Immune Ageing Clocks: A Methods-Oriented Review of Tasks, Modalities, Models, and Recalibration
Abstract
1. Introduction
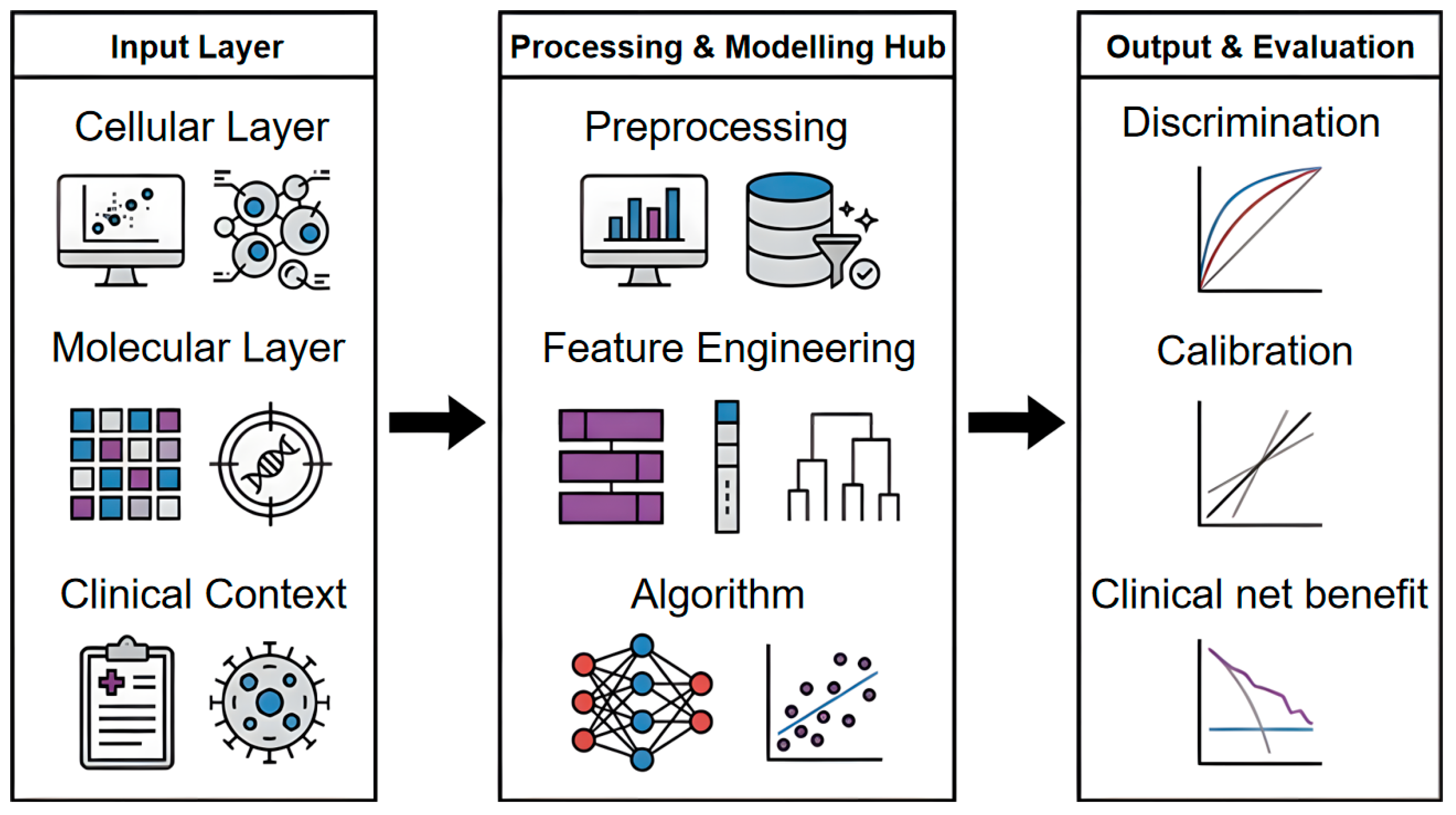
2. Construction and Evaluation Methods for the Immune Ageing Clock
2.1. Core Data Types and Sources
2.1.1. Analytical Units and Cross-Level Mapping
2.1.2. Cellular Modalities
2.1.3. Molecular Modalities
2.2. Modelling Approaches
2.3. Workflow Standardisation
2.3.1. Task Definition and Cohort Design
2.3.2. Preprocessing and Feature Engineering
2.3.3. Validation, Recalibration and Reproducible Release
2.4. Evaluation Framework for Immune Ageing Clocks
2.4.1. Discrimination and Model Fit
2.4.2. Calibration and Probability Accuracy
2.4.3. Clinical Utility and External Validity
3. Task Taxonomy of Immune Ageing Clocks
3.1. Calendar Age-Driven Age-Clock Immune Chronology
3.2. Risk Clock-Type Immune Clocks with Mortality and Multimorbidity as Endpoints
3.3. Cell/Lineage Clocks Based on Cellular States and Adaptive Receptor Repertoires
4. Current Challenges
4.1. Methodological Challenges: Batch Effects, Compositional Confounding and Cross-Cohort Transportability
4.2. Biological Challenges: Heterogeneity, CHIP and Inflammation–Metabolism Networks
4.3. Evidence and Standards: From Correlational Labels to Actionable Metrics
5. Future Outlook
5.1. Multimodal Integration and Deep Models
5.2. Tissue- and Cell-Type-Specific Clocks
5.3. Intervention-Oriented Clocks and Causal Pathways
5.4. Toward a Shared “Immune Age” Language
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organization. World Report on Ageing and Health; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- López-Otín, C.; Blasco, M.A.; Partridge, L.; Serrano, M.; Kroemer, G. The hallmarks of aging. Cell 2013, 153, 1194–1217. [Google Scholar] [CrossRef]
- Barnett, K.; Mercer, S.W.; Norbury, M.; Watt, G.; Wyke, S.; Guthrie, B. Epidemiology of multimorbidity and implications for health care, research, and medical education: A cross-sectional study. Lancet 2012, 380, 37–43. [Google Scholar] [CrossRef]
- Nikolich-Žugich, J. The twilight of immunity: Emerging concepts in aging of the immune system. Nat. Immunol. 2018, 19, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Schaum, N.; Lehallier, B.; Hahn, O.; Pálovics, R.; Hosseinzadeh, S.; Lee, S.E.; Sit, R.; Lee, D.P.; Losada, P.M.; Zardeneta, M.E. Ageing hallmarks exhibit organ-specific temporal signatures. Nature 2020, 583, 596–602. [Google Scholar] [CrossRef] [PubMed]
- Franceschi, C.; Garagnani, P.; Parini, P.; Giuliani, C.; Santoro, A. Inflammaging: A new immune-metabolic viewpoint for age-related diseases. Nat. Rev. Endocrinol. 2018, 14, 576–590. [Google Scholar] [CrossRef]
- Ciabattini, A.; Nardini, C.; Santoro, F.; Garagnani, P.; Franceschi, C.; Medaglini, D. Vaccination in the elderly: The challenge of immune changes with aging. Semin. Immunol. 2018, 40, 83–94. [Google Scholar] [CrossRef]
- Gustafson, C.E.; Kim, C.; Weyand, C.M.; Goronzy, J.J. Influence of immune aging on vaccine responses. J. Allergy Clin. Immunol. 2020, 145, 1309–1321. [Google Scholar] [CrossRef] [PubMed]
- Van Avondt, K.; Strecker, J.K.; Tulotta, C.; Minnerup, J.; Schulz, C.; Soehnlein, O. Neutrophils in aging and aging-related pathologies. Immunol. Rev. 2023, 314, 357–375. [Google Scholar] [CrossRef]
- Hazeldine, J.; Lord, J.M. The impact of ageing on natural killer cell function and potential consequences for health in older adults. Ageing Res. Rev. 2013, 12, 1069–1078. [Google Scholar] [CrossRef]
- Simmons, S.R.; Bhalla, M.; Herring, S.E.; Tchalla, E.Y.I.; Bou Ghanem, E.N. Older but Not Wiser: The Age-Driven Changes in Neutrophil Responses during Pulmonary Infections. Infect. Immun. 2021, 89, e00653-00620. [Google Scholar] [CrossRef]
- Wang, B.; Han, J.; Elisseeff, J.H.; Demaria, M. The senescence-associated secretory phenotype and its physiological and pathological implications. Nat. Rev. Mol. Cell Biol. 2024, 25, 958–978. [Google Scholar] [CrossRef]
- Alpert, A.; Pickman, Y.; Leipold, M.; Rosenberg-Hasson, Y.; Ji, X.; Gaujoux, R.; Rabani, H.; Starosvetsky, E.; Kveler, K.; Schaffert, S. A clinically meaningful metric of immune age derived from high-dimensional longitudinal monitoring. Nat. Med. 2019, 25, 487–495. [Google Scholar] [CrossRef]
- Sayed, N.; Huang, Y.; Nguyen, K.; Krejciova-Rajaniemi, Z.; Grawe, A.P.; Gao, T.; Tibshirani, R.; Hastie, T.; Alpert, A.; Cui, L. An inflammatory aging clock (iAge) based on deep learning tracks multimorbidity, immunosenescence, frailty and cardiovascular aging. Nat. Aging 2021, 1, 598–615. [Google Scholar] [CrossRef]
- Wu, F.; Mu, W.C.; Markov, N.T.; Fuentealba, M.; Halaweh, H.; Senchyna, F.; Manwaring-Mueller, M.N.; Winer, D.A.; Furman, D. Immunological biomarkers of aging. J. Immunol. 2025, 214, 889–902. [Google Scholar] [CrossRef] [PubMed]
- Müezzinler, A.; Zaineddin, A.K.; Brenner, H. A systematic review of leukocyte telomere length and age in adults. Ageing Res. Rev. 2013, 12, 509–519. [Google Scholar] [CrossRef] [PubMed]
- Aviv, A.; Valdes, A.M.; Spector, T.D. Human telomere biology: Pitfalls of moving from the laboratory to epidemiology. Int. J. Epidemiol. 2006, 35, 1424–1429. [Google Scholar] [CrossRef]
- Houseman, E.A.; Accomando, W.P.; Koestler, D.C.; Christensen, B.C.; Marsit, C.J.; Nelson, H.H.; Wiencke, J.K.; Kelsey, K.T. DNA methylation arrays as surrogate measures of cell mixture distribution. BMC Bioinform. 2012, 13, 86. [Google Scholar] [CrossRef]
- Horvath, S. DNA methylation age of human tissues and cell types. Genome Biol. 2013, 14, R115. [Google Scholar] [CrossRef] [PubMed]
- Hannum, G.; Guinney, J.; Zhao, L.; Zhang, L.; Hughes, G.; Sadda, S.; Klotzle, B.; Bibikova, M.; Fan, J.B.; Gao, Y. Genome-wide methylation profiles reveal quantitative views of human aging rates. Mol. Cell 2013, 49, 359–367. [Google Scholar] [CrossRef]
- Levine, M.E.; Lu, A.T.; Quach, A.; Chen, B.H.; Assimes, T.L.; Bandinelli, S.; Hou, L.; Baccarelli, A.A.; Stewart, J.D.; Li, Y. An epigenetic biomarker of aging for lifespan and healthspan. Aging 2018, 10, 573–591. [Google Scholar] [CrossRef]
- Jaffe, A.E.; Irizarry, R.A. Accounting for cellular heterogeneity is critical in epigenome-wide association studies. Genome Biol. 2014, 15, R31. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Zhang, Z.; Kumar, S.; Botey-Bataller, J.; Zoodsma, M.; Ehsani, A.; Zhan, Q.; Alaswad, A.; Zhou, L.; Grondman, I. Single-cell immune aging clocks reveal inter-individual heterogeneity during infection and vaccination. Nat. Aging 2025, 5, 607–621. [Google Scholar] [CrossRef]
- Lehallier, B.; Gate, D.; Schaum, N.; Nanasi, T.; Lee, S.E.; Yousef, H.; Moran Losada, P.; Berdnik, D.; Keller, A.; Verghese, J. Undulating changes in human plasma proteome profiles across the lifespan. Nat. Med. 2019, 25, 1843–1850. [Google Scholar] [CrossRef]
- Gong, Q.; Sharma, M.; Glass, M.C.; Kuan, E.L.; Chander, A.; Singh, M.; Graybuck, L.T.; Thomson, Z.J.; LaFrance, C.M.; Rachid Zaim, S. Multi-omic profiling reveals age-related immune dynamics in healthy adults. Nature 2025, 648, 696–706. [Google Scholar] [CrossRef]
- Mi, T.; Soerens, A.G.; Alli, S.; Kang, T.G.; Vasandan, A.B.; Wang, Z.; Vezys, V.; Kimura, S.; Iacobucci, I.; Baylin, S.B. Conserved epigenetic hallmarks of T cell aging during immunity and malignancy. Nat. Aging 2024, 4, 1053–1063. [Google Scholar] [CrossRef]
- Jaiswal, S.; Ebert, B.L. Clonal hematopoiesis in human aging and disease. Science 2019, 366, eaan4673. [Google Scholar] [CrossRef]
- Jaiswal, S.; Libby, P. Clonal haematopoiesis: Connecting ageing and inflammation in cardiovascular disease. Nat. Rev. Cardiol. 2020, 17, 137–144. [Google Scholar] [CrossRef]
- Van Calster, B.; McLernon, D.J.; van Smeden, M.; Wynants, L.; Steyerberg, E.W.; Topic Group ‘Evaluating Diagnostic Tests and Prediction Models’ of the STRATOS Initiative. Calibration: The Achilles heel of predictive analytics. BMC Med. 2019, 17, 230. [Google Scholar] [CrossRef]
- Ramspek, C.L.; Jager, K.J.; Dekker, F.W.; Zoccali, C.; van Diepen, M. External validation of prognostic models: What, why, how, when and where? Clin. Kidney J. 2020, 14, 49–58. [Google Scholar] [CrossRef]
- Vickers, A.J.; Elkin, E.B. Decision curve analysis: A novel method for evaluating prediction models. Med. Decis. Mak. 2006, 26, 565–574. [Google Scholar] [CrossRef] [PubMed]
- Rutledge, J.; Oh, H.; Wyss-Coray, T. Measuring biological age using omics data. Nat. Rev. Genet. 2022, 23, 715–727. [Google Scholar] [CrossRef]
- Finlayson, S.G.; Subbaswamy, A.; Singh, K.; Bowers, J.; Kupke, A.; Zittrain, J.; Kohane, I.S.; Saria, S. The Clinician and Dataset Shift in Artificial Intelligence. N. Engl. J. Med. 2021, 385, 283–286. [Google Scholar] [CrossRef]
- Jylhävä, J.; Pedersen, N.L.; Hägg, S. Biological age predictors. EBioMedicine 2017, 21, 29–36. [Google Scholar] [CrossRef]
- Mhanna, V.; Bashour, H.; Lê Quý, K.; Barennes, P.; Rawat, P.; Greiff, V.; Mariotti-Ferrandiz, E. Adaptive immune receptor repertoire analysis. Nat. Rev. Methods Primers 2024, 4, 6. [Google Scholar] [CrossRef]
- Luecken, M.D.; Theis, F.J. Current best practices in single-cell RNA-seq analysis: A tutorial. Mol. Syst. Biol. 2019, 15, e8746. [Google Scholar] [CrossRef]
- Stoeckius, M.; Hafemeister, C.; Stephenson, W.; Houck-Loomis, B.; Chattopadhyay, P.K.; Swerdlow, H.; Satija, R.; Smibert, P. Simultaneous epitope and transcriptome measurement in single cells. Nat. Methods 2017, 14, 865–868. [Google Scholar] [CrossRef]
- Buenrostro, J.D.; Wu, B.; Litzenburger, U.M.; Ruff, D.; Gonzales, M.L.; Snyder, M.P.; Chang, H.Y.; Greenleaf, W.J. Single-cell chromatin accessibility reveals principles of regulatory variation. Nature 2015, 523, 486–490. [Google Scholar] [CrossRef]
- Quer, G.; Radin, J.M.; Gadaleta, M.; Baca-Motes, K.; Ariniello, L.; Ramos, E.; Kheterpal, V.; Topol, E.J.; Steinhubl, S.R. Wearable sensor data and self-reported symptoms for COVID-19 detection. Nat. Med. 2021, 27, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Mishra, T.; Wang, M.; Metwally, A.A.; Bogu, G.K.; Brooks, A.W.; Bahmani, A.; Alavi, A.; Celli, A.; Higgs, E.; Dagan-Rosenfeld, O. Pre-symptomatic detection of COVID-19 from smartwatch data. Nat. Biomed. Eng. 2020, 4, 1208–1220. [Google Scholar] [CrossRef] [PubMed]
- Rudin, C. Stop explaining black box machine learning models for high stakes decisions and use interpretable models instead. Nat. Mach. Intell. 2019, 1, 206–215. [Google Scholar] [CrossRef]
- Lundberg, S.M.; Erion, G.; Chen, H.; DeGrave, A.; Prutkin, J.M.; Nair, B.; Katz, R.; Himmelfarb, J.; Bansal, N.; Lee, S.I. From local explanations to global understanding with explainable AI for trees. Nat. Mach. Intell. 2020, 2, 56–67. [Google Scholar] [CrossRef]
- Zhao, Z.; Zobolas, J.; Zucknick, M.; Aittokallio, T. Tutorial on survival modeling with applications to omics data. Bioinformatics 2024, 40, btae132. [Google Scholar] [CrossRef]
- Cui, H.; Wang, C.; Maan, H.; Pang, K.; Luo, F.; Duan, N.; Wang, B. scGPT: Toward building a foundation model for single-cell multi-omics using generative AI. Nat. Methods 2024, 21, 1470–1480. [Google Scholar] [CrossRef] [PubMed]
- Stuart, T.; Butler, A.; Hoffman, P.; Hafemeister, C.; Papalexi, E.; Mauck, W.M., 3rd; Hao, Y.; Stoeckius, M.; Smibert, P.; Satija, R. Comprehensive integration of single-cell data. Cell 2019, 177, 1888–1902.e1821. [Google Scholar] [CrossRef] [PubMed]
- Korsunsky, I.; Millard, N.; Fan, J.; Slowikowski, K.; Zhang, F.; Wei, K.; Baglaenko, Y.; Brenner, M.; Loh, P.R.; Raychaudhuri, S. Fast, sensitive and accurate integration of single-cell data with Harmony. Nat. Methods 2019, 16, 1289–1296. [Google Scholar] [CrossRef]
- Gayoso, A.; Steier, Z.; Lopez, R.; Regier, J.; Nazor, K.L.; Streets, A.; Yosef, N. Joint probabilistic modeling of single-cell multi-omic data with totalVI. Nat. Methods 2021, 18, 272–282. [Google Scholar] [CrossRef]
- Hao, Y.; Hao, S.; Andersen-Nissen, E.; Mauck, W.M., 3rd; Zheng, S.; Butler, A.; Lee, M.J.; Wilk, A.J.; Darby, C.; Zager, M. Integrated analysis of multimodal single-cell data. Cell 2021, 184, 3573–3587.e3529. [Google Scholar] [CrossRef] [PubMed]
- Luecken, M.D.; Büttner, M.; Chaichoompu, K.; Danese, A.; Interlandi, M.; Mueller, M.F.; Strobl, D.C.; Zappia, L.; Dugas, M.; Colomé-Tatché, M.; et al. Benchmarking atlas-level data integration in single-cell genomics. Nat. Methods 2022, 19, 41–50. [Google Scholar] [CrossRef]
- Lea, A.S.; Jones, D.S. Mind the Gap—Machine Learning, Dataset Shift, and History in the Age of Clinical Algorithms. N. Engl. J. Med. 2024, 390, 293–295. [Google Scholar] [CrossRef]
- Bhattacharya, S.; Dunn, P.; Thomas, C.G.; Smith, B.; Schaefer, H.; Chen, J.; Hu, Z.; Zalocusky, K.A.; Shankar, R.D.; Shen-Orr, S.S. ImmPort, toward repurposing of open access immunological assay data for translational and clinical research. Sci. Data 2018, 5, 180015. [Google Scholar] [CrossRef]
- Barrett, T.; Wilhite, S.E.; Ledoux, P.; Evangelista, C.; Kim, I.F.; Tomashevsky, M.; Marshall, K.A.; Phillippy, K.H.; Sherman, P.M.; Holko, M. NCBI GEO: Archive for functional genomics data sets—Update. Nucleic Acids Res. 2013, 41, D991–D995. [Google Scholar] [CrossRef]
- Athar, A.; Füllgrabe, A.; George, N.; Iqbal, H.; Huerta, L.; Ali, A.; Snow, C.; Fonseca, N.A.; Petryszak, R.; Papatheodorou, I. ArrayExpress update—From bulk to single-cell expression data. Nucleic Acids Res. 2019, 47, D711–D715. [Google Scholar] [CrossRef]
- Collins, G.S.; Reitsma, J.B.; Altman, D.G.; Moons, K.G. Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): The TRIPOD statement. BMJ 2015, 350, g7594. [Google Scholar] [CrossRef]
- Collins, G.S.; Moons, K.G.M.; Dhiman, P.; Riley, R.D.; Beam, A.L.; Van Calster, B.; Ghassemi, M.; Liu, X.; Reitsma, J.B.; van Smeden, M. TRIPOD+AI statement: Updated guidance for reporting clinical prediction models that use regression or machine learning methods. BMJ 2024, 385, e078378. [Google Scholar] [CrossRef] [PubMed]
- Moons, K.G.M.; Wolff, R.F.; Riley, R.D.; Whiting, P.F.; Westwood, M.; Collins, G.S.; Reitsma, J.B.; Kleijnen, J.; Mallett, S. PROBAST: A Tool to Assess Risk of Bias and Applicability of Prediction Model Studies: Explanation and Elaboration. Ann. Intern. Med. 2019, 170, W1–W33. [Google Scholar] [CrossRef] [PubMed]
- Moons, K.G.M.; Damen, J.A.A.; Kaul, T.; Hooft, L.; Andaur Navarro, C.; Dhiman, P.; Beam, A.L.; Van Calster, B.; Celi, L.A.; Denaxas, S. PROBAST+AI: An updated quality, risk of bias, and applicability assessment tool for prediction models using regression or artificial intelligence methods. BMJ 2025, 388, e082505. [Google Scholar] [CrossRef]
- Hernandez-Boussard, T.; Bozkurt, S.; Ioannidis, J.P.A.; Shah, N.H. MINIMAR (MINimum Information for Medical AI Reporting): Developing reporting standards for artificial intelligence in health care. J. Am. Med. Inform. Assoc. 2020, 27, 2011–2015. [Google Scholar] [CrossRef]
- Vasey, B.; Nagendran, M.; Campbell, B.; Clifton, D.A.; Collins, G.S.; Denaxas, S.; Denniston, A.K.; Faes, L.; Geerts, B.; Ibrahim, M. Reporting guideline for the early stage clinical evaluation of decision support systems driven by artificial intelligence: DECIDE-AI. BMJ 2022, 377, e070904. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.A.; Shokhirev, M.N. Contextualizing aging clocks and properly describing biological age. Aging Cell 2024, 23, e14377. [Google Scholar] [CrossRef]
- Steyerberg, E.W. Clinical Prediction Models: A Practical Approach to Development, Validation, and Updating, 2nd ed.; Springer: Cham, Switzerland, 2019. [Google Scholar]
- Efthimiou, O.; Seo, M.; Chalkou, K.; Debray, T.; Egger, M.; Salanti, G. Developing clinical prediction models: A step-by-step guide. BMJ 2024, 386, e078276. [Google Scholar] [CrossRef]
- Mogilenko, D.A.; Shpynov, O.; Andhey, P.S.; Arthur, L.; Swain, A.; Esaulova, E.; Brioschi, S.; Shchukina, I.; Kerndl, M.; Bambouskova, M. Comprehensive profiling of an aging immune system reveals clonal GZMK+ CD8+ T cells as conserved hallmark of inflammaging. Immunity 2021, 54, 99–115.e112. [Google Scholar] [CrossRef]
- Wang, Y.; Ke, D.; Chen, Y.; Zhang, C.; Liu, W.; Chen, L.; Pu, J. Decoding immune aging at single-cell resolution. Trends Immunol. 2025, 46, 791–807. [Google Scholar] [CrossRef]
- Newman, A.M.; Steen, C.B.; Liu, C.L.; Gentles, A.J.; Chaudhuri, A.A.; Scherer, F.; Khodadoust, M.S.; Esfahani, M.S.; Luca, B.A.; Steiner, D. Determining cell type abundance and expression from bulk tissues with digital cytometry. Nat. Biotechnol. 2019, 37, 773–782. [Google Scholar] [CrossRef]
- Cossarizza, A.; Chang, H.D.; Radbruch, A.; Acs, A.; Adam, D.; Adam-Klages, S.; Agace, W.W.; Aghaeepour, N.; Akdis, M.; Allez, M. Guidelines for the use of flow cytometry and cell sorting in immunological studies (second edition). Eur. J. Immunol. 2019, 49, 1457–1973. [Google Scholar] [CrossRef] [PubMed]
- Bendall, S.C.; Simonds, E.F.; Qiu, P.; Amir, e.-A.D.; Krutzik, P.O.; Finck, R.; Bruggner, R.V.; Melamed, R.; Trejo, A.; Ornatsky, O.I. Single-cell mass cytometry of differential immune and drug responses across a human hematopoietic continuum. Science 2011, 332, 687–696. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Jin, W.; Ju, L.; Fu, J.; Wang, G.; Yu, M.; Chen, F.; Qian, K.; Wang, X.; Zhang, Y. Tracking single-cell evolution using clock-like chromatin accessibility loci. Nat. Biotechnol. 2025, 43, 784–798. [Google Scholar] [CrossRef]
- Hu, J.; Pan, M.; Reid, B. Quantifiable blood TCR repertoire components associate with immune aging. Nat. Commun. 2024, 15, 8171. [Google Scholar] [CrossRef] [PubMed]
- Corrie, B.D.; Christley, S.; Busse, C.E.; Cowell, L.G.; Neller, K.C.M.; Rubelt, F.; Schwab, N.; Community, A. Data Sharing and Reuse: A Method by the AIRR Community. Methods Mol. Biol. 2022, 2453, 447–476. [Google Scholar] [CrossRef]
- Ren, X.; Kuan, P.F. RNAAgeCalc: A multi-tissue transcriptional age calculator. PLoS ONE 2020, 15, e0237006. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Wang, Z.; Wang, Y.; Zhu, Y.; Zhou, Q.; Jian, X.; Zhao, G.; Qiu, J.; Xia, K.; Tang, B. A metabolomic profile of biological aging in 250,341 individuals from the UK Biobank. Nat. Commun. 2024, 15, 8081. [Google Scholar] [CrossRef]
- Zhu, H.; Chen, J.; Liu, K.; Gao, L.; Wu, H.; Ma, L.; Zhou, J.; Liu, Z.; Han, J.J. Human PBMC scRNA-seq-based aging clocks reveal ribosome to inflammation balance as a single-cell aging hallmark and super longevity. Sci. Adv. 2023, 9, eabq7599. [Google Scholar] [CrossRef]
- Horvath, S.; Raj, K. DNA methylation-based biomarkers and the epigenetic clock theory of ageing. Nat. Rev. Genet. 2018, 19, 371–384. [Google Scholar] [CrossRef]
- The ENCODE Project Consortium. An integrated encyclopedia of DNA elements in the human genome. Nature 2012, 489, 57–74. [Google Scholar] [CrossRef]
- Roadmap Epigenomics Consortium; Kundaje, A.; Meuleman, W.; Ernst, J.; Bilenky, M.; Yen, A.; Heravi-Moussavi, A.; Kheradpour, P.; Zhang, Z.; Wang, J. Integrative analysis of 111 reference human epigenomes. Nature 2015, 518, 317–330. [Google Scholar] [CrossRef]
- Argentieri, M.A.; Xiao, S.; Bennett, D.; Winchester, L.; Nevado-Holgado, A.J.; Ghose, U.; Albukhari, A.; Yao, P.; Mazidi, M.; Lv, J. Proteomic aging clock predicts mortality and risk of common age-related diseases in diverse populations. Nat. Med. 2024, 30, 2450–2460. [Google Scholar] [CrossRef]
- Mutz, J.; Iniesta, R.; Lewis, C.M. Metabolomic age (MileAge) predicts health and life span: A comparison of multiple machine learning algorithms. Sci. Adv. 2024, 10, eadp3743. [Google Scholar] [CrossRef]
- Nie, C.; Li, Y.; Li, R.; Yan, Y.; Zhang, D.; Li, T.; Li, Z.; Sun, Y.; Zhen, H.; Ding, J. Distinct biological ages of organs and systems identified from a multi-omics study. Cell Rep. 2022, 38, 110459. [Google Scholar] [CrossRef]
- Argelaguet, R.; Arnol, D.; Bredikhin, D. MOFA+: A statistical framework for comprehensive integration of multi-modal single-cell data. Genome Biol. 2020, 21, 111. [Google Scholar] [CrossRef] [PubMed]
- Field, A.E.; Robertson, N.A.; Wang, T.; Havas, A.; Ideker, T.; Adams, P.D. DNA methylation clocks in aging: Categories, causes, and consequences. Mol. Cell 2018, 71, 882–895. [Google Scholar] [CrossRef] [PubMed]
- Lu, A.T.; Quach, A.; Wilson, J.G.; Reiner, A.P.; Aviv, A.; Raj, K.; Hou, L.; Baccarelli, A.A.; Li, Y.; Stewart, J.D. DNA methylation GrimAge strongly predicts lifespan and healthspan. Aging 2019, 11, 303–327. [Google Scholar] [CrossRef] [PubMed]
- Marioni, R.E.; Shah, S.; McRae, A.F.; Ritchie, S.J.; Muniz-Terrera, G.; Harris, S.E.; Gibson, J.; Redmond, P.; Cox, S.R.; Pattie, A. The epigenetic clock is correlated with physical and cognitive fitness in the Lothian Birth Cohort 1936. Int. J. Epidemiol. 2015, 44, 1388–1396. [Google Scholar] [CrossRef]
- Johnson, W.E.; Li, C.; Rabinovic, A. Adjusting batch effects in microarray expression data using empirical Bayes methods. Biostatistics 2007, 8, 118–127. [Google Scholar] [CrossRef] [PubMed]
- Leek, J.T.; Johnson, W.E.; Parker, H.S.; Jaffe, A.E.; Storey, J.D. The sva package for removing batch effects and other unwanted variation in high-throughput experiments. Bioinformatics 2012, 28, 882–883. [Google Scholar] [CrossRef]
- Pomponio, R.; Erus, G.; Habes, M.; Doshi, J.; Srinivasan, D.; Mamourian, E.; Bashyam, V.; Nasrallah, I.M.; Satterthwaite, T.D.; Fan, Y. Harmonization of large MRI datasets for the analysis of brain imaging patterns throughout the lifespan. NeuroImage 2020, 208, 116450. [Google Scholar] [CrossRef]
- Van Gassen, S.; Gaudilliere, B.; Angst, M.S.; Saeys, Y.; Aghaeepour, N. CytoNorm: A normalization algorithm for cytometry data. Cytom. A 2020, 97, 268–278. [Google Scholar] [CrossRef]
- Wolock, S.L.; Lopez, R.; Klein, A.M. Scrublet: Computational identification of cell doublets in single-cell transcriptomic data. Cell Syst. 2019, 8, 281–291.e289. [Google Scholar] [CrossRef]
- Sun, X.; Nguyen, T.; Achour, A.; Ko, A.; Cifello, J.; Ling, C.; Sharma, J.; Hiroi, T.; Zhang, Y.; Chia, C.W. Longitudinal analysis reveals age-related changes in the T cell receptor repertoire of human T cell subsets. J. Clin. Investig. 2022, 132, e158122. [Google Scholar] [CrossRef]
- Wang, X.; Park, J.; Susztak, K.; Zhang, N.R.; Li, M. Bulk tissue cell type deconvolution with multi-subject single-cell expression reference. Nat. Commun. 2019, 10, 380. [Google Scholar] [CrossRef]
- Avila Cobos, F.; Alquicira-Hernandez, J.; Powell, J.E.; Mestdagh, P.; De Preter, K. Benchmarking of cell type deconvolution pipelines for transcriptomics data. Nat. Commun. 2020, 11, 5650. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Park, J.E.; Kim, H.; Park, S.H. Review of statistical methods for evaluating the performance of survival or other time-to-event prediction models (from conventional to deep learning approaches). Korean J. Radiol. 2021, 22, 1697–1707. [Google Scholar] [CrossRef] [PubMed]
- Bansal, A.; Heagerty, P.J. A tutorial on evaluating the time-varying discrimination accuracy of survival models used in dynamic decision making. Med. Decis. Mak. 2018, 38, 904–916. [Google Scholar] [CrossRef]
- Huang, Y.; Li, W.; Macheret, F.; Gabriel, R.A.; Ohno-Machado, L. A tutorial on calibration measurements and calibration models for clinical prediction models. J. Am. Med. Inform. Assoc. 2020, 27, 621–633. [Google Scholar] [CrossRef]
- Binuya, M.A.E.; Engelhardt, E.G.; Schats, W.; Schmidt, M.K.; Steyerberg, E.W. Methodological guidance for the evaluation and updating of clinical prediction models: A systematic review. BMC Med. Res. Methodol. 2022, 22, 316. [Google Scholar] [CrossRef]
- Kattan, M.W.; Gerds, T.A. A framework for the evaluation of statistical prediction models. Chest 2020, 158, S29–S38. [Google Scholar] [CrossRef] [PubMed]
- Belsky, D.W.; Caspi, A.; Corcoran, D.L.; Sugden, K.; Poulton, R.; Arseneault, L.; Baccarelli, A.; Chamarti, K.; Gao, X.; Hannon, E. DunedinPACE, a DNA methylation biomarker of the pace of aging. eLife 2022, 11, e73420. [Google Scholar] [CrossRef] [PubMed]
- Silva Filho, T.; Song, H.; Perello-Nieto, M.; Santos-Rodriguez, R.; Kull, M.; Flach, P. Classifier calibration: A survey on how to assess and improve predicted class probabilities. Mach. Learn. 2023, 112, 3211–3260. [Google Scholar] [CrossRef]
- Vickers, A.J.; Holland, F. Decision curve analysis to evaluate the clinical benefit of prediction models. Spine J. 2021, 21, 1643–1648. [Google Scholar] [CrossRef]
- Heller, G. A Modified net reclassification improvement statistic. J. Stat. Plan. Inference 2023, 227, 18–33. [Google Scholar] [CrossRef]
- Palacios-Pedrero, M.Ã.; Jansen, J.M.; Blume, C.; Stanislawski, N.; Jonczyk, R.; Molle, A.; Hernandez, M.G.; Kaiser, F.K.; Jung, K.; Osterhaus, A.D.M.E. Signs of immunosenescence correlate with poor outcome of mRNA COVID-19 vaccination in older adults. Nat. Aging 2022, 2, 896–905. [Google Scholar] [CrossRef]
- Scepanovic, P.; Alanio, C.; Hammer, C.; Hodel, F.; Bergstedt, J.; Patin, E.; Thorball, C.W.; Chaturvedi, N.; Charbit, B.; Abel, L. Human genetic variants and age are the strongest predictors of humoral immune responses to common pathogens and vaccines. Genome Med. 2018, 10, 59. [Google Scholar] [CrossRef]
- Waziry, R.; Ryan, C.P.; Corcoran, D.L.; Huffman, K.M.; Kobor, M.S.; Kothari, M.; Graf, G.H.; Kraus, V.B.; Kraus, W.E.; Lin, D.T.S. Effect of long-term caloric restriction on DNA methylation measures of biological aging in healthy adults from the CALERIE trial. Nat. Aging 2023, 3, 248–257. [Google Scholar] [CrossRef]
- Pribić, T.; Das, J.K.; Đerek, L.; Belsky, D.W.; Orenduff, M.; Huffman, K.M.; Kraus, W.E.; Deriš, H.; Šimunović, J.; Štambuk, T. A 2-year calorie restriction intervention may reduce glycomic biological age biomarkers—A pilot study. NPJ Aging 2025, 11, 71. [Google Scholar] [CrossRef]
- Karagiannis, T.T.; Dowrey, T.W.; Villacorta-Martin, C.; Montano, M.; Reed, E.; Belkina, A.C.; Andersen, S.L.; Perls, T.T.; Monti, S.; Murphy, G.J. Multi-modal profiling of peripheral blood cells across the human lifespan reveals distinct immune cell signatures of aging and longevity. EBioMedicine 2023, 90, 104514. [Google Scholar] [CrossRef] [PubMed]
- Strindhall, J.; Nilsson, B.O.; Löfgren, S.; Ernerudh, J.; Pawelec, G.; Johansson, B.; Wikby, A. No immune risk profile among individuals who reach 100 years of age: Findings from the Swedish NONA immune longitudinal study. Exp. Gerontol. 2007, 42, 753–761. [Google Scholar] [CrossRef] [PubMed]
- Greto, V.L.; Cvetko, A.; Štambuk, T.; Dempster, N.J.; Kifer, D.; Deriš, H.; Cindrić, A.; Vučković, F.; Falchi, M.; Gillies, R.S. Extensive weight loss reduces glycan age by altering IgG N-glycosylation. Int. J. Obes. 2021, 45, 1521–1531. [Google Scholar] [CrossRef]
- Xu, X.; Li, R.; Mo, O.; Liu, K.; Li, J.; Hao, P. Cell-type deconvolution for bulk RNA-seq data using single-cell reference: A comparative analysis and recommendation guideline. Brief. Bioinform. 2025, 26, bbaf031. [Google Scholar] [CrossRef]
- Terekhova, M.; Swain, A.; Bohacova, P.; Aladyeva, E.; Arthur, L.; Laha, A.; Mogilenko, D.A.; Burdess, S.; Sukhov, V.; Kleverov, D. Single-cell atlas of healthy human blood unveils age-related loss of NKG2C+GZMB− CD8+ memory T cells and accumulation of type 2 memory T cells. Immunity 2023, 56, 2836–2854.e2839. [Google Scholar] [CrossRef]
- Lu, J.; Ahmad, R.; Nguyen, T.; Cifello, J.; Hemani, H.; Li, J.; Chen, J.; Li, S.; Wang, J.; Achour, A. Heterogeneity and transcriptome changes of human CD8+ T cells across nine decades of life. Nat. Commun. 2022, 13, 5128. [Google Scholar] [CrossRef]
- Corrie, B.D.; Marthandan, N.; Zimonja, B.; Jaglale, J.; Zhou, Y.; Barr, E.; Knoetze, N.; Breden, F.M.W.; Christley, S.; Scott, J.K. iReceptor: A platform for querying and analyzing antibody/B-cell and T-cell receptor repertoire data across federated repositories. Immunol. Rev. 2018, 284, 24–41. [Google Scholar] [CrossRef]
- Mannick, J.B.; Morris, M.; Hockey, H.P.; Roma, G.; Beibel, M.; Kulmatycki, K.; Watkins, M.; Shavlakadze, T.; Zhou, W.; Quinn, D. TORC1 inhibition enhances immune function and reduces infections in the elderly. Sci. Transl. Med. 2018, 10, eaaq1564. [Google Scholar] [CrossRef] [PubMed]
- Andaur Navarro, C.L.; Damen, J.A.A.; Takada, T.; Nijman, S.W.J.; Dhiman, P.; Ma, J.; Collins, G.S.; Bajpai, R.; Riley, R.D.; Moons, K.G.M.; et al. Completeness of reporting of clinical prediction models developed using supervised machine learning: A systematic review. BMC Med. Res. Methodol. 2022, 22, 12. [Google Scholar] [CrossRef] [PubMed]
- Márquez, E.J.; Chung, C.H.; Marches, R.; Rossi, R.J.; Nehar-Belaid, D.; Eroglu, A.; Mellert, D.J.; Kuchel, G.A.; Banchereau, J.; Ucar, D. Sexual-dimorphism in human immune system aging. Nat. Commun. 2020, 11, 751. [Google Scholar] [CrossRef] [PubMed]
- Spidlen, J.; Breuer, K.; Rosenberg, C.; Kotecha, N.; Brinkman, R.R. FlowRepository: A resource of annotated flow cytometry datasets associated with peer-reviewed publications. Cytometry A 2012, 81, 727–731. [Google Scholar] [CrossRef] [PubMed]
- Čuklina, J.; Lee, C.H.; Williams, E.G.; Sajic, T.; Collins, B.C.; Rodríguez Martínez, M.; Sharma, V.S.; Wendt, F.; Goetze, S.; Keele, G.R. Diagnostics and correction of batch effects in large-scale proteomic studies: A tutorial. Mol. Syst. Biol. 2021, 17, e10240. [Google Scholar] [CrossRef]
- Zhang, Y.; Parmigiani, G.; Johnson, W.E. ComBat-seq: Batch effect adjustment for RNA-seq count data. NAR Genom. Bioinform. 2020, 2, lqaa078. [Google Scholar] [CrossRef]
- Jew, B.; Alvarez, M.; Rahmani, E.; Miao, Z.; Ko, A.; Garske, K.M.; Sul, J.H.; Pietiläinen, K.H.; Pajukanta, P.; Halperin, E. Accurate estimation of cell composition in bulk expression through robust integration of single-cell information. Nat. Commun. 2020, 11, 1971. [Google Scholar] [CrossRef]
- Steyerberg, E.W.; Moons, K.G.; van der Windt, D.A.; Hayden, J.A.; Perel, P.; Schroter, S.; Riley, R.D.; Hemingway, H.; Altman, D.G.; Group, P. Prognosis Research Strategy (PROGRESS) 3: Prognostic model research. PLoS Med. 2013, 10, e1001381. [Google Scholar] [CrossRef]
- Risso, D.; Ngai, J.; Speed, T.P.; Dudoit, S. Normalization of RNA-seq data using factor analysis of control genes or samples. Nat. Biotechnol. 2014, 32, 896–902. [Google Scholar] [CrossRef]
- Parker, H.S.; Corrada Bravo, H.; Leek, J.T. Removing batch effects for prediction problems with frozen surrogate variable analysis. PeerJ 2014, 2, e561. [Google Scholar] [CrossRef]
- Newman, A.M.; Liu, C.L.; Green, M.R.; Gentles, A.J.; Feng, W.; Xu, Y.; Hoang, C.D.; Diehn, M.; Alizadeh, A.A. Robust enumeration of cell subsets from tissue expression profiles. Nat. Methods 2015, 12, 453–457. [Google Scholar] [CrossRef]
- van de Berg, P.J.; Griffiths, S.J.; Yong, S.L.; Macaulay, R.; Bemelman, F.J.; Jackson, S.; Henson, S.M.; ten Berge, I.J.; Akbar, A.N.; van Lier, R.A. Cytomegalovirus infection reduces telomere length of the circulating T cell pool. J. Immunol. 2010, 184, 3417–3423. [Google Scholar] [CrossRef]
- Dowd, J.B.; Bosch, J.A.; Steptoe, A.; Blackburn, E.H.; Lin, J.; Rees-Clayton, E.; Aiello, A.E. Cytomegalovirus is associated with reduced telomerase activity in the Whitehall II cohort. Exp. Gerontol. 2013, 48, 385–390. [Google Scholar] [CrossRef]
- Ahadi, S.; Zhou, W.; Schüssler-Fiorenza Rose, S.M.; Sailani, M.R.; Contrepois, K.; Avina, M.; Ashland, M.; Brunet, A.; Snyder, M. Personal aging markers and ageotypes revealed by deep longitudinal profiling. Nat. Med. 2020, 26, 83–90. [Google Scholar] [CrossRef]
- Rivera, S.C.; Liu, X.; Chan, A.W.; Denniston, A.K.; Calvert, M.J.; Spirit, A.I.; Group, C.-A.W. Guidelines for clinical trial protocols for interventions involving artificial intelligence: The SPIRIT-AI Extension. BMJ 2020, 370, m3210. [Google Scholar] [CrossRef]
- Liu, X.; Rivera, S.C.; Moher, D.; Calvert, M.J.; Denniston, A.K.; Spirit, A.I.; Group, C.-A.W. Reporting guidelines for clinical trial reports for interventions involving artificial intelligence: The CONSORT-AI Extension. BMJ 2020, 370, m3164. [Google Scholar] [CrossRef] [PubMed]
- Niarakis, A.; Laubenbacher, R.; An, G.; Ilan, Y.; Fisher, J.; Flobak, Ã.; Reiche, K.; RodrÃguez MartÃnez, M.; Geris, L.; Ladeira, L. Immune digital twins for complex human pathologies: Applications, limitations, and challenges. npj Syst. Biol. Appl. 2024, 10, 141. [Google Scholar] [CrossRef] [PubMed]
- Regev, A.; Teichmann, S.A.; Lander, E.S.; Amit, I.; Benoist, C.; Birney, E.; Bodenmiller, B.; Campbell, P.; Carninci, P.; Clatworthy, M. The Human Cell Atlas. eLife 2017, 6, e27041. [Google Scholar] [CrossRef] [PubMed]
- Lotfollahi, M.; Naghipourfar, M.; Luecken, M.D.; Khajavi, M.; Büttner, M.; Wagenstetter, M.; Avsec, Å.; Gayoso, A.; Yosef, N.; Interlandi, M. Mapping single-cell data to reference atlases by transfer learning. Nat. Biotechnol. 2022, 40, 121–130. [Google Scholar] [CrossRef]


| Dimension | Prior Reviews (Typical Coverage) | This Review (What We Add) |
|---|---|---|
| Tasks | Often mixed or implicit (age vs. outcome vs. lineage-level) [4,34] | Explicit task taxonomy: age/risk(outcome)/cell lineage (intracellular and repertoire) [13,14,35] |
| Modality | Described by assay types, less by analytical unit [36,37,38] | “Cell layer vs. molecular layer vs. clinical–digital context” as modelling units + cross-level mapping [25,39,40] |
| Models | Broad ML overview, limited linkage to task/modality [41,42] | Model choices mapped to task–modality constraints (statistics/ML/DL) [43,44] |
| Fusion | Mentioned but not systematised [45,46] | Early vs. late fusion as a design decision with pros/cons and failure modes [47,48,49] |
| Evaluation | Emphasis on fit/correlation/AUC [19,20] | Adds calibration, clinical net benefit, and deployment metrics as core [29,30,31] |
| Recalibration/transportability | Often brief or absent | Recalibration + internal–external CV/LOCO as standard practice [33,50] |
| Reporting | General reproducibility statements [51,52,53] | Concrete reporting checklist: leakage control, covariates, missingness, calibration, code/data share [54,55,56,57,58,59] |
| Clock/Framework | Data Modality and Features | Population/Cohort | Primary Target/Outcome | Modelling Strategy | Key Message/Methodological Contribution |
|---|---|---|---|---|---|
| IMM-AGE [13] | Longitudinal high-dimensional immune phenotyping (flow cytometry panels, plasma cytokines), clinical covariates | ~135 adults (8-year follow-up) from the Stanford 1000 Immunomes-related cohorts | Composite “immune age” capturing longitudinal immune remodelling; association with mortality, CMV status, inflammatory burden | Regularised regression and manifold learning on longitudinal immune features | Defines a clinically meaningful immune age metric derived from longitudinal immune system trajectories, integrating multiple immune cell subsets and soluble markers, and linking immune age acceleration to morbidity and mortality. |
| iAge—inflammatory ageing clock [14] | Deep profiling of plasma inflammatory proteome (“blood immunome”, >50 cytokines/chemokines), clinical phenotypes | 1001 individuals aged 8–96 years, multi-cohort; centenarians included | Continuous “inflammatory age” (iAge); multimorbidity index, frailty, immunosenescence, cardiovascular ageing, exceptional longevity | Deep-learning model trained on inflammatory protein patterns; feature attribution to identify key drivers (e.g., CXCL9) | Establishes a deep-learning inflammatory ageing clock that tracks multimorbidity, frailty, and vascular ageing; demonstrates that CXCL9-driven vascular inflammation is a modifiable axis of inflammaging with mechanistic validation. |
| Immune Risk Profile (IRP)—Swedish NONA/OCTO studies [106] | Conventional immunophenotyping: CD4/CD8 ratio, CD8 + CD28– T-cells, naïve/memory subsets, CMV serostatus | Very old Swedish cohorts (OCTO/NONA), individuals in their 80 s–100 s | All-cause mortality and frailty in very old adults | Rule-based composite risk profile (CD4/CD8 < 1, expanded CD8 + CD28–, CMV+) | Classical immune risk score linking T-cell compartment remodelling and CMV-driven immunosenescence to short-term mortality; centenarians lacking the IRP suggest “escaped” immune risk despite extreme age. |
| IgG glycan “glycan age” clock [107] | LC-MS-based profiling of IgG N-glycans; 24 glycan traits summarising Fc glycosylation | >5000 individuals from multiple population cohorts | Chronological age, cardiometabolic risk and lifestyle factors; “glycan age” acceleration | Linear models and age-prediction regression using IgG glycan traits | Demonstrates that IgG glycan patterns constitute a sensitive biomarker of biological and chronological age; links pro-inflammatory glycan shifts to cardiometabolic risk, positioning humoral immune glycome as an immune-centric ageing clock. |
| PBMC scRNA-seq ageing clocks [73] | Single-cell RNA-seq of PBMCs; cell-type composition and gene expression; multi-cohort integration with supercentenarians | Multi-cohort ageing series plus 7 supercentenarians; adult age range plus extreme longevity | Cell composition and transcriptomic ageing clocks at a single-cell resolution; supercentenarian “biological age delay” | Partial least squares regression on cell-type composition; gene-expression-based clocks within major immune lineages | Constructs human PBMC single-cell immune ageing clocks, revealing that supercentenarians exhibit delayed immune ageing and a “ribosome–inflammation” equilibrium; highlights cell-type-specific transcriptional hallmarks of slow immune ageing. |
| sc-ImmuAging—cell-type-specific immune ageing clocks [108] | PBMC scRNA-seq (five large human cohorts); gene-level features in CD4, CD8, B-, NK-cells, and monocytes | 1081 healthy European adults (18–97 years), plus external validation cohorts, COVID-19 and BCG vaccination cohorts | Cell-type-specific transcriptomic immune age; age acceleration/rejuvenation during infection and vaccination | Feature selection (correlation, mutual information, MIRA) + LASSO, random forest and PointNet deep-learning; separate clocks per cell type | Establishes robust cell-type-specific single-cell immune ageing clocks, reveals COVID-19-induced age acceleration (especially in monocytes) and identifies CD8+ T-cell age rejuvenation in individuals with strong interferon signatures after BCG vaccination. |
| Methodological Issue | Tool/Method Category (Representative Tools) | Typical Use Scenarios | Strengths | Key Limitations/Risks (Explicitly Named in the Review) | Representative References (SCI/High-Impact Preferred) |
|---|---|---|---|---|---|
| Batch effects/platform heterogeneity | Empirical Bayes batch correction (ComBat) | Pre-integration correction for “sample-by-feature” matrices in bulk transcriptomics/proteomics/methylation across cohorts | Mature and widely validated; transparent statistical assumptions | Strong dependence on correct specification of batch and covariates; when true age-related biology is entangled with batch, genuine signals may be attenuated (“over-correction”); new external samples often require additional handling | Johnson et al., Biostatistics 2007 [84] |
| Batch effects/platform heterogeneity | Latent factor/surrogate-variable adjustment (SVA) | Unknown or incompletely annotated technical variation | Captures unobserved systematic noise and improves robustness | Can remove biology correlated with phenotype/age; requires careful design matrix specification | Leek et al., Bioinformatics 2012 [85] |
| Batch effects/platform heterogeneity | Removal of unwanted variation (RUV/RUVSeq) | RNA-seq with ERCC spike-ins, technical replicates, or definable negative control genes | Leverages controls; often more stable than naïve regression-only approaches | Mis-specified control sets introduce bias; risk of over-correcting genuine biological gradients | Risso et al., Nature Biotechnology 2014 [120] |
| Batch effects/platform heterogeneity (deployment-oriented) | Prediction-focused batch adjustment (fSVA) | Real-world deployment with single-sample or small-batch prediction (clinical/translational settings) | “Freezes” latent factors learned from training data for new-sample adjustment; closer to actual prediction pipelines | Depends on representativeness of the training cohort; limited benefit under strong target-domain drift | Parker et al., PeerJ 2014 [121] |
| Batch effects/platform heterogeneity (cytometry) | Control-based normalisation (CytoNorm) | Multi-batch flow/mass cytometry with instrument drift; bridge/control samples available | Highly practical for cytometry drift; emphasises control consistency | Requires high-quality controls and consistent gating/labelling; control design errors propagate bias | Van Gassen et al., Cytometry Part A 2020 [87] |
| Compositional confounding (bulk “mixtures”) | Partial deconvolution: proportion estimation (CIBERSORT) | Bulk tissue/whole blood/PBMC transcriptomics: estimate immune cell fractions | Classic, interpretable, broadly used | Limited transferability of signature matrices; unstable for rare subsets or strong inflammatory/therapy-driven compositional shifts | Newman et al., Nat. Biotechnol. 2019 [122] |
| Compositional confounding (bulk + multi-donor references) | Multi-subject scRNA reference deconvolution (MuSiC) | Bulk RNA-seq with multi-donor scRNA references; cross-population/cross-cohort studies | Uses cross-donor stable markers; often more robust across populations | Biassed when reference and target tissue/state mismatch; sensitive to novel states/subtypes | Wang et al., Nature Communications 2019 [90] |
| Compositional confounding (bulk + scRNA reference) | Reference-based decomposition models (BisqueRNA) | Bulk RNA-seq with scRNA reference; relatively stable cell-type labels available | Clear engineering workflow; practical for routine cohorts | Depends on annotation and depth comparability; larger errors for rare populations | Jew et al., Nature Communications 2020 [118] |
| Compositional confounding (method selection) | Benchmarking and comparative evaluations | Supporting a “do not rely on a single algorithm” recommendation in the review | Provides empirical determinants: preprocessing, marker choice, composition, method sensitivity | Conclusions are tissue- and dataset-dependent; cannot be blindly generalised | Avila Cobos et al., Nature Communications 2020 [91] |
| Compositional confounding (epigenomics) | Cell-mixture adjustment (reference-based) (Houseman method) | Whole-blood DNA methylation (DNAm): mixture effects strongly distort age/disease signals | One of the standard approaches in DNAm mixture correction; can be cross-validated with cytometry | Bias under reference mismatch across populations/platforms; incomplete reference panels leave residual confounding | Houseman et al., BMC Bioinformatics 2012 [18] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Yu, G.; Shao, Z.; Zhuo, J.; Chen, Z. Immune Ageing Clocks: A Methods-Oriented Review of Tasks, Modalities, Models, and Recalibration. Cells 2026, 15, 421. https://doi.org/10.3390/cells15050421
Yu G, Shao Z, Zhuo J, Chen Z. Immune Ageing Clocks: A Methods-Oriented Review of Tasks, Modalities, Models, and Recalibration. Cells. 2026; 15(5):421. https://doi.org/10.3390/cells15050421
Chicago/Turabian StyleYu, Gengchen, Zeyu Shao, Jingyu Zhuo, and Zixuan Chen. 2026. "Immune Ageing Clocks: A Methods-Oriented Review of Tasks, Modalities, Models, and Recalibration" Cells 15, no. 5: 421. https://doi.org/10.3390/cells15050421
APA StyleYu, G., Shao, Z., Zhuo, J., & Chen, Z. (2026). Immune Ageing Clocks: A Methods-Oriented Review of Tasks, Modalities, Models, and Recalibration. Cells, 15(5), 421. https://doi.org/10.3390/cells15050421






