Long-Term Treatment with Gadopentetic Acid or Gadodiamide Increases TRPC5 Expression and Decreases Adriamycin Nuclear Accumulation in Breast Cancer Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture and Transfection
2.2. Patch-Clamp Electrophysiology
2.3. Solutions
2.4. Cell Counting Kit-8 Assay
2.5. Immunofluorescence
2.6. Quantitative Real-Time Polymerase Chain Reaction
2.7. The ADM Accumulation Assay
2.8. Calcium Imaging
2.9. Western Blot
2.10. Drugs
2.11. Statistical Methods
3. Results
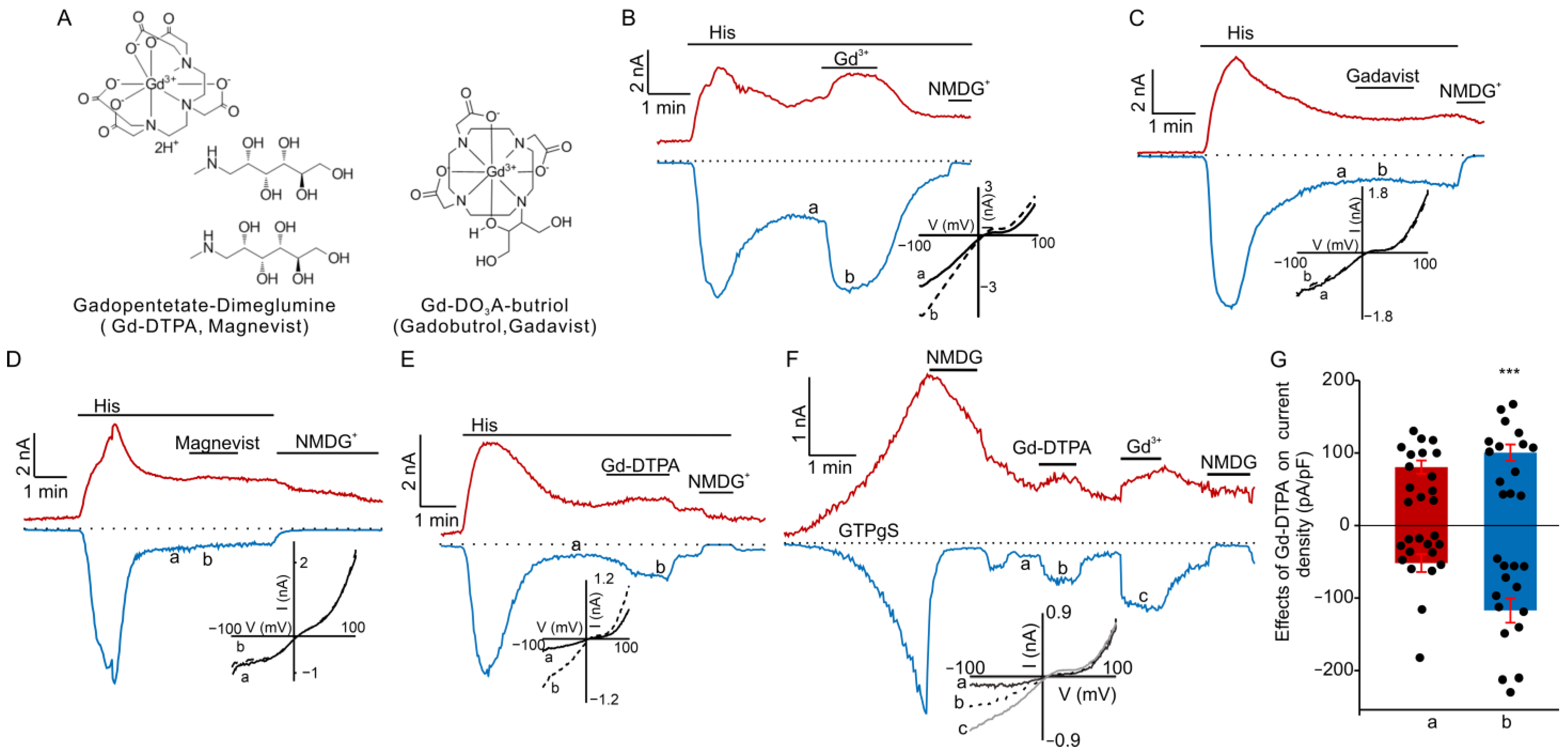
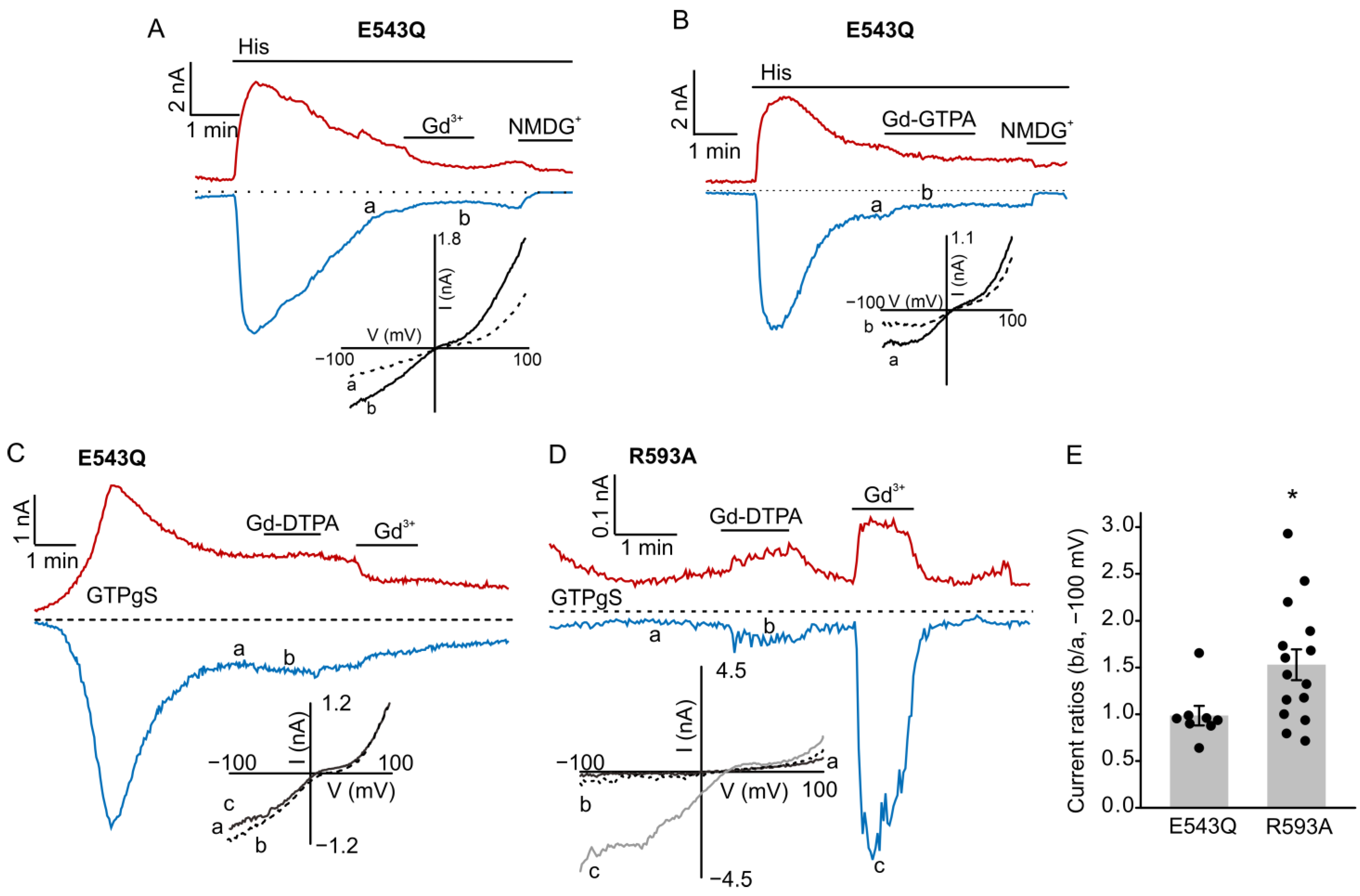
3.1. Gd-DTPA Potentiated TRPC5 Currents in HEK Cells
3.2. Effect of GBCAs on MCF-7, SK-BR-3, and MCF-7 /ADM Cell Viability in the Presence of ADM
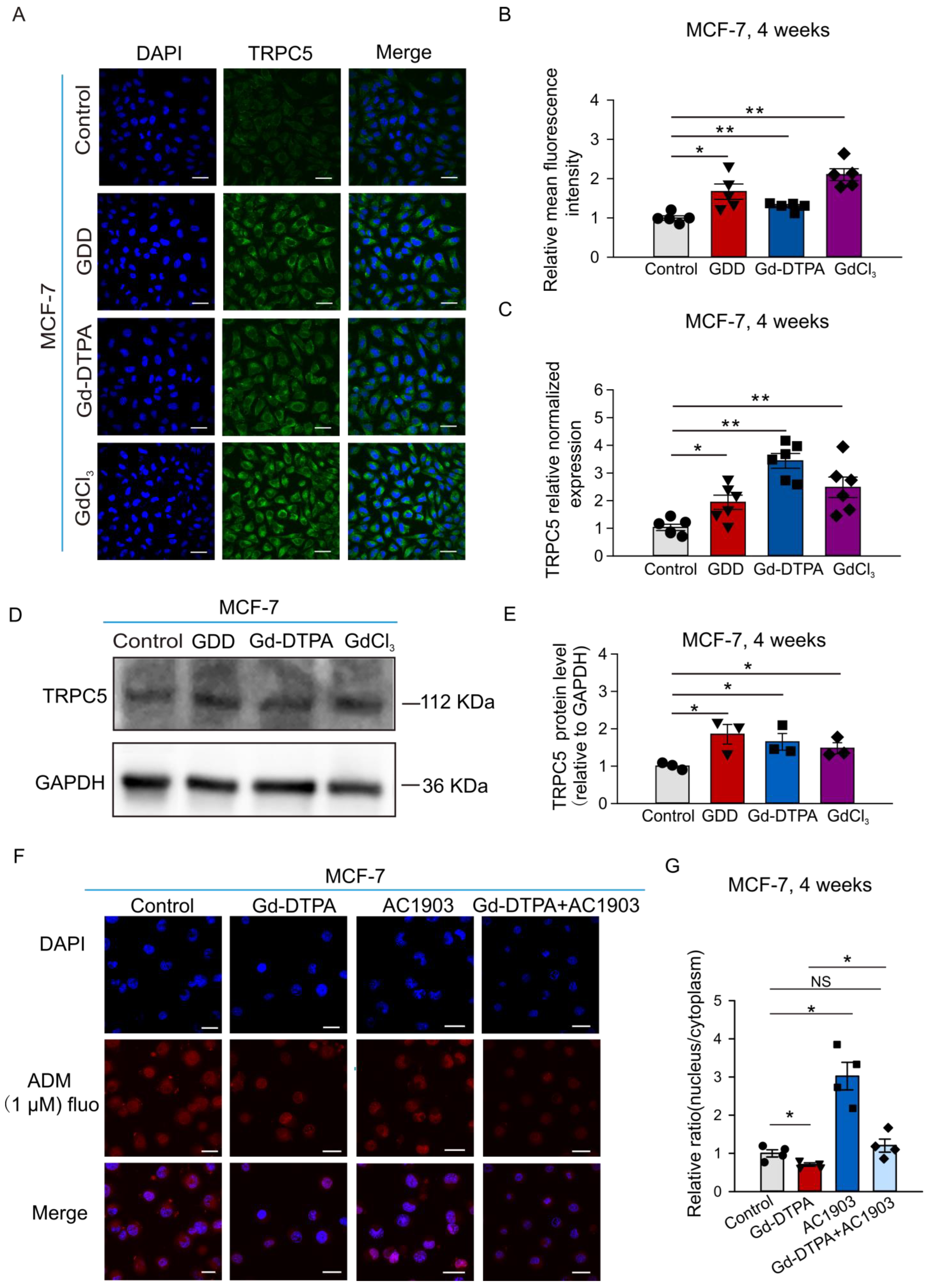
3.3. The Nuclear Accumulation of ADM Was Decreased in the MCF-7 Cells Treated with GCBAs for 4 Weeks
3.4. The Expression of TRPC5 Was Increased after Treatment with GCBAs, and the Inhibition of TRPC5 Increased ADM Nuclear Accumulation
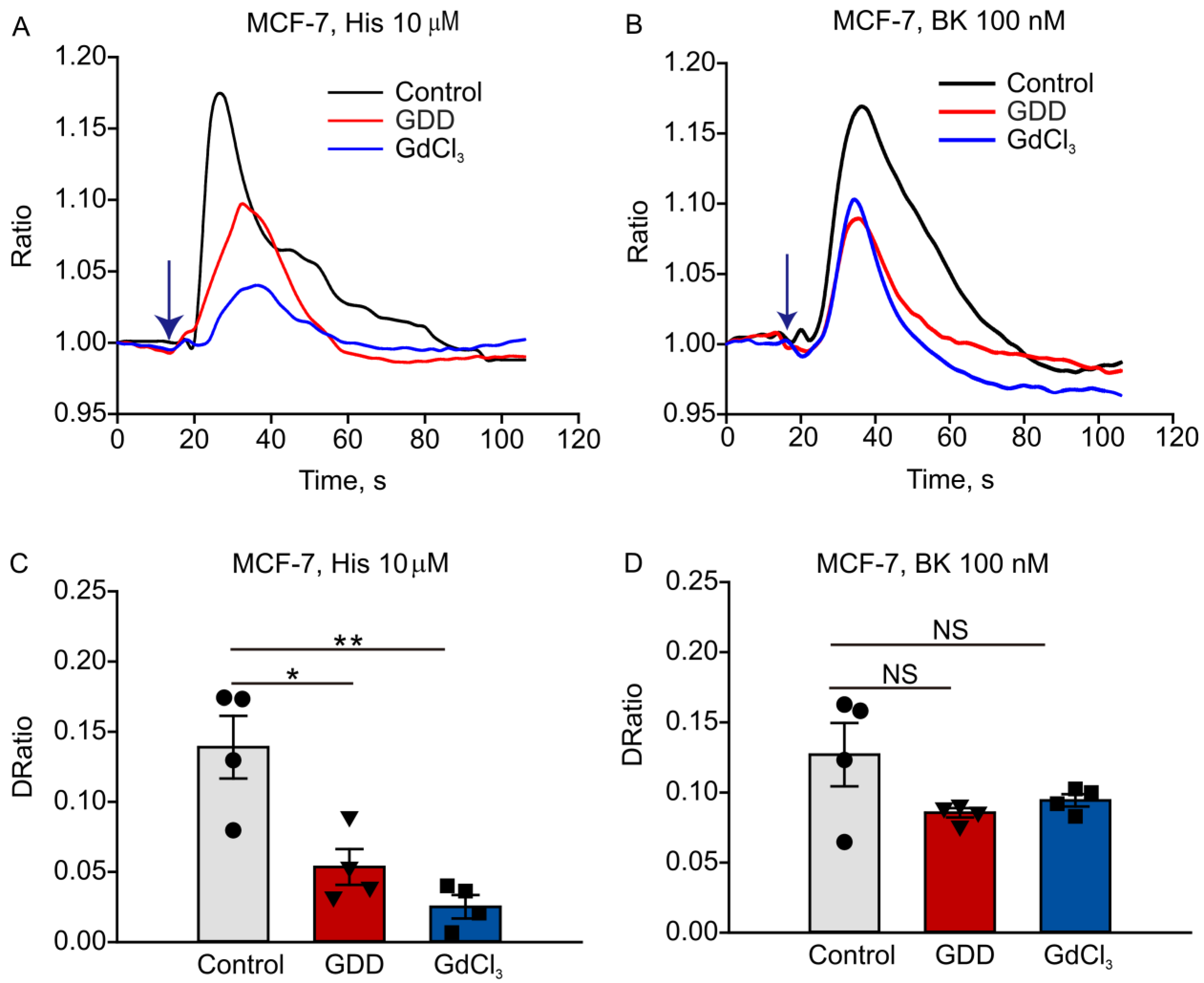
3.5. Long-Term Treatment with GDD Decreased Intracellular Calcium Transients Induced by Histamine and Bradykinin in MCF-7 Cells
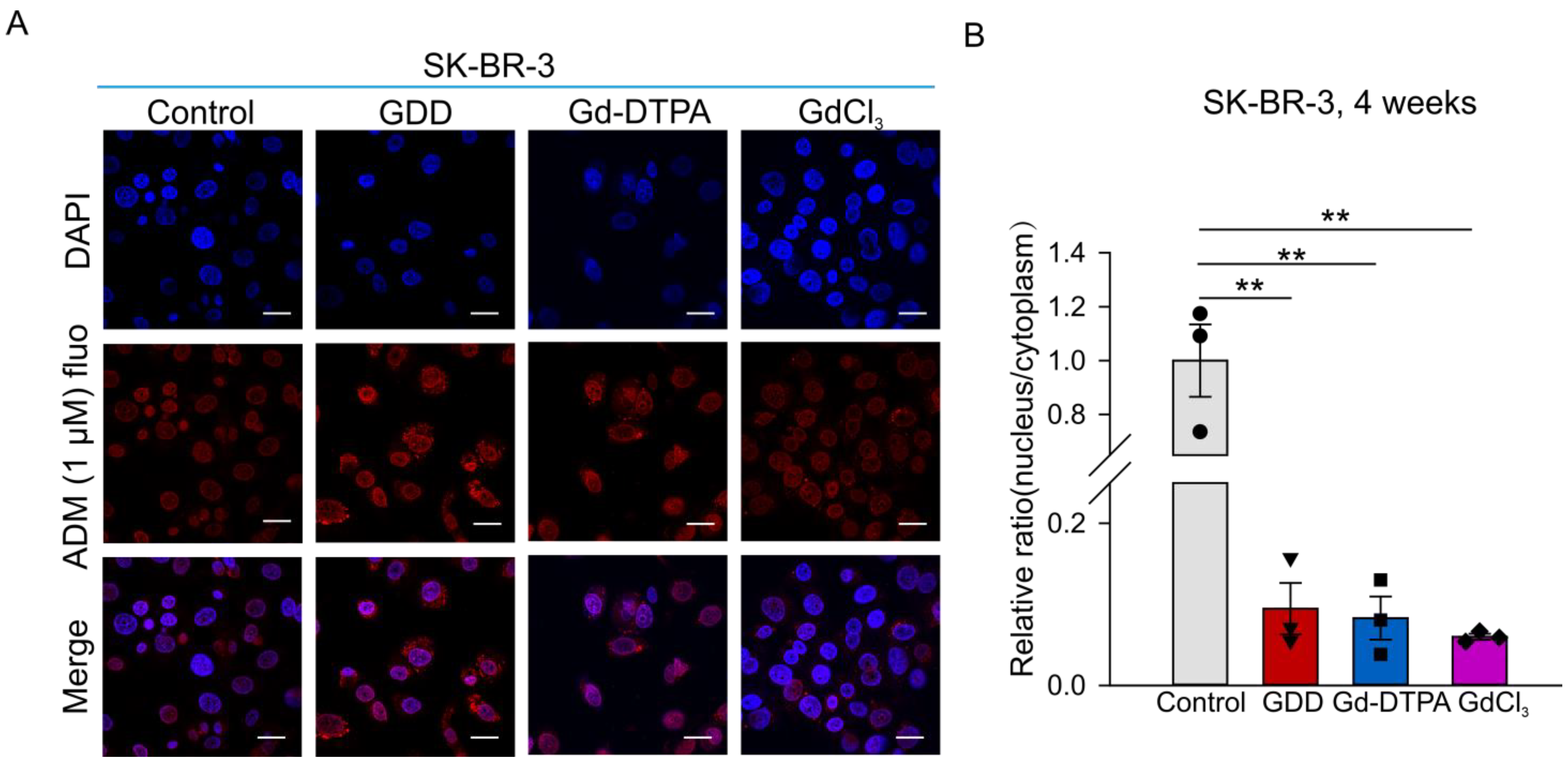
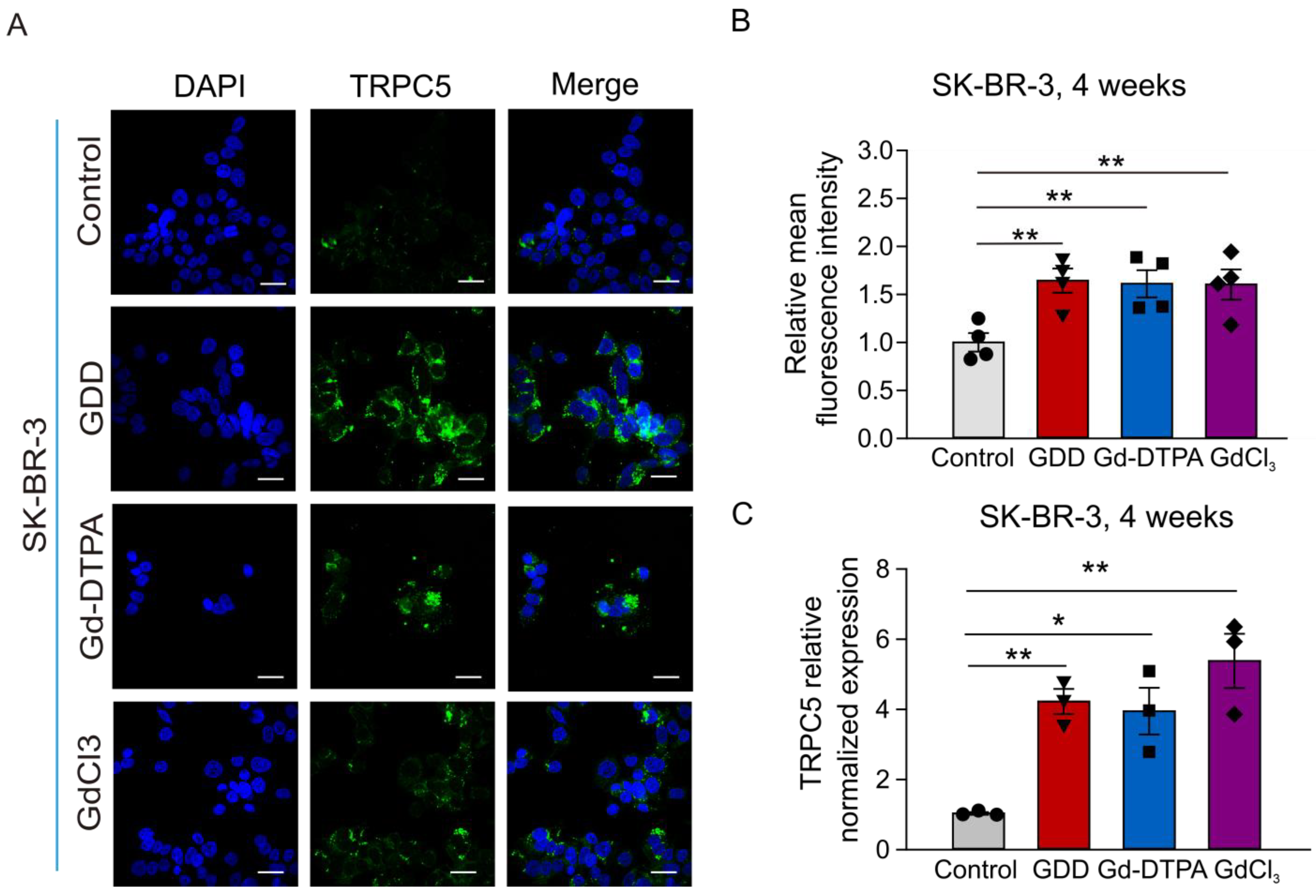
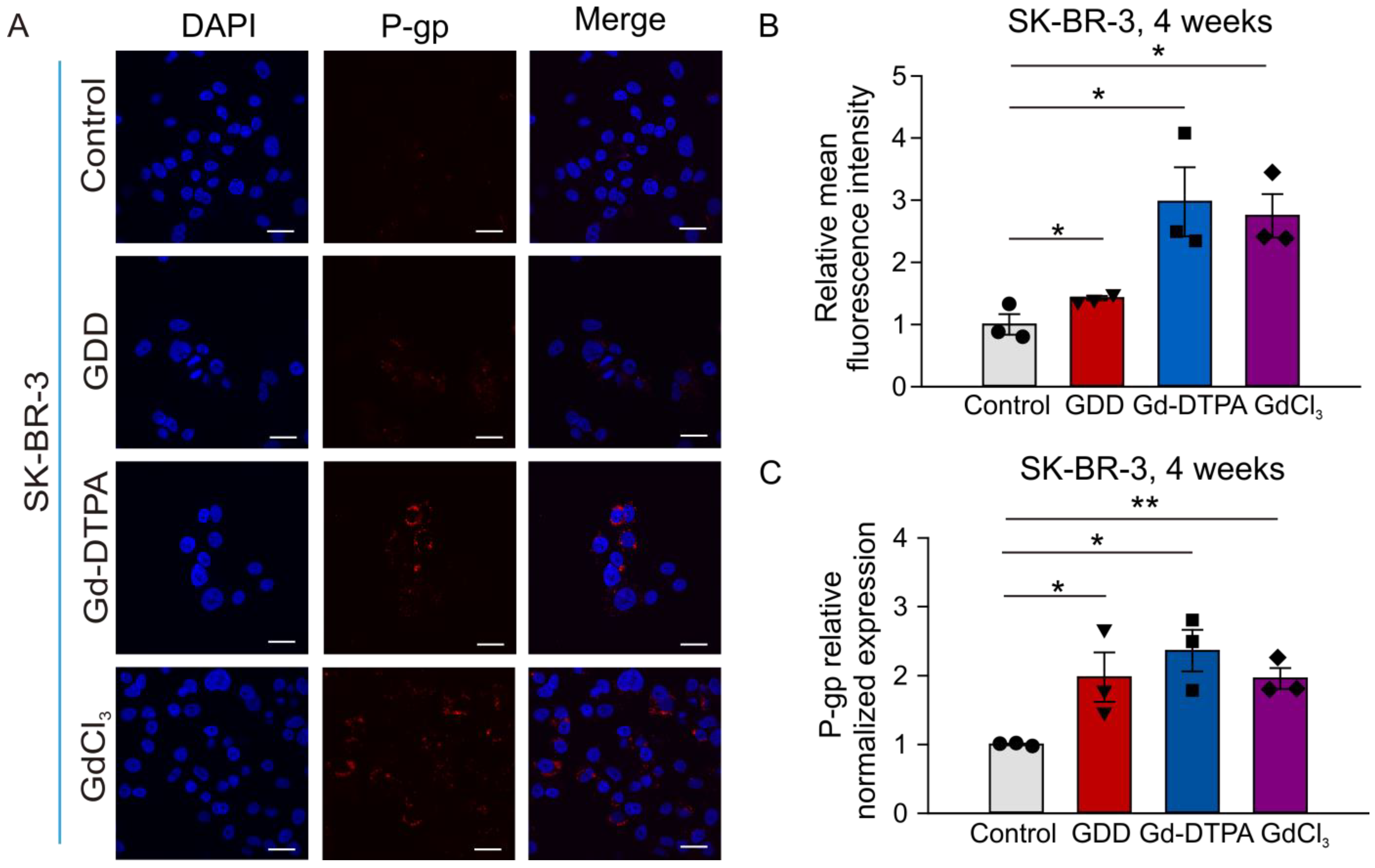
3.6. The Effects of GCBA Treatment on SK-BR-3 Cells
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Li, H.; Zeng, Y.; Zhang, H.; Gu, Z.; Gong, Q.; Luo, K. Functional gadolinium-based nanoscale systems for cancer theranostics. J. Control. Release 2021, 329, 482–512. [Google Scholar] [CrossRef] [PubMed]
- Cheong, B.Y.C.; Wilson, J.M.; Preventza, O.A.; Muthupillai, R. Gadolinium-Based Contrast Agents: Updates and Answers to Typical Questions Regarding Gadolinium Use. Tex. Heart Inst. J. 2022, 49, e217680. [Google Scholar] [CrossRef] [PubMed]
- Rah, Y.C.; Han, E.J.; Park, S.; Rhee, J.; Koun, S.; Park, H.C.; Choi, J. In vivo assay of the potential gadolinium-induced toxicity for sensory hair cells using a zebrafish animal model. J. Appl. Toxicol. JAT 2018, 38, 1398–1404. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Wang, G.; Cao, Q.; Ren, W.; Xu, L.; Bu, S. A risk prediction model for contrast-induced nephropathy associated with gadolinium-based contrast agents. Ren. Fail. 2022, 44, 741–747. [Google Scholar] [CrossRef] [PubMed]
- Turyanskaya, A.; Rauwolf, M.; Pichler, V.; Simon, R.; Burghammer, M.; Fox, O.J.L.; Sawhney, K.; Hofstaetter, J.G.; Roschger, A.; Roschger, P.; et al. Detection and imaging of gadolinium accumulation in human bone tissue by micro- and submicro-XRF. Sci. Rep. 2020, 10, 6301. [Google Scholar] [CrossRef]
- Ramalho, J.; Semelka, R.C.; Ramalho, M.; Nunes, R.H.; AlObaidy, M.; Castillo, M. Gadolinium-Based Contrast Agent Accumulation and Toxicity: An Update. AJNR. Am. J. Neuroradiol. 2016, 37, 1192–1198. [Google Scholar] [CrossRef]
- Ariyani, W.; Miyazaki, W.; Tsushima, Y.; Koibuchi, N. Gadolinium-based contrast agent accelerates the migration of astrocyte via integrin αvβ3 signaling pathway. Sci. Rep. 2022, 12, 5850. [Google Scholar] [CrossRef]
- Funke, S.K.I.; Factor, C.; Rasschaert, M.; Lezius, L.; Sperling, M.; Karst, U.; Robert, P. Long-term Gadolinium Retention in the Healthy Rat Brain: Comparison between Gadopiclenol, Gadobutrol, and Gadodiamide. Radiology 2022, 305, 179–189. [Google Scholar] [CrossRef]
- Nakamura, R.; Takanezawa, Y.; Ohshiro, Y.; Uraguchi, S.; Kiyono, M. Effects of chemical forms of gadolinium on the spleen in mice after single intravenous administration. Biochem. Biophys. Rep. 2022, 29, 101217. [Google Scholar] [CrossRef]
- Richter, H.; Bucker, P.; Martin, L.F.; Dunker, C.; Fingerhut, S.; Xia, A.; Karol, A.; Sperling, M.; Karst, U.; Radbruch, A.; et al. Gadolinium Tissue Distribution in a Large-Animal Model after a Single Dose of Gadolinium-based Contrast Agents. Radiology 2021, 301, 637–642. [Google Scholar] [CrossRef]
- Pasquini, L.; Napolitano, A.; Visconti, E.; Longo, D.; Romano, A.; Toma, P.; Espagnet, M.C.R. Gadolinium-Based Contrast Agent-Related Toxicities. CNS Drugs 2018, 32, 229–240. [Google Scholar] [CrossRef]
- Lee, E.J.; Kim, K.K.; Lee, E.K.; Lee, J.E. Characteristic MRI findings in hyperglycaemia-induced seizures: Diagnostic value of contrast-enhanced fluid-attenuated inversion recovery imaging. Clin. Radiol. 2016, 71, 1240–1247. [Google Scholar] [CrossRef]
- Muldoon, L.L.; Neuwelt, E.A. Dose-Dependent Neurotoxicity (Seizures) Due to Deposition of Gadolinium-based Contrast Agents in the Central Nervous System. Radiology 2015, 277, 925–926. [Google Scholar] [CrossRef]
- Chen, X.; Li, W.; Riley, A.M.; Soliman, M.; Chakraborty, S.; Stamatkin, C.W.; Obukhov, A.G. Molecular Determinants of the Sensitivity to Gq/11-Phospholipase C-dependent Gating, Gd3+ Potentiation, and Ca2+ Permeability in the Transient Receptor Potential Canonical Type 5 (TRPC5) Channel. J. Biol. Chem. 2017, 292, 898–911. [Google Scholar] [CrossRef]
- Semtner, M.; Schaefer, M.; Pinkenburg, O.; Plant, T.D. Potentiation of TRPC5 by protons. J. Biol. Chem. 2007, 282, 33868–33878. [Google Scholar] [CrossRef]
- Strübing, C.; Krapivinsky, G.; Krapivinsky, L.; Clapham, D.E. TRPC1 and TRPC5 form a novel cation channel in mammalian brain. Neuron 2001, 29, 645–655. [Google Scholar] [CrossRef]
- Davare, M.A.; Fortin, D.A.; Saneyoshi, T.; Nygaard, S.; Kaech, S.; Banker, G.; Soderling, T.R.; Wayman, G.A. Transient receptor potential canonical 5 channels activate Ca2+/calmodulin kinase Igamma to promote axon formation in hippocampal neurons. J. Neurosci. 2009, 29, 9794–9808. [Google Scholar] [CrossRef]
- Kumar, S.; Chakraborty, S.; Barbosa, C.; Brustovetsky, T.; Brustovetsky, N.; Obukhov, A.G. Mechanisms controlling neurite outgrowth in a pheochromocytoma cell line: The role of TRPC channels. J. Cell. Physiol. 2012, 227, 1408–1419. [Google Scholar] [CrossRef]
- Zheng, F.; Phelan, K.D. The role of canonical transient receptor potential channels in seizure and excitotoxicity. Cells 2014, 3, 288–303. [Google Scholar] [CrossRef]
- Cai, D.; Li, N.; Jin, L.; Qi, X.; Hua, D.; Wang, T. High CTC-TRPC5 Expression Significantly Associated With Poor Prognosis in Radical Resected Colorectal Cancer Patients. Front. Mol. Biosci. 2021, 8, 727864. [Google Scholar] [CrossRef]
- He, D.X.; Ma, X. Transient receptor potential channel C5 in cancer chemoresistance. Acta Pharmacol. Sin. 2016, 37, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Chen, Z.; Hua, D.; He, D.; Wang, L.; Zhang, P.; Wang, J.; Cai, Y.; Gao, C.; Zhang, X.; et al. Essential role for TrpC5-containing extracellular vesicles in breast cancer with chemotherapeutic resistance. Proc. Natl. Acad. Sci. USA 2014, 111, 6389–6394. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Chen, M.; Zhang, S.; Miao, Z.; Wang, J.; Lu, X.; Zhao, X. TRPC5-induced autophagy promotes the TMZ-resistance of glioma cells via the CAMMKβ/AMPKα/mTOR pathway. Oncol. Rep. 2019, 41, 3413–3423. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Ning, K.; Sun, X.; Zhang, C.; Jin, L.F.; Hua, D. Glycolysis is essential for chemoresistance induced by transient receptor potential channel C5 in colorectal cancer. BMC Cancer 2018, 18, 207. [Google Scholar] [CrossRef]
- Jung, S.; Mühle, A.; Schaefer, M.; Strotmann, R.; Schultz, G.; Plant, T.D. Lanthanides potentiate TRPC5 currents by an action at extracellular sites close to the pore mouth. J. Biol. Chem. 2003, 278, 3562–3571. [Google Scholar] [CrossRef]
- Ma, X.; Cai, Y.; He, D.; Zou, C.; Zhang, P.; Lo, C.Y.; Xu, Z.; Chan, F.L.; Yu, S.; Chen, Y.; et al. Transient receptor potential channel TRPC5 is essential for P-glycoprotein induction in drug-resistant cancer cells. Proc. Natl. Acad. Sci. USA 2012, 109, 16282–16287. [Google Scholar] [CrossRef]
- Thorn, C.F.; Oshiro, C.; Marsh, S.; Hernandez-Boussard, T.; McLeod, H.; Klein, T.E.; Altman, R.B. Doxorubicin pathways: Pharmacodynamics and adverse effects. Pharm. Genom. 2011, 21, 440–446. [Google Scholar] [CrossRef]
- Chen, X.; Sooch, G.; Demaree, I.S.; White, F.A.; Obukhov, A.G. Transient Receptor Potential Canonical (TRPC) Channels: Then and Now. Cells 2020, 9, 1983. [Google Scholar] [CrossRef]
- Santoni, G.; Morelli, M.B.; Marinelli, O.; Nabissi, M.; Santoni, M.; Amantini, C. Calcium Signaling and the Regulation of Chemosensitivity in Cancer Cells: Role of the Transient Receptor Potential Channels. Adv. Exp. Med. Biol. 2020, 1131, 505–517. [Google Scholar] [CrossRef]
- Singh, R.; Adhya, P.; Sharma, S.S. Redox-sensitive TRP channels: A promising pharmacological target in chemotherapy-induced peripheral neuropathy. Expert Opin. Ther. Targets 2021, 25, 529–545. [Google Scholar] [CrossRef]
- Zou, Y.; Liu, Z.; Zhou, Y.; Wang, J.; Xu, Q.; Zhao, X.; Miao, Z. TRPC5 mediates TMZ resistance in TMZ-resistant glioblastoma cells via NFATc3-P-gp pathway. Transl. Oncol. 2021, 14, 101214. [Google Scholar] [CrossRef]
- Perrotta, G.; Metens, T.; Absil, J.; Lemort, M.; Manto, M. Absence of clinical cerebellar syndrome after serial injections of more than 20 doses of gadoterate, a macrocyclic GBCA: A monocenter retrospective study. J. Neurol. 2017, 264, 2277–2283. [Google Scholar] [CrossRef]
- Hylton, N.M.; Blume, J.D.; Bernreuter, W.K.; Pisano, E.D.; Rosen, M.A.; Morris, E.A.; Weatherall, P.T.; Lehman, C.D.; Newstead, G.M.; Polin, S.; et al. Locally advanced breast cancer: MR imaging for prediction of response to neoadjuvant chemotherapy--results from ACRIN 6657/I-SPY TRIAL. Radiology 2012, 263, 663–672. [Google Scholar] [CrossRef]
- Ringler, M.D.; Rhodes, N.G.; Ayers-Ringler, J.R.; Jakaitis, D.R.; McDonald, R.J.; Kallmes, D.F.; McDonald, J.S. Gadolinium retention within multiple rat organs after intra-articular administration of gadolinium-based contrast agents. Skelet. Radiol. 2021, 50, 1419–1425. [Google Scholar] [CrossRef]
- Bezzerides, V.J.; Ramsey, I.S.; Kotecha, S.; Greka, A.; Clapham, D.E. Rapid vesicular translocation and insertion of TRP channels. Nat. Cell Biol. 2004, 6, 709–720. [Google Scholar] [CrossRef]
- Zhang, P.; Liu, X.; Li, H.; Chen, Z.; Yao, X.; Jin, J.; Ma, X. TRPC5-induced autophagy promotes drug resistance in breast carcinoma via CaMKKβ/AMPKα/mTOR pathway. Sci. Rep. 2017, 7, 3158. [Google Scholar] [CrossRef]
- Hittelman, W.N.; Rao, P.N. The nature of adriamycin-induced cytotoxicity in Chinese hamster cells as revealed by premature chromosome condensation. Cancer Res. 1975, 35, 3027–3035. [Google Scholar]
- Machaca, K. Ca(2+) signaling, genes and the cell cycle. Cell Calcium 2010, 48, 243–250. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, W.; Wang, M.; Lv, W.; White, F.A.; Chen, X.; Obukhov, A.G. Long-Term Treatment with Gadopentetic Acid or Gadodiamide Increases TRPC5 Expression and Decreases Adriamycin Nuclear Accumulation in Breast Cancer Cells. Cells 2023, 12, 1304. https://doi.org/10.3390/cells12091304
Zhang W, Wang M, Lv W, White FA, Chen X, Obukhov AG. Long-Term Treatment with Gadopentetic Acid or Gadodiamide Increases TRPC5 Expression and Decreases Adriamycin Nuclear Accumulation in Breast Cancer Cells. Cells. 2023; 12(9):1304. https://doi.org/10.3390/cells12091304
Chicago/Turabian StyleZhang, Weiheng, Mengyuan Wang, Weizhen Lv, Fletcher A. White, Xingjuan Chen, and Alexander G. Obukhov. 2023. "Long-Term Treatment with Gadopentetic Acid or Gadodiamide Increases TRPC5 Expression and Decreases Adriamycin Nuclear Accumulation in Breast Cancer Cells" Cells 12, no. 9: 1304. https://doi.org/10.3390/cells12091304
APA StyleZhang, W., Wang, M., Lv, W., White, F. A., Chen, X., & Obukhov, A. G. (2023). Long-Term Treatment with Gadopentetic Acid or Gadodiamide Increases TRPC5 Expression and Decreases Adriamycin Nuclear Accumulation in Breast Cancer Cells. Cells, 12(9), 1304. https://doi.org/10.3390/cells12091304







