobiol. 2006, 5, 140–149. [Google Scholar] [CrossRef]
- Zhang, H.; Leng, P.; Zhang, J. Enhanced meniscal repair by overexpression of hIGF-1 in a full-thickness model. Clin. Orthop. Relat. Res. 2009, 467, 3165–3174. [Google Scholar] [CrossRef]
- Moriguchi, Y.; Tateishi, K.; Ando, W.; Shimomura, K.; Yonetani, Y.; Tanaka, Y.; Kita, K.; Hart, D.A.; Gobbi, A.; Shino, K.; et al. Repair of meniscal lesions using a scaffold-free tissue-engineered construct derived from allogenic synovial MSCs in a miniature swine model. Biomaterials 2013, 34, 2185–2193. [Google Scholar] [CrossRef]
- Kondo, S.; Muneta, T.; Nakagawa, Y.; Koga, H.; Watanabe, T.; Tsuji, K.; Sotome, S.; Okawa, A.; Kiuchi, S.; Ono, H.; et al. Transplantation of autologous synovial mesenchymal stem cells promotes meniscus regeneration in aged primates. J. Orthop. Res. 2017, 35, 1274–1282. [Google Scholar] [CrossRef] [PubMed]
- Desando, G.; Giavaresi, G.; Cavallo, C.; Bartolotti, I.; Sartoni, F.; Nicoli Aldini, N.; Martini, L.; Parrilli, A.; Mariani, E.; Fini, M.; et al. Autologous Bone Marrow Concentrate in a Sheep Model of Osteoarthritis: New Perspectives for Cartilage and Meniscus Repair. Tissue Eng. 2016, 22, 608–619. [Google Scholar] [CrossRef] [PubMed]
- Marcacci, M.; Berruto, M.; Brocchetta, D.; Delcogliano, A.; Ghinelli, D.; Gobbi, A.; Kon, E.; Pederzini, L.; Rosa, D.; Sacchetti, G.L.; et al. Articular cartilage engineering with Hyalograft® C: 3-Year clinical results. Clin. Orthop. Relat. Res. 2005, 435, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, P.G.; Magna, H.A.; Reeves, L.M.; Lopresti-Morrow, L.L.; Yocum, S.A.; Rosner, P.J.; Geoghegan, K.F.; Hambor, J.E. Cloning, expression, and type II collagenolytic activity of matrix metalloproteinase-13 from human osteoarthritic cartilage. J. Clin. Investig. 1996, 97, 761–768. [Google Scholar] [CrossRef]
- Mengshol, J.A. IL-1 induces collagenase-3 (MMP-13) promoter activity in stably transfected chondrocytic cells: Requirement for Runx-2 and activation by p38 MAPK and JNK pathways. Nucleic Acids Res. 2001, 29, 4361–4372. [Google Scholar] [CrossRef]
- Massoud, D.; Jian, Y.Q. The interleukin 1beta pathway in the pathogenesis of osteoarthritis. J. Rheumatol. 2008, 35, 2306–2312. [Google Scholar]
- Vangsness, C.T.; Farr, J.; Boyd, J.; Dellaero, D.T.; Mills, C.R.; LeRoux-Williams, M. Adult human mesenchymal stem cells delivered via intra-articular injection to the knee following partial medial meniscectomy A Randomized, Double-Blind, Controlled Study. J. Bone Jt. Surg. 2014, 96, 90–98. [Google Scholar] [CrossRef]
- Pak, J.; Lee, J.H.; Lee, S.H. Regenerative repair of damaged meniscus with autologous adipose tissue-derived stem cells. BioMed Res. Int. 2014, 2014, 436029. [Google Scholar] [CrossRef]
- Centeno, C.J.; Busse, D.; Kisiday, J.; Keohan, C.; Freeman, M.; Karli, D. Regeneration of meniscus cartilage in a knee treated with percutaneously implanted autologous mesenchymal stem cells. Med. Hypotheses 2008, 71, 900–908. [Google Scholar] [CrossRef]
- Onoi, Y.; Hiranaka, T.; Nishida, R.; Takase, K.; Fujita, M.; Hida, Y.; Fujishiro, T.; Okamoto, K. Second-look arthroscopic findings of cartilage and meniscus repair after injection of adipose-derived regenerative cells in knee osteoarthrits: Report of two cases. Regen. Ther. 2019, 11, 212–216. [Google Scholar] [CrossRef]
- Sekiya, I.; Koga, H.; Otabe, K.; Nakagawa, Y.; Katano, H.; Ozeki, N.; Mizuno, M.; Horie, M.; Kohno, Y.; Katagiri, K.; et al. Additional Use of Synovial Mesenchymal Stem Cell Transplantation Following Surgical Repair of a Complex Degenerative Tear of the Medial Meniscus of the Knee: A Case Report. Cell Transplant. 2019, 28, 1445–1454. [Google Scholar] [CrossRef] [PubMed]
- Whitehouse, M.R.; Howells, N.R.; Parry, M.C.; Austin, E.; Kafienah, W.; Brady, K.; Goodship, A.E.; Eldridge, J.D.; Blom, A.W.; Hollander, A.P. Repair of torn avascular meniscal cartilage using undifferentiated autologous mesenchymal stem cells: From in vitro optimization to a first-in-human study. Stem Cells Transl. Med. 2017, 6, 1237–1248. [Google Scholar] [CrossRef] [PubMed]
- Olivos-Meza, A.; Pérez Jiménez, F.J.; Granados-Montiel, J.; Landa-Solís, C.; Cortés González, S.; Jiménez Aroche, C.A.; Valdez Chávez, M.; Renán León, S.; Gomez-Garcia, R.; Martínez-López, V.; et al. First Clinical Application of Polyurethane Meniscal Scaffolds with Mesenchymal Stem Cells and Assessment of Cartilage Quality with T2 Mapping at 12 Months. Cartilage 2019. [Google Scholar] [CrossRef] [PubMed]
| MSC Source | Osteogenic | Chondrogenic | Adipogenic | Clinical Advantage | Clinical Disadvantage |
|---|---|---|---|---|---|
| Bone marrow | +++ | +++ | ++ | Aspiration can be done under L/A | Invasive, Painful, Low yield |
| Adipose | + | + | +++ | Less painful than marrow aspiration and high yield | L/A toxic to ASCs therefore harvest preferable under GA |
| Synovium | +++ | +++ | +++ | Painless, Minimally invasive and Minimal tissue requirement | Staged surgery, cells require expansion |
| Author/Year | Animal/Defect Model | Source/Cell number/Method of Delivery | Control | Outcome Measurement/Timeline | Results |
|---|---|---|---|---|---|
| Nakagawa et al. [90]/2015 | Micro minipig/Medial Meniscal full thickness longitudinal tear | Allogeneic Synovial MSCs/20 × 106/Suture repair +MSC suspension injection | Suture repair + Acellular suspension | Macro and Histo analysis, IMHC, TEM, MRI, Biomechanical analysis/12 weeks | Macroscopy: Scores were better in MSC group at all time points compared to control. Histology: Scores were higher in MSC group at all time points compared to control. TEM: dense collagen fibrils in MSC group, none in control. MRI: MSCs group has T1rho values closer to intact meniscus than control. Higher tensile strength in MSC group |
| Hatsushika et al. [91]/2014 | Pig/Medial meniscus anterior half resection | Allogeneic Synovial MSCs/50 × 106 × 3/IA injections x3 with 2-week gaps of synovial MSCs | PBS injection | Macro and Histo analysis. IMHC TEM MRI/16 weeks | Macro: regeneration of anterior medial meniscus in both groups. Histo: better Safranin-O staining in MSC group, COL I and II staining showed larger representation in MSC group. Mod Pauli’s score was higher in MSC group. MRI: regenerate area appeared more organized in MSC group |
| Ferris et al. [93]/2014 | Horse/Meniscal tear | Autologous BMSCs/15–20 × 106/Arthroscopy + IA injection of BMSCs | Previous surgical data | Return to work/24 months | 18/24 (75%) horses with meniscal lesions returned to work. 9 returned to previous level of activity |
| Abdel-Hamid et al. [94]/2005 | Dog/Longitudinal full thickness meniscal tear | Autologous BMSCs/2-4ml aspirate/Injection at tear site | Tear with no treatment | Histo, IMHC/12 weeks | Better healing response in injected group compared to control. Histo: angiogenesis, collagen deposition and fibroblast proliferation in injected compared to control |
| Author/Year | Animal/Defect Model | Source/Cell number/Method of Delivery | Control | Outcome Measurement/Timeline | Results |
|---|---|---|---|---|---|
| Zhang et al. [100]/2009 | Goat/full thickness defect in medial meniscus anterior horn | BMMSC with transfection of hIGF-1/30 × 106/mL/Calcium alginate gel into defect | Defect with nil treatment | Histo, TEM, GAG Assay MRI/16 weeks | BMMSC w/hIGF-1 group had better repair tissue without clear margin. Large number of well aligned cells within repair defect. TEM showed round oval like chondrocyte like cells. MRI: smooth continuous anterior horn Higher GAG content to control |
| Moriguchi et al. [101]/2013 | Pig/4 mm cylindrical defect in medial meniscus | Synovial MSC/0.2 × 106 cells—3 weeks culture/3D matrix construct (TEC) | Nil treatment | Gross morphology Histo/6 months | TEC implanted defects showed fibrocartilaginous repair and integration compared to control. Histo: cartilage like cells with nuclei in lacuna |
| Kondo et al. [102]/2017 | Monkey/Anterior horn of medial meniscus Partial Meniscectomy | Synovial MSCs/0.25 × 106/Aggregates | Nil aggerate | Macro and Histo analysis MRI/8 weeks (n = 3) 16 weeks (n = 4) | Macro: Regeneration in control and MSCs groups with MSC showed larger medial meniscus at 8 and 16 weeks. Histo: Safranin-O slight staining at 8 weeks, positive at 16 weeks. No staining in control MRI: MSC groups closer resembled intact menisci compared to control. |
| Desando et al. [103]/2016 | Sheep/Unilateral medial meniscectomy | Bone marrow concentrate or BMSCs/BMC:39 × 106 BMSCs: 6 × 106/Arthrotomy Bone marrow or BMMSC in HA mesh | Nil treatment | Gross morphology Microtomography Histo Immunohistology/12 weeks | Meniscal tissue regeneration greatest in BMC + HA group. Both BMC and BMSCs group showed good cell density and proteoglycan content compared to control. BMC+ HA group had higher expression of Col II than I compared to BMSCs group. |
| Author/Year | Study type/Patient number | Source/Cell Number | Method of Delivery | Outcome/Follow Up | Results |
|---|---|---|---|---|---|
| Vangsness et al. [108]/2014 | Randomized control trial/55 | Allogeneic MSCs derived from BMAC/A:50 × 106 B:150 × 106 | Percutaneous knee injection | MRI VAS Lysholm knee score/2 years | Significant improvement in scores at 3 months. 12-month MRI at 12 months: significant increase in meniscal volume in MSC groups compared to control |
| Pak et al. [109]/2014 | Case control/1 | Abdominal liposuction/Not reported | Percutaneous knee injection | VAS, Functional rating index, ROM, MRI/18 months | At 3 months MRI showed no evidence of meniscal tear, Symptoms improved and asymptomatic at 18 months |
| Centeno CJ et al. [110]/2008 | Case control/1 | Iliac crest BMAC/45.6 × 106 | Percutaneous knee injection | VAS, Functional rating index, MRI/3 months | Increased meniscus volume on MRI. Decreased VAS Score from 3.33 to 0.13 |
| Onoi et al. [111]/2019 | Case report/2 | Liposuction from thigh/5.5 × 106 | Percutaneous knee injection | MRI KOOS Arthroscopy/6 months | Both patients reported better scores at 6 months follow up. 2nd look arthroscopy showed meniscal tear healing |
| Sekiya et al. [112]/2019 | Case series/5 | Arthroscopically harvested Synovial Tissue/32–70 × 106 | Arthroscopic transplantation of autologous synovial MSC suspension to sutured meniscal lesion | Lysholm knee score KOOS NRS 3D MRI/24 months | Significant improvement of Lysholm score by 1 year. Other scores significantly increased by 2 years 3D MRI: Tears were indistinguishable |
| Author/Year | Study Type/Patient Number | Source/Cell Number | Method of Delivery | Outcome/Follow Up | Results |
|---|---|---|---|---|---|
| Whitehouse et al. [113]/2017 | Case Series/5 | Iliac crest BMAC/1 × 106/cm2 | Arthroscopic MSC injection into Collagen Scaffold | IKDC Score Lysholm Score. ROM MRI/2 years | 3 patients reported significantly improved clinical outcomes and MRI imaging |
| Olivos-Meza [114]/2019 | Case Series/17 | s/c G-CSF x 3 blood draws. Cell separation isolation and culture CD 90+ cells/20 × 106 | Arthroscopic implantation of MSC cell seeded polyurethane scaffold vs. acellular polyurethane scaffold | Lysholm Score MRI/12 months | Both groups improved in Lysholm scores. No intergroup difference was noted. MRI Cartigram: Improved in cell seeded scaffold at 9 months but reduced to initial value at 12 months |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jacob, G.; Shimomura, K.; Krych, A.J.; Nakamura, N. The Meniscus Tear: A Review of Stem Cell Therapies. Cells 2020, 9, 92. https://doi.org/10.3390/cells9010092
Jacob G, Shimomura K, Krych AJ, Nakamura N. The Meniscus Tear: A Review of Stem Cell Therapies. Cells. 2020; 9(1):92. https://doi.org/10.3390/cells9010092
Chicago/Turabian StyleJacob, George, Kazunori Shimomura, Aaron J. Krych, and Norimasa Nakamura. 2020. "The Meniscus Tear: A Review of Stem Cell Therapies" Cells 9, no. 1: 92. https://doi.org/10.3390/cells9010092
APA StyleJacob, G., Shimomura, K., Krych, A. J., & Nakamura, N. (2020). The Meniscus Tear: A Review of Stem Cell Therapies. Cells, 9(1), 92. https://doi.org/10.3390/cells9010092





