Case Report on: Very Early Afterdepolarizations in HiPSC-Cardiomyocytes—An Artifact by Big Conductance Calcium Activated Potassium Current (Ibk,Ca)
Abstract
1. Introduction
2. Methods
2.1. Generation of hiPSC and Engineered Heart Tissue (EHT)
2.2. Human Adult Heart Tissue
2.3. Current Recordings
2.4. AP Recordings
2.5. Molecular Biology
2.6. Mathematical Modelling and Computer Simulations
2.7. Statistical Analysis
2.8. Drugs
3. Results
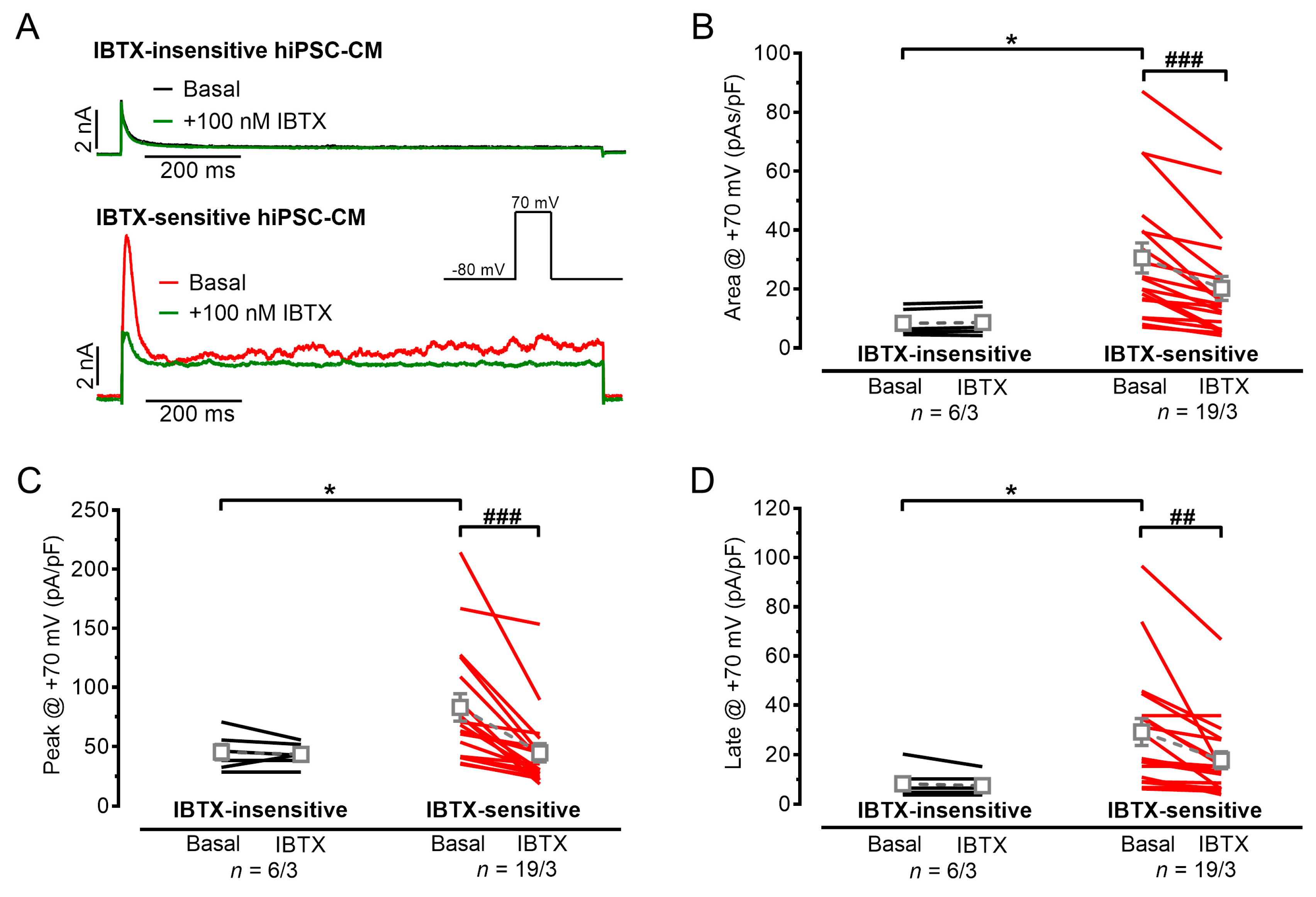
3.1. Outward Potassium Currents in hiPSC-CMs, Appearance of IBK,Ca
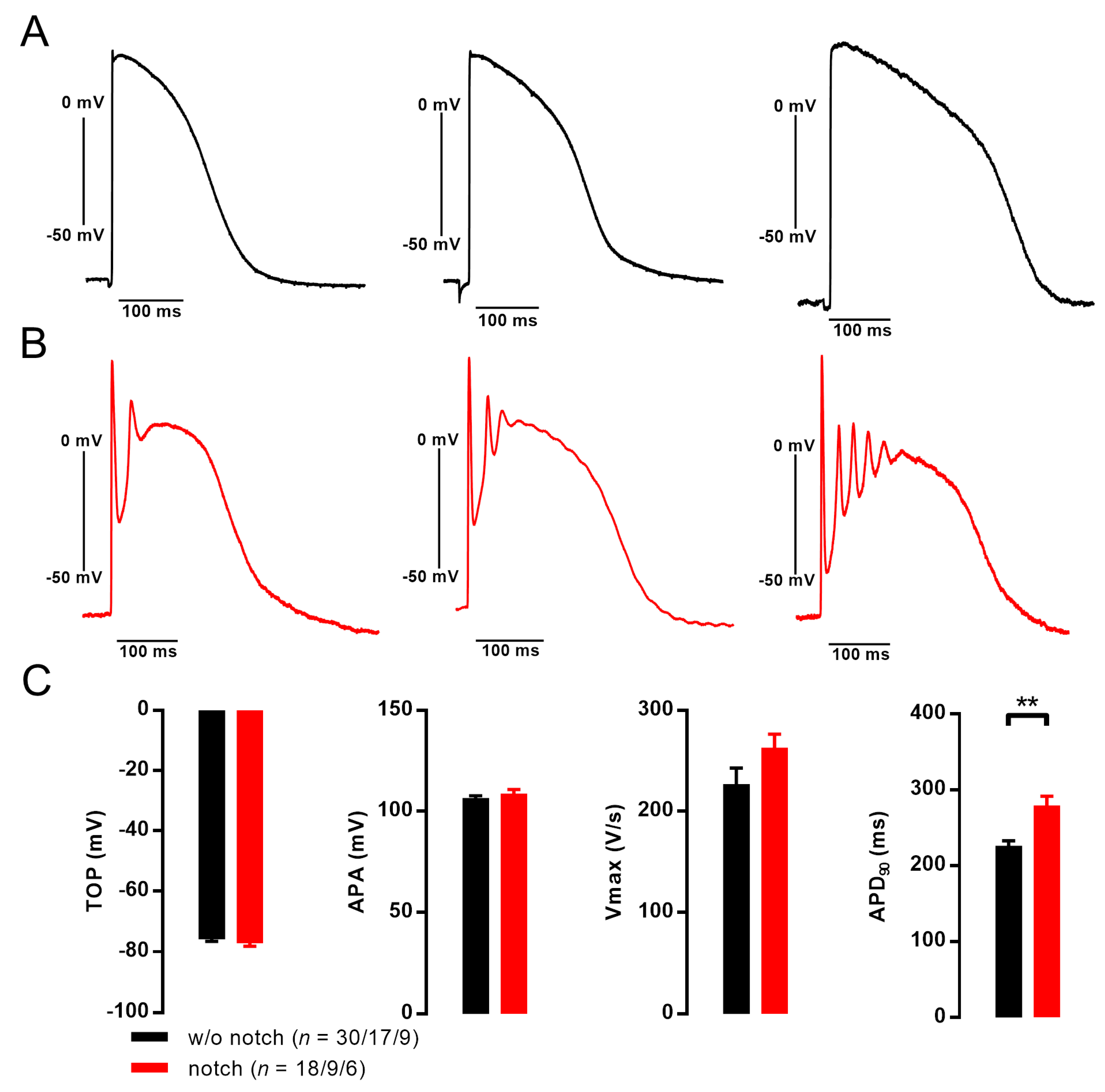
3.2. Action Potentials with Strong Initial Repolarization and Oscillations Are Sensitive to Iberiotoxin
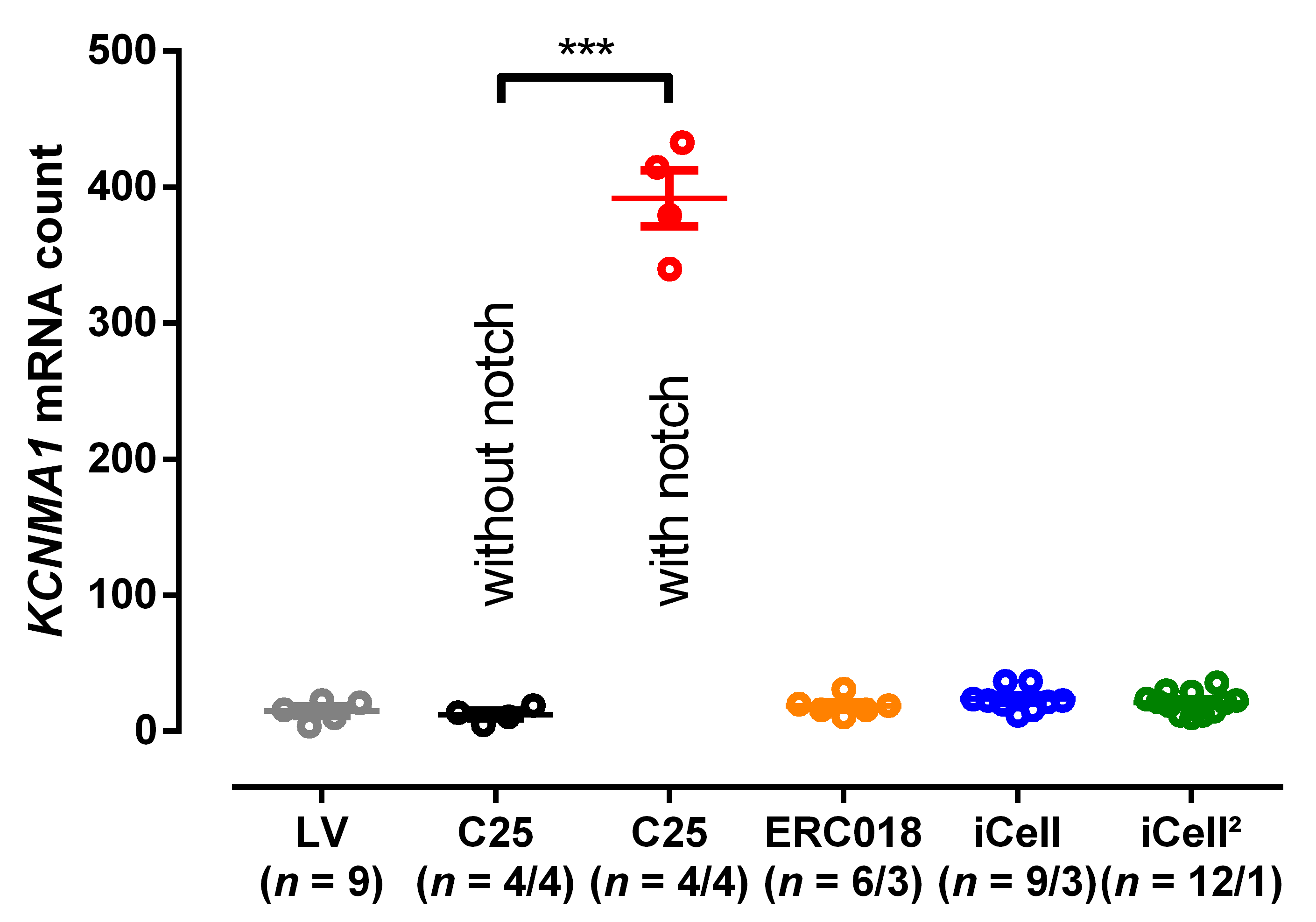
3.3. EHT with Notch/Oscillations in the AP Showed Large BKCa Expression
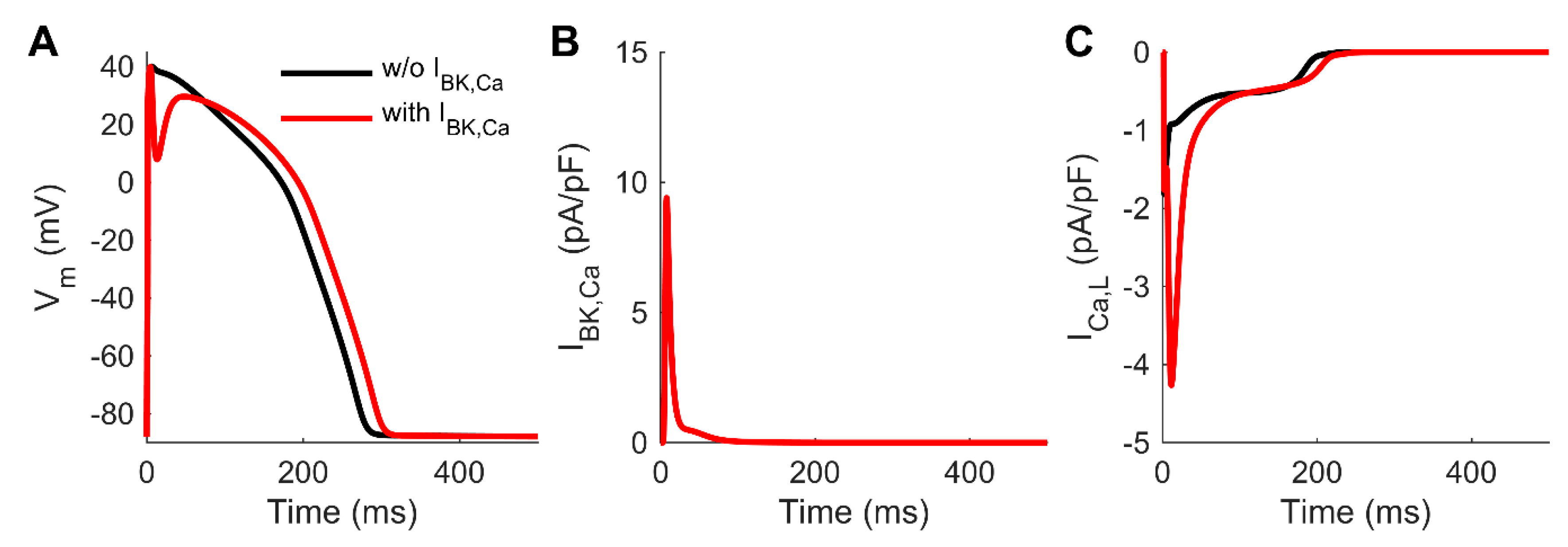
3.4. Computer Simulations Corroborate the Effect of the Non-Cardiac BKCa to Human AP
4. Discussion
- (1)
- HiPSC-CMs could express ion channels otherwise not found in adult cardiomyocytes.
- (2)
- In human adult ventricular myocardium, BKCa did not contribute to AP shape.
- (3)
- In hiPSC-CMs BKCa could induce irregular AP shapes with oscillations resembling very early after depolarizations.
5. Conclusions
6. Limitations
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AP | Action potential |
| APD | Action potential duration |
| APD90 | Action potential duration at 90% repolarization |
| BKCa | Big conductance calcium activated potassium channel, (Maxi-K, slo1, KCa1.1) |
| CM | Cardiomyocytes |
| EAD | Early afterdepolarization |
| EHT | Engineered heart tissue |
| hiPSC-CMs | Human induced pluripotent stem cell-derived cardiomyocytes |
| IBTX | Iberiotoxin |
| IBK,Ca | Big-conductance calcium activated potassium current |
| ICa,L | L-type calcium current |
| LV | Left ventricle |
| RMP | Resting membrane potential |
References
- Moretti, A.; Bellin, M.; Welling, A.; Jung, C.B.; Lam, J.T.; Bott-Flügel, L.; Dorn, T.; Goedel, A.; Höhnke, C.; Hofmann, F.; et al. Patient-Specific Induced Pluripotent Stem-Cell Models for Long-QT Syndrome. N. Engl. J. Med. 2010, 363, 1397–1409. [Google Scholar] [CrossRef] [PubMed]
- Itzhaki, I.; Maizels, L.; Huber, I.; Zwi-Dantsis, L.; Caspi, O.; Winterstern, A.; Feldman, O.; Gepstein, A.; Arbel, G.; Hammerman, H.; et al. Modelling the long QT syndrome with induced pluripotent stem cells. Nature 2011, 471, 225–229. [Google Scholar] [CrossRef] [PubMed]
- Carvajal-vergara, X.; Sevilla, A.; Souza, S.L.D.; Ang, Y.; Lee, D.; Yang, L.; Kaplan, A.D.; Adler, E.D.; Rozov, R.; Ge, Y.; et al. Patient-specific induced pluripotent stem-cell-derived models of LEOPARD syndrome. Nature 2010, 465, 808–812. [Google Scholar] [CrossRef] [PubMed]
- Liang, P.; Lan, F.; Lee, A.S.; Gong, T.; Sanchez-Freire, V.; Wang, Y.; Diecke, S.; Sallam, K.; Knowles, J.W.; Wang, P.J.; et al. Drug screening using a library of human induced pluripotent stem cell-derived cardiomyocytes reveals disease-specific patterns of cardiotoxicity. Circulation 2013, 127, 1677–1691. [Google Scholar] [CrossRef]
- Lemoine, M.D.; Krause, T.; Koivumaki, J.T.; Prondzynski, M.; Schulze, M.L.; Girdauskas, E.; Willems, S.; Hansen, A.; Eschenhagen, T.; Christ, T. Human Induced Pluripotent Stem Cell-Derived Engineered Heart Tissue as a Sensitive Test System for QT Prolongation and Arrhythmic Triggers. Circ. Arrhythmia Electrophysiol. 2018, 11, e006035. [Google Scholar] [CrossRef]
- Weinberger, F.; Breckwoldt, K.; Pecha, S.; Kelly, A.; Geertz, B.; Starbatty, J.; Yorgan, T.; Cheng, K.-H.; Lessmann, K.; Stolen, T.; et al. Cardiac repair in guinea pigs with human engineered heart tissue from induced pluripotent stem cells. Sci. Transl. Med. 2016, 8, ra148–ra363. [Google Scholar] [CrossRef]
- Ma, J.; Guo, L.; Fiene, S.J.; Anson, B.D.; Thomson, J.A.; Kamp, T.J.; Kolaja, K.L.; Swanson, B.J.; January, C.T.; Kl, K.; et al. High purity human-induced pluripotent stem cell-derived cardiomyocytes: Electrophysiological properties of action potentials and ionic currents. Am. J. Physiol. Heart Circ. Physiol. 2011, 301, 2006–2017. [Google Scholar] [CrossRef]
- Herron, T.J.; Da Rocha, A.M.; Campbell, K.F.; Ponce-Balbuena, D.; Willis, B.C.; Guerrero-Serna, G.; Liu, Q.; Klos, M.; Musa, H.; Zarzoso, M.; et al. Extracellular matrix-mediated maturation of human pluripotent stem cell-derived cardiac monolayer structure and electrophysiological function. Circ. Arrhythmia Electrophysiol. 2016, 9, e003638. [Google Scholar] [CrossRef]
- Horváth, A.; Lemoine, M.D.; Löser, A.; Mannhardt, I.; Flenner, F.; Uzun, A.U.; Neuber, C.; Breckwoldt, K.; Hansen, A.; Girdauskas, E.; et al. Low Resting Membrane Potential and Low Inward Rectifier Potassium Currents Are Not Inherent Features of hiPSC-Derived Cardiomyocytes. Stem Cell Rep. 2018, 10, 822–833. [Google Scholar] [CrossRef]
- Vaidyanathan, R.; Markandeya, Y.S.; Kamp, T.J.; Makielski, J.C.; Janaury, C.T.; Eckhardt, L.L. IK1-Enhanced Human Induced Pluripotent Stem Cell-Derived Cardiomyocytes: An Improved Cardiomyocyte Model to Investigate Inherited Arrhythmia Syndromes. Am. J. Physiol. Heart Circ. Physiol. 2016, 310, H1611–H1621. [Google Scholar] [CrossRef]
- Uzun, A.U.; Mannhardt, I.; Breckwoldt, K.; Horváth, A.; Johannsen, S.S.; Hansen, A.; Eschenhagen, T.; Christ, T. Ca2+-currents in human induced pluripotent stem cell-derived cardiomyocytes effects of two different culture conditions. Front. Pharmacol. 2016, 7, 300. [Google Scholar] [CrossRef] [PubMed]
- Ivashchenko, C.Y.; Pipes, G.C.; Lozinskaya, I.M.; Lin, Z.; Xiaoping, X.; Needle, S.; Grygielko, E.T.; Hu, E.; Toomey, J.R.; Lepore, J.J.; et al. Human-induced pluripotent stem cell-derived cardiomyocytes exhibit temporal changes in phenotype. Am. J. Physiol. Heart Circ. Physiol. 2013, 305, H913–H922. [Google Scholar] [CrossRef] [PubMed]
- Stimers, J.R.; Song, L.; Rusch, N.J.; Rhee, S.W. Overexpression of the large-conductance, Ca2+-activated K+(BK) channel shortens action potential duration in HL-1 cardiomyocytes. PLoS ONE 2015, 10, e0130588. [Google Scholar] [CrossRef] [PubMed]
- Mannhardt, I.; Breckwoldt, K.; Letuffe-Brenière, D.; Schaaf, S.; Schulz, H.; Neuber, C.; Benzin, A.; Werner, T.; Eder, A.; Schulze, T.; et al. Human Engineered Heart Tissue: Analysis of Contractile Force. Stem Cell Rep. 2016, 7, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Lanier, M.; Schade, D.; Willems, E.; Tsuda, M.; Spiering, S.; Kalisiak, J.; Mercola, M.; Cashman, J.R. Wnt Inhibition Correlates with Human Embryonic Stem Cell Cardiomyogenesis: A Structure—Activity Relationship Study Based on Inhibitors for the Wnt Response. J. Med. Chem. 2012, 55, 697–708. [Google Scholar] [CrossRef] [PubMed]
- Breckwoldt, K.; Letuffe-Brenière, D.; Mannhardt, I.; Schulze, T.; Ulmer, B.; Werner, T.; Benzin, A.; Klampe, B.; Reinsch, M.C.; Laufer, S.; et al. Differentiation of cardiomyocytes and generation of human engineered heart tissue. Nat. Protoc. 2017, 12, 1177–1197. [Google Scholar] [CrossRef]
- Ulmer, B.M.; Stoehr, A.; Schulze, M.L.; Patel, S.; Gucek, M.; Mannhardt, I.; Funcke, S.; Murphy, E.; Eschenhagen, T.; Hansen, A. Contractile Work Contributes to Maturation of Energy Metabolism in hiPSC-Derived Cardiomyocytes. Stem Cell Rep. 2018, 10, 834–847. [Google Scholar] [CrossRef]
- Schaaf, S.; Eder, A.; Vollert, I.; Stöhr, A.; Hansen, A.; Eschenhagen, T. Generation of Strip-Format Fibrin-Based Engineered Heart Tissue (EHT). In Cardiac Tissue Engineering: Methods and Protocols; Radisic, M., Black, D.L., III, Eds.; Springer: New York, NY, USA, 2014; pp. 121–129. ISBN 978-1-4939-1047-2. [Google Scholar]
- Christ, T.; Boknik, P.; Wöhrl, S.; Wettwer, E.; Graf, E.M.; Bosch, R.F.; Knaut, M.; Schmitz, W.; Ravens, U.; Dobrev, D. L-type Ca2+ current downregulation in chronic human atrial fibrillation is associated with increased activity of protein phosphatases. Circulation 2004, 110, 2651–2657. [Google Scholar] [CrossRef]
- Dobrev, D.; Friedrich, A.; Voigt, N.; Jost, N.; Wettwer, E.; Christ, T.; Knaut, M.; Ravens, U. The G protein-gated potassium current IK,ACh is constitutively active in patients with chronic atrial fibrillation. Circulation 2005, 112, 3697–3706. [Google Scholar] [CrossRef]
- Lemoine, M.D.; Mannhardt, I.; Breckwoldt, K.; Prondzynski, M.; Flenner, F.; Ulmer, B.; Hirt, M.N.; Neuber, C.; Horváth, A.; Kloth, B.; et al. Human iPSC-derived cardiomyocytes cultured in 3D engineered heart tissue show physiological upstroke velocity and sodium current density. Sci. Rep. 2017, 7, 5464. [Google Scholar] [CrossRef]
- Tabak, J.; Tomaiuolo, M.; Gonzalez-Iglesias, A.E.; Milescu, L.S.; Bertram, R. Fast-Activating Voltage- and Calcium-Dependent Potassium (BK) Conductance Promotes Bursting in Pituitary Cells: A Dynamic Clamp Study. J. Neurosci. 2011, 31, 16855–16863. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lin, M.T.; Hessinger, D.A.; Pearce, W.J.; Longo, L.D. Modulation of BK channel calcium affinity by differential phosphorylation in developing ovine basilar artery myocytes. Am. J. Physiol. Heart Circ. Physiol. 2006, 291, H732–H740. [Google Scholar] [CrossRef] [PubMed]
- Ding, J.P.; Li, Z.W.; Lingle, C.J. Inactivating BK Channels in Rat Chromaffin Cells May Arise from Heteromultimeric Assembly of Distinct Inactivation-Competent and Noninactivating Subunits. Biophys. J. 1998, 74, 268–289. [Google Scholar] [CrossRef]
- O’Hara, T.; Virág, L.; Varró, A.; Rudy, Y. Simulation of the undiseased human cardiac ventricular action potential: Model formulation and experimental validation. PLoS Comput. Biol. 2011, 7, e1002061. [Google Scholar] [CrossRef]
- Heubach, J.F.; Graf, E.M.; Leutheuser, J.; Bock, M.; Balana, B.; Zahanich, I.; Christ, T.; Boxberger, S.; Wettwer, E.; Ravens, U. Electrophysiological properties of human mesenchymal stem cells. J. Physiol. 2004, 554, 659–672. [Google Scholar] [CrossRef]
- Schulze, M.L.; Lemoine, M.D.; Fischer, A.W.; Scherschel, K.; David, R.; Hansen, A.; Eschenhagen, T.; Ulmer, B.M. Biomaterials Dissecting hiPSC-CM pacemaker function in a cardiac organoid model. Biomaterials 2019, 206, 133–145. [Google Scholar] [CrossRef]
- Lemme, M.; Ulmer, B.M.; Lemoine, M.D.; Zech, A.T.L.; Flenner, F.; Ravens, U.; Reichenspurner, H.; Rol-Garcia, M.; Smith, G.; Hansen, A.; et al. Atrial-like Engineered heart tissue: An In vitro Model of the Human Atrium, Stem Cell Reports. Stem Cell Rep. 2018, 11, 1–13. [Google Scholar] [CrossRef]
- Lemme, M.; Braren, I.; Prondzynski, M.; Ulmer, M.; Schulze, M.L.; Ismaili, D.; Meyer, C.; Hansen, A.; Christ, T.; Lemoine, M.D.; et al. Chronic intermittent tachypacing by an optogenetic approach induces arrhythmia vulnerability in human engineered heart tissue. Cardiovasc. Res. 2019. [Google Scholar] [CrossRef]
- Prondzynski, M.; Lemoine, M.D.; Zech, A.T.L.; Horvath, A.; Di Mauro, V.; Koivumaki, J.T.; Kresin, N.; Busch, J.; Krause, T.; Kramer, E.; et al. Disease modeling of a mutation in alpha-actinin 2 guides clinical therapy in hypertrophic cardiomyopathy. EMBO Mol. Med. 2019, 11, e11115. [Google Scholar] [CrossRef]
- Jan, L.Y.; Jan, Y.N. Cloned potassium channels from eukaryotes and prokaryotes. Annu. Rev. Neurosci. 1997, 20, 91–123. [Google Scholar] [CrossRef][Green Version]
- Cui, J.; Yang, H.; Lee, U.S. Molecular mechanisms of BK channel activation. Cell. Mol. Life Sci. CMLS 2009, 66, 852–875. [Google Scholar] [PubMed]
- Singh, H.; Stefani, E.; Toro, L. Intracellular BK Ca (iBK Ca ) channels. J. Physiol. 2012, 590, 5937–5947. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Jiang, M.T.; Su, J.; Hutchins, W.; Konorev, E.; Baker, J.E. Mitochondrial big conductance KCa channel and cardioprotection in infant rabbit heart. J. Cardiovasc. Pharmacol. 2007, 50, 497–502. [Google Scholar] [CrossRef] [PubMed]
- Tseng-crank, J.; Godinot, N.; Johansent, T.E.; Ahringt, P.K.; Strobaek, D.; Mertz, R.; Foster, C.D.; Olesen, S.-P.; Reinhart, P.H. Cloning, expression, and distribution of a Ca2+ -activated K+ channel ß-subunit from human brain. PNAS 1996, 93, 9200–9205. [Google Scholar] [CrossRef] [PubMed]
- Ko, J.H.; Ibrahim, M.A.; Park, W.S.; Ko, E.A.; Kim, N.; Warda, M.; Lim, I.; Bang, H.; Han, J. Cloning of large-conductance Ca2+-activated K+ channel α-subunits in mouse cardiomyocytes. Biochem. Biophys. Res. Commun. 2009, 389, 74–79. [Google Scholar] [CrossRef] [PubMed]
- Tseng-crank, J.; Foster, C.D.; Krause, J.D.; Mertz, R.; Godinot, N.; Dichiara, T.J.; Reinhart, P.H.; Drive, M.; Carolina, N. Cloning, Expression, and Distribution of Functionally Distinct Ca2+ -Activated K-Channel Isoforms from Human Brain. Neuron 1994, 13, 1315–1330. [Google Scholar] [CrossRef]
- Liu, S.; Gao, X.; Wu, X.; Yu, Y.; Yu, Z.; Zhao, S.; Zhao, H. BK channels regulate calcium oscillations in ventricular myocytes on different substrate stiffness. Life Sci. 2019, 235, 116802. [Google Scholar]
- Takamatsu, H.; Nagao, T.; Ichijo, H.; Adachi-Akahane, S. L-type Ca2+ channels serve as a sensor of the SR Ca2+ for tuning the efficacy of Ca2+-induced Ca2+ release in rat ventricular myocytes. J. Physiol. 2003, 552, 415–424. [Google Scholar] [CrossRef]
- Son, Y.K.; Hong, D.H.; Choi, T.-H.; Choi, S.W.; Shin, D.H.; Kim, S.J.; Jung, I.D.; Park, Y.-M.; Jung, W.-K.; Kim, D.-J.; et al. The inhibitory effect of BIM (I) on L-type Ca2+ channels in rat ventricular cells. Biochem. Biophys. Res. Commun. 2012, 423, 110–115. [Google Scholar] [CrossRef]
- Candia, S.; Garcia, M.L.; Latorre, R. Mode of action of iberiotoxin, a potent blocker of the large conductance Ca(2+)-activated K+ channel. Biophys. J. 1992, 63, 583–590. [Google Scholar] [CrossRef]
- Lai, M.H.; Wu, Y.; Gao, Z.; Anderson, M.E.; Dalziel, J.E.; Meredith, A.L. BK channels regulate sinoatrial node firing rate and cardiac pacing in vivo. AJP: Heart Circ. Physiol. 2014, 307, H1327–H1338. [Google Scholar] [CrossRef] [PubMed]
- Kilpinen, H.; Goncalves, A.; Leha, A.; Afzal, V.; Alasoo, K.; Ashford, S.; Bala, S.; Bensaddek, D.; Casale, F.P.; Culley, O.J.; et al. Common genetic variation drives molecular heterogeneity in human iPSCs. Nature 2017, 546, 370–375. [Google Scholar] [CrossRef] [PubMed]
- Merkle, F.T.; Ghosh, S.; Kamitaki, N.; Mitchell, J.; Avior, Y.; Mello, C.; Kashin, S.; Mekhoubad, S.; Ilic, D.; Charlton, M.; et al. Human pluripotent stem cells recurrently acquire and expand dominant negative P53 mutations. Nature 2017, 545, 229–233. [Google Scholar] [CrossRef] [PubMed]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Horváth, A.; Christ, T.; Koivumäki, J.T.; Prondzynski, M.; Zech, A.T.L.; Spohn, M.; Saleem, U.; Mannhardt, I.; Ulmer, B.; Girdauskas, E.; et al. Case Report on: Very Early Afterdepolarizations in HiPSC-Cardiomyocytes—An Artifact by Big Conductance Calcium Activated Potassium Current (Ibk,Ca). Cells 2020, 9, 253. https://doi.org/10.3390/cells9010253
Horváth A, Christ T, Koivumäki JT, Prondzynski M, Zech ATL, Spohn M, Saleem U, Mannhardt I, Ulmer B, Girdauskas E, et al. Case Report on: Very Early Afterdepolarizations in HiPSC-Cardiomyocytes—An Artifact by Big Conductance Calcium Activated Potassium Current (Ibk,Ca). Cells. 2020; 9(1):253. https://doi.org/10.3390/cells9010253
Chicago/Turabian StyleHorváth, András, Torsten Christ, Jussi T. Koivumäki, Maksymilian Prondzynski, Antonia T. L. Zech, Michael Spohn, Umber Saleem, Ingra Mannhardt, Bärbel Ulmer, Evaldas Girdauskas, and et al. 2020. "Case Report on: Very Early Afterdepolarizations in HiPSC-Cardiomyocytes—An Artifact by Big Conductance Calcium Activated Potassium Current (Ibk,Ca)" Cells 9, no. 1: 253. https://doi.org/10.3390/cells9010253
APA StyleHorváth, A., Christ, T., Koivumäki, J. T., Prondzynski, M., Zech, A. T. L., Spohn, M., Saleem, U., Mannhardt, I., Ulmer, B., Girdauskas, E., Meyer, C., Hansen, A., Eschenhagen, T., & Lemoine, M. D. (2020). Case Report on: Very Early Afterdepolarizations in HiPSC-Cardiomyocytes—An Artifact by Big Conductance Calcium Activated Potassium Current (Ibk,Ca). Cells, 9(1), 253. https://doi.org/10.3390/cells9010253




