Beyond Circadian Patterns: Mechanistic Insights into Sleep–Epilepsy Interactions and Therapeutic Implications
Abstract
1. Introduction: The Bidirectional Sleep–Epilepsy Nexus
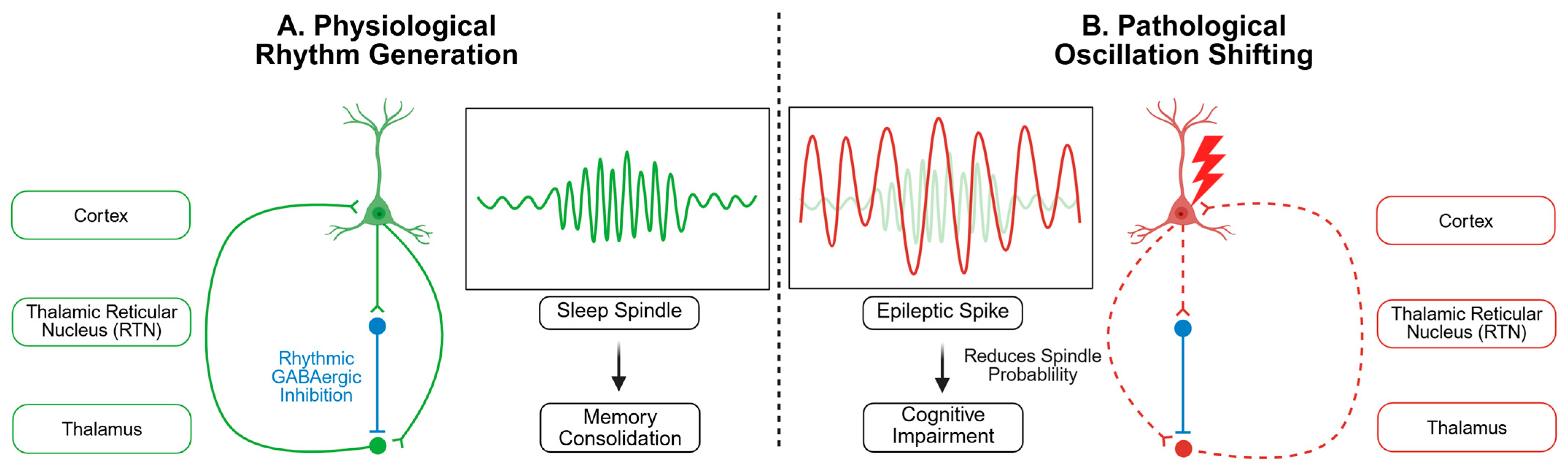
2. Thalamocortical Networks: The Battleground Between Spindles and Spikes
2.1. Sleep Spindles as Guardians of Memory Consolidation
2.2. Epileptic Spikes as Circuit Hijackers
2.3. TRN: The Critical Hub
3. Sleep State-Dependent Seizure Susceptibility: The REM Sleep Paradox
3.1. Protective Power of REM Sleep
3.2. GABA-Mediated Inhibition During REM Sleep
3.3. Sleep Architecture Disruption in Epilepsy
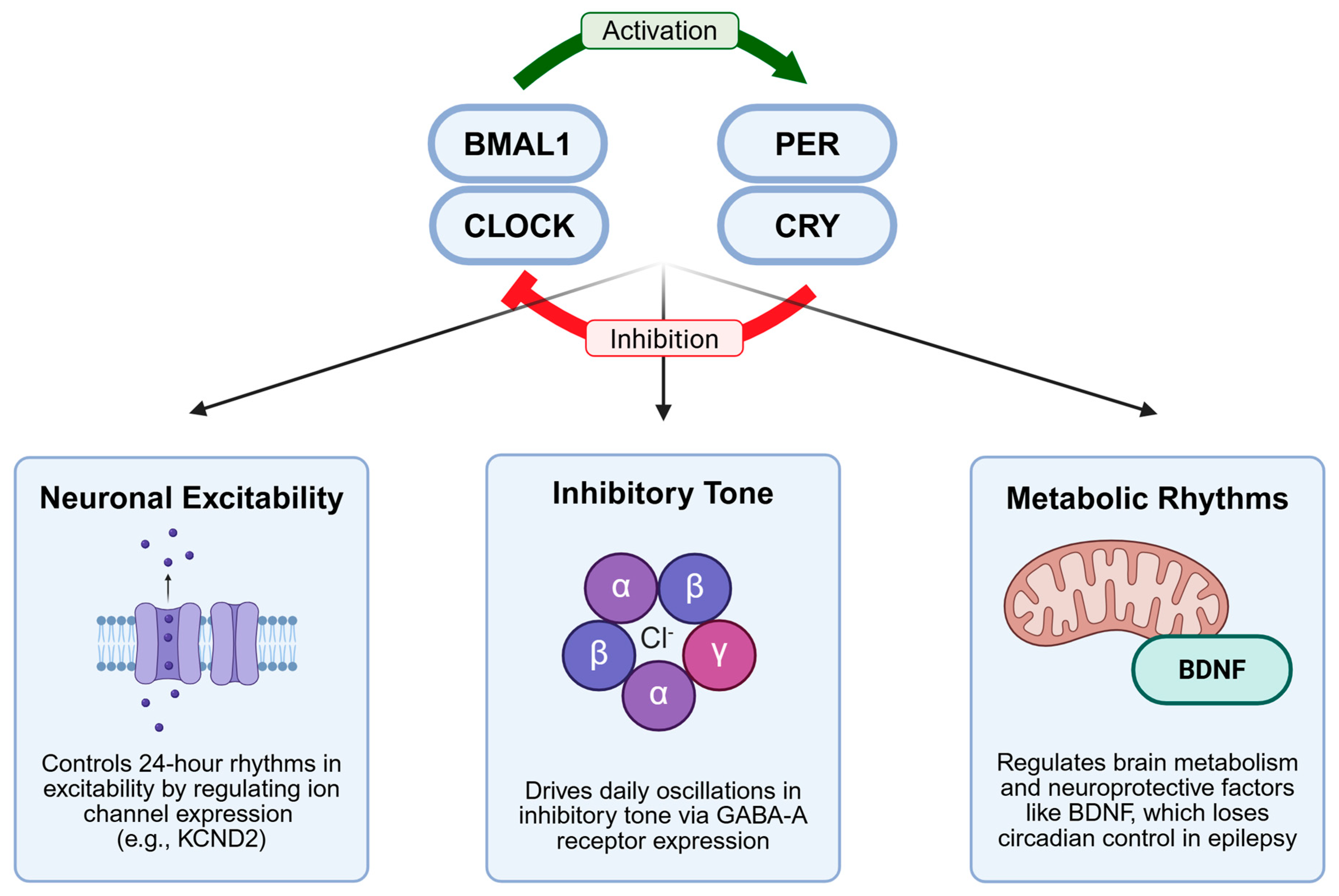
4. Molecular Clock Mechanisms in Epilepsy
4.1. Circadian Clock Genes as Seizure Modulators
4.2. Circadian Control of Neuronal Excitability
4.3. Metabolic Rhythms and Seizure Susceptibility
5. Clinical Syndrome Spotlight: Paradigmatic Disorders
5.1. SHE: A Model Disorder
5.2. Idiopathic Generalized Epilepsy and Sleep Disruption
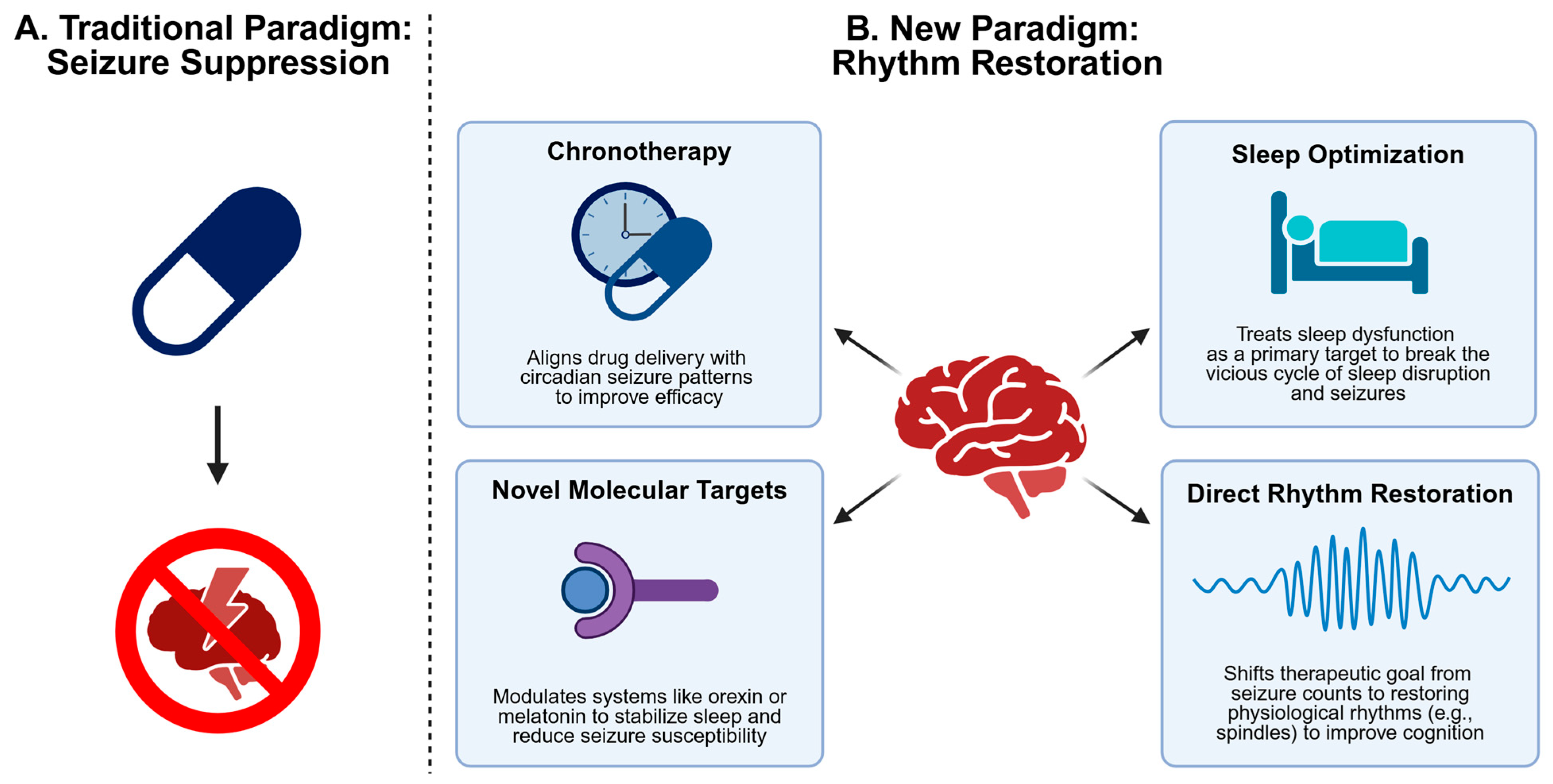
6. Therapeutic Innovations: Chronobiological Approaches
6.1. Precision Timing of Antiseizure Medications
6.2. Sleep Optimization Strategies
6.3. Novel Molecular Targets
6.4. Biomarker-Guided Interventions: The Immense Gap Between Data and the Clinic
- Sleep Spindles: While elegant measure of thalamocortical integrity [28], the use of spindles as clinical biomarkers is fundamentally a niche concept. It requires expert-led polysomnography, which is a costly and labor-intensive procedure that is unavailable to the vast majority of patients. Furthermore, the development of automated spindle-detection algorithms that are robust to artifacts and epileptiform activity endemic to clinical EEG remains a significant computational challenge. Without an inexpensive, scalable, and reliable method of detection, sleep spindles remain a powerful research tool but do not represent a common clinical tool.
- Circadian Biomarkers: Using wearables to guide chronotherapy is technologically feasible [108,109,110], but clinically and commercially stalled. The primary barrier is the lack of clear business cases for developing and validating these strategies. This requires pharmaceutical companies to fund complex and expensive trials to prove that the tailored dosing of their existing drugs is superior to standard regimens, a high-risk, low-reward proposition that few have pursued.
- Neurostimulation: Integrating chronobiological data into responsive neurostimulation devices is at the frontier of personalized therapy [111]. However, its relevance is restricted to a small fraction of the most refractory patients owing to the extreme cost and invasive nature of the therapy. The computational burden of analyzing continuous data streams from such devices is also immense, requiring a dedicated data science infrastructure. This will not be a scalable solution for epilepsy care in the near future.
7. Future Directions: Emerging Horizons
7.1. Personalized Chronotherapy
7.2. Sleep-Based Seizure Prediction
7.3. Gene Therapy: A Distant and Uncertain Horizon
8. Conclusions: From Seizure Suppression to Rhythm Restoration
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| REM | Rapid eye movement |
| TRN | Thalamic reticular nucleus |
| EEG | Electroencephalography |
| BDNF | Brain-derived neurotropic factor |
| SHE | Sleep-Related Hypermotor Epilepsy |
| JME | Juvenile Myoclonic Epilepsy |
References
- Huang, X.; Zhang, Y.; Lin, Q.; Huang, K.; Li, Y.; Liu, P.; Cao, D.; Li, W.; Li, W.; Zhou, D.; et al. The impact of sleep disorders on quality of life in patients with epilepsy. Seizure 2025, 129, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Badawy, R.A.; Curatolo, J.M.; Newton, M.; Berkovic, S.F.; Macdonell, R.A. Sleep deprivation increases cortical excitability in epilepsy: Syndrome-specific effects. Neurology 2006, 67, 1018–1022. [Google Scholar] [CrossRef]
- Wodeyar, A.; Chinappen, D.; Mylonas, D.; Baxter, B.; Manoach, D.S.; Eden, U.T.; Kramer, M.A.; Chu, C.J. Thalamic epileptic spikes disrupt sleep spindles in patients with epileptic encephalopathy. Brain 2024, 147, 2803–2816. [Google Scholar] [CrossRef] [PubMed]
- Kramer, M.A.; Stoyell, S.M.; Chinappen, D.; Ostrowski, L.M.; Spencer, E.R.; Morgan, A.K.; Emerton, B.C.; Jing, J.; Westover, M.B.; Eden, U.T.; et al. Focal Sleep Spindle Deficits Reveal Focal Thalamocortical Dysfunction and Predict Cognitive Deficits in Sleep Activated Developmental Epilepsy. J. Neurosci. 2021, 41, 1816–1829. [Google Scholar] [CrossRef]
- Bernard, C.; Frauscher, B.; Gelinas, J.; Timofeev, I. Sleep, oscillations, and epilepsy. Epilepsia 2023, 64, S3–S12. [Google Scholar] [CrossRef]
- Iber, C. The AASM Manual for the Scoring of Sleep and Associated Events: Rules, Terminology, and Technical Specification; American Academy of Sleep Medicine: Darien, IL, USA, 2007. [Google Scholar]
- Fernandez, L.M.J.; Luthi, A. Sleep Spindles: Mechanisms and Functions. Physiol. Rev. 2020, 100, 805–868. [Google Scholar] [CrossRef]
- Schabus, M.; Hodlmoser, K.; Gruber, G.; Sauter, C.; Anderer, P.; Klosch, G.; Parapatics, S.; Saletu, B.; Klimesch, W.; Zeitlhofer, J. Sleep spindle-related activity in the human EEG and its relation to general cognitive and learning abilities. Eur. J. Neurosci. 2006, 23, 1738–1746. [Google Scholar] [CrossRef]
- Mak-McCully, R.A.; Deiss, S.R.; Rosen, B.Q.; Jung, K.-Y.; Sejnowski, T.J.; Bastuji, H.; Rey, M.; Cash, S.S.; Bazhenov, M.; Halgren, E. Synchronization of isolated downstates (K-complexes) may be caused by cortically-induced disruption of thalamic spindling. PLoS Comput. Biol. 2014, 10, e1003855. [Google Scholar] [CrossRef]
- Koupparis, A.M.; Kokkinos, V.; Kostopoulos, G.K. Spindle power is not affected after spontaneous K-complexes during human NREM sleep. PLoS ONE 2013, 8, e54343. [Google Scholar] [CrossRef]
- Steriade, M.; McCormick, D.A.; Sejnowski, T.J. Thalamocortical oscillations in the sleeping and aroused brain. Science 1993, 262, 679–685. [Google Scholar] [CrossRef] [PubMed]
- Baran, B.; Karahanoglu, F.I.; Mylonas, D.; Demanuele, C.; Vangel, M.; Stickgold, R.; Anticevic, A.; Manoach, D.S. Increased Thalamocortical Connectivity in Schizophrenia Correlates With Sleep Spindle Deficits: Evidence for a Common Pathophysiology. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 2019, 4, 706–714. [Google Scholar] [CrossRef] [PubMed]
- Kwon, H.; Chinappen, D.M.; Kinard, E.A.; Goodman, S.K.; Huang, J.F.; Berja, E.D.; Walsh, K.G.; Shi, W.; Manoach, D.S.; Kramer, M.A.; et al. Association of Sleep Spindle Rate With Memory Consolidation in Children With Rolandic Epilepsy. Neurology 2025, 104, e210232. [Google Scholar] [CrossRef] [PubMed]
- Sheybani, L.; Megevand, P.; Roehri, N.; Spinelli, L.; Kleinschmidt, A.; van Mierlo, P.; Seeck, M.; Vulliemoz, S. Asymmetry of sleep electrophysiological markers in patients with focal epilepsy. Brain Commun. 2023, 5, fcad161. [Google Scholar] [CrossRef]
- Chen, P.; Hao, C.; Ma, N. Sleep spindles consolidate declarative memory with tags: A meta-analysis of adult data. J. Pac. Rim Psychol. 2024, 18. [Google Scholar] [CrossRef]
- Kumral, D.; Matzerath, A.; Leonhart, R.; Schonauer, M. Spindle-dependent memory consolidation in healthy adults: A meta-analysis. Neuropsychologia 2023, 189, 108661. [Google Scholar] [CrossRef]
- Blanco-Duque, C.; Bond, S.A.; Krone, L.B.; Dufour, J.P.; Gillen, E.C.P.; Purple, R.J.; Kahn, M.C.; Bannerman, D.M.; Mann, E.O.; Achermann, P.; et al. Oscillatory-Quality of sleep spindles links brain state with sleep regulation and function. Sci. Adv. 2024, 10, eadn6247. [Google Scholar] [CrossRef] [PubMed]
- Boutin, A.; Gabitov, E.; Pinsard, B.; Bore, A.; Carrier, J.; Doyon, J. Temporal cluster-based organization of sleep spindles underlies motor memory consolidation. Proc. Biol. Sci. 2024, 291, 20231408. [Google Scholar] [CrossRef]
- Gervan, P.; Bocskai, G.; Berencsi, A.; Gombos, F.; Kovacs, I. Visual Training Induced Temporo-Occipital Fast Sleep Spindle Clustering in Humans Revealed by Full-Night HD-EEG Recordings. J. Sleep Res. 2025, e70068. [Google Scholar] [CrossRef]
- Lahlou, S.; Kaminska, M.; Doyon, J.; Carrier, J.; Sharp, M. Sleep spindle density and temporal clustering are associated with sleep-dependent memory consolidation in Parkinson’s disease. J. Clin. Sleep Med. 2024, 20, 1153–1162. [Google Scholar] [CrossRef]
- Staresina, B.P.; Bergmann, T.O.; Bonnefond, M.; van der Meij, R.; Jensen, O.; Deuker, L.; Elger, C.E.; Axmacher, N.; Fell, J. Hierarchical nesting of slow oscillations, spindles and ripples in the human hippocampus during sleep. Nat. Neurosci. 2015, 18, 1679–1686. [Google Scholar] [CrossRef]
- Basha, D.; Azarmehri, A.; Proulx, E.; Chauvette, S.; Ghorbani, M.; Timofeev, I. The reuniens nucleus of the thalamus facilitates hippocampo-cortical dialogue during sleep. eLife 2025, 12, RP90826. [Google Scholar] [CrossRef]
- Leresche, N.; Lambert, R.C.; Errington, A.C.; Crunelli, V. From sleep spindles of natural sleep to spike and wave discharges of typical absence seizures: Is the hypothesis still valid? Pflugers Arch. 2011, 463, 201–212. [Google Scholar] [CrossRef]
- Blumenfeld, H. Cellular and Network Mechanisms of Spike-Wave Seizures. Epilepsia 2005, 46, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Kostopoulos, G.K. Spike-and-wave discharges of absence seizures as a transformation of sleep spindles: The continuing development of a hypothesis. Clin. Neurophysiol. 2000, 111, S27–S38. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Xin, M.; Song, G.; Liang, J. Childhood absence epilepsy patients with cognitive impairment have decreased sleep spindle density. Sleep Med. 2023, 103, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Lehner, J.; Frueh, J.S.; Datta, A.N. Sleep quality and architecture in Idiopathic generalized epilepsy: A systematic review and meta-analysis. Sleep Med. Rev. 2022, 65, 101689. [Google Scholar] [CrossRef]
- McLaren, J.R.; Luo, Y.; Kwon, H.; Shi, W.; Kramer, M.A.; Chu, C.J. Preliminary evidence of a relationship between sleep spindles and treatment response in epileptic encephalopathy. Ann. Clin. Transl. Neurol. 2023, 10, 1513–1524. [Google Scholar] [CrossRef]
- Huang, Y.; Liu, Y.; Song, W.; Liu, Y.; Wang, X.; Han, J.; Ye, J.; Han, H.; Wang, L.; Li, J.; et al. Assessment of Cognitive Function with Sleep Spindle Characteristics in Adults with Epilepsy. Neural Plast. 2023, 2023, 7768980. [Google Scholar] [CrossRef]
- Schiller, K.; Avigdor, T.; Abdallah, C.; Sziklas, V.; Crane, J.; Stefani, A.; Peter-Derex, L.; Frauscher, B. Focal epilepsy disrupts spindle structure and function. Sci. Rep. 2022, 12, 11137. [Google Scholar] [CrossRef]
- Halasz, P.; Szucs, A. Sleep and Epilepsy Link by Plasticity. Front. Neurol. 2020, 11, 911. [Google Scholar] [CrossRef]
- Castelnovo, A.; D’Agostino, A.; Mayeli, A.; Albantakis, L.; Tononi, G.; Ferrarelli, F. Sleep spindle abnormalities as neurophysiological biomarkers of Schizophrenia spectrum disorders: From cellular mechanisms and neural circuits to clinical implications. Biol. Psychiatry, 2025; Online ahead of print. [Google Scholar]
- Li, Q.; Westover, M.B.; Zhang, R.; Chu, C.J. Computational Evidence for a Competitive Thalamocortical Model of Spikes and Spindle Activity in Rolandic Epilepsy. Front. Comput. Neurosci. 2021, 15, 680549. [Google Scholar] [CrossRef] [PubMed]
- Ni, K.M.; Hou, X.J.; Yang, C.H.; Dong, P.; Li, Y.; Zhang, Y.; Jiang, P.; Berg, D.K.; Duan, S.; Li, X.-M. Selectively driving cholinergic fibers optically in the thalamic reticular nucleus promotes sleep. eLife 2016, 5, e10382. [Google Scholar] [CrossRef]
- Lewis, L.D.; Voigts, J.; Flores, F.J.; Schmitt, L.I.; A Wilson, M.; Halassa, M.M.; Brown, E.N. Thalamic reticular nucleus induces fast and local modulation of arousal state. eLife 2015, 4, e08760. [Google Scholar] [CrossRef]
- Ng, M.; Pavlova, M. Why are seizures rare in rapid eye movement sleep? Review of the frequency of seizures in different sleep stages. Epilepsy Res. Treat. 2013, 2013, 932790. [Google Scholar] [CrossRef]
- Luppi, P.-H.; Clément, O.; Fort, P. Paradoxical (REM) sleep genesis by the brainstem is under hypothalamic control. Curr. Opin. Neurobiol. 2013, 23, 786–792. [Google Scholar] [CrossRef] [PubMed]
- Shouse, M.N.; da Silva, A.M.; Sammaritano, M. Circadian rhythm, sleep, and epilepsy. J. Clin. Neurophysiol. 1996, 13, 32–50. [Google Scholar] [CrossRef]
- Brooks, P.L.; Peever, J.H. Identification of the Transmitter and Receptor Mechanisms Responsible for REM Sleep Paralysis. J. Neurosci. 2012, 32, 9785–9795. [Google Scholar] [CrossRef] [PubMed]
- Bryson, A.; Reid, C.; Petrou, S. Fundamental Neurochemistry Review: GABA(A) receptor neurotransmission and epilepsy: Principles, disease mechanisms and pharmacotherapy. J. Neurochem. 2023, 165, 6–28. [Google Scholar] [CrossRef]
- Brickley, S.G.; Mody, I. Extrasynaptic GABAA Receptors: Their Function in the CNS and Implications for Disease. Neuron 2012, 73, 23–34. [Google Scholar] [CrossRef]
- Perucca, E.; Brodie, M.J.; Kwan, P.; Tomson, T. 30 years of second-generation antiseizure medications: Impact and future perspectives. Lancet Neurol. 2020, 19, 544–556. [Google Scholar] [CrossRef]
- Andrew, P.M.; MacMahon, J.A.; Liu, X.; Saito, N.H.; Berger, K.E.; Morgan, J.E.; Dhir, A.; Harvey, D.J.; McCarren, H.S.; Rogawski, M.A. Allopregnanolone as an Adjunct Therapy to Midazolam is More Effective Than Midazolam Alone in Suppressing Soman-Induced Status Epilepticus in Male Rats. CNS Neurosci. Ther. 2025, 31, e70215. [Google Scholar] [CrossRef] [PubMed]
- Frauscher, B.; Iranzo, A.; Gaig, C.; Gschliesser, V.; Guaita, M.; Raffelseder, V.; Ehrmann, L.; Sola, N.; Salamero, M.; Tolosa, E.; et al. Normative EMG values during REM sleep for the diagnosis of REM sleep behavior disorder. Sleep 2012, 35, 835–847. [Google Scholar] [CrossRef] [PubMed]
- Yeh, W.C.; Lin, H.J.; Li, Y.S.; Chien, C.F.; Wu, M.N.; Liou, L.M.; Hsieh, C.F.; Hsu, C.Y. Rapid eye movement sleep reduction in patients with epilepsy: A systematic review and meta-analysis. Seizure 2022, 96, 46–58. [Google Scholar] [CrossRef]
- Calvello, C.; Fernandes, M.; Lupo, C.; Maramieri, E.; Placidi, F.; Izzi, F.; Castelli, A.; Pagano, A.; Mercuri, N.B.; Liguori, C. Sleep architecture in drug-naïve adult patients with epilepsy: Comparison between focal and generalized epilepsy. Epilepsia Open 2023, 8, 165–172. [Google Scholar] [CrossRef]
- Ho, A.; Hannan, S.; Thomas, J.; Avigdor, T.; Abdallah, C.; Dubeau, F.; Gotman, J.; Frauscher, B. Rapid eye movement sleep affects interictal epileptic activity differently in mesiotemporal and neocortical areas. Epilepsia 2023, 64, 3036–3048. [Google Scholar] [CrossRef]
- Sadak, U.; Honrath, P.; Ermis, U.; Heckelmann, J.; Meyer, T.; Weber, Y.; Wolking, S. Reduced REM sleep: A potential biomarker for epilepsy—A retrospective case-control study. Seizure 2022, 98, 27–33. [Google Scholar] [CrossRef]
- Kilgore-Gomez, A.; Norato, G.; Theodore, W.H.; Inati, S.K.; Rahman, S.A. Sleep physiology in patients with epilepsy: Influence of seizures on rapid eye movement (REM) latency and REM duration. Epilepsia 2024, 65, 995–1005. [Google Scholar] [CrossRef]
- Liguori, C.; Toledo, M.; Kothare, S. Effects of anti-seizure medications on sleep architecture and daytime sleepiness in patients with epilepsy: A literature review. Sleep Med. Rev. 2021, 60, 101559. [Google Scholar] [CrossRef]
- Re, C.J.; Batterman, A.I.; Gerstner, J.R.; Buono, R.J.; Ferraro, T.N. The molecular genetic interaction between circadian rhythms and susceptibility to seizures and epilepsy. Front. Neurol. 2020, 11, 520. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Liu, Y.; Liu, L.; Meng, Q.; Du, C.; Li, K.; Dong, S.; Zhang, Y.; Li, H.; Zhang, H. Decreased expression of the clock gene Bmal1 is involved in the pathogenesis of temporal lobe epilepsy. Mol. Brain 2021, 14, 113. [Google Scholar] [CrossRef]
- Martinowich, K.; Manji, H.; Lu, B. New insights into BDNF function in depression and anxiety. Nat. Neurosci. 2007, 10, 1089–1093. [Google Scholar] [CrossRef] [PubMed]
- Matos, H.C.; Koike, B.D.V.; Pereira, W.D.S.; de Andrade, T.G.; Castro, O.W.; Duzzioni, M.; Kodali, M.; Leite, J.P.; Shetty, A.K.; Gitai, D.L.G. Rhythms of Core Clock Genes and Spontaneous Locomotor Activity in Post-Status Epilepticus Model of Mesial Temporal Lobe Epilepsy. Front. Neurol. 2018, 9, 632. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Lahens, N.F.; Ballance, H.I.; Hughes, M.E.; Hogenesch, J.B. A circadian gene expression atlas in mammals: Implications for biology and medicine. Proc. Natl. Acad. Sci. USA 2014, 111, 16219–16224. [Google Scholar] [CrossRef]
- Granados-Fuentes, D.; Hermanstyne, T.O.; Carrasquillo, Y.; Nerbonne, J.M.; Herzog, E.D. IA Channels Encoded by Kv1.4 and Kv4.2 Regulate Circadian Period of PER2 Expression in the Suprachiasmatic Nucleus. J. Biol. Rhythm. 2015, 30, 396–407. [Google Scholar] [CrossRef] [PubMed]
- Walton, J.C.; McNeill, J.K.t.; Oliver, K.A.; Albers, H.E. Temporal Regulation of GABA(A) Receptor Subunit Expression: Role in Synaptic and Extrasynaptic Communication in the Suprachiasmatic Nucleus. eNeuro 2017, 4, ENEURO.0352-16.2017. [Google Scholar] [CrossRef]
- Wang, L.M.; Dragich, J.M.; Kudo, T.; Odom, I.H.; Welsh, D.K.; O’Dell, T.J.; Colwell, C.S. Expression of the circadian clock gene Period2 in the hippocampus: Possible implications for synaptic plasticity and learned behaviour. ASN Neuro 2009, 1, e00012. [Google Scholar] [CrossRef]
- Magnone, M.C.; Langmesser, S.; Bezdek, A.C.; Tallone, T.; Rusconi, S.; Albrecht, U. The Mammalian circadian clock gene per2 modulates cell death in response to oxidative stress. Front. Neurol. 2014, 5, 289. [Google Scholar] [CrossRef]
- Ohdo, S. Chronopharmacology focused on biological clock. Drug Metab. Pharmacokinet. 2007, 22, 3–14. [Google Scholar] [CrossRef]
- Tinuper, P.; Bisulli, F.; Cross, J.H.; Hesdorffer, D.; Kahane, P.; Nobili, L.; Provini, F.; Scheffer, I.E.; Tassi, L.; Vignatelli, L.; et al. Definition and diagnostic criteria of sleep-related hypermotor epilepsy. Neurology 2016, 86, 1834–1842. [Google Scholar] [CrossRef]
- Nielsen, T.O.; Herlin, M.K.; Linnet, K.M.; Beniczky, S.; Sommerlund, M.; Granild-Jensen, J.B.; Gregersen, P.A. Autosomal dominant sleep-related hypermotor epilepsy caused by a previously unreported CHRNA4 variant. Eur. J. Med. Genet. 2022, 65, 104444. [Google Scholar] [CrossRef]
- Brodtkorb, E.; Myren-Svelstad, S.; Knudsen-Baas, K.M.; Nakken, K.O.; Spigset, O. Precision treatment with nicotine in autosomal dominant sleep-related hypermotor epilepsy (ADSHE): An observational study of clinical outcome and serum cotinine levels in 17 patients. Epilepsy Res. 2021, 178, 106792. [Google Scholar] [CrossRef]
- Kasteleijn-Nolst Trenite, D.; Rubboli, G.; Hirsch, E.; Martins da Silva, A.; Seri, S.; Wilkins, A.; Parra, J.; Covanis, A.; Elia, M.; Capovilla, G.; et al. Methodology of photic stimulation revisited: Updated European algorithm for visual stimulation in the EEG laboratory. Epilepsia 2012, 53, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Yegnanarayan, R.; Mahesh, S.D.; Sangle, S. Chronotherapeutic dose schedule of phenytoin and carbamazepine in epileptic patients. Chronobiol. Int. 2006, 23, 1035–1046. [Google Scholar] [CrossRef] [PubMed]
- Ramgopal, S.; Thome-Souza, S.; Loddenkemper, T. Chronopharmacology of anti-convulsive therapy. Curr. Neurol. Neurosci. Rep. 2013, 13, 339. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhuo, Z.; Wang, H. Epilepsy, gut microbiota, and circadian rhythm. Front. Neurol. 2023, 14, 1157358. [Google Scholar] [CrossRef] [PubMed]
- Guilhoto, L.M.; Loddenkemper, T.; Vendrame, M.; Bergin, A.; Bourgeois, B.F.; Kothare, S.V. Higher evening antiepileptic drug dose for nocturnal and early-morning seizures. Epilepsy Behav. 2011, 20, 334–337. [Google Scholar] [CrossRef]
- Cheng, N.; Liu, J.; Kan, X.; Wang, J.; Hui, Z.; Chen, J. Optimizing epilepsy treatment: The impact of circadian rhythms and medication timing on conversion rates and survival. QJM 2025, 118, 33–41. [Google Scholar] [CrossRef]
- Wu, D.; Fei, F.; Zhang, Q.; Wang, X.; Gong, Y.; Chen, X.; Zheng, Y.; Tan, B.; Xu, C.; Xie, H.; et al. Nanoengineered on-demand drug delivery system improves efficacy of pharmacotherapy for epilepsy. Sci. Adv. 2022, 8, eabm3381. [Google Scholar] [CrossRef]
- Wheless, J.W.; Phelps, S.J. A Clinician’s Guide to Oral Extended-Release Drug Delivery Systems in Epilepsy. J. Pediatr. Pharmacol. Ther. 2018, 23, 277–292. [Google Scholar] [CrossRef]
- Fountain, N.B.; Quigg, M.; Murchison, C.F.; Carrazana, E.; Rabinowicz, A.L. Analysis of seizure-cluster circadian periodicity from a long-term, open-label safety study of diazepam nasal spray. Epilepsia 2024, 65, 920–928. [Google Scholar] [CrossRef]
- Grigg-Damberger, M.; Foldvary-Schaefer, N. Bidirectional relationships of sleep and epilepsy in adults with epilepsy. Epilepsy Behav. 2021, 116, 107735. [Google Scholar] [CrossRef]
- Moore, J.L.; Carvalho, D.Z.; St Louis, E.K.; Bazil, C. Sleep and Epilepsy: A Focused Review of Pathophysiology, Clinical Syndromes, Co-morbidities, and Therapy. Neurotherapeutics 2021, 18, 170–180. [Google Scholar] [CrossRef]
- Malow, B.; Passaro, E.; Milling, C.; Minecan, D.; Levy, K. Sleep deprivation does not affect seizure frequency during inpatient video-EEG monitoring. Neurology 2002, 59, 1371–1374. [Google Scholar] [CrossRef]
- Ismayilova, V.; Demir, A.U.; Tezer, F.I. Subjective sleep disturbance in epilepsy patients at an outpatient clinic: A questionnaire-based study on prevalence. Epilepsy Res. 2015, 115, 119–125. [Google Scholar] [CrossRef]
- Zanzmera, P.; Shukla, G.; Gupta, A.; Singh, H.; Goyal, V.; Srivastava, A.; Behari, M. Markedly disturbed sleep in medically refractory compared to controlled epilepsy—A clinical and polysomnography study. Seizure 2012, 21, 487–490. [Google Scholar] [CrossRef] [PubMed]
- Vendrame, M.; Auerbach, S.; Loddenkemper, T.; Kothare, S.; Montouris, G. Effect of continuous positive airway pressure treatment on seizure control in patients with obstructive sleep apnea and epilepsy. Epilepsia 2011, 52, e168–e171. [Google Scholar] [CrossRef] [PubMed]
- Sudbrack-Oliveira, P.; Lima Najar, L.; Foldvary-Schaefer, N.; da Mota Gomes, M. Sleep architecture in adults with epilepsy: A systematic review. Sleep Med. 2019, 53, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Bazil, C.W.; Castro, L.H.; Walczak, T.S. Reduction of rapid eye movement sleep by diurnal and nocturnal seizures in temporal lobe epilepsy. Arch. Neurol. 2000, 57, 363–368. [Google Scholar] [CrossRef]
- Malow, B.A.; Weatherwax, K.J.; Chervin, R.D.; Hoban, T.F.; Marzec, M.L.; Martin, C.; Binns, L.A. Identification and treatment of obstructive sleep apnea in adults and children with epilepsy: A prospective pilot study. Sleep Med. 2003, 4, 509–515. [Google Scholar] [CrossRef]
- Jain, S.V.; Glauser, T.A. Effects of epilepsy treatments on sleep architecture and daytime sleepiness: An evidence-based review of objective sleep metrics. Epilepsia 2014, 55, 26–37. [Google Scholar] [CrossRef] [PubMed]
- Shvarts, V.; Chung, S. Epilepsy, antiseizure therapy, and sleep cycle parameters. Epilepsy Res. Treat. 2013, 2013, 670682. [Google Scholar] [CrossRef] [PubMed]
- Krutoshinskaya, Y.; Coulehan, K.; Pushchinska, G.; Spiegel, R. The reciprocal relationship between sleep and epilepsy. J. Pers. Med. 2024, 14, 118. [Google Scholar] [CrossRef] [PubMed]
- Berteotti, C.; Calvello, C.; Liguori, C. Role of the orexin system in the bidirectional relation between sleep and epilepsy: New chances for patients with epilepsy by the antagonism to orexin receptors? Epilepsia 2023, 64, 1991–2005. [Google Scholar] [CrossRef]
- Asadi, S.; Roohbakhsh, A.; Shamsizadeh, A.; Fereidoni, M.; Kordijaz, E.; Moghimi, A. The effect of intracerebroventricular administration of orexin receptor type 2 antagonist on pentylenetetrazol-induced kindled seizures and anxiety in rats. BMC Neurosci. 2018, 19, 49. [Google Scholar] [CrossRef]
- Sheibani, M.; Shayan, M.; Khalilzadeh, M.; Ghasemi, M.; Dehpour, A.R. Orexin receptor antagonists in the pathophysiology and treatment of sleep disorders and epilepsy. Neuropeptides 2023, 99, 102335. [Google Scholar] [CrossRef]
- Roundtree, H.M.; Simeone, T.A.; Johnson, C.; Matthews, S.A.; Samson, K.K.; Simeone, K.A. Orexin Receptor Antagonism Improves Sleep and Reduces Seizures in Kcna1-null Mice. Sleep 2016, 39, 357–368. [Google Scholar] [CrossRef]
- Samzadeh, M.; Papuc, E.; Furtak-Niczyporuk, M.; Rejdak, K. Decreased Cerebrospinal Fluid Orexin-A (Hypocretin-1) Concentrations in Patients after Generalized Convulsive Status Epilepticus. J. Clin. Med. 2020, 9, 3354. [Google Scholar] [CrossRef]
- Hoyer, D.; Jacobson, L.H. Orexin in sleep, addiction and more: Is the perfect insomnia drug at hand? Neuropeptides 2013, 47, 477–488. [Google Scholar] [CrossRef]
- Sun, Y.; Tisdale, R.K.; Kilduff, T.S. Hypocretin/Orexin Receptor Pharmacology and Sleep Phases. Front. Neurol. Neurosci. 2021, 45, 22–37. [Google Scholar] [CrossRef] [PubMed]
- Elkhayat, H.A.; Hassanein, S.M.; Tomoum, H.Y.; Abd-Elhamid, I.A.; Asaad, T.; Elwakkad, A.S. Melatonin and sleep-related problems in children with intractable epilepsy. Pediatr. Neurol. 2010, 42, 249–254. [Google Scholar] [CrossRef]
- Molina-Carballo, A.; Muñoz-Hoyos, A.; Reiter, R.J.; Sánchez-Forte, M.; Moreno-Madrid, F.; Rufo-Campos, M.; Molina-Font, J.A.; Acuna-Castroviejo, D. Utility of high doses of melatonin as adjunctive anticonvulsant therapy in a child with severe myoclonic epilepsy: Two years’ experience. J. Pineal Res. 1997, 23, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Goldberg-Stern, H.; Oren, H.; Peled, N.; Garty, B.Z. Effect of melatonin on seizure frequency in intractable epilepsy: A pilot study. J. Child. Neurol. 2012, 27, 1524–1528. [Google Scholar] [CrossRef]
- Gupta, M.; Aneja, S.; Kohli, K. Add-on melatonin improves sleep behavior in children with epilepsy: Randomized, double-blind, placebo-controlled trial. J. Child. Neurol. 2005, 20, 112–115. [Google Scholar] [CrossRef]
- Jain, S.V.; Horn, P.S.; Simakajornboon, N.; Beebe, D.W.; Holland, K.; Byars, A.W.; Glauser, T.A. Melatonin improves sleep in children with epilepsy: A randomized, double-blind, crossover study. Sleep Med. 2015, 16, 637–644. [Google Scholar] [CrossRef] [PubMed]
- Verma, N.; Maiti, R.; Mishra, B.R.; Jha, M.; Jena, M.; Mishra, A. Effect of add-on melatonin on seizure outcome, neuronal damage, oxidative stress, and quality of life in generalized epilepsy with generalized onset motor seizures in adults: A randomized controlled trial. J. Neurosci. Res. 2021, 99, 1618–1631. [Google Scholar] [CrossRef]
- Muppa, V.; Mahapatro, S.; Bhoi, S.; Satapathy, A.K.; Saini, L. Melatonin as an add-on anti-seizure medication in children with epilepsy: An open-label randomized controlled trial. J. Neurosci. Rural Pract. 2024, 15, 455–460. [Google Scholar] [CrossRef]
- Fauteck, J.; Schmidt, H.; Lerchl, A.; Kurlemann, G.; Wittkowski, W. Melatonin in epilepsy: First results of replacement therapy and first clinical results. Biol. Signals Recept. 1999, 8, 105–110. [Google Scholar] [CrossRef]
- Maghbooli, M.; Alyan NajafAbadi, S.; MalekMahmoudi, G.; Molseghi, M.H. Effect of add-on melatonin on seizure outcomes and quality of sleep in epilepsy with idiopathic generalized tonic-clonic seizures alone in adult patients: Cross-sectional, randomized, double-blind, placebo-controlled clinical trial. Brain Behav. 2023, 13, e2860. [Google Scholar] [CrossRef]
- Uberos, J.; Augustin-Morales, M.; Molina Carballo, A.; Florido, J.; Narbona, E.; Muñoz-Hoyos, A. Normalization of the sleep–wake pattern and melatonin and 6-sulphatoxy-melatonin levels after a therapeutic trial with melatonin in children with severe epilepsy. J. Pineal Res. 2011, 50, 192–196. [Google Scholar] [CrossRef]
- Peled, N.; Shorer, Z.; Peled, E.; Pillar, G. Melatonin effect on seizures in children with severe neurologic deficit disorders. Epilepsia 2001, 42, 1208–1210. [Google Scholar] [CrossRef]
- Vaitkevicius, H.; Ramsay, R.E.; Swisher, C.B.; Husain, A.M.; Aimetti, A.; Gasior, M. Intravenous ganaxolone for the treatment of refractory status epilepticus: Results from an open-label, dose-finding, phase 2 trial. Epilepsia 2022, 63, 2381–2391. [Google Scholar] [CrossRef]
- Richardson, R.J.; Petrou, S.; Bryson, A. Established and emerging GABA(A) receptor pharmacotherapy for epilepsy. Front. Pharmacol. 2024, 15, 1341472. [Google Scholar] [CrossRef]
- Chuang, S.-H.; Reddy, D.S. 3β-Methyl-neurosteroid analogs are preferential positive allosteric modulators and direct activators of extrasynaptic δ-subunit γ-aminobutyric acid type A receptors in the hippocampus dentate gyrus subfield. J. Pharmacol. Exp. Ther. 2018, 365, 583–601. [Google Scholar] [CrossRef]
- Reddy, D.S.; Carver, C.M.; Clossen, B.; Wu, X. Extrasynaptic gamma-aminobutyric acid type A receptor-mediated sex differences in the antiseizure activity of neurosteroids in status epilepticus and complex partial seizures. Epilepsia 2019, 60, 730–743. [Google Scholar] [CrossRef]
- Gasior, M.; Husain, A.; Barra, M.E.; Raja, S.M.; MacLeod, D.; Guptill, J.T.; Vaitkevicius, H.; Rybak, E. Intravenous Ganaxolone: Pharmacokinetics, Pharmacodynamics, Safety, and Tolerability in Healthy Adults. Clin. Pharmacol. Drug Dev. 2024, 13, 248–258. [Google Scholar] [CrossRef] [PubMed]
- Karoly, P.J.; Goldenholz, D.M.; Freestone, D.R.; Moss, R.E.; Grayden, D.B.; Theodore, W.H.; Cook, M.J. Circadian and circaseptan rhythms in human epilepsy: A retrospective cohort study. Lancet Neurol. 2018, 17, 977–985. [Google Scholar] [CrossRef]
- Baud, M.O.; Kleen, J.K.; Mirro, E.A.; Andrechak, J.C.; King-Stephens, D.; Chang, E.F.; Rao, V.R. Multi-day rhythms modulate seizure risk in epilepsy. Nat. Commun. 2018, 9, 88. [Google Scholar] [CrossRef] [PubMed]
- Karoly, P.J.; Ung, H.; Grayden, D.B.; Kuhlmann, L.; Leyde, K.; Cook, M.J.; Freestone, D.R. The circadian profile of epilepsy improves seizure forecasting. Brain 2017, 140, 2169–2182. [Google Scholar] [CrossRef] [PubMed]
- Charlebois, C.M.; Anderson, D.N.; Smith, E.H.; Davis, T.S.; Newman, B.J.; Peters, A.Y.; Arain, A.M.; Dorval, A.D.; Rolston, J.D.; Butson, C.R. Circadian changes in aperiodic activity are correlated with seizure reduction in patients with mesial temporal lobe epilepsy treated with responsive neurostimulation. Epilepsia 2024, 65, 1360–1373. [Google Scholar] [CrossRef]
- Biondi, A.; Santoro, V.; Viana, P.F.; Laiou, P.; Pal, D.K.; Bruno, E.; Richardson, M.P. Noninvasive mobile EEG as a tool for seizure monitoring and management: A systematic review. Epilepsia 2022, 63, 1041–1063. [Google Scholar] [CrossRef]
- Loddenkemper, T.; Lockley, S.W.; Kaleyias, J.; Kothare, S.V. Chronobiology of epilepsy: Diagnostic and therapeutic implications of chrono-epileptology. J. Clin. Neurophysiol. 2011, 28, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Rao, V.R.; Rolston, J.D. Unearthing the mechanisms of responsive neurostimulation for epilepsy. Commun. Med. 2023, 3, 166. [Google Scholar] [CrossRef] [PubMed]
- Proix, T.; Jirsa, V.K.; Bartolomei, F.; Guye, M.; Truccolo, W. Predicting the spatiotemporal diversity of seizure propagation and termination in human focal epilepsy. Nat. Commun. 2018, 9, 1088. [Google Scholar] [CrossRef]
- Tsiouris, K.; Pezoulas, V.C.; Zervakis, M.; Konitsiotis, S.; Koutsouris, D.D.; Fotiadis, D.I. A Long Short-Term Memory deep learning network for the prediction of epileptic seizures using EEG signals. Comput. Biol. Med. 2018, 99, 24–37. [Google Scholar] [CrossRef]
- Ferrara, R.; Giaquinto, M.; Percannella, G.; Rundo, L.; Saggese, A. Personalizing Seizure Detection for Individual Patients by Optimal Selection of EEG Signals. Sensors 2025, 25, 2715. [Google Scholar] [CrossRef]
- Cook, M.J.; O’Brien, T.J.; Berkovic, S.F.; Murphy, M.; Morokoff, A.; Fabinyi, G.; D’Souza, W.; Yerra, R.; Archer, J.; Litewka, L.; et al. Prediction of seizure likelihood with a long-term, implanted seizure advisory system in patients with drug-resistant epilepsy: A first-in-man study. Lancet Neurol. 2013, 12, 563–571. [Google Scholar] [CrossRef]
- Carpenter, J.C.; Lignani, G. Gene Editing and Modulation: The Holy Grail for the Genetic Epilepsies? Neurotherapeutics 2021, 18, 1515–1523. [Google Scholar] [CrossRef]
- Piguet, F.; de Saint Denis, T.; Audouard, E.; Beccaria, K.; Andre, A.; Wurtz, G.; Schatz, R.; Alves, S.; Sevin, C.; Zerah, M.; et al. The Challenge of Gene Therapy for Neurological Diseases: Strategies and Tools to Achieve Efficient Delivery to the Central Nervous System. Hum. Gene Ther. 2021, 32, 349–374. [Google Scholar] [CrossRef] [PubMed]
| Feature | Wakefulness | Non-REM Sleep | REM Sleep |
|---|---|---|---|
| Cortical EEG (Electroencephalography) Activity | Desynchronized, high frequency | Synchronized oscillations | Desynchronized, high-frequency (wakeful-like) |
| Dominant Neurotransmitters | High cholinergic and monoaminergic tone | Reduced cholinergic and monoaminergic activity | High cholinergic; noradrenergic and serotonergic neurons silent |
| Seizure Susceptibility | Relative seizure vulnerability | High; promotes seizure generation and propagation | Dramatically reduced; natural anticonvulsant state |
| Motor Activity | Normal muscle tone | Reduced muscle tone | Muscle atonia (sleep paralysis) |
| Reference | [37] | [36,37] | [38] |
| Gene | Normal Function in Circadian Rhythm | Dysregulation and Role in Epilepsy | Reference |
|---|---|---|---|
| BMAL1 | Core positive regulator; heterodimerizes with CLOCK to activate target gene transcription | Decreased in temporal lobe epilepsy. Knockout lowers seizure threshold; gene has direct protective role | [51,52] |
| CLOCK | Core positive regulator; heterodimerizes with BMAL1 to drive clock-controlled genes, including BDNF | Dysregulation disrupts CLOCK-BMAL1 complex, causing loss of circadian BDNF regulation in epilepsy | [51,53] |
| PER2 | Core negative regulator in transcriptional–translational feedback loop; inhibits CLOCK/BMAL1 activity | Dysregulated following status epilepticus. Knockout increases seizure susceptibility; overexpression is protective | [51,54] |
| CRY1/2 | Core negative regulators; inhibit CLOCK/BMAL1 activity with PER proteins in feedback loop | Dysregulation creates permissive environment for seizure generation and disrupts epileptic activity patterns | [51] |
| Syndrome | Key Clinical Features | Primary Sleep-Related Characteristics | Proposed Underlying Mechanism |
|---|---|---|---|
| Sleep-related Hypermotor Epilepsy (SHE) | Complex hypermotor seizures arising exclusively from sleep | Seizures emerge from Stage N2 non-REM sleep. Timing coincides with periods when sleep spindles are most prominent | Genetic mutations in nicotinic acetylcholine receptor subunits |
| Juvenile Myoclonic Epilepsy (JME) | Most common idiopathic generalized epilepsy syndrome; characterized by morning seizure clustering | Patients exhibit profound sleep architecture abnormalities, including reduced slow-wave sleep and fragmented sleep continuity. Seizures correlate with sleep-to-wake transition. Sleep deprivation is a potent seizure trigger | Fundamental alterations in sleep regulatory mechanisms |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kwon, K.; Lee, Y.; Kim, M.S. Beyond Circadian Patterns: Mechanistic Insights into Sleep–Epilepsy Interactions and Therapeutic Implications. Cells 2025, 14, 1331. https://doi.org/10.3390/cells14171331
Kwon K, Lee Y, Kim MS. Beyond Circadian Patterns: Mechanistic Insights into Sleep–Epilepsy Interactions and Therapeutic Implications. Cells. 2025; 14(17):1331. https://doi.org/10.3390/cells14171331
Chicago/Turabian StyleKwon, Kanghyun, Yoonsung Lee, and Man S. Kim. 2025. "Beyond Circadian Patterns: Mechanistic Insights into Sleep–Epilepsy Interactions and Therapeutic Implications" Cells 14, no. 17: 1331. https://doi.org/10.3390/cells14171331
APA StyleKwon, K., Lee, Y., & Kim, M. S. (2025). Beyond Circadian Patterns: Mechanistic Insights into Sleep–Epilepsy Interactions and Therapeutic Implications. Cells, 14(17), 1331. https://doi.org/10.3390/cells14171331






