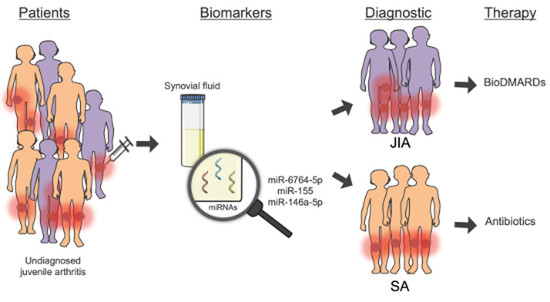
Synovial-Fluid miRNA Signature for Diagnosis of Juvenile Idiopathic Arthritis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Collection and Preparation of Synovial Fluid (SF) and Serum (SE) Samples for miRNA Studies
2.2. miRNA Profiling
2.2.1. HTG EdgeSeq Analysis
2.2.2. Molecular Barcoding and Adapter Addition
2.2.3. PCR Clean-Up
2.2.4. Determination of Library Concentration
2.2.5. Sample Pooling and Sequencing
2.2.6. Sequence Analysis
2.2.7. Normalization
2.3. miRNA Extraction and Quantification by RT-qPCR
2.4. Statistical Analysis
3. Results
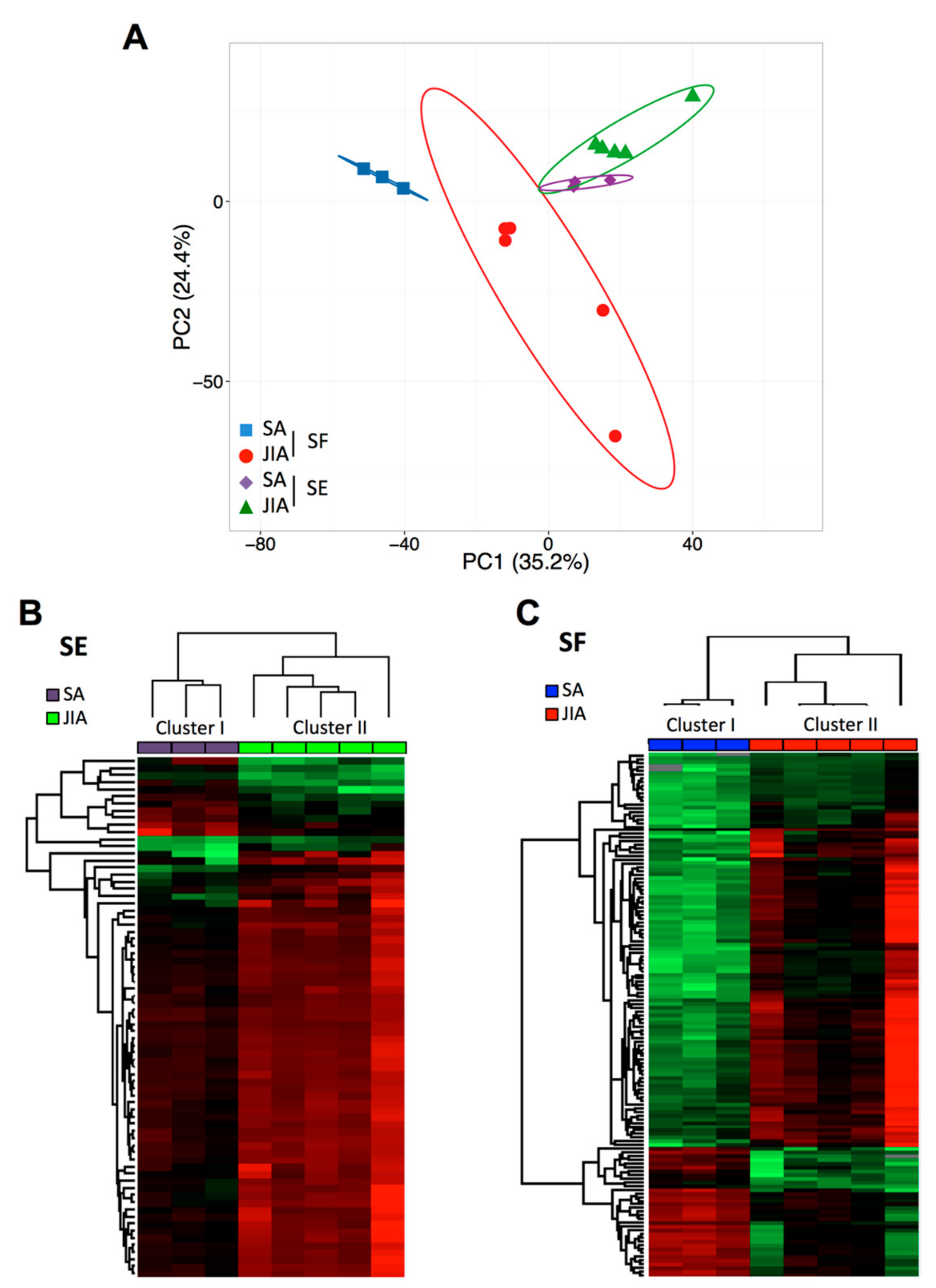
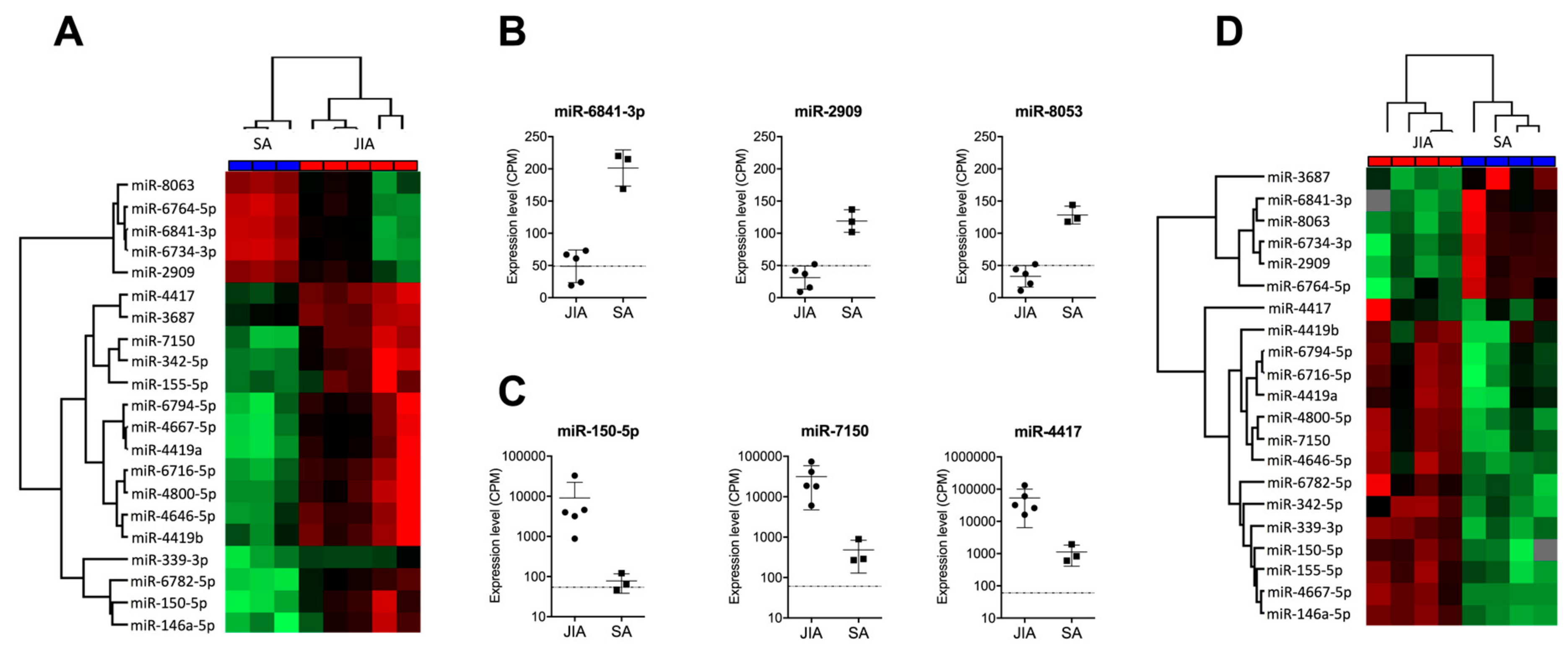
3.1. A Signature Based on 21 miRNAs in SF Distinguishes JIA from SA
3.2. An Independent Cohort Used to Validate the miRNA Signature Specific for JIA
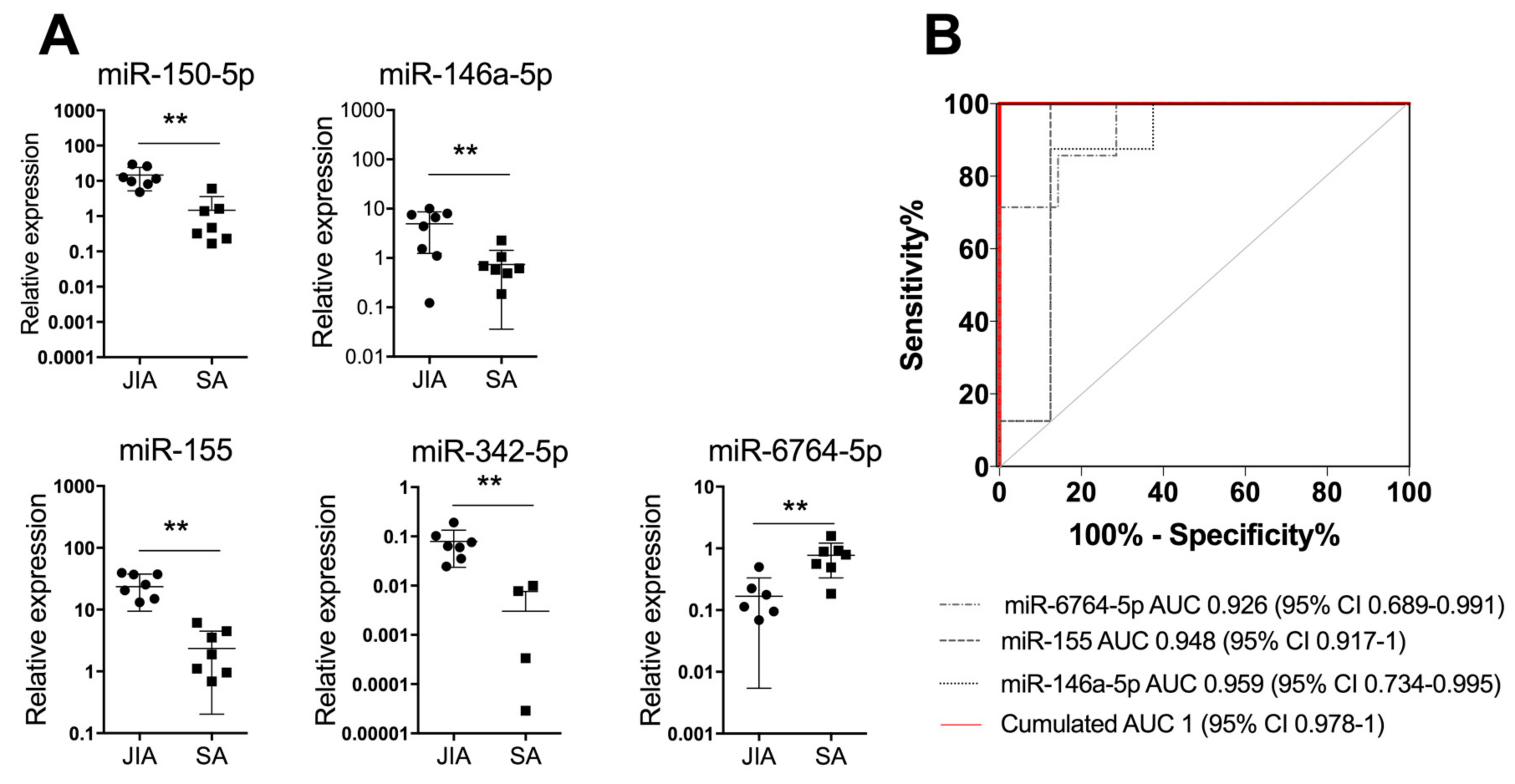
3.3. An SF Combination of Three miRNAs Differentiates JIA from SA at Initial Presentation
3.4. Comparative Pathway Analysis Reveals Key Pathophysiological Mechanisms
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Martini, A.; Ravelli, A.; Avcin, T.; Beresford, M.W.; Burgos-Vargas, R.; Cuttica, R.; Ilowite, N.T.; Khubchandani, R.; Laxer, R.M.; Lovell, D.J.; et al. Toward New Classification Criteria for Juvenile Idiopathic Arthritis: First Steps, Pediatric Rheumatology International Trials Organization International Consensus. J. Rheumatol. 2019, 46, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Giancane, G.; Alongi, A.; Ravelli, A. Update on the pathogenesis and treatment of juvenile idiopathic arthritis. Curr. Opin. Rheumatol. 2017, 29, 523–529. [Google Scholar] [CrossRef] [PubMed]
- Harris, J.G.; Kessler, E.A.; Verbsky, J.W. Update on the Treatment of Juvenile Idiopathic Arthritis. Curr. Allergy Asthma Rep. 2013, 13, 337–346. [Google Scholar] [CrossRef] [PubMed]
- Smolen, J.S.; Aletaha, D.; McInnes, I.B. Rheumatoid arthritis. Lancet 2016, 388, 2023–2038. [Google Scholar] [CrossRef]
- Duroux-Richard, I.; Jorgensen, C.; Apparailly, F. What do microRNAs mean for rheumatoid arthritis? Arthritis Rheum. 2012, 64, 11–20. [Google Scholar] [CrossRef]
- Nakasa, T.; Miyaki, S.; Okubo, A.; Hashimoto, M.; Nishida, K.; Ochi, M.; Asahara, H. Expression of microRNA-146 in rheumatoid arthritis synovial tissue. Arthritis Rheum. 2008, 58, 1284–1292. [Google Scholar] [CrossRef]
- Ono, S.; Lam, S.; Nagahara, M.; Hoon, D.S.B. Circulating microRNA Biomarkers as Liquid Biopsy for Cancer Patients: Pros and Cons of Current Assays. J. Clin. Med. 2015, 4, 1890–1907. [Google Scholar] [CrossRef]
- Stanczyk, J.; Pedrioli, D.M.L.; Brentano, F.; Sanchez-Pernaute, O.; Kolling, C.; Gay, R.E.; Kyburz, D. Altered expression of MicroRNA in synovial fibroblasts and synovial tissue in rheumatoid arthritis. Arthritis Rheum. 2008, 58, 1001–1009. [Google Scholar] [CrossRef]
- Vicente, R.; Noël, D.; Pers, Y.-M.; Apparailly, F.; Jorgensen, C. Deregulation and therapeutic potential of microRNAs in arthritic diseases. Nat. Rev. Rheumatol. 2016, 12, 211–220. [Google Scholar] [CrossRef]
- Li, D.; Duan, M.; Feng, Y.; Geng, L.; Li, X.; Zhang, W. MiR-146a modulates macrophage polarization in systemic juvenile idiopathic arthritis by targeting INHBA. Mol. Immunol. 2016, 77, 205–212. [Google Scholar] [CrossRef]
- Schulert, G.S.; Fall, N.; Harley, J.B.; Shen, N.; Lovell, D.J.; Thornton, S.; Grom, A.A. Monocyte MicroRNA Expression in Active Systemic Juvenile Idiopathic Arthritis Implicates MicroRNA-125a-5p in Polarized Monocyte Phenotypes. Arthritis Rheumatol. 2016, 68, 2300–2313. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Feng, M.; Wu, F.; Ma, X.; Lu, J.; Kang, M.; Liu, Z. Plasma miR-26a as a Diagnostic Biomarker Regulates Cytokine Expression in Systemic Juvenile Idiopathic Arthritis. J. Rheumatol. 2016, 43, 1607–1614. [Google Scholar] [CrossRef] [PubMed]
- Kamiya, Y.; Kawada, J.; Kawano, Y.; Torii, Y.; Kawabe, S.; Iwata, N.; Ito, Y. Serum microRNAs as Potential Biomarkers of Juvenile Idiopathic Arthritis. Clin. Rheumatol. 2015, 34, 1705–1712. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Wu, F.; Xin, L.; Su, G.; He, F.; Yang, Y.; Liu, Z. Differential plasma microRNAs expression in juvenile idiopathic arthritis. Mod. Rheumatol. 2015, 26, 1–9. [Google Scholar] [CrossRef]
- Singh, S.; Rai, G.; Aggarwal, A. Association of microRNA-146a and its target gene IRAK1 polymorphism with enthesitis related arthritis category of juvenile idiopathic arthritis. Rheumatol. Int. 2014, 34, 1395–1400. [Google Scholar] [CrossRef]
- Montgomery, N.I.; Epps, H.R. Pediatric Septic Arthritis. Orthop. Clin. N. Am. 2017, 48, 209–216. [Google Scholar] [CrossRef]
- El-Sayed, A.M.M. Treatment of early septic arthritis of the hip in children: Comparison of results of open arthrotomy versus arthroscopic drainage. J. Child. Orthop. 2008, 2, 229–237. [Google Scholar] [CrossRef]
- Dubnov-Raz, G.; Scheuerman, O.; Chodick, G.; Finkelstein, Y.; Samra, Z.; Garty, B.-Z. Invasive Kingella kingae infections in children: Clinical and laboratory characteristics. Pediatrics 2008, 122, 1305–1309. [Google Scholar] [CrossRef]
- Martinot, M.; Sordet, C.; Soubrier, M.; Puéchal, X.; Saraux, A.; Lioté, F.; Guggenbuhl, P.; Lègre, V.; Jaulhac, B.; Maillefert, J.-F.; et al. Diagnostic value of serum and synovial procalcitonin in acute arthritis: A prospective study of 42 patients. Clin. Exp. Rheumatol. 2005, 23, 303–310. [Google Scholar]
- Honda, T.; Uehara, T.; Matsumoto, G.; Arai, S. Sugano M: Neutrophil left shift and white blood cell count as markers of bacterial infection. Clin. Chim. Acta. 2016, 457, 46–53. [Google Scholar] [CrossRef]
- Bustin, S.A.; Benes, V.; Garson, J.A.; Hellemans, J.; Huggett, J.; Kubista, M.; Mueller, R.; Nolan, T.; Pfaffl, M.W.; Shipley, G.L.; et al. The MIQE guidelines: Minimum information for publication of quantitative real-time PCR experiments. Clin. Chem. 2009, 55, 611–622. [Google Scholar] [CrossRef] [PubMed]
- Tyanova, S.; Temu, T.; Sinitcyn, P.; Carlson, A.; Hein, M.Y.; Geiger, T.; Cox, J. The Perseus computational platform for comprehensive analysis of (prote)omics data. Nat. Methods 2016, 13, 731–740. [Google Scholar] [CrossRef] [PubMed]
- Faraggi, D.; Kramar, A. Methodological issues associated with tumor marker development: Biostatistical aspects. Urol. Oncol. 2000, 5, 211–213. [Google Scholar] [CrossRef]
- Kramar, A.; Faraggi, D.; Fortuné, A.; Reiser, B. mROC: A computer program for combining tumour markers in predicting disease states. Comput. Methods Programs Biomed. 2001, 66, 199–207. [Google Scholar] [CrossRef]
- Murata, K.; Yoshitomi, H.; Tanida, S.; Ishikawa, M.; Nishitani, K.; Ito, H.; Nakamura, T. Plasma and synovial fluid microRNAs as potential biomarkers of rheumatoid arthritis and osteoarthritis. Arthritis Res. Ther. 2010, 12, 86. [Google Scholar] [CrossRef] [PubMed]
- Pritchard, C.C.; Kroh, E.; Wood, B.; Arroyo, J.D.; Dougherty, K.J.; Miyaji, M.M.; Tewari, M. Blood cell origin of circulating microRNAs: A cautionary note for cancer biomarker studies. Cancer Prev. Res. 2012, 5, 492–497. [Google Scholar] [CrossRef]
- Monleau, M.; Bonnel, S.; Gostan, T.; Blanchard, D.; Courgnaud, V.; Lecellier, C.-H. Comparison of different extraction techniques to profile microRNAs from human sera and peripheral blood mononuclear cells. BMC Genom. 2014, 15, 395. [Google Scholar] [CrossRef]
- Kim, Y.-K.; Yeo, J.; Kim, B.; Ha, M.; Kim, V.N. Short structured RNAs with low GC content are selectively lost during extraction from a small number of cells. Mol. Cell 2012, 46, 893–895. [Google Scholar] [CrossRef]
- Rice, J.; Roberts, H.; Burton, J.; Pan, J.; States, V.; Rai, S.N.; Galandiuk, S. Assay reproducibility in clinical studies of plasma miRNA. PLoS ONE 2015, 10, e0121948. [Google Scholar] [CrossRef]
- Mestdagh, P.; Hartmann, N.; Baeriswyl, L.; Andreasen, D.; Bernard, N.; Chen, C.; Cheo, D.; D’Andrade, P.; DeMayo, M.; Dennis, L.; et al. Evaluation of quantitative miRNA expression platforms in the microRNA quality control (miRQC) study. Nat. Methods 2014, 11, 809–815. [Google Scholar] [CrossRef]
- Chen, J.-Q.; Papp, G.; Póliska, S.; Szabó, K.; Tarr, T.; Bálint, B.L.; Zeher, M. MicroRNA expression profiles identify disease-specific alterations in systemic lupus erythematosus and primary Sjögren’s syndrome. PLoS ONE 2017, 12, e0174585. [Google Scholar] [CrossRef] [PubMed]
- Hezova, R.; Slaby, O.; Faltejskova, P.; Mikulkova, Z.; Buresova, I.; Raja, K.R.M.; Michalek, J. microRNA-342, microRNA-191 and microRNA-510 are differentially expressed in T regulatory cells of type 1 diabetic patients. Cell. Immunol. 2010, 260, 70–74. [Google Scholar] [CrossRef] [PubMed]
- Niimoto, T.; Nakasa, T.; Ishikawa, M.; Okuhara, A.; Izumi, B.; Deie, M.; Ochi, M. MicroRNA-146a expresses in interleukin-17 producing T cells in rheumatoid arthritis patients. BMC Musculoskelet. Disord. 2010, 11, 209. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pauley, K.M.; Cha, S. miRNA-146a in rheumatoid arthritis: A new therapeutic strategy. Immunotherapy 2011, 3, 829–831. [Google Scholar] [CrossRef] [Green Version]
- Churov, A.V.; Oleinik, E.K.; Knip, M. MicroRNAs in rheumatoid arthritis: Altered expression and diagnostic potential. Autoimmun. Rev. 2015, 14, 1029–1037. [Google Scholar] [CrossRef]
- Lenert, A.; Fardo, D.W. Detecting novel micro RNAs in rheumatoid arthritis with gene-based association testing. Clin. Exp. Rheumatol. 2017, 35, 586–592. [Google Scholar]
- Malik, D.; Sharma, A.; Raina, A.; Kaul, D. Deregulated Blood Cellular miR-2909 RNomics Observed in Rheumatoid Arthritis Subjects. Arch. Med. 2015, 7, 1–6. [Google Scholar]
- Kaul, D.; Sasikala, M.; Raina, A. Regulatory role of miR-2909 in cell-mediated immune response. Cell Biochem. Funct. 2012, 30, 500–504. [Google Scholar] [CrossRef]
- Taganov, K.D.; Boldin, M.P.; Chang, K.-J.; Baltimore, D. NF-kappaB-dependent induction of microRNA miR-146, an inhibitor targeted to signaling proteins of innate immune responses. Proc. Natl. Acad. Sci. USA 2006, 103, 12481–12486. [Google Scholar] [CrossRef] [Green Version]
- Faraoni, I.; Antonetti, F.R.; Cardone, J.; Bonmassar, E. miR-155 gene: A typical multifunctional microRNA. Biochim. Biophys. Acta 2009, 1792, 497–505. [Google Scholar] [CrossRef]
- Wang, L.; Qin, Y.; Tong, L.; Wu, S.; Wang, Q.; Jiao, Q.; Zhong, Z. MiR-342-5p suppresses coxsackievirus B3 biosynthesis by targeting the 2C-coding region. Antivir. Res. 2012, 93, 270–279. [Google Scholar] [CrossRef] [PubMed]
- Vasilescu, C.; Rossi, S.; Shimizu, M.; Tudor, S.; Veronese, A.; Ferracin, M.; Calin, G.A. MicroRNA Fingerprints Identify miR-150 as a Plasma Prognostic Marker in Patients with Sepsis. PLoS ONE 2009, 4, e7405. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Fang, Y.; Zhu, P.; Ren, C.-Y.; Chen, H.; Gu, J.; Mao, X.-H. Burkholderia pseudomallei survival in lung epithelial cells benefits from miRNA-mediated suppression of ATG10. Autophagy 2015, 11, 1293–1307. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wong, S.C.; Dobie, R.; Altowati, M.A.; Werther, G.A.; Farquharson, C.; Ahmed, S.F. Growth and the Growth Hormone-Insulin Like Growth Factor 1 Axis in Children with Chronic. Dir. Endocr. Rev. 2016, 37, 62–110. [Google Scholar] [CrossRef] [PubMed]
- Wiegering, V.; Girschick, H.J.; Morbach, H. B-cell pathology in juvenile idiopathic arthritis. Arthritis 2010, 2010, 759868. [Google Scholar] [CrossRef] [Green Version]
- Barnes, M.G.; Grom, A.A.; Thompson, S.D.; Griffin, T.A.; Pavlidis, P.; Itert, L.; Colbert, R.A. Subtype-specific peripheral blood gene expression profiles in recent-onset juvenile idiopathic arthritis. Arthritis Rheum. 2009, 60, 2102–2112. [Google Scholar] [CrossRef]
- Koca, B.; Sahin, S.; Adrovic, A.; Barut, K.; Kasapcopur, O. Cardiac involvement in juvenile idiopathic arthritis. Rheumatol. Int. 2017, 37, 137–142. [Google Scholar] [CrossRef]
- Nicolau, J.; Lequerré, T.; Bacquet, H.; Vittecoq, O. Rheumatoid arthritis, insulin resistance, and diabetes. Jt. Bone Spine 2017, 84, 411–416. [Google Scholar] [CrossRef]
- Aupiais, C.; Basmaci, R.; Ilharreborde, B.; Blachier, A.; Desmarest, M.; Job-Deslandre, C.; Lorrot, M. Arthritis in children: Comparison of clinical and biological characteristics of septic arthritis and juvenile idiopathic arthritis. Arch. Dis. Child. 2017, 102, 316–322. [Google Scholar] [CrossRef]
- Basmaci, R.; Ilharreborde, B.; Bonacorsi, S.; Kahil, M.; Mallet, C.; Aupiais, C.; Doit, C.; Dugué, S.; Lorrot, M. Septic arthritis in children with normal initial C-reactive protein: Clinical and biological features. Arch. Pediatr. 2014, 21, 1195–1199. [Google Scholar] [CrossRef]
| Exploration Cohort | Validation Cohort | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Centrifuged | Non-Centrifuged | Centrifuged | |||||||
| JIA (n = 5) | SA (n = 3) | p Value | JIA (n = 4) | SA (n = 4) | p Value | JIA (n = 9) | SA (n = 9) | p Value | |
| Age at sampling (years) | 10.3 ± 3.7 | 1.2 ± 0.1 | 0.036 | 7.5 ± 3.9 | 1.7 ± 0.6 | 0.057 | 8.9 ± 3.7 | 1.2 ± 0.2 | 0.0002 |
| Number of affected joints % (n) | |||||||||
| Monoarthritis | 40 (2) | 100 (3) | / | 50 (2) | 100 (4) | / | 55.5 (5) | 100 (9) | / |
| Oligoarthritis | 60 (3) | 0 (0) | / | 50 (2) | 0 (0) | / | 45.5 (4) | 0 (0) | / |
| Affected joints, % (n) | |||||||||
| Knee | 100 (5) | 100 (3) | / | 4 | 1 | / | 100 (9) | 100 (9) | / |
| Hip | 0 (0) | / | 0 | 2 | / | 0 (0) | 0 (0) | / | |
| Ankle | 40 (2) | / | 0 | 0 | / | 22.2 (2) | 0 (0) | / | |
| Shoulder | 20 (1) | / | 0 | 1 | / | 0 (0) | 0 (0) | / | |
| Bacterial contamination, % (n) | |||||||||
| Kingella kingae | NA | 100 (3) | / | NA | 100 (4) | / | NA | 100 (9) | / |
| C- reactive protein level | 20.3 ± 33.5 | 23.9 ± 4.4 | ns | 5.3 | 48.0 ± 24.6 | NA | 13.6 ± 23.0 | 28.6 ± 15.6 | 0.042 |
| Blood WBC count (×109/L) | 6.8 ± 1.1 | 12.7 ± 1.8 | 0.018 | 4.4 | 10.1 ± 4.3 | NA | 7.1 ± 2.9 | 12.3 ± 3.6 | 0.002 |
| Neutrophil count (×109/L) | 3.8 ± 1.0 | 7.5 ± 1.6 | ns | NA | 5.5 ± 3.6 | NA | 4.0 ± 2.1 | 4.4 ± 3.0 | ns |
| Haemoglobin level (g/dL) | 11.9 ± 1.0 | 11.4 ± 0.9 | ns | NA | 11.1 ± 1 | NA | 11.9 ± 4.0 | 11.4 ± 4.0 | ns |
| Platelet count (×109/L) | 310 ± 65 | 368 ± 29 | ns | NA | 501 ± 176 | NA | 320 ± 119 | 385 ± 144 | ns |
| Centifuged | Non-Centrifuged | |||
|---|---|---|---|---|
| miRNA Name | log2 (FC) | p-Values | log2 (FC) | p-Values |
| miR-146a-5p | 5.08 | 0.010 | 6.90 | 8.8 × 10−6 |
| miR-150-5p | 5.95 | 0.002 | 6.08 | 0.0008 |
| miR-155-5p | 4.00 | 0.022 | 5.22 | 0.0007 |
| miR-2909 | −2.19 | 0.01 | −2.12 | 0.002 |
| miR-339-3p | 4.32 | 0.006 | 7.54 | 0.00008 |
| miR-342-5p | 4.57 | 0.004 | 2.25 | 0.001 |
| miR-3687 | 4.42 | 0.0007 | −1.13 | 0.012 |
| miR-4417 | 5.34 | 0.0005 | 0.42 | 0.395 |
| miR-4419a | 4.21 | 0.002 | 4.20 | 0.020 |
| miR-4419b | 4.19 | 0.005 | 1.68 | 0.105 |
| miR-4646-5p | 4.21 | 0.004 | 1.75 | 0.004 |
| miR-4667-5p | 4.01 | 0.005 | 3.29 | 0.00003 |
| miR-4800-5p | 4.83 | 0.005 | 2.83 | 0.003 |
| miR-6716-5p | 4.89 | 0.002 | 4.86 | 0.009 |
| miR-6734-3p | −2.09 | 0.005 | −1.58 | 0.003 |
| miR-6764-5p | −2.12 | 0.006 | −1.75 | 0.022 |
| miR-6782-5p | 4.76 | 0.0003 | 4.18 | 0.005 |
| miR-6794-5p | 4.16 | 0.004 | 3.37 | 0.010 |
| miR-6841-3p | −2.24 | 0.007 | −1.38 | 0.022 |
| miR-7150 | 5.79 | 0.0007 | 4.90 | 0.004 |
| miR-8063 | −2.14 | 0.009 | −2.37 | 0.002 |
| Path Name | No. of mRNAs | miRNA Names | FDR p-Value |
|---|---|---|---|
| Insulin signaling | 18 | miR-146a-5p, miR-150-5p, miR-155-5p, miR-2909, miR-339-3p, miR-342-5p, miR-4419a, miR-4419b, miR-4646-5p, miR-4667-5p, miR-6716-5p, miR-6734-3p, miR-6764-5p, miR-6782-5p, miR-6794-5p, miR-7150, miR-6841-5p, miR-8063 | 3.4 × 10−2 to 1.3 × 10−7 |
| B-cell receptor signaling | 16 | miR-146a-5p, miR-150-5p, miR-155-5p, miR-2909, miR-339-3p, miR-342-5p, miR-4419a, miR-4419b, miR-4646-5p, miR-4667-5p, miR-4800-5p, miR-6734-3p, miR-6782-5p, miR-6794-5p, miR-7150, miR-8063 | 4.8 × 10−2 to 6.5 × 10−6 |
| EGF EGFR signaling | 16 | miR-146a-5p, miR-150-5p, miR-155-5p, miR-2909, miR-339-3p, miR-342-5p, miR-4419a, miR-4419b, miR-4667-5p, miR-4800-5p, miR-6716-5p, miR-6734-3p, miR-6782-5p, miR-6794-5p, miR-7150, miR-8063 | 4.5 × 10−2 to 2.4 × 10−6 |
| Calcium regulation in cardiac cell | 15 | miR-150-5p, miR-2909, miR-342-5p, miR-4419a, miR-4419b, miR-4646-5p, miR-4667-5p, miR-4800-5p, miR-6716-5p, miR-6734-3p, miR-6764-5p, miR-6782-5p, miR-6794-5p, miR-7150, miR-8063 | 4.5 × 10−2 to 7.9 × 10−8 |
| DNA damage response only ATM-dependent | 15 | miR-146a-5p, miR-150-5p, miR-155-5p, miR-2909, miR-339-3p, miR-342-5p, miR-4419a, miR-4419b, miR-4646-5p, miR-4667-5p, miR-6716-5p, miR-6782-5p, miR-6794-5p, miR-7150, miR-8063 | 4.9 × 10−2 to 8.1 × 10−5 |
| MiRNAs in cardiomyocyte hypertrophy | 15 | miR-146a-5p, miR-150-5p, miR-155-5p, miR-2909, miR-342-5p, miR-4419a, miR-4419b, miR-4646-5p, miR-4667-5p, miR-4800-5p, miR-6782-5p, miR-6794-5p, miR-7150, miR-6841-5p, miR-8063 | 3.2 × 10−2 to 7.9 × 10−7 |
| Adipogenesis | 13 | miR-146a-5p, miR-150-5p, miR-2909, miR-339-3p, miR-342-5p, miR-4419a, miR-4419b, miR-4800-5p, miR-6734-3p, miR-6764-5p, miR-6782-5p, miR-6794-5p, miR-7150, miR-8063 | 4.6 × 10−2 to 5.6 × 10−5 |
| G-protein signaling | 13 | miR-146a-5p, miR-150-5p, miR-2909, miR-339-3p, miR-342-5p, miR-4419a, miR-4419b, miR-4667-5p, miR-4800-5p, miR-6734-3p, miR-6764-5p, miR-6782-5p, miR-7150, miR-8063 | 4.7 × 10−2 to 1 × 10−4 |
| Regulation of actin cytoskeleton | 12 | miR-146a-5p, miR-155-5p, miR-2909, miR-342-5p, miR-4419a, miR-4419b, miR-4646-5p, miR-4667-5p, miR-6782-5p, miR-6794-5p, miR-7150, miR-6841-5p, miR-8063 | 4.1 × 10−2 to 8.3 × 10−6 |
| MAPK signaling | 12 | miR-146a-5p, miR-155-5p, miR-339-3p, miR-342-5p, miR-4419a, miR-4646-5p, miR-4667-5p, miR-6734-3p, miR-6794-5p, miR-7150, miR-6841-5p, miR-8063 | 4.8 × 10−2 to 5 × 10−5 |
| Myometrial relaxation and contraction | 12 | miR-150-5p, miR-2909, miR-342-5p, miR-4419a, miR-4667-5p, miR-4800-5p, miR-6716-5p, miR-6734-3p, miR-6764-5p, miR-6782-5p, miR-7150 | 4.7 × 10−2 to 6 × 10−4 |
| TGF beta signaling pathway 1 | 12 | miR-146a-5p, miR-155-5p, miR-2909, miR-342-5p, miR-4419a, miR-4419b, miR-4646-5p, miR-4667-5p, miR-6734-3p, miR-7150, miR-6841-5p, miR-8063 | 4.8 × 10−2 to 7.6 × 10−9 |
| AMPK signaling | 11 | miR-2909, miR-342-5p, miR-4419a, miR-4419b, miR-4646-5p, miR-6716-5p, miR-6734-3p, miR-6764-5p, miR-6782-5p, miR-6794-5p, miR-8063 | 4 × 10−2 to 8 × 10−4 |
| Leptin signaling | 11 | miR-146a-5p, miR-2909, miR-342-5p, miR-4419b, miR-4646-5p, miR-4667-5p, miR-6734-3p, miR-6782-5p, miR-6794-5p, miR-7150, miR-8063 | 4.8 × 10−2 to 2 × 10−4 |
| Apoptosis | 9 | miR-146a-5p, miR-150-5p, miR-155-5p, miR-2909, miR-339-3p, miR-342-5p, miR-4419b, miR-6794-5p, miR-8063 | 2.7 × 10−2 to 4 × 10−4 |
| RANKL RANK signaling | 9 | miR-150-5p, miR-155-5p, miR-2909, miR-339-3p, miR-342-5p, miR-3687, miR-4667-5p, miR-6794-5p, miR-6841-5p | 4.8 × 10−2 to 5 × 10−4 |
| ErbB signaling | 8 | miR-150-5p, miR-2909, miR-342-5p, miR-4667-5p, miR-6734-3p, miR-6782-5p, miR-6794-5p, miR-7150 | 4.8 × 10−2 to 4 × 10−4 |
| Nuclear receptors | 8 | miR-150-5p, miR-155-5p, miR-342-5p, miR-4419a, miR-4667-5p, miR-6734-3p, miR-6782-5p, miR-6794-5p, miR-7150 | 4.8 × 10−2 to 3.5 × 10−5 |
| Prolactin signaling | 8 | miR-2909, miR-342-5p, miR-4419a, miR-4646-5p, miR-4667-5p, miR-6716-5p, miR-6734-3p | 4.6 × 10−2 to 1 × 10−4 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nziza, N.; Jeziorski, E.; Delpont, M.; Cren, M.; Chevassus, H.; Carbasse, A.; Mahe, P.; Abassi, H.; Joly-Monrigal, P.; Schordan, E.; et al. Synovial-Fluid miRNA Signature for Diagnosis of Juvenile Idiopathic Arthritis. Cells 2019, 8, 1521. https://doi.org/10.3390/cells8121521
Nziza N, Jeziorski E, Delpont M, Cren M, Chevassus H, Carbasse A, Mahe P, Abassi H, Joly-Monrigal P, Schordan E, et al. Synovial-Fluid miRNA Signature for Diagnosis of Juvenile Idiopathic Arthritis. Cells. 2019; 8(12):1521. https://doi.org/10.3390/cells8121521
Chicago/Turabian StyleNziza, Nadége, Eric Jeziorski, Marion Delpont, Maïlys Cren, Hugues Chevassus, Aurélia Carbasse, Perrine Mahe, Hamouda Abassi, Pauline Joly-Monrigal, Eric Schordan, and et al. 2019. "Synovial-Fluid miRNA Signature for Diagnosis of Juvenile Idiopathic Arthritis" Cells 8, no. 12: 1521. https://doi.org/10.3390/cells8121521
APA StyleNziza, N., Jeziorski, E., Delpont, M., Cren, M., Chevassus, H., Carbasse, A., Mahe, P., Abassi, H., Joly-Monrigal, P., Schordan, E., Mangé, A., Jorgensen, C., Apparailly, F., & Duroux-Richard, I. (2019). Synovial-Fluid miRNA Signature for Diagnosis of Juvenile Idiopathic Arthritis. Cells, 8(12), 1521. https://doi.org/10.3390/cells8121521