Phosphorylation of TIP3 Aquaporins during Phaseolus vulgaris Embryo Development
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Peptide Synthesis and Purification
2.3. Membrane Protein Isolation
2.4. Phosphoprotein Detection
2.5. Phosphoamino Acid Detection
2.6. PvTIP3;1 and PvTIP3;2 Immunodetection
2.7. Overexpression of PvTIP3;2 in P. pastoris
2.8. Phosphotyrosine Immunoprecipitation
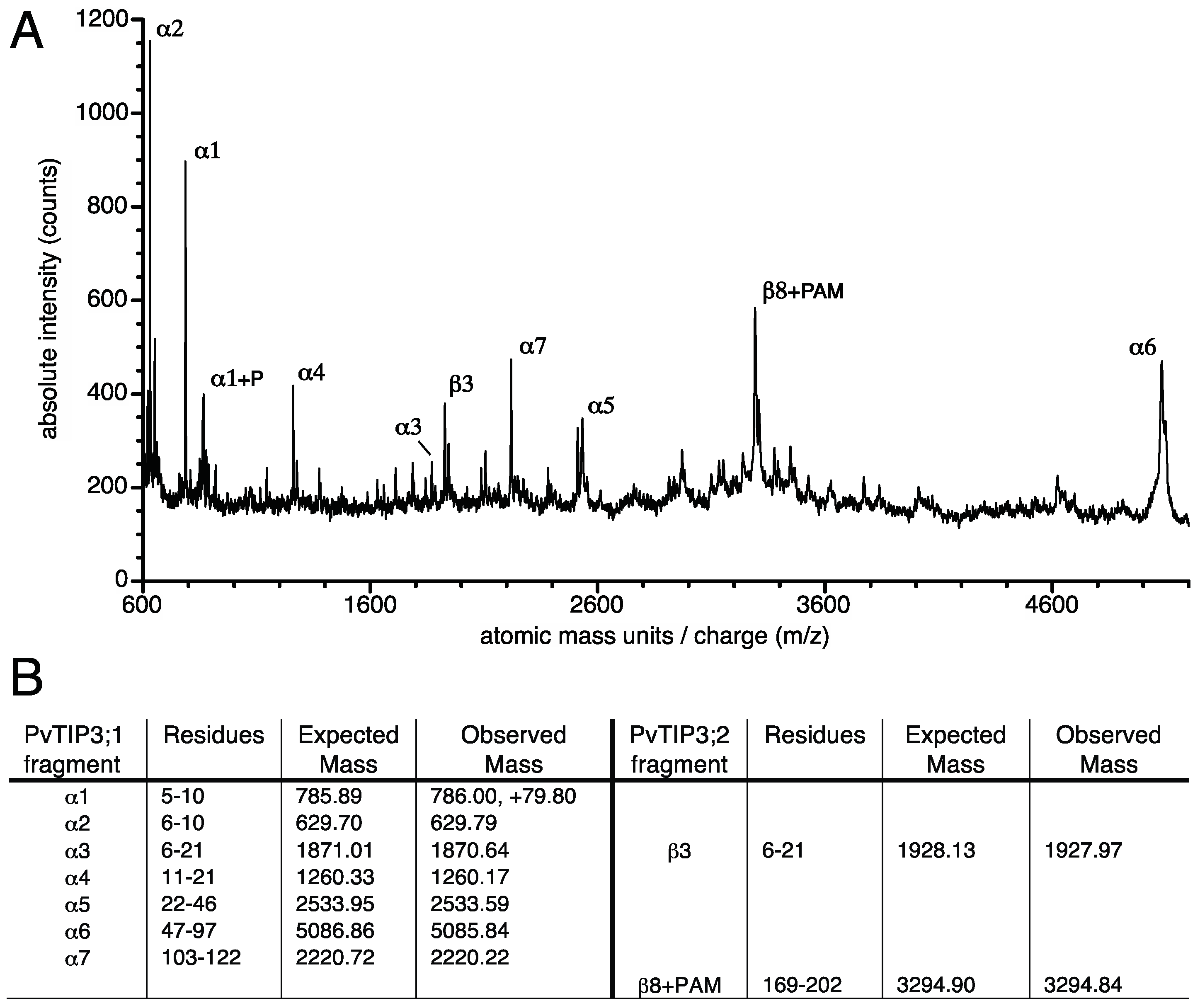
2.9. Mass Spectrometry
2.10. Circular Dichroism (CD) Spectrometry
3. Results
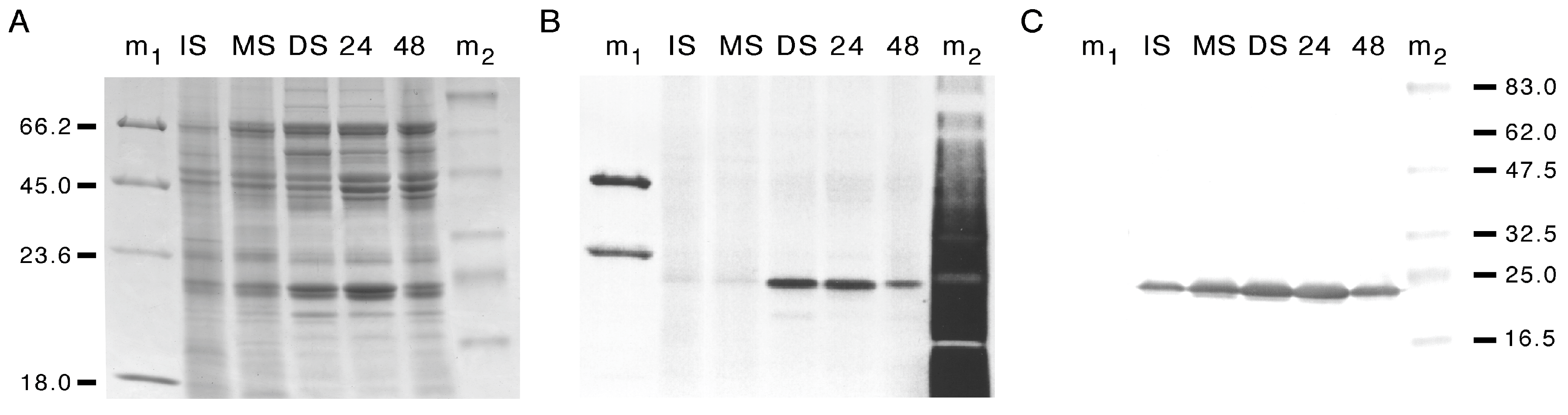
3.1. Developmental Changes in the Abundance of Seed-Membrane Phosphoproteins
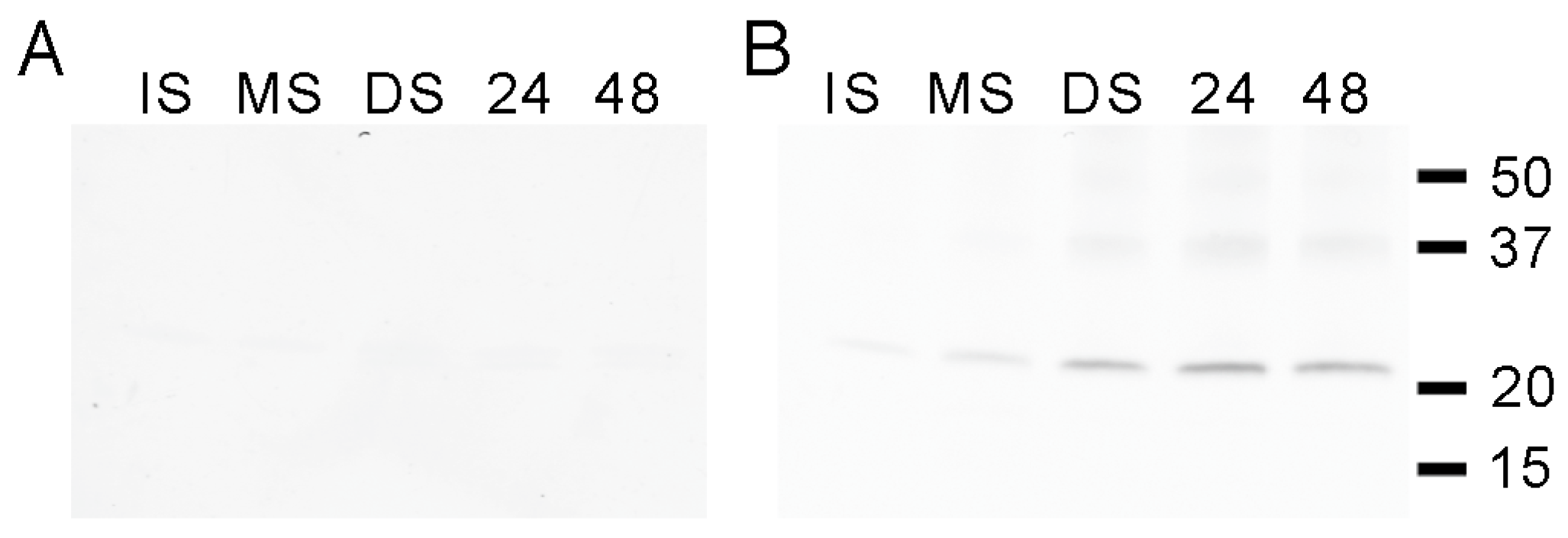
3.2. PvTIP3 Proteins Are the Primary Seed-Membrane Phosphoproteins
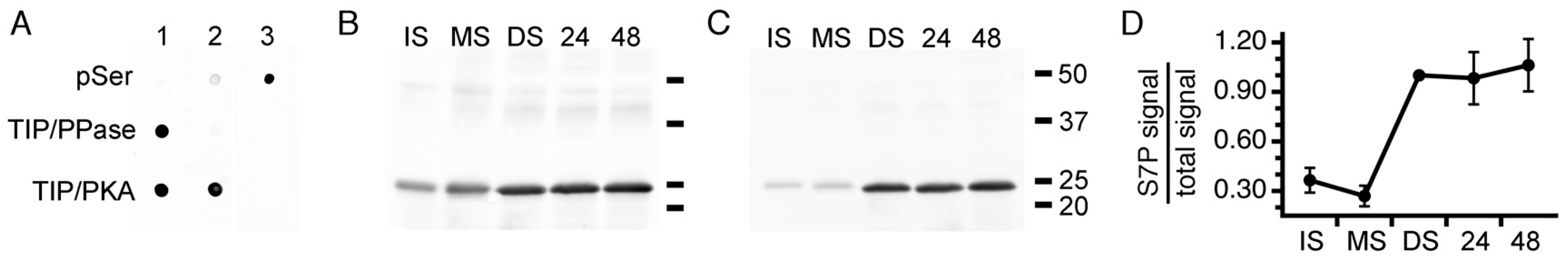
3.3. Expression of PvTIP3;1 and PvTIP3;2 in Seed Development
3.4. Ser7 Phosphorylation in PvTIP3;1 Increases Following Seed Maturation
3.5. Tyrosine Phosphorylation of the PvTIP3;2 Amino-Terminal Peptide
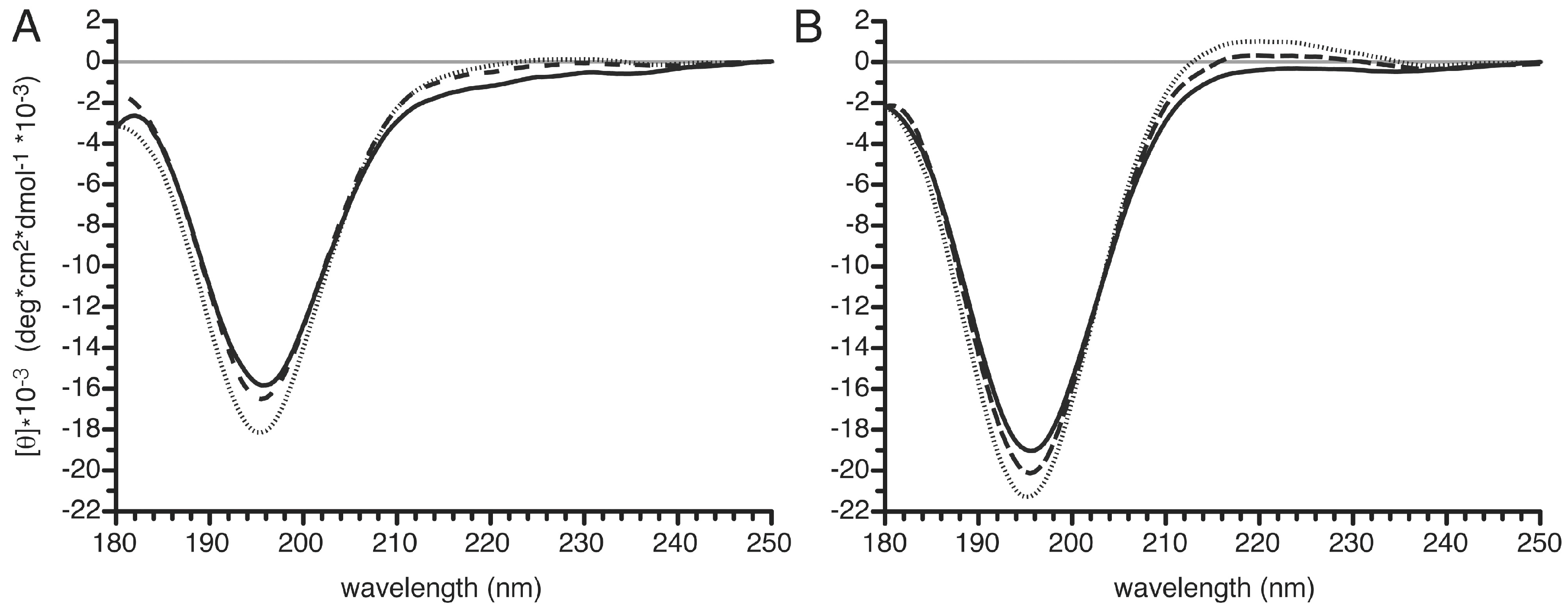
3.6. Structural Changes in the PvTIP3;1 Amino Terminal Peptide upon Phosphorylation
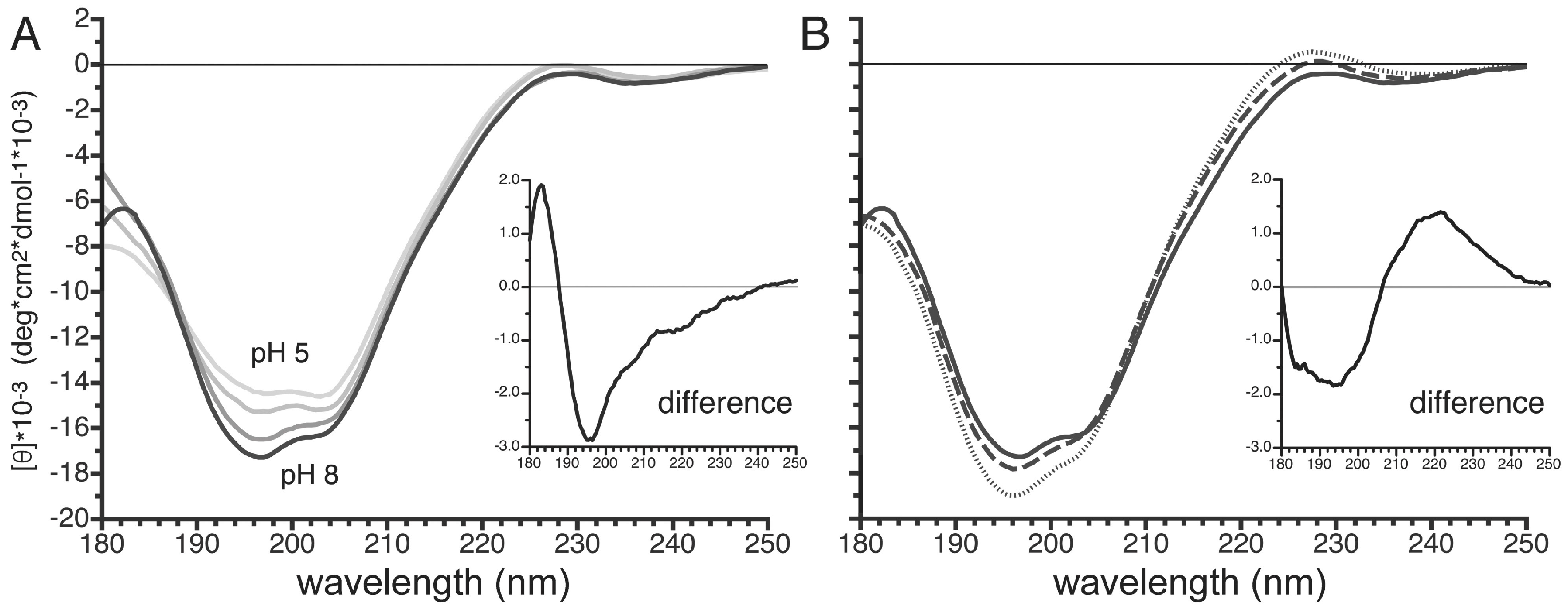
3.7. pH- and Temperature-Dependent Structural Changes in the Carboxy Terminus of PvTIP3;1
4. Discussion
4.1. The Membrane Phosphoproteome Changes Dynamically during Seed Maturation, Dormancy and Germination
4.2. Phosphorylation-Dependent Appearance of Polyproline II Secondary Structure
4.3. Serine Phosphorylation of PvTIP3;1 during Seed Maturation
4.4. Role of PvTIP3;2 in Seed Development
4.5. Possible Roles of PvTIP3;2 Tyrosine Phosphorylation
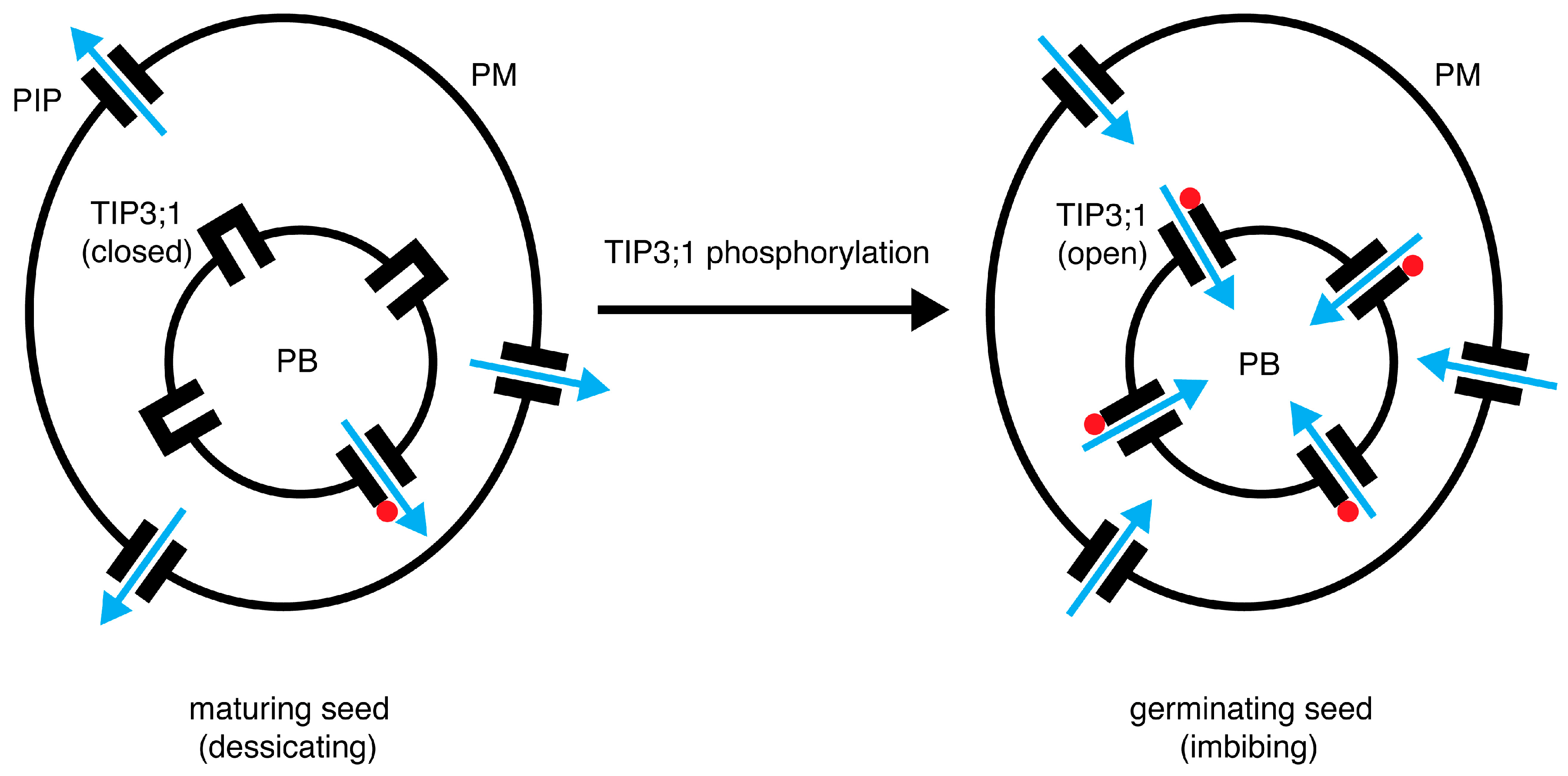
4.6. A Model for the Role of PvTIP3;1 Ser7 Phosphorylation in Seed Development
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gomes, D.; Agasse, A.; Thiébaud, P.; Delrot, S.; Gerós, H.; Chaumont, F. Aquaporins are multifunctional water and solute transporters highly divergent in living organisms. Biochim. Biophys. Acta 2009, 1788, 1213–1228. [Google Scholar] [CrossRef]
- Maurel, C.; Kado, R.T.; Guern, J.; Chrispeels, M.J. Phosphorylation regulates the water channel activity of the seed-specific aquaporin α-TIP. EMBO J. 1995, 14, 3028–3035. [Google Scholar] [CrossRef]
- Maurel, C.; Boursiac, Y.; Luu, D.T.; Santoni, V.; Shahzad, Z.; Verdoucq, L. Aquaporins in plants. Physiol. Rev. 2015, 95, 1321–1358. [Google Scholar] [CrossRef] [PubMed]
- Fox, A.R.; Maistriaux, L.C.; Chaumont, F. Toward understanding of the high number of plant aquaporin isoforms and multiple regulation mechanisms. Plant. Sci. 2017, 264, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.D.; Herman, E.M.; Chrispeels, M.J. An abundant, highly conserved tonoplast protein in seeds. Plant. Physiol. 1989, 91, 1006–1013. [Google Scholar] [CrossRef] [PubMed]
- Gattolin, S.; Sorieul, M.; Frigerio, L. Mapping of tonoplast intrinsic proteins in maturing and germinating Arabidopsis seeds reveals dual localization of embryonic TIPs to the tonoplast and plasma membrane. Mol. Plant. 2011, 4, 180–189. [Google Scholar] [CrossRef]
- Johnson, K.D.; Chrispeels, M.J. Tonoplast-bound protein kinase phosphorylates tonoplast intrinsic protein. Plant. Physiol. 1992, 100, 1787–1795. [Google Scholar] [CrossRef]
- Maurel, C.; Chrispeels, M.; Lurin, C.; Tacnet, F.; Geelen, D.; Ripoche, P.; Guern, J. Function and regulation of seed aquaporins. J. Exp. Bot. 1997, 48, 421–430. [Google Scholar] [CrossRef]
- Daniels, M.J.; Yeager, M. Phosphorylation of aquaporin PvTIP3;1 defined by mass spectrometry and molecular modeling. Biochemistry 2005, 44, 14443–14454. [Google Scholar] [CrossRef]
- Johnson, K.D.; Höfte, H.; Chrispeels, M.J. An intrinsic tonoplast protein of protein storage vacuoles in seeds is structurally related to a bacterial solute transporter (GlpF). Plant Cell 1990, 2, 525–532. [Google Scholar] [CrossRef]
- Ariani, A.; Gepts, P. Genome-wide identification and characterization of aquaporin gene family in common bean (Phaseolus vulgaris L.). Mol. Genet. Genomics 2015, 290, 1771–1785. [Google Scholar] [CrossRef] [PubMed]
- Vander Willigen, C.; Postaire, O.; Tournaire-Roux, C.; Boursiac, Y.; Maurel, C. Expression and inhibition of aquaporins in germinating Arabidopsis seeds. Plant Cell Physiol. 2006, 47, 1241–1250. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Staehelin, L.A. Protein storage vacuoles are transformed into lytic vacuoles in root meristematic cells of germinating seedlings by multiple, cell type-specific mechanisms. Plant Physiol. 2011, 155, 2023–2035. [Google Scholar] [CrossRef] [PubMed]
- Weber, H.; Borisjuk, L.; Wobus, U. Molecular physiology of legume seed development. Ann. Rev. Plant Biol. 2005, 56, 253–279. [Google Scholar] [CrossRef]
- Daniels, M.J.; Chrispeels, M.J.; Yeager, M. Projection structure of a plant vacuole membrane aquaporin by electron cryo-crystallography. J. Mol. Biol. 1999, 294, 1337–1349. [Google Scholar] [CrossRef][Green Version]
- Sambrook, J.F.; Russell, D.W. Molecular Cloning: A Laboratory Manual, 3rd ed.; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 2001. [Google Scholar]
- Steinberg, T.H.; Agnew, B.J.; Gee, K.R.; Leung, W.-Y.; Goodman, T.; Schulenberg, B.; Hendrickson, J.; Beechem, J.M.; Haugland, R.P.; Patton, W.F. Global quantitative phosphoprotein analysis using multiplexed proteomics technology. Proteomics 2003, 3, 1128–1144. [Google Scholar] [CrossRef]
- Makrantoni, V.; Antrobus, R.; Botting, C.H.; Coote, P.J. Rapid enrichment and analysis of yeast phosphoproteins using affinity chromatography, 2D-PAGE and peptide mass fingerprinting. Yeast 2005, 22, 401–414. [Google Scholar] [CrossRef]
- Zhang, G.; Neubert, T.A. Use of detergents to increase selectivity of immunoprecipitation of tyrosine phosphorylated peptides prior to identification by MALDI quadrupole-TOF MS. Proteomics 2006, 6, 571–578. [Google Scholar] [CrossRef]
- Kelly, S.M.; Jess, T.J.; Price, N.C. How to study proteins by circular dichroism. Biochim. Biophys. Acta 2005, 1751, 119–139. [Google Scholar] [CrossRef]
- Pace, C.N.; Vajdos, F.; Fee, L.; Grimsley, G.; Gray, T. How to measure and predict the molar absorption coefficient of a protein. Protein Sci. 1995, 4, 2411–2423. [Google Scholar] [CrossRef]
- Greenfield, N.J. Using circular dichroism spectra to estimate protein secondary structure. Nat. Protoc. 2006, 1, 2876–2890. [Google Scholar] [CrossRef] [PubMed]
- Lees, J.G.; Smith, B.R.; Wien, F.; Miles, A.J.; Wallace, B.A. CDtool- an integrated software package for circular dichroism spectroscopic data processing, analysis, and archiving. Anal. Biochem. 2004, 332, 285–289. [Google Scholar] [CrossRef] [PubMed]
- Krittanai, C.; Johnson, W.C., Jr. Correcting the circular dichroism spectra of peptides for contributions of absorbing side chains. Anal. Biochem. 1997, 253, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Sreerama, N.; Woody, R.W. Computation and analysis of protein circular dichroism spectra. Methods Enzymol. 2004, 383, 318–351. [Google Scholar]
- Whitmore, L.; Wallace, B.A. Protein secondary structure analyses from circular dichroism spectroscopy: Methods and reference databases. Biopolymers 2008, 89, 392–400. [Google Scholar] [CrossRef]
- Abdul-Gader, A.; Miles, A.J.; Wallace, B.A. A reference dataset for the analyses of membrane protein secondary structures and transmembrane residues using circular dichroism spectroscopy. Bioinformatics 2011, 27, 1630–1636. [Google Scholar] [CrossRef]
- Stankiewicz, P.J.; Tracey, A.S.; Crans, D.C. Inhibition of phosphate-metabolizing enzymes by oxovanadium(V) complexes. In Metal Ions in Biological Systems; Sigel, H., Sigel, A., Eds.; Marcel Dekker, Inc.: New York, NY, USA, 1995; Volume 31, pp. 287–324. [Google Scholar]
- Oufattole, M.; Park, J.H.; Poxleitner, M.; Jiang, L.; Rogers, J.C. Selective membrane protein internalization accompanies movement from the endoplasmic reticulum to the protein storage vacuole pathway in Arabidopsis. Plant Cell 2005, 17, 3066–3080. [Google Scholar] [CrossRef]
- Daniels, M.J.; Chaumont, F.; Mirkov, T.E.; Chrispeels, M.J. Characterization of a new vacuolar membrane aquaporin sensitive to mercury at a unique site. Plant Cell 1996, 8, 587–599. [Google Scholar]
- Hunter, P.R.; Craddock, C.P.; Di Benedetto, S.; Roberts, L.M.; Frigerio, L. Fluorescent reporter proteins for the tonoplast and the vacuolar lumen identify a single vacuolar compartment in Arabidopsis cells. Plant Physiol. 2007, 145, 1371–1382. [Google Scholar] [CrossRef]
- Utsugi, S.; Shibasaka, M.; Maekawa, M.; Katsuhara, M. Control of the water transport activity of barley HvTIP3;1 specifically expressed in seeds. Plant Cell Physiol. 2015, 56, 1831–1840. [Google Scholar] [CrossRef][Green Version]
- Aebersold, R.; Patterson, S.D. Current Problems and Technical Solutions in Protein Biochemistry. In Proteins: Analysis and Design; Angeletti, R.H., Ed.; Academic Press: San Diego, CA, USA, 1998; pp. 4–120. [Google Scholar]
- Höfte, H.; Chrispeels, M.J. Protein sorting to the vacuolar membrane. Plant Cell 1992, 4, 995–1004. [Google Scholar] [CrossRef] [PubMed]
- Maurel, C. Plant aquaporins: Novel functions and regulation properties. FEBS Lett. 2007, 581, 2227–2236. [Google Scholar] [CrossRef] [PubMed]
- Hjerrild, M.; Stensballe, A.; Rasmussen, T.E.; Kofoed, C.B.; Blom, N.; Sicheritz-Ponten, T.; Larsen, M.R.; Brunak, S.; Jensen, O.N.; Gammeltoft, S. Identification of phosphorylation sites in protein kinase A substrates using artificial neural networks and mass spectrometry. J. Proteome Res. 2004, 3, 426–433. [Google Scholar] [CrossRef]
- Bah, A.; Forman-Kay, J.D. Modulation of intrinsically disordered protein function by post-translational modifications. J. Biol. Chem. 2016, 291, 6696–6705. [Google Scholar] [CrossRef]
- Sreerama, N.; Woody, R.W. Poly(Pro)II helices in globular proteins: Identification and circular dichroic analysis. Biochemistry 1994, 33, 10022–10025. [Google Scholar] [CrossRef]
- Tiffany, M.L.; Krimm, S. Extended conformations of polypeptides and proteins in urea and guanidine hydrochloride. Biopolymers 1973, 12, 575–587. [Google Scholar] [CrossRef]
- Venyaminov, S.Y.; Baikalov, I.A.; Shen, Z.M.; Wu, C.-S.C.; Yang, J.T. Circular dichroic analysis of denatured proteins: Inclusion of denatured proteins in the reference set. Anal. Biochem. 1993, 214, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Lopes, J.L.S.; Miles, A.J.; Whitmore, L.; Wallace, B.A. Distinct circular dichroism spectroscopic signatures of polyproline II and unordered secondary structures: Applications in secondary structure analyses. Protein Sci. 2014, 23, 1765–1772. [Google Scholar] [CrossRef] [PubMed]
- Machado, E.L.; da Silva, A.C.; da Silva, M.J.; Leite, A.; Ottoboni, L.M.M. Endogenous protein phosphorylation and casein kinase activity during seed development in Araucaria angustifolia. Phytochemistry 2002, 61, 835–842. [Google Scholar] [CrossRef]
- Agrawal, G.K.; Thelen, J.J. Large scale identification and quantitative profiling of phosphoproteins expressed during seed filling in oilseed rape. Mol. Cell Proteomics 2006, 5, 2044–2059. [Google Scholar] [CrossRef]
- Mukhopadhyay, K.; Singh, M. Endogenous protein phosphorylation and protein kinase activity in winged bean. Phytochemistry 1997, 46, 455–459. [Google Scholar] [CrossRef]
- Kovaleva, V.; Cramer, R.; Krynytskyy, H.; Gout, I.; Gout, R. Analysis of tyrosine phosphorylation and phosphotyrosine-binding proteins in germinating seeds from Scots pine. Plant Physiol. Biochem. 2013, 67, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Herman, E.M.; Larkins, B.A. Protein storage bodies and vacuoles. Plant Cell 1999, 11, 601–613. [Google Scholar] [CrossRef] [PubMed]
- Iakoucheva, L.M.; Radivojac, P.; Brown, C.J.; O’Connor, T.R.; Sikes, J.G.; Obradovic, Z.; Dunker, A.K. The importance of intrinsic disorder for protein phosphorylation. Nucleic Acids Res. 2004, 32, 1037–1049. [Google Scholar] [CrossRef] [PubMed]
- Manning, M.C.; Woody, R.W. Theoretical CD studies of polypeptide helices: Examination of important electronic and geometric factors. Biopolymers 1991, 31, 569–586. [Google Scholar] [CrossRef] [PubMed]
- Elam, W.A.; Schrank, T.P.; Campagnolo, A.J.; Hilser, V.J. Evolutionary conservation of the polyproline II conformation surrounding intrinsically disordered phosphorylation sites. Protein Sci. 2013, 22, 405–417. [Google Scholar] [CrossRef]
- Kim, S.-Y.; Jung, Y.; Hwang, G.-S.; Han, H.; Cho, M. Phosphorylation alters backbone conformational preferences of serine and threonine peptides. Proteins 2011, 79, 3155–3165. [Google Scholar] [CrossRef]
- Chaumont, F.; Moshelion, M.; Daniels, M.J. Regulation of plant aquaporin activity. Biol. Cell 2005, 97, 749–764. [Google Scholar] [CrossRef]
- Jolivet, P.; Roux, E.; d’Andrea, S.; Davanture, M.; Negroni, L.; Zivy, M.; Chardot, T. Protein composition of oil bodies in Arabidopsis thaliana ecotype WS. Plant Physiol. Biochem. 2004, 42, 501–509. [Google Scholar] [CrossRef]
- Li, G.-W.; Peng, Y.-H.; Yu, X.; Zhang, M.-H.; Cai, W.-M.; Sun, W.-N.; Su, W.-A. Transport functions and expression analysis of vacuolar membrane aquaporins in response to various stresses in rice. J. Plant Physiol. 2008, 165, 1879–1888. [Google Scholar] [CrossRef]
- Mao, Z.; Sun, W. Arabidopsis seed-specific vacuolar aquaporins are involved in maintaining seed longevity under the control of ABSCISIC ACID INSENSITIVE 3. J. Exp. Bot. 2015, 66, 4781–4794. [Google Scholar] [CrossRef] [PubMed]
- Campbell, E.M.; Birdsell, D.N.; Yool, A.J. The activity of human aquaporin 1 as a cGMP-gated cation channel is regulated by tyrosine phosphorylation in the carboxyl-terminal domain. Mol. Pharmacol. 2012, 81, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Yanochko, G.M.; Yool, A.J. Regulated cationic channel function in Xenopus oocytes expressing Drosophila Big Brain. J. Neurosci. 2002, 22, 2530–2540. [Google Scholar] [CrossRef] [PubMed]
- O’Rourke, J.A.; Iniguez, L.P.; Fu, F.; Bucciarelli, B.; Miller, S.S.; Jackson, S.A.; McClean, P.E.; Li, J.; Dai, X.; Zhao, P.X.; et al. An RNA-Seq based gene expression atlas of the common bean. BMC Genomics 2014, 15, 866–881. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, M.X.; Moon, S.; Jung, K.-H. Genome-wide expression analysis of rice aquaporin genes and development of a functional gene network mediated by aquaporin expression in roots. Planta 2013, 238, 669–681. [Google Scholar] [CrossRef]
- Melroy, D.L.; Herman, E.M. TIP, an integral membrane protein of the protein-storage vacuoles of the soybean cotyledon undergoes developmentally regulated membrane accumulation and removal. Planta 1991, 184, 113–122. [Google Scholar] [CrossRef]
- Robinson, D.G.; Hinz, G. Vacuole biogenesis and protein transport to the plant vacuole: A comparison with the yeast vacuole and the mammalian lysosome. Protoplasma 1997, 197, 1–25. [Google Scholar] [CrossRef]
- Strzalka, K.; Hara-Nishimura, I.; Nishimura, M. Changes in physical properties of vacuolar membrane during transformation of protein bodies into vacuoles in germinating pumpkin seeds. Biochim. Biophys. Acta 1995, 1239, 103–110. [Google Scholar] [CrossRef]
- Capelle, V.; Remoué, C.; Moreau, L.; Reyss, A.; Mahé, A.; Massonneau, A.; Falque, M.; Charcosset, A.; Thévenot, C.; Rogowsky, P.; et al. QTLs and candidate genes for desiccation and abscisic acid content in maize kernels. BMC Plant. Biol. 2010, 10, 2–23. [Google Scholar] [CrossRef]
- Veselova, T.V.; Veselovskii, V.A.; Usmanov, P.D.; Usmanova, O.V.; Kozar, V.I. Hypoxia and imbibition injuries to aging seeds. Russ. J. Plant. Physiol 2003, 50, 835–842. [Google Scholar] [CrossRef]
- Zhou, Y.; Setz, N.; Niemietz, C.; Qu, H.; Offler, C.E.; Tyerman, S.D.; Patrick, J.W. Aquaporins and unloading of phloem-imported water in coats of developing bean seeds. Plant. Cell Environ. 2007, 30, 1566–1577. [Google Scholar] [CrossRef] [PubMed]
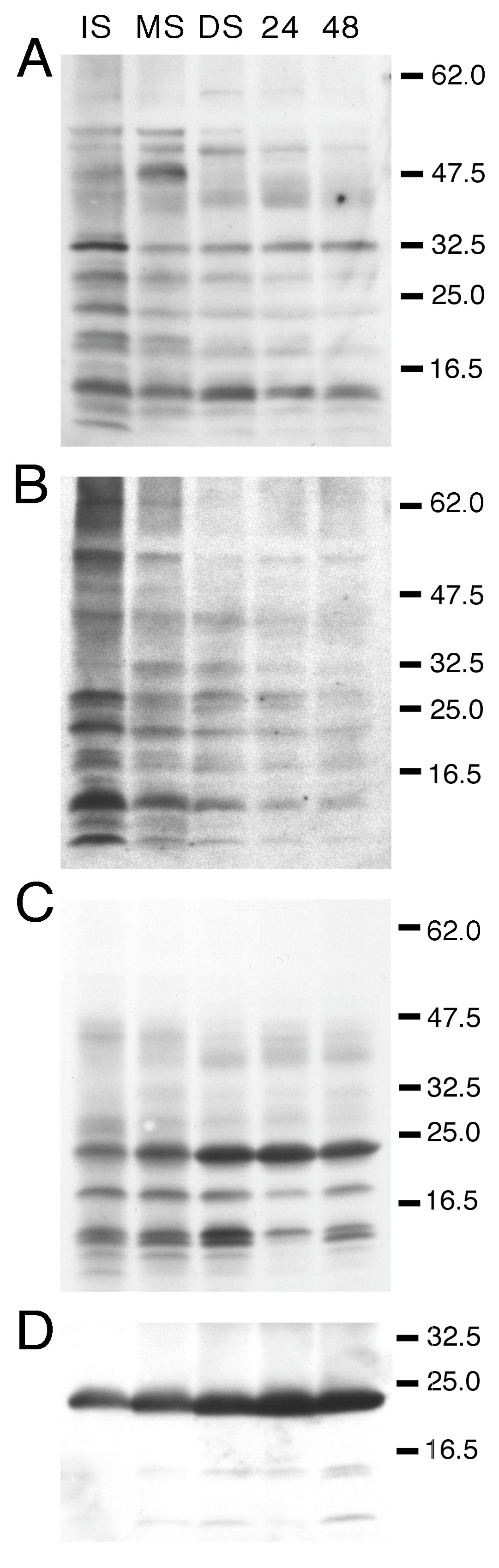

| kD | Immature Seed | Mature Seed | Dry Seed | 24 h Imbibed | 48 h Imbibed |
|---|---|---|---|---|---|
| 70 | + | ++ | ++ | ++ | +++ |
| 67 | +++ | ++++ | ++ | ++ | +++ |
| 60 | - | + | ++ | + | ++ |
| 52 | ++++ | +++ | ++ | +++ | ++++ |
| 48 | ++++ | +++ | ++ | +++ | ++++ |
| 45 | + | + | ++ | ++ | +++ |
| 36 | + | - | - | - | - |
| 33 | + | + | - | + | + |
| 32 | + | ++ | + | + | + |
| 29 | ++ | ++ | + | + | ++ |
| 24 | ++++ | ++++ | ++++ | ++++ | ++++ |
| 22 | ++++ | ++++ | +++ | ++++ | ++++ |
| 19 | - | + | ++ | + | + |
| 15 | - | + | + | + | + |
| 12 | + | + | - | - | - |
| kD | Immature Seed | Mature Seed | Dry Seed | 24 h Imbibed | 48 h Imbibed |
|---|---|---|---|---|---|
| 24 | + | + | ++++ | ++++ | +++ |
| 19 | - | - | + | - | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Daniels, M.J.; Yeager, M. Phosphorylation of TIP3 Aquaporins during Phaseolus vulgaris Embryo Development. Cells 2019, 8, 1362. https://doi.org/10.3390/cells8111362
Daniels MJ, Yeager M. Phosphorylation of TIP3 Aquaporins during Phaseolus vulgaris Embryo Development. Cells. 2019; 8(11):1362. https://doi.org/10.3390/cells8111362
Chicago/Turabian StyleDaniels, Mark J., and Mark Yeager. 2019. "Phosphorylation of TIP3 Aquaporins during Phaseolus vulgaris Embryo Development" Cells 8, no. 11: 1362. https://doi.org/10.3390/cells8111362
APA StyleDaniels, M. J., & Yeager, M. (2019). Phosphorylation of TIP3 Aquaporins during Phaseolus vulgaris Embryo Development. Cells, 8(11), 1362. https://doi.org/10.3390/cells8111362




