Black Poplar (Populus nigra L.) Root Extracellular Trap, Structural and Molecular Remodeling in Response to Osmotic Stress
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
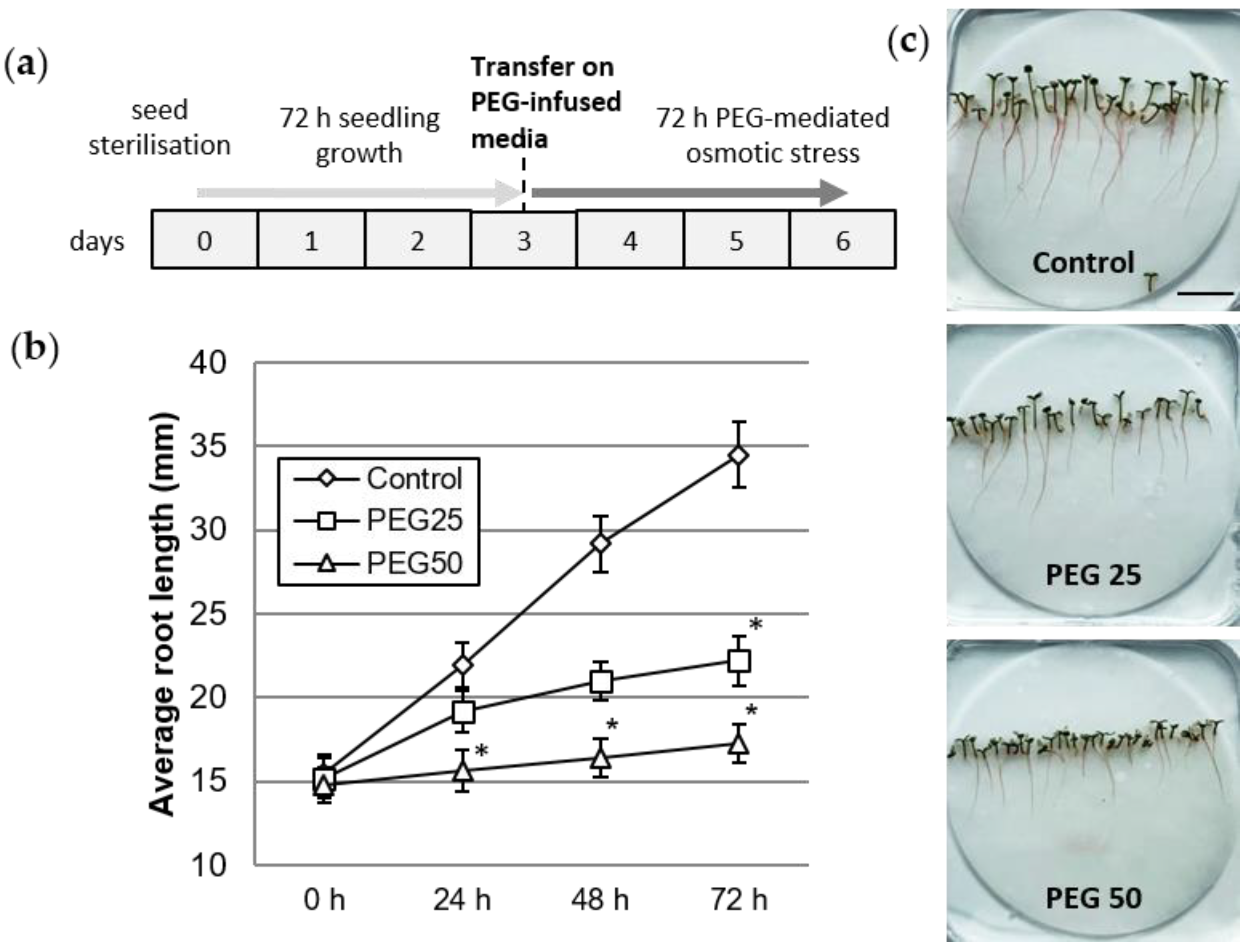
2.2. Application of an Osmotic Constraint on 3-Day-Old Seedlings
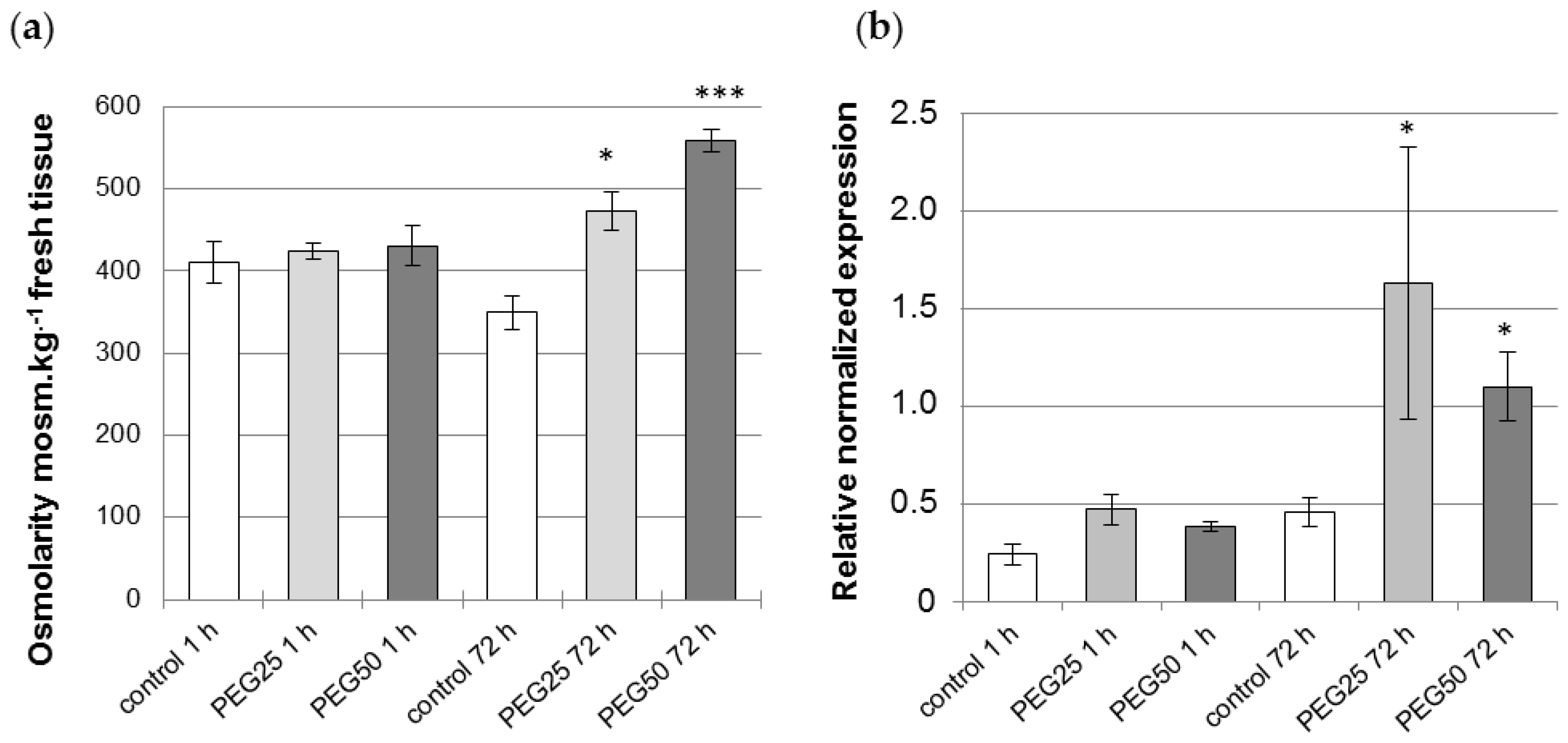
2.3. Plant Osmolarity Measurements
2.4. Delta-1-Pyrroline-5-carboxylate Synthase 2 (P5CS2) Gene Expression Analysis
2.5. Root AC-DC Microscopic Observation and Mucilage Detection Assay
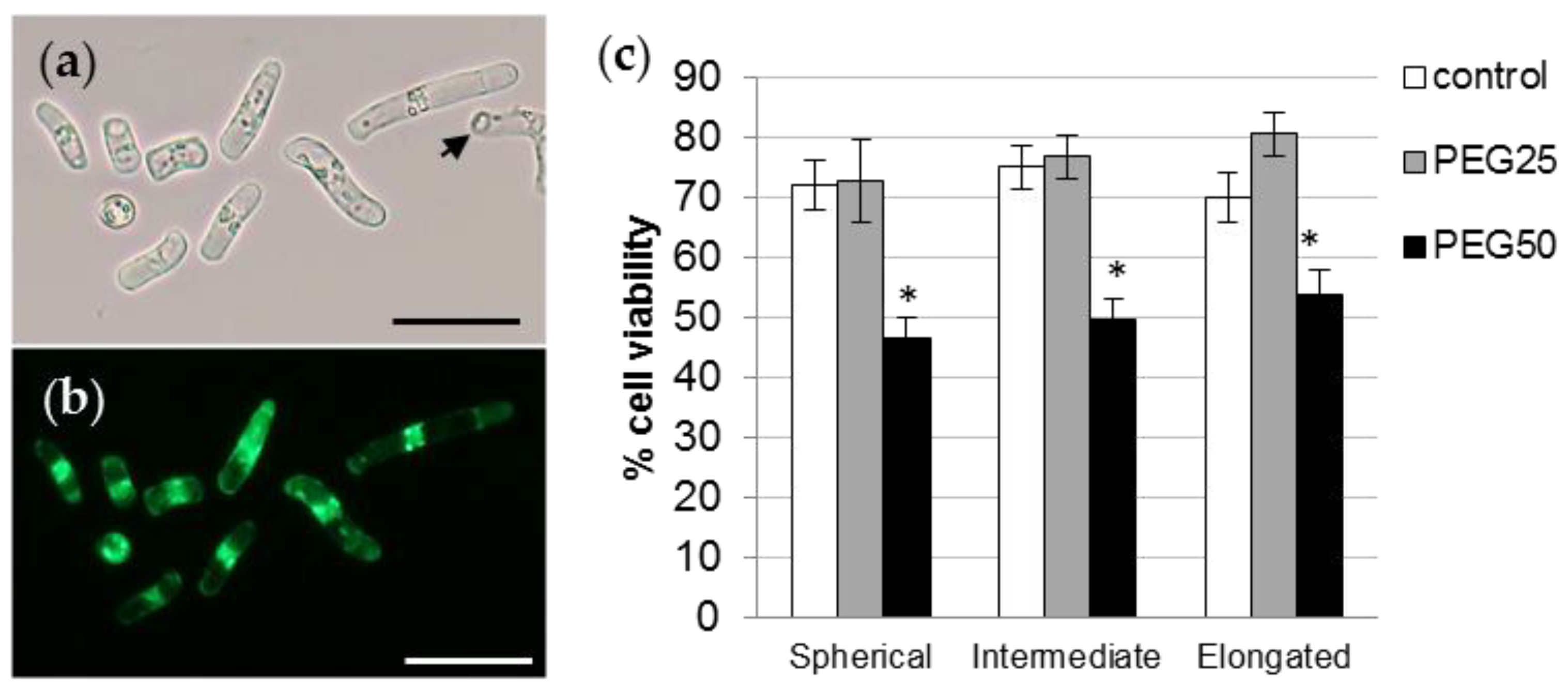
2.6. Root AC-DCs and Root Tip Viability
2.7. Image Analysis
2.8. Statistical Analysis
2.9. Resin Embedding Protocol on Root Tips with RET
2.10. Immunofluorescence Labeling on Resin Sections
3. Results
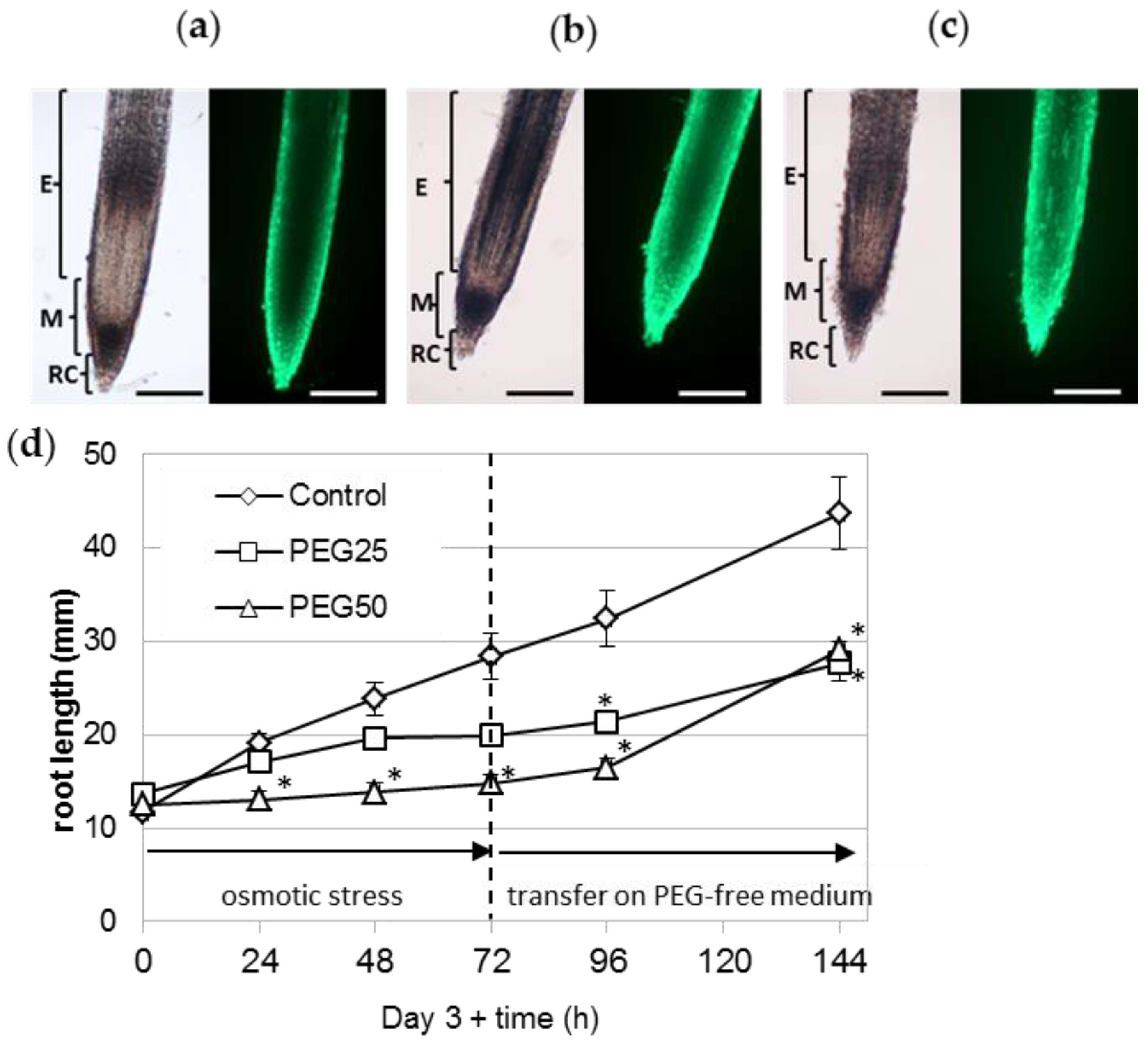
3.1. Physiological Response of P. nigra Seedlings to Osmotic Stress
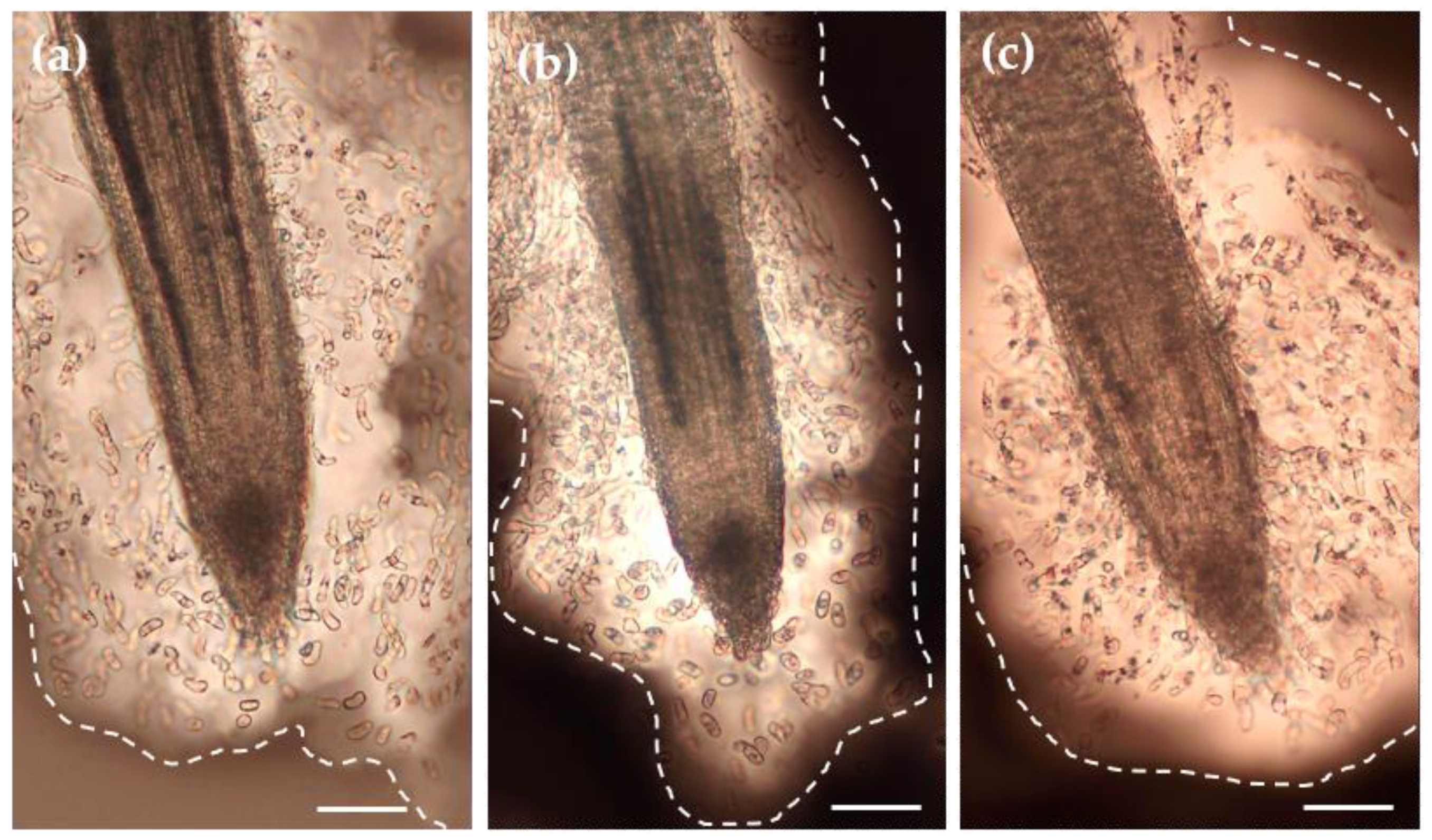
3.2. Light Microscopy Observation of P. nigra Root Extracellular Trap
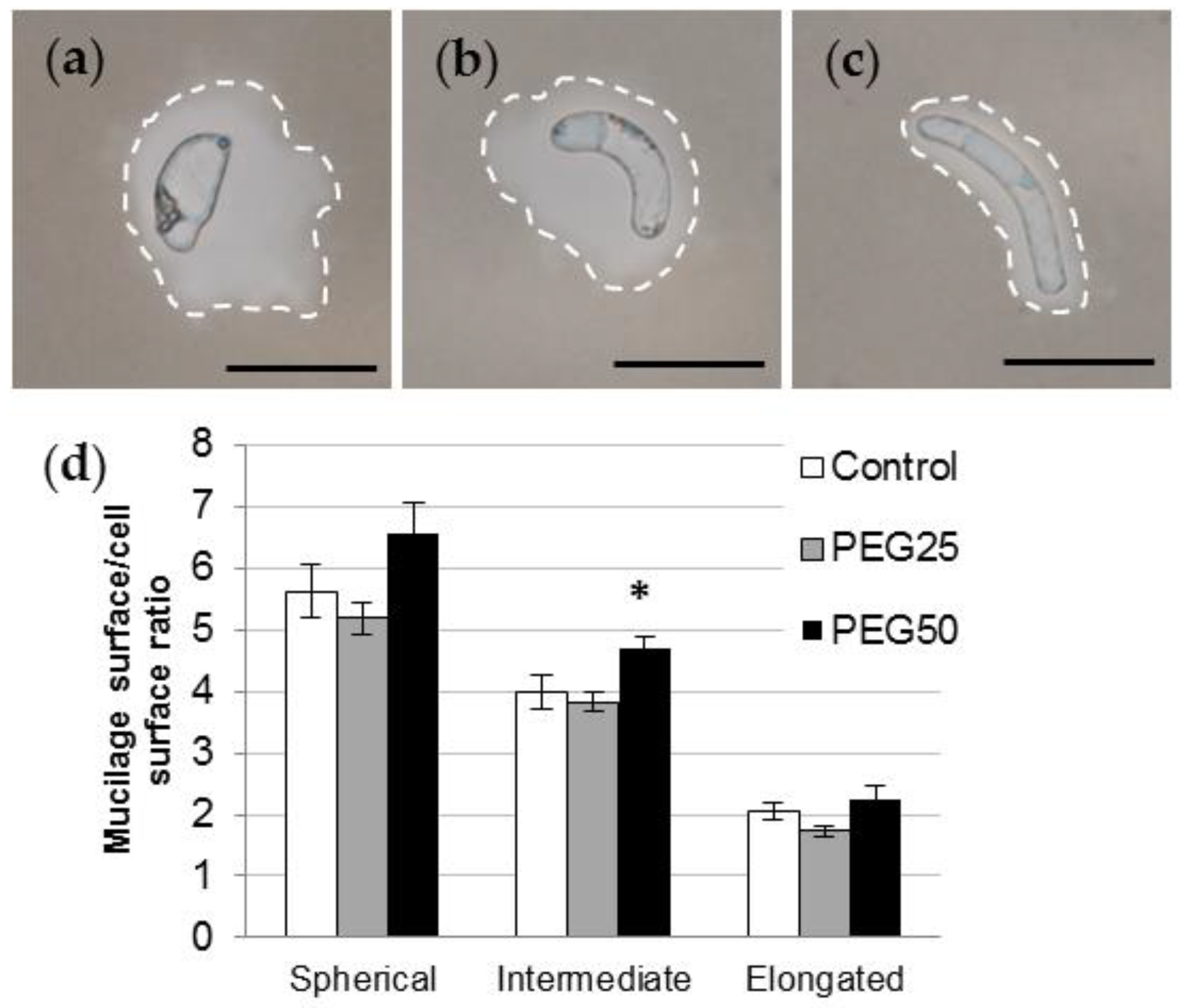
3.3. Characterization of Root AC-DC Morphotypes and Their Mucilage
3.4. Viability of Root AC-DCs and Meristematic Zone under Osmotic Stress
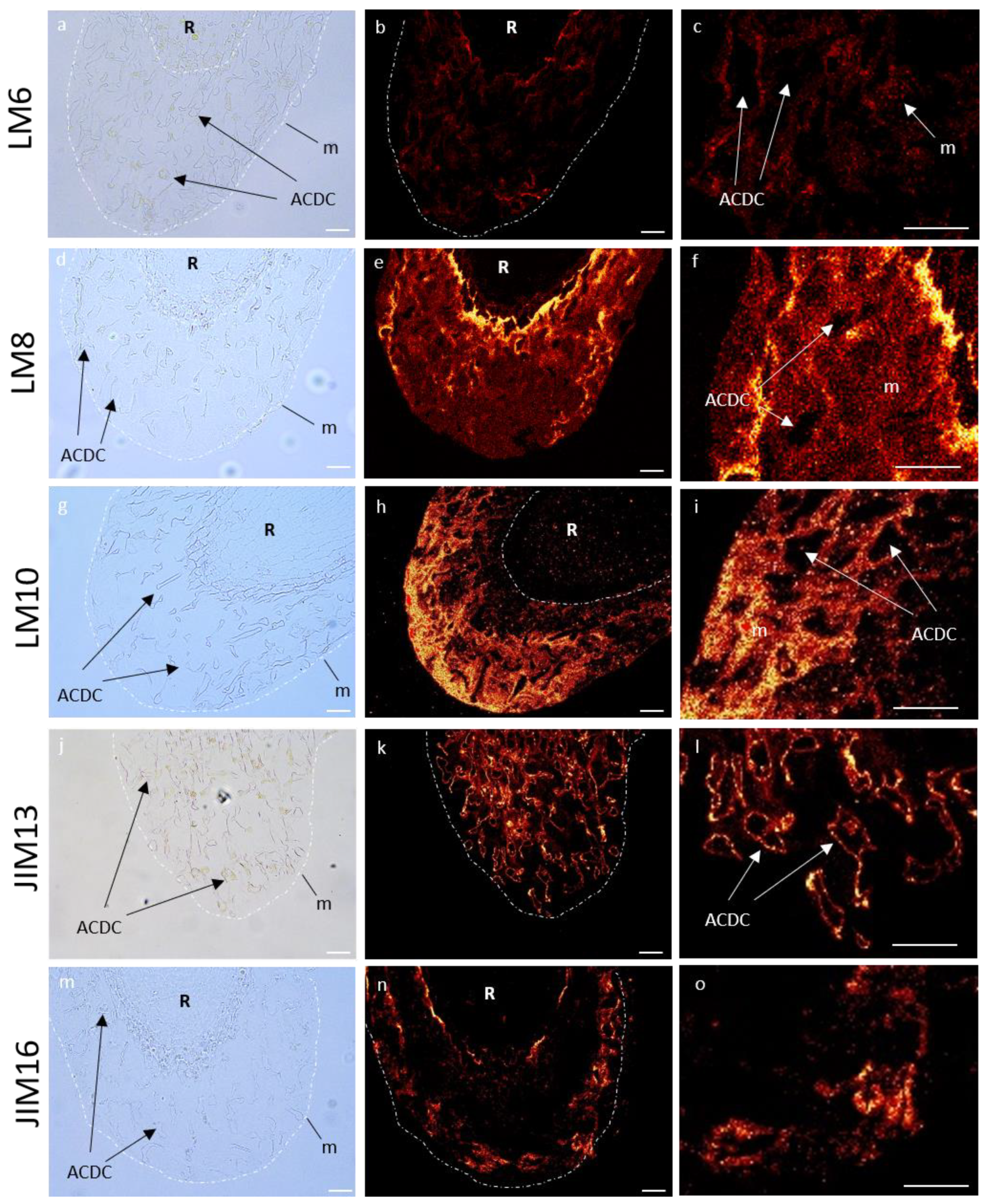
3.5. Immunocytochemical Characterization of Glycopolymers in Control RET and Root Tip
3.5.1. Immunolocalization of Pectin Epitopes
3.5.2. Immunolocalization of Hemicellulose Epitopes
3.5.3. Immunolocalization of Hydroxyprolin Rich Glycoprotein (HRGP) Epitopes
3.6. Immunocytochemical Characterization of Glycopolymers in RET and Root Tips under Osmotic Stress
4. Discussion
4.1. The First Characterization of P. nigra RET and Root Tip Glycopolymers
4.2. Physiological, Structural, and Molecular Remodeling in Response to Osmotic Stress
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Masson-Delmotte, V.; Zhai, P.; Pirani, A.; Connors, S.L.; Péan, C.; Berger, S.; Caud, N.; Chen, Y.; Goldfarb, L.; Gomis, M.I.; et al. (Eds.) IPCC, 2021: Summary for Policymakers. In Climate Change 2021: The Physical Science Basis; Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK, 2021; 2391p. [Google Scholar] [CrossRef]
- Vanden Broeck, A.; Cox, K.; Van Braeckel, A.; Neyrinck, S.; De Regge, N.; Van Looy, K. Reintroduced Native Populus nigra in Restored Floodplain Reduces Spread of Exotic Poplar Species. Front. Plant Sci. 2021, 11, 580653. [Google Scholar] [CrossRef]
- Lefebvre, M.; Villar, M.; Boizot, N.; Delile, A.; Dimouro, B.; Lomelech, A.-M.; Teyssier, C. Variability in seeds’ physicochemical characteristics, germination and seedling growth within and between two French Populus nigra populations. Peer Community J. Peer Community/Cent. Mersenne 2022, 2, e10. [Google Scholar] [CrossRef]
- Mahoney, J.M.; Rood, S.B. Streamflow requirements for cottonwood seedling recruitment: An integrative model. Wetlands 2018, 18, 634–645. [Google Scholar] [CrossRef]
- Corenblit, D.; Steiger, J.; González, E.; Gurnell, A.M.; Charrier, G.; Darrozes, J.; Dousseau, J.; Julien, F.; Lambs, L.; Larrue, S.; et al. The biogeomorphological life cycle of poplars during the fluvial biogeomorphological succession: A special focus on Populus nigra L. Earth Surf. Process. Landforms 2014, 39, 546–563. [Google Scholar] [CrossRef]
- Carminati, A.; Zarebanadkouki, M.; Kroener, E.; Ahmed, M.A.; Holz, M. Biophysical rhizosphere processes affecting root water uptake. Ann. Bot. 2016, 118, 561–571. [Google Scholar] [CrossRef] [PubMed]
- Janiak, A.; Kwaśniewski, M.; Szarejko, I. Gene expression regulation in roots under drought. J. Exp. Bot. 2016, 67, 1003–1014. [Google Scholar] [CrossRef]
- Drisch, R.C.; Stahl, Y. Function and regulation of transcription factors involved in root apical meristem and stem cell maintenance. Front. Plant Sci. 2015, 6, 505. [Google Scholar] [CrossRef]
- Hawes, M.C.; Brigham, L.A.; Wen, F.; Woo, H.H.; Zhu, Y. Function of root border cells in plant health: Pioneers in the rhizosphere. Annu. Rev. Phytopathol. 1998, 36, 311–327. [Google Scholar] [CrossRef]
- Hawes, M.C.; Gunawardena, U.; Miyasaka, S.; Zhao, X. The role of root border cells in plant defense. Trends Plant Sci. 2000, 5, 128–133. [Google Scholar] [CrossRef]
- Vicré, M.; Santaella, C.; Blanchet, S.; Gateau, A.; Driouich, A. Root border-like cells of Arabidopsis. Microscopical characterization and role in the interaction with Rhizobacteria. Plant Physiol. 2005, 138, 998–1008. [Google Scholar] [CrossRef]
- Driouich, A.; Follet-Gueye, M.L.; Vicré-Gibouin, M.; Hawes, M. Root border cells and secretions as critical elements in plant host defense. Curr. Opin. Plant Biol. 2013, 16, 489–495. [Google Scholar] [CrossRef] [PubMed]
- Driouich, A.; Smith, C.; Ropitaux, M.; Chambard, M.; Boulogne, I.; Bernard, S.; Follet-Gueye, M.L.; Vicré, M.; Moore, J.P. Root extracellular traps versus neutrophil extracellular traps in host defence, a case of functional convergence? Biol. Rev. 2019, 94, 1685–1700. [Google Scholar] [CrossRef] [PubMed]
- Nazari, M. Plant mucilage components and their functions in the rhizosphere. Rhizosphere 2021, 18, 100344. [Google Scholar] [CrossRef]
- Carreras, A.; Bernard, S.; Durambur, G.; Gügi, B.; Loutelier, C.; Pawlak, B.; Boulogne, I.; Vicré, M.; Driouich, A.; Goffner, D.; et al. In vitro characterization of root extracellular trap and exudates of three Sahelian woody plant species. Planta 2019, 251, 19. [Google Scholar] [CrossRef]
- Ropitaux, M.; Bernard, S.; Schapman, D.; Follet-Gueye, M.L.; Vicré, M.; Boulogne, I.; Driouich, A. Root border cells and mucilage secretions of soybean, Glycine max (Merr) L.: Characterization and role in interactions with the oomycete Phytophthora parasitica. Cells 2020, 9, 2215. [Google Scholar] [CrossRef]
- Cannesan, M.A.; Durand, C.; Burel, C.; Gangneux, C.; Lerouge, P.; Ishii, T.; Laval, K.; Follet-Gueye, M.L.; Driouich, A.; Vicré-Gibouin, M. Effect of arabinogalactan proteins from the root caps of pea and Brassica napus on Aphanomyces euteiches zoospore chemotaxis and germination. Plant Physiol. 2012, 159, 1658–1670. [Google Scholar] [CrossRef]
- Chambard, M.; Ben Mlouka, M.A.; Jing, L.; Plasson, C.; Cosette, P.; Leprince, J.; Follet-Gueye, M.L.; Driouich, A.; Nguema-Ona, E.; Boulogne, I. Elicitation of Roots and AC-DC with PEP-13 Peptide Shows Differential Defense Responses in Multi-Omics. Cells 2022, 11, 2605. [Google Scholar] [CrossRef]
- Yan, Z.; Bo, C.; Shibin, G.; Tingzhao, R. Biological Characters of Root Border Cells Development in Maize (Zea mays). Biotechnology 2014, 13, 89–98. [Google Scholar] [CrossRef]
- Endo, I.; Tange, T.; Osawa, H. A cell-type-specific defect in border cell formation in the Acacia mangium root cap developing an extraordinary sheath of sloughed-off cells. Ann. Bot. 2011, 108, 279–290. [Google Scholar] [CrossRef]
- Badri, D.V.; Vivanco, J.M. Regulation and function of root exudates. Plant Cell Environ. 2009, 32, 666–681. [Google Scholar] [CrossRef]
- Ma, W.; Muthreich, N.; Liao, C.; Franz-Wachtel, M.; Schütz, W.; Zhang, F.; Hochholdinger, F.; Li, C. The mucilage proteome of maize (Zea mays L.) primary roots. J. Proteome Res. 2010, 9, 2968–2976. [Google Scholar] [CrossRef]
- Weiller, F.; Moore, J.P.; Yopung, P.; Driouich, A.; Vivier, M.A. The Brassicaceae species Heliophila coronopifolia produces root border-like cells that protect the root tip and secrete defensin peptides. Ann. Bot. 2017, 119, 803–813. [Google Scholar] [CrossRef] [PubMed]
- Plancot, B.; Santaella, C.; Jaber, R.; Kiefer-Meyer, M.C.; Follet-Gueye, M.L.; Leprince, J.; Gattin, I.; Souc, C.; Driouich, A.; Vicré-Gibouin, M. Deciphering the responses of root border-like cells of Arabidopsis and flax to pathogen-derived elicitors. Plant Physiol. 2013, 163, 1584–1597. [Google Scholar] [CrossRef]
- Castilleux, R.; Plancot, B.; Ropitaux, M.; Carreras, A.; Leprince, J.; Boulogne, I.; Follet-Gueye, M.L.; Popper, Z.A.; Driouich, A.; Vicré, M. Cell wall extensins in root-microbe interactions and root secretions. J. Exp. Bot. 2018, 69, 4235–4247. [Google Scholar] [CrossRef]
- Wen, F.; White, G.J.; VanEtten, H.D.; Xiong, Z.; Hawes, M.C. Extracellular DNA is required for root tip resistance to fungal infection. Plant Physiol. 2009, 151, 820–829. [Google Scholar] [CrossRef] [PubMed]
- Hawes, M.C.; Curlango-Rivera, G.; Wen, F.; White, G.J.; VanEtten, H.D.; Xiong, Z. Extracellular DNA: The tip of root defenses? Plant Sci. 2011, 180, 741–745. [Google Scholar] [CrossRef]
- Chambard, M.; Plasson, C.; Derambure, C.; Coutant, S.; Tournier, I.; Lefranc, B.; Leprince, J.; Kiefer-Meyer, M.C.; Driouich, A.; Follet-Gueye, M.L.; et al. New Insights into Plant Extracellular DNA. A Study in Soybean Root Extracellular Trap. Cells 2021, 10, 69. [Google Scholar] [CrossRef] [PubMed]
- Curlango-Rivera, G.; Xiong, Z.; Kessler, J.O.; Hawes, M.C. Extracellular trapping of bacteria in plant defense responses: Dynamics and specificity. Phytopathology 2011, 101, S40. [Google Scholar]
- Koroney, A.S.; Plasson, C.; Pawlak, B.; Sidikou, R.; Driouich, A.; Menu-Bouaouiche, L.; Vicré-Gibouin, M. Root exudate of Solanum tuberosum is enriched in galactose-containing molecules and impacts the growth of Pectobacterium atrosepticum. Ann. Bot. 2016, 118, 797–808. [Google Scholar] [CrossRef]
- Goldberg, N.P.; Hawes, M.C.; Stanghellini, M.E. Specific attraction to and infection of cotton root cap cells by zoospores of Pythium dissotocum. Can. J. Bot. 1989, 67, 1760–1767. [Google Scholar] [CrossRef]
- Gunawardena, U.; Rodriguez, M.; Straney, D.; Romeo, J.T.; VanEtten, H.D.; Hawes, M.C. Tissue-specific localization of pea root infection by Nectria haematococca. Mechanisms and consequences. Plant Physiol. 2005, 137, 1363–1674. [Google Scholar] [CrossRef] [PubMed]
- Cannesan, M.A.; Gangneux, C.; Lanoue, A.; Giron, D.; Laval, K.; Hawes, M.; Driouich, A.; Vicré-Gibouin, M. Association between border cell responses and localized root infection by pathogenic Aphanomyces euteiches. Ann. Bot. 2011, 108, 459–469. [Google Scholar] [CrossRef]
- Hawes, M.C.; McLain, J.; Ramirez-Andreotta, M.; Curlango-Rivera, G.; Flores-Lara, Y.; Brigham, L. Extracellular trapping of soil contaminants by root border cells: New insights into plant defense. Agronomy 2016, 6, 5. [Google Scholar] [CrossRef]
- Huskey, D.A.; Curlango-Rivera, G.; Root, R.A.; Wen, F.; Amistadi, M.K.; Chorover, J.; Hawes, M.C. Trapping of lead (Pb) by corn and pea root border cells. Plant Soil 2018, 430, 205–217. [Google Scholar] [CrossRef]
- Feng, Y.; Li, H.; Zhang, X.; Li, X.; Zhang, J.; Shi, L.; Chen, X.; Nong, W.; Wang, C.; Shabala, S.; et al. Effects of Cadmium Stress on Root and Root Border Cells of Some Vegetable Species with Different Types of Root Meristem. Life 2022, 12, 1401. [Google Scholar] [CrossRef]
- De Vries, F.T.; Brown, C.; Stevens, C.J. Grassland species root response to drought: Consequences for soil carbon and nitrogen availability. Plant Soil 2016, 409, 297–312. [Google Scholar] [CrossRef]
- Rabbi, S.M.F.; Tighe, M.K.; Flavel, R.J.; Kaiser, B.N.; Guppy, C.N.; Zhang, X.; Young, I.M. Plant roots redesign the rhizosphere to alter the three-dimensional physical architecture and water dynamics. New Phytol. 2018, 219, 542–550. [Google Scholar] [CrossRef]
- Carter, A.Y.; Ottman, M.J.; Curlango-Rivera, G.; Huskey, D.A.; D’Agostini, B.A.; Hawes, M.C. Drought-Tolerant Barley: II. Root Tip characteristics in Emerging Roots. Agronomy 2019, 9, 220. [Google Scholar] [CrossRef]
- Nazari, M.; Riebeling, S.; Banfield, C.C.; Akale, A.; Crosta, M.; Mason-Jones, K.; Dippold, M.A.; Ahmed, M.A. Mucilage Polysaccharide Composition and Exudation in Maize From Contrasting Climatic Regions. Front. Plant Sci. 2020, 11, 587610. [Google Scholar] [CrossRef]
- Ouedraogo, D.Y.; Mortier, F.; Gourlet-Fleury, S.; Freycon, V.; Picard, N. Slow-growing species cope best with drought: Evidence from long-term measurements in a tropical semi-deciduous moist forest of Central Africa. J. Ecol. 2013, 101, 1459–1470. [Google Scholar] [CrossRef]
- Suszka, J.; Plitta, B.P.; Michalak, M.; Bujarska-Borkowska, B.; Tylkowski, T.; Chmielarz, P. Optimal seed water content and storage temperature for preservation of Populus nigra L. germplasm. Ann. For. Sci. 2014, 71, 543–549. [Google Scholar] [CrossRef]
- Osmolovskaya, N.; Shumilina, J.; Kim, A.; Didio, A.; Grishina, T.; Bilova, T.; Keltsieva, O.A.; Zhukov, V.; Tikhonovich, I.; Tarakhovskaya, E.; et al. Methodology of Drought Stress Research: Experimental Setup and Physiological Characterization. Int. J. Mol. Sci. 2018, 19, 4089. [Google Scholar] [CrossRef] [PubMed]
- Verslues, P.E. Quantification of water stress-induced osmotic adjustment and proline accumulation for Arabidopsis thaliana molecular genetic studies. In Plant Stress Tolerance. Methods in Molecular Biology (Methods and Protocols); Sunkar, R., Ed.; Humana Press: Totowa, NJ, USA, 2010; Volume 639, pp. 301–315. [Google Scholar] [CrossRef]
- Hong, Z.; Lakkineni, K.; Zhang, Z.; Verma, D.P. Removal of feedback inhibition of delta(1)-pyrroline-5-carboxylate synthetase results in increased proline accumulation and protection of plants from osmotic stress. Plant Physiol. 2000, 122, 1129–1136. [Google Scholar] [CrossRef] [PubMed]
- Verhertbruggen, Y.; Marcus, S.E.; Haeger, A.; Ordaz-Ortiz, J.J.; Knox, J.P. An extended set of monoclonal antibodies to pectic homogalacturonan. Carbohydr. Res. 2009, 344, 1858–1862. [Google Scholar] [CrossRef]
- Jones, L.; Seymour, G.B.; Knox, J.P. Localization of pectic galactan in tomato cell walls using a monoclonal antibody specific to (1 [->] 4)-β-D-Galactan. Plant Physiol. 1997, 113, 1405–1412. [Google Scholar] [CrossRef]
- Willats, W.G.; Marcus, S.E.; Knox, J.P. Generation of a monoclonal antibody specific to (1→ 5)-α-l-arabinan. Carbohydr. Res. 1998, 308, 149–152. [Google Scholar] [CrossRef]
- Willats, W.G.; McCartney, L.; Steele-King, C.G.; Marcus, S.E.; Mort, A.; Huisman, M.; van Alebeek, G.J.; Schols, H.A.; Voragen, A.G.; Le Goff, A.; et al. A xylogalacturonan epitope is specifically associated with plant cell detachment. Planta 2004, 218, 673–681. [Google Scholar] [CrossRef]
- Pedersen, H.L.; Fangel, J.U.; McCleary, B.; Ruzanski, C.; Rydahl, M.G.; Ralet, M.C.; Farkas, V.; von Schantz, L.; Marcus, S.E.; Andersen, M.C.; et al. Versatile high resolution oligosaccharide microarrays for plant glycobiology and cell wall research. J. Biol. Chem. 2012, 287, 39429–39438. [Google Scholar] [CrossRef]
- Ruprecht, C.; Bartetzko, M.P.; Senf, D.; Dallabernadina, P.; Boos, I.; Andersen, M.C.; Kotake, T.; Knox, J.P.; Hahn, M.G.; Clausen, M.H.; et al. A synthetic glycan microarray enables epitope mapping of plant cell wall glycan-directed antibodies. Plant Physiol. 2017, 175, 1094–1104. [Google Scholar] [CrossRef]
- Marcus, S.E.; Blake, A.W.; Benians, T.A.; Lee, K.J.; Poyser, C.; Donaldson, L.; Leroux, O.; Rogowski, A.; Petersen, H.L.; Boraston, A.; et al. Restricted access of proteins to mannan polysaccharides in intact plant cell walls. Plant J. 2010, 64, 191–203. [Google Scholar] [CrossRef]
- Pennell, R.I.; Janniche, L.; Kjellbom, P.; Scofield, G.N.; Peart, J.M.; Roberts, K. Developmental regulation of a plasma membrane arabinogalactan protein epitope in oilseed rape flowers. Plant Cell 1991, 3, 1317–1326. [Google Scholar] [CrossRef] [PubMed]
- Knox, J.P.; Linstead, P.J.; Cooper, J.P.C.; Roberts, K. Developmentally regulated epitopes of cell surface arabinogalactan proteins and their relation to root tissue pattern formation. Plant J. 1991, 1, 317–326. [Google Scholar] [CrossRef] [PubMed]
- Yates, E.A.; Valdor, J.F.; Haslam, S.M.; Morris, H.R.; Dell, A.; Mackie, W.; Knox, J.P. Characterization of carbohydrate structural features recognized by anti-arabinogalactan-protein monoclonal antibodies. Glycobiology 1996, 6, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Smallwood, M.; Martin, H.; Knox, J.P. An epitope of rice threonine-and hydroxyproline-rich glycoprotein is common to cell wall and hydrophobic plasma-membrane glycoproteins. Planta 1995, 196, 510–522. [Google Scholar] [CrossRef] [PubMed]
- Smallwood, M.; Beven, A.; Donovan, N.; Neill, S.J.; Peart, J.; Roberts, K.; Knox, J.P. Localization of cell wall proteins in relation to the developmental anatomy of the carrot root apex. Plant J. 1994, 5, 237–246. [Google Scholar] [CrossRef]
- Heimsch, C.; Seago, J.L., Jr. Organization of the root apical meristem in angiosperms. Am. J. Bot. 2008, 95, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Hawes, M.C.; Bengough, G.; Cassab, G.; Ponce, G. Root caps and rhizosphere. J. Plant Growth Regul. 2003, 21, 352–367. [Google Scholar] [CrossRef]
- Marzec-Schmidt, K.; Ludwików, A.; Wojciechowska, N.; Kasprowicz-Maluśki, A.; Mucha, J.; Bagniewska-Zadworna, A. Xylem cell wall formation in pioneer roots and stems of Populus trichocarpa (Torr. & Gray). Front. Plant Sci. 2019, 10, 1419. [Google Scholar] [CrossRef]
- Durand, C.; Vicré-Gibouin, M.; Follet-Gueye, M.L.; Duponchel, L.; Moreau, M.; Lerouge, P.; Driouich, A. The Organization Pattern of Root Border-Like Cells of Arabidopsis Is Dependent on Cell Wall Homogalacturonan. Plant Physiol. 2009, 150, 1411–1421. [Google Scholar] [CrossRef]
- Mravec, J.; Guo, X.; Hansen, A.R.; Schückel, J.; Kračun, S.K.I.; Mikkelsen, M.D.; Mouille, G.; Johansen, I.E.; Ulvskov, P.; Domozych, D.S.; et al. Pea border cell maturation and release involve complex cell wall structural dynamics. Plant Physiol. 2017, 174, 1051–1066. [Google Scholar] [CrossRef]
- Ropitaux, M.; Bernard, S.; Follet-Gueye, M.L.; Vicré, M.; Boulogne, I.; Driouich, A. Xyloglucan and cellulose form molecular cross-bridges connecting root border cells in pea (Pisum sativum). Plant Physiol. Biochem. 2019, 139, 191–196. [Google Scholar] [CrossRef] [PubMed]
- Edashige, Y.; Ishi, T. Extracellular Polysaccharides from the Suspension-Culture of Populus Alba L. Holzforschung 1999, 53, 137–141. [Google Scholar] [CrossRef]
- Macquet, A.; Ralet, M.C.; Kronenberger, J.; Marion-Poll, A.; North, H.M. In situ, chemical and macromolecular study of the composition of Arabidopsis thaliana seed coat mucilage. Plant Cell Physiol. 2007, 48, 984–999. [Google Scholar] [CrossRef] [PubMed]
- Ralet, M.C.; Crépeau, M.J.; Vigouroux, J.; Tran, J.; Berger, A.; Sallé, C.; Granier, F.; Botran, L.; North, H.M. Xylans Provide the Structural Driving Force for Mucilage Adhesion to the Arabidopsis Seed Coat. Plant Physiol. 2016, 171, 165–178. [Google Scholar] [CrossRef] [PubMed]
- Naran, R.; Chen, G.; Carpita, N.C. Novel rhamnogalacturonan I and arabinoxylan polysaccharides of flax seed mucilage. Plant Physiol. 2008, 148, 132–141. [Google Scholar] [CrossRef]
- Yu, L.; Yakubov, G.E.; Zeng, W.; Xing, X.; Stenson, J.; Bulone, V.; Stokes, J.R. Multi-layer mucilage of Plantago ovata seeds: Rheological differences arise from variations in arabinoxylan side chains. Carbohydr. Polym. 2017, 165, 132–141. [Google Scholar] [CrossRef]
- Tan, L.; Eberhard, S.; Pattathil, S.; Warder, C.; Glushka, J.; Yuan, C.; Hao, Z.; Zhu, X.; Avci, U.; Miller, J.S.; et al. An Arabidopsis cell wall proteoglycan consists of pectin and arabinoxylan covalently linked to an arabinogalactan protein. Plant Cell 2013, 25, 270–287. [Google Scholar] [CrossRef]
- Broxterman, S.E.; Schols, H.A. Interactions between pectin and cellulose in primary plant cell walls. Carbohydr. Polym. 2018, 192, 263–272. [Google Scholar] [CrossRef]
- Coenen, G.J.; Bakx, E.J.; Verhoef, R.P.; Schols, H.A.; Voragen, A.G.J. Identification of the connecting linkage between homo- or xylogalacturonan and rhamnogalacturonan type I. Carbohydr. Polym. 2007, 70, 224–235. [Google Scholar] [CrossRef]
- Rowe, J.H.; Topping, J.F.; Liu, J.; Lindsey, K. Abscisic acid regulates root growth under osmotic stress conditions via an interacting hormonal network with cytokinin, ethylene and auxin. New Phytol. 2016, 211, 225–239. [Google Scholar] [CrossRef]
- Verslues, P.E.; Sharp, R.E. Proline accumulation in maize (Zea mays L.) primary roots at low water potentials. II. Metabolic source of increased proline deposition in the elongation zone. Plant Physiol. 1999, 119, 1349–1360. [Google Scholar] [CrossRef] [PubMed]
- Bizet, F.; Hummel, I.; Bogeat-Triboulot, M.B. Length and activity of the root apical meristem revealed in vivo by infrared imaging. J. Exp. Bot. 2015, 66, 1387–1395. [Google Scholar] [CrossRef] [PubMed]
- Le Gall, H.; Philippe, F.; Domon, J.M.; Gillet, F.; Pelloux, J.; Rayon, C. Cell Wall Metabolism in Response to Abiotic Stress. Plants 2015, 4, 112–166. [Google Scholar] [CrossRef] [PubMed]
- Moore, J.P.; Vicré-Gibouin, M.; Farrant, J.M.; Driouich, A. Adaptations of higher plant cell walls to water loss: Drought vs. desiccation. Physiol. Plant. 2008, 134, 237–245. [Google Scholar] [CrossRef]
- Wormit, A.; Usadel, B. The Multifaceted Role of Pectin Methylesterase Inhibitors (PMEIs). Int. J. Mol. Sci. 2018, 19, 2878. [Google Scholar] [CrossRef]
- Guillemin, F.; Guillon, F.; Bonnin, E.; Devaux, M.F.; Chevalier, T.; Knox, J.P.; Liners, F.; Thibault, J.F. Distribution of pectic epitopes in cell walls of the sugar beet root. Planta 2005, 222, 355–371. [Google Scholar] [CrossRef]
- Leucci, M.R.; Lenucci, M.S.; Piro, G.; Dalessandro, G. Water stress and cell wall polysaccharides in the apical root zone of wheat cultivars varying in drought tolerance. J. Plant Physiol. 2008, 165, 1168–1180. [Google Scholar] [CrossRef]
- Klaassen, M.T.; Trindade, L.M. RG-I galactan side-chains are involved in the regulation of the water-binding capacity of potato cell walls. Carbohydr. Polym. 2020, 227, 115353. [Google Scholar] [CrossRef]
- Popper, Z.A.; Fry, S.C. Widespread occurrence of a covalent linkage between xyloglucan and acidic polysaccharides in suspension-cultured angiosperm cells. Ann. Bot. 2005, 96, 91–99. [Google Scholar] [CrossRef]
- Tiika, R.J.; Wei, J.; Cui, G.; Ma, Y.; Yang, H.; Duan, H. Transcriptome-wide characterization and functional analysis of Xyloglucan endo-transglycosylase/hydrolase (XTH) gene family of Salicornia europaea L. under salinity and drought stress. BMC Plant Biol. 2021, 21, 491. [Google Scholar] [CrossRef]
- Choi, J.Y.; Seo, Y.S.; Kim, S.J.; Kim, W.T.; Shin, J.S. Constitutive expression of CaXTH3, a hot pepper xyloglucan endotransglucosylase/hydrolase, enhanced tolerance to salt and drought stresses without phenotypic defects in tomato plants (Solanum lycopersicum cv. Dotaerang). Plant Cell Rep. 2011, 30, 867–877. [Google Scholar] [CrossRef] [PubMed]
- Mareri, L.; Romi, M.; Cai, G. Arabinogalactan proteins: Actors or spectators during abiotic and biotic stress in plants? Plant Biosyst. 2018, 153, 173–185. [Google Scholar] [CrossRef]
- Ninmanont, P.; Wongchai, C.; Pfeiffer, W.; Chaidee, A. Salt stress of two rice varieties: Root border cell response and multi-logistic quantification. Protoplasma 2021, 258, 1119–1131. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.A.; Kroener, E.; Holz, M.; Zarebanadkouki, M.; Carminati, A. Mucilage exudation facilitates root water uptake in dry soils. Funct. Plant Biol. 2014, 41, 1129–1137. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.A.; Sanaullah, M.; Blagodatskaya, E.; Mason-Jones, K.; Jawad, H.; Kuzyakov, Y.; Dippold, M.A. Soil microorganisms exhibit enzymatic and priming response to root mucilage under drought. Soil Biol. Biochem. 2018, 116, 410–418. [Google Scholar] [CrossRef]
- Williams, A.; de Vries, F.T. Plant root exudation under drought: Implications for ecosystem functioning. New Phytol. 2020, 225, 1899–1905. [Google Scholar] [CrossRef]
- Shaw, R.; Tian, X.; Xu, J. Single-Cell Transcriptome Analysis in Plants: Advances and Challenges. Mol. Plant 2021, 14, 115–126. [Google Scholar] [CrossRef]


| RET | Root | |||||
|---|---|---|---|---|---|---|
| Mucilage | AC-DC | Meristematic Zone | Elongation Zone | |||
| Pectins | HG | LM19 | − | − | ++ | ++ |
| LM20 | − | + | ± | ± | ||
| RG-I | LM6 | + | − | ± | ± | |
| LM5 | − | + | − | ++ | ||
| XGA | LM8 | ++ | − | − | − | |
| Hemicelluloses | XyG | LM25 | − | − | + | + |
| Het | LM21 | − | − | − | − | |
| Xylan | LM10 | ++ | − | − | − | |
| Hydroxyprolin Rich Glycoproteins | Arabino- galactan Protein | JIM8 | − | − | + | + |
| JIM13 | − | + | + | + | ||
| JIM14 | − | ± | − | − | ||
| JIM16 | ++ | − | − | − | ||
| Extensin | JIM12 | − | − | − | − | |
| LM1 | − | − | − | − | ||
| RET | Root | |||||
|---|---|---|---|---|---|---|
| Mucilage | AC-DC | Meristematic Zone | Elongation Zone | |||
| Pectins | HG | LM19 | = | = | ||
| LM20 | ↘ | ↗ | ↗ | |||
| RG-I | LM6 | ↘↘ | ↗ | ↗ | ||
| LM5 | ↘ | ↘ | ||||
| XGA | LM8 | ↘↘ | ||||
| Hemicelluloses | XyG | LM25 | ↗ | ↘ | ↘ | |
| Het | LM21 | |||||
| Xylan | LM10 | ↘↘ | ||||
| Hydroxyprolin Rich Glycoproteins | Arabino- galactan Protein | JIM8 | ↗ | ↗ | ||
| JIM13 | ↗ | ↗ | ↗ | |||
| JIM14 | ↘ | |||||
| JIM16 | ↘↘ | |||||
| Extensin | JIM12 | |||||
| LM1 | ||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Busont, O.; Durambur, G.; Bernard, S.; Plasson, C.; Joudiou, C.; Baude, L.; Chefdor, F.; Depierreux, C.; Héricourt, F.; Larcher, M.; et al. Black Poplar (Populus nigra L.) Root Extracellular Trap, Structural and Molecular Remodeling in Response to Osmotic Stress. Cells 2023, 12, 858. https://doi.org/10.3390/cells12060858
Busont O, Durambur G, Bernard S, Plasson C, Joudiou C, Baude L, Chefdor F, Depierreux C, Héricourt F, Larcher M, et al. Black Poplar (Populus nigra L.) Root Extracellular Trap, Structural and Molecular Remodeling in Response to Osmotic Stress. Cells. 2023; 12(6):858. https://doi.org/10.3390/cells12060858
Chicago/Turabian StyleBusont, Océane, Gaëlle Durambur, Sophie Bernard, Carole Plasson, Camille Joudiou, Laura Baude, Françoise Chefdor, Christiane Depierreux, François Héricourt, Mélanie Larcher, and et al. 2023. "Black Poplar (Populus nigra L.) Root Extracellular Trap, Structural and Molecular Remodeling in Response to Osmotic Stress" Cells 12, no. 6: 858. https://doi.org/10.3390/cells12060858
APA StyleBusont, O., Durambur, G., Bernard, S., Plasson, C., Joudiou, C., Baude, L., Chefdor, F., Depierreux, C., Héricourt, F., Larcher, M., Malik, S., Boulogne, I., Driouich, A., Carpin, S., & Lamblin, F. (2023). Black Poplar (Populus nigra L.) Root Extracellular Trap, Structural and Molecular Remodeling in Response to Osmotic Stress. Cells, 12(6), 858. https://doi.org/10.3390/cells12060858








