To See or Not to See: A Systematic Review of the Importance of Human Ocular Surface Cytokine Biosignatures in Ocular Allergy
Abstract
1. Introduction
2. IgE-Mediated Mechanism of Ocular Allergy
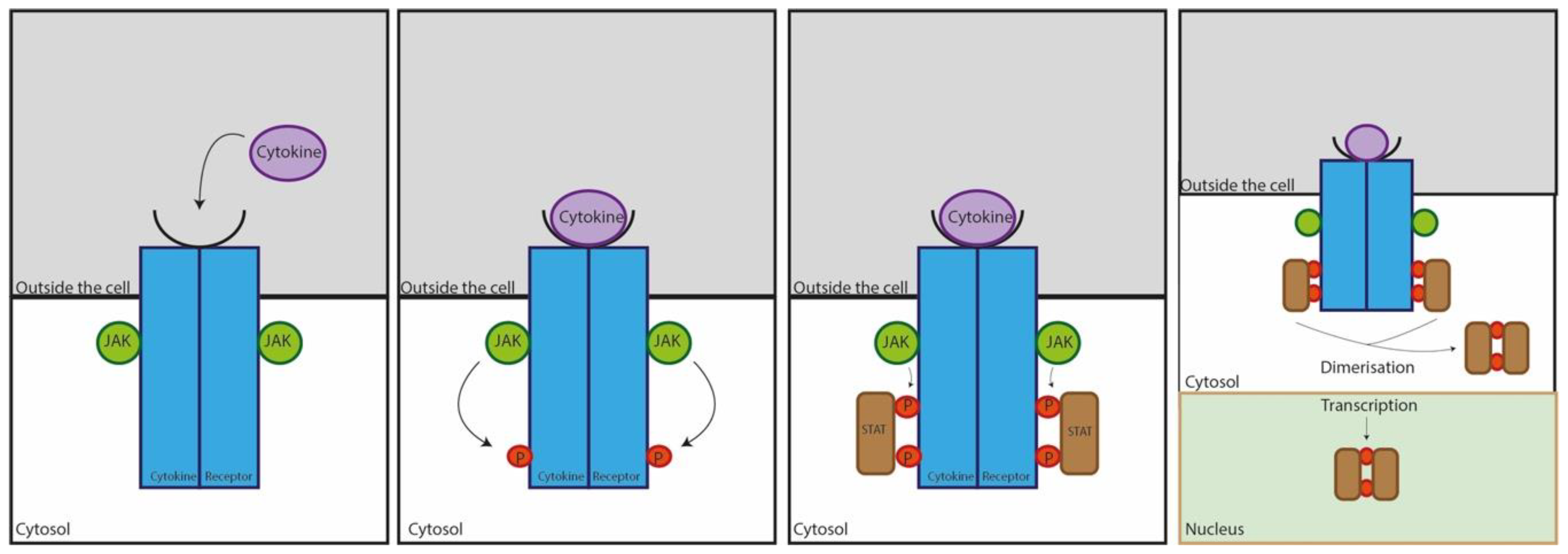
3. Overview of Cytokines in Ocular Allergy
4. Physiology of Ocular Allergy
5. Tear Cytokine Analysis Methods
5.1. Immunoblotting
5.2. ELISA and Multiplex Assays
6. Results of Previous Ocular Allergy Studies
6.1. Normal Tear Cytokine Levels
6.2. Cytokines as Biomarkers
7. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Pawankar, R.; Canonica, G.W.; Holgate, S.T.; Lockey, R.F. White Book on Allergy 2011–2012 Executive Summary; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- Asher, M.I.; Strachan, D.P.; Pearce, N.; García-Marcos, L. The ISAAC Story: The International Study of Asthma and Allergies in Childhood; The International Study of Asthma and Allergies in Childhood: Auckland, New Zealand, 2011. [Google Scholar]
- EAACI. Tackling the Allergy Crisis in Europe-Concerted Policy Action Needed; EAACI: Brussels, Belgium, 2016. [Google Scholar]
- AIHW. Allergic Rhinitis (‘Hay Fever’) in Australia; Australian Government: Canberra, Australia, 2011.
- Leonardi, A.; Doan, S.; Fauquert, J.L.; Bozkurt, B.; Allegri, P.; Marmouz, F.; Rondon, C.; Jedrzejczak, M.; Hellings, P.; Delgado, L.; et al. Diagnostic tools in ocular allergy. Allergy 2017, 72, 1485–1498. [Google Scholar] [CrossRef] [PubMed]
- Burge, H.A.; Rogers, C.A. Outdoor allergens. Environ. Health Perspect. 2000, 108, 653–659. [Google Scholar] [PubMed]
- Mimura, T.; Yamagami, S.; Noma, H.; Kamei, Y.; Goto, M.; Kondo, A.; Matsubara, M. Specific IgE for wheat in tear fluid of patients with allergic conjunctivitis. Cutaneous Ocul. Toxicol. 2015, 34, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Laboratory Tests in the Diagnosis of Allergic Diseases; ASCIA: Balgowla, Australia, 2010.
- Sicherer, S.H.; Wood, R.A. Allergy testing in childhood: Using allergen-specific IgE tests. Pediatrics 2012, 129, 193–197. [Google Scholar] [CrossRef] [PubMed]
- Williams, P.; Barnes, J.H.; Szeinbach, S.L.; Sullivan, T.J. Analytic precision and accuracy of commercial immunoassays for specific IgE: Establishing a standard. J. Allergy Clin. Immunol. 2000, 105, 1221–1230. [Google Scholar] [CrossRef] [PubMed]
- Leonardi, A.; Motterle, L.; Bortolotti, M. Allergy and the eye. Clin. Exp. immunol. 2008, 153, 17–21. [Google Scholar] [CrossRef] [PubMed]
- McMonnies, C.W. Eye rubbing type and prevalence including contact lens ‘removal-relief’ rubbing. Clin. Exp. Optom. 2016, 99, 366–372. [Google Scholar] [CrossRef]
- Yamamoto, T.; Kita, M.; Kimura, I.; Oseko, F.; Terauchi, R.; Takahashi, K.; Kubo, T.; Kanamura, N. Mechanical stress induces expression of cytokines in human periodontal ligament cells. Oral Dis. 2006, 12, 171–175. [Google Scholar] [CrossRef]
- Smith, A.F.; Pitt, A.D.; Rodruiguez, A.E.; Alio, J.L.; Marti, N.; Teus, M.; Guillen, S.; Bataille, L.; Barnes, J.R. The Economic and Quality of Life Impact of Seasonal Allergic Conjunctivitis in a Spanish Setting. Ophthalmic Epidemiol. 2005, 12, 233–242. [Google Scholar] [CrossRef]
- Klossek, J.; Annesi-Maesano, I.; Pribil, C.; Didier, A. The Burden Associated with Ocular Symptoms in Allergic Rhinitis. Int. Arch. Allergy Immunol. 2012, 158, 411–417. [Google Scholar] [CrossRef]
- Leonardi, A. Allergy and allergic mediators in tears. Exp. Eye Res. 2013, 117, 106–117. [Google Scholar] [CrossRef] [PubMed]
- Bielory, L. Allergic conjunctivitis: The evolution of therapeutic options. Allergy Asthma Proc. 2012, 33, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Leonardi, A.; Bogacka, E.; Fauquert, J.L.; Kowalski, M.L.; Groblewska, A.; Jedrzejczak-Czechowicz, M.; Doan, S.; Marmouz, F.; Demoly, P.; Delgado, L. Ocular allergy: Recognizing and diagnosing hypersensitivity disorders of the ocular surface. Allergy 2012, 67, 1327–1337. [Google Scholar] [CrossRef] [PubMed]
- Jalbert, I.; McOptom, F.S. Effect of lens wear on corneal stroma: Preliminary findings. Aust. New Zealand J. Ophthalmol. 1999, 27, 211–213. [Google Scholar] [CrossRef]
- Bielory, L. Allergic and immunologic disorders of the eye. Part II: Ocular allergy. J. Allergy Clin. Immunol. 2000, 106, 1019–1032. [Google Scholar] [CrossRef] [PubMed]
- Delves, P.J.; Roitt, I.M. Roitt’s Essential Immunology; John Wiley & Sons, Inc.: Chichester, UK, 2015. [Google Scholar]
- Secrist, H.; Chelen, C.J.; Wen, Y.; Marshall, J.D.; Umetsu, D.T. Allergen immunotherapy decreases interleukin 4 production in CD4+ T cells from allergic individuals. J. Exp. Med. 1993, 178, 2123–2130. [Google Scholar] [CrossRef]
- Bonini, S.; Lambiase, A.; Sacchetti, M.; Bonini, S. Cytokines in ocular allergy. Int. Ophthalmol. Clin. 2003, 43, 27–32. [Google Scholar] [CrossRef]
- Masi, A.; Glozier, N.; Dale, R.; Guastella, A.J. The Immune System, Cytokines, and Biomarkers in Autism Spectrum Disorder. Neurosci. Bull. 2017, 33, 194–204. [Google Scholar] [CrossRef]
- Cohen, M.C. Cytokine Function: A Study in Biologic Diversity. Am. J. Clin. Pathol. 1996, 105, 589–598. [Google Scholar] [CrossRef]
- O’Sullivan, L.A.; Liongue, C.; Lewis, R.S.; Stephenson, S.E.; Ward, A.C. Cytokine receptor signaling through the Jak–Stat–Socs pathway in disease. Mol. Immunol. 2007, 44, 2497–2506. [Google Scholar] [CrossRef]
- Liongue, C.; Ward, A.C. Evolution of the JAK-STAT pathway. JAKSTAT 2013, 2, e22756. [Google Scholar] [CrossRef] [PubMed]
- Bao, K.; Reinhardt, R.L. The differential expression of IL-4 and IL-13 and its impact on type-2 immunity. Cytokine 2015, 75, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, L.K.; Fonseca, B.P.A.; Barboza, B.; Viola, J.P. The role of interferon-gamma on immune and allergic responses. Memórias do Instituto Oswaldo Cruz 2005, 100, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Broide, D.H. Cytokines, Chemokines, and Adhesion Proteins; Elsevier: Amsterdam, Netherlands, 2006. [Google Scholar]
- Liu, Y.; Shao, Z.; Shangguan, G.; Bie, Q.; Zhang, B. Biological Properties and the Role of IL-25 in Disease Pathogenesis. J. Immunol. Res. 2018, 2018, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Thompson, S.R.; Humphries, S. Interleukin-18 genetics and inflammatory disease susceptibility. Genes Immun. 2007, 8, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Ryan, D.M.; Vincent, T.L.; Salit, J.; Walters, M.S.; Agosto-Perez, F.; Shaykhiev, R.; Strulovici-Barel, Y.; Downey, R.J.; Buro-Auriemma, L.J.; Staudt, M.R.; et al. Smoking Dysregulates the Human Airway Basal Cell Transcriptome at COPD Risk Locus 19q13.2. PLoS ONE 2014, 9, e88051. [Google Scholar] [CrossRef] [PubMed]
- Schultz, C.L.; Kunert, K.S. Interleukin-6 Levels in Tears of Contact Lens Wearers. J. Interf. Cytokine Res. 2000, 20, 309–310. [Google Scholar] [CrossRef] [PubMed]
- Bio, G. Immunoassays: Protein Arrays vs. ELISA and Westerns; Grace Bio-Labs: Bend, OR, USA, 2013. [Google Scholar]
- Carreño, E.; Enríquez-De-Salamanca, A.; Tesón, M.; García-Vázquez, C.; Stern, M.E.; Whitcup, S.M.; Calonge, M. Cytokine and chemokine levels in tears from healthy subjects. Acta Ophthalmol. 2010, 88, e250–e258. [Google Scholar] [CrossRef] [PubMed]
- Al-Shamahy, H.A.; Wright, S.G. Enzyme-linked immunosorbent assay for brucella antigen detection in human sera. J. Med Microbiol. 1998, 47, 169–172. [Google Scholar] [CrossRef]
- González-Pérez, J.; Villa-Collar, C.; Moreiras, T.S.; Gesto, I.L.; Parafita, M.; González-Méijome, J.M.; Rodríguez-Ares, M.T. Tear film inflammatory mediators during continuous wear of contact lenses and corneal refractive therapy. Br. J. Ophthalmol. 2012, 96, 1092–1098. [Google Scholar] [CrossRef]
- Sonoda, S.; Uchino, E.; Nakao, K.; Sakamoto, T. Inflammatory cytokine of basal and reflex tears analysed by multicytokine assay. Br. J. Ophthalmol. 2006, 90, 120–122. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yüksel-Elgin, C.; İskeleli, G.; Talaz, S.; Akyol, S. Comparative analysis of tear film levels of inflammatory mediators in contact lens users. Curr. Eye Res. 2016, 41, 441–447. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, S.; Miller, W.L.; McDermott, A.M. Conjunctival Cytokine Expression in Symptomatic Moderate Dry Eye Subjects. Investig. Opthalmol. Vis. Sci. 2006, 47, 2445–2450. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cook, E.; Stahl, J.; Lowe, L.; Chen, R.; Morgan, E.; Wilson, J.; Varró, R.; Chan, A.; Graziano, F.; Barney, N. Simultaneous measurement of six cytokines in a single sample of human tears using microparticle-based flow cytometry: Allergics vs. non-allergics. J. Immunol. Methods 2001, 254, 109–118. [Google Scholar] [CrossRef]
- Yucekul, B.; Mocan, M.C.; Kocabeyoglu, S.; Tan, C.; Irkec, M. Evaluation of Long-Term Silicone Hydrogel Use on Ocular Surface Inflammation and Tear Function in Patients With and Without Meibomian Gland Dysfunction. Eye Contact Lens Sci. Clin. Pr. 2019, 45, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Muhafiz, E.; Bayhan, H.A.; Şahin, S.; Göçmen, A.Y.; Bayhan, S.A.; Gürdal, C. Evaluation of the Ocular Surface in Different Contact Lens Replacement Schedules. Cornea 2019, 38, 587–594. [Google Scholar] [CrossRef] [PubMed]
- Kothur, K.; Wienholt, L.; Brilot, F.; Dale, R.C. CSF cytokines/chemokines as biomarkers in neuroinflammatory CNS disorders: A systematic review. Cytokine 2016, 77, 227–237. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Sinha, S.; Goyal, M. Diagnosis of human tuberculosis: Identification of new biomarker(s) and biosignature(s). Int. J. Infect. Dis. 2016, 45, 413. [Google Scholar] [CrossRef]
- Konradsen, J.R.; Nordlund, B.; Levänen, B.; Hedlin, G.; Lindén, A. The cytokine interleukin-26 as a biomarker in pediatric asthma. Respir. Res. 2016, 17, 974. [Google Scholar] [CrossRef]
- Ionescu, C.; Corbu, C.G.; Tanase, C.; Jonescu-Cuypers, C.; Nicula, C.; Dascalescu, D.; Cristea, M.; Voinea, L.-M. Inflammatory Biomarkers Profile as Microenvironmental Expression in Keratoconus. Dis. Markers 2016, 2016, 1–8. [Google Scholar] [CrossRef]
- Monastero, R.N.; Pentyala, S. Cytokines as Biomarkers and Their Respective Clinical Cutoff Levels. Int. J. Inflamm. 2017, 2017, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Piyadasa, H.; Altieri, A.; Basu, S.; Schwartz, J.; Halayko, A.J.; Mookherjee, N. Biosignature for airway inflammation in a house dust mite-challenged murine model of allergic asthma. Boil. Open 2016, 5, 112–121. [Google Scholar] [CrossRef] [PubMed]
- Leonardi, A.; Curnow, S.J.; Zhan, H.; Calder, V.L. Multiple cytokines in human tear specimens in seasonal and chronic allergic eye disease and in conjunctival fibroblast cultures. Clin. Exp. Allergy 2006, 36, 777–784. [Google Scholar] [CrossRef] [PubMed]
- Cook, E.B. Tear cytokines in acute and chronic ocular allergic inflammation. Curr. Opin. Allergy Clin. Immunol. 2004, 4, 441–445. [Google Scholar] [CrossRef] [PubMed]
- Boehm, N.; Riechardt, A.I.; Wiegand, M.; Pfeiffer, N.; Grus, F.H. Proinflammatory Cytokine Profiling of Tears from Dry Eye Patients by Means of Antibody Microarrays. Investig. Opthalmol. Vis. Sci. 2011, 52, 7725–7730. [Google Scholar] [CrossRef] [PubMed]
- Enríquez-De-Salamanca, A.; Calonge, M. Cytokines and chemokines in immune-based ocular surface inflammation. Expert Rev. Clin. Immunol. 2008, 4, 457–467. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Li, Q.; Ye, M.; Yu, J. Tear Luminex Analysis in Dry Eye Patients. Med Sci. Monit. 2018, 24, 7595–7602. [Google Scholar] [CrossRef] [PubMed]
- Di Zazzo, A.; Micera, A.; De Piano, M.; Cortes, M.; Bonini, S. Tears and ocular surface disorders: Usefulness of biomarkers. J. Cell. Physiol. 2019, 234, 9982–9993. [Google Scholar] [CrossRef]
- Willcox, M.D.P. Is There a Role for Inflammation in Contact Lens Discomfort? Eye Contact Lens Sci. Clin. Pr. 2017, 43, 5–16. [Google Scholar] [CrossRef]
- Ghasemi, H.; Ghazanfari, T.; Yaraee, R.; Owlia, P.; Hassan, Z.M.; Faghihzadeh, S. Roles of IL-10 in Ocular Inflammations: A Review. Ocul. Immunol. Inflamm. 2012, 20, 406–418. [Google Scholar] [CrossRef]
- Posa, A.; Bräuer, L.; Schicht, M.; Garreis, F.; Beileke, S.; Paulsen, F. Schirmer strip vs. capillary tube method: Non-invasive methods of obtaining proteins from tear fluid. Ann. Anat. Anat. Anz. 2013, 195, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Kornman, K.S.; Di Giovine, F.S. Genetic Variations in Cytokine Expression: A Risk Factor for Severity of Adult Periodontitis. Ann. Periodontol. 1998, 3, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Rohleder, N.; Kudielka, B.M.; Hellhammer, D.H.; Wolf, J.M.; Kirschbaum, C. Age and sex steroid-related changes in glucocorticoid sensitivity of pro-inflammatory cytokine production after psychosocial stress. J. Neuroimmunol. 2002, 126, 69–77. [Google Scholar] [CrossRef]
- Szűcs, D.; Ioannidis, J.P.A. Empirical assessment of published effect sizes and power in the recent cognitive neuroscience and psychology literature. PLoS Boil. 2017, 15, e2000797. [Google Scholar] [CrossRef] [PubMed]
- Willcox, M.D.; Zhao, Z.; Naduvilath, T.; De La Jara, P.L. Cytokine changes in tears and relationship to contact lens discomfort. Mol. Vis. 2015, 21, 293–305. [Google Scholar] [PubMed]
- Ahmed, I.; Sutton, A.J.; Riley, R.D. Assessment of publication bias, selection bias, and unavailable data in meta-analyses using individual participant data: A database survey. BMJ 2012, 344, d7762. [Google Scholar] [CrossRef] [PubMed]
- Carlson, N.B.; Kurtz, D. Clinical Procedures for Ocular Examination; McGraw-Hill Education: New York, NY, USA, 2003. [Google Scholar]
- Stern, M.E.; Siemasko, K.F.; Niederkorn, J.Y. The Th1/Th2 paradigm in ocular allergy. Curr. Opin. Allergy Clin. Immunol. 2005, 5, 446–450. [Google Scholar] [CrossRef]
- Nelson, H.S. The Pharmacotherapy of Rhinitis, Conjunctivitis and Asthma. In Textbook of Allergy for the Clinician; CRC Press: Boca Raton, FL, USA, 2016. [Google Scholar]
- Durham, S.R.; Penagos, M. Sublingual or subcutaneous immunotherapy for allergic rhinitis? J. Allergy Clin. Immunol. 2016, 137, 339–349.e10. [Google Scholar] [CrossRef]
- Rondón, C.; Blanca-López, N.; Campo, P.; Mayorga, C.; Jurado-Escobar, R.; Torres, M.J.; Canto, G.; Blanca, M. Specific immunotherapy in local allergic rhinitis: A randomized, double-blind placebo-controlled trial with Phleum pratense subcutaneous allergen immunotherapy. Allergy 2018, 73, 905–915. [Google Scholar] [CrossRef]
- Chassin, H.; Geering, B.; Schukur, L.; Ausländer, D.; Lang, B.; Fussenegger, M. Sensing and responding to allergic response cytokines through a genetically encoded circuit. Nat. Commun. 2017, 8, 1101. [Google Scholar] [CrossRef]
- Lawrence, M.G.; Steinke, J.W.; Borish, L. Cytokine-targeting biologics for allergic diseases. Ann. Allergy Asthma Immunol. 2018, 120, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Bahn, R.S. Cytokines in Thyroid Eye Disease: Potential for Anticytokine Therapy. Thyroid 1998, 8, 415–418. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Fragala, M.S.; McElhaney, J.E.; Kuchel, G.A. Conceptual and methodological issues relevant to cytokine and inflammatory marker measurements in clinical research. Curr. Opin. Clin. Nutr. Metab. Care 2010, 13, 541–547. [Google Scholar] [CrossRef] [PubMed]
- Solomon, A. Allergic manifestations of contact lens wearing. Curr. Opin. Allergy Clin. Immunol. 2016, 16, 492–497. [Google Scholar] [CrossRef]
- Binder, P.S.; Lindstrom, R.L.; Stulting, R.D.; Donnenfeld, E.; Wu, H.; McDonnell, P.; Rabinowitz, Y. Keratoconus and corneal ectasia after LASIK. J. Cataract. Refract. Surg. 2005, 31, 2035–2038. [Google Scholar] [CrossRef]
| SAC | PAC | VKC | AKC | |
|---|---|---|---|---|
| Presentation | Intermittent | Persistent | Intermittent and persistent phases | Chronic |
| Allergic mechanism | IgE-mediated | IgE-mediated | IgE and non-IgE mediated | IgE and non-IgE mediated |
| Cell types involved | Mast cells | Mast cells | Mast cells, eosinophils, lymphocytes, basophils, plasma cells, and macrophages | Mast cells, eosinophils, and lymphocytes |
| Background | Atopic | Atopic | Childhood and/or atopic | Atopic |
| Eyelids | Oedema | Oedema | Oedema, pseudoptosis (saggy eyelid) | Eczema, meibomitis (tear gland inflammation), blepharitis (oil gland inflammation) |
| Conjunctiva | Follicles and/or papillae | Follicles and/or papillae | Giant papillae | Papillae and/or fibrosis |
| Limbus | No effect | No effect | Thickening, Trantas dots | Thickening, Trantas dots |
| Cornea | No effect | No effect | Ulcer, vernal plaques | Ulcer, vernal plaque, opacities, neovascularization |
| Discharge | Clear mucoid | Clear mucoid | Stringy mucoid | Stringy mucoid |
| Symptoms | Watery eyes, itchy eyes, inflammation, discomfort, conjunctivitis, swollen eyelids, blurred vision | Watery eyes, itchy eyes, inflammation, discomfort, conjunctivitis, swollen eyelids, blurred vision | Itchy eyes, irritation, discomfort, conjunctivitis, photosensitivity, papillae | Itchy eyes, irritation, discomfort, conjunctivitis, swollen eyelids, papillae, photosensitivity |
| Cytokine | Actions |
|---|---|
| IL-1β | Induces fever Proinflammatory Stimulates synthesis of other cytokines |
| IL-2 | Promotes allergic reactions |
| IL-4 | Upregulates allergic reaction mediator cells |
| IL-5 | Increases proliferation of allergic B cells in eosinophil-mediated ocular allergy |
| IL-6 | Promotes allergic cell differentiation |
| IL-8 | Chemotactic |
| IL-10 | Inhibition of allergic responses Downregulation of inflammatory cytokines |
| IL-12 | Supresses allergy Upregulates IFNγ |
| IL-13 | Promotes antibody production Upregulates chemotactic cytokines Inhibits cytokine secretion |
| TNFα | Chemotactic Increases cytokine secretion |
| IFNγ | Inhibits allergic reactions |
| Cytokine | Role within the Eye |
|---|---|
| IL-1 | Triggers production of IL-2, IL-6, IL-8, and TNFα Stimulates cellular proliferation |
| IL-2 | T cell activation and proliferation begins when this cytokine reaches the requisite concentration |
| IL-3 | Growth and proliferation of pluripotent stem cells to replace old or damaged cells |
| IL-4 | Starts off the Th2 allergic response |
| IL-6 | Cellular growth Production of antibodies |
| IL-8 | Chemotaxis—chemical gradient attraction of cells from one location to another |
| IL-10 | Inhibits cytokine synthesis to stop a cellular reaction |
| TNFα | Activates T-cells Stimulates IL-1 and IL-6 synthesis |
| Source | SAC | PAC | VKC | AKC |
|---|---|---|---|---|
| Leonardi, Motterle and Bortolotti, 2008 [11] | - | - | IL-8, IFNγ, IL-4, IL-13 | - |
| Leonardi, 2013 [16] | IL-4, IL-5, IL-13, IL-1β | - | IL-4, IL-5, IL-10, IL-12, IL-13, IFNγ, IL-1β, IL-6, TNFα | IL-2, IL-4, IL-5, IL-10, IFNγ, IL-1β, IL-6, TNFα, IL-8 |
| Bonini et al., 2003 [23] | IL-2, IL-4, IL-5, IL-10, TNFα, IFNγ | IL-2, IL-4, IL-5, IL-10, IFNγ | IL-1β, IL-6 | - |
| Leonardi et al., 2006 [51] | IL-1β, IL-2, IL-4, IL-5, IL-6, IL-10, IL-12, IL-13, IFNγ, | - | IL-1β, IL-2, IL-4, IL-5, IL-6, IL-10, IL-12, IL-13, IFNγ, TNFα | IL-1β, IL-2, IL-5, IL-6, IL-12, IL-13 |
| Cook, 2004 [52] | IL-1β | - | IL-4, IL-5, IL-10, IL-12, IL-13, IFNγ, IL-1β, IL-6, TNFα | IL-2, IL-4, IL-5, IL-10, IFNγ, IL-1β, IL-6, TNFα,IL-8 |
| Enriquez-de-Salamanca and Calonge, 2008 [54] | IL-1β, IL-2, IL-4, IL-5, IL-6, IL-12, IL-13, IFNγ | - | IL-1β, IL-2, IL-4, IL-5, IL-6, IL-12, IL-13, IFNγ, TNFα | IL-1β, IL-2, IL-4, IL-5, IL-6, IL-10, IL- 12, IL-13, IFNγ, TNFα |
| Di Zazzo et al., 2017 [56] | - | - | IL-4, IL-5, TGF-β1, IL-1β, IL-6, TNFα | IL-4, IL-5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aydin, E.; Gokhale, M.; Azizoglu, S.; Suphioglu, C. To See or Not to See: A Systematic Review of the Importance of Human Ocular Surface Cytokine Biosignatures in Ocular Allergy. Cells 2019, 8, 620. https://doi.org/10.3390/cells8060620
Aydin E, Gokhale M, Azizoglu S, Suphioglu C. To See or Not to See: A Systematic Review of the Importance of Human Ocular Surface Cytokine Biosignatures in Ocular Allergy. Cells. 2019; 8(6):620. https://doi.org/10.3390/cells8060620
Chicago/Turabian StyleAydin, Esrin, Moneisha Gokhale, Serap Azizoglu, and Cenk Suphioglu. 2019. "To See or Not to See: A Systematic Review of the Importance of Human Ocular Surface Cytokine Biosignatures in Ocular Allergy" Cells 8, no. 6: 620. https://doi.org/10.3390/cells8060620
APA StyleAydin, E., Gokhale, M., Azizoglu, S., & Suphioglu, C. (2019). To See or Not to See: A Systematic Review of the Importance of Human Ocular Surface Cytokine Biosignatures in Ocular Allergy. Cells, 8(6), 620. https://doi.org/10.3390/cells8060620





