Serum Bile Acids Profiling in Inflammatory Bowel Disease Patients Treated with Anti-TNFs
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Study Outcomes
2.3. sBA Analysis
2.4. Ethical Considerations
2.5. Statistical Analysis
3. Results
3.1. Demographic and Clinical Characteristics
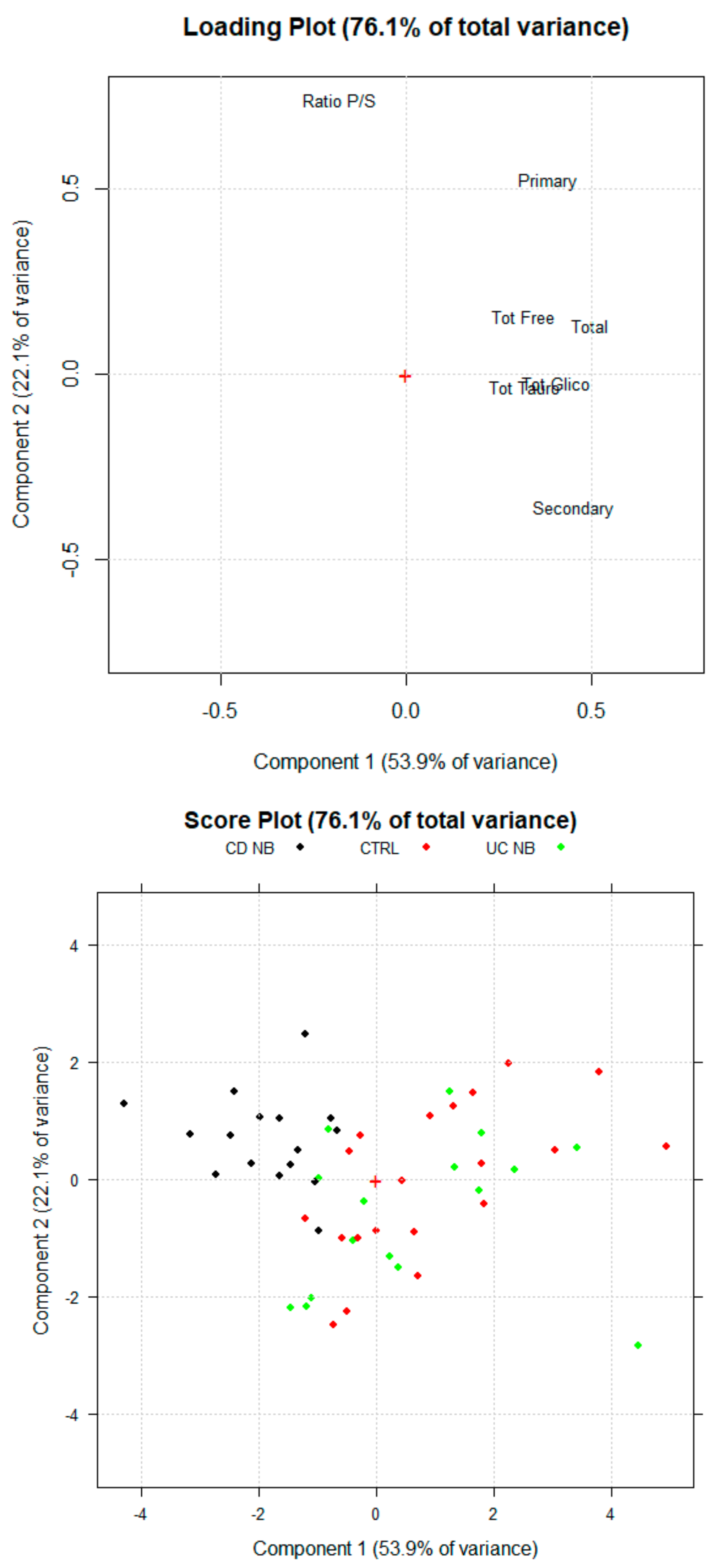
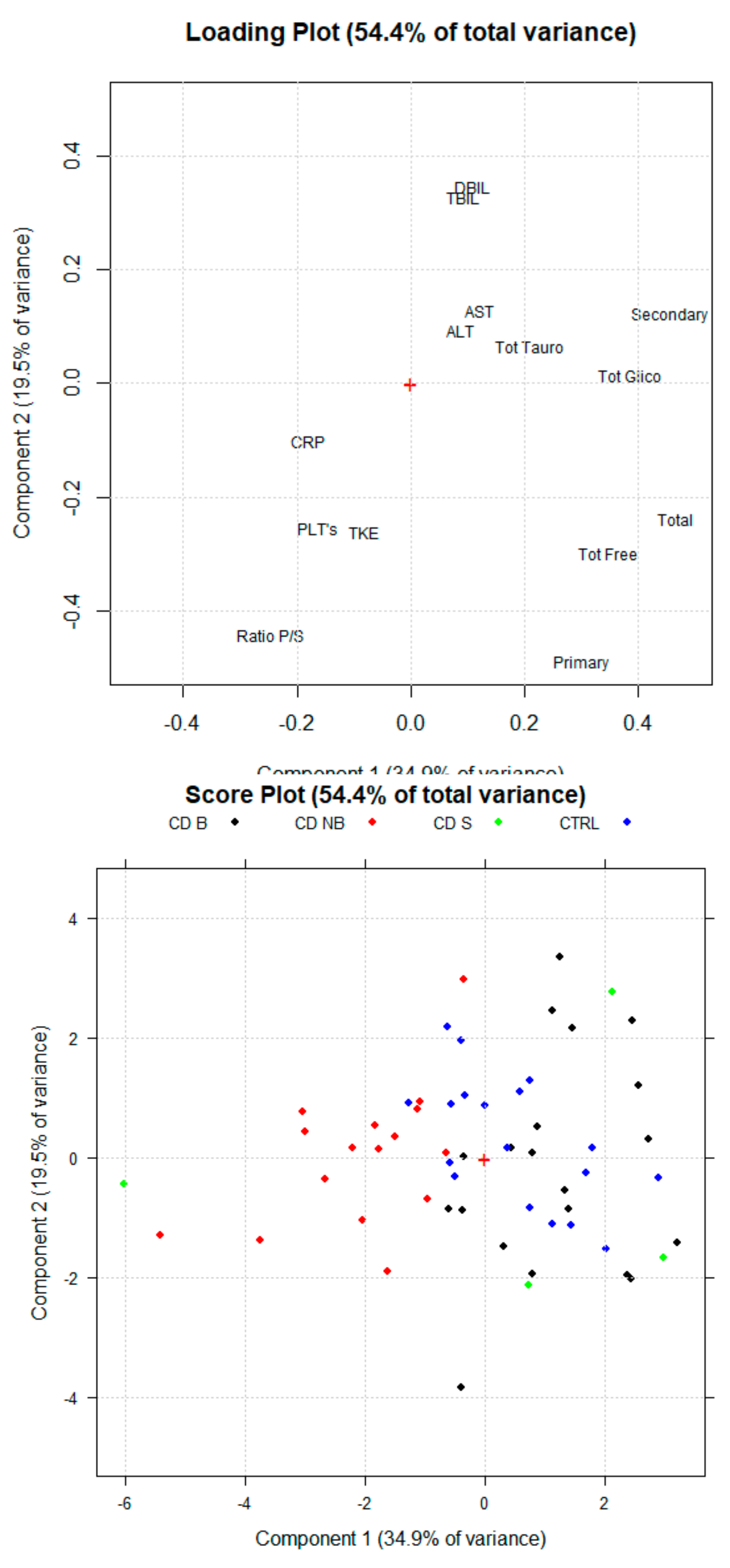
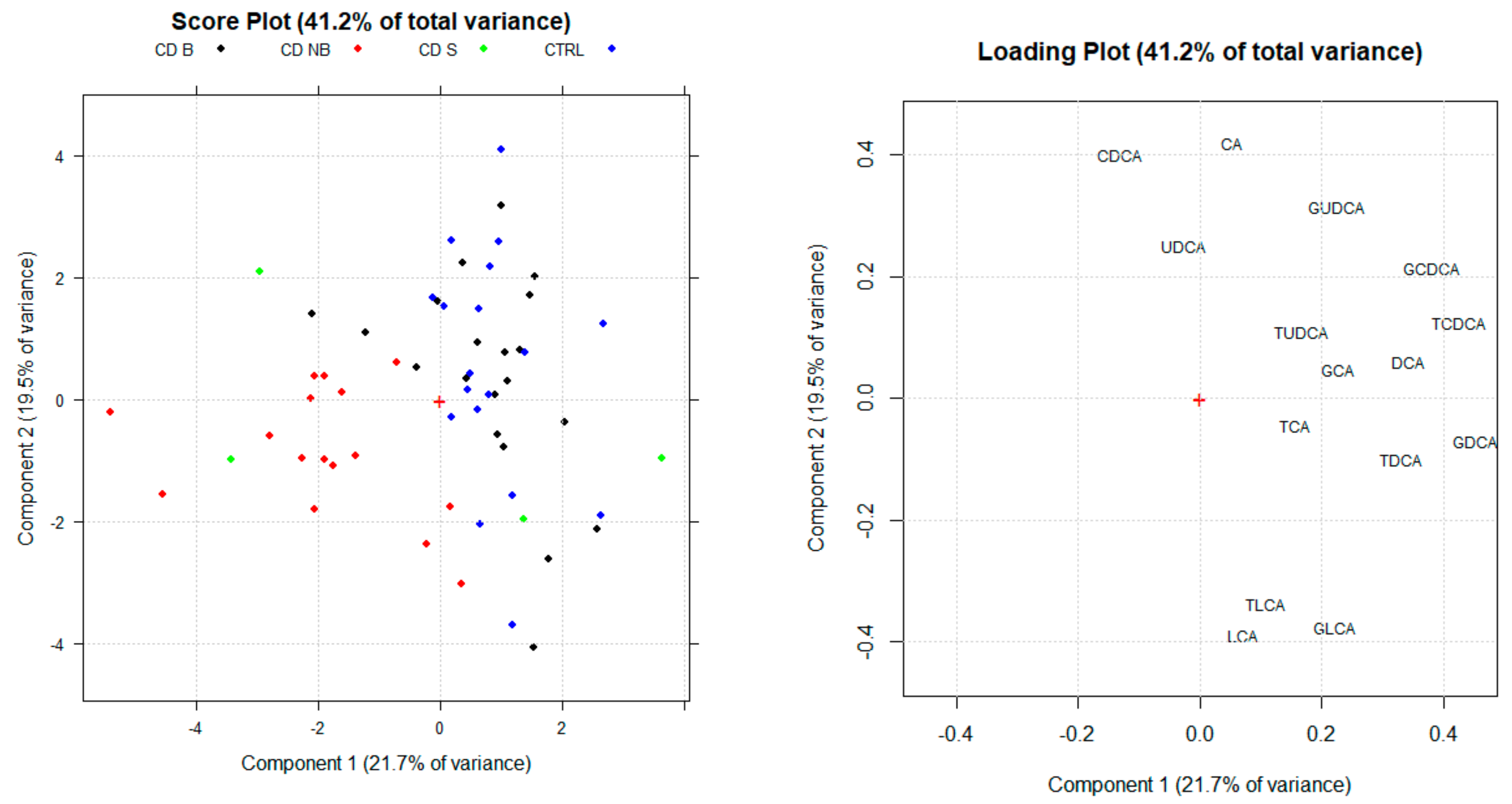
3.2. Principal Component Analysis
3.3. Serum BA Profile in CD Patients Treated with Anti-TNF Alpha
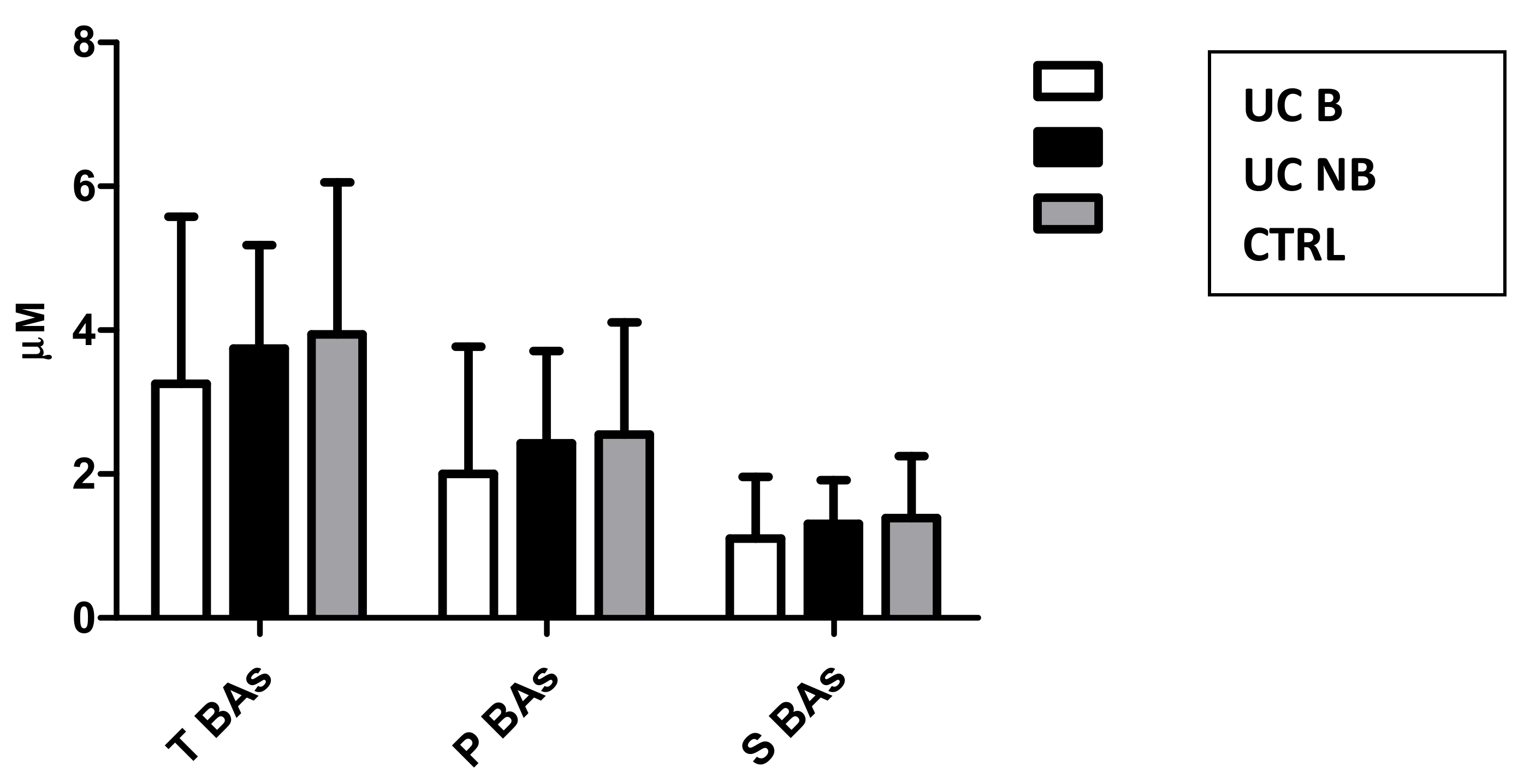
3.4. sBA Profile in UC Patients Treated with Anti-TNF Alpha Therapy
3.5. Secondary Outcomes
3.5.1. Disease Duration
3.5.2. Age at Diagnosis
3.5.3. Disease Extension
3.5.4. Inflammatory State
3.5.5. Steroid Treatment
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Duboc, H.; Rajca, S.; Rainteau, D.; Benarous, D.; Maubert, M.A.; Quervain, E.; Bridonneau, C. Connecting dysbiosis, bile-acid dysmetabolism and gut inflammation in inflammatory bowel diseases. Gut 2013, 62, 531–539. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Houten, S.M.; Mataki, C.; Christoffolete, M.A.; Kim, B.W.; Sato, H.; Schoonjans, K. Bile acids induce energy expenditure by promoting intracellular thyroid hormone activation. Nature 2006, 439, 484–489. [Google Scholar] [CrossRef]
- Aldini, R.; Roda, A.; Festi, D.; Mazzella, G.; Morselli, A.M.; Sama, C.; Barbara, L. Diagnostic value of serum primary bile acids in detecting bile acid malabsorption. Gut 1982, 23, 829–834. [Google Scholar] [CrossRef]
- Kirwan, W.O.; Smith, A.N.; Mitchell, W.D.; Falconer, J.D.; Eastwood, M.A. Bile acids and colonic motility in the rabbit and the human. Gut 1975, 16, 894–902. [Google Scholar] [CrossRef] [PubMed]
- Kullak-Ublick, G.A.; Stieger, B.; Meier, P.J. Enterohepatic bile salt transporters in normal physiology and liver disease. Gastroenterology 2004, 126, 322–342. [Google Scholar] [CrossRef]
- Hofmann, A.F.; Borgström, B. The intraluminal phase of fat digestion in man: The lipid content of the micellar and oil phases of intestinal content obtained during fat digestion and absorption. J. Clin. Investig. 1964, 43, 247–257. [Google Scholar] [CrossRef] [PubMed]
- Parks, D.J.; Blanchard, S.G.; Bledsoe, R.K.; Chandra, G.; Consler, T.G.; Kliewer, S.A.; Lehmann, J.M. Bile acids: Natural ligands for an orphan nuclear receptor. Science 1999, 284, 1365–1368. [Google Scholar] [CrossRef] [PubMed]
- Kawamata, Y.; Fujii, R.; Hosoya, M.; Harada, M.; Yoshida, H.; Miwa, M.; Hinuma, S. AG protein-coupled receptor responsive to bile acids. J. Biol. Chem. 2003, 278, 9435–9440. [Google Scholar] [CrossRef]
- Ridlon, J.M.; Kang, D.J.; Hylemon, P.B. Bile salt biotransformations by human intestinal bacteria. J. Lipid Res. 2006, 47, 241–259. [Google Scholar] [CrossRef] [Green Version]
- Vessey, D.A. The biochemical basis for the conjugation of bile acids with either glycine or taurine. Biochem. J. 1978, 174, 621–626. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ejderhamn, J.; Strandvik, B. Serum bile acids in relation to disease activity and intake of dietary fibers in juvenile ulcerative colitis. Digestion 1991, 50, 162–169. [Google Scholar] [CrossRef]
- Gnewuch, C.; Liebisch, G.; Langmann, T.; Dieplinger, B.; Mueller, T.; Haltmayer, M.; Schmitz, G. Serum bile acid profiling reflects enterohepatic detoxification state and intestinal barrier function in inflammatory bowel disease. World J. Gastroenterol. WJG 2009, 15, 3134. [Google Scholar] [CrossRef]
- Chiang, J.Y. Bile acids: Regulation of synthesis. J. Lipid Res. 2009, 50, 1955–1966. [Google Scholar] [CrossRef]
- Lynch, S.V.; Pedersen, O. The human intestinal microbiome in health and disease. N. Engl. J. Med. 2016, 375, 2369–2379. [Google Scholar] [CrossRef]
- Hofmann, A.F. Chronic diarrhea caused by idiopathic bile acid malabsorption: An explanation at last. Expert Rev. Gastroenterol. Hepatol. 2009, 3, 461–464. [Google Scholar] [CrossRef]
- Monte, M.J.; Marin, J.J.; Antelo, A.; Vazquez Tato, J. Bile acids: Chemistry, physiology, and pathophysiology. World J. Gastroenterol. 2009, 15, 804–816. [Google Scholar] [CrossRef]
- Tiraterra, E.; Franco, P.; Porru, E.; Katsanos, K.H.; Christodoulou, D.K.; Roda, G. Role of bile acids in inflammatory bowel disease. Ann. Gastroenterol. 2018, 31, 266–272. [Google Scholar] [CrossRef]
- Vashist, N.M.; Samaan, M.; Mosli, M.H.; Parker, C.E.; MacDonald, J.K.; Nelson, S.A.; Zou, G.Y.; Feagan, B.G.; Khanna, R.; Jairath, V. Endoscopic scoring indices for evaluation of disease activity in ulcerative colitis. Cochrane Database Syst. Rev. 2018, 16, CD011450. [Google Scholar] [CrossRef]
- Evertsz, F.B.; Hoeks, C.C.; Nieuwkerk, P.T.; Stokkers, P.C.; Ponsioen, C.Y.; Bockting, C.L.; Sanderman, R.; Sprangers, M.A. Development of the patient Harvey Bradshaw index and a comparison with a clinician-based Harvey Bradshaw index assessment of Crohn’s disease activity. J. Clin. Gastroenterol. 2013, 47, 850–856. [Google Scholar] [CrossRef]
- Daperno, M.; D’Haens, G.; Van Assche, G.; Baert, F.; Bulois, P.; Maunoury, V.; Sostegni, R.; Rocca, R.; Pera, A.; Gevers, A. Development and validation of a new, simplified endoscopic activity score for Crohn’s disease: The SES-CD. Gastrointest. Endosc. 2004, 60, 505–512. [Google Scholar] [CrossRef]
- Roda, A.; Pellicciari, R.; Gioiello, A.; Neri, F.; Camborata, C.; Passeri, D.; Montagnani, M. Semisynthetic bile acid FXR and TGR5 agonists: Physicochemical properties, pharmacokinetics, and metabolism in the rat. J. Pharmacol. Exp. Ther. 2014, 350, 56–68. [Google Scholar] [CrossRef]
- Kwon, R.S.; Carey, M.C. Do steroids ameliorate bile acid malabsorption in Crohn’s disease? Gut 2004, 53, 10–11. [Google Scholar] [CrossRef]
- Uchiyama, K.; Kishi, H.; Komatsu, W.; Nagao, M.; Ohhira, S.; Kobashi, G. Lipid and Bile Acid Dysmetabolism in Crohn’s Disease. J. Immunol. Res. 2018, 2018, 1–6. [Google Scholar] [CrossRef]


| PLT’s (×1000) | CRP (0–6 mg/L) | Endoscopic activity (0 = not active, 1 = mild, 2 = moderate, 3 = severe) | Disease activity (0 = not active, 1 = mild, 2 = moderate, 3 = severe) | TBIL | Montreal Classification | Date of Diagnosis | Medication | |
|---|---|---|---|---|---|---|---|---|
| CD1 | 349 | 9 | 1 | 0 | 0.4 | L1B1 | 2012 | infliximab |
| CD2 | 303 | 5 | 0 | 0 | 0.6 | L1B3 | 2011 | infliximab |
| CD3 | 284 | 2 | 2 | 1 | 0.4 | L2B3 | 1990 | infliximab |
| CD4 | 250 | 2 | 0 | 0 | 0.9 | L1B1 | 2009 | infliximab |
| CD5 | 406 | 7 | 1 | 1 | 0.5 | L3B1 | 2011 | infliximab, methotrexate |
| CD6 | 260 | 2 | 1 | 2 | 0.9 | L1B1 | NA | adalumimab |
| CD7 | 287 | 2 | 0 | 0 | 0.4 | L1B1 | 2009 | adalumimab, 5-ASΑ |
| CD8 | 395 | 29 | 2 | 1 | 1.2 | L1B1 | 2011 | infliximab |
| CD9 | 281 | 2 | 0 | 0 | 0.4 | L1B2 | 2013 | infliximab |
| CD10 | 228 | 3 | 0 | 0 | 0.9 | L1B1 | 2003 | infliximab |
| CD11 | 485 | 14 | 2 | 2 | 0.5 | L1B2 | 2006 | infliximab |
| CD12 | 303 | 3 | 2 | 1 | 0.4 | L1B1 | 2010 | infliximab |
| CD13 | 302 | 12 | 1 | 2 | 0.4 | L1B1 | 2000 | adalumimab, 5-ASΑ |
| CD14 | 244 | 1 | 0 | 0 | 1.2 | L1B1 | 1997 | infliximab |
| CD15 | 407 | 5 | 1 | 1 | 0.6 | L3B1 | 2012 | infliximab |
| CD16 | 183 | 2 | 0 | 0 | 0.9 | L3B1 | NA | infliximab |
| CD17 | 162 | 4 | 0 | 0 | 0.5 | L1B1 | 2012 | infliximab, AZA, 5-ASA |
| CD18 | 245 | 1 | 0 | 0 | 0.4 | L1B1 | 2012 | infliximab |
| CD19 | 270 | NA | 0 | 0 | 1.2 | L3B2 | 1997 | adalumimab, 5-ASΑ sus |
| CD20 | 385 | 7 | 1 | 3 | 0.6 | L3B2 | 2014 | adalumimab |
| UC1 | NA | NA | NA | NA | NA | E3 | 2008 | infliximab |
| UC2 | NA | NA | NA | NA | NA | E3 | 2012 | golimumab |
| UC3 | 157 | 7 | 0 | 0 | 0.7 | E2 | NA | golimumab, 5-ASA |
| UC4 | 381 | 21 | 1 | 1 | 0.3 | E3 | 2004 | golimumab, steroids |
| UC5 | 196 | 4 | 0 | 0 | 1.2 | E2 | 2016 | infliximab, AZA, 5-ASA |
| UC6 | 253 | 3 | 0 | 0 | 0.4 | E3 | 1995 | infliximab |
| UC7 | 267 | 2 | 1 | 1 | 0.4 | E3 | 1996 | infliximab, 5-ASA |
| UC8 | 261 | 5 | 0 | 0 | 0.4 | E3 | NA | vedolizumab, AZA, 5-ASA |
| UC9 | 343 | 1 | 0 | 0 | 0.4 | E3 | 2006 | golimumab, steroids, 5-ASA |
| UC10 | 470 | 6 | 1 | 0 | 0.4 | E3 | 2000 | adalimumab, AZA, 5-ASA, ursodeoxycholic acid |
| UC11 | 209 | 5 | 1 | 1 | 0.3 | E3 | NA | infliximab |
| UC12 | 228 | 2 | 1 | 1 | 0.5 | E3 | 1985 | infliximab, steroids |
| UC13 | 176 | 3 | 0 | 0 | 2.5 | E3 | 1984 | infliximab |
| UC14 | 203 | 6 | 2 | 3 | 0.6 | E3 | 2006 | infliximab, 5-ASA |
| UC15 | 242 | 3 | 1 | 1 | 0.3 | E3 | 1997 | infliximab |
| UC16 | 322 | 5 | 1 | 2 | 0.9 | E3 | 2010 | infliximab, Aza adalimumab, 5-ASA |
| UC17 | 275 | 2 | 1 | 1 | 0.6 | E2 | 2013 | golimumab, steroids, AZA, 5-ASA |
| UC18 | 254 | 5 | 2 | 1 | 0.5 | E2 | 1994 | infliximab |
| UC19 | 253 | 5 | 1 | 0 | 1 | E3 | NA | vedolizumab, 5-ASA |
| UC20 | 232 | 2 | 0 | 0 | 0.6 | E2 | NA | adalumimab |
| CD1 | 323 | 1 | 0 | 0 | 0.6 | L3B1 | 2011 | AZA |
| CD2 | 329 | 6 | 0 | 3 | 0.3 | L1B1 | 2016 | no medication |
| CD3 | 222 | 12 | 0 | 0 | 0.2 | L1B1 | 2016 | steroids, 5-ASA |
| CD4 | 257 | 2 | 0 | 0 | 0.4 | L1B1 | 2006 | AZA |
| CD5 | NA | NA | 0 | 0 | NA | L3B1 | 2008 | AZA |
| CD6 | 211 | 3 | 0 | 0 | 0.8 | L3B1 | 1998 | AZA |
| CD7 | NA | 0 | 0 | NA | L2B1 | 1989 | No medication | |
| CD8 | 209 | 4 | 0 | 0 | 1.3 | L1B1 | 2016 | AZA, 5-ASA |
| CD9 | 312 | 3 | 0 | 0 | 0.5 | L1B1 | 1997 | AZA, 5-ASA |
| CD10 | 335 | 5 | 0 | 1 | 0.6 | L1B2 | 2006 | steroids per os/enema |
| CD11 | 279 | 2 | 0 | 0 | 0.8 | L1B1 | 2010 | AZA |
| CD12 | 258 | 7 | 0 | 0 | 0.9 | L1B1 | 2015 | AZA, 5-ASA |
| CD13 | 409 | 27 | 0 | 0 | 0.4 | L4B1 | 2017 | steroids |
| CD14 | 166 | 6 | 1 | 0 | 1 | L2B1 | 1974 | 5-ASA, steroids enema |
| CD15 | NA | 0 | 0 | NA | L2B1 | 2013 | AZA, 5-ASA | |
| CD16 | 455 | 20 | 2 | 2 | 0.3 | L3B3 | NA | no medication |
| CD17 | 326 | 26 | 1 | 0 | 0.3 | L1B1 | 2002 | no medication |
| CD18 | NA | NA | NA | NA | NA | NA | NA | NA |
| CD19 | NA | NA | NA | NA | NA | NA | NA | NA |
| CD20 | NA | NA | NA | NA | NA | NA | NA | NA |
| UC1 | NA | NA | 0 | 0 | 0.15 | E3 | 1993 | methotrexate, 5-ASA |
| UC2 | NA | NA | 0 | 1 | 1.4 | E2 | 2015 | 5-ASA, per os/sus |
| UC3 | 219 | 2 | 0 | 0 | 1.3 | E3 | 2007 | No medication |
| UC4 | 181 | 3 | 0 | 0 | 0.5 | E2 | 2013 | AZA, 5-ASA |
| UC5 | NA | NA | 0 | 0 | ΝΑ | E3 | 2004 | 5-ASA per os/sus |
| UC6 | 457 | 2 | 2 | 1 | 0.5 | E3 | 1990 | 5-ASA per os/sus, steroids |
| UC7 | NA | NA | 1 | 0 | ΝΑ | E2 | 2010 | 5-ASA |
| UC8 | 346 | 5 | 2 | 1 | 0.4 | E3 | 2016 | 5-ASA |
| UC9 | 319 | 13 | 0 | 1 | 0.5 | E2 | 2016 | 5-ASA |
| UC10 | 224 | 3 | 0 | 0 | 1 | E2 | 2017 | 5-ASA per os/sus |
| UC11 | 255 | 2 | 1 | 2 | 0.6 | E3 | 2011 | 5-ASA |
| UC12 | NA | NA | 0 | 0 | NA | E2 | 2000 | NA |
| UC13 | NA | NA | 0 | 0 | NA | E3 | 2009 | no medication |
| UC14 | NA | NA | 0 | 0 | E3 | 2017 | NA | |
| UC15 | 242 | 3 | 0 | 0 | 0.4 | E2 | 2017 | AZA, steroids, 5-ASA |
| UC16 | 279 | 5 | 0 | 1 | 0.5 | E3 | 1997 | AZA, 5-ASA |
| UC17 | 201 | NA | 0 | 1 | 0.4 | E3 | 2007 | no medication |
| UC18 | 401 | 7 | 0 | 0 | 0.6 | E3 | 1989 | methotrexate, steroids |
| UC19 | 199 | 37 | 3 | 3 | 0.4 | E3 | 2013 | 5-ASA (oral and topical), steroids |
| UC20 | 285 | 4 | 1 | 0 | 0.5 | E2 | 2007 | 5-ASA (oral and topical) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roda, G.; Porru, E.; Katsanos, K.; Skamnelos, A.; Kyriakidi, K.; Fiorino, G.; Christodoulou, D.; Danese, S.; Roda, A. Serum Bile Acids Profiling in Inflammatory Bowel Disease Patients Treated with Anti-TNFs. Cells 2019, 8, 817. https://doi.org/10.3390/cells8080817
Roda G, Porru E, Katsanos K, Skamnelos A, Kyriakidi K, Fiorino G, Christodoulou D, Danese S, Roda A. Serum Bile Acids Profiling in Inflammatory Bowel Disease Patients Treated with Anti-TNFs. Cells. 2019; 8(8):817. https://doi.org/10.3390/cells8080817
Chicago/Turabian StyleRoda, Giulia, Emanuele Porru, Konstantinos Katsanos, Alexandros Skamnelos, Kallirroi Kyriakidi, Gionata Fiorino, Dimitrios Christodoulou, Silvio Danese, and Aldo Roda. 2019. "Serum Bile Acids Profiling in Inflammatory Bowel Disease Patients Treated with Anti-TNFs" Cells 8, no. 8: 817. https://doi.org/10.3390/cells8080817






