Effects of Eribulin on the RNA Content of Extracellular Vesicles Released by Metastatic Breast Cancer Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Cultures
2.2. Extracellular Vesicle Isolation
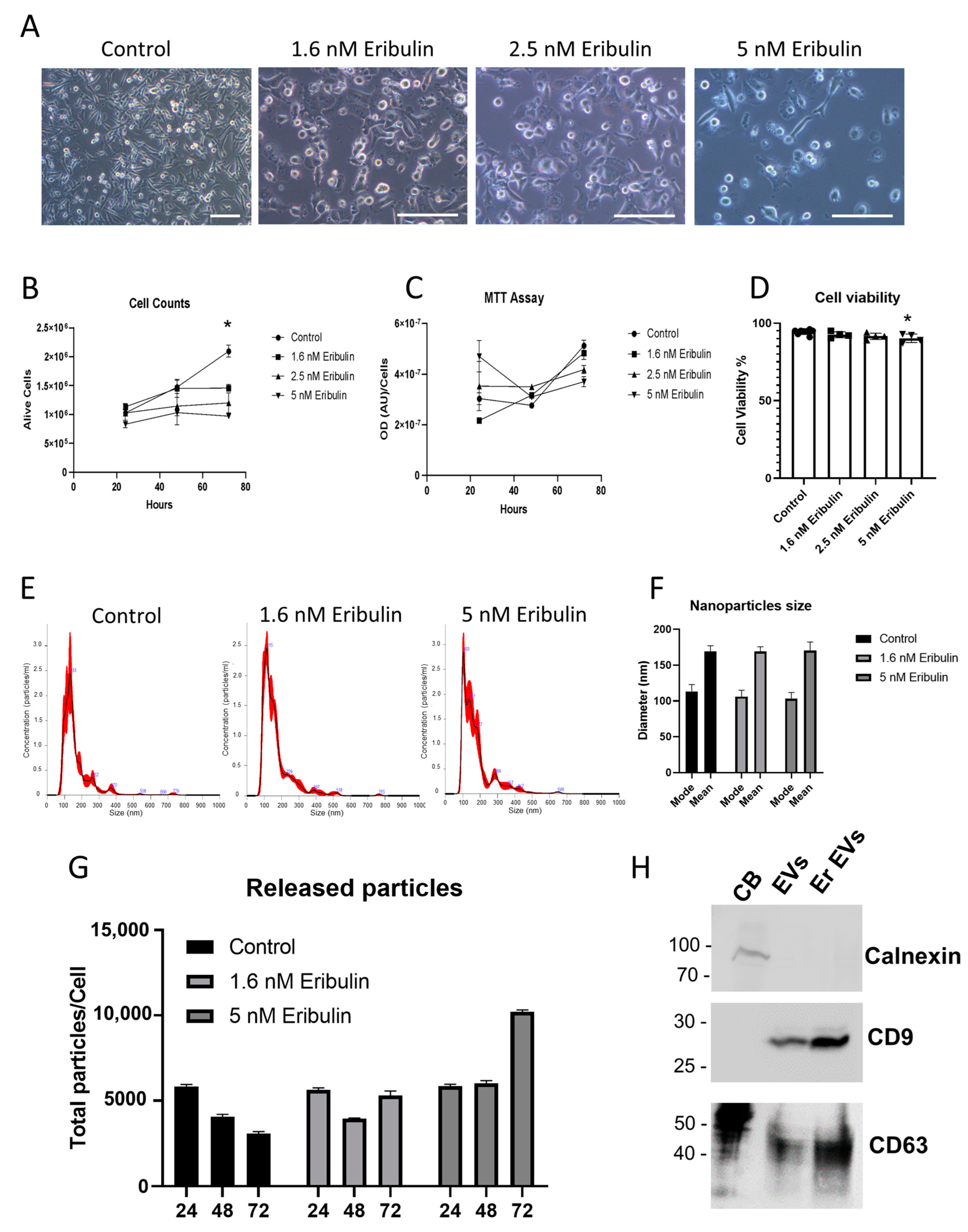
2.3. Nanoparticle Tracking Assay (NTA)
2.4. Western Blotting Analysis
2.5. RNA Sequencing and Data Analysis
2.6. Analysis of Co-Expressed Gene Modules by WGCNA
2.7. Analysis of miRNA Target Genes
2.8. Association of piRNAs, snoRNAs and tRNA Fragments with Breast Cancer
2.9. RNA Isolation and Quantification
3. Results
3.1. EV Isolation and Characterization
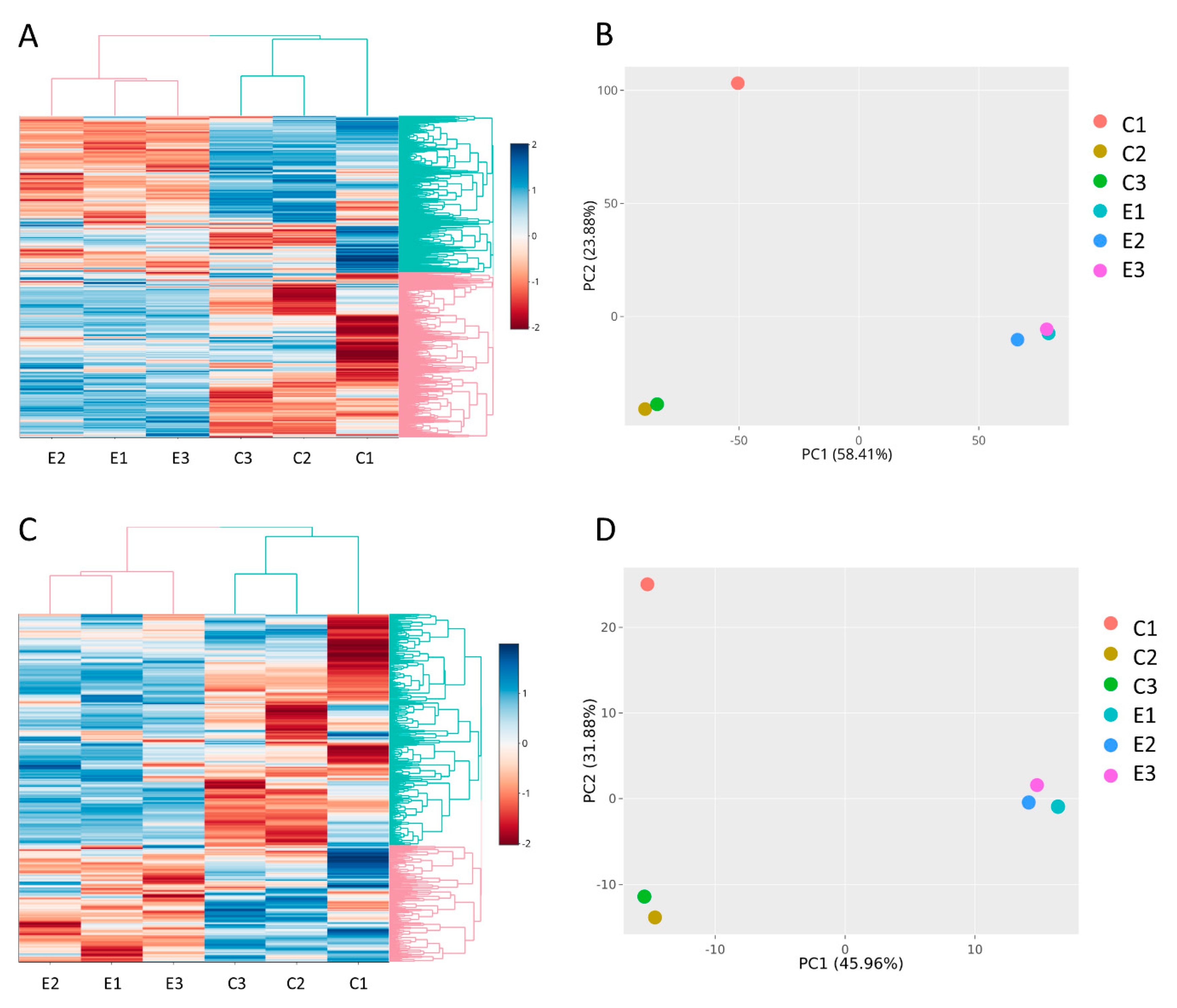
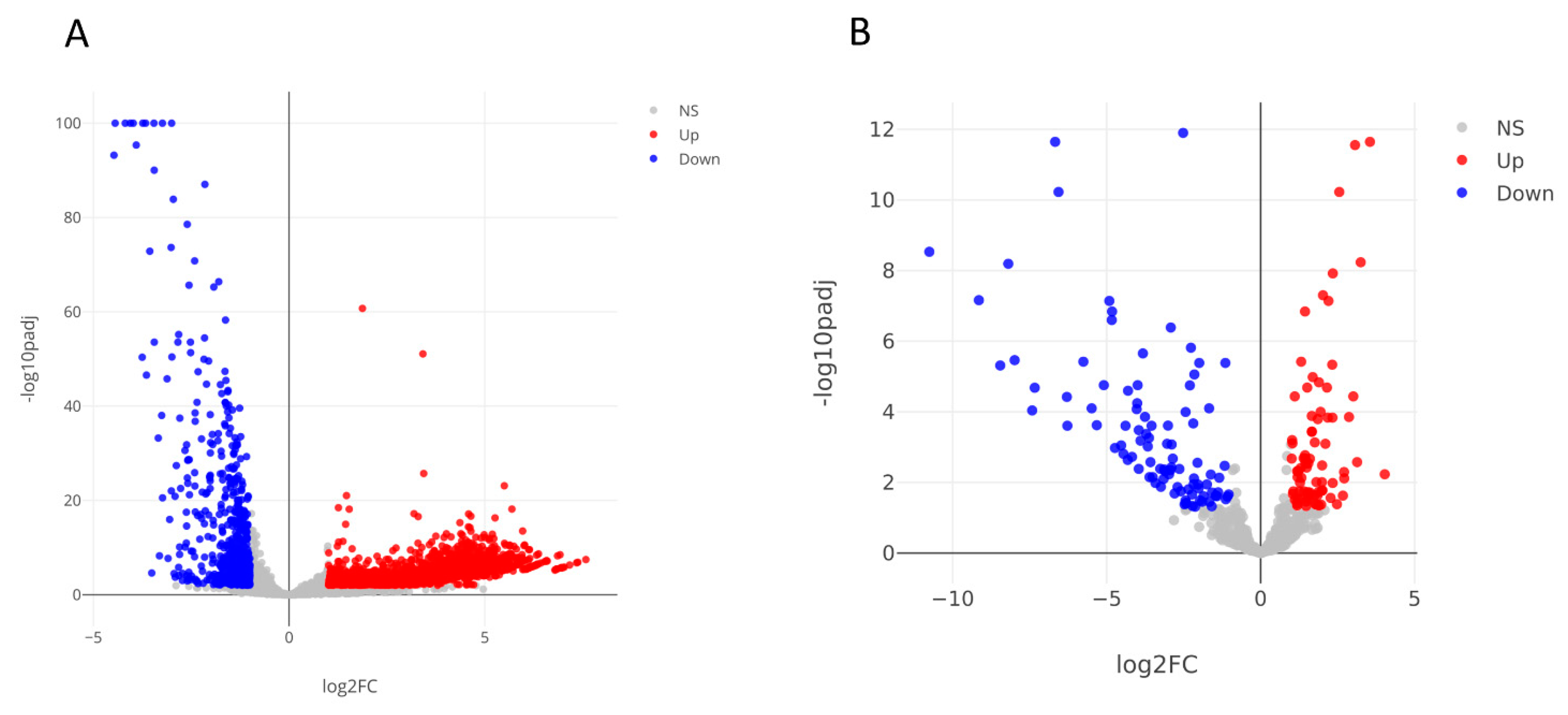
3.2. RNA Sequencing and Data Analysis
3.3. Functional Enrichment Analysis
3.4. Identification of Key Genes Associated with the Eribulin Treatment by WGCNA
3.5. Experimentally Assessed Target Genes of miRNAs in EV
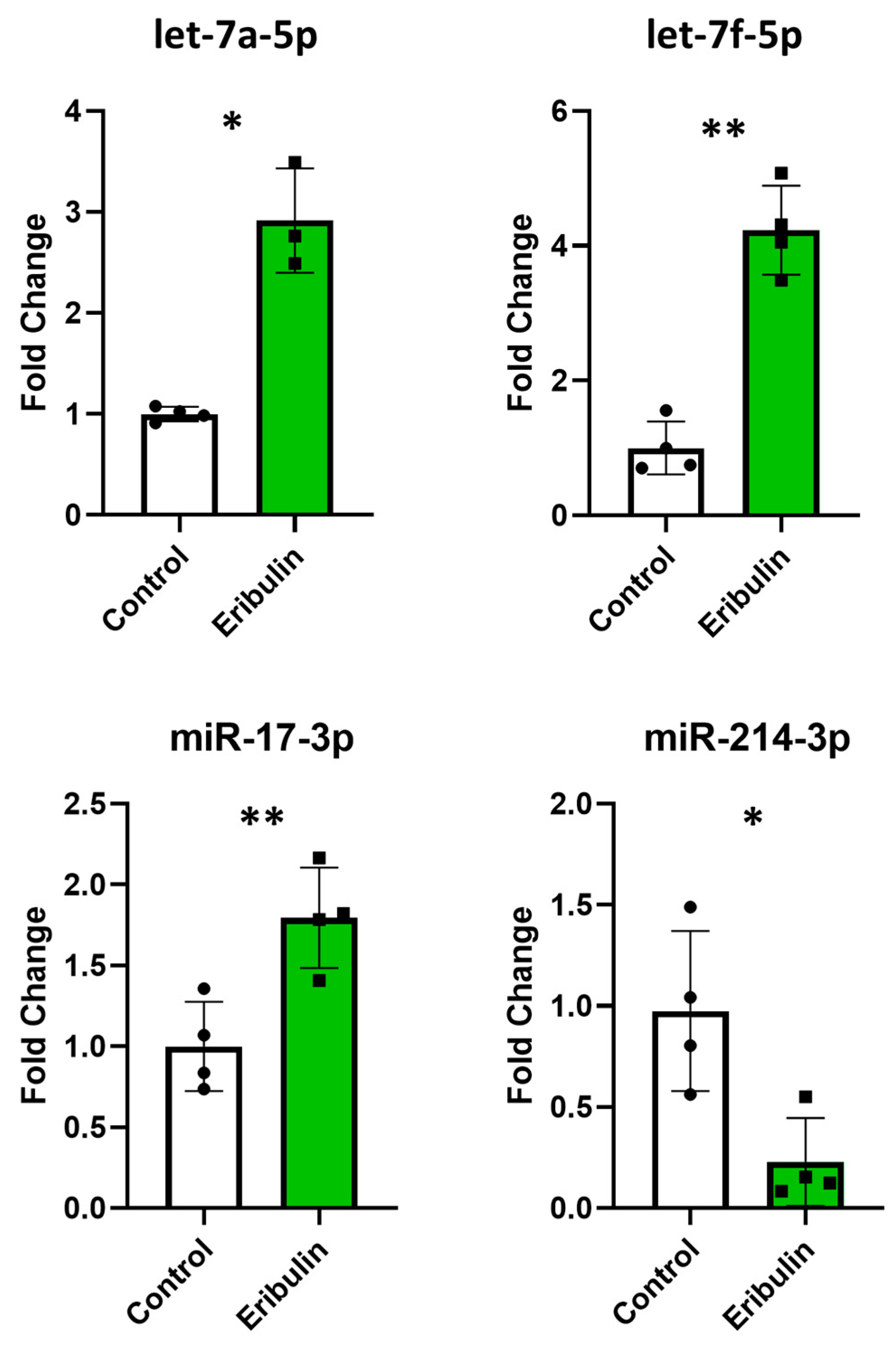
3.6. Comparison of MicroRNA Expression in EVs and Cells
3.7. Association of Other Small RNAs with Breast Cancer
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: Globocan estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Morrow, M.; Schnitt, S.J.; Norton, L. Current management of lesions associated with an increased risk of breast cancer. Nat. Rev. Clin. Oncol. 2015, 12, 227–238. [Google Scholar] [CrossRef]
- Porter, D.A.; Krop, I.E.; Nasser, S.; Sgroi, D.; Kaelin, C.M.; Marks, J.R.; Riggins, G.; Polyak, K. A sage (serial analysis of gene expression) view of breast tumor progression. Cancer Res. 2001, 61, 5697–5702. [Google Scholar]
- Chin, K.; de Solorzano, C.O.; Knowles, D.; Jones, A.; Chou, W.; Rodriguez, E.G.; Kuo, W.L.; Ljung, B.M.; Chew, K.; Myambo, K.; et al. In situ analyses of genome instability in breast cancer. Nat. Genet. 2004, 36, 984–988. [Google Scholar] [CrossRef]
- Salemme, V.; Centonze, G.; Cavallo, F.; Defilippi, P.; Conti, L. The crosstalk between tumor cells and the immune microenvironment in breast cancer: Implications for immunotherapy. Front. Oncol. 2021, 11, 610303. [Google Scholar] [CrossRef] [PubMed]
- Wolff, A.C.; Hammond, M.E.; Hicks, D.G.; Dowsett, M.; McShane, L.M.; Allison, K.H.; Allred, D.C.; Bartlett, J.M.; Bilous, M.; Fitzgibbons, P.; et al. Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American society of clinical oncology/college of American pathologists clinical practice guideline update. J. Clin. Oncol. 2013, 31, 3997–4013. [Google Scholar] [CrossRef] [PubMed]
- Early Breast Cancer Trialists’ Collaborative Group (EBCTCG). Anthracycline-containing and taxane-containing chemotherapy for early-stage operable breast cancer: A patient-level meta-analysis of 100,000 women from 86 randomised trials. Lancet 2023, 401, 1277–1292. [Google Scholar] [CrossRef] [PubMed]
- Camara, R.J.A.; Schwentner, L.; Friedl, T.W.P.; Deniz, M.; Fink, V.; Lato, K.; Widschwendter, P.; Rack, B.; Janni, W.; Singer, S.; et al. Quality of life during and after adjuvant anthracycline-taxane-based chemotherapy with or without gemcitabine in high-risk early breast cancer: Results of the success a trial. Breast Cancer Res. Treat. 2019, 175, 627–635. [Google Scholar] [CrossRef]
- Masuda, N.; Lee, S.J.; Ohtani, S.; Im, Y.H.; Lee, E.S.; Yokota, I.; Kuroi, K.; Im, S.A.; Park, B.W.; Kim, S.B.; et al. Adjuvant capecitabine for breast cancer after preoperative chemotherapy. N. Engl. J. Med. 2017, 376, 2147–2159. [Google Scholar] [CrossRef]
- den Brok, W.D.; Speers, C.H.; Gondara, L.; Baxter, E.; Tyldesley, S.K.; Lohrisch, C.A. Survival with metastatic breast cancer based on initial presentation, de novo versus relapsed. Breast Cancer Res. Treat. 2017, 161, 549–556. [Google Scholar] [CrossRef]
- Okouneva, T.; Azarenko, O.; Wilson, L.; Littlefield, B.A.; Jordan, M.A. Inhibition of centromere dynamics by eribulin (e7389) during mitotic metaphase. Mol. Cancer Ther. 2008, 7, 2003–2011. [Google Scholar] [CrossRef]
- Jordan, M.A.; Kamath, K.; Manna, T.; Okouneva, T.; Miller, H.P.; Davis, C.; Littlefield, B.A.; Wilson, L. The primary antimitotic mechanism of action of the synthetic halichondrin e7389 is suppression of microtubule growth. Mol. Cancer Ther. 2005, 4, 1086–1095. [Google Scholar] [CrossRef]
- Kuznetsov, G.; Towle, M.J.; Cheng, H.; Kawamura, T.; TenDyke, K.; Liu, D.; Kishi, Y.; Yu, M.J.; Littlefield, B.A. Induction of morphological and biochemical apoptosis following prolonged mitotic blockage by halichondrin b macrocyclic ketone analog e7389. Cancer Res. 2004, 64, 5760–5766. [Google Scholar] [CrossRef]
- Kashiwagi, S.; Asano, Y.; Goto, W.; Takada, K.; Takahashi, K.; Hatano, T.; Tanaka, S.; Takashima, T.; Tomita, S.; Motomura, H.; et al. Mesenchymal-epithelial transition and tumor vascular remodeling in eribulin chemotherapy for breast cancer. Anticancer Res. 2018, 38, 401–410. [Google Scholar]
- Agoulnik, S.I.; Kawano, S.; Taylor, N.; Oestreicher, J.; Matsui, J.; Chow, J.; Oda, Y.; Funahashi, Y. Eribulin mesylate exerts specific gene expression changes in pericytes and shortens pericyte-driven capillary network in vitro. Vasc. Cell 2014, 6, 3. [Google Scholar] [CrossRef]
- Funahashi, Y.; Okamoto, K.; Adachi, Y.; Semba, T.; Uesugi, M.; Ozawa, Y.; Tohyama, O.; Uehara, T.; Kimura, T.; Watanabe, H.; et al. Eribulin mesylate reduces tumor microenvironment abnormality by vascular remodeling in preclinical human breast cancer models. Cancer Sci. 2014, 105, 1334–1342. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T.; Ozawa, Y.; Kimura, T.; Sato, Y.; Kuznetsov, G.; Xu, S.; Uesugi, M.; Agoulnik, S.; Taylor, N.; Funahashi, Y.; et al. Eribulin mesilate suppresses experimental metastasis of breast cancer cells by reversing phenotype from epithelial-mesenchymal transition (emt) to mesenchymal-epithelial transition (met) states. Br. J. Cancer 2014, 110, 1497–1505. [Google Scholar] [CrossRef] [PubMed]
- Husemann, L.C.; Reese, A.; Radine, C.; Piekorz, R.P.; Budach, W.; Sohn, D.; Janicke, R.U. The microtubule targeting agents eribulin and paclitaxel activate similar signaling pathways and induce cell death predominantly in a caspase-independent manner. Cell Cycle 2020, 19, 464–478. [Google Scholar] [CrossRef] [PubMed]
- Yoshie, M.; Ishida, A.; Ohashi, H.; Nakachi, N.; Azumi, M.; Tamura, K. Stathmin dynamics modulate the activity of eribulin in breast cancer cells. Pharmacol. Res. Perspect. 2021, 9, e00786. [Google Scholar] [CrossRef] [PubMed]
- Pivot, X.; Marme, F.; Koenigsberg, R.; Guo, M.; Berrak, E.; Wolfer, A. Pooled analyses of eribulin in metastatic breast cancer patients with at least one prior chemotherapy. Ann. Oncol. 2016, 27, 1525–1531. [Google Scholar] [CrossRef] [PubMed]
- Cortes, J.; Vahdat, L.; Blum, J.L.; Twelves, C.; Campone, M.; Roche, H.; Bachelot, T.; Awada, A.; Paridaens, R.; Goncalves, A.; et al. Phase ii study of the halichondrin b analog eribulin mesylate in patients with locally advanced or metastatic breast cancer previously treated with an anthracycline, a taxane, and capecitabine. J. Clin. Oncol. 2010, 28, 3922–3928. [Google Scholar] [CrossRef] [PubMed]
- Vahdat, L.T.; Pruitt, B.; Fabian, C.J.; Rivera, R.R.; Smith, D.A.; Tan-Chiu, E.; Wright, J.; Tan, A.R.; Dacosta, N.A.; Chuang, E.; et al. Phase ii study of eribulin mesylate, a halichondrin b analog, in patients with metastatic breast cancer previously treated with an anthracycline and a taxane. J. Clin. Oncol. 2009, 27, 2954–2961. [Google Scholar] [CrossRef] [PubMed]
- Pellegrino, B.; Cavanna, L.; Boggiani, D.; Zamagni, C.; Frassoldati, A.; Schirone, A.; Caldara, A.; Rocca, A.; Gori, S.; Piacentini, F.; et al. Phase II study of eribulin in combination with gemcitabine for the treatment of patients with locally advanced or metastatic triple negative breast cancer (erige trial). Clinical and pharmacogenetic results on behalf of the gruppo oncologico italiano di ricerca clinica (goirc). ESMO Open 2021, 6, 100019. [Google Scholar]
- Zhang, Z.Y.; King, B.M.; Pelletier, R.D.; Wong, Y.N. Delineation of the interactions between the chemotherapeutic agent eribulin mesylate (e7389) and human cyp3a4. Cancer Chemother. Pharmacol. 2008, 62, 707–716. [Google Scholar] [CrossRef] [PubMed]
- Cortes, J.; Montero, A.J.; Gluck, S. Eribulin mesylate, a novel microtubule inhibitor in the treatment of breast cancer. Cancer Treat. Rev. 2012, 38, 143–151. [Google Scholar] [CrossRef]
- Cortes, J.; Schoffski, P.; Littlefield, B.A. Multiple modes of action of eribulin mesylate: Emerging data and clinical implications. Cancer Treat. Rev. 2018, 70, 190–198. [Google Scholar] [CrossRef]
- Raposo, G.; Stoorvogel, W. Extracellular vesicles: Exosomes, microvesicles, and friends. J. Cell Biol. 2013, 200, 373–383. [Google Scholar] [CrossRef]
- van Niel, G.; D’Angelo, G.; Raposo, G. Shedding light on the cell biology of extracellular vesicles. Nat. Rev. Mol. Cell Biol. 2018, 19, 213–228. [Google Scholar] [CrossRef]
- King, H.W.; Michael, M.Z.; Gleadle, J.M. Hypoxic enhancement of exosome release by breast cancer cells. BMC Cancer 2012, 12, 421. [Google Scholar] [CrossRef]
- Higginbotham, J.N.; Demory Beckler, M.; Gephart, J.D.; Franklin, J.L.; Bogatcheva, G.; Kremers, G.J.; Piston, D.W.; Ayers, G.D.; McConnell, R.E.; Tyska, M.J.; et al. Amphiregulin exosomes increase cancer cell invasion. Curr. Biol. 2011, 21, 779–786. [Google Scholar] [CrossRef]
- Santos, J.C.; Lima, N.D.S.; Sarian, L.O.; Matheu, A.; Ribeiro, M.L.; Derchain, S.F.M. Exosome-mediated breast cancer chemoresistance via mir-155 transfer. Sci. Rep. 2018, 8, 829. [Google Scholar] [CrossRef]
- Thompson, C.A.; Purushothaman, A.; Ramani, V.C.; Vlodavsky, I.; Sanderson, R.D. Heparanase regulates secretion, composition, and function of tumor cell-derived exosomes. J. Biol. Chem. 2013, 288, 10093–10099. [Google Scholar] [CrossRef]
- Yang, Y.; Li, C.W.; Chan, L.C.; Wei, Y.; Hsu, J.M.; Xia, W.; Cha, J.H.; Hou, J.; Hsu, J.L.; Sun, L.; et al. Exosomal pd-l1 harbors active defense function to suppress t cell killing of breast cancer cells and promote tumor growth. Cell Res. 2018, 28, 862–864. [Google Scholar] [CrossRef]
- Hu, W.; Tan, C.; He, Y.; Zhang, G.; Xu, Y.; Tang, J. Functional miRNAs in breast cancer drug resistance. Onco Targets Ther. 2018, 11, 1529–1541. [Google Scholar] [CrossRef] [PubMed]
- Lv, M.M.; Zhu, X.Y.; Chen, W.X.; Zhong, S.L.; Hu, Q.; Ma, T.F.; Zhang, J.; Chen, L.; Tang, J.H.; Zhao, J.H. Exosomes mediate drug resistance transfer in mcf-7 breast cancer cells and a probable mechanism is delivery of p-glycoprotein. Tumour Biol. 2014, 35, 10773–10779. [Google Scholar] [CrossRef] [PubMed]
- Pederson, P.J.; Liang, H.; Filonov, D.; Mooberry, S.L. Eribulin and paclitaxel differentially alter extracellular vesicles and their cargo from triple-negative breast cancer cells. Cancers 2021, 13, 2783. [Google Scholar] [CrossRef] [PubMed]
- Bertolini, I.; Ghosh, J.C.; Kossenkov, A.V.; Mulugu, S.; Krishn, S.R.; Vaira, V.; Qin, J.; Plow, E.F.; Languino, L.R.; Altieri, D.C. Small extracellular vesicle regulation of mitochondrial dynamics reprograms a hypoxic tumor microenvironment. Dev. Cell 2020, 55, 163–177.e166. [Google Scholar] [CrossRef]
- Kreger, B.T.; Johansen, E.R.; Cerione, R.A.; Antonyak, M.A. The enrichment of survivin in exosomes from breast cancer cells treated with paclitaxel promotes cell survival and chemoresistance. Cancers 2016, 8, 111. [Google Scholar] [CrossRef] [PubMed]
- Guescini, M.; Canonico, B.; Lucertini, F.; Maggio, S.; Annibalini, G.; Barbieri, E.; Luchetti, F.; Papa, S.; Stocchi, V. Muscle Releases Alpha-Sarcoglycan Positive Extracellular Vesicles Carrying miRNAs in the Bloodstream. PLoS ONE 2015, 10, e0125094. [Google Scholar] [CrossRef] [PubMed]
- Mensa, E.; Guescini, M.; Giuliani, A.; Bacalini, M.G.; Ramini, D.; Corleone, G.; Ferracin, M.; Fulgenzi, G.; Graciotti, L.; Prattichizzo, F.; et al. Small extracellular vesicles deliver mir-21 and mir-217 as pro-senescence effectors to endothelial cells. J. Extracell. Vesicles 2020, 9, 1725285. [Google Scholar] [CrossRef]
- Coenye, T. Do results obtained with RNA-sequencing require independent verification? Biofilm 2021, 13, 100043. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.; Heger, A.; Sudbery, I. Umi-tools: Modeling sequencing errors in unique molecular identifiers to improve quantification accuracy. Genome Res. 2017, 27, 491–499. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Paggi, J.M.; Park, C.; Bennett, C.; Salzberg, S.L. Graph-based genome alignment and genotyping with hisat2 and hisat-genotype. Nat. Biotechnol. 2019, 37, 907–915. [Google Scholar] [CrossRef] [PubMed]
- Anders, S.; Pyl, P.T.; Huber, W. Htseq—A python framework to work with high-throughput sequencing data. Bioinformatics 2015, 31, 166–169. [Google Scholar] [CrossRef] [PubMed]
- Kozomara, A.; Birgaoanu, M.; Griffiths-Jones, S. Mirbase: From microRNA sequences to function. Nucleic Acids Res. 2019, 47, D155–D162. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhang, P.; Lu, Y.; Li, Y.; Zheng, Y.; Kan, Y.; Chen, R.; He, S. PiRBase: A comprehensive database of piRNA sequences. Nucleic Acids Res. 2019, 47, D175–D180. [Google Scholar] [CrossRef]
- La Ferlita, A.; Alaimo, S.; Veneziano, D.; Nigita, G.; Balatti, V.; Croce, C.M.; Ferro, A.; Pulvirenti, A. Identification of tRNA-derived ncRNAs in tcga and nci-60 panel cell lines and development of the public database trfexplorer. Database 2019, 2019, baz115. [Google Scholar] [CrossRef]
- Kucukural, A.; Yukselen, O.; Ozata, D.M.; Moore, M.J.; Garber, M. Debrowser: Interactive differential expression analysis and visualization tool for count data. BMC Genom. 2019, 20, 6. [Google Scholar] [CrossRef]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with deseq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef]
- Xie, Z.; Bailey, A.; Kuleshov, M.V.; Clarke, D.J.B.; Evangelista, J.E.; Jenkins, S.L.; Lachmann, A.; Wojciechowicz, M.L.; Kropiwnicki, E.; Jagodnik, K.M.; et al. Gene set knowledge discovery with enrichr. Curr. Protoc. 2021, 1, e90. [Google Scholar] [CrossRef]
- Langfelder, P.; Horvath, S. Wgcna: An r package for weighted correlation network analysis. BMC Bioinform. 2008, 9, 559. [Google Scholar] [CrossRef]
- Zhang, B.; Horvath, S. A general framework for weighted gene co-expression network analysis. Stat. Appl. Genet. Mol. Biol. 2005, 4, 17. [Google Scholar] [CrossRef] [PubMed]
- Horvath, S.; Dong, J. Geometric interpretation of gene coexpression network analysis. PLoS Comput. Biol. 2008, 4, e1000117. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.Y.; Lin, Y.C.; Cui, S.; Huang, Y.; Tang, Y.; Xu, J.; Bao, J.; Li, Y.; Wen, J.; Zuo, H.; et al. Mirtarbase update 2022: An informative resource for experimentally validated miRNA-target interactions. Nucleic Acids Res. 2022, 50, D222–D230. [Google Scholar] [CrossRef] [PubMed]
- Thompson, S.L.; Bakhoum, S.F.; Compton, D.A. Mechanisms of chromosomal instability. Curr. Biol. 2010, 20, R285–R295. [Google Scholar] [CrossRef] [PubMed]
- Qin, S.; Xu, L.; Yi, M.; Yu, S.; Wu, K.; Luo, S. Novel immune checkpoint targets: Moving beyond pd-1 and ctla-4. Mol. Cancer 2019, 18, 155. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Xu, Y.; Liu, B. iPiDi-PUL: Identifying Piwi-interacting RNA-disease associations based on positive unlabeled learning. Brief. Bioinform. 2021, 22, bbaa058. [Google Scholar] [CrossRef]
- Chen, J.; Lin, J.; Hu, Y.; Ye, M.; Yao, L.; Wu, L.; Zhang, W.; Wang, M.; Deng, T.; Guo, F.; et al. RNADisease v4.0: An updated resource of RNA-associated diseases, providing RNA-disease analysis, enrichment and prediction. Nucleic Acids Res. 2023, 51, D1397–D1404. [Google Scholar] [CrossRef]
- Guescini, M.; Sisti, D.; Rocchi, M.B.L.; Stocchi, L.; Stocchi, V. A new real-time PCR method to overcome significant quantitative inaccuracy due to slight amplification inhibition. BMC Bioinform. 2008, 30, 326. [Google Scholar] [CrossRef] [PubMed]
- EMA(CHMP). Assessment Report for Halaven Eribulin Procedure No. Emea/h/c/002084; EMA/514195/2011; European Medicines Agency (EMA): London, UK, 2011; Available online: https://www.ema.europa.eu/en/documents/assessment-report/halaven-epar-public-assessment-report_en.pdf (accessed on 10 July 2023).
- Bellio, C.; Emperador, M.; Castellano, P.; Gris-Oliver, A.; Canals, F.; Sanchez-Pla, A.; Zamora, E.; Arribas, J.; Saura, C.; Serra, V.; et al. Gdf15 is an eribulin response biomarker also required for survival of dtp breast cancer cells. Cancers 2022, 14, 2562. [Google Scholar] [CrossRef]
- Oldham, M.C.; Konopka, G.; Iwamoto, K.; Langfelder, P.; Kato, T.; Horvath, S.; Geschwind, D.H. Functional organization of the transcriptome in human brain. Nat. Neurosci. 2008, 11, 1271–1282. [Google Scholar] [CrossRef]
- Giulietti, M.; Occhipinti, G.; Principato, G.; Piva, F. Weighted gene co-expression network analysis reveals key genes involved in pancreatic ductal adenocarcinoma development. Cell. Oncol. 2016, 39, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Giulietti, M.; Occhipinti, G.; Principato, G.; Piva, F. Identification of candidate miRNA biomarkers for pancreatic ductal adenocarcinoma by weighted gene co-expression network analysis. Cell. Oncol. 2017, 40, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Giulietti, M.; Righetti, A.; Principato, G.; Piva, F. LncRNA co-expression network analysis reveals novel biomarkers for pancreatic cancer. Carcinogenesis 2018, 39, 1016–1025. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Wen, J.; Huang, Z.; Chen, X.P.; Zhang, B.X.; Chu, L. Small Nucleolar RNAs: Insight into Their Function in Cancer. Front. Oncol. 2019, 9, 587. [Google Scholar] [CrossRef] [PubMed]
- Pundavela, J.; Roselli, S.; Faulkner, S.; Attia, J.; Scott, R.J.; Thorne, R.F.; Forbes, J.F.; Bradshaw, R.A.; Walker, M.M.; Jobling, P.; et al. Nerve fibers infiltrate the tumor microenvironment and are associated with nerve growth factor production and lymph node invasion in breast cancer. Mol. Oncol. 2015, 9, 1626–1635. [Google Scholar] [CrossRef] [PubMed]
- Demir, I.E.; Reyes, C.M.; Alrawashdeh, W.; Ceyhan, G.O.; Deborde, S.; Friess, H.; Gorgulu, K.; Istvanffy, R.; Jungwirth, D.; Kuner, R.; et al. Clinically actionable strategies for studying neural influences in cancer. Cancer Cell 2020, 38, 11–14. [Google Scholar] [CrossRef]
- Jezequel, P.; Kerdraon, O.; Hondermarck, H.; Guerin-Charbonnel, C.; Lasla, H.; Gouraud, W.; Canon, J.L.; Gombos, A.; Dalenc, F.; Delaloge, S.; et al. Identification of three subtypes of triple-negative breast cancer with potential therapeutic implications. Breast Cancer Res. 2019, 21, 65. [Google Scholar] [CrossRef] [PubMed]
- Abu Rmaileh, A.; Solaimuthu, B.; Khatib, A.; Lavi, S.; Tanna, M.; Hayashi, A.; Ben Yosef, M.; Lichtenstein, M.; Pillar, N.; Shaul, Y.D. Dpysl2 interacts with jak1 to mediate breast cancer cell migration. J. Cell Biol. 2022, 221, e202106078. [Google Scholar] [CrossRef]
- Harburg, G.C.; Hinck, L. Navigating breast cancer: Axon guidance molecules as breast cancer tumor suppressors and oncogenes. J. Mammary Gland. Biol. Neoplasia 2011, 16, 257–270. [Google Scholar] [CrossRef]
- Winkler, J.; Abisoye-Ogunniyan, A.; Metcalf, K.J.; Werb, Z. Concepts of extracellular matrix remodelling in tumour progression and metastasis. Nat. Commun. 2020, 11, 5120. [Google Scholar] [CrossRef] [PubMed]
- Cox, T.R. The matrix in cancer. Nat. Rev. Cancer 2021, 21, 217–238. [Google Scholar] [CrossRef] [PubMed]
- Nallanthighal, S.; Heiserman, J.P.; Cheon, D.J. The role of the extracellular matrix in cancer stemness. Front. Cell Dev. Biol. 2019, 7, 86. [Google Scholar] [CrossRef] [PubMed]
- Mendonsa, A.M.; Na, T.Y.; Gumbiner, B.M. E-cadherin in contact inhibition and cancer. Oncogene 2018, 37, 4769–4780. [Google Scholar] [CrossRef] [PubMed]
- Moh, M.C.; Shen, S. The roles of cell adhesion molecules in tumor suppression and cell migration: A new paradox. Cell Adhes. Migr. 2009, 3, 334–336. [Google Scholar] [CrossRef]
- Harjunpaa, H.; Llort Asens, M.; Guenther, C.; Fagerholm, S.C. Cell adhesion molecules and their roles and regulation in the immune and tumor microenvironment. Front. Immunol. 2019, 10, 1078. [Google Scholar] [CrossRef]
- Kaul, R.; Risinger, A.L.; Mooberry, S.L. Eribulin rapidly inhibits tgf-beta-induced snail expression and can induce slug expression in a smad4-dependent manner. Br. J. Cancer 2019, 121, 611–621. [Google Scholar] [CrossRef]
- Snaebjornsson, M.T.; Janaki-Raman, S.; Schulze, A. Greasing the wheels of the cancer machine: The role of lipid metabolism in cancer. Cell Metab. 2020, 31, 62–76. [Google Scholar] [CrossRef]
- Mashima, T.; Sato, S.; Okabe, S.; Miyata, S.; Matsuura, M.; Sugimoto, Y.; Tsuruo, T.; Seimiya, H. Acyl-coa synthetase as a cancer survival factor: Its inhibition enhances the efficacy of etoposide. Cancer Sci. 2009, 100, 1556–1562. [Google Scholar] [CrossRef]
- Beloribi-Djefaflia, S.; Vasseur, S.; Guillaumond, F. Lipid metabolic reprogramming in cancer cells. Oncogenesis 2016, 5, e189. [Google Scholar] [CrossRef]
- Simeone, P.; Tacconi, S.; Longo, S.; Lanuti, P.; Bravaccini, S.; Pirini, F.; Ravaioli, S.; Dini, L.; Giudetti, A.M. Expanding roles of de novo lipogenesis in breast cancer. Int. J. Environ. Res. Public Health 2021, 18, 3575. [Google Scholar] [CrossRef]
- Nabekura, T.; Kawasaki, T.; Jimura, M.; Mizuno, K.; Uwai, Y. Microtubule-targeting anticancer drug eribulin induces drug efflux transporter p-glycoprotein. Biochem. Biophys. Rep. 2020, 21, 100727. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Seletsky, B.M.; Palme, M.H.; Lydon, P.J.; Singer, L.A.; Chase, C.E.; Lemelin, C.A.; Shen, Y.; Davis, H.; Tremblay, L.; et al. Macrocyclic ketone analogues of halichondrin b. Bioorg. Med. Chem. Lett. 2004, 14, 5551–5554. [Google Scholar] [CrossRef] [PubMed]
- Taur, J.S.; DesJardins, C.S.; Schuck, E.L.; Wong, Y.N. Interactions between the chemotherapeutic agent eribulin mesylate (e7389) and p-glycoprotein in cf-1 abcb1a-deficient mice and caco-2 cells. Xenobiotica 2011, 41, 320–326. [Google Scholar] [CrossRef] [PubMed]
- Oba, T.; Izumi, H.; Ito, K.I. Abcb1 and abcc11 confer resistance to eribulin in breast cancer cell lines. Oncotarget 2016, 7, 70011–70027. [Google Scholar] [CrossRef]
- Nemcova-Furstova, V.; Kopperova, D.; Balusikova, K.; Ehrlichova, M.; Brynychova, V.; Vaclavikova, R.; Daniel, P.; Soucek, P.; Kovar, J. Characterization of acquired paclitaxel resistance of breast cancer cells and involvement of ABC transporters. Toxicol. Appl. Pharmacol. 2016, 310, 215–228. [Google Scholar] [CrossRef] [PubMed]
- Hansen, S.N.; Westergaard, D.; Thomsen, M.B.; Vistesen, M.; Do, K.N.; Fogh, L.; Belling, K.C.; Wang, J.; Yang, H.; Gupta, R.; et al. Acquisition of docetaxel resistance in breast cancer cells reveals upregulation of abcb1 expression as a key mediator of resistance accompanied by discrete upregulation of other specific genes and pathways. Tumour Biol. 2015, 36, 4327–4338. [Google Scholar] [CrossRef]
- Juraszek, B.; Nalecz, K.A. Slc22a5 (octn2) carnitine transporter-indispensable for cell metabolism, a jekyll and hyde of human cancer. Molecules 2019, 25, 14. [Google Scholar] [CrossRef]
- Garcia-Closas, M.; Rothman, N.; Figueroa, J.D.; Prokunina-Olsson, L.; Han, S.S.; Baris, D.; Jacobs, E.J.; Malats, N.; De Vivo, I.; Albanes, D.; et al. Common genetic polymorphisms modify the effect of smoking on absolute risk of bladder cancer. Cancer Res. 2013, 73, 2211–2220. [Google Scholar] [CrossRef]
- Rajput, S.; Guo, Z.; Li, S.; Ma, C.X. Pi3k inhibition enhances the anti-tumor effect of eribulin in triple negative breast cancer. Oncotarget 2019, 10, 3667–3680. [Google Scholar] [CrossRef]
- Gris-Oliver, A.; Ibrahim, Y.H.; Rivas, M.A.; Garcia-Garcia, C.; Sanchez-Guixe, M.; Ruiz-Pace, F.; Viaplana, C.; Perez-Garcia, J.M.; Llombart-Cussac, A.; Grueso, J.; et al. Pi3k activation promotes resistance to eribulin in her2-negative breast cancer. Br. J. Cancer 2021, 124, 1581–1591. [Google Scholar] [CrossRef]
- Tanaka, S.; Ishii, T.; Sato, F.; Toi, M.; Itou, J. Eribulin mesylate-induced c-fos upregulation enhances cell survival in breast cancer cell lines. Biochem. Biophys. Res. Commun. 2020, 526, 154–157. [Google Scholar] [CrossRef]
- Liu, Y.; Dou, M.; Song, X.; Dong, Y.; Liu, S.; Liu, H.; Tao, J.; Li, W.; Yin, X.; Xu, W. The emerging role of the piRNA/piwi complex in cancer. Mol. Cancer 2019, 18, 123. [Google Scholar] [CrossRef] [PubMed]
- Guo, B.; Li, D.; Du, L.; Zhu, X. PiRNAs: Biogenesis and their potential roles in cancer. Cancer Metastasis Rev. 2020, 39, 567–575. [Google Scholar] [CrossRef] [PubMed]
- Weng, W.; Li, H.; Goel, A. Piwi-interacting RNAs (piRNAs) and cancer: Emerging biological concepts and potential clinical implications. Biochim. Biophys. Acta Rev. Cancer 2019, 1871, 160–169. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.; Hu, H.; Xue, X.; Shen, S.; Gao, E.; Guo, G.; Shen, X.; Zhang, X. Altered expression of piRNAs and their relation with clinicopathologic features of breast cancer. Clin. Transl. Oncol. 2013, 15, 563–568. [Google Scholar] [CrossRef]
- Wang, Y.; Weng, Q.; Ge, J.; Zhang, X.; Guo, J.; Ye, G. TRNA-derived small RNAs: Mechanisms and potential roles in cancers. Genes. Dis. 2022, 9, 1431–1442. [Google Scholar] [CrossRef]
- Zhang, Y.; Qian, H.; He, J.; Gao, W. Mechanisms of tRNA-derived fragments and tRNA halves in cancer treatment resistance. Biomark. Res. 2020, 8, 52. [Google Scholar] [CrossRef]
- Sun, C.; Huang, X.; Li, J.; Fu, Z.; Hua, Y.; Zeng, T.; He, Y.; Duan, N.; Yang, F.; Liang, Y.; et al. Exosome-transmitted trf-16-k8j7k1b promotes tamoxifen resistance by reducing drug-induced cell apoptosis in breast cancer. Cancers 2023, 15, 899. [Google Scholar] [CrossRef]
- Farina, N.H.; Scalia, S.; Adams, C.E.; Hong, D.; Fritz, A.J.; Messier, T.L.; Balatti, V.; Veneziano, D.; Lian, J.B.; Croce, C.M.; et al. Identification of tRNA-derived small RNA (tsRNA) responsive to the tumor suppressor, runx1, in breast cancer. J. Cell Physiol. 2020, 235, 5318–5327. [Google Scholar] [CrossRef]
- Krishnan, P.; Ghosh, S.; Wang, B.; Heyns, M.; Graham, K.; Mackey, J.R.; Kovalchuk, O.; Damaraju, S. Profiling of small nucleolar RNAs by next generation sequencing: Potential new players for breast cancer prognosis. PLoS ONE 2016, 11, e0162622. [Google Scholar] [CrossRef]
- Uhr, K.; Prager-van der Smissen, W.J.C.; Heine, A.A.J.; Ozturk, B.; van Jaarsveld, M.T.M.; Boersma, A.W.M.; Jager, A.; Wiemer, E.A.C.; Smid, M.; Foekens, J.A.; et al. MicroRNAs as possible indicators of drug sensitivity in breast cancer cell lines. PLoS ONE 2019, 14, e0216400. [Google Scholar] [CrossRef] [PubMed]
- Bockhorn, J.; Dalton, R.; Nwachukwu, C.; Huang, S.; Prat, A.; Yee, K.; Chang, Y.F.; Huo, D.; Wen, Y.; Swanson, K.E.; et al. MicroRNA-30c inhibits human breast tumour chemotherapy resistance by regulating twf1 and il-11. Nat. Commun. 2013, 4, 1393. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Fu, Y.; Chen, L.; Lee, W.; Lai, Y.; Rezaei, K.; Tabbara, S.; Latham, P.; Teal, C.B.; Man, Y.G.; et al. Mir-671-5p inhibits epithelial-to-mesenchymal transition by downregulating foxm1 expression in breast cancer. Oncotarget 2016, 7, 293–307. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Zhang, Y.; Jasper, J.; Lykken, E.; Alexander, P.B.; Markowitz, G.J.; McDonnell, D.P.; Li, Q.J.; Wang, X.F. Mir-148a functions to suppress metastasis and serves as a prognostic indicator in triple-negative breast cancer. Oncotarget 2016, 7, 20381–20394. [Google Scholar] [CrossRef] [PubMed]
- Melo, S.A.; Sugimoto, H.; O’Connell, J.T.; Kato, N.; Villanueva, A.; Vidal, A.; Qiu, L.; Vitkin, E.; Perelman, L.T.; Melo, C.A.; et al. Cancer exosomes perform cell-independent microRNA biogenesis and promote tumorigenesis. Cancer Cell 2014, 26, 707–721. [Google Scholar] [CrossRef]
- Pasculli, B.; Barbano, R.; Fontana, A.; Biagini, T.; Di Viesti, M.P.; Rendina, M.; Valori, V.M.; Morritti, M.; Bravaccini, S.; Ravaioli, S.; et al. Hsa-mir-155-5p up-regulation in breast cancer and its relevance for treatment with poly[adp-ribose] polymerase 1 (parp-1) inhibitors. Front. Oncol. 2020, 10, 1415. [Google Scholar] [CrossRef]
- Xu, W.; Song, C.; Wang, X.; Li, Y.; Bai, X.; Liang, X.; Wu, J.; Liu, J. Downregulation of mir-155-5p enhances the anti-tumor effect of cetuximab on triple-negative breast cancer cells via inducing cell apoptosis and pyroptosis. Aging 2021, 13, 228–240. [Google Scholar] [CrossRef]
- Zhou, M.; Liu, Z.; Zhao, Y.; Ding, Y.; Liu, H.; Xi, Y.; Xiong, W.; Li, G.; Lu, J.; Fodstad, O.; et al. MicroRNA-125b confers the resistance of breast cancer cells to paclitaxel through suppression of pro-apoptotic bcl-2 antagonist killer 1 (bak1) expression. J. Biol. Chem. 2010, 285, 21496–21507. [Google Scholar] [CrossRef]
- Yang, Q.; Wang, Y.; Lu, X.; Zhao, Z.; Zhu, L.; Chen, S.; Wu, Q.; Chen, C.; Wang, Z. Mir-125b regulates epithelial-mesenchymal transition via targeting sema4c in paclitaxel-resistant breast cancer cells. Oncotarget 2015, 6, 3268–3279. [Google Scholar] [CrossRef]
- Yu, X.; Luo, A.; Liu, Y.; Wang, S.; Li, Y.; Shi, W.; Liu, Z.; Qu, X. Mir-214 increases the sensitivity of breast cancer cells to tamoxifen and fulvestrant through inhibition of autophagy. Mol. Cancer 2015, 14, 208. [Google Scholar] [CrossRef]
- Fong, M.Y.; Zhou, W.; Liu, L.; Alontaga, A.Y.; Chandra, M.; Ashby, J.; Chow, A.; O’Connor, S.T.; Li, S.; Chin, A.R.; et al. Breast-cancer-secreted mir-122 reprograms glucose metabolism in premetastatic niche to promote metastasis. Nat. Cell Biol. 2015, 17, 183–194. [Google Scholar] [CrossRef] [PubMed]
- Ferragut Cardoso, A.P.; Banerjee, M.; Nail, A.N.; Lykoudi, A.; States, J.C. MiRNA dysregulation is an emerging modulator of genomic instability. Semin. Cancer Biol. 2021, 76, 120–131. [Google Scholar] [CrossRef] [PubMed]
- Chevillet, J.R.; Kang, Q.; Ruf, I.K.; Briggs, H.A.; Vojtech, L.N.; Hughes, S.M.; Cheng, H.H.; Arroyo, J.D.; Meredith, E.K.; Gallichotte, E.N.; et al. Quantitative and stoichiometric analysis of the microRNA content of exosomes. Proc. Natl. Acad. Sci. USA 2014, 111, 14888–14893. [Google Scholar] [CrossRef] [PubMed]
- Luo, T.; Chen, S.Y.; Qiu, Z.X.; Miao, Y.R.; Ding, Y.; Pan, X.Y.; Li, Y.; Lei, Q.; Guo, A.Y. Transcriptomic features in a single extracellular vesicle via single-cell RNA sequencing. Small Methods 2022, 6, e2200881. [Google Scholar] [CrossRef]

| Gene Name | Category | log2 (Fold-Change) | p Adjusted |
|---|---|---|---|
| AQP4-AS1 | lncRNA | 7.59 | 2.71 × 10−8 |
| AC002463.3 | lncRNA | 7.39 | 1.02 × 10−7 |
| GABRG3 | protein_coding | 7.36 | 1.31 × 10−7 |
| CASS4 | protein_coding | 7.18 | 4.00 × 10−7 |
| NEFM | protein_coding | 6.94 | 2.53 × 10−7 |
| MLIP | protein_coding | 6.87 | 4.40 × 10−7 |
| CALN1 | protein_coding | 6.61 | 5.66 × 10−8 |
| FER1L5 | protein_coding | 6.59 | 4.47 × 10−8 |
| OTOG | protein_coding | 6.55 | 7.29 × 10−8 |
| CELF2-AS2 | lncRNA | 6.45 | 1.49 × 10−7 |
| ATP10B | protein_coding | 6.39 | 1.63 × 10−9 |
| RP11-799O21.2 | lncRNA | 6.37 | 2.70 × 10−7 |
| FAM83F | protein_coding | 6.36 | 2.54 × 10−7 |
| TDRD12 | protein_coding | 6.36 | 2.82 × 10−7 |
| RP4-715N11.2 | lncRNA | 6.35 | 3.15 × 10−7 |
| POU2F3 | protein_coding | 6.35 | 3.02 × 10−7 |
| GRM8 | protein_coding | 6.34 | 3.00 × 10−9 |
| NID2 | protein_coding | 6.33 | 3.84 × 10−7 |
| LINC02822 | lncRNA | 6.33 | 4.40 × 10−7 |
| MMP16 | protein_coding | 6.32 | 3.76 × 10−7 |
| H3C10 | protein_coding | −3.22 | 5.04 × 10−21 |
| ANP32B | protein_coding | −3.22 | 5.72 × 10−117 |
| TP53INP2 | protein_coding | −3.24 | 1.82 × 10−39 |
| IGIP | protein_coding | −3.30 | 5.91 × 10−9 |
| BCL2L2 | protein_coding | −3.33 | 1.19 × 10−33 |
| NUDT16 | protein_coding | −3.43 | 1.84 × 10−55 |
| SH3PXD2A | protein_coding | −3.43 | 2.76 × 10−96 |
| CRTAP | protein_coding | −3.44 | 9.71 × 10−143 |
| H4C11 | protein_coding | −3.49 | 2.83 × 10−5 |
| PGPEP1 | protein_coding | −3.54 | 7.71 × 10−73 |
| ZSWIM9 | protein_coding | −3.63 | 5.21 × 10−47 |
| C22orf46 | transcribed unitary pseudogene | −3.65 | 3.46 × 10−167 |
| CTD-3099C6.13 | unknown | −3.72 | 8.58 × 10−120 |
| FBXW4 | protein_coding | −3.74 | 2.42 × 10−50 |
| RAB13 | protein_coding | −3.89 | 2.47 × 10−96 |
| RASSF3 | protein_coding | −3.97 | 9.52 × 10−168 |
| KIF1C | protein_coding | −4.05 | 0.00 × 10−0 |
| TRAK2 | protein_coding | −4.17 | 5.02 × 10−249 |
| NET1 | protein_coding | −4.43 | 0.00 × 10 |
| RP11-603J24.7 | processed_pseudogene | −4.46 | 3.44 × 10−95 |
| Gene Name | Category | log2 (FoldChange) | p Adjusted |
|---|---|---|---|
| SNORD98 | snoRNA | 4.03 | 1.95 × 10−3 |
| SNORA47 | snoRNA | 3.55 | 2.34 × 10−11 |
| SNORD71 | snoRNA | 3.25 | 2.83 × 10−8 |
| SNORD12C | snoRNA | 3.13 | 3.25 × 10−3 |
| SNORD28 | snoRNA | 3.07 | 2.34 × 10−11 |
| piR-hsa-23566 | piRNA | 3.01 | 1.54 × 10−5 |
| SNORD37 | snoRNA | 2.87 | 4.50 × 10−4 |
| SNORD90 | snoRNA | 2.72 | 4.61 × 10−3 |
| SNORD18B | snoRNA | 2.71 | 6.04 × 10−3 |
| SNORD127 | snoRNA | 2.67 | 1.26 × 10−2 |
| SNORA62 | snoRNA | 2.55 | 2.61 × 10−10 |
| SNORD19B | snoRNA | 2.48 | 2.94 × 10−2 |
| SNORD83A | snoRNA | 2.35 | 3.60 × 10−8 |
| piR-hsa-1834 | piRNA | 2.34 | 1.15 × 10−2 |
| SNORD46 | snoRNA | 2.33 | 1.39 × 10−4 |
| piR-hsa-26039 | piRNA | 2.32 | 1.01 × 10−5 |
| SNORD34 | snoRNA | 2.27 | 3.36 × 10−2 |
| SNORD78 | snoRNA | 2.20 | 1.64 × 10−7 |
| SNORD123 | snoRNA | 2.18 | 1.40 × 10−4 |
| SNORD49A | snoRNA | 2.15 | 1.54 × 10−5 |
| piR-hsa-27513 | piRNA | −4.53 | 7.26 × 10−4 |
| tRFdb-3004a-617 | tRNA-derived ncRNA | −4.73 | 4.84 × 10−5 |
| hsa-miR-155-5p | miRNA | −4.82 | 7.75 × 10−6 |
| ts-112 | tRNA-derived ncRNA | −4.83 | 1.59 × 10−5 |
| piR-hsa-7116 | piRNA | −4.91 | 7.12 × 10−6 |
| piR-hsa-28382 | piRNA | −5.09 | 1.18 × 10−4 |
| piR-hsa-17793 | piRNA | −5.32 | 8.62 × 10−4 |
| piR-hsa-2467 | piRNA | −5.49 | 3.59 × 10−5 |
| piR-hsa-28205 | piRNA | −5.76 | 1.09 × 10−4 |
| 5P_tRNA-His-GTG-1-8 | tRNA-derived ncRNA | −6.28 | 4.44 × 10−5 |
| piR-hsa-28478 | piRNA | −6.29 | 5.08 × 10−4 |
| piR-hsa−5939 | piRNA | −6.56 | 3.80 × 10−7 |
| piR-hsa-25046 | piRNA | −6.67 | 7.45 × 10−8 |
| ts-44 | tRNA-derived ncRNA | −7.33 | 1.02 × 10−5 |
| piR-hsa-12789 | piRNA | −7.42 | 1.48 × 10−4 |
| hsa-miR-6087 | miRNA | −7.99 | 2.68 × 10−5 |
| hsa-miR-1306-5p | miRNA | −8.19 | 5.79 × 10−16 |
| hsa-miR-196b-5p | miRNA | −8.45 | 2.07 × 10−5 |
| piR-hsa-9105 | piRNA | −9.15 | 1.64 × 10−7 |
| piR-hsa-12275 | piRNA | −10.76 | 2.15 × 10−7 |
| Gene Symbol | Gene Name | Module | GS Score | Functions * |
|---|---|---|---|---|
| PXDN | Peroxidasin | turquoise | 0.9949 | Peroxidase secreted into the extracellular matrix, contributes to the collagen IV network-dependent fibronectin/FN and laminin assembly, which is required for full extracellular matrix (ECM)-mediated signaling. |
| PVT1 | Pvt1 oncogene | turquoise | 0.9946 | LncRNA oncogene, also in breast cancer. It also promotes extracellular matrix degradation (PMID: 35399100) |
| WDR27 | WD repeat domain 27 | turquoise | 0.9944 | Scaffolding for proteins |
| SLC22A5 | Solute carrier family 22 member 5 | turquoise | 0.9944 | Carnitine and polyspecific organic cation transporter, critical for elimination of many endogenous cations, toxins and drugs, including etoposide, oxaliplatin, imatinib, vincristine, vinblastine, paclitaxel, sunitinib, vinorelbine, cisplatin, oxaliplatin (PMID: 31861504) |
| MYO1G | Myosin IG | turquoise | 0.9943 | Unconventional myosin required during immune response |
| LDLRAD4 | Low density lipoprotein receptor class A domain containing 4 | turquoise | 0.9941 | Involved in negative regulation of cell migration; negative regulation of epithelial to mesenchymal transition. Functions as a negative regulator of TGF-beta signaling and thereby probably plays a role in cell proliferation, differentiation, apoptosis, motility, extracellular matrix production and immunosuppression. |
| CAPN11 | Calpain 11 | turquoise | 0.9941 | Calcium-dependent cysteine proteases of substrates involved in cytoskeletal remodeling and signal transduction |
| PHIP | Pleckstrin homology domain interacting protein | turquoise | 0.9933 | Regulates glucose transporter translocation |
| KTN1-AS1 | KTN1 antisense RNA 1 | turquoise | 0.9929 | LncRNA |
| DSE | Dermatan sulfate epimerase | turquoise | 0.9929 | Biosynthesis of the dermatan sulfate, a component of extracellular matrix (ECM) |
| TRIOBP | TRIO and F-actin binding protein | blue | 0.9999 | Associates with and stabilizes F-actin structures |
| KIF1C | Kinesin family member 1C | blue | 0.9999 | Microtubule-dependent molecular motor that transport organelles and move chromosomes during cell division |
| NET1 | Neuroepithelial cell transforming 1 | blue | 0.9999 | Guanine nucleotide exchange factor (GEF) |
| MAP4 | Microtubule associated protein 4 | blue | 0.9999 | Microtubule-associated protein, promotes microtubule assembly |
| CDA | Cytidine deaminase | blue | 0.9999 | Enzyme involved in pyrimidine salvaging |
| ABLIM3 | Actin binding LIM protein family member 3 | blue | 0.9998 | Interacts with actin filaments and is a component of adherens junctions |
| CARD19 | Caspase recruitment domain family member 19 | blue | 0.9998 | Mitochondrial protein |
| ACTG1 | Actin gamma 1 | blue | 0.9998 | Actin gamma |
| TCEAL3 | Transcription elongation factor A like 3 | blue | 0.9998 | Transcription elongation factor |
| TOMM20 | Translocase of outer mitochondrial membrane 20 | blue | 0.9998 | Mitochondrial import receptor |
| Up-Regulated miRNAs in EVs after Eribulin-Treatment | Validated Targets | Role * |
|---|---|---|
| hsa-let-7a-5p | AURKA | CIN |
| hsa-miR-29a-3p | KLF4 | CIN |
| hsa-miR-29a-3p | MDM2 | CIN |
| Down-Regulated miRNAs in EVs after Eribulin-Treatment | Validated Targets | Role |
| hsa-miR-125a-5p | TP53 | CIN |
| hsa-miR-144-3p | FBXW7 | CIN |
| hsa-miR-155-5p | APC | CIN |
| hsa-miR-214-3p | CTNNB1 | CIN |
| hsa-miR-214-3p | ABCB1 (MDR1, P-glycoprotein) | MDR |
| hsa-miR-214-3p | CD274 (PD-L1) | IMMUNE |
| hsa-miR-328-3p | ABCG2 (BCRP) | MDR |
| hsa-miR-381-3p | ID1 | CIN |
| hsa-miR-451a | ABCB1 (MDR1, P-glycoprotein) | MDR |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giulietti, M.; Piva, F.; Cecati, M.; Maggio, S.; Guescini, M.; Saladino, T.; Scortichini, L.; Crocetti, S.; Caramanti, M.; Battelli, N.; et al. Effects of Eribulin on the RNA Content of Extracellular Vesicles Released by Metastatic Breast Cancer Cells. Cells 2024, 13, 479. https://doi.org/10.3390/cells13060479
Giulietti M, Piva F, Cecati M, Maggio S, Guescini M, Saladino T, Scortichini L, Crocetti S, Caramanti M, Battelli N, et al. Effects of Eribulin on the RNA Content of Extracellular Vesicles Released by Metastatic Breast Cancer Cells. Cells. 2024; 13(6):479. https://doi.org/10.3390/cells13060479
Chicago/Turabian StyleGiulietti, Matteo, Francesco Piva, Monia Cecati, Serena Maggio, Michele Guescini, Tiziana Saladino, Laura Scortichini, Sonia Crocetti, Miriam Caramanti, Nicola Battelli, and et al. 2024. "Effects of Eribulin on the RNA Content of Extracellular Vesicles Released by Metastatic Breast Cancer Cells" Cells 13, no. 6: 479. https://doi.org/10.3390/cells13060479
APA StyleGiulietti, M., Piva, F., Cecati, M., Maggio, S., Guescini, M., Saladino, T., Scortichini, L., Crocetti, S., Caramanti, M., Battelli, N., & Romagnoli, E. (2024). Effects of Eribulin on the RNA Content of Extracellular Vesicles Released by Metastatic Breast Cancer Cells. Cells, 13(6), 479. https://doi.org/10.3390/cells13060479









