Expression of Checkpoint Molecules in the Tumor Microenvironment of Intrahepatic Cholangiocarcinoma: Implications for Immune Checkpoint Blockade Therapy
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Recruitment
2.2. Sample Collection
2.3. Whole Slide Multiplexed Immunofluorescence (mIF)
2.4. Statistical Analysis
3. Results
3.1. Patient Characteristics and Clinical Data
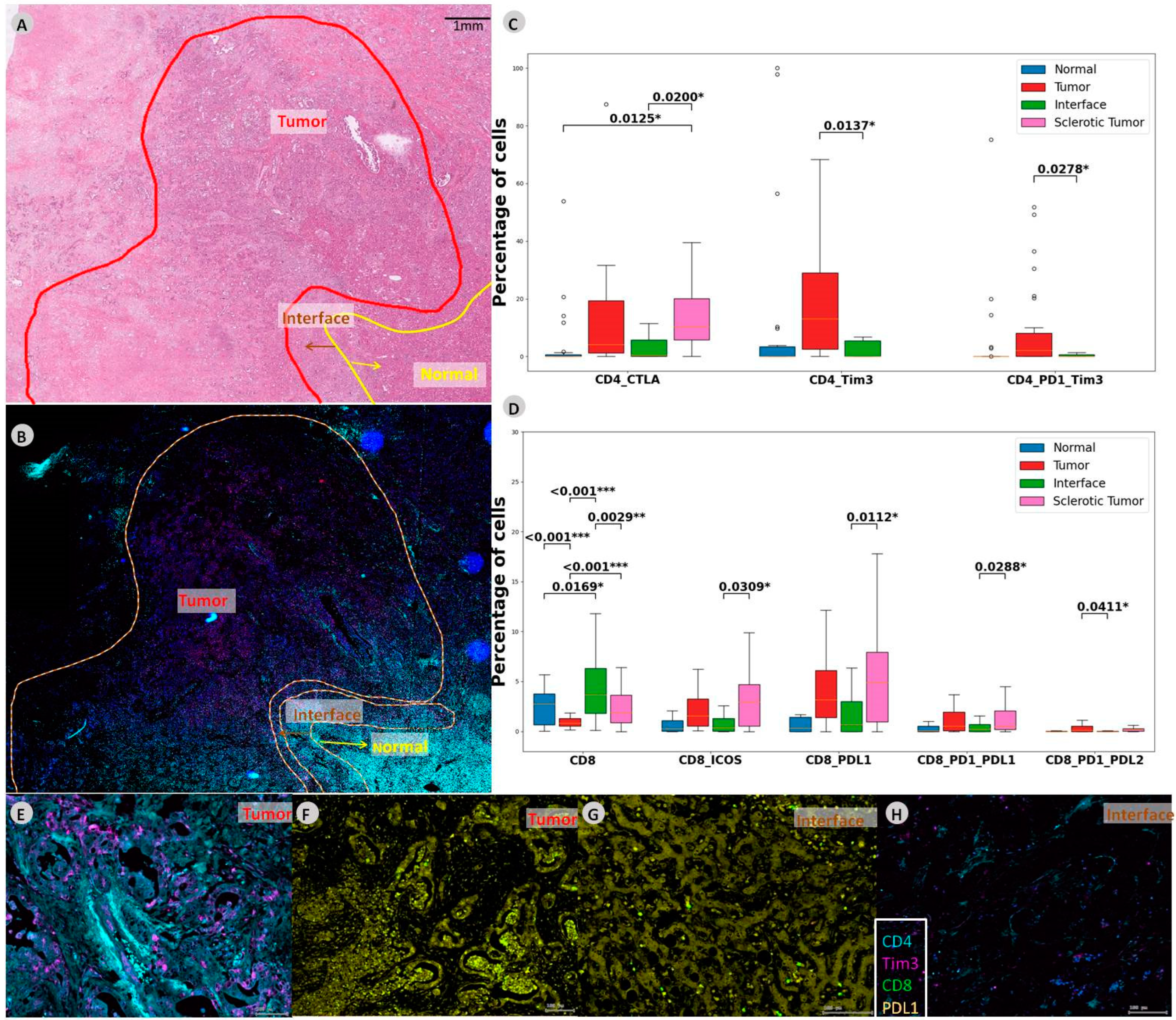
3.2. Sclerotic Tumor Demonstrates Higher Expression of CD8 with PD-L1
3.3. Binary Logistic Regression for Nodal Status with Respect to Immune Cell Subsets
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bertuccio, P.; Malvezzi, M.; Carioli, G.; Hashim, D.; Boffetta, P.; El-Serag, H.B.; La Vecchia, C.; Negri, E. Global trends in mortality from intrahepatic and extrahepatic cholangiocarcinoma. J. Hepatol. 2019, 71, 104–114. [Google Scholar] [CrossRef] [PubMed]
- Primrose, J.N.; Fox, R.P.; Palmer, D.H.; Malik, H.Z.; Prasad, R.; Mirza, D.; Anthony, A.; Corrie, P.; Falk, S.; Finch-Jones, M.; et al. Capecitabine compared with observation in resected biliary tract cancer (BILCAP): A randomised, controlled, multicentre, phase 3 study. Lancet Oncol. 2019, 20, 663–673. [Google Scholar] [CrossRef] [PubMed]
- Valle, J.W.; Lamarca, A.; Goyal, L.; Barriuso, J.; Zhu, A.X. New Horizons for Precision Medicine in Biliary Tract Cancers. Cancer Discov. 2017, 7, 943–962. [Google Scholar] [CrossRef] [PubMed]
- Valle, J.; Wasan, H.; Palmer, D.H.; Cunningham, D.; Anthoney, A.; Maraveyas, A.; Madhusudan, S.; Iveson, T.; Hughes, S.; Pereira, S.P.; et al. Cisplatin plus gemcitabine versus gemcitabine for biliary tract cancer. N. Engl. J. Med. 2010, 362, 1273–1281. [Google Scholar] [CrossRef] [PubMed]
- Martin-Serrano, M.A.; Kepecs, B.; Torres-Martin, M.; Bramel, E.R.; Haber, P.K.; Merritt, E.; Rialdi, A.; Param, N.J.; Maeda, M.; Lindblad, K.E.; et al. Novel microenvironment-based classification of intrahepatic cholangiocarcinoma with therapeutic implications. Gut 2022. [Google Scholar] [CrossRef]
- Cabrita, R.; Lauss, M.; Sanna, A.; Donia, M.; Skaarup Larsen, M.; Mitra, S.; Johansson, I.; Phung, B.; Harbst, K.; Vallon-Christersson, J.; et al. Tertiary lymphoid structures improve immunotherapy and survival in melanoma. Nature 2020, 577, 561–565. [Google Scholar] [CrossRef]
- Brahmer, J.; Reckamp, K.L.; Baas, P.; Crino, L.; Eberhardt, W.E.; Poddubskaya, E.; Antonia, S.; Pluzanski, A.; Vokes, E.E.; Holgado, E.; et al. Nivolumab versus Docetaxel in Advanced Squamous-Cell Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2015, 373, 123–135. [Google Scholar] [CrossRef]
- Yuan, J.; Hegde, P.S.; Clynes, R.; Foukas, P.G.; Harari, A.; Kleen, T.O.; Kvistborg, P.; Maccalli, C.; Maecker, H.T.; Page, D.B.; et al. Novel technologies and emerging biomarkers for personalized cancer immunotherapy. J. Immunother. Cancer 2016, 4, 3. [Google Scholar] [CrossRef]
- Gutiérrez-Larrañaga, M.; González-López, E.; Roa-Bautista, A.; Rodrigues, P.M.; Díaz-González, Á.; Banales, J.M.; López-Hoyos, M.; Santos-Laso, A.; Crespo, J. Immune Checkpoint Inhibitors: The Emerging Cornerstone in Cholangiocarcinoma Therapy? Liver Cancer 2021, 10, 545–560. [Google Scholar] [CrossRef]
- Jusakul, A.; Cutcutache, I.; Yong, C.H.; Lim, J.Q.; Huang, M.N.; Padmanabhan, N.; Nellore, V.; Kongpetch, S.; Ng, A.W.T.; Ng, L.M.; et al. Whole-Genome and Epigenomic Landscapes of Etiologically Distinct Subtypes of Cholangiocarcinoma. Cancer Discov. 2017, 7, 1116–1135. [Google Scholar] [CrossRef]
- Fabris, L.; Sato, K.; Alpini, G.; Strazzabosco, M. The Tumor Microenvironment in Cholangiocarcinoma Progression. Hepatology 2021, 73 (Suppl. 1), 75–85. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Loeuillard, E.; Gores, G.J.; Ilyas, S.I. Cholangiocarcinoma: What are the most valuable therapeutic targets—Cancer-associated fibroblasts, immune cells, or beyond T cells? Expert Opin. Ther. Targets 2021, 25, 835–845. [Google Scholar] [CrossRef]
- Fabris, L.; Perugorria, M.J.; Mertens, J.; Björkström, N.K.; Cramer, T.; Lleo, A.; Solinas, A.; Sänger, H.; Lukacs-Kornek, V.; Moncsek, A.; et al. The tumour microenvironment and immune milieu of cholangiocarcinoma. Liver Int. 2019, 39 (Suppl. S1), 63–78. [Google Scholar] [CrossRef] [PubMed]
- Loeuillard, E.; Conboy, C.B.; Gores, G.J.; Rizvi, S. Immunobiology of cholangiocarcinoma. JHEP Rep. Innov. Hepatol. 2019, 1, 297–311. [Google Scholar] [CrossRef] [PubMed]
- Kato, S.; Okamura, R.; Kumaki, Y.; Ikeda, S.; Nikanjam, M.; Eskander, R.; Goodman, A.; Lee, S.; Glenn, S.T.; Dressman, D.; et al. Expression of TIM3/VISTA checkpoints and the CD68 macrophage-associated marker correlates with anti-PD1/PDL1 resistance: Implications of immunogram heterogeneity. Oncoimmunology 2020, 9, 1708065. [Google Scholar] [CrossRef] [PubMed]
- Tian, L.; Ma, J.; Ma, L.; Zheng, B.; Liu, L.; Song, D.; Wang, Y.; Zhang, Z.; Gao, Q.; Song, K.; et al. PD-1/PD-L1 expression profiles within intrahepatic cholangiocarcinoma predict clinical outcome. World J. Surg. Oncol. 2020, 18, 303. [Google Scholar] [CrossRef] [PubMed]
- Oh, D.-Y.; He, A.R.; Qin, S.; Chen, L.-T.; Okusaka, T.; Vogel, A.; Kim, J.W.; Suksombooncharoen, T.; Lee, M.A.; Kitano, M.; et al. Durvalumab plus Gemcitabine and Cisplatin in Advanced Biliary Tract Cancer. NEJM Evid. 2022, 1, EVIDoa2200015. [Google Scholar] [CrossRef]
- Kam, A.E.; Masood, A.; Shroff, R.T. Current and emerging therapies for advanced biliary tract cancers. Lancet Gastroenterol. Hepatol. 2021, 6, 956–969. [Google Scholar] [CrossRef]
- Kim, H.; Kim, J.; Byeon, S.; Jang, K.T.; Hong, J.Y.; Lee, J.; Park, S.H.; Park, J.O.; Park, Y.S.; Lim, H.Y.; et al. Programmed Death Ligand 1 Expression as a Prognostic Marker in Patients with Advanced Biliary Tract Cancer. Oncology 2021, 99, 365–372. [Google Scholar] [CrossRef]
- Dievernich, A.; Achenbach, P.; Davies, L.; Klinge, U. Tissue remodeling macrophages morphologically dominate at the interface of polypropylene surgical meshes in the human abdomen. Hernia 2020, 24, 1175–1189. [Google Scholar] [CrossRef]
- Ma, L.; Wang, L.; Khatib, S.A.; Chang, C.W.; Heinrich, S.; Dominguez, D.A.; Forgues, M.; Candia, J.; Hernandez, M.O.; Kelly, M.; et al. Single-cell atlas of tumor cell evolution in response to therapy in hepatocellular carcinoma and intrahepatic cholangiocarcinoma. J. Hepatol. 2021, 75, 1397–1408. [Google Scholar] [CrossRef] [PubMed]
- Carapeto, F.; Bozorgui, B.; Shroff, R.T.; Chagani, S.; Solis Soto, L.; Foo, W.C.; Wistuba, I.; Meric-Bernstam, F.; Shalaby, A.; Javle, M.; et al. The Immunogenomic Landscape of Resected Intrahepatic Cholangiocarcinoma. Hepatology 2021, 75, 297–308. [Google Scholar] [CrossRef] [PubMed]
- Rodenhiser, D.; Mann, M. Epigenetics and human disease: Translating basic biology into clinical applications. CMAJ 2006, 174, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Yang, H.; Wan, L.; Wang, Z.; Wang, H.; Ge, C.; Liu, Y.; Hao, Y.; Zhang, D.; Shi, G.; et al. Single-cell transcriptomic architecture and intercellular crosstalk of human intrahepatic cholangiocarcinoma. J. Hepatol. 2020, 73, 1118–1130. [Google Scholar] [CrossRef] [PubMed]
- Goeppert, B.; Frauenschuh, L.; Zucknick, M.; Stenzinger, A.; Andrulis, M.; Klauschen, F.; Joehrens, K.; Warth, A.; Renner, M.; Mehrabi, A.; et al. Prognostic impact of tumour-infiltrating immune cells on biliary tract cancer. Br. J. Cancer 2013, 109, 2665–2674. [Google Scholar] [CrossRef] [PubMed]
- Kitano, Y.; Okabe, H.; Yamashita, Y.I.; Nakagawa, S.; Saito, Y.; Umezaki, N.; Tsukamoto, M.; Yamao, T.; Yamamura, K.; Arima, K.; et al. Tumour-infiltrating inflammatory and immune cells in patients with extrahepatic cholangiocarcinoma. Br. J. Cancer 2018, 118, 171–180. [Google Scholar] [CrossRef]
- Job, S.; Rapoud, D.; Dos Santos, A.; Gonzalez, P.; Desterke, C.; Pascal, G.; Elarouci, N.; Ayadi, M.; Adam, R.; Azoulay, D.; et al. Identification of Four Immune Subtypes Characterized by Distinct Composition and Functions of Tumor Microenvironment in Intrahepatic Cholangiocarcinoma. Hepatology 2020, 72, 965–981. [Google Scholar] [CrossRef] [PubMed]
- Konishi, D.; Umeda, Y.; Yoshida, K.; Shigeyasu, K.; Yano, S.; Toji, T.; Takeda, S.; Yoshida, R.; Yasui, K.; Fuji, T.; et al. Regulatory T cells induce a suppressive immune milieu and promote lymph node metastasis in intrahepatic cholangiocarcinoma. Br. J. Cancer 2022, 127, 757–765. [Google Scholar] [CrossRef]
- Zhang, X.F.; Xue, F.; Dong, D.H.; Weiss, M.; Popescu, I.; Marques, H.P.; Aldrighetti, L.; Maithel, S.K.; Pulitano, C.; Bauer, T.W.; et al. Number and Station of Lymph Node Metastasis After Curative-intent Resection of Intrahepatic Cholangiocarcinoma Impact Prognosis. Ann. Surg. 2021, 274, e1187–e1195. [Google Scholar] [CrossRef]
- Sahara, K.; Tsilimigras, D.I.; Merath, K.; Bagante, F.; Guglielmi, A.; Aldrighetti, L.; Weiss, M.; Bauer, T.W.; Alexandrescu, S.; Poultsides, G.A.; et al. Therapeutic Index Associated with Lymphadenectomy Among Patients with Intrahepatic Cholangiocarcinoma: Which Patients Benefit the Most from Nodal Evaluation? Ann. Surg. Oncol. 2019, 26, 2959–2968. [Google Scholar] [CrossRef]
- Weber, S.M.; Ribero, D.; O’Reilly, E.M.; Kokudo, N.; Miyazaki, M.; Pawlik, T.M. Intrahepatic cholangiocarcinoma: Expert consensus statement. HPB 2015, 17, 669–680. [Google Scholar] [CrossRef] [PubMed]
- Bagante, F.; Spolverato, G.; Weiss, M.; Alexandrescu, S.; Marques, H.P.; Aldrighetti, L.; Maithel, S.K.; Pulitano, C.; Bauer, T.W.; Shen, F.; et al. Assessment of the Lymph Node Status in Patients Undergoing Liver Resection for Intrahepatic Cholangiocarcinoma: The New Eighth Edition AJCC Staging System. J. Gastrointest. Surg. 2018, 22, 52–59. [Google Scholar] [CrossRef] [PubMed]


| ICCA | ||
|---|---|---|
| Total | Percentage | |
| Included patients | 50 | 100% |
| Gender | ||
| %Male | 24 | 48% |
| %Female | 26 | 52% |
| Median age (years) | 67 | |
| Multifocal tumor | ||
| %Yes | 17 | 34% |
| %No | 33 | 66% |
| Tumor stadium (T) | ||
| %UICC T1 | 17 | 34% |
| %UICC T2 | 24 | 48% |
| %UICC T3 | 6 | 12% |
| %UICC T4 | 3 | 6% |
| Nodal status (N) | ||
| %N0 | 32 | 64% |
| %N1 + N2 | 16 | 32% |
| %not known | 2 | 4% |
| Tumor grading (G) | ||
| %G1 | 0 | 0% |
| %G2 | 37 | 74% |
| %G3 | 10 | 20% |
| %not known | 3 | 6% |
| Perineural invasion (Pn) | ||
| %Pn0 | 10 | 20% |
| %Pn1 | 13 | 26% |
| %not known | 27 | 54% |
| Residual tumor (R) | ||
| %R0 | 46 | 92% |
| %R1 | 4 | 8% |
| Lymphovascular invasion (L) | ||
| %L0 | 38 | 76% |
| %L1 | 9 | 18% |
| %not known | 3 | 6% |
| Median overall survival (months) | 32 | |
| iCCA-ROI | Number |
|---|---|
| Normal | 30 |
| Tumor | 45 |
| Interface | 30 |
| Scleotic tumor | 32 |
| Immune Cell Subset | Perihilar Cholangiocarcinoma | Intrahepatic Cholangiocarcinoma | ||
|---|---|---|---|---|
| OR (95%CI) | p-Value | OR (95%CI) | p-Value | |
| Univariate analysis | ||||
| Tumor_CD68 | 4.09 (1.16–14.43) | 0.028 | ||
| Tumor_CD8_PD-L1_PD-L2 | 4.72 (1.07–20.89) | 0.041 | ||
| Tumor_CD8_PD-L2 | 4.72 (1.07–20.89) | 0.041 | ||
| Tumor_CD4_ICOS_TIGIT | 5.36 (1.25–23.04) | 0.024 | ||
| Tumor_CD4_TIGIT | 6.11 (1.41–26.41) | 0.015 | ||
| Tumor_CD8_ICOS_TIGIT | 6.11 (1.41–26.41) | 0.015 | ||
| Tumor_CD8_ICOS_CTLA-4 | 6.11 (1.41–26.41) | 0.015 | ||
| Liver_CD8_CTLA-4 | 0.13 (0.20–0.78) | 0.026 | ||
| Liver_CD8_TIM3 | 0.13 (0.20–0.78) | 0.026 | ||
| Multivariate analysis | ||||
| Tumor_CD8_PD-L2 | 5.24 (1.01–27.18) | 0.048 | ||
| Tumor_CD8_ICOS_TIGIT | 8.05 (1.60–40.53) | 0.011 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Heij, L.; Bednarsch, J.; Tan, X.; Rosin, M.; Appinger, S.; Reichel, K.; Pecina, D.; Doukas, M.; van Dam, R.M.; Garcia Vallejo, J.; et al. Expression of Checkpoint Molecules in the Tumor Microenvironment of Intrahepatic Cholangiocarcinoma: Implications for Immune Checkpoint Blockade Therapy. Cells 2023, 12, 851. https://doi.org/10.3390/cells12060851
Heij L, Bednarsch J, Tan X, Rosin M, Appinger S, Reichel K, Pecina D, Doukas M, van Dam RM, Garcia Vallejo J, et al. Expression of Checkpoint Molecules in the Tumor Microenvironment of Intrahepatic Cholangiocarcinoma: Implications for Immune Checkpoint Blockade Therapy. Cells. 2023; 12(6):851. https://doi.org/10.3390/cells12060851
Chicago/Turabian StyleHeij, Lara, Jan Bednarsch, Xiuxiang Tan, Mika Rosin, Simone Appinger, Konrad Reichel, Dana Pecina, Michail Doukas, Ronald M. van Dam, Juan Garcia Vallejo, and et al. 2023. "Expression of Checkpoint Molecules in the Tumor Microenvironment of Intrahepatic Cholangiocarcinoma: Implications for Immune Checkpoint Blockade Therapy" Cells 12, no. 6: 851. https://doi.org/10.3390/cells12060851
APA StyleHeij, L., Bednarsch, J., Tan, X., Rosin, M., Appinger, S., Reichel, K., Pecina, D., Doukas, M., van Dam, R. M., Garcia Vallejo, J., Ulmer, F., Lang, S., Luedde, T., Rocha, F. G., Sivakumar, S., & Neumann, U. P. (2023). Expression of Checkpoint Molecules in the Tumor Microenvironment of Intrahepatic Cholangiocarcinoma: Implications for Immune Checkpoint Blockade Therapy. Cells, 12(6), 851. https://doi.org/10.3390/cells12060851






