Biomarkers of Neurological Damage: From Acute Stage to Post-Acute Sequelae of COVID-19
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Data and Sample Collection
2.3. Microfluidic Next Generation Enzyme-Linked Immunosorbent Assay (ELISA)
2.4. Statistical Analysis
3. Results
3.1. Study Population
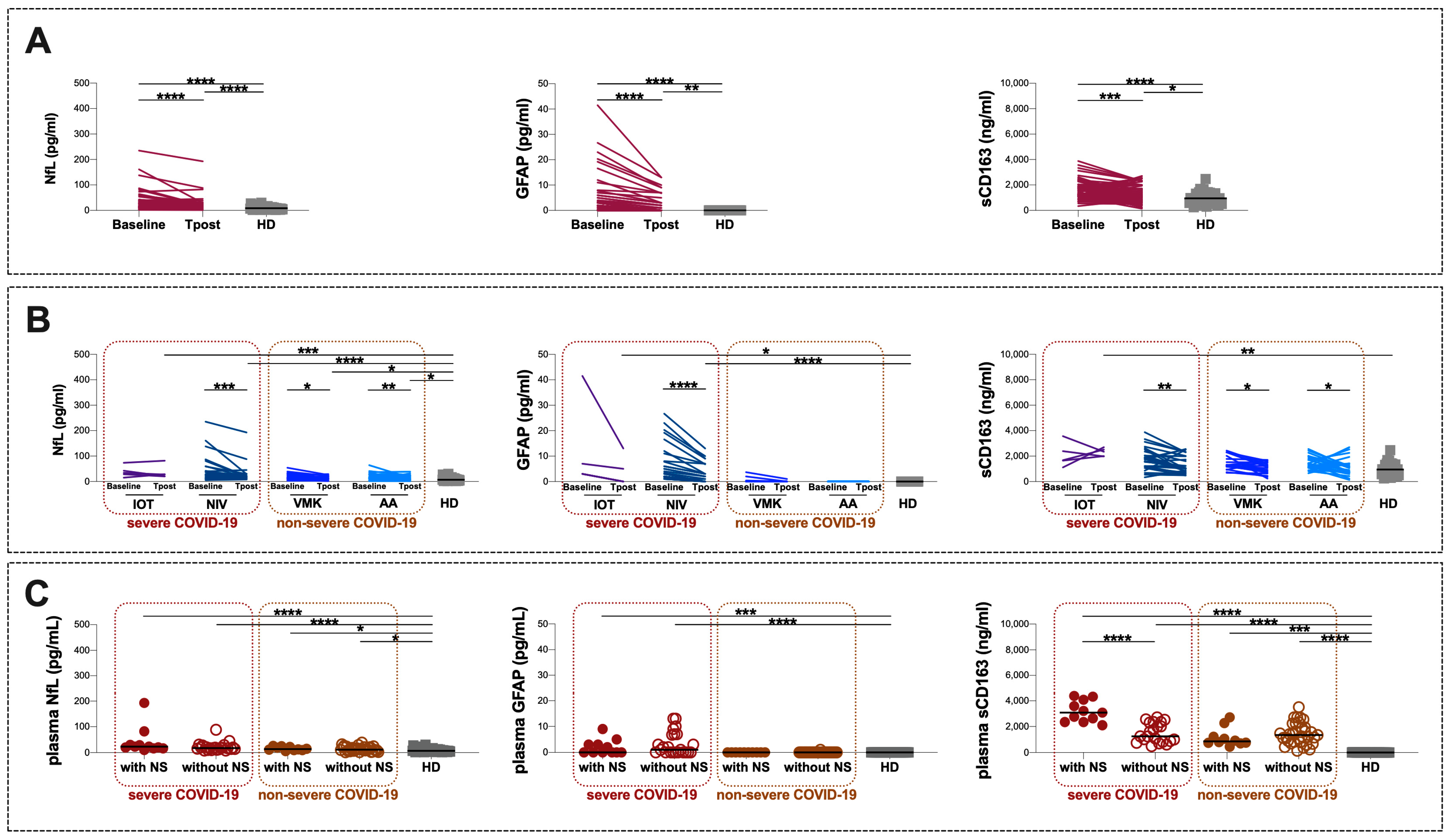
3.2. Plasma Biomarkers of Brain Injury on Hospital Admission
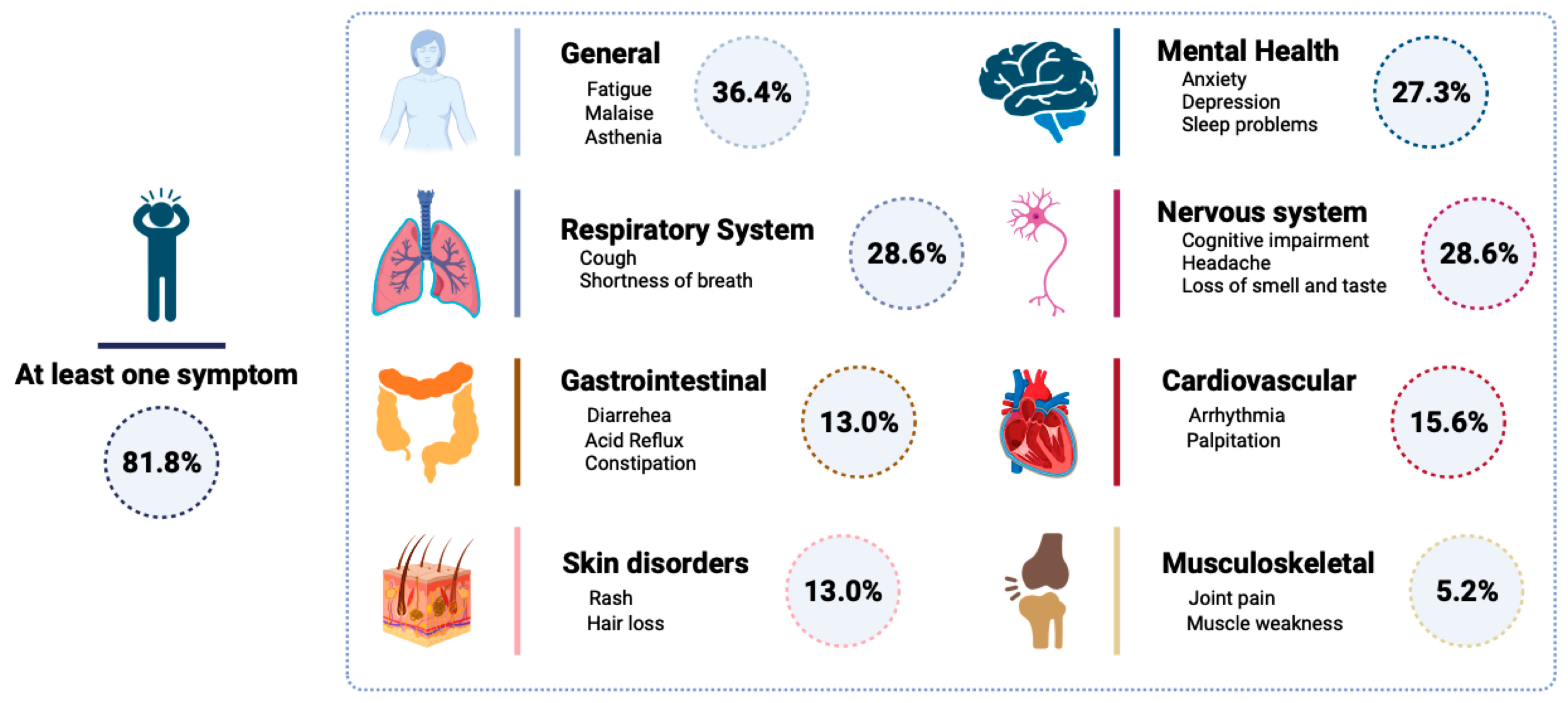
3.3. Post-COVID Symptoms
3.4. Plasma Biomarkers of Brain Injury in Long COVID
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wiersinga, W.J.; Rhodes, A.; Cheng, A.C.; Peacock, S.J.; Prescott, H.C. Pathophysiology, Transmission, Diagnosis, and Treatment of Coronavirus Disease 2019 (COVID-19): A Review. JAMA 2020, 324, 782–793. [Google Scholar] [CrossRef]
- Zingaropoli, M.A.; Iannetta, M.; Piermatteo, L.; Pasculli, P.; Latronico, T.; Mazzuti, L.; Campogiani, L.; Duca, L.; Ferraguti, G.; De Michele, M.; et al. Neuro-Axonal Damage and Alteration of Blood-Brain Barrier Integrity in COVID-19 Patients. Cells 2022, 11, 2480. [Google Scholar] [CrossRef]
- Nordvig, A.S.; Fong, K.T.; Willey, J.Z.; Thakur, K.T.; Boehme, A.K.; Vargas, W.S.; Smith, C.J.; Elkind, M.S.V. Potential Neurologic Manifestations of COVID-19. Neurol. Clin. Pract. 2021, 11, e135–e146. [Google Scholar] [CrossRef]
- Solomon, T. Neurological Infection with SARS-CoV-2—The Story so Far. Nat. Rev. Neurol. 2021, 17, 65–66. [Google Scholar] [CrossRef]
- Savarraj, J.; Park, E.S.; Colpo, G.D.; Hinds, S.N.; Morales, D.; Ahnstedt, H.; Paz, A.S.; Assing, A.; Liu, F.; Juneja, S.; et al. Brain Injury, Endothelial Injury and Inflammatory Markers Are Elevated and Express Sex-Specific Alterations after COVID-19. J. Neuroinflamm. 2021, 18, 277. [Google Scholar] [CrossRef] [PubMed]
- Moriguchi, T.; Harii, N.; Goto, J.; Harada, D.; Sugawara, H.; Takamino, J.; Ueno, M.; Sakata, H.; Kondo, K.; Myose, N.; et al. A First Case of Meningitis/Encephalitis Associated with SARS-Coronavirus-2. Int. J. Infect. Dis. 2020, 94, 55–58. [Google Scholar] [CrossRef] [PubMed]
- Helms, J.; Kremer, S.; Merdji, H.; Clere-Jehl, R.; Schenck, M.; Kummerlen, C.; Collange, O.; Boulay, C.; Fafi-Kremer, S.; Ohana, M.; et al. Neurologic Features in Severe SARS-CoV-2 Infection. N. Engl. J. Med. 2020, 382, 2268–2270. [Google Scholar] [CrossRef] [PubMed]
- Mao, L.; Jin, H.; Wang, M.; Hu, Y.; Chen, S.; He, Q.; Chang, J.; Hong, C.; Zhou, Y.; Wang, D.; et al. Neurologic Manifestations of Hospitalized Patients with Coronavirus Disease 2019 in Wuhan, China. JAMA Neurol. 2020, 77, 683–690. [Google Scholar] [CrossRef]
- Ellul, M.A.; Benjamin, L.; Singh, B.; Lant, S.; Michael, B.D.; Easton, A.; Kneen, R.; Defres, S.; Sejvar, J.; Solomon, T. Neurological Associations of COVID-19. Lancet Neurol. 2020, 19, 767–783. [Google Scholar] [CrossRef]
- Premraj, L.; Kannapadi, N.V.; Briggs, J.; Seal, S.M.; Battaglini, D.; Fanning, J.; Suen, J.; Robba, C.; Fraser, J.; Cho, S.-M. Mid and Long-Term Neurological and Neuropsychiatric Manifestations of Post-COVID-19 Syndrome: A Meta-Analysis. J. Neurol. Sci. 2022, 434, 120162. [Google Scholar] [CrossRef]
- Liotta, E.M.; Batra, A.; Clark, J.R.; Shlobin, N.A.; Hoffman, S.C.; Orban, Z.S.; Koralnik, I.J. Frequent Neurologic Manifestations and Encephalopathy-Associated Morbidity in COVID-19 Patients. Ann. Clin. Transl. Neurol. 2020, 7, 2221–2230. [Google Scholar] [CrossRef] [PubMed]
- Newcombe, V.F.J.; Dangayach, N.S.; Sonneville, R. Neurological Complications of COVID-19. Intensive Care Med. 2021, 47, 1021–1023. [Google Scholar] [CrossRef]
- Yassin, A.; Nawaiseh, M.; Shaban, A.; Alsherbini, K.; El-Salem, K.; Soudah, O.; Abu-Rub, M. Neurological Manifestations and Complications of Coronavirus Disease 2019 (COVID-19): A Systematic Review and Meta-Analysis. BMC Neurol. 2021, 21, 138. [Google Scholar] [CrossRef] [PubMed]
- Thakur, K.T.; Miller, E.H.; Glendinning, M.D.; Al-Dalahmah, O.; Banu, M.A.; Boehme, A.K.; Boubour, A.L.; Bruce, S.S.; Chong, A.M.; Claassen, J.; et al. COVID-19 Neuropathology at Columbia University Irving Medical Center/New York Presbyterian Hospital. Brain 2021, 144, 2696–2708. [Google Scholar] [CrossRef]
- Garcia, M.A.; Barreras, P.V.; Lewis, A.; Pinilla, G.; Sokoll, L.J.; Kickler, T.; Mostafa, H.; Caturegli, M.; Moghekar, A.; Fitzgerald, K.C.; et al. Cerebrospinal Fluid in COVID-19 Neurological Complications: Neuroaxonal Damage, Anti-SARS-Cov-2 Antibodies but No Evidence of Cytokine Storm. J. Neurol. Sci. 2021, 427, 117517. [Google Scholar] [CrossRef]
- Virhammar, J.; Nääs, A.; Fällmar, D.; Cunningham, J.L.; Klang, A.; Ashton, N.J.; Jackmann, S.; Westman, G.; Frithiof, R.; Blennow, K.; et al. Biomarkers for Central Nervous System Injury in Cerebrospinal Fluid Are Elevated in COVID-19 and Associated with Neurological Symptoms and Disease Severity. Eur. J. Neurol. 2021, 28, 3324–3331. [Google Scholar] [CrossRef]
- Bozzetti, S.; Ferrari, S.; Zanzoni, S.; Alberti, D.; Braggio, M.; Carta, S.; Piraino, F.; Gabbiani, D.; Girelli, D.; Nocini, R.; et al. Neurological Symptoms and Axonal Damage in COVID-19 Survivors: Are There Sequelae? Immunol. Res. 2021, 69, 553–557. [Google Scholar] [CrossRef]
- Kanberg, N.; Ashton, N.J.; Andersson, L.-M.; Yilmaz, A.; Lindh, M.; Nilsson, S.; Price, R.W.; Blennow, K.; Zetterberg, H.; Gisslén, M. Neurochemical Evidence of Astrocytic and Neuronal Injury Commonly Found in COVID-19. Neurology 2020, 95, e1754–e1759. [Google Scholar] [CrossRef]
- Aamodt, A.H.; Høgestøl, E.A.; Popperud, T.H.; Holter, J.C.; Dyrhol-Riise, A.M.; Tonby, K.; Stiksrud, B.; Quist-Paulsen, E.; Berge, T.; Barratt-Due, A.; et al. Blood Neurofilament Light Concentration at Admittance: A Potential Prognostic Marker in COVID-19. J. Neurol. 2021, 268, 3574–3583. [Google Scholar] [CrossRef]
- Ameres, M.; Brandstetter, S.; Toncheva, A.A.; Kabesch, M.; Leppert, D.; Kuhle, J.; Wellmann, S. Association of Neuronal Injury Blood Marker Neurofilament Light Chain with Mild-to-Moderate COVID-19. J. Neurol. 2020, 267, 3476–3478. [Google Scholar] [CrossRef]
- Zetterberg, H.; Blennow, K. Fluid Biomarkers for Mild Traumatic Brain Injury and Related Conditions. Nat. Rev. Neurol. 2016, 12, 563–574. [Google Scholar] [CrossRef] [PubMed]
- Giovannoni, G. Peripheral Blood Neurofilament Light Chain Levels: The Neurologist’s C-Reactive Protein? Brain 2018, 141, 2235–2237. [Google Scholar] [CrossRef] [PubMed]
- Aamodt, W.W.; Waligorska, T.; Shen, J.; Tropea, T.F.; Siderowf, A.; Weintraub, D.; Grossman, M.; Irwin, D.; Wolk, D.A.; Xie, S.X.; et al. Neurofilament Light Chain as a Biomarker for Cognitive Decline in Parkinson Disease. Mov. Disord. 2021, 36, 2945–2950. [Google Scholar] [CrossRef] [PubMed]
- Cooper, M.A.; Fehniger, T.A.; Turner, S.C.; Chen, K.S.; Ghaheri, B.A.; Ghayur, T.; Carson, W.E.; Caligiuri, M.A. Human Natural Killer Cells: A Unique Innate Immunoregulatory Role for the CD56(Bright) Subset. Blood 2001, 97, 3146–3151. [Google Scholar] [CrossRef]
- De Lorenzo, R.; Loré, N.I.; Finardi, A.; Mandelli, A.; Cirillo, D.M.; Tresoldi, C.; Benedetti, F.; Ciceri, F.; Rovere-Querini, P.; Comi, G.; et al. Blood Neurofilament Light Chain and Total Tau Levels at Admission Predict Death in COVID-19 Patients. J. Neurol. 2021, 268, 4436–4442. [Google Scholar] [CrossRef]
- Chmielewska, N.; Szyndler, J.; Makowska, K.; Wojtyna, D.; Maciejak, P.; Płaźnik, A. Looking for Novel, Brain-Derived, Peripheral Biomarkers of Neurological Disorders. Neurol. Neurochir. Pol. 2018, 52, 318–325. [Google Scholar] [CrossRef] [PubMed]
- Brunkhorst, R.; Pfeilschifter, W.; Foerch, C. Astroglial Proteins as Diagnostic Markers of Acute Intracerebral Hemorrhage-Pathophysiological Background and Clinical Findings. Transl. Stroke Res. 2010, 1, 246–251. [Google Scholar] [CrossRef]
- Liddelow, S.A.; Barres, B.A. Reactive Astrocytes: Production, Function, and Therapeutic Potential. Immunity 2017, 46, 957–967. [Google Scholar] [CrossRef]
- Rosenberg, G.A. Matrix Metalloproteinases in Neuroinflammation. Glia 2002, 39, 279–291. [Google Scholar] [CrossRef]
- Rempe, R.G.; Hartz, A.M.S.; Bauer, B. Matrix Metalloproteinases in the Brain and Blood-Brain Barrier: Versatile Breakers and Makers. J. Cereb. Blood Flow. Metab. 2016, 36, 1481–1507. [Google Scholar] [CrossRef]
- Ji, Y.; Gao, Q.; Ma, Y.; Wang, F.; Tan, X.; Song, D.; Hoo, R.L.C.; Wang, Z.; Ge, X.; Han, H.; et al. An MMP-9 Exclusive Neutralizing Antibody Attenuates Blood-Brain Barrier Breakdown in Mice with Stroke and Reduces Stroke Patient-Derived MMP-9 Activity. Pharmacol. Res. 2023, 190, 106720. [Google Scholar] [CrossRef] [PubMed]
- Mohammadhosayni, M.; Sadat Mohammadi, F.; Ezzatifar, F.; Mahdavi Gorabi, A.; Khosrojerdi, A.; Aslani, S.; Hemmatzadeh, M.; Yazdani, S.; Arabi, M.; Marofi, F.; et al. Matrix Metalloproteinases Are Involved in the Development of Neurological Complications in Patients with Coronavirus Disease 2019. Int. Immunopharmacol. 2021, 100, 108076. [Google Scholar] [CrossRef] [PubMed]
- Polivka, J.; Polivka, J.; Krakorova, K.; Peterka, M.; Topolcan, O. Current Status of Biomarker Research in Neurology. EPMA J. 2016, 7, 14. [Google Scholar] [CrossRef][Green Version]
- Stilund, M.; Reuschlein, A.-K.; Christensen, T.; Møller, H.J.; Rasmussen, P.V.; Petersen, T. Soluble CD163 as a Marker of Macrophage Activity in Newly Diagnosed Patients with Multiple Sclerosis. PLoS ONE 2014, 9, e98588. [Google Scholar] [CrossRef]
- Nissen, S.K.; Ferreira, S.A.; Nielsen, M.C.; Schulte, C.; Shrivastava, K.; Hennig, D.; Etzerodt, A.; Graversen, J.H.; Berg, D.; Maetzler, W.; et al. Soluble CD163 Changes Indicate Monocyte Association With Cognitive Deficits in Parkinson’s Disease. Mov. Disord. 2021, 36, 963–976. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Zhang, X.; Ma, J.; Liu, Z.; Qi, Y.; Fang, L.; Zheng, Y.; Cai, Z. Circulating Soluble CD163: A Potential Predictor for the Functional Outcome of Acute Ischemic Stroke. Front. Neurol. 2021, 12, 740420. [Google Scholar] [CrossRef] [PubMed]
- Rezaee, M.; Foroutan, F.; Etemad, L.; Razban, V.; Tajbakhsh, A.; Savardashtaki, A. Clinical Utility of Soluble CD163 and Its Diagnostic and Prognostic Value in a Variety of Neurological Disorders. Mini Rev. Med. Chem. 2023, 23, 1592–1607. [Google Scholar] [CrossRef]
- Fabriek, B.O.; Van Haastert, E.S.; Galea, I.; Polfliet, M.M.J.; Döpp, E.D.; Van Den Heuvel, M.M.; Van Den Berg, T.K.; De Groot, C.J.A.; Van Der Valk, P.; Dijkstra, C.D. CD163-Positive Perivascular Macrophages in the Human CNS Express Molecules for Antigen Recognition and Presentation. Glia 2005, 51, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Galea, J.; Cruickshank, G.; Teeling, J.L.; Boche, D.; Garland, P.; Perry, V.H.; Galea, I. The Intrathecal CD163-Haptoglobin-Hemoglobin Scavenging System in Subarachnoid Hemorrhage. J. Neurochem. 2012, 121, 785–792. [Google Scholar] [CrossRef]
- Weaver, L.K.; Hintz-Goldstein, K.A.; Pioli, P.A.; Wardwell, K.; Qureshi, N.; Vogel, S.N.; Guyre, P.M. Pivotal Advance: Activation of Cell Surface Toll-like Receptors Causes Shedding of the Hemoglobin Scavenger Receptor CD163. J. Leukoc. Biol. 2006, 80, 26–35. [Google Scholar] [CrossRef]
- Etzerodt, A.; Maniecki, M.B.; Møller, K.; Møller, H.J.; Moestrup, S.K. Tumor Necrosis Factor α-Converting Enzyme (TACE/ADAM17) Mediates Ectodomain Shedding of the Scavenger Receptor CD163. J. Leukoc. Biol. 2010, 88, 1201–1205. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Rial, J.; Currás-Tuala, M.J.; Rivero-Calle, I.; Gómez-Carballa, A.; Cebey-López, M.; Rodríguez-Tenreiro, C.; Dacosta-Urbieta, A.; Rivero-Velasco, C.; Rodríguez-Núñez, N.; Trastoy-Pena, R.; et al. Increased Serum Levels of SCD14 and SCD163 Indicate a Preponderant Role for Monocytes in COVID-19 Immunopathology. Front. Immunol. 2020, 11, 560381. [Google Scholar] [CrossRef]
- Zingaropoli, M.A.; Nijhawan, P.; Carraro, A.; Pasculli, P.; Zuccalà, P.; Perri, V.; Marocco, R.; Kertusha, B.; Siccardi, G.; Del Borgo, C.; et al. Increased SCD163 and SCD14 Plasmatic Levels and Depletion of Peripheral Blood Pro-Inflammatory Monocytes, Myeloid and Plasmacytoid Dendritic Cells in Patients With Severe COVID-19 Pneumonia. Front. Immunol. 2021, 12, 627548. [Google Scholar] [CrossRef] [PubMed]
- Marocco, R.; Carraro, A.; Zingaropoli, M.A.; Nijhawan, P.; Tortellini, E.; Guardiani, M.; Mengoni, F.; Zuccalà, P.; Belvisi, V.; Kertusha, B.; et al. Role of Tocilizumab in Down Regulating SCD163 Plasmatic Levels in a Cohort of COVID-19 Patients. Front. Immunol. 2022, 13, 871592. [Google Scholar] [CrossRef]
- Pasculli, P.; Zingaropoli, M.A.; Masci, G.M.; Mazzuti, L.; Perri, V.; Paribeni, F.; Russo, G.; Arcari, G.; Iafrate, F.; Vullo, F.; et al. Chest Computed Tomography Score, Cycle Threshold Values and Secondary Infection in Predicting COVID-19 Mortality. New Microbiol. 2021, 44, 145–154. [Google Scholar]
- Zingaropoli, M.A.; Latronico, T.; Pasculli, P.; Masci, G.M.; Merz, R.; Ciccone, F.; Dominelli, F.; Del Borgo, C.; Lichtner, M.; Iafrate, F.; et al. Tissue Inhibitor of Matrix Metalloproteinases-1 (TIMP-1) and Pulmonary Involvement in COVID-19 Pneumonia. Biomolecules 2023, 13, 1040. [Google Scholar] [CrossRef] [PubMed]
- Petrella, C.; Zingaropoli, M.A.; Ceci, F.M.; Pasculli, P.; Latronico, T.; Liuzzi, G.M.; Ciardi, M.R.; Angeloni, A.; Ettorre, E.; Menghi, M.; et al. COVID-19 Affects Serum Brain-Derived Neurotrophic Factor and Neurofilament Light Chain in Aged Men: Implications for Morbidity and Mortality. Cells 2023, 12, 655. [Google Scholar] [CrossRef]
- Chung, H.-Y.; Neu, C.; Wickel, J.; Kuckertz, S.L.; Coldewey, S.M. Neurofilament Light Chain in Patients with COVID-19 and Bacterial Pneumonia. Ann. Neurol. 2021, 90, 174–175. [Google Scholar] [CrossRef]
- Geis, T.; Brandstetter, S.; Toncheva, A.A.; Laub, O.; Leipold, G.; Wagner, R.; Kabesch, M.; Kasser, S.; Kuhle, J.; Wellmann, S.; et al. Serum Neurofilament Light Chain (SNfL) Values in a Large Cross-Sectional Population of Children with Asymptomatic to Moderate COVID-19. J. Neurol. 2021, 268, 3969–3974. [Google Scholar] [CrossRef] [PubMed]
- Kanberg, N.; Simrén, J.; Edén, A.; Andersson, L.-M.; Nilsson, S.; Ashton, N.J.; Sundvall, P.-D.; Nellgård, B.; Blennow, K.; Zetterberg, H.; et al. Neurochemical Signs of Astrocytic and Neuronal Injury in Acute COVID-19 Normalizes during Long-Term Follow-Up. eBioMedicine 2021, 70, 103512. [Google Scholar] [CrossRef]
- Sutter, R.; Hert, L.; De Marchis, G.M.; Twerenbold, R.; Kappos, L.; Naegelin, Y.; Kuster, G.M.; Benkert, P.; Jost, J.; Maceski, A.M.; et al. Serum Neurofilament Light Chain Levels in the Intensive Care Unit: Comparison between Severely Ill Patients with and without Coronavirus Disease 2019. Ann. Neurol. 2021, 89, 610–616. [Google Scholar] [CrossRef]
- Leng, A.; Shah, M.; Ahmad, S.A.; Premraj, L.; Wildi, K.; Li Bassi, G.; Pardo, C.A.; Choi, A.; Cho, S.-M. Pathogenesis Underlying Neurological Manifestations of Long COVID Syndrome and Potential Therapeutics. Cells 2023, 12, 816. [Google Scholar] [CrossRef] [PubMed]
- Hiraoka, A.; Horiike, N.; Akbar, S.M.F.; Michitaka, K.; Matsuyama, T.; Onji, M. Soluble CD163 in Patients with Liver Diseases: Very High Levels of Soluble CD163 in Patients with Fulminant Hepatic Failure. J. Gastroenterol. 2005, 40, 52–56. [Google Scholar] [CrossRef]
- Møller, H.J.; de Fost, M.; Aerts, H.; Hollak, C.; Moestrup, S.K. Plasma Level of the Macrophage-Derived Soluble CD163 Is Increased and Positively Correlates with Severity in Gaucher’s Disease. Eur. J. Haematol. 2004, 72, 135–139. [Google Scholar] [CrossRef]
- Rajamanickam, A.; Kumar, N.P.; Pandiarajan, A.N.; Selvaraj, N.; Munisankar, S.; Renji, R.M.; Venkatramani, V.; Murhekar, M.; Thangaraj, J.W.V.; Kumar, M.S.; et al. Dynamic Alterations in Monocyte Numbers, Subset Frequencies and Activation Markers in Acute and Convalescent COVID-19 Individuals. Sci. Rep. 2021, 11, 20254. [Google Scholar] [CrossRef]
- Etzerodt, A.; Rasmussen, M.R.; Svendsen, P.; Chalaris, A.; Schwarz, J.; Galea, I.; Møller, H.J.; Moestrup, S.K. Structural Basis for Inflammation-Driven Shedding of CD163 Ectodomain and Tumor Necrosis Factor-α in Macrophages. J. Biol. Chem. 2014, 289, 778–788. [Google Scholar] [CrossRef]
- Burdo, T.H.; Lentz, M.R.; Autissier, P.; Krishnan, A.; Halpern, E.; Letendre, S.; Rosenberg, E.S.; Ellis, R.J.; Williams, K.C. Soluble CD163 Made by Monocyte/Macrophages Is a Novel Marker of HIV Activity in Early and Chronic Infection Prior to and after Anti-Retroviral Therapy. J. Infect. Dis. 2011, 204, 154–163. [Google Scholar] [CrossRef] [PubMed]
- Silver, J.; Miller, J.H. Regeneration beyond the Glial Scar. Nat. Rev. Neurosci. 2004, 5, 146–156. [Google Scholar] [CrossRef] [PubMed]
- Wanner, I.B.; Anderson, M.A.; Song, B.; Levine, J.; Fernandez, A.; Gray-Thompson, Z.; Ao, Y.; Sofroniew, M.V. Glial Scar Borders Are Formed by Newly Proliferated, Elongated Astrocytes That Interact to Corral Inflammatory and Fibrotic Cells via STAT3-Dependent Mechanisms after Spinal Cord Injury. J. Neurosci. 2013, 33, 12870–12886. [Google Scholar] [CrossRef]
- Huang, L.; Wu, Z.-B.; Zhuge, Q.; Zheng, W.; Shao, B.; Wang, B.; Sun, F.; Jin, K. Glial Scar Formation Occurs in the Human Brain after Ischemic Stroke. Int. J. Med. Sci. 2014, 11, 344–348. [Google Scholar] [CrossRef]
- Cai, H.; Ma, Y.; Jiang, L.; Mu, Z.; Jiang, Z.; Chen, X.; Wang, Y.; Yang, G.-Y.; Zhang, Z. Hypoxia Response Element-Regulated MMP-9 Promotes Neurological Recovery via Glial Scar Degradation and Angiogenesis in Delayed Stroke. Mol. Ther. 2017, 25, 1448–1459. [Google Scholar] [CrossRef] [PubMed]
- Mosser, D.M.; Edwards, J.P. Exploring the Full Spectrum of Macrophage Activation. Nat. Rev. Immunol. 2008, 8, 958–969. [Google Scholar] [CrossRef] [PubMed]
- Ellwardt, E.; Zipp, F. Molecular Mechanisms Linking Neuroinflammation and Neurodegeneration in MS. Exp. Neurol. 2014, 262 Pt A, 8–17. [Google Scholar] [CrossRef]
- Theoharides, T.C.; Kempuraj, D. Role of SARS-CoV-2 Spike-Protein-Induced Activation of Microglia and Mast Cells in the Pathogenesis of Neuro-COVID. Cells 2023, 12, 688. [Google Scholar] [CrossRef]
- Hampshire, A.; Chatfield, D.A.; MPhil, A.M.; Jolly, A.; Trender, W.; Hellyer, P.J.; Giovane, M.D.; Newcombe, V.F.J.; Outtrim, J.G.; Warne, B.; et al. Multivariate Profile and Acute-Phase Correlates of Cognitive Deficits in a COVID-19 Hospitalised Cohort. eClinicalMedicine 2022, 47, 101417. [Google Scholar] [CrossRef] [PubMed]

| Severe (n = 74) | Non-Severe (n = 70) | ||||
|---|---|---|---|---|---|
| IOT (n = 19) | NIV (n = 55) | VMK (n = 31) | AA (n = 39) | HD (n = 53) | |
| Female, n (%) | 2 (10.5) | 19 (34.5) | 13 (41.9) | 28 (71.8) | 30 (56.6) |
| Age, median (IQR) (years) | 72 (62–78) | 71 (58–82) | 60 (56–70) | 58 (49–65) | 64 (59–69) |
| Deaths, n (%) | 14 (73.7) | 13 (26.6) | 0 (0) | 0 (0) | - |
| Comorbidity | |||||
| Any, n (%) | 12 (63.2) | 37 (67.3) | 16 (51.6) | 18 (46.2) | - |
| Hypertension, n (%) | 8 (42.1) | 20 (36.4) | 8 (25.8) | 14 (35.9) | - |
| Cardiovascular, n (%) | 7 (36.8) | 11 (20.0) | 4 (12.9) | 4 (10.3) | - |
| Diabetes, n (%) | 5 (26.3) | 9 (16.4) | 3 (9.7) | 3 (7.7) | - |
| Pulmonary, n (%) | 3 (15.8) | 8 (14.5) | 3 (9.7) | 4 (10.3) | - |
| Cancer, n (%) | 1 (5.3) | 10 (18.2) | 2 (6.5) | 1 (2.6) | - |
| Renal, n (%) | 0 (0) | 5 (9.1) | 0 (0) | 1 (2.6) | - |
| Symptoms | |||||
| Fever, n (%) | 14 (73.7) | 38 (69.1) | 22 (71.0) | 27 (69.2) | - |
| Cough, n (%) | 7 (36.8) | 23 (41.8) | 10 (32.3) | 15 (38.5) | - |
| Shortness of breath, n (%) | 9 (47.4) | 20 (36.4) | 6 (19.4) | 7 (17.9) | - |
| Myalgia or arthralgia, n (%) | 5 (26.3) | 9 (16.4) | 11 (35.5) | 10 (25.6) | - |
| Diarrhea, n (%) | 9 (47.4) | 6 (10.9) | 6 (19.4) | 3 (7.7) | - |
| Anosmia and ageusia, n (%) | 1 (5.3) | 2 (3.6) | 6 (19.4) | 8 (20.5) | - |
| Sputum production, n (%) | 0 (0) | 2 (3.6) | 0 (0) | 1 (2.6) | - |
| Neurological, n (%) | 2 (10.5) | 7 (12.7) | 4 (12.9) | 2 (5.1) | - |
| Severe (n = 74) | Non-Severe (n = 70) | ||||
|---|---|---|---|---|---|
| IOT (n = 19) | NIV (n = 55) | VMK (n = 31) | AA (n = 39) | HD (n = 53) | |
| Plasma NfL (pg/mL) | 75 (34–99) | 43 (22–97) | 25 (14–54) | 18 (12–31) | 7 (4–11) |
| Plasma GFAP (pg/mL) | 16 (6–25) | 6 (2–12) | 0 (0–0) | 0 (0–0) | 0 (0–0) |
| Plasma sCD163 (ng/mL) | 2011 (1336–3360) | 1867 (1226–2486) | 1268 (1023–1743) | 1208 (946–1711) | 952 (590–1303) |
| Severe (n = 74) | Non-Severe (n = 70) | ||||
|---|---|---|---|---|---|
| with NS (n = 9) | without NS (n = 65) | with NS (n = 6) | without NS (n = 64) | HD (n = 53) | |
| Plasma NfL (pg/mL) | 90 (52–102) | 43 (22–97) | 30 (25–91) | 18 (12–33) | 7 (4–11) |
| Plasma GFAP (pg/mL) | 16 (13–17) | 6 (2–12) | 1 (10–13) | 0 (0–0) | 0 (0–0) |
| Plasma sCD163 (ng/mL) | 1353 (1284–2050) | 1944 (1278–2503) | 1415 (1204–1879) | 1234 (994–1716) | 952 (590–1303) |
| NfL (pg/mL) | GFAP (pg/mL) | sCD163 (ng/mL) | MMP-9 (pg/mL) | TIMP-1 (pg/mL) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Patient | Gender | Age | NS on Hospital Admission | Ventilation Support | CSF | Plasma | CSF | Plasma | CSF | Plasma | CSF | |
| 1 | male | 67 | confusion | VMK | 330 | 30 | 650 | 10 | 23 | 1621 | 1219 | 1346 |
| 2 | male | 83 | confusion, syncope | NIV | 889 | 84 | 764 | 12 | 43 | 1576 | 9 | 49,004 |
| 3 | female | 70 | headache, confusion | AA | 622 | 27 | 662 | 10 | 38 | 1232 | 28 | 48,354 |
| 4 | female | 61 | nystagmus, seizure, forced deviation of the left to the left | VMK | 1591 | 89 | 708 | 13 | 73 | 2654 | 104 | 68,715 |
| 5 | male | 86 | weakness, headache, gaze deviation to the right | NIV | 7961 | 140 | 4336 | 16 | 285 | 1345 | 733 | 106,787 |
| 6 | female | 58 | headache, confusion | AA | 318 | 20 | 570 | 13 | 32 | 1120 | 71 | 134 |
| 7 | male | 36 | headache, confusion | IOT | 4998 | 90 | 80,754 | 18 | 245 | 1336 | 15,871 | 455,431 |
| 8 | male | 69 | lower limb paresthesia | NIV | 18,555 | 103 | 1004 | 12 | 365 | 1231 | 9118 | 697,953 |
| 9 | female | 67 | headache, confusion | VMK | 6661 | 97 | 995 | 11 | 216 | 1453 | 228 | 315,212 |
| 10 | female | 62 | impaired bilateral vision and frontal headache | NIV | 8497 | 60 | 3560 | 14 | 51 | 2910 | 4468 | 32,304 |
| 11 | male | 78 | headache, confusion | IOT | 1720 | 97 | 2150 | 16 | 86 | 1353 | 18 | 138,117 |
| 12 | male | 60 | headache, confusion | VMK | 266 | 30 | 567 | 10 | 44 | 1376 | 151 | 71,671 |
| 13 | male | 50 | headache, confusion | NIV | 456 | 45 | 4560 | 17 | 47 | 1523 | 526 | 98,811 |
| 14 | male | 43 | confusion | NIV | 432 | 43 | 5432 | 16 | 146 | 2524 | 548 | 100,108 |
| 15 | male | 54 | headache, confusion | NIV | 5998 | 101 | 50,754 | 19 | 342 | 1009 | 45,900 | 1346 |
| Severe (n = 34) | Non-Severe (n = 43) | |||||||
|---|---|---|---|---|---|---|---|---|
| Baseline | Tpost | Baseline | Tpost | |||||
| Plasma NfL (pg/mL) | 28 (15–59) | 19 (14–28) | 15 (10–29) | 13 (8–21) | ||||
| Plasma GFAP (pg/mL) | 3 (2–9) | 1 (0–6) | 0 (0–0) | 0 (0–0) | ||||
| Plasma sCD163 (ng/mL) | 1746 (1203–2380) | 1238 (912–2307) | 1296 (1109–1925) | 1062 (708–1436) | ||||
| IOT (n = 5) | NIV (n = 29) | VMK (n = 19) | AA (n = 24) | |||||
| Baseline | Tpost | Baseline | Tpost | Baseline | Tpost | Baseline | Tpost | |
| Plasma NfL (pg/mL) | 34 (22–58) | 28 (21–54) | 27 (15–60) | 18 (13–23) | 16 (10–30) | 13 (10–19) | 15 (11–29) | 13 (7–21) |
| Plasma GFAP (pg/mL) | 0 (0–3) | 0 (0–9) | 3 (2–10) | 1 (0–5) | 0 (0–0) | 0 (0–0) | 0 (0–0) | 0 (0–0) |
| Plasma sCD163 (ng/mL) | 3078 (2345–4084) | 2345 (1996–2613) | 1781 (1200–2360) | 1065 (826–2009) | 1273 (1176–2026) | 1123 (708–1506) | 1368 (1099–1898) | 981 (702–1396) |
| Severe (n = 34) | Non-Severe (n = 43) | ||||
|---|---|---|---|---|---|
| with NS (n = 11) | without NS (n = 23) | with NS (n = 10) | without NS (n = 33) | HD (n = 53) | |
| Plasma NfL (pg/mL) | 23 (17–30) | 18 (13–21) | 14 (10–23) | 11 (8–21) | 7 (4–11) |
| Plasma GFAP (pg/mL) | 0 (0–3) | 1 (0–7) | 0 (0–0) | 0 (0–0) | 0 (0–0) |
| Plasma sCD163 (ng/mL) | 3078 (2345–4084) | 1243 (854–2319) | 856 (741–1473) | 1351 (804–2086) | 952 (590–1303) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zingaropoli, M.A.; Pasculli, P.; Barbato, C.; Petrella, C.; Fiore, M.; Dominelli, F.; Latronico, T.; Ciccone, F.; Antonacci, M.; Liuzzi, G.M.; et al. Biomarkers of Neurological Damage: From Acute Stage to Post-Acute Sequelae of COVID-19. Cells 2023, 12, 2270. https://doi.org/10.3390/cells12182270
Zingaropoli MA, Pasculli P, Barbato C, Petrella C, Fiore M, Dominelli F, Latronico T, Ciccone F, Antonacci M, Liuzzi GM, et al. Biomarkers of Neurological Damage: From Acute Stage to Post-Acute Sequelae of COVID-19. Cells. 2023; 12(18):2270. https://doi.org/10.3390/cells12182270
Chicago/Turabian StyleZingaropoli, Maria Antonella, Patrizia Pasculli, Christian Barbato, Carla Petrella, Marco Fiore, Federica Dominelli, Tiziana Latronico, Federica Ciccone, Michele Antonacci, Grazia Maria Liuzzi, and et al. 2023. "Biomarkers of Neurological Damage: From Acute Stage to Post-Acute Sequelae of COVID-19" Cells 12, no. 18: 2270. https://doi.org/10.3390/cells12182270
APA StyleZingaropoli, M. A., Pasculli, P., Barbato, C., Petrella, C., Fiore, M., Dominelli, F., Latronico, T., Ciccone, F., Antonacci, M., Liuzzi, G. M., Talarico, G., Bruno, G., Galardo, G., Pugliese, F., Lichtner, M., Mastroianni, C. M., Minni, A., & Ciardi, M. R. (2023). Biomarkers of Neurological Damage: From Acute Stage to Post-Acute Sequelae of COVID-19. Cells, 12(18), 2270. https://doi.org/10.3390/cells12182270










