Antioxidant and Pro-Oxidant Activities of Melatonin in the Presence of Copper and Polyphenols In Vitro and In Vivo
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Drugs
2.2. ROS Detection
2.3. Reduction of Cu(II) to Cu(I) by Melatonin
2.4. Determination of Hydrogen Peroxide
2.5. Hydroxyl Radical Detection
2.6. Detection of DNA Damage
2.7. Animals and Treatments
2.8. Enzymatic Activity Assays
2.9. Copper Measurement
2.10. Western Blotting
2.11. RNA Isolation, Reverse Transcriptase Polymerase Chain Reaction (RT-PCR) and qPCR
- Thbs1
- Sense: CCCCTACAACCACAACCCTGAC
- Antisense: ACTGATCTCCAACCCCATCCAT
- p21
- Sense: GACTTCTCCCATTTCTTAGTAGCAG
- Antisense: TGACACCCACGGTATTCAACAC
- Bax
- Sense: TGGAGATGAACTGGACAGCAATAT
- Antisense: GCAAAGTAGAAGAGGGCAACCAC
- β-actin
- Sense: GCTGAGAGGGAAATCGTGCGT
- Antisense: ACCGCTCGTTGCCAATAGTGA
2.12. Statistical Analysis
3. Results
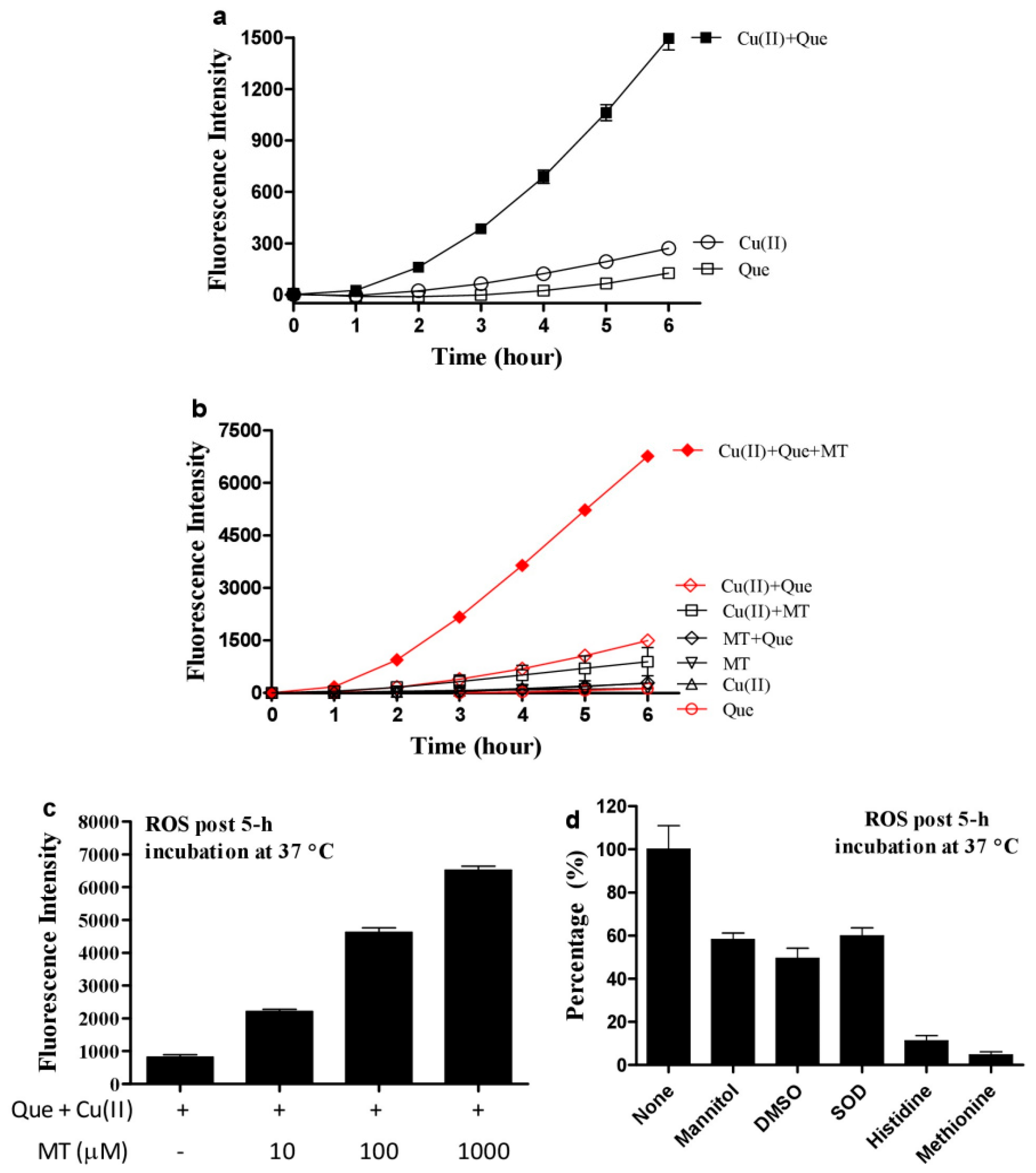
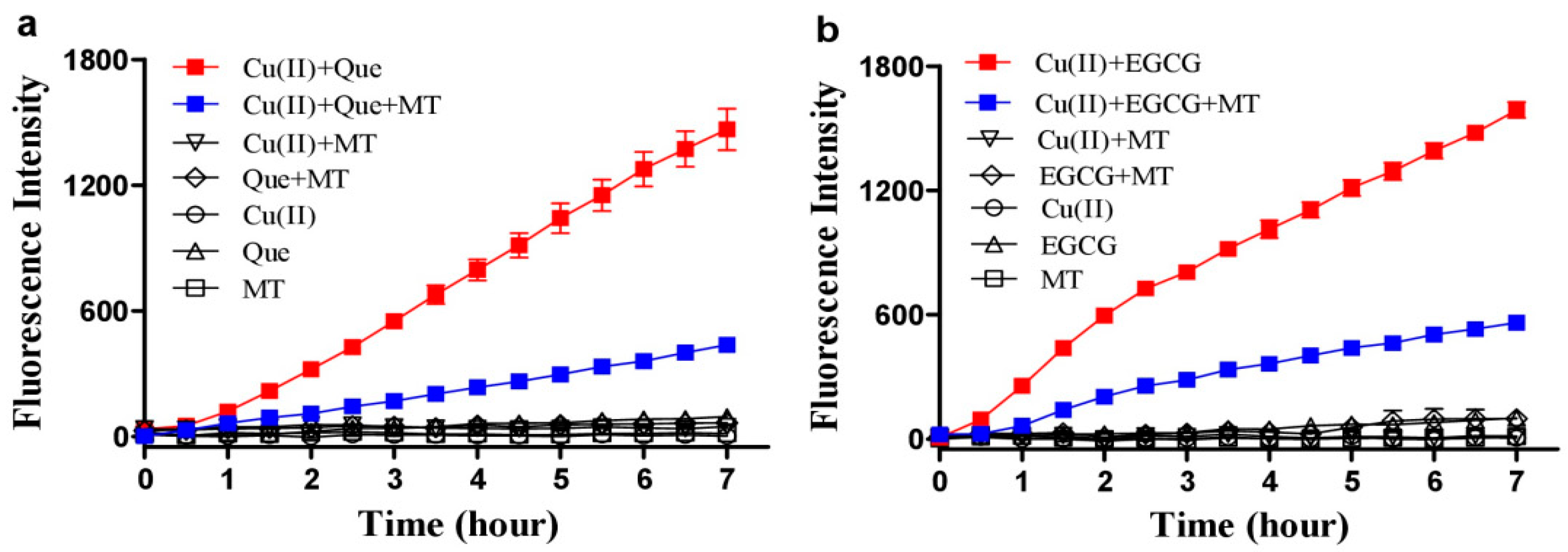
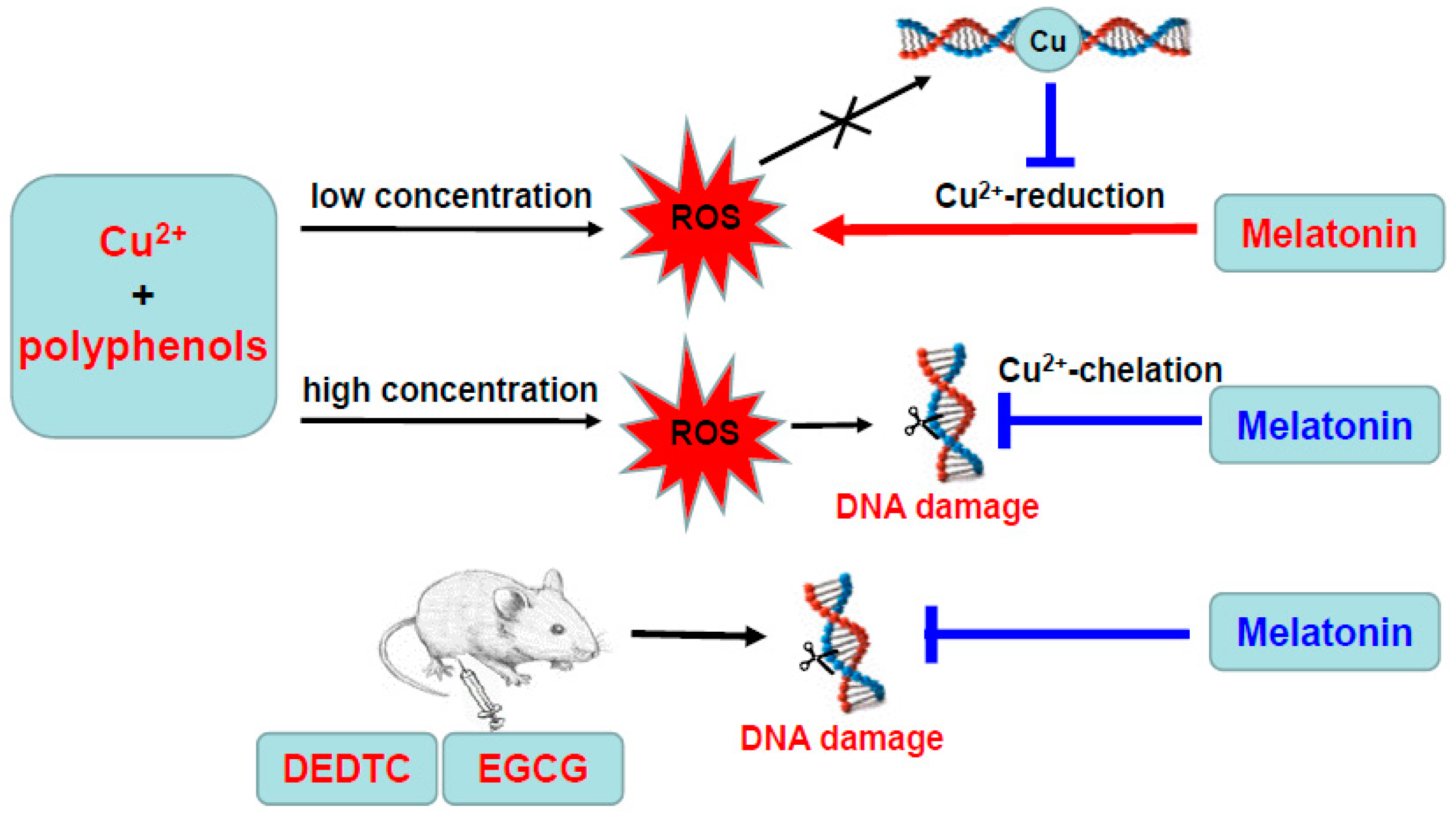
3.1. Melatonin, Copper and Quercetin Synergistically Produce ROS
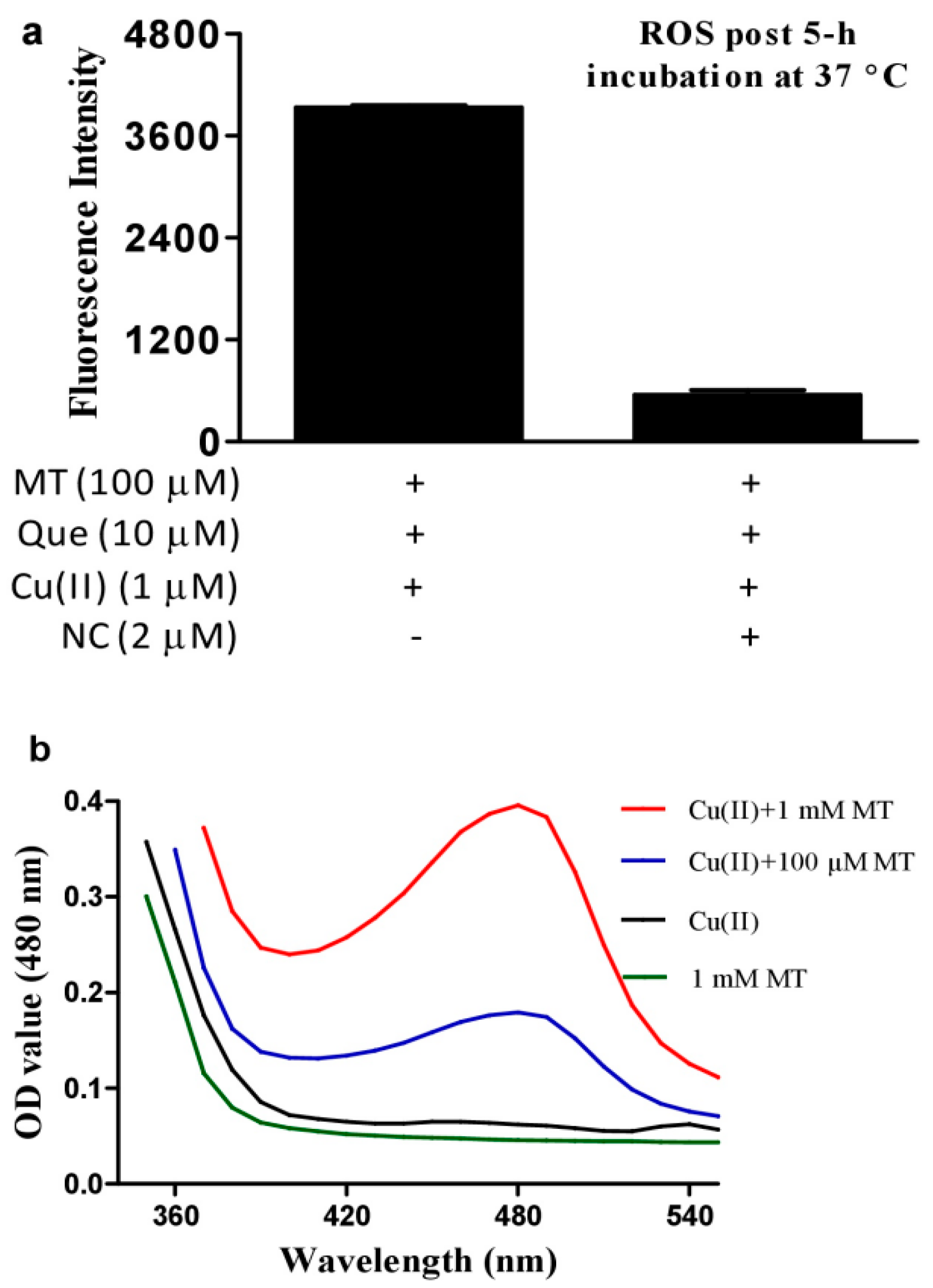
3.2. Hydrogen Peroxide Plays an Important Role for Cooperative ROS Production by Melatonin, Copper and Polyphenols
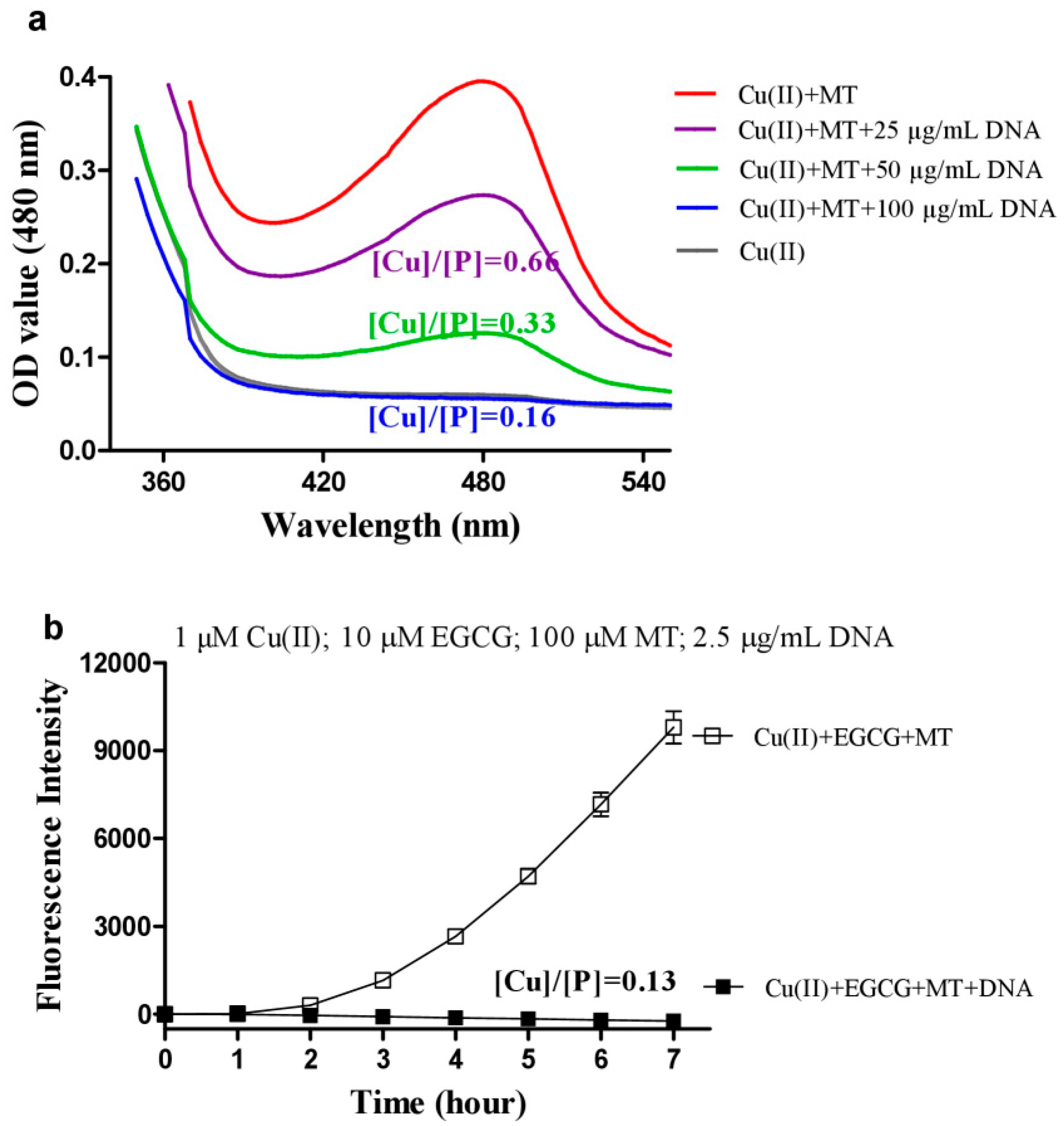
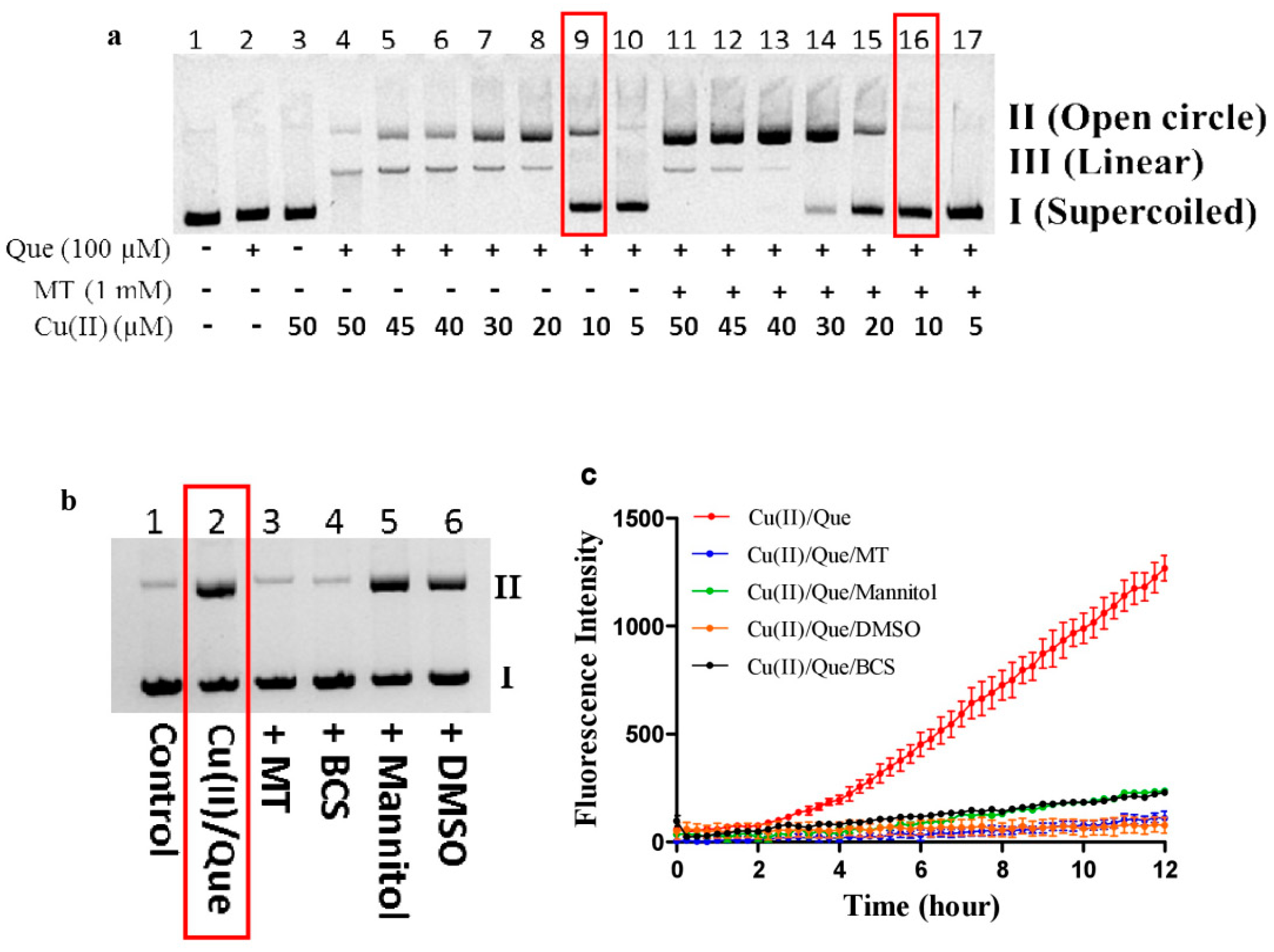
3.3. DNA Inhibits Melatonin’s Action to Reduce Cu(II) into Cu(I)
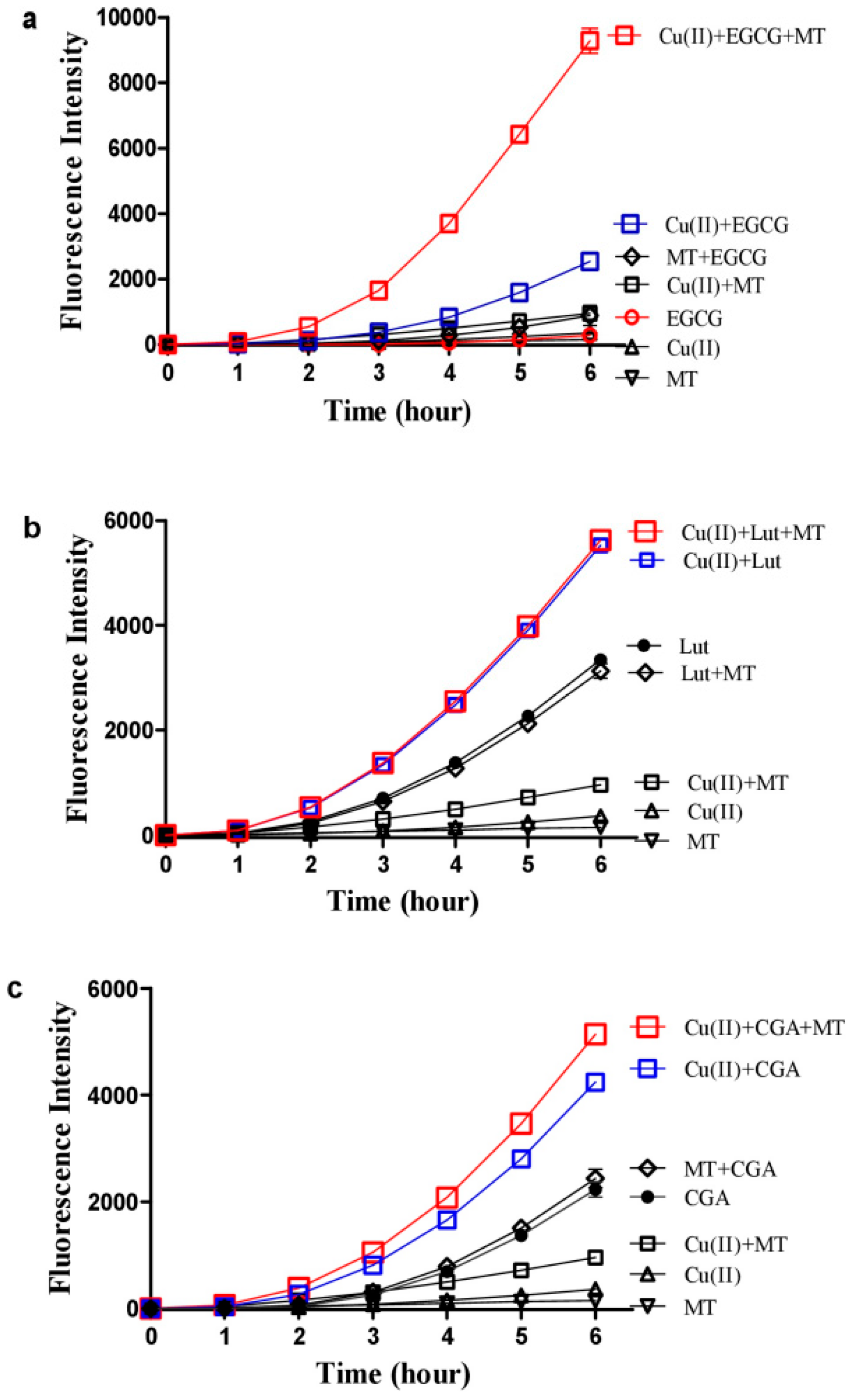
3.4. Melatonin Inhibits Hydroxyl Radical Formed by High Concentrations of Copper/Quercetin or EGCG
3.5. Melatonin Inhibits DNA Damage Caused by Copper/Quercetin via Chelating Copper
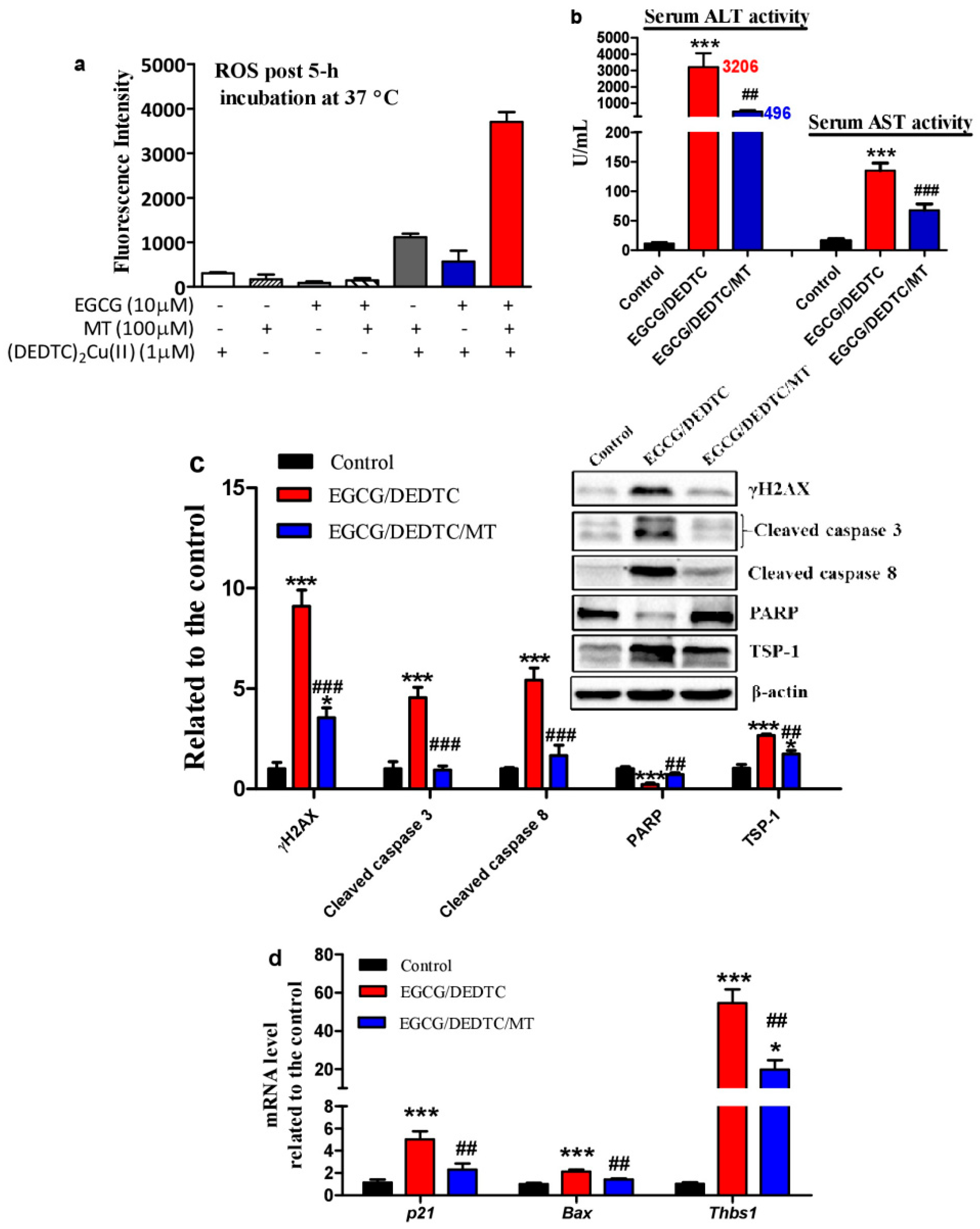
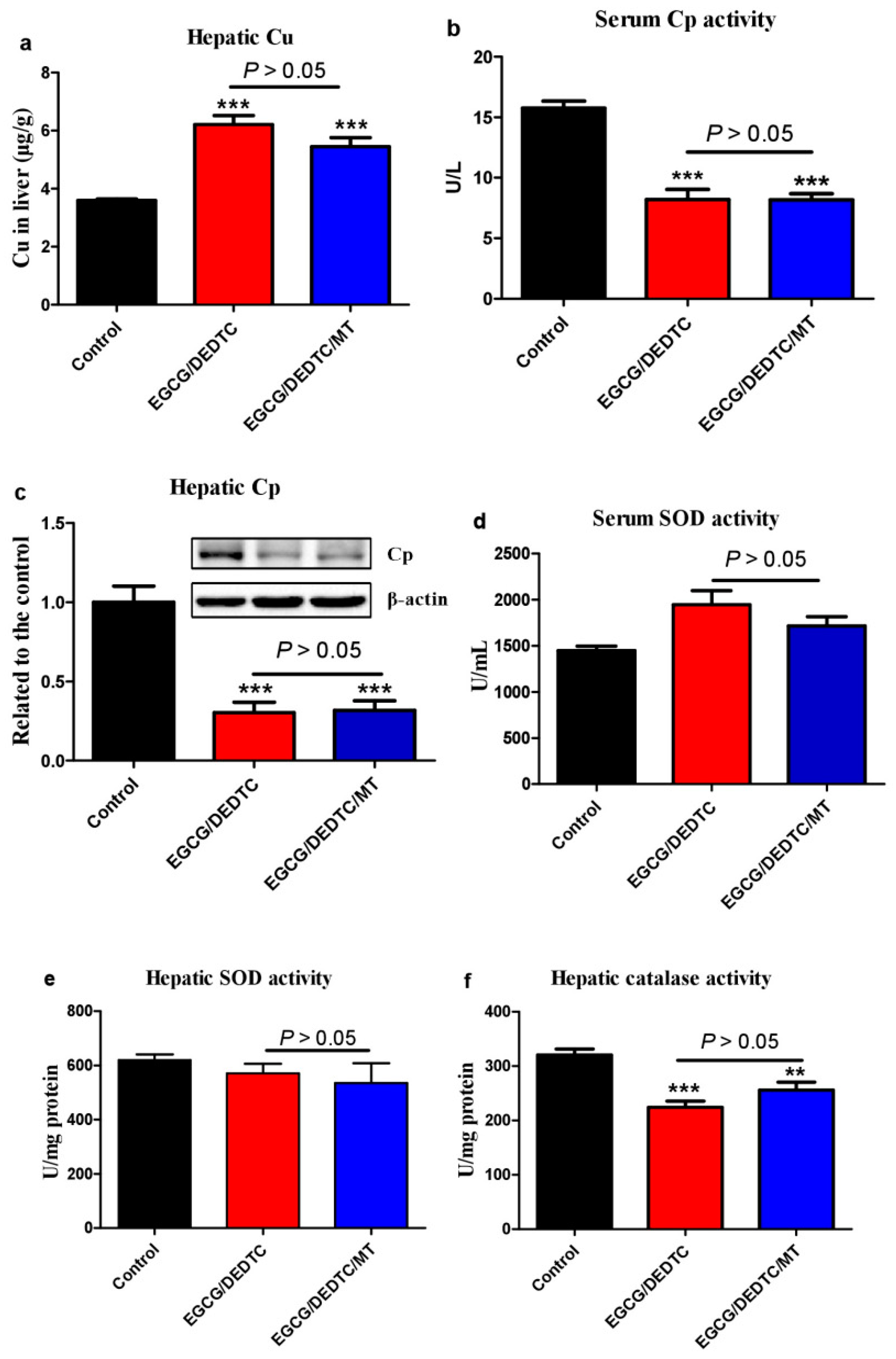
3.6. Melatonin Attenuates the Synergistic Hepatoxicity Induced by EGCG and DEDTC
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Favero, G.; Franceschetti, L.; Buffoli, B.; Moghadasian, M.H.; Reiter, R.J.; Rodella, L.F.; Rezzani, R. Melatonin: Protection against age-related cardiac pathology. Ageing Res. Rev. 2017, 35, 336–349. [Google Scholar] [CrossRef] [PubMed]
- Bonomini, F.; Borsani, E.; Favero, G.; Rodella, L.F.; Rezzani, R. Dietary Melatonin Supplementation Could Be a Promising Preventing/Therapeutic Approach for a Variety of Liver Diseases. Nutrients 2018, 10, 1135. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Sharopov, F.; Fokou, P.V.T.; Kobylinska, A.; Jonge, L.; Tadio, K.; Sharifi-Rad, J.; Posmyk, M.M.; Martorell, M.; Martins, N.; et al. Melatonin in Medicinal and Food Plants: Occurrence, Bioavailability, and Health Potential for Humans. Cells 2019, 8, 681. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Reiter, R.J. Mitochondria: The birth place, battle ground and the site of melatonin metabolism in cells. Melatonin Res. 2019, 2, 44–66. [Google Scholar] [CrossRef]
- Galano, A.; Medina, M.E.; Tan, D.X.; Reiter, R.J. Melatonin and its metabolites as copper chelating agents and their role in inhibiting oxidative stress: A physicochemical analysis. J. Pineal Res. 2015, 58, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Mayo, J.C.; Tan, D.; Sainz, R.M.; Natarajan, M.; Lopez-Burillo, S.; Reiter, R. Protection against oxidative protein damage induced by metal-catalyzed reaction or alkylperoxyl radicals: Comparative effects of melatonin and other antioxidants. Biochim. Biophys. Acta (BBA) Gen. Subj. 2003, 1620, 139–150. [Google Scholar] [CrossRef]
- Kładna, A.; Aboul-Enein, H.Y.; Kruk, I. Enhancing effect of melatonin on chemiluminescence accompanying decomposition of hydrogen peroxide in the presence of copper. Free. Radic. Biol. Med. 2003, 34, 1544–1554. [Google Scholar] [CrossRef]
- Hadi, N.; Singh, S.; Ahmad, A.; Zaidi, R. Strand scission in DNA induced by 5-hydroxytryptamine (Serotonin) in the presence of copper ions. Neurosci. Lett. 2001, 308, 83–86. [Google Scholar] [CrossRef]
- Hadi, N.; Malik, A.; Azam, S.; Khan, N.; Iqbal, J. Serotonin–Cu(II)-mediated DNA cleavage: Mechanism of copper binding by serotonin. Toxicol. Vitr. 2002, 16, 669–674. [Google Scholar] [CrossRef]
- Sakano, K.; Oikawa, S.; Hiraku, Y.; Kawanishi, S. Oxidative DNA damage induced by a melatonin metabolite, 6-hydroxymelatonin, via a unique non-o-quinone type of redox cycle. Biochem. Pharmacol. 2004, 68, 1869–1878. [Google Scholar] [CrossRef]
- Chen, Z.; Gu, Q.; Yu, X.; Huang, L.; Xu, S.; Wang, R.; Shen, W.; Shen, W. Hydrogen peroxide acts downstream of melatonin to induce lateral root formation. Ann. Bot. 2018. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Rosales-Corral, S.A.; Tan, D.X.; Acuna-Castroviejo, D.; Qin, L.; Yang, S.F.; Xu, K. Melatonin, a Full Service Anti-Cancer Agent: Inhibition of Initiation, Progression and Metastasis. Int. J. Mol. Sci. 2017, 18, 843. [Google Scholar] [CrossRef] [PubMed]
- Maitra, S.; Bhattacharya, D.; Das, S.; Bhattacharya, S. Melatonin and its anti-glioma functions: A comprehensive review. Rev. Neurosci. 2019, 30, 527–541. [Google Scholar] [CrossRef] [PubMed]
- Bedini, A.; Fraternale, A.; Crinelli, R.; Mari, M.; Bartolucci, S.; Chiarantini, L.; Spadoni, G. Design, Synthesis, and Biological Activity of Hydrogen Peroxide Responsive Arylboronate Melatonin Hybrids. Chem. Res. Toxicol. 2019, 32, 100–112. [Google Scholar] [CrossRef] [PubMed]
- Lecour, S.; Lamont, K.T. Natural Polyphenols and Cardioprotection. Mini-Reviews Med. Chem. 2011, 11, 1191–1199. [Google Scholar]
- Cory, H.; Passarelli, S.; Szeto, J.; Tamez, M.; Mattei, J. The Role of Polyphenols in Human Health and Food Systems: A Mini-Review. Front. Nutr. 2018, 5, 87. [Google Scholar] [CrossRef] [PubMed]
- Ho, C.T.; Zhang, J.; Wan, X.; Zhang, K.; Lim, J.; Yang, C.S. Antioxidants: Differing Meanings in Food Science and Health Science. J. Agric. Food Chem. 2018, 66, 3063–3068. [Google Scholar]
- Mazzanti, G.; Di Sotto, A.; Vitalone, A. Hepatotoxicity of green tea: An update. Arch. Toxicol. 2015, 89, 1175–1191. [Google Scholar] [CrossRef]
- Schönthal, A.H. Adverse effects of concentrated green tea extracts. Mol. Nutr. Food Res. 2011, 55, 874–885. [Google Scholar] [CrossRef]
- Mazzanti, G.; Menniti-Ippolito, F.; Moro, P.A.; Cassetti, F.; Raschetti, R.; Santuccio, C.; Mastrangelo, S. Hepatotoxicity from green tea: A review of the literature and two unpublished cases. Eur. J. Clin. Pharmacol. 2009, 65, 331–341. [Google Scholar] [CrossRef]
- Bagdas, D.; Çam Etöz, B.; Gul, Z.; Ziyanok, S.; Inan, S.; Turacozen, O.; Gul, N.Y.; Topal, A.; Cinkilic, N.; Taş, S.; et al. In vivo systemic chlorogenic acid therapy under diabetic conditions: Wound healing effects and cytotoxicity/genotoxicity profile. Food Chem. Toxicol. 2015, 81, 54–61. [Google Scholar] [CrossRef] [PubMed]
- León-González, A.J.; Auger, C.; Schini-Kerth, V.B. Pro-oxidant activity of polyphenols and its implication on cancer chemoprevention and chemotherapy. Biochem. Pharmacol. 2015, 98, 371–380. [Google Scholar] [CrossRef] [PubMed]
- Grzesik, M.; Bartosz, G.; Stefaniuk, I.; Pichla, M.; Namiesnik, J.; Sadowska-Bartosz, I. Dietary antioxidants as a source of hydrogen peroxide. Food Chem. 2019, 278, 692–699. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Wang, Y.; Wan, X.; Yang, C.S.; Zhang, J. Green tea polyphenol (−)-epigallocatechin-3-gallate triggered hepatotoxicity in mice: Responses of major antioxidant enzymes and the Nrf2 rescue pathway. Toxicol. Appl. Pharmacol. 2015, 283, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Dong, R.; Sun, K.; Wang, X.; Wang, J.; Yang, C.S.; Zhang, J. Synergistic toxicity of epigallocatechin-3-gallate and diethyldithiocarbamate, a lethal encounter involving redox-active copper. Free. Radic. Biol. Med. 2017, 113, 143–156. [Google Scholar] [CrossRef] [PubMed]
- Hadi, S.; Bhat, S.; Azmi, A.; Hanif, S.; Shamim, U.; Ullah, M. Oxidative breakage of cellular DNA by plant polyphenols: A putative mechanism for anticancer properties. Semin. Cancer Biol. 2007, 17, 370–376. [Google Scholar] [CrossRef] [PubMed]
- Yoshino, M.; Haneda, M.; Naruse, M.; Murakami, K. Prooxidant Activity of Flavonoids: Copper-Dependent Strand Breaks and the Formation of 8-Hydroxy-2′-Deoxyguanosine in DNA. Mol. Genet. Metab. 1999, 68, 468–472. [Google Scholar] [CrossRef]
- Yamashita, N.; Tanemura, H.; Kawanishi, S. Mechanism of oxidative DNA damage induced by quercetin in the presence of Cu(II). Mutat. Res. Mol. Mech. Mutagen. 1999, 425, 107–115. [Google Scholar] [CrossRef]
- Li, Y.; A Trush, M. Reactive oxygen-dependent DNA damage resulting from the oxidation of phenolic compounds by a copper-redox cycle mechanism. Cancer Res. 1994, 54, 1895–1898. [Google Scholar]
- Patel, R.P.; Svistunenko, D.; Wilson, M.T.; Darley-Usmar, V.M. Reduction of Cu(II) by lipid hydroperoxides: Implications for the copper-dependent oxidation of low-density lipoprotein. Biochem. J. 1997, 322, 425–433. [Google Scholar] [CrossRef]
- Shao, B.; Mao, L.; Qu, N.; Wang, Y.F.; Gao, H.Y.; Li, F.; Qin, L.; Shao, J.; Huang, C.H.; Xu, D.; et al. Mechanism of synergistic DNA damage induced by the hydroquinone metabolite of brominated phenolic environmental pollutants and Cu(II): Formation of DNA-Cu complex and site-specific production of hydroxyl radicals. Free. Radic. Biol. Med. 2017, 104, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Sun, K.; Eriksson, S.E.; Tan, Y.; Zhang, L.; Arnér, E.S.; Zhang, J. Serum thioredoxin reductase levels increase in response to chemically induced acute liver injury. Biochim. Biophys. Acta (BBA) Gen. Subj. 2014, 1840, 2105–2111. [Google Scholar] [CrossRef] [PubMed]
- Fazal, F.; Greensill, J.; Ainley, K.; Parish, J.; Rahman, A.; Hadi, S. Strand scission in DNA by quercetin and Cu(II): Identification of free radical intermediates and biological consequences of scission. Carcinogenesis 1990, 11, 2005–2008. [Google Scholar] [CrossRef] [PubMed]
- Andrushchenko, V.; van de Sande, J.H.; Wieser, H. Vibrational circular dichroism and IR absorption of DNA complexes with Cu2+ ions. Biopolymers 2003, 72, 374–390. [Google Scholar] [CrossRef] [PubMed]
- Tardito, S.; Bassanetti, I.; Bignardi, C.; Elviri, L.; Tegoni, M.; Mucchino, C.; Bussolati, O.; Franchi-Gazzola, R.; Marchiὸ, L. Copper Binding Agents Acting as Copper Ionophores Lead to Caspase Inhibition and Paraptotic Cell Death in Human Cancer Cells. J. Am. Chem. Soc. 2011, 133, 6235–6242. [Google Scholar] [CrossRef] [PubMed]
- Skrott, Z.; Mistrik, M.; Andersen, K.K.; Friis, S.; Majera, D.; Gursky, J.; Ozdian, T.; Bartkova, J.; Turi, Z.; Moudry, P.; et al. Alcohol-abuse drug disulfiram targets cancer via p97 segregase adapter NPL4. Nature 2017, 552, 194–199. [Google Scholar] [CrossRef] [PubMed]
- Isenberg, J.S.; Ridnour, L.A.; Perruccio, E.M.; Espey, M.G.; Wink, D.A.; Roberts, D.D. Thrombospondin-1 inhibits endothelial cell responses to nitric oxide in a cGMP-dependent manner. Proc. Natl. Acad. Sci. USA 2005, 102, 13141–13146. [Google Scholar] [CrossRef]
- Isenberg, J.S.; Ridnour, L.A.; Dimitry, J.; Frazier, W.A.; Wink, D.A.; Roberts, D.D. CD47 Is Necessary for Inhibition of Nitric Oxide-stimulated Vascular Cell Responses by Thrombospondin-1. J. Biol. Chem. 2006, 281, 26069–26080. [Google Scholar] [CrossRef] [PubMed]
- Hou, Z.; Sang, S.; You, H.; Lee, M.J.; Hong, J.; Chin, K.V.; Yang, C.S. Mechanism of Action of (−)-Epigallocatechin-3-Gallate: Auto-oxidation–Dependent Inactivation of Epidermal Growth Factor Receptor and Direct Effects on Growth Inhibition in Human Esophageal Cancer KYSE 150 Cells. Cancer Res. 2005, 65, 8049–8056. [Google Scholar] [CrossRef]
- Li, G.X.; Chen, Y.K.; Hou, Z.; Xiao, H.; Jin, H.; Lu, G.; Lee, M.J.; Liu, B.; Guan, F.; Yang, Z.; et al. Pro-oxidative activities and dose–response relationship of (−)-epigallocatechin-3-gallate in the inhibition of lung cancer cell growth: A comparative study in vivo and in vitro. Carcinogenesis 2010, 31, 902–910. [Google Scholar] [CrossRef]
- Wei, Y.; Chen, P.; Ling, T.; Wang, Y.; Dong, R.; Zhang, C.; Zhang, L.; Han, M.; Wang, D.; Wan, X.; et al. Certain (−)-epigallocatechin-3-gallate (EGCG) auto-oxidation products (EAOPs) retain the cytotoxic activities of EGCG. Food Chem. 2016, 204, 218–226. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Wei, Y.; Wang, T.; Wan, X.; Yang, C.S.; Reiter, R.J.; Zhang, J. Melatonin attenuates (-)-epigallocatehin-3-gallate-triggered hepatotoxicity without compromising its downregulation of hepatic gluconeogenic and lipogenic genes in mice. J. Pineal Res. 2015, 59, 497–507. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; He, Y.; Wu, X.; Zhao, G.; Zhang, K.; Yang, C.S.; Reiter, R.J.; Zhang, J. Melatonin and (-)-Epigallocatechin-3-Gallate: Partners in Fighting Cancer. Cells 2019, 8, 745. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Chen, L.D.; Poeggeler, B.; Manchester, L.C.; Reiter, R.J. Melatonin: A potent endogenous hydroxyl radical scavenger. Endocr. J. 1993, 1, 57–60. [Google Scholar]
- Galano, A. On the direct scavenging activity of melatonin towards hydroxyl and a series of peroxyl radicals. Phys. Chem. Chem. Phys. 2011, 13, 7178. [Google Scholar] [CrossRef] [PubMed]
- Galano, A.; Tan, D.X.; Reiter, R.J. Melatonin: A Versatile Protector against Oxidative DNA Damage. Molecules 2018, 23, 530. [Google Scholar] [CrossRef]
- Galano, A.; Reiter, R.J. Melatonin and its metabolites vs oxidative stress: From individual actions to collective protection. J. Pineal Res. 2018, 65, e12514. [Google Scholar] [CrossRef] [PubMed]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, J.; Wang, X.; He, Y.; Jia, L.; Yang, C.S.; Reiter, R.J.; Zhang, J. Antioxidant and Pro-Oxidant Activities of Melatonin in the Presence of Copper and Polyphenols In Vitro and In Vivo. Cells 2019, 8, 903. https://doi.org/10.3390/cells8080903
Wang J, Wang X, He Y, Jia L, Yang CS, Reiter RJ, Zhang J. Antioxidant and Pro-Oxidant Activities of Melatonin in the Presence of Copper and Polyphenols In Vitro and In Vivo. Cells. 2019; 8(8):903. https://doi.org/10.3390/cells8080903
Chicago/Turabian StyleWang, Jiajia, Xiaoxiao Wang, Yufeng He, Lijie Jia, Chung S. Yang, Russel J. Reiter, and Jinsong Zhang. 2019. "Antioxidant and Pro-Oxidant Activities of Melatonin in the Presence of Copper and Polyphenols In Vitro and In Vivo" Cells 8, no. 8: 903. https://doi.org/10.3390/cells8080903
APA StyleWang, J., Wang, X., He, Y., Jia, L., Yang, C. S., Reiter, R. J., & Zhang, J. (2019). Antioxidant and Pro-Oxidant Activities of Melatonin in the Presence of Copper and Polyphenols In Vitro and In Vivo. Cells, 8(8), 903. https://doi.org/10.3390/cells8080903






