Protective Effects and Target Network Analysis of Ginsenoside Rg1 in Cerebral Ischemia and Reperfusion Injury: A Comprehensive Overview of Experimental Studies
Abstract
1. Introduction
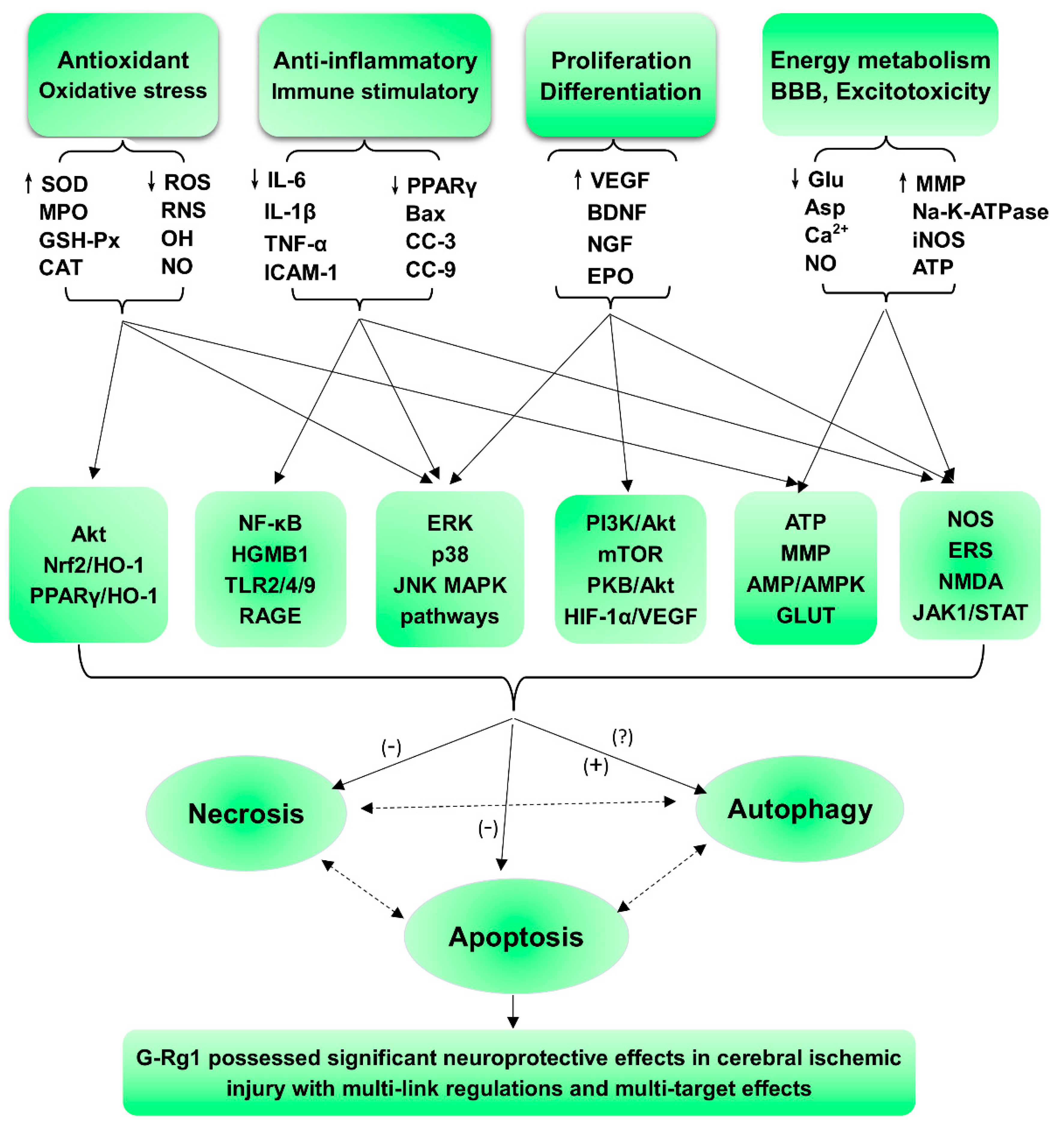
2. Protective Effects and Mechanisms
2.1. Regulation of Oxidative Stress and Apoptosis
2.2. Regulation of Necrosis and Apoptosis Associated with Anti-Inflammatory Activity
2.3. Regulation of the Neural Cycle, Proliferation, Differentiation, and Regeneration
2.4. Regulation of Energy Metabolism and the Blood-Brain Barrier and Other Effects
3. Conclusions and Remarks
Author Contributions
Funding
Conflicts of Interest
References
- Woodruff, T.M.; Thundyil, J.; Tang, S.-C.; Sobey, C.G.; Taylor, S.M.; Arumugam, T.V. Pathophysiology, treatment, and animal and cellular models of human ischemic stroke. Mol. Neurodegener. 2011, 6, 11. [Google Scholar] [CrossRef] [PubMed]
- Eltzschig, H.K.; Eckle, T. Ischemia and reperfusion—From mechanism to translation. Nat. Med. 2011, 17, 1391–1401. [Google Scholar] [CrossRef] [PubMed]
- Turley, K.R.; Toledo-Pereyra, L.H.; Kothari, R.U. Molecular mechanisms in the pathogenesis and treatment of acute ischemic stroke. J. Investig. Surg. 2005, 18, 207–218. [Google Scholar] [CrossRef] [PubMed]
- Raimondo, D.; Tuttolomondo, D.A.; Buttà, C.; Miceli, S.; Licata, G.; Pinto, A. Effects of ACE-inhibitors and angiotensin receptor blockers on inflammation. Curr. Pharm. Des. 2012, 18, 4385–4413. [Google Scholar] [CrossRef] [PubMed]
- Tuttolomondo, A.; Di, S.R.; Di, R.D.; Pedone, C.; La, P.S.; Pinto, A.; Licata, G. Effects of clinical and laboratory variables and of pretreatment with cardiovascular drugs in acute ischaemic stroke: A retrospective chart review from the GIFA study. Int. J. Cardiol. 2011, 151, 318–322. [Google Scholar] [CrossRef]
- Bolaños, J.P.; Moro, M.A.; Lizasoain, I.; Almeida, A. Mitochondria and reactive oxygen and nitrogen species in neurological disorders and stroke: Therapeutic implications. Adv. Drug Deliv. Rev. 2009, 61, 1299–1315. [Google Scholar] [CrossRef]
- Crack, P.J.; Taylor, J.M. Reactive oxygen species and the modulation of stroke. Free Radic. Biol. Med. 2005, 38, 1433–1444. [Google Scholar] [CrossRef]
- Fraser, P.A. The role of free radical generation in increasing cerebrovascular permeability. Free Radic. Boil. Med. 2011, 51, 967–977. [Google Scholar] [CrossRef]
- Eckert, B. Acute Stroke Therapy 1981–2009. Clin. Neuroradiol. 2009, 19, 8. [Google Scholar] [CrossRef]
- Xie, W.; Meng, X.; Zhai, Y.; Zhou, P.; Ye, T.; Wang, Z.; Sun, G.; Sun, X. Panax Notoginseng Saponins: A Review of Its Mechanisms of Antidepressant or Anxiolytic Effects and Network Analysis on Phytochemistry and Pharmacology. Molecules 2018, 23. [Google Scholar] [CrossRef]
- Chen, Y.Q.; Rong, L.; Qiao, J.O. Antiinflammatory effects of Panax notoginseng saponins ameliorate acute lung injury induced by oleic acid and lipopolysaccharide in rats. Mol. Med. Rep. 2014, 10, 1400–1408. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, H.G.; Jia, Y.; Li, X.H. Panax notoginseng saponins attenuate atherogenesis accelerated by zymosan in rabbits. Biol. Pharm. Bull. 2010, 33, 1324–1330. [Google Scholar] [CrossRef]
- Gao, Y.T.; Zhang, W.B.; Yang, L.R.; Wang, X.M.; Yang, Y.L. Effects of Panax notoginseng saponins on antioxidation and preventing DNA damage caused by hydroxyl radical. Zhong Yao Cai 2008, 31, 1399–1402. [Google Scholar]
- Zhu, L.Q.; Fan, J.P.; Huang, Q.F.; Sun, S.L.; Gao, Y.; Zou, Y.H.; Zhang, Z.; He, L.Y.; Zheng, H. Study on the anti-apopotosis induced by hypoxia/hypoglycemia and reoxygenation of panax notoginseng saponins in cultured rat hippocampal neurons. Zhongguo Zhong Yao Za Zhi 2003, 28, 52–55. [Google Scholar]
- Zhou, Y.; Li, H.Q.; Lu, L.; Fu, D.L.; Liu, A.J.; Li, J.H.; Zheng, G.Q. Ginsenoside Rg1 provides neuroprotection against blood brain barrier disruption and neurological injury in a rat model of cerebral ischemia/reperfusion through downregulation of aquaporin 4 expression. Phytomedicine 2014, 21, 998–1003. [Google Scholar] [CrossRef]
- Guo, C.; Gao, Y.G.; Zang, P.; He, Z.M.; Zhao, Y.; Zhu, H.Y.; Yang, H.; Dong, W.; Zhang, L.X. Simultaneous determination of sixteen ginsenosides in Panax ginseng and its preparation by HPLC. J. Ginseng Res. 2014, 45, 2009–2013. [Google Scholar]
- Park, H.-W.; In, G.; Han, S.-T.; Lee, M.-W.; Kim, S.-Y.; Kim, K.; Cho, B.-G.; Han, G.-H.; Chang, I.-M. Simultaneous determination of 30 ginsenosides in Panax ginseng preparations using ultra performance liquid chromatography. J. Ginseng Res. 2013, 37, 457–467. [Google Scholar] [CrossRef]
- Joo, K.-M.; Park, C.-W.; Jeong, H.-J.; Lee, S.J.; Chang, I.S. Simultaneous determination of two Amadori compounds in Korean red ginseng (Panax ginseng) extracts and rat plasma by high-performance anion-exchange chromatography with pulsed amperometric detection. J. Chromatogr. B 2008, 865, 159–166. [Google Scholar] [CrossRef]
- Wu, S.; Cui, X.M.; Guo, C.L.; Wang, C.X.; Yang, Y.; Qu, Y.; Yang, X.Y. Effects of different steaming methods on saponins in taproots of Panax notoginseng. Chinese Traditional & Herbal Drugs. 2015, 46, 3352–3356. [Google Scholar]
- Anggelia, M.R.; Chan, W.H. Impairment of preimplantation and postimplantation embryonic development through intrinsic apoptotic processes by ginsenoside Rg1 in vitro and in vivo. Environ. Toxicol. 2017, 32, 1937–1951. [Google Scholar] [CrossRef]
- Fan, C.; Zhu, X.; Song, Q.; Wang, P.; Liu, Z.; Yu, S.Y. MiR-134 modulates chronic stress-induced structural plasticity and depression-like behaviors via downregulation of Limk1/cofilin signaling in rats. Neuropharmacology 2018, 131, 364–376. [Google Scholar] [CrossRef]
- Ghaeminia, M.; Rajkumar, R.; Koh, H.L.; Dawe, G.S.; Tan, C.H. Ginsenoside Rg1 modulates medial prefrontal cortical firing and suppresses the hippocampo-medial prefrontal cortical long-term potentiation. J. Ginseng Res. 2018, 42, 298–303. [Google Scholar] [CrossRef]
- Guo, Y.H.; Zhao, S.; Du, Y.X.; Xing, Q.J.; Chen, B.L.; Yu, C.Q. Effects of ginsenoside Rg1-loaded alginate-chitosan microspheres on human bone marrow stromal cells. Biosci. Rep. 2017, 37. [Google Scholar] [CrossRef]
- Li, Y.; Guan, Y.; Wang, Y.; Yu, C.L.; Zhai, F.G.; Guan, L.X. Neuroprotective Effect of the Ginsenoside Rg1 on Cerebral Ischemic Injury In Vivo and In Vitro Is Mediated by PPARgamma-Regulated Antioxidative and Anti-Inflammatory Pathways. Evid. Based Complement. Altern. Med. 2017, 2017, 7842082. [Google Scholar]
- Song, L.; Xu, M.B.; Zhou, X.L.; Zhang, D.P.; Zhang, S.L.; Zheng, G.Q. A Preclinical Systematic Review of Ginsenoside-Rg1 in Experimental Parkinson’s Disease. Oxid. Med. Cell. Longev. 2017, 2017, 2163053. [Google Scholar] [CrossRef]
- Wang, L.; Zhao, H.; Zhai, Z.Z.; Qu, L.X. Protective effect and mechanism of ginsenoside Rg1 in cerebral ischaemia-reperfusion injury in mice. Biomed. Pharm. 2018, 99, 876–882. [Google Scholar] [CrossRef]
- Xia, C.Y.; Chu, S.F.; Zhang, S.; Gao, Y.; Ren, Q.; Lou, Y.X.; Luo, P.; Tian, M.T.; Wang, Z.Q.; Du, G.H.; et al. Ginsenoside Rg1 alleviates corticosterone-induced dysfunction of gap junctions in astrocytes. J. Ethnopharmacol. 2017, 208, 207–213. [Google Scholar] [CrossRef]
- Yu, H.; Fan, C.; Yang, L.; Yu, S.; Song, Q.; Wang, P.; Mao, X. Ginsenoside Rg1 Prevents Chronic Stress-Induced Depression-Like Behaviors and Neuronal Structural Plasticity in Rats. Cell Physiol. Biochem. 2018, 48, 2470–2482. [Google Scholar] [CrossRef]
- Zhang, Y.; Hu, W.; Zhang, B.; Yin, Y.; Zhang, J.; Huang, D.; Huang, R.; Li, W.; Li, W. Ginsenoside Rg1 protects against neuronal degeneration induced by chronic dexamethasone treatment by inhibiting NLRP-1 inflammasomes in mice. Int. J. Mol. Med. 2017, 40, 1134–1142. [Google Scholar] [CrossRef]
- Cheng, Y.; Shen, L.H.; Zhang, J.T. Anti-amnestic and anti-aging effects of ginsenoside Rg1 and Rb1 and its mechanism of action. Acta Pharm. Sin. 2005, 26, 143–149. [Google Scholar] [CrossRef]
- Li, T.; Shu, Y.J.; Cheng, J.Y.; Liang, R.C.; Dian, S.N.; Lv, X.X.; Yang, M.Q.; Huang, S.L.; Chen, G.; Yang, F. Pharmacokinetics and efficiency of brain targeting of ginsenosides Rg1 and Rb1 given as Nao-Qing microemulsion. Drug Dev. Ind. Pharm. 2015, 41, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.; Sun, W.; Gong, W.; Ding, Y.; Zhuang, Y.; Hou, Q. Ginsenoside Rg1 protects against transient focal cerebral ischemic injury and suppresses its systemic metabolic changes in cerabral injury rats. Acta Pharm. Sin. B 2015, 5, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, Y.B.; Stary, C.M.; White, R.E.; Giffard, R.G. The use of microRNAs to modulate redox and immune response to stroke. Antioxid. Redox Signal. 2015, 22, 187–202. [Google Scholar] [CrossRef] [PubMed]
- Rodrigo, R.; Fernandez-Gajardo, R.; Gutierrez, R.; Matamala, J.M.; Carrasco, R.; Miranda-Merchak, A.; Feuerhake, W. Oxidative stress and pathophysiology of ischemic stroke: Novel therapeutic opportunities. CNS Neurol. Disord. Drug Targets 2013, 12, 698–714. [Google Scholar] [CrossRef] [PubMed]
- Zuluaga, M.; Gueguen, V.; Letourneur, D.; Pavon-Djavid, G. Astaxanthin-antioxidant impact on excessive Reactive Oxygen Species generation induced by ischemia and reperfusion injury. Chem. Biol. Interact. 2018, 279, 145–158. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Suo, L.; Liu, Y.; Li, H.; Xue, W. Protective effects of ginsenoside Rg1 against oxygen-glucose-deprivation-induced apoptosis in neural stem cells. J. Neurol. Sci. 2017, 373, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Minutoli, L.; Puzzolo, D.; Rinaldi, M.; Irrera, N.; Marini, H.; Arcoraci, V.; Bitto, A.; Crea, G.; Pisani, A.; Squadrito, F.; et al. ROS-Mediated NLRP3 Inflammasome Activation in Brain, Heart, Kidney, and Testis Ischemia/Reperfusion Injury. Oxid. Med. Cell. Longev. 2016, 2016, 2183026. [Google Scholar] [CrossRef] [PubMed]
- Xie, C.L.; Li, J.H.; Wang, W.W.; Zheng, G.Q.; Wang, L.X. Neuroprotective effect of ginsenoside-Rg1 on cerebral ischemia/reperfusion injury in rats by downregulating protease-activated receptor-1 expression. Life Sci. 2015, 121, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Xie, C.L.; Wang, W.W.; Xue, X.D.; Zhang, S.F.; Gan, J.; Liu, Z.G. A systematic review and meta-analysis of Ginsenoside-Rg1 (G-Rg1) in experimental ischemic stroke. Sci. Rep. 2015, 5, 7790. [Google Scholar] [CrossRef] [PubMed]
- Cadenas, S. ROS and redox signaling in myocardial ischemia-reperfusion injury and cardioprotection. Free Radic. Biol. Med. 2018, 117, 76–89. [Google Scholar] [CrossRef]
- Forrester, S.J; Kikuchi, D.S.; Hernandes, M.S.; Xu, Q.; Griendling, K.K. Reactive Oxygen Species in Metabolic and Inflammatory Signaling. Circ. Res. 2018, 122, 877–902. [Google Scholar] [CrossRef]
- Bystrom, P.; Foley, N.; Toledo-Pereyra, L.; Quesnelle, K. Ischemic preconditioning modulates ROS to confer protection in liver ischemia and reperfusion. EXCLI J. 2017, 16, 483–496. [Google Scholar]
- Granger, D.N.; Kvietys, P.R. Reperfusion therapy-What’s with the obstructed, leaky and broken capillaries? Pathophysiology 2017, 24, 213–228. [Google Scholar] [CrossRef]
- Lavie, L. Oxidative stress in obstructive sleep apnea and intermittent hypoxia--revisited--the bad ugly and good: Implications to the heart and brain. Sleep Med. Rev. 2015, 20, 27–45. [Google Scholar] [CrossRef]
- Quesnelle, K.M.; Bystrom, P.V.; Toledo-Pereyra, L.H. Molecular responses to ischemia and reperfusion in the liver. Arch. Toxicol. 2015, 89, 651–657. [Google Scholar] [CrossRef]
- Zhou, D.; Fang, T.; Lu, L.Q.; Yi, L. Neuroprotective potential of cerium oxide nanoparticles for focal cerebral ischemic stroke. J. Huazhong Univ. Sci. Technol. Med. Sci. 2016, 36, 480–486. [Google Scholar] [CrossRef]
- Bayir, H.; Fadeel, B.; Palladino, M.J.; Witasp, E.; Kurnikov, I.V.; Tyurina, Y.Y.; Tyurin, V.A.; Amoscato, A.A.; Jiang, J.; Kochanek, P.M.; et al. Apoptotic interactions of cytochrome c: Redox flirting with anionic phospholipids within and outside of mitochondria. Biochim. Biophys. Acta 2006, 1757, 648–659. [Google Scholar] [CrossRef]
- Guaragnella, N.; Antonacci, L.; Passarella, S.; Marra, E.; Giannattasio, S. Achievements and perspectives in yeast acetic acid-induced programmed cell death pathways. Biochem. Soc. Trans. 2011, 39, 1538–1543. [Google Scholar] [CrossRef]
- Islam, M.T. Oxidative stress and mitochondrial dysfunction-linked neurodegenerative disorders. Neurol. Res. 2017, 39, 73–82. [Google Scholar] [CrossRef]
- Negrette-Guzman, M.; Huerta-Yepez, S.; Tapia, E.; Pedraza-Chaverri, J. Modulation of mitochondrial functions by the indirect antioxidant sulforaphane: A seemingly contradictory dual role and an integrative hypothesis. Free Radic. Biol. Med. 2013, 65, 1078–1089. [Google Scholar] [CrossRef]
- Rhim, T.; Lee, D.Y.; Lee, M. Hypoxia as a target for tissue specific gene therapy. J. Control. Release 2013, 172, 484–494. [Google Scholar] [CrossRef]
- Sun, K.; Fan, J.; Han, J. Ameliorating effects of traditional Chinese medicine preparation, Chinese materia medica and active compounds on ischemia/reperfusion-induced cerebral microcirculatory disturbances and neuron damage. Acta Pharm. Sin. B 2015, 5, 8–24. [Google Scholar] [CrossRef]
- Zhang, J.; Han, X.; Li, X.; Luo, Y.; Zhao, H.; Yang, M.; Ni, B.; Liao, Z. Core-shell hybrid liposomal vesicles loaded with panax notoginsenoside: Preparation, characterization and protective effects on global cerebral ischemia/reperfusion injury and acute myocardial ischemia in rats. Int. J. Nanomed. 2012, 7, 4299–4310. [Google Scholar] [CrossRef]
- Wang, Q.Y.; Liu, F.; Wu, F.J.; Li, J.L. Effects of ginsenoside Rg1 on the expressions of p-eRK1/2 and p-JNK in local cerebral ischemia/reperfusion injury rats. Zhongguo Zhong Xi Yi Jie He Za Zhi 2013, 33, 229–234. [Google Scholar]
- Jin, Z.; Liang, J.; Wang, J.; Kolattukudy, P.E. MCP-induced protein 1 mediates the minocycline-induced neuroprotection against cerebral ischemia/reperfusion injury in vitro and in vivo. J. Neuroinflammation 2015, 12, 39. [Google Scholar] [CrossRef]
- Yang, Y.; Li, X.; Zhang, L.; Liu, L.; Jing, G.; Cai, H. Ginsenoside Rg1 suppressed inflammation and neuron apoptosis by activating PPARgamma/HO-1 in hippocampus in rat model of cerebral ischemia-reperfusion injury. Int. J. Clin. Exp. Pathol. 2015, 8, 2484–2494. [Google Scholar]
- Deng, Y.; Yang, M.; Xu, F.; Zhang, Q.; Zhao, Q.; Yu, H.; Li, D.; Zhang, G.; Lu, A.; Cho, K.; et al. Combined Salvianolic Acid B and Ginsenoside Rg1 Exerts Cardioprotection against Ischemia/Reperfusion Injury in Rats. PLoS ONE 2015, 10, e0135435. [Google Scholar] [CrossRef]
- Dong, G.; Chen, T.; Ren, X.; Zhang, Z.; Huang, W.; Liu, L.; Luo, P.; Zhou, H. Rg1 prevents myocardial hypoxia/reoxygenation injury by regulating mitochondrial dynamics imbalance via modulation of glutamate dehydrogenase and mitofusin 2. Mitochondrion 2016, 26, 7–18. [Google Scholar] [CrossRef]
- Liu, Q.; Kou, J.P.; Yu, B.Y. Ginsenoside Rg1 protects against hydrogen peroxide-induced cell death in PC12 cells via inhibiting NF-kappaB activation. Neurochem. Int. 2011, 58, 119–125. [Google Scholar] [CrossRef]
- Sun, Z.G.; Chen, L.P.; Wang, F.W.; Xu, C.Y.; Geng, M. Protective effects of ginsenoside Rg1 against hydrogen peroxide-induced injury in human neuroblastoma cells. Neural Regen. Res. 2016, 11, 1159–1164. [Google Scholar]
- Li, F.; Lv, Y.N.; Tan, Y.S.; Shen, K.; Zhai, K.F.; Chen, H.L.; Kou, J.P.; Yu, B.Y. An integrated pathway interaction network for the combination of four effective compounds from ShengMai preparations in the treatment of cardio-cerebral ischemic diseases. Acta Pharm. Sin. 2015, 36, 1337–1348. [Google Scholar] [CrossRef]
- Huang, X.P.; Qiu, Y.Y.; Wang, B.; Ding, H.; Tang, Y.H.; Zeng, R.; Deng, C.Q. Effects of Astragaloside IV combined with the active components of Panax notoginseng on oxidative stress injury and nuclear factor-erythroid 2-related factor 2/heme oxygenase-1 signaling pathway after cerebral ischemia-reperfusion in mice. Pharm. Mag. 2014, 10, 402–409. [Google Scholar]
- Wang, M.; Xu, X.; Xu, H.; Wen, F.; Zhang, X.; Sun, H.; Yao, F.; Sun, G.; Sun, X. Effect of the total saponins of Aralia elata (Miq) Seem on cardiac contractile function and intracellular calcium cycling regulation. J. Ethnopharmacol. 2014, 155, 240–247. [Google Scholar] [CrossRef]
- Yang, X.; Xiong, X.; Wang, H.; Wang, J. Protective Effects of Panax Notoginseng Saponins on Cardiovascular Diseases: A Comprehensive Overview of Experimental Studies. Evid. Based Complement. Altern. Med. 2014, 2014, 204840. [Google Scholar] [CrossRef]
- Shen, K.; Wang, Y.; Zhang, Y.; Zhou, H.; Song, Y.; Cao, Z.; Kou, J.; Yu, B. Cocktail of Four Active Components Derived from Sheng Mai San Inhibits Hydrogen Peroxide-Induced PC12 Cell Apoptosis Linked with the Caspase-3/ROCK1/MLC Pathway. Rejuv. Res. 2015, 18, 517–527. [Google Scholar] [CrossRef]
- Wang, J.; Hou, J.; Zhao, H.; Liu, J. Synergistic Use of Geniposide and Ginsenoside Rg1 Balance Microglial TNF-alpha and TGF-beta1 following Oxygen-Glucose Deprivation In Vitro: A Genome-Wide Survey. Evid. Based Complement. Altern. Med. 2015, 2015, 756346. [Google Scholar] [CrossRef]
- Sun, C.; Lai, X.; Huang, X.; Zeng, Y. Protective effects of ginsenoside Rg1 on astrocytes and cerebral ischemic-reperfusion mice. Biol. Pharm. Bull. 2014, 37, 1891–1898. [Google Scholar] [CrossRef]
- Boehncke, W.H. Systemic Inflammation and Cardiovascular Comorbidity in Psoriasis Patients: Causes and Consequences. Front. Immunol. 2018, 9, 579. [Google Scholar] [CrossRef]
- O’Connell, G.C.; Chantler, P.D.; Barr, T.L. Stroke-associated pattern of gene expression previously identified by machine-learning is diagnostically robust in an independent patient population. Genom. Data 2017, 14, 47–52. [Google Scholar] [CrossRef]
- Alawieh, A.; Andersen, M.; Adkins, D.L.; Tomlinson, S. Acute Complement Inhibition Potentiates Neurorehabilitation and Enhances tPA-Mediated Neuroprotection. J. Neurosci. 2018, 38, 6527–6545. [Google Scholar] [CrossRef]
- Xu, H.; Qin, W.; Hu, X.; Mu, S.; Zhu, J.; Lu, W.; Luo, Y. Lentivirus-mediated overexpression of OTULIN ameliorates microglia activation and neuroinflammation by depressing the activation of the NF-kappaB signaling pathway in cerebral ischemia/reperfusion rats. J. Neuroinflammation 2018, 15, 83. [Google Scholar] [CrossRef]
- Iadecola, C.; Anrather, J. The immunology of stroke: From mechanisms to translation. Nat. Med. 2011, 17, 796–808. [Google Scholar] [CrossRef]
- Si, Y.C.; Li, Q.; Xie, C.E.; Niu, X.; Xia, X.H.; Yu, C.Y. Chinese herbs and their active ingredients for activating xue (blood) promote the proliferation and differentiation of neural stem cells and mesenchymal stem cells. Chin. Med. 2014, 9, 13. [Google Scholar] [CrossRef]
- He, P.; Li, P.; Hua, Q.; Liu, Y.; Staufenbiel, M.; Li, R.; Shen, Y. Chronic administration of anti-stroke herbal medicine TongLuoJiuNao reduces amyloidogenic processing of amyloid precursor protein in a mouse model of Alzheimer’s disease. PLoS ONE 2013, 8, e58181. [Google Scholar] [CrossRef]
- Wu, M.Y.; Yiang, G.T.; Liao, W.T.; Tsai, A.P.; Cheng, Y.L.; Cheng, P.W.; Li, C.Y.; Li, C.J. Current Mechanistic Concepts in Ischemia and Reperfusion Injury. Cell. Physiol. Biochem. 2018, 46, 1650–1667. [Google Scholar] [CrossRef]
- Tao, T.; Chen, F.; Bo, L.; Xie, Q.; Yi, W.; Zou, Y.; Hu, B.; Li, J.; Deng, X. Ginsenoside Rg1 protects mouse liver against ischemia-reperfusion injury through anti-inflammatory and anti-apoptosis properties. J. Surg. Res. 2014, 191, 231–238. [Google Scholar] [CrossRef]
- Li, P.; Lv, B.; Jiang, X.; Wang, T.; Ma, X.; Chang, N.; Wang, X.; Gao, X. Identification of NF-kappaB inhibitors following Shenfu injection and bioactivity-integrated UPLC/Q-TOF-MS and screening for related anti-inflammatory targets in vitro and in silico. J. Ethnopharmacol. 2016, 194, 658–667. [Google Scholar] [CrossRef]
- Van Golen, R.F.; Reiniers, M.J.; Olthof, P.B.; van Gulik, T.M.; Heger, M. Sterile inflammation in hepatic ischemia/reperfusion injury: Present concepts and potential therapeutics. J. Gastroenterol. Hepatol. 2013, 28, 394–400. [Google Scholar] [CrossRef]
- Wang, J.; Li, D.; Hou, J.; Lei, H. Protective effects of geniposide and ginsenoside Rg1 combination treatment on rats following cerebral ischemia are mediated via microglial microRNA1555p inhibition. Mol. Med. Rep. 2018, 17, 3186–3193. [Google Scholar]
- Huang, X.P.; Ding, H.; Lu, J.D.; Tang, Y.H.; Deng, B.X.; Deng, C.Q. Effects of the Combination of the Main Active Components of Astragalus and Panax notoginseng on Inflammation and Apoptosis of Nerve Cell after Cerebral Ischemia-Reperfusion. Am. J. Chin. Med. 2015, 43, 1419–1438. [Google Scholar] [CrossRef]
- Wang, J.; Hou, J.C.; Xiang, L.H.; Zhang, J.; Ju, D.H. Compatibility of geniposide and ginsenoside rgl: Their regulating roles in secretion of anoxia induction injured microglia inflammatory cytokines. Hong Zhong Xi Yi Jie He Za Zhi 2014, 34, 91–95. [Google Scholar]
- Ceccatelli, S.; Dare, E.; Moors, M. Methylmercury-induced neurotoxicity and apoptosis. Chem. Biol. Interact. 2010, 188, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, A.A.; Lopez-Granero, C.; Farina, M.; Rocha, J.B.T.; Bowman, A.B.; Aschner, M. Oxidative stress, caspase-3 activation and cleavage of ROCK-1 play an essential role in MeHg-induced cell death in primary astroglial cells. Food Chem. Toxicol. 2018, 113, 328–336. [Google Scholar] [CrossRef] [PubMed]
- Fujimura, M.; Usuki, F. Site-specific neural hyperactivity via the activation of MAPK and PKA/CREB pathways triggers neuronal degeneration in methylmercury-intoxicated mice. Toxicol. Lett. 2017, 271, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Guida, N.; Laudati, G.; Anzilotti, S.; Sirabella, R.; Cuomo, O.; Brancaccio, P.; Santopaolo, M.; Galgani, M.; Montuori, P.; di Renzo, G.; et al. Methylmercury upregulates RE-1 silencing transcription factor (REST) in SH-SY5Y cells and mouse cerebellum. Neurotoxicology 2016, 52, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Yang, X.; Chen, X.; Li, Z.; Fang, M.; Bai, B.; Tan, D. Hydrogen sulfide may attenuate methylmercury-induced neurotoxicity via mitochondrial preservation. Chem. Biol. Interact. 2017, 263, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Takanezawa, Y.; Nakamura, R.; Sone, Y.; Uraguchi, S.; Kiyono, M. Atg5-dependent autophagy plays a protective role against methylmercury-induced cytotoxicity. Toxicol. Lett. 2016, 262, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Qin, L.; Fan, S.; Jia, R.; Liu, Y. Ginsenoside Rg1 protects cardiomyocytes from hypoxia-induced injury through the PI3K/AKT/mTOR pathway. Pharmazie 2018, 73, 349–355. [Google Scholar] [PubMed]
- Maslov, L.N.; Mrochek, A.G.; Shchepetkin, I.A.; Headrick, J.P.; Hanus, L.; Barzakh, E.I.; Lishmanov, A.; Gorbunov, A.S.; Tsybul’nikov, S.; et al. Protein kinases role in adaptive phenomenon of heart ischemic postconditioning development. Ross Fiziol Zh Im I M Sechenova 2013, 99, 433–452. [Google Scholar]
- Tang, B.; Wang, D.; Li, M.; Wu, Q.; Yang, Q.; Shi, W.; Chen, C. An in vivo study of hypoxia-inducible factor-1alpha signaling in ginsenoside Rg1-mediated brain repair after hypoxia/ischemia brain injury. Pediatr. Res. 2017, 81, 120–126. [Google Scholar] [CrossRef]
- Mei, Y.; Lei, C.; Min, L.; Yalin, H.; Hui, G.; Shiduo, L.; Jian, F.; Fang, H. Alteration of NRSF expression exacerbating 1-methyl-4-phenyl-pyridinium ion-induced cell death of SH-SY5Y cells. Neurosci. Res. 2009, 65, 236–244. [Google Scholar]
- Tang, B.; Qu, Y.; Wang, D.; Mu, D. Targeting hypoxia inducible factor-1alpha: A novel mechanism of ginsenoside Rg1 for brain repair after hypoxia/ischemia brain damage. CNS Neurol. Disord. Drug Targets 2011, 10, 235–238. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.J.; Li, Q.Y.; Xu, S.J.; Zeng, N. Effect of ginsenoside Rg1 on angiogenesis after neonatal hypoxia ischemia brain damage in rats. Sichuan Da Xue Xue Bao Yi Xue Ban 2011, 42, 503–507. [Google Scholar] [PubMed]
- Bao, C.; Wang, Y.; Min, H.; Zhang, M.; Du, X.; Han, R.; Liu, X. Combination of ginsenoside Rg1 and bone marrow mesenchymal stem cell transplantation in the treatment of cerebral ischemia reperfusion injury in rats. Cell. Physiol. Biochem. 2015, 37, 901–910. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Zhang, J. Ginsenoside Rg1 increases ischemia-induced cell proliferation and survival in the dentate gyrus of adult gerbils. Neurosci. Lett. 2003, 344, 1–4. [Google Scholar] [CrossRef]
- Huang, X.P.; Ding, H.; Wang, B.; Qiu, Y.Y.; Tang, Y.H.; Zeng, R.; Deng, C.Q. Effects of the main active components combinations of Astragalus and Panax notoginseng on energy metabolism in brain tissues after cerebral ischemia-reperfusion in mice. Pharm. Mag. 2015, 11, 732–739. [Google Scholar]
- Wang, J.; Hou, J.; Lei, H.; Fu, J.; Pan, Y.; Liu, J. Synergistic neuroprotective effect of microglialconditioned media treated with geniposide and ginsenoside Rg1 on hypoxia injured neurons. Mol. Med. Rep. 2015, 12, 5328–5334. [Google Scholar] [CrossRef] [PubMed]
- He, Q.; Sun, J.; Wang, Q.; Wang, W.; He, B. Neuroprotective effects of ginsenoside Rg1 against oxygen-glucose deprivation in cultured hippocampal neurons. J. Chin. Med. Assoc. 2014, 77, 142–149. [Google Scholar] [CrossRef]
- Shen, L.; Zhang, J. NMDA receptor and iNOS are involved in the effects of ginsenoside Rg1 on hippocampal neurogenesis in ischemic gerbils. Neurol. Res. 2007, 29, 270–273. [Google Scholar] [CrossRef]
- Korver, S.; Vergouwe, M.; Hollak, C.E.M.; van Schaik, I.N.; Langeveld, M. Development and clinical consequences of white matter lesions in Fabry disease: A systematic review. Mol. Genet. MeTable 2018, 125, 205–216. [Google Scholar] [CrossRef]
- Marchesoni, C.; Cisneros, E.; Pfister, P.; Yanez, P.; Rollan, C.; Romero, C.; Kisinovsky, I.; Rattagan, L.; Cejas, L.L.; Pardal, A.; et al. Brain MRI findings in children and adolescents with Fabry disease. J. Neurol. Sci. 2018, 395, 131–134. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.F.; Song, X.Y.; Chen, N.H. Advances in pharmacological studies of Panax notoginseng saponins on brain ischemia-reperfusion injury. Yao Xue Xue Bao 2016, 51, 1039–1046. [Google Scholar] [PubMed]
- Meng, X.; Xie, W.; Xu, Q.; Liang, T.; Xu, X.; Sun, G.; Sun, X. Neuroprotective Effects of Radix Scrophulariae on Cerebral Ischemia and Reperfusion Injury via MAPK Pathways. Molecules 2018, 23, 2401. [Google Scholar] [CrossRef] [PubMed]
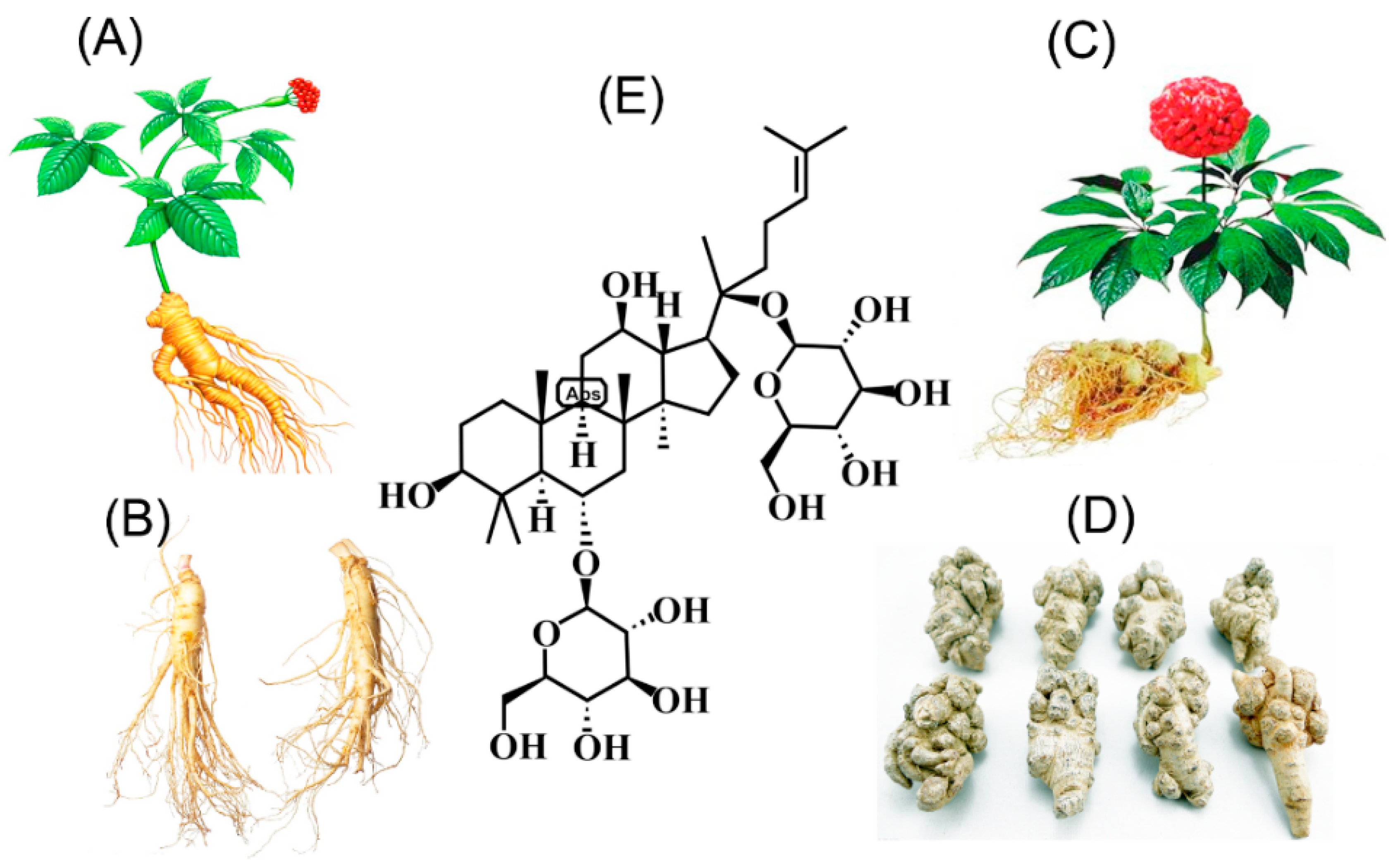
| N | Model | Tissue Sites | Effects | Mechanisms | RF |
|---|---|---|---|---|---|
| 1 | Middle cerebral artery I/R injury model rats; OGD-induced cortical neuronal model | Cerebral I/R injury | ↓ Indicators of oxidative stress and inflammation ↑ Neurological function; MPO and SOD activity, and CAT content ↓ Brain edema, TNF-α and IL-6 content | ↑ Expression of PPARγ and NF-κB 65; Via PPARγ-regulated anti-oxidative and anti-inflammatory pathways | [24] |
| 2 | OGD-induced NSCs | Cerebral I/R injury | ↓ OGD-induced apoptosis in NSCs ↓ OGD-induced oxidative stress ↓ Expression of CC-3 and Bax | ↑ Expression of anti-apoptotic protein Bcl-2; ↓ OGD-induced oxidative stress; ↓ p38/JNK2 phosphorylation; | [36] |
| 3 | H2O2-induced SH-SY5Y cells | Ischemic cerebrovascular disease | ↑ Survival rate of SH-SY5Y cells ↓ Amount of leaked LDH ↑ SOD activity | ↓ Caspase-3 immunoreactivity; ↑ HSP 70 expression; Via anti-oxidation and inhibition of apoptosis; | [60] |
| 4 | OGD-induced BV2 microglial cells | Ischemic stroke | ↓ NO level, TNF-α content and expression ↑ Cell viability, the content and expression of TGF-β1 | Fcγ-receptor-mediated phagocytosis pathway; | [66] |
| 5 | H2O2-treated PC12 cells; TNF-α-treated EA.hy926 cells | Cardio-cerebral ischemic diseases | ↓ H2O2-induced cytotoxicity ↑ Nerve cell survival rate | ↓ Phosphorylation of NF-κB, p50, p65 and IKKα/β; Regulates the Nrf2/HO-1 pathway; | [61] |
| 6 | MCAO-induced SD rat models | Cerebral I/R injury | Effect similar to that produced by ROZ in activating PPARγ/HO-1 | By activating PPARγ/HO-1 to protect against apoptosis and inflammation | [56] |
| 7 | BALB/c mouse t-MCAO model, H2O2-induced mouse cultured astrocytes | Cerebral I/R | ↓ H2O2-induced apoptosis ↓ Ca 2+ overload, loss of MMP ↓ ROS production in astrocytes | Prevents astrocytes from undergoing apoptosis | [67] |
| 8 | MCAO/R-induced C57BL/6 mice | Cerebral I/R | ↑ Nerve cell survival rate ↓ MDA and NO contents ↑ SOD activity and glutathione levels ↓ Nrf2 in the cytoplasm and ↑ in nucleus | ↑ Nuclear translocation rate and HO-1 mRNA levels; By jointly activating the Nrf2/HO-1 signaling pathway | [62] |
| 9 | H2O2-induced PC12 cells | Oxidative stress-induced neuronal injury | ↓ H2O2-induced cytotoxicity ↓ Phosphorylation and nuclear translocation of NF-κB/p65 ↓ Phosphorylation and degradation of IκB | ↓ Activation of Akt and ERK1/2; Via anti-oxidative stress and the regulatory effects of H2O2 on the NF-κB pathway; | [59] |
| N | Model | Tissue Sites | Effects | Mechanisms | RF |
|---|---|---|---|---|---|
| 1 | Cerebral I/R-induced model, C57BL/6 mice | Cerebral I/R | ↓ Infarct volume, neurological deficit scores ↓ IL-1β, TNF-α and IL-6 contents in serum ↓ Glu and Asp contents | ↑ BDNF expression ↓ Expression of IL-1β, IL-6 and TNF-α in serum ↓ Glu and Asp contents | [1] |
| 2 | OGD-injured BV2 microglial cells, MCAO-induced male rat model | Focal cerebral ischemic stroke | ↓ Neurobehavioral deficits, infarct volume, and brain edema ↓ CD11b-positive cell numbers and miR-155-5p levels | ↓ Expression levels of miR-155-5p, pri-miR-155 and pre-miR-155 Inhibition of microglial miR-155-5p following ischemic injury | [2] |
| 3 | MCA I/R model in rats, OGD-induced cortical neurons | Cerebral I/R injury. | ↓ Indicators of oxidative stress and inflammation ↑ Neurological function, CAT content, MPO, and SOD activity ↓ Brain edema, TNF-α and IL-6 contents | ↑ Expression of PPAR-γ and NF-κB/65 PPARγ-regulated anti-oxidative and anti-inflammatory pathways | [3] |
| 4 | OGD-induced BV2 microglial cells | Ischemic stroke | ↑ Expression levels of PPAR-γ, Bcl-2 ↓ Expression levels of CC-3, CC-9, IL-1β, TNF-α, HMGB1, and RAGE in the hippocampus | Fcγ receptor-mediated phagocytosis pathway | [6] |
| 5 | Cerebral I/R injury-induced C57BL/6 mouse model | Cerebral I/R injury | ↑ Neurocyte survival rate ↓ Apoptotic rate, expression levels of CC-3 and ICAM-1, and TNF-α ↓ p-IκBα levels and nuclear translocation of NF-κB ↓ Phosphorylation of JAK1, expression of p-STAT1 | ↑ GRP78 expression ↓ Activation of NF-κB Anti-apoptotic and anti-inflammatory mechanisms JAK1/STAT1 signaling pathways and the regulation of ERS | [7] |
| 6 | H2O2-treated rat PC12 cells, TNF-α treated EA.hy926 cells, | Cardio-cerebral ischemic diseases | ↓ NO level and protection of Bed cell viability ↑ Content and expression of TGF-β1 ↓ Content and expression of TNF-α | ↓ Phosphorylation of NF-κB, p50, p65 and IKKα/β via the Nrf2/HO-1 pathway | [8] |
| 7 | MCAO-induced SD rat models | Cerebral I/R injury | Effect similar to that of ROZ in activating PPARγ/HO-1. | ↓ Apoptosis and inflammation via the activation of PPARγ/HO-1 | [9] |
| 8 | OGD-injured microglia model | mimics ischemia-injured microglia | ↓ NO release ↑ TGF-β level ↓ TNF-α content | The up-regulation of TGF-β expression ↓ TNF-α expression | [10] |
| 9 | H2O2-induced PC12 cells | Oxidative stress-induced neuron injury | ↓ Cytotoxicity induced by H2O2 ↓ Phosphorylation and nuclear translocation of NF-kB/p65 ↓ Phosphorylation and degradation of IκB | ↓ Activation of Akt and ERK1/2 Anti-oxidative stress and the regulation of the NF-κB pathway | [11] |
| N | Model | Tissue Sites | Effects | Mechanisms | RF |
|---|---|---|---|---|---|
| 1 | Modified Rice–Vannucci model | Hypoxia-ischemia brain injury | ↓ Neurological impairment and pathologic damage ↑ Angiogenesis after HI | ↓ CC-3 ↑ Expression of VEGF and HIF-1α signaling pathways | [12] |
| 2 | HIBD-induced SD rat model | Hypoxia ischemia brain damage (HIBD) | ↓ Apoptotic index (AI) of neurons ↑ Protein expression of p-ERK1/2 and HIF-1α ↓ Expression of CC-3 | ↑ HIF-1α expression ↓ Activation of CC-3 by the Erk1/2 signaling pathway | [13] |
| 3 | I/R-induced SD rat model | Cerebral I/R injury | ↓ Cell apoptosis ↑ ischemic conditions and cerebral recovery; ↓ Cell apoptosis and facilitating; | ↓ Cell apoptosis ↑ Differentiation of BMSCs into neurons and glial cells | [14] |
| 4 | Cerebral I/R SD rat model | Focal cerebral I/R | ↓ Changing trend in neurological deficit scores ↓ Evans blue content and aquaporin 4 expression | ↓ Aquaporin 4 expression | [14] |
| 5 | MCAO and 24-h reperfusion SD rat model | Cerebral I/R | ↓ Scores on neurofunction and the apoptosis rate ↑ Number of surviving pyramidal cells ↓ Expression of p-JNK | ↑ Expression level of p-ERK1/2 ↓ Neuronal apoptosis Regulation of the expression levels of p-ERK1/2 and p-JNK | [16] |
| 6 | HIBD-induced SD rat model | Hypoxia-ischemia brain damage | ↑ Protein expression of HIF-1α and VEGF ↑ Number of vwf-positive cells | ↑ Angiogenesis after HIBD ↑ Strengthening and stabilizing the HIF-1alpha/VEGF signaling pathways | [17] |
| 7 | HIBD-induced SD rat model | HIBD | ↑ Neural viability ↑ Angiogenesis ↑ Neurogenesis | ↑ Potential regulator of HIF-1α expression | [18] |
| 8 | Normal adult mice, global ischemia gerbil model | Cerebral I/R | ↑ Proliferation and differentiation of neural progenitor cells ↑ Neural plasticity in efficacy and structure | ↑ Expression of BDNF, Bcl-2 and anti-oxidant enzymes ↑ New synapse formation ↓ Apoptosis and calcium overload | [19] |
| 9 | Male Mongolian gerbils | Transient global ischemia | ↑ Proliferation of cells ↑ Number of surviving BrdU-positive cells | ↑ Proliferation of cells in the SGZ of adult gerbils at 11 DAI | [20] |
| N | Model | Tissue Sites | Effects | Mechanisms | RF |
|---|---|---|---|---|---|
| 1 | CI/RI-induced- C57BL/6 mouse model | Cerebral ischemia-reperfusion injury (CI/RI) | ↑ ATP, ADP, and AMP contents and the level of TAN ↑ Phosphorylation of p-AMPKα1/2 | ↑ mRNA and protein levels of GLUT3 ↑ Uptake of glucose into nerve cells via AMPKα1/2 | [96] |
| 2 | CI/RI-induced-C57BL/6 mouse model | Cerebral ischemia-reperfusion | ↑ Neurocyte survival rate ↓ Apoptotic rate and CC-3 levels ↓ Level of TNF-α and ICAM-1 mRNA ↓ Phosphorylation of IκBα and JAK1 and expression of p-STAT1 ↓ Rate of nuclear translocation of NF-κB | ↓ Activation of NF-κB and JAK1/STAT1 pathways ↑ GRP78 expression via anti-apoptosis and anti-inflammation The regulation of ERS after cerebral ischemia | [75] |
| 3 | OGD-induced BV2 microglial cells and N2a neuronal cells, | Cerebral ischemia-reperfusion | ↓ LDH leakage ↑ Neuronal cell viability ↑ Mitochondrial ultrastructure | ↓ Expression of NMDA receptor subunit 1 and activated caspase-3 | [97] |
| 4 | Meta-analysis, animal models of focal cerebral ischemia | Human ischemic stroke | ↓ Infarct volume ↑ Neurological function scores | G-Rg1 exhibited marked efficacy against acute ischemic stroke | [39] |
| 5 | MCAO model | Focal cerebral ischemia/reperfusion | ↓ Neurobehavioral function scores and infarct volume ↓ Permeability of the BBB | ↓ Expression of PAR-1 | [38] |
| 6 | OGD/R-induced cultured hippocampal neurons | Cultured hippocampal cells | ↓ Cell viability loss and cell apoptosis ↓ nNOS activity and free Ca2+ concentration | ↓ Calcium over-influx into neuronal cells ↓ nNOS activity | [98] |
| 7 | Transient global ischemia-induced adult gerbil model | Transient global ischemia | ↑ iNOS activity ↑ Hippocampal progenitor cell proliferation | Activation of iNOS activity and NMDA receptors | [99] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xie, W.; Zhou, P.; Sun, Y.; Meng, X.; Dai, Z.; Sun, G.; Sun, X. Protective Effects and Target Network Analysis of Ginsenoside Rg1 in Cerebral Ischemia and Reperfusion Injury: A Comprehensive Overview of Experimental Studies. Cells 2018, 7, 270. https://doi.org/10.3390/cells7120270
Xie W, Zhou P, Sun Y, Meng X, Dai Z, Sun G, Sun X. Protective Effects and Target Network Analysis of Ginsenoside Rg1 in Cerebral Ischemia and Reperfusion Injury: A Comprehensive Overview of Experimental Studies. Cells. 2018; 7(12):270. https://doi.org/10.3390/cells7120270
Chicago/Turabian StyleXie, Weijie, Ping Zhou, Yifan Sun, Xiangbao Meng, Ziru Dai, Guibo Sun, and Xiaobo Sun. 2018. "Protective Effects and Target Network Analysis of Ginsenoside Rg1 in Cerebral Ischemia and Reperfusion Injury: A Comprehensive Overview of Experimental Studies" Cells 7, no. 12: 270. https://doi.org/10.3390/cells7120270
APA StyleXie, W., Zhou, P., Sun, Y., Meng, X., Dai, Z., Sun, G., & Sun, X. (2018). Protective Effects and Target Network Analysis of Ginsenoside Rg1 in Cerebral Ischemia and Reperfusion Injury: A Comprehensive Overview of Experimental Studies. Cells, 7(12), 270. https://doi.org/10.3390/cells7120270






