Ezrin and Its Phosphorylated Thr567 Form Are Key Regulators of Human Extravillous Trophoblast Motility and Invasion
Abstract
1. Introduction
2. Materials and Methods
2.1. Human Placental Tissues
2.2. Cell Lines and Culture
2.3. EVT Purification
2.4. siRNA Ezrin and Control Delivery
2.5. Western Blotting
2.6. Immunofluorescent Staining
2.7. Immunohistochemistry
2.8. Motility/Invasion Assay
2.9. Trypan Blue Exclusion/MTT Conversion
3. Results
3.1. Ezrin Is Expressed in Human Extravillous Trophoblasts in Anchoring Columns In Vivo
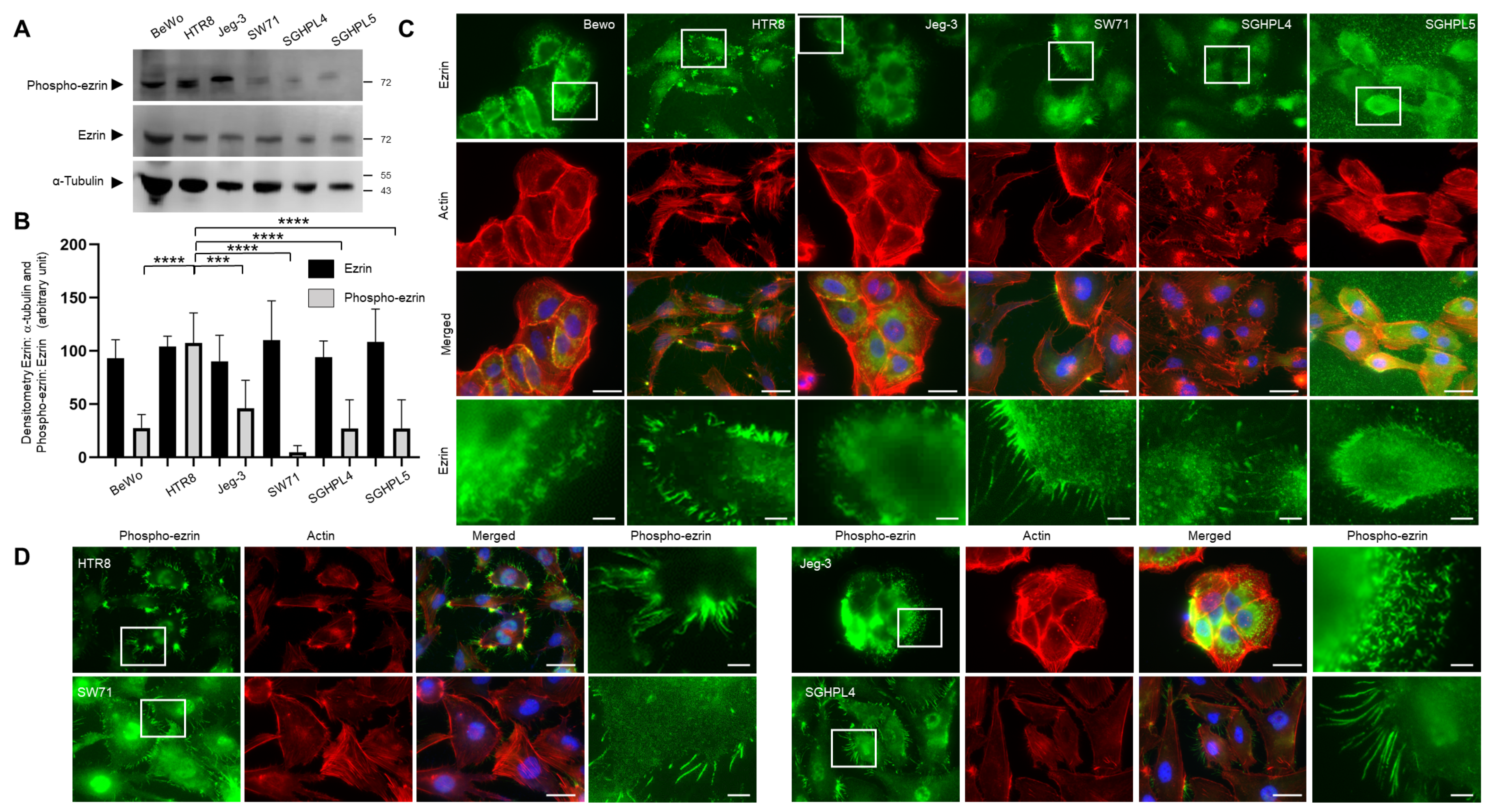
3.2. Expression and Localisation of Ezrin in Trophoblast Cell Lines
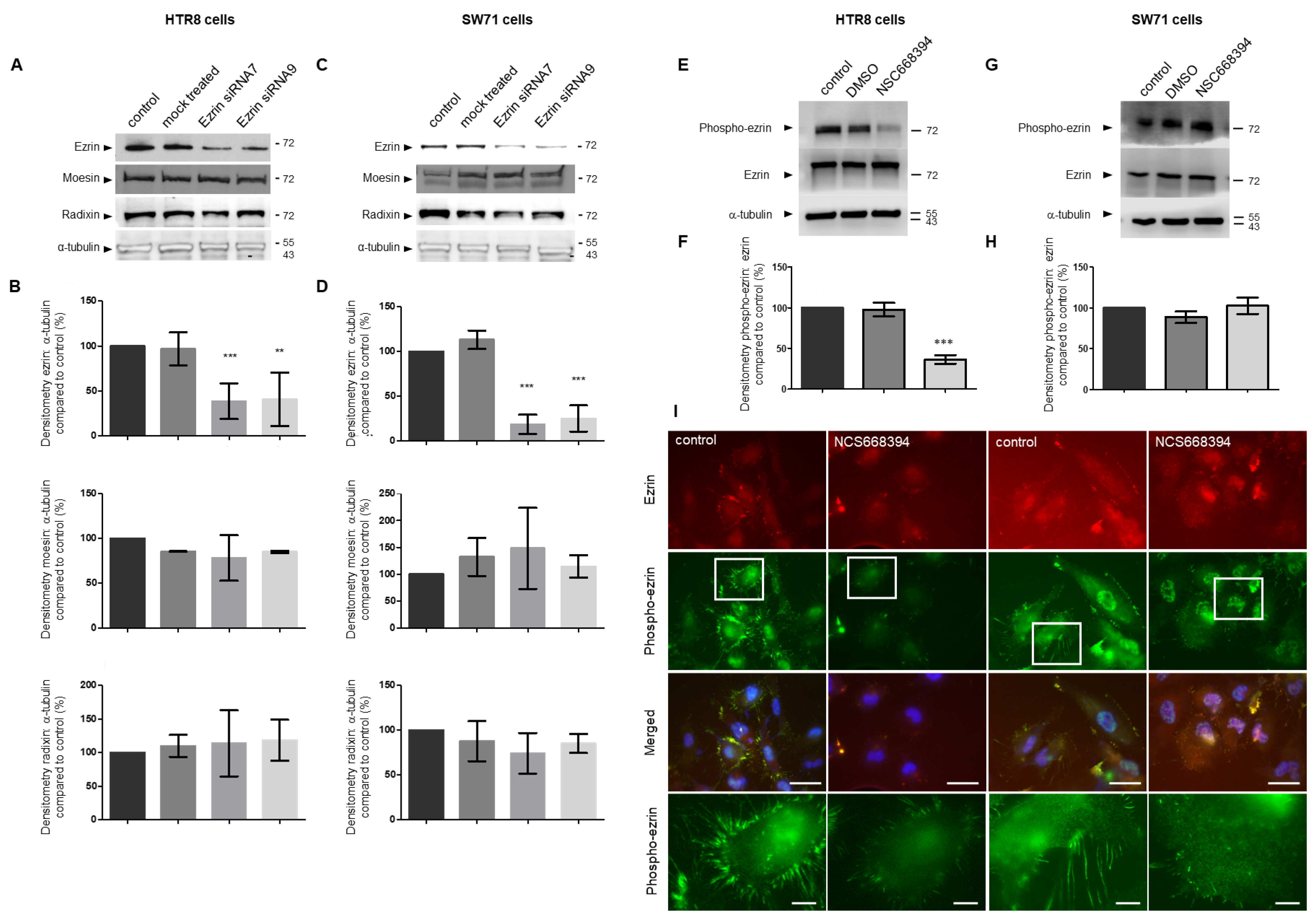
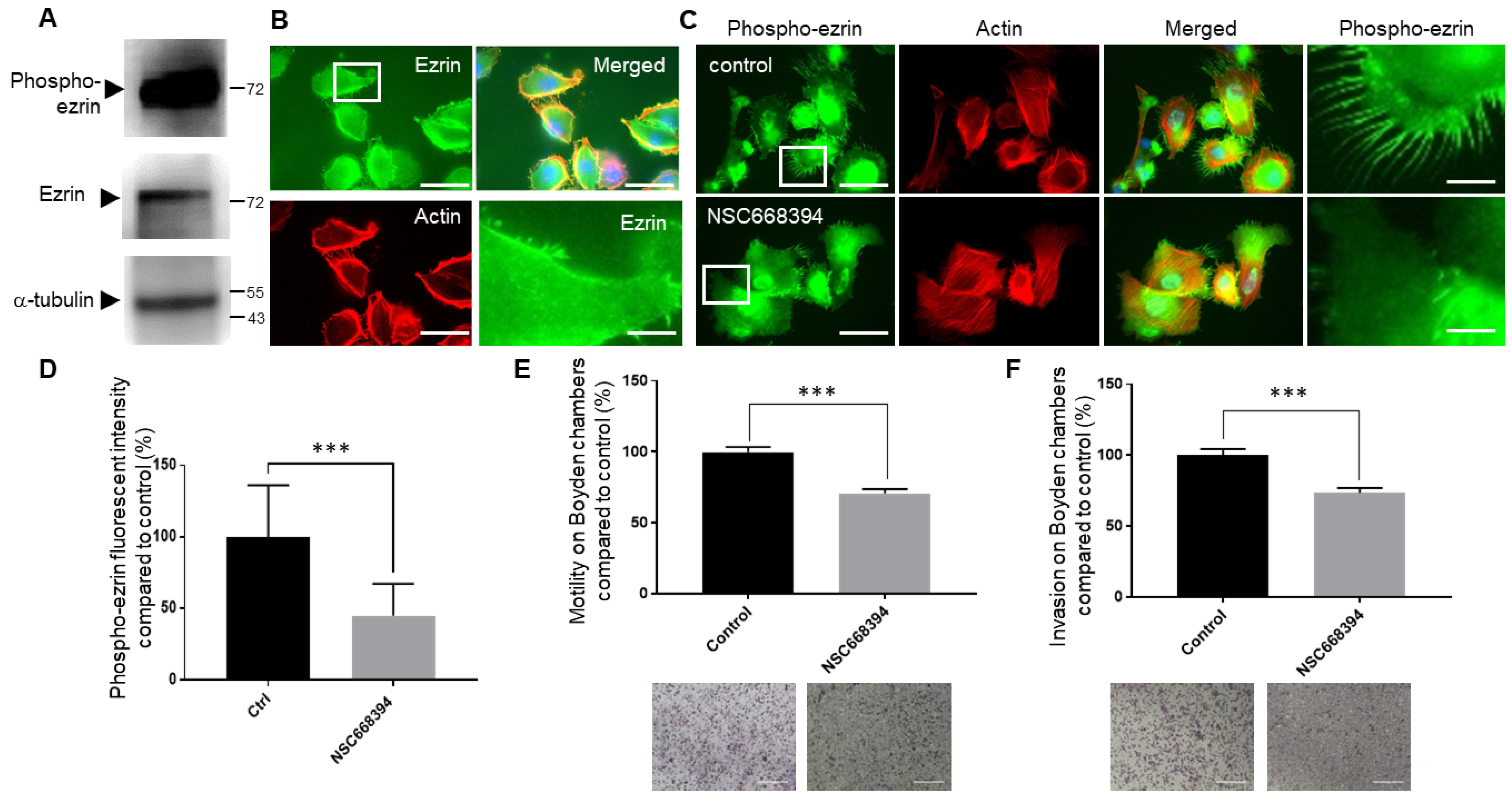
3.3. Regulating the Expression and Activity Levels of Ezrin in EVT Cell Lines
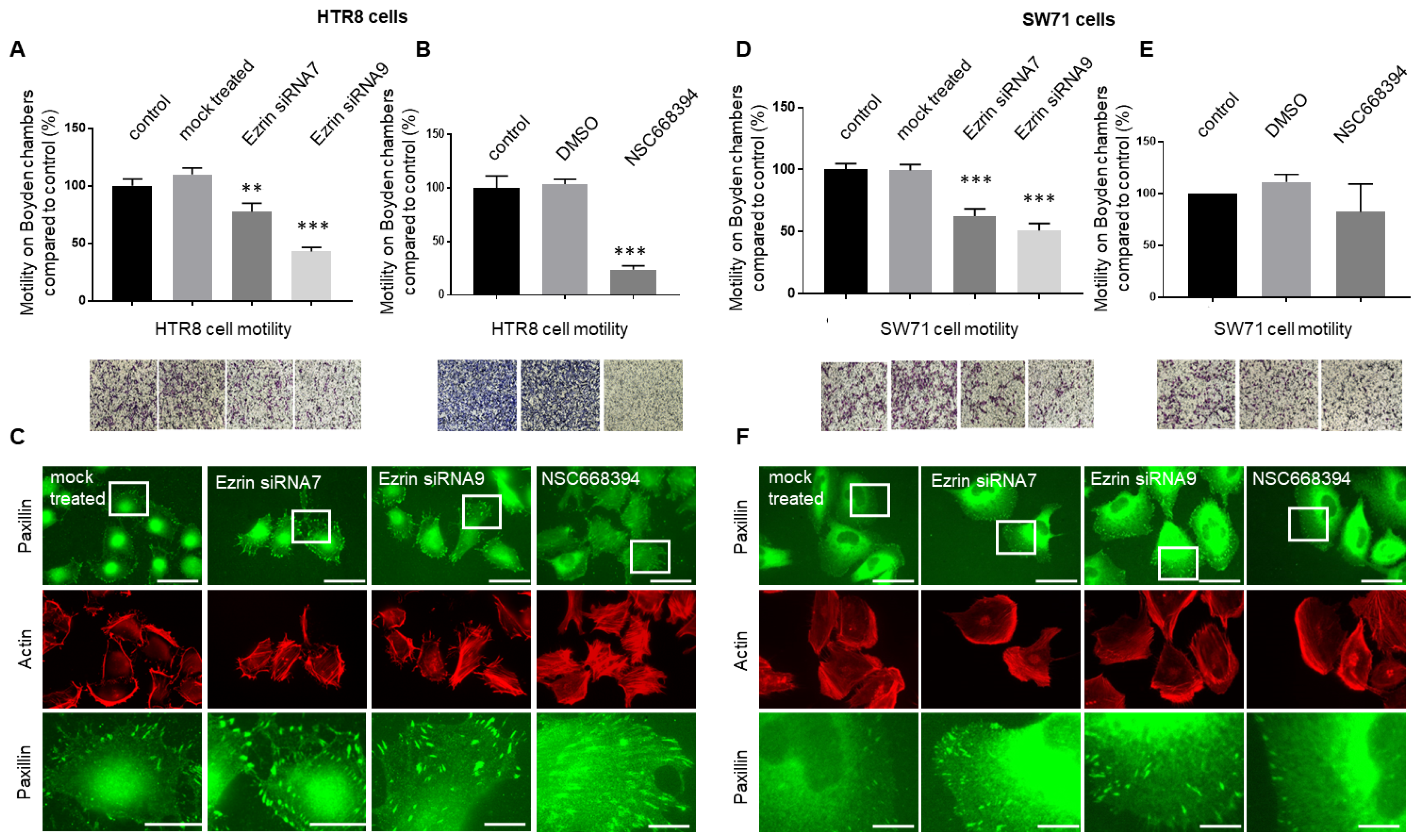
3.4. The Knock-Down or Inactivation of Ezrin Results in Significant Reductions in EVT-like Cell Motility
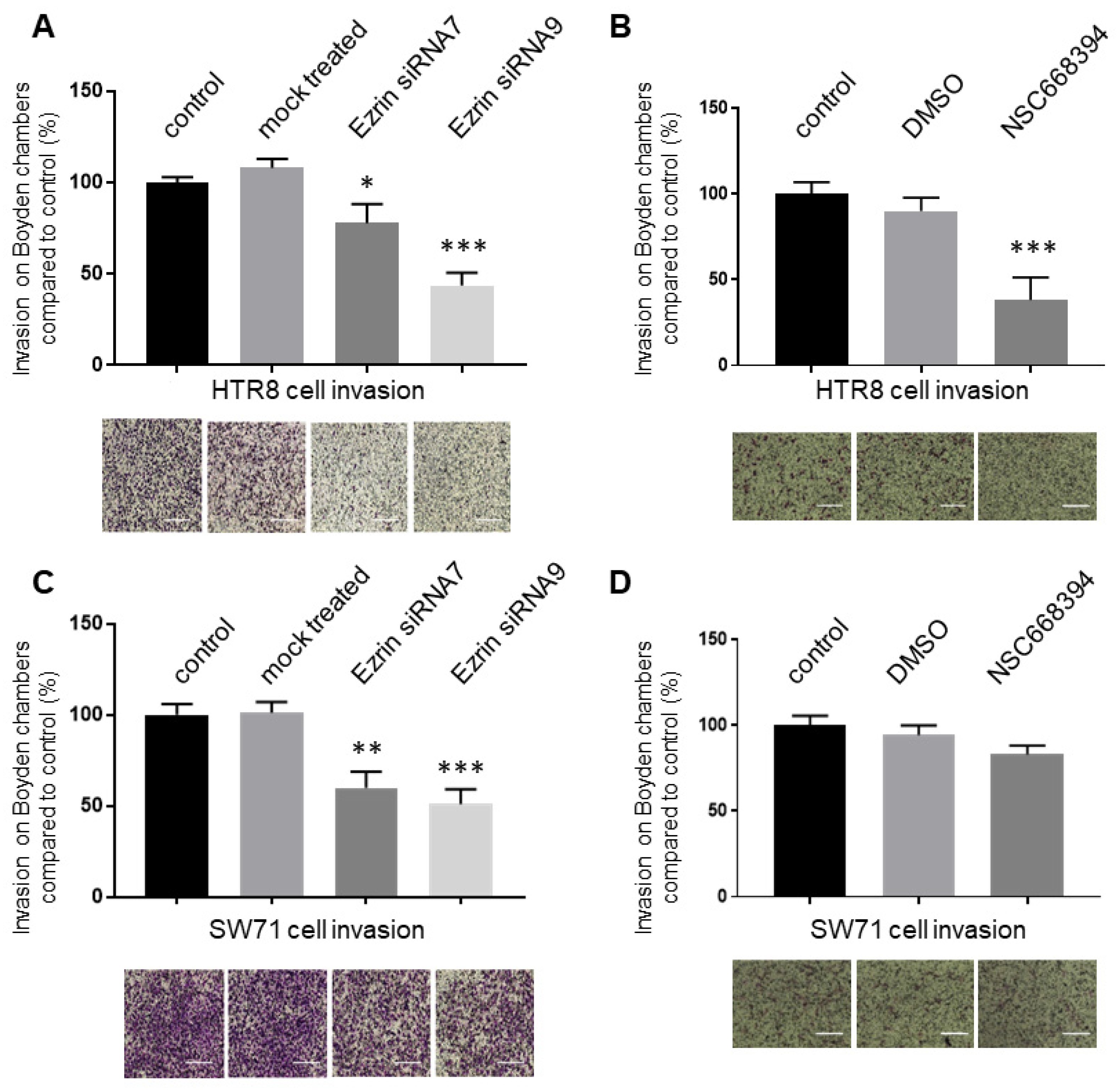
3.5. The Knock-Down or Inactivation of Ezrin Results in Significant Reductions in EVT Cell Invasion
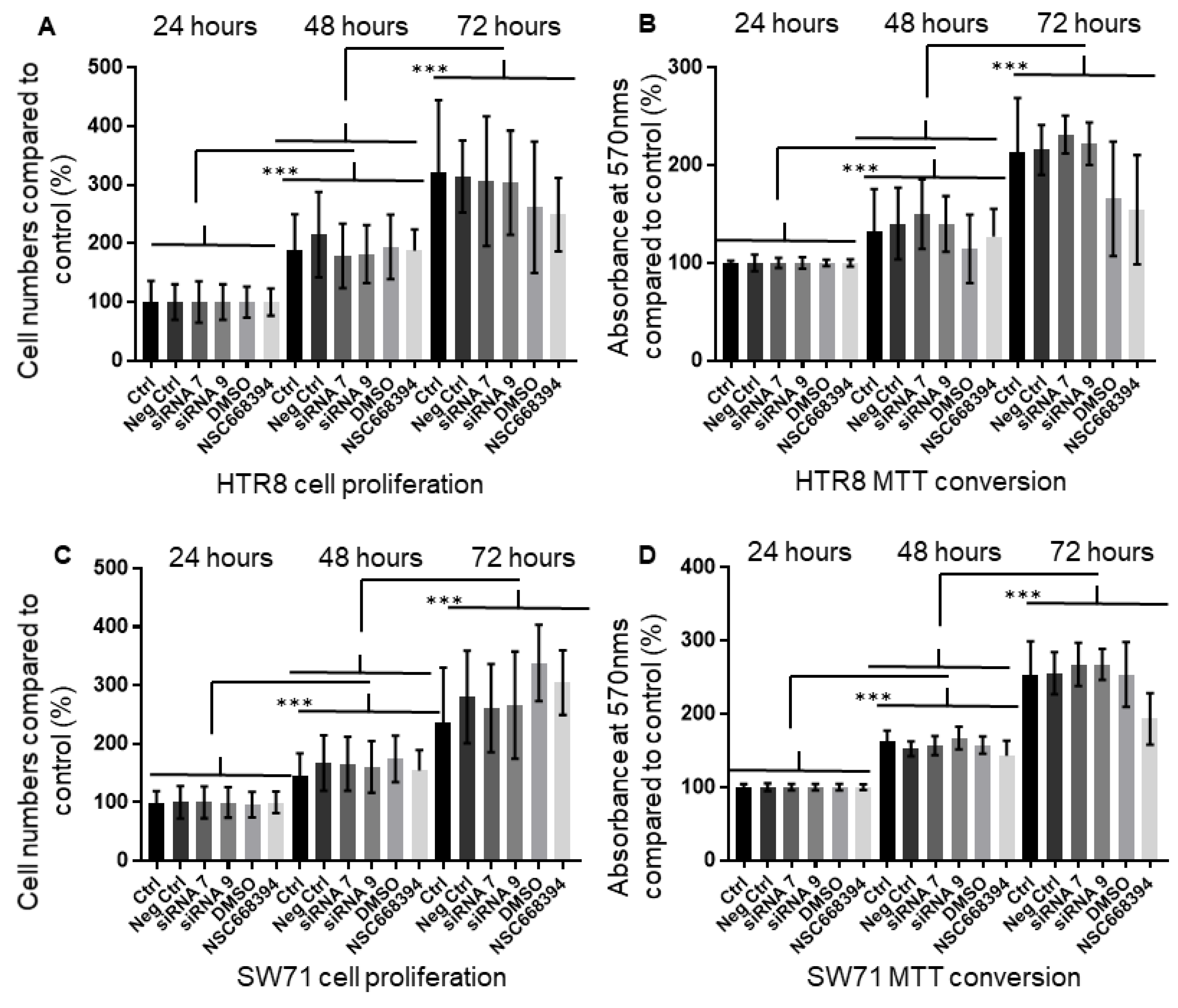
3.6. The Knock-Down or Inactivation of Ezrin Does Not Lead to Changes in Cell Viability and Proliferation
3.7. Inhibiting Ezrin Phosphorylation in Primary Extravillous Trophoblasts Results in a Significant Reduction in Motility and Invasion
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nishimura, T.; Higuchi, K.; Sai, Y.; Sugita, Y.; Yoshida, Y.; Tomi, M.; Wada, M.; Wakayama, T.; Tamura, A.; Tsukita, S.; et al. Fetal growth retardation and lack of hypotaurine in ezrin knockout mice. PLoS ONE 2014, 9, e105423. [Google Scholar] [CrossRef] [PubMed]
- Riquelme, G.; Vallejos, C.; de Gregorio, N.; Morales, B.; Godoy, V.; Berrios, M.; Bastias, N.; Rodriguez, C. Lipid rafts and cytoskeletal proteins in placental microvilli membranes from preeclamptic and IUGR pregnancies. J. Membr. Biol. 2011, 241, 127–140. [Google Scholar] [CrossRef] [PubMed]
- Kawaguchi, K.; Asano, S. Pathophysiological Roles of Actin-Binding Scaffold Protein, Ezrin. Int. J. Mol. Sci. 2022, 23, 3246. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Flores, L.R.; Keeling, M.C.; Sliogeryte, K.; Gavara, N. Ezrin Phosphorylation at T567 Modulates Cell Migration, Mechanical Properties, and Cytoskeletal Organization. Int. J. Mol. Sci. 2020, 21, 435. [Google Scholar] [CrossRef]
- Rouven Bruckner, B.; Pietuch, A.; Nehls, S.; Rother, J.; Janshoff, A. Ezrin is a Major Regulator of Membrane Tension in Epithelial Cells. Sci. Rep. 2015, 5, 14700. [Google Scholar] [CrossRef]
- Poullet, P.; Gautreau, A.; Kadare, G.; Girault, J.A.; Louvard, D.; Arpin, M. Ezrin interacts with focal adhesion kinase and induces its activation independently of cell-matrix adhesion. J. Biol. Chem. 2001, 276, 37686–37691. [Google Scholar] [CrossRef]
- Maniti, O.; Carvalho, K.; Picart, C. Model membranes to shed light on the biochemical and physical properties of ezrin/radixin/moesin. Biochimie 2013, 95, 3–11. [Google Scholar] [CrossRef]
- Fehon, R.G.; McClatchey, A.I.; Bretscher, A. Organizing the cell cortex: The role of ERM proteins. Nat. Rev. Mol. Cell Biol. 2010, 11, 276–287. [Google Scholar] [CrossRef]
- Garbett, D.; Bretscher, A. PDZ interactions regulate rapid turnover of the scaffolding protein EBP50 in microvilli. J. Cell Biol. 2012, 198, 195–203. [Google Scholar] [CrossRef]
- Gary, R.; Bretscher, A. Ezrin self-association involves binding of an N-terminal domain to a normally masked C-terminal domain that includes the F-actin binding site. Mol. Biol. Cell 1995, 6, 1061–1075. [Google Scholar] [CrossRef]
- Janke, M.; Herrig, A.; Austermann, J.; Gerke, V.; Steinem, C.; Janshoff, A. Actin binding of ezrin is activated by specific recognition of PIP2-functionalized lipid bilayers. Biochemistry 2008, 47, 3762–3769. [Google Scholar] [CrossRef] [PubMed]
- Fievet, B.T.; Gautreau, A.; Roy, C.; Del Maestro, L.; Mangeat, P.; Louvard, D.; Arpin, M. Phosphoinositide binding and phosphorylation act sequentially in the activation mechanism of ezrin. J. Cell Biol. 2004, 164, 653–659. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Zhou, R.; Mettler, S.; Wu, T.; Abbas, A.; Delaney, J.; Forte, J.G. High turnover of ezrin T567 phosphorylation: Conformation, activity, and cellular function. Am. J. Physiol. Cell Physiol. 2007, 293, C874–C884. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Ma, X.; Zhang, M.; Wang, M.; Wang, G.; Ye, Y.; Xia, W. Ezrin Mediates Invasion and Metastasis in Tumorigenesis: A Review. Front. Cell Dev. Biol. 2020, 8, 588801. [Google Scholar] [CrossRef] [PubMed]
- Valdman, A.; Fang, X.; Pang, S.T.; Nilsson, B.; Ekman, P.; Egevad, L. Ezrin expression in prostate cancer and benign prostatic tissue. Eur. Urol. 2005, 48, 852–857. [Google Scholar] [CrossRef]
- Chuan, Y.C.; Iglesias-Gato, D.; Fernandez-Perez, L.; Cedazo-Minguez, A.; Pang, S.T.; Norstedt, G.; Pousette, A.; Flores-Morales, A. Ezrin mediates c-Myc actions in prostate cancer cell invasion. Oncogene 2010, 29, 1531–1542. [Google Scholar] [CrossRef] [PubMed]
- Meng, Y.; Lu, Z.; Yu, S.; Zhang, Q.; Ma, Y.; Chen, J. Ezrin promotes invasion and metastasis of pancreatic cancer cells. J. Transl. Med. 2010, 8, 61. [Google Scholar] [CrossRef]
- Ghaffari, A.; Hoskin, V.; Turashvili, G.; Varma, S.; Mewburn, J.; Mullins, G.; Greer, P.A.; Kiefer, F.; Day, A.G.; Madarnas, Y.; et al. Intravital imaging reveals systemic ezrin inhibition impedes cancer cell migration and lymph node metastasis in breast cancer. Breast Cancer Res. 2019, 21, 12. [Google Scholar] [CrossRef]
- Hunter, K.W. Ezrin, a key component in tumor metastasis. Trends Mol. Med. 2004, 10, 201–204. [Google Scholar] [CrossRef]
- Sarrio, D.; Rodriguez-Pinilla, S.M.; Dotor, A.; Calero, F.; Hardisson, D.; Palacios, J. Abnormal ezrin localization is associated with clinicopathological features in invasive breast carcinomas. Breast Cancer Res. Treat. 2006, 98, 71–79. [Google Scholar] [CrossRef]
- Matsumoto, H.; Daikoku, T.; Wang, H.; Sato, E.; Dey, S.K. Differential expression of ezrin/radixin/moesin (ERM) and ERM-associated adhesion molecules in the blastocyst and uterus suggests their functions during implantation. Biol. Reprod. 2004, 70, 729–736. [Google Scholar] [CrossRef]
- Berryman, M.; Gary, R.; Bretscher, A. Ezrin oligomers are major cytoskeletal components of placental microvilli: A proposal for their involvement in cortical morphogenesis. J. Cell Biol. 1995, 131, 1231–1242. [Google Scholar] [CrossRef] [PubMed]
- Higuchi, K.; Iizasa, H.; Sai, Y.; Horieya, S.; Lee, K.E.; Wada, M.; Deguchi, M.; Nishimura, T.; Wakayama, T.; Tamura, A.; et al. Differential expression of ezrin and CLP36 in the two layers of syncytiotrophoblast in rats. Biol. Pharm. Bull. 2010, 33, 1400–1406. [Google Scholar] [CrossRef] [PubMed]
- Paradela, A.; Bravo, S.B.; Henriquez, M.; Riquelme, G.; Gavilanes, F.; Gonzalez-Ros, J.M.; Albar, J.P. Proteomic analysis of apical microvillous membranes of syncytiotrophoblast cells reveals a high degree of similarity with lipid rafts. J. Proteome Res. 2005, 4, 2435–2441. [Google Scholar] [CrossRef]
- Narvanen, A. Purification, from cultured human choriocarcinoma cells, of a 75000-Mr protein reacting with antibodies to a synthetic peptide based on a cloned human endogenous provirus nucleotide sequence. Biochem. J. 1985, 231, 53–57. [Google Scholar] [CrossRef] [PubMed]
- Miura, S.; Sato, K.; Kato-Negishi, M.; Teshima, T.; Takeuchi, S. Fluid shear triggers microvilli formation via mechanosensitive activation of TRPV6. Nat. Commun. 2015, 6, 8871. [Google Scholar] [CrossRef] [PubMed]
- Karmakar, S.; Das, C. Modulation of ezrin and E-cadherin expression by IL-1beta and TGF-beta1 in human trophoblasts. J. Reprod. Immunol. 2004, 64, 9–29. [Google Scholar] [CrossRef] [PubMed]
- Ferretti, C.; Bruni, L.; Dangles-Marie, V.; Pecking, A.P.; Bellet, D. Molecular circuits shared by placental and cancer cells, and their implications in the proliferative, invasive and migratory capacities of trophoblasts. Hum. Reprod. Update 2007, 13, 121–141. [Google Scholar] [CrossRef]
- Costanzo, V.; Bardelli, A.; Siena, S.; Abrignani, S. Exploring the links between cancer and placenta development. Open Biol. 2018, 8, 180081. [Google Scholar] [CrossRef]
- Tabrizi, M.E.A.; Lancaster, T.L.; Ismail, T.M.; Georgiadou, A.; Ganguly, A.; Mistry, J.J.; Wang, K.; Rudland, P.S.; Ahmad, S.; Gross, S.R. S100P enhances the motility and invasion of human trophoblast cell lines. Sci. Rep. 2018, 8, 11488. [Google Scholar] [CrossRef]
- Straszewski-Chavez, S.L.; Abrahams, V.M.; Alvero, A.B.; Aldo, P.B.; Ma, Y.; Guller, S.; Romero, R.; Mor, G. The isolation and characterization of a novel telomerase immortalized first trimester trophoblast cell line, Swan 71. Placenta 2009, 30, 939–948. [Google Scholar] [CrossRef] [PubMed]
- McCormick, J.; Whitley, G.S.; Le Bouteiller, P.; Cartwright, J.E. Soluble HLA-G regulates motility and invasion of the trophoblast-derived cell line SGHPL-4. Hum. Reprod. 2009, 24, 1339–1345. [Google Scholar] [CrossRef] [PubMed]
- Haslinger, P.; Haider, S.; Sonderegger, S.; Otten, J.V.; Pollheimer, J.; Whitley, G.; Knofler, M. AKT isoforms 1 and 3 regulate basal and epidermal growth factor-stimulated SGHPL-5 trophoblast cell migration in humans. Biol. Reprod. 2013, 88, 54. [Google Scholar] [CrossRef] [PubMed]
- Bulut, G.; Hong, S.H.; Chen, K.; Beauchamp, E.M.; Rahim, S.; Kosturko, G.W.; Glasgow, E.; Dakshanamurthy, S.; Lee, H.S.; Daar, I.; et al. Small molecule inhibitors of ezrin inhibit the invasive phenotype of osteosarcoma cells. Oncogene 2012, 31, 269–281. [Google Scholar] [CrossRef]
- Male, V.; Gardner, L.; Moffett, A. Isolation of cells from the feto-maternal interface. Curr. Protoc. Immunol. 2012, 97, 7.40.1–7.40.11. [Google Scholar] [CrossRef]
- Goh Then Sin, C.; Hersch, N.; Rudland, P.S.; Barraclough, R.; Hoffmann, B.; Gross, S.R. S100A4 downregulates filopodia formation through increased dynamic instability. Cell Adh. Migr. 2011, 5, 439–447. [Google Scholar] [CrossRef]
- Louvet, S.; Aghion, J.; Santa-Maria, A.; Mangeat, P.; Maro, B. Ezrin becomes restricted to outer cells following asymmetrical division in the preimplantation mouse embryo. Dev. Biol. 1996, 177, 568–579. [Google Scholar] [CrossRef]
- Hanono, A.; Garbett, D.; Reczek, D.; Chambers, D.N.; Bretscher, A. EPI64 regulates microvillar subdomains and structure. J. Cell Biol. 2006, 175, 803–813. [Google Scholar] [CrossRef]
- Proudfit, A.; Bhunia, N.; Pore, D.; Parker, Y.; Lindner, D.; Gupta, N. Pharmacologic Inhibition of Ezrin-Radixin-Moesin Phosphorylation is a Novel Therapeutic Strategy in Rhabdomyosarcoma. Sarcoma 2020, 2020, 9010496. [Google Scholar] [CrossRef]
- Morales, F.C.; Takahashi, Y.; Kreimann, E.L.; Georgescu, M.M. Ezrin-radixin-moesin (ERM)-binding phosphoprotein 50 organizes ERM proteins at the apical membrane of polarized epithelia. Proc. Natl. Acad. Sci. USA 2004, 101, 17705–17710. [Google Scholar] [CrossRef]
- Kano, T.; Wada, S.; Morimoto, K.; Kato, Y.; Ogihara, T. Effect of knockdown of ezrin, radixin, and moesin on P-glycoprotein function in HepG2 cells. J. Pharm. Sci. 2011, 100, 5308–5314. [Google Scholar] [CrossRef]
- Xiao, Y.; Wu, J.; Yuan, Y.; Guo, X.; Chen, B.; Huang, Q. Effect of moesin phosphorylation on high-dose sphingosine-1-phosphate-induced endothelial responses. Mol. Med. Rep. 2018, 17, 1933–1939. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Kong, J.; Lin, Z.; Yang, Y.; Jin, T.; Xu, M.; Sun, J.; Chen, L. Ezrin promotes breast cancer progression by modulating AKT signals. Br. J. Cancer 2019, 120, 703–713. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, K.; McNamara, K.M.; Miki, Y.; Iwabuchi, E.; Kanai, A.; Miyashita, M.; Ishida, T.; Sasano, H. S100P and Ezrin promote trans-endothelial migration of triple negative breast cancer cells. Cell. Oncol. 2019, 42, 67–80. [Google Scholar] [CrossRef]
- Kong, J.; Di, C.; Piao, J.; Sun, J.; Han, L.; Chen, L.; Yan, G.; Lin, Z. Ezrin contributes to cervical cancer progression through induction of epithelial-mesenchymal transition. Oncotarget 2016, 7, 19631–19642. [Google Scholar] [CrossRef] [PubMed]
- Hoskin, V.; Szeto, A.; Ghaffari, A.; Greer, P.A.; Cote, G.P.; Elliott, B.E. Ezrin regulates focal adhesion and invadopodia dynamics by altering calpain activity to promote breast cancer cell invasion. Mol. Biol. Cell 2015, 26, 3464–3479. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Ma, G.; Qian, J.; Zhu, Y.; Liang, M.; Yao, N.; Ding, Q.; Chen, L.; Liu, X.; Xia, T.; et al. Interaction Between Ezrin and Cortactin in Promoting Epithelial to Mesenchymal Transition in Breast Cancer Cells. Med. Sci. Monit. 2017, 23, 1583–1596. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Belkina, N.V.; Park, C.; Nambiar, R.; Loughhead, S.M.; Patino-Lopez, G.; Ben-Aissa, K.; Hao, J.J.; Kruhlak, M.J.; Qi, H.; et al. Constitutively active ezrin increases membrane tension, slows migration, and impedes endothelial transmigration of lymphocytes in vivo in mice. Blood 2012, 119, 445–453. [Google Scholar] [CrossRef]
- Antelmi, E.; Cardone, R.A.; Greco, M.R.; Rubino, R.; Di Sole, F.; Martino, N.A.; Casavola, V.; Carcangiu, M.; Moro, L.; Reshkin, S.J. ss1 integrin binding phosphorylates ezrin at T567 to activate a lipid raft signalsome driving invadopodia activity and invasion. PLoS ONE 2013, 8, e75113. [Google Scholar] [CrossRef]
- Zaidel-Bar, R.; Itzkovitz, S.; Ma’ayan, A.; Iyengar, R.; Geiger, B. Functional atlas of the integrin adhesome. Nat. Cell Biol. 2007, 9, 858–867. [Google Scholar] [CrossRef]
- Wehrle-Haller, B. Structure and function of focal adhesions. Curr. Opin. Cell Biol. 2012, 24, 116–124. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, D.; Guo, Z.; Zhao, J.; Wu, B.; Deng, H.; Zhou, T.; Xiang, H.; Gao, F.; Yu, X.; et al. Rho kinase phosphorylation promotes ezrin-mediated metastasis in hepatocellular carcinoma. Cancer Res. 2011, 71, 1721–1729. [Google Scholar] [CrossRef]
- Leiphrakpam, P.D.; Rajput, A.; Mathiesen, M.; Agarwal, E.; Lazenby, A.J.; Are, C.; Brattain, M.G.; Chowdhury, S. Ezrin expression and cell survival regulation in colorectal cancer. Cell. Signal. 2014, 26, 868–879. [Google Scholar] [CrossRef]
- Quan, C.; Sun, J.; Lin, Z.; Jin, T.; Dong, B.; Meng, Z.; Piao, J. Ezrin promotes pancreatic cancer cell proliferation and invasion through activating the Akt/mTOR pathway and inducing YAP translocation. Cancer Manag. Res. 2019, 11, 6553–6566. [Google Scholar] [CrossRef] [PubMed]
- Berryman, M.; Franck, Z.; Bretscher, A. Ezrin is concentrated in the apical microvilli of a wide variety of epithelial cells whereas moesin is found primarily in endothelial cells. J. Cell Sci. 1993, 105, 1025–1043. [Google Scholar] [CrossRef] [PubMed]
- Pidoux, G.; Gerbaud, P.; Dompierre, J.; Lygren, B.; Solstad, T.; Evain-Brion, D.; Tasken, K. A PKA-ezrin-Cx43 signaling complex controls gap junction communication and thereby trophoblast cell fusion. J. Cell Sci. 2014, 127, 4172–4185. [Google Scholar] [CrossRef] [PubMed]
- Bretscher, A. Purification of an 80,000-dalton protein that is a component of the isolated microvillus cytoskeleton, and its localization in nonmuscle cells. J. Cell Biol. 1983, 97, 425–432. [Google Scholar] [CrossRef]
- Crepaldi, T.; Gautreau, A.; Comoglio, P.M.; Louvard, D.; Arpin, M. Ezrin is an effector of hepatocyte growth factor-mediated migration and morphogenesis in epithelial cells. J. Cell Biol. 1997, 138, 423–434. [Google Scholar] [CrossRef]
- Al-Zuhair, A.G.; Ibrahim, M.E.; Mughal, S.; Abdulla, M.A. Loss and regeneration of the microvilli of human placental syncytiotrophoblast. Arch. Gynecol. 1987, 240, 147–151. [Google Scholar] [CrossRef]
- Zaman, R.; Lombardo, A.; Sauvanet, C.; Viswanatha, R.; Awad, V.; Bonomo, L.E.; McDermitt, D.; Bretscher, A. Effector-mediated ERM activation locally inhibits RhoA activity to shape the apical cell domain. J. Cell Biol. 2021, 220, e202007146. [Google Scholar] [CrossRef]
- Watanabe, K.; Tachibana, M.; Kim, S.; Watarai, M. Participation of ezrin in bacterial uptake by trophoblast giant cells. Reprod. Biol. Endocrinol. 2009, 7, 95. [Google Scholar] [CrossRef]
- Ng, T.; Parsons, M.; Hughes, W.E.; Monypenny, J.; Zicha, D.; Gautreau, A.; Arpin, M.; Gschmeissner, S.; Verveer, P.J.; Bastiaens, P.I.; et al. Ezrin is a downstream effector of trafficking PKC-integrin complexes involved in the control of cell motility. EMBO J. 2001, 20, 2723–2741. [Google Scholar] [CrossRef]
- Jokela, T.; Oikari, S.; Takabe, P.; Rilla, K.; Karna, R.; Tammi, M.; Tammi, R. Interleukin-1beta-induced Reduction of CD44 Ser-325 Phosphorylation in Human Epidermal Keratinocytes Promotes CD44 Homomeric Complexes, Binding to Ezrin, and Extended, Monocyte-adhesive Hyaluronan Coats. J. Biol. Chem. 2015, 290, 12379–12393. [Google Scholar] [CrossRef]
- Jeong, J.; Choi, J.; Kim, W.; Dann, P.; Takyar, F.; Gefter, J.V.; Friedman, P.A.; Wysolmerski, J.J. Inhibition of ezrin causes PKCalpha-mediated internalization of erbb2/HER2 tyrosine kinase in breast cancer cells. J. Biol. Chem. 2019, 294, 887–901. [Google Scholar] [CrossRef]
- Franck, Z.; Gary, R.; Bretscher, A. Moesin, like ezrin, colocalizes with actin in the cortical cytoskeleton in cultured cells, but its expression is more variable. J. Cell Sci. 1993, 105, 219–231. [Google Scholar] [CrossRef]
- Chang, W.L.; Liu, Y.W.; Dang, Y.L.; Jiang, X.X.; Xu, H.; Huang, X.; Wang, Y.L.; Wang, H.; Zhu, C.; Xue, L.Q.; et al. PLAC8, a new marker for human interstitial extravillous trophoblast cells, promotes their invasion and migration. Development 2018, 145, dev148932. [Google Scholar] [CrossRef]
- Garcia-Ortiz, A.; Serrador, J.M. ERM Proteins at the Crossroad of Leukocyte Polarization, Migration and Intercellular Adhesion. Int. J. Mol. Sci. 2020, 21, 1502. [Google Scholar] [CrossRef] [PubMed]
- Makitie, T.; Carpen, O.; Vaheri, A.; Kivela, T. Ezrin as a prognostic indicator and its relationship to tumor characteristics in uveal malignant melanoma. Investig. Ophthalmol. Vis. Sci. 2001, 42, 2442–2449. [Google Scholar]
- Weng, W.H.; Ahlen, J.; Astrom, K.; Lui, W.O.; Larsson, C. Prognostic impact of immunohistochemical expression of ezrin in highly malignant soft tissue sarcomas. Clin. Cancer Res. 2005, 11, 6198–6204. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.L.; Chen, D.F.; Lan, C.H.; Liu, K.Y.; Fang, D.C. Knockdown of ezrin via RNA interference suppresses Helicobacter pylori-enhanced invasion of gastric cancer cells. Cancer Biol. Ther. 2011, 11, 746–752. [Google Scholar] [CrossRef]
- Tang, Y.; Sun, X.; Yu, S.; Bie, X.; Wang, J.; Ren, L. Inhibition of Ezrin suppresses cell migration and invasion in human nasopharyngeal carcinoma. Oncol. Lett. 2019, 18, 553–560. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Li, G.; Du, M.; Bao, S. Effect of Ezrin on regulating trophoblast cell invasion via PKC signaling pathway in unexplained recurrent spontaneous abortion. Reprod. Biol. 2022, 22, 100634. [Google Scholar] [CrossRef] [PubMed]
- Bonilha, V.L.; Finnemann, S.C.; Rodriguez-Boulan, E. Ezrin promotes morphogenesis of apical microvilli and basal infoldings in retinal pigment epithelium. J. Cell Biol. 1999, 147, 1533–1548. [Google Scholar] [CrossRef] [PubMed]
- Dard, N.; Louvet-Vallee, S.; Santa-Maria, A.; Maro, B. Phosphorylation of ezrin on threonine T567 plays a crucial role during compaction in the mouse early embryo. Dev. Biol. 2004, 271, 87–97. [Google Scholar] [CrossRef]
- Ren, L.; Hong, S.H.; Cassavaugh, J.; Osborne, T.; Chou, A.J.; Kim, S.Y.; Gorlick, R.; Hewitt, S.M.; Khanna, C. The actin-cytoskeleton linker protein ezrin is regulated during osteosarcoma metastasis by PKC. Oncogene 2009, 28, 792–802. [Google Scholar] [CrossRef]
- Pelaseyed, T.; Viswanatha, R.; Sauvanet, C.; Filter, J.J.; Goldberg, M.L.; Bretscher, A. Ezrin activation by LOK phosphorylation involves a PIP2-dependent wedge mechanism. eLife 2017, 6, e22759. [Google Scholar] [CrossRef]
- Patel, A.; Dash, P.R. Formation of atypical podosomes in extravillous trophoblasts regulates extracellular matrix degradation. Eur. J. Cell Biol. 2012, 91, 171–179. [Google Scholar] [CrossRef]
- Viswanatha, R.; Wayt, J.; Ohouo, P.Y.; Smolka, M.B.; Bretscher, A. Interactome analysis reveals ezrin can adopt multiple conformational states. J. Biol. Chem. 2013, 288, 35437–35451. [Google Scholar] [CrossRef]
- Lipreri da Silva, J.C.; Coelho-Silva, J.L.; Lima, K.; Vicari, H.P.; Lazarini, M.; Costa-Lotufo, L.V.; Traina, F.; Machado-Neto, J.A. Comprehensive analysis of cytoskeleton regulatory genes identifies ezrin as a prognostic marker and molecular target in acute myeloid leukemia. Cell. Oncol. 2021, 44, 1105–1117. [Google Scholar] [CrossRef]
- Curto, M.; McClatchey, A.I. Ezrin… a metastatic detERMinant? Cancer Cell 2004, 5, 113–114. [Google Scholar] [CrossRef]
- Cortesio, C.L.; Chan, K.T.; Perrin, B.J.; Burton, N.O.; Zhang, S.; Zhang, Z.Y.; Huttenlocher, A. Calpain 2 and PTP1B function in a novel pathway with Src to regulate invadopodia dynamics and breast cancer cell invasion. J. Cell Biol. 2008, 180, 957–971. [Google Scholar] [CrossRef] [PubMed]
- Yao, X.; Thibodeau, A.; Forte, J.G. Ezrin-calpain I interactions in gastric parietal cells. Am. J. Physiol. 1993, 265, C36–C46. [Google Scholar] [CrossRef] [PubMed]
- Shuster, C.B.; Herman, I.M. Indirect association of ezrin with F-actin: Isoform specificity and calcium sensitivity. J. Cell Biol. 1995, 128, 837–848. [Google Scholar] [CrossRef] [PubMed]
- Dewitt, S.; Hallett, M. Leukocyte membrane “expansion”: A central mechanism for leukocyte extravasation. J. Leukoc. Biol. 2007, 81, 1160–1164. [Google Scholar] [CrossRef] [PubMed]
- Goto, S.; Ozaki, Y.; Ozawa, F.; Mizutani, E.; Kitaori, T.; Suzumori, N.; Blomgren, K.; Furuno, T.; Sugiura-Ogasawara, M. The investigation of calpain in human placenta with fetal growth restriction. Am. J. Reprod. Immunol. 2021, 85, e13325. [Google Scholar] [CrossRef] [PubMed]
- Nicola, C.; Timoshenko, A.V.; Dixon, S.J.; Lala, P.K.; Chakraborty, C. EP1 receptor-mediated migration of the first trimester human extravillous trophoblast: The role of intracellular calcium and calpain. J. Clin. Endocrinol. Metab. 2005, 90, 4736–4746. [Google Scholar] [CrossRef]
- Meng, Y.H.; Zhang, J.B.; Sun, Y.L.; Liu, X.L. ADATMS-7 regulates the focal adhesion kinase signaling and promotes invasiveness of trophoblasts in early pregnancy. Placenta 2020, 92, 54–61. [Google Scholar] [CrossRef]
- Mak, H.; Naba, A.; Varma, S.; Schick, C.; Day, A.; SenGupta, S.K.; Arpin, M.; Elliott, B.E. Ezrin phosphorylation on tyrosine 477 regulates invasion and metastasis of breast cancer cells. BMC Cancer 2012, 12, 82. [Google Scholar] [CrossRef]
- Naba, A.; Reverdy, C.; Louvard, D.; Arpin, M. Spatial recruitment and activation of the Fes kinase by ezrin promotes HGF-induced cell scattering. EMBO J. 2008, 27, 38–50. [Google Scholar] [CrossRef]
- Zhou, R.; Cao, X.; Watson, C.; Miao, Y.; Guo, Z.; Forte, J.G.; Yao, X. Characterization of protein kinase A-mediated phosphorylation of ezrin in gastric parietal cell activation. J. Biol. Chem. 2003, 278, 35651–35659. [Google Scholar] [CrossRef]
- Yang, H.S.; Hinds, P.W. Increased ezrin expression and activation by CDK5 coincident with acquisition of the senescent phenotype. Mol. Cell 2003, 11, 1163–1176. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, J.; Elliott, B.E.; Louvard, D.; Arpin, M. Src-dependent ezrin phosphorylation in adhesion-mediated signaling. Mol. Biol. Cell 2005, 16, 1481–1490. [Google Scholar] [CrossRef] [PubMed]
- Gautreau, A.; Poullet, P.; Louvard, D.; Arpin, M. Ezrin, a plasma membrane-microfilament linker, signals cell survival through the phosphatidylinositol 3-kinase/Akt pathway. Proc. Natl. Acad. Sci. USA 1999, 96, 7300–7305. [Google Scholar] [CrossRef] [PubMed]

| Percentage Ezrin Pixel: Total Pixels in Images ± SEM (n = 50) | Percentage Ezrin Pixel in STB: Total Pixels in Images ± SEM (n = 50) | Percentage Ezrin Pixel in CTB: Total Pixels in Images ± SEM (n = 50) | Percentage Ezrin Pixel in pcEVT: Total Pixels in Images ± SEM (n = 50) | |
|---|---|---|---|---|
| First Trimester | 34.69 ± 3.28 | 56.24 ± 7.24 | 81.68 ± 8.72 | 94.20 ± 5.87 |
| Second Trimester | 28.24 ± 2.37 | 27.24 ± 19.34 | 55.36 ± 13.30 | 75.49 ± 10.52 |
| p < 0.05 a | p > 0.05 a | p < 0.05 a | p > 0.05 a | |
| Full Term | 12.33 ± 3.41 | 3.76 ± 5.32 | 1.52 ± 2.15 | 29.24 ± 225.53 |
| p < 0.001 a | p < 0.01 a | p < 0.001 a | p < 0.01 a | |
| p < 0.01 b | p < 0.05 b | p < 0.001 b | p < 0.05 b |
| Cell Lines | Phospho-Ezrin Protrusions Per Cell ± SEM (n = 50) | p-Value |
|---|---|---|
| HTR8/SVneo control | 41.00 ± 1.26 | |
| HTR8/SVneo treated with NSC668394 | 16.41 ± 0.96 | p < 0.001 |
| Swan71 control | 40.81 ± 1.23 | |
| Swan71 treated with NSC668394 | 35.45 ± 0.91 | p < 0.05 |
| Cell Lines | Percentage Motility ± SEM (n = 50) | p-Value a | p-Value b |
|---|---|---|---|
| HTR8/SVneo control | 100.00 ± 3.46 | ||
| HTR8/SVneo treated with ezrin siRNA7 | 73.92 ± 8.59 | p < 0.01 | p < 0.001 |
| HTR8/SVneo treated with ezrin siRNA9 | 48.40 ± 7.01 | p < 0.001 | p < 0.05 |
| HTR8/SVneo treated with NSC668394 | 24.28 ± 3.91 | p < 0.001 | |
| Swan71 control | 100.98 ± 8.29 | ||
| Swan71 treated with ezrin siRNA7 | 52.17 ± 6.72 | p < 0.001 | p < 0.01 |
| Swan71 treated with ezrin siRNA9 | 39.57 ± 5.59 | p < 0.001 | p < 0.01 |
| Swan71 treated with NSC668394 | 79.88 ± 15.59 | p > 0.05 |
| Cell Lines | Percentage Focal Adhesions Per Cell ± SEM (n = 50) | p-Value a | p-Value b |
|---|---|---|---|
| HTR8/SVneo control | 100 ± 5.22 | ||
| HTR8/SVneo treated with ezrin siRNA7 | 170.32 ± 11.34 | p < 0.0001 | |
| HTR8/SVneo treated with ezrin siRNA9 | 192.48 ± 14.43 | p < 0.0001 | 0.0089 |
| Swan71 control | 100 ± 4.81 | ||
| Swan71 treated with ezrin siRNA7 | 145.88 ± 10.23 | 0.0078 | |
| Swan71 treated with ezrin siRNA9 | 172.78 ± 12.13 | p < 0.0001 | 0.1219 |
| Cell Lines | Percentage Focal Adhesions Per Cell ± SEM (n = 50) | p-Value |
|---|---|---|
| HTR8/SVneo control | 100 ± 5.47 | |
| HTR8/SVneo treated with NSC668394 | 162.11 ± 7.94 | p < 0.0001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tabrizi, M.E.A.; Gupta, J.K.; Gross, S.R. Ezrin and Its Phosphorylated Thr567 Form Are Key Regulators of Human Extravillous Trophoblast Motility and Invasion. Cells 2023, 12, 711. https://doi.org/10.3390/cells12050711
Tabrizi MEA, Gupta JK, Gross SR. Ezrin and Its Phosphorylated Thr567 Form Are Key Regulators of Human Extravillous Trophoblast Motility and Invasion. Cells. 2023; 12(5):711. https://doi.org/10.3390/cells12050711
Chicago/Turabian StyleTabrizi, Maral E. A., Janesh K. Gupta, and Stephane R. Gross. 2023. "Ezrin and Its Phosphorylated Thr567 Form Are Key Regulators of Human Extravillous Trophoblast Motility and Invasion" Cells 12, no. 5: 711. https://doi.org/10.3390/cells12050711
APA StyleTabrizi, M. E. A., Gupta, J. K., & Gross, S. R. (2023). Ezrin and Its Phosphorylated Thr567 Form Are Key Regulators of Human Extravillous Trophoblast Motility and Invasion. Cells, 12(5), 711. https://doi.org/10.3390/cells12050711






