Roles of Podoplanin in Malignant Progression of Tumor
Abstract
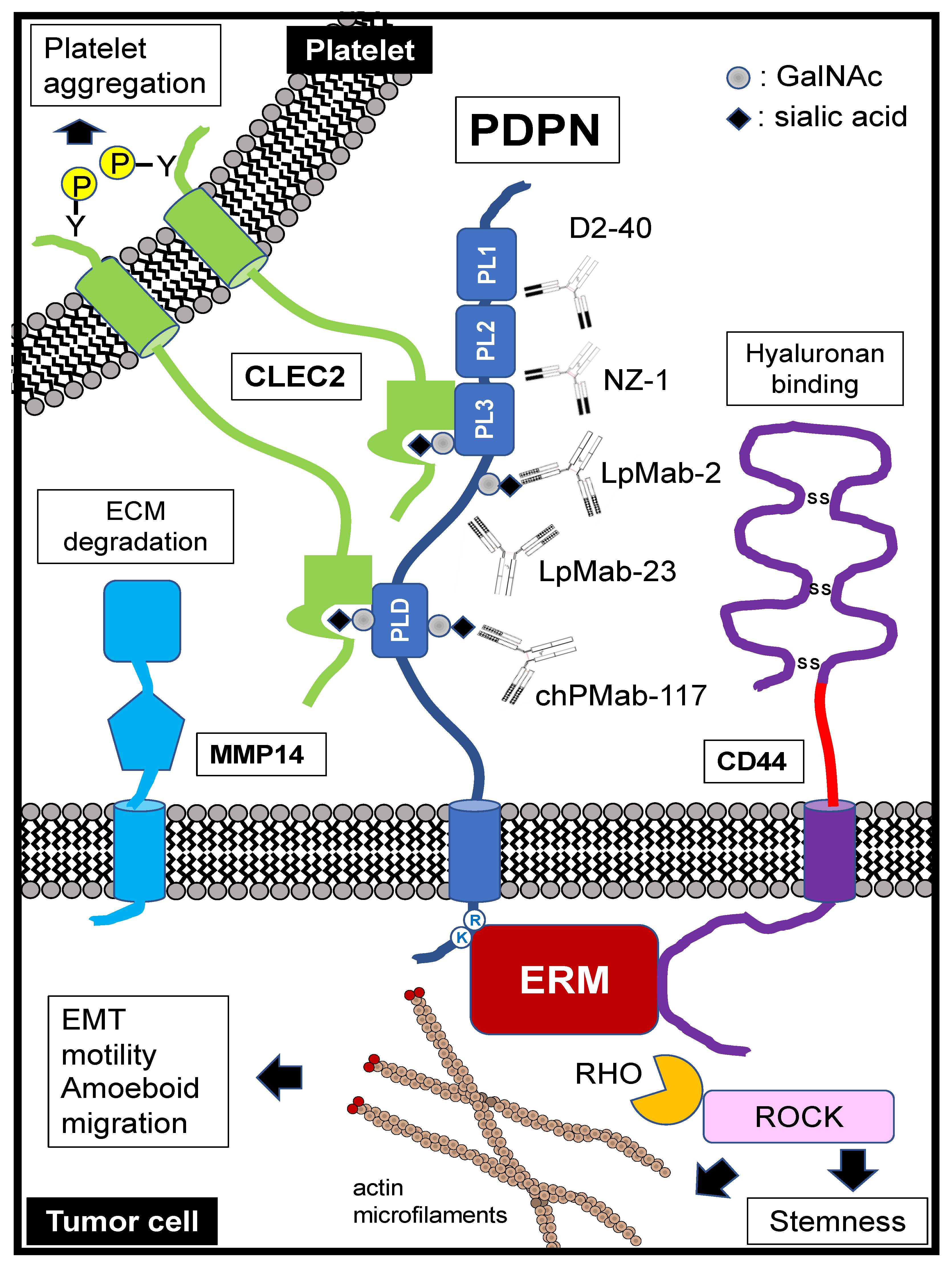
1. PDPN Structure and Functions
1.1. PDPN Structure
1.2. Molecular Functions of PDPN Extracellular Domain
1.3. Molecular Functions of PDPN Intracellular Domain
2. Physiological Roles of PDPN
2.1. Lungs
2.2. Lymphatic Endothelial Cells
2.3. Podocyte
3. PDPN Overexpression in Cancer as a Diagnostic Marker
3.1. PDPN Overexpression in Tumors
3.2. Pathological Diagnosis by Specific Antibodies
3.3. The Mechanism of PDPN Overexpression in Tumors
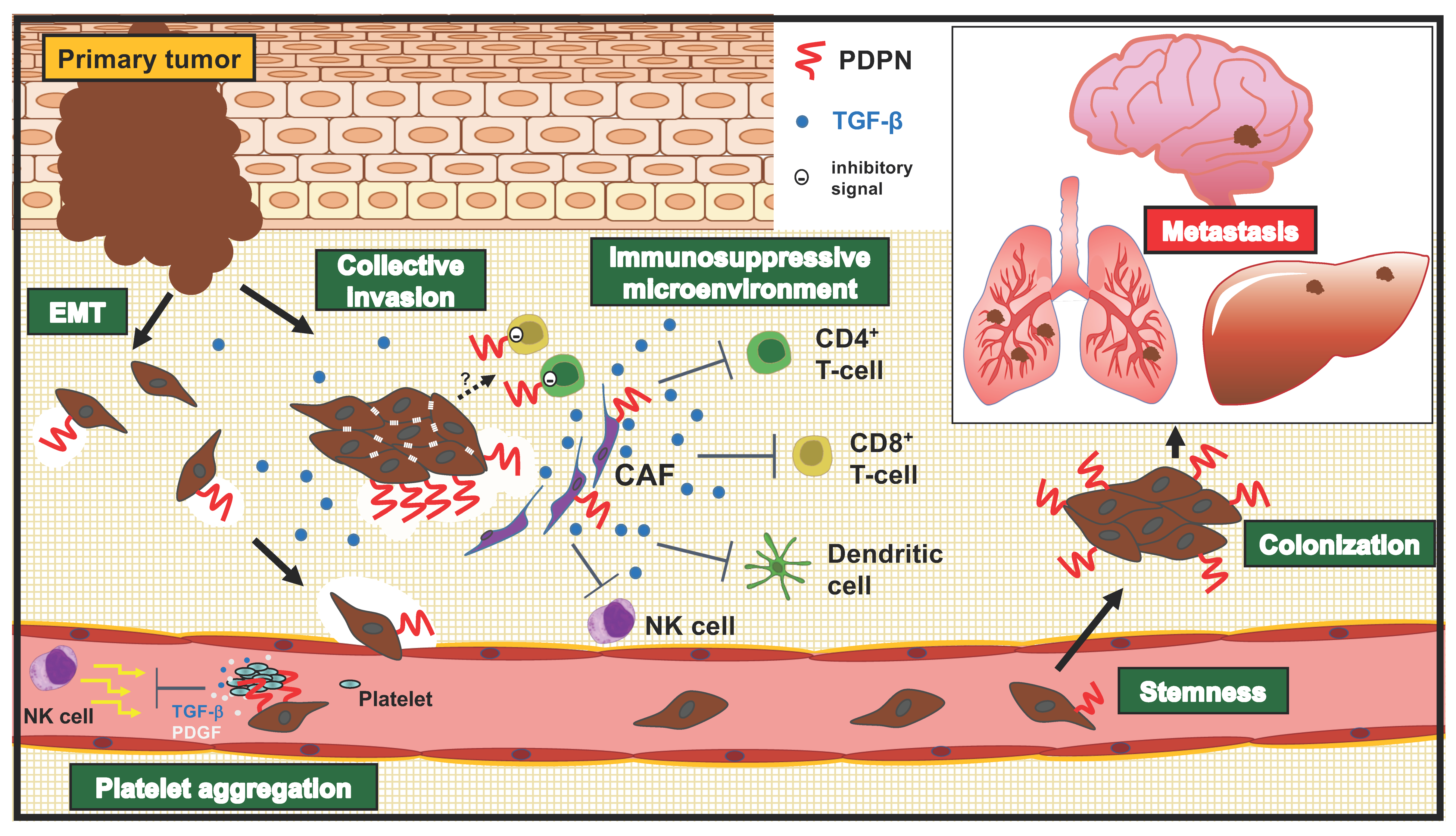
4. Roles of PDPN in Invasion-Metastatic Cascade
4.1. Migration and Invasion
4.2. Platelet Aggregation
4.3. Stemness (Colonization)
4.4. Stromal Expression of PDPN and Its Roles in Tumors
4.5. Roles of PDPN in T Cell Immunity
5. Therapeutic Strategies to PDPN-Overexpressing Tumors
5.1. Anti-PDPN Monoclonal Antibodies (mAbs)
5.2. Radioimmunotherapy (RIT)
5.3. Cancer-Specific Anti-PDPN mAbs
5.4. Chimeric Antigen Receptor (CAR)-T
6. Concluding Remarks and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Krishnan, H.; Rayes, J.; Miyashita, T.; Ishii, G.; Retzbach, E.P.; Sheehan, S.A.; Takemoto, A.; Chang, Y.W.; Yoneda, K.; Asai, J.; et al. Podoplanin: An emerging cancer biomarker and therapeutic target. Cancer Sci. 2018, 109, 1292–1299. [Google Scholar] [CrossRef]
- Quintanilla, M.; Montero-Montero, L.; Renart, J.; Martín-Villar, E. Podoplanin in Inflammation and Cancer. Int. J. Mol. Sci. 2019, 20, 707. [Google Scholar] [CrossRef]
- Kato, Y.; Fujita, N.; Kunita, A.; Sato, S.; Kaneko, M.; Osawa, M.; Tsuruo, T. Molecular identification of Aggrus/T1alpha as a platelet aggregation-inducing factor expressed in colorectal tumors. J. Biol. Chem. 2003, 278, 51599–51605. [Google Scholar] [CrossRef]
- Takei, J.; Itai, S.; Harada, H.; Furusawa, Y.; Miwa, T.; Fukui, M.; Nakamura, T.; Sano, M.; Sayama, Y.; Yanaka, M.; et al. Characterization of Anti-Goat Podoplanin Monoclonal Antibody PMab-235 Using Immunohistochemistry Against Goat Tissues. Monoclon. Antib. Immunodiagn. Immunother. 2019, 38, 213–219. [Google Scholar] [CrossRef]
- Kato, Y.; Takei, J.; Furusawa, Y.; Sayama, Y.; Sano, M.; Konnai, S.; Kobayashi, A.; Harada, H.; Takahashi, M.; Suzuki, H.; et al. Epitope Mapping of Anti-Bear Podoplanin Monoclonal Antibody PMab-247. Monoclon. Antib. Immunodiagn. Immunother. 2019, 38, 230–233. [Google Scholar] [CrossRef]
- Sayama, Y.; Sano, M.; Furusawa, Y.; Kaneko, M.K.; Kato, Y. Epitope Mapping of PMab-225 an Anti-Alpaca Podoplanin Monoclonal Antibody Using Flow Cytometry. Monoclon. Antib. Immunodiagn. Immunother. 2019, 38, 255–260. [Google Scholar] [CrossRef]
- Sano, M.; Kaneko, M.K.; Kato, Y. Epitope Mapping of Monoclonal Antibody PMab-233 Against Tasmanian Devil Podoplanin. Monoclon. Antib. Immunodiagn. Immunother. 2019, 38, 261–265. [Google Scholar] [CrossRef]
- Kato, Y.; Sayama, Y.; Sano, M.; Kaneko, M.K. Epitope Analysis of an Antihorse Podoplanin Monoclonal Antibody PMab-219. Monoclon. Antib. Immunodiagn. Immunother. 2019, 38, 266–270. [Google Scholar] [CrossRef]
- Sayama, Y.; Sano, M.; Kaneko, M.K.; Kato, Y. Epitope Analysis of an Anti-Whale Podoplanin Monoclonal Antibody, PMab-237, Using Flow Cytometry. Monoclon. Antib. Immunodiagn. Immunother. 2020, 39, 17–22. [Google Scholar] [CrossRef]
- Sayama, Y.; Sano, M.; Asano, T.; Furusawa, Y.; Takei, J.; Nakamura, T.; Yanaka, M.; Okamoto, S.; Handa, S.; Komatsu, Y.; et al. Epitope Mapping of PMab-241, a Lymphatic Endothelial Cell-Specific Anti-Bear Podoplanin Monoclonal Antibody. Monoclon. Antib. Immunodiagn. Immunother. 2020, 39, 77–81. [Google Scholar] [CrossRef]
- Kato, Y.; Sano, M.; Asano, T.; Sayama, Y.; Kaneko, M.K. Thr80 of Sheep Podoplanin Is a Critical Epitope of the Antisheep Podoplanin Monoclonal Antibody: PMab-256. Monoclon. Antib. Immunodiagn. Immunother. 2020, 39, 95–100. [Google Scholar] [CrossRef]
- Sekiguchi, T.; Takemoto, A.; Takagi, S.; Takatori, K.; Sato, S.; Takami, M.; Fujita, N. Targeting a novel domain in podoplanin for inhibiting platelet-mediated tumor metastasis. Oncotarget 2016, 7, 3934–3946. [Google Scholar] [CrossRef]
- Kaneko, M.; Kato, Y.; Kunita, A.; Fujita, N.; Tsuruo, T.; Osawa, M. Functional sialylated O-glycan to platelet aggregation on Aggrus (T1alpha/Podoplanin) molecules expressed in Chinese hamster ovary cells. J. Biol. Chem. 2004, 279, 38838–38843. [Google Scholar] [CrossRef]
- Kaneko, M.K.; Kato, Y.; Kameyama, A.; Ito, H.; Kuno, A.; Hirabayashi, J.; Kubota, T.; Amano, K.; Chiba, Y.; Hasegawa, Y.; et al. Functional glycosylation of human podoplanin: Glycan structure of platelet aggregation-inducing factor. FEBS Lett. 2007, 581, 331–336. [Google Scholar] [CrossRef]
- Suzuki-Inoue, K.; Kato, Y.; Inoue, O.; Kaneko, M.K.; Mishima, K.; Yatomi, Y.; Yamazaki, Y.; Narimatsu, H.; Ozaki, Y. Involvement of the snake toxin receptor CLEC-2, in podoplanin-mediated platelet activation, by cancer cells. J. Biol. Chem. 2007, 282, 25993–26001. [Google Scholar] [CrossRef]
- Kato, Y.; Kaneko, M.K.; Kunita, A.; Ito, H.; Kameyama, A.; Ogasawara, S.; Matsuura, N.; Hasegawa, Y.; Suzuki-Inoue, K.; Inoue, O.; et al. Molecular analysis of the pathophysiological binding of the platelet aggregation-inducing factor podoplanin to the C-type lectin-like receptor CLEC-2. Cancer Sci. 2008, 99, 54–61. [Google Scholar] [CrossRef]
- Meng, D.; Luo, M.; Liu, B. The Role of CLEC-2 and Its Ligands in Thromboinflammation. Front. Immunol. 2021, 12, 688643. [Google Scholar] [CrossRef]
- Suzuki-Inoue, K.; Fuller, G.L.; García, A.; Eble, J.A.; Pöhlmann, S.; Inoue, O.; Gartner, T.K.; Hughan, S.C.; Pearce, A.C.; Laing, G.D.; et al. A novel Syk-dependent mechanism of platelet activation by the C-type lectin receptor CLEC-2. Blood 2006, 107, 542–549. [Google Scholar] [CrossRef]
- Watson, A.A.; Brown, J.; Harlos, K.; Eble, J.A.; Walter, T.S.; O’Callaghan, C.A. The crystal structure and mutational binding analysis of the extracellular domain of the platelet-activating receptor CLEC-2. J. Biol. Chem. 2007, 282, 3165–3172. [Google Scholar] [CrossRef]
- Watson, A.A.; O’Callaghan, C.A. Crystallization and X-ray diffraction analysis of human CLEC-2. Acta Cryst. Sect. F Struct. Biol. Cryst. Commun. 2005, 61, 1094–1096. [Google Scholar] [CrossRef]
- Fuller, G.L.; Williams, J.A.; Tomlinson, M.G.; Eble, J.A.; Hanna, S.L.; Pöhlmann, S.; Suzuki-Inoue, K.; Ozaki, Y.; Watson, S.P.; Pearce, A.C. The C-type lectin receptors CLEC-2 and Dectin-1, but not DC-SIGN, signal via a novel YXXL-dependent signaling cascade. J. Biol. Chem. 2007, 282, 12397–12409. [Google Scholar] [CrossRef] [PubMed]
- Hughes, C.E.; Auger, J.M.; McGlade, J.; Eble, J.A.; Pearce, A.C.; Watson, S.P. Differential roles for the adapters Gads and LAT in platelet activation by GPVI and CLEC-2. J. Thromb. Haemost. 2008, 6, 2152–2159. [Google Scholar] [CrossRef]
- Ozaki, Y.; Suzuki-Inoue, K.; Inoue, O. Novel interactions in platelet biology: CLEC-2/podoplanin and laminin/GPVI. J. Thromb. Haemost. 2009, 7 (Suppl 1), 191–194. [Google Scholar] [CrossRef]
- Kaneko, M.K.; Kato, Y.; Kitano, T.; Osawa, M. Conservation of a platelet activating domain of Aggrus/podoplanin as a platelet aggregation-inducing factor. Gene 2006, 378, 52–57. [Google Scholar] [CrossRef]
- Nagae, M.; Morita-Matsumoto, K.; Kato, M.; Kaneko, M.K.; Kato, Y.; Yamaguchi, Y. A platform of C-type lectin-like receptor CLEC-2 for binding O-glycosylated podoplanin and nonglycosylated rhodocytin. Structure 2014, 22, 1711–1721. [Google Scholar] [CrossRef] [PubMed]
- Scholl, F.G.; Gamallo, C.; Vilaró, S.; Quintanilla, M. Identification of PA2.26 antigen as a novel cell-surface mucin-type glycoprotein that induces plasma membrane extensions and increased motility in keratinocytes. J. Cell Sci. 1999, 112, 4601–4613. [Google Scholar] [CrossRef]
- Martín-Villar, E.; Megías, D.; Castel, S.; Yurrita, M.M.; Vilaró, S.; Quintanilla, M. Podoplanin binds ERM proteins to activate RhoA and promote epithelial-mesenchymal transition. J. Cell Sci. 2006, 119, 4541–4553. [Google Scholar] [CrossRef]
- Pecora, A.; Laprise, J.; Dahmene, M.; Laurin, M. Skin Cancers and the Contribution of Rho GTPase Signaling Networks to Their Progression. Cancers 2021, 13, 4362. [Google Scholar] [CrossRef]
- Zhang, Z.; Liu, M.; Zheng, Y. Role of Rho GTPases in stem cell regulation. Biochem. Soc. Trans. 2021, 49, 2941–2955. [Google Scholar] [CrossRef]
- Astarita, J.L.; Acton, S.E.; Turley, S.J. Podoplanin: Emerging functions in development, the immune system, and cancer. Front. Immunol. 2012, 3, 283. [Google Scholar] [CrossRef]
- Krishnan, H.; Retzbach, E.P.; Ramirez, M.I.; Liu, T.; Li, H.; Miller, W.T.; Goldberg, G.S. PKA and CDK5 can phosphorylate specific serines on the intracellular domain of podoplanin (PDPN) to inhibit cell motility. Exp. Cell Res. 2015, 335, 115–122. [Google Scholar] [CrossRef]
- Rishi, A.K.; Joyce-Brady, M.; Fisher, J.; Dobbs, L.G.; Floros, J.; VanderSpek, J.; Brody, J.S.; Williams, M.C. Cloning, characterization, and development expression of a rat lung alveolar type I cell gene in embryonic endodermal and neural derivatives. Dev. Biol. 1995, 167, 294–306. [Google Scholar] [CrossRef] [PubMed]
- Dobbs, L.G.; Williams, M.C.; Gonzalez, R. Monoclonal antibodies specific to apical surfaces of rat alveolar type I cells bind to surfaces of cultured, but not freshly isolated, type II cells. Biochim. Biophys. Acta 1988, 970, 146–156. [Google Scholar] [CrossRef]
- Williams, M.C.; Cao, Y.; Hinds, A.; Rishi, A.K.; Wetterwald, A. T1 alpha protein is developmentally regulated and expressed by alveolar type I cells, choroid plexus, and ciliary epithelia of adult rats. Am. J. Respir. Cell Mol. Biol. 1996, 14, 577–585. [Google Scholar] [CrossRef] [PubMed]
- Schacht, V.; Ramirez, M.I.; Hong, Y.K.; Hirakawa, S.; Feng, D.; Harvey, N.; Williams, M.; Dvorak, A.M.; Dvorak, H.F.; Oliver, G.; et al. T1alpha/podoplanin deficiency disrupts normal lymphatic vasculature formation and causes lymphedema. Embo J. 2003, 22, 3546–3556. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, M.I.; Millien, G.; Hinds, A.; Cao, Y.; Seldin, D.C.; Williams, M.C. T1alpha, a lung type I cell differentiation gene, is required for normal lung cell proliferation and alveolus formation at birth. Dev. Biol. 2003, 256, 61–72. [Google Scholar] [CrossRef]
- Wetterwald, A.; Hoffstetter, W.; Cecchini, M.G.; Lanske, B.; Wagner, C.; Fleisch, H.; Atkinson, M. Characterization and cloning of the E11 antigen, a marker expressed by rat osteoblasts and osteocytes. Bone 1996, 18, 125–132. [Google Scholar] [CrossRef]
- Hirakawa, S.; Hong, Y.K.; Harvey, N.; Schacht, V.; Matsuda, K.; Libermann, T.; Detmar, M. Identification of vascular lineage-specific genes by transcriptional profiling of isolated blood vascular and lymphatic endothelial cells. Am. J. Pathol. 2003, 162, 575–586. [Google Scholar] [CrossRef]
- Petrova, T.V.; Mäkinen, T.; Mäkelä, T.P.; Saarela, J.; Virtanen, I.; Ferrell, R.E.; Finegold, D.N.; Kerjaschki, D.; Ylä-Herttuala, S.; Alitalo, K. Lymphatic endothelial reprogramming of vascular endothelial cells by the Prox-1 homeobox transcription factor. Embo J. 2002, 21, 4593–4599. [Google Scholar] [CrossRef]
- Adams, R.H.; Alitalo, K. Molecular regulation of angiogenesis and lymphangiogenesis. Nat. Rev. Mol. Cell Biol. 2007, 8, 464–478. [Google Scholar] [CrossRef]
- Alitalo, K.; Carmeliet, P. Molecular mechanisms of lymphangiogenesis in health and disease. Cancer Cell 2002, 1, 219–227. [Google Scholar] [CrossRef]
- Uhrin, P.; Zaujec, J.; Breuss, J.M.; Olcaydu, D.; Chrenek, P.; Stockinger, H.; Fuertbauer, E.; Moser, M.; Haiko, P.; Fässler, R.; et al. Novel function for blood platelets and podoplanin in developmental separation of blood and lymphatic circulation. Blood 2010, 115, 3997–4005. [Google Scholar] [CrossRef] [PubMed]
- Hess, P.R.; Rawnsley, D.R.; Jakus, Z.; Yang, Y.; Sweet, D.T.; Fu, J.; Herzog, B.; Lu, M.; Nieswandt, B.; Oliver, G.; et al. Platelets mediate lymphovenous hemostasis to maintain blood-lymphatic separation throughout life. J. Clin. Investig. 2014, 124, 273–284. [Google Scholar] [CrossRef]
- Koop, K.; Eikmans, M.; Wehland, M.; Baelde, H.; Ijpelaar, D.; Kreutz, R.; Kawachi, H.; Kerjaschki, D.; de Heer, E.; Bruijn, J.A. Selective loss of podoplanin protein expression accompanies proteinuria and precedes alterations in podocyte morphology in a spontaneous proteinuric rat model. Am. J. Pathol. 2008, 173, 315–326. [Google Scholar] [CrossRef] [PubMed]
- Ijpelaar, D.H.; Schulz, A.; Koop, K.; Schlesener, M.; Bruijn, J.A.; Kerjaschki, D.; Kreutz, R.; de Heer, E. Glomerular hypertrophy precedes albuminuria and segmental loss of podoplanin in podocytes in Munich-Wistar-Frömter rats. Am. J. Physiol. Ren. Physiol. 2008, 294, F758–F767. [Google Scholar] [CrossRef] [PubMed]
- Mishima, K.; Kato, Y.; Kaneko, M.K.; Nakazawa, Y.; Kunita, A.; Fujita, N.; Tsuruo, T.; Nishikawa, R.; Hirose, T.; Matsutani, M. Podoplanin expression in primary central nervous system germ cell tumors: A useful histological marker for the diagnosis of germinoma. Acta Neuropathol. 2006, 111, 563–568. [Google Scholar] [CrossRef] [PubMed]
- Mishima, K.; Kato, Y.; Kaneko, M.K.; Nishikawa, R.; Hirose, T.; Matsutani, M. Increased expression of podoplanin in malignant astrocytic tumors as a novel molecular marker of malignant progression. Acta Neuropathol. 2006, 111, 483–488. [Google Scholar] [CrossRef]
- Abe, S.; Morita, Y.; Kaneko, M.K.; Hanibuchi, M.; Tsujimoto, Y.; Goto, H.; Kakiuchi, S.; Aono, Y.; Huang, J.; Sato, S.; et al. A novel targeting therapy of malignant mesothelioma using anti-podoplanin antibody. J. Immunol. 2013, 190, 6239–6249. [Google Scholar] [CrossRef]
- Takagi, S.; Oh-hara, T.; Sato, S.; Gong, B.; Takami, M.; Fujita, N. Expression of Aggrus/podoplanin in bladder cancer and its role in pulmonary metastasis. Int. J. Cancer 2014, 134, 2605–2614. [Google Scholar] [CrossRef]
- Ariizumi, T.; Ogose, A.; Kawashima, H.; Hotta, T.; Li, G.; Xu, Y.; Umezu, H.; Sugai, M.; Endo, N. Expression of podoplanin in human bone and bone tumors: New marker of osteogenic and chondrogenic bone tumors. Pathol. Int. 2010, 60, 193–202. [Google Scholar] [CrossRef]
- Chang, M.C.; Vargas, S.O.; Hornick, J.L.; Hirsch, M.S.; Crum, C.P.; Nucci, M.R. Embryonic stem cell transcription factors and D2-40 (podoplanin) as diagnostic immunohistochemical markers in ovarian germ cell tumors. Int. J. Gynecol. Pathol. 2009, 28, 347–355. [Google Scholar] [CrossRef]
- Kato, Y.; Sasagawa, I.; Kaneko, M.; Osawa, M.; Fujita, N.; Tsuruo, T. Aggrus: A diagnostic marker that distinguishes seminoma from embryonal carcinoma in testicular germ cell tumors. Oncogene 2004, 23, 8552–8556. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, H.; Onimaru, M.; Koga, T.; Takeshita, M.; Yano, T.; Maehara, Y.; Nakamura, S.; Sueishi, K. High podoplanin expression in cancer cells predicts lower incidence of nodal metastasis in patients with lung squamous cell carcinoma. Pathol. Res. Pr. 2011, 207, 111–115. [Google Scholar] [CrossRef] [PubMed]
- Shimada, Y.; Ishii, G.; Nagai, K.; Atsumi, N.; Fujii, S.; Yamada, A.; Yamane, Y.; Hishida, T.; Nishimura, M.; Yoshida, J.; et al. Expression of podoplanin, CD44, and p63 in squamous cell carcinoma of the lung. Cancer Sci. 2009, 100, 2054–2059. [Google Scholar] [CrossRef] [PubMed]
- Hoshino, A.; Ishii, G.; Ito, T.; Aoyagi, K.; Ohtaki, Y.; Nagai, K.; Sasaki, H.; Ochiai, A. Podoplanin-positive fibroblasts enhance lung adenocarcinoma tumor formation: Podoplanin in fibroblast functions for tumor progression. Cancer Res. 2011, 71, 4769–4779. [Google Scholar] [CrossRef]
- Sasaki, K.; Sugai, T.; Ishida, K.; Osakabe, M.; Amano, H.; Kimura, H.; Sakuraba, M.; Kashiwa, K.; Kobayashi, S. Analysis of cancer-associated fibroblasts and the epithelial-mesenchymal transition in cutaneous basal cell carcinoma, squamous cell carcinoma, and malignant melanoma. Hum. Pathol. 2018, 79, 1–8. [Google Scholar] [CrossRef]
- Suzuki, J.; Aokage, K.; Neri, S.; Sakai, T.; Hashimoto, H.; Su, Y.; Yamazaki, S.; Nakamura, H.; Tane, K.; Miyoshi, T.; et al. Relationship between podoplanin-expressing cancer-associated fibroblasts and the immune microenvironment of early lung squamous cell carcinoma. Lung Cancer 2021, 153, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Pula, B.; Jethon, A.; Piotrowska, A.; Gomulkiewicz, A.; Owczarek, T.; Calik, J.; Wojnar, A.; Witkiewicz, W.; Rys, J.; Ugorski, M.; et al. Podoplanin expression by cancer-associated fibroblasts predicts poor outcome in invasive ductal breast carcinoma. Histopathology 2011, 59, 1249–1260. [Google Scholar] [CrossRef]
- Shindo, K.; Aishima, S.; Ohuchida, K.; Fujiwara, K.; Fujino, M.; Mizuuchi, Y.; Hattori, M.; Mizumoto, K.; Tanaka, M.; Oda, Y. Podoplanin expression in cancer-associated fibroblasts enhances tumor progression of invasive ductal carcinoma of the pancreas. Mol. Cancer 2013, 12, 168. [Google Scholar] [CrossRef]
- Yoshida, T.; Ishii, G.; Goto, K.; Neri, S.; Hashimoto, H.; Yoh, K.; Niho, S.; Umemura, S.; Matsumoto, S.; Ohmatsu, H.; et al. Podoplanin-positive cancer-associated fibroblasts in the tumor microenvironment induce primary resistance to EGFR-TKIs in lung adenocarcinoma with EGFR mutation. Clin. Cancer Res. 2015, 21, 642–651. [Google Scholar] [CrossRef]
- Sakai, T.; Aokage, K.; Neri, S.; Nakamura, H.; Nomura, S.; Tane, K.; Miyoshi, T.; Sugano, M.; Kojima, M.; Fujii, S.; et al. Link between tumor-promoting fibrous microenvironment and an immunosuppressive microenvironment in stage I lung adenocarcinoma. Lung Cancer 2018, 126, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Kalof, A.N.; Cooper, K. D2-40 immunohistochemistry—So far! Adv. Anat. Pathol. 2009, 16, 62–64. [Google Scholar] [CrossRef] [PubMed]
- Marks, A.; Sutherland, D.R.; Bailey, D.; Iglesias, J.; Law, J.; Lei, M.; Yeger, H.; Banerjee, D.; Baumal, R. Characterization and distribution of an oncofetal antigen (M2A antigen) expressed on testicular germ cell tumours. Br. J. Cancer 1999, 80, 569–578. [Google Scholar] [CrossRef] [PubMed]
- Schacht, V.; Dadras, S.S.; Johnson, L.A.; Jackson, D.G.; Hong, Y.K.; Detmar, M. Up-regulation of the lymphatic marker podoplanin, a mucin-type transmembrane glycoprotein, in human squamous cell carcinomas and germ cell tumors. Am. J. Pathol. 2005, 166, 913–921. [Google Scholar] [CrossRef]
- Ogasawara, S.; Kaneko, M.K.; Price, J.E.; Kato, Y. Characterization of anti-podoplanin monoclonal antibodies: Critical epitopes for neutralizing the interaction between podoplanin and CLEC-2. Hybridoma 2008, 27, 259–267. [Google Scholar] [CrossRef]
- Kato, Y.; Kaneko, M.K.; Kuno, A.; Uchiyama, N.; Amano, K.; Chiba, Y.; Hasegawa, Y.; Hirabayashi, J.; Narimatsu, H.; Mishima, K.; et al. Inhibition of tumor cell-induced platelet aggregation using a novel anti-podoplanin antibody reacting with its platelet-aggregation-stimulating domain. Biochem. Biophys. Res. Commun. 2006, 349, 1301–1307. [Google Scholar] [CrossRef]
- Kato, Y.; Vaidyanathan, G.; Kaneko, M.K.; Mishima, K.; Srivastava, N.; Chandramohan, V.; Pegram, C.; Keir, S.T.; Kuan, C.T.; Bigner, D.D.; et al. Evaluation of anti-podoplanin rat monoclonal antibody NZ-1 for targeting malignant gliomas. Nucl. Med. Biol. 2010, 37, 785–794. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, M.K.; Kunita, A.; Abe, S.; Tsujimoto, Y.; Fukayama, M.; Goto, K.; Sawa, Y.; Nishioka, Y.; Kato, Y. Chimeric anti-podoplanin antibody suppresses tumor metastasis through neutralization and antibody-dependent cellular cytotoxicity. Cancer Sci. 2012, 103, 1913–1919. [Google Scholar] [CrossRef]
- Kaji, C.; Tsujimoto, Y.; Kato Kaneko, M.; Kato, Y.; Sawa, Y. Immunohistochemical Examination of Novel Rat Monoclonal Antibodies against Mouse and Human Podoplanin. Acta Histochem. Cytochem. 2012, 45, 227–237. [Google Scholar] [CrossRef]
- Chandramohan, V.; Bao, X.; Kato Kaneko, M.; Kato, Y.; Keir, S.T.; Szafranski, S.E.; Kuan, C.T.; Pastan, I.H.; Bigner, D.D. Recombinant anti-podoplanin (NZ-1) immunotoxin for the treatment of malignant brain tumors. Int. J. Cancer 2013, 132, 2339–2348. [Google Scholar] [CrossRef]
- Bieniasz-Krzywiec, P.; Martin-Perez, R.; Ehling, M.; Garcia-Caballero, M.; Pinioti, S.; Pretto, S.; Kroes, R.; Aldeni, C.; Di Matteo, M.; Prenen, H.; et al. Podoplanin-Expressing Macrophages Promote Lymphangiogenesis and Lymphoinvasion in Breast Cancer. Cell Metab. 2019, 30, 917–936. [Google Scholar] [CrossRef] [PubMed]
- Kuwata, T.; Yoneda, K.; Mori, M.; Kanayama, M.; Kuroda, K.; Kaneko, M.K.; Kato, Y.; Tanaka, F. Detection of Circulating Tumor Cells (CTCs) in Malignant Pleural Mesothelioma (MPM) with the "Universal" CTC-Chip and An Anti-Podoplanin Antibody NZ-1.2. Cells 2020, 9, 888. [Google Scholar] [CrossRef] [PubMed]
- Nishinaga, Y.; Sato, K.; Yasui, H.; Taki, S.; Takahashi, K.; Shimizu, M.; Endo, R.; Koike, C.; Kuramoto, N.; Nakamura, S.; et al. Targeted Phototherapy for Malignant Pleural Mesothelioma: Near-Infrared Photoimmunotherapy Targeting Podoplanin. Cells 2020, 9, 1019. [Google Scholar] [CrossRef]
- Ando, K.; Fujino, N.; Mitani, K.; Ota, C.; Okada, Y.; Kondo, T.; Mizobuchi, T.; Kurihara, M.; Suzuki, K.; Hoshika, Y.; et al. Isolation of individual cellular components from lung tissues of patients with lymphangioleiomyomatosis. Am. J. Physiol. Lung Cell Mol. Physiol. 2016, 310, L899–L908. [Google Scholar] [CrossRef]
- Fujii, Y.; Kaneko, M.; Neyazaki, M.; Nogi, T.; Kato, Y.; Takagi, J. PA tag: A versatile protein tagging system using a super high affinity antibody against a dodecapeptide derived from human podoplanin. Protein Expr. Purif. 2014, 95, 240–247. [Google Scholar] [CrossRef]
- Fujii, Y.; Matsunaga, Y.; Arimori, T.; Kitago, Y.; Ogasawara, S.; Kaneko, M.K.; Kato, Y.; Takagi, J. Tailored placement of a turn-forming PA tag into the structured domain of a protein to probe its conformational state. J. Cell Sci. 2016, 129, 1512–1522. [Google Scholar] [CrossRef]
- Tamura, R.; Oi, R.; Akashi, S.; Kaneko, M.K.; Kato, Y.; Nogi, T. Application of the NZ-1 Fab as a crystallization chaperone for PA tag-inserted target proteins. Protein Sci. 2019, 28, 823–836. [Google Scholar] [CrossRef]
- Yoneda, K.; Kuwata, T.; Chikaishi, Y.; Mori, M.; Kanayama, M.; Takenaka, M.; Oka, S.; Hirai, A.; Imanishi, N.; Kuroda, K.; et al. Detection of circulating tumor cells with a novel microfluidic system in malignant pleural mesothelioma. Cancer Sci. 2019, 110, 726–733. [Google Scholar] [CrossRef]
- Yoneda, K.; Chikaishi, Y.; Kuwata, T.; Ohnaga, T.; Tanaka, F. Capture of mesothelioma cells with 'universal' CTC-chip. Oncol. Lett. 2018, 15, 2635–2640. [Google Scholar] [CrossRef] [PubMed]
- Kanayama, M.; Oyama, R.; Mori, M.; Taira, A.; Shinohara, S.; Kuwata, T.; Takenaka, M.; Yoneda, K.; Kuroda, K.; Ohnaga, T.; et al. Novel circulating tumor cell-detection chip combining conventional podoplanin and EGFR antibodies for all histological malignant pleural mesothelioma. Oncol. Lett. 2021, 22, 522. [Google Scholar] [CrossRef]
- Tanaka, M.; Kijima, H.; Shimada, H.; Makuuchi, H.; Ozawa, S.; Inokuchi, S. Expression of podoplanin and vimentin is correlated with prognosis in esophageal squamous cell carcinoma. Mol. Med. Rep. 2015, 12, 4029–4036. [Google Scholar] [CrossRef] [PubMed]
- Rahadiani, N.; Ikeda, J.; Makino, T.; Tian, T.; Qiu, Y.; Mamat, S.; Wang, Y.; Doki, Y.; Aozasa, K.; Morii, E. Tumorigenic role of podoplanin in esophageal squamous-cell carcinoma. Ann. Surg. Oncol. 2010, 17, 1311–1323. [Google Scholar] [CrossRef]
- Nakashima, Y.; Yoshinaga, K.; Kitao, H.; Ando, K.; Kimura, Y.; Saeki, H.; Oki, E.; Morita, M.; Kakeji, Y.; Hirahashi, M.; et al. Podoplanin is expressed at the invasive front of esophageal squamous cell carcinomas and is involved in collective cell invasion. Cancer Sci. 2013, 104, 1718–1725. [Google Scholar] [CrossRef] [PubMed]
- Kreppel, M.; Scheer, M.; Drebber, U.; Ritter, L.; Zöller, J.E. Impact of podoplanin expression in oral squamous cell carcinoma: Clinical and histopathologic correlations. Virchows Arch. 2010, 456, 473–482. [Google Scholar] [CrossRef] [PubMed]
- Kreppel, M.; Drebber, U.; Wedemeyer, I.; Eich, H.T.; Backhaus, T.; Zöller, J.E.; Scheer, M. Podoplanin expression predicts prognosis in patients with oral squamous cell carcinoma treated with neoadjuvant radiochemotherapy. Oral Oncol. 2011, 47, 873–878. [Google Scholar] [CrossRef]
- Vinicius de, L.V.; Scapulatempo, C.; Perpetuo, N.M.; Mohamed, F.; de Carvalho, T.S.; de Oliveira, A.T.; Segalla, J.G.; Carvalho, A.L. Prognostic and risk factors in patients with locally advanced cutaneous squamous cell carcinoma of the trunk and extremities. J. Ski. Cancer 2011, 2011, 420796. [Google Scholar] [CrossRef]
- Kim, H.Y.; Rha, K.S.; Shim, G.A.; Kim, J.H.; Kim, J.M.; Huang, S.M.; Koo, B.S. Podoplanin is involved in the prognosis of head and neck squamous cell carcinoma through interaction with VEGF-C. Oncol. Rep. 2015, 34, 833–842. [Google Scholar] [CrossRef][Green Version]
- Xia, Y.; Liu, L.; Xiong, Y.; Bai, Q.; Wang, J.; Xi, W.; Qu, Y.; Xu, J.; Guo, J. Podoplanin associates with adverse postoperative prognosis of patients with clear cell renal cell carcinoma. Cancer Sci. 2016, 107, 1243–1249. [Google Scholar] [CrossRef]
- Motomura, K.; Natsume, A.; Watanabe, R.; Ito, I.; Kato, Y.; Momota, H.; Nishikawa, R.; Mishima, K.; Nakasu, Y.; Abe, T.; et al. Immunohistochemical analysis-based proteomic subclassification of newly diagnosed glioblastomas. Cancer Sci. 2012, 103, 1871–1879. [Google Scholar] [CrossRef]
- Schoppmann, S.F.; Jesch, B.; Riegler, M.F.; Maroske, F.; Schwameis, K.; Jomrich, G.; Birner, P. Podoplanin expressing cancer associated fibroblasts are associated with unfavourable prognosis in adenocarcinoma of the esophagus. Clin. Exp. Metastasis 2013, 30, 441–446. [Google Scholar] [CrossRef]
- Obulkasim, H.; Shi, X.; Wang, J.; Li, J.; Dai, B.; Wu, P.; Wang, S.; Wang, X.; Ding, Y. Podoplanin is an important stromal prognostic marker in perihilar cholangiocarcinoma. Oncol. Lett. 2018, 15, 137–146. [Google Scholar] [CrossRef] [PubMed]
- Hantusch, B.; Kalt, R.; Krieger, S.; Puri, C.; Kerjaschki, D. Sp1/Sp3 and DNA-methylation contribute to basal transcriptional activation of human podoplanin in MG63 versus Saos-2 osteoblastic cells. BMC Mol. Biol. 2007, 8, 20. [Google Scholar] [CrossRef] [PubMed]
- Durchdewald, M.; Guinea-Viniegra, J.; Haag, D.; Riehl, A.; Lichter, P.; Hahn, M.; Wagner, E.F.; Angel, P.; Hess, J. Podoplanin is a novel fos target gene in skin carcinogenesis. Cancer Res. 2008, 68, 6877–6883. [Google Scholar] [CrossRef] [PubMed]
- Kunita, A.; Kashima, T.G.; Ohazama, A.; Grigoriadis, A.E.; Fukayama, M. Podoplanin is regulated by AP-1 and promotes platelet aggregation and cell migration in osteosarcoma. Am. J. Pathol. 2011, 179, 1041–1049. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Chen, C.S.; Ichikawa, H.; Goldberg, G.S. SRC induces podoplanin expression to promote cell migration. J. Biol. Chem. 2010, 285, 9649–9656. [Google Scholar] [CrossRef]
- Peterziel, H.; Müller, J.; Danner, A.; Barbus, S.; Liu, H.K.; Radlwimmer, B.; Pietsch, T.; Lichter, P.; Schütz, G.; Hess, J.; et al. Expression of podoplanin in human astrocytic brain tumors is controlled by the PI3K-AKT-AP-1 signaling pathway and promoter methylation. Neuro. Oncol. 2012, 14, 426–439. [Google Scholar] [CrossRef] [PubMed]
- Mei, Y.; Zhang, P.; Zuo, H.; Clark, D.; Xia, R.; Li, J.; Liu, Z.; Mao, L. Ebp1 activates podoplanin expression and contributes to oral tumorigenesis. Oncogene 2014, 33, 3839–3850. [Google Scholar] [CrossRef]
- Kunita, A.; Baeriswyl, V.; Meda, C.; Cabuy, E.; Takeshita, K.; Giraudo, E.; Wicki, A.; Fukayama, M.; Christofori, G. Inflammatory Cytokines Induce Podoplanin Expression at the Tumor Invasive Front. Am. J. Pathol. 2018, 188, 1276–1288. [Google Scholar] [CrossRef]
- Ohta, M.; Abe, A.; Ohno, F.; Hasegawa, Y.; Tanaka, H.; Maseki, S.; Kondo, E.; Kurita, K.; Nakanishi, H. Positive and negative regulation of podoplanin expression by TGF-β and histone deacetylase inhibitors in oral and pharyngeal squamous cell carcinoma cell lines. Oral Oncol. 2013, 49, 20–26. [Google Scholar] [CrossRef]
- Suzuki, H.; Kato, Y.; Kaneko, M.K.; Okita, Y.; Narimatsu, H.; Kato, M. Induction of podoplanin by transforming growth factor-beta in human fibrosarcoma. FEBS Lett. 2008, 582, 341–345. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Lambert, A.W.; Weinberg, R.A. Linking EMT programmes to normal and neoplastic epithelial stem cells. Nat. Rev. Cancer 2021, 21, 325–338. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Antin, P.; Berx, G.; Blanpain, C.; Brabletz, T.; Bronner, M.; Campbell, K.; Cano, A.; Casanova, J.; Christofori, G.; et al. Guidelines and definitions for research on epithelial-mesenchymal transition. Nat. Rev. Mol. Cell Biol. 2020, 21, 341–352. [Google Scholar] [CrossRef] [PubMed]
- Renart, J.; Carrasco-Ramírez, P.; Fernández-Muñoz, B.; Martín-Villar, E.; Montero, L.; Yurrita, M.M.; Quintanilla, M. New insights into the role of podoplanin in epithelial-mesenchymal transition. Int. Rev. Cell Mol. Biol. 2015, 317, 185–239. [Google Scholar] [CrossRef]
- Senbanjo, L.T.; Chellaiah, M.A. CD44: A Multifunctional Cell Surface Adhesion Receptor Is a Regulator of Progression and Metastasis of Cancer Cells. Front. Cell Dev. Biol. 2017, 5, 18. [Google Scholar] [CrossRef] [PubMed]
- Martín-Villar, E.; Fernández-Muñoz, B.; Parsons, M.; Yurrita, M.M.; Megías, D.; Pérez-Gómez, E.; Jones, G.E.; Quintanilla, M. Podoplanin associates with CD44 to promote directional cell migration. Mol. Biol. Cell 2010, 21, 4387–4399. [Google Scholar] [CrossRef]
- Li, Y.Y.; Zhou, C.X.; Gao, Y. Podoplanin promotes the invasion of oral squamous cell carcinoma in coordination with MT1-MMP and Rho GTPases. Am. J. Cancer Res. 2015, 5, 514–529. [Google Scholar] [PubMed]
- Asif, P.J.; Longobardi, C.; Hahne, M.; Medema, J.P. The Role of Cancer-Associated Fibroblasts in Cancer Invasion and Metastasis. Cancers 2021, 13, 4720. [Google Scholar] [CrossRef]
- Li, Y.Y.; Zhou, C.X.; Gao, Y. Interaction between oral squamous cell carcinoma cells and fibroblasts through TGF-β1 mediated by podoplanin. Exp. Cell Res. 2018, 369, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Masi, I.; Caprara, V.; Bagnato, A.; Rosanò, L. Tumor Cellular and Microenvironmental Cues Controlling Invadopodia Formation. Front. Cell Dev. Biol. 2020, 8, 584181. [Google Scholar] [CrossRef]
- Aseervatham, J. Cytoskeletal Remodeling in Cancer. Biology 2020, 9, 385. [Google Scholar] [CrossRef]
- Martín-Villar, E.; Borda-d’Agua, B.; Carrasco-Ramirez, P.; Renart, J.; Parsons, M.; Quintanilla, M.; Jones, G.E. Podoplanin mediates ECM degradation by squamous carcinoma cells through control of invadopodia stability. Oncogene 2015, 34, 4531–4544. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.S.; Xianglan, Z.; Park, K.K.; Chung, W.Y. Functional invadopodia formation through stabilization of the PDPN transcript by IMP-3 and cancer-stromal crosstalk for PDPN expression. Carcinogenesis 2012, 33, 2135–2146. [Google Scholar] [CrossRef] [PubMed]
- Zhao, P.; Xu, Y.; Wei, Y.; Qiu, Q.; Chew, T.L.; Kang, Y.; Cheng, C. The CD44s splice isoform is a central mediator for invadopodia activity. J. Cell Sci. 2016, 129, 1355–1365. [Google Scholar] [CrossRef]
- Grass, G.D.; Tolliver, L.B.; Bratoeva, M.; Toole, B.P. CD147, CD44, and the epidermal growth factor receptor (EGFR) signaling pathway cooperate to regulate breast epithelial cell invasiveness. J. Biol. Chem. 2013, 288, 26089–26104. [Google Scholar] [CrossRef]
- Te Boekhorst, V.; Preziosi, L.; Friedl, P. Plasticity of Cell Migration In Vivo and In Silico. Annu. Rev. Cell Dev. Biol. 2016, 32, 491–526. [Google Scholar] [CrossRef] [PubMed]
- Van Helvert, S.; Storm, C.; Friedl, P. Mechanoreciprocity in cell migration. Nat. Cell Biol. 2018, 20, 8–20. [Google Scholar] [CrossRef]
- Stuelten, C.H.; Parent, C.A.; Montell, D.J. Cell motility in cancer invasion and metastasis: Insights from simple model organisms. Nat. Rev. Cancer 2018, 18, 296–312. [Google Scholar] [CrossRef]
- Wicki, A.; Lehembre, F.; Wick, N.; Hantusch, B.; Kerjaschki, D.; Christofori, G. Tumor invasion in the absence of epithelial-mesenchymal transition: Podoplanin-mediated remodeling of the actin cytoskeleton. Cancer Cell 2006, 9, 261–272. [Google Scholar] [CrossRef]
- Tozluoğlu, M.; Tournier, A.L.; Jenkins, R.P.; Hooper, S.; Bates, P.A.; Sahai, E. Matrix geometry determines optimal cancer cell migration strategy and modulates response to interventions. Nat. Cell Biol. 2013, 15, 751–762. [Google Scholar] [CrossRef]
- Pandya, P.; Orgaz, J.L.; Sanz-Moreno, V. Modes of invasion during tumour dissemination. Mol. Oncol. 2017, 11, 5–27. [Google Scholar] [CrossRef]
- Wyckoff, J.B.; Pinner, S.E.; Gschmeissner, S.; Condeelis, J.S.; Sahai, E. ROCK- and myosin-dependent matrix deformation enables protease-independent tumor-cell invasion in vivo. Curr. Biol. 2006, 16, 1515–1523. [Google Scholar] [CrossRef] [PubMed]
- De Winde, C.M.; George, S.L.; Crosas-Molist, E.; Hari-Gupta, Y.; Arp, A.B.; Benjamin, A.C.; Millward, L.J.; Makris, S.; Carver, A.; Imperatore, V.; et al. Podoplanin drives dedifferentiation and amoeboid invasion of melanoma. iScience 2021, 24, 102976. [Google Scholar] [CrossRef] [PubMed]
- Braun, A.; Anders, H.J.; Gudermann, T.; Mammadova-Bach, E. Platelet-Cancer Interplay: Molecular Mechanisms and New Therapeutic Avenues. Front. Oncol. 2021, 11, 665534. [Google Scholar] [CrossRef] [PubMed]
- Takemoto, A.; Miyata, K.; Fujita, N. Platelet-activating factor podoplanin: From discovery to drug development. Cancer Metastasis Rev. 2017, 36, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Fujita, N.; Takagi, S. The impact of Aggrus/podoplanin on platelet aggregation and tumour metastasis. J. Biochem. 2012, 152, 407–413. [Google Scholar] [CrossRef] [PubMed]
- David, C.J.; Massagué, J. Contextual determinants of TGFβ action in development, immunity and cancer. Nat. Rev. Mol. Cell Biol. 2018, 19, 419–435. [Google Scholar] [CrossRef]
- Wilson, M.M.; Weinberg, R.A.; Lees, J.A.; Guen, V.J. Emerging Mechanisms by which EMT Programs Control Stemness. Trends Cancer 2020, 6, 775–780. [Google Scholar] [CrossRef]
- Atsumi, N.; Ishii, G.; Kojima, M.; Sanada, M.; Fujii, S.; Ochiai, A. Podoplanin, a novel marker of tumor-initiating cells in human squamous cell carcinoma A431. Biochem. Biophys. Res. Commun. 2008, 373, 36–41. [Google Scholar] [CrossRef]
- Miyashita, T.; Higuchi, Y.; Kojima, M.; Ochiai, A.; Ishii, G. Single cell time-lapse analysis reveals that podoplanin enhances cell survival and colony formation capacity of squamous cell carcinoma cells. Sci. Rep. 2017, 7, 39971. [Google Scholar] [CrossRef]
- Miyashita, T.; Neri, S.; Hashimoto, H.; Akutsu, A.; Sugano, M.; Fujii, S.; Ochiai, A.; Ishii, G. Fibroblasts-dependent invasion of podoplanin-positive cancer stem cells in squamous cell carcinoma. J. Cell Physiol. 2020, 235, 7251–7260. [Google Scholar] [CrossRef] [PubMed]
- Ito, M.; Ishii, G.; Nagai, K.; Maeda, R.; Nakano, Y.; Ochiai, A. Prognostic impact of cancer-associated stromal cells in patients with stage I lung adenocarcinoma. Chest 2012, 142, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Ito, S.; Ishii, G.; Hoshino, A.; Hashimoto, H.; Neri, S.; Kuwata, T.; Higashi, M.; Nagai, K.; Ochiai, A. Tumor promoting effect of podoplanin-positive fibroblasts is mediated by enhanced RhoA activity. Biochem. Biophys. Res. Commun. 2012, 422, 194–199. [Google Scholar] [CrossRef] [PubMed]
- Neri, S.; Ishii, G.; Taira, T.; Hishida, T.; Yoshida, J.; Nishimura, M.; Nagai, K.; Ochiai, A. Recruitment of podoplanin positive cancer-associated fibroblasts in metastatic lymph nodes predicts poor prognosis in pathological N2 stage III lung adenocarcinoma. Ann. Surg. Oncol. 2012, 19, 3953–3962. [Google Scholar] [CrossRef]
- Nakamura, H.; Ichikawa, T.; Nakasone, S.; Miyoshi, T.; Sugano, M.; Kojima, M.; Fujii, S.; Ochiai, A.; Kuwata, T.; Aokage, K.; et al. Abundant tumor promoting stromal cells in lung adenocarcinoma with hypoxic regions. Lung Cancer 2018, 115, 56–63. [Google Scholar] [CrossRef]
- Komohara, Y.; Jinushi, M.; Takeya, M. Clinical significance of macrophage heterogeneity in human malignant tumors. Cancer Sci. 2014, 105, 1–8. [Google Scholar] [CrossRef]
- Fridman, W.H.; Pagès, F.; Sautès-Fridman, C.; Galon, J. The immune contexture in human tumours: Impact on clinical outcome. Nat. Rev. Cancer 2012, 12, 298–306. [Google Scholar] [CrossRef]
- Chen, W.; Ten Dijke, P. Immunoregulation by members of the TGFβ superfamily. Nat. Rev. Immunol. 2016, 16, 723–740. [Google Scholar] [CrossRef]
- Liu, S.; Ren, J.; Ten Dijke, P. Targeting TGFβ signal transduction for cancer therapy. Signal. Transduct. Target 2021, 6, 8. [Google Scholar] [CrossRef]
- Chihara, N.; Madi, A.; Kondo, T.; Zhang, H.; Acharya, N.; Singer, M.; Nyman, J.; Marjanovic, N.D.; Kowalczyk, M.S.; Wang, C.; et al. Induction and transcriptional regulation of the co-inhibitory gene module in T cells. Nature 2018, 558, 454–459. [Google Scholar] [CrossRef]
- Peters, A.; Burkett, P.R.; Sobel, R.A.; Buckley, C.D.; Watson, S.P.; Bettelli, E.; Kuchroo, V.K. Podoplanin negatively regulates CD4+ effector T cell responses. J. Clin. Investig. 2015, 125, 129–140. [Google Scholar] [CrossRef]
- Li, F.; Chen, Y.; Pang, M.; Yang, P.; Jing, H. Immune checkpoint inhibitors and cellular treatment for lymphoma immunotherapy. Clin. Exp. Immunol. 2021, 205, 1–11. [Google Scholar] [CrossRef]
- Qin, S.; Xu, L.; Yi, M.; Yu, S.; Wu, K.; Luo, S. Novel immune checkpoint targets: Moving beyond PD-1 and CTLA-4. Mol. Cancer 2019, 18, 155. [Google Scholar] [CrossRef] [PubMed]
- Astarita, J.L.; Keerthivasan, S.; Husain, B.; Şenbabaoğlu, Y.; Verschueren, E.; Gierke, S.; Pham, V.C.; Peterson, S.M.; Chalouni, C.; Pierce, A.A.; et al. The neutrophil protein CD177 is a novel PDPN receptor that regulates human cancer-associated fibroblast physiology. PLoS ONE 2021, 16, e0260800. [Google Scholar] [CrossRef]
- Tsao, L.C.; Force, J.; Hartman, Z.C. Mechanisms of Therapeutic Antitumor Monoclonal Antibodies. Cancer Res. 2021, 81, 4641–4651. [Google Scholar] [CrossRef] [PubMed]
- Li, B.T.; Smit, E.F.; Goto, Y.; Nakagawa, K.; Udagawa, H.; Mazières, J.; Nagasaka, M.; Bazhenova, L.; Saltos, A.N.; Felip, E.; et al. Trastuzumab Deruxtecan in HER2-Mutant Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2021, 386, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Modi, S.; Saura, C.; Yamashita, T.; Park, Y.H.; Kim, S.B.; Tamura, K.; Andre, F.; Iwata, H.; Ito, Y.; Tsurutani, J.; et al. Trastuzumab Deruxtecan in Previously Treated HER2-Positive Breast Cancer. N. Engl. J. Med. 2020, 382, 610–621. [Google Scholar] [CrossRef] [PubMed]
- Shitara, K.; Bang, Y.J.; Iwasa, S.; Sugimoto, N.; Ryu, M.H.; Sakai, D.; Chung, H.C.; Kawakami, H.; Yabusaki, H.; Lee, J.; et al. Trastuzumab Deruxtecan in Previously Treated HER2-Positive Gastric Cancer. N. Engl. J. Med. 2020, 382, 2419–2430. [Google Scholar] [CrossRef]
- Heath, E.I.; Rosenberg, J.E. The biology and rationale of targeting nectin-4 in urothelial carcinoma. Nat. Rev. Urol. 2021, 18, 93–103. [Google Scholar] [CrossRef]
- Bardia, A.; Hurvitz, S.A.; Tolaney, S.M.; Loirat, D.; Punie, K.; Oliveira, M.; Brufsky, A.; Sardesai, S.D.; Kalinsky, K.; Zelnak, A.B.; et al. Sacituzumab Govitecan in Metastatic Triple-Negative Breast Cancer. N. Engl. J. Med. 2021, 384, 1529–1541. [Google Scholar] [CrossRef]
- Goldenberg, D.M.; Stein, R.; Sharkey, R.M. The emergence of trophoblast cell-surface antigen 2 (TROP-2) as a novel cancer target. Oncotarget 2018, 9, 28989–29006. [Google Scholar] [CrossRef] [PubMed]
- Nakazawa, Y.; Takagi, S.; Sato, S.; Oh-hara, T.; Koike, S.; Takami, M.; Arai, H.; Fujita, N. Prevention of hematogenous metastasis by neutralizing mice and its chimeric anti-Aggrus/podoplanin antibodies. Cancer Sci. 2011, 102, 2051–2057. [Google Scholar] [CrossRef] [PubMed]
- Takagi, S.; Sato, S.; Oh-hara, T.; Takami, M.; Koike, S.; Mishima, Y.; Hatake, K.; Fujita, N. Platelets promote tumor growth and metastasis via direct interaction between Aggrus/podoplanin and CLEC-2. PLoS ONE 2013, 8, e73609. [Google Scholar] [CrossRef] [PubMed]
- Ukaji, T.; Takemoto, A.; Katayama, R.; Takeuchi, K.; Fujita, N. A safety study of newly generated anti-podoplanin-neutralizing antibody in cynomolgus monkey (Macaca fascicularis). Oncotarget 2018, 9, 33322–33336. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kato, Y.; Kunita, A.; Abe, S.; Ogasawara, S.; Fujii, Y.; Oki, H.; Fukayama, M.; Nishioka, Y.; Kaneko, M.K. The chimeric antibody chLpMab-7 targeting human podoplanin suppresses pulmonary metastasis via ADCC and CDC rather than via its neutralizing activity. Oncotarget 2015, 6, 36003–36018. [Google Scholar] [CrossRef]
- Scott, A.M.; Wolchok, J.D.; Old, L.J. Antibody therapy of cancer. Nat. Rev. Cancer 2012, 12, 278–287. [Google Scholar] [CrossRef]
- Zahavi, D.; Weiner, L. Monoclonal Antibodies in Cancer Therapy. Antibodies 2020, 9, 34. [Google Scholar] [CrossRef]
- Kaneko, M.K.; Honma, R.; Ogasawara, S.; Fujii, Y.; Nakamura, T.; Saidoh, N.; Takagi, M.; Kagawa, Y.; Konnai, S.; Kato, Y. PMab-38 Recognizes Canine Podoplanin of Squamous Cell Carcinomas. Monoclon. Antib. Immunodiagn. Immunother. 2016, 35, 263–266. [Google Scholar] [CrossRef]
- Ito, A.; Ohta, M.; Kato, Y.; Inada, S.; Kato, T.; Nakata, S.; Yatabe, Y.; Goto, M.; Kaneda, N.; Kurita, K.; et al. A Real-Time Near-Infrared Fluorescence Imaging Method for the Detection of Oral Cancers in Mice Using an Indocyanine Green-Labeled Podoplanin Antibody. Technol. Cancer Res. Treat. 2018, 17, 1533033818767936. [Google Scholar] [CrossRef]
- Kato, Y.; Ohishi, T.; Kawada, M.; Maekawa, N.; Konnai, S.; Itai, S.; Yamada, S.; Kaneko, M.K. The mouse-canine chimeric anti-dog podoplanin antibody P38B exerts antitumor activity in mouse xenograft models. Biochem. Biophys. Rep. 2019, 17, 23–26. [Google Scholar] [CrossRef]
- Kato, Y.; Ito, Y.; Ohishi, T.; Kawada, M.; Nakamura, T.; Sayama, Y.; Sano, M.; Asano, T.; Yanaka, M.; Okamoto, S.; et al. Antibody-Drug Conjugates Using Mouse-Canine Chimeric Anti-Dog Podoplanin Antibody Exerts Antitumor Activity in a Mouse Xenograft Model. Monoclon. Antib. Immunodiagn. Immunother. 2020, 39, 37–44. [Google Scholar] [CrossRef]
- Pantelyushin, S.; Ranninger, E.; Guerrera, D.; Hutter, G.; Maake, C.; Markkanen, E.; Bettschart-Wolfensberger, R.; Rohrer Bley, C.; Läubli, H.; Vom Berg, J. Cross-Reactivity and Functionality of Approved Human Immune Checkpoint Blockers in Dogs. Cancers 2021, 13, 785. [Google Scholar] [CrossRef]
- Maekawa, N.; Konnai, S.; Nishimura, M.; Kagawa, Y.; Takagi, S.; Hosoya, K.; Ohta, H.; Kim, S.; Okagawa, T.; Izumi, Y.; et al. PD-L1 immunohistochemistry for canine cancers and clinical benefit of anti-PD-L1 antibody in dogs with pulmonary metastatic oral malignant melanoma. NPJ Precis. Oncol. 2021, 5, 10. [Google Scholar] [CrossRef] [PubMed]
- Klingemann, H. Immunotherapy for Dogs: Running Behind Humans. Front. Immunol. 2018, 9, 133. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Asano, T.; Sano, M.; Takei, J.; Hosono, H.; Nanamiya, R.; Nakamura, T.; Yanaka, M.; Harada, H.; Fukui, M.; et al. Development of Monoclonal Antibody PMab-269 Against California Sea Lion Podoplanin. Monoclon. Antib. Immunodiagn. Immunother. 2021, 40, 124–133. [Google Scholar] [CrossRef] [PubMed]
- Hosono, H.; Asano, T.; Takei, J.; Sano, M.; Tanaka, T.; Kaneko, M.K.; Kato, Y. Development of an Anti-Elephant Podoplanin Monoclonal Antibody PMab-265 for Flow Cytometry. Monoclon. Antib. Immunodiagn. Immunother. 2021, 40, 141–145. [Google Scholar] [CrossRef]
- Kato, Y.; Furusawa, Y.; Sano, M.; Takei, J.; Nakamura, T.; Yanaka, M.; Okamoto, S.; Handa, S.; Komatsu, Y.; Asano, T.; et al. Development of an Anti-Sheep Podoplanin Monoclonal Antibody PMab-256 for Immunohistochemical Analysis of Lymphatic Endothelial Cells. Monoclon. Antib. Immunodiagn. Immunother. 2020, 39, 82–90. [Google Scholar] [CrossRef]
- Takei, J.; Furusawa, Y.; Yamada, S.; Nakamura, T.; Sayama, Y.; Sano, M.; Konnai, S.; Kobayashi, A.; Harada, H.; Kaneko, M.K.; et al. PMab-247 Detects Bear Podoplanin in Immunohistochemical Analysis. Monoclon. Antib. Immunodiagn. Immunother. 2019, 38, 171–174. [Google Scholar] [CrossRef] [PubMed]
- Kato, Y.; Yamada, S.; Furusawa, Y.; Itai, S.; Nakamura, T.; Yanaka, M.; Sano, M.; Harada, H.; Fukui, M.; Kaneko, M.K. PMab-213: A monoclonal antibody for immunohistochemical analysis against pig podoplanin. Monoclon. Antib. Immunodiagn. Immunother. 2019, 38, 18–24. [Google Scholar] [CrossRef]
- Kato, Y.; Furusawa, Y.; Itai, S.; Takei, J.; Nakamura, T.; Sano, M.; Harada, H.; Yamada, S.; Kaneko, M.K. Establishment of an Anticetacean Podoplanin Monoclonal Antibody PMab-237 for Immunohistochemical Analysis. Monoclon. Antib. Immunodiagn. Immunother. 2019, 38, 108–113. [Google Scholar] [CrossRef]
- Furusawa, Y.; Yamada, S.; Nakamura, T.; Sano, M.; Sayama, Y.; Itai, S.; Takei, J.; Harada, H.; Fukui, M.; Kaneko, M.K.; et al. PMab-235: A monoclonal antibody for immunohistochemical analysis against goat podoplanin. Heliyon 2019, 5, e02063. [Google Scholar] [CrossRef] [PubMed]
- Furusawa, Y.; Yamada, S.; Itai, S.; Nakamura, T.; Takei, J.; Sano, M.; Harada, H.; Fukui, M.; Kaneko, M.K.; Kato, Y. Establishment of a monoclonal antibody PMab-233 for immunohistochemical analysis against Tasmanian devil podoplanin. Biochem. Biophys. Rep. 2019, 18, 100631. [Google Scholar] [CrossRef] [PubMed]
- Furusawa, Y.; Kaneko, M.K.; Nakamura, T.; Itai, S.; Fukui, M.; Harada, H.; Yamada, S.; Kato, Y. Establishment of a Monoclonal Antibody PMab-231 for Tiger Podoplanin. Monoclon. Antib. Immunodiagn. Immunother. 2019, 38, 89–95. [Google Scholar] [CrossRef]
- Furusawa, Y.; Yamada, S.; Itai, S.; Sano, M.; Nakamura, T.; Yanaka, M.; Handa, S.; Mizuno, T.; Maeda, K.; Fukui, M.; et al. Establishment of Monoclonal Antibody PMab-202 Against Horse Podoplanin. Monoclon. Antib. Immunodiagn. Immunother. 2018, 37, 233–237. [Google Scholar] [CrossRef] [PubMed]
- Yamada, S.; Itai, S.; Nakamura, T.; Yanaka, M.; Saidoh, N.; Chang, Y.W.; Handa, S.; Harada, H.; Kagawa, Y.; Ichii, O.; et al. PMab-52: Specific and Sensitive Monoclonal Antibody Against Cat Podoplanin for Immunohistochemistry. Monoclon. Antib. Immunodiagn. Immunother. 2017, 36, 224–230. [Google Scholar] [CrossRef]
- Honma, R.; Ogasawara, S.; Kaneko, M.; Fujii, Y.; Oki, H.; Nakamura, T.; Takagi, M.; Konnai, S.; Kato, Y. PMab-44 Detects Bovine Podoplanin in Immunohistochemistry. Monoclon. Antib. Immunodiagn. Immunother. 2016, 35, 186–190. [Google Scholar] [CrossRef]
- Honma, R.; Kaneko, M.K.; Ogasawara, S.; Fujii, Y.; Konnai, S.; Takagi, M.; Kato, Y. Specific Detection of Dog Podoplanin Expressed in Renal Glomerulus by a Novel Monoclonal Antibody PMab-38 in Immunohistochemistry. Monoclon. Antib. Immunodiagn. Immunother. 2016, 35, 212–216. [Google Scholar] [CrossRef]
- Honma, R.; Fujii, Y.; Ogasawara, S.; Oki, H.; Liu, X.; Nakamura, T.; Kaneko, M.K.; Takagi, M.; Kato, Y. Establishment of a novel monoclonal antibody PMab-32 against rabbit podoplanin. Monoclon. Antib. Immunodiagn. Immunother. 2016, 35, 41–47. [Google Scholar] [CrossRef]
- Oki, H.; Honma, R.; Ogasawara, S.; Fujii, Y.; Liu, X.; Takagi, M.; Kaneko, M.K.; Kato, Y. Development of Sensitive Monoclonal Antibody PMab-2 Against Rat Podoplanin. Monoclon. Antib. Immunodiagn. Immunother. 2015, 34, 396–403. [Google Scholar] [CrossRef]
- Mitsunaga, M.; Ogawa, M.; Kosaka, N.; Rosenblum, L.T.; Choyke, P.L.; Kobayashi, H. Cancer cell-selective in vivo near infrared photoimmunotherapy targeting specific membrane molecules. Nat. Med. 2011, 17, 1685–1691. [Google Scholar] [CrossRef]
- Kobayashi, H.; Furusawa, A.; Rosenberg, A.; Choyke, P.L. Near-infrared photoimmunotherapy of cancer: A new approach that kills cancer cells and enhances anti-cancer host immunity. Int. Immunol. 2021, 33, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Larson, S.M.; Carrasquillo, J.A.; Cheung, N.K.; Press, O.W. Radioimmunotherapy of human tumours. Nat. Rev. Cancer 2015, 15, 347–360. [Google Scholar] [CrossRef]
- Gudkov, S.V.; Shilyagina, N.Y.; Vodeneev, V.A.; Zvyagin, A.V. Targeted Radionuclide Therapy of Human Tumors. Int. J. Mol. Sci. 2015, 17, 33. [Google Scholar] [CrossRef]
- Sudo, H.; Tsuji, A.B.; Sugyo, A.; Saga, T.; Kaneko, M.K.; Kato, Y.; Higashi, T. Therapeutic efficacy evaluation of radioimmunotherapy with (90) Y-labeled anti-podoplanin antibody NZ-12 for mesothelioma. Cancer Sci. 2019, 110, 1653–1664. [Google Scholar] [CrossRef]
- Miederer, M.; Scheinberg, D.A.; McDevitt, M.R. Realizing the potential of the Actinium-225 radionuclide generator in targeted alpha particle therapy applications. Adv. Drug Deliv. Rev. 2008, 60, 1371–1382. [Google Scholar] [CrossRef] [PubMed]
- Tafreshi, N.K.; Doligalski, M.L.; Tichacek, C.J.; Pandya, D.N.; Budzevich, M.M.; El-Haddad, G.; Khushalani, N.I.; Moros, E.G.; McLaughlin, M.L.; Wadas, T.J.; et al. Development of Targeted Alpha Particle Therapy for Solid Tumors. Molecules 2019, 24, 4314. [Google Scholar] [CrossRef] [PubMed]
- Sudo, H.; Tsuji, A.B.; Sugyo, A.; Kaneko, M.K.; Kato, Y.; Nagatsu, K.; Suzuki, H.; Higashi, T. Preclinical Evaluation of Podoplanin-Targeted Alpha-Radioimmunotherapy with the Novel Antibody NZ-16 for Malignant Mesothelioma. Cells 2021, 10, 2503. [Google Scholar] [CrossRef] [PubMed]
- Kato, Y.; Kaneko, M.K. A cancer-specific monoclonal antibody recognizes the aberrantly glycosylated podoplanin. Sci. Rep. 2014, 4, 5924. [Google Scholar] [CrossRef]
- Miyazaki, A.; Nakai, H.; Sonoda, T.; Hirohashi, Y.; Kaneko, M.K.; Kato, Y.; Sawa, Y.; Hiratsuka, H. LpMab-23-recognizing cancer-type podoplanin is a novel predictor for a poor prognosis of early stage tongue cancer. Oncotarget 2018, 9, 21156–21165. [Google Scholar] [CrossRef][Green Version]
- Kaneko, M.K.; Ohishi, T.; Kawada, M.; Kato, Y. A cancer-specific anti-podocalyxin monoclonal antibody (60-mG(2a)-f) exerts antitumor effects in mouse xenograft models of pancreatic carcinoma. Biochem. Biophys. Rep. 2020, 24, 100826. [Google Scholar] [CrossRef]
- Kaneko, M.K.; Nakamura, T.; Kunita, A.; Fukayama, M.; Abe, S.; Nishioka, Y.; Yamada, S.; Yanaka, M.; Saidoh, N.; Yoshida, K.; et al. ChLpMab-23: Cancer-Specific Human-Mouse Chimeric Anti-Podoplanin Antibody Exhibits Antitumor Activity via Antibody-Dependent Cellular Cytotoxicity. Monoclon. Antib. Immunodiagn. Immunother. 2017, 36, 104–112. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, M.K.; Yamada, S.; Nakamura, T.; Abe, S.; Nishioka, Y.; Kunita, A.; Fukayama, M.; Fujii, Y.; Ogasawara, S.; Kato, Y. Antitumor activity of chLpMab-2, a human-mouse chimeric cancer-specific antihuman podoplanin antibody, via antibody-dependent cellular cytotoxicity. Cancer Med. 2017, 6, 768–777. [Google Scholar] [CrossRef] [PubMed]
- Ward, P.L.; Koeppen, H.; Hurteau, T.; Schreiber, H. Tumor antigens defined by cloned immunological probes are highly polymorphic and are not detected on autologous normal cells. J. Exp. Med. 1989, 170, 217–232. [Google Scholar] [CrossRef] [PubMed]
- Schietinger, A.; Philip, M.; Yoshida, B.A.; Azadi, P.; Liu, H.; Meredith, S.C.; Schreiber, H. A mutant chaperone converts a wild-type protein into a tumor-specific antigen. Science 2006, 314, 304–308. [Google Scholar] [CrossRef]
- Brooks, C.L.; Schietinger, A.; Borisova, S.N.; Kufer, P.; Okon, M.; Hirama, T.; Mackenzie, C.R.; Wang, L.X.; Schreiber, H.; Evans, S.V. Antibody recognition of a unique tumor-specific glycopeptide antigen. Proc. Natl. Acad. Sci. USA 2010, 107, 10056–10061. [Google Scholar] [CrossRef]
- June, C.H.; O’Connor, R.S.; Kawalekar, O.U.; Ghassemi, S.; Milone, M.C. CAR T cell immunotherapy for human cancer. Science 2018, 359, 1361–1365. [Google Scholar] [CrossRef]
- Waseda, M.; Kaneko, S. Podoplanin as an Attractive Target of CAR T Cell Therapy. Cells 2020, 9, 1971. [Google Scholar] [CrossRef]
- Shiina, S.; Ohno, M.; Ohka, F.; Kuramitsu, S.; Yamamichi, A.; Kato, A.; Motomura, K.; Tanahashi, K.; Yamamoto, T.; Watanabe, R.; et al. CAR T Cells Targeting Podoplanin Reduce Orthotopic Glioblastomas in Mouse Brains. Cancer Immunol. Res. 2016, 4, 259–268. [Google Scholar] [CrossRef]
- He, Y.; Schreiber, K.; Wolf, S.P.; Wen, F.; Steentoft, C.; Zerweck, J.; Steiner, M.; Sharma, P.; Shepard, H.M.; Posey, A.; et al. Multiple cancer-specific antigens are targeted by a chimeric antigen receptor on a single cancer cell. JCI Insight 2019, 4, e130416. [Google Scholar] [CrossRef]
- Sharma, P.; Marada, V.; Cai, Q.; Kizerwetter, M.; He, Y.; Wolf, S.P.; Schreiber, K.; Clausen, H.; Schreiber, H.; Kranz, D.M. Structure-guided engineering of the affinity and specificity of CARs against Tn-glycopeptides. Proc. Natl. Acad. Sci. USA 2020, 117, 15148–15159. [Google Scholar] [CrossRef]
| Organ | Tumor Type | PDPN Expression | Functional and/or Clinical Significance | Detection (mAb) | Ref |
|---|---|---|---|---|---|
| Esophageal | SCC 1 | Tumor | PDPN membrane expression is correlated with vimentin cytoplasmic expression. | IHC 12 (D2-40) | [81] |
| SCC | Tumor | PDPN knockdown suppresses tumor formation in mice and enhances chemosensitivity. | IHC (D2-40) | [82] | |
| SCC | Tumor | PDPN is involved in collective cell invasion in the absence of EMT 7. | IHC (D2-40) | [83] | |
| Oral | SCC | Tumor | PDPN expression correlates with cervical lymph node metastases and clinical outcome. | IHC (D2-40) | [84] |
| SCC | Tumor | High PDPN expression in the biopsy specimen predicts poor response to neoadjuvant radiochemotherapy with carboplatin. | IHC (D2-40) | [85] | |
| Skin | SCC | Tumor | High PDPN expression in the primary tumor predicts poor clinical outcomes. | IHC (D2-40) | [86] |
| Head & neck | SCC | Tumor | PDPN knockdown suppresses tumor migration and invasion. | IHC (NR 14) | [87] |
| Kidney | ccRCC 2 | Tumor | High PDPN expression was an independent adverse prognostic factor for patient survival. | IHC (18H5) | [88] |
| Brain | GBM 3 | Tumor | PDPN is expressed in the mesenchymal type of GBM, which presents the worst prognosis. | IHC (NZ-1.2) | [89] |
| Breast | AC 4 | CAF 6 | Tumors with a negative ER 8 status yielded the highest number of PDPN-expressing CAFs. | IHC (D2-40) | [58] |
| Lung | SCC | CAF | PDPN-positive CAFs express high TGF-β and are associated with the immunosuppressive TME 9. | TCGA13microarray | [57] |
| AC | CAF | PDPN-positive vascular adventitial fibroblasts enhance tumor formation in mice. | IHC (D2-40) | [55] | |
| AC | CAF | PDPN-positive CAFs cases display high CD204 TAMs 10 and low CD8/FOXP3 T cells, associated with the immunosuppressive TME. | TCGA microarray | [61] | |
| AC | CAF | PDPN-positive CAFs promote tumor cell resistance to EGFR TKIs 11. | IHC (D2-40) | [60] | |
| Pancreas | AC | CAF | PDPN-positive CAFs enhance the invasion of cancer cells more effectively than PDPN-negative CAFs. | IHC (D2-40) | [59] |
| Esophageal | AC | CAF | PDPN-expressing CAFs were observed in invasive AC, but not in precursor lesions. | IHC (D2-40) | [90] |
| Bile duct | CCA 5 | CAF | Association between lymphatic vessel density and PDPN expression in CAFs. PDPN promotes the migratory ability of CAFs. | IHC (sc-134482) | [91] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suzuki, H.; Kaneko, M.K.; Kato, Y. Roles of Podoplanin in Malignant Progression of Tumor. Cells 2022, 11, 575. https://doi.org/10.3390/cells11030575
Suzuki H, Kaneko MK, Kato Y. Roles of Podoplanin in Malignant Progression of Tumor. Cells. 2022; 11(3):575. https://doi.org/10.3390/cells11030575
Chicago/Turabian StyleSuzuki, Hiroyuki, Mika K. Kaneko, and Yukinari Kato. 2022. "Roles of Podoplanin in Malignant Progression of Tumor" Cells 11, no. 3: 575. https://doi.org/10.3390/cells11030575
APA StyleSuzuki, H., Kaneko, M. K., & Kato, Y. (2022). Roles of Podoplanin in Malignant Progression of Tumor. Cells, 11(3), 575. https://doi.org/10.3390/cells11030575







