Preclinical Study of Pain Neuropeptide Expression in Murine Sensory Neurons Induced by Irradiated Osteoclasts in the Context of Stereotactic Body Radiation Therapy
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Radiation Treatment
2.3. Sensory Neuronal Cultures from Mice Dorsal Root Ganglia (DRG)
2.4. Sensory Neuronal Cultures from Non-Human Primate (NHP) DRGs
2.5. Culturing and Differentiation of Osteoclast
2.6. Tartrate-Resistant Acid Phosphatase (TRAP) Staining and Image Analysis
2.7. Resorption Pit Assay for Osteoclast
2.8. Conditioned Media (C.M) Generation
2.9. RT-qPCR
2.10. Immunofluorescence Staining
2.11. Neurite Length Analysis
2.12. Statistical Analysis
3. Results
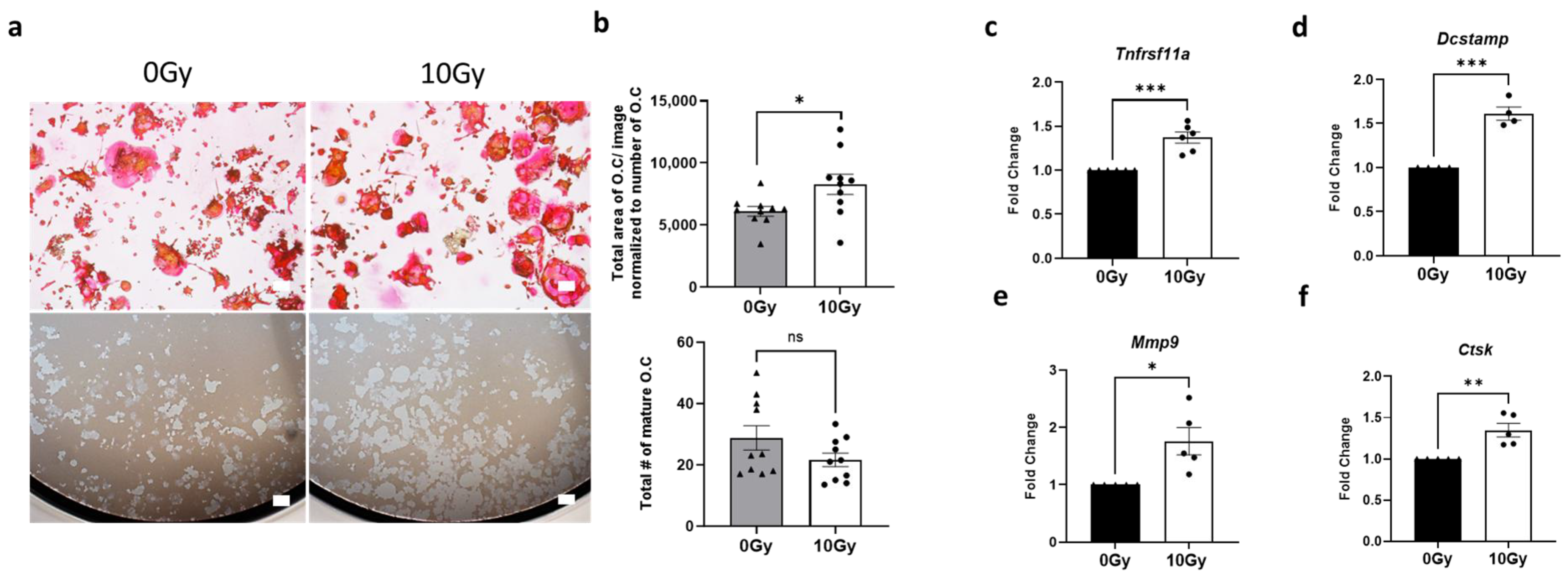
3.1. Impact of High-Dose Radiation on Osteoclast Maturation and Activity
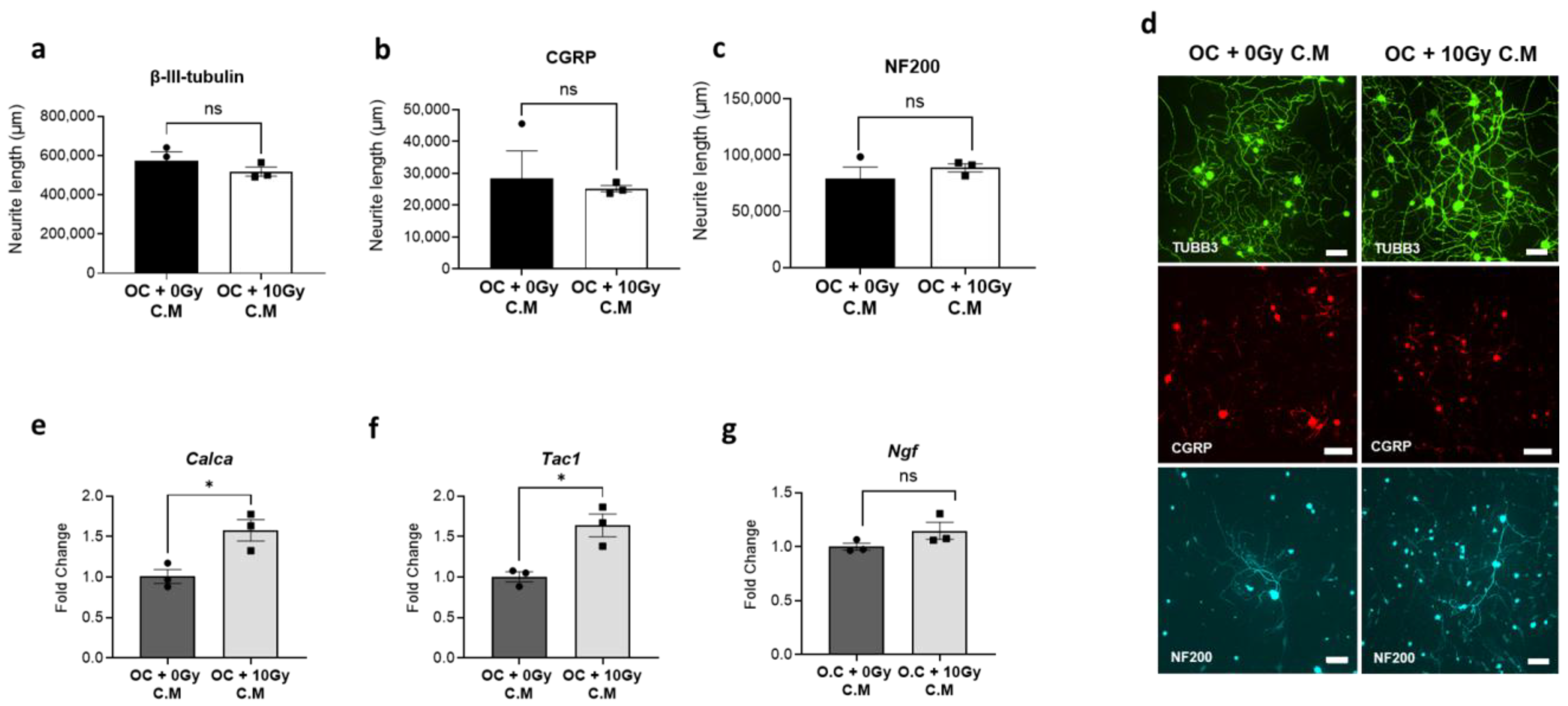
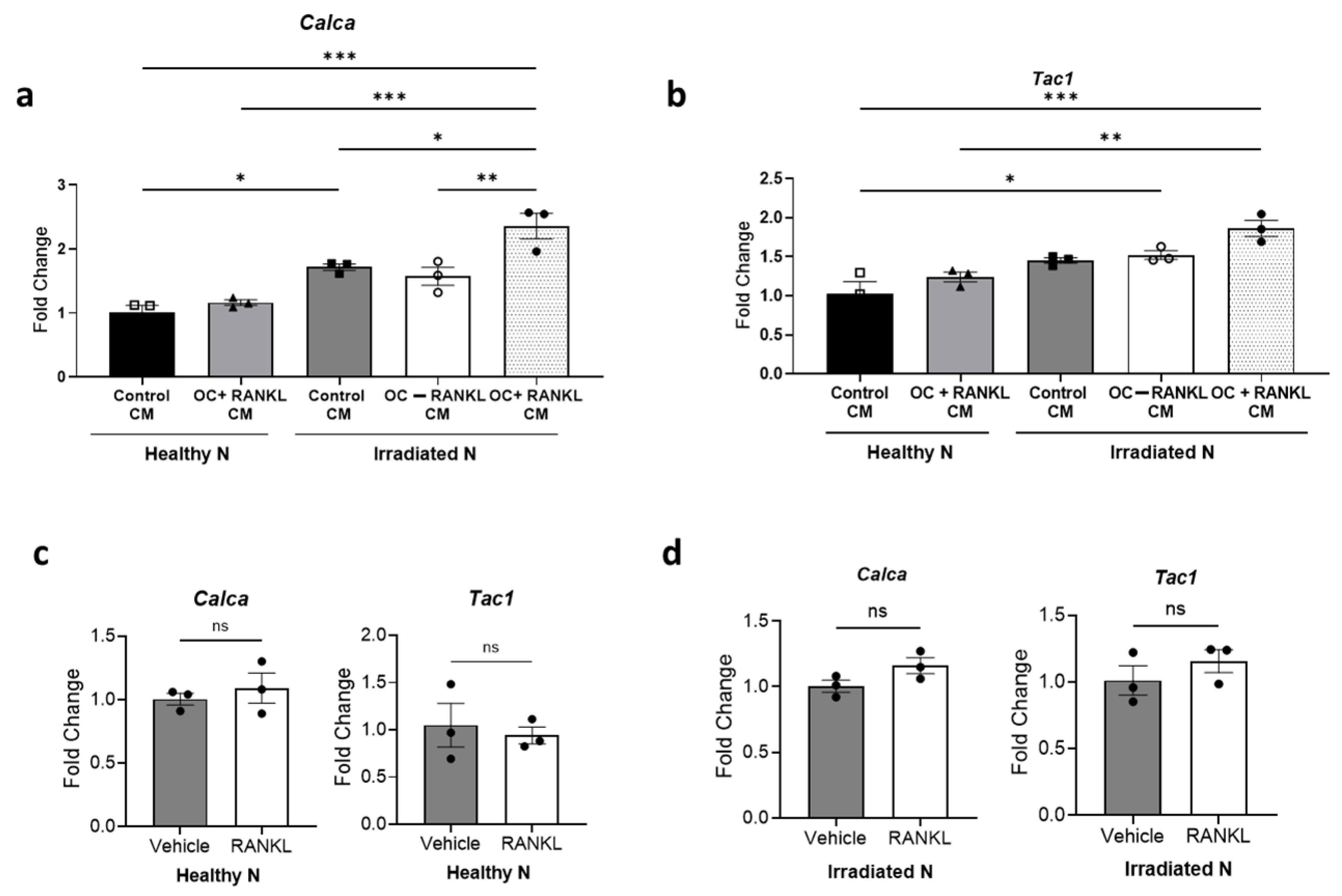
3.2. Impact of Radiation-Activated Osteoclast-Derived Secretory Factors on Pain-Associated Neuropeptides in Sensory Neurons
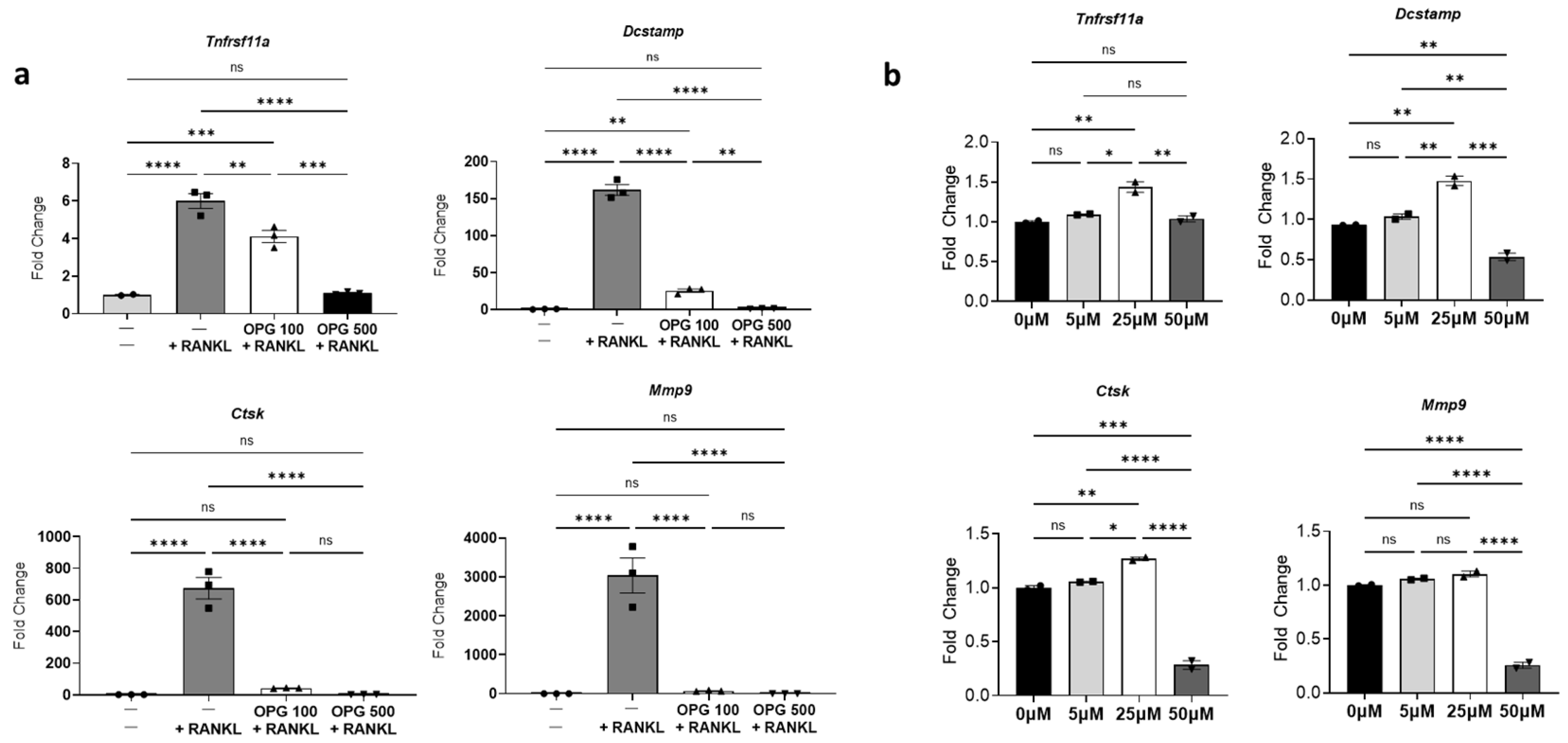
3.3. Modulation of Radiation-Induced Osteoclast Activation and Neuronal Responses by OPG and Bisphosphonate
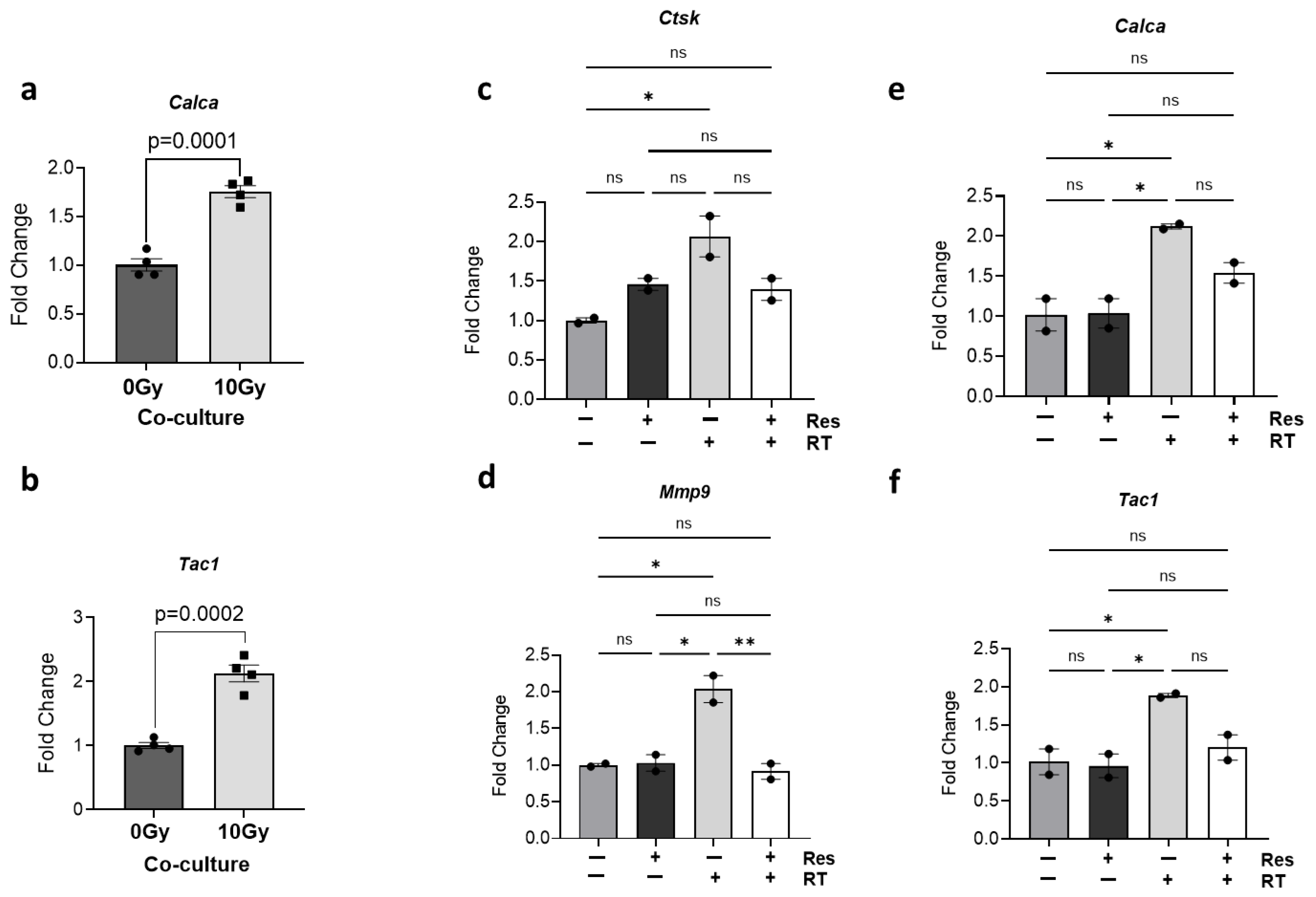
3.4. Effect of Radiation Exposure on Osteoclast-Sensory Neuron Co-Cultures
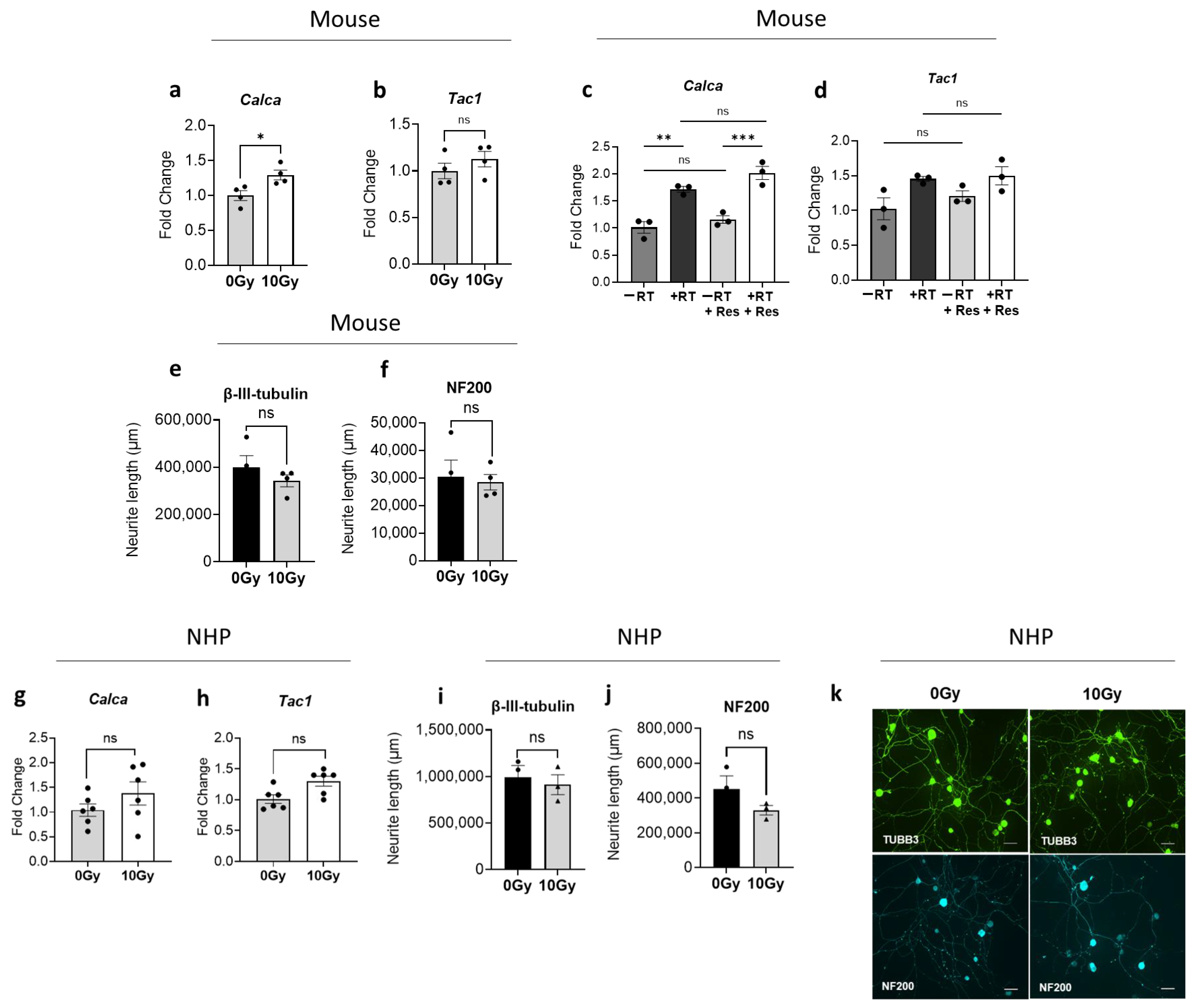
3.5. Effect of Direct Radiation Exposure on Sensory Neurons and Neuropeptide Expression
3.6. Impact of Osteoclast-Derived Secretory Factors on Irradiated Neurons
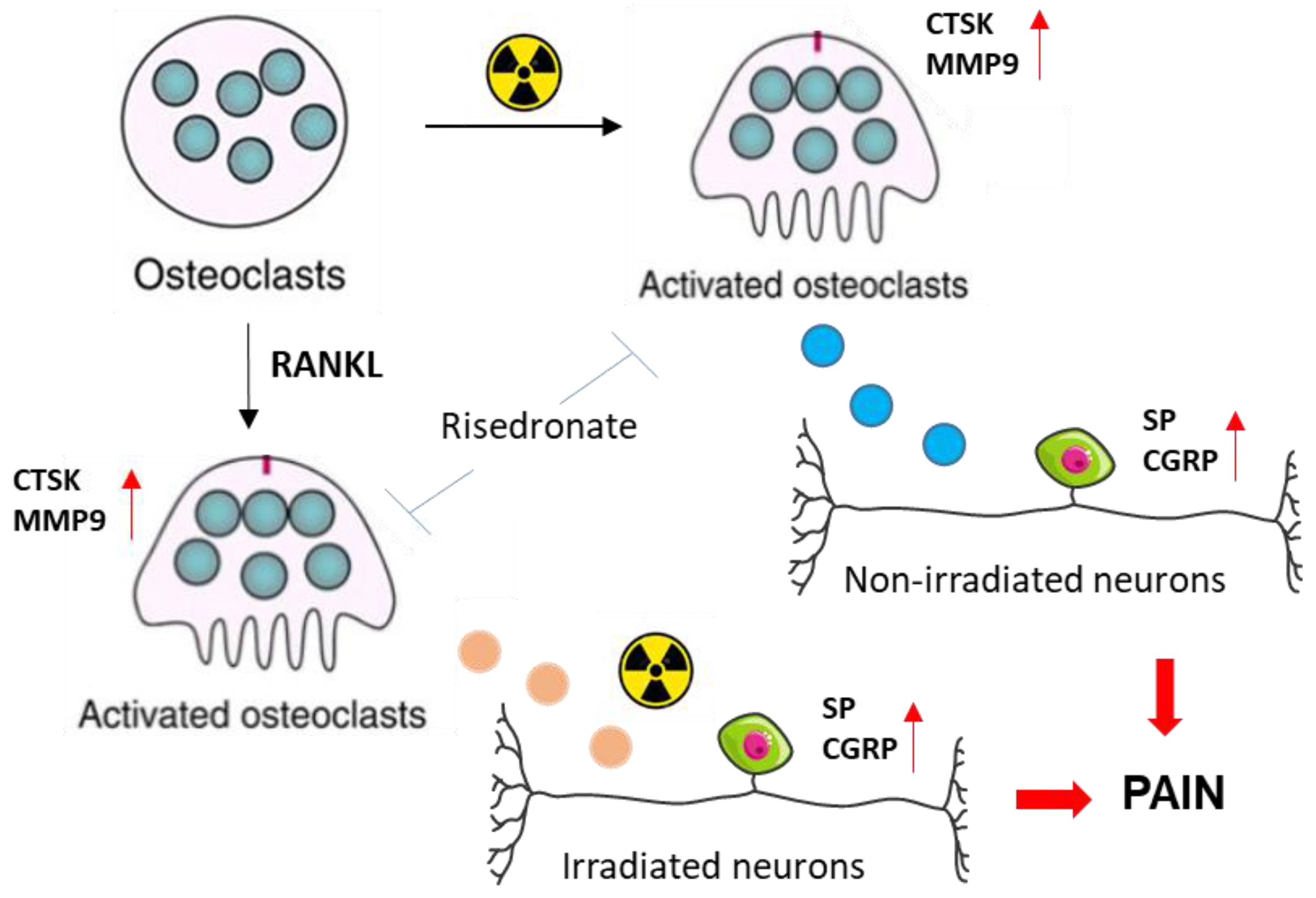
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vlaskou Badra, E.; Baumgartl, M.; Fabiano, S.; Jongen, A.; Guckenberger, M. Stereotactic radiotherapy for early-stage non-small cell lung cancer: Current standards and ongoing research. Transl. Lung Cancer Res. 2021, 10, 1930–1949. [Google Scholar] [CrossRef] [PubMed]
- Chipko, C.; Ojwang, J.; Rezai Gharai, L.; Deng, X.; Mukhopadhyay, N.; Weiss, E. Characterization of chest wall toxicity during long-term follow up after thoracic stereotactic body radiation therapy. Pract. Radiat. Oncol. 2019, 9, e338–e346. [Google Scholar] [CrossRef] [PubMed]
- Albers, J.; Parker, W.; Kildea, J.; Pembroke, C.; Faria, S. Chest wall pain following lung stereotactic body radiation therapy using 48 Gy in three fractions: A search for predictors. Cancer Radiother. 2019, 23, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Dunlap, N.E.; Cai, J.; Biedermann, G.B.; Yang, W.; Benedict, S.H.; Sheng, K.; Schefter, T.E.; Kavanagh, B.D.; Larner, J.M. Chest wall volume receiving >30 Gy predicts risk of severe pain and/or rib fracture after lung stereotactic body radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 2010, 76, 796–801. [Google Scholar] [CrossRef]
- Steber, C.R.; Hughes, R.T.; Soike, M.H.; Jacobson, T.; Helis, C.A.; Farris, J.C.; Farris, M.K. Five- versus ten-fraction regimens of stereotactic body radiation therapy for primary and metastatic NSCLC. Clin. Lung Cancer 2021, 22, e122–e131. [Google Scholar] [CrossRef]
- Tomé, W.A.; Hodge, C.W.; Mehta, M.P.; Bentzen, S.M. Incidence of rib fractures after stereotactic body radiotherapy for peripheral lung lesions: Clinical experience and dose response estimation. J. Radiosurg. SBRT 2011, 1, 155–161. [Google Scholar] [CrossRef]
- Stephans, K.L.; Djemil, T.; Tendulkar, R.D.; Robinson, C.G.; Reddy, C.A.; Videtic, G.M.M. Prediction of chest wall toxicity from lung stereotactic body radiotherapy (SBRT). Int. J. Radiat. Oncol. Biol. Phys. 2012, 82, 974–980. [Google Scholar] [CrossRef]
- Welsh, J.; Thomas, J.; Shah, D.; Allen, P.K.; Wei, X.; Mitchell, K.; Gao, S.; Balter, P.; Komaki, R.; Chang, J.Y. Obesity increases the risk of chest wall pain from thoracic stereotactic body radiation therapy. Int. J. Radiat. Oncol. Biol. Phys. 2011, 81, 91–96. [Google Scholar] [CrossRef]
- Andolino, D.L.; Forquer, J.A.; Henderson, M.A.; Barriger, R.B.; Shapiro, R.H.; Brabham, J.G.; Johnstone, P.A.S.; Cardenes, H.R.; Fakiris, A.J. Chest wall toxicity after stereotactic body radiotherapy for malignant lesions of the lung and liver. Int. J. Radiat. Oncol. Biol. Phys. 2011, 80, 692–697. [Google Scholar] [CrossRef]
- Thompson, M.; Rosenzweig, K.E. The evolving toxicity profile of SBRT for lung cancer. Transl. Lung Cancer Res. 2019, 8, 48–57. [Google Scholar] [CrossRef]
- Voruganti, I.S.; Donovan, E.; Walker-Dilks, C.; Swaminath, A. Chest wall toxicity after stereotactic radiation in early lung cancer: A systematic review. Curr. Oncol. 2020, 27, 179–189. [Google Scholar] [CrossRef] [PubMed]
- Mutter, R.W.; Liu, F.; Abreu, A.; Yorke, E.; Jackson, A.; Rosenzweig, K.E. Dose-volume parameters predict for the development of chest wall pain after stereotactic body radiation for lung cancer. Int. J. Radiat. Oncol. Biol. Phys. 2012, 82, 1783–1790. [Google Scholar] [CrossRef]
- Barnea, Y.; Kashtan, H.; Skornick, Y.; Werbin, N. Isolated rib fractures in elderly patients: Mortality and morbidity. Can. J. Surg. 2002, 45, 43–46. [Google Scholar] [PubMed] [PubMed Central]
- DeNunzio, N.; Milano, M.; Management of Chest Wall Toxicity After SBRT. ARROCase Presentation, American Society for Radiation Oncology (ASTRO). 2020. Available online: https://www.astro.org/ASTRO/media/ASTRO/AffiliatePages/arro/PDFs/ChestWallToxicity.pdf (accessed on 1 January 2025).
- Wang, Q.; Stone, K.; Kern, J.A.; Slatore, C.G.; Swanson, S.; Blackstock, W.; Khan, R.S.; Smith, C.B.; Veluswamy, R.R.; Chidel, M.; et al. Adverse events following limited resection versus stereotactic body radiation therapy for early-stage lung cancer. Ann. Am. Thorac. Soc. 2022, 19, 2053–2061. [Google Scholar] [CrossRef] [PubMed]
- Woody, N.M.; Videtic, G.M.M.; Stephans, K.L.; Djemil, T.; Kim, Y.; Xia, P. Predicting chest wall pain from lung stereotactic body radiotherapy for different fractionation schemes. Int. J. Radiat. Oncol. Biol. Phys. 2012, 83, 427–434. [Google Scholar] [CrossRef]
- Lloyd, S.; Decker, R.H.; Evans, S.B. Bone scan findings of chest wall pain syndrome after stereotactic body radiation therapy: Implications for the pathophysiology of the syndrome. J. Thorac. Dis. 2013, 5, E41–E44. [Google Scholar] [CrossRef]
- Green, D.E.; Adler, B.J.; Chan, M.E.; Rubin, C.T. Devastation of adult stem cell pools by irradiation precedes collapse of trabecular bone quality and quantity. J. Bone Miner. Res. 2012, 27, 749–759. [Google Scholar] [CrossRef]
- Yumoto, K.; Globus, R.K.; Mojarrab, R.; Arakaki, J.; Wang, A.; Searby, N.D.; Almeida, E.A.C.; Limoli, C.L. Short-term effects of whole-body exposure to (56)Fe ions in combination with musculoskeletal disuse on bone cells. Radiat. Res. 2010, 173, 494–504. [Google Scholar] [CrossRef]
- Kondo, H.; Searby, N.D.; Mojarrab, R.; Phillips, J.; Alwood, J.S.; Yumoto, K.; Almeida, E.A.C.; Limoli, C.L.; Globus, R.K. Total-body irradiation of postpubertal mice with (137)Cs acutely compromises the microarchitecture of cancellous bone and increases osteoclasts. Radiat. Res. 2009, 171, 283–289. [Google Scholar] [CrossRef]
- Keenawinna, L.; Oest, M.E.; Mann, K.A.; Spadaro, J.; Damron, T.A. Zoledronic acid prevents loss of trabecular bone after focal irradiation in mice. Radiat. Res. 2013, 180, 89–99. [Google Scholar] [CrossRef]
- Alwood, J.S.; Shahnazari, M.; Chicana, B.; Schreurs, A.S.; Kumar, A.; Bartolini, A.; Shirazi-Fard, Y.; Globus, R.K. Ionizing radiation stimulates expression of pro-osteoclastogenic genes in marrow and skeletal tissue. J. Interferon Cytokine Res. 2015, 35, 480–487. [Google Scholar] [CrossRef]
- Oest, M.E.; Damron, T.A. Focal therapeutic irradiation induces an early transient increase in bone glycation. Radiat. Res. 2014, 181, 439–443. [Google Scholar] [CrossRef]
- Willey, J.S.; Livingston, E.W.; Robbins, M.E.; Bourland, J.D.; Tirado-Lee, L.; Smith-Sielicki, H.; Bateman, T.A. Risedronate prevents early radiation-induced osteoporosis in mice at multiple skeletal locations. Bone 2010, 46, 101–111. [Google Scholar] [CrossRef]
- Aoki, M.; Sato, M.; Hirose, K.; Akimoto, H.; Kawaguchi, H.; Hatayama, Y.; Ono, S.; Takai, Y. Radiation-induced rib fracture after stereotactic body radiotherapy with a total dose of 54–56 Gy given in 9–7 fractions for patients with peripheral lung tumor: Impact of maximum dose and fraction size. Radiat. Oncol. 2015, 10, 99. [Google Scholar] [CrossRef]
- Rydzewski, N.R.; Yadav, P.; Musunuru, H.B.; Condit, K.M.; Francis, D.; Zhao, S.G.; Baschnagel, A.M. Radiomic modeling of bone density and rib fracture risk after stereotactic body radiation therapy for early-stage non-small cell lung cancer. Adv. Radiat. Oncol. 2022, 7, 100884. [Google Scholar] [CrossRef]
- Farris, M.; National Cancer Institute (NCI). A Randomized Clinical Trial of Prophylactic Risedronate for Patients with Peripheral Lung Tumors Treated with SBRT; Wake Forest Baptist Comprehensive Cancer Center: Winston-Salem, NC, USA, 2019.
- Neto, E.; Leitão, L.; Mateus, J.C.; Sousa, D.M.; Alves, C.J.; Aroso, M.; Monteiro, A.C.; Conceição, F.; Oreffo, R.O.C.; West, J.; et al. Osteoclast-derived extracellular vesicles are implicated in sensory neurons sprouting through the activation of epidermal growth factor signaling. Cell Biosci. 2022, 12, 127. [Google Scholar] [CrossRef]
- Yoneda, T.; Hiasa, M.; Nagata, Y.; Okui, T.; White, F.A. Acidic microenvironment and bone pain in cancer-colonized bone. BoneKEy Rep. 2015, 4, 690. [Google Scholar] [CrossRef]
- Hu, Y.; Chen, X.; Wang, S.; Jing, Y.; Su, J. Subchondral bone microenvironment in osteoarthritis and pain. Bone Res. 2021, 9, 20. [Google Scholar] [CrossRef] [PubMed]
- Park, S.H.; Eber, M.R.; das Dores, M.; Fonseca, M.; Patel, C.M.; Cunnane, K.A.; Ding, H.; Hsu, F.-C.; Peters, C.M.; Ko, M.-C.; et al. Usefulness of the measurement of neurite outgrowth of primary sensory neurons to study cancer-related painful complications. Biochem. Pharmacol. 2021, 188, 114520. [Google Scholar] [CrossRef] [PubMed]
- Johnson, B.J.; Barcus, R.A.; Olson, J.D.; Lipford, M.E.; Andrews, R.N.; Dugan, G.O.; Tooze, J.A. Total-body irradiation alters white matter volume and microstructural integrity in rhesus macaques. Int. J. Radiat. Oncol. Biol. Phys. 2024, 119, 208–218. [Google Scholar] [CrossRef] [PubMed]
- Basbaum, A.I.; Bautista, D.M.; Scherrer, G.; Julius, D. Cellular and molecular mechanisms of pain. Cell 2009, 139, 267–284. [Google Scholar] [CrossRef] [PubMed]
- Barker, P.A.; Mantyh, P.; Arendt-Nielsen, L.; Viktrup, L.; Tive, L. Nerve growth factor signaling and its contribution to pain. J. Pain Res. 2020, 13, 1223–1241. [Google Scholar] [CrossRef] [PubMed]
- Willey, J.S.; Lloyd, S.A.J.; Robbins, M.E.; Bourland, J.D.; Smith-Sielicki, H.; Bowman, L.C.; Norrdin, R.W.; Bateman, T.A. Early increase in osteoclast number in mice after whole-body irradiation with 2 Gy X rays. Radiat. Res. 2008, 170, 388–392. [Google Scholar] [CrossRef] [PubMed]
- Taremi, M.; Hope, A.; Lindsay, P.; Dahele, M.; Fung, S.; Purdie, T.G.; Jaffray, D.; Dawson, L.; Bezjak, A. Predictors of radiotherapy-induced bone injury (RIBI) after stereotactic lung radiotherapy. Radiat. Oncol. 2012, 7, 159. [Google Scholar] [CrossRef]
- U.S. Department of Health and Human Services. Pain Management Best Practices Inter-Agency Task Force. In Pain Management Best Practices Inter-Agency Task Force Report: Updates, Gaps, Inconsistencies, and Recommendations; U.S. Department of Health and Human Services: Washington, DC, USA, 2019. Available online: https://www.hhs.gov/sites/default/files/pmtf-final-report-2019-05-23.pdf (accessed on 1 January 2025).
- Shaikh, T.; Turaka, A. Predictors and management of chest wall toxicity after lung stereotactic body radiotherapy. Cancer Treat. Rev. 2014, 40, 1215–1220. [Google Scholar] [CrossRef]
- McNicol, E.; Horowicz-Mehler, N.; Fisk, R.A.; Bennett, K.; Gialeli-Goudas, M.; Chew, P.W.; Lau, J.; Carr, D. Management of opioid side effects in cancer-related and chronic noncancer pain: A systematic review. J. Pain 2003, 4, 231–256. [Google Scholar] [CrossRef]
- Wiffen, P.J.; Derry, S.; Bell, R.F.; Rice, A.S.C.; Tölle, T.R.; Phillips, T.; Moore, R.A. Gabapentin for chronic neuropathic pain in adults. Cochrane Database Syst. Rev. 2017, 6, CD007938. [Google Scholar] [CrossRef]
- Fu, K.; Robbins, S.R.; McDougall, J.J. Osteoarthritis: The genesis of pain. Rheumatology 2018, 57 (Suppl. S4), iv43–iv50. [Google Scholar] [CrossRef]
- Wood, M.J.; Miller, R.E.; Malfait, A.-M. The genesis of pain in osteoarthritis: Inflammation as a mediator of osteoarthritis pain. Clin. Geriatr. Med. 2022, 38, 221–238. [Google Scholar] [CrossRef]
- Suzuki, M.; Millecamps, M.; Ohtori, S.; Mori, C.; Stone, L.S. Anti–nerve growth factor therapy attenuates cutaneous hypersensitivity and musculoskeletal discomfort in mice with osteoporosis. Pain Rep. 2018, 3, e652. [Google Scholar] [CrossRef]
- Ni, S.; Ling, Z.; Wang, X.; Cao, Y.; Wu, T.; Deng, R.; Crane, J.L.; Skolasky, R.; Demehri, S.; Zhen, G.; et al. Sensory innervation in porous endplates by Netrin-1 from osteoclasts mediates PGE2-induced spinal hypersensitivity in mice. Nat. Commun. 2019, 10, 5643. [Google Scholar] [CrossRef] [PubMed]
- Andriessen, A.S.; Donnelly, C.R.; Ji, R.-R. Reciprocal interactions between osteoclasts and nociceptive sensory neurons in bone cancer pain. Pain Rep. 2021, 6, e867. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Farris, M.; McTyre, E.R.; Okoukoni, C.; Dugan, G.; Johnson, B.J.; Blackstock, A.W.; Munley, M.T.; Bourland, J.D.; Cline, J.M.; Willey, J.S. Cortical thinning and structural bone changes in non-human primates after single-fraction whole-chest irradiation. Radiat. Res. 2018, 190, 63–71. [Google Scholar] [CrossRef]
- Richardson, J.D.; Vasko, M.R. Cellular mechanisms of neurogenic inflammation. J. Pharmacol. Exp. Ther. 2002, 302, 839–845. [Google Scholar] [CrossRef]
- Chiu, I.M.; von Hehn, C.A.; Woolf, C.J. Neurogenic inflammation and the peripheral nervous system in host defense and immunopathology. Nat. Neurosci. 2012, 15, 10631067. [Google Scholar] [CrossRef]
- Gutierrez, H.; O’Keeffe, G.W.; Gavaldà, N.; Gallagher, D.; Davies, A.M. Nuclear factor κB signaling either stimulates or inhibits neurite growth depending on the phosphorylation status of p65/RelA. J. Neurosci. 2008, 28, 8246–8256. [Google Scholar] [CrossRef]
- Ji, R.-R.; Samad, T.A.; Jin, S.-X.; Schmoll, R.; Woolf, C.J. p38 MAPK activation by NGF in primary sensory neurons after inflammation increases TRPV1 levels and maintains heat hyperalgesia. Neuron 2002, 36, 57–68. [Google Scholar] [CrossRef]
- Jin, X.; Gereau IV, R.W. Acute p38-mediated modulation of tetrodotoxin-resistant sodium channels in mouse sensory neurons by tumor necrosis factor-α. J. Neurosci. 2006, 26, 246–255. [Google Scholar] [CrossRef]
- Lai, Y.-H.E.; Bäumer, W.; Meneses, C.; Roback, D.M.; Robertson, J.B.; Mishra, S.K.; Lascelles, B.D.X.; Nolan, M.W. Irradiation of the normal murine tongue causes upregulation and activation of transient receptor potential (TRP) ion channels. Radiat. Res. 2021, 196, 331–344. [Google Scholar] [CrossRef]
- Meneses, C.S.; Gidcumb, E.M.; Marcus, K.L.; Gonzalez, Y.; Lai, Y.H.; Mishra, S.K.; Lascelles, B.D.X.; Nolan, M.W. Acute radiotherapy-associated oral pain may promote tumor growth at distant sites. Front. Oncol. 2023, 13, 1029108. [Google Scholar] [CrossRef]
- Scholz, J.; Woolf, C.J. The neuropathic pain triad: Neurons, immune cells and glia. Nat. Neurosci. 2007, 10, 1361–1368. [Google Scholar] [CrossRef]
- Ji, R.-R.; Berta, T.; Nedergaard, M. Glia and pain: Is chronic pain a gliopathy? Pain 2013, 154 (Suppl. S1), S10S28. [Google Scholar] [CrossRef]
- Farris, M.; Hughes, R.T.; Pacholke, H.D.; Levine, B.; Black, P.J.; Razavian, N.; Ververs, J.D.; Ponnatapura, J.; Farris, J.C.; Glynn, S.; et al. The Impact of Prophylactic Bisphosphonates on Radiation-Induced Rib Fractures and Chest Wall Pain in Peripheral Lung Tumor SBRT: A Randomized, Double-Blind, Placebo-Controlled Study. Int. J. Radiat. Oncol. Biol. Phys. 2024, 120, S85–S86. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, S.H.; Peters, M.; Aguayo, C.; Farris, M.K.; Hughes, R.T.; Moore, J.; Munley, M.T.; Reno, K.E.; Foster, J.A.; Gardin, J.; et al. Preclinical Study of Pain Neuropeptide Expression in Murine Sensory Neurons Induced by Irradiated Osteoclasts in the Context of Stereotactic Body Radiation Therapy. Cells 2025, 14, 1324. https://doi.org/10.3390/cells14171324
Park SH, Peters M, Aguayo C, Farris MK, Hughes RT, Moore J, Munley MT, Reno KE, Foster JA, Gardin J, et al. Preclinical Study of Pain Neuropeptide Expression in Murine Sensory Neurons Induced by Irradiated Osteoclasts in the Context of Stereotactic Body Radiation Therapy. Cells. 2025; 14(17):1324. https://doi.org/10.3390/cells14171324
Chicago/Turabian StylePark, Sun H., Megan Peters, Caleb Aguayo, Michael K. Farris, Ryan T. Hughes, Joseph Moore, Michael T. Munley, Kaitlyn E. Reno, Jeffrey A. Foster, Jean Gardin, and et al. 2025. "Preclinical Study of Pain Neuropeptide Expression in Murine Sensory Neurons Induced by Irradiated Osteoclasts in the Context of Stereotactic Body Radiation Therapy" Cells 14, no. 17: 1324. https://doi.org/10.3390/cells14171324
APA StylePark, S. H., Peters, M., Aguayo, C., Farris, M. K., Hughes, R. T., Moore, J., Munley, M. T., Reno, K. E., Foster, J. A., Gardin, J., Schaaf, G. W., Cline, J. M., Peters, C. M., & Willey, J. S. (2025). Preclinical Study of Pain Neuropeptide Expression in Murine Sensory Neurons Induced by Irradiated Osteoclasts in the Context of Stereotactic Body Radiation Therapy. Cells, 14(17), 1324. https://doi.org/10.3390/cells14171324






