Effects of Air Pollution on Cellular Senescence and Skin Aging
Abstract
1. Introduction
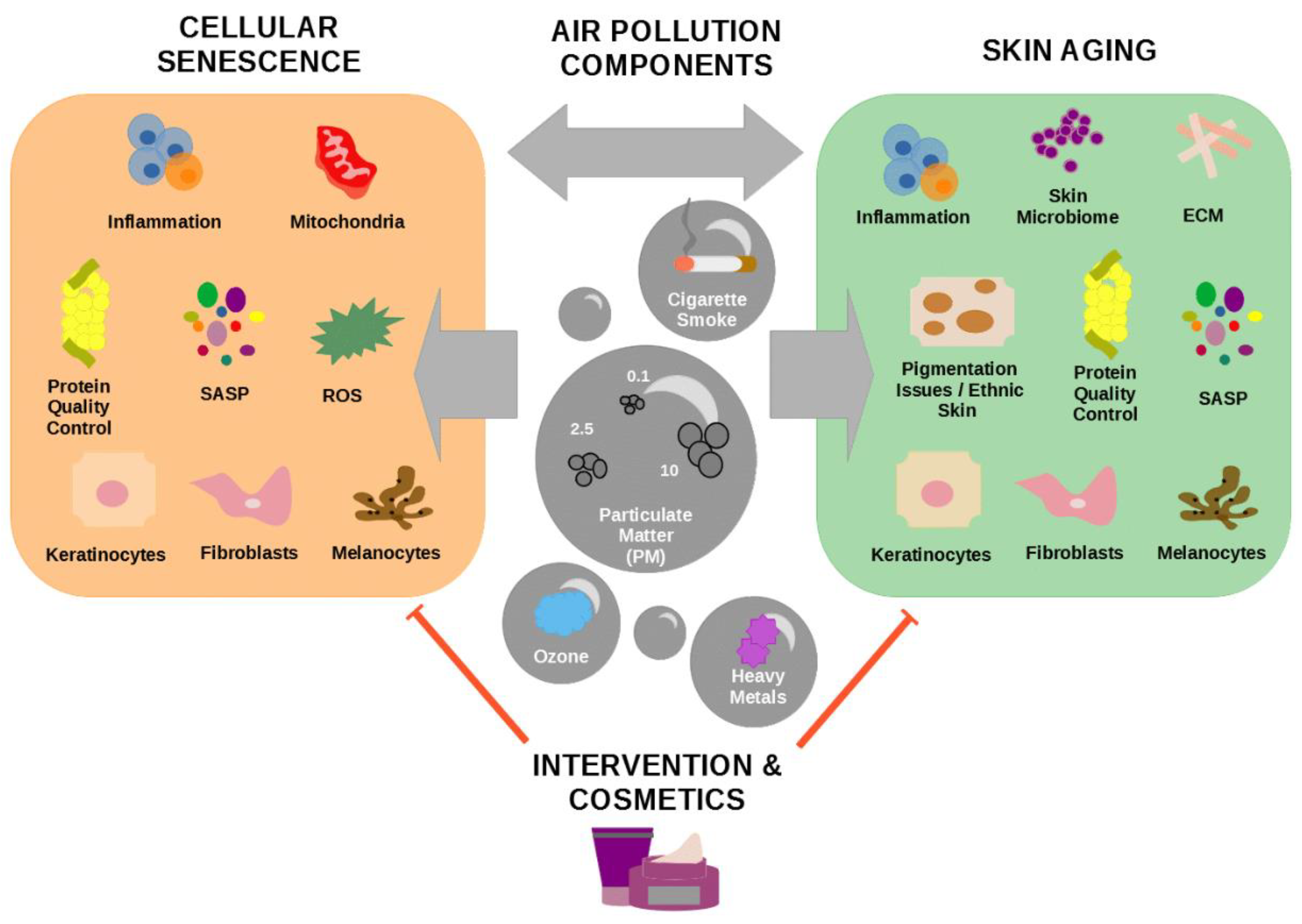
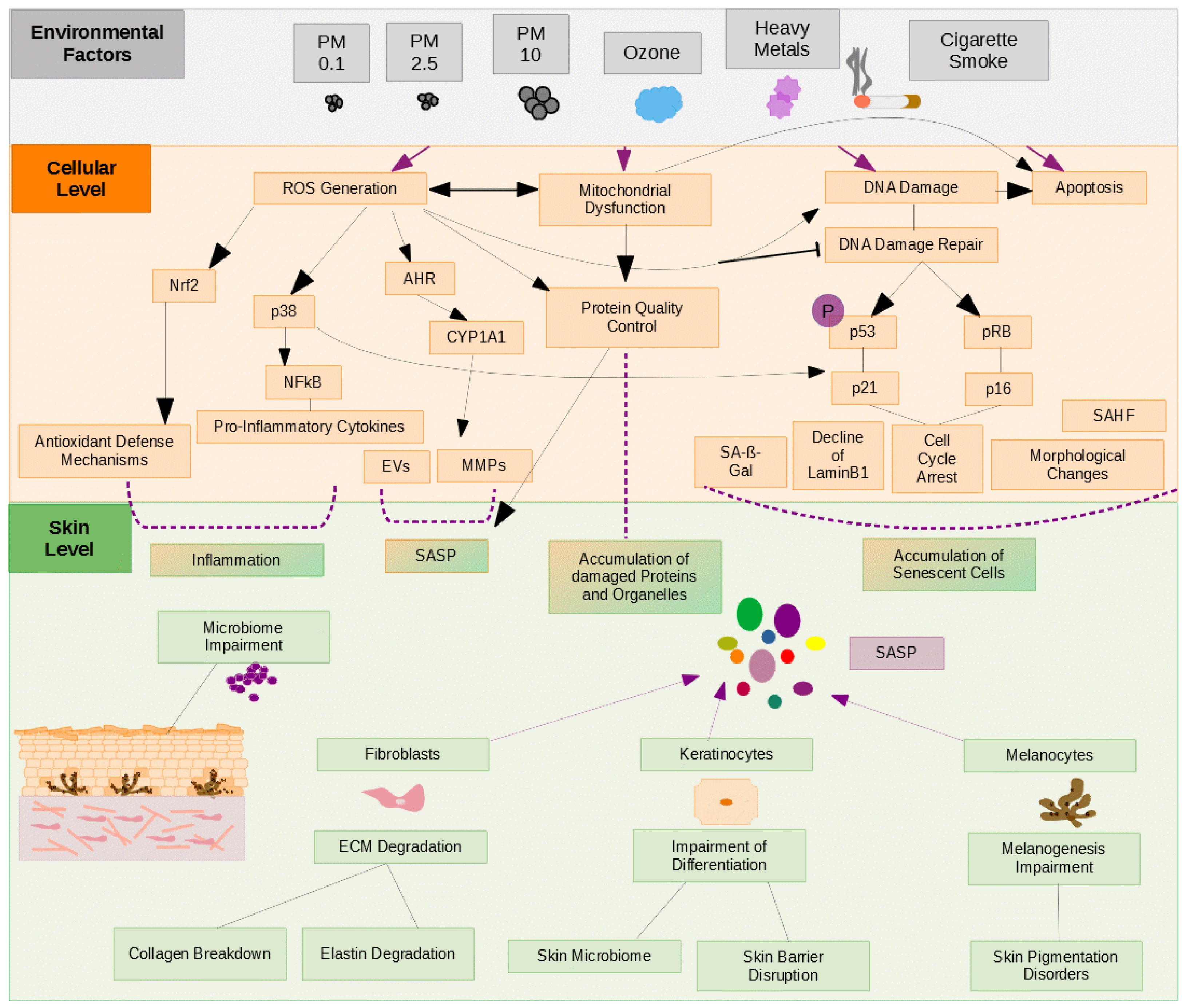
2. Cellular Senescence
3. Cellular Senescence and Skin Aging
3.1. Main Characteristics of Skin Aging
3.2. Inflammation and Skin Aging
3.3. Senescence of Skin Cells and Skin Aging
3.4. Microbioma and Skin Aging
3.5. Skin Aging in Different Ethnicities and Phototypes
4. Major Components of Air Pollution Affecting Skin Appearance
4.1. Particulate Matter
4.2. Cigarette Smoke
4.3. Ozone
4.4. Heavy Metals
5. Therapeutics and Cosmetics
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| AD | atopic dermatitis |
| AhR | aryl hydrocarbon receptor |
| ARE | antioxidant response element |
| BAFFR | B-cell activating factor receptor |
| BDDI | E/Z-2-benzylidene-5,6-dimethoxy-3,3-dimethyl-indan-1-one |
| CD | cluster of differentiation |
| COL1A1 | collagen type 1 alpha chain |
| COX-1 and -2 | cyclooxygenases-1 and −2 |
| CYP1A | cytochrome P450 1A |
| DNA | deoxyribonucleic acid |
| DNMT | DNA methyltransferase |
| Dp | aerodynamic diameter |
| DPE | diesel particulate extract |
| ECM | extracellular matrix |
| EVs | extracellular vesicles |
| HaCaT | immortalized human keratinocytes |
| IL | interleukin |
| LC3 | light chain 3 |
| LTβR | lymphotoxin β receptor |
| MAPK | mitogen-activated protein kinase |
| MMP | matrix metalloproteases |
| NAC | N-acetylcysteine |
| NF-κB | nuclear factor kappa-light-chain-enhancer of activated B cells |
| NOX | nicotinamide adenine dinucleotide phosphate oxidase |
| Nrf2 | nuclear factor erythroid 2-related factor 2 |
| O3 | ozone |
| PAH | polyaromatic hydrocarbons |
| PM | particulate matter |
| pRB | phosphorylated retinoblastoma protein |
| PRRs | pattern-recognition receptors |
| RANK | receptor activator of NF-κB |
| ROS | reactive oxygen species |
| SAHF | senescence-associated heterochromatin foci |
| SA-β-Gal | senescence-associated β-Galactosidase |
| SASP | senescence-associated secretory phenotype |
| tBHP | tert-butyl hydroperoxide |
| TCR | T-cell receptor |
| TET1 | DNA demethylase 1 |
| TGF | transforming growth factor |
| TNF-α | tumor necrosis factor alpha |
| TNFR | tumor necrosis factor receptor |
| UV | ultraviolet |
| WHO | world health organization |
References
- Cavinato, M. Cosmetics and Cosmeceuticals. In Reference Module in Biomedical Sciences; Ratan, S.I., Ed.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 446–461. ISBN 9780128160756. [Google Scholar]
- Vierkötter, A.; Krutmann, J. Environmental Influences on Skin Aging and Ethnic-Specific Manifestations. Dermato-Endocrinology 2012, 4, 227–231. [Google Scholar] [CrossRef] [PubMed]
- Cavinato, M.; Jansen-Dürr, P. Molecular Mechanisms of UVB-Induced Senescence of Dermal Fibroblasts and Its Relevance for Photoaging of the Human Skin. Exp. Gerontol. 2017, 94, 78–82. [Google Scholar] [CrossRef] [PubMed]
- Cavinato, M.; Wedel, S.; Jansen-Dürr, P. Aging of Cells In Vitro. In Reference Module in Biomedical Sciences; Elsevier: Amsterdam, The Netherlands, 2019; pp. 138–148. ISBN 9780128160763. [Google Scholar]
- Kim, K.E.; Cho, D.; Park, H.J. Air Pollution and Skin Diseases: Adverse Effects of Airborne Particulate Matter on Various Skin Diseases. Life Sci. 2016, 152, 126–134. [Google Scholar] [CrossRef] [PubMed]
- WHO. WHO Releases Country Estimates on Air Pollution Exposure and Health Impact (outdoor air pollution) in, Asia and Western Pacific regions. Available online: https://www.who.int/news-room/fact-sheets/detail/ambient- (accessed on 30 April 2022).
- McDaniel, D.; Farris, P.; Valacchi, G. Atmospheric Skin Aging-Contributors and Inhibitors. J. Cosmet. Dermatol. 2018, 17, 124–137. [Google Scholar] [CrossRef]
- Puri, P.; Nandar, S.; Kathuria, S.; Ramesh, V. Effects of Air Pollution on the Skin: A Review. Indian J. Dermatol. Venereol. Leprol. 2017, 83, 415. [Google Scholar] [CrossRef]
- Krutmann, J.; Liu, W.; Li, L.; Pan, X.; Crawford, M.; Sore, G.; Seite, S. Pollution and Skin: From Epidemiological and Mechanistic Studies to Clinical Implications. J. Dermatol. Sci. 2014, 76, 163–168. [Google Scholar] [CrossRef]
- Kampa, M.; Castanas, E. Human Health Effects of Air Pollution. Environ. Pollut. 2008, 151, 362–367. [Google Scholar] [CrossRef]
- Grether-Beck, S.; Felsner, I.; Brenden, H.; Marini, A.; Jaenicke, T.; Aue, N.; Welss, T.; Uthe, I.; Krutmann, J. Air Pollution-induced Tanning of Human Skin*. Br. J. Dermatol. 2021, 185, 1026–1034. [Google Scholar] [CrossRef]
- Shi, Y.; Zeng, Z.; Liu, J.; Pi, Z.; Zou, P.; Deng, Q.; Ma, X.; Qiao, F.; Xiong, W.; Zhou, C.; et al. Particulate Matter Promotes Hyperpigmentation via AhR/MAPK Signaling Activation and by Increasing α-MSH Paracrine Levels in Keratinocytes. Environ. Pollut. 2021, 278, 116850. [Google Scholar] [CrossRef]
- Diao, P.; He, H.; Tang, J.; Xiong, L.; Li, L. Natural Compounds Protect the Skin from Airborne Particulate Matter by Attenuating Oxidative Stress. Biomed. Pharmacother. 2021, 138, 111534. [Google Scholar] [CrossRef]
- Mohamad Kamal, N.S.; Safuan, S.; Shamsuddin, S.; Foroozandeh, P. Aging of the Cells: Insight into Cellular Senescence and Detection Methods. Eur. J. Cell Biol. 2020, 99, 151108. [Google Scholar] [CrossRef] [PubMed]
- Bellei, B.; Picardo, M. Premature Cell Senescence in Human Skin: Dual Face in Chronic Acquired Pigmentary Disorders. Ageing Res. Rev. 2020, 57, 100981. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.S.; Dreesen, O. Biomarkers of Cellular Senescence and Skin Aging. Front. Genet. 2018, 9, 247. [Google Scholar] [CrossRef] [PubMed]
- Martic, I.; Wedel, S.; Jansen-Dürr, P.; Cavinato, M. A New Model to Investigate UVB-Induced Cellular Senescence and Pigmentation in Melanocytes. Mech. Ageing Dev. 2020, 190, 111322. [Google Scholar] [CrossRef]
- Ho, C.Y.; Dreesen, O. Faces of Cellular Senescence in Skin Aging. Mech. Ageing Dev. 2021, 198, 111525. [Google Scholar] [CrossRef]
- Van Deursen, J.M. The Role of Senescent Cells in Ageing. Nature 2014, 509, 439–446. [Google Scholar] [CrossRef]
- Vasileiou, P.; Evangelou, K.; Vlasis, K.; Fildisis, G.; Panayiotidis, M.; Chronopoulos, E.; Passias, P.-G.; Kouloukoussa, M.; Gorgoulis, V.; Havaki, S. Mitochondrial Homeostasis and Cellular Senescence. Cells 2019, 8, 686. [Google Scholar] [CrossRef]
- Eckhart, L.; Tschachler, E.; Gruber, F. Autophagic Control of Skin Aging. Front. Cell Dev. Biol. 2019, 7, 143. [Google Scholar] [CrossRef]
- Lee, Y.I.; Choi, S.; Roh, W.S.; Lee, J.H.; Kim, T.-G. Cellular Senescence and Inflammaging in the Skin Microenvironment. Int. J. Mol. Sci. 2021, 22, 3849. [Google Scholar] [CrossRef]
- Davalli, P.; Mitic, T.; Caporali, A.; Lauriola, A.; D’Arca, D. ROS, Cell Senescence, and Novel Molecular Mechanisms in Aging and Age-Related Diseases. Oxidative Med. Cell. Longev. 2016, 2016, 3565127. [Google Scholar] [CrossRef]
- Da Silva, P.F.L.; Schumacher, B. Principles of the Molecular and Cellular Mechanisms of Aging. J. Investig. Dermatol. 2021, 141, 951–960. [Google Scholar] [CrossRef] [PubMed]
- Toussaint, O.; Medrano, E.; von Zglinicki, T. Cellular and Molecular Mechanisms of Stress-Induced Premature Senescence (SIPS) of Human Diploid Fibroblasts and Melanocytes. Exp. Gerontol. 2000, 35, 927–945. [Google Scholar] [CrossRef]
- Birch, J.; Gil, J. Senescence and the SASP: Many Therapeutic Avenues. Genes Dev. 2020, 34, 1565–1576. [Google Scholar] [CrossRef] [PubMed]
- Ritschka, B.; Storer, M.; Mas, A.; Heinzmann, F.; Ortells, M.C.; Morton, J.P.; Sansom, O.J.; Zender, L.; Keyes, W.M. The Senescence-Associated Secretory Phenotype Induces Cellular Plasticity and Tissue Regeneration. Genes Dev. 2017, 31, 172–183. [Google Scholar] [CrossRef] [PubMed]
- Cuollo, L.; Antonangeli, F.; Santoni, A.; Soriani, A. The Senescence-Associated Secretory Phenotype (SASP) in the Challenging Future of Cancer Therapy and Age-Related Diseases. Biology 2020, 9, 485. [Google Scholar] [CrossRef] [PubMed]
- Dumont, P.; Balbeur, L.; Remacle, J.; Toussaint, O. Appearance of Biomarkers of in Vitro Ageing after Successive Stimulation of WI-38 Fibroblasts with IL-1alpha and TNF-Alpha: Senescence Associated Beta-Galactosidase Activity and Morphotype Transition. J. Anat. 2000, 197, 529–537. [Google Scholar] [CrossRef]
- Roy, A.; Sil, P.C. Tertiary Butyl Hydroperoxide Induced Oxidative Damage in Mice Erythrocytes: Protection by Taurine. Pathophysiology 2012, 19, 137–148. [Google Scholar] [CrossRef]
- Cavinato, M.; Koziel, R.; Romani, N.; Weinmüllner, R.; Jenewein, B.; Hermann, M.; Dubrac, S.; Ratzinger, G.; Grillari, J.; Schmuth, M.; et al. UVB-Induced Senescence of Human Dermal Fibroblasts Involves Impairment of Proteasome and Enhanced Autophagic Activity. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2016, 72, glw150. [Google Scholar] [CrossRef][Green Version]
- Tang, L.; Wu, W.; Fu, W.; Hu, Y. The Effects of Phototherapy and Melanocytes on Keratinocytes. Exp. Ther. Med. 2018, 15, 3459–3466. [Google Scholar] [CrossRef]
- Von Zglinicki, T.; Wan, T.; Miwa, S. Senescence in Post-Mitotic Cells: A Driver of Aging? Antioxid. Redox Signal. 2021, 34, 308–323. [Google Scholar] [CrossRef]
- Wu, S.; Zhou, F.; Zhang, Z.; Xing, D. Mitochondrial Oxidative Stress Causes Mitochondrial Fragmentation via Differential Modulation of Mitochondrial Fission-Fusion Proteins. FEBS J. 2011, 278, 941–954. [Google Scholar] [CrossRef] [PubMed]
- Jung, T.; Höhn, A.; Grune, T. The Proteasome and the Degradation of Oxidized Proteins: Part III—Redox Regulation of the Proteasomal System. Redox Biol. 2014, 2, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Löw, P. The Role of Ubiquitin–Proteasome System in Ageing. Gen. Comp. Endocrinol. 2011, 172, 39–43. [Google Scholar] [CrossRef] [PubMed]
- Bulteau, A.-L.; Moreau, M.; Nizard, C.; Friguet, B. Impairment of Proteasome Function upon UVA- and UVB-Irradiation of Human Keratinocytes. Free Radic. Biol. Med. 2002, 32, 1157–1170. [Google Scholar] [CrossRef]
- Aman, Y.; Schmauck-Medina, T.; Hansen, M.; Morimoto, R.I.; Simon, A.K.; Bjedov, I.; Palikaras, K.; Simonsen, A.; Johansen, T.; Tavernarakis, N.; et al. Autophagy in Healthy Aging and Disease. Nat. Aging 2021, 1, 634–650. [Google Scholar] [CrossRef]
- Jeong, D.; Qomaladewi, N.P.; Lee, J.; Park, S.H.; Cho, J.Y. The Role of Autophagy in Skin Fibroblasts, Keratinocytes, Melanocytes, and Epidermal Stem Cells. J. Investig. Dermatol. 2020, 140, 1691–1697. [Google Scholar] [CrossRef]
- Tashiro, K.; Shishido, M.; Fujimoto, K.; Hirota, Y.; Yo, K.; Gomi, T.; Tanaka, Y. Age-Related Disruption of Autophagy in Dermal Fibroblasts Modulates Extracellular Matrix Components. Biochem. Biophys. Res. Commun. 2014, 443, 167–172. [Google Scholar] [CrossRef]
- Zhang, C.-F.; Gruber, F.; Ni, C.; Mildner, M.; Koenig, U.; Karner, S.; Barresi, C.; Rossiter, H.; Narzt, M.-S.; Nagelreiter, I.M.; et al. Suppression of Autophagy Dysregulates the Antioxidant Response and Causes Premature Senescence of Melanocytes. J. Investig. Dermatol. 2015, 135, 1348–1357. [Google Scholar] [CrossRef]
- Kim, J.Y.; Kim, J.; Ahn, Y.; Lee, E.J.; Hwang, S.; Almurayshid, A.; Park, K.; Chung, H.; Kim, H.J.; Lee, S.; et al. Autophagy Induction Can Regulate Skin Pigmentation by Causing Melanosome Degradation in Keratinocytes and Melanocytes. Pigment Cell Melanoma Res. 2020, 33, 403–415. [Google Scholar] [CrossRef]
- Pfisterer, K.; Shaw, L.E.; Symmank, D.; Weninger, W. The Extracellular Matrix in Skin Inflammation and Infection. Front. Cell Dev. Biol. 2021, 9, 1578. [Google Scholar] [CrossRef]
- Stefanovic, N.; Irvine, A.D.; Flohr, C. The Role of the Environment and Exposome in Atopic Dermatitis. Curr. Treat. Options Allergy 2021, 8, 222–241. [Google Scholar] [CrossRef] [PubMed]
- Celebi Sozener, Z.; Ozdel Ozturk, B.; Cerci, P.; Turk, M.; Gorgulu Akin, B.; Akdis, M.; Altiner, S.; Ozbey, U.; Ogulur, I.; Mitamura, Y.; et al. Epithelial Barrier Hypothesis: Effect of the External Exposome on the Microbiome and Epithelial Barriers in Allergic Disease. Allergy 2022, 77, 1418–1449. [Google Scholar] [CrossRef]
- Fernández-Gallego, N.; Sánchez-Madrid, F.; Cibrian, D. Role of AHR Ligands in Skin Homeostasis and Cutaneous Inflammation. Cells 2021, 10, 3176. [Google Scholar] [CrossRef] [PubMed]
- Kyoreva, M.; Li, Y.; Hoosenally, M.; Hardman-Smart, J.; Morrison, K.; Tosi, I.; Tolaini, M.; Barinaga, G.; Stockinger, B.; Mrowietz, U.; et al. CYP1A1 Enzymatic Activity Influences Skin Inflammation Via Regulation of the AHR Pathway. J. Investig. Dermatol. 2021, 141, 1553–1563.e3. [Google Scholar] [CrossRef] [PubMed]
- Esser, C.; Bargen, I.; Weighardt, H.; Haarmann-Stemmann, T.; Krutmann, J. Functions of the Aryl Hydrocarbon Receptor in the Skin. Semin. Immunopathol. 2013, 35, 677–691. [Google Scholar] [CrossRef]
- Haas, K.; Weighardt, H.; Deenen, R.; Köhrer, K.; Clausen, B.; Zahner, S.; Boukamp, P.; Bloch, W.; Krutmann, J.; Esser, C. Aryl Hydrocarbon Receptor in Keratinocytes Is Essential for Murine Skin Barrier Integrity. J. Investig. Dermatol. 2016, 136, 2260–2269. [Google Scholar] [CrossRef]
- Di Meglio, P.; Duarte, J.H.; Ahlfors, H.; Owens, N.D.L.; Li, Y.; Villanova, F.; Tosi, I.; Hirota, K.; Nestle, F.O.; Mrowietz, U.; et al. Activation of the Aryl Hydrocarbon Receptor Dampens the Severity of Inflammatory Skin Conditions. Immunity 2014, 40, 989–1001. [Google Scholar] [CrossRef]
- Nakamura, M.; Morita, A.; Seité, S.; Haarmann-Stemmann, T.; Grether-Beck, S.; Krutmann, J. Environment-Induced Lentigines: Formation of Solar Lentigines beyond Ultraviolet Radiation. Exp. Dermatol. 2015, 24, 407–411. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.-C. NF-ΚB Signaling in Inflammation. Signal Transduct. Target. Ther. 2017, 2, 17023. [Google Scholar] [CrossRef]
- Burke, K.E. Mechanisms of Aging and Development—A New Understanding of Environmental Damage to the Skin and Prevention with Topical Antioxidants. Mech. Ageing Dev. 2018, 172, 123–130. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, L.; Wen, X.; Hao, D.; Zhang, N.; He, G.; Jiang, X. NF-ΚB Signaling in Skin Aging. Mech. Ageing Dev. 2019, 184, 111160. [Google Scholar] [CrossRef] [PubMed]
- Kobielak, A.; Boddupally, K. Junctions and Inflammation in the Skin. Cell Commun. Adhes. 2014, 21, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Saha, S.; Buttari, B.; Panieri, E.; Profumo, E.; Saso, L. An Overview of Nrf2 Signaling Pathway and Its Role in Inflammation. Molecules 2020, 25, 5474. [Google Scholar] [CrossRef]
- Rajagopalan, P.; Jain, A.P.; Nanjappa, V.; Patel, K.; Mangalaparthi, K.K.; Babu, N.; Cavusoglu, N.; Roy, N.; Soeur, J.; Breton, L.; et al. Proteome-Wide Changes in Primary Skin Keratinocytes Exposed to Diesel Particulate Extract—A Role for Antioxidants in Skin Health. J. Dermatol. Sci. 2018, 91, 239–249. [Google Scholar] [CrossRef] [PubMed]
- Ko, H.; Kim, M.-M. H2O2 Promotes the Aging Process of Melanogenesis through Modulation of MITF and Nrf2. Mol. Biol. Rep. 2019, 46, 2461–2471. [Google Scholar] [CrossRef]
- Gęgotek, A.; Skrzydlewska, E. The Role of Transcription Factor Nrf2 in Skin Cells Metabolism. Arch. Dermatol. Res. 2015, 307, 385–396. [Google Scholar] [CrossRef] [PubMed]
- Waaijer, M.E.C.; Gunn, D.A.; Adams, P.D.; Pawlikowski, J.S.; Griffiths, C.E.M.; van Heemst, D.; Slagboom, P.E.; Westendorp, R.G.J.; Maier, A.B. P16INK4a Positive Cells in Human Skin Are Indicative of Local Elastic Fiber Morphology, Facial Wrinkling, and Perceived Age. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2016, 71, 1022–1028. [Google Scholar] [CrossRef] [PubMed]
- Wedel, S.; Martic, I.; Hrapovic, N.; Fabre, S.; Madreiter-Sokolowski, C.T.; Haller, T.; Pierer, G.; Ploner, C.; Jansen-Dürr, P.; Cavinato, M. TBHP Treatment as a Model for Cellular Senescence and Pollution-Induced Skin Aging. Mech. Ageing Dev. 2020, 190, 111318. [Google Scholar] [CrossRef]
- Rorteau, J.; Chevalier, F.P.; Bonnet, S.; Barthélemy, T.; Lopez-Gaydon, A.; Martin, L.S.; Bechetoille, N.; Lamartine, J. Maintenance of Chronological Aging Features in Culture of Normal Human Dermal Fibroblasts from Old Donors. Cells 2022, 11, 858. [Google Scholar] [CrossRef]
- Victorelli, S.; Lagnado, A.; Halim, J.; Moore, W.; Talbot, D.; Barrett, K.; Chapman, J.; Birch, J.; Ogrodnik, M.; Meves, A.; et al. Senescent Human Melanocytes Drive Skin Ageing via Paracrine Telomere Dysfunction. EMBO J. 2019, 38, e101982. [Google Scholar] [CrossRef]
- Barysch, M.J.; Braun, R.P.; Kolm, I.; Ahlgrimm-Siesz, V.; Hofmann-Wellenhof, R.; Duval, C.; Warrick, E.; Bernerd, F.; Nouveau, S.; Dummer, R. Keratinocytic Malfunction as a Trigger for the Development of Solar Lentigines. Dermatopathology 2019, 6, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Yoon, J.E.; Kim, Y.; Kwon, S.; Kim, M.; Kim, Y.H.; Kim, J.-H.; Park, T.J.; Kang, H.Y. Senescent Fibroblasts Drive Ageing Pigmentation: A Potential Therapeutic Target for Senile Lentigo. Theranostics 2018, 8, 4620–4632. [Google Scholar] [CrossRef] [PubMed]
- Carmona-Cruz, S.; Orozco-Covarrubias, L.; Sáez-de-Ocariz, M. The Human Skin Microbiome in Selected Cutaneous Diseases. Front. Cell. Infect. Microbiol. 2022, 12, 834135. [Google Scholar] [CrossRef]
- Kim, M.; Park, T.; Yun, J.I.; Lim, H.W.; Han, N.R.; Lee, S.T. Investigation of Age-Related Changes in the Skin Microbiota of Korean Women. Microorganisms 2020, 8, 1581. [Google Scholar] [CrossRef]
- D’Mello, S.; Finlay, G.; Baguley, B.; Askarian-Amiri, M. Signaling Pathways in Melanogenesis. Int. J. Mol. Sci. 2016, 17, 1144. [Google Scholar] [CrossRef]
- Yamaguchi, Y.; Brenner, M.; Hearing, V.J. The Regulation of Skin Pigmentation. J. Biol. Chem. 2007, 282, 27557–27561. [Google Scholar] [CrossRef] [PubMed]
- Serre, C.; Busuttil, V.; Botto, J.-M. Intrinsic and Extrinsic Regulation of Human Skin Melanogenesis and Pigmentation. Int. J. Cosmet. Sci. 2018, 40, 328–347. [Google Scholar] [CrossRef] [PubMed]
- Venkatesh, S.; Maymone, M.B.C.; Vashi, N.A. Aging in Skin of Color. Clin. Dermatol. 2019, 37, 351–357. [Google Scholar] [CrossRef]
- Vierkötter, A.; Hüls, A.; Yamamoto, A.; Stolz, S.; Krämer, U.; Matsui, M.S.; Morita, A.; Wang, S.; Li, Z.; Jin, L.; et al. Extrinsic Skin Ageing in German, Chinese and Japanese Women Manifests Differently in All Three Groups Depending on Ethnic Background, Age and Anatomical Site. J. Dermatol. Sci. 2016, 83, 219–225. [Google Scholar] [CrossRef]
- Wong, Q.Y.A.; Chew, F.T. Defining Skin Aging and Its Risk Factors: A Systematic Review and Meta-Analysis. Sci. Rep. 2021, 11, 22075. [Google Scholar] [CrossRef]
- Vashi, N.A.; de Castro Maymone, M.B.; Kundu, R.V. Aging Differences in Ethnic Skin. J. Clin. Aesthet. Dermatol. 2016, 9, 31–38. [Google Scholar] [PubMed]
- Langton, A.K.; Alessi, S.; Hann, M.; Chien, A.L.-L.; Kang, S.; Griffiths, C.E.M.; Watson, R.E.B. Aging in Skin of Color: Disruption to Elastic Fiber Organization Is Detrimental to Skin’s Biomechanical Function. J. Investig. Dermatol. 2019, 139, 779–788. [Google Scholar] [CrossRef] [PubMed]
- Chien, A.L.; Suh, J.; Cesar, S.S.A.; Fischer, A.H.; Cheng, N.; Poon, F.; Rainer, B.; Leung, S.; Martin, J.; Okoye, G.A.; et al. Pigmentation in African American Skin Decreases with Skin Aging. J. Am. Acad. Dermatol. 2016, 75, 782–787. [Google Scholar] [CrossRef]
- Del Bino, S.; Duval, C.; Bernerd, F. Clinical and Biological Characterization of Skin Pigmentation Diversity and Its Consequences on UV Impact. Int. J. Mol. Sci. 2018, 19, 2668. [Google Scholar] [CrossRef]
- Numan, M.; Brown, J.; Michou, L. Impact of Air Pollutants on Oxidative Stress in Common Autophagy-Mediated Aging Diseases. Int. J. Environ. Res. Public Health 2015, 12, 2289–2305. [Google Scholar] [CrossRef] [PubMed]
- Ryu, Y.S.; Kang, K.A.; Piao, M.J.; Ahn, M.J.; Yi, J.M.; Bossis, G.; Hyun, Y.-M.; Park, C.O.; Hyun, J.W. Particulate Matter-Induced Senescence of Skin Keratinocytes Involves Oxidative Stress-Dependent Epigenetic Modifications. Exp. Mol. Med. 2019, 51, 1–14. [Google Scholar] [CrossRef]
- Shin, K.-O.; Uchida, Y.; Park, K. Diesel Particulate Extract Accelerates Premature Skin Aging in Human Fibroblasts via Ceramide-1-Phosphate-Mediated Signaling Pathway. Int. J. Mol. Sci. 2022, 23, 2691. [Google Scholar] [CrossRef]
- Lee, C.-W.; Lin, Z.-C.; Hu, S.C.-S.; Chiang, Y.-C.; Hsu, L.-F.; Lin, Y.-C.; Lee, I.-T.; Tsai, M.-H.; Fang, J.-Y. Urban Particulate Matter Down-Regulates Filaggrin via COX2 Expression/PGE2 Production Leading to Skin Barrier Dysfunction. Sci. Rep. 2016, 6, 27995. [Google Scholar] [CrossRef]
- Park, S.-Y.; Byun, E.; Lee, J.; Kim, S.; Kim, H. Air Pollution, Autophagy, and Skin Aging: Impact of Particulate Matter (PM10) on Human Dermal Fibroblasts. Int. J. Mol. Sci. 2018, 19, 2727. [Google Scholar] [CrossRef]
- Patatian, A.; Delestre-Delacour, C.; Percoco, G.; Ramdani, Y.; Di Giovanni, M.; Peno-Mazzarino, L.; Bader, T.; Bénard, M.; Driouich, A.; Lati, E.; et al. Skin Biological Responses to Urban Pollution in an Ex Vivo Model. Toxicol. Lett. 2021, 348, 85–96. [Google Scholar] [CrossRef]
- Vierkötter, A.; Schikowski, T.; Ranft, U.; Sugiri, D.; Matsui, M.; Krämer, U.; Krutmann, J. Airborne Particle Exposure and Extrinsic Skin Aging. J. Investig. Dermatol. 2010, 130, 2719–2726. [Google Scholar] [CrossRef] [PubMed]
- Damevska, K.; Simeonovski, V.; Darlenski, R.; Damevska, S. How to Prevent Skin Damage from Air Pollution Part 2: Current Treatment Options. Dermatol. Ther. 2021, 34, e15132. [Google Scholar] [CrossRef] [PubMed]
- Mancebo, S.E.; Wang, S.Q. Recognizing the Impact of Ambient Air Pollution on Skin Health. J. Eur. Acad. Dermatol. Venereol. 2015, 29, 2326–2332. [Google Scholar] [CrossRef] [PubMed]
- Peng, F.; Xue, C.-H.; Hwang, S.K.; Li, W.-H.; Chen, Z.; Zhang, J.-Z. Exposure to Fine Particulate Matter Associated with Senile Lentigo in Chinese Women: A Cross-Sectional Study. J. Eur. Acad. Dermatol. Venereol. 2017, 31, 355–360. [Google Scholar] [CrossRef]
- Drakaki, E.; Dessinioti, C.; Antoniou, C.V. Air Pollution and the Skin. Front. Environ. Sci. 2014, 2, 11. [Google Scholar] [CrossRef]
- Bae, Y.J.; Park, K.Y.; Han, H.S.; Kim, Y.S.; Hong, J.Y.; Han, T.Y.; Seo, S.J. Effects of Particulate Matter in a Mouse Model of Oxazolone-Induced Atopic Dermatitis. Ann. Dermatol. 2020, 32, 496. [Google Scholar] [CrossRef]
- Pan, S.; Qiu, Y.; Li, M.; Yang, Z.; Liang, D. Recent Developments in the Determination of PM2.5 Chemical Composition. Bull. Environ. Contam. Toxicol. 2022, 108, 819–823. [Google Scholar] [CrossRef]
- Arias-Pérez, R.D.; Taborda, N.A.; Gómez, D.M.; Narvaez, J.F.; Porras, J.; Hernandez, J.C. Inflammatory Effects of Particulate Matter Air Pollution. Environ. Sci. Pollut. Res. 2020, 27, 42390–42404. [Google Scholar] [CrossRef]
- Soeur, J.; Belaïdi, J.-P.; Chollet, C.; Denat, L.; Dimitrov, A.; Jones, C.; Perez, P.; Zanini, M.; Zobiri, O.; Mezzache, S.; et al. Photo-Pollution Stress in Skin: Traces of Pollutants (PAH and Particulate Matter) Impair Redox Homeostasis in Keratinocytes Exposed to UVA1. J. Dermatol. Sci. 2017, 86, 162–169. [Google Scholar] [CrossRef]
- Fernando, I.P.S.; Jayawardena, T.U.; Kim, H.-S.; Vaas, A.P.J.P.; De Silva, H.I.C.; Nanayakkara, C.M.; Abeytunga, D.T.U.; Lee, W.; Ahn, G.; Lee, D.-S.; et al. A Keratinocyte and Integrated Fibroblast Culture Model for Studying Particulate Matter-Induced Skin Lesions and Therapeutic Intervention of Fucosterol. Life Sci. 2019, 233, 116714. [Google Scholar] [CrossRef]
- Estrella, B.; Naumova, E.N.; Cepeda, M.; Voortman, T.; Katsikis, P.D.; Drexhage, H.A. Effects of Air Pollution on Lung Innate Lymphoid Cells: Review of In Vitro and In Vivo Experimental Studies. Int. J. Environ. Res. Public Health 2019, 16, 2347. [Google Scholar] [CrossRef] [PubMed]
- Laing, S.; Wang, G.; Briazova, T.; Zhang, C.; Wang, A.; Zheng, Z.; Gow, A.; Chen, A.F.; Rajagopalan, S.; Chen, L.C.; et al. Airborne Particulate Matter Selectively Activates Endoplasmic Reticulum Stress Response in the Lung and Liver Tissues. Am. J. Physiol. Physiol. 2010, 299, C736–C749. [Google Scholar] [CrossRef] [PubMed]
- Marchini, T.; D’Annunzio, V.; Paz, M.L.; Cáceres, L.; Garcés, M.; Perez, V.; Tasat, D.; Vanasco, V.; Magnani, N.; Gonzalez Maglio, D.; et al. Selective TNF-α Targeting with Infliximab Attenuates Impaired Oxygen Metabolism and Contractile Function Induced by an Acute Exposure to Air Particulate Matter. Am. J. Physiol. Circ. Physiol. 2015, 309, H1621–H1628. [Google Scholar] [CrossRef] [PubMed]
- Mylonas, A.; Conrad, C. Psoriasis: Classical vs. Paradoxical. The Yin-Yang of TNF and Type I Interferon. Front. Immunol. 2018, 9, 2746. [Google Scholar] [CrossRef] [PubMed]
- Pieters, N.; Janssen, B.G.; Dewitte, H.; Cox, B.; Cuypers, A.; Lefebvre, W.; Smeets, K.; Vanpoucke, C.; Plusquin, M.; Nawrot, T.S. Biomolecular Markers within the Core Axis of Aging and Particulate Air Pollution Exposure in the Elderly: A Cross-Sectional Study. Environ. Health Perspect. 2016, 124, 943–950. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.; Guo, Z.; Zhang, R.; Deng, C.; Xu, J.; Dong, W.; Hong, Z.; Yu, H.; Situ, H.; Liu, C.; et al. Nasal Epithelial Barrier Disruption by Particulate Matter ≤2.5 Μm via Tight Junction Protein Degradation. J. Appl. Toxicol. 2018, 38, 678–687. [Google Scholar] [CrossRef] [PubMed]
- Dijkhoff, I.M.; Drasler, B.; Karakocak, B.B.; Petri-Fink, A.; Valacchi, G.; Eeman, M.; Rothen-Rutishauser, B. Impact of Airborne Particulate Matter on Skin: A Systematic Review from Epidemiology to in Vitro Studies. Part. Fibre Toxicol. 2020, 17, 35. [Google Scholar] [CrossRef]
- Pan, T.-L.; Wang, P.-W.; Aljuffali, I.A.; Huang, C.-T.; Lee, C.-W.; Fang, J.-Y. The Impact of Urban Particulate Pollution on Skin Barrier Function and the Subsequent Drug Absorption. J. Dermatol. Sci. 2015, 78, 51–60. [Google Scholar] [CrossRef]
- Kim, S.; Kim, J.; Lee, Y.I.; Jang, S.; Song, S.Y.; Lee, W.J.; Lee, J.H. Particulate Matter-induced Atmospheric Skin Aging Is Aggravated by UVA and Inhibited by a Topical L-ascorbic Acid Compound. Photodermatol. Photoimmunol. Photomed. 2022, 38, 123–131. [Google Scholar] [CrossRef]
- Long, E.; Schwartz, C.; Carlsten, C. Controlled Human Exposure to Diesel Exhaust: A Method for Understanding Health Effects of Traffic-Related Air Pollution. Part. Fibre Toxicol. 2022, 19, 15. [Google Scholar] [CrossRef]
- Molina-García, M.; Malvehy, J.; Granger, C.; Garre, A.; Trullàs, C.; Puig, S. Exposome and Skin. Part 2. The Influential Role of the Exposome, Beyond UVR, in Actinic Keratosis, Bowen’s Disease and Squamous Cell Carcinoma: A Proposal. Dermatol. Ther. 2022, 12, 361–380. [Google Scholar] [CrossRef] [PubMed]
- Bashir, M.M.; Sharma, M.R.; Werth, V.P. TNF-α Production in the Skin. Arch. Dermatol. Res. 2009, 301, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Mansuri, M.S.; Kadam, A.; Palit, S.P.; Dwivedi, M.; Laddha, N.C.; Begum, R. Tumor Necrosis Factor-Alpha Affects Melanocyte Survival and Melanin Synthesis via Multiple Pathways in Vitiligo. Cytokine 2021, 140, 155432. [Google Scholar] [CrossRef]
- Kim, H.; Park, S.-Y.; Moon, S.; Lee, J.; Kim, S. Autophagy in Human Skin Fibroblasts: Impact of Age. Int. J. Mol. Sci. 2018, 19, 2254. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.; Lim, C.; Chung, H.-J.; Kim, J.-H.; Huh, Y.; Park, K.; Jeong, S. Autophagy Activation by Crepidiastrum Denticulatum Extract Attenuates Environmental Pollutant-Induced Damage in Dermal Fibroblasts. Int. J. Mol. Sci. 2019, 20, 517. [Google Scholar] [CrossRef] [PubMed]
- Mokrzyński, K.; Krzysztyńska-Kuleta, O.; Zawrotniak, M.; Sarna, M.; Sarna, T. Fine Particulate Matter-Induced Oxidative Stress Mediated by UVA-Visible Light Leads to Keratinocyte Damage. Int. J. Mol. Sci. 2021, 22, 10645. [Google Scholar] [CrossRef] [PubMed]
- Dimitrov, A.; Zanini, M.; Zucchi, H.; Boudah, S.; Lima, J.; Soeur, J.; Marrot, L. Vitamin C Prevents Epidermal Damage Induced by PM-associated Pollutants and UVA1 Combined Exposure. Exp. Dermatol. 2021, 30, 1693–1698. [Google Scholar] [CrossRef]
- Suo, D.; Zeng, S.; Zhang, J.; Meng, L.; Weng, L. PM2.5 Induces Apoptosis, Oxidative Stress Injury and Melanin Metabolic Disorder in Human Melanocytes. Exp. Ther. Med. 2020, 19, 3227. [Google Scholar] [CrossRef]
- Hieda, D.S.; Anastacio da Costa Carvalho, L.; Vaz de Mello, B.; de Oliveira, E.A.; Romano de Assis, S.; Wu, J.; Du-Thumm, L.; Viana da Silva, C.L.; Roubicek, D.A.; Maria-Engler, S.S.; et al. Air Particulate Matter Induces Skin Barrier Dysfunction and Water Transport Alteration on a Reconstructed Human Epidermis Model. J. Investig. Dermatol. 2020, 140, 2343–2352.e3. [Google Scholar] [CrossRef]
- Sarama, R.; Matharu, P.K.; Abduldaiem, Y.; Corrêa, M.P.; Gil, C.D.; Greco, K.V. In Vitro Disease Models for Understanding Psoriasis and Atopic Dermatitis. Front. Bioeng. Biotechnol. 2022, 10, 803218. [Google Scholar] [CrossRef]
- Percoco, G.; Patatian, A.; Eudier, F.; Grisel, M.; Bader, T.; Lati, E.; Savary, G.; Picard, C.; Benech, P. Impact of Cigarette Smoke on Physical-chemical and Molecular Proprieties of Human Skin in an Ex Vivo Model. Exp. Dermatol. 2021, 30, 1610–1618. [Google Scholar] [CrossRef] [PubMed]
- Ono, Y.; Torii, K.; Fritsche, E.; Shintani, Y.; Nishida, E.; Nakamura, M.; Shirakata, Y.; Haarmann-Stemmann, T.; Abel, J.; Krutmann, J.; et al. Role of the Aryl Hydrocarbon Receptor in Tobacco Smoke Extract-Induced Matrix Metalloproteinase-1 Expression. Exp. Dermatol. 2013, 22, 349–353. [Google Scholar] [CrossRef] [PubMed]
- Hoskin, R.; Pambianchi, E.; Pecorelli, A.; Grace, M.; Therrien, J.-P.; Valacchi, G.; Lila, M.A. Novel Spray Dried Algae-Rosemary Particles Attenuate Pollution-Induced Skin Damage. Molecules 2021, 26, 3781. [Google Scholar] [CrossRef] [PubMed]
- D’Agostini, F.; Balansky, R.; Pesce, C.; Fiallo, P.; Lubet, R.A.; Kelloff, G.J.; De Flora, S. Induction of Alopecia in Mice Exposed to Cigarette Smoke. Toxicol. Lett. 2000, 114, 117–123. [Google Scholar] [CrossRef]
- Lecas, S.; Boursier, E.; Fitoussi, R.; Vié, K.; Momas, I.; Seta, N.; Achard, S. In Vitro Model Adapted to the Study of Skin Ageing Induced by Air Pollution. Toxicol. Lett. 2016, 259, 60–68. [Google Scholar] [CrossRef]
- Langton, A.K.; Tsoureli-Nikita, E.; Merrick, H.; Zhao, X.; Antoniou, C.; Stratigos, A.; Akhtar, R.; Derby, B.; Sherratt, M.J.; Watson, R.E.B.; et al. The Systemic Influence of Chronic Smoking on Skin Structure and Mechanical Function. J. Pathol. 2020, 251, 420–428. [Google Scholar] [CrossRef]
- Petracca, B.; Nădăban, A.; Eeman, M.; Gooris, G.S.; Bouwstra, J.A. Effects of Ozone on Stratum Corneum Lipid Integrity and Assembly. Chem. Phys. Lipids 2021, 240, 105121. [Google Scholar] [CrossRef]
- Valacchi, G.; Pagnin, E.; Corbacho, A.M.; Olano, E.; Davis, P.A.; Packer, L.; Cross, C.E. In Vivo Ozone Exposure Induces Antioxidant/Stress-Related Responses in Murine Lung and Skin. Free Radic. Biol. Med. 2004, 36, 673–681. [Google Scholar] [CrossRef]
- Valacchi, G.; van der Vliet, A.; Schock, B.C.; Okamoto, T.; Obermuller-Jevic, U.; Cross, C.E.; Packer, L. Ozone Exposure Activates Oxidative Stress Responses in Murine Skin. Toxicology 2002, 179, 163–170. [Google Scholar] [CrossRef]
- Valacchi, G.; Sticozzi, C.; Belmonte, G.; Cervellati, F.; Demaude, J.; Chen, N.; Krol, Y.; Oresajo, C. Vitamin C Compound Mixtures Prevent Ozone-Induced Oxidative Damage in Human Keratinocytes as Initial Assessment of Pollution Protection. PLoS ONE 2015, 10, e0131097. [Google Scholar] [CrossRef]
- Krutmann, J.; Bouloc, A.; Sore, G.; Bernard, B.A.; Passeron, T. The Skin Aging Exposome. J. Dermatol. Sci. 2017, 85, 152–161. [Google Scholar] [CrossRef]
- Valacchi, G.; Pecorelli, A.; Belmonte, G.; Pambianchi, E.; Cervellati, F.; Lynch, S.; Krol, Y.; Oresajo, C. Protective Effects of Topical Vitamin C Compound Mixtures against Ozone-Induced Damage in Human Skin. J. Investig. Dermatol. 2017, 137, 1373–1375. [Google Scholar] [CrossRef] [PubMed]
- Chavatte, L.; Juan, M.; Mounicou, S.; Leblanc Noblesse, E.; Pays, K.; Nizard, C.; Bulteau, A.-L. Elemental and Molecular Imaging of Human Full Thickness Skin after Exposure to Heavy Metals. Metallomics 2020, 12, 1555–1562. [Google Scholar] [CrossRef] [PubMed]
- Juliano, C.; Magrini, G. Cosmetic Functional Ingredients from Botanical Sources for Anti-Pollution Skincare Products. Cosmetics 2018, 5, 19. [Google Scholar] [CrossRef]
- Mousavi, S.E.; Delgado-Saborit, J.M.; Adivi, A.; Pauwels, S.; Godderis, L. Air Pollution and Endocrine Disruptors Induce Human Microbiome Imbalances: A Systematic Review of Recent Evidence and Possible Biological Mechanisms. Sci. Total Environ. 2022, 816, 151654. [Google Scholar] [CrossRef]
- Charoenchon, N.; Rhodes, L.E.; Nicolaou, A.; Williamson, G.; Watson, R.E.B.; Farrar, M.D. Ultraviolet Radiation-induced Degradation of Dermal Extracellular Matrix and Protection by Green Tea Catechins: A Randomized Controlled Trial. Clin. Exp. Dermatol. 2022, 47, 1314–1323. [Google Scholar] [CrossRef]
- Tigges, J.; Haarmann-Stemmann, T.; Vogel, C.F.A.; Grindel, A.; Hübenthal, U.; Brenden, H.; Grether-Beck, S.; Vielhaber, G.; Johncock, W.; Krutmann, J.; et al. The New Aryl Hydrocarbon Receptor Antagonist E/Z-2-Benzylindene-5,6-Dimethoxy-3,3-Dimethylindan-1-One Protects against UVB-Induced Signal Transduction. J. Investig. Dermatol. 2014, 134, 556–559. [Google Scholar] [CrossRef]
- Khmaladze, I.; Österlund, C.; Smiljanic, S.; Hrapovic, N.; Lafon-Kolb, V.; Amini, N.; Xi, L.; Fabre, S. A Novel Multifunctional Skin Care Formulation with a Unique Blend of Antipollution, Brightening and Antiaging Active Complexes. J. Cosmet. Dermatol. 2020, 19, 1415–1425. [Google Scholar] [CrossRef]
| Intervention | Active Ingredients | Mechanism | References |
|---|---|---|---|
| Sunscreen | Oxybenzone Zinc oxide |
| [1] |
| Washing and air filters |
| [8,85] | |
| Dietary habits | Phenolic compounds in plants |
| [13] |
| Rinse-off, film-forming cosmetics, and emollients | BDDI Aleurites fordii oil copolymer Kaolin |
| [85] |
| Antioxidants | Vitamin C Vitamin E Ferulic acid |
| [1,85] |
| Botanicals | Algae |
| [127] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martic, I.; Jansen-Dürr, P.; Cavinato, M. Effects of Air Pollution on Cellular Senescence and Skin Aging. Cells 2022, 11, 2220. https://doi.org/10.3390/cells11142220
Martic I, Jansen-Dürr P, Cavinato M. Effects of Air Pollution on Cellular Senescence and Skin Aging. Cells. 2022; 11(14):2220. https://doi.org/10.3390/cells11142220
Chicago/Turabian StyleMartic, Ines, Pidder Jansen-Dürr, and Maria Cavinato. 2022. "Effects of Air Pollution on Cellular Senescence and Skin Aging" Cells 11, no. 14: 2220. https://doi.org/10.3390/cells11142220
APA StyleMartic, I., Jansen-Dürr, P., & Cavinato, M. (2022). Effects of Air Pollution on Cellular Senescence and Skin Aging. Cells, 11(14), 2220. https://doi.org/10.3390/cells11142220






