Validating Discriminative Signatures for Obstructive Sleep Apnea in Exhaled Breath
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Participants and Clinical Data
2.2. Direct Breath Analysis
2.3. Data Preprocessing
2.4. Satistical Analysis and Classification
3. Results
3.1. Study Participants
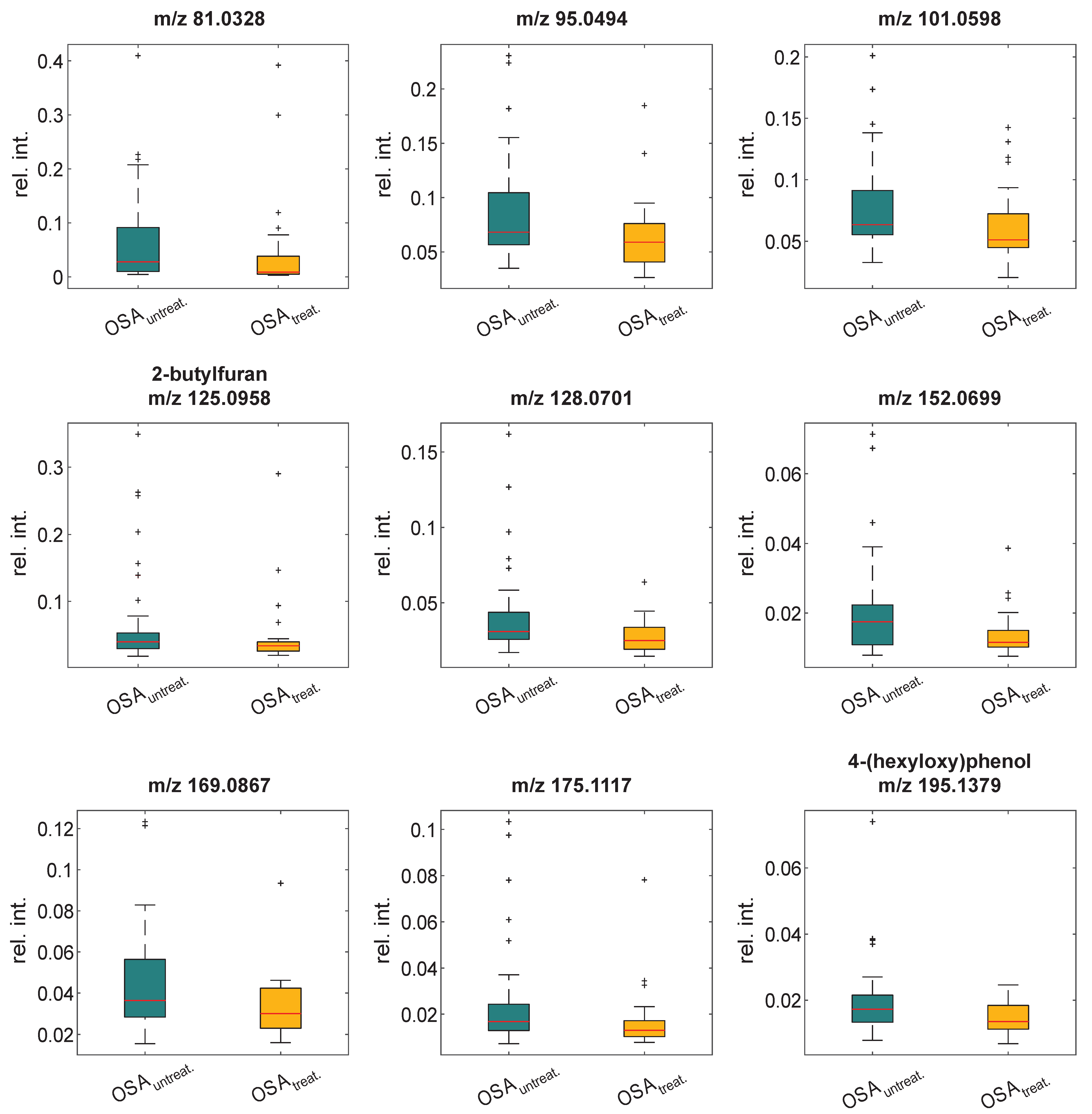
3.2. Validation of Breath Signatures
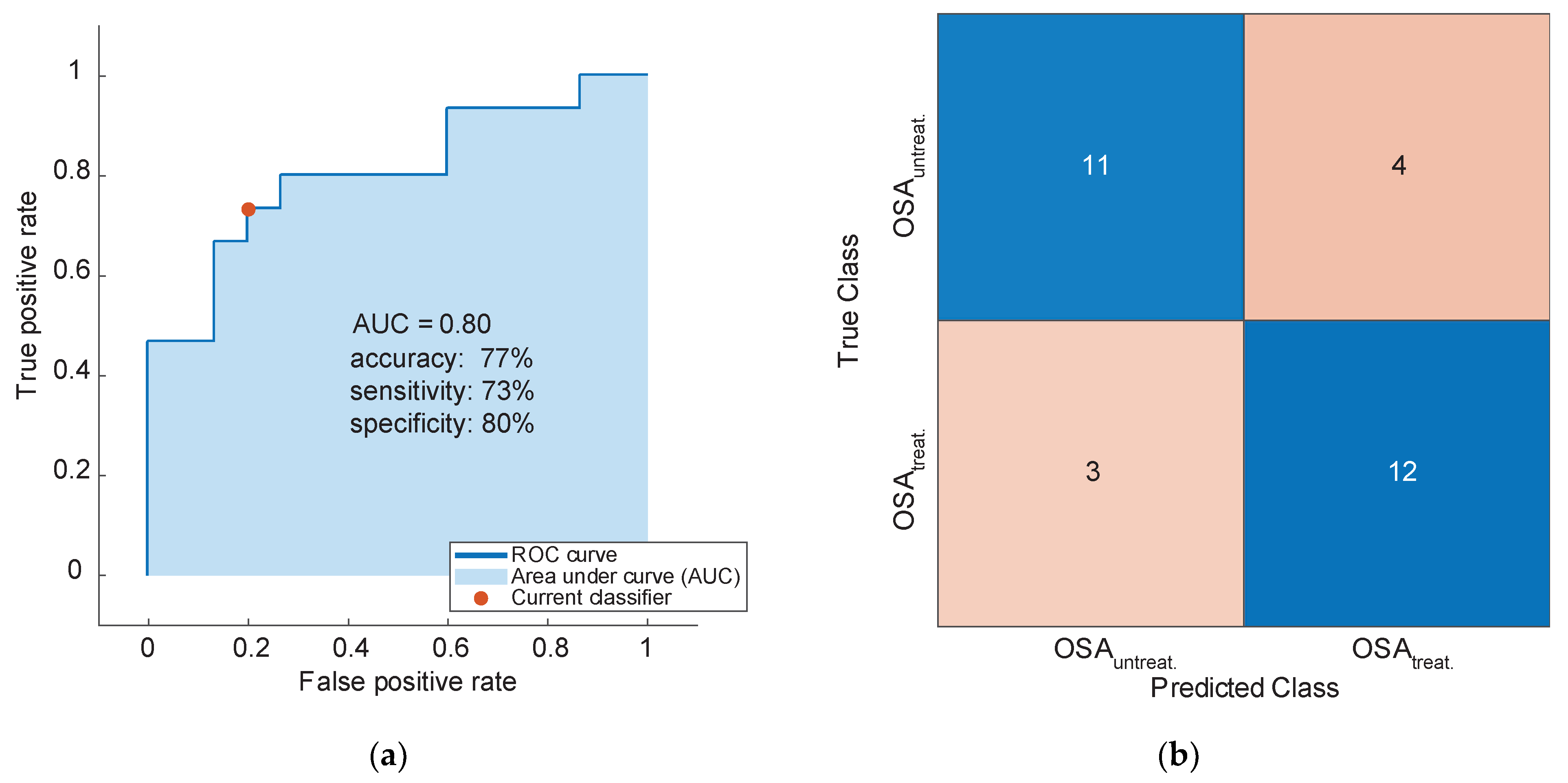
3.3. Classification Based on Breath Signatures
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Senaratna, C.V.; Perret, J.L.; Lodge, C.J.; Lowe, A.J.; Campbell, B.E.; Matheson, M.C.; Hamilton, G.S.; Dharmage, S.C. Prevalence of Obstructive Sleep Apnea in the General Population: A Systematic Review. Sleep Med. Rev. 2017, 34, 70–81. [Google Scholar] [CrossRef] [PubMed]
- Lévy, P.; Kohler, M.; McNicholas, W.T.; Barbé, F.; McEvoy, R.D.; Somers, V.K.; Lavie, L.; Pépin, J.-L. Obstructive Sleep Apnoea Syndrome. Nat. Rev. Dis. Prim. 2015, 1, 15015. [Google Scholar] [CrossRef] [PubMed]
- George, C.F.P. Sleep Apnea, Alertness, and Motor Vehicle Crashes. Am. J. Respir. Crit. Care Med. 2007, 176, 954–956. [Google Scholar] [CrossRef] [PubMed]
- Reichmuth, K.J.; Austin, D.; Skatrud, J.B.; Young, T. Association of Sleep Apnea and Type II Diabetes: A population-based study. Am. J. Respir. Crit. Care Med. 2005, 172, 1590–1595. [Google Scholar] [CrossRef] [PubMed]
- Peppard, P.E.; Young, T.; Palta, M.; Skatrud, J. Prospective Study of the Association between Sleep-Disordered Breathing and Hypertension. N. Engl. J. Med. 2000, 342, 1378–1384. [Google Scholar] [CrossRef] [PubMed]
- Gonzaga, C.; Bertolami, A.; Bertolami, M.; Amodeo, C.; Calhoun, D. Obstructive Sleep Apnea, Hypertension and Cardiovascular Diseases. J. Hum. Hypertens. 2015, 29, 705–712. [Google Scholar] [CrossRef]
- Gay, P.; Weaver, T.; Loube, D.; Iber, C. Evaluation of Positive Airway Pressure Treatment for Sleep Related Breathing Disorders in Adults. Sleep 2006, 29, 381–401. [Google Scholar] [CrossRef]
- Rolfe, I.; Olson, L.G.; Saunders, N.A. Long-Term Acceptance of Continuous Positive Airway Pressure in Obstructive Sleep Apnea. Am. Rev. Respir. Dis. 1991, 144, 1130–1133. [Google Scholar] [CrossRef]
- Kohler, M.; Smith, D.; Tippett, V.; Stradling, J.R. Predictors of Long-Term Compliance with Continuous Positive Airway Pressure. Thorax 2010, 65, 829–832. [Google Scholar] [CrossRef]
- Park, J.G.; Ramar, K.; Olson, E.J. Updates on Definition, Consequences, and Management of Obstructive Sleep Apnea. Mayo Clin. Proc. 2011, 86, 549–555. [Google Scholar] [CrossRef]
- Netzer, N.C.; Stoohs, R.A.; Netzer, C.M.; Clark, K.; Strohl, K.P. Using the Berlin Questionnaire to Identify Patients at Risk for the Sleep Apnea Syndrome. Ann. Intern. Med. 1999, 131, 485–491. [Google Scholar] [CrossRef] [PubMed]
- Nagappa, M.; Liao, P.; Wong, J.; Auckley, D.; Ramachandran, S.K.; Memtsoudis, S.; Mokhlesi, B.; Chung, F. Validation of the Stop-Bang Questionnaire as a Screening Tool for Obstructive Sleep Apnea among Different Populations: A Systematic Review and Meta-Analysis. PLoS ONE 2015, 10, e0143697. [Google Scholar] [CrossRef] [PubMed]
- Marti-Soler, H.; Hirotsu, C.; Marques-Vidal, P.; Vollenweider, P.; Waeber, G.; Preisig, M.; Tafti, M.; Tufik, S.B.; Bittencourt, L.; Tufik, S.; et al. The NoSAS Score for Screening of Sleep-Disordered Breathing: A Derivation and Validation Study. Lancet Respir. Med. 2016, 4, 742–748. [Google Scholar] [CrossRef]
- Johns, M.W. A New Method for Measuring Daytime Sleepiness: The Epworth Sleepiness Scale. Sleep 1991, 14, 540–545. [Google Scholar] [CrossRef] [PubMed]
- Grewe, F.A.; Roeder, M.; Bradicich, M.; Schwarz, E.I.; Held, U.; Thiel, S.; Gaisl, T.; Sievi, N.A.; Kohler, M. Low Repeatability of Epworth Sleepiness Scale after Short Intervals in a Sleep Clinic Population. J. Clin. Sleep Med. 2020, 16, 757–764. [Google Scholar] [CrossRef] [PubMed]
- Tan, A.; Hong, Y.; Tan, L.W.L.; van Dam, R.M.; Cheung, Y.Y.; Lee, C.-H. Validation of NoSAS Score for Screening of Sleep-Disordered Breathing in a Multiethnic Asian Population. Sleep Breath. 2017, 21, 1033–1038. [Google Scholar] [CrossRef] [PubMed]
- Coutinho Costa, J.; Rebelo-Marques, A.; Machado, J.N.; Gama, J.M.R.; Santos, C.; Teixeira, F.; Moita, J. Validation of NoSAS (Neck, Obesity, Snoring, Age, Sex) Score as a Screening Tool for Obstructive Sleep Apnea: Analysis in a Sleep Clinic. Pulmonology 2019, 25, 263–270. [Google Scholar] [CrossRef]
- Duarte, R.L.M.; Magalhães-Da-silveira, F.J.; Oliveira-E-sá, T.S.; Silva, J.A.; Mello, F.C.Q.; Gozal, D. Obstructive Sleep Apnea Screening with a 4-Item Instrument, Named GOAL Questionnaire: Development, Validation and Comparative Study with No-Apnea, STOP-Bang, and NoSAS. Nat. Sci. Sleep 2020, 12, 57–67. [Google Scholar] [CrossRef]
- Duarte, R.L.M.; Rabahi, M.F.; Magalhães-da-Silveira, F.J.; de Oliveira-e-Sá, T.S.; Mello, F.C.Q.; Gozal, D. Simplifying the Screening of Obstructive Sleep Apnea With a 2-Item Model, No-Apnea: A Cross-Sectional Study. J. Clin. Sleep Med. 2018, 14, 1097–1107. [Google Scholar] [CrossRef] [PubMed]
- Rosen, I.M.; Kirsch, D.B.; Chervin, R.D.; Carden, K.A.; Ramar, K.; Aurora, R.N.; Kristo, D.A.; Malhotra, R.K.; Martin, J.L.; Olson, E.J.; et al. Clinical Use of a Home Sleep Apnea Test: An American Academy of Sleep Medicine Position Statement. J. Clin. Sleep Med. 2017, 13, 1205–1207. [Google Scholar] [CrossRef]
- Martinez-Garcia, M.A.; Campos-Rodriguez, F.; Barbé, F.; Gozal, D.; Agustí, A. Precision Medicine in Obstructive Sleep Apnoea. Lancet Respir. Med. 2019, 7, 456–464. [Google Scholar] [CrossRef]
- Bruderer, T.; Gaisl, T.; Gaugg, M.T.; Nowak, N.; Streckenbach, B.; Müller, S.; Moeller, A.; Kohler, M.; Zenobi, R. On-Line Analysis of Exhaled Breath. Chem. Rev. 2019, 119, 10803–10828. [Google Scholar] [CrossRef] [PubMed]
- Dweik, R.A.; Boggs, P.B.; Erzurum, S.C.; Irvin, C.G.; Leigh, M.W.; Lundberg, J.O.; Olin, A.-C.; Plummer, A.L.; Taylor, D.R. An Official ATS Clinical Practice Guideline: Interpretation of Exhaled Nitric Oxide Levels (FeNO) for Clinical Applications. Am. J. Respir. Crit. Care Med. 2011, 184, 602–615. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, E.I.; Martinez-Lozano Sinues, P.; Bregy, L.; Gaisl, T.; Garcia Gomez, D.; Gaugg, M.T.; Suter, Y.; Stebler, N.; Nussbaumer-Ochsner, Y.; Bloch, K.E.; et al. Effects of CPAP Therapy Withdrawal on Exhaled Breath Pattern in Obstructive Sleep Apnoea. Thorax 2016, 71, 110–117. [Google Scholar] [CrossRef]
- Nowak, N.; Engler, A.; Thiel, S.; Stöberl, A.S.; Sinues, P.; Zenobi, R.; Kohler, M. Validation of Breath Biomarkers for Obstructive Sleep Apnea. Sleep Med. 2021, 85, 75–86. [Google Scholar] [CrossRef]
- Bloch, K.E.; Schoch, O.D.; Zhang, J.N.; Russi, E.W. German Version of the Epworth Sleepiness Scale. Respiration 1999, 66, 440–447. [Google Scholar] [CrossRef]
- Kessner, D.; Chambers, M.; Burke, R.; Agus, D.; Mallick, P. ProteoWizard: Open Source Software for Rapid Proteomics Tools Development. Bioinformatics 2008, 24, 2534–2536. [Google Scholar] [CrossRef]
- Singh, K.D.; del Miguel, G.V.; Gaugg, M.T.; Ibañez, A.J.; Zenobi, R.; Kohler, M.; Frey, U.; Sinues, P.M.-L. Translating Secondary Electrospray Ionization–High-Resolution Mass Spectrometry to the Clinical Environment. J. Breath Res. 2018, 12, 027113. [Google Scholar] [CrossRef]
- García-Gómez, D.; Bregy, L.; Barrios-Collado, C.; Vidal-de-Miguel, G.; Zenobi, R. Real-Time High-Resolution Tandem Mass Spectrometry Identifies Furan Derivatives in Exhaled Breath. Anal. Chem. 2015, 87, 6919–6924. [Google Scholar] [CrossRef]
- García-Gómez, D.; Martínez-Lozano Sinues, P.; Barrios-Collado, C.; Vidal-de-Miguel, G.; Gaugg, M.; Zenobi, R. Identification of 2-Alkenals, 4-Hydroxy-2-Alkenals, and 4-Hydroxy-2,6-Alkadienals in Exhaled Breath Condensate by UHPLC-HRMS and in Breath by Real-Time HRMS. Anal. Chem. 2015, 87, 3087–3093. [Google Scholar] [CrossRef]
- Garner, C.E.; Smith, S.; de Lacy Costello, B.; White, P.; Spencer, R.; Probert, C.S.J.; Ratcliffe, N.M. Volatile Organic Compounds from Feces and Their Potential for Diagnosis of Gastrointestinal Disease. FASEB J. 2007, 21, 1675–1688. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Indias, I.; Torres, M.; Montserrat, J.M.; Sanchez-Alcoholado, L.; Cardona, F.; Tinahones, F.J.; Gozal, D.; Poroyko, V.A.; Navajas, D.; Queipo-Ortuño, M.I.; et al. Intermittent Hypoxia Alters Gut Microbiota Diversity in a Mouse Model of Sleep Apnoea. Eur. Respir. J. 2015, 45, 1055–1065. [Google Scholar] [CrossRef] [PubMed]
- Belizário, J.E.; Faintuch, J.; Malpartida, M.G. Breath Biopsy and Discovery of Exclusive Volatile Organic Compounds for Diagnosis of Infectious Diseases. Front. Cell. Infect. Microbiol. 2021, 10, 564194. [Google Scholar] [CrossRef] [PubMed]
- Castellanos, M.; Suñer, R.; Fernández-Real, J.M.; Sanchez, J.M. 2,5-Dimethylfuran as a Validated Biomarker of Smoking Status. Nicotine Tob. Res. 2019, 21, 828–834. [Google Scholar] [CrossRef] [PubMed]
- Papaefstathiou, E.; Stylianou, M.; Andreou, C.; Agapiou, A. Breath Analysis of Smokers, Non-Smokers, and e-Cigarette Users. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2020, 1160, 122349. [Google Scholar] [CrossRef]
- Demirci-Çekiç, S.; Özkan, G.; Avan, A.N.; Uzunboy, S.; Çapanoğlu, E.; Apak, R. Biomarkers of Oxidative Stress and Antioxidant Defense. J. Pharm. Biomed. Anal. 2022, 209, 114477. [Google Scholar] [CrossRef]
- Ibrahim, W.; Natarajan, S.; Wilde, M.; Cordell, R.; Monks, P.S.; Greening, N.; Brightling, C.E.; Evans, R.; Siddiqui, S. A Systematic Review of the Diagnostic Accuracy of Volatile Organic Compounds in Airway Diseases and Their Relation to Markers of Type-2 Inflammation. ERJ Open Res. 2021, 7. [Google Scholar] [CrossRef]
- Vabalas, A.; Gowen, E.; Poliakoff, E.; Casson, A.J. Machine Learning Algorithm Validation with a Limited Sample Size. PLoS ONE 2019, 14, e0224365. [Google Scholar] [CrossRef]
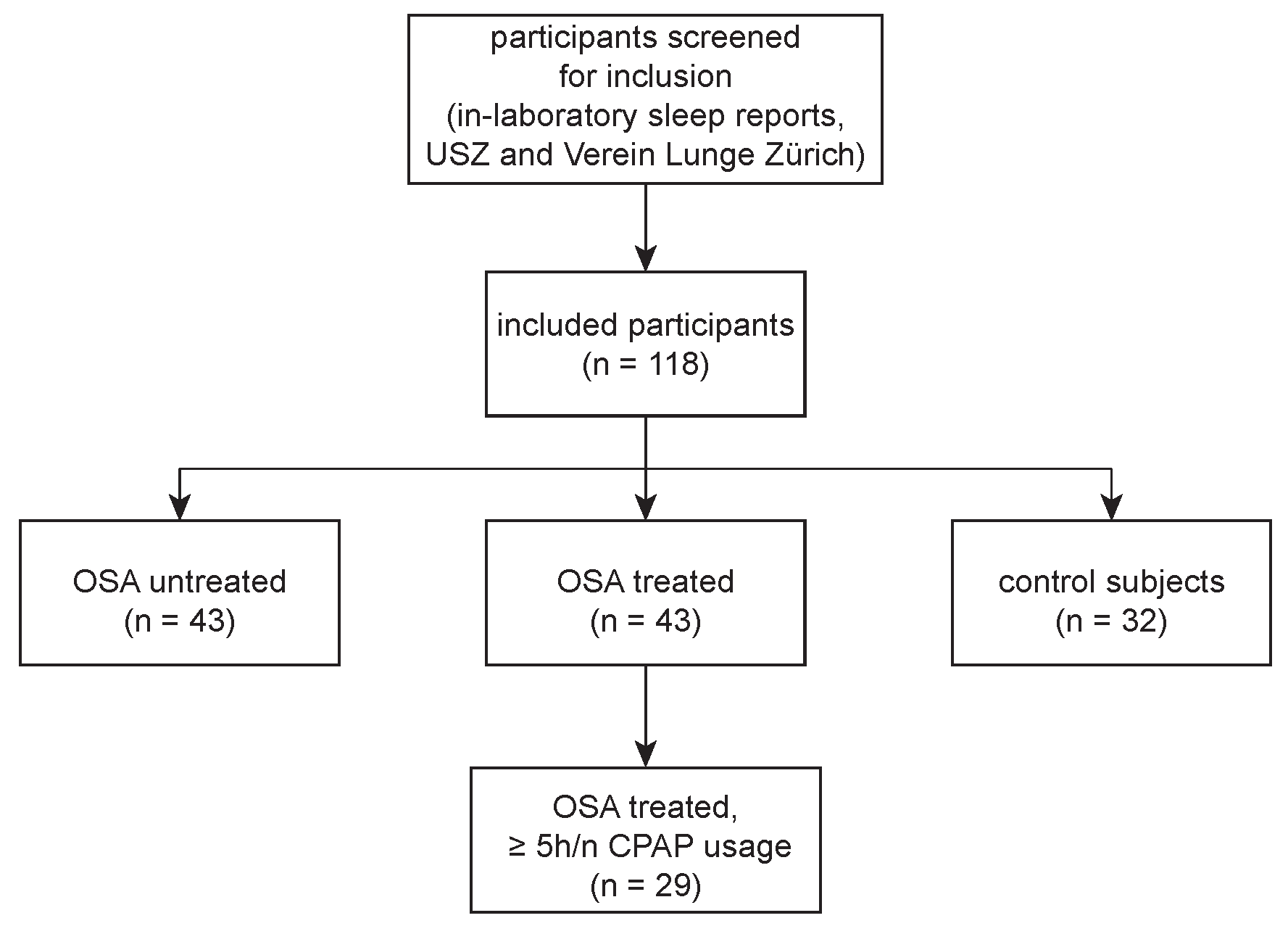
| OSA Untreated | OSA Treated | Control Subjects | p Value 1 | |
|---|---|---|---|---|
| N | 43 | 43 | 32 | - |
| Age (y) | 61 (55, 69) | 61 (53, 69) | 50 (39, 56) | 0.0002 |
| Sex, male, N (%) | 36 (84%) | 36 (84%) | 19 (59%) | 0.020 |
| BMI (kg/m2) | 29.5 (27.0, 34.0) | 30.1 (26.9, 32.3) | 29.7 [27.4, 32.4) | 0.86 |
| Smoker, N (%) | 28 (65%) | 29 (67%) | 18 (56%) | 0.59 |
| Smoking, py | 0.0 (0.0, 25.0) | 1.0 (0.0, 18.0) | 0.0 (0.0, 9.3) | 0.85 |
| AHI at diagnosis (events/h) | 30.2 (24.0, 45.0) | 35.5 (24.0, 44.5) | 3.0 (2.0, 4.0) | 0.0001 |
| ODI at diagnosis (events/h) | 32.0 (23.0, 48.2) | 29.0 (16.0, 46.0) | 4.5 (2.5, 8.5) | 0.0001 |
| AHI at visit (events/h) | 28.0 (18.8, 40.0) | 1.3 (0.8, 3.6) | n.a. | 0.0001 |
| ODI at visit (events/h) | 29.9 (13.9, 41.4) | n.a. | n.a. | - |
| ESS at visit, points | 5.0 (3.0, 9.0) | 6.0 (3.0, 9.0) | 7.5 (6.0, 11.0) | 0.055 |
| Significant Markers | Marker Detection in Previous Study [25] | ||||||
|---|---|---|---|---|---|---|---|
| Stratification 1 a | Stratification 2 b | ||||||
| m/z | Elemental Composition c | Metabolite | Behavior | Sign. | Behavior | Sign. | Behavior |
| 81.0328 | C5H5O | n.a. | increased | no | n.a. | no | n.a. |
| 95.0494 | C6H7O | n.a. | increased | no | n.a. | no | n.a. |
| 101.0598 | C5H9O2 | n.a. | increased | no | n.a. | no | n.a. |
| 125.0958 | C8H13O | 2-butylfuran | increased | yes | increased | yes | increased |
| 128.0701 | C6H10NO2 | n.a. | increased | yes | increased | no | n.a. |
| 152.0699 | C8H10NO2 | n.a. | increased | no | n.a. | no | n.a. |
| 169.0867 | C9H13O3 | n.a. | increased | no | n.a. | no | n.a. |
| 175.1117 | C12H15O | n.a. | increased | yes | increased | yes | increased |
| 195.1379 | C12H19O2 | 4-(hexyloxy)phenol | increased | no | n.a. | yes | increased |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Streckenbach, B.; Osswald, M.; Malesevic, S.; Zenobi, R.; Kohler, M. Validating Discriminative Signatures for Obstructive Sleep Apnea in Exhaled Breath. Cells 2022, 11, 2982. https://doi.org/10.3390/cells11192982
Streckenbach B, Osswald M, Malesevic S, Zenobi R, Kohler M. Validating Discriminative Signatures for Obstructive Sleep Apnea in Exhaled Breath. Cells. 2022; 11(19):2982. https://doi.org/10.3390/cells11192982
Chicago/Turabian StyleStreckenbach, Bettina, Martin Osswald, Stefan Malesevic, Renato Zenobi, and Malcolm Kohler. 2022. "Validating Discriminative Signatures for Obstructive Sleep Apnea in Exhaled Breath" Cells 11, no. 19: 2982. https://doi.org/10.3390/cells11192982
APA StyleStreckenbach, B., Osswald, M., Malesevic, S., Zenobi, R., & Kohler, M. (2022). Validating Discriminative Signatures for Obstructive Sleep Apnea in Exhaled Breath. Cells, 11(19), 2982. https://doi.org/10.3390/cells11192982





