Role of Plant-Derived Active Constituents in Cancer Treatment and Their Mechanisms of Action
Abstract
1. Introduction
2. Methodology
Data Collection
3. Data Analysis
3.1. Important Anticancer Phytochemicals from the Clinical Trials and Their Structure–Activity Relationship Data
3.2. Curcumin
3.3. Epigallocatechin Gallate (EGCG)
3.4. Genistein
3.5. Lycopene
3.6. Resveratrol
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AIF | Apoptosis-inducing factor | MUC1-C | Mucin 1, cell surface associated protein |
| Apaf-1 | Apoptotic protease activating factor 1 | NAF-1 | Nuclear assembly factor 1 |
| ATF4 | Activating transcription factor 4 | NAG-1 | NSAID activated gene 1 |
| Bcl-XL | B-cell lymphoma-extra large | NBR1 | Neighbor of BRCA1 gene 1 |
| CCL2 | Chemokine (C-C motif) ligand 2 | Nrf2 | Nuclear factor erythroid 2–related factor 2 |
| CDK | Cyclin-dependent kinases | PD-L1 | Programmed death-ligand 1 |
| CHOP | C/EBP homologous protein | PKM2 | Pyruvate kinase M2 |
| CREB | cAMP-response element binding protein | PLK1 | Polo-like kinase 1 |
| CXCR4 | C-X-C chemokine receptor type 4 | PPARγ | Peroxisome proliferator- activated receptor gamma |
| DR5 | Death receptor 5 | PTEN | Phosphatase and tensin homolog deleted in chromosome 10 |
| ER | Endoplasmic reticulum | Raf | Rapidly accelerated aibrosarcoma |
| FAK | Focal adhesion kinase | RASSF6 | Ras-association domain family |
| FOXA2 | Forkhead box protein A2 | RHAMM | HMMR hyaluronan-mediated motility receptor |
| GADD45B | Growth arrest and DNA-damage-inducible, beta protein | RhoA | Ras-homolog family member A |
| GLUT1 | Glucose transporter 1 | RIP1 | Receptor interacting protein 1 |
| H2AX | H2A histone family member X | ROCK1 | Rho-associated protein kinase 1 |
| HIF-2α | Hypoxia inducible factor 2 alpha | ROS | Reactive oxygen species |
| HMGB1 | High mobility group box 1 protein | SGK1 | Serum/glucocorticoid regulated kinase 1 |
| HOXD3 | Homeobox D3 | Skp2 | S-phase kinase associated protein 2 |
| HSP90 | Heat shock protein 90 | TASK-3 | Two-pore-domain acid sensitive K+ channel 3 TASK-3 |
| hTERT | Human telomerase reverse transcriptase | TGF-β1 | Transforming growth factor-beta1 |
| iNOS | Inducible nitric oxide synthase | TNF-α | Tumor necrosis factor alpha |
| IκBα | IkappaB alpha | Top1 | Topoisomerase 1 |
| IκK-α | Inhibitory-κB kinase alpha | TRAIL | TNF-related apoptosis-inducing ligand |
| JNK | Jun N-terminal kinase | TRIM16 | Tripartite motif-containing protein 16 |
| Keap1 | Kelch-like ECH-associated protein 1 | uPA | Urokinase-type plasminogen activator |
| LOX | Lysyl oxidase | USP14 | Ubiquitin specific peptidase 14 |
| MEK | MAPK/ERK kinase | Wnt | Wingless-related integration site |
| mTOR | Mammalian target of rapamycin | XIAP | X-linked inhibitor of apoptosis protein |
References
- World Health Organization. International Agency for Research on Cancer; World Health Organization: Geneva, Switzerland, 2019. [Google Scholar]
- Cao, W.; Chen, H.-D.; Yu, Y.-W.; Li, N.; Chen, W.-Q. Changing profiles of cancer burden worldwide and in China: A secondary analysis of the global cancer statistics 2020. Chin. Med. J. 2021, 134, 783. [Google Scholar] [CrossRef] [PubMed]
- Martin, T.A.; Ye, L.; Sanders, A.J.; Lane, J.; Jiang, W.G. Cancer invasion and metastasis: Molecular and cellular perspective. In Madame Curie Bioscience Database [Internet]; Landes Bioscience: Austin, TX, USA, 2013. [Google Scholar]
- Garcia-Oliveira, P.; Otero, P.; Pereira, A.G.; Chamorro, F.; Carpena, M.; Echave, J.; Fraga-Corral, M.; Simal-Gandara, J.; Prieto, M.A. Status and challenges of plant-anticancer compounds in cancer treatment. Pharmaceuticals 2021, 14, 157. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Zhao, G.D.; Shi, Z.; Qi, L.L.; Zhou, L.Y.; Fu, Z.X. The Ras/Raf/MEK/ERK signaling pathway and its role in the occurrence and development of HCC. Oncol. Lett. 2016, 12, 3045–3050. [Google Scholar] [CrossRef]
- Farooq, M.; Khan, A.W.; Kim, M.S.; Choi, S. The Role of Fibroblast Growth Factor (FGF) Signaling in Tissue Repair and Regeneration. Cells 2021, 10, 3242. [Google Scholar] [CrossRef]
- Zhao, Y.; Hu, X.; Liu, Y.; Dong, S.; Wen, Z.; He, W.; Zhang, S.; Huang, Q.; Shi, M. ROS signaling under metabolic stress: Cross-talk between AMPK and AKT pathway. Mol. Cancer 2017, 16, 1–12. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, X. Targeting the Wnt/β-catenin signaling pathway in cancer. J. Hematol. Oncol. 2020, 13, 1–16. [Google Scholar] [CrossRef]
- Khan, A.W.; Khan, A.U.; Shah, S.M.M.; Ullah, A.; Faheem, M.; Saleem, M. An updated list of neuromedicinal plants of Pakistan, their uses, and phytochemistry. Evid. Based Complement. Alternat. Med. 2019, 2019, 6191505. [Google Scholar] [CrossRef]
- Catalano, E. Role of phytochemicals in the chemoprevention of tumors. arXiv 2016, arXiv:1605.04519. [Google Scholar]
- Alhasan, L.; Addai, Z.R. Allicin-induced modulation of angiogenesis in lung cancer cells (A549). Trop. J. Pharm. Res. 2018, 17, 2129–2134. [Google Scholar] [CrossRef]
- Zhang, X.; Zhu, Y.; Duan, W.; Feng, C.; He, X. Allicin induces apoptosis of the MGC-803 human gastric carcinoma cell line through the p38 mitogen-activated protein kinase/caspase-3 signaling pathway. Mol. Med. Rep. 2015, 11, 2755–2760. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Liu, H.; Chen, Y.; Yan, W. The effect of allicin on cell proliferation and apoptosis compared to blank control and cis-platinum in oral tongue squamous cell carcinoma. Onco Targets Ther. 2020, 13, 13183. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Jing, H.; Ma, G.; Liang, P. Allicin induces apoptosis through activation of both intrinsic and extrinsic pathways in glioma cells. Mol. Med. Rep. 2018, 17, 5976–5981. [Google Scholar] [CrossRef] [PubMed]
- Qiu, M.; Liu, J.; Su, Y.; Liu, J.; Wu, C.; Zhao, B. Aloperine induces apoptosis by a reactive oxygen species activation mechanism in human ovarian cancer cells. Protein Pept. Lett. 2020, 27, 860–869. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.-R.; Chen, S.-H.; Lin, C.-Y.; Chao, W.-Y.; Lim, Y.-P.; Yu, H.-I.; Lu, C.-H. In vitro antitumor activity of aloperine on human thyroid cancer cells through caspase-dependent apoptosis. Int. J. Mol. Sci. 2018, 19, 312. [Google Scholar] [CrossRef] [PubMed]
- Ling, Z.; Guan, H.; You, Z.; Wang, C.; Hu, L.; Zhang, L.; Wang, Y.; Chen, S.; Xu, B.; Chen, M. Aloperine executes antitumor effects through the induction of apoptosis and cell cycle arrest in prostate cancer In Vitro and In Vivo. Onco Targets Ther. 2018, 11, 2735. [Google Scholar] [CrossRef]
- Zhang, L.; Liang, J.; Liu, X.; Wu, J.; Tan, D.; Hu, W. Aloperine exerts antitumor effects on bladder cancer In Vitro. Onco Targets Ther. 2020, 13, 10351. [Google Scholar] [CrossRef]
- Zhang, L.; Zheng, Y.; Deng, H.; Liang, L.; Peng, J. Aloperine induces G2/M phase cell cycle arrest and apoptosis in HCT116 human colon cancer cells. Int. J. Mol. Med. 2014, 33, 1613–1620. [Google Scholar] [CrossRef]
- Chen, S.; Jin, Z.; Dai, L.; Wu, H.; Wang, J.; Wang, L.; Zhou, Z.; Yang, L.; Gao, W. Aloperine induces apoptosis and inhibits invasion in MG-63 and U2OS human osteosarcoma cells. Biomed. Pharmacother. 2018, 97, 45–52. [Google Scholar] [CrossRef]
- Li, D.; Li, X.; Li, G.; Meng, Y.; Jin, Y.; Shang, S.; Li, Y. Alpinumisoflavone causes DNA damage in colorectal cancer cells via blocking DNA repair mediated by RAD51. Life Sci. 2019, 216, 259–270. [Google Scholar] [CrossRef]
- Han, Y.; Yang, X.; Zhao, N.; Peng, J.; Gao, H.; Qiu, X. Alpinumisoflavone induces apoptosis in esophageal squamous cell carcinoma by modulating miR-370/PIM1 signaling. Am. J. Cancer Res. 2016, 6, 2755. [Google Scholar] [PubMed]
- Zhao, X.; Zhang, T.; Jiang, K.; Gao, H. Alpinumisoflavone exhibits anticancer activities in glioblastoma multiforme by suppressing glycolysis (Retraction of Vol 11, Pg 631, 2019). Anat. Rec. (Hoboken) 2020, 303, 2192–2201. [Google Scholar] [CrossRef] [PubMed]
- Makarević, J.; Rutz, J.; Juengel, E.; Kaulfuss, S.; Tsaur, I.; Nelson, K.; Pfitzenmaier, J.; Haferkamp, A.; Blaheta, R.A. Amygdalin influences bladder cancer cell adhesion and invasion In Vitro. PLoS ONE 2014, 9, e110244. [Google Scholar] [CrossRef]
- Lee, H.M.; Moon, A. Amygdalin regulates apoptosis and adhesion in Hs578T triple-negative breast cancer cells. Biomol. Ther. 2016, 24, 62. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.-K.; Shin, M.-S.; Yang, H.-Y.; Lee, J.-W.; Kim, Y.-S.; Lee, M.-H.; Kim, J.; Kim, K.-H.; Kim, C.-J. Amygdalin induces apoptosis through regulation of Bax and Bcl-2 expressions in human DU145 and LNCaP prostate cancer cells. Biol. Pharm. Bull. 2006, 29, 1597–1602. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Ma, J.; Wang, F.; Hu, J.; Cui, A.; Wei, C.; Yang, Q.; Li, F. Amygdalin induces apoptosis in human cervical cancer cell line HeLa cells. Immunopharmacol. Immunotoxicol. 2013, 35, 43–51. [Google Scholar] [CrossRef]
- Khan, I.; Khan, F.; Farooqui, A.; Ansari, I.A. Andrographolide exhibits anticancer potential against human colon cancer cells by inducing cell cycle arrest and programmed cell death via augmentation of intracellular reactive oxygen species level. Nutr. Cancer 2018, 70, 787–803. [Google Scholar] [CrossRef]
- Liu, G.; Chu, H. Andrographolide inhibits proliferation and induces cell cycle arrest and apoptosis in human melanoma cells. Oncol. Lett. 2018, 15, 5301–5305. [Google Scholar] [CrossRef]
- Peng, Y.; Wang, Y.; Tang, N.; Sun, D.; Lan, Y.; Yu, Z.; Zhao, X.; Feng, L.; Zhang, B.; Jin, L. Andrographolide inhibits breast cancer through suppressing COX-2 expression and angiogenesis via inactivation of p300 signaling and VEGF pathway. J. Exp. Clin. Cancer Res. 2018, 37, 1–14. [Google Scholar] [CrossRef]
- Forestier-Román, I.S.; López-Rivas, A.; Sánchez-Vázquez, M.M.; Rohena-Rivera, K.; Nieves-Burgos, G.; Ortiz-Zuazaga, H.; Torres-Ramos, C.A.; Martínez-Ferrer, M. Andrographolide induces DNA damage in prostate cancer cells. Oncotarget 2019, 10, 1085. [Google Scholar] [CrossRef]
- Pearngam, P.; Kumkate, S.; Okada, S.; Janvilisri, T. Andrographolide inhibits cholangiocarcinoma cell migration by down-regulation of claudin-1 via the p-38 signaling pathway. Front. Pharmacol. 2019, 10, 827. [Google Scholar] [CrossRef] [PubMed]
- Beesetti, S.L.; Jayadev, M.; Subhashini, G.V.; Mansour, L.; Alwasel, S.; Harrath, A.H. Andrographolide as a therapeutic agent against breast and ovarian cancers. Open Life Sci. 2019, 14, 462–469. [Google Scholar] [CrossRef] [PubMed]
- Shao, H.; Jing, K.; Mahmoud, E.; Huang, H.; Fang, X.; Yu, C. Apigenin sensitizes colon cancer cells to antitumor activity of ABT-263. Mol. Cancer Ther. 2013, 12, 2640–2650. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Wang, X.; Zha, D.; Cai, F.; Zhang, W.; He, Y.; Huang, Q.; Zhuang, H.; Hua, Z.-C. Apigenin potentiates TRAIL therapy of non-small cell lung cancer via upregulating DR4/DR5 expression in a p53-dependent manner. Sci. Rep. 2016, 6, 1–17. [Google Scholar] [CrossRef]
- Yang, J.; Pi, C.; Wang, G. Inhibition of PI3K/Akt/mTOR pathway by apigenin induces apoptosis and autophagy in hepatocellular carcinoma cells. Biomed. Pharmacother. 2018, 103, 699–707. [Google Scholar] [CrossRef]
- Ujiki, M.B.; Ding, X.-Z.; Salabat, M.R.; Bentrem, D.J.; Golkar, L.; Milam, B.; Talamonti, M.S.; Bell, R.H.; Iwamura, T.; Adrian, T.E. Apigenin inhibits pancreatic cancer cell proliferation through G2/M cell cycle arrest. Mol. Cancer 2006, 5, 1–8. [Google Scholar] [CrossRef]
- Li, Y.-W.; Xu, J.; Zhu, G.-Y.; Huang, Z.-J.; Lu, Y.; Li, X.-Q.; Wang, N.; Zhang, F.-X. Apigenin suppresses the stem cell-like properties of triple-negative breast cancer cells by inhibiting YAP/TAZ activity. Cell Death Discov. 2018, 4, 1–9. [Google Scholar] [CrossRef]
- Gupta, S.; Afaq, F.; Mukhtar, H. Involvement of nuclear factor-kappa B, Bax and Bcl-2 in induction of cell cycle arrest and apoptosis by apigenin in human prostate carcinoma cells. Oncogene 2002, 21, 3727–3738. [Google Scholar] [CrossRef]
- Liu, X.; Li, L.; Lv, L.; Chen, D.; Shen, L.; Xie, Z. Apigenin inhibits the proliferation and invasion of osteosarcoma cells by suppressing the Wnt/β-catenin signaling pathway. Oncol. Rep. 2015, 34, 1035–1041. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, J.; Shi, Y.; Xu, C.; Zhang, C.; Wong, Y.K.; Lee, Y.M.; Krishna, S.; He, Y.; Lim, T.K. Mechanistic investigation of the specific anticancer property of artemisinin and its combination with aminolevulinic acid for enhanced anticolorectal cancer activity. ACS Cent. Sci. 2017, 3, 743–750. [Google Scholar] [CrossRef]
- Yu, C.; Sun, P.; Zhou, Y.; Shen, B.; Zhou, M.; Wu, L.; Kong, M. Inhibition of AKT enhances the anti-cancer effects of Artemisinin in clear cell renal cell carcinoma. Biomed. Pharmacother. 2019, 118, 109383. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhou, Y.; Liu, Y.; Zhang, X.; Chen, T.; Chen, K.; Ba, Q.; Li, J.; Liu, H.; Wang, H. Preclinical efficacy and safety assessment of artemisinin-chemotherapeutic agent conjugates for ovarian cancer. EBioMedicine 2016, 14, 44–54. [Google Scholar] [CrossRef] [PubMed]
- Jia, J.; Qin, Y.; Zhang, L.; Guo, C.; Wang, Y.; Yue, X.; Qian, J. Artemisinin inhibits gallbladder cancer cell lines through triggering cell cycle arrest and apoptosis. Mol. Med. Rep. 2016, 13, 4461–4468. [Google Scholar] [CrossRef] [PubMed]
- Cathcart, M.-C.; Useckaite, Z.; Drakeford, C.; Semik, V.; Lysaght, J.; Gately, K.; O’Byrne, K.J.; Pidgeon, G.P. Anti-cancer effects of baicalein in non-small cell lung cancer in-vitro and in-vivo. BMC Cancer 2016, 16, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Palko-Labuz, A.; Sroda-Pomianek, K.; Uryga, A.; Kostrzewa-Suslow, E.; Michalak, K. Anticancer activity of baicalein and luteolin studied in colorectal adenocarcinoma LoVo cells and in drug-resistant LoVo/Dx cells. Biomed. Pharmacother. 2017, 88, 232–241. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.-Y.; Tsai, K.-W.; Li, Y.-Z.; Chang, Y.-S.; Lai, Y.-C.; Laio, Y.-H.; Wu, J.-D.; Liu, Y.-W. Anti-bladder-tumor effect of baicalein from Scutellaria baicalensis Georgi and its application In Vivo. Evid. Based Complement. Alternat. Med. 2013, 2013. [Google Scholar] [CrossRef]
- Tian, Y.; Zhen, L.; Bai, J.a.; Mei, Y.; Li, Z.; Lin, A.; Li, X. Anticancer effects of Baicalein in pancreatic Neuroendocrine tumors In Vitro and In Vivo. Pancreas 2017, 46, 1076. [Google Scholar] [CrossRef]
- Bie, B.; Sun, J.; Li, J.; Guo, Y.; Jiang, W.; Huang, C.; Yang, J.; Li, Z. Baicalein, a natural anti-cancer compound, alters microRNA expression profiles in Bel-7402 human hepatocellular carcinoma cells. Cell. Physiol. Biochem. 2017, 41, 1519–1531. [Google Scholar] [CrossRef]
- Guo, Z.; Hu, X.; Xing, Z.; Xing, R.; Lv, R.; Cheng, X.; Su, J.; Zhou, Z.; Xu, Z.; Nilsson, S. Baicalein inhibits prostate cancer cell growth and metastasis via the caveolin-1/AKT/mTOR pathway. Mol. Cell. Biochem. 2015, 406, 111–119. [Google Scholar] [CrossRef]
- Yu, X.; Cao, Y.; Tang, L.; Yang, Y.; Chen, F.; Xia, J. Baicalein inhibits breast cancer growth via activating a novel isoform of the long noncoding RNA PAX8-AS1-N. J. Cell. Biochem. 2018, 119, 6842–6856. [Google Scholar] [CrossRef]
- Li, Y.; Wang, D.; Liu, J.; Li, Y.; Chen, D.; Zhou, L.; Lang, T.; Zhou, Q. Baicalin Attenuates YAP Activity to Suppress Ovarian Cancer Stemness. Onco Targets Ther. 2020, 13, 7151. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Peng, B.; Nayak, Y.; Wang, C.; Si, F.; Liu, X.; Dou, J.; Xu, H.; Peng, G. Baicalein and baicalin promote melanoma apoptosis and senescence via metabolic inhibition. Front. Cell Dev. Biol. 2020, 8, 836. [Google Scholar] [CrossRef] [PubMed]
- Dong, Q.; Zheng, S.; Xu, R.; Lu, Q.; He, L. Study on effect of berbamine on multidrug resistance leukemia K562/Adr cells. Chin. J. Integr. Med. 2004, 24, 820–822. [Google Scholar]
- Meng, Z.; Li, T.; Ma, X.; Wang, X.; Van Ness, C.; Gan, Y.; Zhou, H.; Tang, J.; Lou, G.; Wang, Y. Berbamine inhibits the growth of liver cancer cells and cancer-initiating cells by targeting Ca2+/calmodulin-dependent protein kinase II. Mol. Cancer Ther. 2013, 12, 2067–2077. [Google Scholar] [CrossRef]
- Zhang, H.; Jiao, Y.; Shi, C.; Song, X.; Chang, Y.; Ren, Y.; Shi, X. Berbamine suppresses cell proliferation and promotes apoptosis in ovarian cancer partially via the inhibition of Wnt/β-catenin signaling. Acta Biochim. Biophys. Sin. 2018, 50, 532–539. [Google Scholar] [CrossRef]
- Mou, L.; Liang, B.; Liu, G.; Jiang, J.; Liu, J.; Zhou, B.; Huang, J.; Zang, N.; Liao, Y.; Ye, L. Berbamine exerts anticancer effects on human colon cancer cells via induction of autophagy and apoptosis, inhibition of cell migration and MEK/ERK signalling pathway. J. BUON 2019, 24, 1870–1875. [Google Scholar]
- Zhu, H.; Ruan, S.; Jia, F.; Chu, J.; Zhu, Y.; Huang, Y.; Liu, G. In vitro and In Vivo superior radiosensitizing effect of berbamine for head and neck squamous cell carcinoma. Onco Targets Ther. 2018, 11, 8117. [Google Scholar] [CrossRef]
- Chen, M.; Xiao, C.; Jiang, W.; Yang, W.; Qin, Q.; Tan, Q.; Lian, B.; Liang, Z.; Wei, C. Capsaicin Inhibits Proliferation and Induces Apoptosis in Breast Cancer by Down-Regulating FBI-1-Mediated NF-κB Pathway. Drug Des. Devel. Ther. 2021, 15, 125. [Google Scholar] [CrossRef]
- Anandakumar, P.; Kamaraj, S.; Jagan, S.; Ramakrishnan, G.; Asokkumar, S.; Naveenkumar, C.; Raghunandhakumar, S.; Vanitha, M.K.; Devaki, T. The anticancer role of capsaicin in experimentallyinduced lung carcinogenesis. J. Pharmacopunct. 2015, 18, 19. [Google Scholar] [CrossRef]
- Ramos-Torres, Á.; Bort, A.; Morell, C.; Rodríguez-Henche, N.; Díaz-Laviada, I. The pepper’s natural ingredient capsaicin induces autophagy blockage in prostate cancer cells. Oncotarget 2016, 7, 1569. [Google Scholar] [CrossRef]
- Jin, J.; Lin, G.; Huang, H.; Xu, D.; Yu, H.; Ma, X.; Zhu, L.; Ma, D.; Jiang, H. Capsaicin mediates cell cycle arrest and apoptosis in human colon cancer cells via stabilizing and activating p53. Int. J. Biol. Sci. 2014, 10, 285. [Google Scholar] [CrossRef] [PubMed]
- Mao, X.; Zhu, H.; Luo, D.; Ye, L.; Yin, H.; Zhang, J.; Zhang, Y.; Zhang, Y. Capsaicin inhibits glycolysis in esophageal squamous cell carcinoma by regulating hexokinase-2 expression. Mol. Med. Rep. 2018, 17, 6116–6121. [Google Scholar] [CrossRef] [PubMed]
- Shin, D.-H.; Kim, O.-H.; Jun, H.-S.; Kang, M.-K. Inhibitory effect of capsaicin on B16-F10 melanoma cell migration via the phosphatidylinositol 3-kinase/Akt/Rac1 signal pathway. Exp. Mol. Med. 2008, 40, 486–494. [Google Scholar] [CrossRef] [PubMed]
- Rattanawong, A.; Payon, V.; Limpanasittikul, W.; Boonkrai, C.; Mutirangura, A.; Wonganan, P. Cepharanthine exhibits a potent anticancer activity in p53-mutated colorectal cancer cells through upregulation of p21Waf1/Cip1. Oncol. Rep. 2018, 39, 227–238. [Google Scholar] [CrossRef]
- Gao, S.; Li, X.; Ding, X.; Qi, W.; Yang, Q. Cepharanthine induces autophagy, apoptosis and cell cycle arrest in breast cancer cells. Cell. Physiol. Biochem. 2017, 41, 1633–1648. [Google Scholar] [CrossRef]
- Payon, V.; Kongsaden, C.; Ketchart, W.; Mutirangura, A.; Wonganan, P. Mechanism of cepharanthine cytotoxicity in human ovarian cancer cells. Planta Med. 2019, 85, 41–47. [Google Scholar] [CrossRef]
- Biswas, K.K.; Tancharon, S.; Sarker, K.P.; Kawahara, K.I.; Hashiguchi, T.; Maruyama, I. Cepharanthine triggers apoptosis in a human hepatocellular carcinoma cell line (HuH-7) through the activation of JNK1/2 and the downregulation of Akt. FEBS Lett. 2006, 580, 703–710. [Google Scholar] [CrossRef]
- Liu, Y.; Feng, Y.; Li, Y.; Hu, Y.; Zhang, Q.; Huang, Y.; Shi, K.; Ran, C.; Hou, J.; Zhou, G. Chlorogenic acid decreases malignant characteristics of hepatocellular carcinoma cells by inhibiting DNMT1 expression. Front. Pharmacol. 2020, 11, 867. [Google Scholar] [CrossRef]
- Gouthamchandra, K.; Sudeep, H.; Venkatesh, B.; Prasad, K.S. Chlorogenic acid complex (CGA7), standardized extract from green coffee beans exerts anticancer effects against cultured human colon cancer HCT-116 cells. Food Sci. Hum. Wellness. 2017, 6, 147–153. [Google Scholar] [CrossRef]
- Changizi, Z.; Moslehi, A.; Rohani, A.H.; Eidi, A. Chlorogenic acid inhibits growth of 4T1 breast cancer cells through involvement in Bax/Bcl2 pathway. J. Cancer Res. Ther. 2020, 16, 1435. [Google Scholar]
- Zhang, T.; Chen, W.; Jiang, X.; Liu, L.; Wei, K.; Du, H.; Wang, H.; Li, J. Anticancer effects and underlying mechanism of Colchicine on human gastric cancer cell lines In Vitro and In Vivo. Biosci. Rep. 2019, 39, BSR20181802. [Google Scholar] [CrossRef]
- Cho, J.H.; Joo, Y.H.; Shin, E.Y.; Park, E.J.; Kim, M.S. Anticancer effects of colchicine on hypopharyngeal cancer. Anticancer Res. 2017, 37, 6269–6280. [Google Scholar]
- Bakar-Ateş, F.; Özmen, N.; Kaya-Sezginer, E.; Kurt, E.E. Effects of colchicine on cell cycle arrest and MMP-2 mRNA expression in MCF-7 breast adenocarcinoma cells. Turk. Hij. Den. Biyol. Derg 2018, 75, 239–244. [Google Scholar] [CrossRef]
- Huang, Z.; Xu, Y.; Peng, W. Colchicine induces apoptosis in HT-29 human colon cancer cells via the AKT and c-Jun N-terminal kinase signaling pathways. Mol. Med. Rep. 2015, 12, 5939–5944. [Google Scholar] [CrossRef] [PubMed]
- Boehle, A.S.; Sipos, B.; Kliche, U.; Kalthoff, H.; Dohrmann, P. Combretastatin A-4 prodrug inhibits growth of human non–small cell lung cancer in a murine xenotransplant model. Ann. Thorac. Surg. 2001, 71, 1657–1665. [Google Scholar] [CrossRef]
- Shen, C.H.; Shee, J.J.; Wu, J.Y.; Lin, Y.W.; Wu, J.D.; Liu, Y.W. Combretastatin A-4 inhibits cell growth and metastasis in bladder cancer cells and retards tumour growth in a murine orthotopic bladder tumour model. Br. J. Pharmacol. 2010, 160, 2008–2027. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Li, W.; Xu, J.; Zhang, T.; Zuo, D.; Zhou, Z.; Lin, B.; Wang, G.; Wang, Z.; Sun, W. NDRG1 inhibition sensitizes osteosarcoma cells to combretastatin A-4 through targeting autophagy. Cell Death Dis. 2017, 8, e3048. [Google Scholar] [CrossRef]
- Zhang, B.Y.; Zhang, L.; Chen, Y.M.; Qiao, X.; Zhao, S.L.; Li, P.; Liu, J.F.; Wen, X.; Yang, J. Corosolic acid inhibits colorectal cancer cells growth as a novel HER2/HER3 heterodimerization inhibitor. Br. J. Pharmacol. 2021, 178, 1475–1491. [Google Scholar] [CrossRef]
- Park, J.B.; Lee, J.S.; Lee, M.S.; Cha, E.Y.; Kim, S.; Sul, J.Y. Corosolic acid reduces 5-FU chemoresistance in human gastric cancer cells by activating AMPK. Mol. Med. Rep. 2018, 18, 2880–2888. [Google Scholar] [CrossRef]
- Zhang, C.; Niu, Y.; Wang, Z.; Xu, X.; Li, Y.; Ma, L.; Wang, J.; Yu, Y. Corosolic acid inhibits cancer progression by decreasing the level of CDK19-mediated O-GlcNAcylation in liver cancer cells. Cell Death Dis. 2021, 12, 1–11. [Google Scholar] [CrossRef]
- Ma, B.; Zhang, H.; Wang, Y.; Zhao, A.; Zhu, Z.; Bao, X.; Sun, Y.; Li, L.; Zhang, Q. Corosolic acid, a natural triterpenoid, induces ER stress-dependent apoptosis in human castration resistant prostate cancer cells via activation of IRE-1/JNK, PERK/CHOP and TRIB3. J. Exp. Clin. Cancer Res. 2018, 37, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.Q.; Zhang, J.H.; Yang, X.S. Corosolic acid induces potent anti-cancer effects in CaSki cervical cancer cells through the induction of apoptosis, cell cycle arrest and PI3K/Akt signalling pathway. Bangladesh J. Pharmacol. 2016, 11, 453–459. [Google Scholar] [CrossRef]
- Woo, S.M.; Seo, S.U.; Min, K.-j.; Im, S.-S.; Nam, J.-O.; Chang, J.-S.; Kim, S.; Park, J.-W.; Kwon, T.K. Corosolic acid induces non-apoptotic cell death through generation of lipid reactive oxygen species production in human renal carcinoma caki cells. Int. J. Mol. Sci. 2018, 19, 1309. [Google Scholar] [CrossRef] [PubMed]
- Son, K.H.; Hwang, J.H.; Kim, D.H.; Cho, Y.-E. Effect of corosolic acid on apoptosis and angiogenesis in MDA-MB-231 human breast cancer cells. J. Nutr. Health 2020, 53, 111–120. [Google Scholar] [CrossRef]
- Cui, A.; Li, X.; Ma, X.; Wang, X.; Liu, C.; Song, Z.; Pan, F.; Xia, Y.; Li, C. Transcriptome and Proteome Analysis Reveals Corosolic Acid Inhibiting Bladder Cancer via Targeting Cell Cycle and Inducing Mitophagy In Vitro and In Vivo. Res. Sq. 2021. [Google Scholar] [CrossRef]
- Festuccia, C.; Mancini, A.; Gravina, G.L.; Scarsella, L.; Llorens, S.; Alonso, G.L.; Tatone, C.; Di Cesare, E.; Jannini, E.A.; Lenzi, A. Antitumor effects of saffron-derived carotenoids in prostate cancer cell models. BioMed Res. Int. 2014, 2014, 135048. [Google Scholar] [CrossRef]
- Ray, P.; Guha, D.; Chakraborty, J.; Banerjee, S.; Adhikary, A.; Chakraborty, S.; Das, T.; Sa, G. Crocetin exploits p53-induced death domain (PIDD) and FAS-associated death domain (FADD) proteins to induce apoptosis in colorectal cancer. Sci. Rep. 2016, 6, 1–11. [Google Scholar] [CrossRef]
- Dhar, A.; Mehta, S.; Dhar, G.; Dhar, K.; Banerjee, S.; Van Veldhuizen, P.; Campbell, D.R.; Banerjee, S.K. Crocetin inhibits pancreatic cancer cell proliferation and tumor progression in a xenograft mouse model. Mol. Cancer Ther. 2009, 8, 315–323. [Google Scholar] [CrossRef]
- Bathaie, S.Z.; Hoshyar, R.; Miri, H.; Sadeghizadeh, M. Anticancer effects of crocetin in both human adenocarcinoma gastric cancer cells and rat model of gastric cancer. Biochem. Cell Biol. 2013, 91, 397–403. [Google Scholar] [CrossRef]
- Chai, Y.; Xiang, K.; Wu, Y.; Zhang, T.; Liu, Y.; Liu, X.; Zhen, W.; Si, Y. Cucurbitacin B inhibits the Hippo-YAP signaling pathway and exerts anticancer activity in colorectal cancer cells. Med. Sci. Monit. 2018, 24, 9251. [Google Scholar] [CrossRef]
- Si, W.; Lyu, J.; Liu, Z.; Wang, C.; Huang, J.; Jiang, L.; Ma, T. Cucurbitacin E inhibits cellular proliferation and enhances the chemo-response in gastric cancer by suppressing AKt activation. J. Cancer 2019, 10, 5843. [Google Scholar] [CrossRef] [PubMed]
- Obchoei, S.; Wongkham, S.; Aroonkesorn, A.; Suebsakwong, P.; Suksamrarn, A. Anti-cancer effect of cucurbitacin B on cholangiocarcinoma cells. BMB 2018. [Google Scholar] [CrossRef]
- Ku, J.M.; Hong, S.H.; Kim, H.I.; Lim, Y.S.; Lee, S.J.; Kim, M.; Seo, H.S.; Shin, Y.C.; Ko, S.-G. Cucurbitacin D exhibits its anti-cancer effect in human breast cancer cells by inhibiting Stat3 and Akt signaling. Eur. J. Inflam. 2018, 16, 1721727X17751809. [Google Scholar] [CrossRef]
- Wang, X.; Hang, Y.; Liu, J.; Hou, Y.; Wang, N.; Wang, M. Anticancer effect of curcumin inhibits cell growth through miR-21/PTEN/Akt pathway in breast cancer cell. Oncol. Lett. 2017, 13, 4825–4831. [Google Scholar] [CrossRef]
- Maulina, T.; Hadikrishna, I.; Hardianto, A.; Sjamsudin, E.; Pontjo, B.; Yusuf, H.Y. The therapeutic activity of curcumin through its anti-cancer potential on oral squamous cell carcinoma: A study on Sprague Dawley rat. SAGE Open Med. 2019, 7, 2050312119875982. [Google Scholar] [CrossRef]
- Killian, P.H.; Kronski, E.; Michalik, K.M.; Barbieri, O.; Astigiano, S.; Sommerhoff, C.P.; Pfeffer, U.; Nerlich, A.G.; Bachmeier, B.E. Curcumin inhibits prostate cancer metastasis In Vivo by targeting the inflammatory cytokines CXCL1 and-2. Carcinogenesis 2012, 33, 2507–2519. [Google Scholar] [CrossRef]
- Tong, W.; Wang, Q.; Sun, D.; Suo, J. Curcumin suppresses colon cancer cell invasion via AMPK-induced inhibition of NF-κB, uPA activator and MMP9. Oncol. Lett. 2016, 12, 4139–4146. [Google Scholar] [CrossRef]
- Kim, M.J.; Park, K.-S.; Kim, K.-T.; Gil, E.Y. The inhibitory effect of curcumin via fascin suppression through JAK/STAT3 pathway on metastasis and recurrence of ovary cancer cells. BMC Womens Health 2020, 20, 1–9. [Google Scholar] [CrossRef]
- Tang, L.; Liu, J.; Zhu, L.; Chen, Q.; Meng, Z.; Sun, L.; Hu, J.; Ni, Z.; Wang, X. Curcumin inhibits growth of human NCI-H292 lung squamous cell carcinoma cells by increasing FOXA2 expression. Front. Pharmacol. 2018, 9, 60. [Google Scholar] [CrossRef]
- Liu, Y.; Zhou, Z.; Yan, J.; Wu, X.; Xu, G. Diosgenin exerts antitumor activity via downregulation of Skp2 in breast cancer cells. Biomed Res. Int. 2020, 2020, 8072639. [Google Scholar] [CrossRef]
- Li, Y.; Wang, X.; Cheng, S.; Du, J.; Deng, Z.; Zhang, Y.; Liu, Q.; Gao, J.; Cheng, B.; Ling, C. Diosgenin induces G2/M cell cycle arrest and apoptosis in human hepatocellular carcinoma cells. Oncol. Rep. 2015, 33, 693–698. [Google Scholar] [CrossRef] [PubMed]
- Jia, S.-S.; Xi, G.-P.; Zhang, M.; Chen, Y.-B.; Lei, B.; Dong, X.-S.; Yang, Y.-M. Induction of apoptosis by D-limonene is mediated by inactivation of Akt in LS174T human colon cancer cells. Oncol. Rep. 2013, 29, 349–354. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Lin, H.; Wang, Y.; Lv, W.; Zhang, S.; Qian, Y.; Deng, X.; Feng, N.; Yu, H.; Qian, B. D-limonene exhibits antitumor activity by inducing autophagy and apoptosis in lung cancer. Onco Targets Ther. 2018, 11, 1833. [Google Scholar] [CrossRef] [PubMed]
- Rabi, T.; Bishayee, A. d-Limonene sensitizes docetaxel-induced cytotoxicity in human prostate cancer cells: Generation of reactive oxygen species and induction of apoptosis. J. Carcinog. 2009, 8. [Google Scholar] [CrossRef]
- Zhang, N.; Wang, J.; Sheng, A.; Huang, S.; Tang, Y.; Ma, S.; Hong, G. Emodin Inhibits the proliferation of MCF-7 human breast cancer cells through activation of aryl hydrocarbon receptor (AhR). Front. Pharmacol. 2021, 2372. [Google Scholar] [CrossRef]
- Li, M.; Jin, S.; Cao, Y.; Xu, J.; Zhu, S.; Li, Z. Emodin regulates cell cycle of non-small lung cancer (NSCLC) cells through hyaluronan synthase 2 (HA2)-HA-CD44/receptor for hyaluronic acid-mediated motility (RHAMM) interaction-dependent signaling pathway. Cancer Cell Int. 2021, 21, 1–12. [Google Scholar] [CrossRef]
- Lin, S.-Z.; Wei, W.-T.; Chen, H.; Chen, K.-J.; Tong, H.-F.; Wang, Z.-H.; Ni, Z.-L.; Liu, H.-B.; Guo, H.-C.; Liu, D.-L. Antitumor activity of emodin against pancreatic cancer depends on its dual role: Promotion of apoptosis and suppression of angiogenesis. PLoS ONE 2012, 7, e42146. [Google Scholar] [CrossRef]
- Saunders, I.T.; Mir, H.; Kapur, N.; Singh, S. Emodin inhibits colon cancer by altering BCL-2 family proteins and cell survival pathways. Cancer Cell Int. 2019, 19, 1–15. [Google Scholar] [CrossRef]
- Deng, G.; Ju, X.; Meng, Q.; Yu, Z.J.; Ma, L.B. Emodin inhibits the proliferation of PC3 prostate cancer cells In Vitro via the Notch signaling pathway. Mol. Med. Rep. 2015, 12, 4427–4433. [Google Scholar] [CrossRef]
- Kwak, T.W.; Park, S.B.; Kim, H.-J.; Jeong, Y.-I.; Kang, D.H. Anticancer activities of epigallocatechin-3-gallate against cholangiocarcinoma cells. Onco Targets Ther. 2017, 10, 137. [Google Scholar] [CrossRef]
- Chen, B.-H.; Hsieh, C.-H.; Tsai, S.-Y.; Wang, C.-Y.; Wang, C.-C. Anticancer effects of epigallocatechin-3-gallate nanoemulsion on lung cancer cells through the activation of AMP-activated protein kinase signaling pathway. Sci. Rep. 2020, 10, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.D.; Pagidas, K. Epigallocatechin-3-gallate, a natural polyphenol, inhibits cell proliferation and induces apoptosis in human ovarian cancer cells. Anticancer Res. 2010, 30, 2519–2523. [Google Scholar] [PubMed]
- Moses, M.A.; Henry, E.C.; Ricke, W.A.; Gasiewicz, T.A. The heat shock protein 90 inhibitor,(−)-epigallocatechin gallate, has anticancer activity in a novel human prostate cancer progression model. Cancer Prev. Res. 2015, 8, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y.S.; Kang, S.U.; Park, J.K.; Kim, Y.E.; Kim, Y.S.; Baek, S.J.; Lee, S.-H.; Kim, C.-H. Anti-cancer effect of (−)-epigallocatechin-3-gallate (EGCG) in head and neck cancer through repression of transactivation and enhanced degradation of β-catenin. Phytomedicine 2016, 23, 1344–1355. [Google Scholar] [CrossRef]
- Md Nesran, Z.N.; Shafie, N.H.; Ishak, A.H.; Mohd Esa, N.; Ismail, A.; Md Tohid, S.F. Induction of endoplasmic reticulum stress pathway by green tea epigallocatechin-3-gallate (EGCG) in colorectal cancer cells: Activation of PERK/p-eIF2α/ATF4 and IRE1α. Biomed Res. Int. 2019, 2019. [Google Scholar] [CrossRef]
- Xu, Y.; Fang, R.; Shao, J.; Cai, Z. Erianin induces triple-negative breast cancer cells apoptosis by activating PI3K/Akt pathway. Biosci. Rep. 2021, 41, BSR20210093. [Google Scholar] [CrossRef]
- Chen, P.; Wu, Q.; Feng, J.; Yan, L.; Sun, Y.; Liu, S.; Xiang, Y.; Zhang, M.; Pan, T.; Chen, X. Erianin, a novel dibenzyl compound in Dendrobium extract, inhibits lung cancer cell growth and migration via calcium/calmodulin-dependent ferroptosis. Signal Transduct. Target. Ther. 2020, 5, 1–11. [Google Scholar] [CrossRef]
- Chen, Y.-T.; Hsieh, M.-J.; Chen, P.-N.; Weng, C.-J.; Yang, S.-F.; Lin, C.-W. Erianin induces apoptosis and autophagy in oral squamous cell carcinoma cells. Am. J. Chin. Med. 2020, 48, 183–200. [Google Scholar] [CrossRef]
- Zhu, Q.; Sheng, Y.; Li, W.; Wang, J.; Ma, Y.; Du, B.; Tang, Y. Erianin, a novel dibenzyl compound in Dendrobium extract, inhibits bladder cancer cell growth via the mitochondrial apoptosis and JNK pathways. Toxicol. Appl. Pharmacol. 2019, 371, 41–54. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, T.; Sun, W.; Wang, Z.; Zuo, D.; Zhou, Z.; Li, S.; Xu, J.; Yin, F.; Hua, Y. Erianin induces G2/M-phase arrest, apoptosis, and autophagy via the ROS/JNK signaling pathway in human osteosarcoma cells In Vitro and In Vivo. Cell Death Dis. 2016, 7, e2247. [Google Scholar] [CrossRef]
- Tang, J.; Liu, J.; Zhang, C.; Zhou, C.; Chen, J. Erianin induces apoptosis of colorectal cancer cells via activation of JNK signaling pathways. Int. J. Clin. Exp. Med. 2019, 12, 11404–11411. [Google Scholar]
- Li, M.; He, Y.; Peng, C.; Xie, X.; Hu, G. Erianin inhibits human cervical cancer cell through regulation of tumor protein p53 via the extracellular signal-regulated kinase signaling pathway. Oncol. Lett. 2018, 16, 5006–5012. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Z.-B.; Huang, J.-M.; Xie, Y.-J.; Zhang, Y.-Z.; Chang, C.; Lai, H.-L.; Wang, W.; Yao, X.-J.; Fan, X.-X.; Wu, Q.-B. Evodiamine suppresses non-small cell lung cancer by elevating CD8+ T cells and downregulating the MUC1-C/PD-L1 axis. J. Exp. Clin. Cancer Res. 2020, 39, 1–16. [Google Scholar] [CrossRef]
- Chen, M.C.; Yu, C.H.; Wang, S.W.; Pu, H.F.; Kan, S.F.; Lin, L.C.; Chi, C.W.; Ho, L.L.T.; Lee, C.H.; Wang, P.S. Anti-proliferative effects of evodiamine on human thyroid cancer cell line ARO. J. Cell. Biochem. 2010, 110, 1495–1503. [Google Scholar] [CrossRef] [PubMed]
- Kan, S.F.; Yu, C.H.; Pu, H.F.; Hsu, J.M.; Chen, M.J.; Wang, P.S. Anti-proliferative effects of evodiamine on human prostate cancer cell lines DU145 and PC3. J. Cell. Biochem. 2007, 101, 44–56. [Google Scholar] [CrossRef] [PubMed]
- Jia, J.; Kang, X.; Liu, Y.; Zhang, J. Inhibition of human liver cancer cell growth by evodiamine involves apoptosis and deactivation of PI3K/AKT pathway. Appl. Biol. Chem. 2020, 63, 1–8. [Google Scholar] [CrossRef]
- Shi, C.-S.; Li, J.-M.; Chin, C.-C.; Kuo, Y.-H.; Lee, Y.-R.; Huang, Y.-C. Evodiamine induces cell growth arrest, apoptosis and suppresses tumorigenesis in human urothelial cell carcinoma cells. Anticancer Res. 2017, 37, 1149–1159. [Google Scholar]
- Zhou, P.; Li, X.-P.; Jiang, R.; Chen, Y.; Lv, X.-T.; Guo, X.-X.; Tian, K.; Yuan, D.-Z.; Lv, Y.-W.; Ran, J.-H. Evodiamine inhibits migration and invasion by Sirt1-mediated post-translational modulations in colorectal cancer. Anticancer Drugs 2019, 30, 611. [Google Scholar] [CrossRef]
- Zhong, Z.-F.; Tan, W.; Wang, S.-P.; Qiang, W.-A.; Wang, Y.-T. Anti-proliferative activity and cell cycle arrest induced by evodiamine on paclitaxel-sensitive and-resistant human ovarian cancer cells. Sci. Rep. 2015, 5, 1–12. [Google Scholar] [CrossRef]
- Khan, M.; Qazi, J.I.; Rasul, A.; Zheng, Y.; Ma, T. Evodiamine induces apoptosis in pancreatic carcinoma PANC-1 cells via NF-κB inhibition. Bangladesh J. Pharmacol. 2013, 8, 8–14. [Google Scholar] [CrossRef]
- Wang, S.; Wang, K.; Wang, H.; Han, J.; Sun, H. Autophagy is essential for flavopiridol-induced cytotoxicity against MCF-7 breast cancer cells. Mol. Med. Rep. 2017, 16, 9715–9720. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pinto, N.; Prokopec, S.D.; Ghasemi, F.; Meens, J.; Ruicci, K.M.; Khan, I.M.; Mundi, N.; Patel, K.; Han, M.W.; Yoo, J. Flavopiridol causes cell cycle inhibition and demonstrates anti-cancer activity in anaplastic thyroid cancer models. PLoS ONE 2020, 15, e0239315. [Google Scholar] [CrossRef] [PubMed]
- Saisomboon, S.; Kariya, R.; Vaeteewoottacharn, K.; Wongkham, S.; Sawanyawisuth, K.; Okada, S. Antitumor effects of flavopiridol, a cyclin-dependent kinase inhibitor, on human cholangiocarcinoma In Vitro and in an In Vivo xenograft model. Heliyon 2019, 5, e01675. [Google Scholar] [CrossRef] [PubMed]
- Patel, V.; Senderowicz, A.M.; Pinto, D.; Igishi, T.; Raffeld, M.; Quintanilla-Martinez, L.; Ensley, J.F.; Sausville, E.A.; Gutkind, J.S. Flavopiridol, a novel cyclin-dependent kinase inhibitor, suppresses the growth of head and neck squamous cell carcinomas by inducing apoptosis. J. Clin. Investig. 1998, 102, 1674–1681. [Google Scholar] [CrossRef]
- Cetintas, V.B.; Acikgoz, E.; Yigitturk, G.; Demir, K.; Oktem, G.; Kaymaz, B.T.; Oltulu, F.; Aktug, H. Effects of flavopiridol on critical regulation pathways of CD133high/CD44high lung cancer stem cells. Medicine 2016, 95. [Google Scholar] [CrossRef]
- Hassan, M.S.; Cwidak, N.; Johnson, C.; Däster, S.; Eppenberger-Castori, S.; Awasthi, N.; Li, J.; Schwarz, M.A.; von Holzen, U. Therapeutic Potential of the Cyclin-Dependent Kinase Inhibitor Flavopiridol on c-Myc Overexpressing Esophageal Cancer. Front. Pharmacol. 2021, 2589. [Google Scholar] [CrossRef]
- Ko, E.-B.; Jang, Y.-G.; Kim, C.-W.; Go, R.-E.; Lee, H.K.; Choi, K.-C. Gallic Acid Hindered Lung Cancer Progression by Inducing Cell Cycle Arrest and Apoptosis in A549 Lung Cancer Cells via PI3K/Akt Pathway. Biomol. Ther. 2021, 30, 151–161. [Google Scholar] [CrossRef]
- Shi, C.-j.; Zheng, Y.B.; Pan, F.F.; Zhang, F.W.; Zhuang, P.; Fu, W.M. Gallic Acid Suppressed Tumorigenesis by an LncRNA MALAT1-Wnt/β-Catenin Axis in Hepatocellular Carcinoma. Front. Pharmacol. 2021, 12. [Google Scholar] [CrossRef]
- Moghtaderi, H.; Sepehri, H.; Delphi, L.; Attari, F. Gallic acid and curcumin induce cytotoxicity and apoptosis in human breast cancer cell MDA-MB-231. BioImpacts BI 2018, 8, 185. [Google Scholar] [CrossRef]
- Lin, X.; Wang, G.; Liu, P.; Han, L.; Wang, T.; Chen, K.; Gao, Y. Gallic acid suppresses colon cancer proliferation by inhibiting SRC and EGFR phosphorylation. Exp. Ther. Med. 2021, 21, 1–11. [Google Scholar] [CrossRef]
- Tsai, C.-L.; Chiu, Y.-M.; Ho, T.-Y.; Hsieh, C.-T.; Shieh, D.-C.; Lee, Y.-J.; Tsay, G.J.; Wu, Y.-Y. Gallic acid induces apoptosis in human gastric adenocarcinoma cells. Anticancer Res. 2018, 38, 2057–2067. [Google Scholar] [PubMed]
- Chen, H.-M.; Wu, Y.-C.; Chia, Y.-C.; Chang, F.-R.; Hsu, H.-K.; Hsieh, Y.-C.; Chen, C.-C.; Yuan, S.-S. Gallic acid, a major component of Toona sinensis leaf extracts, contains a ROS-mediated anti-cancer activity in human prostate cancer cells. Cancer Lett. 2009, 286, 161–171. [Google Scholar] [CrossRef] [PubMed]
- Varela-Rodríguez, L.; Sánchez-Ramírez, B.; Hernández-Ramírez, V.I.; Varela-Rodríguez, H.; Castellanos-Mijangos, R.D.; González-Horta, C.; Chávez-Munguía, B.; Talamás-Rohana, P. Effect of Gallic acid and Myricetin on ovarian cancer models: A possible alternative antitumoral treatment. BMC Complement. Med. Ther. 2020, 20, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Li, D.; Yu, L.; Niu, F. Gallic acid as a cancer-selective agent induces apoptosis in pancreatic cancer cells. Chemotherapy 2012, 58, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Hatami, E.; Nagesh, P.K.; Jaggi, M.; Chauhan, S.C.; Yallapu, M.M. Gambogic acid potentiates gemcitabine induced anticancer activity in non-small cell lung cancer. Eur. J. Pharmacol. 2020, 888, 173486. [Google Scholar] [CrossRef]
- Zhou, J.; Luo, Y.-H.; Wang, J.-R.; Lu, B.-B.; Wang, K.-M.; Tian, Y. Gambogenic acid induction of apoptosis in a breast cancer cell line. Asian Pac. J. Cancer Prev. 2013, 14, 7601–7605. [Google Scholar] [CrossRef]
- Lee, P.N.H.; Ho, W.S. Antiproliferative activity of gambogic acid isolated from Garcinia hanburyi in Hep3B and Huh7 cancer cells. Oncol. Rep. 2013, 29, 1744–1750. [Google Scholar] [CrossRef]
- Pan, H.; Jansson, K.H.; Beshiri, M.L.; Yin, J.; Fang, L.; Agarwal, S.; Nguyen, H.; Corey, E.; Zhang, Y.; Liu, J. Gambogic acid inhibits thioredoxin activity and induces ROS-mediated cell death in castration-resistant prostate cancer. Oncotarget 2017, 8, 77181. [Google Scholar] [CrossRef]
- Zhang, H.; Lei, Y.; Yuan, P.; Li, L.; Luo, C.; Gao, R.; Tian, J.; Feng, Z.; Nice, E.C.; Sun, J. ROS-mediated autophagy induced by dysregulation of lipid metabolism plays a protective role in colorectal cancer cells treated with gambogic acid. PLoS ONE 2014, 9, e96418. [Google Scholar] [CrossRef]
- Lin, D.; Lin, X.; He, T.; Xie, G. Gambogic Acid Inhibits the Progression of Gastric Cancer via circRNA_ASAP2/miR-33a-5p/CDK7 Axis. Cancer Manag. Res. 2020, 12, 9221. [Google Scholar] [CrossRef]
- Zhang, Q.; Bao, J.; Yang, J. Genistein-triggered anticancer activity against liver cancer cell line HepG2 involves ROS generation, mitochondrial apoptosis, G2/M cell cycle arrest and inhibition of cell migration. Arch. Med. Sci. 2019, 15, 1001. [Google Scholar] [CrossRef] [PubMed]
- Qin, J.; Chen, J.X.; Zhu, Z.; Teng, J.A. Genistein inhibits human colorectal cancer growth and suppresses miR-95, Akt and SGK1. Cell. Physiol. Biochem. 2015, 35, 2069–2077. [Google Scholar] [CrossRef] [PubMed]
- Pavese, J.M.; Krishna, S.N.; Bergan, R.C. Genistein inhibits human prostate cancer cell detachment, invasion, and metastasis. Am. J. Clin. Nutr. 2014, 100, 431S–436S. [Google Scholar] [CrossRef] [PubMed]
- Fu, Z.; Cao, X.; Liu, L.; Cao, X.; Cui, Y.; Li, X.; Quan, M.; Ren, K.; Chen, A.; Xu, C. Genistein inhibits lung cancer cell stem-like characteristics by modulating MnSOD and FoxM1 expression. Oncol. Lett. 2020, 20, 2506–2515. [Google Scholar] [CrossRef]
- Sp, N.; Kang, D.Y.; Lee, J.-M.; Bae, S.W.; Jang, K.-J. Potential antitumor effects of 6-gingerol in p53-dependent mitochondrial apoptosis and inhibition of tumor sphere formation in breast cancer cells. Int. J. Mol. Sci. 2021, 22, 4660. [Google Scholar] [CrossRef]
- Kapoor, V.; Aggarwal, S.; Das, S.N. 6-Gingerol mediates its anti tumor activities in human oral and cervical cancer cell lines through apoptosis and cell cycle arrest. Phytother. Res. 2016, 30, 588–595. [Google Scholar] [CrossRef]
- Tsai, Y.; Xia, C.; Sun, Z. The Inhibitory Effect of 6-Gingerol on Ubiquitin-Specific Peptidase 14 Enhances Autophagy-Dependent Ferroptosis and Anti-Tumor In Vivo and In Vitro. Front. Pharmacol. 2020, 11, 1792. [Google Scholar] [CrossRef]
- Park, Y.J.; Wen, J.; Bang, S.; Park, S.W.; Song, S.Y. [6]-Gingerol induces cell cycle arrest and cell death of mutant p53-expressing pancreatic cancer cells. Yonsei Med. J. 2006, 47, 688–697. [Google Scholar] [CrossRef]
- Park, Y.; Woo, S.H.; Seo, S.K.; Kim, H.; Noh, W.C.; Lee, J.K.; Kwon, B.M.; Min, K.N.; Choe, T.B.; Park, I.C. Ginkgetin induces cell death in breast cancer cells via downregulation of the estrogen receptor. Oncol. Lett. 2017, 14, 5027–5033. [Google Scholar] [CrossRef]
- Lou, J.-S.; Bi, W.-C.; Chan, G.K.; Jin, Y.; Wong, C.-W.; Zhou, Z.-Y.; Wang, H.-Y.; Yao, P.; Dong, T.T.; Tsim, K.W. Ginkgetin induces autophagic cell death through p62/SQSTM1-mediated autolysosome formation and redox setting in non-small cell lung cancer. Oncotarget 2017, 8, 93131. [Google Scholar] [CrossRef]
- Jeon, Y.J.; Jung, S.N.; Yun, J.; Lee, C.W.; Choi, J.; Lee, Y.J.; Han, D.C.; Kwon, B.M. Ginkgetin inhibits the growth of DU− 145 prostate cancer cells through inhibition of signal transducer and activator of transcription 3 activity. Cancer Sci. 2015, 106, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Xiong, M.; Wang, L.; Yu, H.L.; Han, H.; Mao, D.; Chen, J.; Zeng, Y.; He, N.; Liu, Z.G.; Wang, Z.Y. Ginkgetin exerts growth inhibitory and apoptotic effects on osteosarcoma cells through inhibition of STAT3 and activation of caspase-3/9. Oncol. Rep. 2016, 35, 1034–1040. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.; Sun, C.-M.; Chuang, H.-H.; Chang, P.-T. Studies on the cytotoxic mechanisms of ginkgetin in a human ovarian adenocarcinoma cell line. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2000, 362, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Huang, S.S.; Wang, X.; Lou, Z.G.; Yao, X.P.; Weng, G.B. Ginkgetin induces apoptosis in 786-O cell line via suppression of JAK2-STAT3 pathway. Iran. J. Basic Med. Sci. 2016, 19, 1245. [Google Scholar]
- Lin, S.-C.; Chu, P.-Y.; Liao, W.-T.; Wu, M.-Y.; Tsui, K.-H.; Lin, L.-T.; Huang, C.-H.; Chen, L.-L.; Li, C.-J. Glycyrrhizic acid induces human MDA-MB-231 breast cancer cell death and autophagy via the ROS-mitochondrial pathway. Oncol. Rep. 2018, 39, 703–710. [Google Scholar] [CrossRef]
- Wang, H.; Ge, X.; Qu, H.; Wang, N.; Zhou, J.; Xu, W.; Xie, J.; Zhou, Y.; Shi, L.; Qin, Z. Glycyrrhizic Acid Inhibits Proliferation of Gastric Cancer Cells by Inducing Cell Cycle Arrest and Apoptosis. Cancer Manag. Res. 2020, 12, 2853. [Google Scholar] [CrossRef]
- Thirugnanam, S.; Xu, L.; Ramaswamy, K.; Gnanasekar, M. Glycyrrhizin induces apoptosis in prostate cancer cell lines DU-145 and LNCaP. Oncol. Rep. 2008, 20, 1387–1392. [Google Scholar]
- Haghshenas, V.; Fakhari, S.; Mirzaie, S.; Rahmani, M.; Farhadifar, F.; Pirzadeh, S.; Jalili, A. Glycyrrhetinic Acid inhibits cell growth and induces apoptosis in ovarian cancer a2780 cells. Adv. Pharm. Bull. 2014, 4, 437. [Google Scholar]
- Cao, H.; Sethumadhavan, K.; Cao, F.; Wang, T.T. Gossypol decreased cell viability and down-regulated the expression of a number of genes in human colon cancer cells. Sci. Rep. 2021, 11, 1–16. [Google Scholar] [CrossRef]
- Messeha, S.S.; Zarmouh, N.O.; Mendonca, P.; Cotton, C.; Soliman, K.F. Molecular mechanism of gossypol mediating CCL2 and IL-8 attenuation in triple-negative breast cancer cells. Mol. Med. Rep. 2020, 22, 1213–1226. [Google Scholar] [CrossRef]
- Wang, Y.; Lai, H.; Fan, X.; Luo, L.; Duan, F.; Jiang, Z.; Wang, Q.; Leung, E.L.H.; Liu, L.; Yao, X. Gossypol inhibits non-small cell lung cancer cells proliferation by targeting EGFRL858R/T790M. Front. Pharmacol. 2018, 9, 728. [Google Scholar] [CrossRef] [PubMed]
- Volate, S.R.; Kawasaki, B.T.; Hurt, E.M.; Milner, J.A.; Kim, Y.S.; White, J.; Farrar, W.L. Gossypol Induces Apoptosis by Activating p53 in Prostate Cancer Cells and Prostate Tumor–Initiating Cells. Mol. Cancer Ther. 2010, 9, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Jin, L.; Li, X.; Deng, H.; Chen, Y.; Lian, Q.; Ge, R.; Deng, H. Gossypol induces apoptosis in ovarian cancer cells through oxidative stress. Mol. Biosyst. 2013, 9, 1489–1497. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, Y.-S.; Chu, S.-C.; Huang, S.-C.; Kao, S.-H.; Lin, M.-S.; Chen, P.-N. Gossypol Reduces Metastasis and Epithelial-Mesenchymal Transition by Targeting Protease in Human Cervical Cancer. Am. J. Chin. Med. 2021, 49, 181–198. [Google Scholar] [CrossRef]
- Wolter, K.G.; Wang, S.J.; Henson, B.S.; Wang, S.; Griffith, K.A.; Kumar, B.; Chen, J.; Carey, T.E.; Bradford, C.R.; D’Silva, N.J. (−)-gossypol inhibits growth and promotes apoptosis of human head and neck squamous cell carcinoma In Vivo. Neoplasia 2006, 8, 163–172. [Google Scholar] [CrossRef]
- Haasler, L.; Kondadi, A.K.; Tsigaras, T.; von Montfort, C.; Graf, P.; Stahl, W.; Brenneisen, P. The BH3 mimetic (±) gossypol induces ROS-independent apoptosis and mitochondrial dysfunction in human A375 melanoma cells In Vitro. Arch. Toxicol. 2021, 95, 1349–1365. [Google Scholar] [CrossRef]
- Ding, Y.; He, J.; Huang, J.; Yu, T.; Shi, X.; Zhang, T.; Yan, G.; Chen, S.; Peng, C. Harmine induces anticancer activity in breast cancer cells via targeting TAZ. Int. J. Oncol. 2019, 54, 1995–2004. [Google Scholar] [CrossRef]
- Ruan, S.; Jia, F.; Li, J. Potential antitumor effect of harmine in the treatment of thyroid cancer. Evid. Based Complement. Alternat. Med. 2017, 2017. [Google Scholar] [CrossRef]
- Li, C.; Wang, Y.; Wang, C.; Yi, X.; Li, M.; He, X. Anticancer activities of harmine by inducing a pro-death autophagy and apoptosis in human gastric cancer cells. Phytomedicine 2017, 28, 10–18. [Google Scholar] [CrossRef]
- Wu, L.-W.; Zhang, J.-K.; Rao, M.; Zhang, Z.-Y.; Zhu, H.-J.; Zhang, C. Harmine suppresses the proliferation of pancreatic cancer cells and sensitizes pancreatic cancer to gemcitabine treatment. Onco Targets Ther. 2019, 12, 4585. [Google Scholar] [CrossRef]
- Gao, J.; Zhu, H.; Wan, H.; Zou, X.; Ma, X.; Gao, G. Harmine suppresses the proliferation and migration of human ovarian cancer cells through inhibiting ERK/CREB pathway. Oncol. Rep. 2017, 38, 2927–2934. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.-F.; Sun, R.Q.; Jia, Y.F.; Chen, Q.; Tu, R.-F.; Li, K.K.; Zhang, X.-D.; Du, R.-L.; Cao, R.H. Synthesis and mechanisms of action of novel harmine derivatives as potential antitumor agents. Sci. Rep. 2016, 6, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Cincin, Z.B.; Unlu, M.; Kiran, B.; Bireller, E.S.; Baran, Y.; Cakmakoglu, B. Anti-proliferative, apoptotic and signal transduction effects of hesperidin in non-small cell lung cancer cells. Cell. Oncol. 2015, 38, 195–204. [Google Scholar] [CrossRef]
- Banjerdpongchai, R.; Wudtiwai, B.; Khaw-On, P.; Rachakhom, W.; Duangnil, N.; Kongtawelert, P. Hesperidin from Citrus seed induces human hepatocellular carcinoma HepG2 cell apoptosis via both mitochondrial and death receptor pathways. Tumor Biol. 2016, 37, 227–237. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Chen, Y.; Zhang, X. Hesperidin-triggered necrosis-like cell death in skin cancer cell line A431 might be prompted by ROS mediated alterations in mitochondrial membrane potential. Int. J. Clin. Exp. Med. 2018, 11, 1948–1954. [Google Scholar]
- Ning, L.; Zhao, W.; Gao, H.; Wu, Y. Hesperidin induces anticancer effects on human prostate cancer cells via ROS-mediated necrosis like cell death. J. BUON 2020, 25, 2629–2634. [Google Scholar]
- Kongtawelert, P.; Wudtiwai, B.; Shwe, T.H.; Pothacharoen, P.; Phitak, T. Inhibitory effect of Hesperidin on the expression of programmed death ligand (PD-L1) in breast Cancer. Molecules 2020, 25, 252. [Google Scholar] [CrossRef]
- Lv, L.; Zhang, W.; Li, T.; Jiang, L.; Lu, X.; Lin, J. Hispidulin exhibits potent anticancer activity In Vitro and In Vivo through activating ER stress in non-small-cell lung cancer cells. Oncol. Rep. 2020, 43, 1995–2003. [Google Scholar] [CrossRef]
- Han, M.; Gao, H.; Ju, P.; Gao, M.Q.; Yuan, Y.P.; Chen, X.H.; Liu, K.L.; Han, Y.T.; Han, Z.W. Hispidulin inhibits hepatocellular carcinoma growth and metastasis through AMPK and ERK signaling mediated activation of PPARγ. Biomed. Pharmacother. 2018, 103, 272–283. [Google Scholar] [CrossRef]
- Gao, H.; Gao, M.Q.; Peng, J.J.; Han, M.; Liu, K.L.; Han, Y.T. Hispidulin mediates apoptosis in human renal cell carcinoma by inducing ceramide accumulation. Acta Pharmacol. Sin. 2017, 38, 1618–1631. [Google Scholar] [CrossRef]
- Yu, C.Y.; Su, K.-Y.; Lee, P.-L.; Jhan, J.-Y.; Tsao, P.-H.; Chan, D.-C.; Chen, Y.-L.S. Potential therapeutic role of hispidulin in gastric cancer through induction of apoptosis via NAG-1 signaling. Evid. Based Complement. Alternat. Med. 2013, 2013, 518301. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Xue, L. Kaempferol suppresses proliferation and induces cell cycle arrest, apoptosis, and DNA damage in breast cancer cells. Oncol. Res. 2019, 27, 629. [Google Scholar] [CrossRef] [PubMed]
- Han, B.; Yu, Y.-Q.; Yang, Q.-L.; Shen, C.-Y.; Wang, X.-J. Kaempferol induces autophagic cell death of hepatocellular carcinoma cells via activating AMPK signaling. Oncotarget 2017, 8, 86227. [Google Scholar] [CrossRef] [PubMed]
- Hung, T.-W.; Chen, P.-N.; Wu, H.-C.; Wu, S.-W.; Tsai, P.-Y.; Hsieh, Y.-S.; Chang, H.-R. Kaempferol inhibits the invasion and migration of renal cancer cells through the downregulation of AKT and FAK pathways. Int. J. Med. Sci. 2017, 14, 984. [Google Scholar] [CrossRef]
- Tu, L.Y.; Bai, H.H.; Cai, J.Y.; Deng, S.P. The mechanism of kaempferol induced apoptosis and inhibited proliferation in human cervical cancer SiHa cell: From macro to nano. Scanning 2016, 38, 644–653. [Google Scholar] [CrossRef]
- Wang, F.; Wang, L.; Qu, C.; Chen, L.; Geng, Y.; Cheng, C.; Yu, S.; Wang, D.; Yang, L.; Meng, Z. Kaempferol induces ROS-dependent apoptosis in pancreatic cancer cells via TGM2-mediated Akt/mTOR signaling. BMC Cancer 2021, 21, 1–11. [Google Scholar] [CrossRef]
- Li, W.; Du, B.; Wang, T.; Wang, S.; Zhang, J. Kaempferol induces apoptosis in human HCT116 colon cancer cells via the Ataxia-Telangiectasia Mutated-p53 pathway with the involvement of p53 Upregulated Modulator of Apoptosis. Chem. Biol. Interact. 2009, 177, 121–127. [Google Scholar] [CrossRef]
- Yang, J.; Chen, H.; Wang, Q.; Deng, S.; Huang, M.; Ma, X.; Song, P.; Du, J.; Huang, Y.; Wen, Y. Inhibitory effect of kurarinone on growth of human non-small cell lung cancer: An experimental study both In Vitro and In Vivo studies. Front. Pharmacol. 2018, 9, 252. [Google Scholar] [CrossRef]
- Zhou, W.; Cao, A.; Wang, L.; Wu, D. Kurarinone synergizes TRAIL-induced apoptosis in gastric cancer cells. Cell Biochem. Biophys. 2015, 72, 241–249. [Google Scholar] [CrossRef]
- De Naeyer, A.; Vanden Berghe, W.; Pocock, V.; Milligan, S.; Haegeman, G.; De Keukeleire, D. Estrogenic and Anticarcinogenic Properties of Kurarinone, a Lavandulyl Flavanone from the Roots of Sophora f lavescens. J. Nat. Prod. 2004, 67, 1829–1832. [Google Scholar] [CrossRef]
- Qu, D.; Zhang, X.; Sang, C.; Zhou, Y.; Ma, J.; Hui, L. Lappaconitine sulfate induces apoptosis in human colon cancer HT-29 cells and down-regulates PI3K/AKT/GSK3β signaling pathway. Med. Chem. Res. 2019, 28, 907–916. [Google Scholar] [CrossRef]
- Sheng, L.-H.; Xu, M.; Xu, L.-Q.; Xiong, F. Cytotoxic effect of lappaconitine on non-small cell lung cancer In Vitro and its molecular mechanism. J. Chin. Med. Mater. 2014, 37, 840–843. [Google Scholar]
- Song, N.; Ma, J.; Zhang, X.; Qu, D.; Hui, L.; Sang, C.; Li, H. Lappaconitine hydrochloride induces apoptosis and S phase cell cycle arrest through MAPK signaling pathway in human liver cancer HepG2 cells. Pharmacogn. Mag. 2021, 17, 334. [Google Scholar]
- Xue, L.; Zhang, W.J.; Fan, Q.X.; Wang, L.X. Licochalcone A inhibits PI3K/Akt/mTOR signaling pathway activation and promotes autophagy in breast cancer cells. Oncol. Lett. 2018, 15, 1869–1873. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.; Li, D.; Zhao, H.; Jiang, J.; Wang, P.; Ma, X.; Sun, X.; Zheng, Q. Licochalcone A-induced human bladder cancer T24 cells apoptosis triggered by mitochondria dysfunction and endoplasmic reticulum stress. Biomed Res. Int. 2013, 2013, 911–919. [Google Scholar] [CrossRef] [PubMed]
- Luo, W.; Sun, R.; Chen, X.; Li, J.; Jiang, J.; He, Y.; Shi, S.; Wen, H. ERK activation-mediated autophagy induction resists licochalcone A-induced anticancer activities in lung cancer cells In Vitro. Onco Targets Ther. 2020, 13, 13437. [Google Scholar] [CrossRef]
- Tsai, J.-P.; Hsiao, P.-C.; Yang, S.-F.; Hsieh, S.-C.; Bau, D.-T.; Ling, C.-L.; Pai, C.-L.; Hsieh, Y.-H. Licochalcone A suppresses migration and invasion of human hepatocellular carcinoma cells through downregulation of MKK4/JNK via NF-κB mediated urokinase plasminogen activator expression. PLoS ONE 2014, 9, e86537. [Google Scholar] [CrossRef]
- Li, Z.H.; Gao, J.; Hu, P.H.; Xiong, J.P. Anticancer effects of liriodenine on the cell growth and apoptosis of human breast cancer MCF-7 cells through the upregulation of p53 expression. Oncol. Lett. 2017, 14, 1979–1984. [Google Scholar] [CrossRef]
- Chang, H.-C.; Chang, F.-R.; Wu, Y.-C.; Lai, Y.-H. Anti-cancer effect of liriodenine on human lung cancer cells. Kaohsiung J. Med. Sci. 2004, 20, 365–371. [Google Scholar] [CrossRef]
- Nordin, N.; Majid, N.A.; Hashim, N.M.; Abd Rahman, M.; Hassan, Z.; Ali, H.M. Liriodenine, an aporphine alkaloid from Enicosanthellum pulchrum, inhibits proliferation of human ovarian cancer cells through induction of apoptosis via the mitochondrial signaling pathway and blocking cell cycle progression. Drug Des. Devel. Ther. 2015, 9, 1437. [Google Scholar]
- Cao, Z.; Zhang, H.; Cai, X.; Fang, W.; Chai, D.; Wen, Y.; Chen, H.; Chu, F.; Zhang, Y. Luteolin promotes cell apoptosis by inducing autophagy in hepatocellular carcinoma. Cell. Physiol. Biochem. 2017, 43, 1803–1812. [Google Scholar] [CrossRef] [PubMed]
- Kang, K.A.; Piao, M.J.; Hyun, Y.J.; Zhen, A.X.; Cho, S.J.; Ahn, M.J.; Yi, J.M.; Hyun, J.W. Luteolin promotes apoptotic cell death via upregulation of Nrf2 expression by DNA demethylase and the interaction of Nrf2 with p53 in human colon cancer cells. Exp. Mol. Med. 2019, 51, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Song, S.; Su, Z.; Xu, H.; Niu, M.; Chen, X.; Min, H.; Zhang, B.; Sun, G.; Xie, S.; Wang, H. Correction: Luteolin selectively kills STAT3 highly activated gastric cancer cells through enhancing the binding of STAT3 to SHP-1. Cell Death Dis. 2018, 9, 1. [Google Scholar] [CrossRef] [PubMed]
- Park, B.-S.; Kil, J.-J.; Kang, H.-M.; Yu, S.-B.; Park, D.-B.; Park, J.-A.; Kim, I.-R. Luteolin Induces Apoptosis via Mitochondrial Pathway and Inhibits Invasion and Migration of Oral Squamous Cell Carcinoma by Suppressing Epithelial-Mesenchymal Transition Induced Transcription Factors. Int. J. Oral Biol. 2018, 43, 69–76. [Google Scholar] [CrossRef]
- Huang, L.; Jin, K.; Lan, H. Luteolin inhibits cell cycle progression and induces apoptosis of breast cancer cells through downregulation of human telomerase reverse transcriptase. Oncol. Lett. 2019, 17, 3842–3850. [Google Scholar] [CrossRef]
- Cai, X.; Lu, W.; Ye, T.; Lu, M.; Wang, J.; Huo, J.; Qian, S.; Wang, X.; Cao, P. The molecular mechanism of luteolin-induced apoptosis is potentially related to inhibition of angiogenesis in human pancreatic carcinoma cells. Oncol. Rep. 2012, 28, 1353–1361. [Google Scholar] [CrossRef]
- Masraksa, W.; Tanasawet, S.; Hutamekalin, P.; Wongtawatchai, T.; Sukketsiri, W. Luteolin attenuates migration and invasion of lung cancer cells via suppressing focal adhesion kinase and non-receptor tyrosine kinase signaling pathway. Nutr. Res. Pract. 2020, 14, 127–133. [Google Scholar] [CrossRef]
- Takeshima, M.; Ono, M.; Higuchi, T.; Chen, C.; Hara, T.; Nakano, S. Anti-proliferative and apoptosis-inducing activity of lycopene against three subtypes of human breast cancer cell lines. Cancer Sci. 2014, 105, 252–257. [Google Scholar] [CrossRef]
- Jiang, L.-N.; Liu, Y.-B.; Li, B.-H. Lycopene exerts anti-inflammatory effect to inhibit prostate cancer progression. Asian J. Androl. 2019, 21, 80. [Google Scholar]
- Cha, J.H.; Kim, W.K.; Ha, A.W.; Kim, M.H.; Chang, M.J. Anti-inflammatory effect of lycopene in SW480 human colorectal cancer cells. Nutr. Res. Pract. 2017, 11, 90–96. [Google Scholar] [CrossRef]
- Jeong, Y.; Lim, J.W.; Kim, H. Lycopene inhibits reactive oxygen species-mediated NF-κB signaling and induces apoptosis in pancreatic cancer cells. Nutrients 2019, 11, 762. [Google Scholar] [CrossRef]
- Cheng, J.; Miller, B.; Balbuena, E.; Eroglu, A. Lycopene protects against smoking-induced lung cancer by inducing base excision repair. Antioxidants 2020, 9, 643. [Google Scholar] [CrossRef] [PubMed]
- Luo, C.; Wu, X.-G. Lycopene enhances antioxidant enzyme activities and immunity function in N-Methyl-N′-nitro-N-nitrosoguanidine–induced gastric cancer rats. Int. J. Mol. Sci. 2011, 12, 3340–3351. [Google Scholar] [CrossRef] [PubMed]
- Aktepe, O.H.; Şahin, T.K.; Güner, G.; Arik, Z.; Yalçin, Ş. Lycopene sensitizes the cervical cancer cells to cisplatin via targeting nuclear factor NF-kappa B. Turk. J. Med. Sci. 2021, 51, 368–374. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Burke, K.E.; Wang, Y.; Wei, H. Dietary lycopene protects SkH-1 mice against ultraviolet B-induced photocarcinogenesis. J. Drugs Dermatol. 2019, 18, 1244–1254. [Google Scholar]
- Czarnik-Kwaśniak, J.; Kwaśniak, K.; Kwasek, P.; Świerzowska, E.; Strojewska, A.; Tabarkiewicz, J. The influence of lycopene,[6]-gingerol, and silymarin on the apoptosis on U-118MG glioblastoma cells In Vitro model. Nutrients 2020, 12, 96. [Google Scholar] [CrossRef] [PubMed]
- Holzapfel, N.P.; Shokoohmand, A.; Wagner, F.; Landgraf, M.; Champ, S.; Holzapfel, B.M.; Clements, J.A.; Hutmacher, D.W.; Loessner, D. Lycopene reduces ovarian tumor growth and intraperitoneal metastatic load. Am. J. Cancer Res. 2017, 7, 1322. [Google Scholar]
- Wang, J.; Xu, J.; Xing, G. Lycorine inhibits the growth and metastasis of breast cancer through the blockage of STAT3 signaling pathway. Acta Biochim. Biophys. Sin. 2017, 49, 771–779. [Google Scholar] [CrossRef]
- Hu, M.; Peng, S.; He, Y.; Qin, M.; Cong, X.; Xing, Y.; Liu, M.; Yi, Z. Lycorine is a novel inhibitor of the growth and metastasis of hormone-refractory prostate cancer. Oncotarget 2015, 6, 15348. [Google Scholar] [CrossRef]
- Sun, Y.; Wu, P.; Sun, Y.; Sharopov, F.S.; Yang, Q.; Chen, F.; Wang, P.; Liang, Z. Lycorine possesses notable anticancer potentials in on-small cell lung carcinoma cells via blocking Wnt/β-catenin signaling and epithelial-mesenchymal transition (EMT). Biochem. Biophys. Res. Commun. 2018, 495, 911–921. [Google Scholar] [CrossRef]
- Liu, W.; Zhang, Q.; Tang, Q.; Hu, C.; Huang, J.; Liu, Y.; Lu, Y.; Wang, Q.; Li, G.; Zhang, R. [Corrigendum] Lycorine inhibits cell proliferation and migration by inhibiting ROCK1/cofilin-induced actin dynamics in HepG2 hepatoblastoma cells. Oncol. Rep. 2019, 42, 2856. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Ma, H.; Zhang, T.; Liu, H.; Yu, L.; Li, G.; Li, H.; Hu, M. Magnolol inhibits the growth of non-small cell lung cancer via inhibiting microtubule polymerization. Cell. Physiol. Biochem. 2017, 42, 1789–1801. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Zhang, F.; Wang, X.A.; Wu, X.; Zhang, B.; Zhang, N.; Wu, W.; Wang, Z.; Weng, H.; Liu, S. Magnolol inhibits growth of gallbladder cancer cells through the p53 pathway. Cancer Sci. 2015, 106, 1341–1350. [Google Scholar] [CrossRef] [PubMed]
- Kuan, L.-Y.; Chen, W.-L.; Chen, J.-H.; Hsu, F.-T.; Liu, T.-T.; Chen, W.-T.; Wang, K.-L.; Chen, W.-C.; Liu, Y.-C.; Wang, W.-S. Magnolol induces apoptosis and inhibits ERK-modulated metastatic potential in hepatocellular carcinoma cells. Vivo 2018, 32, 1361–1368. [Google Scholar] [CrossRef] [PubMed]
- Hwang, E.-S.; Park, K.-K. Magnolol suppresses metastasis via inhibition of invasion, migration, and matrix metalloproteinase-2/-9 activities in PC-3 human prostate carcinoma cells. Biosci. Biotechnol. Biochem. 2010, 74, 961–967. [Google Scholar] [CrossRef]
- Chen, Y.; Huang, K.; Ding, X.; Tang, H.; Xu, Z. Magnolol inhibits growth and induces apoptosis in esophagus cancer KYSE-150 cell lines via the MAP kinase pathway. J. Thorac. Dis. 2019, 11, 3030. [Google Scholar] [CrossRef]
- Huang, H.; Wang, Q.; Du, T.; Lin, C.; Lai, Y.; Zhu, D.; Wu, W.; Ma, X.; Bai, S.; Li, Z. Matrine inhibits the progression of prostate cancer by promoting expression of GADD45B. Prostate 2018, 78, 327–335. [Google Scholar] [CrossRef]
- Zhang, X.; Hou, G.; Liu, A.; Xu, H.; Guan, Y.; Wu, Y.; Deng, J.; Cao, X. Matrine inhibits the development and progression of ovarian cancer by repressing cancer associated phosphorylation signaling pathways. Cell Death Dis. 2019, 10, 1–17. [Google Scholar] [CrossRef]
- Chang, C.; Liu, S.P.; Fang, C.H.; He, R.S.; Wang, Z.; Zhu, Y.Q.; Jiang, S.W. Effects of matrine on the proliferation of HT29 human colon cancer cells and its antitumor mechanism. Oncol. Lett. 2013, 6, 699–704. [Google Scholar] [CrossRef]
- Lin, S.; Zhuang, J.; Zhu, L.; Jiang, Z. Matrine inhibits cell growth, migration, invasion and promotes autophagy in hepatocellular carcinoma by regulation of circ_0027345/miR-345-5p/HOXD3 axis. Cancer Cell Int. 2020, 20, 1–12. [Google Scholar] [CrossRef]
- Pu, J.; Tang, X.; Zhuang, X.; Hu, Z.; He, K.; Wu, Y.; Dai, T. Matrine induces apoptosis via targeting CCR7 and enhances the effect of anticancer drugs in non-small cell lung cancer In Vitro. Innate Immun. 2018, 24, 394–399. [Google Scholar] [CrossRef] [PubMed]
- Ha, T.K.; Jung, I.; Kim, M.E.; Bae, S.K.; Lee, J.S. Anti-cancer activity of myricetin against human papillary thyroid cancer cells involves mitochondrial dysfunction–mediated apoptosis. Biomed. Pharmacother. 2017, 91, 378–384. [Google Scholar] [CrossRef] [PubMed]
- Sun, F.; Zheng, X.Y.; Ye, J.; Wu, T.T.; Wang, J.l.; Chen, W. Potential anticancer activity of myricetin in human T24 bladder cancer cells both In Vitro and In Vivo. Nutr. Cancer 2012, 64, 599–606. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.E.; Ha, T.K.; Yoon, J.H.; Lee, J.S. Myricetin induces cell death of human colon cancer cells via BAX/BCL2-dependent pathway. Anticancer Res. 2014, 34, 701–706. [Google Scholar]
- Ye, C.; Zhang, C.; Huang, H.; Yang, B.; Xiao, G.; Kong, D.; Tian, Q.; Song, Q.; Song, Y.; Tan, H. The natural compound myricetin effectively represses the malignant progression of prostate cancer by inhibiting PIM1 and disrupting the PIM1/CXCR4 interaction. Cell. Physiol. Biochem. 2018, 48, 1230–1244. [Google Scholar] [CrossRef]
- Knickle, A.; Fernando, W.; Greenshields, A.L.; Rupasinghe, H.V.; Hoskin, D.W. Myricetin-induced apoptosis of triple-negative breast cancer cells is mediated by the iron-dependent generation of reactive oxygen species from hydrogen peroxide. Food Chem. Toxicol. 2018, 118, 154–167. [Google Scholar] [CrossRef]
- Kang, H.R.; Moon, J.Y.; Ediriweera, M.K.; Song, Y.W.; Cho, M.; Kasiviswanathan, D.; Cho, S.K. Dietary flavonoid myricetin inhibits invasion and migration of radioresistant lung cancer cells (A549-IR) by suppressing MMP-2 and MMP-9 expressions through inhibition of the FAK-ERK signaling pathway. Food Sci. Nutr. 2020, 8, 2059–2067. [Google Scholar] [CrossRef]
- Subramani, R.; Gonzalez, E.; Arumugam, A.; Nandy, S.; Gonzalez, V.; Medel, J.; Camacho, F.; Ortega, A.; Bonkoungou, S.; Narayan, M. Nimbolide inhibits pancreatic cancer growth and metastasis through ROS-mediated apoptosis and inhibition of epithelial-to-mesenchymal transition. Sci. Rep. 2016, 6, 1–12. [Google Scholar] [CrossRef]
- Gupta, S.C.; Prasad, S.; Sethumadhavan, D.R.; Nair, M.S.; Mo, Y.-Y.; Aggarwal, B.B. Nimbolide, a limonoid triterpene, inhibits growth of human colorectal cancer xenografts by suppressing the proinflammatory microenvironment. Clin. Cancer Res. 2013, 19, 4465–4476. [Google Scholar] [CrossRef]
- Shin, S.-S.; Hwang, B.; Muhammad, K.; Gho, Y.; Song, J.-H.; Kim, W.-J.; Kim, G.; Moon, S.-K. Nimbolide represses the proliferation, migration, and invasion of bladder carcinoma cells via Chk2-mediated G2/M phase cell cycle arrest, altered signaling pathways, and reduced transcription factors-associated MMP-9 expression. Evid. Based Complement. Alternat. Med. 2019, 2019, 3753587. [Google Scholar] [CrossRef]
- Tian, X.; Liu, M.; Huang, X.; Zhu, Q.; Liu, W.; Chen, W.; Zou, Y.; Cai, Y.; Huang, S.; Chen, A. Noscapine induces apoptosis in human colon cancer cells by regulating mitochondrial damage and warburg effect via PTEN/PI3K/mTOR signaling pathway. Onco Targets Ther. 2020, 13, 5419. [Google Scholar] [CrossRef]
- Quisbert-Valenzuela, E.O.; Calaf, G.M. Apoptotic effect of noscapine in breast cancer cell lines. Int. J. Oncol. 2016, 48, 2666–2674. [Google Scholar] [CrossRef] [PubMed]
- Jackson, T.; Chougule, M.B.; Ichite, N.; Patlolla, R.R.; Singh, M. Antitumor activity of noscapine in human non-small cell lung cancer xenograft model. Cancer Chemother. Pharmacol. 2008, 63, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Barken, I.; Geller, J.; Rogosnitzky, M. Prophylactic noscapine therapy inhibits human prostate cancer progression and metastasis in a mouse model. Anticancer Res. 2010, 30, 399–401. [Google Scholar] [PubMed]
- Yao, Z.; Xie, F.; Li, M.; Liang, Z.; Xu, W.; Yang, J.; Liu, C.; Li, H.; Zhou, H.; Qu, L.-H. Oridonin induces autophagy via inhibition of glucose metabolism in p53-mutated colorectal cancer cells. Cell Death Dis. 2017, 8, e2633. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Chen, W.; Liu, K.; Zhang, S.; Yang, R.; Liu, K.; Li, D.; Huang, Y. Oridonin sensitizes hepatocellular carcinoma to the anticancer effect of sorafenib by targeting the Akt pathway. Cancer Manag. Res. 2020, 12, 8081. [Google Scholar] [CrossRef]
- Wang, Y.; Zhu, Z. Oridonin inhibits metastasis of human ovarian cancer cells by suppressing the mTOR pathway. Arch. Med. Sci. 2019, 15, 1017. [Google Scholar] [CrossRef]
- Che, X.; Zhan, J.; Zhao, F.; Zhong, Z.; Chen, M.; Han, R.; Wang, Y. Oridonin Promotes Apoptosis and Restrains the Viability and Migration of Bladder Cancer by Impeding TRPM7 Expression via the ERK and AKT Signaling Pathways. Biomed Res. Int. 2021, 2021, 4340950. [Google Scholar] [CrossRef]
- Song, M.; Liu, X.; Liu, K.; Zhao, R.; Huang, H.; Shi, Y.; Zhang, M.; Zhou, S.; Xie, H.; Chen, H. Targeting AKT with oridonin inhibits growth of esophageal squamous cell carcinoma In Vitro and patient-derived xenografts In Vivo. Mol. Cancer Ther. 2018, 17, 1540–1553. [Google Scholar] [CrossRef]
- Wang, S.; Zhong, Z.; Wan, J.; Tan, W.; Wu, G.; Chen, M.; Wang, Y. Oridonin induces apoptosis, inhibits migration and invasion on highly-metastatic human breast cancer cells. Am. J. Chin. Med. 2013, 41, 177–196. [Google Scholar] [CrossRef]
- Lu, Y.; Sun, Y.; Zhu, J.; Yu, L.; Jiang, X.; Zhang, J.; Dong, X.; Ma, B.; Zhang, Q. Oridonin exerts anticancer effect on osteosarcoma by activating PPAR-γ and inhibiting Nrf2 pathway. Cell Death Dis. 2018, 9, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.J.; Guo, Y.J.; Yang, X.L.; Ou, Z.L. Anti-cervical cancer role of matrine, oxymatrine and sophora flavescens alkaloid gels and its mechanism. J. Cancer 2018, 9, 1357. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Yang, T. Oxymatrine inhibits the proliferation and invasion of breast cancer cells via the PI3K pathway. Cancer Manag. Res. 2019, 11, 10499. [Google Scholar] [CrossRef] [PubMed]
- Ling, Q.; Xu, X.; Wei, X.; Wang, W.; Zhou, B.; Wang, B.; Zheng, S. Oxymatrine induces human pancreatic cancer PANC-1 cells apoptosis via regulating expression of Bcl-2 and IAP families, and releasing of cytochrome c. J. Exp. Clin. Cancer Res. 2011, 30, 66. [Google Scholar] [CrossRef]
- Wu, C.; Huang, W.; Guo, Y.; Xia, P.; Sun, X.; Pan, X.; Hu, W. Oxymatrine inhibits the proliferation of prostate cancer cells In Vitro and In Vivo. Mol. Med. Rep. 2015, 11, 4129–4134. [Google Scholar] [CrossRef]
- Zhao, X.; Huang, L.; Xu, W.; Chen, X.; Shen, Y.; Zeng, W.; Chen, X. Physapubescin B inhibits tumorgenesis and circumvents taxol resistance of ovarian cancer cells through STAT3 signaling. Oncotarget 2017, 8, 70130. [Google Scholar] [CrossRef]
- Chen, L.; Xia, G.; Qiu, F.; Wu, C.; Denmon, A.P.; Zi, X. Physapubescin selectively induces apoptosis in VHL-null renal cell carcinoma cells through down-regulation of HIF-2α and inhibits tumor growth. Sci. Rep. 2016, 6, 1–12. [Google Scholar] [CrossRef]
- Jaudan, A.; Sharma, S.; Malek, S.N.A.; Dixit, A. Induction of apoptosis by pinostrobin in human cervical cancer cells: Possible mechanism of action. PLoS ONE 2018, 13, e0191523. [Google Scholar] [CrossRef]
- Jones, A.A.; Gehler, S. Acacetin and pinostrobin inhibit malignant breast epithelial cell adhesion and focal adhesion formation to attenuate cell migration. Integr. Cancer Ther. 2020, 19, 1534735420918945. [Google Scholar] [CrossRef]
- Roman, W.A.; Gomes, D.B.; Zanchet, B.; Schönell, A.P.; Diel, K.A.; Banzato, T.P.; Ruiz, A.L.; Carvalho, J.E.; Neppel, A.; Barison, A. Antiproliferative effects of pinostrobin and 5, 6-dehydrokavain isolated from leaves of Alpinia zerumbet. Rev. Bras. Farmacogn. 2017, 27, 592–598. [Google Scholar]
- Haddad, A.; Venkateswaran, V.; Viswanathan, L.; Teahan, S.; Fleshner, N.; Klotz, L. Novel antiproliferative flavonoids induce cell cycle arrest in human prostate cancer cell lines. Prostate Cancer Prostatic Dis. 2006, 9, 68–76. [Google Scholar] [CrossRef] [PubMed]
- de Almeida, G.C.; Oliveira, L.F.; Predes, D.; Fokoue, H.H.; Kuster, R.M.; Oliveira, F.L.; Mendes, F.A.; Abreu, J.G. Piperine suppresses the Wnt/β-catenin pathway and has anti-cancer effects on colorectal cancer cells. Sci. Rep. 2020, 10, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Xu, J.; Liao, H.; Li, L.; Pan, L. Piperine induces apoptosis of lung cancer A549 cells via p53-dependent mitochondrial signaling pathway. Tumor Biol. 2014, 35, 3305–3310. [Google Scholar] [CrossRef] [PubMed]
- Lai, L.H.; Fu, Q.H.; Liu, Y.; Jiang, K.; Guo, Q.M.; Chen, Q.Y.; Yan, B.; Wang, Q.Q.; Shen, J.G. Piperine suppresses tumor growth and metastasis In Vitro and In Vivo in a 4T1 murine breast cancer model. Acta Pharmacol. Sin. 2012, 33, 523–530. [Google Scholar] [CrossRef]
- Ouyang, D.-Y.; Zeng, L.H.; Pan, H.; Xu, L.H.; Wang, Y.; Liu, K.P.; He, X.H. Piperine inhibits the proliferation of human prostate cancer cells via induction of cell cycle arrest and autophagy. Food Chem. Toxicol. 2013, 60, 424–430. [Google Scholar] [CrossRef]
- Zhang, C.; He, L.-J.; Zhu, Y.-B.; Fan, Q.-Z.; Miao, D.-D.; Zhang, S.-P.; Zhao, W.-Y.; Liu, X.-P. Piperlongumine inhibits Akt phosphorylation to reverse resistance to cisplatin in human non-small cell lung cancer cells via ROS regulation. Front. Pharmacol. 2019, 10, 1178. [Google Scholar] [CrossRef]
- Zhang, D.F.; Yang, Z.C.; Chen, J.Q.; Jin, X.X.; Chen, X.J.; Shi, H.Y.; Liu, Z.G.; Wang, M.S.; Liang, G.; Zheng, X.H. Piperlongumine inhibits migration and proliferation of castration-resistant prostate cancer cells via triggering persistent DNA damage. BMC Complement. Med. Ther. 2021, 21, 1–15. [Google Scholar] [CrossRef]
- Machado, F.D.S.; Munari, F.M.; Scariot, F.J.; Echeverrigaray, S.; Aguzzoli, C.; Pich, C.T.; Kato, M.J.; Yamaguchi, L.; Moura, S.; Henriques, J.A.P. Piperlongumine induces apoptosis in colorectal cancer HCT 116 cells independent of Bax, p21 and p53 status. Anticancer Res. 2018, 38, 6231–6236. [Google Scholar] [CrossRef]
- Zhang, X.; Yang, C.; Rao, X.; Xiong, J. Plumbagin shows anti-cancer activity in human breast cancer cells by the upregulation of p53 and p21 and suppression of G1 cell cycle regulators. Eur. J. Gynaecol. Oncol. 2016, 37, 30–35. [Google Scholar]
- Eldhose, B.; GUNAwAN, M.; Rahman, M.; Latha, M.S.; Notario, V. Plumbagin reduces human colon cancer cell survival by inducing cell cycle arrest and mitochondria-mediated apoptosis. Int. J. Oncol. 2014, 45, 1913–1920. [Google Scholar] [CrossRef]
- Li, T.; Lv, M.; Chen, X.; Yu, Y.; Zang, G.; Tang, Z. Plumbagin inhibits proliferation and induces apoptosis of hepatocellular carcinoma by downregulating the expression of SIVA. Drug Des. Devel. Ther. 2019, 13, 1289. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Xie, H.; Pan, Y.; Zheng, K.; Xia, Y.; Chen, W. Plumbagin triggers ER stress-mediated apoptosis in prostate cancer cells via induction of ROS. Cell. Physiol. Biochem. 2018, 45, 267–280. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, S.K.; Rengasamy, K.R.; Biswal, B.K. Plumbagin engenders apoptosis in lung cancer cells via caspase-9 activation and targeting mitochondrial-mediated ROS induction. Arch. Pharm. Res. 2020, 43, 242–256. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.-Y.; Yu, J.; Liu, T.-T.; Yang, K.-X.; Yang, L.-Y.; Chen, Q.; Shi, F.; Hao, J.-J.; Cai, Y.; Wang, M.-R. Plumbagin inhibits the proliferation and survival of esophageal cancer cells by blocking STAT3-PLK1-AKT signaling. Cell Death Dis. 2018, 9, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Yan, C.-H.; Li, F.; Ma, Y.-C. Plumbagin shows anticancer activity in human osteosarcoma (MG-63) cells via the inhibition of S-Phase checkpoints and down-regulation of c-myc. Int. J. Clin. Exp. Med. 2015, 8, 14432. [Google Scholar] [PubMed]
- Jaiswal, A.; Sabarwal, A.; Mishra, J.P.N.; Singh, R.P. Plumbagin induces ROS-mediated apoptosis and cell cycle arrest and inhibits EMT in human cervical carcinoma cells. RSC Adv. 2018, 8, 32022–32037. [Google Scholar] [CrossRef]
- Seo, H.W.; Park, J.-H.; Lee, J.Y.; Park, H.-J.; Kim, J.-K. Pristimerin, a Naturally Occurring Triterpenoid, Exerts Potent Anticancer Effect in Colon Cancer Cells. Biomed. Sci. 2018, 24, 15–22. [Google Scholar] [CrossRef]
- Wu, H.; Li, L.; Ai, Z.; Yin, J.; Chen, L. Pristimerin induces apoptosis of oral squamous cell carcinoma cells via G1 phase arrest and MAPK/Erk1/2 and Akt signaling inhibition. Oncol. Lett. 2019, 17, 3017–3025. [Google Scholar] [CrossRef]
- Lee, S.-O.; Kim, J.-S.; Lee, M.-S.; Lee, H.-J. Anti-cancer effect of pristimerin by inhibition of HIF-1α involves the SPHK-1 pathway in hypoxic prostate cancer cells. BMC Cancer 2016, 16, 1–10. [Google Scholar] [CrossRef]
- Li, J.; Guo, Q.; Lei, X.; Zhang, L.; Su, C.; Liu, Y.; Zhou, W.; Chen, H.; Wang, H.; Wang, F. Pristimerin induces apoptosis and inhibits proliferation, migration in H1299 Lung Cancer Cells. J. Cancer 2020, 11, 6348. [Google Scholar] [CrossRef]
- Deeb, D.; Gao, X.; Liu, Y.B.; Pindolia, K.; Gautam, S.C. Pristimerin, a quinonemethide triterpenoid, induces apoptosis in pancreatic cancer cells through the inhibition of pro-survival Akt/NF-κB/mTOR signaling proteins and anti-apoptotic Bcl-2. Int. J. Oncol. 2014, 44, 1707–1715. [Google Scholar] [CrossRef] [PubMed]
- Pei, H.L.; Mu, D.M.; Zhang, B. Anticancer activity of pterostilbene in human ovarian cancer cell lines. Med. Sci. Monit. Basic Res. 2017, 23, 3192. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ma, Z.; Yang, Y.; Di, S.; Feng, X.; Liu, D.; Jiang, S.; Hu, W.; Qin, Z.; Li, Y.; Lv, J. Pterostilbene exerts anticancer activity on non-small-cell lung cancer via activating endoplasmic reticulum stress. Sci. Rep. 2017, 7, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Wakimoto, R.; Ono, M.; Takeshima, M.; Higuchi, T.; Nakano, S. Differential anticancer activity of pterostilbene against three subtypes of human breast cancer cells. Anticancer Res. 2017, 37, 6153–6159. [Google Scholar]
- Zhang, Y.; Li, Y.; Sun, C.; Chen, X.; Han, L.; Wang, T.; Liu, J.; Chen, X.; Zhao, D. Effect of Pterostilbene, a Natural Derivative of Resveratrol, in the Treatment of Colorectal Cancer through Top1/Tdp1-Mediated DNA Repair Pathway. Cancers 2021, 13, 4002. [Google Scholar] [CrossRef]
- Yu, Z.; Li, W. Induction of apoptosis by puerarin in colon cancer HT-29 cells. Cancer Lett. 2006, 238, 53–60. [Google Scholar] [CrossRef]
- Li, J.; Xiong, C.; Xu, P.; Luo, Q.; Zhang, R. Puerarin induces apoptosis in prostate cancer cells via inactivation of the Keap1/Nrf2/ARE signaling pathway. Bioengineered 2021, 12, 402–413. [Google Scholar] [CrossRef]
- Hu, Y.; Li, X.; Lin, L.; Liang, S.; Yan, J. Puerarin inhibits non-small cell lung cancer cell growth via the induction of apoptosis. Oncol. Rep. 2018, 39, 1731–1738. [Google Scholar] [CrossRef]
- Zhang, W.-G.; Yin, X.-C.; Liu, X.-F.; Meng, K.-W.; Tang, K.; Huang, F.-L.; Xu, G.; Gao, J. Puerarin induces hepatocellular carcinoma cell apoptosis modulated by MAPK signaling pathways in a dose-dependent manner. Anticancer Res. 2017, 37, 4425–4431. [Google Scholar]
- Yang, J.-A.; Li, J.-Q.; Shao, L.-M.; Yang, Q.; Liu, B.-H.; Wu, T.-F.; Wu, P.; Yi, W.; Chen, Q.-X. Puerarin inhibits proliferation and induces apoptosis in human glioblastoma cell lines. Int. J. Clin. Exp. Med. 2015, 8, 10132. [Google Scholar]
- Hong, Y.; Lee, J.; Moon, H.; Ryu, C.H.; Seok, J.; Jung, Y.; Ryu, J.; Baek, S.J. Quercetin Induces Anticancer Activity by Upregulating Pro-NAG-1/GDF15 in Differentiated Thyroid Cancer Cells. Cancers 2021, 13, 3022. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, H.A.; Sulaiman, G.M.; Anwar, S.S.; Tawfeeq, A.T.; Khan, R.A.; Mohammed, S.A.; Al-Omar, M.S.; Alsharidah, M.; Rugaie, O.A.; Al-Amiery, A.A. Quercetin against MCF7 and CAL51 breast cancer cell lines: Apoptosis, gene expression and cytotoxicity of nano-quercetin. Nanomedicine 2021, 16, 1937–1961. [Google Scholar] [CrossRef] [PubMed]
- Hisaka, T.; Sakai, H.; Sato, T.; Goto, Y.; Nomura, Y.; Fukutomi, S.; Fujita, F.; Mizobe, T.; Nakashima, O.; Tanigawa, M. Quercetin suppresses proliferation of liver cancer cell lines In Vitro. Anticancer Res. 2020, 40, 4695–4700. [Google Scholar] [CrossRef] [PubMed]
- Nair, H.K.; Rao, K.V.; Aalinkeel, R.; Mahajan, S.; Chawda, R.; Schwartz, S.A. Inhibition of prostate cancer cell colony formation by the flavonoid quercetin correlates with modulation of specific regulatory genes. Clin. Vaccine Immunol. 2004, 11, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Youn, H.; Jeong, J.-C.; Jeong, Y.S.; Kim, E.-J.; Um, S.-J. Quercetin potentiates apoptosis by inhibiting nuclear factor-kappaB signaling in H460 lung cancer cells. Biol. Pharm. Bull. 2013, 36, 944–951. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.-H.; Zhou, L.-Y.; Chen, Q.-Z.; Li, Y.; Shao, Y.; Ren, W.-Y.; Liao, Y.-P.; Wang, H.; Zhu, J.-H.; Huang, M. Resveratrol inactivates PI3K/Akt signaling through upregulating BMP7 in human colon cancer cells. Oncol. Rep. 2017, 38, 456–464. [Google Scholar] [CrossRef]
- Miksits, M.; Wlcek, K.; Svoboda, M.; Kunert, O.; Haslinger, E.; Thalhammer, T.; Szekeres, T.; Jäger, W. Antitumor activity of resveratrol and its sulfated metabolites against human breast cancer cells. Planta Med. 2009, 75, 1227–1230. [Google Scholar] [CrossRef]
- Zhong, L.; Zhang, Y.; Wu, M.; Liu, Y.; Zhang, P.; Chen, X.; Kong, Q.; Liu, J.; Li, H. Resveratrol and STAT inhibitor enhance autophagy in ovarian cancer cells. Cell Death Discov. 2016, 2, 1–8. [Google Scholar] [CrossRef]
- Cheng, L.; Yan, B.; Chen, K.; Jiang, Z.; Zhou, C.; Cao, J.; Qian, W.; Li, J.; Sun, L.; Ma, J. Resveratrol-induced downregulation of NAF-1 enhances the sensitivity of pancreatic cancer cells to gemcitabine via the ROS/Nrf2 signaling pathways. Oxid. Med. Cell. Longev. 2018, 2018. [Google Scholar] [CrossRef]
- Wu, X.; Xu, Y.; Zhu, B.; Liu, Q.; Yao, Q.; Zhao, G. Resveratrol induces apoptosis in SGC-7901 gastric cancer cells. Oncol. Lett. 2018, 16, 2949–2956. [Google Scholar] [CrossRef]
- Yousef, M.; Vlachogiannis, I.A.; Tsiani, E. Effects of resveratrol against lung cancer: In vitro and In Vivo studies. Nutrients 2017, 9, 1231. [Google Scholar] [CrossRef] [PubMed]
- Chao, S.-C.; Chen, Y.-J.; Huang, K.-H.; Kuo, K.-L.; Yang, T.-H.; Huang, K.-Y.; Wang, C.-C.; Tang, C.-H.; Yang, R.-S.; Liu, S.-H. Induction of sirtuin-1 signaling by resveratrol induces human chondrosarcoma cell apoptosis and exhibits antitumor activity. Sci. Rep. 2017, 7, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Jayameena, P.; Sivakumari, K.; Ashok, K.; Rajesh, S. Rutin: A potential anticancer drug against human colon cancer (HCT116) cells. Int. J. Biol. Pharm. Allied Sci. 2018, 7, 1731–1745. [Google Scholar]
- Pinzaru, I.; Chioibas, R.; Marcovici, I.; Coricovac, D.; Susan, R.; Predut, D.; Georgescu, D.; Dehelean, C. Rutin Exerts Cytotoxic and Senescence-Inducing Properties in Human Melanoma Cells. Toxics 2021, 9, 226. [Google Scholar] [CrossRef] [PubMed]
- Elsayed, H.E.; Ebrahim, H.Y.; Mohyeldin, M.M.; Siddique, A.B.; Kamal, A.M.; Haggag, E.G.; El Sayed, K.A. Rutin as a novel c-Met inhibitory lead for the control of triple negative breast malignancies. Nutr. Cancer 2017, 69, 1256–1271. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, Y.; Guo, J.; Cui, H.; Liu, S. Anticancer activity of safranal against colon carcinoma is due to induction of apoptosis and G2/M cell cycle arrest mediated by suppression of mTOR/PI3K/Akt pathway. JBU ON 2018, 23, 574–578. [Google Scholar]
- Chaiboonchoe, A.; Khraiwesh, B.; Murali, C.; Baig, B.; El-Awady, R.; Tarazi, H.; Alzahmi, A.; Nelson, D.R.; Greish, Y.E.; Ramadan, W. Safranal induces DNA double-strand breakage and ER-stress-mediated cell death in hepatocellular carcinoma cells. Sci. Rep. 2018, 8, 1–15. [Google Scholar]
- Jiang, X.; Li, Y.; Feng, J.L.; Nik Nabil, W.N.; Wu, R.; Lu, Y.; Liu, H.; Xi, Z.C.; Xu, H.X. Safrana l prevents prostate cancer recurrence by blocking the Re-activation of quiescent cancer cells via downregulation of S-phase kinase-associated protein 2. Front. Cell Dev. Biol. 2020, 8, 1553. [Google Scholar] [CrossRef]
- Chryssanthi, D.G.; Lamari, F.N.; Iatrou, G.; Pylara, A.; Karamanos, N.K.; Cordopatis, P. Inhibition of breast cancer cell proliferation by style constituents of different Crocus species. Anticancer Res. 2007, 27, 357–362. [Google Scholar]
- Sha, L.; Lv, Z.; Liu, Y.; Zhang, Y.; Sui, X.; Wang, T.; Zhang, H. Shikonin inhibits the Warburg effect, cell proliferation, invasion and migration by downregulating PFKFB2 expression in lung cancer. Mol. Med. Rep. 2021, 24, 1–10. [Google Scholar] [CrossRef]
- Chandimali, N.; Sun, H.-N.; Kong, L.-Z.; Zhen, X.; Liu, R.; Kwon, T.; Lee, D.-S. Shikonin-induced apoptosis of colon cancer cells is reduced by peroxiredoxin V expression. Anticancer Res. 2019, 39, 6115–6123. [Google Scholar] [CrossRef] [PubMed]
- Markowitsch, S.D.; Juetter, K.M.; Schupp, P.; Hauschulte, K.; Vakhrusheva, O.; Slade, K.S.; Thomas, A.; Tsaur, I.; Cinatl, J.; Michaelis, M. Shikonin Reduces Growth of Docetaxel-Resistant Prostate Cancer Cells Mainly through Necroptosis. Cancers 2021, 13, 882. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Li, S.; Wu, L.; Yu, Q.; Li, J.; Feng, J.; Zhang, J.; Chen, J.; Zhou, Y.; Ji, J. Experimental study of hepatocellular carcinoma treatment by shikonin through regulating PKM2. J. Hepatocell. Carcinoma 2020, 7, 19. [Google Scholar] [CrossRef] [PubMed]
- Shilnikova, K.; Piao, M.J.; Kang, K.A.; Ryu, Y.S.; Park, J.E.; Hyun, Y.J.; Zhen, A.X.; Jeong, Y.J.; Jung, U.; Kim, I.G. Shikonin induces mitochondria-mediated apoptosis and attenuates epithelial-mesenchymal transition in cisplatin-resistant human ovarian cancer cells. Oncol. Lett. 2018, 15, 5417–5424. [Google Scholar] [CrossRef]
- Lee, J.H.; Han, S.H.; Kim, Y.M.; Kim, S.H.; Yoo, E.S.; Woo, J.S.; Jung, G.H.; Jung, S.H.; Kim, B.S.; Jung, J.Y. Shikonin inhibits proliferation of melanoma cells by MAPK pathway-mediated induction of apoptosis. Biosci. Rep. 2021, 41, BSR20203834. [Google Scholar] [CrossRef]
- Thonsri, U.; Seubwai, W.; Waraasawapati, S.; Wongkham, S.; Boonmars, T.; Cha’on, U.; Wongkham, C. Antitumor Effect of Shikonin, a PKM2 Inhibitor, in Cholangiocarcinoma Cell Lines. Anticancer Res. 2020, 40, 5115–5124. [Google Scholar] [CrossRef]
- Hou, Y.; Guo, T.; Wu, C.; He, X.; Zhao, M. Effect of shikonin on human breast cancer cells proliferation and apoptosis In Vitro. Yakugaku Zasshi 2006, 126, 1383–1386. [Google Scholar] [CrossRef]
- Bawadood, A.S.; Al-Abbasi, F.A.; Anwar, F.; El-Halawany, A.M.; Al-Abd, A.M. 6-Shogaol suppresses the growth of breast cancer cells by inducing apoptosis and suppressing autophagy via targeting notch signaling pathway. Biomed. Pharmacother. 2020, 128, 110302. [Google Scholar] [CrossRef]
- Saha, A.; Blando, J.; Silver, E.; Beltran, L.; Sessler, J.; DiGiovanni, J. 6-Shogaol from dried ginger inhibits growth of prostate cancer cells both In Vitro and In Vivo through inhibition of STAT3 and NF-κB signaling. Cancer Prev. Res. 2014, 7, 627–638. [Google Scholar] [CrossRef]
- Hsu, Y.-L.; Hung, J.-Y.; Tsai, Y.-M.; Tsai, E.-M.; Huang, M.-S.; Hou, M.-F.; Kuo, P.-L. 6-shogaol, an active constituent of dietary ginger, impairs cancer development and lung metastasis by inhibiting the secretion of CC-chemokine ligand 2 (CCL2) in tumor-associated dendritic cells. J. Agric. Food Chem. 2015, 63, 1730–1738. [Google Scholar] [CrossRef]
- Liu, Q.; Peng, Y.-B.; Qi, L.-W.; Cheng, X.-L.; Xu, X.J.; Liu, L.-L.; Liu, E.-H.; Li, P. The cytotoxicity mechanism of 6-shogaol-treated HeLa human cervical cancer cells revealed by label-free shotgun proteomics and bioinformatics analysis. Evid. Based Complement. Alternat. Med. 2012, 2012, 278652. [Google Scholar] [CrossRef] [PubMed]
- Kil, W.H.; Kim, S.M.; Lee, J.E.; Park, K.S.; Nam, S.J. Anticancer effect of silibinin on the xenograft model using MDA-MB-468 breast cancer cells. Ann. Surg. Treat. Res. 2014, 87, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Hou, X.; Du, H.; Quan, X.; Shi, L.; Zhang, Q.; Wu, Y.; Liu, Y.; Xiao, J.; Li, Y.; Lu, L. Silibinin inhibits NSCLC metastasis by targeting the EGFR/LOX pathway. Front. Pharmacol. 2018, 9, 21. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.J.; Suh, D.S.; Moon, S.H.; Song, Y.J.; Yoon, M.S.; Park, D.Y.; Choi, K.U.; Kim, Y.K.; Kim, K.H. Silibinin inhibits tumor growth through downregulation of extracellular signal-regulated kinase and Akt In Vitro and In Vivo in human ovarian cancer cells. J. Agric. Food Chem. 2013, 61, 4089–4096. [Google Scholar] [CrossRef]
- Deep, G.; Singh, R.; Agarwal, C.; Kroll, D.; Agarwal, R. Silymarin and silibinin cause G1 and G2–M cell cycle arrest via distinct circuitries in human prostate cancer PC3 cells: A comparison of flavanone silibinin with flavanolignan mixture silymarin. Oncogene 2006, 25, 1053–1069. [Google Scholar] [CrossRef]
- Sati, J.; Mohanty, B.P.; Garg, M.L.; Koul, A. Pro-oxidant role of silibinin in DMBA/TPA induced skin cancer: 1H NMR metabolomic and biochemical study. PLoS ONE 2016, 11, e0158955. [Google Scholar] [CrossRef]
- Sameri, S.; Mohammadi, C.; Mehrabani, M.; Najafi, R. Targeting the hallmarks of cancer: The effects of silibinin on proliferation, cell death, angiogenesis, and migration in colorectal cancer. BMC Complement. Med. Ther. 2021, 21, 1–9. [Google Scholar] [CrossRef]
- Wang, Y.-X.; Cai, H.; Jiang, G.; Zhou, T.-B.; Wu, H. Silibinin inhibits proliferation, induces apoptosis and causes cell cycle arrest in human gastric cancer MGC803 cells via STAT3 pathway inhibition. Asian Pac. J. Cancer Prev. 2014, 15, 6791–6798. [Google Scholar] [CrossRef]
- Kim, S.H.; Choo, G.S.; Yoo, E.S.; Woo, J.S.; Han, S.H.; Lee, J.H.; Jung, J.Y. Silymarin induces inhibition of growth and apoptosis through modulation of the MAPK signaling pathway in AGS human gastric cancer cells. Oncol. Rep. 2019, 42, 1904–1914. [Google Scholar] [CrossRef]
- Kim, S.-H.; Choo, G.-S.; Yoo, E.-S.; Woo, J.-S.; Lee, J.-H.; Han, S.-H.; Jung, S.-H.; Kim, H.-J.; Jung, J.-Y. Silymarin inhibits proliferation of human breast cancer cells via regulation of the MAPK signaling pathway and induction of apoptosis. Oncol. Lett. 2021, 21, 1–10. [Google Scholar] [CrossRef]
- Wu, Y.-F.; Fu, S.-L.; Kao, C.-H.; Yang, C.-W.; Lin, C.-H.; Hsu, M.-T.; Tsai, T.-F. Chemopreventive effect of silymarin on liver pathology in HBV X protein transgenic mice. Cancer Res. 2008, 68, 2033–2042. [Google Scholar] [CrossRef][Green Version]
- Fu, R.; Wang, X.; Hu, Y.; Du, H.; Dong, B.; Ao, S.; Zhang, L.; Sun, Z.; Zhang, L.; Lv, G. Solamargine inhibits gastric cancer progression by regulating the expression of lncNEAT1_2 via the MAPK signaling pathway. Int. J. Oncol. 2019, 54, 1545–1554. [Google Scholar] [CrossRef]
- Al Sinani, S.S.; Eltayeb, E.A.; Coomber, B.L.; Adham, S.A. Solamargine triggers cellular necrosis selectively in different types of human melanoma cancer cells through extrinsic lysosomal mitochondrial death pathway. Cancer Cell Int. 2016, 16, 1–12. [Google Scholar] [CrossRef]
- Liu, J.; Wang, Z.; Xu, C.; Qi, Y.; Zhang, Q. Solamargine inhibits proliferation and promotes apoptosis of CM-319 human chordoma cells through suppression of notch pathway. Transl. Cancer Res. 2019, 8, 509–519. [Google Scholar] [CrossRef]
- Fekry, M.I.; Ezzat, S.M.; Salama, M.M.; Alshehri, O.Y.; Al-Abd, A.M. Bioactive glycoalkaloides isolated from Solanum melongena fruit peels with potential anticancer properties against hepatocellular carcinoma cells. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef]
- Xiang, S.; Zhang, Q.; Tang, Q.; Zheng, F.; Wu, J.; Yang, L.; Hann, S.S. Activation of AMPKα mediates additive effects of solamargine and metformin on suppressing MUC1 expression in castration-resistant prostate cancer cells. Sci. Rep. 2016, 6, 1–14. [Google Scholar] [CrossRef]
- Wang, M.; Shu, Z.-J.; Wang, Y.; Peng, W. Stachydrine hydrochloride inhibits proliferation and induces apoptosis of breast cancer cells via inhibition of Akt and ERK pathways. Am. J. Transl. Res. 2017, 9, 1834. [Google Scholar]
- Rathee, P.; Rathee, D.; Rathee, D.; Rathee, S. In vitro anticancer activity of stachydrine isolated from Capparis decidua on prostate cancer cell lines. Nat. Prod. Res. 2012, 26, 1737–1740. [Google Scholar] [CrossRef]
- Wang, Y.; Shi, L.-Y.; Qi, W.-H.; Yang, J.; Qi, Y. Anticancer activity of sugiol against ovarian cancer cell line SKOV3 involves mitochondrial apoptosis, cell cycle arrest and blocking of the RAF/MEK/ERK signalling pathway. Arch. Med. Sci. 2020, 16, 428. [Google Scholar] [CrossRef]
- Jung, S.-N.; Shin, D.-S.; Kim, H.-N.; Jeon, Y.J.; Yun, J.; Lee, Y.-J.; Kang, J.S.; Han, D.C.; Kwon, B.-M. Sugiol inhibits STAT3 activity via regulation of transketolase and ROS-mediated ERK activation in DU145 prostate carcinoma cells. Biochem. Pharmacol. 2015, 97, 38–50. [Google Scholar] [CrossRef]
- Hao, C.; Zhang, X.; Zhang, H.; Shang, H.; Bao, J.; Wang, H.; Li, Z. Sugiol (12-hydroxyabieta-8, 11, 13-trien-7-one) targets hu-man pancreatic carcinoma cells (Mia-PaCa2) by inducing ap-optosis, G2/M cell cycle arrest, ROS production and inhibi-tion of cancer cell migration. J. BUON 2018, 23, 205–210. [Google Scholar] [PubMed]
- Zhao, H.; Zhang, X. Sugiol suppresses the growth, migration, and invasion of human endometrial cancer cells via induction of apoptosis and autophagy. 3 Biotech 2021, 11, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.-Y.; Sher, H.-F.; Chen, H.-W.; Liu, C.-C.; Chen, C.-H.; Lin, C.-S.; Yang, P.-C.; Tsay, H.-S.; Chen, J.J. Anticancer effects of tanshinone I in human non-small cell lung cancer. Mol. Cancer Ther. 2008, 7, 3527–3538. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Guo, S.; Fang, J.; Peng, B.; Zhang, Y.; Cao, T. Tanshinone IIA inhibits cell proliferation and tumor growth by downregulating STAT3 in human gastric cancer. Exp. Ther. Med. 2018, 16, 2931–2937. [Google Scholar] [CrossRef]
- Li, G.; Shan, C.; Liu, L.; Zhou, T.; Zhou, J.; Hu, X.; Chen, Y.; Cui, H.; Gao, N. Tanshinone IIA inhibits HIF-1α and VEGF expression in breast cancer cells via mTOR/p70S6K/RPS6/4E-BP1 signaling pathway. PLoS ONE 2015, 10, e0117440. [Google Scholar] [CrossRef]
- Zhou, J.; Jiang, Y.-Y.; Wang, X.-X.; Wang, H.-P.; Chen, H.; Wu, Y.-C.; Wang, L.; Pu, X.; Yue, G.-Z.; Zhang, L. Tanshinone IIA suppresses ovarian cancer growth through inhibiting malignant properties and angiogenesis. Ann. Transl. Med. 2020, 8, 1295. [Google Scholar] [CrossRef]
- Chiu, S.-C.; Huang, S.-Y.; Chang, S.-F.; Chen, S.-P.; Chen, C.-C.; Lin, T.-H.; Liu, H.-H.; Tsai, T.-H.; Lee, S.-S.; Pang, C.-Y. Potential therapeutic roles of tanshinone IIA in human bladder cancer cells. Int. J. Mol. Sci. 2014, 15, 15622–15637. [Google Scholar] [CrossRef]
- Li, M.; Wang, G.; Zhang, R.; Duan, S.; Chen, J. Tanshinone IIA inhibits proliferation and activates apoptosis in C4-1 cervical carcinoma cells In Vitro. Biotechnol. Biotechnol. Equip. 2019, 33, 1599–1607. [Google Scholar] [CrossRef]
- Park, M.H.; Hong, J.E.; Park, E.S.; Yoon, H.S.; Seo, D.W.; Hyun, B.K.; Han, S.-B.; Ham, Y.W.; Hwang, B.Y.; Hong, J.T. Anticancer effect of tectochrysin in colon cancer cell via suppression of NF-kappaB activity and enhancement of death receptor expression. Mol. Cancer 2015, 14, 1–12. [Google Scholar] [CrossRef]
- Wang, Y.; Ke, R.-J.; Jiang, P.-R.; Ying, J.-H.; Lou, E.-Z.; Chen, J.-Y. The effects of tectochrysin on prostate cancer cells apoptosis and its mechanism. Chin. J. Appl. Physiol. 2019, 35, 283. [Google Scholar]
- Oh, S.-B.; Hwang, C.J.; Song, S.-Y.; Jung, Y.Y.; Yun, H.-M.; Sok, C.H.; Sung, H.C.; Yi, J.-M.; Park, D.H.; Ham, Y.W. Anti-cancer effect of tectochrysin in NSCLC cells through overexpression of death receptor and inactivation of STAT3. Cancer Lett. 2014, 353, 95–103. [Google Scholar] [CrossRef]
- Zhang, H.; Xie, B.; Zhang, Z.; Sheng, X.; Zhang, S. Tetrandrine suppresses cervical cancer growth by inducing apoptosis In Vitro and In Vivo. Drug Des. Devel. Ther. 2019, 13, 119. [Google Scholar] [CrossRef]
- Wang, C.H.; Yang, J.M.; Guo, Y.B.; Shen, J.; Pei, X.H. Anticancer activity of tetrandrine by inducing apoptosis in human breast cancer cell line MDA-MB-231 In Vivo. Evid. Based Complement. Alternat. Med. 2020, 2020, 6823520. [Google Scholar] [CrossRef]
- Qin, R.; Shen, H.; Cao, Y.; Fang, Y.; Li, H.; Chen, Q.; Xu, W. Tetrandrine induces mitochondria-mediated apoptosis in human gastric cancer BGC-823 cells. PLoS ONE 2013, 8, e76486. [Google Scholar] [CrossRef]
- Tsai, S.-C.; Wu, W.-C.; Yang, J.-S. Tetrandrine Inhibits Epithelial-Mesenchymal Transition in IL-6-Induced HCT116 Human Colorectal Cancer Cells. Onco Targets Ther. 2021, 14, 4523. [Google Scholar] [CrossRef]
- Shishodia, G.; Koul, S.; Dong, Q.; Koul, H.K. Tetrandrine (TET) induces death receptors Apo Trail R1 (DR4) and Apo Trail R2 (DR5) and sensitizes prostate cancer cells to TRAIL-induced apoptosis. Mol. Cancer Ther. 2018, 17, 1217–1228. [Google Scholar] [CrossRef]
- Wang, N.; Yang, S.; Tan, T.; Huang, Y.; Chen, Y.; Dong, C.; Chen, J.; Luo, X. Tetrandrine suppresses the growth of human osteosarcoma cells by regulating multiple signaling pathways. Bioengineered 2021, 12, 5870–5882. [Google Scholar] [CrossRef]
- Elbe, H.; Yigitturk, G.; Cavusoglu, T.; Uyanikgil, Y.; Ozturk, F. Apoptotic effects of thymol, a novel monoterpene phenol, on different types of cancer. Bratisl. Lek. Listy 2020, 121, 122–128. [Google Scholar] [CrossRef]
- Zeng, Q.; Che, Y.; Zhang, Y.; Chen, M.; Guo, Q.; Zhang, W. Thymol Isolated from Thymus vulgaris L. inhibits colorectal cancer cell growth and metastasis by suppressing the Wnt/β-catenin pathway. Drug Des. Devel. Ther. 2020, 14, 2535. [Google Scholar] [CrossRef]
- Kang, S.-H.; Kim, Y.-S.; Kim, E.-K.; Hwang, J.-W.; Jeong, J.-H.; Dong, X.; Lee, J.-W.; Moon, S.-H.; Jeon, B.-T.; Park, P.-J. Anticancer effect of thymol on AGS human gastric carcinoma cells. J. Microbiol. Biotechnol. 2016, 26, 28–37. [Google Scholar] [CrossRef]
- Dera, A.; Rajagopalan, P. Thymoquinone attenuates phosphorylation of AKT to inhibit kidney cancer cell proliferation. J. Food Biochem. 2019, 43, e12793. [Google Scholar] [CrossRef]
- Woo, C.C.; Hsu, A.; Kumar, A.P.; Sethi, G.; Tan, K.H.B. Thymoquinone inhibits tumor growth and induces apoptosis in a breast cancer xenograft mouse model: The role of p38 MAPK and ROS. PLoS ONE 2013, 8, e75356. [Google Scholar] [CrossRef]
- Iskender, B.; Izgi, K.; Hizar, E.; Jauch, J.; Arslanhan, A.; Yuksek, E.H.; Canatan, H. Inhibition of epithelial-mesenchymal transition in bladder cancer cells via modulation of mTOR signalling. Tumor Biol. 2016, 37, 8281–8291. [Google Scholar] [CrossRef]
- Kundu, J.; Choi, B.Y.; Jeong, C.-H.; Kundu, J.K.; Chun, K.-S. Thymoquinone induces apoptosis in human colon cancer HCT116 cells through inactivation of STAT3 by blocking JAK2-and Src-mediated phosphorylation of EGF receptor tyrosine kinase. Oncol. Rep. 2014, 32, 821–828. [Google Scholar] [CrossRef]
- Zhu, W.-Q.; Wang, J.; Guo, X.-F.; Liu, Z.; Dong, W.-G. Thymoquinone inhibits proliferation in gastric cancer via the STAT3 pathway In Vivo and In Vitro. World J. Gastroenterol. 2016, 22, 4149. [Google Scholar] [CrossRef]
- Ashour, A.E.; Abd-Allah, A.R.; Korashy, H.M.; Attia, S.M.; Alzahrani, A.Z.; Saquib, Q.; Bakheet, S.A.; Abdel-Hamied, H.E.; Jamal, S.; Rishi, A.K. Thymoquinone suppression of the human hepatocellular carcinoma cell growth involves inhibition of IL-8 expression, elevated levels of TRAIL receptors, oxidative stress and apoptosis. Mol. Cell. Biochem. 2014, 389, 85–98. [Google Scholar] [CrossRef]
- Yang, J.; Kuang, X.R.; Lv, P.T.; Yan, X.X. Thymoquinone inhibits proliferation and invasion of human nonsmall-cell lung cancer cells via ERK pathway. Tumor Biol. 2015, 36, 259–269. [Google Scholar] [CrossRef]
- Abdelfadil, E.; Cheng, Y.-H.; Bau, D.-T.; Ting, W.-J.; Chen, L.-M.; Hsu, H.-H.; Lin, Y.-M.; Chen, R.-J.; Tsai, F.-J.; Tsai, C.-H. Thymoquinone induces apoptosis in oral cancer cells through p38β inhibition. Am. J. Chin. Med. 2013, 41, 683–696. [Google Scholar] [CrossRef]
- Torres, M.P.; Ponnusamy, M.P.; Chakraborty, S.; Smith, L.M.; Das, S.; Arafat, H.A.; Batra, S.K. Effects of thymoquinone in the expression of mucin 4 in pancreatic cancer cells: Implications for the development of novel cancer therapies. Mol. Cancer Ther. 2010, 9, 1419–1431. [Google Scholar] [CrossRef]
- Lin, W.; Ye, H. Anticancer activity of ursolic acid on human ovarian cancer cells via ROS and MMP mediated apoptosis, cell cycle arrest and downregulation of PI3K/AKT pathway. J. BUON 2020, 25, 750–756. [Google Scholar]
- Chen, C.-J.; Shih, Y.-L.; Yeh, M.-Y.; Liao, N.-C.; Chung, H.-Y.; Liu, K.-L.; Lee, M.-H.; Chou, P.-Y.; Hou, H.-Y.; Chou, J.-S. Ursolic acid induces apoptotic cell death through AIF and endo G release through a mitochondria-dependent pathway in NCI-H292 human lung cancer cells In Vitro. In Vivo 2019, 33, 383–391. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Zhao, C.; Jou, D.; Lü, J.; Zhang, C.; Lin, L.; Lin, J. Ursolic acid inhibits the growth of colon cancer-initiating cells by targeting STAT3. Anticancer Res. 2013, 33, 4279–4284. [Google Scholar] [PubMed]
- Zhang, X.; Li, T.; Gong, E.S.; Liu, R.H. Antiproliferative activity of ursolic acid in MDA-MB-231 human breast cancer cells through Nrf2 pathway regulation. J. Agric. Food Chem. 2020, 68, 7404–7415. [Google Scholar] [CrossRef] [PubMed]
- Prasad, S.; Yadav, V.R.; Sung, B.; Gupta, S.C.; Tyagi, A.K.; Aggarwal, B.B. Ursolic acid inhibits the growth of human pancreatic cancer and enhances the antitumor potential of gemcitabine in an orthotopic mouse model through suppression of the inflammatory microenvironment. Oncotarget 2016, 7, 13182. [Google Scholar] [CrossRef]
- Weng, H.; Tan, Z.-J.; Hu, Y.-P.; Shu, Y.-J.; Bao, R.-F.; Jiang, L.; Wu, X.-S.; Li, M.-L.; Ding, Q.; Wang, X.-a. Ursolic acid induces cell cycle arrest and apoptosis of gallbladder carcinoma cells. Cancer Cell Int. 2014, 14, 1–10. [Google Scholar] [CrossRef]
- Zúñiga, R.; Concha, G.; Cayo, A.; Cikutović-Molina, R.; Arevalo, B.; González, W.; Catalán, M.A.; Zúñiga, L. Withaferin A suppresses breast cancer cell proliferation by inhibition of the two-pore domain potassium (K2P9) channel TASK-3. Biomed. Pharmacother. 2020, 129, 110383. [Google Scholar] [CrossRef]
- Yang, I.-H.; Kim, L.-H.; Shin, J.-A.; Cho, S.-D. Chemotherapeutic effect of withaferin A in human oral cancer cells. J. Cancer Ther. 2015, 6, 735. [Google Scholar] [CrossRef][Green Version]
- Nagy, Z.; Cheung, B.B.; Tsang, W.; Tan, O.; Herath, M.; Ciampa, O.C.; Shadma, F.; Carter, D.R.; Marshall, G.M. Withaferin A activates TRIM16 for its anti-cancer activity in melanoma. Sci. Rep. 2020, 10, 1–9. [Google Scholar] [CrossRef]
- Chen, Y.; Han, X.Z.; Wang, W.; Zhao, R.T.; Li, X. Withaferin A inhibits osteosarcoma cells through inactivation of Notch-1 signaling. Bangladesh J. Pharmacol. 2014, 9, 364–370. [Google Scholar] [CrossRef]
- Choi, B.Y.; Kim, B.-W. Withaferin-A inhibits colon cancer cell growth by blocking STAT3 transcriptional activity. J. Cancer Prev. 2015, 20, 185. [Google Scholar] [CrossRef]
- Feng, Q.; Wang, H.; Pang, J.; Ji, L.; Han, J.; Wang, Y.; Qi, X.; Liu, Z.; Lu, L. Prevention of wogonin on colorectal cancer tumorigenesis by regulating p53 nuclear translocation. Front. Pharmacol. 2018, 9, 1356. [Google Scholar] [CrossRef]
- Yang, L.; Zhang, H.W.; Hu, R.; Yang, Y.; Qi, Q.; Lu, N.; Liu, W.; Chu, Y.Y.; You, Q.D.; Guo, Q.L. Wogonin induces G1 phase arrest through inhibiting Cdk4 and cyclin D1 concomitant with an elevation in p21Cip1 in human cervical carcinoma HeLa cells. Biochem. Cell Biol. 2009, 87, 933–942. [Google Scholar] [CrossRef]
- Shi, G.; Wang, Q.; Zhou, X.; Li, J.; Liu, H.; Gu, J.; Wang, H.; Wu, Y.; Ding, L.; Ni, S. Response of human non-small-cell lung cancer cells to the influence of Wogonin with SGK1 dynamics. Acta Biochim. Biophys. Sin. 2017, 49, 302–310. [Google Scholar] [CrossRef]
- Koh, H.; Sun, H.-N.; Xing, Z.; Liu, R.; Chandimali, N.; Kwon, T.; Lee, D.-S. Wogonin Influences Osteosarcoma Stem Cell Stemness Through ROS-dependent Signaling. Vivo 2020, 34, 1077–1084. [Google Scholar] [CrossRef]
- Yu, J.S.; Kim, A.K. Wogonin induces apoptosis by activation of ERK and p38 MAPKs signaling pathways and generation of reactive oxygen species in human breast cancer cells. Mol. Cells 2011, 31, 327–335. [Google Scholar] [CrossRef]
- Ruibin, J.; Bo, J.; Danying, W.; Chihong, Z.; Jianguo, F.; Linhui, G. Therapy effects of wogonin on ovarian cancer cells. BioMed Res. Int. 2017, 2017, 9381513. [Google Scholar] [CrossRef]
- Li, W.D.; Wu, Y.; Zhang, L.; Yan, L.G.; Yin, F.Z.; Ruan, J.S.; Chen, Z.P.; Yang, G.M.; Yan, C.P.; Zhao, D. Characterization of xanthatin: Anticancer properties and mechanisms of inhibited murine melanoma In Vitro and In Vivo. Phytomedicine 2013, 20, 865–873. [Google Scholar] [CrossRef]
- Tao, L.; Sheng, X.; Zhang, L.; Li, W.; Wei, Z.; Zhu, P.; Zhang, F.; Wang, A.; Woodgett, J.R.; Lu, Y. Xanthatin anti-tumor cytotoxicity is mediated via glycogen synthase kinase-3β and β-catenin. Biochem. Pharmacol. 2016, 115, 18–27. [Google Scholar] [CrossRef]
- Yu, Y.; Yu, J.; Pei, C.G.; Li, Y.Y.; Tu, P.; Gao, G.P.; Shao, Y. Xanthatin, a novel potent inhibitor of VEGFR2 signaling, inhibits angiogenesis and tumor growth in breast cancer cells. Int. J. Clin. Exp. Pathol. 2015, 8, 10355. [Google Scholar]
- Li, L.; Liu, P.; Xie, Y.; Liu, Y.; Chen, Z.; Geng, Y.; Zhang, L. Xanthatin inhibits human colon cancer cells progression via mTOR signaling mediated energy metabolism alteration. Drug Dev. Res. 2021, 83, 119–130. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs from 1981 to 2014. J. Nat. Prod. 2016, 79, 629–661. [Google Scholar] [CrossRef]
- Vallianou, N.G.; Evangelopoulos, A.; Schizas, N.; Kazazis, C. Potential anticancer properties and mechanisms of action of curcumin. Anticancer Res. 2015, 35, 645–651. [Google Scholar]
- Zheng, B.; McClements, D.J. Formulation of more efficacious curcumin delivery systems using colloid science: Enhanced solubility, stability, and bioavailability. Molecules 2020, 25, 2791. [Google Scholar] [CrossRef]
- Venkatesan, P.; Rao, M. Structure-activity relationships for the inhibition of lipid peroxidation and the scavenging of free radicals by synthetic symmetrical curcumin analogues. J. Pharm. Pharmacol. 2000, 52, 1123–1128. [Google Scholar] [CrossRef]
- Youssef, K.M.; El-Sherbeny, M.A.; El-Shafie, F.S.; Farag, H.A.; Al-Deeb, O.A.; Awadalla, S.A.A. Synthesis of curcumin analogues as potential antioxidant, cancer chemopreventive agents. Arch. Pharm. 2004, 337, 42–54. [Google Scholar] [CrossRef]
- Ohtsu, H.; Itokawa, H.; Xiao, Z.; Su, C.-Y.; Shih, C.C.-Y.; Chiang, T.; Chang, E.; Lee, Y.; Chiu, S.-Y.; Chang, C. Antitumor agents 222. Synthesis and anti-androgen activity of new diarylheptanoids. Biorg. Med. Chem. 2003, 11, 5083–5090. [Google Scholar] [CrossRef]
- Ohtsu, H.; Xiao, Z.; Ishida, J.; Nagai, M.; Wang, H.-K.; Itokawa, H.; Su, C.-Y.; Shih, C.; Chiang, T.; Chang, E. Antitumor agents. 217. Curcumin analogues as novel androgen receptor antagonists with potential as anti-prostate cancer agents. J. Med. Chem. 2002, 45, 5037–5042. [Google Scholar] [CrossRef]
- Itokawa, H.; Shi, Q.; Akiyama, T.; Morris-Natschke, S.L.; Lee, K.H. Recent advances in the investigation of curcuminoids. Chin. Med. 2008, 3, 1–13. [Google Scholar] [CrossRef]
- Lin, L.; Lee, K.-H. Structure-activity relationships of curcumin and its analogs with different biological activities. Stud. Nat. Prod. Chem. 2006, 33, 785–812. [Google Scholar]
- Morris, J.; Moseley, V.R.; Cabang, A.B.; Coleman, K.; Wei, W.; Garrett-Mayer, E.; Wargovich, M.J. Reduction in promotor methylation utilizing EGCG (epigallocatechin-3-gallate) restores RXRα expression in human colon cancer cells. Oncotarget 2016, 7, 35313. [Google Scholar] [CrossRef]
- Lambert, J.D.; Elias, R.J. The antioxidant and pro-oxidant activities of green tea polyphenols: A role in cancer prevention. Arch. Biochem. Biophys. 2010, 501, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Min, K.-j.; Kwon, T.K. Anticancer effects and molecular mechanisms of epigallocatechin-3-gallate. Integr. Med. Res. 2014, 3, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Landis-Piwowar, K.R.; Kuhn, D.J.; Wan, S.B.; Chen, D.; Chan, T.H.; Dou, Q.P. Evaluation of proteasome-inhibitory and apoptosis-inducing potencies of novel (-)-EGCG analogs and their prodrugs. Int. J. Mol. Med. 2005, 15, 735–742. [Google Scholar] [CrossRef]
- Khandelwal, A.; Hall, J.A.; Blagg, B.S. Synthesis and structure–activity relationships of EGCG analogues, a recently identified Hsp90 inhibitor. J. Org. Chem. 2013, 78, 7859–7884. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, S.; Shibata, H.; Ishikawa, F.; Yokokura, T.; Takahashi, M.; Sugimura, T.; Wakabayashi, K. Suppression of Helicobacter pylori-induced gastritis by green tea extract in Mongolian gerbils. Biochem. Biophys. Res. Commun. 2003, 310, 715–719. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, J.; Abbasi, B.A.; Mahmood, T.; Kanwal, S.; Ali, B.; Shah, S.A.; Khalil, A.T. Plant-derived anticancer agents: A green anticancer approach. Asian Pac. J. Trop. Biomed. 2017, 7, 1129–1150. [Google Scholar] [CrossRef]
- Yang, Z.; Kulkarni, K.; Zhu, W.; Hu, M. Bioavailability and pharmacokinetics of genistein: Mechanistic studies on its. Anticancer Agents Med. Chem. 2012, 12, 1264–1280. [Google Scholar] [CrossRef]
- Papaj, K.; Kasprzycka, A.; Góra, A.; Grajoszek, A.; Rzepecka, G.; Stojko, J.; Barski, J.-J.; Szeja, W.; Rusin, A. Structure–bioavailability relationship study of genistein derivatives with antiproliferative activity on human cancer cell. J. Pharm. Biomed. Anal. 2020, 185, 113216. [Google Scholar] [CrossRef]
- Byczek, A.; Zawisza-Puchalka, J.; Gruca, A.; Papaj, K.; Grynkiewicz, G.; Rusin, M.; Szeja, W.; Rusin, A. Genistein derivatives regioisomerically substituted at 7-O-and 4′-O-have different effect on the cell cycle. J. Chem. 2013, 2013, 191563. [Google Scholar] [CrossRef]
- Szeja, W.; Grynkiewicz, G.; Bieg, T.; Swierk, P.; Byczek, A.; Papaj, K.; Kitel, R.; Rusin, A. Synthesis and cytotoxicity of 2, 3-enopyranosyl C-linked conjugates of genistein. Molecules 2014, 19, 7072–7093. [Google Scholar] [CrossRef]
- Nahum, A.; Hirsch, K.; Danilenko, M.; Watts, C.K.; Prall, O.W.; Levy, J.; Sharoni, Y. Lycopene inhibition of cell cycle progression in breast and endometrial cancer cells is associated with reduction in cyclin D levels and retention of p27 Kip1 in the cyclin E–cdk2 complexes. Oncogene 2001, 20, 3428–3436. [Google Scholar] [CrossRef]
- Carini, F.; David, S.; Tomasello, G.; Mazzola, M.; Damiani, P.; Rappa, F.; Battaglia, L.; Cappello, F.; Jurjus, A.; Geagea, A.G. Colorectal cancer: An update on the effects of lycopene on tumor progression and cell proliferation. J. Biol. Regul. Homeost. Agents 2017, 31, 769–774. [Google Scholar] [PubMed]
- Ben-Dor, A.; Steiner, M.; Gheber, L.; Danilenko, M.; Dubi, N.; Linnewiel, K.; Zick, A.; Sharoni, Y.; Levy, J. Carotenoids activate the antioxidant response element transcription system. Mol. Cancer Ther. 2005, 4, 177–186. [Google Scholar]
- Linnewiel, K.; Ernst, H.; Caris-Veyrat, C.; Ben-Dor, A.; Kampf, A.; Salman, H.; Danilenko, M.; Levy, J.; Sharoni, Y. Structure activity relationship of carotenoid derivatives in activation of the electrophile/antioxidant response element transcription system. Free Radic. Biol. Med. 2009, 47, 659–667. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, G.; Li, C.; Wang, S.; Zhu, M.; Wang, J.; Yue, H.; Ma, X.; Zhen, Y.; Shu, X. Metabolic profile and structure–activity relationship of resveratrol and its analogs in human bladder cancer cells. Cancer Manag. Res. 2019, 11, 4631. [Google Scholar] [CrossRef]
- Herath, W.; Khan, S.I.; Khan, I.A. Microbial metabolism. Part 14. Isolation and bioactivity evaluation of microbial metabolites of resveratrol. Nat. Prod. Res. 2013, 27, 1437–1444. [Google Scholar] [CrossRef]
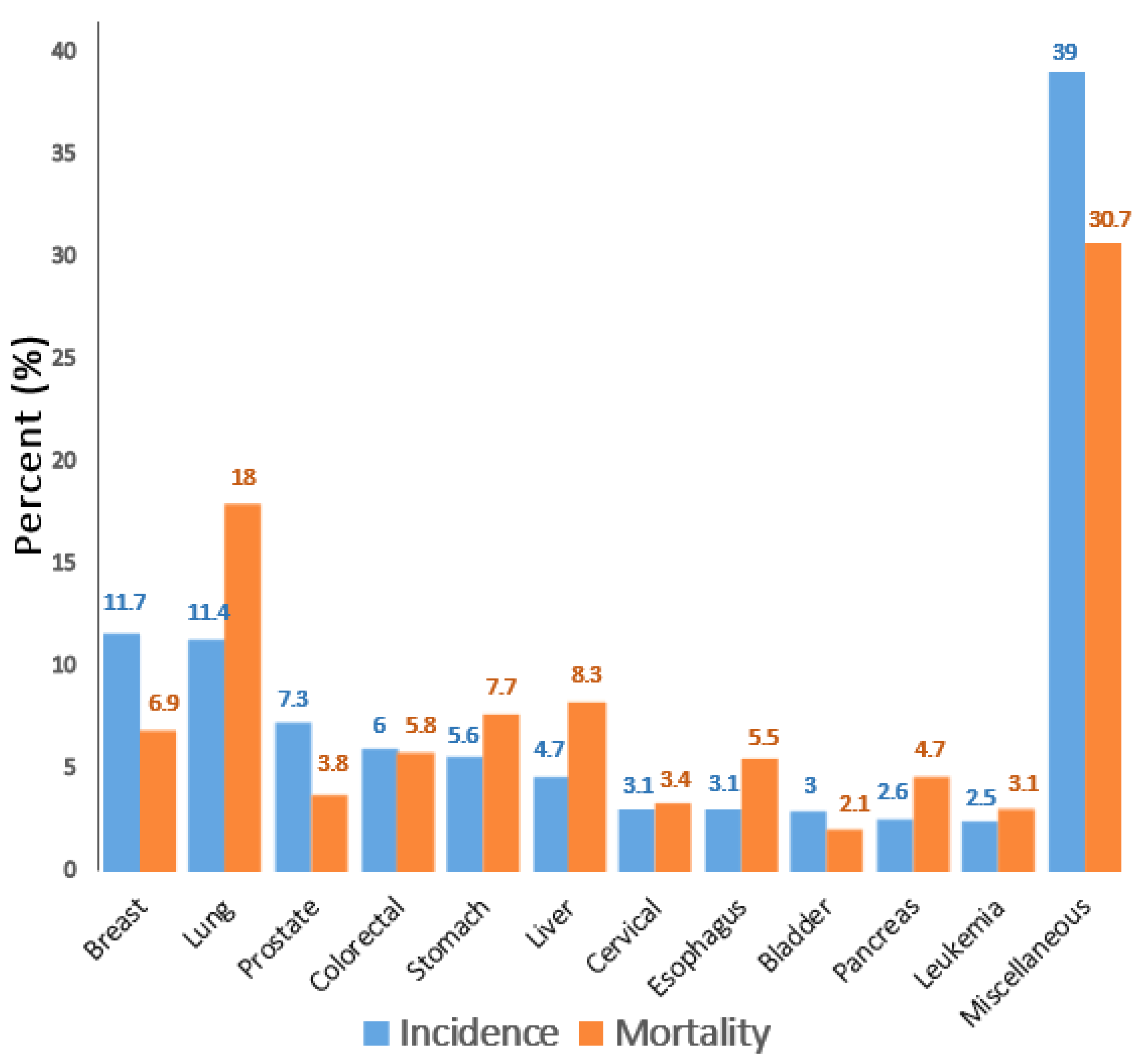
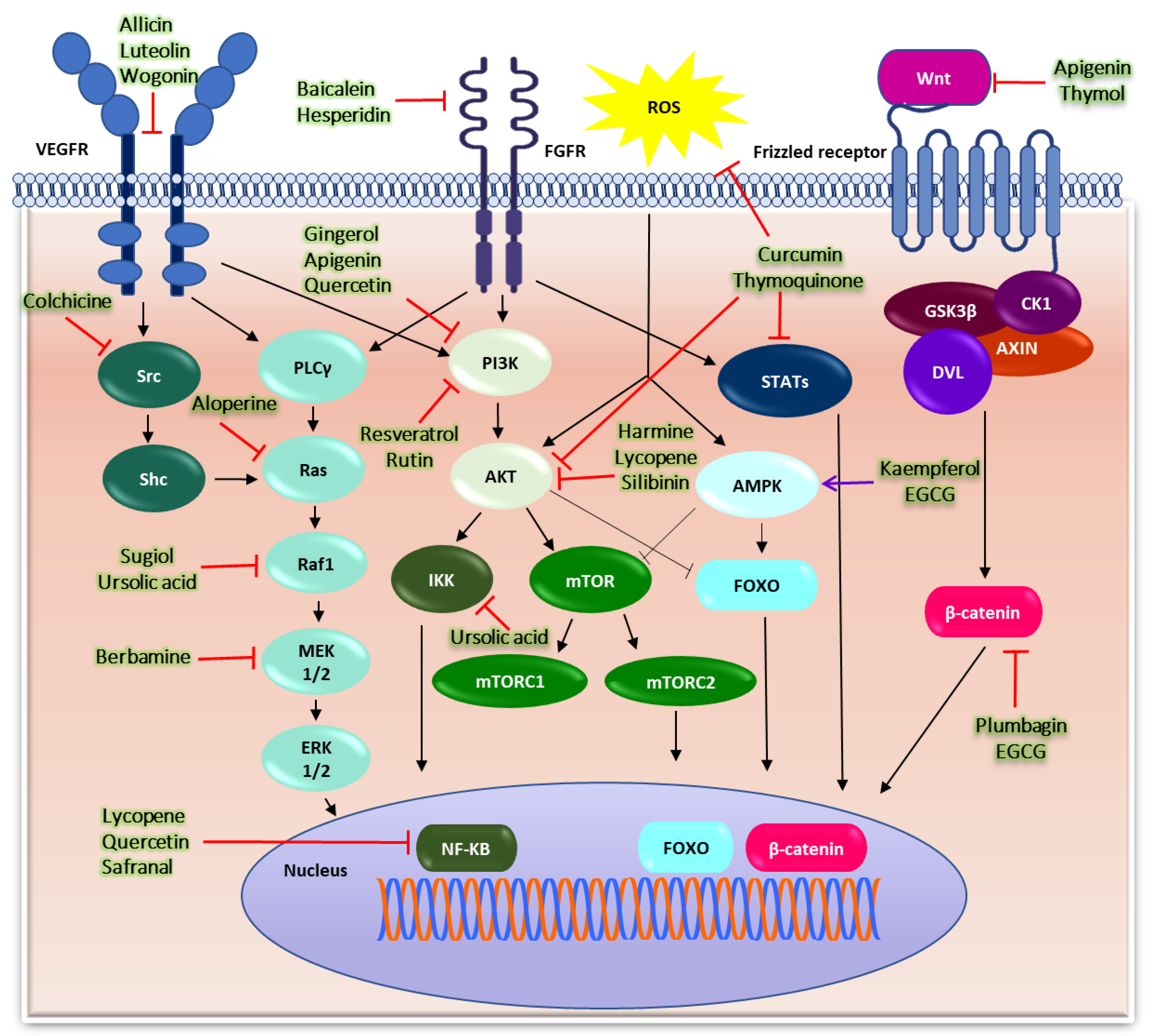
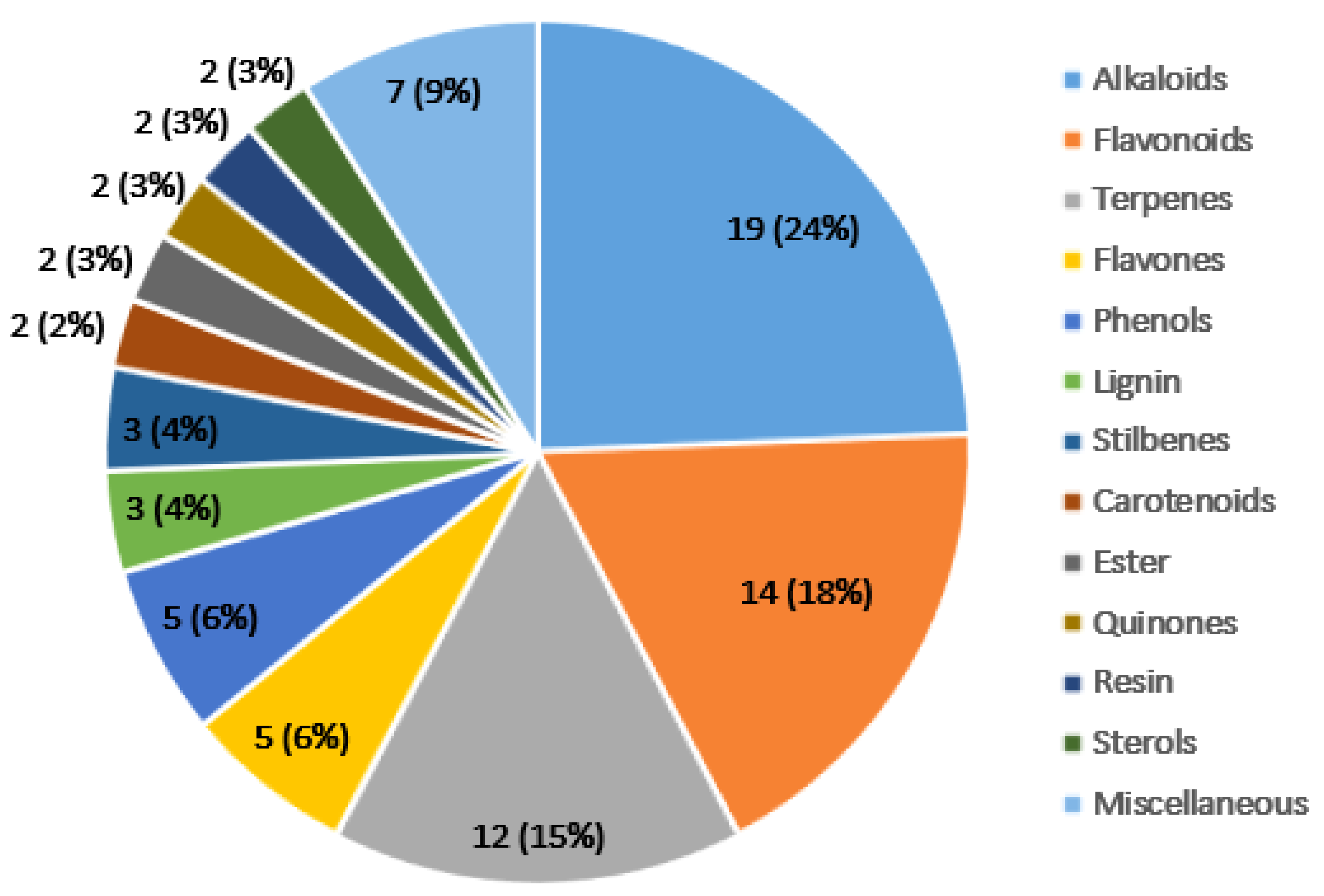
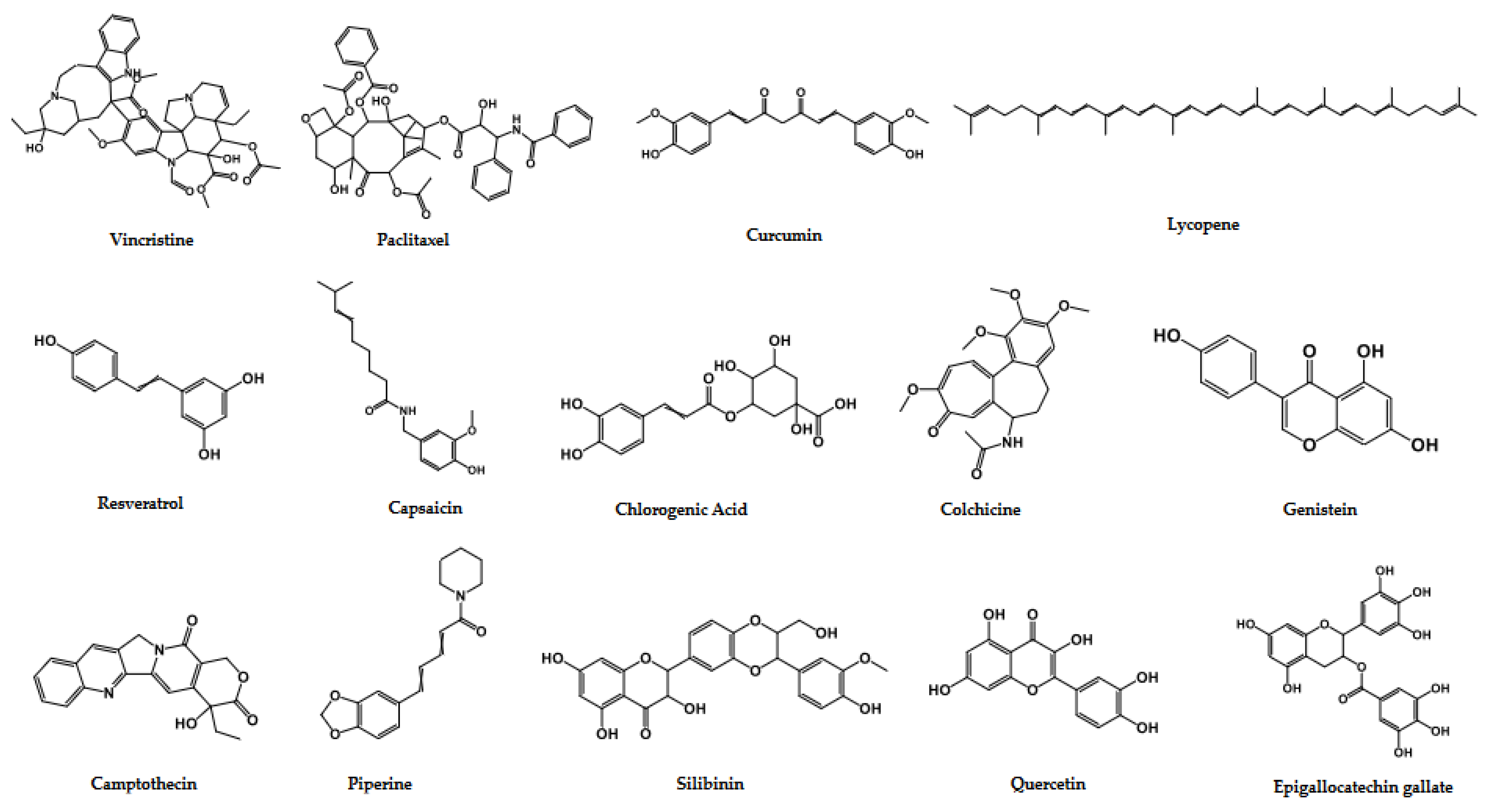
| Continents | Incidence | Rank | Mortality | Rank |
|---|---|---|---|---|
| Worldwide | 247.5 | – | 127.8 | – |
| Asia | 204.8 | – | 125.2 | – |
| Japan | 813.3 | 1 | 332.2 | 3 |
| China | 315.6 | 57 | 207.5 | 42 |
| India | 96 | 121 | 61.5 | 122 |
| South Korea | 449.2 | 42 | 172.8 | 56 |
| Europe | 587.4 | – | 261.1 | – |
| Germany | 750.2 | 4 | 300.9 | 10 |
| France | 716.9 | 9 | 284.4 | 17 |
| Italy | 686.8 | 13 | 289.0 | 15 |
| North America | 693.2 | – | 189.6 | – |
| USA | 689.3 | 12 | 185.0 | 54 |
| Canada | 726.9 | 7 | 229.7 | 33 |
| South America | 224.8 | – | 109.1 | – |
| Brazil | 278.6 | 63 | 122.3 | 72 |
| Argentina | 289.6 | 60 | 155.0 | 63 |
| Colombia | 222.5 | 75 | 108.1 | 81 |
| Africa | 82.7 | – | 53.1 | – |
| South Africa | 182.4 | 83 | 95.8 | 87 |
| Morocco | 160.8 | 93 | 95.5 | 88 |
| Ethiopia | 67.3 | 158 | 45.1 | 155 |
| Australia | 784.4 | 2 | 189.2 | 51 |
| New Zealand | 745.2 | 5 | 217.9 | 38 |
| Sr # | Phytochemicals | Chemical Nature | Plant’s Source/Origin | Chemical Structure | M: Weight (g/mol) | Cancer Type | Study Type | Targets and Mechanisms |
|---|---|---|---|---|---|---|---|---|
| 1 | Allicin | Thioester | Allium sativum | C6H10OS2 | 162.3 | Lung cancer | In vitro | Downregulation of VEGF expression [12] |
| Gastric cancer | In vitro | Enhanced expression of p38 and cleavage caspase-3 [13] | ||||||
| Oral cancer | In vitro | Upregulation of and cleaved caspase-3 [14] | ||||||
| Brain cancer | In vitro | Elevation in Fas/FasL expression [15] | ||||||
| 2 | Aloperine | Alkaloid | Sophora alopecuroides | C15H24N2 | 232.36 | Ovarian cancer | In vitro | Reactive oxygen species activation [16] |
| Thyroid cancer | In vitro | Suppression of Akt pathway and downstream B-cell lymphoma (Bcl-2) expression [17] | ||||||
| Prostate cancer | In vitro, in vivo | Inhibition of Akt and ERK phosphorylation [18] | ||||||
| Bladder cancer | In vitro | Downregulation of Ras, p-Raf1 and p-Erk1/2 expression [19] | ||||||
| Colon cancer | In vitro | Inhibition of JAK/Stat3 and PI3K/Akt pathways [20] | ||||||
| Bones cancer | In vitro | Suppression of PI3K/AKT signaling [21] | ||||||
| 3 | Alpinumisoflavone | Isoflavone | Derris eriocarpa | C20H16O5 | 336.3 | Colon cancer | In vitro | Blockage of DNA repairing [22] |
| Esophageal cancer | In vitro, in vivo, ex-vivo | Upregulation of miR-370 and suppression of PIM1 signaling [23] | ||||||
| Brain cancer | In vitro | Suppression of glycolysis and cyclin D1 expression and activation of caspase-9 [24] | ||||||
| 4 | Amygdalin | Diglucoside | Rosaceae kernels | C20H27NO11 | 457.4 | Bladder cancer | In vitro | Modulation of β1 or β4 integrin expression [25] |
| Breast cancer | In vitro | Downregulation of Bcl-2, upregulation of Bax and p38 MAPK signaling pathways [26] | ||||||
| Prostate cancer | In vitro | Activation of caspase-3 through downregulation of Bcl-2 and up-regulation of Bax [27] | ||||||
| Cervical cancer | In vitro | Downregulation of Bcl-2 and upregulation of Bax protein [28] | ||||||
| 5 | Andrographolide | Diterpenoid | Andrographis paniculata | C20H30O5 | 350.4 | Colon cancer | In vitro | Increase intracellular ROS level [29] |
| Skin cancer | In vitro | Activation of JNK and p38 signaling pathway [30] | ||||||
| Breast cancer | In vitro, in vivo | Suppressing of COX-2 and VEGF pathway [31] | ||||||
| Prostate cancer | In vitro, in vivo | Facilitate DNA damage [32] | ||||||
| Bile duct cancer | In vitro | Suppression of Claudin-1 via p-38 pathway [33] | ||||||
| Ovarian cancer | In vitro | Upregulation of TIMP1 expression [34] | ||||||
| 6 | Apigenin | Flavonoid | Matricaria chamomilla | C15H10O5 | 270.24 | Colon cancer | In vitro, in vivo | Inhibition of the Mcl-1, AKT, and ERK pro-survival regulators [35] |
| Lung cancer | In vitro, in vivo | Inhibition of NF-κB, AKT and ERK pathway [36] | ||||||
| Liver cancer | In vitro, in vivo | Inhibition of PI3K/Akt/mTOR signaling [37] | ||||||
| Pancreatic cancer | In vitro | Through G2/M cell cycle arrest [38] | ||||||
| Breast cancer | In vitro | Inhibition of YAP/TAZ activity [39] | ||||||
| Prostate cancer | In vitro, in vivo | Suppression of NF-κB/p65 expression [40] | ||||||
| Bone cancer | In vitro | Suppression of Wnt/β-catenin signaling [41] | ||||||
| 7 | Artemisinin | Alkaloid | Artemisia annua | C15H22O5 | 282.33 | Colon cancer | In vitro and in vivo | Increase in ROS production [42] |
| Kidney cancer | In vitro, in vivo | Inhibition of AKT signaling [43] | ||||||
| Ovarian cancer | In vitro, in vivo | Suppression of AKT/ERK/mTOR pathway [44] | ||||||
| Gallbladder cancer | In vitro, in vivo | Inhibition of ERK1/2 pathway [45] | ||||||
| 8 | Baicalein | Flavonoid | Scutellaria baicalensis | C15H10O5 | 270.24 | Lung cancer | In vitro, in vivo | Suppression of VEGF, FGFR-2, and RB-1 pathways [46] |
| Colon cancer | In vitro | Activation of caspase-3 [47] | ||||||
| Bladder cancer | In vitro, in vivo | Inhibition of cyclin B1, MMP-2 and MMP-9 mRNA expressions [48] | ||||||
| Pancreatic cancer | In vitro, in vivo | Increase caspase-3 and Bax, while decrease survivin and Bcl-2 expressions [49] | ||||||
| Liver cancer | In vitro | Suppression of PI3K/Akt pathway [50] | ||||||
| Prostate cancer | In vitro | Inhibition of caveolin-1/AKT/mTOR pathway [51] | ||||||
| Breast cancer | In vitro, in vivo | Activation of PAX8-AS1-N activation [52] | ||||||
| Ovarian cancer | In vitro, in vivo | Inhibition of YAP and RASSF6 expressions [53] | ||||||
| Skin cancer | In vitro, in vivo | Inhibition of glucose uptake and metabolism of tumor cells [54] | ||||||
| 9 | Berbamine | Alkaloid | Berberis amurensis | C37H40N2O6 | 608.7 | Blood cancer | In vitro | Upregulation of caspase-3 and downregulation of MDR-1 gene expression [55] |
| Liver cancer | In vitro, in vivo, ex vivo | Inhibition of Ca2+/Calmodulin-dependent protein Kinase II expression [56] | ||||||
| Ovarian cancer | In vitro, in vivo | Inhibition of Wnt/β-catenin signaling [57] | ||||||
| Colon cancer | In vitro | Inhibition of MEK/ERK signaling [58] | ||||||
| Head & neck cancer | In vitro | Inhibition of STAT3 activation [59] | ||||||
| 10 | Capsaicin | Capsaicinoid | Capsicum annuum | C18H27NO3 | 305.4 | Breast cancer | In vitro, in vivo | Downregulation of FBI-1-mediated NF-κB pathway [60] |
| Lung cancer | In vivo | Downregulation of MMP-2 and -9 levels [61] | ||||||
| Prostate cancer | In vitro | Increases protein light chain 3-II (autophagy marker) and ROS levels [62] | ||||||
| Colon cancer | In vitro | Stabilization and activation of p53 [63] | ||||||
| Esophageal cancer | In vitro | Decrease hexokinase-2 (HK-2) expression [64] | ||||||
| Skin cancer | In vitro | Downregulation of PI3-K/Akt/Rac1 pathway [65] | ||||||
| 11 | Cepharanthine | Alkaloid | Stephania cepharantha | C37H38N2O6 | 606.7 | Colon cancer | In vitro | Upregulation of p21Waf1/Cip1 pathway [66] |
| Breast cancer | In vitro | Inhibition of AKT/mTOR signaling [67] | ||||||
| Ovarian cancer | In vitro | Increases expression of p21Waf1 and decreasing expression of cyclins A and D proteins [68] | ||||||
| Liver cancer | In vitro | Activation of JNK1/2 signaling and downregulation of Akt pathway [69] | ||||||
| 12 | Chlorogenic Acid | Ester | Etlingera elatior | C16H18O9 | 354.31 | Liver cancer | In vitro, in vivo | Inhibition of DNMT1 expression [70] |
| Colon cancer | In vitro | Activation of PARP-1, and caspase-9 [71] | ||||||
| Breast cancer | In vitro | Upregulation of Bax and downregulation of Bcl-2 expressions [72] | ||||||
| 13 | Colchicine | Alkaloid | Colchicum automnale | C22H25NO6 | 399.4 | Gastric cancer | In vitro, in vivo | Induce caspase-3-mediated mitochondrial apoptosis [73] |
| Hypopharyngeal cancer | In vitro, in vivo | Inhibition of phosphorylated FAK/SRC complex and paxillin [74] | ||||||
| Breast cancer | In vitro | Inhibition of MMP-2 expression [75] | ||||||
| Colon cancer | In vitro | Decrease in AKT phosphorylation [76] | ||||||
| 14 | Combretastatin A4 | Stilbene | Combretum caffrum | C18H20O5 | 316.3 | Lung cancer | In vitro, in vivo | Disruption of microtubule assembly [77] |
| Bladder cancer | In vitro, in vivo | Activation of caspase-3 and reduction in BubR1 and Bub3 expressions [78] | ||||||
| Bone cancer | In vitro | Inhibition of NDRG1 [79] | ||||||
| 15 | Corosolic acid | Tripernoid | Lagerstroemia speciosa | C30H48O4 | 472.7 | Lung cancer | In vitro, in vivo | Inhibition of VEGFR2 kinase activity [33] |
| Colon cancer | In vitro, in vivo | Inhibition of HER2/HER3 receptors’ heterodimerization [80] | ||||||
| Gastric cancer | In vitro | Activation of AMPK pathway [81] | ||||||
| Liver cancer | In vitro, in vivo, ex vivo | Inactivation of CDK19/YAP/O-GlcNAcylation pathway [82] | ||||||
| Prostate cancer | In vitro, in vivo | Activation of IRE-1/JNK, PERK/CHOP and TRIB3 [83] | ||||||
| Cervical cancer | In vitro | Downregulation of PI3K and Akt signaling [84] | ||||||
| Kidney cancer | In vitro | Induction of lipid ROS [85] | ||||||
| Breast cancer | In vitro | Increase in ROS production and decrease in VEGF concentration [86] | ||||||
| Bladder cancer | In vitro, in vivo | Upregulation of SQSTM1/P62, NBR1, and UBB expression [87] | ||||||
| 16 | Crocetin | Carotenoid | Crocus sativus | C20H24O4 | 328.4 | Prostate cancer | In vitro, in vivo | Induce DNA damage and apoptosis [88] |
| Colon cancer | In vitro | Upregulation FAS/FADD death receptor [89] | ||||||
| Pancreatic cancer | In vitro, in vivo | Upregulation of Bax and downregulation of Bcl-2 protein [90] | ||||||
| Gastric cancer | In vitro, in vivo | Upregulation of caspase-3, -8 and -9 [91] | ||||||
| 17 | Cucurbitacin | Triterpene | Cucumis sativus | C32H46O8 | 558.7 | Colon cancer | In vitro | Inhibition of Hippo-YAP Signaling Pathway [92] |
| Gastric cancer | In vitro, in vivo | Suppression of Akt expression [93] | ||||||
| Bile duct cancer | In vitro | Downregulation of pRB, cyclin D1 and cyclin E expression [94] | ||||||
| Breast cancer | In vitro | Inhibition of Stat3 and Akt signaling [95] | ||||||
| 18 | Curcumin | Curcuminoids | Curcuma longa | C21H20O6 | 368.38 | Breast cancer | In vitro | Upregulation of PTEN/Akt signaling pathway [96] |
| Gastric cancer | In vitro | Suppression of PI3K/Akt/mTOR signaling pathway [49] | ||||||
| Oral cancer | In vivo | Suppression of NF-κB, and COX-2 expression [97] | ||||||
| Prostate cancer | In vitro | Downregulation of NF-κB, and CXCL1 and -2 expressions [98] | ||||||
| Colon cancer | In vitro | Inhibition of AMPK-induced NF-κB, uPA, and MMP9 activation [99] | ||||||
| Ovarian cancer | In vitro | JAK/STAT3 pathway inhibition [100] | ||||||
| Lung cancer | In vitro | Increase in FOXA2 expression [101] | ||||||
| 19 | Diosgenin | Saponin | Dioscorea villosa | C27H42O3 | 414.6 | Breast cancer | In vitro | Downregulation of Skp2 [102] |
| Liver cancer | In vitro | Inhibition of Akt and upregulation of p21 and p27 expression [103] | ||||||
| 20 | D-limonene | Terpene | Citrus aurantium | C10H16 | 136.23 | Colon cancer | In vitro | Inactivation of Akt pathway [104] |
| Lung cancer | In vitro | Upregulation of Atg5 [105] | ||||||
| Prostate cancer | In vitro | Generation of ROS, and activation of caspase-3 and -9 [106] | ||||||
| 21 | Emodin | Resin | Rheum palmatum | C15H10O5 | 270.24 | Breast cancer | In vitro | Activation of AhR-CYP1A1 signaling pathway [107] |
| Lung cancer | In vitro | Suppression of HAS2-HA-CD44/RHAMM pathway [108] | ||||||
| Pancreatic cancer | In vitro, in vivo | Downregulation of NF-κB, VEGF, MMP-2, and -9 [109] | ||||||
| Colon cancer | In vitro | Suppression of PI3K/AKT signaling [110] | ||||||
| Prostate cancer | In vitro | Downregulation of VEGF [111] | ||||||
| 22 | Epigallocatechin gallate (EGCG) | Catechin | Camellia sinensis | C22H18O11 | 458.4 | Bile duct cancer | In vitro, in vivo | Suppression of Notch1, MMP-2, and -9 signaling [112] |
| Lung cancer | In vitro | Activation of AMPK signaling pathway [113] | ||||||
| Ovarian cancer | In vitro | Induce DNA damage [114] | ||||||
| Prostate cancer | In vitro, in vivo | Inhibition of HSP90 function [115] | ||||||
| Head & neck cancer | In vitro, in vivo | Inhibition of beta-catenin expression [116] | ||||||
| Colon cancer | In vitro | Induction of ER stress through PERK/p-eIF2α/ATF4 and IRE1α pathways activation [117] | ||||||
| 23 | Erianin | Bisbenzyl | Dendrobium chrysotoxum | C18H22O5 | 318.4 | Breast cancer | In vitro | Activation PI3K/Akt pathway [118] |
| Lung cancer | In vitro, in vivo | Induction of Ca2+/CaM-dependent ferroptosis [119] | ||||||
| Liver cancer | In vitro, in vivo | Induction of oxidative stress-mediated mitochondrial apoptosis [73] | ||||||
| Oral cancer | In vitro | Regulation of MAPK pathway [120] | ||||||
| Bladder cancer | In vitro, in vivo | Increase in p-JNK level and induce c-Jun and Bcl-2 phosphorylation [121] | ||||||
| Bone cancer | In vitro, in vivo | Activation of ROS/JNK signaling [122] | ||||||
| Colon cancer | In vitro | Activation of JNK pathway [123] | ||||||
| Cervical cancer | In vitro | Regulation of ERK1/2 signaling [124] | ||||||
| 24 | Evodiamine | Alkaloid | Evodia rutaecarpa | C19H17N3O | 303.4 | Lung cancer | In vitro, in vivo | Elevation of CD8+ T cells and downregulation of MUC1-C/PD-L1 axis [125] |
| Thyroid cancer | In vitro | Through M phase cell cycle arrest and apoptosis’s induction [126] | ||||||
| Prostate cancer | In vitro | Activation of caspase-3 and -9 [127] | ||||||
| Liver cancer | In vitro | Deactivation of PI3K/AKT pathway [128] | ||||||
| Bladder cancer | In vitro | Enhance activation of P38 and JNK signaling [129] | ||||||
| Colon cancer | In vitro, in vivo | Inhibition of acetyl-NF-κB, p65 and MMP-9 expression [130] | ||||||
| Ovarian cancer | In vitro | Elevation of p27 and p21, and inhibition of Cdc2 expression [131] | ||||||
| Pancreatic cancer | In vitro | Inhibition of NF-κB, p65, and Bcl-2 expression, while activate Bax and cleaved caspase-3 [132] | ||||||
| 25 | Flavopiridol | Flavonoids | Dysoxylum binectariferum | C21H20ClNO5 | 41.8 | Breast cancer | In vitro | Inhibition of cyclin-dependent kinases [133] |
| Thyroid cancer | In vitro, in vivo | Reduction in Cyclin-dependent kinases (CDK) and MCL1 levels [134] | ||||||
| Bile duct cancer | In vitro, in vivo | Suppression of cyclin-dependent kinase pathway [135] | ||||||
| Head & neck cancer | In vitro, in vivo | Reduction in cyclin D1 expression [136] | ||||||
| Lung cancer | In vitro | Reduction in E-cadherin level [137] | ||||||
| Esophageal cancer | In vitro, in vivo | Decrease in c-Myc expression [138] | ||||||
| 26 | Gallic Acid | Phenolic acid | Galanthus nivalis | C7H6O5 | 170.12 | Lung cancer | In vitro, in vivo | Inhibition of PI3K/Akt pathway [139] |
| Liver cancer | In vitro | Suppression of Wnt/β-catenin signaling [140] | ||||||
| Breast cancer | In vitro, in vivo | Increases expression of cleaved caspase-7, -9, and p53, while reduces expression of Bcl-2, and PARP [141] | ||||||
| Colon cancer | In vitro, in vivo | Inhibition of SRC and EGFR phosphorylation [142] | ||||||
| Gastric cancer | In vitro | Increases expression of caspase-3, -8, and P53 gene [143] | ||||||
| Prostate cancer | In vitro | Generation of ROS [144] | ||||||
| Ovarian cancer | In vitro, in vivo | Inhibition of carbonic anhydrase IX protein [145] | ||||||
| Pancreatic cancer | In vitro | Downregulation of protein Bcl-,2 while increases in BAX expression [146] | ||||||
| 27 | Gambogic acid | Resin | Garcinia hanburyi | C38H44O8 | 628.7 | Lung cancer | In vitro, in vivo | Downregulation of Bcl-2, and upregulation of Bax expression [147] |
| Breast cancer | In vitro, in vivo | Increase the expression of Fas, cleaved caspase-3, -8, -9 and Bax proteins [148] | ||||||
| Liver cancer | In vitro | Induces apoptosis through caspases 3, -7, -8 and -9 [149] | ||||||
| Prostate cancer | In vitro | Induction of ROS production [150] | ||||||
| Colon cancer | In vitro, in vivo | Inhibition of Akt-mTOR signaling [151] | ||||||
| Gastric cancer | In vitro, in vivo | Downregulation of circ_ASAP2 and CDK7, while upregulation of miR-33a-5p expression [152] | ||||||
| 28 | Genistein | Isoflavones | Glycine max | C15H10O5 | 270.24 | Liver cancer | In vitro | Upregulation of Bax, cleaved caspase-3 and -9 and downregulation of Bcl-2 expression [153] |
| Colon cancer | In vitro, in vivo | Suppression of MiR-95, Akt and SGK1 signaling [154] | ||||||
| Prostate cancer | In vitro, in vivo | Decrease MMP-2 expression [155] | ||||||
| Lung cancer | In vitro | Downregulation of FoxM1 [156] | ||||||
| 29 | Gingerol | Phenol | Zingiber officinale | C17H26O4 | 294.4 | Breast cancer | In vitro | Induction of p53-dependent intrinsic apoptosis [157] |
| Oral cancer | In vitro | Activate caspases and increase Apaf-1 expression [158] | ||||||
| Cervical cancer | ||||||||
| Lung cancer | In vitro, in vivo | Reduction in ROS and iron accumulation and suppression of USP14 expression [159] | ||||||
| Pancreatic cancer | In vitro | Inhibition of PI3K/AKT signaling [160] | ||||||
| 30 | Ginkgetin | Flavonoid | Ginkgo biloba | C32H22O10 | 566.5 | Breast cancer | In vitro | Downregulation of estrogen receptor [161] |
| Lung cancer | In vitro, in vivo | Inhibition of p62/SQSTM1 signaling [162] | ||||||
| Prostate cancer | In vitro, in vivo | Suppression of STAT3 expression [163] | ||||||
| Bone cancer | In vitro | Inhibition of STAT3 and activation of caspase-3/9 [164] | ||||||
| Ovarian cancer | In vitro | Induction of apoptosis by activation of caspase-3 [165] | ||||||
| Kidney cancer | In vitro | Suppression of JAK2-STAT3 pathway [166] | ||||||
| 31 | Glycyrrhizin | Triterpenes | Glycyrrhiza glabra | C42H62O16 | 822.9 | Breast cancer | In vitro, in vivo | Induces ROS-mediated apoptosis [167] |
| Gastric cancer | In vitro | Downregulation of PI3K/AKT pathway [168] | ||||||
| Prostate cancer | In vitro | Induces DNA damage [169] | ||||||
| Ovarian cancer | In vitro | Upregulation of Fas and FasL expression [170] | ||||||
| 32 | Gossypol | Phenol | Gossypium hirsutum | C30H30O8 | 518.6 | Colon cancer | In vitro | Suppression of genes coding expression for CLAUDIN1, FAS, IL2, and IL8 [171] |
| Breast cancer | In vitro | Suppression of IKBKE, CCL2 and MAPK1 expression [172] | ||||||
| Lung cancer | In vitro | Decrease EGFR phosphorylation and AKT/ERK signaling [173] | ||||||
| Prostate cancer | In vitro | Activation of p53 protein [174] | ||||||
| Ovarian cancer | In vitro | Cause changes in thiol/redox states of proteins associated with glycolysis and stress responses [175] | ||||||
| Cervical cancer | In vitro, in vivo | Inhibition of FAK signaling and reversing TGF-β1-induced EMT [176] | ||||||
| Head & neck cancer | In vivo | Inhibition of Bcl-XL expression [177] | ||||||
| Skin cancer | In vitro | Induces mitochondria-dependent apoptosis [178] | ||||||
| 33 | Harmine | Alkaloid | Peganum harmala | C13H12N2O | 212.25 | Breast cancer | In vitro, in vivo | Downregulation of TAZ [179] |
| Thyroid cancer | In vitro, in vivo | Downregulation of Bcl-2 and upregulation of Bax expression [180] | ||||||
| Gastric cancer | In vitro | Inhibition of Akt/mTOR/p70S6K signaling [181] | ||||||
| Pancreatic cancer | In vitro | Suppression of AKT/mTOR pathway [182] | ||||||
| Ovarian cancer | In vitro | Inhibition of ERK/CREB pathway [183] | ||||||
| Lung cancer | In vitro | Suppression of AKT phosphorylation and enhances ROS generation [184] | ||||||
| 34 | Hesperidin | Flavonoid | Citrus limon | C28H34O15 | 610.6 | Lung cancer | In vitro | Downregulation of FGF and NF-κB signal transduction pathways [185] |
| Gastric cancer | In vitro | Increase in ROS levels and regulation of MAPK signaling [135] | ||||||
| Liver cancer | In vitro | Downregulation of Bcl-xL and upregulation of Bax, Bak, and tBid proteins [186] | ||||||
| Skin cancer | In vitro | Induces DNA damage [187] | ||||||
| Prostate cancer | In vitro | Induces apoptosis triggered by ROS generation [188] | ||||||
| Breast cancer | In vitro | Inhibition of PD-L1 expression via downregulation of Akt and NF-κB signaling [189] | ||||||
| 35 | Hispidulin | Flavone | Salvia involucrate | C16H12O6 | 300.26 | Lung cancer | In vitro, in vivo | Induces ROS-mediated apoptosis via ER stress pathway [190] |
| Liver cancer | In vitro, in vivo | Upregulation of PPARγ signaling [191] | ||||||
| Kidney cancer | In vitro, in vivo | Activation of ROS/JNK signaling [192] | ||||||
| Gastric cancer | In vitro | Activate ERK1/2 and NAG-1 signaling [193] | ||||||
| 36 | Kaempferol | Flavonoid | Spinacia oleracea | C15H10O6 | 286.24 | Breast cancer | In vitro | Increase expression of H2AX, caspase-3, and -9 [194] |
| Liver cancer | In vitro | Activation of AMPK signaling [195] | ||||||
| Kidney cancer | In vitro | Downregulation of AKT and FAK pathways [196] | ||||||
| Cervical cancer | In vitro | Disruption of mitochondrial membrane potential and intracellular free Ca2+ concentration [197] | ||||||
| Pancreatic cancer | In vitro | Inhibition of TGM2 expression [198] | ||||||
| Colon cancer | In vitro | Activation of ATM and p53-Bax axis [199] | ||||||
| 37 | Kurarinone | Flavonoid | Sophora flavescens | C26H30O6 | 438.5 | Lung cancer | In vitro, in vivo | Suppression of caspase-7 and -12, and AKT pathway [200] |
| Gastric cancer | In vitro | Inhibition of STAT3 signaling [201] | ||||||
| Breast cancer | In vitro | Inhibition of NF-κB activation [202] | ||||||
| 38 | Lappaconitine | Diterpenoid | Aconitum sinomontanum | C32H44N2O8 | 584.7 | Colon cancer | In vitro | Downregulation of PI3K/AKT/GSK3β signaling [203] |
| Lung cancer | In vitro | Downregulation of Cyclin E1 expression [204] | ||||||
| Liver cancer | In vitro | Upregulation of Bax, P53, and downregulation of Bcl-2 expressions [205] | ||||||
| 39 | Licochalcone A | Chalcone | Glycyrrhiza glabra | C21H22O4 | 338.4 | Breast cancer | In vitro | Inhibition of PI3K/Akt/mTOR pathway [206] |
| Bladder cancer | In vitro | Induces ER stress-dependent apoptosis caused by activation of ER-specific caspase-12 [207] | ||||||
| Lung cancer | In vitro | Induces ERK and p38 activation while suppresses JNK signaling [208] | ||||||
| Liver cancer | In vitro | Downregulation of MKK4/JNK [209] | ||||||
| 40 | Liriodenine | Alkaloid | Enicosanthellum pulchrum | C17H9NO3 | 275.26 | Breast cancer | In vitro | Upregulation of p53 [210] |
| Lung cancer | In vitro | Lockage of cell cycle progression at the G2/M phase [211] | ||||||
| Ovarian cancer | In vitro | Inhibition of progression of CAOV-3 cell cycle in S phase [212] | ||||||
| 41 | Luteolin | Flavonoid | Reseda luteola | C15H10O6 | 286.24 | Liver cancer | In vitro | Increases caspase-8 and decreases Bcl-2 expression [213] |
| Colon cancer | In vitro | Upregulation of Nrf2 expression [214] | ||||||
| Gastric cancer | In vitro | Inhibition of STAT3 phosphorylation [215] | ||||||
| Oral cancer | In vitro | Suppression of EMT-induced transcription factors [216] | ||||||
| Breast cancer | In vitro | Suppression of NF-κB/c-Myc activation and hTERT transcription [217] | ||||||
| Pancreatic cancer | In vitro | Inhibition of VEGF expression [218] | ||||||
| Lung cancer | In vitro | Inhibition of FAK-Src signaling [219] | ||||||
| 42 | Lycopene | Carotenoid | Solanum lycopersicum | C40H56 | 536.9 | Breast cancer | In vitro | Inhibition of Akt phosphorylation [220] |
| Prostate cancer | In vitro, in vivo | Downregulation of IL1, IL6, IL8, and TNF-α levels [221] | ||||||
| Colon cancer | In vitro | Suppression of NF-κB and JNK signaling [222] | ||||||
| Pancreatic cancer | In vitro | Inhibition of ROS-Mediated NF-κB Signaling [223] | ||||||
| Lung cancer | In vitro, in vivo | Induction of RARβ expression [224] | ||||||
| Gastric cancer | In vivo | Increase in SOD, and CAT, while decrease in MDA levels [225] | ||||||
| Cervical cancer | In vitro | Upregulation of Bax, and downregulation of Bcl-2 expression [226] | ||||||
| Skin cancer | In vivo | Inhibition of PCNA expression [227] | ||||||
| Brain cancer | In vitro | Activation of caspase-3 pathway [228] | ||||||
| Ovarian cancer | In vitro, in vivo | Decrease in integrin α5 expression and MAPK activation [229] | ||||||
| 43 | Lycorine | Alkaloid | Crinum asiaticum | C16H17NO4 | 287.31 | Breast cancer | In vitro, in vivo | Inhibition of STAT3 signaling pathway [230] |
| Gastric cancer | In vitro, in vivo | Enhances FBXW7-MCL1 axis level [224] | ||||||
| Prostate cancer | In vitro, in vivo | Inhibition of JAK/STAT signaling [231] | ||||||
| Lung cancer | In vitro, in vivo | Inhibition of Wnt/β-catenin signaling [232] | ||||||
| Liver cancer | In vitro | inhibition of ROCK1/cofilin-induced actin dynamics [233] | ||||||
| 44 | Magnolol | Lignan | Magnolia officinalis | C18H18O2 | 266.3 | Lung cancer | In vitro, in vivo | Downregulation of Akt/mTOR pathway [234] |
| Gallbladder cancer | In vitro, in vivo | Increase in p53 expression [235] | ||||||
| Liver cancer | In vitro | Inhibition of ERK-modulated metastatic process [236] | ||||||
| Prostate cancer | In vitro | Downregulation of MMP-2 and MMP-9 expression [237] | ||||||
| Esophageal cancer | In vitro | Activation of MAPK pathway [238] | ||||||
| 45 | Matrine | Alkaloid | Sophora flavescens | C15H24N2O | 248.36 | Prostate cancer | In vitro | Enhances expression of GADD45B, tumor suppresser gene or AKT/GSK3β/β-catenin [239] |
| Ovarian cancer | In vitro, in vivo | Suppression of PI3K/AKT/mTOR pathway expression [240] | ||||||
| Colon cancer | In vitro | Upregulation of Bax, downregulation of Bcl-2, and activation of caspase-3 and -9 [241] | ||||||
| Liver cancer | In vitro, in vivo | Upregulation of miR-345-5p and downregulation of circ_0027345 and HOXD3 [242] | ||||||
| Lung cancer | In vitro | Downregulation of C-C chemokine receptor type 7 (CCR7) [243] | ||||||
| 46 | Myricetin | Flavonoid | Myrica nagi Thunb | C15H10O8 | 318.23 | Thyroid cancer | In vitro | DNA damaging and inducing the release of apoptosis-inducing factor (AIF) [244] |
| Bladder cancer | In vitro, in vivo | Activation of caspase-3, and inhibition of Akt and MMP-9 expression [245] | ||||||
| Colon cancer | In vitro | Increases BAX/BCL2 ratio and AIF release [246] | ||||||
| Prostate cancer | In vitro | Inhibition of PIM1 and disruption of PIM1/CXCR4 interaction [247] | ||||||
| Breast cancer | In vitro | Enhances intracellular ROS production [248] | ||||||
| Lung cancer | In vitro | Inhibition of FAK-ERK signaling pathway [249] | ||||||
| 47 | Nimbolide | Limonoid triterpene | Azadirachta indica | C27H30O7 | 466.5 | Pancreatic cancer | In vitro, in vivo | Reduction in PI3K/AKT/mTOR and ERK signaling [250] |
| Colon cancer | In vitro, in vivo | Inhibition of Bcl-x, CXCR4, VEGF, and NF-κB [251] | ||||||
| Bladder cancer | In vitro | Stimulation of p38 MAPK and AKT phosphorylation [252] | ||||||
| 48 | Noscapine | Alkaloid | Papaver somniferum | C22H23NO7 | 413.4 | Colon cancer | In vitro | Inhibition of PI3K/AKT/mTOR pathway [253] |
| Breast cancer | In vitro | Decreases NF-κB and increases IκBα expression [254] | ||||||
| Lung cancer | In vitro, in vivo | Upregulation of PARP, Bax, and repression of Bcl2 expression [255] | ||||||
| Prostate cancer | In vivo | Suppression of microtubule dynamics [256] | ||||||
| 49 | Oridonin | Diterpenoid | Rabdosia rubescens | C20H28O6 | 364.4 | Colon cancer | In vitro, in vivo | Downregulation of GLUT1 and induction of autophagy [257] |
| Liver cancer | In vitro, in vivo | Inhibition of Akt pathway [258] | ||||||
| Ovarian cancer | In vitro | Suppression of mTOR pathway [259] | ||||||
| Bladder cancer | In vitro, in vivo | Inactivation of ERK and AKT signaling pathways [260] | ||||||
| Esophageal cancer | In vitro, in vivo | Suppression of AKT signaling [261] | ||||||
| Breast cancer | In vitro | Decrease in expression of MMPs and regulation of Integrin β1/FAK pathway [262] | ||||||
| Bone cancer | In vitro, in vivo | Activation of PPAR-γ and inhibition of Nrf2 pathways [263] | ||||||
| 50 | Oxymatrine | Alkaloid | Sophora flavescens | C15H24N2O2 | 264.36 | Cervical cancer | In vitro | Suppression of AKT/mTOR [264] |
| Breast cancer | In vitro | Suppress the PI3K/Akt [265] | ||||||
| Pancreatic cancer | Ion vitro | Downregulation of Livin and Survivin expression and upregulation of Bax/Bcl-2 ratio [266] | ||||||
| Prostate cancer | In vitro, in vivo | Increase in expression of p53 and Bax, and decrease in Bcl-2 level [267] | ||||||
| 51 | Physapubescin B | Steroid | Physalis pubescens | C30H42O8 | 530.6 | Ovarian cancer | In vitro | Suppress transcriptional activity of STAT3 [268] |
| Kidney cancer | In vitro, in vivo | Decreases expression of HIF-2α and activation of caspase-3 and -8 [269] | ||||||
| 52 | Pinostrobin | Flavonoid | Boesenbergia rotunda | C16H14O4 | 270.28 | Cervical cancer | In vitro | Increases expressions of TRAIL, FADD and production of ROS [270] |
| Breast cancer | In vitro | Downregulation of FAK and RhoA signaling [271] | ||||||
| Lung cancer | In vitro | Via promoting apoptosis [272] | ||||||
| Prostate cancer | In vitro | Decrease in cyclins B expression [273] | ||||||
| 53 | Piperine | Alkaloid | Piper nigrum | C17H19NO3 | 285.34 | Colon cancer | In vitro | Suppression of Wnt/β-catenin pathway [274] |
| Lung cancer | In vitro | Induces p53-mediated cell cycle arrest and apoptosis via activation of caspase-3 and -9 cascades [275] | ||||||
| Breast cancer | In vitro, in vivo | Induction of cell apoptosis and cell cycle blockage [276] | ||||||
| Prostate cancer | In vitro | Downregulation of cyclin A & D1 [277] | ||||||
| 54 | Piperlongumine | Alkaloid | Piper longum | C17H19NO5 | 317.34 | Lung cancer | In vitro | Inhibition of Akt phosphorylation [278] |
| Prostate cancer | In vitro | Induces DNA damage [279] | ||||||
| Colon cancer | In vitro | Induces DNA damage via increasing ROS production [280] | ||||||
| 55 | Plumbagin | Alkaloid | Plumbago zeylinica | C11H8O3 | 188.18 | Breast cancer | In vitro | Upregulation of p53 and p21 [281] |
| Colon cancer | In vitro | Induction of ROS formation [282] | ||||||
| Liver cancer | In vitro, in vivo | Downregulation of SIVA/mTOR signaling [283] | ||||||
| Prostate cancer | In vitro, in vivo | Induction of ROS production, and activation of ER stress [284] | ||||||
| Lung cancer | In vitro | Activation of caspase-9 and ROS production [285] | ||||||
| Esophageal cancer | In vitro, in vivo | Inhibition of STAT3-PLK1-AKT signaling [286] | ||||||
| Bone cancer | In vitro | Downregulation of c-Myc expression [287] | ||||||
| Cervical cancer | In vitro | Downregulation of MMP 2, 9, β-catenin and N-cadherin, while upregulation of E-cadherin signaling [288] | ||||||
| 56 | Pristimerin | Triterpenoid | Mortonia greggii | C30H40O4 | 464.6 | Colon cancer | In vitro | Decreases in AKT expression [289] |
| Oral cancer | In vitro | Inhibition of MAPK/Erk1/2 and Akt signaling [290] | ||||||
| Prostate cancer | In vitro | Inhibition of HIF-1α [291] | ||||||
| Lung cancer | In vitro | Downregulation of integrin β1 and MMP2 expression [292] | ||||||
| Pancreatic cancer | In vitro | Inhibition of Akt/NF-κB/mTOR signaling [293] | ||||||
| 57 | Pterostilbene | Stilbenoid | Polygonum cuspidatum | C16H16O3 | 256.3 | Ovarian cancer | In vitro | Decreases release of NF-κB p50, and NF-κB p65 [294] |
| Lung cancer | In vitro, in vivo | Enhance ROS generation, caspase-3 activity and ER stress [295] | ||||||
| Breast cancer | In vitro | Inactivate AKT and mTOR signaling pathways [296] | ||||||
| Colon cancer | In vitro, in vivo | Facilitate DNA repairing mediated through Top1/Tdp1 pathway [297] | ||||||
| 58 | Puerarin | Isoflavone | Pueraria radix | C21H20O9 | 416.4 | Colon cancer | In vitro | Increase Bax expression and caspase-3 activation [298] |
| Prostate cancer | In vitro | Inhibition of Keap1/Nrf2/ARE signaling pathways [299] | ||||||
| Lung cancer | In vitro, in vivo | Inhibition of PI3K/Akt pathway [300] | ||||||
| Liver cancer | In vitro | Modulation of MAPK signaling pathway [301] | ||||||
| Brain cancer | In vitro | Suppression of p-Akt and Bcl-2, while enhancement of Bax and cleaved caspase-3 expression [302] | ||||||
| 59 | Quercetin | Flavonoid | Allium cepa | C15H10O7 | 302.23 | Thyroid cancer | In vitro | Upregulation of Pro-NAG-1/GDF15 [303] |
| Breast cancer | In vitro | Inactivation of caspase-3 pathway [304] | ||||||
| Liver cancer | In vitro | Inhibition of PI3K/Akt and ERK pathways [305] | ||||||
| Prostate cancer | In vitro | Enhances release of tumor suppressor genes i.e., PTEN, p53 and TSC [306] | ||||||
| Lung cancer | In vitro | Inhibition of NF-κB Signaling [307] | ||||||
| 60 | Resveratrol | Stilbenoid | Polygonum cuspidatum | C14H12O3 | 228.24 | Colon cancer | In vitro | Inactivates PI3K/Akt signaling [308] |
| Breast cancer | In vitro | Suppression of Integrin αvβ3 expression [309] | ||||||
| Ovarian cancer | In vitro | Inactivation of STAT3 signaling [310] | ||||||
| Pancreatic cancer | In vitro | Suppression of NAF-1 expression, induces ROS accumulation, and activation of Nrf2 signaling [311] | ||||||
| Gastric cancer | In vitro | Upregulation of Bax, cleaved caspase-3 and -8 while suppression of NF-κB activation [312] | ||||||
| Lung cancer | In vitro, in vivo | Decreases SIRT1-mediated NF-κB activation [313] | ||||||
| Skin cancer | In vitro, in vivo | Deacetylation of SIRT1-activated NF-κB [314] | ||||||
| 61 | Rutin | Flavonoid | Ruta graveolens | C27H30O16 | 610.5 | Colon cancer | In vitro | Inhibition of caspase-3 expression [315] |
| Brain cancer | In vitro | Upregulation of P53 expression [265] | ||||||
| Skin cancer | In vitro | Suppression of PI3K/Akt and Wnt/β-catenin signaling [316] | ||||||
| Breast cancer | In vitro, in vivo | Inhibition of tyrosine kinase c-Met receptor [317] | ||||||
| 62 | Safranal | Alkaloid | Crocus sativus | C10H14O | 150.22 | Colon cancer | In vitro | Suppression of PI3K/Akt/ mTOR pathway [318] |
| Liver cancer | In vitro | Activation of caspases-8 and -9 [319] | ||||||
| Prostate cancer | In vitro, in vivo | Downregulation of AKT and NF-κB signaling [320] | ||||||
| Breast cancer | In vitro | Inhibits DNA and RNA synthesis [321] | ||||||
| 63 | Shikonin | Quinone | Lithospermum erythrorhizon | C16H16O5 | 288.29 | Lung cancer | In vitro | Downregulation of PFKFB2 expression [322] |
| Colon cancer | In vitro | Reduction in peroxiredoxin V (PrxV) expression [323] | ||||||
| Prostate cancer | In vitro | Induces necroptosis by decreasing caspase-8 and increasing pRIP1 and pRIP3 [324] | ||||||
| Liver cancer | In vitro, in vivo | Inhibition of PKM2 expression [325] | ||||||
| Ovarian cancer | In vitro | Decreases Bcl-2 expression and increases BAX, caspase-3 and -9 expression [326] | ||||||
| Skin cancer | In vitro, in vivo | Inhibition of MAPK pathway-mediated induction of apoptosis [327] | ||||||
| Bile duct cancer | In vitro | Inhibitions of PKM2 expression [328] | ||||||
| Breast cancer | In vitro | Inhibition of epidermal growth factor receptor signaling [329] | ||||||
| 64 | Shogaol | Phenol | Zingiber officinale | C17H24O3 | 276.4 | Breast cancer | In vitro | Inhibition Akt and STAT signaling pathway [330] |
| Prostate cancer | In vitro, in vivo | Inhibition of STAT3 and NF-κB signaling [331] | ||||||
| Lung cancer | In vitro, in vivo | Inhibits secretion of CCL2 [332] | ||||||
| Cervical cancer | In vitro | Induces apoptosis and G2/M cell cycle arrest [333] | ||||||
| 65 | Silibinin | Flavonolignan | Silybum marianum | C25H22O10 | 482.4 | Breast cancer | In vivo | Inhibition of EGF–EGFR signaling pathway [334] |
| Lung cancer | In vitro, in vivo | Activation of EGFR/LOX pathway [335] | ||||||
| Ovarian cancer | In vitro, in vivo | Inhibition of ERK and Akt pathway [336] | ||||||
| Prostate cancer | In vitro | Suppression of vimentin and MMP-2 expression [337] | ||||||
| Skin cancer | In vivo | Via Pro-Oxidant activity [338] | ||||||
| Colon cancer | In vitro | Downregulation of COX-2, VEGF, MMP-2, & -9, and CXCR-4 expression [339] | ||||||
| Gastric cancer | In vitro | Inhibition of STAT3 pathway [340] | ||||||
| 66 | Silymarin | Flavonolignan | Silybum marianum | C25H22O10 | 482.4 | Oral cancer | In vitro, in vivo | Induction of DR5/caspase-8 apoptotic signaling [289] |
| Gastric cancer | In vitro | Inhibition of p-ERK and activation of p-p38 and p-JNK pathways [341] | ||||||
| Colon cancer | In vitro | Increases ATF3 transcription through activation of JNK and IκK-α [291] | ||||||
| Prostate cancer | In vitro | Inhibition of cyclins (A, B1, D, E) and cyclin-dependent kinase pathway [337] | ||||||
| Breast cancer | In vitro, in vivo | Regulation of MAPK signaling pathway [342] | ||||||
| Liver cancer | In vivo | Reduction in ROS levels [343] | ||||||
| 67 | Solamargine | Alkaloid | Solanum nigrum L. | C45H73NO15 | 868.1 | Gastric cancer | In vitro, in vivo | Inhibition of Erk1/2 MAPK phosphorylation [344] |
| Skin cancer | In vitro | Downregulation of hILP/XIAP [345] | ||||||
| Bone cancer | In vitro | Suppression of notch pathway [346] | ||||||
| Liver cancer | In vitro | Induction of apoptosis [347] | ||||||
| Prostate cancer | In vitro, in vivo | Suppression of MUC1 expression [348] | ||||||
| 68 | Stachydrine | Alkaloid | Herba Leonuri | C7H13NO2 | 143.18 | Breast cancer | In vitro | Inhibition of Akt/ERK pathways [349] |
| Prostate cancer | In vitro | Inhibits CXCR3 and CXCR4 expressions [350] | ||||||
| 69 | Sugiol | Diterpene | Salvia prionitis | C20H28O2 | 300.4 | Ovarian cancer | In vitro | Blockage of RAF/MEK/ERK signaling pathway [351] |
| Prostate cancer | In vitro, in vivo | Inhibits STAT3 activity and increase ROS level [352] | ||||||
| Pancreatic cancer | In vitro | Induces ROS-mediated alterations in MMP [353] | ||||||
| Uterine cancer | In vitro | Increases Bax and decreases Bcl-2 expressions [354] | ||||||
| 70 | Tanshinone | Terpenoids | Salvia miltiorrhiza | C18H12O3 | 276.3 | Lung cancer | In vitro, in vivo | Suppression of IL-8 through NF-κB and AP-1 Pathways [355] |
| Gastric cancer | In vitro, in vivo | Downregulation of STAT3 pathway [356] | ||||||
| Breast cancer | In vitro | Suppression of HIF-1α and VEGF [357] | ||||||
| Ovarian cancer | In vitro, in vivo | Downregulation of Bcl-2, VEGF, COX2 and upregulation of Bax expressions [358] | ||||||
| Bladder cancer | In vitro | Activation of caspases 3 and -9 [359] | ||||||
| Cervical cancer | In vitro | Decrease in Bcl-2, HPV 16 and E7 protein levels, while increase in Bax and caspase-3 expressions [360] | ||||||
| 71 | Tectochrysin | Flavonoids | Alpinia oxyphylla | C16H12O4 | 268.26 | Colon cancer | In vitro | Inhibition of NF-κB signaling [361] |
| Prostate cancer | In vitro | Suppression of PI3K/AKT pathway [362] | ||||||
| Lung cancer | In vitro | Inhibition of STAT3 signaling [363] | ||||||
| 72 | Tetrandrine | Alkaloid | Stephania tetrandra | C38H42N2O6 | 622.7 | Cervical cancer | In vitro, in vivo | Downregulation of MMP2 and MMP9 [364] |
| Breast cancer | In vivo | Upregulation of Caspase-3, Bax, and downregulation of Bcl-2, Survivin, and PARP signaling [365] | ||||||
| Gastric cancer | In vitro, in vivo | Activation of caspase-3 and -9, and upregulation of apaf-1 [366] | ||||||
| Colon cancer | In vitro | Inhibition of EMT transition [367] | ||||||
| Prostate cancer | In vitro | Induction of DR4 and DR5 expression, and TRAIL-mediated apoptosis [368] | ||||||
| Bone cancer | In vitro, in vivo | Inhibition of PTEN/Akt, MAPK/Erk and Wnt signaling pathways [369] | ||||||
| 73 | Thymol | Phenol | Thymus vulgaris | C10H14O | 150.22 | Lung cancer | In vitro | Enhances cytoplasmic membrane permeability and cell apoptosis [370] |
| Breast cancer | ||||||||
| Prostate cancer | ||||||||
| Colon cancer | In vitro | Suppression of Wnt/β-Catenin pathway [371] | ||||||
| Gastric cancer | In vitro | Activation of Bax, PARP, and caspase-8 proteins [372] | ||||||
| 74 | Thymoquinone | Quinone | Nigella sativa | C10H12O2 | 164.2 | Kidney cancer | In vitro | Inhibition of AKT phosphorylation [373] |
| Breast cancer | In vitro, in vivo | Through phosphorylation of p38 via ROS generation [374] | ||||||
| Bladder cancer | In vitro | Inhibition of mTOR signaling [375] | ||||||
| Colon cancer | In vitro | Inhibition of STAT3, JAK2- and EGF receptor tyrosine kinase [376] | ||||||
| Gastric cancer | In vitro, in vivo | Inhibition of STAT3 pathway [377] | ||||||
| Liver cancer | In vitro | Inhibition of IL-8 expression, and activation of TRAIL receptors [378] | ||||||
| Lung cancer | In vitro | Reduction in ERK1/2 phosphorylation [379] | ||||||
| Oral cancer | In vitro | Downregulation of p38β MAPK [380] | ||||||
| Pancreatic cancer | In vitro | Downregulation of mucin 4 expression [381] | ||||||
| 75 | Ursolic acid | Triterpenoids | Oldenlandia diffusa | C30H48O3 | 456.7 | Ovarian cancer | In vitro | Downregulation of PI3K/AKT pathway [382] |
| Lung cancer | In vitro | Enhances apoptosis-inducing factor (AIF) and endonuclease G release [383] | ||||||
| Colon cancer | In vitro, in vivo | Inhibition of IL-6-mediated STAT3 pathway [384] | ||||||
| Breast cancer | In vitro | Downregulation of Nrf2 expression [385] | ||||||
| Pancreatic cancer | In vitro, in vivo | Inhibition of NF-κB and STAT3 pathways [386] | ||||||
| Gallbladder cancer | In vitro | Activation of caspase-3, -9 and PARP pathway [387] | ||||||
| 76 | Withaferin-A | steroidal lactone | Withania somnifera | C28H38O6 | 470.6 | Breast cancer | In vitro | Inhibition of TASK-3 expression [388] |
| Oral cancer | In vitro | Upregulation of Bim and Bax expression [389] | ||||||
| Skin cancer | In vitro | Activation of TRIM16 [390] | ||||||
| Bone cancer | In vitro | Inactivation of Notch-1 signaling [391] | ||||||
| Colon cancer | In vitro, in vivo | Inhibition of STAT3 Transcriptional activity [392] | ||||||
| 77 | Wogonin | Flavonoid | Scutellaria baicalensis | C16H12O5 | 284.26 | Colon cancer | In vitro | Increases ER stress, and mediates p53 phosphorylation [393] |
| Cervical cancer | In vitro | Inhibition of Cdk4 and cyclin D1 [394] | ||||||
| Lung cancer | In vitro | Downregulation of SGK1 protein levels [395] | ||||||
| Bone cancer | In vitro | Increases ROS level [396] | ||||||
| Breast cancer | In vitro | Activation of ERK and p38 MAPKs pathways [397] | ||||||
| Ovarian cancer | In vitro | Increase in p53 and decrease in VEGF proteins expression [398] | ||||||
| 78 | Xanthatin | Sesquiterpene lactone | Xanthium strumarium | C15H18O3 | 246.3 | Skin cancer | In vitro, in vivo | Inhibition of Wnt/β-catenin pathway [399] |
| Lung cancer | In vitro, in vivo | Inhibition of GSK-3β signaling [400] | ||||||
| Breast cancer | In vitro, in vivo | Inhibition of VEGFR2 signaling [401] | ||||||
| Colon cancer | In vitro | Inhibition of mTOR pathway [402] |
| Cancer Type | Number of Phytochemicals | Cancer Type | Number of Phytochemicals | Cancer Type | Number of Phytochemicals |
|---|---|---|---|---|---|
| Breast cancer | 55 | Pancreatic cancer | 18 | Esophageal cancer | 6 |
| Colon cancer | 53 | Cervical cancer | 14 | Thyroid Cancer | 6 |
| Lung cancer | 53 | Bladder cancer | 13 | Bile duct cancer | 5 |
| Prostate cancer | 45 | Bladder cancer | 13 | Brain cancer | 5 |
| Liver cancer | 30 | Skin cancer | 11 | Miscellaneous | 10 |
| Ovarian Cancer | 27 | Oral cancer | 9 | NA | NA |
| Gastric cancer | 24 | Kidney cancer | 7 | NA | NA |
| Sr # | Phytochemicals | Effective against Number of Cancer Types |
|---|---|---|
| 1 | Lycopene | 10 |
| 2 | Baicalin, Corosolic acid, Plumbagin, Shikonin, Thymoquinone | 9 |
| 3 | Erianin, Evodiamine, Gallic acid, Gossypol | 8 |
| 4 | Apigenin, Curcumin, Luteolin, Oridonin, Resveratrol, Silibinin | 7 |
| 5 | Other phytochemicals | ≤6 |
| Sr # | Phytochemicals | Source | Cancer Type | Development Stage | Status | Trade Name | NCT Number |
|---|---|---|---|---|---|---|---|
| 1 | Vincristine | Catharanthus roseus | Acute leukemia | FDA approved | 1963 | Oncovin | NA |
| 2 | Paclitaxel | Taxus braciola | Late-stage pancreatic cancer | FDA approved | 2013 | Abraxane® | NA |
| Advanced non-small cell lung cancer | FDA approved | 2012 | Abraxane® | NA | |||
| Metastatic breast cancer | FDA approved | 2005 | Abraxane® | NA | |||
| 3 | Curcumin | Curcuma longa | Prostate cancer | Phase 3 | Recruiting, 15 June 2021 | Biocurcumax (BCM-95) ® | NCT03769766 |
| Cervical cancer | Phase 2 | Not yet recruiting, 25 June 2021 | Curcugreen (BCM-95) ® | NCT04294836 | |||
| Pancreatic cancer | Phase 2 | Recruiting, 2020 | NA | NCT00094445 | |||
| Gastric cancer | Phase 2 | Not yet recruiting, 13 January 2022 | Meriva® | NCT02782949 | |||
| Breast cancer | Phase 1 | Recruiting, 23 February 2021 | NA | NCT03980509 | |||
| 4 | Lycopene | Solanum lycopersicum | Prostate cancer | Phase 3 | Completed, 23 January 2018 | NA | NCT01105338 |
| 5 | Resveratrol | Polygonum cuspidatum | Multiple myeloma cancer | Phase 2 | Terminated (collecting more data) 27 February 2019 | SRT501 | NCT00920556 |
| Colon cancer | Phase 1 | Completed, 14 June 2017 | SRT501 | NCT00920803 | |||
| Neuroendocrine cancer | NA | Completed, 18 November 2019 | NA | NCT01476592 | |||
| 6 | Capsaicin | Capsicum annuum | Breast cancer | Phase 3 | Recruiting, 29 December 2021 | Qutenza® | NCT03794388 |
| Head and neck cancer | Phase 2 | Recruiting, 5 August 2021 | Qutenza® | NCT04704453 | |||
| Prostate cancer | Phase 2 | Not yet recruiting, 16 January 2014 | Cayenne | NCT02037464 | |||
| 7 | Chlorogenic acid | Etlingera elatior | Lung cancer | Phase 2 | Recruiting, 26 November 2018 | NA | NCT03751592 |
| 8 | Colchicine | Colchicum autumnale | Liver cancer | Phase 2 | Recruiting, 11 February 2020 | Colchicine | NCT04264260 |
| 9 | Genistein | Glycine max | Prostate cancer | Phase 2 | Temporarily suspended, 4 December 2020 | NA | NCT02766478 |
| Colorectal cancer | Phase 2 | Completed, 10 May 2019 | Bonistein | NCT01985763 | |||
| Prostate cancer | Phase 2 | Completed, 6 August 2019 | Novasoy 400 | NCT01036321 | |||
| Bladder cancer | Phase 2 | Completed, 10 June 2021 | NA | NCT00118040 | |||
| 10 | Camptothecin | Camptotheca acuminata | Solid tumor | Phase 2 | Completed, 28 May 2020 | CRLX101 | NCT00333502 |
| Stomach and esophageal cancer | Phase 2 | Completed, 1 February 2018 | CRLX101 | NCT01612546 | |||
| Advanced non-small cell lung cancer | Phase 2 | Completed, 28 May 2020 | CRLX101 | NCT01380769 | |||
| 11 | Piperine | Piper nigrum | Prostate cancer | Phase 2 | Not yet recruiting, 3 November 2021 | NA | NCT04731844 |
| 12 | Silibinin | Silybum marianum | Prostate cancer | Phase 2 | Completed, 31 March 2014 | Silibin-Phytosome | NCT00487721 |
| 13 | Quercetin | Allium cepa | Squamous cell carcinoma | Phase 2 | Recruiting, 28 October 2021 | NA | NCT03476330 |
| 14 | Epigallocatechin gallate | Camellia sinensis | Colon cancer | Phase 1 | Recruiting, 15 December 2021 | Teavigo™ | NCT02891538 |
| Esophageal cancer | Phase 1 | Recruiting, 10 September 2021 | NA | NCT05039983 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, A.W.; Farooq, M.; Haseeb, M.; Choi, S. Role of Plant-Derived Active Constituents in Cancer Treatment and Their Mechanisms of Action. Cells 2022, 11, 1326. https://doi.org/10.3390/cells11081326
Khan AW, Farooq M, Haseeb M, Choi S. Role of Plant-Derived Active Constituents in Cancer Treatment and Their Mechanisms of Action. Cells. 2022; 11(8):1326. https://doi.org/10.3390/cells11081326
Chicago/Turabian StyleKhan, Abdul Waheed, Mariya Farooq, Muhammad Haseeb, and Sangdun Choi. 2022. "Role of Plant-Derived Active Constituents in Cancer Treatment and Their Mechanisms of Action" Cells 11, no. 8: 1326. https://doi.org/10.3390/cells11081326
APA StyleKhan, A. W., Farooq, M., Haseeb, M., & Choi, S. (2022). Role of Plant-Derived Active Constituents in Cancer Treatment and Their Mechanisms of Action. Cells, 11(8), 1326. https://doi.org/10.3390/cells11081326







