Inhibition of the Adenosine A2A Receptor Mitigates Excitotoxic Injury in Organotypic Tissue Cultures of the Rat Cochlea
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Tissue Preparation and Organotypic Tissue Cultures
2.3. Induction of Excitotoxic Injury and Istradefylline Treatment
2.4. Immunohistochemistry
2.5. Imaging
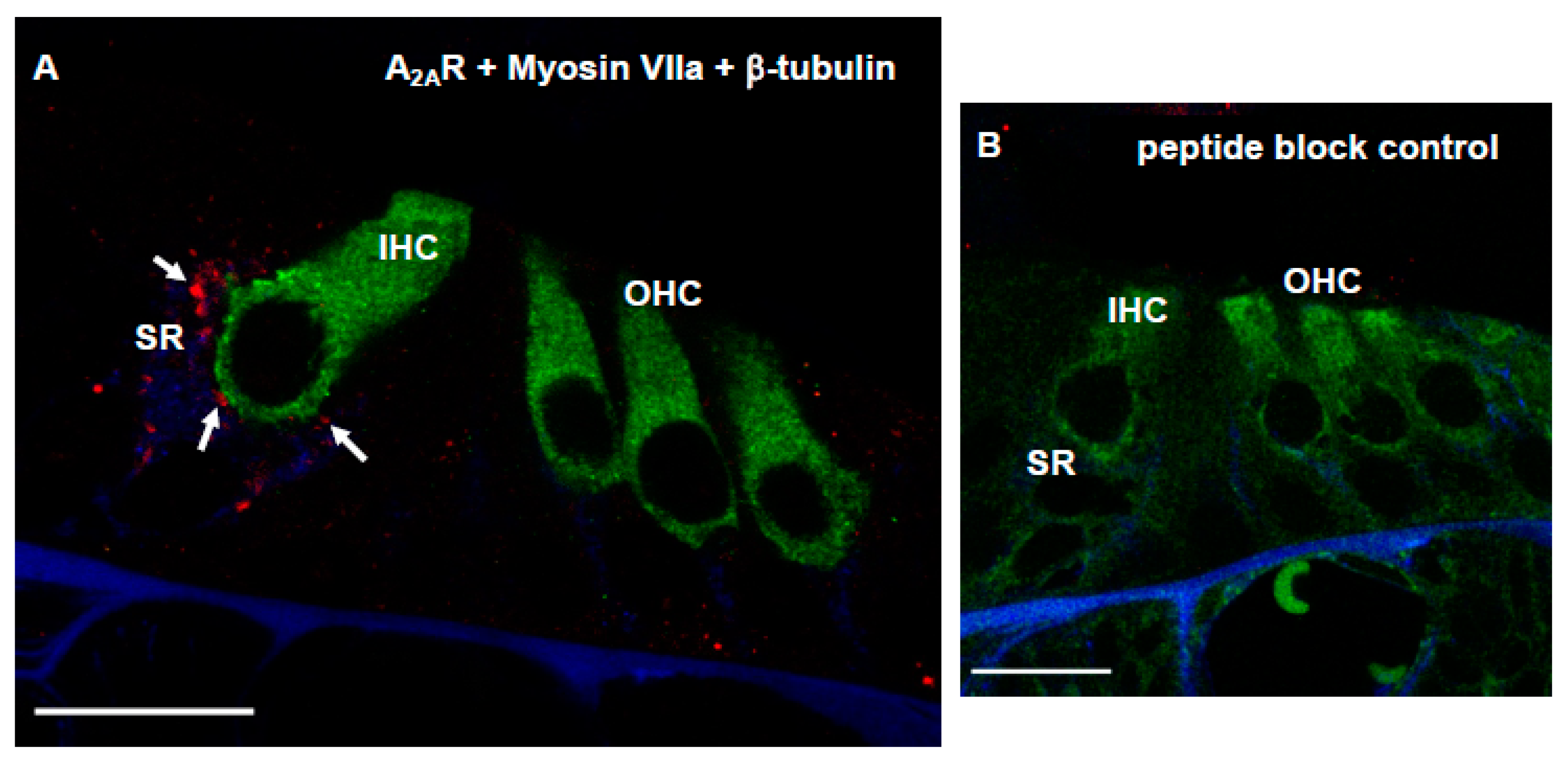
2.6. Adenosine A2AR Distribution
2.7. Statistical Analysis
3. Results
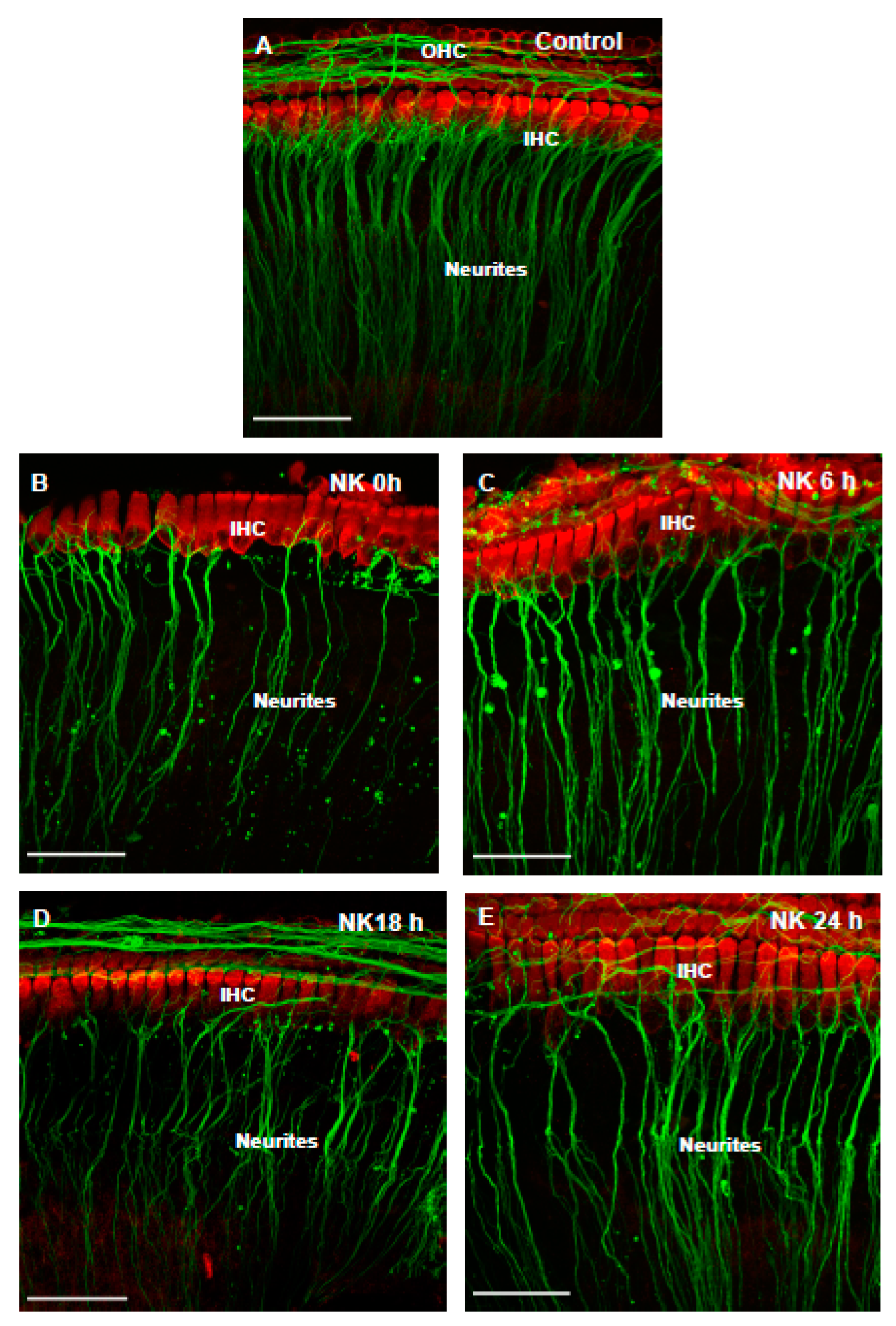
3.1. The Effect of NMDA/Kainic Acid (NK) Treatment on Cochlear Explants
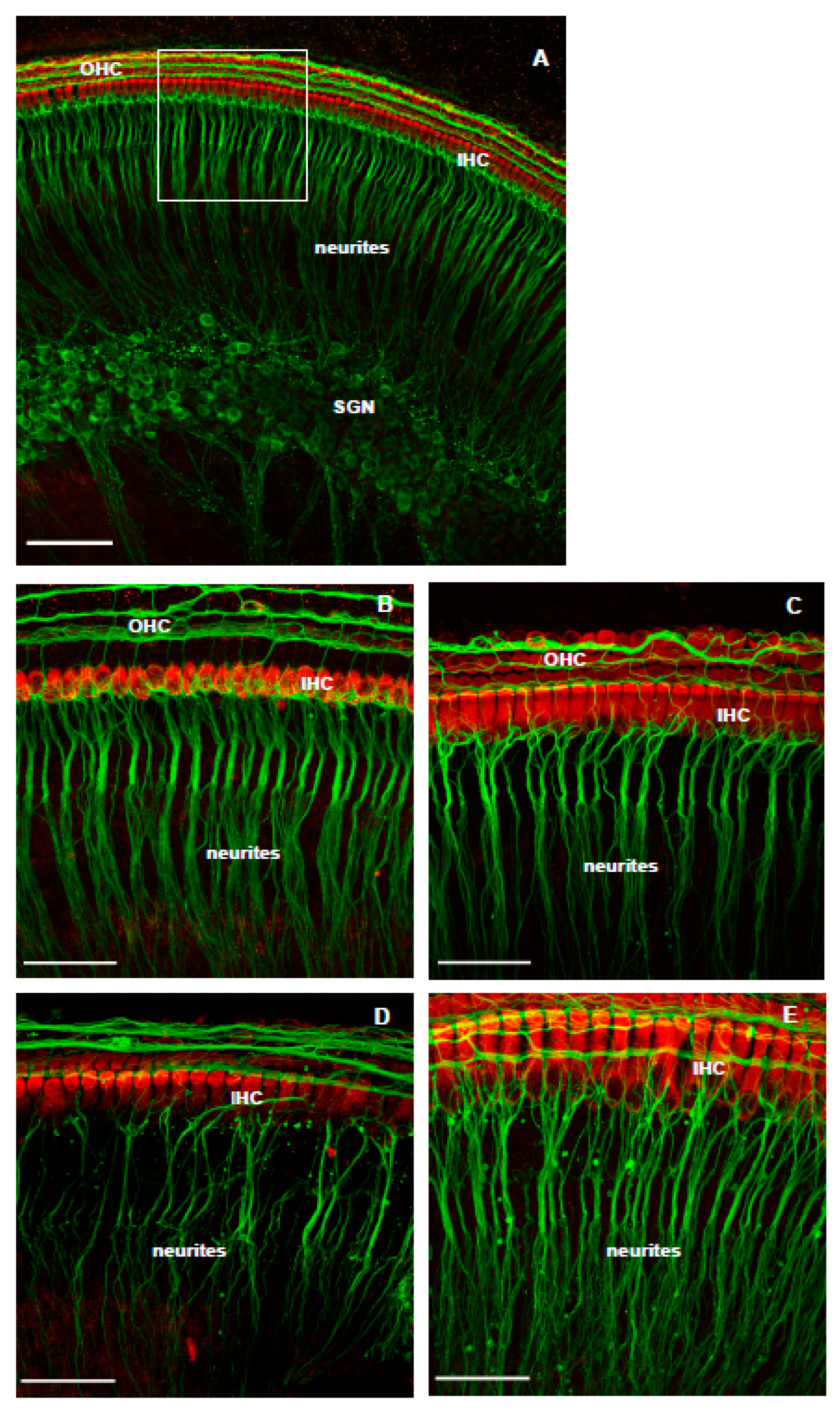
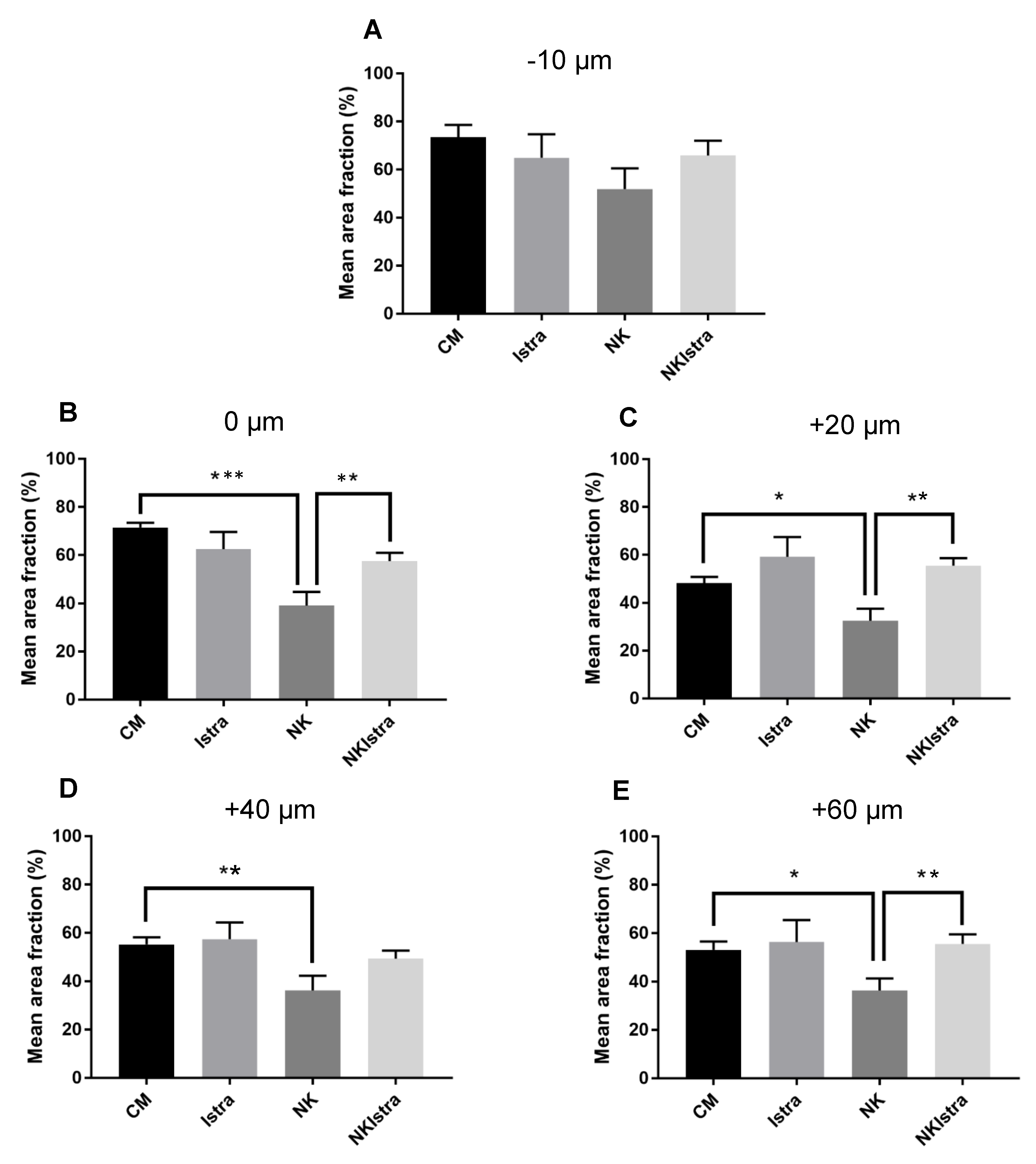
3.2. The Effect of Istradefylline on NK-Induced Loss of SGN Neurites
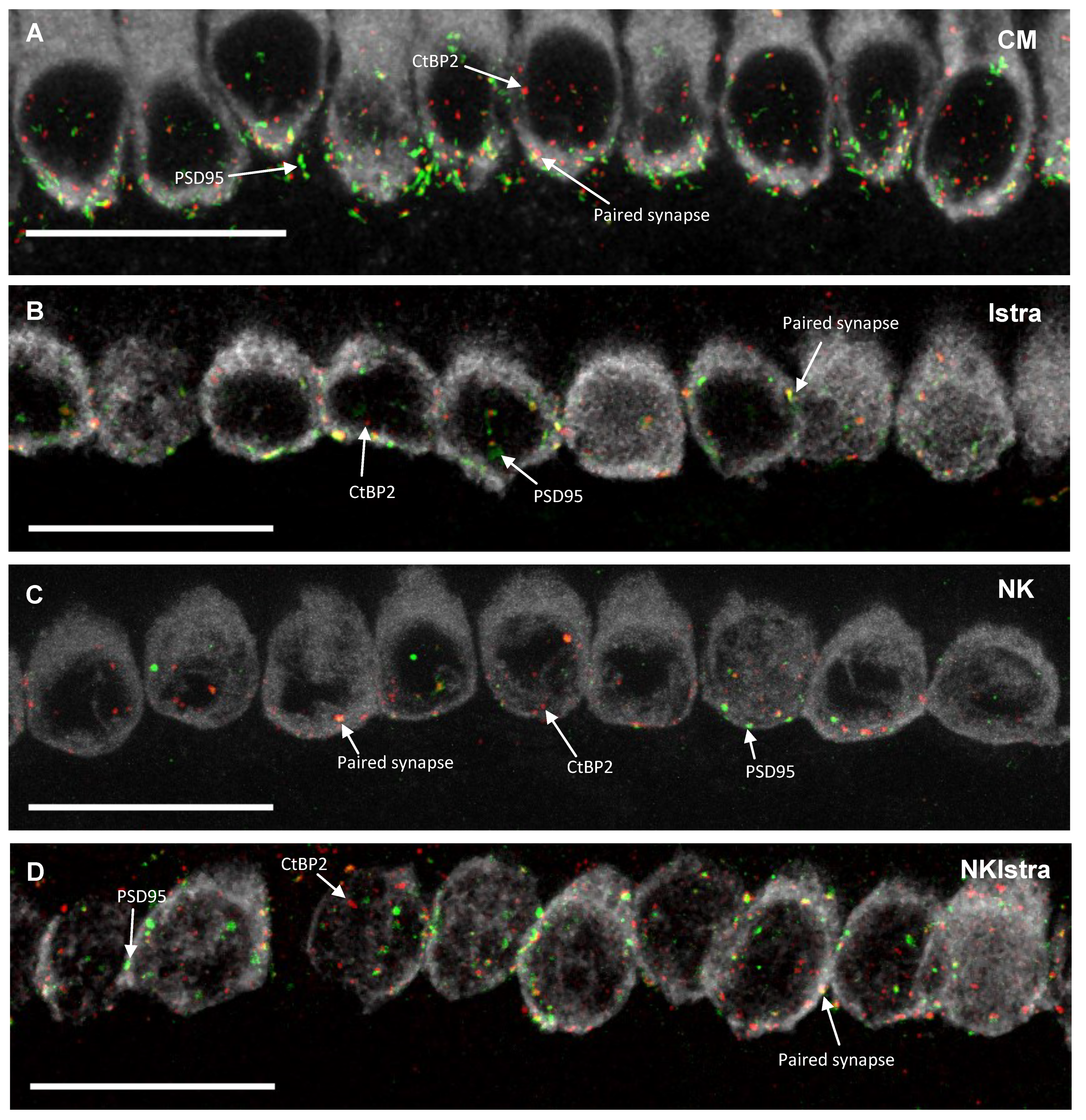
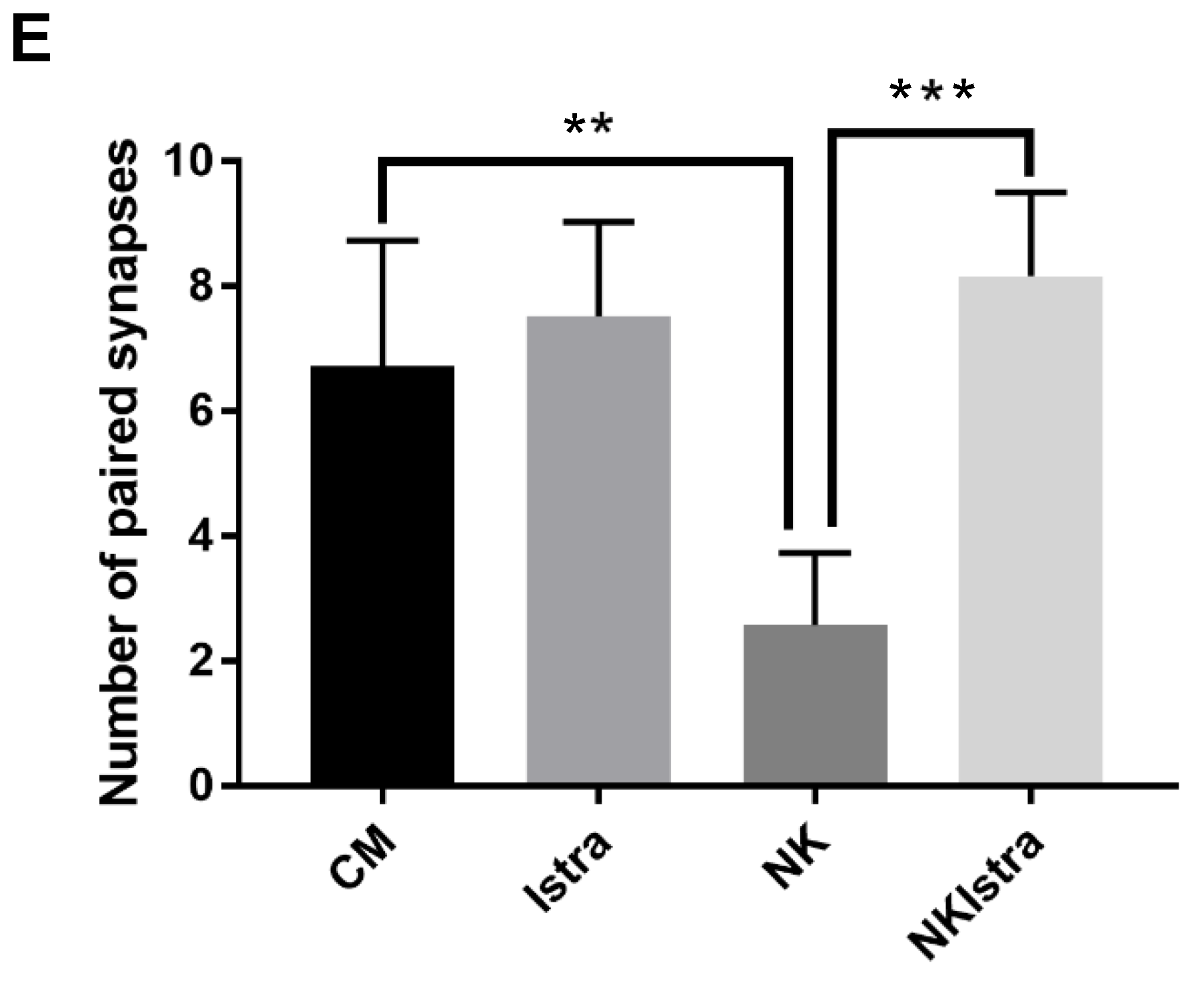
3.3. The Effect of Istradefylline on Ribbon Synapse Counts After Excitotoxic Injury
3.4. Distribution of Adenosine A2AR in Afferent Synapses of the Developing Rat Cochlea
4. Discussion
4.1. The Effect of NK-Induced Injury and Treatment with Istradefylline on SGN Neurites
4.2. The Effect of NK-Induced Injury and Istradefylline Treatment on Ribbon Synapses
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bohne, B.A.; Harding, G.W. Degeneration in the cochlea after noise damage: primary versus secondary events. Am. J. Otol. 2000, 21, 505–509. [Google Scholar] [PubMed]
- Kujawa, S.G.; Liberman, M.C. Adding insult to injury: cochlear nerve degeneration after “temporary” noise-induced hearing loss. J. Neurosci. 2009, 29, 14077–14085. [Google Scholar] [CrossRef] [PubMed]
- Kujawa, S.G.; Liberman, M.C. Synaptopathy in the noise-exposed and ageing cochlea: Primary neural degeneration in acquired sensorineural hearing loss. Hear. Res. 2015, 330 (Pt B), 191–199. [Google Scholar] [CrossRef]
- Fernandez, K.A.; Jeffers, P.W.; Lall, K.; Liberman, M.C.; Kujawa, S.G. Ageing after noise exposure: acceleration of cochlear synaptopathy in “recovered” ears. J. Neurosci. 2015, 35, 7509–7520. [Google Scholar] [CrossRef] [PubMed]
- Viana, L.M.; O’Malley, J.T.; Burgess, B.J.; Jones, D.D.; Oliveira, C.A.; Santos, F.; Merchant, S.N.; Liberman, L.D.; Liberman, M.C. Cochlear neuropathy in human presbycusis: Confocal analysis of hidden hearing loss in post-mortem tissue. Hear. Res. 2015, 327, 78–88. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liberman, L.D.; Liberman, M.C. Dynamics of cochlear synaptopathy after acoustic overexposure. J. Assoc. Res. Otolaryngol. 2015, 16, 205–219. [Google Scholar] [CrossRef] [PubMed]
- Sergeyenko, Y.; Lall, K.; Liberman, M.C.; Kujawa, S.G. Age-related cochlear synaptopathy: An early-onset contributor to auditory functional decline. J. Neurosci. 2013, 33, 13686–13694. [Google Scholar] [CrossRef]
- Schaette, R.; McAlpine, D. Tinnitus with a normal audiogram: Physiological evidence for hidden hearing loss and computational model. J. Neurosci. 2011, 31, 13452–13457. [Google Scholar] [CrossRef]
- Wan, G.; Gomez-Casati, M.E.; Gigliello, A.R.; Liberman, M.C.; Corfas, G. Neurotrophin-3 regulates ribbon synapse density in the cochlea and induces synapse regeneration after acoustic trauma. eLife 2014. [Google Scholar] [CrossRef]
- Sly, D.J.; Campbell, L.; Uschakov, A.; Saief, S.T.; Lam, M.; O’Leary, S.J. Applying neurotrophins to the round window rescues auditory function and reduces inner hair cell synaptopathy after noise-induced hearing loss. Otol. Neurotol. 2016, 37, 1223–1230. [Google Scholar] [CrossRef]
- Chen, J.F.; Eltzschig, H.K.; Fredholm, B.B. Adenosine receptors as drug targets—What are the challenges? Nature Rev. 2013, 12, 265–286. [Google Scholar] [CrossRef] [PubMed]
- Vlajkovic, S.M.; Abi, S.; Wang, C.J.H.; Housley, G.D.; Thorne, P.R. Differential distribution of adenosine receptors in the rat cochlea. Cell Tissue Res. 2007, 328, 461–471. [Google Scholar] [CrossRef] [PubMed]
- Wong, A.C.Y.; Guo, C.X.; Gupta, R.; Housley, G.D.; Thorne, P.R.; Vlajkovic, S.M. Post-exposure administration of A1 adenosine receptor agonists attenuates noise-induced hearing loss. Hear. Res. 2010, 260, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Vlajkovic, S.M.; Lee, K.H.; Wong, A.C.Y.; Guo, C.X.; Gupta, R.; Housley, G.D.; Thorne, P.R. Adenosine amine congener mitigates noise-induced cochlear injury. Purinergic Signal. 2010, 6, 273–281. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaur, T.; Borse, V.; Sheth, S.; Sheehan, K.; Ghosh, S.; Tupal, S.; Jajoo, S.; Mukherjea, D.; Rybak, L.P.; Ramkumar, V. Adenosine A1 receptor protects against cisplatin ototoxicity by suppressing the NOX3/STAT1 inflammatory pathway in the cochlea. J. Neurosci. 2016, 36, 3962–3977. [Google Scholar] [CrossRef]
- Lin, S.C.Y.; Thorne, P.R.; Housley, G.D.; Vlajkovic, S.M. Purinergic signalling and aminoglycoside ototoxicity: The opposing roles of P1 (adenosine) and P2 (ATP) receptors on cochlear hair cell survival. Front. Cell Neurosci. 2019, 13, 207. [Google Scholar] [CrossRef] [PubMed]
- Vlajkovic, S.M.; Housley, G.D.; Thorne, P.R. Adenosine and the auditory system. Curr. Neuropharmacol. 2009, 7, 246–256. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.F. Adenosine receptor control of cognition in normal and disease. Int. Rev. Neurobiol. 2014, 119, 257–307. [Google Scholar]
- Woods, L.T.; Ajit, D.; Camden, J.M.; Erb, L.; Weisman, G.A. Purinergic receptors as potential therapeutic targets in Alzheimer’s disease. Neuropharmacol. 2016, 104, 169–179. [Google Scholar] [CrossRef]
- Vlajkovic, S.M.; Ambepitiya, K.; Barclay, M.; Boison, D.; Housley, G.D.; Thorne, P.R. Adenosine receptors regulate susceptibility to noise-induced neural injury in the mouse cochlea and hearing loss. Hear. Res. 2017, 345, 43–51. [Google Scholar] [CrossRef]
- Jenner, P. Istradefylline, a novel adenosine A2A receptor antagonist, for the treatment of Parkinson’s disease. Expert Opin. Investig. Drugs 2005, 14, 729–738. [Google Scholar] [CrossRef]
- Dungo, R.; Deeks, E.D. Istradefylline: first global approval. Drugs 2013, 73, 875–882. [Google Scholar] [CrossRef]
- Pinna, A. Adenosine A2A receptor antagonists in Parkinson’s disease: progress in clinical trials from the newly approved istradefylline to drugs in early development and those already discontinued. CNS Drugs 2014, 28, 455–474. [Google Scholar] [CrossRef]
- Wang, Q.; Green, S.H. Functional role of neurotrophin-3 in synapse regeneration by spiral ganglion neurons on inner hair cells after excitotoxic trauma in vitro. J. Neurosci. 2011, 31, 7938–7949. [Google Scholar] [CrossRef]
- Munnamalai, V.; Fekete, D.M. Organotypic culture of the mouse cochlea from embryonic day 12 to the neonate. Methods Mol. Biol. 2016, 1427, 293–303. [Google Scholar]
- Landegger, L.D.; Dilwali, S.; Stankovic, K.M. Neonatal murine cochlear explant technique as an in vitro screening tool in hearing research. J. Vis. Exp. 2017, 124. [Google Scholar] [CrossRef]
- Eybalin, M. Neurotransmitters and neuromodulators of the mammalian cochlea. Physiol. Rev. 1993, 73, 309–373. [Google Scholar] [CrossRef]
- Niedzielski, A.S.; Wenthold, R.J. Expression of AMPA, kainate and NMDA receptor subunits in cochlear and vestibular ganglia. J. Neurosci. 1995, 15, 2338–2353. [Google Scholar] [CrossRef]
- Fujikawa, T.; Petralia, R.S.; Fitzgerald, T.S.; Wang, Y.; Millis, B.; Morgado-Diaz, J.A.; Kitamura, K.; Kachar, B. Localization of kainate receptors in inner and outer hair cell synapses. Hear. Res. 2014, 314, 20–32. [Google Scholar] [CrossRef] [Green Version]
- Knipper, M.; Köpschall, I.; Rohbock, K.; Köpke, A.K.; Bonk, I.; Zimmermann, U.; Zenner, H. Transient expression of NMDA receptors during rearrangement of AMPA-receptor-expressing fibers in the developing inner ear. Cell Tissue Res. 1997, 287, 23–41. [Google Scholar] [CrossRef]
- Zhang-Hooks, Y.; Agarwal, A.; Mishina, M.; Bergles, D.E. NMDA receptors enhance spontaneous activity and promote neuronal survival in the developing cochlea. Neuron 2016, 89, 337–350. [Google Scholar] [CrossRef]
- Takago, H.; Oshima-Takago, T. Pre- and postsynaptic ionotropic glutamate receptors in the auditory system of mammals. Hear. Res. 2018, 362, 1–13. [Google Scholar] [CrossRef]
- Pujol, R.; Puel, J.L. Excitotoxicity, synaptic repair, and functional recovery in the mammalian cochlea: A review of recent findings. Ann. N.Y. Acad. Sci. 1999, 884, 249–254. [Google Scholar] [CrossRef]
- Rego, A.; Oliveira, C. Mitochondrial dysfunction and reactive oxygen species in excitotoxicity and apoptosis: Implications for the pathogenesis of neurodegenerative diseases. Neurochem. Res. 2003, 28, 1563–1574. [Google Scholar] [CrossRef]
- Wang, Y.; Gu, Z.; Cao, Y.; Liang, Z.; Han, R.; Bennett, M.C.; Qin, Z. Lysosomal enzyme cathepsin B is involved in kainic acid-induced excitotoxicity in rat striatum. Brain Res. 2006, 1071, 245–249. [Google Scholar] [CrossRef]
- Pujol, R.; Lenoir, M.; Robertson, D.; Eybalin, M.; Johnstone, B.M. Kainic acid selectively alters auditory dendrites connected with cochlear inner hair cells. Hear. Res. 1985, 18, 145–151. [Google Scholar] [CrossRef]
- Roth, B.; Bruns, V. Postnatal development of the rat organ of Corti. II. Hair cell receptors and their supporting elements. Anat. Embryol. 1992, 185, 571–581. [Google Scholar] [CrossRef]
- Sichardt, K.; Nieber, K. Adenosine A1 receptor: Functional receptor-receptor interactions in the brain. Purinergic Signal. 2007, 3, 285–298. [Google Scholar] [CrossRef]
- Borea, P.A.; Gessi, S.; Merighi, S.; Vincenzi, F.; Varani, K. Pharmacology of adenosine receptors: The state of the art. Physiol. Rev. 2018, 98, 1591–1625. [Google Scholar] [CrossRef]
- Cunha, R.A. Neuroprotection by adenosine in the brain: From A1 receptor activation to A2A receptor blockade. Purinerg. Signal. 2005, 1, 111–134. [Google Scholar] [CrossRef]
- Zhao, Z.; Zhao, Y.; Ning, Y.; Yang, N.; Peng, Y.; Li, P.; Chen, X.Y.; Liu, D.; Wang, H.; Chen, X.; et al. Adenosine A2A receptor inactivation alleviates early-onset cognitive dysfunction after traumatic brain injury involving an inhibition of tau hyperphosphorylation. Transl. Psychiatry 2017, 7, e1123. [Google Scholar] [CrossRef]
- Leite, M.R.; Wilhelm, E.A.; Jesse, C.R.; Brandao, R.; Nogueira, C.W. Protective effect of caffeine and a selective A2A receptor antagonist on impairment of memory and oxidative stress of aged rats. Exp. Gerontol. 2011, 46, 309–315. [Google Scholar] [CrossRef]
- Dall’Igna, O.P.; Fett, P.; Gomes, M.W.; Souza, D.O.; Cunha, R.A.; Lara, D.R. Caffeine and adenosine A2A receptor antagonists prevent beta-amyloid (25–35)-induced cognitive deficits in mice. Exp. Neurol. 2007, 203, 241–245. [Google Scholar]
- Armentero, M.T.; Pinna, A.; Ferré, S.; Lanciego, J.L.; Müller, C.E.; Franco, R. Past, present and future of A2A adenosine receptor antagonists in the therapy of Parkinson’s disease. Pharmacol. Therap. 2011, 132, 280. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, B.R.; Lin, S.C.; Espinosa, K.; Thorne, P.R.; Vlajkovic, S.M. Inhibition of the Adenosine A2A Receptor Mitigates Excitotoxic Injury in Organotypic Tissue Cultures of the Rat Cochlea. Cells 2019, 8, 877. https://doi.org/10.3390/cells8080877
Han BR, Lin SC, Espinosa K, Thorne PR, Vlajkovic SM. Inhibition of the Adenosine A2A Receptor Mitigates Excitotoxic Injury in Organotypic Tissue Cultures of the Rat Cochlea. Cells. 2019; 8(8):877. https://doi.org/10.3390/cells8080877
Chicago/Turabian StyleHan, Belinda RX, Shelly CY Lin, Kristan Espinosa, Peter R Thorne, and Srdjan M Vlajkovic. 2019. "Inhibition of the Adenosine A2A Receptor Mitigates Excitotoxic Injury in Organotypic Tissue Cultures of the Rat Cochlea" Cells 8, no. 8: 877. https://doi.org/10.3390/cells8080877




