Short-Term Autophagy Preconditioning Upregulates the Expression of COX2 and PGE2 and Alters the Immune Phenotype of Human Adipose-Derived Stem Cells In Vitro
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cells and Cell Culture
2.2. Western Blot Analysis
2.3. Flow Cytometry
2.4. Colony Forming Unit-Fibroblast (CFU-F) Assay
2.5. RNA Isolation and Quantitative Reverse-Transcription PCR (qRT-PCR)
2.6. Enzyme-Linked Immunosorbent Assay (ELISA)
2.7. Statistical Analysis
3. Results
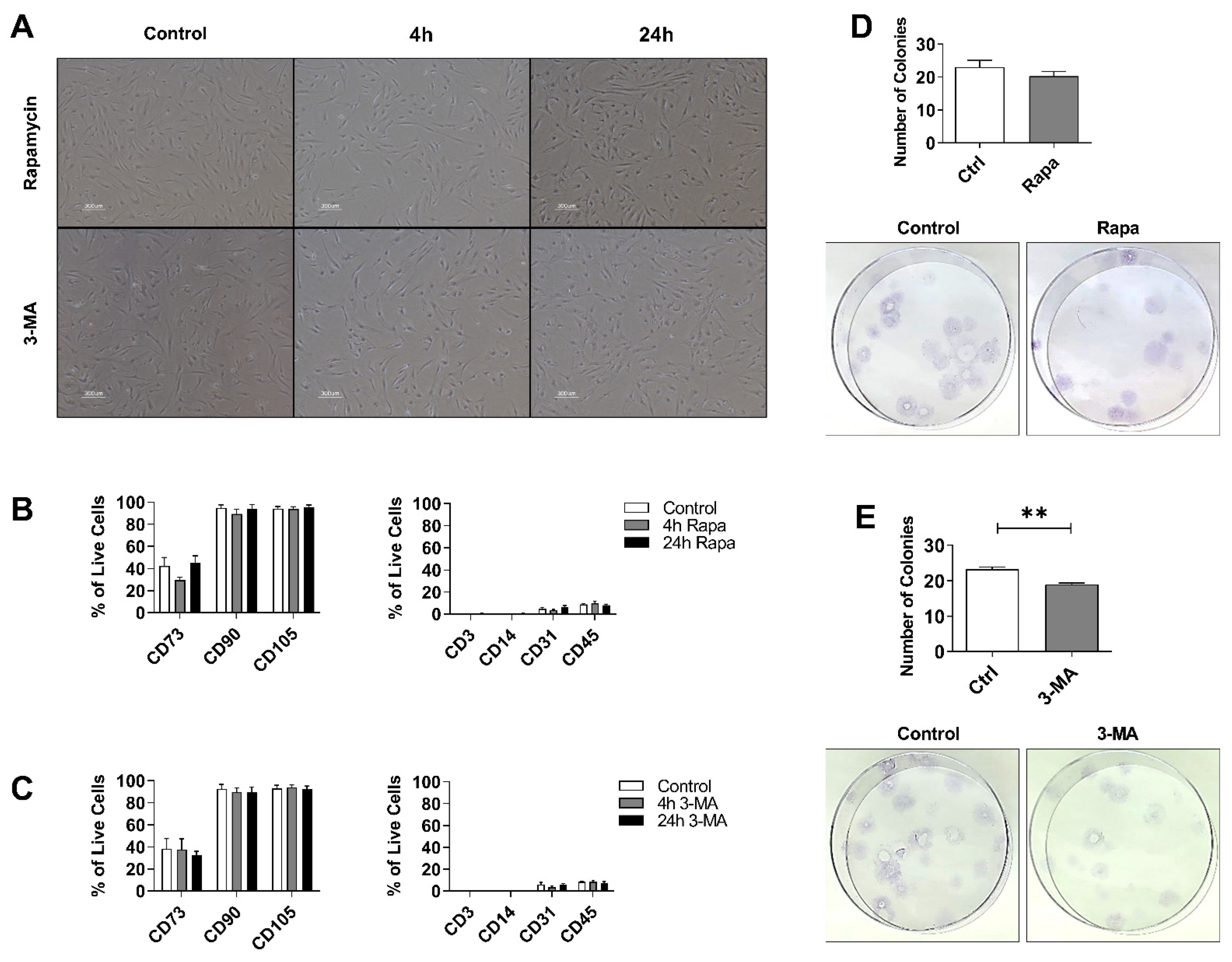
3.1. The mTOR Inhibitor Rapamycin Induces, While the PI3K Inhibitor 3-MA Suppresses, Autophagy in hASCs
3.2. Autophagy Preconditioning Does Not Alter hASCs Stem Cell Properties
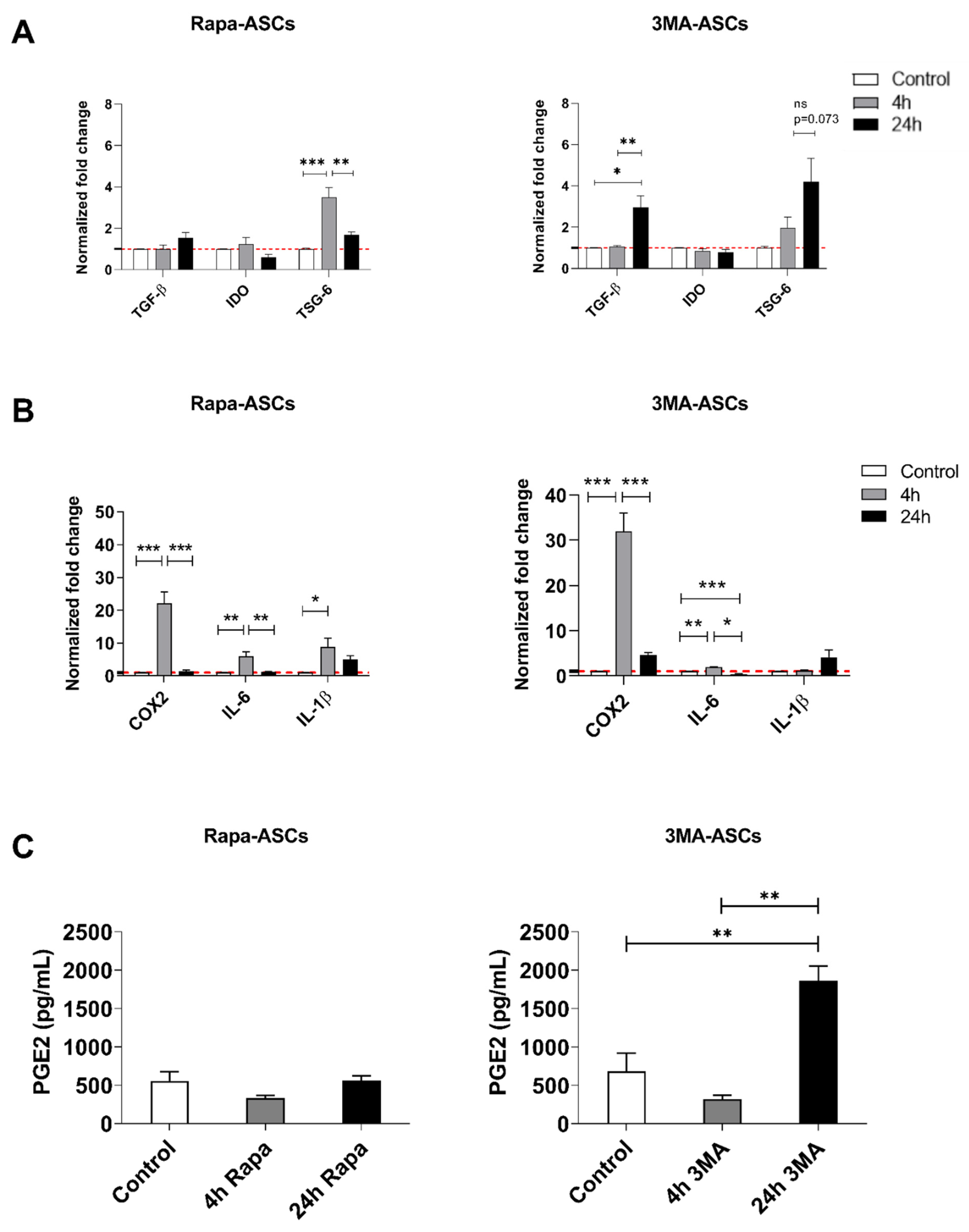
3.3. Autophagy Preconditioning of hASCs Alters Expression of Both Anti-Inflammatory and Pro-Inflammatory Mediators
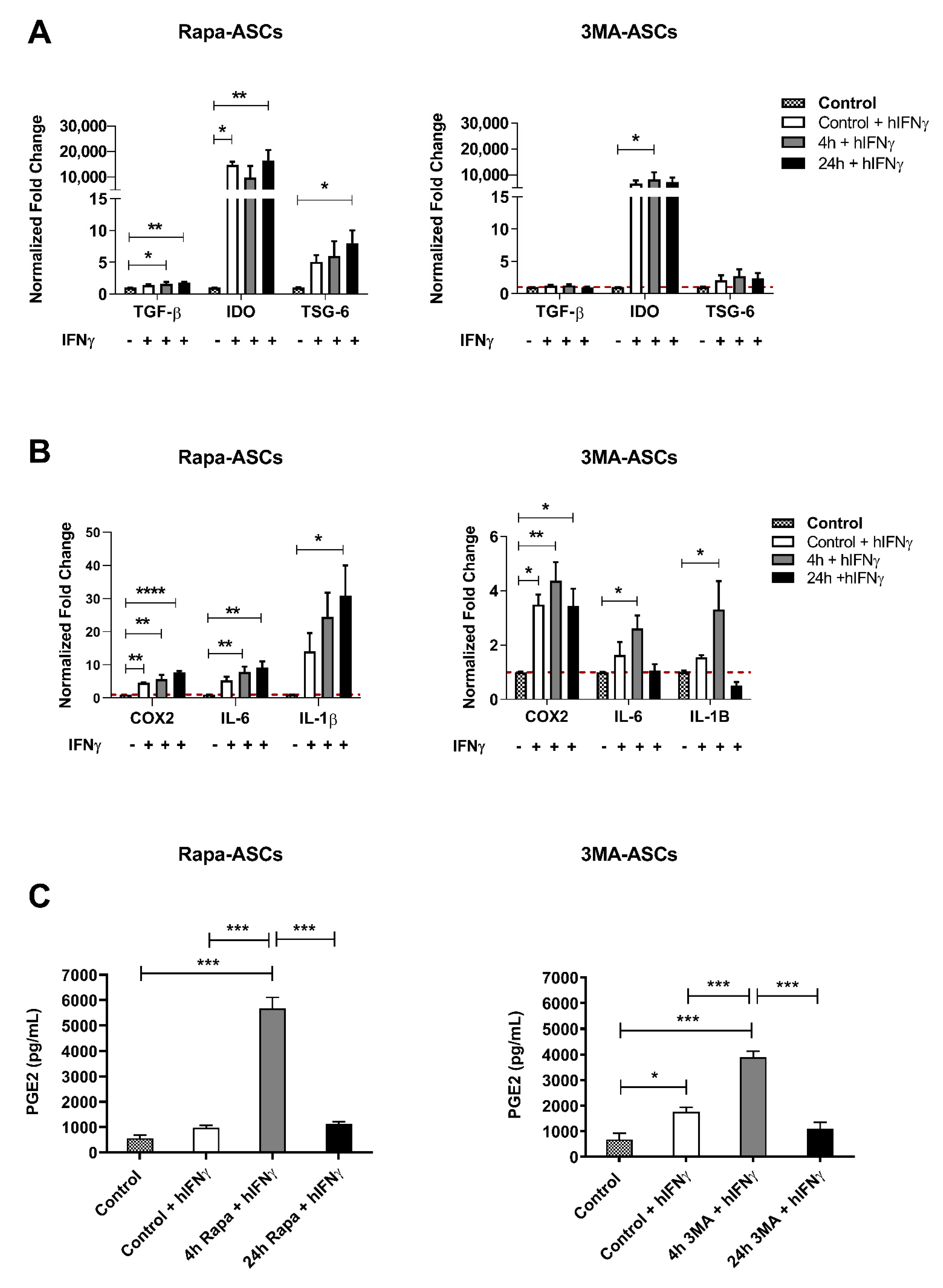
3.4. Autophagy Preconditioning of hASCs Alters Response to Pro-Inflammatory Stimulation with hIFNγ
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zheng, G.; Qiu, G.; Ge, M.; He, J.; Huang, L.; Chen, P.; Wang, W.; Xu, Q.; Hu, Y.; Shu, Q.; et al. Human Adipose-Derived Mesenchymal Stem Cells Alleviate Obliterative Bronchiolitis in a Murine Model via IDO. Respir. Res. 2017, 18, 119. [Google Scholar] [CrossRef] [Green Version]
- Gimble, J.M.; Katz, A.J.; Bunnell, B.A. Adipose-Derived Stem Cells for Regenerative Medicine. Circ. Res. 2007, 100, 1249–1260. [Google Scholar] [CrossRef]
- Melief, S.M.; Zwaginga, J.J.; Fibbe, W.E.; Roelofs, H. Adipose Tissue-Derived Multipotent Stromal Cells Have a Higher Immunomodulatory Capacity Than Their Bone Marrow-Derived Counterparts. Stem Cells Transl. Med. 2013, 2, 455–463. [Google Scholar] [CrossRef]
- Ceccarelli, S.; Pontecorvi, P.; Anastasiadou, E.; Napoli, C.; Marchese, C. Immunomodulatory Effect of Adipose-Derived Stem Cells: The Cutting Edge of Clinical Application. Front. Cell Dev. Biol. 2020, 236. [Google Scholar] [CrossRef] [PubMed]
- Serejo, T.R.T.; Silva-Carvalho, A.É.; Braga, L.D.d.C.F.; Neves, F.d.A.R.; Pereira, R.W.; Carvalho, J.L.d.; Saldanha-Araujo, F. Assessment of the Immunosuppressive Potential of INF-γ Licensed Adipose Mesenchymal Stem Cells, Their Secretome and Extracellular Vesicles. Cells 2019, 8, 22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, D.; Wang, W.; Li, L.; Peng, Y.; Chen, P.; Huang, H.; Guo, Y.; Xia, X.; Wang, Y.; Wang, H.; et al. The Relative Contribution of Paracine Effect versus Direct Differentiation on Adipose-Derived Stem Cell Transplantation Mediated Cardiac Repair. PLoS ONE 2013, 8, e59020. [Google Scholar] [CrossRef]
- Fontanilla, C.V.; Gu, H.; Liu, Q.; Zhu, T.Z.; Zhou, C.; Johnstone, B.H.; March, K.L.; Pascuzzi, R.M.; Farlow, M.R.; Du, Y. Adipose-Derived Stem Cell Conditioned Media Extends Survival Time of a Mouse Model of Amyotrophic Lateral Sclerosis. Sci. Rep. 2015, 5, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Crop, M.J.; Baan, C.C.; Korevaar, S.S.; IJzermans, J.N.M.; Pescatori, M.; Stubbs, A.P.; van IJcken, W.F.J.; Dahlke, M.H.; Eggenhofer, E.; Weimar, W.; et al. Inflammatory Conditions Affect Gene Expression and Function of Human Adipose Tissue-Derived Mesenchymal Stem Cells. Clin. Exp. Immunol. 2010, 162, 474–486. [Google Scholar] [CrossRef]
- Domenis, R.; Cifù, A.; Quaglia, S.; Pistis, C.; Moretti, M.; Vicario, A.; Parodi, P.C.; Fabris, M.; Niazi, K.R.; Soon-Shiong, P.; et al. Pro Inflammatory Stimuli Enhance the Immunosuppressive Functions of Adipose Mesenchymal Stem Cells-Derived Exosomes. Sci. Rep. 2018, 8, 1–11. [Google Scholar] [CrossRef]
- Wobma, H.M.; Tamargo, M.A.; Goeta, S.; Brown, L.M.; Duran-Struuck, R.; Vunjak-Novakovic, G. The Influence of Hypoxia and IFN-γ on the Proteome and Metabolome of Therapeutic Mesenchymal Stem Cells. Biomaterials 2018, 167, 226–234. [Google Scholar] [CrossRef] [PubMed]
- Roemeling-Van Rhijn, M.; Mensah, F.K.F.; Korevaar, S.S.; Leijs, M.J.; Van Osch, G.J.V.M.; IJzermans, J.N.M.; Betjes, M.G.H.; Baan, C.C.; Weimar, W.; Hoogduijn, M.J. Effects of Hypoxia on the Immunomodulatory Properties of Adipose Tissue-Derived Mesenchymal Stem Cells. Front. Immunol. 2013, 4, 203. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seo, Y.; Shin, T.-H.; Kim, H.-S. Molecular Sciences Current Strategies to Enhance Adipose Stem Cell Function: An Update. Int. J. Mol. Sci. 2019, 20, 3827. [Google Scholar] [CrossRef] [Green Version]
- Hsiao, S.T.; Lokmic, Z.; Peshavariya, H.; Abberton, K.M.; Dusting, G.J.; Lim, S.Y.; Dilley, R.J. Hypoxic Conditioning Enhances the Angiogenic Paracrine Activity of Human Adipose-Derived Stem Cells. Stem Cells Dev. 2013, 22, 1614–1623. [Google Scholar] [CrossRef] [Green Version]
- Németh, K.; Leelahavanichkul, A.; Yuen, P.S.T.; Mayer, B.; Parmelee, A.; Doi, K.; Robey, P.G.; Leelahavanichkul, K.; Koller, B.H.; Brown, J.M.; et al. Bone Marrow Stromal Cells Attenuate Sepsis via Prostaglandin E 2-Dependent Reprogramming of Host Macrophages to Increase Their Interleukin-10 Production. Nat. Med. 2009, 15, 42–49. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, L.; He, Y.R.; Liu, S.J.; Hu, L.; Liang, L.C.; Liu, D.L.; Liu, L.; Zhu, Z.Q. Enhanced Effect of IL-1 β-Activated Adipose-Derived MSCs (ADMSCs) on Repair of Intestinal Ischemia-Reperfusion Injury via COX-2-PGE 2 Signaling. Stem Cells Int. 2020, 2020, 2803747. [Google Scholar] [CrossRef] [Green Version]
- Bouffi, C.; Bony, C.; Courties, G.; Jorgensen, C.; Noël, D. IL-6-Dependent PGE2 Secretion by Mesenchymal Stem Cells Inhibits Local Inflammation in Experimental Arthritis. PLoS ONE 2010, 5, e14247. [Google Scholar] [CrossRef]
- González, M.A.; González-Rey, E.; Rico, L.; Büscher, D.; Delgado, M. Treatment of Experimental Arthritis by Inducing Immune Tolerance with Human Adipose-Derived Mesenchymal Stem Cells. Arthritis Rheum. 2009, 60, 1006–1019. [Google Scholar] [CrossRef]
- Matysiak, M.; Orlowski, W.; Fortak-Michalska, M.; Jurewicz, A.; Selmaj, K. Immunoregulatory Function of Bone Marrow Mesenchymal Stem Cells in EAE Depends on Their Differentiation State and Secretion of PGE2. J. Neuroimmunol. 2011, 233, 106–111. [Google Scholar] [CrossRef] [PubMed]
- Dang, S.; Xu, H.; Xu, C.; Cai, W.; Li, Q.; Cheng, Y.; Jin, M.; Wang, R.-X.; Peng, Y.; Zhang, Y.; et al. Autophagy Regulates the Therapeutic Potential of Mesenchymal Stem Cells in Experimental Autoimmune Encephalomyelitis. Autophagy 2014, 10, 1301–1315. [Google Scholar] [CrossRef] [Green Version]
- Yang, F.Y.; Chen, R.; Zhang, X.; Huang, B.; Tsang, L.L.; Li, X.; Jiang, X. Preconditioning Enhances the Therapeutic Effects of Mesenchymal Stem Cells on Colitis Through PGE2-Mediated T-Cell Modulation. Cell Transplant. 2018, 27, 1352–1367. [Google Scholar] [CrossRef] [Green Version]
- Park, H.J.; Kim, J.; Saima, F.T.; Rhee, K.J.; Hwang, S.; Kim, M.Y.; Baik, S.K.; Eom, Y.W.; Kim, H.S. Adipose-Derived Stem Cells Ameliorate Colitis by Suppression of Inflammasome Formation and Regulation of M1-Macrophage Population through Prostaglandin E2. Biochem. Biophys. Res. Commun. 2018, 498, 988–995. [Google Scholar] [CrossRef]
- Ceccariglia, S.; Cargnoni, A.; Silini, A.R.; Parolini, O. Autophagy: A Potential Key Contributor to the Therapeutic Action of Mesenchymal Stem Cells. Autophagy 2019, 16, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Jakovljevic, J.; Harrell, C.R.; Fellabaum, C.; Arsenijevic, A.; Jovicic, N.; Volarevic, V. Modulation of Autophagy as New Approach in Mesenchymal Stem Cell-Based Therapy. Biomed. Pharmacother. 2018, 104, 404–410. [Google Scholar] [CrossRef]
- Glick, D.; Barth, S.; Macleod, K.F. Autophagy: Cellular and Molecular Mechanisms. J. Pathol. 2010, 221, 3–12. [Google Scholar] [CrossRef] [Green Version]
- Chen, X.; He, Y.; Lu, F. Autophagy in Stem Cell Biology: A Perspective on Stem Cell Self-Renewal and Differentiation. Stem Cells Int. 2018, 2018, 9131397. [Google Scholar] [CrossRef]
- Weiss, D.J.; English, K.; Krasnodembskaya, A.; Isaza-Correa, J.M.; Hawthorne, I.J.; Mahon, B.P. The Necrobiology of Mesenchymal Stromal Cells Affects Therapeutic Efficacy. Front. Immunol. 2019, 10, 1228. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Kim, S.G.; Blenis, J. Rapamycin: One Drug, Many Effects. Cell Metab. 2014, 19, 373–379. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pyo, J.O.; Nah, J.; Jung, Y.K. Molecules and Their Functions in Autophagy. Exp. Mol. Med. 2012, 44, 73–80. [Google Scholar] [CrossRef] [Green Version]
- Kim, K.-W.; Moon, S.-J.; Park, M.-J.; Kim, B.-M.; Kim, E.-K.; Lee, S.-H.; Lee, E.-J.; Chung, B.-H.; Yang, C.-W.; Cho, M.-L. Optimization of Adipose Tissue-Derived Mesenchymal Stem Cells by Rapamycin in a Murine Model of Acute Graft-versus-Host Disease. Stem Cell Res. Ther. 2015, 6, 202. [Google Scholar] [CrossRef] [Green Version]
- Javorkova, E.; Vackova, J.; Hajkova, M.; Hermankova, B.; Zajicova, A.; Holan, V.; Krulova, M. The Effect of Clinically Relevant Doses of Immunosuppressive Drugs on Human Mesenchymal Stem Cells. Biomed. Pharmacother. 2018, 97, 402–411. [Google Scholar] [CrossRef]
- Li, C.; Ye, L.; Yang, L.; Yu, X.; He, Y.; Chen, Z.; Li, L.; Zhang, D. Rapamycin Promotes the Survival and Adipogenesis of Ischemia-Challenged Adipose Derived Stem Cells by Improving Autophagy. Cell. Physiol. Biochem. 2017, 44, 1762–1774. [Google Scholar] [CrossRef]
- Strong, A.L.; Bowles, A.C.; Wise, R.M.; Morand, J.P.; Dutreil, M.F.; Gimble, J.M.; Bunnell, B.A. Human Adipose Stromal/Stem Cells from Obese Donors Show Reduced Efficacy in Halting Disease Progression in the Experimental Autoimmune Encephalomyelitis Model of Multiple Sclerosis. Stem Cells 2016, 34, 614–626. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Al-Ghadban, S.; Diaz, Z.T.; Singer, H.J.; Mert, K.B.; Bunnell, B.A. Increase in Leptin and PPAR-γ Gene Expression in Lipedema Adipocytes Differentiated in Vitro from Adipose-Derived Stem Cells. Cells 2020, 9, 430. [Google Scholar] [CrossRef] [Green Version]
- Sabol, R.A.; Villela, V.A.; Denys, A.; Freeman, B.T.; Hartono, A.B.; Wise, R.M.; Harrison, M.A.A.; Sandler, M.B.; Hossain, F.; Miele, L.; et al. Obesity-Altered Adipose Stem Cells Promote Radiation Resistance of Estrogen Receptor Positive Breast Cancer through Paracrine Signaling. Int. J. Mol. Sci. 2020, 21, 2722. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Strong, A.L.; Hunter, R.S.; Jones, R.B.; Bowles, A.C.; Dutreil, M.F.; Gaupp, D.; Hayes, D.J.; Gimble, J.M.; Levi, B.; McNulty, M.A.; et al. Obesity Inhibits the Osteogenic Differentiation of Human Adipose-Derived Stem Cells. J. Transl. Med. 2016, 14, 27. [Google Scholar] [CrossRef] [Green Version]
- Scruggs, B.A.; Semon, J.A.; Zhang, X.; Zhang, S.; Bowles, A.C.; Pandey, A.C.; Imhof, K.M.P.; Kalueff, A.V.; Gimble, J.M.; Bunnell, B.A. Age of the Donor Reduces the Ability of Human Adipose-Derived Stem Cells to Alleviate Symptoms in the Experimental Autoimmune Encephalomyelitis Mo. Stem Cells Transl. Med. 2013, 2, 797–807. [Google Scholar] [CrossRef]
- Wu, Y.-T.; Tan, H.-L.; Shui, G.; Bauvy, C.; Huang, Q.; Wenk, M.R.; Ong, C.-N.; Codogno, P.; Shen, H.-M. Dual Role of 3-Methyladenine in Modulation of Autophagy via Different Temporal Patterns of Inhibition on Class I and III Phosphoinositide 3-Kinase. J. Biol. Chem. 2010, 285, 10850–10861. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Uccelli, A.; Moretta, L.; Pistoia, V. Mesenchymal Stem Cells in Health and Disease. Nat. Rev. Immunol. 2008, 8, 726–736. [Google Scholar] [CrossRef]
- Patrikoski, M.; Mannerström, B.; Miettinen, S. Perspectives for Clinical Translation of Adipose Stromal/Stem Cells. Stem Cells Int. 2019, 2019, 5858247. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wise, R.M.; Harrison, M.A.A.; Sullivan, B.N.; Al-Ghadban, S.; Aleman, S.J.; Vinluan, A.T.; Monaco, E.R.; Donato, U.M.; Pursell, I.A.; Bunnell, B.A. Short-Term Rapamycin Preconditioning Diminishes Therapeutic Efficacy of Human Adipose-Derived Stem Cells in a Murine Model of Multiple Sclerosis. Cells 2020, 9, 2218. [Google Scholar] [CrossRef]
- Li, Z.; Wang, Y.; Wang, H.; Wu, J.; Tan, Y. Rapamycin-Preactivated Autophagy Enhances Survival and Differentiation of Mesenchymal Stem Cells After Transplantation into Infarcted Myocardium. Stem Cell Rev. Rep. 2020, 16, 344–356. [Google Scholar] [CrossRef] [Green Version]
- Zheng, J.; Li, H.; He, L.; Huang, Y.; Cai, J.; Chen, L.; Zhou, C.; Fu, H.; Lu, T.; Zhang, Y.; et al. Preconditioning of Umbilical Cord-derived Mesenchymal Stem Cells by Rapamycin Increases Cell Migration and Ameliorates Liver Ischaemia/Reperfusion Injury in Mice via the CXCR4/CXCL12 Axis. Cell Prolif. 2019, 52, 12546. [Google Scholar] [CrossRef]
- Sarbassov, D.D.; Ali, S.M.; Sengupta, S.; Sheen, J.H.; Hsu, P.P.; Bagley, A.F.; Markhard, A.L.; Sabatini, D.M. Prolonged Rapamycin Treatment Inhibits MTORC2 Assembly and Akt/PKB. Mol. Cell 2006, 22, 159–168. [Google Scholar] [CrossRef]
- Najar, M.; Raicevic, G.; Boufker, H.I.; Kazan, H.F.; De Bruyn, C.; Meuleman, N.; Bron, D.; Toungouz, M.; Lagneaux, L. Mesenchymal Stromal Cells Use PGE2 to Modulate Activation and Proliferation of Lymphocyte Subsets: Combined Comparison of Adipose Tissue, Wharton’s Jelly and Bone Marrow Sources. Cell. Immunol. 2010, 264, 171–179. [Google Scholar] [CrossRef]
- Yañez, R.; Oviedo, A.; Aldea, M.; Bueren, J.A.; Lamana, M.L. Prostaglandin E2 Plays a Key Role in the Immunosuppressive Properties of Adipose and Bone Marrow Tissue-Derived Mesenchymal Stromal Cells. Exp. Cell Res. 2010, 316, 3109–3123. [Google Scholar] [CrossRef] [PubMed]
- Puissant, B.; Barreau, C.; Bourin, P.; Clavel, C.; Corre, J.; Bousquet, C.; Taureau, C.; Cousin, B.; Abbal, M.; Laharrague, P.; et al. Immunomodulatory Effect of Human Adipose Tissue-Derived Adult Stem Cells: Comparison with Bone Marrow Mesenchymal Stem Cells. Br. J. Haematol. 2005, 129, 118–129. [Google Scholar] [CrossRef]
- Cui, L.; Shuo, Y.; Liu, W.; Li, N.; Zhang, W.; Cao, Y. Expanded Adipose-Derived Stem Cells Suppress Mixed Lymphocyte Reaction by Secretion of Prostaglandin E2. Tissue Eng. 2007, 13, 1185–1195. [Google Scholar] [CrossRef]
- Manferdini, C.; Paolella, F.; Gabusi, E.; Gambari, L.; Piacentini, A.; Filardo, G.; Fleury-Cappellesso, S.; Barbero, A.; Murphy, M.; Lisignoli, G. Adipose Stromal Cells Mediated Switching of the Pro-Inflammatory Profile of M1-like Macrophages Is Facilitated by PGE2: In Vitro Evaluation. Osteoarthr. Cartil. 2017, 25, 1161–1171. [Google Scholar] [CrossRef] [Green Version]
- Spaggiari, G.M.; Capobianco, A.; Abdelrazik, H.; Becchetti, F.; Mingari, M.C.; Moretta, L. Mesenchymal Stem Cells Inhibit Natural Killer-Cell Proliferation, Cytotoxicity, and Cytokine Production: Role of Indoleamine 2,3-Dioxygenase and Prostaglandin E2. Blood 2008, 111, 1327–1333. [Google Scholar] [CrossRef] [PubMed]
- Spaggiari, G.M.; Abdelrazik, H.; Becchetti, F.; Moretta, L. MSCs Inhibit Monocyte-Derived DC Maturation and Function by Selectively Interfering with the Generation of Immature DCs: Central Role of MSC-Derived Prostaglandin E2. Blood 2009, 113, 6576–6583. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Gao, L.; Zang, D. Elevated Levels of IFN-γ in CSF and Serum of Patients with Amyotrophic Lateral Sclerosis. PLoS ONE 2015, 10, e0136937. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kallaur, A.P.; Oliveira, S.R.; Simao, A.N.C.; De Almeida, E.R.D.; Morimoto, H.K.; Lopes, J.; De Carvalho Jennings Pereira, W.L.; Andrade, R.M.; Pelegrino, L.M.; Borelli, S.D.; et al. Cytokine Profile in Relapsing-Remitting Multiple Sclerosis Patients and the Association between Progression and Activity of the Disease. Mol. Med. Rep. 2013, 7, 1010–1020. [Google Scholar] [CrossRef] [Green Version]
- Oke, V.; Gunnarsson, I.; Dorschner, J.; Eketjäll, S.; Zickert, A.; Niewold, T.B.; Svenungsson, E. High Levels of Circulating Interferons Type I, Type II and Type III Associate with Distinct Clinical Features of Active Systemic Lupus Erythematosus. Arthritis Res. Ther. 2019, 21, 107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Belkhelfa, M.; Rafa, H.; Medjeber, O.; Arroul-Lammali, A.; Behairi, N.; Abada-Bendib, M.; Makrelouf, M.; Belarbi, S.; Masmoudi, A.N.; Tazir, M.; et al. IFN-γ and TNF-α Are Involved during Alzheimer Disease Progression and Correlate with Nitric Oxide Production: A Study in Algerian Patients. J. Interf. Cytokine Res. 2014, 34, 839–847. [Google Scholar] [CrossRef]
- Wang, B.; Lin, Y.; Hu, Y.; Shan, W.; Liu, S.; Xu, Y.; Zhang, H.; Cai, S.; Yu, X.; Cai, Z.; et al. MTOR Inhibition Improves the Immunomodulatory Properties of Human Bone Marrow Mesenchymal Stem Cells by Inducing COX-2 and PGE2. Stem Cell Res. Ther. 2017, 8, 292. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, Y.-C.; Kuo, H.-C.; Wang, J.-S.; Lin, W.-W. Regulation of Inflammatory Response by 3-Methyladenine Involves the Coordinative Actions on Akt and Glycogen Synthase Kinase 3β Rather than Autophagy. J. Immunol. 2012, 189, 4154–4164. [Google Scholar] [CrossRef] [Green Version]
- Saxton, R.A.; Sabatini, D.M. MTOR Signaling in Growth, Metabolism, and Disease. Cell 2017, 168, 960. [Google Scholar] [CrossRef] [Green Version]
- Banaszynski, L.A.; Liu, C.W.; Wandless, T.J. Characterization of the FKBP-Rapamycin-FRB Ternary Complex. J. Am. Chem. Soc. 2005, 127, 4715–4721. [Google Scholar] [CrossRef]
- Zhang, J.; Xu, K.; Liu, P.; Geng, Y.; Wang, B.; Gan, W.; Guo, J.; Wu, F.; Chin, Y.R.; Berrios, C.; et al. Inhibition of Rb Phosphorylation Leads to MTORC2-Mediated Activation of Akt. Mol. Cell 2016, 62, 929–942. [Google Scholar] [CrossRef] [Green Version]
- Wang, Q.; Zhou, Y.; Wang, X.; Evers, B.M. Glycogen Synthase Kinase-3 Is a Negative Regulator of Extracellular Signal-Regulated Kinase. Oncogene 2005, 25, 43–50. [Google Scholar] [CrossRef] [Green Version]
- Chinnadurai, R.; Copland, I.B.; Ng, S.; Garcia, M.; Prasad, M.; Arafat, D.; Gibson, G.; Kugathasan, S.; Galipeau, J. Mesenchymal Stromal Cells Derived from Crohn’s Patients Deploy Indoleamine 2,3-Dioxygenase-Mediated Immune Suppression, Independent of Autophagy. Mol. Ther. 2015, 23, 1248–1261. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Delarosa, O.; Lombardo, E.; Beraza, A.; Mancheño-Corvo, P.; Ramirez, C.; Menta, R.; Rico, L.; Camarillo, E.; García, L.; Abad, J.L.; et al. Requirement of IFN-γ-Mediated Indoleamine 2,3-Dioxygenase Expression in the Modulation of Lymphocyte Proliferation by Human Adipose-Derived Stem Cells. Tissue Eng. Part A 2009, 15, 2795–2806. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Sun, L.; Huang, C.; Chen, B.; Zhou, Z. Induction of Macrophage M2b/c Polarization by Adipose Tissue-Derived Mesenchymal Stem Cells. J. Immunol. Res. 2019, 2019, 7059680. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- DelaRosa, O.; Sánchez-Correa, B.; Morgado, S.; Ramírez, C.; Del Río, B.; Menta, R.; Lombardo, E.; Tarazona, R.; Casado, J.G. Human Adipose-Derived Stem Cells Impair Natural Killer Cell Function and Exhibit Low Susceptibility to Natural Killer-Mediated Lysis. Stem Cells Dev. 2012, 21, 1333–1343. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, W.J.; Li, Q.; Ryu, M.O.; Ahn, J.O.; Ha Bhang, D.; Chan Jung, Y.; Youn, H.Y. TSG-6 Secreted by Human Adipose Tissue-Derived Mesenchymal Stem Cells Ameliorates DSS-Induced Colitis by Inducing M2 Macrophage Polarization in Mice. Sci. Rep. 2017, 7, 5187. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Song, W.J.; Ryu, M.O.; Nam, A.; An, J.H.; Ahn, J.O.; Bhang, D.H.; Jung, Y.C.; Youn, H.Y. TSG-6 Secreted by Human Adipose Tissue-Derived Mesenchymal Stem Cells Ameliorates Severe Acute Pancreatitis via ER Stress Downregulation in Mice. Stem Cell Res. Ther. 2018, 9, 255. [Google Scholar] [CrossRef]
- Romano, B.; Elangovan, S.; Erreni, M.; Sala, E.; Petti, L.; Kunderfranco, P.; Massimino, L.; Restelli, S.; Sinha, S.; Lucchetti, D.; et al. TNF-Stimulated Gene-6 Is a Key Regulator in Switching Stemness and Biological Properties of Mesenchymal Stem Cells. Stem Cells 2019, 37, 973–987. [Google Scholar] [CrossRef]
- Wang, Y.C.; Chen, R.F.; Brandacher, G.; Lee, W.P.A.; Kuo, Y.R. The Suppression Effect of Dendritic Cells Maturation by Adipose-Derived Stem Cells through TGF-Β1 Related Pathway. Exp. Cell Res. 2018, 370, 708–717. [Google Scholar] [CrossRef]
- English, K.; Ryan, J.M.; Tobin, L.; Murphy, M.J.; Barry, F.P.; Mahon, B.P. Cell Contact, Prostaglandin E2 and Transforming Growth Factor Beta 1 Play Non-Redundant Roles in Human Mesenchymal Stem Cell Induction of CD4+CD25Highforkhead Box P3+ Regulatory T Cells. Clin. Exp. Immunol. 2009, 156, 149–160. [Google Scholar] [CrossRef] [Green Version]
- Gao, L.; Cen, S.; Wang, P.; Xie, Z.; Liu, Z.; Deng, W.; Su, H.; Wu, X.; Wang, S.; Li, J.; et al. Autophagy Improves the Immunosuppression of CD4+ T Cells by Mesenchymal Stem Cells Through Transforming Growth Factor-Β1. Stem Cells Transl. Med. 2016, 5, 1496. [Google Scholar] [CrossRef] [Green Version]
- Scheller, J.; Chalaris, A.; Schmidt-Arras, D.; Rose-John, S. The Pro- and Anti-Inflammatory Properties of the Cytokine Interleukin-6. Biochim. Biophys. Acta Mol. Cell Res. 2011, 1813, 878–888. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schett, G. Physiological Effects of Modulating the Interleukin-6 Axis. Rheumatology 2018, 57 (Suppl. S2), ii43–ii50. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vasilev, G.; Ivanova, M.; Ivanova-Todorova, E.; Tumangelova-Yuzeir, K.; Krasimirova, E.; Stoilov, R.; Kyurkchiev, D. Secretory Factors Produced by Adipose Mesenchymal Stem Cells Downregulate Th17 and Increase Treg Cells in Peripheral Blood Mononuclear Cells from Rheumatoid Arthritis Patients. Rheumatol. Int. 2019, 39, 819–826. [Google Scholar] [CrossRef] [PubMed]
- Ivanova-Todorova, E.; Bochev, I.; Dimitrov, R.; Belemezova, K.; Mourdjeva, M.; Kyurkchiev, S.; Kinov, P.; Altankova, I.; Kyurkchiev, D. Conditioned Medium from Adipose Tissue-Derived Mesenchymal Stem Cells Induces CD4+FOXP3+ Cells and Increases IL-10 Secretion. J. Biomed. Biotechnol. 2012, 2012, 295167. [Google Scholar] [CrossRef]
- Pilny, E.; Smolarczyk, R.; Jarosz-Biej, M.; Hadyk, A.; Skorupa, A.; Ciszek, M.; Krakowczyk, Ł.; Kułach, N.; Gillner, D.; Sokół, M.; et al. Human ADSC Xenograft through IL-6 Secretion Activates M2 Macrophages Responsible for the Repair of Damaged Muscle Tissue. Stem Cell Res. Ther. 2019, 10, 93. [Google Scholar] [CrossRef]
- Heo, S.C.; Jeon, E.S.; Lee, I.H.; Kim, H.S.; Kim, M.B.; Kim, J.H. Tumor Necrosis Factor-α-Activated Human Adipose Tissue-Derived Mesenchymal Stem Cells Accelerate Cutaneous Wound Healing through Paracrine Mechanisms. J. Invest. Dermatol. 2011, 131, 1559–1567. [Google Scholar] [CrossRef] [Green Version]
- Sempere, J.M.; Martinez-Peinado, P.; Arribas, M.I.; Reig, J.A.; De La Sen, M.L.; Zubcoff, J.J.; Fraga, M.F.; Fernández, A.F.; Santana, A.; Roche, E. Single Cell-Derived Clones from Human Adipose Stem Cells Present Different Immunomodulatory Properties. Clin. Exp. Immunol. 2014, 176, 255–265. [Google Scholar] [CrossRef] [Green Version]
- Girdlestone, J.; Pido-Lopez, J.; Srivastava, S.; Chai, J.; Leaver, N.; Galleu, A.; Lombardi, G.; Navarrete, C.V. Enhancement of the Immunoregulatory Potency of Mesenchymal Stromal Cells by Treatment with Immunosuppressive Drugs. Cytotherapy 2015, 17, 1188–1199. [Google Scholar] [CrossRef] [Green Version]
| Donor | Age | BMI |
|---|---|---|
| 1 | 34 | 20.34 |
| 2 | 40 | 21.18 |
| 3 | 39 | 23.4 |
| 4 | 25 | 22.0 |
| 5 | 40 | 21.19 |
| Average ± SD | 36.5 ± 2.87 | 21.62 ± 0.52 |
| Gene | Forward (5′-3′) | Reverse (5′-3′) |
|---|---|---|
| Beta-actin | ACGTTGCTATCCAGGCTGTGCTAT | TTAATGTCACGCACGATTTCCCGC |
| ATG7 | ATGATCCCTGTAACTTAGCCCA | CACGGAAGCAAACAACTTCAAC |
| LC3B | AAGGCGCTTACAGCTCAATG | CTGGGAGGCATAGACCATGT |
| P62 | GCACCCCAATGTGATCTGC | CGCTACACAAGTCGTAGTCTGG |
| TGF-β | CAGTCACCATAGCAACACTC | CCTGGCCTGAACTACTATCT |
| IDO | TCTCATTTCGTGATGGAGACTGC | GTGTCCCGTTCTTGCATTTGC |
| TSG-6 | AGAATTTGTGAGCAGCCCCT | GGCTGCTCGTTCAAGCCATA |
| IL-1β | CATGGGATAACGAGGCTTATG | CCACTTGTTGCTCCATATCC |
| IL-6 | CCTTCCAAAGATGGCTGAAA | TGGCTTGTTCCTCACTACT |
| COX2 | TTGCTGGCAGGGTTGCTGGTGGTA | CATCTGCCTGCTCTGGTCAATCGAA |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wise, R.M.; Al-Ghadban, S.; Harrison, M.A.A.; Sullivan, B.N.; Monaco, E.R.; Aleman, S.J.; Donato, U.M.; Bunnell, B.A. Short-Term Autophagy Preconditioning Upregulates the Expression of COX2 and PGE2 and Alters the Immune Phenotype of Human Adipose-Derived Stem Cells In Vitro. Cells 2022, 11, 1376. https://doi.org/10.3390/cells11091376
Wise RM, Al-Ghadban S, Harrison MAA, Sullivan BN, Monaco ER, Aleman SJ, Donato UM, Bunnell BA. Short-Term Autophagy Preconditioning Upregulates the Expression of COX2 and PGE2 and Alters the Immune Phenotype of Human Adipose-Derived Stem Cells In Vitro. Cells. 2022; 11(9):1376. https://doi.org/10.3390/cells11091376
Chicago/Turabian StyleWise, Rachel M., Sara Al-Ghadban, Mark A. A. Harrison, Brianne N. Sullivan, Emily R. Monaco, Sarah J. Aleman, Umberto M. Donato, and Bruce A. Bunnell. 2022. "Short-Term Autophagy Preconditioning Upregulates the Expression of COX2 and PGE2 and Alters the Immune Phenotype of Human Adipose-Derived Stem Cells In Vitro" Cells 11, no. 9: 1376. https://doi.org/10.3390/cells11091376
APA StyleWise, R. M., Al-Ghadban, S., Harrison, M. A. A., Sullivan, B. N., Monaco, E. R., Aleman, S. J., Donato, U. M., & Bunnell, B. A. (2022). Short-Term Autophagy Preconditioning Upregulates the Expression of COX2 and PGE2 and Alters the Immune Phenotype of Human Adipose-Derived Stem Cells In Vitro. Cells, 11(9), 1376. https://doi.org/10.3390/cells11091376







