Wnt Signaling in Neural Crest Ontogenesis and Oncogenesis
Abstract
1. Introduction
2. Wnt Signaling Pathways
2.1. β-Catenin-Dependent Canonical Wnt Signaling Pathway
2.2. β-Catenin-Independent Non-Canonical Wnt Signaling Pathways
3. Wnt Signaling in NPB Formation and NC Induction
3.1. Wnt Signaling in NPB Formation
3.2. Wnt Signaling in NC Induction and Specification
3.3. Crosstalk of Wnt Signaling with Upstream Modulators in NC Induction
4. Wnt Signaling and Crosstalk with Other Signaling Pathways in NC Delamination and EMT
4.1. Wnt Signaling and Crosstalk in Trunk NC Delamination and EMT
4.2. Wnt Signaling and Crosstalk in Cranial NC Delamination and EMT
5. Wnt Signaling in NC Migration
5.1. Non-Canonical Wnt Signaling in NC Migration
5.2. Canonical Wnt Signaling in NC Migration
6. Wnt in NC Multipotency and Fate Determination
6.1. Wnt Signaling and Crosstalk in Maintaining NC Multipotency
6.2. Wnt Signaling in NC Fate Specification and Differentiation
7. Wnt Signaling in NC-Derived Cancers
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bronner, M.E. Formation and migration of neural crest cells in the vertebrate embryo. Histochem. Cell Biol. 2012, 138, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Neural crest cells: Evolution, Development and Disease; Trainor, P.A., Ed.; Academic Press: London, UK, 2014; pp. 1–455. [Google Scholar]
- Bae, C.J.; Saint-Jeannet, J.P. Induction and specification of neural crest cells: Extracellular signals and transcriptional switches. In Neural crest cells: Evolution, Development and Disease; Trainor, P.A., Ed.; Academic Press: London, UK, 2014; pp. 27–49. [Google Scholar]
- Bronner, M.E.; Simoes-Costa, M. The Neural Crest Migrating into the Twenty-First Century. Curr. Top. Dev. Biol. 2016, 116, 115–134. [Google Scholar] [PubMed]
- Szabo, A.; Mayor, R. Mechanisms of Neural Crest Migration. Annu. Rev. Genet. 2018, 52, 43–63. [Google Scholar] [CrossRef] [PubMed]
- Mayor, R.; Theveneau, E. The neural crest. Development 2013, 140, 2247–2251. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, S.; Diaz, R.; Trainor, P.A. Signals and switches in Mammalian neural crest cell differentiation. Cold Spring Harb. Perspect. Biol. 2013, 5. [Google Scholar] [CrossRef] [PubMed]
- Simoes-Costa, M.; Bronner, M.E. Insights into neural crest development and evolution from genomic analysis. Genome Res. 2013, 23, 1069–1080. [Google Scholar] [CrossRef] [PubMed]
- Lumb, R.; Buckberry, S.; Secker, G.; Lawrence, D.; Schwarz, Q. Transcriptome profiling reveals expression signatures of cranial neural crest cells arising from different axial levels. BMC Dev. Biol. 2017, 17, 5. [Google Scholar] [CrossRef]
- La Noce, M.; Mele, L.; Tirino, V.; Paino, F.; De Rosa, A.; Naddeo, P.; Papagerakis, P.; Papaccio, G.; Desiderio, V. Neural crest stem cell population in craniomaxillofacial development and tissue repair. Eur. Cell. Mater. 2014, 28, 348–357. [Google Scholar] [CrossRef]
- Knight, R.D.; Schilling, T.F. Cranial neural crest and development of the head skeleton. Adv. Exp. Med. Biol. 2006, 589, 120–133. [Google Scholar]
- Kuo, B.R.; Erickson, C.A. Vagal neural crest cell migratory behavior: A transition between the cranial and trunk crest. Dev. Dyn. 2011, 240, 2084–2100. [Google Scholar] [CrossRef]
- Hutchins, E.J.; Kunttas, E.; Piacentino, M.L.; Howard, A.G.A.t.; Bronner, M.E.; Uribe, R.A. Migration and diversification of the vagal neural crest. Dev. Biol. 2018, 444, S98–S109. [Google Scholar] [CrossRef] [PubMed]
- Ezin, A.M.; Sechrist, J.W.; Zah, A.; Bronner, M.; Fraser, S.E. Early regulative ability of the neuroepithelium to form cardiac neural crest. Dev. Biol. 2011, 349, 238–249. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Plein, A.; Fantin, A.; Ruhrberg, C. Neural crest cells in cardiovascular development. Curr. Top. Dev. Biol. 2015, 111, 183–200. [Google Scholar] [PubMed]
- Jiang, X.; Rowitch, D.H.; Soriano, P.; McMahon, A.P.; Sucov, H.M. Fate of the mammalian cardiac neural crest. Development 2000, 127, 1607–1616. [Google Scholar] [PubMed]
- Krotoski, D.M.; Fraser, S.E.; Bronner-Fraser, M. Mapping of neural crest pathways in Xenopus laevis using inter- and intra-specific cell markers. Dev. Biol. 1988, 127, 119–132. [Google Scholar] [CrossRef]
- Vega-Lopez, G.A.; Cerrizuela, S.; Aybar, M.J. Trunk neural crest cells: Formation, migration and beyond. Int. J. Dev. Biol. 2017, 61, 5–15. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, Y.; Sipp, D.; Enomoto, H. Tissue interactions in neural crest cell development and disease. Science 2013, 341, 860–863. [Google Scholar] [CrossRef]
- Vega-Lopez, G.A.; Cerrizuela, S.; Tribulo, C.; Aybar, M.J. Neurocristopathies: New insights 150 years after the neural crest discovery. Dev. Biol. 2018, 444, S110–S143. [Google Scholar] [CrossRef]
- Trainor, P.A. Craniofacial birth defects: The role of neural crest cells in the etiology and pathogenesis of Treacher Collins syndrome and the potential for prevention. Am. J. Med. Genet. A 2010, 152, 2984–2994. [Google Scholar] [CrossRef]
- Etchevers, H.C.; Dupin, E.; Le Douarin, N.M. The diverse neural crest: From embryology to human pathology. Development 2019, 146, dev169821. [Google Scholar] [CrossRef]
- Amiel, J.; Sproat-Emison, E.; Garcia-Barcelo, M.; Lantieri, F.; Burzynski, G.; Borrego, S.; Pelet, A.; Arnold, S.; Miao, X.; Griseri, P.; et al. Hirschsprung disease, associated syndromes and genetics: A review. J. Med. Genet. 2008, 45, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Langer, J.C. Hirschsprung disease. Curr. Opin. Pediatr. 2013, 25, 368–374. [Google Scholar] [CrossRef] [PubMed]
- Epstein, J.A. Developing models of DiGeorge syndrome. Trends Genet. 2001, 17, S13–S17. [Google Scholar] [CrossRef]
- Schulz, Y.; Wehner, P.; Opitz, L.; Salinas-Riester, G.; Bongers, E.M.; van Ravenswaaij-Arts, C.M.; Wincent, J.; Schoumans, J.; Kohlhase, J.; Borchers, A.; et al. CHD7, the gene mutated in CHARGE syndrome, regulates genes involved in neural crest cell guidance. Hum. Genet. 2014, 133, 997–1009. [Google Scholar] [CrossRef] [PubMed]
- Fujita, K.; Ogawa, R.; Kawawaki, S.; Ito, K. Roles of chromatin remodelers in maintenance mechanisms of multipotency of mouse trunk neural crest cells in the formation of neural crest-derived stem cells. Mech. Dev. 2014, 133, 126–145. [Google Scholar] [CrossRef] [PubMed]
- Pauli, S.; Bajpai, R.; Borchers, A. CHARGEd with neural crest defects. Am. J. Med. Genet. C Semin. Med. Genet. 2017, 175, 478–486. [Google Scholar] [CrossRef] [PubMed]
- Trainor, P.; Krumlauf, R. Development. Riding the crest of the Wnt signaling wave. Science 2002, 297, 781–783. [Google Scholar] [CrossRef][Green Version]
- Schmidt, C.; Patel, K. Wnts and the neural crest. Anat. Embryol. 2005, 209, 349–355. [Google Scholar] [CrossRef]
- Yanfeng, W.; Saint-Jeannet, J.P.; Klein, P.S. Wnt-frizzled signaling in the induction and differentiation of the neural crest. Bioessays 2003, 25, 317–325. [Google Scholar] [CrossRef]
- Gomez, G.A.; Prasad, M.S.; Sandhu, N.; Shelar, P.B.; Leung, A.W.; Garcia-Castro, M.I. Human neural crest induction by temporal modulation of WNT activation. Dev. Biol. 2019, 449, 99–106. [Google Scholar] [CrossRef]
- Jones, N.C.; Trainor, P.A. Role of morphogens in neural crest cell determination. J. Neurobiol. 2005, 64, 388–404. [Google Scholar] [CrossRef]
- Devotta, A.; Hong, C.S.; Saint-Jeannet, J.P. Dkk2 promotes neural crest specification by activating Wnt/beta-catenin signaling in a GSK3beta independent manner. Elife 2018, 7, e34404. [Google Scholar] [CrossRef]
- Hong, C.S.; Saint-Jeannet, J.P. The b-HLH transcription factor Hes3 participates in neural plate border formation by interfering with Wnt/beta-catenin signaling. Dev. Biol. 2018, 442, 162–172. [Google Scholar] [CrossRef]
- Cheung, M.; Chaboissier, M.C.; Mynett, A.; Hirst, E.; Schedl, A.; Briscoe, J. The transcriptional control of trunk neural crest induction, survival, and delamination. Dev. Cell 2005, 8, 179–192. [Google Scholar] [CrossRef]
- De Calisto, J.; Araya, C.; Marchant, L.; Riaz, C.F.; Mayor, R. Essential role of non-canonical Wnt signalling in neural crest migration. Development 2005, 132, 2587–2597. [Google Scholar] [CrossRef]
- Ossipova, O.; Kerney, R.; Saint-Jeannet, J.P.; Sokol, S.Y. Regulation of neural crest development by the formin family protein Daam1. Genesis 2018, 56, e23108. [Google Scholar] [CrossRef]
- Hari, L.; Miescher, I.; Shakhova, O.; Suter, U.; Chin, L.; Taketo, M.; Richardson, W.D.; Kessaris, N.; Sommer, L. Temporal control of neural crest lineage generation by Wnt/beta-catenin signaling. Development 2012, 139, 2107–2117. [Google Scholar] [CrossRef]
- Hari, L.; Brault, V.; Kleber, M.; Lee, H.Y.; Ille, F.; Leimeroth, R.; Paratore, C.; Suter, U.; Kemler, R.; Sommer, L. Lineage-specific requirements of beta-catenin in neural crest development. J. Cell Biol. 2002, 159, 867–880. [Google Scholar] [CrossRef]
- Nusse, R.; Clevers, H. Wnt/beta-Catenin Signaling, Disease, and Emerging Therapeutic Modalities. Cell 2017, 169, 985–999. [Google Scholar] [CrossRef]
- Bhanot, P.; Brink, M.; Samos, C.H.; Hsieh, J.C.; Wang, Y.; Macke, J.P.; Andrew, D.; Nathans, J.; Nusse, R. A new member of the frizzled family from Drosophila functions as a Wingless receptor. Nature 1996, 382, 225–230. [Google Scholar] [CrossRef]
- Janda, C.Y.; Waghray, D.; Levin, A.M.; Thomas, C.; Garcia, K.C. Structural basis of Wnt recognition by Frizzled. Science 2012, 337, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Ke, J.; Xu, H.E.; Williams, B.O. Lipid modification in Wnt structure and function. Curr. Opin. Lipidol. 2013, 24, 129–133. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Bubeck, D.; MacDonald, B.T.; Liang, W.X.; Mao, J.H.; Malinauskas, T.; Llorca, O.; Aricescu, A.R.; Siebold, C.; He, X.; et al. Structural and functional studies of LRP6 ectodomain reveal a platform for Wnt signaling. Dev. Cell 2011, 21, 848–861. [Google Scholar] [CrossRef] [PubMed]
- Chu, M.L.; Ahn, V.E.; Choi, H.J.; Daniels, D.L.; Nusse, R.; Weis, W.I. structural Studies of Wnts and identification of an LRP6 binding site. Structure 2013, 21, 1235–1242. [Google Scholar] [CrossRef] [PubMed]
- Tamai, K.; Semenov, M.; Kato, Y.; Spokony, R.; Liu, C.; Katsuyama, Y.; Hess, F.; Saint-Jeannet, J.P.; He, X. LDL-receptor-related proteins in Wnt signal transduction. Nature 2000, 407, 530–535. [Google Scholar] [CrossRef] [PubMed]
- Li, V.S.; Ng, S.S.; Boersema, P.J.; Low, T.Y.; Karthaus, W.R.; Gerlach, J.P.; Mohammed, S.; Heck, A.J.; Maurice, M.M.; Mahmoudi, T.; et al. Wnt signaling through inhibition of beta-catenin degradation in an intact Axin1 complex. Cell 2012, 149, 1245–1256. [Google Scholar] [CrossRef] [PubMed]
- Cselenyi, C.S.; Jernigan, K.K.; Tahinci, E.; Thorne, C.A.; Lee, L.A.; Lee, E. LRP6 transduces a canonical Wnt signal independently of Axin degradation by inhibiting GSK3’s phosphorylation of beta-catenin. Proc. Natl. Acad. Sci. 2008, 105, 8032–8037. [Google Scholar] [CrossRef] [PubMed]
- Wodarz, A.; Nusse, R. Mechanisms of Wnt signaling in development. Annu. Rev. Cell. Dev. Biol. 1998, 14, 59–88. [Google Scholar] [CrossRef]
- Steinhart, Z.; Angers, S. Wnt signaling in development and tissue homeostasis. Development 2018, 145, dev146589. [Google Scholar] [CrossRef]
- McManus, E.J.; Sakamoto, K.; Armit, L.J.; Ronaldson, L.; Shpiro, N.; Marquez, R.; Alessi, D.R. Role that phosphorylation of GSK3 plays in insulin and Wnt signalling defined by knockin analysis. EMBO J. 2005, 24, 1571–1583. [Google Scholar] [CrossRef]
- Humphries, A.C.; Mlodzik, M. From instruction to output: Wnt/PCP signaling in development and cancer. Curr. Opin. Cell. Biol. 2018, 51, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Mayor, R.; Theveneau, E. The role of the non-canonical Wnt-planar cell polarity pathway in neural crest migration. Biochem. J. 2014, 457, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, A.; Yamamoto, H.; Sato, A. Selective activation mechanisms of Wnt signaling pathways. Trends Cell Biol. 2009, 19, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Simons, M.; Mlodzik, M. Planar cell polarity signaling: From fly development to human disease. Annu. Rev. Genet. 2008, 42, 517–540. [Google Scholar] [CrossRef] [PubMed]
- Ridley, A.J. Life at the leading edge. Cell 2011, 145, 1012–1022. [Google Scholar] [CrossRef]
- De, A. Wnt/Ca2+ signaling pathway: A brief overview. Acta Biochim. Biophys. Sin. 2011, 43, 745–756. [Google Scholar] [CrossRef] [PubMed]
- Kuhl, M.; Sheldahl, L.C.; Malbon, C.C.; Moon, R.T. Ca(2+)/calmodulin-dependent protein kinase II is stimulated by Wnt and Frizzled homologs and promotes ventral cell fates in Xenopus. J. Biol. Chem. 2000, 275, 12701–12711. [Google Scholar] [CrossRef]
- Sheldahl, L.C.; Park, M.; Malbon, C.C.; Moon, R.T. Protein kinase C is differentially stimulated by Wnt and Frizzled homologs in a G-protein-dependent manner. Curr. Biol. 1999, 9, 695–698. [Google Scholar] [CrossRef]
- Lilienbaum, A.; Israel, A. From calcium to NF-kappa B signaling pathways in neurons. Mol. Cell. Biol. 2003, 23, 2680–2698. [Google Scholar] [CrossRef]
- Simoes-Costa, M.; Bronner, M.E. Establishing neural crest identity: A gene regulatory recipe. Development 2015, 142, 242–257. [Google Scholar] [CrossRef]
- Pla, P.; Monsoro-Burq, A.H. The neural border: Induction, specification and maturation of the territory that generates neural crest cells. Dev. Biol. 2018, 444, S36–S46. [Google Scholar] [CrossRef] [PubMed]
- Plouhinec, J.L.; Medina-Ruiz, S.; Borday, C.; Bernard, E.; Vert, J.P.; Eisen, M.B.; Harland, R.M.; Monsoro-Burq, A.H. A molecular atlas of the developing ectoderm defines neural, neural crest, placode, and nonneural progenitor identity in vertebrates. PLoS Biol. 2017, 15, e2004045. [Google Scholar] [CrossRef] [PubMed]
- Pera, E.; Stein, S.; Kessel, M. Ectodermal patterning in the avian embryo: Epidermis versus neural plate. Development 1999, 126, 63–73. [Google Scholar] [PubMed]
- Maharana, S.K.; Schlosser, G. A gene regulatory network underlying the formation of pre-placodal ectoderm in Xenopus laevis. BMC Biol. 2018, 16, 79. [Google Scholar] [CrossRef] [PubMed]
- Masek, J.; Machon, O.; Korinek, V.; Taketo, M.M.; Kozmik, Z. Tcf7l1 protects the anterior neural fold from adopting the neural crest fate. Development 2016, 143, 2206–2216. [Google Scholar] [CrossRef]
- Carmona-Fontaine, C.; Acuna, G.; Ellwanger, K.; Niehrs, C.; Mayor, R. Neural crests are actively precluded from the anterior neural fold by a novel inhibitory mechanism dependent on Dickkopf1 secreted by the prechordal mesoderm. Dev. Biol. 2007, 309, 208–221. [Google Scholar] [CrossRef] [PubMed]
- Hong, C.S.; Saint-Jeannet, J.P. The activity of Pax3 and Zic1 regulates three distinct cell fates at the neural plate border. Mol. Biol. Cell 2007, 18, 2192–2202. [Google Scholar] [CrossRef]
- Nichane, M.; Ren, X.; Souopgui, J.; Bellefroid, E.J. Hairy2 functions through both DNA-binding and non DNA-binding mechanisms at the neural plate border in Xenopus. Dev. Biol. 2008, 322, 368–380. [Google Scholar] [CrossRef]
- Milet, C.; Monsoro-Burq, A.H. Neural crest induction at the neural plate border in vertebrates. Dev. Biol. 2012, 366, 22–33. [Google Scholar] [CrossRef]
- Khudyakov, J.; Bronner-Fraser, M. Comprehensive spatiotemporal analysis of early chick neural crest network genes. Dev. Dyn. 2009, 238, 716–723. [Google Scholar] [CrossRef]
- Bronner, M.E.; LeDouarin, N.M. Development and evolution of the neural crest: An overview. Dev. Biol. 2012, 366, 2–9. [Google Scholar] [CrossRef]
- Meulemans, D.; Bronner-Fraser, M. Gene-regulatory interactions in neural crest evolution and development. Dev. Cell. 2004, 7, 291–299. [Google Scholar] [CrossRef]
- Wei, S.; Xu, G.; Bridges, L.C.; Williams, P.; White, J.M.; DeSimone, D.W. ADAM13 induces cranial neural crest by cleaving class B Ephrins and regulating Wnt signaling. Dev. Cell. 2010, 19, 345–352. [Google Scholar] [CrossRef]
- Li, J.; Perfetto, M.; Neuner, R.; Bahudhanapati, H.; Christian, L.; Mathavan, K.; Bridges, L.C.; Alfandari, D.; Wei, S. Xenopus ADAM19 regulates Wnt signaling and neural crest specification by stabilizing ADAM13. Development 2018, 145, dev158154. [Google Scholar] [CrossRef]
- Ribes, V.; Le Roux, I.; Rhinn, M.; Schuhbaur, B.; Dolle, P. Early mouse caudal development relies on crosstalk between retinoic acid, Shh and Fgf signalling pathways. Development 2009, 136, 665–676. [Google Scholar] [CrossRef]
- Yokota, C.; Astrand, C.; Takahashi, S.; Hagey, D.W.; Stenman, J.M. Apolipoprotein C-I mediates Wnt/Ctnnb1 signaling during neural border formation and is required for neural crest development. Int. J. Dev. Biol. 2017, 61, 415–425. [Google Scholar] [CrossRef]
- Seo, J.H.; Park, D.S.; Hong, M.; Chang, E.J.; Choi, S.C. Essential role of AWP1 in neural crest specification in Xenopus. Int. J. Dev. Biol. 2013, 57, 829–836. [Google Scholar] [CrossRef]
- Simoes-Costa, M.; Stone, M.; Bronner, M.E. Axud1 Integrates Wnt Signaling and Transcriptional Inputs to Drive Neural Crest Formation. Dev. Cell. 2015, 34, 544–554. [Google Scholar] [CrossRef]
- Koehler, A.; Schlupf, J.; Schneider, M.; Kraft, B.; Winter, C.; Kashef, J. Loss of Xenopus cadherin-11 leads to increased Wnt/beta-catenin signaling and up-regulation of target genes c-myc and cyclin D1 in neural crest. Dev. Biol. 2013, 383, 132–145. [Google Scholar] [CrossRef]
- Wu, J.; Yang, J.; Klein, P.S. Neural crest induction by the canonical Wnt pathway can be dissociated from anterior-posterior neural patterning in Xenopus. Dev. Biol. 2005, 279, 220–232. [Google Scholar] [CrossRef]
- Hong, C.S.; Park, B.Y.; Saint-Jeannet, J.P. Fgf8a induces neural crest indirectly through the activation of Wnt8 in the paraxial mesoderm. Development 2008, 135, 3903–3910. [Google Scholar] [CrossRef] [PubMed]
- Ossipova, O.; Sokol, S.Y. Neural crest specification by noncanonical Wnt signaling and PAR-1. Development 2011, 138, 5441–5450. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Deardorff, M.A.; Tan, C.; Saint-Jeannet, J.P.; Klein, P.S. A role for frizzled 3 in neural crest development. Development 2001, 128, 3655–3663. [Google Scholar] [PubMed]
- Abu-Elmagd, M.; Garcia-Morales, C.; Wheeler, G.N. Frizzled7 mediates canonical Wnt signaling in neural crest induction. Dev. Biol. 2006, 298, 285–298. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Kuriyama, S.; Moreno, M.; Mayor, R. The posteriorizing gene Gbx2 is a direct target of Wnt signalling and the earliest factor in neural crest induction. Development 2009, 136, 3267–3278. [Google Scholar] [CrossRef] [PubMed]
- Saint-Jeannet, J.P.; He, X.; Varmus, H.E.; Dawid, I.B. Regulation of dorsal fate in the neuraxis by Wnt-1 and Wnt-3a. Proc. Natl. Acad. Sci. 1997, 94, 13713–13718. [Google Scholar] [CrossRef] [PubMed]
- Dutta, S.; Dawid, I.B. Kctd15 inhibits neural crest formation by attenuating Wnt/beta-catenin signaling output. Development 2010, 137, 3013–3018. [Google Scholar] [CrossRef] [PubMed]
- Hassler, C.; Cruciat, C.M.; Huang, Y.L.; Kuriyama, S.; Mayor, R.; Niehrs, C. Kremen is required for neural crest induction in Xenopus and promotes LRP6-mediated Wnt signaling. Development 2007, 134, 4255–4263. [Google Scholar] [CrossRef]
- Park, D.S.; Seo, J.H.; Hong, M.; Choi, S.C. Role of the Rap2/TNIK kinase pathway in regulation of LRP6 stability for Wnt signaling. Biochem. Biophys. Res. Commun. 2013, 436, 338–343. [Google Scholar] [CrossRef]
- Martinez-Morales, P.L.; Diez del Corral, R.; Olivera-Martinez, I.; Quiroga, A.C.; Das, R.M.; Barbas, J.A.; Storey, K.G.; Morales, A.V. FGF and retinoic acid activity gradients control the timing of neural crest cell emigration in the trunk. J. Cell Biol. 2011, 194, 489–503. [Google Scholar] [CrossRef]
- Lin, S.J.; Chiang, M.C.; Shih, H.Y.; Hsu, L.S.; Yeh, T.H.; Huang, Y.C.; Lin, C.Y.; Cheng, Y.C. Regulator of G protein signaling 2 (Rgs2) regulates neural crest development through Ppardelta-Sox10 cascade. Biochim. Biophys. Acta Mol. Cell. Res. 2017, 1864, 463–474. [Google Scholar] [CrossRef]
- Guemar, L.; de Santa Barbara, P.; Vignal, E.; Maurel, B.; Fort, P.; Faure, S. The small GTPase RhoV is an essential regulator of neural crest induction in Xenopus. Dev. Biol. 2007, 310, 113–128. [Google Scholar] [CrossRef][Green Version]
- Schille, C.; Bayerlova, M.; Bleckmann, A.; Schambony, A. Ror2 signaling is required for local upregulation of GDF6 and activation of BMP signaling at the neural plate border. Development 2016, 143, 3182–3194. [Google Scholar] [CrossRef]
- Wang, Y.; Fu, Y.; Gao, L.; Zhu, G.; Liang, J.; Gao, C.; Huang, B.; Fenger, U.; Niehrs, C.; Chen, Y.G.; et al. Xenopus skip modulates Wnt/beta-catenin signaling and functions in neural crest induction. J. Biol. Chem. 2010, 285, 10890–10901. [Google Scholar] [CrossRef]
- Park, D.S.; Seo, J.H.; Hong, M.; Bang, W.; Han, J.K.; Choi, S.C. Role of Sp5 as an essential early regulator of neural crest specification in xenopus. Dev. Dyn. 2013, 242, 1382–1394. [Google Scholar] [CrossRef]
- Heeg-Truesdell, E.; LaBonne, C. Neural induction in Xenopus requires inhibition of Wnt-beta-catenin signaling. Dev. Biol. 2006, 298, 71–86. [Google Scholar] [CrossRef]
- Garcia-Castro, M.I.; Marcelle, C.; Bronner-Fraser, M. Ectodermal Wnt function as a neural crest inducer. Science 2002, 297, 848–851. [Google Scholar]
- Schmidt, C.; McGonnell, I.M.; Allen, S.; Otto, A.; Patel, K. Wnt6 controls amniote neural crest induction through the non-canonical signaling pathway. Dev. Dyn. 2007, 236, 2502–2511. [Google Scholar] [CrossRef]
- Ikeya, M.; Lee, S.M.; Johnson, J.E.; McMahon, A.P.; Takada, S. Wnt signalling required for expansion of neural crest and CNS progenitors. Nature 1997, 389, 966–970. [Google Scholar] [CrossRef]
- Chang, C.; Hemmati-Brivanlou, A. Neural crest induction by Xwnt7B in Xenopus. Dev. Biol. 1998, 194, 129–134. [Google Scholar] [CrossRef]
- LaBonne, C.; Bronner-Fraser, M. Neural crest induction in Xenopus: Evidence for a two-signal model. Development 1998, 125, 2403–2414. [Google Scholar] [PubMed]
- Summerhurst, K.; Stark, M.; Sharpe, J.; Davidson, D.; Murphy, P. 3D representation of Wnt and Frizzled gene expression patterns in the mouse embryo at embryonic day 11.5 (Ts19). Gene Expr. Patterns 2008, 8, 331–348. [Google Scholar] [CrossRef] [PubMed]
- Duester, G. Retinoic acid synthesis and signaling during early organogenesis. Cell 2008, 134, 921–931. [Google Scholar] [CrossRef] [PubMed]
- Bolanos-Garcia, V.M.; Miguel, R.N. On the structure and function of apolipoproteins: More than a family of lipid-binding proteins. Prog. Biophys. Mol. Biol. 2003, 83, 47–68. [Google Scholar] [CrossRef]
- Fujimura, N.; Vacik, T.; Machon, O.; Vlcek, C.; Scalabrin, S.; Speth, M.; Diep, D.; Krauss, S.; Kozmik, Z. Wnt-mediated down-regulation of Sp1 target genes by a transcriptional repressor Sp5. J. Biol. Chem. 2007, 282, 1225–1237. [Google Scholar] [CrossRef] [PubMed]
- Duan, W.; Sun, B.; Li, T.W.; Tan, B.J.; Lee, M.K.; Teo, T.S. Cloning and characterization of AWP1, a novel protein that associates with serine/threonine kinase PRK1 in vivo. Gene 2000, 256, 113–121. [Google Scholar] [CrossRef]
- Oishi, I.; Suzuki, H.; Onishi, N.; Takada, R.; Kani, S.; Ohkawara, B.; Koshida, I.; Suzuki, K.; Yamada, G.; Schwabe, G.C.; et al. The receptor tyrosine kinase Ror2 is involved in non-canonical Wnt5a/JNK signalling pathway. Genes Cells 2003, 8, 645. [Google Scholar] [CrossRef]
- Borday, C.; Parain, K.; Thi Tran, H.; Vleminckx, K.; Perron, M.; Monsoro-Burq, A.H. An atlas of Wnt activity during embryogenesis in Xenopus tropicalis. PLoS ONE 2018, 13, e0193606. [Google Scholar] [CrossRef]
- Weisz Hubsman, M.; Volinsky, N.; Manser, E.; Yablonski, D.; Aronheim, A. Autophosphorylation-dependent degradation of Pak1, triggered by the Rho-family GTPase, Chp. Biochem. J. 2007, 404, 487–497. [Google Scholar] [CrossRef]
- Yang, Z.; Rayala, S.; Nguyen, D.; Vadlamudi, R.K.; Chen, S.; Kumar, R. Pak1 phosphorylation of snail, a master regulator of epithelial-to-mesenchyme transition, modulates snail’s subcellular localization and functions. Cancer Res. 2005, 65, 3179–3184. [Google Scholar] [CrossRef]
- Faure, S.; Fort, P. Atypical RhoV and RhoU GTPases control development of the neural crest. Small GTPases 2011, 2, 310–313. [Google Scholar] [CrossRef] [PubMed]
- Quinlan, R.; Graf, M.; Mason, I.; Lumsden, A.; Kiecker, C. Complex and dynamic patterns of Wnt pathway gene expression in the developing chick forebrain. Neural Dev. 2009, 4, 35. [Google Scholar] [CrossRef] [PubMed]
- Brault, V.; Moore, R.; Kutsch, S.; Ishibashi, M.; Rowitch, D.H.; McMahon, A.P.; Sommer, L.; Boussadia, O.; Kemler, R. Inactivation of the beta-catenin gene by Wnt1-Cre-mediated deletion results in dramatic brain malformation and failure of craniofacial development. Development 2001, 128, 1253–1264. [Google Scholar] [PubMed]
- Stuhlmiller, T.J.; Garcia-Castro, M.I. Current perspectives of the signaling pathways directing neural crest induction. Cell. Mol. Life Sci. 2012, 69, 3715–3737. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Glinka, A.; Delius, H.; Niehrs, C. Mutual antagonism between dickkopf1 and dickkopf2 regulates Wnt/beta-catenin signalling. Curr. Biol. 2000, 10, 1611–1614. [Google Scholar] [CrossRef]
- Li, L.; Mao, J.; Sun, L.; Liu, W.; Wu, D. Second cysteine-rich domain of Dickkopf-2 activates canonical Wnt signaling pathway via LRP-6 independently of dishevelled. J. Biol. Chem. 2002, 277, 5977–5981. [Google Scholar] [CrossRef] [PubMed]
- Brott, B.K.; Sokol, S.Y. Regulation of Wnt/LRP signaling by distinct domains of Dickkopf proteins. Mol. Cell. Biol. 2002, 22, 6100–6110. [Google Scholar] [CrossRef]
- Aybar, M.J.; Mayor, R. Early induction of neural crest cells: Lessons learned from frog, fish and chick. Curr. Opin. Genet. Dev. 2002, 12, 452–458. [Google Scholar] [CrossRef]
- Villanueva, S.; Glavic, A.; Ruiz, P.; Mayor, R. Posteriorization by FGF, Wnt, and retinoic acid is required for neural crest induction. Dev. Biol. 2002, 241, 289–301. [Google Scholar] [CrossRef]
- Domingos, P.M.; Itasaki, N.; Jones, C.M.; Mercurio, S.; Sargent, M.G.; Smith, J.C.; Krumlauf, R. The Wnt/beta-catenin pathway posteriorizes neural tissue in Xenopus by an indirect mechanism requiring FGF signalling. Dev. Biol. 2001, 239, 148–160. [Google Scholar] [CrossRef]
- Poliakov, A.; Cotrina, M.; Wilkinson, D.G. Diverse roles of eph receptors and ephrins in the regulation of cell migration and tissue assembly. Dev. Cell 2004, 7, 465–480. [Google Scholar] [CrossRef] [PubMed]
- Borchers, A.; David, R.; Wedlich, D. Xenopus cadherin-11 restrains cranial neural crest migration and influences neural crest specification. Development 2001, 128, 3049–3060. [Google Scholar] [PubMed]
- Mao, B.; Wu, W.; Davidson, G.; Marhold, J.; Li, M.; Mechler, B.M.; Delius, H.; Hoppe, D.; Stannek, P.; Walter, C.; et al. Kremen proteins are Dickkopf receptors that regulate Wnt/beta-catenin signalling. Nature 2002, 417, 664–667. [Google Scholar] [CrossRef] [PubMed]
- Leong, G.M.; Subramaniam, N.; Figueroa, J.; Flanagan, J.L.; Hayman, M.J.; Eisman, J.A.; Kouzmenko, A.P. Ski-interacting protein interacts with Smad proteins to augment transforming growth factor-beta-dependent transcription. J. Biol. Chem. 2001, 276, 18243–18248. [Google Scholar] [CrossRef]
- Heffer, A.; Marquart, G.D.; Aquilina-Beck, A.; Saleem, N.; Burgess, H.A.; Dawid, I.B. Generation and characterization of Kctd15 mutations in zebrafish. PLoS ONE 2017, 12. [Google Scholar] [CrossRef]
- Zarelli, V.E.; Dawid, I.B. Inhibition of neural crest formation by Kctd15 involves regulation of transcription factor AP-2. Proc. Natl. Acad. Sci. 2013, 110, 2870–2875. [Google Scholar] [CrossRef]
- Wong, T.C.; Rebbert, M.; Wang, C.; Chen, X.; Heffer, A.; Zarelli, V.E.; Dawid, I.B.; Zhao, H. Genes regulated by potassium channel tetramerization domain containing 15 (Kctd15) in the developing neural crest. Int. J. Dev. Biol. 2016, 60, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Shy, B.R.; Wu, C.I.; Khramtsova, G.F.; Zhang, J.Y.; Olopade, O.I.; Goss, K.H.; Merrill, B.J. Regulation of Tcf7l1 DNA binding and protein stability as principal mechanisms of Wnt/beta-catenin signaling. Cell Rep. 2013, 4, 1–9. [Google Scholar] [CrossRef]
- Theveneau, E.; Mayor, R. Neural crest delamination and migration: From epithelium-to-mesenchyme transition to collective cell migration. Dev. Biol. 2012, 366, 34–54. [Google Scholar] [CrossRef]
- Nichols, D.H. Neural crest formation in the head of the mouse embryo as observed using a new histological technique. J. Embryol. Exp. Morphol. 1981, 64, 105–120. [Google Scholar]
- Nichols, D.H. Ultrastructure of neural crest formation in the midbrain/rostral hindbrain and preotic hindbrain regions of the mouse embryo. Am. J. Anat. 1987, 179, 143–154. [Google Scholar] [CrossRef]
- Sadaghiani, B.; Thiebaud, C.H. Neural crest development in the Xenopus laevis embryo, studied by interspecific transplantation and scanning electron microscopy. Dev. Biol. 1987, 124, 91–110. [Google Scholar] [CrossRef]
- Burstyn-Cohen, T.; Kalcheim, C. Association between the cell cycle and neural crest delamination through specific regulation of G1/S transition. Dev. Cell 2002, 3, 383–395. [Google Scholar] [CrossRef]
- Hutchins, E.J.; Bronner, M.E. Draxin acts as a molecular rheostat of canonical Wnt signaling to control cranial neural crest EMT. J. Cell Biol. 2018, 217, 3683–3697. [Google Scholar] [CrossRef] [PubMed]
- Burstyn-Cohen, T.; Stanleigh, J.; Sela-Donenfeld, D.; Kalcheim, C. Canonical Wnt activity regulates trunk neural crest delamination linking BMP/noggin signaling with G1/S transition. Development 2004, 131, 5327–5339. [Google Scholar] [CrossRef] [PubMed]
- Padmanabhan, L.A.T.a.R. The Cell Biology of Neural Crest Cell Delamination and EMT. In Neural Crest Cells: Evolution, Development and Disease; Trainor, P.A., Ed.; Academic Press: London, UK, 2014; pp. 51–72. [Google Scholar]
- Kumar, D.; Nitzan, E.; Kalcheim, C. YAP promotes neural crest emigration through interactions with BMP and Wnt activities. Cell Commun. Signal. 2019, 17, 69. [Google Scholar] [CrossRef] [PubMed]
- Clay, M.R.; Halloran, M.C. Regulation of cell adhesions and motility during initiation of neural crest migration. Curr. Opin. Neurobiol. 2011, 21, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Chalpe, A.J.; Prasad, M.; Henke, A.J.; Paulson, A.F. Regulation of cadherin expression in the chicken neural crest by the Wnt/beta-catenin signaling pathway. Cell Adh. Migr. 2010, 4, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Abbruzzese, G.; Cousin, H.; Salicioni, A.M.; Alfandari, D. GSK3 and Polo-like kinase regulate ADAM13 function during cranial neural crest cell migration. Mol. Biol. Cell 2014, 25, 4072–4082. [Google Scholar] [CrossRef]
- Shoval, I.; Ludwig, A.; Kalcheim, C. Antagonistic roles of full-length N-cadherin and its soluble BMP cleavage product in neural crest delamination. Development 2007, 134, 491–501. [Google Scholar] [CrossRef]
- Xu, X.; Li, W.E.; Huang, G.Y.; Meyer, R.; Chen, T.; Luo, Y.; Thomas, M.P.; Radice, G.L.; Lo, C.W. Modulation of mouse neural crest cell motility by N-cadherin and connexin 43 gap junctions. J. Cell Biol. 2001, 154, 217–230. [Google Scholar] [CrossRef]
- Ulmer, B.; Hagenlocher, C.; Schmalholz, S.; Kurz, S.; Schweickert, A.; Kohl, A.; Roth, L.; Sela-Donenfeld, D.; Blum, M. Calponin 2 acts as an effector of noncanonical Wnt-mediated cell polarization during neural crest cell migration. Cell Rep. 2013, 3, 615–621. [Google Scholar] [CrossRef][Green Version]
- Rabadan, M.A.; Herrera, A.; Fanlo, L.; Usieto, S.; Carmona-Fontaine, C.; Barriga, E.H.; Mayor, R.; Pons, S.; Marti, E. Delamination of neural crest cells requires transient and reversible Wnt inhibition mediated by Dact1/2. Development 2016, 143, 2194–2205. [Google Scholar] [CrossRef]
- Tuttle, A.M.; Hoffman, T.L.; Schilling, T.F. Rabconnectin-3a regulates vesicle endocytosis and canonical Wnt signaling in zebrafish neural crest migration. PLoS Biol. 2014, 12, e1001852. [Google Scholar] [CrossRef]
- Hutchins, E.J.; Bronner, M.E. Draxin alters laminin organization during basement membrane remodeling to control cranial neural crest EMT. Dev. Biol. 2019, 446, 151–158. [Google Scholar] [CrossRef]
- Zhao, Y.; Shi, J.; Winey, M.; Klymkowsky, M.W. Identifying domains of EFHC1 involved in ciliary localization, ciliogenesis, and the regulation of Wnt signaling. Dev. Biol. 2016, 411, 257–265. [Google Scholar] [CrossRef]
- de Melker, A.A.; Desban, N.; Duband, J.L. Cellular localization and signaling activity of beta-catenin in migrating neural crest cells. Dev. Dyn. 2004, 230, 708–726. [Google Scholar] [CrossRef]
- Maj, E.; Kunneke, L.; Loresch, E.; Grund, A.; Melchert, J.; Pieler, T.; Aspelmeier, T.; Borchers, A. Controlled levels of canonical Wnt signaling are required for neural crest migration. Dev. Biol. 2016, 417, 77–90. [Google Scholar] [CrossRef]
- Willems, B.; Tao, S.; Yu, T.; Huysseune, A.; Witten, P.E.; Winkler, C. The Wnt Co-Receptor Lrp5 Is Required for Cranial Neural Crest Cell Migration in Zebrafish. PLoS ONE 2015, 10, e0131768. [Google Scholar] [CrossRef][Green Version]
- Banerjee, S.; Gordon, L.; Donn, T.M.; Berti, C.; Moens, C.B.; Burden, S.J.; Granato, M. A novel role for MuSK and non-canonical Wnt signaling during segmental neural crest cell migration. Development 2011, 138, 3287–3296. [Google Scholar] [CrossRef]
- Piloto, S.; Schilling, T.F. Ovo1 links Wnt signaling with N-cadherin localization during neural crest migration. Development 2010, 137, 1981–1990. [Google Scholar] [CrossRef]
- Gessert, S.; Maurus, D.; Rossner, A.; Kuhl, M. Pescadillo is required for Xenopus laevis eye development and neural crest migration. Dev. Biol. 2007, 310, 99–112. [Google Scholar] [CrossRef]
- Podleschny, M.; Grund, A.; Berger, H.; Rollwitz, E.; Borchers, A. A PTK7/Ror2 Co-Receptor Complex Affects Xenopus Neural Crest Migration. PLoS ONE 2015, 10, e0145169. [Google Scholar] [CrossRef]
- Shnitsar, I.; Borchers, A. PTK7 recruits dsh to regulate neural crest migration. Development 2008, 135, 4015–4024. [Google Scholar] [CrossRef]
- Groysman, M.; Shoval, I.; Kalcheim, C. A negative modulatory role for rho and rho-associated kinase signaling in delamination of neural crest cells. Neural Dev. 2008, 3, 27. [Google Scholar] [CrossRef]
- Tao, W.; Pennica, D.; Xu, L.; Kalejta, R.F.; Levine, A.J. Wrch-1, a novel member of the Rho gene family that is regulated by Wnt-1. Genes Dev. 2001, 15, 1796–1807. [Google Scholar] [CrossRef]
- Fort, P.; Guemar, L.; Vignal, E.; Morin, N.; Notarnicola, C.; de Santa Barbara, P.; Faure, S. Activity of the RhoU/Wrch1 GTPase is critical for cranial neural crest cell migration. Dev. Biol. 2011, 350, 451–463. [Google Scholar] [CrossRef]
- Gorny, A.K.; Kaufmann, L.T.; Swain, R.K.; Steinbeisser, H. A secreted splice variant of the Xenopus frizzled-4 receptor is a biphasic modulator of Wnt signalling. Cell Commun. Signal. 2013, 11, 82. [Google Scholar] [CrossRef]
- Liu, J.A.; Wu, M.H.; Yan, C.H.; Chau, B.K.; So, H.; Ng, A.; Chan, A.; Cheah, K.S.; Briscoe, J.; Cheung, M. Phosphorylation of Sox9 is required for neural crest delamination and is regulated downstream of BMP and canonical Wnt signaling. Proc. Natl. Acad. Sci. 2013, 110, 2882–2887. [Google Scholar] [CrossRef]
- Matthews, H.K.; Marchant, L.; Carmona-Fontaine, C.; Kuriyama, S.; Larrain, J.; Holt, M.R.; Parsons, M.; Mayor, R. Directional migration of neural crest cells in vivo is regulated by Syndecan-4/Rac1 and non-canonical Wnt signaling/RhoA. Development 2008, 135, 1771–1780. [Google Scholar] [CrossRef]
- Simon, E.; Theze, N.; Fedou, S.; Thiebaud, P.; Faucheux, C. Vestigial-like 3 is a novel Ets1 interacting partner and regulates trigeminal nerve formation and cranial neural crest migration. Biol. Open 2017, 6, 1528–1540. [Google Scholar] [CrossRef]
- Sinnberg, T.; Levesque, M.P.; Krochmann, J.; Cheng, P.F.; Ikenberg, K.; Meraz-Torres, F.; Niessner, H.; Garbe, C.; Busch, C. Wnt-signaling enhances neural crest migration of melanoma cells and induces an invasive phenotype. Mol. Cancer 2018, 17, 59. [Google Scholar] [CrossRef]
- Matthews, H.K.; Broders-Bondon, F.; Thiery, J.P.; Mayor, R. Wnt11r is required for cranial neural crest migration. Dev. Dyn. 2008, 237, 3404–3409. [Google Scholar] [CrossRef]
- Hatta, K.; Nose, A.; Nagafuchi, A.; Takeichi, M. Cloning and expression of cDNA encoding a neural calcium-dependent cell adhesion molecule: Its identity in the cadherin gene family. J. Cell Biol. 1988, 106, 873–881. [Google Scholar] [CrossRef]
- Tepass, U.; Truong, K.; Godt, D.; Ikura, M.; Peifer, M. Cadherins in embryonic and neural morphogenesis. Nat. Rev. Mol. Cell Biol. 2000, 1, 91–100. [Google Scholar] [CrossRef]
- Varelas, X. The Hippo pathway effectors TAZ and YAP in development, homeostasis and disease. Development 2014, 141, 1614–1626. [Google Scholar] [CrossRef]
- Zhang, L.; Gao, X.; Wen, J.; Ning, Y.; Chen, Y.G. Dapper 1 antagonizes Wnt signaling by promoting dishevelled degradation. J. Biol. Chem. 2006, 281, 8607–8612. [Google Scholar] [CrossRef]
- Habas, R.; Kato, Y.; He, X. Wnt/Frizzled activation of Rho regulates vertebrate gastrulation and requires a novel Formin homology protein Daam1. Cell 2001, 107, 843–854. [Google Scholar] [CrossRef]
- Mayor, E.T.a.R. Neural Crest Cell Migration: Guidance, Pathways, and Cell–Cell Interactions. In Neural Crest Cells: Evolution, Development and Disease; Trainor, P.A., Ed.; Academic Press: London, UK, 2014; pp. 73–88. [Google Scholar]
- Clay, M.R.; Halloran, M.C. Control of neural crest cell behavior and migration: Insights from live imaging. Cell Adh. Migr. 2010, 4, 586–594. [Google Scholar] [CrossRef]
- Shellard, A.; Mayor, R. Integrating chemical and mechanical signals in neural crest cell migration. Curr. Opin. Genet. Dev. 2019, 57, 16–24. [Google Scholar] [CrossRef]
- Jaffe, A.B.; Hall, A. Rho GTPases: Biochemistry and biology. Annu. Rev. Cell Dev. Biol. 2005, 21, 247–269. [Google Scholar] [CrossRef] [PubMed]
- Kulesa, P.M.; McLennan, R. Neural crest migration: Trailblazing ahead. F1000Prime Rep. 2015, 7, 2. [Google Scholar] [CrossRef] [PubMed]
- Abercrombie, M.; Heaysman, J.E. Observations on the social behaviour of cells in tissue culture. I. Speed of movement of chick heart fibroblasts in relation to their mutual contacts. Exp. Cell Res. 1953, 5, 111–131. [Google Scholar] [CrossRef]
- Theveneau, E.; Steventon, B.; Scarpa, E.; Garcia, S.; Trepat, X.; Streit, A.; Mayor, R. Chase-and-run between adjacent cell populations promotes directional collective migration. Nat. Cell Biol. 2013, 15, 763–772. [Google Scholar] [CrossRef]
- Carmona-Fontaine, C.; Matthews, H.K.; Kuriyama, S.; Moreno, M.; Dunn, G.A.; Parsons, M.; Stern, C.D.; Mayor, R. Contact inhibition of locomotion in vivo controls neural crest directional migration. Nature 2008, 456, 957–961. [Google Scholar] [CrossRef]
- Hayes, M.; Naito, M.; Daulat, A.; Angers, S.; Ciruna, B. Ptk7 promotes non-canonical Wnt/PCP-mediated morphogenesis and inhibits Wnt/beta-catenin-dependent cell fate decisions during vertebrate development. Development 2013, 140, 1807–1818. [Google Scholar] [CrossRef] [PubMed]
- Aspenstrom, P.; Fransson, A.; Saras, J. Rho GTPases have diverse effects on the organization of the actin filament system. Biochem. J. 2004, 377, 327–337. [Google Scholar] [CrossRef]
- Vidal, C.; Geny, B.; Melle, J.; Jandrot-Perrus, M.; Fontenay-Roupie, M. Cdc42/Rac1-dependent activation of the p21-activated kinase (PAK) regulates human platelet lamellipodia spreading: Implication of the cortical-actin binding protein cortactin. Blood 2002, 100, 4462–4469. [Google Scholar] [CrossRef]
- Adams, C.C.; Jakovljevic, J.; Roman, J.; Harnpicharnchai, P.; Woolford, J.L., Jr. Saccharomyces cerevisiae nucleolar protein Nop7p is necessary for biogenesis of 60S ribosomal subunits. RNA 2002, 8, 150–165. [Google Scholar] [CrossRef][Green Version]
- Sikorski, E.M.; Uo, T.; Morrison, R.S.; Agarwal, A. Pescadillo interacts with the cadmium response element of the human heme oxygenase-1 promoter in renal epithelial cells. J. Biol. Chem. 2006, 281, 24423–24430. [Google Scholar] [CrossRef]
- Rickmann, M.; Fawcett, J.W.; Keynes, R.J. The migration of neural crest cells and the growth of motor axons through the rostral half of the chick somite. J. Embryol. Exp. Morphol. 1985, 90, 437–455. [Google Scholar] [PubMed]
- Serbedzija, G.N.; Fraser, S.E.; Bronner-Fraser, M. Pathways of trunk neural crest cell migration in the mouse embryo as revealed by vital dye labelling. Development 1990, 108, 605–612. [Google Scholar] [PubMed]
- Raible, D.W.; Wood, A.; Hodsdon, W.; Henion, P.D.; Weston, J.A.; Eisen, J.S. Segregation and early dispersal of neural crest cells in the embryonic zebrafish. Dev. Dyn. 1992, 195, 29–42. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Perfetto, M.; Materna, C.; Li, R.; Thi Tran, H.; Vleminckx, K.; Duncan, M.K.; Wei, S. A new transgenic reporter line reveals Wnt-dependent Snai2 re-expression and cranial neural crest differentiation in Xenopus. Sci. Rep. 2019, 9, 11191. [Google Scholar] [CrossRef]
- Abbruzzese, G.; Becker, S.F.; Kashef, J.; Alfandari, D. ADAM13 cleavage of cadherin-11 promotes CNC migration independently of the homophilic binding site. Dev. Biol. 2016, 415, 383–390. [Google Scholar] [CrossRef] [PubMed]
- McCusker, C.; Cousin, H.; Neuner, R.; Alfandari, D. Extracellular cleavage of cadherin-11 by ADAM metalloproteases is essential for Xenopus cranial neural crest cell migration. Mol. Biol. Cell 2009, 20, 78–89. [Google Scholar] [CrossRef]
- Cousin, H.; Abbruzzese, G.; Kerdavid, E.; Gaultier, A.; Alfandari, D. Translocation of the cytoplasmic domain of ADAM13 to the nucleus is essential for Calpain8-a expression and cranial neural crest cell migration. Dev. Cell 2011, 20, 256–263. [Google Scholar] [CrossRef] [PubMed]
- Swain, R.K.; Katoh, M.; Medina, A.; Steinbeisser, H. Xenopus frizzled-4S, a splicing variant of Xfz4 is a context-dependent activator and inhibitor of Wnt/beta-catenin signaling. Cell Commun. Signal. 2005, 3, 12. [Google Scholar] [CrossRef]
- Nie, S.; Kee, Y.; Bronner-Fraser, M. Myosin-X is critical for migratory ability of Xenopus cranial neural crest cells. Dev. Biol. 2009, 335, 132–142. [Google Scholar] [CrossRef][Green Version]
- Li, B.; Mackay, D.R.; Dai, Q.; Li, T.W.; Nair, M.; Fallahi, M.; Schonbaum, C.P.; Fantes, J.; Mahowald, A.P.; Waterman, M.L.; et al. The LEF1/beta -catenin complex activates movo1, a mouse homolog of Drosophila ovo required for epidermal appendage differentiation. Proc. Natl. Acad. Sci. 2002, 99, 6064–6069. [Google Scholar] [CrossRef]
- Noren, N.K.; Liu, B.P.; Burridge, K.; Kreft, B. p120 catenin regulates the actin cytoskeleton via Rho family GTPases. J. Cell Biol. 2000, 150, 567–580. [Google Scholar] [CrossRef] [PubMed]
- Dupin, E.; Sommer, L. Neural crest progenitors and stem cells: From early development to adulthood. Dev. Biol. 2012, 366, 83–95. [Google Scholar] [CrossRef] [PubMed]
- Zurkirchen, L.; Sommer, L. Quo vadis: Tracing the fate of neural crest cells. Curr. Opin. Neurobiol. 2017, 47, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Bringas, P., Jr.; Chai, Y. An in vitro model for characterizing the post-migratory cranial neural crest cells of the first branchial arch. Dev. Dyn. 2006, 235, 1433–1440. [Google Scholar] [CrossRef] [PubMed]
- Shakhova, O.; Sommer, L. In Vitro Derivation of Melanocytes from Embryonic Neural Crest Stem Cells. Methods Mol. Biol. 2015, 1–8. [Google Scholar] [CrossRef]
- Replogle, M.R.; Sreevidya, V.S.; Lee, V.M.; Laiosa, M.D.; Svoboda, K.R.; Udvadia, A.J. Establishment of a murine culture system for modeling the temporal progression of cranial and trunk neural crest cell differentiation. Dis. Model. Mech. 2018, 11, dmm035097. [Google Scholar] [CrossRef] [PubMed]
- Bronner-Fraser, M.; Fraser, S.E. Cell lineage analysis reveals multipotency of some avian neural crest cells. Nature 1988, 335, 161–164. [Google Scholar] [CrossRef] [PubMed]
- Krispin, S.; Nitzan, E.; Kassem, Y.; Kalcheim, C. Evidence for a dynamic spatiotemporal fate map and early fate restrictions of premigratory avian neural crest. Development 2010, 137, 585–595. [Google Scholar] [CrossRef] [PubMed]
- Nitzan, E.; Krispin, S.; Pfaltzgraff, E.R.; Klar, A.; Labosky, P.A.; Kalcheim, C. A dynamic code of dorsal neural tube genes regulates the segregation between neurogenic and melanogenic neural crest cells. Development 2013, 140, 2269–2279. [Google Scholar] [CrossRef] [PubMed]
- Baggiolini, A.; Varum, S.; Mateos, J.M.; Bettosini, D.; John, N.; Bonalli, M.; Ziegler, U.; Dimou, L.; Clevers, H.; Furrer, R.; et al. Premigratory and migratory neural crest cells are multipotent in vivo. Cell Stem Cell 2015, 16, 314–322. [Google Scholar] [CrossRef] [PubMed]
- Kleber, M.; Lee, H.Y.; Wurdak, H.; Buchstaller, J.; Riccomagno, M.M.; Ittner, L.M.; Suter, U.; Epstein, D.J.; Sommer, L. Neural crest stem cell maintenance by combinatorial Wnt and BMP signaling. J. Cell Biol. 2005, 169, 309–320. [Google Scholar] [CrossRef] [PubMed]
- Nagoshi, N.; Shibata, S.; Kubota, Y.; Nakamura, M.; Nagai, Y.; Satoh, E.; Morikawa, S.; Okada, Y.; Mabuchi, Y.; Katoh, H.; et al. Ontogeny and multipotency of neural crest-derived stem cells in mouse bone marrow, dorsal root ganglia, and whisker pad. Cell Stem Cell 2008, 2, 392–403. [Google Scholar] [CrossRef] [PubMed]
- Li, H.Y.; Say, E.H.; Zhou, X.F. Isolation and characterization of neural crest progenitors from adult dorsal root ganglia. Stem Cells 2007, 25, 2053–2065. [Google Scholar] [CrossRef] [PubMed]
- Joseph, N.M.; Mukouyama, Y.S.; Mosher, J.T.; Jaegle, M.; Crone, S.A.; Dormand, E.L.; Lee, K.F.; Meijer, D.; Anderson, D.J.; Morrison, S.J. Neural crest stem cells undergo multilineage differentiation in developing peripheral nerves to generate endoneurial fibroblasts in addition to Schwann cells. Development 2004, 131, 5599–5612. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, D.; Rothstein, M.; Azambuja, A.P.; Simoes-Costa, M. Control of neural crest multipotency by Wnt signaling and the Lin28/let-7 axis. Elife 2018, 7, e40556. [Google Scholar] [CrossRef]
- Newman, M.A.; Thomson, J.M.; Hammond, S.M. Lin-28 interaction with the Let-7 precursor loop mediates regulated microRNA processing. RNA 2008, 14, 1539–1549. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Quarto, N.; Senarath-Yapa, K.; Grey, N.; Bai, X.; Longaker, M.T. Enhanced Activation of Canonical Wnt Signaling Confers Mesoderm-Derived Parietal Bone with Similar Osteogenic and Skeletal Healing Capacity to Neural Crest-Derived Frontal Bone. PLoS ONE 2015, 10, e0138059. [Google Scholar] [CrossRef]
- Malik, Z.; Alexiou, M.; Hallgrimsson, B.; Economides, A.N.; Luder, H.U.; Graf, D. Bone Morphogenetic Protein 2 Coordinates Early Tooth Mineralization. J. Dent. Res. 2018, 97, 835–843. [Google Scholar] [CrossRef]
- Jia, S.; Kwon, H.E.; Lan, Y.; Zhou, J.; Liu, H.; Jiang, R. Bmp4-Msx1 signaling and Osr2 control tooth organogenesis through antagonistic regulation of secreted Wnt antagonists. Dev. Biol. 2016, 420, 110–119. [Google Scholar] [CrossRef]
- Dorsky, R.I.; Moon, R.T.; Raible, D.W. Control of neural crest cell fate by the Wnt signalling pathway. Nature 1998, 396, 370–373. [Google Scholar] [CrossRef]
- Lee, H.Y.; Kleber, M.; Hari, L.; Brault, V.; Suter, U.; Taketo, M.M.; Kemler, R.; Sommer, L. Instructive role of Wnt/beta-catenin in sensory fate specification in neural crest stem cells. Science 2004, 303, 1020–1023. [Google Scholar] [CrossRef] [PubMed]
- DiNuoscio, G.; Atit, R.P. Wnt/beta-catenin signaling in the mouse embryonic cranial mesenchyme is required to sustain the emerging differentiated meningeal layers. Genesis 2019, 57, e23279. [Google Scholar] [CrossRef] [PubMed]
- Zacharias, A.L.; Gage, P.J. Canonical Wnt/beta-catenin signaling is required for maintenance but not activation of Pitx2 expression in neural crest during eye development. Dev. Dyn. 2010, 239, 3215–3225. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Lan, Y.; Chai, Y.; Jiang, R. Antagonistic actions of Msx1 and Osr2 pattern mammalian teeth into a single row. Science 2009, 323, 1232–1234. [Google Scholar] [CrossRef]
- Gou, Y.; Li, J.; Jackson-Weaver, O.; Wu, J.; Zhang, T.; Gupta, R.; Cho, I.; Ho, T.V.; Chen, Y.; Li, M.; et al. Protein Arginine Methyltransferase PRMT1 Is Essential for Palatogenesis. J. Dent. Res. 2018, 97, 1510–1518. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Iseki, S.; Maxson, R.E.; Sucov, H.M.; Morriss-Kay, G.M. Tissue origins and interactions in the mammalian skull vault. Dev. Biol. 2002, 241, 106–116. [Google Scholar] [CrossRef]
- Bikkavilli, R.K.; Avasarala, S.; Vanscoyk, M.; Sechler, M.; Kelley, N.; Malbon, C.C.; Winn, R.A. Dishevelled3 is a novel arginine methyl transferase substrate. Sci. Rep. 2012, 2, 805. [Google Scholar] [CrossRef]
- Cha, B.; Kim, W.; Kim, Y.K.; Hwang, B.N.; Park, S.Y.; Yoon, J.W.; Park, W.S.; Cho, J.W.; Bedford, M.T.; Jho, E.H. Methylation by protein arginine methyltransferase 1 increases stability of Axin, a negative regulator of Wnt signaling. Oncogene 2011, 30, 2379–2389. [Google Scholar] [CrossRef]
- Bikkavilli, R.K.; Malbon, C.C. Arginine methylation of G3BP1 in response to Wnt3a regulates beta-catenin mRNA. J. Cell Sci. 2011, 124, 2310–2320. [Google Scholar] [CrossRef]
- Bikkavilli, R.K.; Malbon, C.C. Wnt3a-stimulated LRP6 phosphorylation is dependent upon arginine methylation of G3BP2. J. Cell Sci. 2012, 125, 2446–2456. [Google Scholar] [CrossRef]
- Maguire, L.H.; Thomas, A.R.; Goldstein, A.M. Tumors of the neural crest: Common themes in development and cancer. Dev. Dyn. 2015, 244, 311–322. [Google Scholar] [CrossRef] [PubMed]
- Miller, A.J.; Mihm, M.C., Jr. Melanoma. N. Engl. J. Med. 2006, 355, 51–65. [Google Scholar] [CrossRef] [PubMed]
- Powell, D.R.; O’Brien, J.H.; Ford, H.L.; Artinger, K.B. Neural Crest Cells and Cancer: Insights into Tumor Progression. In Neural Crest Cells: Evolution, Development and Disease; Trainor, P.A., Ed.; Academic Press: London, UK, 2014; pp. 335–357. [Google Scholar]
- Rubinfeld, B.; Robbins, P.; El-Gamil, M.; Albert, I.; Porfiri, E.; Polakis, P. Stabilization of beta-catenin by genetic defects in melanoma cell lines. Science 1997, 275, 1790–1792. [Google Scholar] [CrossRef] [PubMed]
- Rimm, D.L.; Caca, K.; Hu, G.; Harrison, F.B.; Fearon, E.R. Frequent nuclear/cytoplasmic localization of beta-catenin without exon 3 mutations in malignant melanoma. Am. J. Pathol. 1999, 154, 325–329. [Google Scholar] [CrossRef]
- Demunter, A.; Libbrecht, L.; Degreef, H.; De Wolf-Peeters, C.; van den Oord, J.J. Loss of membranous expression of beta-catenin is associated with tumor progression in cutaneous melanoma and rarely caused by exon 3 mutations. Mod. Pathol. 2002, 15, 454–461. [Google Scholar] [CrossRef] [PubMed]
- Xue, G.; Romano, E.; Massi, D.; Mandala, M. Wnt/beta-catenin signaling in melanoma: Preclinical rationale and novel therapeutic insights. Cancer Treat. Rev. 2016, 49, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Saitoh, A.; Hansen, L.A.; Vogel, J.C.; Udey, M.C. Characterization of Wnt gene expression in murine skin: Possible involvement of epidermis-derived Wnt-4 in cutaneous epithelial-mesenchymal interactions. Exp Cell Res. 1998, 243, 150–160. [Google Scholar] [CrossRef] [PubMed]
- Chien, A.J.; Moore, E.C.; Lonsdorf, A.S.; Kulikauskas, R.M.; Rothberg, B.G.; Berger, A.J.; Major, M.B.; Hwang, S.T.; Rimm, D.L.; Moon, R.T. Activated Wnt/beta-catenin signaling in melanoma is associated with decreased proliferation in patient tumors and a murine melanoma model. Proc. Natl. Acad. Sci. 2009, 106, 1193–1198. [Google Scholar] [CrossRef] [PubMed]
- Damsky, W.E.; Curley, D.P.; Santhanakrishnan, M.; Rosenbaum, L.E.; Platt, J.T.; Gould Rothberg, B.E.; Taketo, M.M.; Dankort, D.; Rimm, D.L.; McMahon, M.; et al. beta-catenin signaling controls metastasis in Braf-activated Pten-deficient melanomas. Cancer Cell 2011, 20, 741–754. [Google Scholar] [CrossRef]
- Gurney, J.G.; Ross, J.A.; Wall, D.A.; Bleyer, W.A.; Severson, R.K.; Robison, L.L. Infant cancer in the U.S.: Histology-specific incidence and trends, 1973 to 1992. J. Pediatr. Hematol. Oncol. 1997, 19, 428–432. [Google Scholar] [CrossRef]
- London, W.B.; Castleberry, R.P.; Matthay, K.K.; Look, A.T.; Seeger, R.C.; Shimada, H.; Thorner, P.; Brodeur, G.; Maris, J.M.; Reynolds, C.P.; et al. Evidence for an age cutoff greater than 365 days for neuroblastoma risk group stratification in the Children’s Oncology Group. J. Clin. Oncol. 2005, 23, 6459–6465. [Google Scholar] [CrossRef] [PubMed]
- Maris, J.M. Recent advances in neuroblastoma. N. Engl. J. Med. 2010, 362, 2202–2211. [Google Scholar] [CrossRef] [PubMed]
- Brodeur, G.M. Neuroblastoma: Biological insights into a clinical enigma. Nat. Rev. Cancer 2003, 3, 203–216. [Google Scholar] [CrossRef] [PubMed]
- Becker, J.; Wilting, J. WNT signaling, the development of the sympathoadrenal-paraganglionic system and neuroblastoma. Cell. Mol. Life. Sci. 2018, 75, 1057–1070. [Google Scholar] [CrossRef] [PubMed]
- Becker, J.; Wilting, J. WNT Signaling in Neuroblastoma. Cancers 2019, 11, 1013. [Google Scholar] [CrossRef]
- Brodeur, G.M.; Seeger, R.C.; Schwab, M.; Varmus, H.E.; Bishop, J.M. Amplification of N-myc in untreated human neuroblastomas correlates with advanced disease stage. Science 1984, 224, 1121–1124. [Google Scholar] [CrossRef]
- Schwab, M.; Alitalo, K.; Klempnauer, K.H.; Varmus, H.E.; Bishop, J.M.; Gilbert, F.; Brodeur, G.; Goldstein, M.; Trent, J. Amplified DNA with limited homology to myc cellular oncogene is shared by human neuroblastoma cell lines and a neuroblastoma tumour. Nature 1983, 305, 245–248. [Google Scholar] [CrossRef]
- Blanc, E.; Roux, G.L.; Benard, J.; Raguenez, G. Low expression of Wnt-5a gene is associated with high-risk neuroblastoma. Oncogene 2005, 24, 1277–1283. [Google Scholar] [CrossRef]
- Dyberg, C.; Papachristou, P.; Haug, B.H.; Lagercrantz, H.; Kogner, P.; Ringstedt, T.; Wickstrom, M.; Johnsen, J.I. Planar cell polarity gene expression correlates with tumor cell viability and prognostic outcome in neuroblastoma. BMC Cancer 2016, 16, 259. [Google Scholar] [CrossRef]
- Duffy, D.J.; Krstic, A.; Schwarzl, T.; Halasz, M.; Iljin, K.; Fey, D.; Haley, B.; Whilde, J.; Haapa-Paananen, S.; Fey, V.; et al. Wnt signalling is a bi-directional vulnerability of cancer cells. Oncotarget 2016, 7, 60310–60331. [Google Scholar] [CrossRef]
- Fishbein, L.; Leshchiner, I.; Walter, V.; Danilova, L.; Robertson, A.G.; Johnson, A.R.; Lichtenberg, T.M.; Murray, B.A.; Ghayee, H.K.; Else, T.; et al. Comprehensive Molecular Characterization of Pheochromocytoma and Paraganglioma. Cancer Cell 2017, 31, 181–193. [Google Scholar] [CrossRef] [PubMed]
- Dolecek, T.A.; Propp, J.M.; Stroup, N.E.; Kruchko, C. CBTRUS statistical report: Primary brain and central nervous system tumors diagnosed in the United States in 2005-2009. Neuro Oncol 2012, 14, v1–v49. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Lee, J.K.; Ahn, S.H.; Lee, J.; Nam, D.H. WNT signaling in glioblastoma and therapeutic opportunities. Lab. Invest. 2016, 96, 137–150. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.M.; Jun, E.S.; Jung, J.S.; Suh, S.Y.; Han, J.Y.; Kim, J.Y.; Kim, K.W.; Jung, J.S. Role of Wnt5a in the proliferation of human glioblastoma cells. Cancer Lett. 2007, 257, 172–181. [Google Scholar] [CrossRef] [PubMed]
- Ferletta, M.; Uhrbom, L.; Olofsson, T.; Ponten, F.; Westermark, B. Sox10 has a broad expression pattern in gliomas and enhances platelet-derived growth factor-B--induced gliomagenesis. Mol. Cancer Res. 2007, 5, 891–897. [Google Scholar] [CrossRef] [PubMed]
- Gallik, K.L.; Treffy, R.W.; Nacke, L.M.; Ahsan, K.; Rocha, M.; Green-Saxena, A.; Saxena, A. Neural crest and cancer: Divergent travelers on similar paths. Mech. Dev. 2017, 148, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Powell, D.R.; Blasky, A.J.; Britt, S.G.; Artinger, K.B. Riding the crest of the wave: Parallels between the neural crest and cancer in epithelial-to-mesenchymal transition and migration. Wiley Interdiscip. Rev. Syst. Biol. Med. 2013, 5, 511–522. [Google Scholar] [CrossRef] [PubMed]
- Zhan, T.; Rindtorff, N.; Boutros, M. Wnt signaling in cancer. Oncogene 2017, 36, 1461–1473. [Google Scholar] [CrossRef] [PubMed]
- Reya, T.; Clevers, H. Wnt signalling in stem cells and cancer. Nature 2005, 434, 843–850. [Google Scholar] [CrossRef]
- Kerosuo, L.; Bronner-Fraser, M. What is bad in cancer is good in the embryo: Importance of EMT in neural crest development. Semin. Cell Dev. Biol. 2012, 23, 320–332. [Google Scholar] [CrossRef]
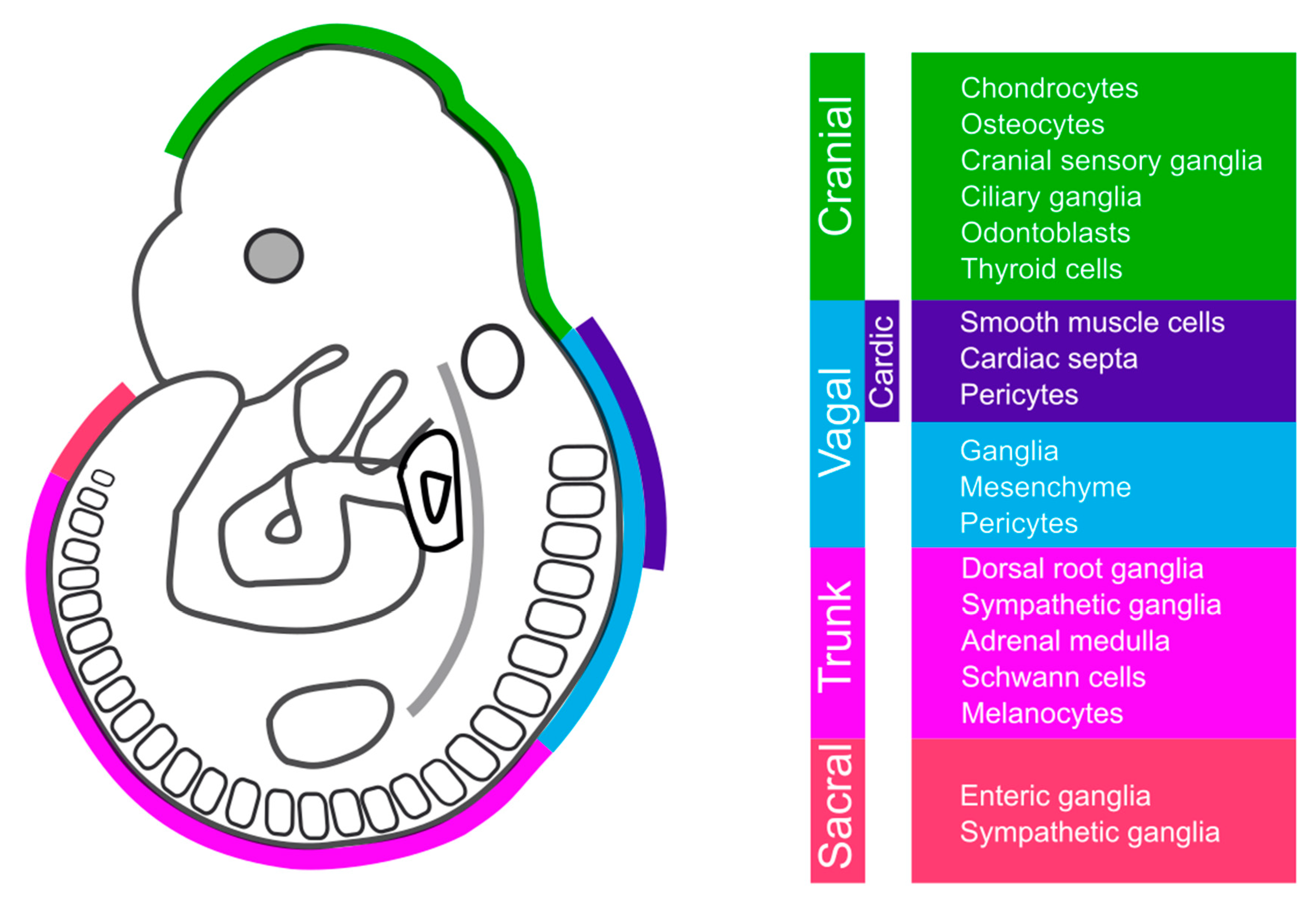
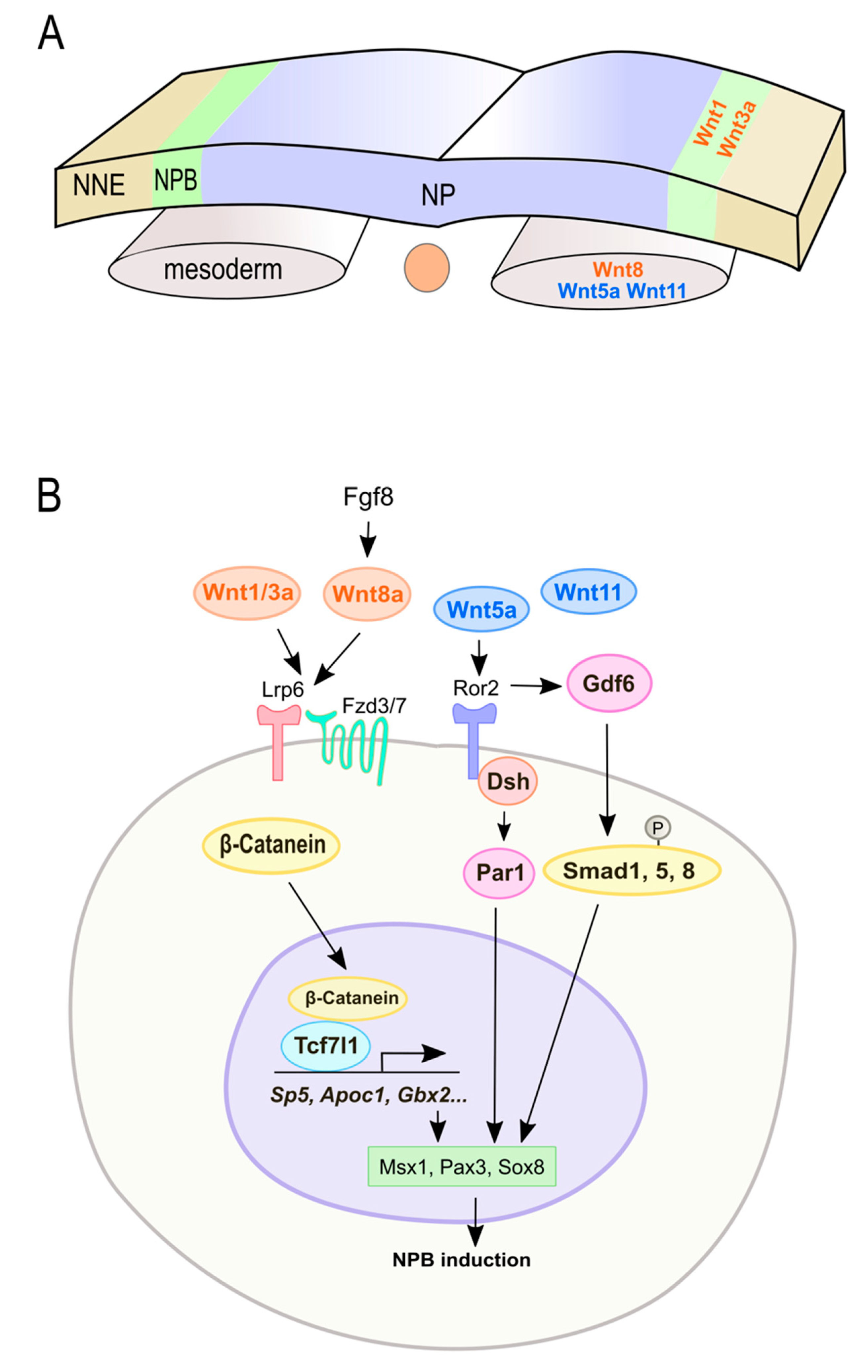


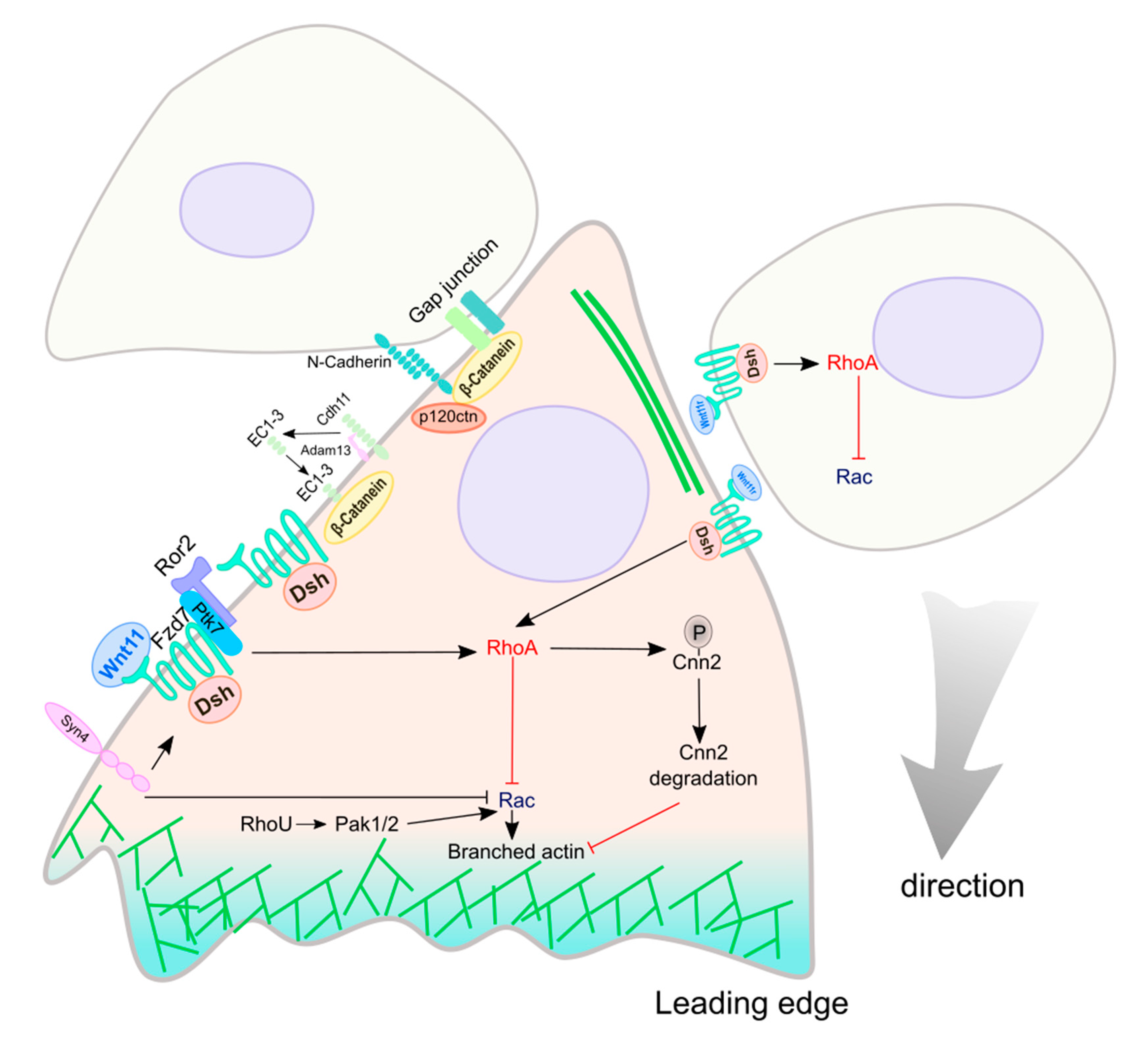
| Molecule | Role in Wnt Signaling | Experimental Approach | Function | Region | Species | Phenotype | Reference |
|---|---|---|---|---|---|---|---|
| ADAM13 | upregulates canonical Wnt signaling by cleaving class B Ephrins | MO | LOF | cranial | Xenopus | defective NC induction (Snai2, Sox9, Foxd3) and head cartilage | [75] |
| ADAM19 | upregulates canonical Wnt signaling by stabilizing ADAM13 | MO | LOF | cranial | Xenopus | defective NC induction (Snai2, Sox9, Foxd3) and head cartilage | [76] |
| Aldh1a2 | delay Wnt3a and Wnt8a expression | KO | LOF | trunk | mouse | diminished NPB specification (Msx1, Pax3) | [77] |
| Apoc1 | downstream effector of canonical Wnt signaling | MO | LOF | cranial | Xenopus | defective NPB induction (Msx1, Pax3, Zic1), eyes and head deformation | [78] |
| Awp1 | stabilizes Ctnnb1 | MO | LOF | cranial | Xenopus | defective NPB induction (Msx1, Pax3), pigmentation and craniofacial cartilage | [79] |
| Axud1 | downstream effector of canonical Wnt signaling | MO, dominant-negative construct | LOF | trunk | chick | defective NC inductions (Foxd3, Sox9, Sox10, Ets1) | [80] |
| Cdh11 | competitive binding with Ctnnb1 to repress Wnt/Ctnnb1 signaling | MO | LOF | cranial | Xenopus | increased Wnt/Ctnnb1 signaling and NC induction (Sox10, Ap2) | [81] |
| Ctnnb1 | coactivator for Tcf/Lef1 transcription factor | RNA injection, MO | GOF; LOF | cranial | Xenopus | expanded (GOF) or diminished (LOF) NC induction (Snai2, Twist) | [82,83] |
| Daam1 | mediator of non-canonical Wnt signaling and actin polymerization | MO, mutations | LOF | cranial | Xenopus | defective NC induction (Twist, Sox8, Snai2, Sox10) | [38] |
| Dkk1 | antagonist of canonical Wnt signaling | blocking antibody, Dkk1-null mouse | LOF | cranial | Xenopus, mouse | NC generated in the anterior neural fold, expanded cranial cartilages | [68] |
| Dkk2 | positive regulator of canonical Wnt signaling independent of Gsk3b | MO | LOF | cranial | Xenopus | defective NC induction (Snai2, Twist1, Sox10), reduced craniofacial cartilages | [34] |
| Dvl (Dsh) | canonical and non-canonical Wnt signaling | mutants (dd1, dd2) | LOF | cranial | Xenopus | repressed NC induction (Snai2) | [37] |
| PCP signaling | PCP mutants: ΔN-Dsh, Dsh-DEP+ | GOF, LOF | cranial | Xenopus | expanded (GOF) or decreased (LOF) NC induction (Foxd3, Sox8, Snai2) | [84] | |
| Fgf8a | induce Wnt8 in the paraxial mesoderm | MO, RNA injection | LOF, GOF | cranial | Xenopus | defective (LOF) NPB induction (Pax3), induced (GOF) NC in anterior neural plate | [83] |
| Fzd3 | receptor for Wnt1 | RNA injection, MO | GOF, LOF | cranial | Xenopus | Induced (GOF) or diminished (LOF) NC induction (Snai, Twist) | [85] |
| Fzd7 | receptor in canonical Wnt/Ctnnb1 signaling | MO | LOF | cranial | Xenopus | inhibited NC induction, loss of pigment cells | [86] |
| Gbx2 | direct target of canonical Wnt signaling | MO, RNA injection | LOF, GOF | cranial | Xenopus | diminished (LOF) or rescued (GOF) NPB specifiers (Pax3, Msx1) | [87] |
| Gsk3b | phosphorylation and degradation of Ctnnb1 | RNA Injections | GOF | cranial | Xenopus | increased NC induction (Krox20, Ap2, Snai2) | [88] |
| Hes3 | inhibition of Wnt/Ctnnb1 signaling | expression constructs | GOF | cranial, trunk | Xenopus | blocked NC specifiers (Snai2, Sox10), supernumerary pigment cells | [35] |
| kctd15 | attenuate canonical Wnt/Ctnnb1 signaling | MO, mRNA injection | LOF | cranial | zebrafish | defective NC induction (Sox10, Foxd3), increased pigmentation, loss of jaw elements | [89] |
| expression constructs | GOF | cranial, trunk | zebrafish | increased NC induction (Foxd3, Sox10), loss of pigmentation, small head | [89] | ||
| Krm2 | promotes Lrp6-mediated Wnt signaling in the absence of Dkks | MO, RNA microinjection | LOF, GOF | cranial | Xenopus | diminished (LOF) NC markers, ectopic NC-derived structures (GOF) | [90] |
| Lrp6 | Wnt co-receptor | normal and mutant RNA injection | GOF, LOF | cranial | Xenopus | induced (GOF) or diminished (LOF) NC specifier Snai2 | [47,90] |
| Mark2 (Par-1) | bind to Dvl and regulated by Wnt5a/Wnt11 | MO, mRNA injection | LOF | cranial | Xenopus | repressed (LOF) or enhanced NC specification (Sox8, Foxd3, Snai2) | [84] |
| Rap2 | stabilizes Lrp6 through TNIK kinase | MO, siRNA | LOF | cranial | Xenopus | abrogate ectopic expression of NC markers Snai, Foxd3 | [91] |
| retinoic acid | regulate Wnt1 and Wnt3a expression | vitamin A deficiency | LOF | trunk | quail | defective NPB (Pax7) and NC induction (Snai2 and Sox9) | [92] |
| Rgs2 | regulates Wnt-Ppard-Sox10 signaling cascade | MO, dominant negative rgs2 construct | LOF | cranial | zebrafish | increased NC induction (Sox10, Snail1b), reduced cranial cartilage formation | [93] |
| RhoV | downstream effector of canonical Wnt signaling | MO | LOF | cranial | Xenopus | defective NC induction (Sox9, Sox10, Snai), abnormal craniofacial skeletons | [94] |
| Ror2 | co-receptor in non-canonical Wnt/PCP signaling | MO | LOF | cranial | Xenopus | defective NPB induction (Gbx2, Zic1, Msx1, Msx2), decreased BMP signaling at NPB | [84,95] |
| Skip | a potential scaffold in Ctnnb1/Tcf transcriptional regulation | MO, siRNA | LOF | cranial | Xenopus | defective NC induction (Snai2, Sox3, Foxd3), loss of pigment cells | [96] |
| Sp5 | downstream effector of canonical Wnt and Fgf pathways | MO | LOF | cranial | Xenopus | defective NPB induction (Msx1, Pax3); defects in craniofacial cartilage and pigmentation | [97] |
| Tcf7l1 | transcription factor | cKO by AP2α-Cre | LOF | cranial | mouse | anteriorly expanded NC specifiers (Foxd3, Sox9, Sox10, Pax3); exencephaly | [67] |
| inhibitory mutants | LOF | cranial | Xenopus | defective NPB induction (Msx1) | [98] | ||
| THVGR (hormone-inducible Tcf7l1) | GOF | cranial | Xenopus | increased NC induction (Snai2, Twist) | [82] | ||
| Wnt1 | ligand, canonical pathway | dominant negative Wnt1 | LOF | trunk | chick | repressed NC induction (Snai2) | [99] |
| Wnt1 expressing cells | GOF | trunk | chick | inhibited NC induction | [100] | ||
| Wnt1/Wnt3a | ligand, canonical pathway | RNA and DNA Injections | GOF | cranial | Xenopus | increased NC induction (Krox20, Ap2, Snai2) | [88] |
| Wnt1 and/or Wnt3a knockouts | LOF | cranial, trunk | mouse | defective NPB induction (Pax3), cranial skeletons, cranial ganglia | [101] | ||
| Wnt5a | ligand, non-canonical pathway | MO, dominant negative, RNA injection | LOF, GOF | cranial | Xenopus | defective (LOF) or enhanced (GOF) NC specification (Pax3, Foxd3, Sox8) | [84] |
| Wnt6 | ligand, upstream of Dvl-Rho-JNK in PCP signaling | Wnt6 cell implantation, siRNA, | GOF, LOF | trunk | chick | induced (GOF) or diminished (LOF) NC induction | [100] |
| Wnt7b | ligand | RNA microinjection | GOF | cranial | Xenopus | increased NC induction (Snai2, Twist) | [102] |
| Wnt8 | ligand, canonical pathway, downstream of Fgf8a | MO, mRNA injection | LOF, GOF | cranial | Xenopus | defective (LOF) NPB induction (Pax3, Sox8), rescued (GOF) NC in Fgf8a-deficient embryos | [83] |
| dominant negative, RNA microinjection | LOF, GOF | cranial | Xenopus | defective (LOF) or increased (GOF) NC induction (Snai2) | [103] | ||
| Wnt11 | ligand, non-canonical pathway | MO, dominant negative, RNA injection | LOF | cranial | Xenopus | defective (LOF) or enhanced (GOF) NC specification (Pax3, Foxd3, Sox8) | [84] |
| Wnt11r | ligand, non-canonical pathway | MO | LOF | cranial | Xenopus | defective NC specification (Foxd3, Sox8) | [84] |
| Molecule | Role in Wnt Signaling | Experimental Approach | Function | Region | Species | Phenotype | Reference |
|---|---|---|---|---|---|---|---|
| ADAM13 | regulated by Gsk3 and Plk | MO | LOF | cranial | Xenopus | inhibited NC migration | [142] |
| Bmp4 | stimulate Wnt1 expression | BMP4-coated microbeads | GOF | trunk | chick, quail | promoted G1/S transition and NC delamination | [137] |
| Cdh2 | cleaved product CTF2 induces Ctnnb1 expression | expression vectors | GOF | trunk | quail | enhanced NC delamination | [143] |
| KO | LOF | cardiac | mouse | elevated NC proliferation and reduced NC migration | [144] | ||
| Cnn2 | downstream PCP signaling, actin dynamics | MO | LOF | cranial | chick, Xenopus | inhibited NC migration and reduced cartilage | [145] |
| Ctnnb1 | coactivator for Tcf/Lef1 transcription factor | overexpression | GOF | trunk | chick | rescued NC delamination in Noggin-treated neural tubes | [143] |
| Dact1 | repress Ctnnb1 as the transcriptional coactivator | MO, expression vectors | LOF, GOF | cranial | Xenopus | blocked (LOF) or enhanced (GOF) NC delamination | [146] |
| Dact2 | repress Ctnnb1 as the transcriptional coactivator | RNAi, expression vectors | LOF, GOF | truck | chick | blocked (LOF) or enhanced (GOF) NC delamination | [146] |
| Dmxl2 (Rbc3a) | regulate Fzd7 endocytosis and enhance Wnt signaling | MO | LOF | cranial, trunk | zebrafish | defective NC migration, cardiac edema, reduced melanocytes | [147] |
| Draxin | repress Wnt signaling via Lrp5, modulate laminin | MO, CRISPR; expression vectors | LOF, GOF | cranial | chick | premature NC delamination (LOF), inhibited EMT (GOF) | [136,148] |
| Dvl (Dsh) | PCP signaling | PCP mutants (Dsh-DN, Dsh-DEP+) | LOF | cranial | Xenopus | repressed NC migration | [37] |
| Efhc1 (Efhc1b) | downregulate Wnt8a | MO | LOF | cranial | Xenopus | upregulated Wnt signaling and defective NC migration | [149] |
| Fgf8/4 | inhibit Wnt1 expression | Fgf8/4-soaked beads | GOF | trunk | chick | repressed NC emigration or delamination | [92] |
| Fgfr1 | inhibit Wnt1 expression | dominant-negative, inhibitor | LOF | trunk | chick | premature NC emigration | [92] |
| Gsk3 | phosphorylation and degradation of Ctnnb1 | Gsk3 inhibitors LiCl, BIO | Wnt GOF | trunk, cranial | chick, Xenopus | inhibited NC delamination and migration | [150,151] |
| Lef1 | transcription factor, canonical pathway | inducible Lef1-GR | GOF | cranial | Xenopus | repressed NC migration | [151] |
| Lrp5 | co-receptor | MO, CRISPR | LOF | cranial | zebrafish | defective NC migration, cranial skeleton malformation | [152] |
| Musk | downstream of non-canonical Wnt11r-Dsh signaling | transgenic fish, KO mouse | LOF | trunk | zebrafish, mouse | defective segmental NC migration | [153] |
| Noggin | inhibit Wnt1 expression | CHO-Noggin cells | LOF | trunk | Chick | inhibited G1/S transition and NC delamination | [143] |
| Ovo1 | Wnt target, regulate intracellular trafficking of Cdh2 | MO | LOF | cranial | zebrafish | defective migration of NC-derived pigment precursors | [154] |
| Pes1 (Pescadillo) | downstream of Wnt4/Fzd3 | MO | LOF | cranial | Xenopus | increased apoptosis; defective NC migration, eye and craniofacial cartilage | [155] |
| Ptk7 | co-receptor, interact with Ror2 in Wnt/PCP signaling | MO | LOF | cranial | Xenopus | defective NC migration | [156] |
| required for Fzd7-mediated Dsh localization | MO | LOF | cranial | Xenopus | defective NC migration | [157] | |
| Rara | regulate Wnt1/Wnt3a expression | dominant negative/active RA receptors | LOF, GOF | trunk | chick | diminished (LOF) or enhanced (GOF) NC emigration | [92] |
| RhoA, RhoB | regulated by Wnt6 [100] | dominant-negative, inhibitor, activator | LOF, GOF | trunk | chick | enhanced (LOF) or repressed (GOF) NC EMT/delamination | [158] |
| RhoU | activated by Wnt1 [159] | mutant construct, MO, RNA injection | LOF, GOF | cranial | chick, Xenopus | blocked (LOF) or impaired NC migration, reduced cartilages | [160] |
| Ror2 | co-receptor, interact with Ptk in Wnt/PCP signaling | expression vector | GOF | cranial | Xenopus | rescued migration defect in Ptk7-deficient NC cells | [156] |
| Sfrp (Fz4-v1) | secreted splice variant of fz4 receptor | MO, mRNA injection | LOF, GOF | cranial, trunk | Xenopus | defective NC migration (LOF), altered Wnt signaling | [161] |
| Sox9 | phosphorylated by Wnt1 and Bmp signaling | MO; point mutations | LOF | trunk | chick | failed to initiate NC delamination | [162] |
| Syn4 (Syndecan4) | interact with Wnt5/Dvl/PCP signaling | MO | LOF | trunk | Xenopus, zebrafish | diminished NC migration, reduced cartilage and melanocytes | [163] |
| Tcf7l1 (Tcf3) | transcription factor, canonical pathway | inducible Tcf3-VP16-GR, Tcf3ΔC-GR | GOF, LOF | cranial | Xenopus | impaired NC migration | [151] |
| Vgll3 | induce Wnt5a and Wnt8b expression | MO, mRNA injection | LOF, GOF | cranial | Xenopus | impaired NC migration, trigeminal and profundal placodes | [164] |
| Wnt1 | ligand, canonical pathway | Wnt1 producing cells | GOF | trunk | chick | inhibited NC delamination and migration | [150] |
| Wnt1 DNA electroporation | GOF | trunk | chick | enhanced NC delamination | [143] | ||
| Wnt3a | ligand, canonical pathway | Wnt3a cells, melanoma cells | GOF | trunk | chick, human cell line | enhanced EMT and NC migration | [165] |
| Wnt5 | ligand, PCP signaling | MO | LOF | trunk | Xenopus | defective NC migration | [163] |
| Wnt11 | ligand, PCP signaling | expression vectors | LOF, GOF | cranial | Xenopus | inhibited NC migration | [37] |
| Wnt11r | ligand | MO, mRNA injection | LOF, GOF | cranial | Xenopus | repressed or rescued NC migrating | [166] |
| Yap (Yap1) | bidirectional crosstalk with Wnt and Bmp signaling | expression vectors, mutants, siRNA | GOF, LOF | trunk | chick, quail | stimulated (GOF) or inhibited (LOF) NC EMT and emigration | [139] |
| Molecule | Role in Wnt Signaling | Experimental Approach | Function | Region | Species | Phenotype | Reference |
|---|---|---|---|---|---|---|---|
| Axin2 | scaffold protein for Ctnnb1 degradation | KO | LOF | cranial | mouse | enhanced osteogenic potential and regeneration of NC-derived frontal bone | [211] |
| Bmp2 | crosstalk with Wnt/Ctnnb1 signaling | protein to NC culture | GOF | truck | mouse | suppressed sensory neurogenesis of early NC stem cells | [205] |
| repress Wnt antagonists Dkk1 and Sost | cKO by Wnt1-Cre | LOF | tooth | mouse | early tooth mineralization defects | [212] | |
| Bmp4 | repress Wnt antagonists Dkk2 and Sfrp2 | cKO by Wnt1-Cre | LOF | tooth | mouse | bud-stage arrest of the mandibular molar tooth germs | [213] |
| Chd7 | chromatin remodeler, activated by Wnt/Bmp | siRNA, DN, WT expression | LOF, GOF | trunk, DRG | mouse | inhibited (LOF) or maintained (GOF) undifferentiated state (Sox10, p75) of NCSC | [27] |
| Ctnnb1 | coactivator for Tcf/Lef1 transcription factor | mRNA injection | GOF | cranial | zebrafish | promoted pigment and cartilage fates from NC cells | [214] |
| ca by Wnt1-Cre in premigratory NC cells | GOF | trunk | mouse | suppressed melanocyte (Dct, Mitf) differentiation from premigratory NC cells | [39] | ||
| ca by Sox10-Cre in migratory NC cells | GOF | trunk | mouse | ectopic melanocytes, inhibited other lineages from migratory NC cells | [39] | ||
| cKO by Wnt1-cre | LOF | trunk | mouse | lack melanocytes and dorsal root ganglia | [40] | ||
| ca, cKO by Wnt1-Cre | GOF, LOF | trunk, cranial | mouse | promoted (GOF) or blocked (LOF) sensory neurogenesis of NC stem cells | [215] | ||
| cKO by PdgfraCreErt2, Dermo1Cre | LOF | cranial | mouse | forebrain meningeal hypoplasia derived from NC cells | [216] | ||
| Wnt1-Cre | LOF | cranial | mouse | defective maintenance of Pitx2 expression in NC cells and abnormal eyes | [217] | ||
| Wnt1-cre | LOF | cranial | mouse | affected NC survival and differentiation; failure of craniofacial development | [115] | ||
| let-7 miRNA | repressed by Wnt/Lin28a | electroporation of let-7 mimic | GOF | trunk | chick | down-regulation of NC multipotency, promoted differentiation | [209] |
| Lin28a | activated by Wnt | siRNA; MO; CRISPR | LOF | trunk | chick | suppressed NC multipotency (Sox10, Foxd3) | [209] |
| Msx1 | repress Dkk2 and Sfrp2, interact Bmp4 | KO | LOF | tooth | mouse | bud-stage arrest of the mandibular molar tooth germs | [213] |
| Osr2 | upregulate Dkk2 and Sfrp2 expression | KO | LOF | tooth | mouse | supernumerary teeth | [213,218] |
| Prmt1 | inhibit Wnt, Bmp and other signaling | cKO by Wnt1-Cre | LOF | cranial | mouse | decreased mesenchymal proliferation, cleft palate, craniofacial anomalies | [219] |
| Tcf7l1 (Tcf3) | transcription factor of Wnt/Ctnnb1 signaling | mutant mRNA injection | LOF | cranial | zebrafish | promoted neural fates, repressed pigment cells | [214] |
| Wnt1 | ligand, Ctnnb1-dependent | Wnt1-expressing fibroblasts | GOF | trunk | mouse | promoted sensory neurogenesis of early NC stem cells | [215] |
| Wnt1 and Bmp2 | ligands | Wnt1 cell and Bmp2 protein | GOF | truck | mouse | repressed neurogenesis and maintained multipotency of NC stem cells | [205] |
| Wnt3a and Bmp2 | ligands | proteins to NC culture | GOF | trunk, DRG | mouse | maintained multipotency (Sox10 or p75) of NC stem cells | [27] |
| Wnt inhibitors | stabilize or elevate Axin | XAV939, IWR1 | LOF | cranial | Xenopus | repressed NC differentiation and defective cartilage | [188] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ji, Y.; Hao, H.; Reynolds, K.; McMahon, M.; Zhou, C.J. Wnt Signaling in Neural Crest Ontogenesis and Oncogenesis. Cells 2019, 8, 1173. https://doi.org/10.3390/cells8101173
Ji Y, Hao H, Reynolds K, McMahon M, Zhou CJ. Wnt Signaling in Neural Crest Ontogenesis and Oncogenesis. Cells. 2019; 8(10):1173. https://doi.org/10.3390/cells8101173
Chicago/Turabian StyleJi, Yu, Hongyan Hao, Kurt Reynolds, Moira McMahon, and Chengji J. Zhou. 2019. "Wnt Signaling in Neural Crest Ontogenesis and Oncogenesis" Cells 8, no. 10: 1173. https://doi.org/10.3390/cells8101173
APA StyleJi, Y., Hao, H., Reynolds, K., McMahon, M., & Zhou, C. J. (2019). Wnt Signaling in Neural Crest Ontogenesis and Oncogenesis. Cells, 8(10), 1173. https://doi.org/10.3390/cells8101173





