Open-Label Sulforaphane Trial in FMR1 Premutation Carriers with Fragile-X-Associated Tremor and Ataxia Syndrome (FXTAS)
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Protocol
2.2. Biomarkers
2.3. Bioenergetics Assessment
2.4. Statistical Analysis
3. Results
3.1. Participants
3.2. Analysis of Outcome Measures
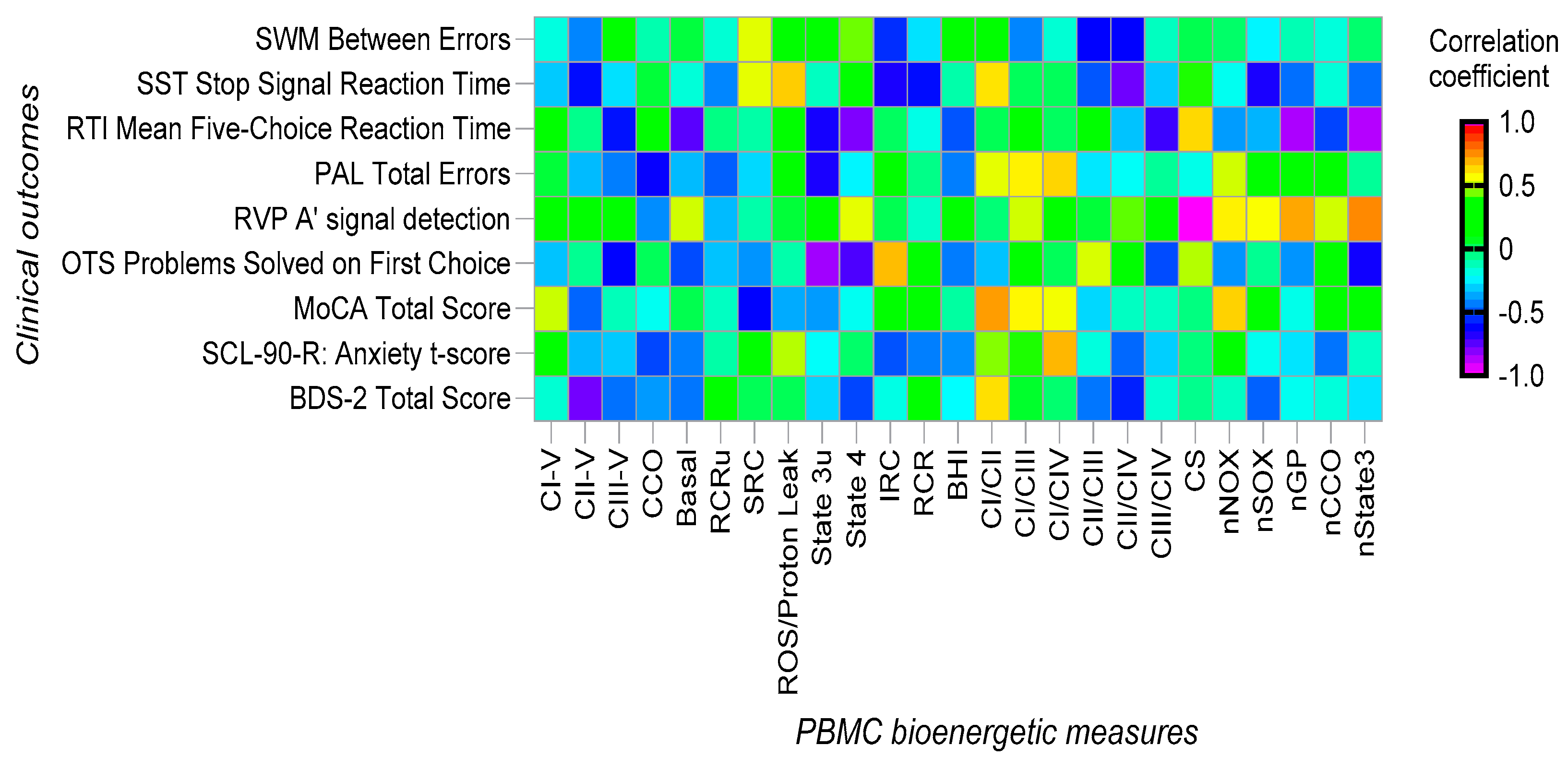
3.3. Bioenergetics Results in PBMC
4. Discussion
4.1. Limitations
4.2. Sulforaphane in Neurogenerative Disorders
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tassone, F.; Hagerman, R. The fragile X-associated tremor ataxia syndrome. In Modeling Fragile X Syndrome; Denman, R., Ed.; Results and Problems in Cell Differentiation; Springer: Berlin/Heidelberg, Germany; Volume 54, pp. 337–357.
- Saldarriaga, W.; Forero-Forero, J.V.; González-Teshima, L.Y.; Fandiño-Losada, A.; Isaza, C.; Tovar-Cuevas, J.R.; Silva, M.; Choudhary, N.S.; Tang, H.-T.; Aguilar-Gaxiola, S.; et al. Genetic cluster of fragile X syndrome in a Colombian district. J. Hum. Genet. 2018, 63, 509–516. [Google Scholar] [CrossRef]
- Hagerman, R.J.; Leehey, M.; Heinrichs, W.; Tassone, F.; Wilson, R.; Hills, J.; Grigsby, J.; Gage, B.; Hagerman, P.J. Intention tremor, parkinsonism, and generalized brain atrophy in male carriers of fragile X. Neurology 2001, 57, 127–130. [Google Scholar] [CrossRef] [PubMed]
- Jacquemont, S.; Hagerman, R.J.; Leehey, M.; Grigsby, J.; Zhang, L.; Brunberg, J.A.; Greco, C.; Portes, V.D.; Jardini, T.; Levine, R.; et al. Fragile X Premutation Tremor/Ataxia Syndrome: Molecular, Clinical, and Neuroimaging Correlates. Am. J. Hum. Genet. 2003, 72, 869–878. [Google Scholar] [CrossRef]
- Hagerman, R.J.; Hagerman, P. Fragile X-associated tremor/ataxia syndrome—Features, mechanisms and management. Nat. Rev. Neurol. 2016, 12, 403–412. [Google Scholar] [CrossRef]
- Jacquemont, S.; Hagerman, R.J.; Leehey, M.A.; Hall, D.A.; Levine, R.A.; Brunberg, J.A.; Zhang, L.; Jardini, T.; Gane, L.W.; Harris, S.W.; et al. Penetrance of the Fragile X–Associated Tremor/Ataxia Syndrome in a Premutation Carrier Population. JAMA 2004, 291, 460–469. [Google Scholar] [CrossRef] [PubMed]
- Hagerman, R.; Hagerman, P. Advances in clinical and molecular understanding of the FMR1 premutation and fragile X-associated tremor/ataxia syndrome. Lancet Neurol. 2013, 12, 786–798. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Revenga, L.; Madrigal, I.; Pagonabarraga, J.; Xuncla, M.; Badenas, C.; Kulisevsky, J.; Gomez, B.; Mila, M. Penetrance of FMR1 premutation associated pathologies in fragile X syndrome families. Eur. J. Hum. Genet. 2009, 17, 1359–1362. [Google Scholar] [CrossRef]
- Leehey, M.A.; Berry-Kravis, E.; Min, S.; Hall, D.A.; Rice, C.D.; Zhang, L.; Grigsby, J.; Greco, C.M.; Reynolds, A.; Lara, R. Progression of tremor and ataxia in male carriers of the FMR1 premutation. Mov. Disord. Off. J. Mov. Disord. Soc. 2007, 22, 203–206. [Google Scholar] [CrossRef] [PubMed]
- Skomorowsky, A. The Carriers. Sci. Am. Mind 2016, 27, 34–41. [Google Scholar] [CrossRef]
- Tassone, F.; Hall, D.A. FXTAS, FXPOI, and Other Premutation Disorders; Springer: Cham, Switzerland, 2016; ISBN 3-319-33898-6. [Google Scholar]
- Ross-Inta, C.; Omanska-Klusek, A.; Wong, S.; Barrow, C.; Garcia-Arocena, D.; Iwahashi, C.; Berry-Kravis, E.; Hagerman, R.J.; Hagerman, P.J.; Giulivi, C. Evidence of mitochondrial dysfunction in fragile X-associated tremor/ataxia syndrome. Biochem. J. 2010, 429, 545–552. [Google Scholar] [CrossRef]
- Song, G.; Napoli, E.; Wong, S.; Hagerman, R.; Liu, S.; Tassone, F.; Giulivi, C. Altered redox mitochondrial biology in the neurodegenerative disorder fragile X-tremor/ataxia syndrome: Use of antioxidants in precision medicine. Mol. Med. 2016, 22, 548–559. [Google Scholar] [CrossRef]
- Alvarez-Mora, M.; Rodriguez-Revenga, L.; Madrigal, I.; Guitart-Mampel, M.; Garrabou, G.; Mila, M. Impaired mitochondrial function and dynamics in the pathogenesis of FXTAS. Mol. Neurobiol. 2017, 54, 6896–6902. [Google Scholar] [CrossRef]
- Norat, P.; Soldozy, S.; Sokolowski, J.D.; Gorick, C.M.; Kumar, J.S.; Chae, Y.; Yağmurlu, K.; Prada, F.; Walker, M.; Levitt, M.R.; et al. Mitochondrial dysfunction in neurological disorders: Exploring mitochondrial transplantation. npj Regen. Med. 2020, 5, 22. [Google Scholar] [CrossRef]
- Jardim, F.R.; de Almeida, F.J.S.; Luckachaki, M.D.; de Oliveira, M.R. Effects of sulforaphane on brain mitochondria: Mechanistic view and future directions. J. Zhejiang Univ.–Sci. B 2020, 21, 263–279. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Zhao, Q.; Zhang, Y.; Shi, W.; Wang, H.; Zheng, Z.; Meng, L.; Xin, Y.; Jiang, X. Sulforaphane-mediated Nrf2 activation prevents radiation-induced skin injury through inhibiting the oxidative-stress-activated DNA damage and NLRP3 inflammasome. Antioxidants 2021, 10, 1850. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.; Nioi, P.; Pickett, C.B. The Nrf2-antioxidant response element signaling pathway and its activation by oxidative stress. J. Biol. Chem. 2009, 284, 13291–13295. [Google Scholar] [CrossRef] [PubMed]
- Hayes, J.D.; Dinkova-Kostova, A.T. The Nrf2 regulatory network provides an interface between redox and intermediary metabolism. Trends Biochem. Sci. 2014, 39, 199–218. [Google Scholar] [CrossRef]
- O’Mealey, G.B.; Berry, W.L.; Plafker, S.M. Sulforaphane is a Nrf2-independent inhibitor of mitochondrial fission. Redox Biol. 2017, 11, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Napoli, E.; Flores, A.; Mansuri, Y.; Hagerman, R.J.; Giulivi, C. Sulforaphane improves mitochondrial metabolism in fibroblasts from patients with fragile X-associated tremor and ataxia syndrome. Neurobiol. Dis. 2021, 157, 105427. [Google Scholar] [CrossRef] [PubMed]
- Hall, D.A.; Birch, R.C.; Anheim, M.; Jønch, A.E.; Pintado, E.; O’Keefe, J.; Trollor, J.N.; Stebbins, G.T.; Hagerman, R.J.; Fahn, S. Emerging topics in FXTAS. J. Neurodev. Disord. 2014, 6, 31. [Google Scholar] [CrossRef] [PubMed]
- Bacalman, S.; Farzin, F.; Bourgeois, J.; Cogswell, J.; Goodlin-Jones, B.; Gane, L.; Grigsby, J.; Leehey, M.; Tassone, F.; Hagerman, R. Psychiatric Phenotype of the Fragile X-Associated Tremor/Ataxia Syndrome (FXTAS) in Males: Newly Described Fronto-Subcortical Dementia. J. Clin. Psychiatry 2006, 67, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Wild, K.; Howieson, D.; Webbe, F.; Seelye, A.; Kaye, J. Status of computerized cognitive testing in aging: A systematic review. Alzheimer’s Dement. 2008, 4, 428–437. [Google Scholar] [CrossRef] [PubMed]
- Saunders, N.L.; Summers, M.J. Attention and working memory deficits in mild cognitive impairment. J. Clin. Exp. Neuropsychol. 2010, 32, 350–357. [Google Scholar] [CrossRef] [PubMed]
- Derogatis, L.R.; Kathryn, L. The SCL-90-R and Brief Symptom Inventory (BSI) in primary care. In Handbook of Psychological Assessment in Primary Care Settings; Routledge: London, UK, 2000; pp. 310–347. ISBN 1-315-82734-4. [Google Scholar]
- Grigsby, J.; Kaye, K.; Robbins, L.J. Reliabilities, norms and factor structure of the Behavioral Dyscontrol Scale. Percept. Mot. Ski. 1992, 74, 883–892. [Google Scholar] [CrossRef] [PubMed]
- Shura, R.D.; Rowland, J.A.; Yoash-Gantz, R.E. The Behavioral Dyscontrol Scale-II with non-elderly veterans. Arch. Clin. Neuropsychol. 2014, 29, 409–414. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nasreddine, Z.S.; Phillips, N.A.; Bédirian, V.; Charbonneau, S.; Whitehead, V.; Collin, I.; Cummings, J.L.; Chertkow, H. The Montreal Cognitive Assessment, MoCA: A brief screening tool for mild cognitive impairment. J. Am. Geriatr. Soc. 2005, 53, 695–699. [Google Scholar] [CrossRef] [PubMed]
- Tassone, F.; Pan, R.; Amiri, K.; Taylor, A.K.; Hagerman, P.J. A rapid polymerase chain reaction-based screening method for identification of all expanded alleles of the fragile X (FMR1) gene in newborn and high-risk populations. J. Mol. Diagn. 2008, 10, 43–49. [Google Scholar] [CrossRef]
- Filipovic-Sadic, S.; Sah, S.; Chen, L.; Krosting, J.; Sekinger, E.; Zhang, W.; Hagerman, P.J.; Stenzel, T.T.; Hadd, A.G.; Latham, G.J. A novel FMR1 PCR method for the routine detection of low abundance expanded alleles and full mutations in fragile X syndrome. Clin. Chem. 2010, 56, 399–408. [Google Scholar] [CrossRef]
- Tassone, F.; Hagerman, R.J.; Iklé, D.N.; Dyer, P.N.; Lampe, M.; Willemsen, R.; Oostra, B.A.; Taylor, A.K. FMRP expression as a potential prognostic indicator in fragile X syndrome. Am. J. Med. Genet. 1999, 84, 250–261. [Google Scholar] [CrossRef]
- Tassone, F.; Hagerman, R.J.; Taylor, A.K.; Gane, L.W.; Godfrey, T.E.; Hagerman, P.J. Elevated levels of FMR1 mRNA in carrier males: A new mechanism of involvement in the fragile-X syndrome. Am. J. Hum. Genet. 2000, 66, 6–15. [Google Scholar] [CrossRef]
- Kim, K.; Hessl, D.; Randol, J.L.; Espinal, G.M.; Schneider, A.; Protic, D.; Aydin, E.Y.; Hagerman, R.J.; Hagerman, P.J. Association between IQ and FMR1 protein (FMRP) across the spectrum of CGG repeat expansions. PLoS ONE 2019, 14, e0226811. [Google Scholar] [CrossRef] [PubMed]
- Blommer, J.; Pitcher, T.; Mustapic, M.; Eren, E.; Yao, P.J.; Vreones, M.P.; Pucha, K.A.; Dalrymple-Alford, J.; Shoorangiz, R.; Meissner, W.G. Extracellular vesicle biomarkers for cognitive impairment in Parkinson’s disease. Brain 2023, 146, 195–208. [Google Scholar] [CrossRef] [PubMed]
- Mustapic, M.; Eitan, E.; Werner, J.K., Jr.; Berkowitz, S.T.; Lazaropoulos, M.P.; Tran, J.; Goetzl, E.J.; Kapogiannis, D. Plasma extracellular vesicles enriched for neuronal origin: A potential window into brain pathologic processes. Front. Neurosci. 2017, 11, 278. [Google Scholar] [CrossRef] [PubMed]
- Napoli, E.; Song, G.; Schneider, A.; Hagerman, R.; Eldeeb, M.A.A.A.; Azarang, A.; Tassone, F.; Giulivi, C. Warburg effect linked to cognitive-executive deficits in FMR1 premutation. FASEB J. 2016, 30, 3334. [Google Scholar] [CrossRef]
- Wang, J.Y.; Napoli, E.; Kim, K.; McLennan, Y.A.; Hagerman, R.J.; Giulivi, C. Brain atrophy and white matter damage linked to peripheral bioenergetic deficits in the neurodegenerative disease FXTAS. Int. J. Mol. Sci. 2021, 22, 9171. [Google Scholar] [CrossRef]
- Giulivi, C.; Zhang, Y.-F.; Omanska-Klusek, A.; Ross-Inta, C.; Wong, S.; Hertz-Picciotto, I.; Tassone, F.; Pessah, I.N. Mitochondrial dysfunction in autism. JAMA 2010, 304, 2389–2396. [Google Scholar] [CrossRef]
- Napoli, E.; Tassone, F.; Wong, S.; Angkustsiri, K.; Simon, T.J.; Song, G.; Giulivi, C. Mitochondrial citrate transporter-dependent metabolic signature in the 22q11.2 deletion syndrome. J. Biol. Chem. 2015, 290, 23240–23253. [Google Scholar] [CrossRef]
- Napoli, E.; Wong, S.; Hung, C.; Ross-Inta, C.; Bomdica, P.; Giulivi, C. Defective mitochondrial disulfide relay system, altered mitochondrial morphology and function in Huntington’s disease. Hum. Mol. Genet. 2013, 22, 989–1004. [Google Scholar] [CrossRef]
- Napoli, E.; Wong, S.; Giulivi, C. Evidence of reactive oxygen species-mediated damage to mitochondrial DNA in children with typical autism. Mol. Autism 2013, 4, 2. [Google Scholar] [CrossRef]
- Napoli, E.; Hung, C.; Wong, S.; Giulivi, C. Toxicity of the flame-retardant BDE-49 on brain mitochondria and neuronal progenitor striatal cells enhanced by a PTEN-deficient background. Toxicol. Sci. 2013, 132, 196–210. [Google Scholar] [CrossRef]
- Giulivi, C.; Napoli, E.; Schwartzer, J.; Careaga, M.; Ashwood, P. Gestational exposure to a viral mimetic poly (i: C) results in long-lasting changes in mitochondrial function by leucocytes in the adult offspring. Mediat. Inflamm. 2013, 2013, 609602. [Google Scholar] [CrossRef] [PubMed]
- Napoli, E.; Ross-Inta, C.; Wong, S.; Hung, C.; Fujisawa, Y.; Sakaguchi, D.; Angelastro, J.; Omanska-Klusek, A.; Schoenfeld, R.; Giulivi, C. Mitochondrial dysfunction in Pten haplo-insufficient mice with social deficits and repetitive behavior: Interplay between Pten and p 53. PLoS ONE 2012, 7, e42504. [Google Scholar] [CrossRef] [PubMed]
- Napoli, E.; Ross-Inta, C.; Wong, S.; Omanska-Klusek, A.; Barrow, C.; Iwahashi, C.; Garcia-Arocena, D.; Sakaguchi, D.; Berry-Kravis, E.; Hagerman, R. Altered zinc transport disrupts mitochondrial protein processing/import in fragile X-associated tremor/ataxia syndrome. Hum. Mol. Genet. 2011, 20, 3079–3092. [Google Scholar] [CrossRef] [PubMed]
- Napoli, E.; Song, G.; Wong, S.; Hagerman, R.; Giulivi, C. Altered bioenergetics in primary dermal fibroblasts from adult carriers of the FMR1 premutation before the onset of the neurodegenerative disease fragile X-associated tremor/ataxia syndrome. Cerebellum 2016, 15, 552–564. [Google Scholar] [CrossRef]
- Nolin, S.L.; Napoli, E.; Flores, A.; Hagerman, R.J.; Giulivi, C. Deficits in prenatal serine biosynthesis underlie the mitochondrial dysfunction associated with the autism-linked FMR1 gene. Int. J. Mol. Sci. 2021, 22, 5886. [Google Scholar] [CrossRef] [PubMed]
- Hong, F.; Freeman, M.L.; Liebler, D.C. Identification of sensor cysteines in human Keap1 modified by the cancer chemopreventive agent sulforaphane. Chem. Res. Toxicol. 2005, 18, 1917–1926. [Google Scholar] [CrossRef]
- Ma, Q. Role of nrf2 in oxidative stress and toxicity. Annu. Rev. Pharmacol. Toxicol. 2013, 53, 401–426. [Google Scholar] [CrossRef]
- Kubo, E.; Chhunchha, B.; Singh, P.; Sasaki, H.; Singh, D.P. Sulforaphane reactivates cellular antioxidant defense by inducing Nrf2/ARE/Prdx6 activity during aging and oxidative stress. Sci. Rep. 2017, 7, 14130. [Google Scholar] [CrossRef]
- Zimmerman, A.W.; Singh, K.; Connors, S.L.; Liu, H.; Panjwani, A.A.; Lee, L.-C.; Diggins, E.; Foley, A.; Melnyk, S.; Singh, I.N. Randomized controlled trial of sulforaphane and metabolite discovery in children with Autism Spectrum Disorder. Mol. Autism 2021, 12, 38. [Google Scholar] [CrossRef]
- Gwon, Y.; Oh, J.; Kim, J.-S. Sulforaphane induces colorectal cancer cell proliferation through Nrf2 activation in a p53-dependent manner. Appl. Biol. Chem. 2020, 63, 86. [Google Scholar] [CrossRef]
- Uddin, M.S.; Al Mamun, A.; Jakaria, M.; Thangapandiyan, S.; Ahmad, J.; Rahman, M.A.; Mathew, B.; Abdel-Daim, M.M.; Aleya, L. Emerging promise of sulforaphane-mediated Nrf2 signaling cascade against neurological disorders. Sci. Total Environ. 2020, 707, 135624. [Google Scholar] [CrossRef] [PubMed]
- Mangla, B.; Javed, S.; Sultan, M.H.; Kumar, P.; Kohli, K.; Najmi, A.; Alhazmi, H.A.; Al Bratty, M.; Ahsan, W. Sulforaphane: A review of its therapeutic potentials, advances in its nanodelivery, recent patents, and clinical trials. Phytother. Res. 2021, 35, 5440–5458. [Google Scholar] [CrossRef] [PubMed]
- Miyakawa-Liu, M.; Feehan, A.K.; Pai, S.; Garcia-Diaz, J. Rates of Cognitive Decline in 100 Patients with Alzheimer Disease. Ochsner J. 2022, 22, 129–133. [Google Scholar] [CrossRef]
- Salvadori, E.; Poggesi, A.; Pracucci, G.; Chiti, A.; Ciolli, L.; Del Bene, A.; Di Donato, I.; Marini, S.; Nannucci, S.; Orlandi, G. Longitudinal changes in MoCA performances in patients with mild cognitive impairment and small vessel disease. Results from the VMCI-Tuscany Study. Cereb. Circ.–Cogn. Behav. 2021, 2, 100008. [Google Scholar] [CrossRef]
- Garber, K.; Smith, K.T.; Reines, D.; Warren, S.T. Transcription, translation and fragile X syndrome. Curr. Opin. Genet. Dev. 2006, 16, 270–275. [Google Scholar] [CrossRef] [PubMed]
- Villavicencio Tejo, F.; Quintanilla, R.A. Contribution of the Nrf2 pathway on oxidative damage and mitochondrial failure in Parkinson and Alzheimer’s disease. Antioxidants 2021, 10, 1069. [Google Scholar] [CrossRef] [PubMed]
- Lasbleiz, C.; Peyrel, A.; Tarot, P.; Sarniguet, J.; Crouzier, L.; Cubedo, N.; Delprat, B.; Rossel, M.; Maurice, T.; Liévens, J.-C. Sigma-1 receptor agonist PRE-084 confers protection against TAR DNA-binding protein-43 toxicity through NRF2 signalling. Redox Biol. 2022, 58, 102542. [Google Scholar] [CrossRef]
- Shimizu, S.; Kasai, S.; Yamazaki, H.; Tatara, Y.; Mimura, J.; Engler, M.J.; Tanji, K.; Nikaido, Y.; Inoue, T.; Suganuma, H. Sulforaphane increase mitochondrial biogenesis-related gene expression in the hippocampus and suppresses age-related cognitive decline in mice. Int. J. Mol. Sci. 2022, 23, 8433. [Google Scholar] [CrossRef]
- Bose, C.; Alves, I.; Singh, P.; Palade, P.T.; Carvalho, E.; Børsheim, E.; Jun, S.-R.; Cheema, A.; Boerma, M.; Awasthi, S. Sulforaphane prevents age-associated cardiac and muscular dysfunction through Nrf2 signaling. Aging Cell 2020, 19, e13261. [Google Scholar] [CrossRef]
- Bent, S.; Lawton, B.; Warren, T.; Widjaja, F.; Dang, K.; Fahey, J.W.; Cornblatt, B.; Kinchen, J.M.; Delucchi, K.; Hendren, R.L. Identification of urinary metabolites that correlate with clinical improvements in children with autism treated with sulforaphane from broccoli. Mol. Autism 2018, 9, 35. [Google Scholar] [CrossRef]
- Shekarri, Q.; Dekker, M. A physiological-based model for simulating the bioavailability and kinetics of sulforaphane from broccoli products. Foods 2021, 10, 2761. [Google Scholar] [CrossRef] [PubMed]
- Schepici, G.; Bramanti, P.; Mazzon, E. Efficacy of sulforaphane in neurodegenerative diseases. Int. J. Mol. Sci. 2020, 21, 8637. [Google Scholar] [CrossRef] [PubMed]
- Lynch, D.R.; Chin, M.P.; Delatycki, M.B.; Subramony, S.H.; Corti, M.; Hoyle, J.C.; Boesch, S.; Nachbauer, W.; Mariotti, C.; Mathews, K.D. Safety and efficacy of omaveloxolone in Friedreich ataxia (MOXIe study). Ann. Neurol. 2021, 89, 212–225. [Google Scholar] [CrossRef] [PubMed]


| Variable | N | Mean (SD) or Frequency (%) | Median (Q1, Q3) | Range |
|---|---|---|---|---|
| Age (years) | 11 | 73.47 (8.95) | 74.37 (67.33, 78.2) | 60.73–88.66 |
| Gender | 11 | |||
| Male | 6 (54.5%) | |||
| Female | 5 (45.5%) | |||
| FXTAS diagnosis | 11 | |||
| Definite | 6 (54.5%) | |||
| Probable | 3 (27.3%) | |||
| Possible | 2 (18.2%) | |||
| FXTAS stage | 11 | 3.05 (0.79) | 3 (2.5, 3.75) | 2–4 |
| 2 | 3 (27.3%) | |||
| 3 | 4 (36.4%) | |||
| 3.5 | 1 (9.1%) | |||
| 4 | 3 (27.3%) | |||
| Age of onset: tremor | 10 | 59.25 (7.31) | 58.5 (53.38, 64.5) | 49–73 |
| Age of onset: ataxia | 8 | 66.38 (5.93) | 66 (62.75, 70) | 57–75 |
| AGG interruptions | 11 | 1.09 (0.54) | 1 (1, 1) | 0–2 |
| 0 | 1 (9.1%) | |||
| 1 | 8 (72.7%) | |||
| 2 | 2 (18.2%) | |||
| CGG repeats | 11 | 85.27 (13.16) | 87 (72, 92.5) | 70–110 |
| Activation ratio (in females) | 5 | 0.54 (0.14) | 0.49 (0.46, 0.53) | 0.44–0.78 |
| FMR1 mRNA level | 11 | 2.50 (0.34) | 2.57 (2.19, 2.77) | 1.99–2.94 |
| Variable | N | Pre-Sulforaphane | Post-Sulforaphane | Change from Baseline (Pre-Post) | Cohen’s d Effect Size | p-Value * | FDR # |
|---|---|---|---|---|---|---|---|
| Mean (SD) | Mean (SD) | Mean (SD) | |||||
| CANTAB | |||||||
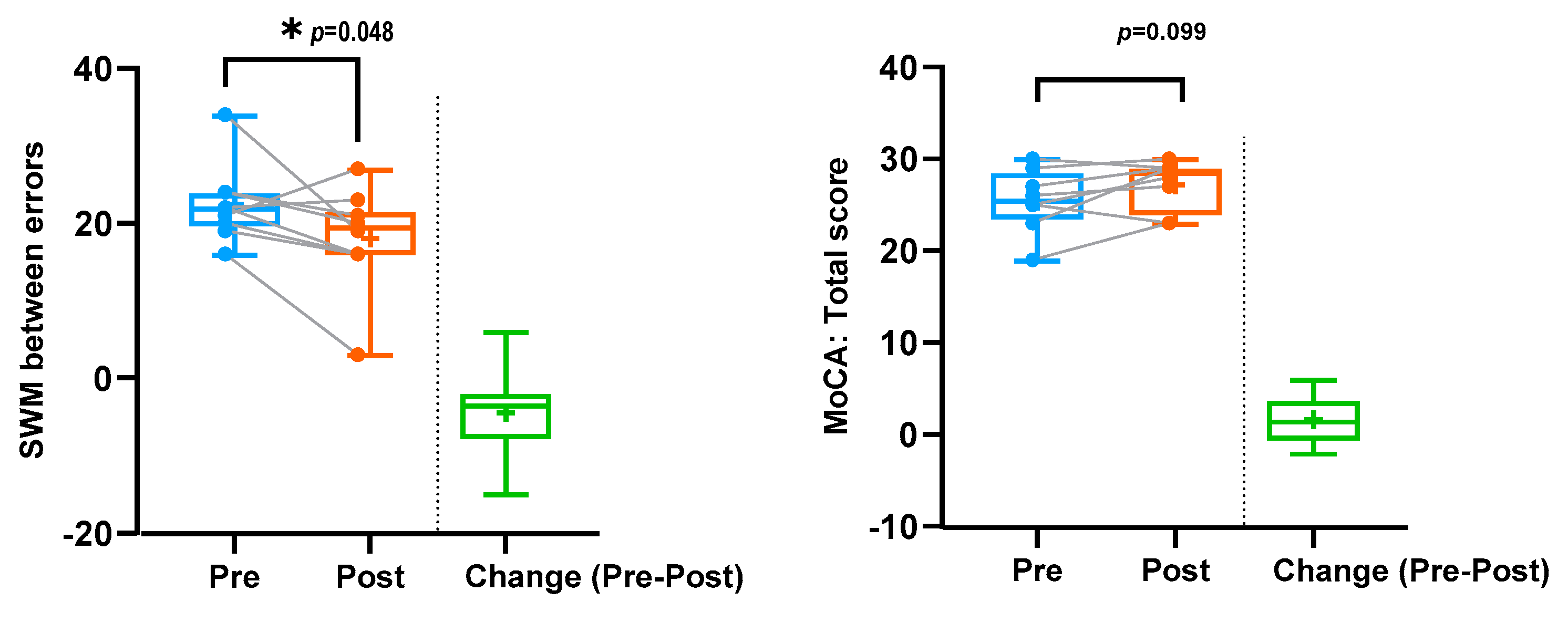
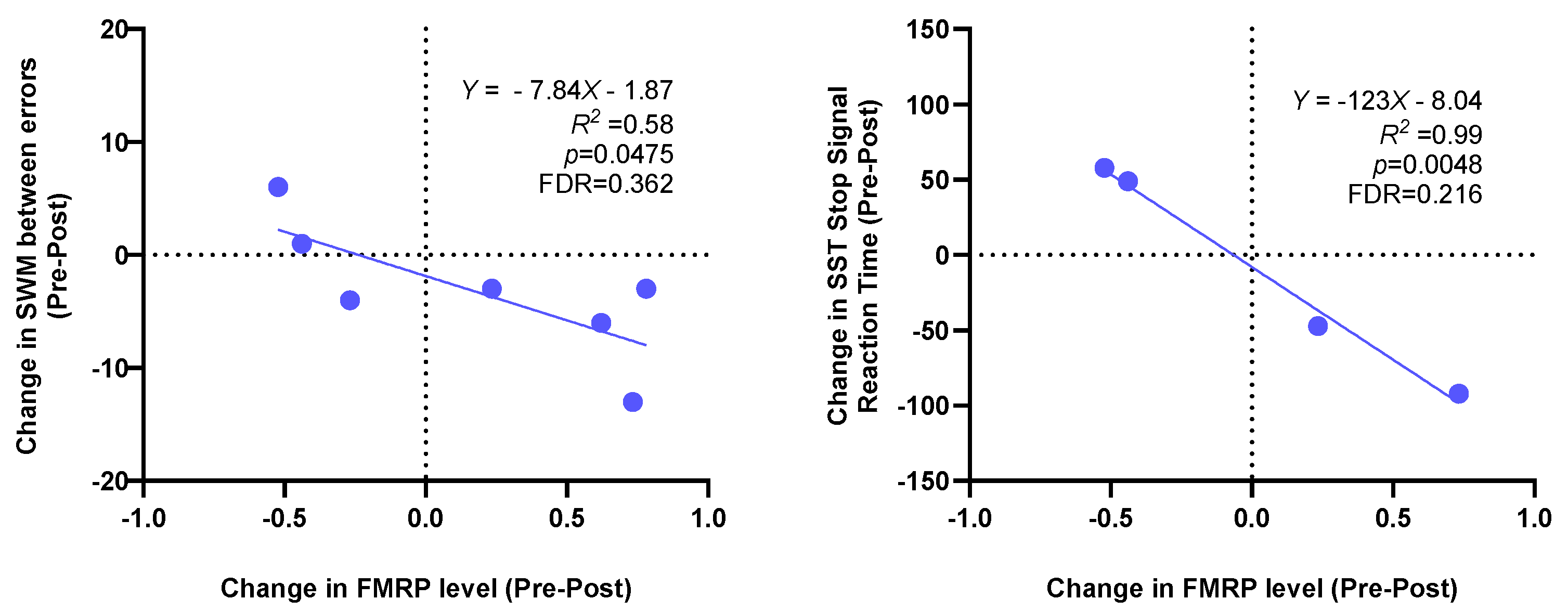
| SWM between errors | 10 | 22.6 (4.74) | 18.2 (6.37) | −4.4 (6.08) | −0.72 | 0.048 1 | 0.43 |
| SST Stop Signal Reaction Time | 7 | 303.7 (43.75) | 283.58 (59.67) | −20.11 (58.21) | −0.35 | 0.396 1 | 0.713 |
| RTI Mean Five-Choice Reaction Time | 10 | 417.22 (68.97) | 436.07 (89.78) | 18.85 (73.97) | 0.25 | 1 2 | 1 |
| PAL Total Errors | 8 | 16.88 (6.71) | 17.12 (7.75) | 0.25 (9.05) | 0.03 | 0.94 1 | 1 |
| RVP A’ signal detection | 8 | 0.87 (0.04) | 0.89 (0.03) | 0.02 (0.03) | 0.67 | 0.159 1 | 0.476 |
| OTS Problems Solved on First Choice | 7 | 8.71 (3.2) | 7.43 (3.95) | −1.29 (2.98) | −0.43 | 0.2981 | 0.67 |
| MoCA total score | 8 | 25.5 (3.46) | 27.25 (2.76) | 1.75 (2.6) | 0.67 | 0.099 1 | 0.446 |
| SCL-90-R: Anxiety t-score | 9 | 57.78 (9) | 57.44 (8.22) | −0.33 (4.53) | −0.07 | 0.831 1 | 1 |
| BDS-II total score | 11 | 23.36 (2.38) | 23.27 (2) | −0.09 (3.14) | −0.03 | 0.926 1 | 1 |
| Variable | N | Pre-Sulforaphane | Post-Sulforaphane | Change from Baseline (Pre-Post) | Cohen’s d Effect Size | p-Value * | FDR # |
|---|---|---|---|---|---|---|---|
| Mean (SD) | Mean (SD) | Mean (SD) | |||||
| Complex IV quantity | 11 | 29.2 (17.46) | 39.77 (35.14) | 10.57 (31.78) | 0.33 | 0.52 2 | 0.866 |
| Complex IV specific activity | 11 | 0.45 (0.2) | 0.44 (0.32) | −0.01 (0.34) | −0.03 | 0.918 1 | 0.918 |
| ATP synthase quantity | 11 | 35.81 (11.98) | 41.8 (17.35) | 5.99 (15.05) | 0.40 | 0.216 1 | 0.866 |
| ATP synthase specific activity | 11 | 0.00225 (0.00128) | 0.00262 (0.00246) | 0.00037 (0.00251) | 0.15 | 0.765 2 | 0.918 |
| FMRP level | 7 | 1.03 (0.445) | 1.193 (0.715) | 0.162 (0.569) | 0.28 | 0.478 1 | 0.866 |
| Variable | N | Pre-Sulforaphane | Post-Sulforaphane | Change from Baseline (Pre-Post) | log2 (Fold Change) | p-Value * | FDR |
|---|---|---|---|---|---|---|---|
| Mean (SD) | Mean (SD) | Mean (SD) | |||||
| CI-V | 11 | 0.358 (0.197) | 0.453 (0.206) | 0.095 (0.248) | 0.34 | 0.232 1 | 0.572 |
| CII-V | 11 | 0.455 (0.275) | 0.507 (0.351) | 0.052 (0.403) | 0.16 | 0.678 1 | 0.771 |
| CIII-V | 11 | 0.42 (0.393) | 0.388 (0.325) | −0.032 (0.379) | −0.11 | 0.966 2 | 0.966 |
| CCO | 11 | 0.998 (1.301) | 0.887 (0.481) | −0.11 (1.015) | −0.17 | 0.32 2 | 0.572 |
| Basal | 11 | 0.686 (0.52) | 0.558 (0.362) | −0.128 (0.397) | −0.30 | 0.31 1 | 0.572 |
| RCRu | 11 | 1.651 (0.813) | 1.35 (0.733) | −0.301 (1.054) | −0.29 | 0.366 1 | 0.61 |
| SRC | 11 | 1.908 (1.257) | 1.828 (1.335) | −0.08 (1.493) | −0.06 | 0.413 2 | 0.645 |
| ROS/proton leak | 11 | 1.327 (1.054) | 1.281 (0.391) | −0.046 (0.789) | −0.05 | 0.465 2 | 0.684 |
| State 3u | 11 | 1.04 (0.571) | 0.834 (0.542) | −0.206 (0.653) | −0.32 | 0.319 1 | 0.572 |
| State 4 | 11 | 0.715 (0.413) | 0.676 (0.437) | −0.039 (0.47) | −0.08 | 0.79 1 | 0.823 |
| IRC | 11 | 0.651 (0.234) | 0.761 (0.35) | 0.11 (0.303) | 0.23 | 0.255 1 | 0.572 |
| RCR | 11 | 1.046 (0.617) | 0.86 (0.238) | −0.186 (0.583) | −0.28 | 0.52 2 | 0.684 |
| BHI | 11 | 1.067 (0.75) | 0.671 (0.46) | −0.396 (0.661) | −0.67 | 0.24 2 | 0.572 |
| CI/CII | 11 | 1.054 (0.82) | 1.466 (1.426) | 0.412 (1.695) | 0.48 | 0.52 2 | 0.684 |
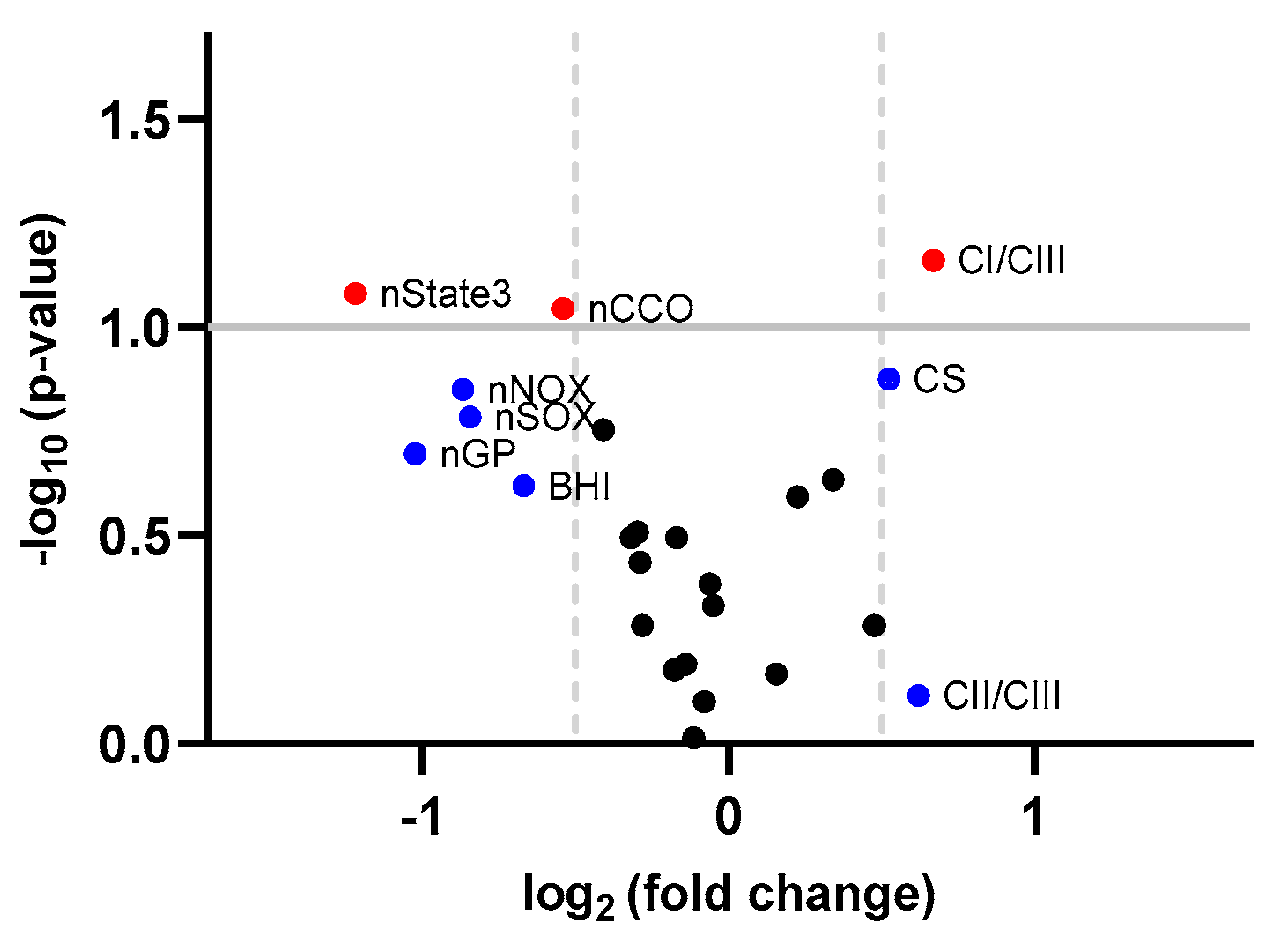
| CI/CIII | 11 | 1.435 (1.261) | 2.28 (1.789) | 0.846 (1.377) | 0.67 | 0.069 1 | 0.572 |
| CI/CIV | 11 | 0.818 (0.881) | 0.742 (0.78) | −0.076 (0.528) | −0.14 | 0.643 1 | 0.771 |
| CII/CIII | 11 | 1.431 (0.667) | 2.199 (2.233) | 0.768 (2.412) | 0.62 | 0.765 2 | 0.823 |
| CII/CIV | 11 | 0.772 (0.727) | 0.682 (0.391) | −0.09 (0.672) | −0.18 | 0.666 1 | 0.771 |
| CIII/CIV | 11 | 0.692 (0.624) | 0.521 (0.415) | −0.171 (0.391) | −0.41 | 0.176 1 | 0.572 |
| CS | 11 | 2.003 (1.458) | 2.879 (1.115) | 0.876 (1.777) | 0.52 | 0.133 1 | 0.572 |
| nNOX | 11 | 0.336 (0.346) | 0.184 (0.109) | −0.152 (0.315) | −0.87 | 0.141 1 | 0.572 |
| nSOX | 11 | 0.352 (0.325) | 0.196 (0.134) | −0.156 (0.344) | −0.84 | 0.164 1 | 0.572 |
| nGP | 11 | 0.356 (0.4) | 0.175 (0.242) | −0.181 (0.437) | −1.02 | 0.201 1 | 0.572 |
| nCCO | 11 | 0.56 (0.397) | 0.385 (0.326) | −0.175 (0.308) | −0.54 | 0.09 1 | 0.572 |
| nState3 | 11 | 0.582 (0.677) | 0.25 (0.298) | −0.332 (0.64) | −1.22 | 0.083 1 | 0.572 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santos, E.; Clark, C.; Biag, H.M.B.; Tang, S.J.; Kim, K.; Ponzini, M.D.; Schneider, A.; Giulivi, C.; Montanaro, F.A.M.; Gipe, J.T.-E.; et al. Open-Label Sulforaphane Trial in FMR1 Premutation Carriers with Fragile-X-Associated Tremor and Ataxia Syndrome (FXTAS). Cells 2023, 12, 2773. https://doi.org/10.3390/cells12242773
Santos E, Clark C, Biag HMB, Tang SJ, Kim K, Ponzini MD, Schneider A, Giulivi C, Montanaro FAM, Gipe JT-E, et al. Open-Label Sulforaphane Trial in FMR1 Premutation Carriers with Fragile-X-Associated Tremor and Ataxia Syndrome (FXTAS). Cells. 2023; 12(24):2773. https://doi.org/10.3390/cells12242773
Chicago/Turabian StyleSantos, Ellery, Courtney Clark, Hazel Maridith B. Biag, Si Jie Tang, Kyoungmi Kim, Matthew D. Ponzini, Andrea Schneider, Cecilia Giulivi, Federica Alice Maria Montanaro, Jesse Tran-Emilia Gipe, and et al. 2023. "Open-Label Sulforaphane Trial in FMR1 Premutation Carriers with Fragile-X-Associated Tremor and Ataxia Syndrome (FXTAS)" Cells 12, no. 24: 2773. https://doi.org/10.3390/cells12242773
APA StyleSantos, E., Clark, C., Biag, H. M. B., Tang, S. J., Kim, K., Ponzini, M. D., Schneider, A., Giulivi, C., Montanaro, F. A. M., Gipe, J. T.-E., Dayton, J., Randol, J. L., Yao, P. J., Manolopoulos, A., Kapogiannis, D., Hwang, Y. H., Hagerman, P., Hagerman, R., & Tassone, F. (2023). Open-Label Sulforaphane Trial in FMR1 Premutation Carriers with Fragile-X-Associated Tremor and Ataxia Syndrome (FXTAS). Cells, 12(24), 2773. https://doi.org/10.3390/cells12242773







