Transcription Factor NFE2L1 Decreases in Glomerulonephropathies after Podocyte Damage
Abstract
1. Introduction
2. Materials and Methods
2.1. Biopsies from Patients with Renal Disease
2.2. Ex Vivo Tissue Slice Preparation
2.3. Cell Culture
2.4. Protein Extraction and Western Blotting
2.5. Cell Fractionation
2.6. Multiplex Immunofluorescence
2.7. Direct Immunofluorescence
2.8. Image Acquisition and Analysis
2.9. Statistical Analysis
3. Results
3.1. Use of Whole-Slide Image Analysis Allows for Measurements of Protein Expression and Glomeruli Features
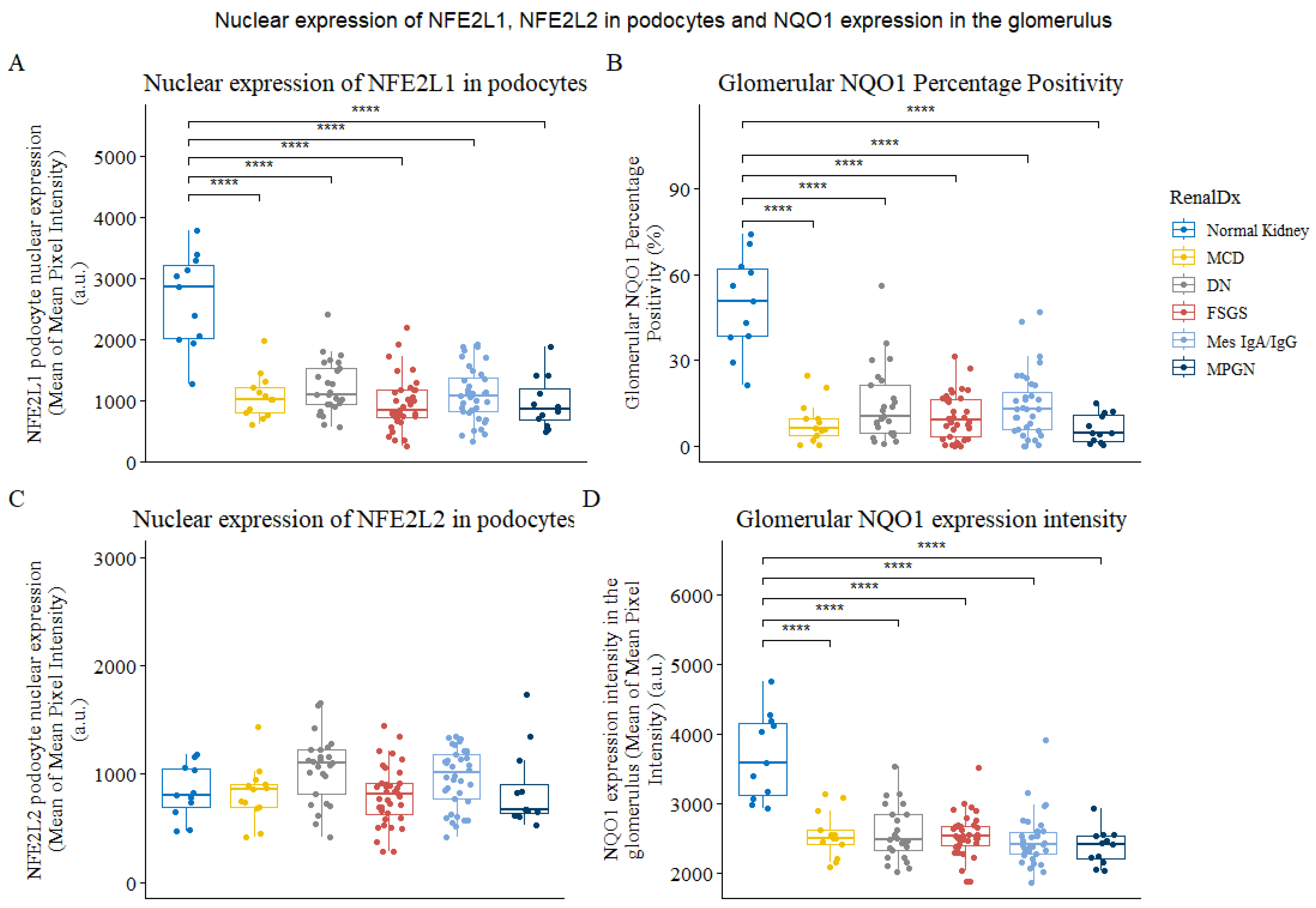
3.2. NFE2L1 Podocyte Nuclear Protein Expression Is Reduced in Kidney Disease
3.3. Protein Expression Levels of NFE2L1, Synaptopodin and NQO1 Are Reduced in PAN-Treated Ex Vivo Renal Tissue Slices
3.4. Protein Expression Levels of Nuclear NFE2L1 Isoform Are Reduced in PAN-Treated Podocyte Cell Line
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kopp, J.B.; Anders, H.-J.; Susztak, K.; Podestà, M.A.; Remuzzi, G.; Hildebrandt, F.; Romagnani, P. Podocytopathies. Nat. Rev. Dis. Primers 2020, 6, 68. [Google Scholar] [CrossRef] [PubMed]
- Perico, L.; Conti, S.; Benigni, A.; Remuzzi, G. Podocyte–Actin Dynamics in Health and Disease. Nat. Rev. Nephrol. 2016, 12, 692–710. [Google Scholar] [CrossRef] [PubMed]
- Haley, K.E.; Kronenberg, N.M.; Liehm, P.; Elshani, M.; Bell, C.; Harrison, D.J.; Gather, M.C.; Reynolds, P.A. Podocyte Injury Elicits Loss and Recovery of Cellular Forces. Sci. Adv. 2018, 4, eaap8030. [Google Scholar] [CrossRef] [PubMed]
- Hogan, J.; Radhakrishnan, J. The Treatment of Minimal Change Disease in Adults. JASN 2013, 24, 702–711. [Google Scholar] [CrossRef] [PubMed]
- Kriz, W.; Lemley, K.V. A Potential Role for Mechanical Forces in the Detachment of Podocytes and the Progression of CKD. JASN 2015, 26, 258–269. [Google Scholar] [CrossRef]
- Susztak, K.; Raff, A.C.; Schiffer, M. Glucose-Induced Reactive Oxygen Species Cause Apoptosis of Podocytes and Podocyte Depletion at the Onset of Diabetic Nephropathy. Diabetes 2006, 55, 9. [Google Scholar] [CrossRef]
- Calizo, R.C.; Bhattacharya, S.; van Hasselt, J.G.C.; Wei, C.; Wong, J.S.; Wiener, R.J.; Ge, X.; Wong, N.J.; Lee, J.-J.; Cuttitta, C.M.; et al. Disruption of Podocyte Cytoskeletal Biomechanics by Dasatinib Leads to Nephrotoxicity. Nat. Commun. 2019, 10, 2061. [Google Scholar] [CrossRef]
- Puelles, V.G.; Cullen-McEwen, L.A.; Taylor, G.E.; Li, J.; Hughson, M.D.; Kerr, P.G.; Hoy, W.E.; Bertram, J.F. Human Podocyte Depletion in Association with Older Age and Hypertension. Am. J. Physiol. Ren. Physiol. 2016, 310, F656–F668. [Google Scholar] [CrossRef]
- Xu, Z.; Chen, L.; Leung, L.; Yen, T.S.B.; Lee, C.; Chan, J.Y. Liver-Specific Inactivation of the Nrf1 Gene in Adult Mouse Leads to Nonalcoholic Steatohepatitis and Hepatic Neoplasia. Proc. Natl. Acad. Sci. USA 2005, 102, 4120–4125. [Google Scholar] [CrossRef]
- Chan, J.Y.; Kwong, M.; Lu, R.; Chang, J.; Wang, B.; Yen, T.S.B.; Kan, Y.W. Targeted Disruption of the Ubiquitous CNC-BZIP Transcription Factor, Nrf-1, Results in Anemia and Embryonic Lethality in Mice. EMBO J. 1998, 17, 1779–1787. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, S.; Xiang, Y.; Qiu, L.; Zhao, H.; Hayes, J.D. The Selective Post-Translational Processing of Transcription Factor Nrf1 Yields Distinct Isoforms That Dictate Its Ability to Differentially Regulate Gene Expression. Sci. Rep. 2015, 5, 12983. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, Y.; Morita, T.; Kim, M.; Iemura, S.; Natsume, T.; Yamamoto, M.; Kobayashi, A. Dual Regulation of the Transcriptional Activity of Nrf1 by β-TrCP- and Hrd1-Dependent Degradation Mechanisms. Mol. Cell Biol. 2011, 31, 4500–4512. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Lucocq, J.M.; Yamamoto, M.; Hayes, J.D. The NHB1 (N-Terminal Homology Box 1) Sequence in Transcription Factor Nrf1 Is Required to Anchor It to the Endoplasmic Reticulum and Also to Enable Its Asparagine-Glycosylation. Biochem. J. 2007, 408, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Radhakrishnan, S.K.; den Besten, W.; Deshaies, R.J. P97-Dependent Retrotranslocation and Proteolytic Processing Govern Formation of Active Nrf1 upon Proteasome Inhibition. eLife 2014, 3, e01856. [Google Scholar] [CrossRef]
- Northrop, A.; Vangala, J.R.; Feygin, A.; Radhakrishnan, S.K. Disabling the Protease DDI2 Attenuates the Transcriptional Activity of NRF1 and Potentiates Proteasome Inhibitor Cytotoxicity. Int. J. Mol. Sci. 2020, 21, 327. [Google Scholar] [CrossRef]
- Johnsen, O.; Skammelsrud, N.; Luna, L.; Nishizawa, M.; Prydz, H.; Kolstø, A.B. Small Maf Proteins Interact with the Human Transcription Factor TCF11/Nrf1/LCR-F1. Nucleic Acids Res. 1996, 24, 4289–4297. [Google Scholar] [CrossRef]
- Siegel, D.; Dehn, D.D.; Bokatzian, S.S.; Quinn, K.; Backos, D.S.; Di Francesco, A.; Bernier, M.; Reisdorph, N.; de Cabo, R.; Ross, D. Redox Modulation of NQO1. PLoS ONE 2018, 13, e0190717. [Google Scholar] [CrossRef]
- Zappa, F.; Ward, T.; Pedrinis, E.; Butler, J.; McGown, A. NAD(P)H:Quinone Oxidoreductase 1 Expression in Kidney Podocytes. J. Histochem. Cytochem. 2003, 51, 297–302. [Google Scholar] [CrossRef]
- Katsuoka, F.; Otsuki, A.; Hatanaka, N.; Okuyama, H.; Yamamoto, M. Target Gene Diversity of the Nrf1-MafG Transcription Factor Revealed by a Tethered Heterodimer. Mol. Cell. Biol. 2022, 42, e00520–e00521. [Google Scholar] [CrossRef]
- Harder, J.L.; Menon, R.; Otto, E.A.; Zhou, J.; Eddy, S.; Wys, N.L.; O’Connor, C.; Luo, J.; Nair, V.; Cebrian, C.; et al. Organoid Single Cell Profiling Identifies a Transcriptional Signature of Glomerular Disease. JCI Insight 2019, 4, e122697. [Google Scholar] [CrossRef]
- Saleem, M.A.; O’Hare, M.J.; Reiser, J.; Coward, R.J.; Inward, C.D.; Farren, T.; Xing, C.Y.; Ni, L.; Mathieson, P.W.; Mundel, P. A Conditionally Immortalized Human Podocyte Cell Line Demonstrating Nephrin and Podocin Expression. JASN 2002, 13, 630–638. [Google Scholar] [CrossRef] [PubMed]
- Bankhead, P.; Loughrey, M.B.; Fernández, J.A.; Dombrowski, Y.; McArt, D.G.; Dunne, P.D.; McQuaid, S.; Gray, R.T.; Murray, L.J.; Coleman, H.G.; et al. QuPath: Open Source Software for Digital Pathology Image Analysis. Sci. Rep. 2017, 7, 16878. [Google Scholar] [CrossRef] [PubMed]
- Nagata, M. Cyclin-Dependent Kinase Inhibitors: P27kip1 and P57kip2 Expression during Human Podocyte Differentiation. Nephrol. Dial. Transplant. 1999, 14, 48–51. [Google Scholar] [CrossRef] [PubMed]
- Moon, S.-J.; Jeong, J.Y.; Kim, J.-H.; Choi, D.-H.; Choi, H.; Chang, Y.-K.; Na, K.R.; Lee, K.W.; Lee, C.-H.; Choi, D.E.; et al. The Potential Roles of NAD(P)H:Quinone Oxidoreductase 1 in the Development of Diabetic Nephropathy and Actin Polymerization. Sci. Rep. 2020, 10, 17735. [Google Scholar] [CrossRef] [PubMed]
- Puelles, V.G.; Bertram, J.F.; Moeller, M.J. Quantifying Podocyte Depletion: Theoretical and Practical Considerations. Cell Tissue Res. 2017, 369, 229–236. [Google Scholar] [CrossRef]
- Roelants, C.; Pillet, C.; Franquet, Q.; Sarrazin, C.; Peilleron, N.; Giacosa, S.; Guyon, L.; Fontanell, A.; Fiard, G.; Long, J.-A.; et al. Ex-Vivo Treatment of Tumor Tissue Slices as a Predictive Preclinical Method to Evaluate Targeted Therapies for Patients with Renal Carcinoma. Cancers 2020, 12, 232. [Google Scholar] [CrossRef]
- Xiang, Y.; Wang, M.; Hu, S.; Qiu, L.; Yang, F.; Zhang, Z. Mechanisms Controlling the Multistage Post-Translational Processing of Endogenous Nrf1 α/TCF11 Proteins to Yield Distinct Isoforms within the Coupled Positive and Negative Feedback Circuits. Toxicol. Appl. Pharmacol. 2018, 360, 212–235. [Google Scholar] [CrossRef]
- Tsujita, T.; Baird, L.; Furusawa, Y.; Katsuoka, F.; Hou, Y.; Gotoh, S.; Kawaguchi, S.; Yamamoto, M. Discovery of an NRF1-Specific Inducer from a Large-Scale Chemical Library Using a Direct NRF1-Protein Monitoring System. Genes. Cells 2015, 20, 563–577. [Google Scholar] [CrossRef]
- Rascle, A.; Suleiman, H.; Neumann, T.; Witzgall, R. Role of Transcription Factors in Podocytes. Nephron Exp. Nephrol. 2007, 106, e60–e66. [Google Scholar] [CrossRef]
- Usui, T.; Morito, N.; Shawki, H.H.; Sato, Y.; Tsukaguchi, H.; Hamada, M.; Jeon, H.; Yadav, M.K.; Kuno, A.; Tsunakawa, Y.; et al. Transcription Factor MafB in Podocytes Protects against the Development of Focal Segmental Glomerulosclerosis. Kidney Int. 2020, 98, 391–403. [Google Scholar] [CrossRef]
- Maezawa, Y.; Onay, T.; Scott, R.P.; Keir, L.S.; Dimke, H.; Li, C.; Eremina, V.; Maezawa, Y.; Jeansson, M.; Shan, J.; et al. Loss of the Podocyte-Expressed Transcription Factor Tcf21/Pod1 Results in Podocyte Differentiation Defects and FSGS. JASN 2014, 25, 2459–2470. [Google Scholar] [CrossRef] [PubMed]
- Endlich, N.; Kliewe, F.; Kindt, F.; Schmidt, K.; Kotb, A.M.; Artelt, N.; Lindenmeyer, M.T.; Cohen, C.D.; Döring, F.; Kuss, A.W.; et al. The Transcription Factor Dach1 Is Essential for Podocyte Function. J. Cell Mol. Med. 2018, 22, 2656–2669. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Zhang, D.; Motojima, M.; Kume, T.; Hou, Q.; Pan, Y.; Duan, A.; Zhang, M.; Jiang, S.; Hou, J.; et al. Super-Enhancer-Associated Transcription Factors Maintain Transcriptional Regulation in Mature Podocytes. JASN 2021, 32, 1323–1337. [Google Scholar] [CrossRef]
- Koizumi, S.; Hamazaki, J.; Murata, S. Transcriptional Regulation of the 26S Proteasome by Nrf1. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci. 2018, 94, 325–336. [Google Scholar] [CrossRef] [PubMed]
- Biswas, M.; Chan, J.Y. Role of Nrf1 in Antioxidant Response Element-Mediated Gene Expression and Beyond. Toxicol. Appl. Pharmacol. 2010, 244, 16–20. [Google Scholar] [CrossRef]
- Xing, W.; Singgih, A.; Kapoor, A.; Alarcon, C.M.; Baylink, D.J.; Mohan, S. Nuclear Factor-E2-Related Factor-1 Mediates Ascorbic Acid Induction of Osterix Expression via Interaction with Antioxidant-Responsive Element in Bone Cells. J. Biol. Chem. 2007, 282, 22052–22061. [Google Scholar] [CrossRef]
- Lindström, N.O.; De Sena Brandine, G.; Tran, T.; Ransick, A.; Suh, G.; Guo, J.; Kim, A.D.; Parvez, R.K.; Ruffins, S.W.; Rutledge, E.A.; et al. Progressive Recruitment of Mesenchymal Progenitors Reveals a Time-Dependent Process of Cell Fate Acquisition in Mouse and Human Nephrogenesis. Dev. Cell 2018, 45, 651–660.e4. [Google Scholar] [CrossRef]
- Lake, B.B.; Menon, R.; Winfree, S.; Hu, Q.; Ferreira, R.M.; Kalhor, K.; Barwinska, D.; Otto, E.A.; Ferkowicz, M.; Diep, D.; et al. An Atlas of Healthy and Injured Cell States and Niches in the Human Kidney. Nature 2023, 619, 585–594. [Google Scholar] [CrossRef]
- Widenmaier, S.B.; Snyder, N.A.; Nguyen, T.B.; Arduini, A.; Lee, G.Y.; Arruda, A.P.; Saksi, J.; Bartelt, A.; Hotamisligil, G.S. NRF1 Is an ER Membrane Sensor That Is Central to Cholesterol Homeostasis. Cell 2017, 171, 1094–1109.e15. [Google Scholar] [CrossRef]
- Moorhead, J.F.; El-Nahas, M.; Chan, M.K.; Varghese, Z. Lipid Nephrotoxicity in Chronic Progressive Glomerular and Tubulo-Interstitial Disease. Lancet 1982, 320, 1309–1311. [Google Scholar] [CrossRef]
- Wahl, P.; Ducasa, G.M.; Fornoni, A. Systemic and Renal Lipids in Kidney Disease Development and Progression. Am. J. Physiol. Ren. Physiol. 2016, 310, F433–F445. [Google Scholar] [CrossRef] [PubMed]
- Tufro, A. Cholesterol Accumulation in Podocytes: A Potential Novel Targetable Pathway in Diabetic Nephropathy. Diabetes 2013, 62, 3661–3662. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hua, W.; Huang, H.; Tan, L.; Wan, J.; Gui, H.; Zhao, L.; Ruan, X.; Chen, X.; Du, X. CD36 Mediated Fatty Acid-Induced Podocyte Apoptosis via Oxidative Stress. PLoS ONE 2015, 10, e0127507. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.S.; Kruth, H.S. Accumulation of Cholesterol in the Lesions of Focal Segmental Glomerulosclerosis. Nephrology 2003, 8, 223–224. [Google Scholar] [CrossRef]
- Herman-Edelstein, M.; Scherzer, P.; Tobar, A.; Levi, M.; Gafter, U. Altered Renal Lipid Metabolism and Renal Lipid Accumulation in Human Diabetic Nephropathy. J. Lipid Res. 2014, 55, 561–572. [Google Scholar] [CrossRef]
- Ross, D.; Siegel, D. Functions of NQO1 in Cellular Protection and CoQ10 Metabolism and Its Potential Role as a Redox Sensitive Molecular Switch. Front. Physiol. 2017, 8, 595. [Google Scholar] [CrossRef]
- Kobayashi, A.; Tsukide, T.; Miyasaka, T.; Morita, T.; Mizoroki, T.; Saito, Y.; Ihara, Y.; Takashima, A.; Noguchi, N.; Fukamizu, A.; et al. Central Nervous System-Specific Deletion of Transcription Factor Nrf1 Causes Progressive Motor Neuronal Dysfunction: CNS-Specific Deletion of Transcription Factor Nrf1. Genes. Cells 2011, 16, 692–703. [Google Scholar] [CrossRef] [PubMed]
- Miyazaki, Y.; Shimizu, A.; Pastan, I.; Taguchi, K.; Naganuma, E.; Suzuki, T.; Hosoya, T.; Yokoo, T.; Saito, A.; Miyata, T.; et al. Keap1 Inhibition Attenuates Glomerulosclerosis. Nephrol. Dial. Transplant. 2014, 29, 783–791. [Google Scholar] [CrossRef]
- Liu, B.-C.; Song, X.; Lu, X.-Y.; Li, D.T.; Eaton, D.C.; Shen, B.-Z.; Li, X.-Q.; Ma, H.-P. High Glucose Induces Podocyte Apoptosis by Stimulating TRPC6 via Elevation of Reactive Oxygen Species. Biochim. Biophys. Acta (BBA) Mol. Cell Res. 2013, 1833, 1434–1442. [Google Scholar] [CrossRef]
- Wang, C.; Li, C.; Peng, H.; Ye, Z.; Zhang, J.; Liu, X.; Lou, T. Activation of the Nrf2-ARE Pathway Attenuates Hyperglycemia-Mediated Injuries in Mouse Podocytes. Cell Physiol. Biochem. 2014, 34, 891–902. [Google Scholar] [CrossRef]
- Zheng, H.; Whitman, S.A.; Wu, W.; Wondrak, G.T.; Wong, P.K.; Fang, D.; Zhang, D.D. Therapeutic Potential of Nrf2 Activators in Streptozotocin-Induced Diabetic Nephropathy. Diabetes 2011, 60, 3055–3066. [Google Scholar] [CrossRef]
- Zhou, S.; Wang, P.; Qiao, Y.; Ge, Y.; Wang, Y.; Quan, S.; Yao, R.; Zhuang, S.; Wang, L.J.; Du, Y.; et al. Genetic and Pharmacologic Targeting of Glycogen Synthase Kinase 3 β Reinforces the Nrf2 Antioxidant Defense against Podocytopathy. JASN 2016, 27, 2289–2308. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Ge, Y.; Dworkin, L.; Peng, A.; Gong, R. The β Isoform of GSK3 Mediates Podocyte Autonomous Injury in Proteinuric Glomerulopathy: GSK3β Mediates Podocyte Autonomous Injury. J. Pathol. 2016, 239, 23–35. [Google Scholar] [CrossRef] [PubMed]
- de Zeeuw, D.; Akizawa, T.; Audhya, P.; Bakris, G.L.; Chin, M.; Christ-Schmidt, H.; Goldsberry, A.; Houser, M.; Krauth, M.; Lambers Heerspink, H.J.; et al. Bardoxolone Methyl in Type 2 Diabetes and Stage 4 Chronic Kidney Disease. N. Engl. J. Med. 2013, 369, 2492–2503. [Google Scholar] [CrossRef] [PubMed]







| Characteristic | All (n = 127) | MCD | DN | FSGS | MesIgA/IgG | MCGN | |
|---|---|---|---|---|---|---|---|
| Sex | F | 52 (40.9%) | 3 (23.1%) | 7 (25.9%) | 19 (51.4%) | 18 (48.6%) | 5 (38.5%) |
| M | 75 (59%) | 10 (76.9%) | 20 (74.1%) | 18 (48.6%) | 19 (51.4%) | 8 (61.5%) | |
| * Age | 49.0 ± 15.8 | 51.2 ± 21.2 | 62.2 ± 11.8 | 52.0 ± 17.5 | 44 ± 15.4 | 49.3 ± 13.6 | |
| * s-Creatinine (µmol/L) | 229.5 ± 192.7 | 110.9 ± 113.1 | 203.7 ± 115.4 | 183.2 ± 105.2 | 184.5 ± 216.0 | 225.8 ± 156.6 | |
| * Urea (mmol/L) | 12.2 ± 7.6 | 9.1 ± 8.7 | 13.1 ± 6.7 | 10.7 ± 5 | 10.3 ± 9.3 | 12.1 ± 4.6 | |
| * Albumin | 27.6 ± 18.5 | 16.7 ± 16.4 | 24.6 ± 16.9 | 24.9 ± 16.6 | 31.1 ± 18.6 | 27.3 ± 19.2 | |
| * eGFR (mL/min/1.73 m2) | 35.5 ± 8.3 | 53.3 ± 7.6 | 34.7 ± 10.9 | 36.7 ± 10.6 | 44.4 ± 7.0 | 33.3 ± 3.5 | |
| * Blood Glucose (mmol/L) | 7.3 ± 4.3 | 7.3 ± 3.0 | 11.3 ± 6 | 6.1 ± 1.6 | 5.8 ± 3.6 | 5.33 ± 0.6 | |
| Feature | Description |
|---|---|
| Number of glomeruli | Sum of annotated glomeruli per case |
| Area of annotated glomeruli | Mean area of all annotated glomeruli (μm2) |
| Number of podocytes | Sum of p57-positive podocytes per case |
| Podocytes per glomerulus | Total number of p57-positive podocyte/total number of glomeruli in a case |
| Area of NQO1 positivity. | Mean of NQO1-positive area within glomerulus (μm2) |
| Glomerular NQO1 percentage positivity | Mean percentage of NQO1-positive area/total glomerulus area (%) |
| NQO1 expression intensity | Mean of total mean NQO1 pixel intensity within NQO1-positive area in the glomerulus |
| NFE2L2 intensity within NQO1-positive area | Mean of total mean NFE2L2 pixel intensity within NQO1-positive area in the glomerulus |
| NFE2L1 intensity within NQO1-positive area | Mean of total mean NFE2L1 pixel intensity within NQO1-positive area in the glomerulus |
| Podocyte nuclear area | Mean of Podocyte nuclear area (μm2) area in a case |
| p57 podocyte nuclear expression | Mean of total mean p57 pixel intensity within podocyte nucleus |
| NFE2L1 podocyte nuclear expression | Mean of total mean NFE2L1 pixel intensity within podocyte nucleus |
| NFE2L2 podocyte nuclear expression | Mean of total mean NFE2L2 pixel intensity within podocyte nucleus |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elshani, M.; Um, I.H.; Leung, S.; Reynolds, P.A.; Chapman, A.; Kudsy, M.; Harrison, D.J. Transcription Factor NFE2L1 Decreases in Glomerulonephropathies after Podocyte Damage. Cells 2023, 12, 2165. https://doi.org/10.3390/cells12172165
Elshani M, Um IH, Leung S, Reynolds PA, Chapman A, Kudsy M, Harrison DJ. Transcription Factor NFE2L1 Decreases in Glomerulonephropathies after Podocyte Damage. Cells. 2023; 12(17):2165. https://doi.org/10.3390/cells12172165
Chicago/Turabian StyleElshani, Mustafa, In Hwa Um, Steve Leung, Paul A. Reynolds, Alex Chapman, Mary Kudsy, and David J. Harrison. 2023. "Transcription Factor NFE2L1 Decreases in Glomerulonephropathies after Podocyte Damage" Cells 12, no. 17: 2165. https://doi.org/10.3390/cells12172165
APA StyleElshani, M., Um, I. H., Leung, S., Reynolds, P. A., Chapman, A., Kudsy, M., & Harrison, D. J. (2023). Transcription Factor NFE2L1 Decreases in Glomerulonephropathies after Podocyte Damage. Cells, 12(17), 2165. https://doi.org/10.3390/cells12172165






