The Immune Response to SARS-CoV-2 Vaccine in a Cohort of Family Pediatricians from Southern Italy
Abstract
1. Introduction
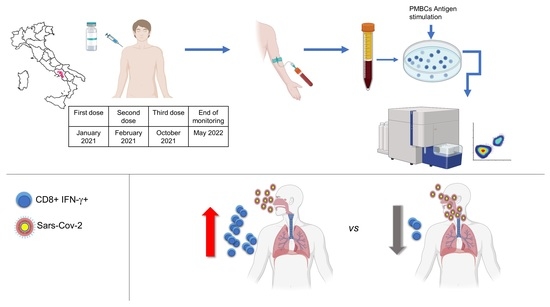
2. Materials and Methods
2.1. Study Participants
2.2. Evaluation of SARS-CoV-2-Specific IgG
2.3. Sample Collection and Storage
2.4. Antigen Stimulation
2.5. Flow Cytometry
2.6. Cytokines Release Assay
2.7. Statistical Analyses
3. Results
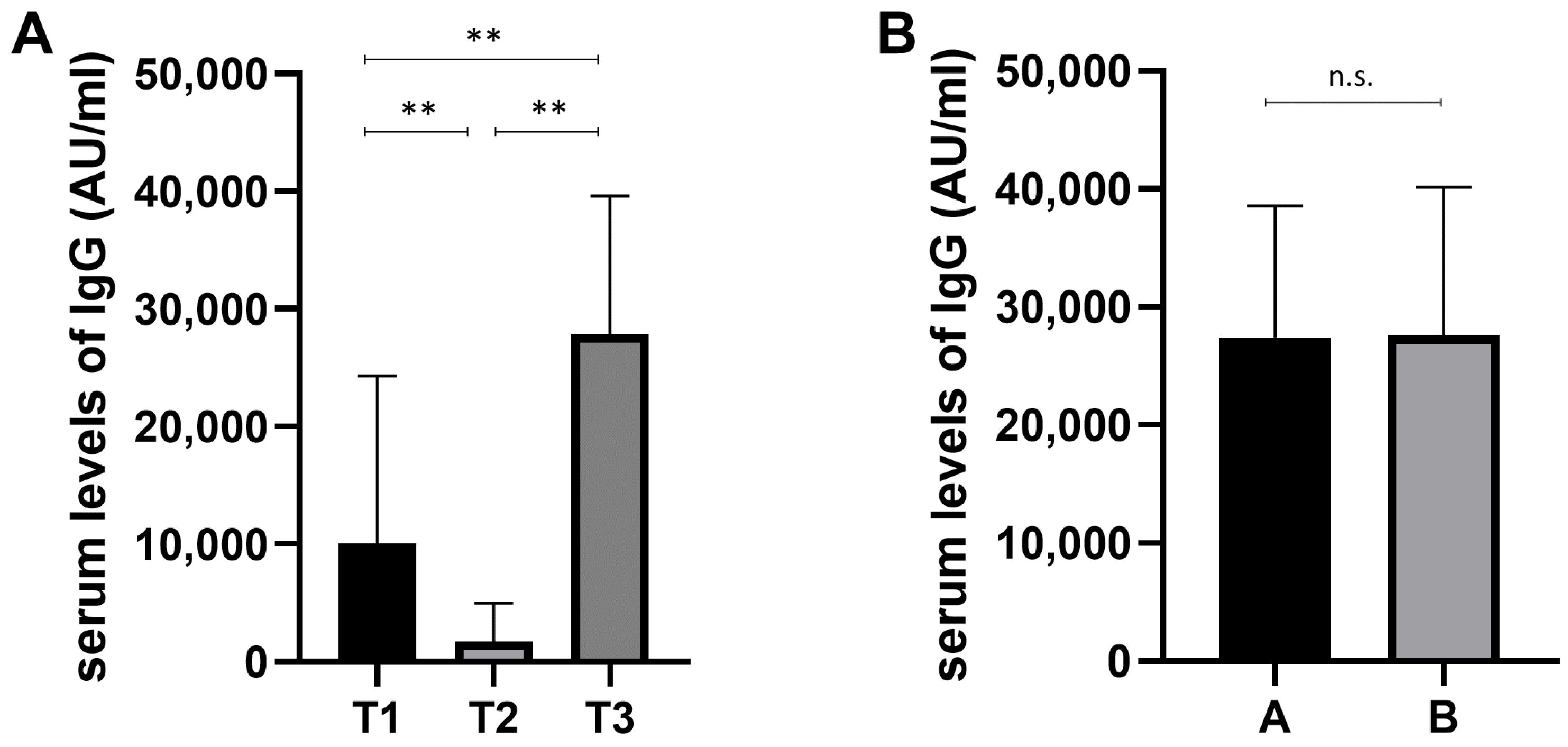
3.1. Study Group and Analysis of Humoral Response
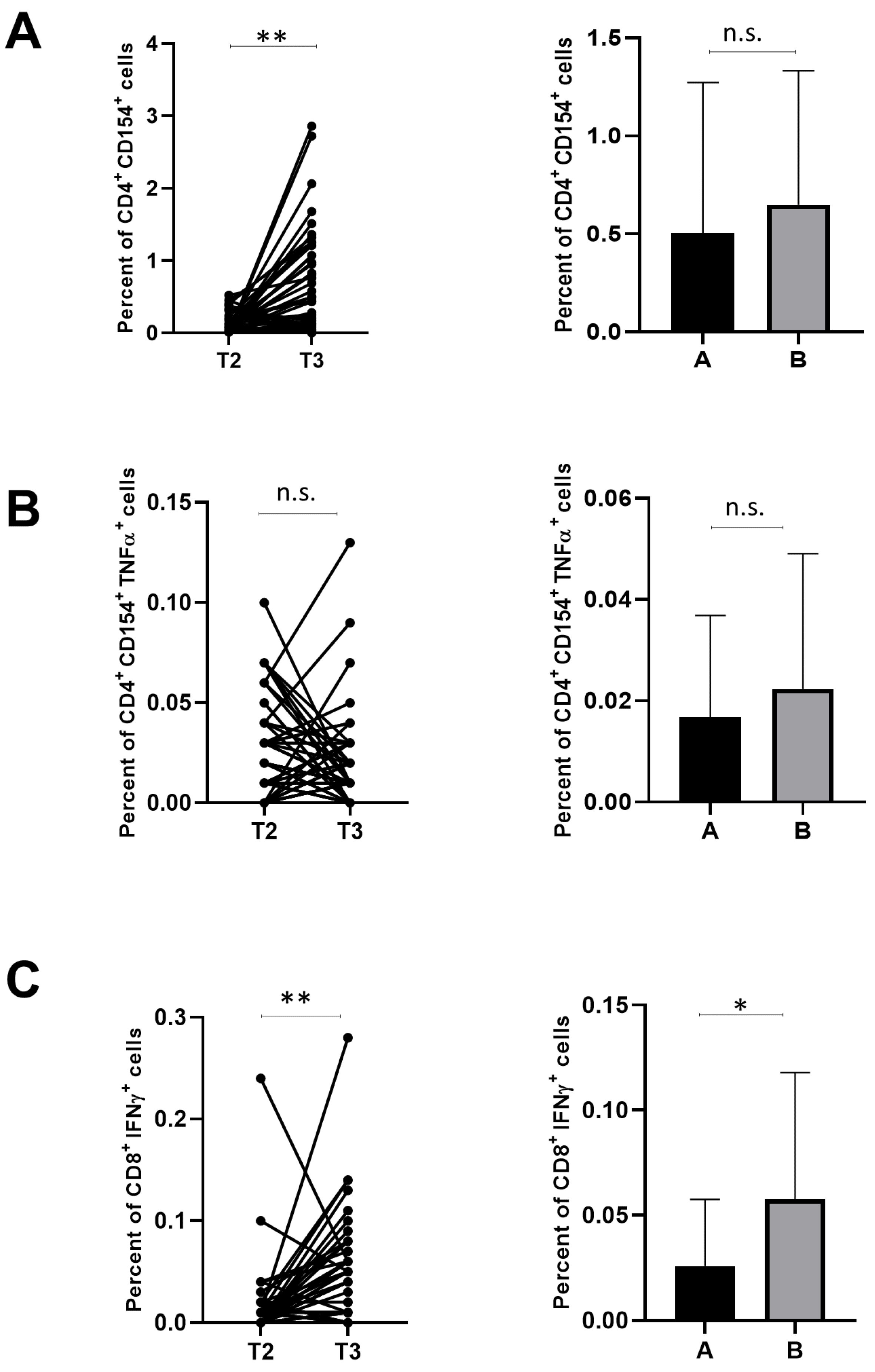
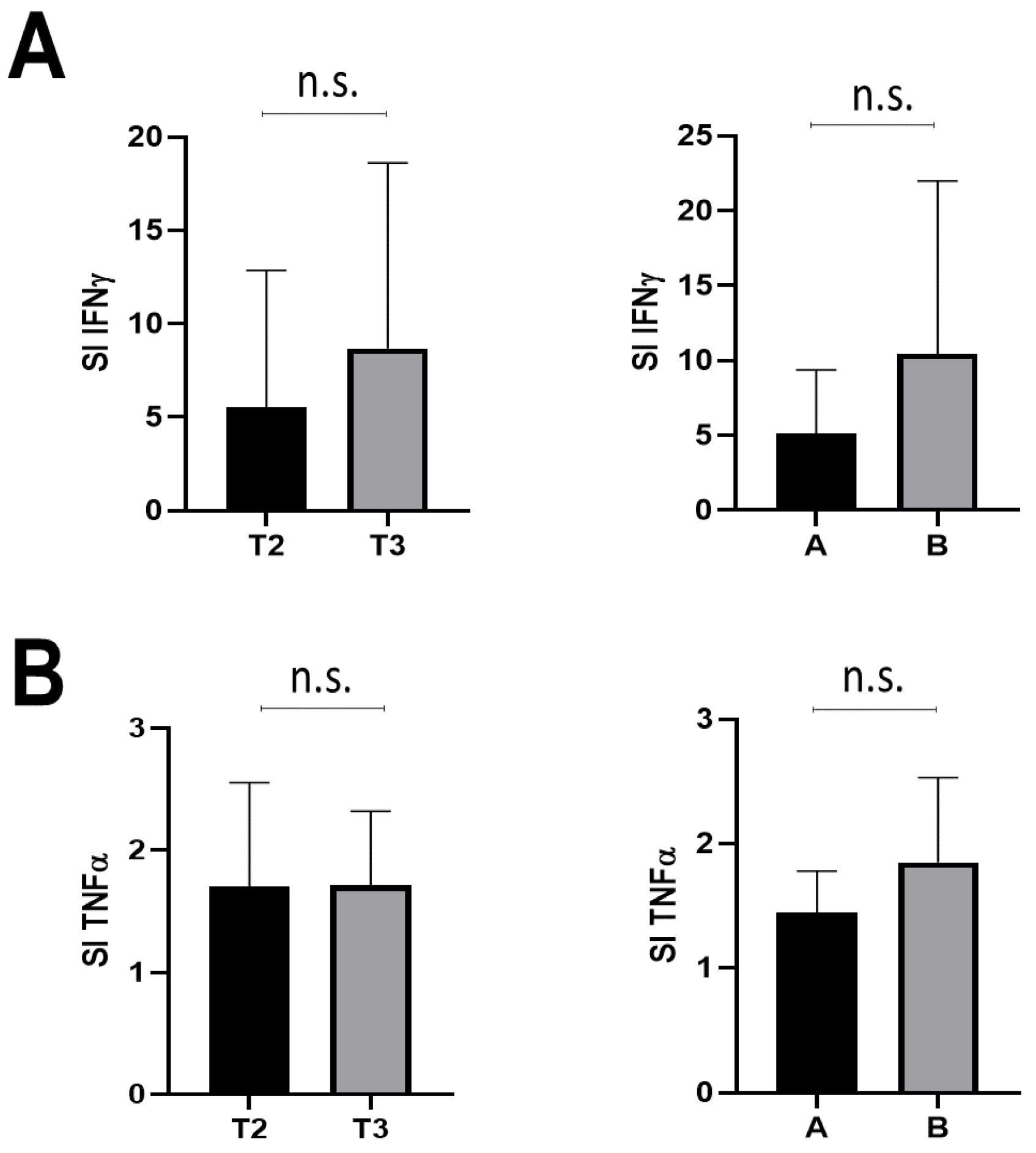
3.2. T-Cell Response
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- Sahin, U.; Muik, A.; Vogler, I.; Derhovanessian, E.; Kranz, L.M.; Vormehr, M.; Quandt, J.; Bidmon, N.; Ulges, A.; Baum, A.; et al. BNT162b2 vaccine induces neutralizing antibodies and poly-specific T cells in humans. Nature 2021, 595, 572–577. [Google Scholar] [CrossRef] [PubMed]
- GeurtsvanKessel, C.H.; Geers, D.; Schmitz, K.S.; Mykytyn, A.Z.; Lamers, M.M.; Bogers, S.; Scherbeijn, S.; Gommers, L.; Sablerolles, R.S.G.; Nieuwkoop, N.N.; et al. Divergent SARS-CoV-2 Omicron-reactive T and B cell responses in COVID-19 vaccine recipients. Sci. Immunol. 2022, 7, eabo2202. [Google Scholar] [CrossRef] [PubMed]
- Araf, Y.; Akter, F.; Tang, Y.D.; Fatemi, R.; Parvez, M.S.A.; Zheng, C.; Hossain, M.G. Omicron variant of SARS-CoV-2: Genomics, transmissibility, and responses to current COVID-19 vaccines. J. Med. Virol. 2022, 94, 1825–1832. [Google Scholar] [CrossRef]
- Menni, C.; Valdes, A.M.; Polidori, L.; Antonelli, M.; Penamakuri, S.; Nogal, A.; Louca, P.; May, A.; Figueiredo, J.C.; Hu, C.; et al. Symptom prevalence, duration, and risk of hospital admission in individuals infected with SARS-CoV-2 during periods of omicron and delta variant dominance: A prospective observational study from the ZOE COVID Study. Lancet 2022, 399, 1618–1624. [Google Scholar] [CrossRef] [PubMed]
- Simon, B.; Rubey, H.; Gromann, M.; Knopf-Volkerer, A.; Hemedi, B.; Zehetmayer, S.; Kirsch, B. SARS-CoV-2 Antibody and T Cell Response after a Third Vaccine Dose in Hemodialysis Patients Compared with Healthy Controls. Vaccines 2022, 10, 694. [Google Scholar] [CrossRef]
- Panizo, N.; Gimenez, E.; Albert, E.; Zulaica, J.; Rodriguez-Moreno, A.; Rusu, L.; Gimenez-Civera, E.; Puchades, M.J.; D’Marco, L.; Gandia-Salmeron, L.; et al. SARS-CoV-2-Spike Antibody and T-Cell Responses Elicited by a Homologous Third mRNA COVID-19 Dose in Hemodialysis and Kidney Transplant Recipients. Microorganisms 2022, 10, 2275. [Google Scholar] [CrossRef]
- Guerrera, G.; Picozza, M.; D’Orso, S.; Placido, R.; Pirronello, M.; Verdiani, A.; Termine, A.; Fabrizio, C.; Giannessi, F.; Sambucci, M.; et al. BNT162b2 vaccination induces durable SARS-CoV-2-specific T cells with a stem cell memory phenotype. Sci. Immunol. 2021, 6, eabl5344. [Google Scholar] [CrossRef]
- Tarke, A.; Coelho, C.H.; Zhang, Z.; Dan, J.M.; Yu, E.D.; Methot, N.; Bloom, N.I.; Goodwin, B.; Phillips, E.; Mallal, S.; et al. SARS-CoV-2 vaccination induces immunological T cell memory able to cross-recognize variants from Alpha to Omicron. Cell 2022, 185, 847–859.e811. [Google Scholar] [CrossRef]
- Painter, M.M.; Mathew, D.; Goel, R.R.; Apostolidis, S.A.; Pattekar, A.; Kuthuru, O.; Baxter, A.E.; Herati, R.S.; Oldridge, D.A.; Gouma, S.; et al. Rapid induction of antigen-specific CD4(+) T cells is associated with coordinated humoral and cellular immunity to SARS-CoV-2 mRNA vaccination. Immunity 2021, 54, 2133–2142.e2133. [Google Scholar] [CrossRef]
- Moss, P. The T cell immune response against SARS-CoV-2. Nat. Immunol. 2022, 23, 186–193. [Google Scholar] [CrossRef]
- Jacobsen, H.; Cobos Jimenez, V.; Sitaras, I.; Bar-Zeev, N.; Cicin-Sain, L.; Higdon, M.M.; Deloria-Knoll, M. Post-vaccination T cell immunity to omicron. Front. Immunol. 2022, 13, 944713. [Google Scholar] [CrossRef] [PubMed]
- Noor, R. Host Protective Immunity against Severe Acute Respiratory Coronavirus 2 (SARS-CoV-2) and the COVID-19 Vaccine-Induced Immunity against SARS-CoV-2 and Its Variants. Viruses 2022, 14, 2541. [Google Scholar] [CrossRef]
- Jeong, S.; Lee, N.; Lee, S.K.; Cho, E.J.; Hyun, J.; Park, M.J.; Song, W.; Kim, H.S. Humoral and Cellular Responses to BNT162b2 as a Booster Following Two Doses of ChAdOx1 nCov-19 Determined Using Three SARS-CoV-2 Antibody Assays and an Interferon-Gamma Release Assay: A Prospective Longitudinal Study in Healthcare Workers. Front. Immunol. 2022, 13, 859019. [Google Scholar] [CrossRef]
- Murugesan, K.; Jagannathan, P.; Pham, T.D.; Pandey, S.; Bonilla, H.F.; Jacobson, K.; Parsonnet, J.; Andrews, J.R.; Weiskopf, D.; Sette, A.; et al. Interferon-gamma Release Assay for Accurate Detection of Severe Acute Respiratory Syndrome Coronavirus 2 T-Cell Response. Clin. Infect. Dis. 2021, 73, e3130–e3132. [Google Scholar] [CrossRef]
- Clemenceau, B.; Guillaume, T.; Coste-Burel, M.; Peterlin, P.; Garnier, A.; Le Bourgeois, A.; Jullien, M.; Ollier, J.; Grain, A.; Bene, M.C.; et al. SARS-CoV-2 T-Cell Responses in Allogeneic Hematopoietic Stem Cell Recipients following Two Doses of BNT162b2 mRNA Vaccine. Vaccines 2022, 10, 448. [Google Scholar] [CrossRef]
- Valyi-Nagy, I.; Matula, Z.; Gonczi, M.; Tasnady, S.; Beko, G.; Reti, M.; Ajzner, E.; Uher, F. Comparison of antibody and T cell responses elicited by BBIBP-CorV (Sinopharm) and BNT162b2 (Pfizer-BioNTech) vaccines against SARS-CoV-2 in healthy adult humans. Geroscience 2021, 43, 2321–2331. [Google Scholar] [CrossRef]
- Gimenez, E.; Albert, E.; Torres, I.; Remigia, M.J.; Alcaraz, M.J.; Galindo, M.J.; Blasco, M.L.; Solano, C.; Forner, M.J.; Redon, J.; et al. SARS-CoV-2-reactive interferon-gamma-producing CD8+ T cells in patients hospitalized with coronavirus disease 2019. J. Med. Virol. 2021, 93, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Thieme, C.J.; Anft, M.; Paniskaki, K.; Blazquez-Navarro, A.; Doevelaar, A.; Seibert, F.S.; Hoelzer, B.; Konik, M.J.; Berger, M.M.; Brenner, T.; et al. Robust T Cell Response Toward Spike, Membrane, and Nucleocapsid SARS-CoV-2 Proteins Is Not Associated with Recovery in Critical COVID-19 Patients. Cell Rep. Med. 2020, 1, 100092. [Google Scholar] [CrossRef] [PubMed]
- Gelzo, M.; Cacciapuoti, S.; Pinchera, B.; De Rosa, A.; Cernera, G.; Scialo, F.; Mormile, M.; Fabbrocini, G.; Parrella, R.; Gentile, I.; et al. Prognostic Role of Neutrophil to Lymphocyte Ratio in COVID-19 Patients: Still Valid in Patients That Had Started Therapy? Front. Public Health 2021, 9, 664108. [Google Scholar] [CrossRef]
- Scalia, G.; Raia, M.; Gelzo, M.; Cacciapuoti, S.; Rosa, A.; Pinchera, B.; Scotto, R.; Tripodi, L.; Mormile, M.; Fabbrocini, G.; et al. Lymphocyte Population Changes at Two Time Points during the Acute Period of COVID-19 Infection. J. Clin. Med. 2022, 11, 4306. [Google Scholar] [CrossRef]
- Hurme, A.; Jalkanen, P.; Heroum, J.; Liedes, O.; Vara, S.; Melin, M.; Terasjarvi, J.; He, Q.; Poysti, S.; Hanninen, A.; et al. Long-Lasting T Cell Responses in BNT162b2 COVID-19 mRNA Vaccinees and COVID-19 Convalescent Patients. Front. Immunol. 2022, 13, 869990. [Google Scholar] [CrossRef]
- Havervall, S.; Marking, U.; Svensson, J.; Greilert-Norin, N.; Bacchus, P.; Nilsson, P.; Hober, S.; Gordon, M.; Blom, K.; Klingstrom, J.; et al. Anti-Spike Mucosal IgA Protection against SARS-CoV-2 Omicron Infection. N. Engl. J. Med. 2022, 387, 1333–1336. [Google Scholar] [CrossRef]
- Berni Canani, R.; Comegna, M.; Paparo, L.; Cernera, G.; Bruno, C.; Strisciuglio, C.; Zollo, I.; Gravina, A.G.; Miele, E.; Cantone, E.; et al. Age-Related Differences in the Expression of Most Relevant Mediators of SARS-CoV-2 Infection in Human Respiratory and Gastrointestinal Tract. Front. Pediatr. 2021, 9, 697390. [Google Scholar] [CrossRef] [PubMed]
- Naaber, P.; Tserel, L.; Kangro, K.; Punapart, M.; Sepp, E.; Jurjenson, V.; Karner, J.; Haljasmagi, L.; Haljasorg, U.; Kuusk, M.; et al. Protective antibodies and T cell responses to Omicron variant after the booster dose of BNT162b2 vaccine. Cell Rep. Med. 2022, 3, 100716. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Han, J.; Zhang, Y.; He, J.; Yu, W.; Zhang, X.; Wu, J.; Zhang, S.; Kong, Y.; Guo, Y.; et al. SARS-CoV-2 Omicron Variant: Epidemiological Features, Biological Characteristics, and Clinical Significance. Front. Immunol. 2022, 13, 877101. [Google Scholar] [CrossRef]
- Moller, J.F.; Moller, B.; Wiedenmann, B.; Berg, T.; Schott, E. CD154, a marker of antigen-specific stimulation of CD4 T cells, is associated with response to treatment in patients with chronic HCV infection. J. Viral. Hepat 2011, 18, e341–e349. [Google Scholar] [CrossRef]
- De Rosa, S.C.; Cohen, K.W.; Bonaparte, M.; Fu, B.; Garg, S.; Gerard, C.; Goepfert, P.A.; Huang, Y.; Larocque, D.; McElrath, M.J.; et al. Whole-blood cytokine secretion assay as a high-throughput alternative for assessing the cell-mediated immunity profile after two doses of an adjuvanted SARS-CoV-2 recombinant protein vaccine candidate. Clin. Transl. Immunol. 2022, 11, e1360. [Google Scholar] [CrossRef]
- Riou, C.; du Bruyn, E.; Stek, C.; Daroowala, R.; Goliath, R.T.; Abrahams, F.; Said-Hartley, Q.; Allwood, B.W.; Hsiao, N.Y.; Wilkinson, K.A.; et al. Relationship of SARS-CoV-2-specific CD4 response to COVID-19 severity and impact of HIV-1 and tuberculosis coinfection. J. Clin. Investig. 2021, 131, 1–15. [Google Scholar] [CrossRef]
- Sekine, T.; Perez-Potti, A.; Rivera-Ballesteros, O.; Stralin, K.; Gorin, J.B.; Olsson, A.; Llewellyn-Lacey, S.; Kamal, H.; Bogdanovic, G.; Muschiol, S.; et al. Robust T Cell Immunity in Convalescent Individuals with Asymptomatic or Mild COVID-19. Cell 2020, 183, 158–168.e114. [Google Scholar] [CrossRef] [PubMed]
- Naranbhai, V.; Nathan, A.; Kaseke, C.; Berrios, C.; Khatri, A.; Choi, S.; Getz, M.A.; Tano-Menka, R.; Ofoman, O.; Gayton, A.; et al. T cell reactivity to the SARS-CoV-2 Omicron variant is preserved in most but not all individuals. Cell 2022, 185, 1041–1051.e1046. [Google Scholar] [CrossRef] [PubMed]
- Scurr, M.J.; Lippiatt, G.; Capitani, L.; Bentley, K.; Lauder, S.N.; Smart, K.; Somerville, M.S.; Rees, T.; Stanton, R.J.; Gallimore, A.; et al. Magnitude of venous or capillary blood-derived SARS-CoV-2-specific T cell response determines COVID-19 immunity. Nat. Commun. 2022, 13, 5422. [Google Scholar] [CrossRef] [PubMed]
- van den Dijssel, J.; Hagen, R.R.; de Jongh, R.; Steenhuis, M.; Rispens, T.; Geerdes, D.M.; Mok, J.Y.; Kragten, A.H.; Duurland, M.C.; Verstegen, N.J.; et al. Parallel detection of SARS-CoV-2 epitopes reveals dynamic immunodominance profiles of CD8(+) T memory cells in convalescent COVID-19 donors. Clin. Transl. Immunol. 2022, 11, e1423. [Google Scholar] [CrossRef] [PubMed]
- Ishii, H.; Nomura, T.; Yamamoto, H.; Nishizawa, M.; Thu Hau, T.T.; Harada, S.; Seki, S.; Nakamura-Hoshi, M.; Okazaki, M.; Daigen, S.; et al. Neutralizing-antibody-independent SARS-CoV-2 control correlated with intranasal-vaccine-induced CD8(+) T cell responses. Cell Rep. Med. 2022, 3, 100520. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Yamasaki, S. Current understanding of T cell immunity against SARS-CoV-2. Inflamm. Regen 2022, 42, 51. [Google Scholar] [CrossRef]
- Niessl, J.; Sekine, T.; Buggert, M. T cell immunity to SARS-CoV-2. Semin Immunol. 2021, 55, 101505. [Google Scholar] [CrossRef]
- Tan, A.T.; Linster, M.; Tan, C.W.; Le Bert, N.; Chia, W.N.; Kunasegaran, K.; Zhuang, Y.; Tham, C.Y.L.; Chia, A.; Smith, G.J.D.; et al. Early induction of functional SARS-CoV-2-specific T cells associates with rapid viral clearance and mild disease in COVID-19 patients. Cell Rep. 2021, 34, 108728. [Google Scholar] [CrossRef] [PubMed]
- Toor, S.M.; Saleh, R.; Sasidharan Nair, V.; Taha, R.Z.; Elkord, E. T-cell responses and therapies against SARS-CoV-2 infection. Immunology 2021, 162, 30–43. [Google Scholar] [CrossRef]
- McMahan, K.; Yu, J.; Mercado, N.B.; Loos, C.; Tostanoski, L.H.; Chandrashekar, A.; Liu, J.; Peter, L.; Atyeo, C.; Zhu, A.; et al. Correlates of protection against SARS-CoV-2 in rhesus macaques. Nature 2021, 590, 630–634. [Google Scholar] [CrossRef]
- Zhuang, Z.; Lai, X.; Sun, J.; Chen, Z.; Zhang, Z.; Dai, J.; Liu, D.; Li, Y.; Li, F.; Wang, Y.; et al. Mapping and role of T cell response in SARS-CoV-2-infected mice. J. Exp. Med. 2021, 218, 1–13. [Google Scholar] [CrossRef]
- Castaldo, A.; D’Anna, C.; Gelzo, M.; Giannattasio, A.; Maglione, M.; Muzzica, S.; Raia, M.; Scalia, G.; Tripodi, L.; Castaldo, G.; et al. Immunophenotyping of peripheral blood cells allows to discriminate MIS-C and Kawasaki disease. Transl. Med. Commun. 2022, 7, 22. [Google Scholar] [CrossRef] [PubMed]
- Gelzo, M.; Castaldo, A.; Giannattasio, A.; Scalia, G.; Raia, M.; Esposito, M.V.; Maglione, M.; Muzzica, S.; D’Anna, C.; Grieco, M.; et al. MIS-C: A COVID-19-as sociated condition between hypoimmunity and hyperimmunity. Front. Immunol. 2022, 13, 985433. [Google Scholar] [CrossRef]
- Gao, F.; Mallajoysula, V.; Arunachalam, P.S.; van der Ploeg, K.; Manohar, M.; Roltgen, K.; Yang, F.; Wirz, O.; Hoh, R.; Haraguchi, E.; et al. Spheromers reveal robust T cell responses to the Pfizer/BioNTech vaccine and attenuated peripheral CD8(+) T cell responses post SARS-CoV-2 infection. Immunity 2023, 56, 864–878.e864. [Google Scholar] [CrossRef] [PubMed]
- Collaborators, C.-C.I. Estimating global, regional, and national daily and cumulative infections with SARS-CoV-2 through Nov 14, 2021: A statistical analysis. Lancet 2022, 399, 2351–2380. [Google Scholar] [CrossRef]

| Population | |
|---|---|
| Number of subjects included | 53 |
| Median age and range (years) | 63 (38–71) |
| Male/female | 17/36 |
| Vaccination | |
| First dose of vaccine | January 2021 |
| Second dose of vaccine | February 2021 |
| Third dose of vaccine (booster dose) | October 2021 |
| Sampling | |
| First sampling (T1) | March 2021 |
| Second sampling (T2) | September 2021 |
| Third sampling (T3) | November 2021 |
| End of the monitoring | May 2022 |
| Time Period | Alpha (B.1.1.7) | Beta (B.1.351) | Gamma (P.1) | Delta (B.1.617.2) | Eta (B.1.525) | Kappa (B.167.1) | Omicron (B.1.1.529) | Other |
|---|---|---|---|---|---|---|---|---|
| January–March 2021 | 73.0 | 0.83 | 6.0 | 1.0 | 1.0 | 1.2 | 0 | 17 |
| September 2021 | 2.3 | 0.1 | 0.4 | 88.1 | 0.1 | 1.2 | 0.1 | 8 |
| October 2021 | 0.1 | 0.1 | 0.1 | 89.6 | 0 | 0 | 0 | 10 |
| November 2021 | 0.3 | 0 | 0.1 | 91.4 | 0 | 0.9 | 0 | 7 |
| December 2021 | 0.1 | 0 | 0.1 | 86.6 | 0 | 0.9 | 0.1 | 12 |
| January 2022 | 0.1 | 0 | 0 | 79 | 0 | 0 | 21 | |
| February 2022 | 0 | 0 | 0 | 7.1 | 0 | 0 | 71.4 | 22 |
| March 2022 | 0 | 0 | 0 | 0.7 | 0 | 0.1 | 90.9 | 8 |
| April 2022 | 0 | 0 | 0 | 0.2 | 0 | 0 | 89.7 | 10 |
| May 2022 | 0 | 0 | 0 | 0 | 0 | 0 | 93.6 | 6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cortese, P.; Amato, F.; Davino, A.; De Franchis, R.; Esposito, S.; Zollo, I.; Di Domenico, M.; Solito, E.; Zarrilli, F.; Gentile, L.; et al. The Immune Response to SARS-CoV-2 Vaccine in a Cohort of Family Pediatricians from Southern Italy. Cells 2023, 12, 1447. https://doi.org/10.3390/cells12111447
Cortese P, Amato F, Davino A, De Franchis R, Esposito S, Zollo I, Di Domenico M, Solito E, Zarrilli F, Gentile L, et al. The Immune Response to SARS-CoV-2 Vaccine in a Cohort of Family Pediatricians from Southern Italy. Cells. 2023; 12(11):1447. https://doi.org/10.3390/cells12111447
Chicago/Turabian StyleCortese, Paolo, Felice Amato, Antonio Davino, Raffaella De Franchis, Speranza Esposito, Immacolata Zollo, Marina Di Domenico, Egle Solito, Federica Zarrilli, Laura Gentile, and et al. 2023. "The Immune Response to SARS-CoV-2 Vaccine in a Cohort of Family Pediatricians from Southern Italy" Cells 12, no. 11: 1447. https://doi.org/10.3390/cells12111447
APA StyleCortese, P., Amato, F., Davino, A., De Franchis, R., Esposito, S., Zollo, I., Di Domenico, M., Solito, E., Zarrilli, F., Gentile, L., Cernera, G., & Castaldo, G. (2023). The Immune Response to SARS-CoV-2 Vaccine in a Cohort of Family Pediatricians from Southern Italy. Cells, 12(11), 1447. https://doi.org/10.3390/cells12111447