Prokaryotic Aquaporins
Abstract
1. Introduction
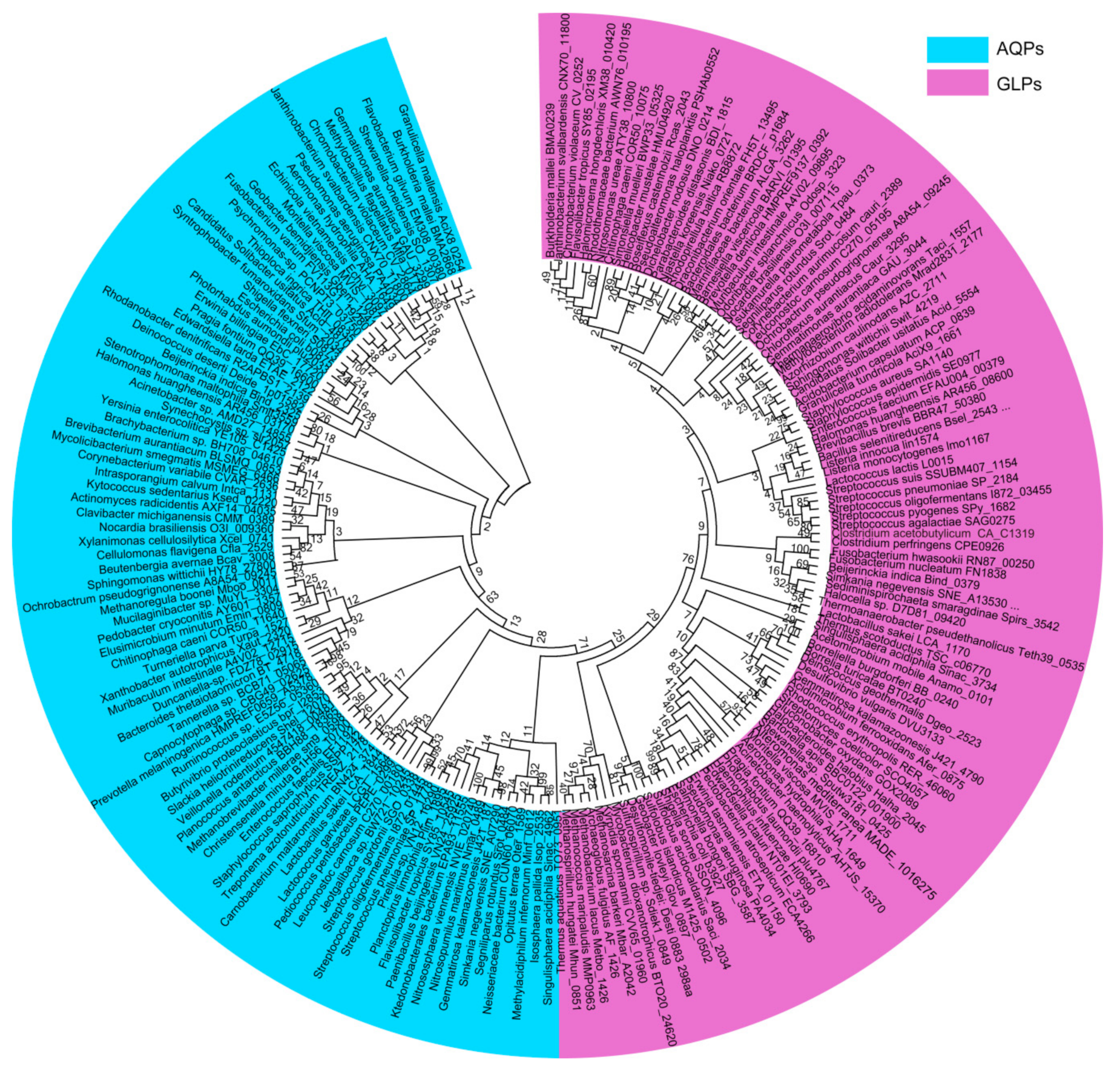
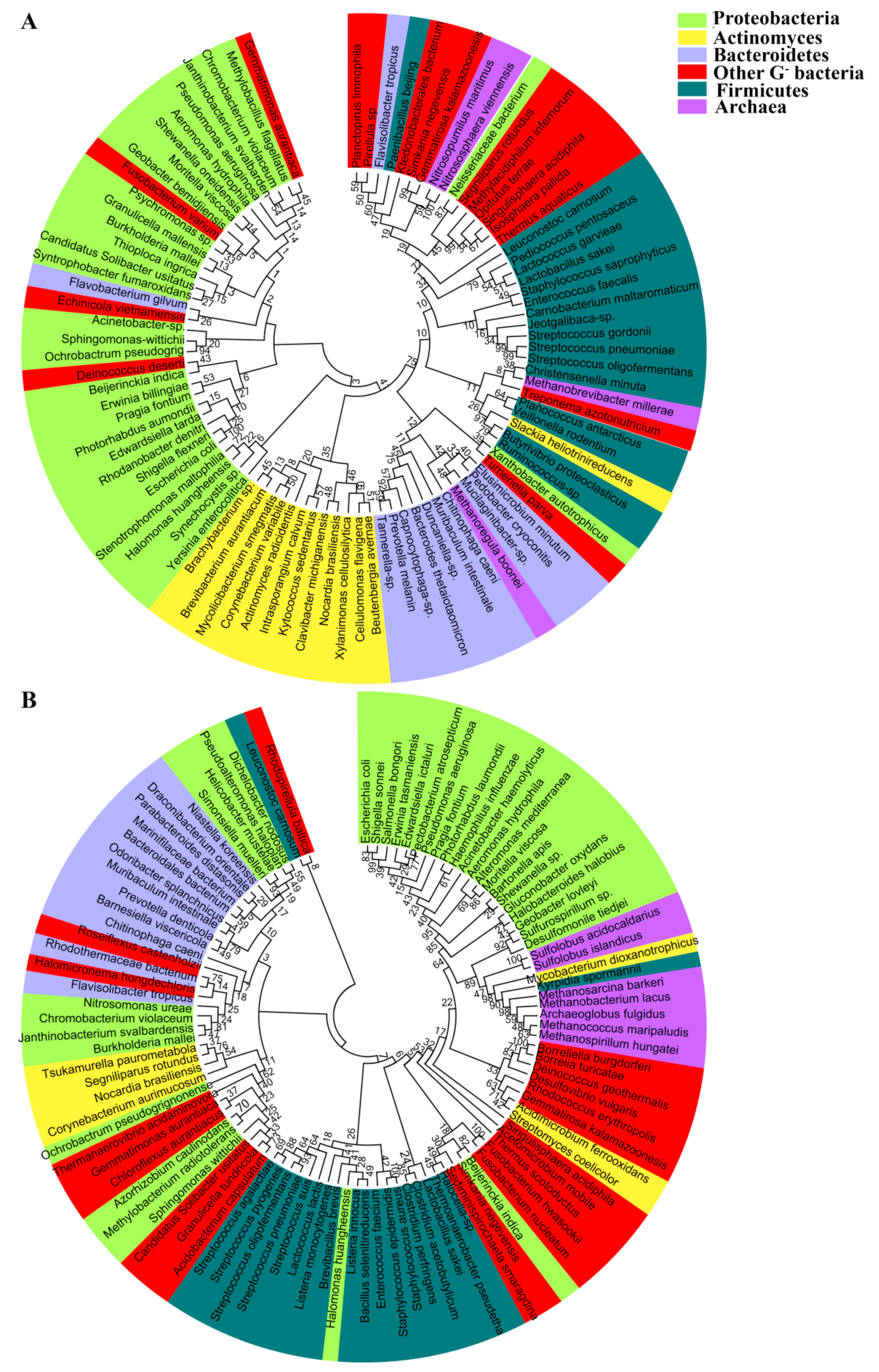
2. Distribution and Phylogeny of the Aquaporin Homologues in Prokaryotes
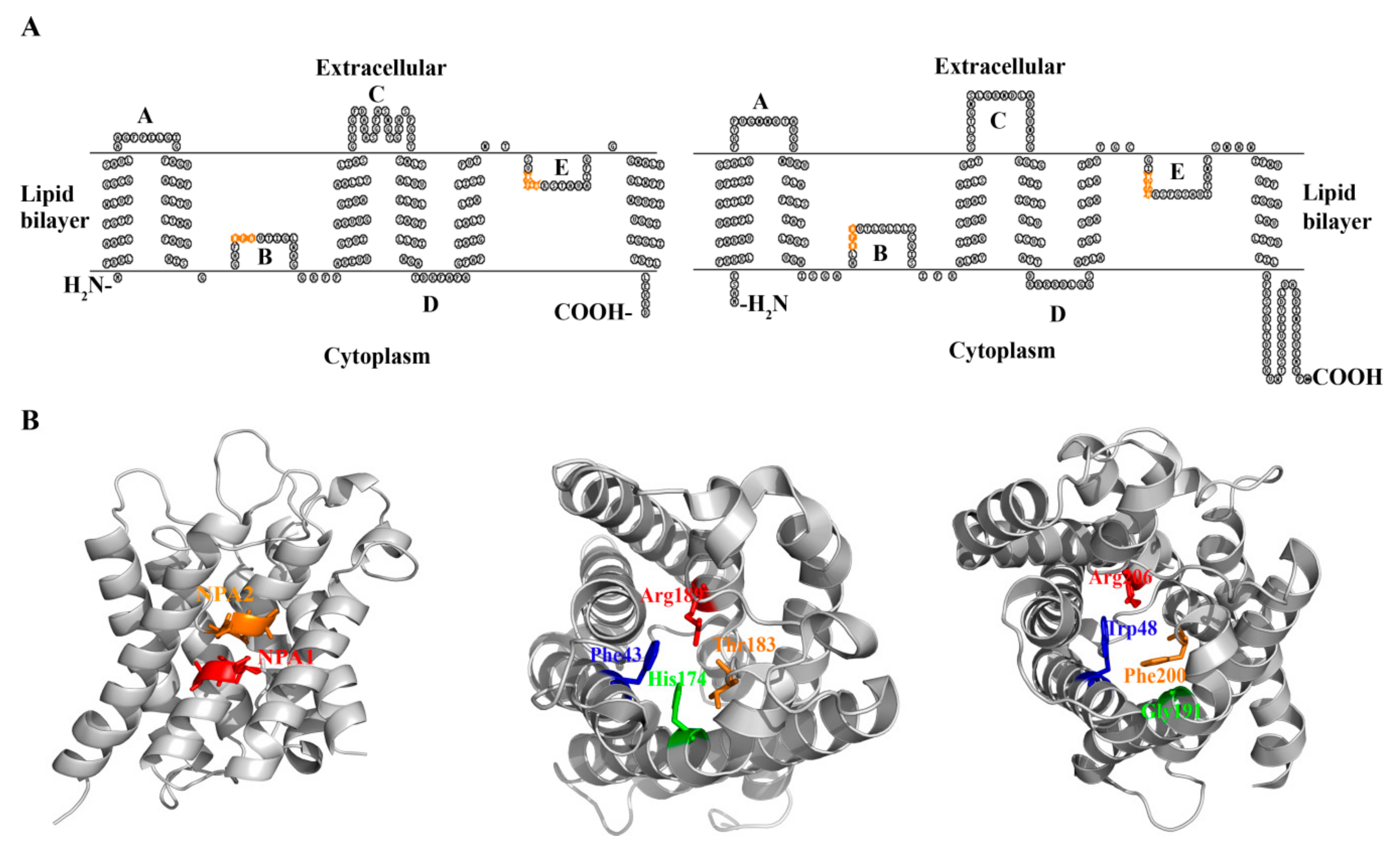
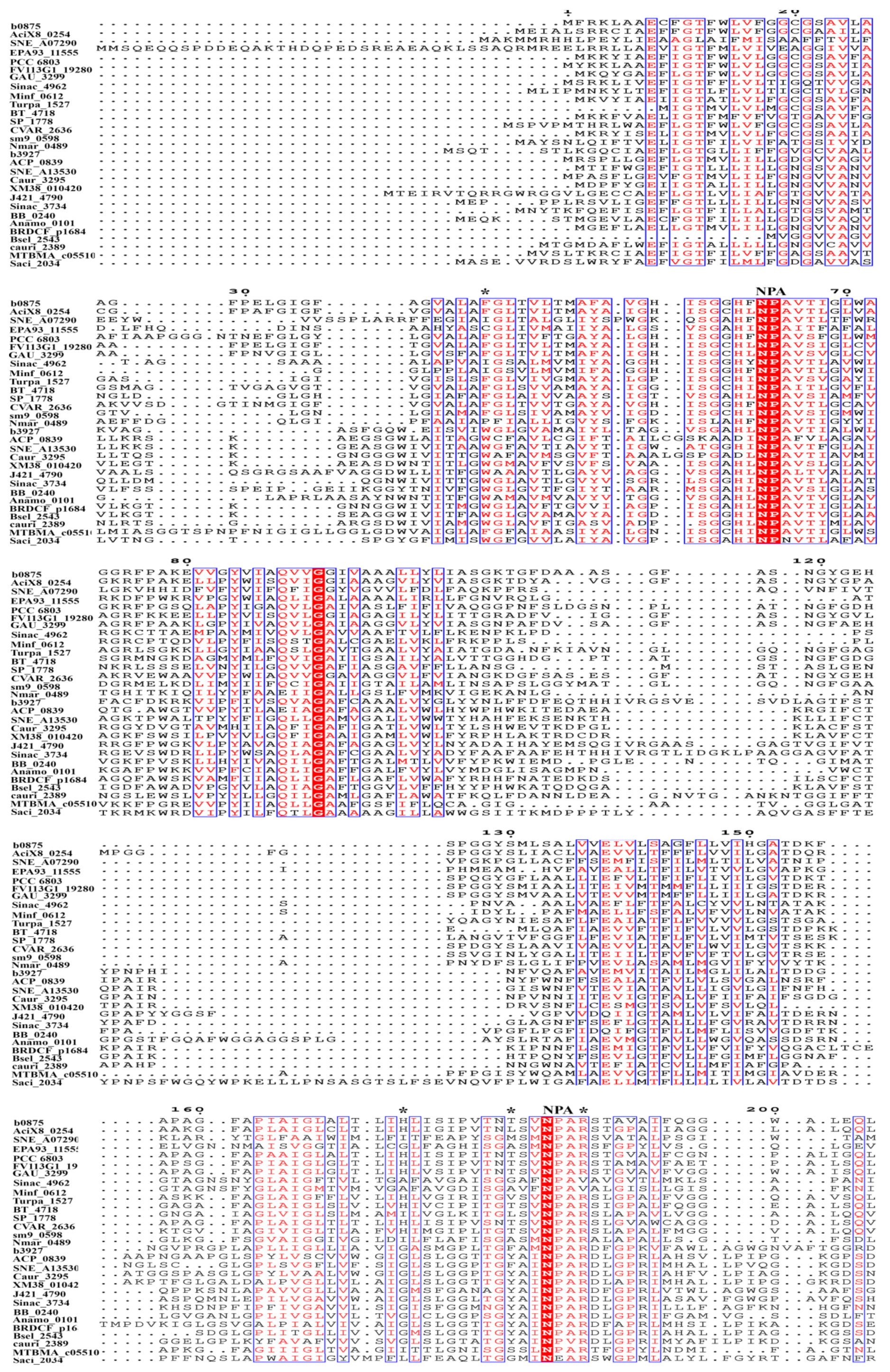
3. The Conserved Amino Acid Motifs and Topological Characteristics of Prokaryotic Aquaporins
4. The Protein Structures and Substrate Selectivities of Prokaryotic Aquaporins
5. Research Approaches Used for Determining the Transport Properties of Prokaryotic Aquaporins
6. Physiological Functions of Prokaryotic Aquaporins
7. Mechanisms of the Regulatory Expression of Prokaryotic Aquaporins
8. Future Research Directions
Funding
Conflicts of Interest
References
- Preston, G.M.; Carroll, T.P.; Guggino, W.B.; Agre, P. Appearance of water channels in Xenopus oocytes expressing red cell CHIP28 protein. Science 1992, 256, 385–387. [Google Scholar] [CrossRef] [PubMed]
- Borgnia, M.J.; Agre, P. Reconstitution and functional comparison of purified GlpF and AqpZ, the glycerol and water channels from Escherichia coli. Proc. Natl. Acad. Sci. USA 2001, 98, 2888–2893. [Google Scholar] [CrossRef] [PubMed]
- Ishibashi, K.; Morishita, Y.; Tanaka, Y. The evolutionary aspects of aquaporin family. Adv. Exp. Med. Biol. 2017, 969, 35–50. [Google Scholar] [PubMed]
- Calamita, I. Understanding microbial MIP channels. Trends Microbiol. 2000, 8, 104–105. [Google Scholar] [CrossRef]
- Agre, P.; Bonhivers, M.; Borgnia, M.J. The aquaporins, blueprints for cellular plumbing systems. J. Biol. Chem. 1998, 273, 14659–14662. [Google Scholar] [CrossRef]
- Abascal, F.; Irisarri, I.; Zardoya, R. Diversity and evolution of membrane intrinsic proteins. Biochim. Biophys. Acta 2014, 1840, 1468–1481. [Google Scholar] [CrossRef]
- Zardoya, R. Phylogeny and evolution of the major intrinsic protein family. Biol. Cell 2005, 97, 397–414. [Google Scholar] [CrossRef]
- Ishibashi, K.; Hara, S.; Kondo, S. Aquaporin water channels in mammals. Clin. Exp. Nephrol. 2009, 13, 107–117. [Google Scholar] [CrossRef]
- Savage, D.F.; O’Connell, J.D.; Miercke, L.J.; Finer-Moore, J.; Stroud, R.M. Structural context shapes the aquaporin selectivity filter. Proc. Natl. Acad. Sci. USA 2010, 107, 17164–17169. [Google Scholar] [CrossRef]
- Sonah, H.; Deshmukh, R.K.; Labbe, C.; Belanger, R.R. Analysis of aquaporins in Brassicaceae species reveals high-level of conservation and dynamic role against biotic and abiotic stress in canola. Sci. Rep. 2017, 7, 2771. [Google Scholar] [CrossRef]
- Calamita, G.; Perret, J.; Delporte, C. Aquaglyceroporins: Drug targets for metabolic diseases? Front. Physiol. 2018, 9, 851. [Google Scholar] [CrossRef] [PubMed]
- Siefritz, F.; Tyree, M.T.; Lovisolo, C.; Schubert, A.; Kaldenhoff, R. PIP1 plasma membrane aquaporins in tobacco: From cellular effects to function in plants. Plant. Cell 2002, 14, 869–876. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, O.; Reshetnyak, G.; Grondin, A.; Saijo, Y.; Leonhardt, N.; Maurel, C.; Verdoucq, L. Aquaporins facilitate hydrogen peroxide entry into guard cells to mediate ABA- and pathogen-triggered stomatal closure. Proc. Natl. Acad. Sci. USA 2017, 114, 9200–9205. [Google Scholar] [CrossRef] [PubMed]
- Nesverova, V.; Tornroth-Horsefield, S. Phosphorylation-dependent regulation of mammalian aquaporins. Cells 2019, 8, 82. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Chen, L.; Dong, H. Plant aquaporins in infection by and immunity against pathogens - A critical review. Front. Plant. Sci. 2019, 10, 632. [Google Scholar] [CrossRef] [PubMed]
- Murata, K.; Mitsuoka, K.; Hirai, T.; Walz, T.; Agre, P.; Heymann, J.B.; Engel, A.; Fujiyoshi, Y. Structural determinants of water permeation through aquaporin-1. Nature 2000, 407, 599–605. [Google Scholar] [CrossRef]
- Maurel, C.; Verdoucq, L.; Luu, D.T.; Santoni, V. Plant aquaporins: Membrane channels with multiple integrated functions. Annu. Rev. Plant. Biol. 2008, 59, 595–624. [Google Scholar] [CrossRef]
- Ishibashi, K. New members of mammalian aquaporins: AQP10-AQP12. Handb. Exp. Pharm. 2009, 190, 251–262. [Google Scholar]
- Deshmukh, R.K.; Sonah, H.; Belanger, R.R. Plant aquaporins: Genome-wide identification, transcriptomics, proteomics, and advanced analytical tools. Front. Plant. Sci. 2016, 7, 1896. [Google Scholar] [CrossRef]
- Wu, B.; Beitz, E. Aquaporins with selectivity for unconventional permeants. Cell Mol. Life Sci. 2007, 64, 2413–2421. [Google Scholar] [CrossRef]
- Bienert, G.P.; Chaumont, F. Aquaporin-facilitated transmembrane diffusion of hydrogen peroxide. Biochim. Biophys. Acta 2014, 1840, 1596–1604. [Google Scholar] [CrossRef] [PubMed]
- Yusupov, M.; Razzokov, J.; Cordeiro, R.M.; Bogaerts, A. Transport of reactive oxygen and nitrogen species across aquaporin: A molecular level picture. Oxid. Med. Cell Longev. 2019, 2019, 2930504. [Google Scholar] [CrossRef] [PubMed]
- Uehlein, N.; Lovisolo, C.; Siefritz, F.; Kaldenhoff, R. The tobacco aquaporin NtAQP1 is a membrane CO2 pore with physiological functions. Nature 2003, 425, 734–737. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, R.; Li, D.; Jia, X.; Zhou, D.; Li, J.; Lyi, S.M.; Hou, S.; Huang, Y.; Kochian, L.V.; et al. NIP1;2 is a plasma membrane-localized transporter mediating aluminum uptake, translocation, and tolerance in Arabidopsis. Proc. Natl. Acad. Sci. USA 2017, 114, 5047–5052. [Google Scholar] [CrossRef] [PubMed]
- Zwiazek, J.J.; Xu, H.; Tan, X.; Navarro-Rodenas, A.; Morte, A. Significance of oxygen transport through aquaporins. Sci. Rep. 2017, 7, 40411. [Google Scholar] [CrossRef]
- Yool, A.J.; Campbell, E.M. Structure, function and translational relevance of aquaporin dual water and ion channels. Mol. Asp. Med. 2012, 33, 553–561. [Google Scholar] [CrossRef]
- Miller, E.W.; Dickinson, B.C.; Chang, C.J. Aquaporin-3 mediates hydrogen peroxide uptake to regulate downstream intracellular signaling. Proc. Natl. Acad. Sci. USA 2010, 107, 15681–15686. [Google Scholar] [CrossRef]
- Al Ghouleh, I.; Frazziano, G.; Rodriguez, A.I.; Csanyi, G.; Maniar, S.; St Croix, C.M.; Kelley, E.E.; Egana, L.A.; Song, G.J.; Bisello, A.; et al. Aquaporin 1, Nox1, and Ask1 mediate oxidant-induced smooth muscle cell hypertrophy. Cardiovasc. Res. 2013, 97, 134–142. [Google Scholar] [CrossRef]
- Medrano-Fernandez, I.; Bestetti, S.; Bertolotti, M.; Bienert, G.P.; Bottino, C.; Laforenza, U.; Rubartelli, A.; Sitia, R. Stress regulates Aquaporin-8 permeability to impact cell growth and survival. Antioxid. Redox Signal. 2016, 24, 1031–1044. [Google Scholar] [CrossRef]
- Madeira, A.; Moura, T.F.; Soveral, G. Aquaglyceroporins: Implications in adipose biology and obesity. Cell Mol. Life Sci. 2015, 72, 759–771. [Google Scholar] [CrossRef]
- Verkman, A.S. More than just water channels: Unexpected cellular roles of aquaporins. J. Cell Sci. 2005, 118, 3225–3232. [Google Scholar] [CrossRef] [PubMed]
- Verkman, A.S.; Anderson, M.O.; Papadopoulos, M.C. Aquaporins: Important but elusive drug targets. Nat. Rev. Drug Discov. 2014, 13, 259–277. [Google Scholar] [CrossRef] [PubMed]
- Tanghe, A.; Van Dijck, P.; Thevelein, J.M. Why do microorganisms have aquaporins? Trends Microbiol. 2006, 14, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Hohmann, I.; Bill, R.M.; Kayingo, I.; Prior, B.A. Microbial MIP channels. Trends Microbiol. 2000, 8, 33–38. [Google Scholar] [CrossRef]
- Tong, H.; Wang, X.; Dong, Y.; Hu, Q.; Zhao, Z.; Zhu, Y.; Dong, L.; Bai, F.; Dong, X. A Streptococcus aquaporin acts as peroxiporin for efflux of cellular hydrogen peroxide and alleviation of oxidative stress. J. Biol. Chem. 2019, 294, 4583–4595. [Google Scholar] [CrossRef]
- Calamita, G.; Bishai, W.R.; Preston, G.M.; Guggino, W.B.; Agre, P. Molecular cloning and characterization of AqpZ, a water channel from Escherichia coli. J. Biol. Chem. 1995, 270, 29063–29066. [Google Scholar] [CrossRef]
- Sweet, G.; Gandor, C.; Voegele, R.; Wittekindt, N.; Beuerle, J.; Truniger, V.; Lin, E.C.; Boos, W. Glycerol facilitator of Escherichia coli: Cloning of glpF and identification of the glpF product. J. Bacteriol. 1990, 172, 424–430. [Google Scholar] [CrossRef]
- Park, J.H.; Saier, M.H., Jr. Phylogenetic characterization of the MIP family of transmembrane channel proteins. J. Membr. Biol. 1996, 153, 171–180. [Google Scholar] [CrossRef]
- Savage, D.F.; Egea, P.F.; Robles-Colmenares, Y.; O’Connell, J.D.; Stroud, R.M. Architecture and selectivity in aquaporins: 2.5 A X-ray structure of aquaporin Z. PLoS Biol. 2003, 1, E72. [Google Scholar] [CrossRef]
- Fu, D.; Libson, A.; Miercke, L.J.; Weitzman, C.; Nollert, P.; Krucinski, J.; Stroud, R.M. Structure of a glycerol-conducting channel and the basis for its selectivity. Science 2000, 290, 481–486. [Google Scholar] [CrossRef]
- Froger, A.; Tallur, B.; Thomas, D.; Delamarche, C. Prediction of functional residues in water channels and related proteins. Protein Sci. 1998, 7, 1458–1468. [Google Scholar] [CrossRef] [PubMed]
- Lagree, V.; Froger, A.; Deschamps, S.; Hubert, J.F.; Delamarche, C.; Bonnec, G.; Thomas, D.; Gouranton, J.; Pellerin, I. Switch from an aquaporin to a glycerol channel by two amino acids substitution. J. Biol. Chem. 1999, 274, 6817–6819. [Google Scholar] [CrossRef]
- Jung, J.S.; Preston, G.M.; Smith, B.L.; Guggino, W.B.; Agre, P. Molecular structure of the water channel through aquaporin CHIP. The hourglass model. J. Biol. Chem. 1994, 269, 14648–14654. [Google Scholar] [PubMed]
- Borgnia, M.J.; Kozono, D.; Calamita, G.; Maloney, P.C.; Agre, P. Functional reconstitution and characterization of AqpZ, the E. coli water channel protein. J. Mol. Biol. 1999, 291, 1169–1179. [Google Scholar] [CrossRef] [PubMed]
- Kozono, D.; Ding, X.; Iwasaki, I.; Meng, X.; Kamagata, Y.; Agre, P.; Kitagawa, Y. Functional expression and characterization of an archaeal aquaporin. AqpM from methanothermobacter marburgensis. J. Biol. Chem. 2003, 278, 10649–10656. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M.O.; Mouritsen, O.G. Single-channel water permeabilities of Escherichia coli aquaporins AqpZ and GlpF. Biophys. J. 2006, 90, 2270–2284. [Google Scholar] [CrossRef] [PubMed]
- de Groot, B.L.; Grubmuller, H. Water permeation across biological membranes: Mechanism and dynamics of aquaporin-1 and GlpF. Science 2001, 294, 2353–2357. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M.O.; Tajkhorshid, E.; Schulten, K. The mechanism of glycerol conduction in aquaglyceroporins. Structure 2001, 9, 1083–1093. [Google Scholar] [CrossRef]
- Hub, J.S.; de Groot, B.L. Mechanism of selectivity in aquaporins and aquaglyceroporins. Proc. Natl. Acad. Sci. USA 2008, 105, 1198–1203. [Google Scholar] [CrossRef]
- Mathai, J.C.; Missner, A.; Kugler, P.; Saparov, S.M.; Zeidel, M.L.; Lee, J.K.; Pohl, P. No facilitator required for membrane transport of hydrogen sulfide. Proc. Natl. Acad. Sci. USA 2009, 106, 16633–16638. [Google Scholar] [CrossRef]
- Lee, J.K.; Kozono, D.; Remis, J.; Kitagawa, Y.; Agre, P.; Stroud, R.M. Structural basis for conductance by the archaeal aquaporin AqpM at 1.68 A. Proc. Natl. Acad. Sci. USA 2005, 102, 18932–18937. [Google Scholar] [CrossRef] [PubMed]
- Araya-Secchi, R.; Garate, J.A.; Holmes, D.S.; Perez-Acle, T. Molecular dynamics study of the archaeal aquaporin AqpM. BMC Genom. 2011, 12, S8. [Google Scholar] [CrossRef] [PubMed]
- Beitz, E.; Becker, D.; von Bulow, J.; Conrad, C.; Fricke, N.; Geadkaew, A.; Krenc, D.; Song, J.; Wree, D.; Wu, B. In vitro analysis and modification of aquaporin pore selectivity. Handb. Exp. Pharm. 2009, 190, 77–92. [Google Scholar]
- Madeira, A.; Moura, T.F.; Soveral, G. Detecting aquaporin function and regulation. Front. Chem. 2016, 4, 3. [Google Scholar] [CrossRef]
- Pettersson, N.; Hagstrom, J.; Bill, R.M.; Hohmann, S. Expression of heterologous aquaporins for functional analysis in Saccharomyces cerevisiae. Curr. Genet. 2006, 50, 247–255. [Google Scholar] [CrossRef]
- Zeidel, M.L.; Ambudkar, S.V.; Smith, B.L.; Agre, P. Reconstitution of functional water channels in liposomes containing purified red cell CHIP28 protein. Biochemistry 1992, 31, 7436–7440. [Google Scholar] [CrossRef]
- Verkman, A.S. Water permeability measurement in living cells and complex tissues. J. Membr. Biol. 2000, 173, 73–87. [Google Scholar] [CrossRef]
- Farinas, J.; Verkman, A.S. Cell volume and plasma membrane osmotic water permeability in epithelial cell layers measured by interferometry. Biophys. J. 1996, 71, 3511–3522. [Google Scholar] [CrossRef]
- Levin, M.H.; de la Fuente, R.; Verkman, A.S. Urearetics: A small molecule screen yields nanomolar potency inhibitors of urea transporter UT-B. FASEB J. 2007, 21, 551–563. [Google Scholar] [CrossRef]
- Verkman, A.S.; Dix, J.A.; Seifter, J.L. Water and urea transport in renal microvillus membrane vesicles. Am. J. Physiol. 1985, 248, F650–F655. [Google Scholar] [CrossRef]
- Soveral, G.; Macey, R.I.; Moura, T.F. Water permeability of brush border membrane vesicles from kidney proximal tubule. J. Membr. Biol. 1997, 158, 219–228. [Google Scholar] [CrossRef] [PubMed]
- Soveral, G.; Madeira, A.; Loureiro-Dias, M.C.; Moura, T.F. Membrane tension regulates water transport in yeast. Biochim. Biophys. Acta 2008, 1778, 2573–2579. [Google Scholar] [CrossRef] [PubMed]
- Mallo, R.C.; Ashby, M.T. AqpZ-mediated water permeability in Escherichia coli measured by stopped-flow spectroscopy. J. Bacteriol. 2006, 188, 820–822. [Google Scholar] [CrossRef] [PubMed]
- Madeira, A.; Camps, M.; Zorzano, A.; Moura, T.F.; Soveral, G. Biophysical assessment of human aquaporin-7 as a water and glycerol channel in 3T3-L1 adipocytes. PLoS ONE 2013, 8, e83442. [Google Scholar] [CrossRef]
- Solenov, E.; Watanabe, H.; Manley, G.T.; Verkman, A.S. Sevenfold-reduced osmotic water permeability in primary astrocyte cultures from AQP-4-deficient mice, measured by a fluorescence quenching method. Am. J. Physiol. Cell Physiol. 2004, 286, C426–C432. [Google Scholar] [CrossRef]
- Soveral, G.; Madeira, A.; Loureiro-Dias, M.C.; Moura, T.F. Water transport in intact yeast cells as assessed by fluorescence self-quenching. Appl. Env. Microbiol. 2007, 73, 2341–2343. [Google Scholar] [CrossRef]
- Galietta, L.J.; Haggie, P.M.; Verkman, A.S. Green fluorescent protein-based halide indicators with improved chloride and iodide affinities. FEBS Lett. 2001, 499, 220–224. [Google Scholar] [CrossRef]
- Heller, K.B.; Lin, E.C.; Wilson, T.H. Substrate specificity and transport properties of the glycerol facilitator of Escherichia coli. J. Bacteriol. 1980, 144, 274–278. [Google Scholar]
- Sanno, Y.; Wilson, T.H.; Lin, E.C. Control of permeation to glycerol in cells of Escherichia coli. Biochem. Biophys. Res. Commun. 1968, 32, 344–349. [Google Scholar] [CrossRef]
- Maurel, C.; Reizer, J.; Schroeder, J.I.; Chrispeels, M.J.; Saier, M.H., Jr. Functional characterization of the Escherichia coli glycerol facilitator, GlpF, in Xenopus oocytes. J. Biol. Chem. 1994, 269, 11869–11872. [Google Scholar]
- Will, Y.; Hynes, J.; Ogurtsov, V.I.; Papkovsky, D.B. Analysis of mitochondrial function using phosphorescent oxygen-sensitive probes. Nat. Protoc. 2006, 1, 2563–2572. [Google Scholar] [CrossRef] [PubMed]
- Belousov, V.V.; Fradkov, A.F.; Lukyanov, K.A.; Staroverov, D.B.; Shakhbazov, K.S.; Terskikh, A.V.; Lukyanov, S. Genetically encoded fluorescent indicator for intracellular hydrogen peroxide. Nat. Methods 2006, 3, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.B.; Barker, K.A.; Huang, B.K.; Sikes, H.D. In-depth characterization of the fluorescent signal of HyPer, a probe for hydrogen peroxide, in bacteria exposed to external oxidative stress. J. Microbiol. Methods 2014, 106, 33–39. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bilan, D.S.; Belousov, V.V. In vivo imaging of hydrogen peroxide with HyPer probes. Antioxid. Redox Signal. 2018, 29, 569–584. [Google Scholar] [CrossRef] [PubMed]
- Oparka, M.; Walczak, J.; Malinska, D.; van Oppen, L.; Szczepanowska, J.; Koopman, W.J.H.; Wieckowski, M.R. Quantifying ROS levels using CM-H2DCFDA and HyPer. Methods 2016, 109, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Bienert, G.P.; Moller, A.L.; Kristiansen, K.A.; Schulz, A.; Moller, I.M.; Schjoerring, J.K.; Jahn, T.P. Specific aquaporins facilitate the diffusion of hydrogen peroxide across membranes. J. Biol. Chem. 2007, 282, 1183–1192. [Google Scholar] [CrossRef] [PubMed]
- Almasalmeh, A.; Krenc, D.; Wu, B.; Beitz, E. Structural determinants of the hydrogen peroxide permeability of aquaporins. FEBS J. 2014, 281, 647–656. [Google Scholar] [CrossRef]
- Tong, H.; Chen, W.; Merritt, J.; Qi, F.; Shi, W.; Dong, X. Streptococcus oligofermentans inhibits Streptococcus mutans through conversion of lactic acid into inhibitory H2O2: A possible counteroffensive strategy for interspecies competition. Mol. Microbiol. 2007, 63, 872–880. [Google Scholar] [CrossRef]
- Liu, L.; Tong, H.; Dong, X. Function of the pyruvate oxidase-lactate oxidase cascade in interspecies competition between Streptococcus oligofermentans and Streptococcus mutans. Appl. Environ. Microbiol. 2012, 78, 2120–2127. [Google Scholar] [CrossRef]
- Delamarche, C.; Thomas, D.; Rolland, J.P.; Froger, A.; Gouranton, J.; Svelto, M.; Agre, P.; Calamita, G. Visualization of AqpZ-mediated water permeability in Escherichia coli by cryoelectron microscopy. J. Bacteriol. 1999, 181, 4193–4197. [Google Scholar]
- Calamita, G.; Kempf, B.; Bonhivers, M.; Bishai, W.R.; Bremer, E.; Agre, P. Regulation of the Escherichia coli water channel gene aqpZ. Proc. Natl. Acad. Sci. USA 1998, 95, 3627–3631. [Google Scholar] [CrossRef] [PubMed]
- Voegele, R.T.; Sweet, G.D.; Boos, W. Glycerol kinase of Escherichia coli is activated by interaction with the glycerol facilitator. J. Bacteriol. 1993, 175, 1087–1094. [Google Scholar] [CrossRef] [PubMed]
- Schweizer, H.P.; Jump, R.; Po, C. Structure and gene-polypeptide relationships of the region encoding glycerol diffusion facilitator (glpF) and glycerol kinase (glpK) of Pseudomonas aeruginosa. Microbiology 1997, 143, 1287–1297. [Google Scholar] [CrossRef] [PubMed]
- Bienert, G.P.; Desguin, B.; Chaumont, F.; Hols, P. Channel-mediated lactic acid transport: A novel function for aquaglyceroporins in bacteria. Biochem. J. 2013, 454, 559–570. [Google Scholar] [CrossRef]
- Kreida, S.; Tornroth-Horsefield, S. Structural insights into aquaporin selectivity and regulation. Curr. Opin. Struct. Biol. 2015, 33, 126–134. [Google Scholar] [CrossRef]
- Roche, J.V.; Tornroth-Horsefield, S. Aquaporin protein-protein interactions. Int. J. Mol. Sci. 2017, 18, 2255. [Google Scholar] [CrossRef]
- Tornroth-Horsefield, S.; Hedfalk, K.; Fischer, G.; Lindkvist-Petersson, K.; Neutze, R. Structural insights into eukaryotic aquaporin regulation. FEBS Lett. 2010, 584, 2580–2588. [Google Scholar] [CrossRef]
- Mosca, A.F.; de Almeida, A.; Wragg, D.; Martins, A.P.; Sabir, F.; Leoni, S.; Moura, T.F.; Prista, C.; Casini, A.; Soveral, G. Molecular basis of Aquaporin-7 permeability regulation by pH. Cells 2018, 7, 207. [Google Scholar] [CrossRef]
- Zelenina, M.; Bondar, A.A.; Zelenin, S.; Aperia, A. Nickel and extracellular acidification inhibit the water permeability of human aquaporin-3 in lung epithelial cells. J. Biol. Chem. 2003, 278, 30037–30043. [Google Scholar] [CrossRef]
- Leitao, L.; Prista, C.; Loureiro-Dias, M.C.; Moura, T.F.; Soveral, G. The grapevine tonoplast aquaporin TIP2;1 is a pressure gated water channel. Biochem. Biophys. Res. Commun. 2014, 450, 289–294. [Google Scholar] [CrossRef]
- Jiang, J.; Daniels, B.V.; Fu, D. Crystal structure of AqpZ tetramer reveals two distinct Arg-189 conformations associated with water permeation through the narrowest constriction of the water-conducting channel. J. Biol. Chem. 2006, 281, 454–460. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Xie, H.; Wang, L.; Shen, Y.; Chen, W.; Song, B.; Zhang, Z.; Zheng, A.; Lin, Q.; Fu, R.; et al. Gating Mechanism of Aquaporin Z in synthetic bilayers and native membranes revealed by solid-state NMR spectroscopy. J. Am. Chem. Soc. 2018, 140, 7885–7895. [Google Scholar] [CrossRef] [PubMed]
- Trefz, M.; Keller, R.; Vogt, M.; Schneider, D. The GlpF residue Trp219 is part of an amino-acid cluster crucial for aquaglyceroporin oligomerization and function. Biochim. Biophys. Acta 2018, 1860, 887–894. [Google Scholar] [CrossRef] [PubMed]
- Klein, N.; Hellmann, N.; Schneider, D. Anionic lipids modulate the activity of the aquaglyceroporin GlpF. Biophys. J. 2015, 109, 722–731. [Google Scholar] [CrossRef][Green Version]
- Klein, N.; Trefz, M.; Schneider, D. Covalently linking oligomerization-impaired GlpF protomers does not completely re-establish wild-Type channel activity. Int. J. Mol. Sci. 2019, 20, 927. [Google Scholar] [CrossRef]
- Laganowsky, A.; Reading, E.; Allison, T.M.; Ulmschneider, M.B.; Degiacomi, M.T.; Baldwin, A.J.; Robinson, C.V. Membrane proteins bind lipids selectively to modulate their structure and function. Nature 2014, 510, 172–175. [Google Scholar] [CrossRef]
- Schmidt, V.; Sidore, M.; Bechara, C.; Duneau, J.P.; Sturgis, J.N. The lipid environment of Escherichia coli Aquaporin Z. Biochim. Biophys. Acta. 2019, 1861, 431–440. [Google Scholar] [CrossRef]
- Agre, P. Aquaporin water channels (Nobel Lecture). Angew. Chem. Int. Ed. Engl. 2004, 43, 4278–4290. [Google Scholar] [CrossRef]
- Gotfryd, K.; Mosca, A.F.; Missel, J.W.; Truelsen, S.F.; Wang, K.; Spulber, M.; Krabbe, S.; Helix-Nielsen, C.; Laforenza, U.; Soveral, G.; et al. Human adipose glycerol flux is regulated by a pH gate in AQP10. Nat. Commun. 2018, 9, 4749. [Google Scholar] [CrossRef]
- Ishibashi, K.; Tanaka, Y.; Morishita, Y. The role of mammalian superaquaporins inside the cell. Biochim. Biophys. Acta 2014, 1840, 1507–1512. [Google Scholar] [CrossRef]
| Organism | Gene Number | Uniprot Accession Number | Transported Substrate | Heterologously Expressed Host | Expression Sites in Itself | Suggested Physiological Functions | References |
|---|---|---|---|---|---|---|---|
| Eukaryote | |||||||
| Human | |||||||
| AQP0 | 4284 | P30301 | Water | Not determined | Lens | congenital cataract (loss of function mutation) | [32,98] |
| AQP1 | 358 | P29972 | Water, O2, H2O2, CO2, NO | Xenopus oocytes; yeast; | Renal tubules, red blood cells | Water permeability | [1,25,32,98] |
| AQP2 | 359 | P41181 | Water | Not determined | Renal collecting duct | nephrogenic diabetes insipidus (loss of function mutation) | [32,98] |
| AQP3 | 360 | Q92482 | Glycerol, urea, H2O2, water | Not determined | Renal collecting duct, adipocytes | Glycerol permeability | [11,27,32,98] |
| AQP4 | 361 | P55087 | Water, CO2, NO, O2 | Not determined | Astrocytes | Cerebrospinal fluid flux | [32,98] |
| AQP5 | 362 | P55064 | Water, H2O2, CO2 | Not dermined | Glandular tissues such as salivary gland | Saliva secretion | [32,98] |
| AQP6 | 363 | Q13520 | Water, nitrate | Not determined | Intracellular vesicles in renal collecting duct | Acid secretion | [32,98] |
| AQP7 | 364 | O14520 | Glycerol, urea, water | Not determined | Fat cells, renal proximal tubule | Glycerol permeability | [11,32,98] |
| AQP8 | 343 | O94778 | Water, H2O2 | Yeast | Intestinal epithelium | None identified | [27,32,76,98] |
| AQP9 | 366 | O43315 | Glycerol, urea, H2O2, water | Not determined | Hepatocytes, erythrocytes | Glycerol permeability | [11,32,98] |
| AQP10 | 89872 | Q96PS8 | Glycerol, urea, water | Protepolymersome, Yeast | adipose tissue | None identified | [11,98,99] |
| AQP11 | 282679 | Q8NBQ7 | Water | Proteoliposome | Liver, testis | None identified | [32,100] |
| AQP12 | 375318 | Q8IXF9 | Unknown | Not dermined | Exocrine pancreas | None identified | [32,100] |
| Plant | |||||||
| Nicotiana tabacum | NtAQP1 | Q9ZR68 | CO2 | Xenopus oocytes | Leaf | Increasing leaf growth | [23] |
| PIP1;3 | Q40595 | O2 | Yeast | Root | Increase of ATP levels in the apical root segments | [25] | |
| Arabidopsis thaliana | NIP1;2 | Q8LFP7 | Aluminum-malate | Yeast | Root | Aluminum uptake, translocation and tolerance | [24] |
| PIP2;1 | P43286 | H2O2 | Not determined | Guard cell | Stomatal closure | [13] | |
| Prokaryote | |||||||
| Bacteria | |||||||
| Escherichia coli | aqpZ | P60844 | Water | Xenopus oocytes | Cellular membrane | Osmostic stress resistance | [39,44,80] |
| glpF | P0AER0 | Glycerol | Xenopus oocytes | Cellular membrane | Growth on low concentration glycerol | [40,70] | |
| Streptococcus oligofermentans | aqpA | I872_01445 | H2O2 | E. coli, Yeast | Cellular membrane | H2O2 detoxification and interspecies competition | [35] |
| Lactobacillus plantarum | glpF1 | F9UST3 | Lactic acid, urea, H2O2 | Xenopus oocytes, Yeast | Cellular membrane | Lactic acid stress tolerance | [84] |
| glpF2 | F9USY3 | Water, glycerol, dihydroxyacetone, H2O2 | Xenopus oocytes, Yeast | Cellular membrane | None identified | [84] | |
| glpF3 | F9UTW9 | Water, glycerol, dihydroxyacetone, H2O2 | Xenopus oocytes, Yeast | Cellular membrane | None identified | [84] | |
| glpF4 | F9UMX3 | Water, glycerol, dihydroxyacetone, lactic acid, urea, H2O2 | Xenopus oocytes, Yeast | Cellular membrane | Lactic acid stress tolerance | [84] | |
| Pseudomonas aeruginosa | glpF | Q51389 | Glycerol | Not determined | None identified | Growth on glycerol | [83] |
| Archaea | |||||||
| Methanothermobacter marburgensis | aqpM | Q9C4Z5 | Water, glycerol | Proteoliposomes | Cellular membrane | None identified | [45,51,52] |
| Archaeoglobus fulgidus | AfAQP | O28846 | Water | Proteoliposomes | Cellular membrane | None identified | [50] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tong, H.; Hu, Q.; Zhu, L.; Dong, X. Prokaryotic Aquaporins. Cells 2019, 8, 1316. https://doi.org/10.3390/cells8111316
Tong H, Hu Q, Zhu L, Dong X. Prokaryotic Aquaporins. Cells. 2019; 8(11):1316. https://doi.org/10.3390/cells8111316
Chicago/Turabian StyleTong, Huichun, Qingqing Hu, Lin Zhu, and Xiuzhu Dong. 2019. "Prokaryotic Aquaporins" Cells 8, no. 11: 1316. https://doi.org/10.3390/cells8111316
APA StyleTong, H., Hu, Q., Zhu, L., & Dong, X. (2019). Prokaryotic Aquaporins. Cells, 8(11), 1316. https://doi.org/10.3390/cells8111316






