Deficiency in Androgen Receptor Aggravates the Depressive-Like Behaviors in Chronic Mild Stress Model of Depression
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Behavior Experiment
2.2.1. Chronic Mild Stress (CMS) Protocol
2.2.2. Sucrose Preference Test
2.2.3. Forced Swimming Test
2.2.4. Fluoxetine and 7,8-Dihydroxyflavone (7,8-DHF) Treatments for ARKO Mice
2.3. Immunohistochemistry (IHC)
2.4. In Situ Hybridization (ISH) for BDNF
2.5. Laser Capture Microdissection, and RNA Isolation
2.6. Cell Culture
2.7. Transient Transfection with siRNA
2.8. Quantitative Real-Time PCR (qRT-PCR)
2.9. Statistical and Data Analysis
3. Results
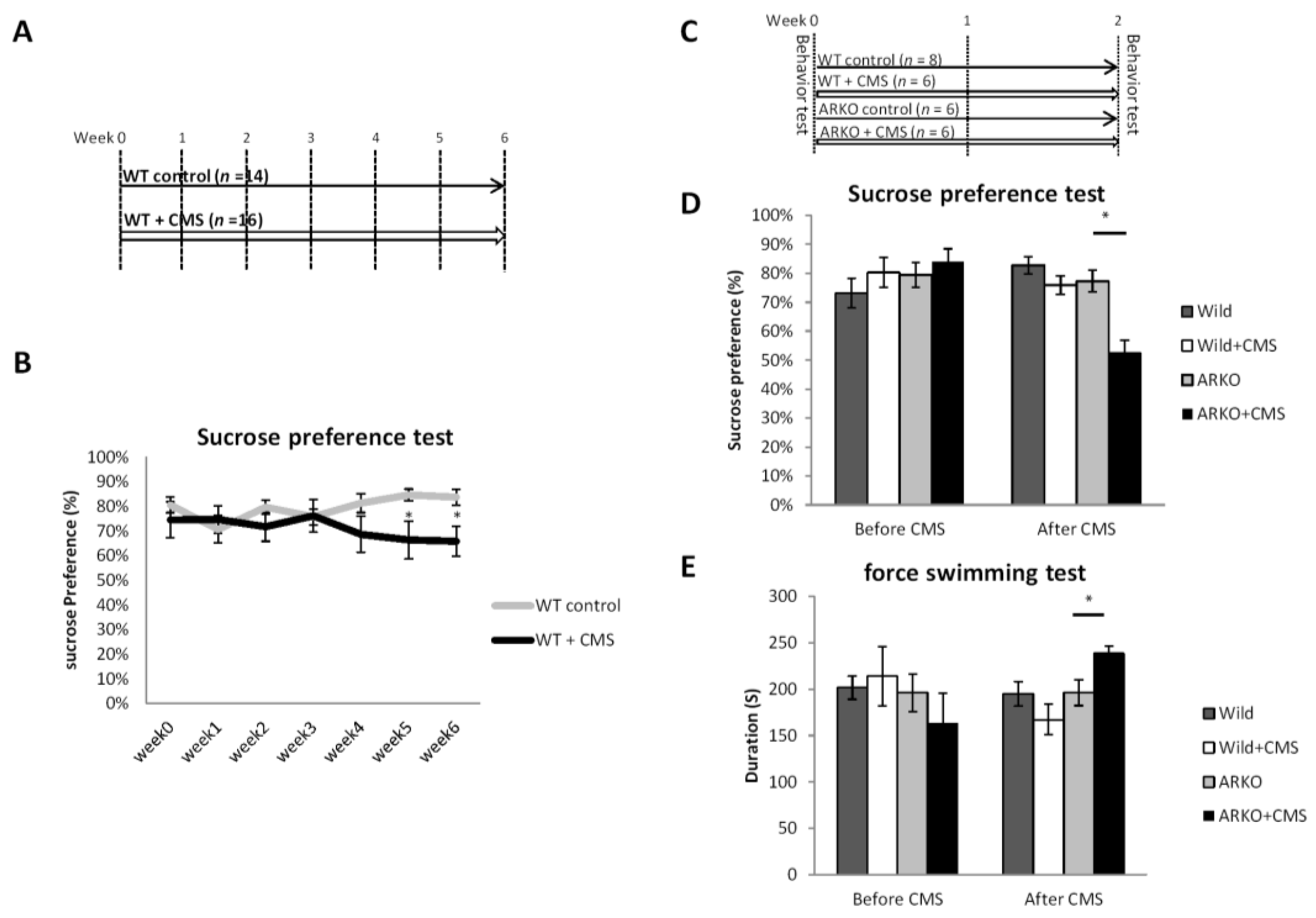
3.1. Loss of AR Accelerated the CMS-Mediated Depressive-Like Behavior in Mice
3.2. Mechanism Dissection of Why Loss of AR Accelerated the CMS-Mediated Depressive-Like Behavior: Via Decreasing the BDNF Expression in the CA1 Region of Mouse Hippocampus in Response to CMS
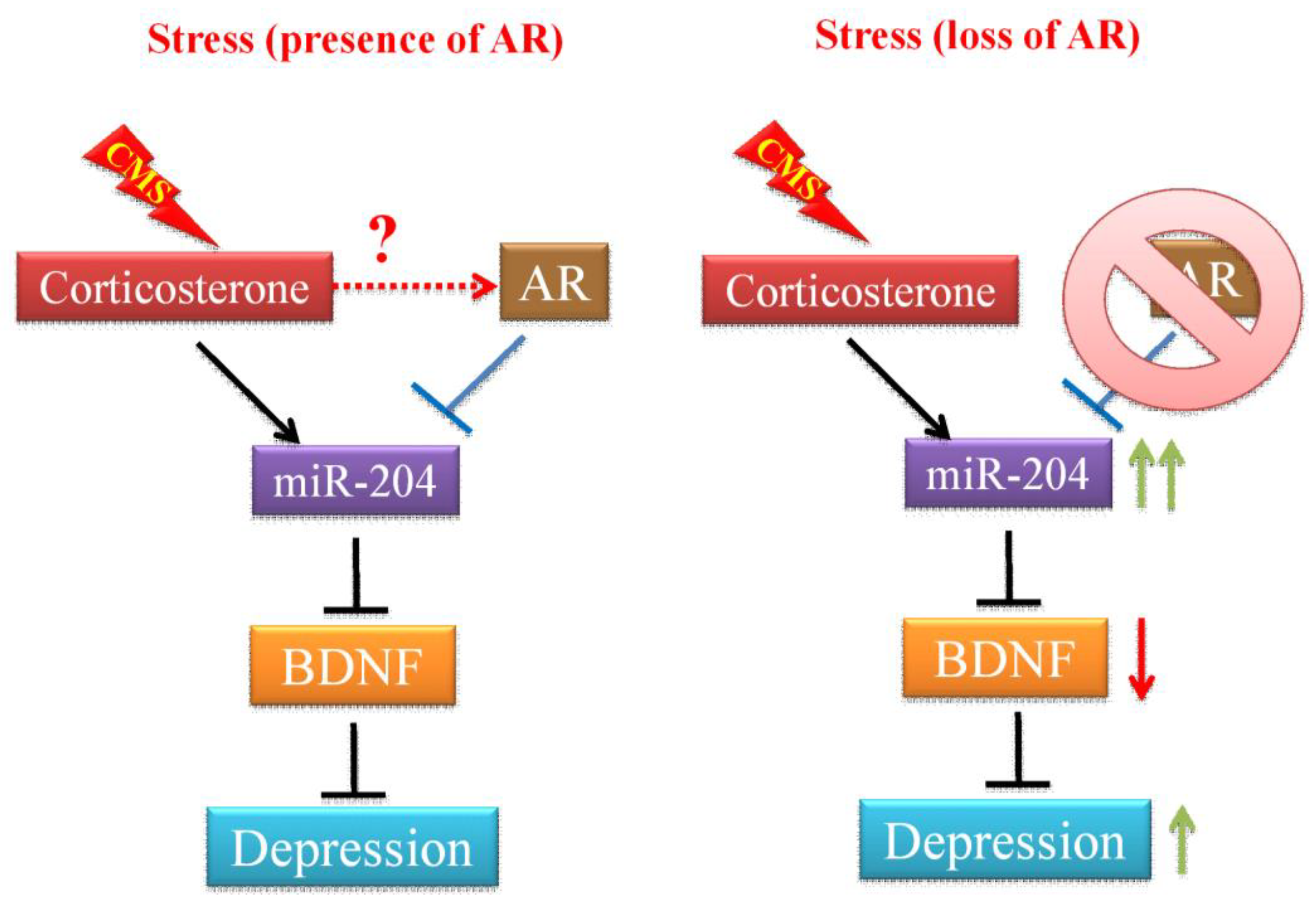
3.3. Mechanism Dissection of How Androgens/AR Increases the BDNF Expression: Via Altering the miRNA-204-5p Expression
3.4. Targeting the AR/BDNF/TrkB-Mediated pAKT/p-p38 Signals with 7,8-DHF and Fluoxetine Attenuates the Depressive-Like Behavior in ARKO Mice with CMS Exposure
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Alonso, J.; Angermeyer, M.C.; Bernert, S.; Bruffaerts, R.; Brugha, T.S.; Bryson, H.; de Girolamo, G.; Graaf, R.; Demyttenaere, K.; Gasquet, I.; et al. Disability and quality of life impact of mental disorders in Europe: Results from the European Study of the Epidemiology of Mental Disorders (ESEMeD) project. Acta Psychiatr. Scand. Suppl. 2004, 109, 38–46. [Google Scholar]
- Gu, L.; Xie, J.; Long, J.; Chen, Q.; Chen, Q.; Pan, R.; Yan, Y.; Wu, G.; Liang, B.; Tan, J.; et al. Epidemiology of major depressive disorder in mainland china: A systematic review. PLoS ONE 2013, 8, e65356. [Google Scholar] [CrossRef] [PubMed]
- Djukanovic, I.; Sorjonen, K.; Peterson, U. Association between depressive symptoms and age, sex, loneliness and treatment among older people in Sweden. Aging Ment. Health 2014, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Burris, A.S.; Banks, S.M.; Carter, C.S.; Davidson, J.M.; Sherins, R.J. A long-term, prospective study of the physiologic and behavioral effects of hormone replacement in untreated hypogonadal men. J. Androl. 1992, 13, 297–304. [Google Scholar]
- Wang, C.; Alexander, G.; Berman, N.; Salehian, B.; Davidson, T.; McDonald, V.; Steiner, B.; Hull, L.; Callegari, C.; Swerdloff, R.S. Testosterone replacement therapy improves mood in hypogonadal men—A clinical research center study. J. Clin. Endocrinol. Metab. 1996, 81, 3578–3583. [Google Scholar] [PubMed]
- Snyder, P.J.; Bhasin, S.; Cunningham, G.R.; Matsumoto, A.M.; Stephens-Shields, A.J.; Cauley, J.A.; Gill, T.M.; Barrett-Connor, E.; Swerdloff, R.S.; Wang, C.; et al. Effects of Testosterone Treatment in Older Men. N. Engl. J. Med. 2016, 374, 611–624. [Google Scholar] [CrossRef] [PubMed]
- Chamberlain, N.L.; Driver, E.D.; Miesfeld, R.L. The length and location of CAG trinucleotide repeats in the androgen receptor N-terminal domain affect transactivation function. Nucleic Acids Res. 1994, 22, 3181–3186. [Google Scholar] [CrossRef] [PubMed]
- Beilin, J.; Ball, E.M.; Favaloro, J.M.; Zajac, J.D. Effect of the androgen receptor CAG repeat polymorphism on transcriptional activity: Specificity in prostate and non-prostate cell lines. J. Mol. Endocrinol. 2000, 25, 85–96. [Google Scholar] [CrossRef]
- Sankar, J.S.; Hampson, E. Testosterone levels and androgen receptor gene polymorphism predict specific symptoms of depression in young men. Gend. Med. 2012, 9, 232–243. [Google Scholar] [CrossRef]
- Cherrier, M.M.; Aubin, S.; Higano, C.S. Cognitive and mood changes in men undergoing intermittent combined androgen blockade for non-metastatic prostate cancer. Psychooncology 2009, 18, 237–247. [Google Scholar] [CrossRef]
- Lee, M.; Jim, H.S.; Fishman, M.; Zachariah, B.; Heysek, R.; Biagioli, M.; Jacobsen, P.B. Depressive symptomatology in men receiving androgen deprivation therapy for prostate cancer: A controlled comparison. Psychooncology 2015, 24, 472–477. [Google Scholar] [CrossRef] [PubMed]
- Fliegner, M.; Krupp, K.; Brunner, F.; Rall, K.; Brucker, S.Y.; Briken, P.; Richter-Appelt, H. Sexual life and sexual wellness in individuals with complete androgen insensitivity syndrome (CAIS) and Mayer-Rokitansky-Kuster-Hauser Syndrome (MRKHS). J. Sex. Med. 2014, 11, 729–742. [Google Scholar] [CrossRef] [PubMed]
- Mueller, S.C.; Grissom, E.M.; Dohanich, G.P. Assessing gonadal hormone contributions to affective psychopathologies across humans and animal models. Psychoneuroendocrinology 2014, 46, 114–128. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.S.; Kamphuis, W.; Huitinga, I.; Zhou, J.N.; Swaab, D.F. Gene expression analysis in the human hypothalamus in depression by laser microdissection and real-time PCR: The presence of multiple receptor imbalances. Mol. Psychiatry 2008, 13, 741, 786–799. [Google Scholar] [CrossRef] [PubMed]
- Selakovic, D.; Joksimovic, J.; Jovicic, N.; Mitrovic, S.; Mihailovic, V.; Katanic, J.; Milovanovic, D.; Pantovic, S.; Mijailovic, N.; Rosic, G. The Impact of Hippocampal Sex Hormones Receptors in Modulation of Depressive-Like Behavior Following Chronic Anabolic Androgenic Steroids and Exercise Protocols in Rats. Front. Behav. Neurosci. 2019, 13, 19. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.L.; Lee, C.T.; Liu, Y.L. Serum brain-derived neurotrophic factor levels in patients with major depression: Effects of antidepressants. J. Psychiatr. Res. 2008, 42, 521–525. [Google Scholar] [CrossRef] [PubMed]
- Bus, B.A.; Molendijk, M.L.; Tendolkar, I.; Penninx, B.W.; Prickaerts, J.; Elzinga, B.M.; Voshaar, R.C. Chronic depression is associated with a pronounced decrease in serum brain-derived neurotrophic factor over time. Mol. Psychiatry 2015, 20, 602–608. [Google Scholar] [CrossRef]
- Castren, E.; Rantamaki, T. The role of BDNF and its receptors in depression and antidepressant drug action: Reactivation of developmental plasticity. Dev. Neurobiol. 2010, 70, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Autry, A.E.; Monteggia, L.M. Brain-derived neurotrophic factor and neuropsychiatric disorders. Pharm. Rev. 2012, 64, 238–258. [Google Scholar] [CrossRef] [PubMed]
- Castren, E.; Voikar, V.; Rantamaki, T. Role of neurotrophic factors in depression. Curr. Opin. Pharm. 2007, 7, 18–21. [Google Scholar] [CrossRef] [PubMed]
- Licinio, J.; Wong, M.L. Brain-derived neurotrophic factor in depression: A male problem? Mol. Psychiatry 2010, 15, 227. [Google Scholar] [CrossRef] [PubMed]
- Verhagen, M.; van der Meij, A.; van Deurzen, P.A.; Janzing, J.G.; Arias-Vasquez, A.; Buitelaar, J.K.; Franke, B. Meta-analysis of the BDNF Val66Met polymorphism in major depressive disorder: Effects of gender and ethnicity. Mol. Psychiatry 2010, 15, 260–271. [Google Scholar] [CrossRef] [PubMed]
- Monteggia, L.M.; Luikart, B.; Barrot, M.; Theobold, D.; Malkovska, I.; Nef, S.; Parada, L.F.; Nestler, E.J. Brain-derived neurotrophic factor conditional knockouts show gender differences in depression-related behaviors. Biol. Psychiatry 2007, 61, 187–197. [Google Scholar] [CrossRef] [PubMed]
- Hamson, D.K.; Jones, B.A.; Csupity, A.S.; Ali, F.M.; Watson, N.V. Androgen insensitive male rats display increased anxiety-like behavior on the elevated plus maze. Behav. Brain Res. 2014, 259, 158–163. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Masugi-Tokita, M.; Takanami, K.; Yamada, S.; Kawata, M. Testosterone has sublayer-specific effects on dendritic spine maturation mediated by BDNF and PSD-95 in pyramidal neurons in the hippocampus CA1 area. Brain Res. 2012, 1484, 76–84. [Google Scholar] [CrossRef]
- Shi, J. Regulatory networks between neurotrophins and miRNAs in brain diseases and cancers. Acta Pharm. Sin. 2015, 36, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Imam, J.S.; Plyler, J.R.; Bansal, H.; Prajapati, S.; Bansal, S.; Rebeles, J.; Chen, H.I.; Chang, Y.F.; Panneerdoss, S.; Zoghi, B.; et al. Genomic loss of tumor suppressor miRNA-204 promotes cancer cell migration and invasion by activating AKT/mTOR/Rac1 signaling and actin reorganization. PLoS ONE 2012, 7, e52397. [Google Scholar] [CrossRef] [PubMed]
- Dwivedi, Y. Emerging role of microRNAs in major depressive disorder: Diagnosis and therapeutic implications. Dialogues Clin. Neurosci. 2014, 16, 43–61. [Google Scholar] [PubMed]
- Yeh, S.; Tsai, M.Y.; Xu, Q.; Mu, X.M.; Lardy, H.; Huang, K.E.; Lin, H.; Yeh, S.D.; Altuwaijri, S.; Zhou, X.; et al. Generation and characterization of androgen receptor knockout (ARKO) mice: An in vivo model for the study of androgen functions in selective tissues. Proc. Natl. Acad. Sci. USA 2002, 99, 13498–13503. [Google Scholar] [CrossRef] [PubMed]
- Cheng, B.H.; Chu, T.M.; Chang, C.; Kang, H.Y.; Huang, K.E. Testosterone delivered with a scaffold is as effective as bone morphologic protein-2 in promoting the repair of critical-size segmental defect of femoral bone in mice. PLoS ONE 2013, 8, e70234. [Google Scholar] [CrossRef] [PubMed]
- Schweizer, M.C.; Henniger, M.S.; Sillaber, I. Chronic mild stress (CMS) in mice: Of anhedonia, ‘anomalous anxiolysis’ and activity. PLoS ONE 2009, 4, e4326. [Google Scholar] [CrossRef] [PubMed]
- Herrera-Perez, J.J.; Martinez-Mota, L.; Fernandez-Guasti, A. Aging increases the susceptibility to develop anhedonia in male rats. Prog. Neuropsychopharmacol. Biol. Psychiatry 2008, 32, 1798–1803. [Google Scholar] [CrossRef] [PubMed]
- Couillard-Despres, S.; Wuertinger, C.; Kandasamy, M.; Caioni, M.; Stadler, K.; Aigner, R.; Bogdahn, U.; Aigner, L. Ageing abolishes the effects of fluoxetine on neurogenesis. Mol. Psychiatry 2009, 14, 856–864. [Google Scholar] [CrossRef] [PubMed]
- Fedchenko, N.; Reifenrath, J. Different approaches for interpretation and reporting of immunohistochemistry analysis results in the bone tissue—A review. Diagn. Pathol. 2014, 9, 221. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.Y.; Yin, C.Y.; Zhu, L.J.; Zhu, X.H.; Xu, C.; Luo, C.X.; Chen, H.; Zhu, D.Y.; Zhou, Q.G. Sucrose preference test for measurement of stress-induced anhedonia in mice. Nat. Protoc. 2018, 13, 1686–1698. [Google Scholar] [CrossRef] [PubMed]
- Kerr, J.E.; Allore, R.J.; Beck, S.G.; Handa, R.J. Distribution and hormonal regulation of androgen receptor (AR) and AR messenger ribonucleic acid in the rat hippocampus. Endocrinology 1995, 136, 3213–3221. [Google Scholar] [CrossRef] [PubMed]
- Taliaz, D.; Loya, A.; Gersner, R.; Haramati, S.; Chen, A.; Zangen, A. Resilience to chronic stress is mediated by hippocampal brain-derived neurotrophic factor. J. Neurosci. 2011, 31, 4475–4483. [Google Scholar] [CrossRef]
- Verhovshek, T.; Sengelaub, D.R. Androgen action at the target musculature regulates brain-derived neurotrophic factor protein in the spinal nucleus of the bulbocavernosus. Dev. Neurobiol. 2013, 73, 587–598. [Google Scholar] [CrossRef]
- Halievski, K.; Henley, C.L.; Domino, L.; Poort, J.E.; Fu, M.; Katsuno, M.; Adachi, H.; Sobue, G.; Breedlove, S.M.; Jordan, C.L. Androgen-dependent loss of muscle BDNF mRNA in two mouse models of SBMA. Exp. Neurol. 2015, 269, 224–232. [Google Scholar] [CrossRef]
- Rodgers, A.B.; Morgan, C.P.; Bronson, S.L.; Revello, S.; Bale, T.L. Paternal stress exposure alters sperm microRNA content and reprograms offspring HPA stress axis regulation. J. Neurosci. 2013, 33, 9003–9012. [Google Scholar] [CrossRef]
- Ding, M.; Lin, B.; Li, T.; Liu, Y.; Li, Y.; Zhou, X.; Miao, M.; Gu, J.; Pan, H.; Yang, F.; et al. A dual yet opposite growth-regulating function of miR-204 and its target XRN1 in prostate adenocarcinoma cells and neuroendocrine-like prostate cancer cells. Oncotarget 2015, 6, 7686–7700. [Google Scholar] [CrossRef] [PubMed]
- Duman, R.S.; Voleti, B. Signaling pathways underlying the pathophysiology and treatment of depression: Novel mechanisms for rapid-acting agents. Trends Neurosci. 2012, 35, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Nibuya, M.; Nestler, E.J.; Duman, R.S. Chronic antidepressant administration increases the expression of cAMP response element binding protein (CREB) in rat hippocampus. J. Neurosci. 1996, 16, 2365–2372. [Google Scholar] [CrossRef] [PubMed]
- Miller, K.K.; Wexler, T.L.; Zha, A.M.; Lawson, E.A.; Meenaghan, E.M.; Misra, M.; Binstock, A.B.; Herzog, D.B.; Klibanski, A. Androgen deficiency: Association with increased anxiety and depression symptom severity in anorexia nervosa. J. Clin. Psychiatry 2007, 68, 959–965. [Google Scholar] [CrossRef] [PubMed]
- Schneider, G.; Nienhaus, K.; Gromoll, J.; Heuft, G.; Nieschlag, E.; Zitzmann, M. Depressive symptoms in men aged 50 years and older and their relationship to genetic androgen receptor polymorphism and sex hormone levels in three different samples. Am. J. Geriatr. Psychiatry 2011, 19, 274–283. [Google Scholar] [CrossRef] [PubMed]
- Schneider, G.; Nienhaus, K.; Gromoll, J.; Heuft, G.; Nieschlag, E.; Zitzmann, M. Sex hormone levels, genetic androgen receptor polymorphism, and anxiety in >/=50-year-old males. J. Sex. Med. 2011, 8, 3452–3464. [Google Scholar] [CrossRef] [PubMed]
- Herrera-Perez, J.J.; Martinez-Mota, L.; Chavira, R.; Fernandez-Guasti, A. Testosterone prevents but not reverses anhedonia in middle-aged males and lacks an effect on stress vulnerability in young adults. Horm. Behav. 2012, 61, 623–630. [Google Scholar] [CrossRef]
- Aydogan, U.; Aydogdu, A.; Akbulut, H.; Sonmez, A.; Yuksel, S.; Basaran, Y.; Uzun, O.; Bolu, E.; Saglam, K. Increased frequency of anxiety, depression, quality of life and sexual life in young hypogonadotropic hypogonadal males and impacts of testosterone replacement therapy on these conditions. Endocr. J. 2012, 59, 1099–1105. [Google Scholar] [CrossRef]
- Zuloaga, D.G.; Poort, J.E.; Jordan, C.L.; Breedlove, S.M. Male rats with the testicular feminization mutation of the androgen receptor display elevated anxiety-related behavior and corticosterone response to mild stress. Horm. Behav. 2011, 60, 380–388. [Google Scholar] [CrossRef]
- Hung, C.I.; Liu, C.Y.; Chen, C.Y.; Yang, C.H.; Wang, S.J. The impacts of migraine and anxiety disorders on painful physical symptoms among patients with major depressive disorder. J. Headache Pain 2014, 15, 73. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, Z.; Dai, J.; Chen, L.; Huang, Y.; Zhan, Z. Beneficial effects of benzodiazepine diazepam on chronic stress-induced impairment of hippocampal structural plasticity and depression-like behavior in mice. Behav. Brain Res. 2012, 228, 339–350. [Google Scholar] [CrossRef] [PubMed]
- Haridas, S.; Kumar, M.; Manda, K. Melatonin ameliorates chronic mild stress induced behavioral dysfunctions in mice. Physiol. Behav. 2013, 119, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Small, S.A.; Schobel, S.A.; Buxton, R.B.; Witter, M.P.; Barnes, C.A. A pathophysiological framework of hippocampal dysfunction in ageing and disease. Nat. Rev. Neurosci. 2011, 12, 585–601. [Google Scholar] [CrossRef] [PubMed]
- Joels, M.; Karst, H.; Alfarez, D.; Heine, V.M.; Qin, Y.; van Riel, E.; Verkuyl, M.; Lucassen, P.J.; Krugers, H.J. Effects of chronic stress on structure and cell function in rat hippocampus and hypothalamus. Stress 2004, 7, 221–231. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Liu, L.; Liu, Y.Y.; Luo, J.; Lin, J.Y.; Li, X.; Wang, B.; Min, S. Effects of electroconvulsive stimulation on long-term potentiation and synaptophysin in the hippocampus of rats with depressive behavior. J. Ect. 2012, 28, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Ishii, H.; Tsurugizawa, T.; Ogiue-Ikeda, M.; Asashima, M.; Mukai, H.; Murakami, G.; Hojo, Y.; Kimoto, T.; Kawato, S. Local production of sex hormones and their modulation of hippocampal synaptic plasticity. Neuroscientist 2007, 13, 323–334. [Google Scholar] [CrossRef] [PubMed]
- Ooishi, Y.; Kawato, S.; Hojo, Y.; Hatanaka, Y.; Higo, S.; Murakami, G.; Komatsuzaki, Y.; Ogiue-Ikeda, M.; Kimoto, T.; Mukai, H. Modulation of synaptic plasticity in the hippocampus by hippocampus-derived estrogen and androgen. J. Steroid Biochem. Mol. Biol. 2012, 131, 37–51. [Google Scholar] [CrossRef] [PubMed]
- Filova, B.; Ostatnikova, D.; Celec, P.; Hodosy, J. The effect of testosterone on the formation of brain structures. Cells Tissues Organs 2013, 197, 169–177. [Google Scholar] [CrossRef]
- Zhang, Y.; Gu, F.; Chen, J.; Dong, W. Chronic antidepressant administration alleviates frontal and hippocampal BDNF deficits in CUMS rat. Brain Res. 2010, 1366, 141–148. [Google Scholar] [CrossRef]
- Stajic, D.; Selakovic, D.; Jovicic, N.; Joksimovic, J.; Arsenijevic, N.; Lukic, M.L.; Rosic, G. The role of galectin-3 in modulation of anxiety state level in mice. Brainbehaviorand Immun. 2019, 78, 177–187. [Google Scholar] [CrossRef]
- Oh, H.; Piantadosi, S.C.; Rocco, B.R.; Lewis, D.A.; Watkins, S.C.; Sibille, E. The Role of Dendritic Brain-Derived Neurotrophic Factor Transcripts on Altered Inhibitory Circuitry in Depression. Biol. Psychiatry 2019, 85, 517–526. [Google Scholar] [CrossRef] [PubMed]
- Van der Loos, C.M. Multiple immunoenzyme staining: Methods and visualizations for the observation with spectral imaging. J. Histochem. Cytochem. Off. J. Histochem. Soc. 2008, 56, 313–328. [Google Scholar] [CrossRef] [PubMed]
- Su, Q.R.; Su, L.Y.; Su, H.R.; Chen, Q.; Ren, G.Y.; Yin, Y.; Shen, S.Q.; Yu, A.Y.; Xia, G.Y. Polymorphisms of androgen receptor gene in childhood and adolescent males with first-onset major depressive disorder and association with related symptomatology. Int. J. Neurosci. 2007, 117, 903–917. [Google Scholar] [CrossRef] [PubMed]
- Lima Giacobbo, B.; Doorduin, J.; Klein, H.C.; Dierckx, R.; Bromberg, E.; de Vries, E.F.J. Brain-Derived Neurotrophic Factor in Brain Disorders: Focus on Neuroinflammation. Mol. Neurobiol. 2019, 56, 3295–3312. [Google Scholar] [CrossRef] [PubMed]
- Calmarza-Font, I.; Lagunas, N.; Garcia-Segura, L.M. Antidepressive and anxiolytic activity of selective estrogen receptor modulators in ovariectomized mice submitted to chronic unpredictable stress. Behav. Brain Res. 2012, 227, 287–290. [Google Scholar] [CrossRef] [PubMed]
- Tsien, J.Z.; Huerta, P.T.; Tonegawa, S. The essential role of hippocampal CA1 NMDA receptor-dependent synaptic plasticity in spatial memory. Cell 1996, 87, 1327–1338. [Google Scholar] [CrossRef]
- Zhang, J.M.; Konkle, A.T.; Zup, S.L.; McCarthy, M.M. Impact of sex and hormones on new cells in the developing rat hippocampus: A novel source of sex dimorphism? Eur. J. Neurosci. 2008, 27, 791–800. [Google Scholar] [CrossRef] [PubMed]
- MacLusky, N.J.; Hajszan, T.; Johansen, J.A.; Jordan, C.L.; Leranth, C. Androgen effects on hippocampal CA1 spine synapse numbers are retained in Tfm male rats with defective androgen receptors. Endocrinology 2006, 147, 2392–2398. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Altarejos, J.Y.; Montminy, M. CREB and the CRTC co-activators: Sensors for hormonal and metabolic signals. Nat. Rev. Mol. Cell Biol. 2011, 12, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Osborne, M.C.; Verhovshek, T.; Sengelaub, D.R. Androgen regulates trkB immunolabeling in spinal motoneurons. J. Neurosci. Res. 2007, 85, 303–309. [Google Scholar] [CrossRef]
- Radin, D.P.; Patel, P. BDNF: An Oncogene or Tumor Suppressor? Anticancer Res. 2017, 37, 3983–3990. [Google Scholar] [PubMed]
- Polakowski, N.; Terol, M.; Hoang, K.; Nash, I.; Laverdure, S.; Gazon, H.; Belrose, G.; Mesnard, J.M.; Cesaire, R.; Peloponese, J.M.; et al. HBZ stimulates brain-derived neurotrophic factor/TrkB autocrine/paracrine signaling to promote survival of human T-cell leukemia virus type 1-Infected T cells. J. Virol. 2014, 88, 13482–13494. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Zhang, B.; Sun, X.; Lu, S.; Liu, Z.; Liu, Y.; Li, H.; Wang, L.; Wang, X.; Zhao, C. MiR-204 inhibits human NSCLC metastasis through suppression of NUAK1. Br. J. Cancer 2014, 111, 2316–2327. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, C.P.; Rhee, H.; Phee, B.K.; Kim, K.; Kim, H.J.; Lee, H.; Park, J.H.; Jung, J.H.; Kim, J.Y.; Kim, H.C.; et al. miR-204 downregulates EphB2 in aging mouse hippocampal neurons. Aging Cell 2016, 15, 380–388. [Google Scholar] [CrossRef] [PubMed]
- Janowsky, J.S. The role of androgens in cognition and brain aging in men. Neuroscience 2006, 138, 1015–1020. [Google Scholar] [CrossRef] [PubMed]
- Tornese, P.; Sala, N.; Bonini, D.; Bonifacino, T.; La Via, L.; Milanese, M.; Treccani, G.; Seguini, M.; Ieraci, A.; Mingardi, J.; et al. Chronic mild stress induces anhedonic behavior and changes in glutamate release, BDNF trafficking and dendrite morphology only in stress vulnerable rats. The rapid restorative action of ketamine. Neurobiol. Stress 2019, 10, 100160. [Google Scholar] [CrossRef] [PubMed]
- Fuchikami, M.; Yamamoto, S.; Morinobu, S.; Takei, S.; Yamawaki, S. Epigenetic regulation of BDNF gene in response to stress. Psychiatry Investig. 2010, 7, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Barnum, C.J.; Pace, T.W.; Hu, F.; Neigh, G.N.; Tansey, M.G. Psychological stress in adolescent and adult mice increases neuroinflammation and attenuates the response to LPS challenge. J. Neuroinflammation 2012, 9, 9. [Google Scholar] [CrossRef] [PubMed]
- Strekalova, T.; Couch, Y.; Kholod, N.; Boyks, M.; Malin, D.; Leprince, P.; Steinbusch, H.M. Update in the methodology of the chronic stress paradigm: Internal control matters. Behav. Brain Funct. 2011, 7, 9. [Google Scholar] [CrossRef] [PubMed]
- Ren, Z.; Yan, P.; Zhu, L.; Yang, H.; Zhao, Y.; Kirby, B.P.; Waddington, J.L.; Zhen, X. Dihydromyricetin exerts a rapid antidepressant-like effect in association with enhancement of BDNF expression and inhibition of neuroinflammation. Psychopharmacology 2018, 235, 233–244. [Google Scholar] [CrossRef] [PubMed]



© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hung, Y.-Y.; Huang, Y.-L.; Chang, C.; Kang, H.-Y. Deficiency in Androgen Receptor Aggravates the Depressive-Like Behaviors in Chronic Mild Stress Model of Depression. Cells 2019, 8, 1021. https://doi.org/10.3390/cells8091021
Hung Y-Y, Huang Y-L, Chang C, Kang H-Y. Deficiency in Androgen Receptor Aggravates the Depressive-Like Behaviors in Chronic Mild Stress Model of Depression. Cells. 2019; 8(9):1021. https://doi.org/10.3390/cells8091021
Chicago/Turabian StyleHung, Yi-Yung, Ya-Ling Huang, Chawnshang Chang, and Hong-Yo Kang. 2019. "Deficiency in Androgen Receptor Aggravates the Depressive-Like Behaviors in Chronic Mild Stress Model of Depression" Cells 8, no. 9: 1021. https://doi.org/10.3390/cells8091021
APA StyleHung, Y.-Y., Huang, Y.-L., Chang, C., & Kang, H.-Y. (2019). Deficiency in Androgen Receptor Aggravates the Depressive-Like Behaviors in Chronic Mild Stress Model of Depression. Cells, 8(9), 1021. https://doi.org/10.3390/cells8091021





