The Effect of Neurotoxin MPTP and Neuroprotector Isatin on the Profile of Ubiquitinated Brain Mitochondrial Proteins
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Animals, MPTP, and Isatin Administration, and Behavioral Tests
2.3. Isolation of Mitochondrial Fraction and Sample Preparation for Mass Spectrometry
2.4. Mass Spectrometry and Liquid Chromatography
2.5. Protein Identification and GO Annotation
3. Results
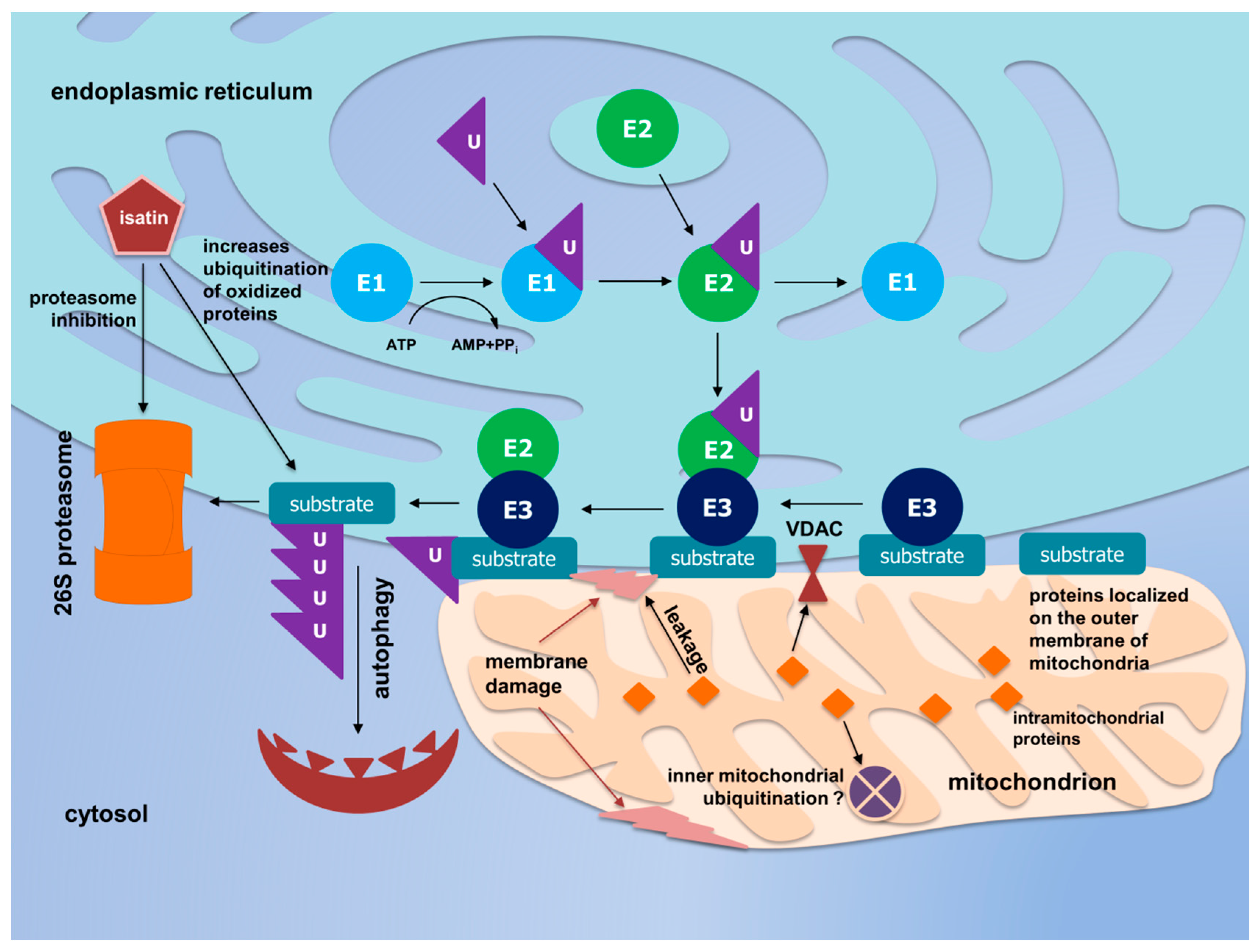
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cheng, A.; Hou, Y.; Mattson, M.P. Mitochondria and neuroplasticity. ASN Neuro 2010, 2, AN20100019. [Google Scholar] [CrossRef] [PubMed]
- Cookson, M.R. The biochemistry of Parkinson’s disease. Annu. Rev. Biochem. 2005, 74, 9–52. [Google Scholar] [CrossRef] [PubMed]
- Buneeva, O.A.; Medvedev, A.E. Mitochondrial Disfunction in Parkinson’s Disease. Biochem. (Mosc.) Suppl. Ser. B Biomed. Chem. 2011, 5, 313–336. [Google Scholar] [CrossRef]
- Maret, G.; Testa, B.; Jenner, P.; el Tayar, N.; Carrupt, P.A. The MPTP story: MAO activates tetrahydropyridine derivatives to toxins causing parkinsonism. Drug Metab. Rev. 1990, 22, 291–332. [Google Scholar] [CrossRef] [PubMed]
- Park, J.S.; Davis, R.L.; Sue, C.M. Mitochondrial Dysfunction in Parkinson’s Disease: New Mechanistic Insights and Therapeutic Perspectives. Curr. Neurol. Neurosci. Rep. 2018, 18, 21. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Zhao, Z.Q.; Xie, J.X. Effect of isatin on rotational behavior and DA levels in caudate putamen in Parkinsonian rats. Brain Res. 2001, 917, 127–132. [Google Scholar] [CrossRef]
- Hamaue, N.; Minami, M.; Terado, M.; Hirafuji, M.; Endo, T.; Machida, M.; Hiroshige, T.; Ogata, A.; Tashiro, K.; Saito, H.; et al. Comparative study of the effects of isatin, an endogenous MAO-inhibitor, and selegiline on bradykinesia and dopamine levels in a rat model of Parkinson’s disease induced by the Japanese Encephalitis virus. NeuroToxicology 2004, 25, 205–213. [Google Scholar] [CrossRef]
- Melamed, E.; Youdim, M.B.H. Prevention of dopaminergic toxicity of MPTP in mice by phenylethylamine, a specific substrate of type B monoamine oxidase. Br. J. Pharmacol. 1985, 86, 529–531. [Google Scholar] [CrossRef] [PubMed]
- Youdim, M.; Edmondson, D.; Tipton, K. The therapeutic potential of monoamine oxidase inhibitors. Nat. Rev. Neurosci. 2006, 7, 295–308. [Google Scholar] [CrossRef] [PubMed]
- Hoglinger, G.U.; Carrard, G.; Michel, P.P.; Medja, F.; Lombes, A.; Ruberg, M.; Friguet, B.; Hirsch, E.C. Dysfunction of mitochondrial complex I and the proteasome: Interactions between two biochemical deficits in a cellular model of Parkinson’s disease. J. Neurochem. 2003, 86, 1297–1307. [Google Scholar] [CrossRef] [PubMed]
- Fornai, F.; Schluter, O.M.; Lenzi, P.; Gesi, M.; Ruffoli, R.; Ferrucci, M.; Lazzeri, G.; Busceti, C.L.; Pontarelli, F.; Battaglia, G.; et al. Parkinson-like syndrome induced by continuous MPTP infusion: Convergent roles of the ubiquitin proteasome system and α-synuclein. Proc. Natl. Acad. Sci. USA 2005, 102, 3413–3418. [Google Scholar] [CrossRef] [PubMed]
- Hershko, A.; Ciechanover, A. The ubiquitin system. Annu. Rev. Biochem. 1998, 67, 425–479. [Google Scholar] [CrossRef] [PubMed]
- Hershko, A.; Ciechanover, A.; Varshavsky, A. The ubiquitin system. Nat. Med. 2000, 6, 1073–1081. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, A.L.; Ciechanover, A. Targeting proteins for destruction by the ubiquitin system: Implications for human pathobiology. Annu. Rev. Pharmacol. Toxicol. 2009, 49, 73–96. [Google Scholar] [CrossRef] [PubMed]
- Hamazaki, J.; Sasaki, K.; Kawahara, H.; Hisanaga, S.; Tanaka, K.; Murata, S. Rpn10-Mediated degradation of ubiquitinated proteins is essential for mouse development. Mol. Cell. Biol. 2007, 19, 6629–6638. [Google Scholar] [CrossRef] [PubMed]
- Saeki, Y. Ubiquitin recognition by the proteasome. J. Biochem. 2017, 161, 113–124. [Google Scholar] [CrossRef] [PubMed]
- Medvedev, A.E.; Buneeva, O.A.; Kopylov, A.T.; Tikhonova, O.V.; Medvedeva, M.V.; Nerobkova, L.N.; Kapitsa, I.G.; Zgoda, V.G. The brain mitochondrial subproteome of Rpn10-binding proteins and its changes induced by the neurotoxin MPTP and the neuroprotector Isatin. Biochemistry (Moscow) 2017, 82, 330–333. [Google Scholar] [CrossRef] [PubMed]
- Buneeva, O.A.; Gnedenko, O.V.; Kopylov, A.T.; Medvedeva, M.V.; Zgoda, V.G.; Ivanov, A.S.; Medvedev, A.E. Quantitative affinity interaction of ubiquitinated and non-ubiquitinated proteins with proteasome subunit Rpn10. Biochemistry (Moscow) 2017, 82, 1042–1047. [Google Scholar] [CrossRef] [PubMed]
- Prut, L.; Belzung, C. The open field as a paradigm to measure the effects of drugs on anxiety-like behaviors: A review. Eur. J. Pharmacol. 2003, 463, 3–33. [Google Scholar] [CrossRef]
- Buneeva, O.; Medvedeva, M.; Kopylov, A.; Zgoda, V.; Medvsedev, A. Use of biotinylated ubiquitin for analysis of rat brain mitochondrial proteome and interactome. Int. J. Mol. Sci. 2012, 3, 11593–11609. [Google Scholar] [CrossRef] [PubMed]
- Medvedev, A.; Buneeva, O.; Kopylov, A.; Gnedenko, O.; Ivanov, A.; Zgoda, V.; Makarov, A.A. Amyloid-binding proteins: Affinity-based separation, proteomic identification, and optical biosensor validation. Methods Mol. Biol. 2015, 1295, 465–477. [Google Scholar] [PubMed]
- Vaudel, M.; Burkhart, J.M.; Zahedi, R.P.; Oveland, E.; Berven, F.S.; Sickmann, A.; Martens, L.; Barsnes, H. PeptideShaker enables reanalysis of MS-derived proteomics data sets. Nat. Biotechnol. 2015, 33, 22–24. [Google Scholar] [CrossRef] [PubMed]
- Vaudel, M.; Barsnes, H.; Berven, F.S.; Sickmann, A.; Martens, L. SearchGUI: An open-source graphical user interface for simultaneous OMSSA and X!Tandem searches. Proteomics 2011, 11, 996–999. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, V.N.; Perlman, D.H.; Costello, C.E.; McComb, M.E. Software tool for researching annotations of proteins: Open-source protein annotation software with data visualization. Anal. Chem. 2009, 81, 9819–9823. [Google Scholar] [CrossRef] [PubMed]
- Buneeva, O.A.; Kopylov, A.T.; Nerobkova, L.N.; Kapitsa, I.G.; Zgoda, V.G.; Medvedev, A.E. The effect of neurotoxin MPTP administration to mice on the proteomic profile of brain isatin-binding proteins. Biochem. (Mosc.) Suppl. Ser. B Biomed. Chem. 2018, 12, 22–26. [Google Scholar] [CrossRef]
- Medvedev, A.; Buneeva, O.; Gnedenko, O.; Ershov, P.; Ivanov, A. Isatin, an endogenous non-peptide biofactor: A review of its molecular targets, mechanisms of actions and their biomedical implications. Biofactors 2018, 44, 95–108. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.S.; Jeong, J.H.; Min, H.K.; Jung, H.J.; Hwang, D.; Lee, S.W.; Pak, Y.K. Shot-gun proteomic analysis of mitochondrial D-loop DNA binding proteins: Identification of mitochondrial histones. Mol. BioSyst. 2011, 7, 1523–1536. [Google Scholar] [CrossRef] [PubMed]
- Cascone, A.; Bruelle, C.; Lindholm, D.; Bernardi, P.; Eriksson, O. Destabilization of the outer and inner mitochondrial membranes by core and linker histones. PLoS ONE 2012, 7, e35357. [Google Scholar] [CrossRef] [PubMed]
- Bragoszewski, P.; Gornicka, A.; Sztolsztener, M.E.; Chacinska, A. The ubiquitin-proteasome system regulates mitochondrial intermembrane space proteins. Mol. Cell. Biol. 2013, 33, 2136–2148. [Google Scholar] [CrossRef] [PubMed]
- Bragoszewski, P.; Turek, M.; Chacinska, A. Control of mitochondrial biogenesis and function by the ubiquitin–proteasome system. Open Biol. 2017, 7, 170007. [Google Scholar] [CrossRef] [PubMed]
- Brand, M.D.; Esteves, T.C. Physiological functions of the mitochondrial uncoupling proteins UCP2 and UCP3. Cell Metab. 2005, 2, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Azzi, V.; Brandt, M.D. Degradation of an intramitochondrial protein by the cytosolic proteasome. J. Cell Sci. 2009, 123, 578–585. [Google Scholar] [CrossRef] [PubMed]
- Vertegaal, A.C. Uncovering ubiquitin and ubiquitin-like signaling networks. Chem. Rev. 2011, 111, 7923–7940. [Google Scholar] [CrossRef] [PubMed]
- Buneeva, O.A.; Medvedev, A.E. The role of atypical ubiquitination in cell regulation. Biochem. (Mosc.) Suppl. Ser. B Biomed. Chem. 2017, 11, 16–31. [Google Scholar] [CrossRef]
- Marchi, S.; Patergnani, S.; Pinton, P. The endoplasmic reticulum–mitochondria connection: One touch, multiple functions. Biochim. Biophys. Acta 2014, 1837, 461–469. [Google Scholar] [CrossRef] [PubMed]
- Plotegher, N.; Duchen, M.R. Crosstalk between lysosomes and mitochondria in Parkinson’s disease. Front. Cell Dev. Biol. 2017, 5, 110. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, G.; Udasin, R.G.; Ciechanover, A. On the linkage between the ubiquitin-proteasome system and the mitochondria. Biochem. Biophys. Res. Commun. 2016, 473, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, G.; Ziv, T.; Braten, O.; Admon, A.; Udasin, R.G.; Ciechanover, A. Ubiquitination of specific mitochondrial matrix proteins. Biochem. Biophys. Res. Commun. 2016, 475, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Cassandri, M.; Smirnov, A.; Novelli, F.; Pitolli, C.; Agostini, M.; Malewicz, M.; Melino, G.; Raschellà, G. Zinc-finger proteins in health and disease. Cell Death Discov. 2017, 3, 17071. [Google Scholar] [CrossRef] [PubMed]
- Hanz, S.; Perlson, E.; Willis, D.; Zheng, J.Q.; Massarwa, R.; Huerta, J.J. Axoplasmic importins enable retrograde injury signaling in lesioned nerve. Neuron 2003, 40, 1095–1104. [Google Scholar] [CrossRef]
- Perez, M.; Santa-Maria, I.; Gomez de Barreda, E.; Zhu, X.; Cuadros, R.; Cabrero, J.R.; Sanchez-Madrid, F.; Dawson, H.N.; Vitek, M.P.; Perry, G.; et al. Tau—An inhibitor of deacetylase HDAC6 function. J. Neurochem. 2009, 109, 1756–1766. [Google Scholar] [CrossRef] [PubMed]
- Doyle, J.M.; Gao, J.; Wang, J.; Yang, M.; Potts, P.R. MAGE-RING protein complexes comprise a family of E3 ubiquitin ligases. Mol. Cell 2010, 39, 963–974. [Google Scholar] [CrossRef] [PubMed]
- Liu, A.; Yu, Q.; Peng, Z.; Huang, Y.; Diao, S.; Cheng, J.; Wang, W.; Hong, M. Mir-200b inhibits CD133+ glioma cells by targeting the AKT pathway. Oncol. Lett. 2017, 13, 4701–4707. [Google Scholar] [CrossRef] [PubMed]
- Grigaravicius, P.; von Deimling, A.; Frappart, P.-O. RINT1 functions as a multitasking protein at the crossroads between genomic stability, ER homeostasis, and autophagy. Autophagy 2016, 12, 1413–1415. [Google Scholar] [CrossRef] [PubMed]
- Damerla, R.R.; Cui, C.; Gabriel, G.C.; Liu, X.; Craige, B.; Gibbs, B.C.; Francis, R.; Li, Y.; Chatterjee, B.; San Agustin, J.T.; et al. Novel Jbts17 mutant mouse model of Joubert syndrome with cilia transition zone defects and cerebellar and other ciliopathy related anomalies. Hum. Mol. Genet. 2015, 24, 3994–4005. [Google Scholar] [CrossRef] [PubMed]
- Gardoni, F.; Marcello, E.; Di Luca, M. Postsynaptic density-membrane associated guanylate kinase proteins (PSD-MAGUKs) and their role in CNS disorders. Neuroscience 2009, 158, 324–333. [Google Scholar] [CrossRef] [PubMed]
- Qin, H.; Buckley, J.A.; Li, X.; Liu, Y.; Fox, T.H., III; Meares, G.P.; Yu, H.; Yan, Z.; Harms, A.S.; Li, Y.; et al. Inhibition of the JAK/STAT pathway protects against α-synuclein-induced neuroinflammation and dopaminergic neurodegeneration. J. Neurosci. 2016, 26, 5144–5159. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, M.; Bogdanovic, N.; Nakagawa, H.; Volkmann, I.; Aoki, M.; Winblad, B.; Sakai, J.; Tjernberg, L.O. Analysis of microdissected neurons by 18O mass spectrometry reveals altered protein expression in Alzheimer’s disease. J. Cell. Mol. Med. 2012, 16, 1686–1700. [Google Scholar] [CrossRef] [PubMed]
- Chano, T.; Okabe, H.; Hulette, C.M. RB1CC1 insufficiency causes neuronal atrophy through mTOR signaling alteration and involved in the pathology of Alzheimer’s diseases. Brain Res. 2007, 1168, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.M.; Jiang, J.; Wilson, B.; Zhang, W.; Hong, J.S.; Liu, B. Microglial activation-mediated delayed and progressive degeneration of rat nigral dopaminergic neurons: Relevance to Parkinson’s disease. J. Neurochem. 2002, 81, 1285–1297. [Google Scholar] [CrossRef] [PubMed]
- Martin, H.L.; Teismann, P. Glutathione—A review on its role and significance in Parkinson’s disease. FASEB J. 2009, 23, 3263–3272. [Google Scholar] [CrossRef] [PubMed]
- Hamdane, M.; Smet, C.; Sambo, A.V.; Leroy, A.; Wieruszeski, J.M.; Delobel, P.; Maurage, C.A.; Ghestem, A.; Wintjens, R.; Bégard, S.; et al. Pin1: A therapeutic target in Alzheimer neurodegeneration. J. Mol. Neurosci. 2002, 19, 275–287. [Google Scholar] [CrossRef]
- Ryu, S.J.; Park, S.C. Targeting major vault protein in senescence-associated apoptosis resistance. Expert Opin. Ther. Targets 2009, 13, 479–484. [Google Scholar] [CrossRef] [PubMed]
- Guven, A.; Tolun, A. TBC1D24 truncating mutation resulting in severe neurodegeneration. J. Med. Genet. 2013, 50, 199–202. [Google Scholar] [CrossRef] [PubMed]
- Alkallas, R.; Fish, L.; Goodarzi, H.; Najafabadi, H.S. Inference of RNA decay rate from transcriptional profiling highlights the regulatory programs of Alzheimer’s disease. Nat. Commun. 2017, 8, 909. [Google Scholar] [CrossRef] [PubMed]
- Kepa, A.; Medina, L.M.; Erk, S.; Srivastava, D.P.; Fernandes, A.; Toro, R.; Lévi, S.; Ruggeri, B.; Fernandes, C.; Degenhardt, F.; et al. Associations of the intellectual disability gene MYT1L with helixloop-helix gene expression, hippocampus volume and hippocampus activation during memory retrieval. Neuropsychopharmacology 2017, 42, 2516–2526. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, D.P.; Copits, B.A.; Xie, Z.; Huda, R.; Jones, K.A.; Mukherji, S.; Cahill, M.E.; VanLeeuwen, J.-E.; Woolfrey, K.M.; Rafalovich, I.; et al. Afadin is required for maintenance of dendritic structure and excitatory tone. J. Biol. Chem. 2012, 287, 35964–35974. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Li, J.; Wang, H.; Wang, G. NUCB2/nesfatin-1: Expression and functions in the regulation of emotion and stress. Prog. Neuropsychopharmacol. Biol. Psychiatr. 2018, 81, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Budanov, A.V.; Park, E.J.; Birse, R.; Kim, T.E.; Perkins, G.A.; Ocorr, K.; Ellisman, M.H.; Bodmer, R.; Bier, E.; et al. Sestrin as a feedback inhibitor of tor that prevents age-related pathologies. Science 2010, 327, 1223–1228. [Google Scholar] [CrossRef] [PubMed]
- Henderson-Smith, A.; Jason, J.; Corneveaux, J.J.; De Both, M.; Cuyugan, L.; Liang, W.S.; Huentelman, M.; Adler, C.; Driver-Dunckley, E.; Beach, T.G.; et al. Next-generation profiling to identify the molecular etiology of Parkinson dementia. Neurol. Genet. 2016, 2, e75. [Google Scholar] [CrossRef] [PubMed]
- Neisch, A.L.; Neufeld, T.P.; Thomas, S.; Hays, T.S. A STRIPAK complex mediates axonal transport of autophagosomes and dense core vesicles through PP2A regulation. J. Cell Biol. 2017, 216, 441–461. [Google Scholar] [CrossRef] [PubMed]
- Bishop, P.; Dan Rocca, D.; Henley, J.M. Ubiquitin C-terminal hydrolase L1 (UCH-L1): Structure, distribution and roles in brain function and dysfunction. Biochem. J. 2016, 473, 2453–2462. [Google Scholar] [CrossRef] [PubMed]
- Ko, H.S.; Kim, S.W.; Sriram, S.R.; Dawson, V.L.; Dawson, T.M. Identification of far upstream element-binding protein-1 as an authentic Parkin substrate. J. Biol. Chem. 2006, 281, 16193–16196. [Google Scholar] [CrossRef] [PubMed]
- Ginguay, A.; Cynober, L.; Curis, E.; Nicolis, I. Ornithine aminotransferase, an important glutamate-metabolizing enzyme at the crossroads of multiple metabolic pathways. Biology 2017, 6, 18. [Google Scholar] [CrossRef] [PubMed]
- Park, M.H.; Jo, M.; Kim, Y.R.; Lee, C.K.; Hong, J.T. Roles of peroxiredoxins in cancer, neurodegenerative diseases and inflammatory diseases. Pharmacol. Ther. 2016, 163, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Marat, A.M.; Dokainish, H.; McPherson, P.S. DENN domain proteins: Regulators of Rab GTPases. J. Biol. Chem. 2011, 286, 13791–13800. [Google Scholar] [CrossRef] [PubMed]
- Dallas, S.; Miller, D.S.; Bendayan, R. A novel member of the multidrug resistance-associated protein (MRP) family, contributing to decreased drug accumulation. Pharmacol. Rev. 2006, 58, 140–161. [Google Scholar] [CrossRef] [PubMed]
- Tao, R.-R.; Wang, H.; Hong, L.-J.; Huang, J.-Y.; Lu, Y.-M.; Liao, M.-H.; Ye, W.-F.; Lu, N.-N.; Zhu, D.-Y.; Huang, Q.; et al. Nitrosative stress induces peroxiredoxin 1 ubiquitination during ischemic insult via E6AP activation in endothelial cells both in vitro and in vivo. Antioxid. Redox Signal. 2014, 21, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Van Vliet, A.R.; Verfaillie, T.; Agostinis, P. New functions of mitochondria associated membranes in cellular signaling. Biochim. Biophys. Acta 2014, 1843, 2253–2262. [Google Scholar] [CrossRef] [PubMed]
- Yan, M.H.; Wang, X.; Zhu, X. Mitochondrial defects and oxidative stress in Alzheimer disease and Parkinson disease. Free Radic. Biol. Med. 2013, 62, 90–101. [Google Scholar] [CrossRef] [PubMed]
- Hirayama, K.; Aoki, S.; Nishikawa, K.; Matsumoto, T.; Wada, K. Identification of novel chemical inhibitors for ubiquitin C-terminal hydrolase-L3 by virtual screening. Bioorg. Med. Chem. 2007, 15, 6810–6818. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Bi, C.; Schmitt, S.M.; Li, X.; Fan, Y.; Zhang, N.; Dou, Q.P. Metal-based 2,3-indolinedione derivatives as proteasome inhibitors and inducers of apoptosis in human cancer cells. Int. J. Mol. Med. 2014, 34, 870–879. [Google Scholar] [CrossRef] [PubMed]
- Shaid, S.; Brandts, C.H.; Serve, H.; Dikic, I. Ubiquitination and selective autophagy. Cell Death Differ. 2013, 20, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Ji, C.H.; Kwon, Y.T. Crosstalk and interplay between the ubiquitin-proteasome system and autophagy. Mol. Cells 2017, 40, 441–449. [Google Scholar] [PubMed]
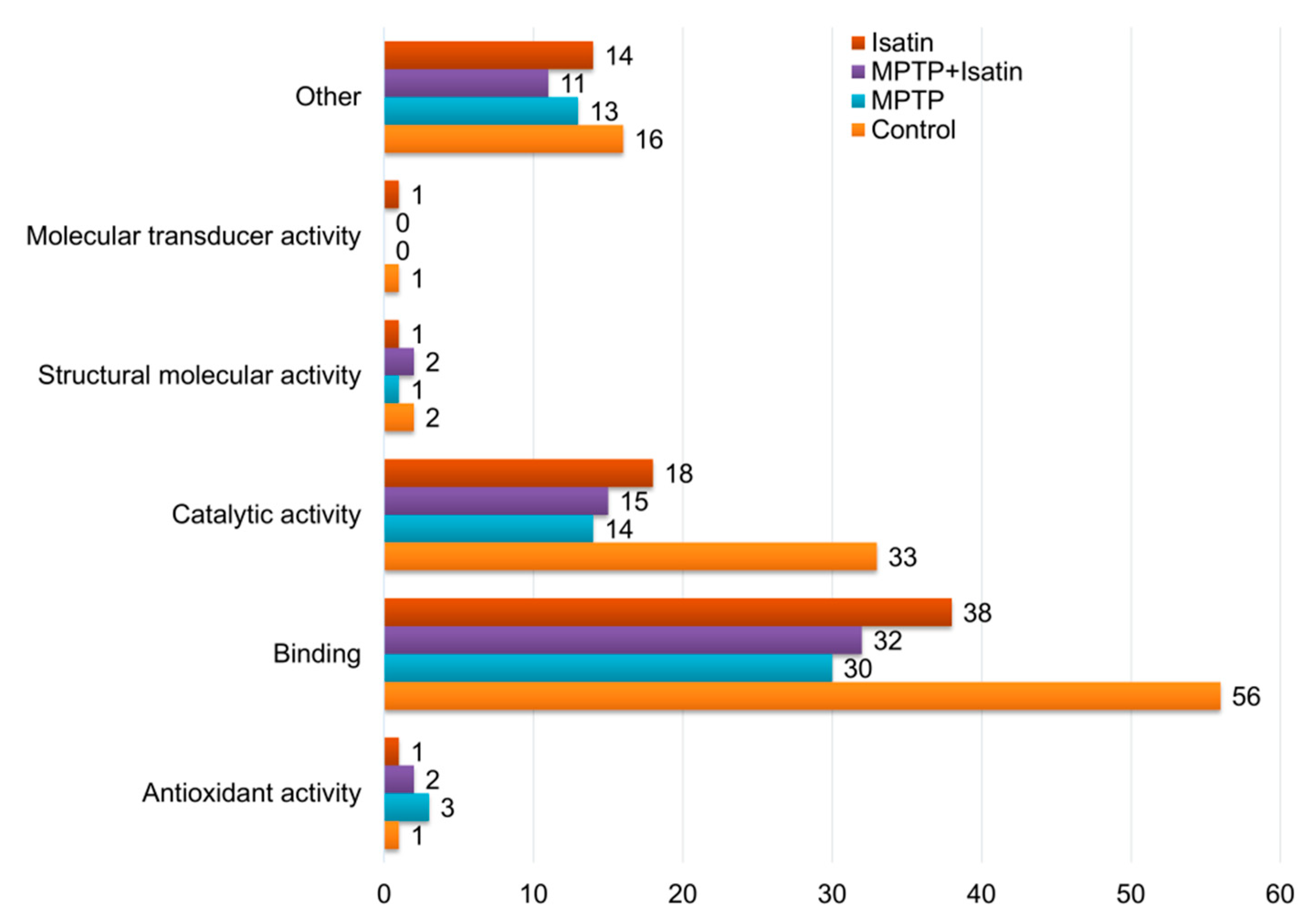
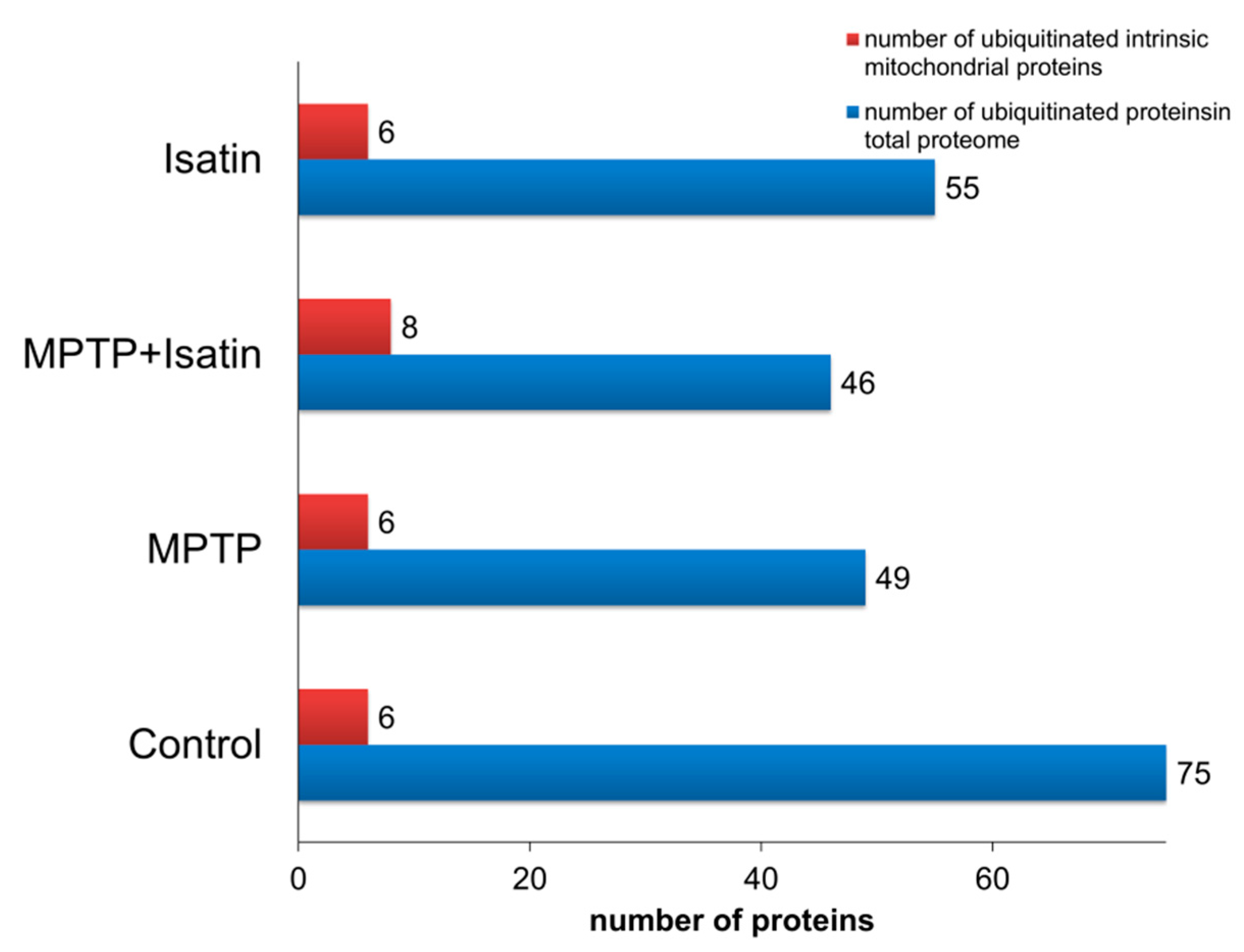
| Accession Number | UniProt ID | Recommended Protein Name | Sample Type | Extramitochondrial Compartment | Intramitochondrial Compartment | Molecular Function | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Control | MPTP | MPTP+isatin | Isatin | Antioxidant Activity | Binding | Catalytic Activity | Other | |||||
| CATD_MOUSE | P18242 | Cathepsin D | ● | ● | ● | |||||||
| APR_MOUSE | Q9JM54 | Phorbol-12-myristate-13-acetate-induced protein 1 | ● | ● | ||||||||
| TAP1_MOUSE | P21958 | Antigen peptide transporter 1 | ● | ● | ● | ● | ● | |||||
| AKT1_MOUSE | P31750 | RAC-alpha serine/threonine-protein kinase | ● | ● | ● | ● | ● | |||||
| KPYM_MOUSE | P52480 | Pyruvate kinase PKM | ● | ● | ● | ● | ||||||
| ACSS3_MOUSE | Q14DH7 | Acyl-CoA synthetase short-chain family member 3, mitochondrial | ● | ● | ● | ● | ||||||
| AL4A1_MOUSE | Q8CHT0 | Delta-1-pyrroline-5-carboxylate dehydrogenase, mitochondrial | ● | ● | ● | ● | ||||||
| NALP5_MOUSE | Q9R1M5 | NACHT, LRR and PYD domains-containing protein 5 | ● | ● | ● | |||||||
| AT2A1_MOUSE | Q8R429 | Sarcoplasmic/endoplasmic reticulum calcium ATPase 1 | ● | ● | ● | ● | ● | |||||
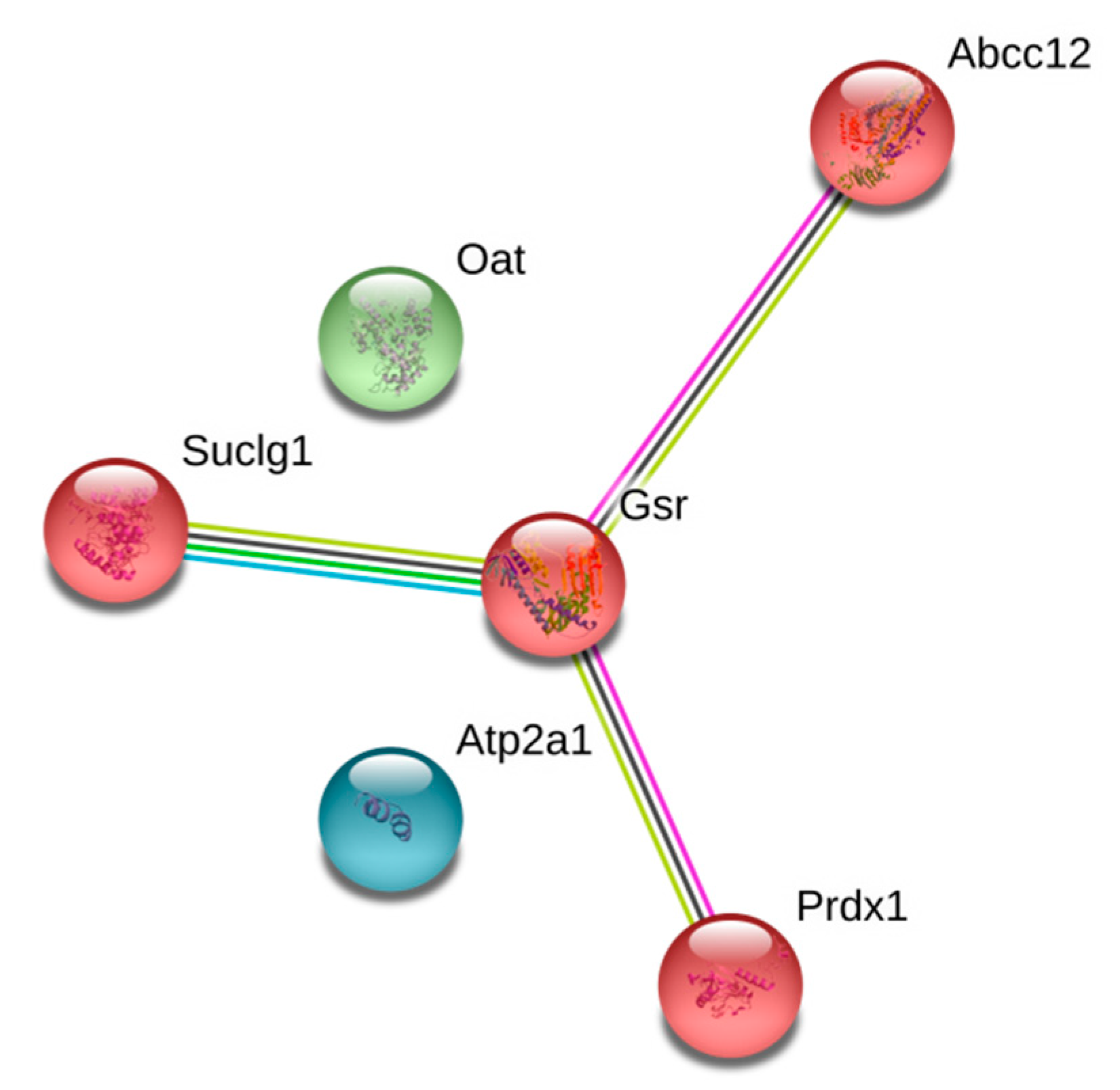
| SUCA_MOUSE | Q9WUM5 | Succinate-CoA ligase (ADP/GDP-forming) subunit alpha, mitochondrial | ● | ● | ● | ● | ● | |||||
| OAT_MOUSE | P29758 | Ornithine aminotransferase, mitochondrial | ● | ● | ● | ● | ||||||
| MRP9_MOUSE | Q80WJ6 | Multidrug resistance-associated protein 9 | ● | ● | ● | ● | ||||||
| DLDH_MOUSE | O08749 | Dihydrolipoyl dehydrogenase, mitochondrial | ● | ● | ● | ● | ● | |||||
| CATB_MOUSE | P10605 | Cathepsin B | ● | ● | ● | ● | ||||||
| CLH1_MOUSE | Q68FD5 | Clathrin heavy chain 1 | ● | ● | ● | |||||||
| ADCYA_MOUSE | Q8C0T9 | Adenylate cyclase type 10 | ● | ● | ● | ● | ||||||
| FPPS_MOUSE | Q920E5 | Farnesyl pyrophosphate synthase | ● | ● | ● | ● | ||||||
| AATM_MOUSE | P05202 | Aspartate aminotransferase, mitochondrial | ● | ● | ● | ● | ||||||
| ERAL1_MOUSE | Q9CZU4 | GTPase Era, mitochondrial | ● | ● | ● | |||||||
| TO20L_MOUSE | Q9D4V6 | TOMM20-like protein 1 | ● | ● | ● | ● | ||||||
| PRDX1_MOUSE | P35700 | Peroxiredoxin-1 | ● | ● | ● | ● | ● | ● | ||||
| GSHR_MOUSE | P47791 | Glutathione reductase, mitochondrial | ● | ● | ● | ● | ● | |||||
| UniProt ID | Protein Name | Gene Name | Sequence | Ubiquitination | Oxidized Residue | Confidence, % |
|---|---|---|---|---|---|---|
| O08749 | Dihydrolipoyl-dehydrogenase, mitochondrial | Dld | IPVNNRFQTKSTDR | K446 | 98.99 | |
| P29758 | Ornithine aminotransferase, mitochondrial | Oat | LFNYNKVLPMNTGVEAGETACK | K135 | M139 | 93.59 |
| Q14DH7 | Acyl-CoA synthetase short-chain family member 3, mitochondrial | Acss3 | TPPPGQAGK | K472 | 89.51 | |
| Q8CHT0 | Delta-1-pyrroline-5-carboxylate dehydrogenase, mitochondrial | Aldh4a1 | NESVGYYVEPCIIESKDPQEPIMK | K437 | 95.86 | |
| Q9CZU4 * | GTPase Era, mitochondrial | Eral1 | LNPQVLQCLTKFSQVPSILVLN | K225 | 93.35 | |
| Q9WUM5 | Succinate-CoA ligase [ADP/GDP-forming] subunit alpha, mitochondrial | Suclg1 | KAKPVVSFIAGITAPPGR | K280 | 100.00 | |
| P05202 | Aspartate aminotransferase, mitochondrial | Got2 | GINVCLCQSYAKNMGLYGER | K279 | 93.83 | |
| P47791 | Glutathione reductase, mitochondrial | Gsr | RDAYVSRLNTIYQNNLTK | K141 | 92.89 | |
| P35700 | Peroxiredoxin-1 | Prdx1 | GSDTIKPDVNK | K185 | 99.48 |
| UNIPROT Accession Number | Protein Name | Involvement in Neurodegeneration | Reference |
|---|---|---|---|
| Q6ZPY5 | Zinc finger protein 507 | Alterations in ZNFs are involved in the development of neurodegeneration | [39] |
| Q91YE6 | Importin-9 | Regeneration of injured neurons | [40] |
| P68368 | Tubulin alpha-4A chain | Alpha-tubulin levels decreased mainly in neurons containing neurofibrillary tau pathology | [41] |
| Q9QZ04 | MAGE-like protein 2 | MAGE proteins form complexes with E3 ubiquitin ligases | [42] |
| Q9R0G7 | Zinc finger E-box-binding homeobox 2 | It is involved in the regulation of microRNA in glioma stem cells | [43] |
| Q8BZ36 | RAD50-interacting protein 1 | It functions as a multitask protein, and is involved in genomic stability, ER homeostasis, and autophagy | [44] |
| Q8CE72 | Protein JBTS17 | Jbts17 mutant mice have cilia transition zone defects and related cerebellar anomalies | [45] |
| Q6P7F1 | MAGUK p55 subfamily member 4 | Plays a role in several CNS disorders | [46] |
| P43300 | Early growth response protein 3 | Gene encoding this protein is induced by alpha-synuclein | [47] |
| Q8C4A5 | Putative Polycomb group protein ASXL3 | Is upregulated in Alzheimer’s disease | [48] |
| Q9ESK9 | RB1-inducible coiled-coil protein 1 | Its insufficiency causes neuronal atrophy and is involved in the pathology of Alzheimer’s diseases | [49] |
| P47791 | Glutathione reductase, mitochondrial | Is implicated in glutathione reduction. GSH is important for pathogenesis of Parkinson’s disease | [50,51] |
| Q9CR16 | Peptidyl-prolyl cis-trans isomerase D | Binds to hyperphosphorylated Tau proteins in degenerating neurons | [52] |
| Q9EQK5 | Major vault protein | Is implicated in senescence-associated apoptosis resistance | [53] |
| Q3UUG6 | TBC1 domain family member 24 | Truncating mutation results in severe neurodegeneration | [54] |
| P23950 | mRNA decay activator protein ZFP36L1 | Involved in mRNA stability in the human brain | [55] |
| P97500 | Myelin transcription factor 1-like protein | Influences memory-related processes | [56] |
| Q9QZQ1 | Afadin | Maintenance of dendritic structure and excitatory tone | [57] |
| P81117 | Nucleobindin-2 | Altered levels found in neuropsychiatric disorders | [58] |
| P58006 | Sestrin-1 | A negative feedback regulator of TOR; its loss results in various TOR-dependent, age-related pathologies | [59] |
| Q9D2H8 | Fibronectin type III domain-containing protein 8 | Its expression is decreased in patients with PD with dementia | [60] |
| Q8C079 | Striatin-interacting protein 1 | Involved in the targeting, attachment, and cytoskeletal transport of autophagosomes, which are accumulated in neurodegenerative neurons | [61] |
| Q8BJQ2 | Ubiquitin carboxyl-terminal hydrolase 1 | Undergoes oxidative modification in both Alzheimer’s disease and Parkinson’s disease | [62] |
| Q91WJ8 | Far upstream element-binding protein 1 | Being a substrate for ubiquitination by Parkin, it plays an important role in development of Parkinson disease | [63] |
| P29758 | OAT, mitochondrial | Ornithine aminotransferase deficiency causes gyrate atrophy | [64] |
| P35700 | Peroxiredoxin-1 | Plays a protective role in counteracting Aβ injury by increasing cell viability preserving neurites, and decreasing cell death | [65] |
| Q8C4S8 | DENN domain-containing protein 2A | DENN proteins regulate autophagy | [66] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Buneeva, O.; Kopylov, A.; Kapitsa, I.; Ivanova, E.; Zgoda, V.; Medvedev, A. The Effect of Neurotoxin MPTP and Neuroprotector Isatin on the Profile of Ubiquitinated Brain Mitochondrial Proteins. Cells 2018, 7, 91. https://doi.org/10.3390/cells7080091
Buneeva O, Kopylov A, Kapitsa I, Ivanova E, Zgoda V, Medvedev A. The Effect of Neurotoxin MPTP and Neuroprotector Isatin on the Profile of Ubiquitinated Brain Mitochondrial Proteins. Cells. 2018; 7(8):91. https://doi.org/10.3390/cells7080091
Chicago/Turabian StyleBuneeva, Olga, Arthur Kopylov, Inga Kapitsa, Elena Ivanova, Victor Zgoda, and Alexei Medvedev. 2018. "The Effect of Neurotoxin MPTP and Neuroprotector Isatin on the Profile of Ubiquitinated Brain Mitochondrial Proteins" Cells 7, no. 8: 91. https://doi.org/10.3390/cells7080091
APA StyleBuneeva, O., Kopylov, A., Kapitsa, I., Ivanova, E., Zgoda, V., & Medvedev, A. (2018). The Effect of Neurotoxin MPTP and Neuroprotector Isatin on the Profile of Ubiquitinated Brain Mitochondrial Proteins. Cells, 7(8), 91. https://doi.org/10.3390/cells7080091





